Abstract
Compounds with propargylamine moiety are useful synthetic precursors of several important classes of nitrogen-containing heterocycles. The title compound, methyl (2E)-3-[3-benzyl-2-(3-methoxy-3-oxoprop-1-yn-1-yl)-2-(1-naphthyl)imidazolidine-1-yl]acrylate, has been prepared by domino-reaction, employing easily available 1-benzyl-2-(1-naphthyl)-4,5-dihydro-1H-imidazole and methyl propiolate in a high 92% yield. The structure of title compound was determined using 1H-NMR, 13C-NMR, UV, FT-IR and HRMS (High-Resolution Mass Spectrometry).
1. Introduction
Propargylamines are widely used as convenient synthetic intermediates for the preparation of various heterocyclic and pharmaceutically relevant compounds [1,2,3]. A special place among propargylamines is occupied by N-vinylpropargylamines due to the fact that the profile of their reactivity includes [3,3]-sigmatropic aza-Claisen rearrangement as well as 5-exo-dig- and 6-endo-dig-cyclizations, leading to such attractive heterocyclic compounds as pyrroles and pyridines [4,5,6,7,8,9,10,11,12,13,14]. Recently, we have proposed a new type of N-vinylpropargylamines, which can be easily obtained by the reaction of 1,2-disubstituted 2-imidazolines with such electron-deficient alkynes as methyl propiolate or acetylacetylene [15]. It was also shown that refluxing these compounds in o-xylene solution under aerobic conditions led to polysubstituted pyrroles through a domino-sequence starting with [3,3]-sigmatropic aza-Claisen rearrangement and final oxidative step. At the same time, the synthetic potential of such propargylamines, in our opinion, is far from being exhausted. Herein, we report a convenient preparative synthesis of a new derivative from the aforementioned group of compounds, containing a condensed aromatic fragment as a substituent.
2. Results and Discussion
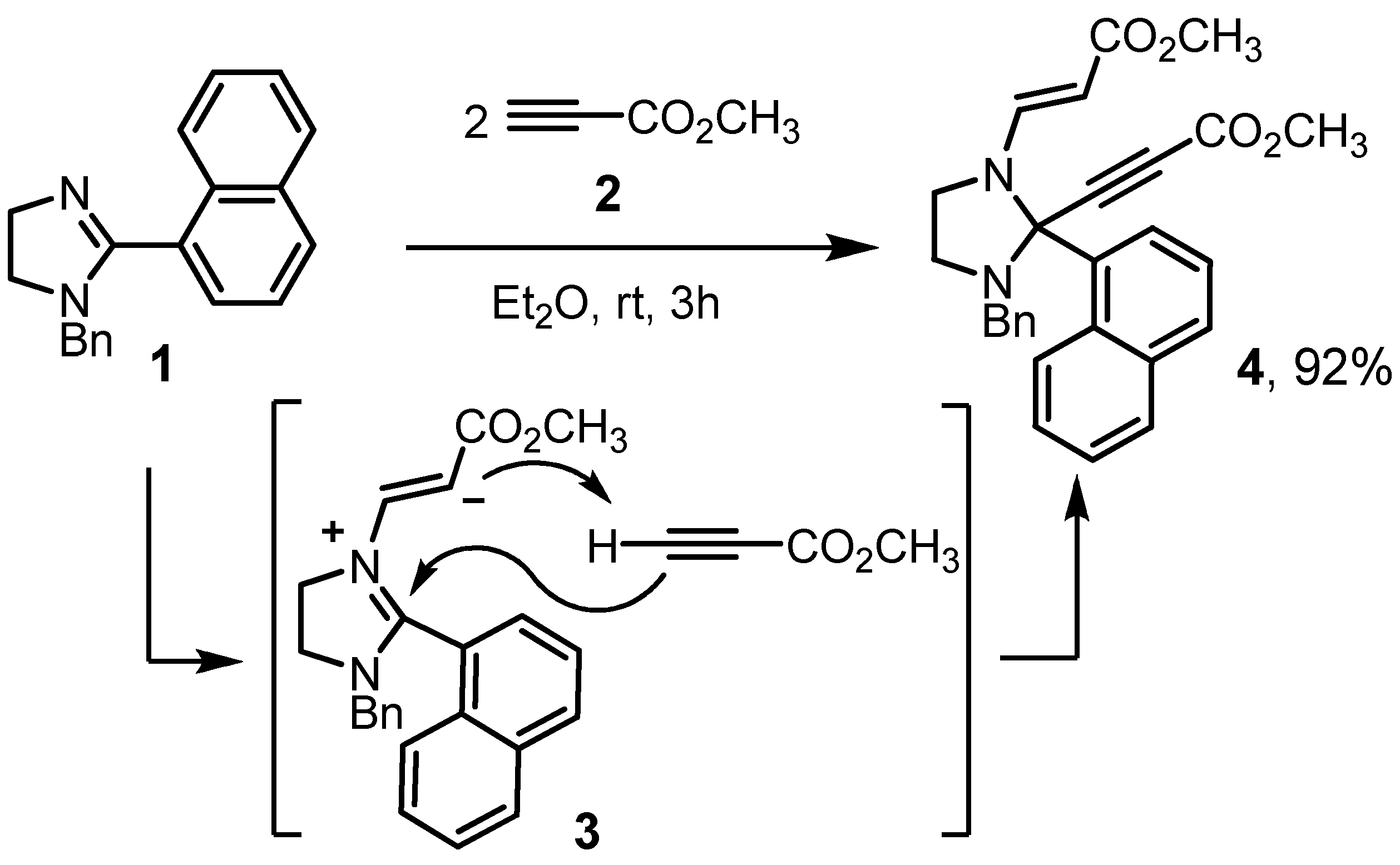
The starting material, 1-benzyl-2-(1-naphthyl)-4,5-dihydro-1H-imidazole (1), was obtained through a slightly modified Fujioka’s procedure by condensation of N-benzylethane-1,2-diamine (CAS 4152-09-4) with 1-naphthaldehyde (CAS 66-77-3) and subsequent oxidation of cyclic aminal by NBS (CAS 128-08-5) [16,17]. The reaction of imidazoline 1 with 2 equivalents of methyl propiolate (2, CAS 922-67-8) in diethyl ether at room temperature was completed in 3 h according to TLC (Scheme 1). Target methyl (2E)-3-[3-benzyl-2-(3-methoxy-3-oxoprop-1-yn-1-yl)-2-(1-naphthyl)imidazolidin-1-yl]acrylate (4) was isolated in 85% yield by concentrating the reaction mixture and crystallization from diethyl ether at 0–5 °C. Isolation of the product by column chromatography using chloroform: hexane, 100:50 as an eluent led to an increase in yield up to 92%.
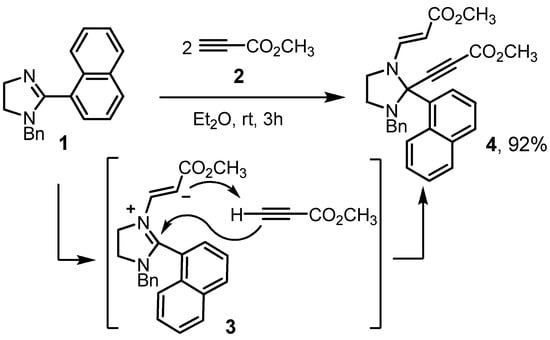
Scheme 1.
Preparation of the title compound.
The mechanism of imidazolidine 4 formation includes the conjugated addition of 2-imidazoline (1) at the triple bond of the first alkyne molecule, leading to the zwitterion (3), which then deprotonates the second methylpropiolate molecule to form an acetylenide ion. At the final stage, nucleophilic addition of the obtained acetylenide ion occurs at the position 2 of the 2-imidazolinium ion. Thus, this interaction is an example of a pseudo three-component domino reaction.
The structure determination of the title compound 4 was performed with 1H- and 13C-NMR spectroscopy, IR and UV spectroscopy, and also mass spectrometry. In the 1H-NMR spectrum, six protons of the two methoxy groups of ester fragments of propiolic and aminoacrylic acids appeared as two peaks at 3.51 ppm (s, 3H) and 3.89 ppm (s, 3H). Two protons of the methylene group of the benzyl fragment appeared in the spectrum as two doublets at 3.56 ppm (1H) and 4.68 ppm (1H), which confirms the presence of chiral carbon atom in the product structure. CH-protons of the double bond of the aminoacrylic acid residue, containing an electron-withdrawing ester group on the one hand and an electron-donating nitrogen atom of the imidazolidine fragment on the other, appeared as two doublets at 4.68 ppm and 7.11 ppm with constant 13.1 Hz which is characteristic for CH-protons of double bond in trans-configuration. In the IR spectra of adduct 4, there is a band at 2220 cm−1, characteristic for stretching vibrations of the triple bond; a high-intensity band at 1715 cm−1, characteristic of vibrations of the carbonyl groups of ester fragments; and at 1614 cm−1, characteristic for stretching vibrations of the double bond. The brutto formula was devised with the help of high-resolution mass spectrometry. The spectral characteristics of α-naphthyl derivative 4 were in good agreement with the data of previously obtained analogous compounds.
In summary, using the elaborated method, compound 4 has been synthesized for the first time in a high yield. These results can be useful for further research on propargylamines and their subsequent transformations.
3. Materials and Methods
3.1. General
Starting reagents were purchased from commercial sources and were used without any additional purification (Acros Organics, Geel, Belgium). 1H and 13C-NMR spectra were acquired on a Jeol JNM-ECA 600 spectrometer (Jeol Ltd, Tokyo, Japan) (with operating frequencies of 600 and 150 MHz, respectively) at room temperature and referenced to the residual signals of the solvent. The solvent used for NMR was CDCl3. Chemical shifts are reported in parts per million (δ/ppm). Coupling constants are reported in Hertz (J/Hz). The peak patterns are indicated as follows: s, singlet; d, doublet; t, triplet; q, quadruplet; m, multiplet; dd, doublet of doublets and br s, broad singlet. Infrared spectra were measured on an Infralum FT-801 FT/IR instrument (Simex, Novosibirsk, Russia). The wavelengths are reported in reciprocal centimeters (νmax/cm−1). UV spectra were recorded using Varian Cary 50 (Agilent, Santa Clara, CA, USA). Mass spectra were recorded with LCMS-8040 Triple quadrupole liquid chromatograph mass-spectrometer from Shimadzu (ESI) and Kratos MS-30 mass-spectrometer (EI, 70 eV) (Shimadzu, Tokyo, Japan). HRMS spectra were recorded on a Bruker MicrOTOF-Q II (Bruker, Billerica, MA, USA). The reaction progress was monitored by TLC and the spots were visualized under UV light (254 or 365 nm) (UVGL-25, UVP, Upland, CA, USA). Column chromatography was performed using silica gel (60–75 mesh) (Merck KGaA, Darmstadt, Germany). Melting points were determined on a SMP-10 apparatus (Stuart, Stone, UK) and were uncorrected. Solvents were distilled and dried according to standard procedures.
3.2. Methyl (2E)-3-[3-Benzyl-2-(3-methoxy-3-oxoprop-1-yn-1-yl)-2-(1-naphthyl)imidazolidin-1-yl]acrylate
Methyl propiolate (0.182 mL, 2.2 mmol) was added to a solution of 1-benzyl-2-(1-naphthyl)-4,5-dihydro-1H-imidazole (0.3 g, 1 mmol) in dry ether (4 mL) and the resulting solution was stirred for 3h at rt. After the interaction completion (TLC monitoring) the solution was concentrated approximately two times in vacuo and cooled to 0–5 °C. The resultant crystals were filtered and dried at rt to give the title compound 3 (404 mg, 85%) as a yellow powder; m.p. 142–143 °C (Et2O). Purification by column chromatography on SiO2 (CHCl3/Hexane, 100:50) instead of crystallization gave 440 mg (92%) of the title compound 3. TLC: Rf =0.35 (CHCl3). 1H-NMR (600 MHz, CDCl3) δ, ppm: 2.95 (q, 1H, J = 9.1 Hz), 3.32 (d, 1H, J = 13.1 Hz), 3.38 (t, 1H, J = 8.3 Hz), 3.50 (t, 1H, J = 9.1 Hz), 3.51 (s, 3H), 3.56 (d, 1H, J = 12.6 Hz), 3.69 (q, 1H, J = 17.7 Hz), 3.89 (s, 3H), 4.68 (d, 1H, J = 13.1 Hz), 6.98 (dd, 2H, J = 6.3, J = 2.8 Hz), 7.11 (d, 1H, J = 13.1 Hz), 7.14–7.16 (m, 3H), 7.48–7.52 (m, 3H), 7.88–7.89 (m, 1H), 7.95 (d, 1H, J = 8.1 Hz), 8.28 (d, 1H, J = 7.6 Hz), 8.45 (d, 1H, J = 8.6 Hz). 13C-NMR (150 MHz, CDCl3) δ, ppm: 168.9 (Cq), 153.8 (Cq), 144.1 (CH), 137.3 (CH), 135.2 (CH), 132.4 (CH), 131.3 (CH), 130.1 (CH), 129.4 (Cq), 129.2 (Cq), 128.8 (2C, CH), 128.4 (2C, CH), 127.4 (CH), 126.2 (CH), 126.2 (Cq), 125.9 (CH), 124.5 (Cq), 89.8 (CH), 85.0 (Cq), 81.9 (Cq), 81.3 (Cq), 53.7 (CH2), 53.2 (CH2), 50.7 (CH3), 48.0 (CH3), 44.9 (CH2). EI-MS (m/z, M+): 455 (100%), 456 (30%), 457 (6%). HRMS Electrospray ionization (ESI) m/z calcd for C28H27N2O4+ = 455.1965, found 455.1965. λmax, MeCN: 220 (ε = 48,800), 275 (ε = 29,800) nm. IR (KBr), ν (cm−1): 3028 (Ar-H), 2948 (C-H), 2835 (C-H), 2220 (C≡C), 1715 (C=O), 1614 (C=C), 1245 (C-O ether), 1158 (C-O ether).
Copies of all spectra and ESI-HRMS (Figures S1–S6) are provided in the Supplementary Materials file.
Supplementary Materials
Copies of 1H-, 13C-NMR, IR, UV and HRMS spectra are available online.
Author Contributions
Conceptualization, N.E.G. and L.G.V.; methodology, N.E.G. and A.S.G.; investigation, A.S.G.; writing–original draft preparation, N.E.G.; writing–review and editing, A.S.G. and L.G.V.; funding acquisition, N.E.G. All authors have read and agreed to the published version of the manuscript.
Funding
This work has been funded by the Russian Foundation for Basic Research (project № 19-03-00502 a).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lauder, K.; Toscani, A.; Scalacci, N.; Castagnolo, D. Synthesis and Reactivity of Propargylamines in Organic Chemistry. Chem. Rev. 2017, 117, 14091–14200. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Wang, T. Gold-Catalyzed Transformations of Propargyl Alcohols and Propargyl Amines. Asian J. Org. Chem. 2018, 7, 1758–1783. [Google Scholar] [CrossRef]
- Sheng, X.; Chen, K.; Shi, C.; Huang, D. Recent Advances in Reactions of Propargylamines. Synthesis 2020, 52, 1–20. [Google Scholar] [CrossRef]
- Cossy, J.; Poitevin, C.; Sallé, L.; Gomez Pardo, D. The Thermal Rearrangement of N-Alkyl-N-Vinylpropargylamines into 2-Methylpyrroles. A New Synthesis of Annulated[b]Pyrroles. Tetrahedron Lett. 1996, 37, 6709–6710. [Google Scholar] [CrossRef]
- Minnihan, E.C.; Colletti, S.L.; Toste, F.D.; Shen, H.C. Gold(I)-catalyzed regioselective cyclizations of silyl ketene amides and carbamates with alkynes. J. Org. Chem. 2007, 72, 6287–6289. [Google Scholar] [CrossRef] [PubMed]
- Bremner, W.S.; Organ, M.G. Formation of Substituted Pyrroles via an Imine Condensation/Aza-Claisen Rearrangement/Imine-Allene Cyclization Process by MAOS. J. Comb. Chem. 2008, 10, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Cacchi, S.; Fabrizi, G.; Filisti, E. N-Propargylic β-Enaminones: Common Intermediates for the Synthesis of Polysubstituted Pyrroles and Pyridines. Org. Lett. 2008, 10, 2629–2632. [Google Scholar] [CrossRef] [PubMed]
- Weng, J.; Chen, Y.; Yue, B.; Xu, M.; Jin, H. Synthesis of Polysubstituted Pyrroles from Activated Alkynes and N-Propargylamines through Base-Catalyzed Cascade Reaction. Eur. J. Org. Chem. 2015, 3164–3170. [Google Scholar] [CrossRef]
- Saito, A.; Konishi, T.; Hanzawa, Y. Synthesis of Pyrroles by Gold(I)-Catalyzed Amino-Claisen Rearrangement of N-Propargyl Enaminone Derivatives. Org. Lett. 2010, 12, 372–374. [Google Scholar] [CrossRef] [PubMed]
- Xin, X.; Wang, D.; Li, X.; Wan, B. Highly regioselective migration of the sulfonyl group: Easy access to functionalized pyrroles. Angew. Chem. Int. Ed. 2012, 51, 1693–1697. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wang, Y.; Hu, F.; Kan, X.; Yang, C.; Liu, J.; Liu, P.; Zhang, Q. Base-promoted intramolecular cyclization of: N-alkyl, N-propargylic β-enaminones for the synthesis of polysubstituted pyrroles. RSC Adv. 2016, 6, 68454–68459. [Google Scholar] [CrossRef]
- Ge, B.; Lv, W.; Yu, J.; Xiao, S.; Cheng, G. Base-promoted C-C bond cleavage for the synthesis of 2,3,4-trisubstituted pyrroles from: N-propargyl β-enaminones. Org. Chem. Front. 2018, 5, 3103–3107. [Google Scholar] [CrossRef]
- Chikayuki, Y.; Miyashige, T.; Yonekawa, S.; Kirita, A.; Matsuo, N.; Teramoto, H.; Sasaki, S.; Higashiyama, K.; Yamauchi, T. Transition-Metal-Free Synthesis of Pyridine Derivatives by Thermal Cyclization of N-Propargyl Enamines. Synthesis 2020, 52, 1113–1121. [Google Scholar] [CrossRef]
- Matouš, P.; Kadaník, M.; Timoracký, M.; Kuneš, J.; Maříková, J.; Růžička, A.; Kočovský, P.; Pour, M. Nucleophile-assisted cyclization of β-propargylamino acrylic compounds catalyzed by gold(i): A rapid construction of multisubstituted tetrahydropyridines and their fused derivatives. Org. Chem. Front. 2020, 7, 3356–3367. [Google Scholar] [CrossRef]
- Golantsov, N.E.; Golubenkova, A.S.; Festa, A.A.; Varlamov, A.V.; Voskressensky, L.G. A Domino Route toward Polysubstituted Pyrroles from 2-Imidazolines and Electron-Deficient Alkynes. Org. Lett. 2020, 22, 4726–4731. [Google Scholar] [CrossRef] [PubMed]
- Fujioka, H.; Murai, K.; Kubo, O.; Ohba, Y.; Kita, Y. One-pot synthesis of imidazolines from aldehydes: Detailed study about solvents and substrates. Tetrahedron 2007, 63, 638–643. [Google Scholar] [CrossRef]
- Golubenkova, A.S.; Golantsov, N.E.; Festa, A.A.; Voskressensky, L.G. 1-Benzyl-2-(Thien-2-yl)-4,5-dihydro-1H-imidazole. MolBank 2020, 2020, M1137. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).