Abstract
A new suitable method of synthesis of methyl 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate by condensation of methyl 2-isothiocyanatobenzoate and methyl malonate is described. The structure of the compound both and by-product methyl 2-(methoxycarbonothioylamino)benzoate was confirmed by means of elemental analysis, 1H NMR, 13C NMR, LC/MS and single crystal X-ray diffraction. UV/Vis and IR spectra of compounds are described. The presence of a strong intramolecular hydrogen bond between the hydroxy group and the carbonyl oxygen atom of the ester group in methyl 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate is shown. The crystal structure of product was stabilized through intermolecular hydrogen bonds.
1. Introduction
Derivatives of 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylic acid, especially containing fluorine atom, are known as highly effective antibiotics, which are similar to commonly used quinolones [1,2,3,4,5,6,7]. The sulfur atom in the 2 position of quinoline moiety improves antibacterial activity [4]. Hence, this scaffold is interesting for development of new effective bactericide compounds. However, known synthetic routes for 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylates have stages requiring stringent conditions like high temperature or strong base catalyst. Usual way of their synthesis is condensation of appropriate isothiocyanatobenzene 1 with anion of malonic ester 2 results in formation of methylenemalonate derivatives 3. Subsequent thermal or acid catalyzed cyclization gives 4-hydroxy-2-(alkylthio)quinoline-3-carboxylate 4 (Scheme 1) [1,2,3,4,5,6]. In similar way alkyl phenyldithiocarbamates 5, which can be easy obtained in situ from corresponding aniline, CS2 and alkylating reagent, react with anion of malonic ester 2 producing methylenemalonate derivatives 3. Further desalkylation of alkylthioderivatives 4 by strong acid leads to 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate 6 (Scheme 1) [1,2,3,4,5,6].

Scheme 1.
General route to 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate 6 synthesis.
Alternative way of synthesis starting from anthranilic acid 7 was suggested (Scheme 2) [7]. Anthranilic acid 7 was converted into 4,5-difluoro-2-{[(methylthio)carbonothioyl]amino}benzoic acid 8 by action of CS2 and MeI. After reflux in Ac2O 6,7-difluoro-2-(methylthio)-4H-3,1-benzothiazin-4-one 9 was obtained. The latter was condensed with EtOAc by LiN(i-Pr)2 as catalyst producing ethyl 3-(4,5-difluoro-2-{[(methylthio)carbonothioyl]amino}phenyl)-3-oxopropanoate 10. Followed intramolecular condensation by EtONa lead to ethyl 6,7-difluoro-4-hydroxy-2-thioxo-1.2-dihydroquinoline-3-carboxylate 11.
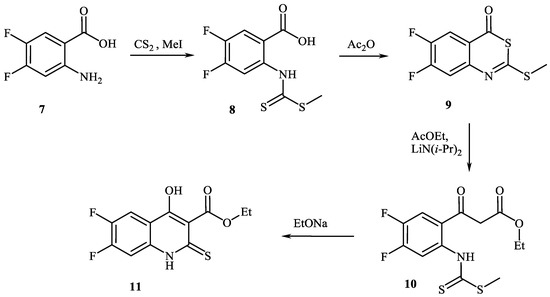
Scheme 2.
Synthesis of 6,7-difluoro-4-hydroxy-2-thioxo-1.2-dihydroquinoline-3-carboxylate 11 starting from anthranilic acid derivative.
The interaction of the methyl 2-isothiocyanatobenzoate 12 with primary amines or hydrazine derivatives is a high-performance method for synthesis of 3-substituted 4-oxo-2-thioxo-1,2,3,4-tetrahydroquinazolines containing various substituents in 3 position of quinazoline ring (alkyl, aryl, hetaryl, etc.) in mild condition with high yields [8,9,10,11,12,13,14,15,16,17,18]. In continuation of these studies, we investigated the interaction of methyl 2-isothiocyanatobenzoate 12 with C-nucleophiles on the example of malonic ester.
2. Results and Discussions
By analogy with N-nucleophiles [8,9,10,11,12,13,14,15,16,17,18], malonic ester as C-nucleophile reagent could also react with methyl 2-isothiocyanatobenzoate 12 in relatively mild condition, directly producing 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate structure, which would be useful for development of new effective antibacterial drugs.
2.1. Synthesis
Trivial reflux of methyl 2-isothiocyanatobenzoate 12 and dimethyl malonate in usual organic solvents (MeOH, THF, dioxane, DMF) did not lead to significant changes in the reaction medium. When this reaction was carried out in methanol with the presence of sodium methanolate, it was found that the reaction process was completed at reflux for 4 h. After neutralization of the reaction mixture with AcOH, the precipitate containing 2 products was formed. These pure substances were isolated by fractional crystallization from aqueous MeOH. The major product was methyl 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate 13 with yield of 70% and the minor was methyl 2-(methoxycarbonothioylamino)benzoate 14 with yield of 20% (Scheme 3).

Scheme 3.
The reaction of methyl 2-isothiocyanatobenzoate 12 with dimethyl malonate.
Control reflux of methyl 2-isothiocyanatobenzoate 12 in MeOH with the presence of sodium methanolate for 4 h led to methyl 2-(methoxycarbonothioylamino)benzoate 14 with yield of 85%. To avoid the formation of a by-product 14, we obtained pure sodium dimethyl malonate preliminary, and the reaction of methyl 2-isothiocyanatobenzoate 12 with preliminary obtained sodium dimethyl malonate was carried out in an aprotic solvent dioxane at reflux for 4 h. After acidification of the reaction mixture with AcOH, pure methyl 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate 13 was isolated with yield of 67%.
The purity and structures of methyl 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate 13 were confirmed by LC/MS data, which presented the signal of [M + H]+ ion at 236.2 and signal of decomposition product at 204.2 (Figure S3). The only one peak of OCH3 group as a singlet at 3.78 ppm and a singlet of the thioamide proton at 13.14 ppm that is characteristic for cyclic thioamides, in 1H NMR spectrum of the product 13 are the confirmation of this structure also. However, the peak of the OH proton at 11.20 ppm was very broad. This may indicate the presence of strong intramolecular hydrogen bond (Figure S1). Accordingly, the sole peak of the carbon atom of OCH3 group at 51.5 ppm both and the peak of the carbon atom of thioamide fragment at 177.7 ppm in 13C NMR spectrum of the product 13 are proof of this structure too (Figure S2). The presence of thioamide fragments is confirmed by occurrence the absorption band at 379 nm in UV/Vis spectrum of compound 13 (Figure S4). The interesting feature of compound 13 takes place in its IR spectrum. The strong absorption band of the ester carbonyl group did not appear near 1700 cm−1 but was drastically shifted to 1626 cm−1. As well, absorption band of the OH group usually near 3400 cm−1 is very weak and practically did not appear (Figure S5). Such behavior indicates the presence of strong intramolecular hydrogen bond between the OH group and the oxygen atom of the C=O group. X-ray analysis data given below confirm this assumption. (The spectral data may be accessed in the Supplementary Materials).
1H NMR spectrum of the methyl 2-(methoxycarbonothioylamino)benzoate 14 shows two distinct peaks of OCH3 group as a singlets at 3.81 ppm and 3.94 ppm and peak of the thioamide proton at 11.23 ppm (Figure S6). Different peaks of carbon atoms of OCH3 group at 52.3 ppm and 57.4 ppm in 13C NMR spectrum of the product 14 are proof of this structure also (Figure S7). The peak of C=S carbon atom is shifted to 188.8 ppm, which can be explained by the presence of acceptor OCH3 group. The signal of molecular [M + H]+ ion at 226.2 in LC/MS spectrum of the product 14 is corresponding for this structure (Figure S8). In UV/Vis spectrum of compound 14 the absorption band, which is connected with C=S group appears at 315 nm (Figure S9). In IR spectrum of compound 14 the strong absorption band of the ester carbonyl group appears at 1686 cm−1 (Figure S10), As well, absorption band of the NH group are present at 3447 cm−1 and at 3256 cm−1, which are additional confirmation of its structure.
2.2. X-ray Analysis
The final structure of synthesized compounds was confirmed by X-ray analysis (Figure 1, Figure 2 and Figure 3).

Figure 1.
Molecular structure of compound 13 according to X-ray diffraction data. Thermal displacement ellipsoids are shown at the 50% probability level.

Figure 2.
Centrosymmetric dimer of molecules 13 in the crystal phase. Hydrogen bonds are shown by dashed blue lines.

Figure 3.
Molecular structure of compound 14 according to X-ray diffraction data. Thermal displacementellipsoids are shown at the 50% probability level.
All non-hydrogen atoms of compound 13 lie within the plane with accuracy of 0.02 Ǻ. Such a conformation is stabilized additionally by the O1-H…O2 strong intramolecular hydrogen bond (H…O 1.45 Ǻ, O-H…O 161°). This hydrogen bond causes also some electron density redistribution within this fragment: the C10-O2 (1.234(5) Ǻ) and C7-C8 (1.389(4) Ǻ) bonds are elongated as compared to their mean values [19] 1.210 Ǻ and 1.326 Ǻ respectively while the C7-O1 (1.340(5) Ǻ) bond is shortened (its mean value is 1.363 Ǻ) (Figure 1).
In the crystal phase molecules 13 form centrosymmetric dimers which are bound by the N1-H…S1′ (-x,1-y,1-z) intermolecular hydrogen bond (H…S 2.53 Ǻ, N-H…S 161°) (Figure 2).
The colourless crystals of methyl 2-(methoxycarbonothioylamino)benzoate 14 are monoclinic with one molecule in the independent part of the unit cell. The substituent at C1 atom has ap-conformation relatively phenyl cycle and it is turned relatively the C1-C2 endocyclic bond (the C2-C1-N1-C7 torsion angle is 30.5(8)°). Such orientation is stabilized by the formation of the C2-H2…S1 (H…S 2.67 Å, C-H…S 119°) intramolecular hydrogen bond. The carbonyl group of the substituent at the C6 atom is slightly turned relatively the C1-C6 endocyclic bond (the C1-C6-C9-O1 torsion angle is -15.0(8)°) due to steric repulsion between O2 oxygen atom of the substituent and hydrogen atoms of phenyl cycle (the shortened intramolecular contact is: O2…H5 2.36 Å (2.45 Å)) on the one hand and by the formation of the N1-H1…O1 intramolecular hydrogen bond (H…O 2.05 Å, N-H…O 130°), on the other hand (Figure 3).
In the crystal phase molecules 14 form zigzag chains along [0 1 0] crystallographic direction due to the C10-H10B…S1′ (0.5 + x, 0.5 − y, 0.5 + z) intermolecular hydrogen bond (H…S 2.99 Å, C-H…S 161°). The neighboring chains are bound by the C5-H5…C5π’ (0.5 − x, 0.5 + y, 1.5 − z) and N1-H1…O2′ (x, −1 + y, z) hydrogen bonds (H…O 2.62 Å, N-H…O 128°).
3. Materials and Methods
3.1. General Information
All NMR spectra were recorded on a Varian MR-400 spectrometer (Varian, Inc., Walnut Creek, CA, USA) with standard pulse sequences operating at 400 MHz for 1H NMR and 100 MHz for 13C NMR. For all NMR spectra, DMSO-d6 was used as solvent. Chemical shift values are referenced to residual protons (δ 2.49 ppm) and carbons (δ 39.6 ppm) of the solvent as an internal standard. Elemental analysis was performed on EuroEA-3000 CHNS-O Analyzer (Euro Vector, Milan, Italy). Melting points were measured with a Buchi B-520 melting point apparatus (Buchi AG, Flawil, Switzerland). LC/MS spectra were recorded with ELSD Alltech 3300 liquid chromatograph (Buchi AG, Flawil, Switzerland) equipped with a UV detector (λmax 254 nm), API-150EX mass-spectrometer and using a Zorbax SB-C18 column, Phenomenex (100 × 4 mm) Rapid Resolution HT Cartrige 4.6 × 30 mm, 1.8-Micron. Elution started with 0.1 M solution of HCOOH in water and ended with 0.1 M solution of HCOOH in acetonitrile used a linear gradient at a flow rate of 0.15 mL/min and an analysis cycle time of 25 min. UV/Vis spectra of solutions in CH3CN were recorded on a Specord 200 spectrometer (Analytik Jena AG, Jena, Germany). IR spectra in KBr pellets were recorded on a Bruker Vertex 70 FTIR spectrometer (Bruker Optik GmbH, Ettlingen, Germany).
3.2. Synthesis
Starting methyl 2-isothiocyanatobenzoate 12 and dimethyl malonate, as well as NaOH and solvents are commercially available and were reagent grade.
3.2.1. Reaction of Methyl 2-Isothiocyanatobenzoate 12 with Dimethyl Malonate with the Presence of Sodium Methanolate
The mixture of methyl 2-isothio-cyanatobenzoate 12 (19.3 g, 0.1 mol) and dimethyl malonate (12.5 mL, 0.11 mol) was added to the solution of MeONa (from 5.06 g, 0.22 mol Na) in MeOH (400 mL) with stirring. The reaction mixture was refluxed for 4 h. Then NaOH (3.6 g, 0.09 mol) was added and the reaction mixture was stirred at 50 °C for 2 h. After cooling, a solution of AcOH (18 mL, 0.32 mol) in 100 mL of water was added. The precipitate that formed was filtered, washed with water (50 mL) and fractional crystallized from methanol (100 mL). Yield of methyl 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate 13 16.4 g (70%) and yield of methyl 2-(methoxycarbonothioylamino)benzoate 14 4.57 g (20%).
3.2.2. Synthesis of Methyl 4-Hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate 13
The mixture of methyl 2-isothiocyanatobenzoate 12 (19.3 g, 0.1 mol) and sodium dimethyl malonate (15.4 g, 0.1 mol) in dry dioxane (400 mL) was refluxed for 4 h with stirring. After cooling, a solution of AcOH (6.2 mL, 0.1 mol) in 200 mL of water was added. The precipitate that formed was filtered, washed with water (50 mL) and recrystallized from methanol (100 mL). Yield 15.7 g (67%), white solids, m.p. 187–188 °C. 1H NMR spectrum δ, ppm (J, Hz): 3.78 (3H, s, OCH3); 7.33 (1H, t, J = 7.4, H Ar); 7.57–7.67 (2H, m, H Ar); 8.14 (1H, d, J = 8.0, H Ar); 11.20 (1H, very broad, OH); 13.14 (1H, s, NH). 13C NMR spectrum, δ, ppm: 51.5 (OCH3); 116.4; 117.0; 118.9; 123.6; 123.7; 132.5; 140.0; 156.5; 165.8 (CO); 177.7 (CS). LC/MS m/z (%): 236.2 [M + H]+ (90.0); 204.2 (100). UV (CH3CN) λmax (ε): 233 nm (30300); 294 nm (22500); 324 nm (12000); 379 nm (5800). IR (KBr, ν cm−1): 3136 (w) (N-H), 3080 (w) (N-H), 3009 (m) (C-H), 2952 (m) (C-H), 1626 (vs) (C=O), 1568 (vs), 1491 (s), 1439 (s), 1334 (m) (C=S). Anal. calcd. for C11H9NO3S, %: C 56.16; H 3.86; N 5.95; S 13.63. Found, %: C 56.28; H 3.90; N 6.03; S 13.58.
3.2.3. Synthesis of Methyl 2-(Methoxycarbonothioylamino)benzoate 14
Methyl 2-isothiocyanatobenzoate 12 (19.3 g, 0.1 mol) was added to the solution of MeONa (from 5.06 g, 0.22 mol Na) in dry MeOH (400 mL) with stirring. The reaction mixture was refluxed for 4 h. After cooling, a solution of AcOH (12.4 mL, 0.22 mol) in 100 mL of water was added. The precipitate that formed was filtered, washed with water (50 mL) and recrystallized from methanol (100 mL). Yield 19.1 g (85%), white solids, m.p. 67–68 °C. 1H NMR spectrum δ, ppm (J, Hz): 3.81 (3H, s, OCH3); 3.94 (3H, s, OCH3); 7.33 (1H, t, J = 8.0, H Ar); 7.57–7.63 (2H, m, H Ar); 7.84 (1H, d, J = 8.0, H Ar); 11.23 (1H, s, NH). 13C NMR spectrum, δ, ppm: 52.3 (OCH3); 57.4 (OCH3); 123.3; 125.0; 125.5; 130.3; 133.0; 137.5; 166.6 (CO); 188.8 (CS). LC/MS m/z (%): 226.2 [M + H]+ (100). UV (CH3CN) λmax (ε): 209 nm (23000); 238 nm (15100); 281 nm (14900); 315 nm (9100). IR (KBr, ν cm−1): 3447 (s) (N-H), 3256 (s) (N-H), 2940 (m) (C-H), 1686 (vs) (C=O), 1587 (s), 1529 (vs), 1379 (s) (C=S), 1268 (vs). Anal. calcd. for C10H11NO3S, %: C 53.32; H 4.92; N 6.22; S 14.23. Found, %: C 53.42; H 4.94; N 6.23; S 14.18.
3.3. X-ray Diffraction Study
3.3.1. Methyl 4-Hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate 13
Single crystals for X-ray diffraction study were grown from MeOH. The colourless crystals of 13 (C11H9NO3S) are triclinic. At 293° Ka = 4.5062(7), b = 10.812(1), c = 12.198(1) Å, α = 115.73(1)°, β = 93.12(1)°, γ = 99.73(1), V = 522.2(1) Å3, Mr = 235.25, Z = 2, space group P, dcalc = 1.496 g/cm3, μ(MoKα) = 0.299 mm−1, F(000) = 244. Intensities of 3308 reflections (1778 independent, Rint = 0.116) were measured on the “Xcalibur-3” diffractometer (graphite monochromated MoKα radiation, CCD detector, ω-scaning, 2Θmax = 50°). The structure was solved by direct method using SHELXTL package [20]. Positions of the hydrogen atoms were located from electron density difference maps and refined by “riding” model with Uiso = nUeq (n = 1.5 for methyl group and n = 1.2 for other hydrogen atoms) of the carrier atom. Hydrogen atoms taken part in the hydrogen bond formation were refined using isotropic approximation. Full-matrix least-squares refinement against F2 in anisotropic approximation for non-hydrogen atoms using 1778 reflections was converged to wR2 = 0.071 (R1 = 0.053 for 1024 reflections with F > 4σ(F), S = 0.804). The final atomic coordinates, and crystallographic data for molecule 13 have been deposited to with the Cambridge Crystallographic Data Centre, 12 Union Road, CB2 1EZ, UK (fax: +44-1223-336033; e-mail: deposit@ccdc.cam.ac.uk) and are available on request quoting the deposition numbers CCDC 1954902).
3.3.2. Methyl 2-(Methoxycarbonothioylamino)benzoate 14
The colourless crystals of 14 (C10H11NO3S) are monoclinic. At 293° Ka = 14.042(4) Å, b = 5.5052(14) Å, c = 14.226(5) Å, β = 101.64(3)°, V = 1077.2(5) Å3, Mr = 225.26, Z = 4, space group P21/n, dcalc = 1.389 g/cm3, μ(MoKα) = 0.286 mm−1, F(000) = 472. Intensities of 7077 reflections (1896 independent, Rint = 0.132) were measured on the “Xcalibur-3” diffractometer (graphite monochromated MoKα radiation, CCD detector, ω-scaning, 2Θmax = 50°). The structure was solved by direct method using SHELXTL package [20]. Positions of the hydrogen atoms were located from electron density difference maps and refined by “riding” model with Uiso = nUeq (n = 1.5 for methyl group and n = 1.2 for other hydrogen atoms) of the carrier atom. Hydrogen atoms taken part in the hydrogen bond formation were refined using isotropic approximation. Full-matrix least-squares refinement against F2 in anisotropic approximation for non-hydrogen atoms using 1896 reflections was converged to wR2 = 0.152 (R1 = 0.077 for 846 reflections with F > 4σ(F), S = 0.949). The final atomic coordinates, and crystallographic data for molecule 13 have been deposited to with the Cambridge Crystallographic Data Centre, 12 Union Road, CB2 1EZ, UK (fax: +44-1223-336033; e-mail: deposit@ccdc.cam.ac.uk) and are available on request quoting the deposition numbers CCDC 1954903).
4. Conclusions
We have suggested a facile one-pot method of synthesis of methyl 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate by condensation of methyl 2-isothiocyanatobenzoate and methyl malonate. According single crystal X-ray diffraction data, there is the strong intramolecular hydrogen bond between the hydroxy group and the carbonyl oxygen atom of the ester group in methyl 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate and the crystal structure of the product was stabilized through the intermolecular hydrogen bonds between the NH group and the sulfur atom of another molecule.
Supplementary Materials
The following are available online: Figure S1. 1H NMR spectrum (400 MHz, DMSO-d6) of methyl 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate 13; Figure S2. 13C NMR spectrum (100 MHz, DMSO-d6) of methyl 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate 13; Figure S3. LC/MS data for methyl 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate 13; Figure S4. UV/Vis spectrum (CH3CN) of methyl 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate 13; Figure S5. IR spectrum of methyl 4-hydroxy-2-thioxo-1,2-dihydroquinoline-3-carboxylate 13 (KBr pellet); Table S1. Coordinates (×104) and equivalent isotropic thermal parameters (Å2 × 103) of atoms in structure 13; Table S2. Bond lengths (Å) in structure 13; Table S3. Valence angles (deg.) in structure 13; Figure S6. 1H NMR spectrum (400 MHz, DMSO-d6) of methyl 2-(methoxycarbonothioylamino)benzoate 14; Figure S7. 13C NMR spectrum (100 MHz, DMSO-d6) of methyl 2-(methoxycarbonothioylamino)benzoate 14; Figure S8. LC/MS data for methyl 2-(methoxycarbonothioylamino)benzoate 14; Figure S9. UV/Vis spectrum (CH3CN) of methyl 2-(methoxycarbonothioylamino)benzoate 14; Figure S10. IR spectrum of methyl 2-(methoxycarbonothioylamino)benzoate 14 (KBr pellet); Table S4. Coordinates (×104) and equivalent isotropic thermal parameters (Å2 × 103) of atoms in structure 14; Table S5. Bond lengths (Å) in structure 14; Table S6. Valence angles (deg.) in structure 14.
Author Contributions
Conceptualization, S.M.K. and D.V.K.; synthesis of compounds, I.O.M., NMR spectra characterization and X-ray analysis, I.S.K.; writing—original draft preparation, O.G.D. and S.M.K.; writing—review and editing, O.D.M.; supervision and project administration, A.V.I.; funding acquisition, D.V.K.
Funding
The work was supported by the Ministry of Science and Higher Education of the Russian Federation in frames of agreement on reimbursement of costs associated with the development of a platform for biologically active compound libraries and the design of actual biotargets, including platform testing on the example of invention and preparation of candidate libraries for HBV treatment designed as inhibitors of viral penetration and assembly of viral core particles (RFMEFI57917 × 0154).
Acknowledgments
The authors wish to thank the Ministry of Science and Higher Education of the Russian Federation for their financial support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kazuno, K.; Kise, M.; Kitano, M.; Ozaki, M.; Segawa, J.; Shirahase, I.; Tomii, Y. Preparation of Oxothiazetoquinolinecarboxylates as Bactericides. GB Patent 2190376B, 18 November 1987. [Google Scholar]
- Taguchi, M.; Kondo, H.; Inoue, Y.; Kawahata, Y.; Tsukamoto, G. Epoxymethanothiazoloquinolonecarboxylic Acid Derivatives as Medical Bactericides, Their Preparation, and Formulations Containing Them. EPA 286089A1, 12 October 1988. [Google Scholar]
- Kise, M.; Kitano, M.; Ozaki, M.; Kazuno, K.; Matsuda, M.; Shirahase, I.; Segawa, J. Preparation and Testing of 6-Fluoro-7-piperazino-4-oxo-4H-[1,3]thiazeto[3,2-a]quinolinecarboxylate Derivatives as Antibactericides. EPA 315828A1, 17 May 1989. [Google Scholar]
- Segawa, J.; Kitano, M.; Kazuno, K.; Tsuda, M.; Shirahase, I.; Ozaki, M.; Matsuda, M.; Kise, M. Studies on pyridonecarboxylic acids. 2. Synthesis and antibacterial activity of 8-substituted 7-fluoro-5-oxo-5H-thiazolo[3,2-a]quinoline-4-carboxylic acids. J. Heterocycl. Chem. 1992, 29, 1117–1123. [Google Scholar] [CrossRef]
- Segawa, J.; Kazuno, K.; Matsuoka, M.; Shirahase, I.; Ozaki, M.; Matsuda, M.; Tomii, Y.; Kitano, M.; Kise, M. Studies on pyridonecarboxylic acids. III. Synthesis and antibacterial activity evaluation of 1,8-disubstituted 6-fluoro-4-oxo-7-piperazinyl-4H-[1,3]thiazeto[3,2-a]quinoline-3-carboxylic acid derivatives. Chem. Pharm. Bull. 1995, 43, 63–70. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ahmed, A.; Daneshtalab, M. Polycyclic quinolones (part 1) - Thieno[2,3-b]benzo[h]quinoline derivatives: Design, synthesis, preliminary in vitro and in silico studies. Heterocycles 2012, 85, 103–122. [Google Scholar] [CrossRef]
- Segawa, J. Process for Producing Quinolinecarboxylic Acid Derivative. WO 9404506A1, 3 March 1994. [Google Scholar]
- Ivachtchenko, A.; Kovalenko, S.; Drushlyak, O. Synthesis of substituted 4-oxo-2-thioxo-1,2,3,4-tetrahydroquinazolines and 4-oxo-3,4-dihydroquinazoline-2-thioles. J. Comb. Chem. 2003, 5, 775–788. [Google Scholar] [CrossRef] [PubMed]
- Urleb, U. The reactions of heterocyclic isothiocyanates bearing an ortho ester group with N-nucleophiles. The scope and some limitations of the reaction. J. Heterocycl. Chem. 1995, 32, 69–71. [Google Scholar] [CrossRef]
- Ghorab, M.M.; Ragab, F.A.; Heiba, H.I.; Bayomi, A.A. Utility of methyl 2-isothiocyanatobenzoate in the synthesis of some new quinazoline derivatives as potential anticancer and radiosensitizing agents. Arzneimittelforschung 2011, 61, 719–726. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Ganesh, T.; Diebold, B.A.; Zhu, Y.; McCoy, J.W.; Smith, S.M.E.; Sun, A.; Lambeth, J.D. Thioxo-dihydroquinazolin-one Compounds as Novel Inhibitors of Myeloperoxidase. ACS Med. Chem. Lett. 2015, 6, 1047–1052. [Google Scholar] [CrossRef] [PubMed]
- Cherbuliez, E.; Willhalm, B.; Jaccard, S.; Rabinowitz, J. Formation and rearrangement of esters. LXXVIII. Reaction of o-methoxycarbonylphenylisothiocyanate with diamines and hydrazine, and cyclization of some N-phenyl-N’-hydroxyalkylthioureas. Helv. Chim. Acta 1967, 50, 2563–2569. [Google Scholar] [CrossRef]
- Pathak, U.S.; Alagarsamy, V. Synthesis and pharmacological investigation of some 2-substituted [1,3,4]thiadiazolo[2,3-b]quinazolin-5(4H)-ones as antihypertensive agents. Indian Drugs 2000, 37, 51–55. [Google Scholar]
- Gakhar, H.K.; Shama, A.; Gill, G.K. Thiazolo[2’,3’:3,4]-s-triazolo[5,1-b]quinazolin-10-ones. J. Indian Chem. Soc. 1983, 60, 803–804. [Google Scholar]
- Verma, J.K. Coordination polymers of cobalt(II), nickel(II), copper(II), zinc(II) and cadmium(II) with 3-allyl-1-(2-mercapto-4-oxo-3(4H)-quinazolinyl)-2-thiopseudourea. Curr. Sci. 1987, 56, 1168–1171. [Google Scholar]
- Il’chenko, O.V.; Zaremba, O.V.; Kovalenko, S.M.; Sherakov, A.A.; Chernykh, V.P. Synthesis of 3-Substituted 2-Thioxo-2,3-dihydro-1H-benzofuro[3,2-d]pyrimidin-4(1H)-ones. Synth. Commun. 2007, 37, 2559–2568. [Google Scholar] [CrossRef]
- Tkachenko, O.V.; Vlasov, S.V.; Kovalenko, S.M.; Zhuravel, I.O.; Chernykh, V.P. Synthesis and the antimicrobial activity of ethyl 5-methyl-2-(alkylthio)-4-oxo-3,4-dihydrothieno [2,3-d]pyrimidine-6-carboxylates. Zh. Org. Farm. Khim. 2013, 11, 9–15. [Google Scholar] [CrossRef]
- Parkhomenko, O.O.; Kovalenko, S.M.; Chernykh, V.P.; Osolodchenko, T.P. Synthesis and antimicrobial activity of 5-oxo-1-thioxo-4,5-dihydro[1,3]thiazolo[3,4-a]quinazolines. ARKIVOC 2005, 8, 82–88. [Google Scholar] [CrossRef]
- Burgi, H.-B.; Dunitz, J.D. Structure Correlation; VCH: Weinheim, Germany, 1994; Volume 2, p. 741. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. 2008, A64, 112–122. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).