5-Amino-3-(diethylamino)-5H-benzo[4,5]imidazo[1,2-b][1,2,4,6]thiatriazine 1,1-Dioxide
Abstract
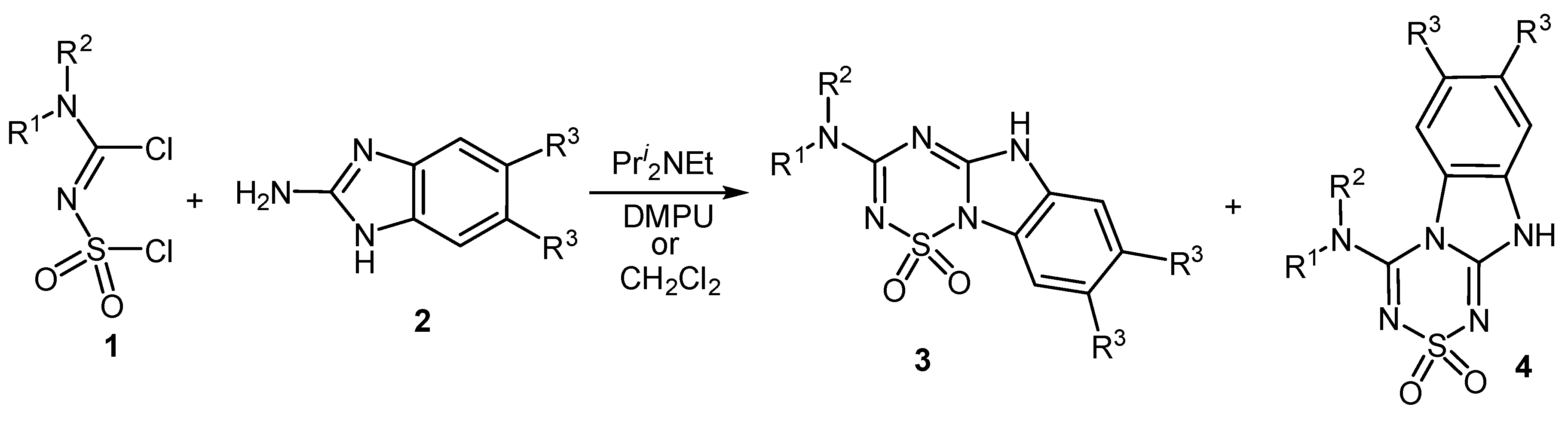
1. Introduction
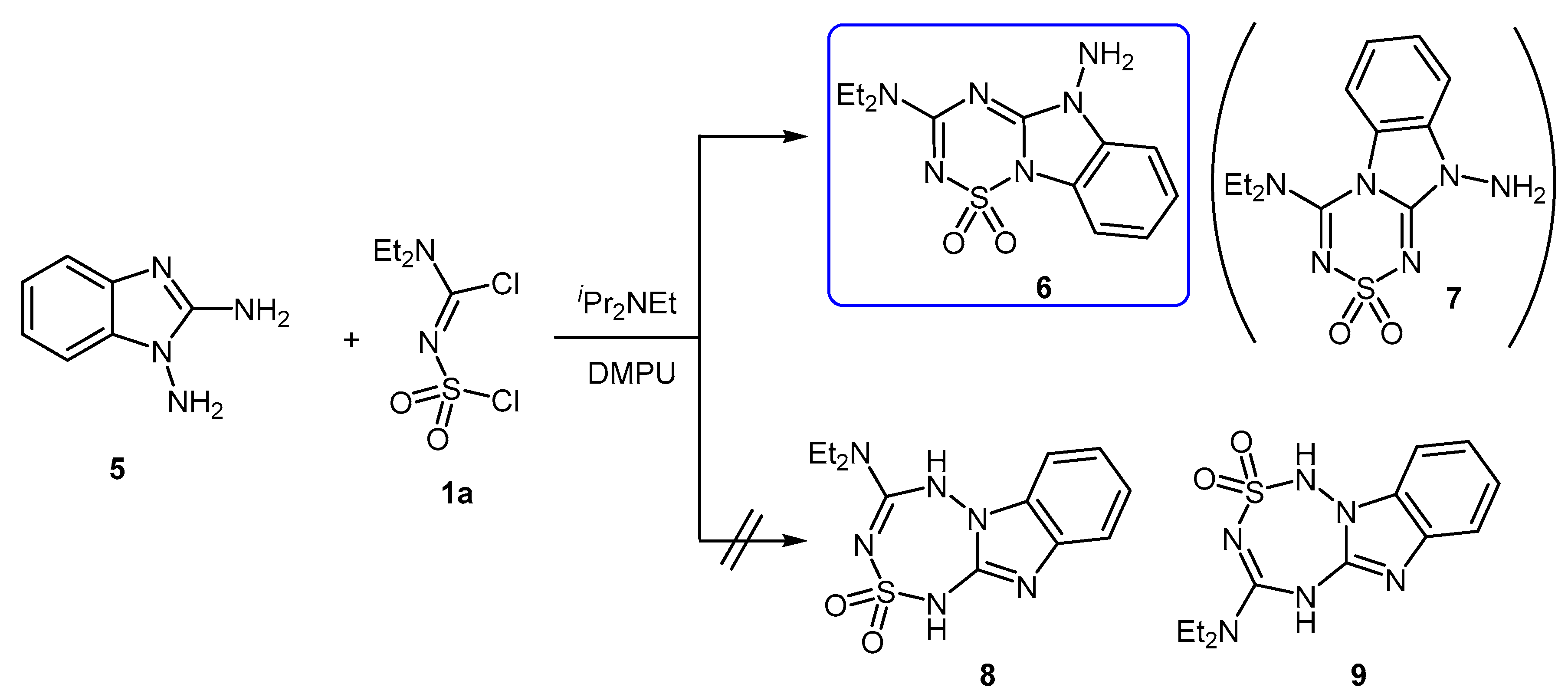
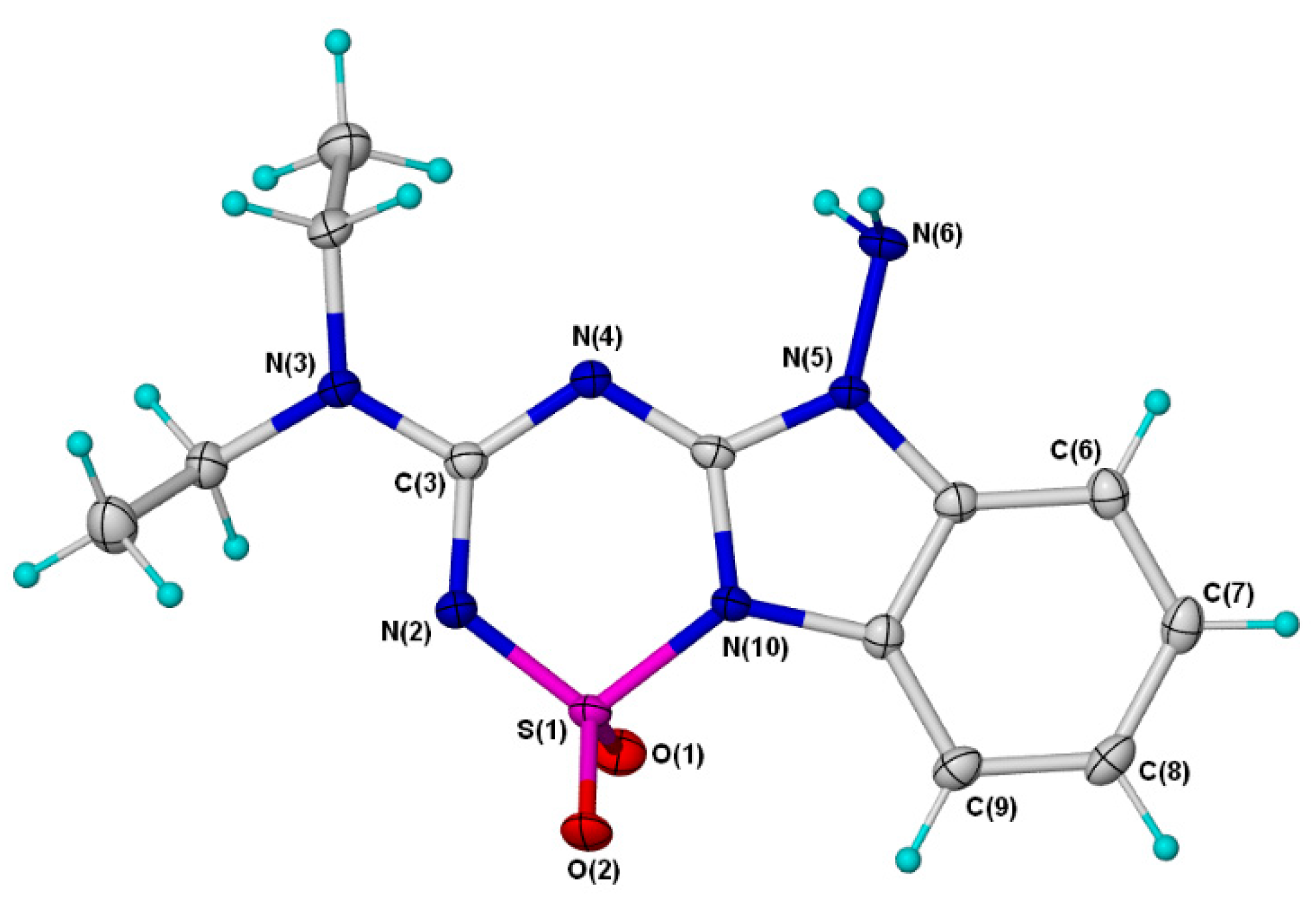
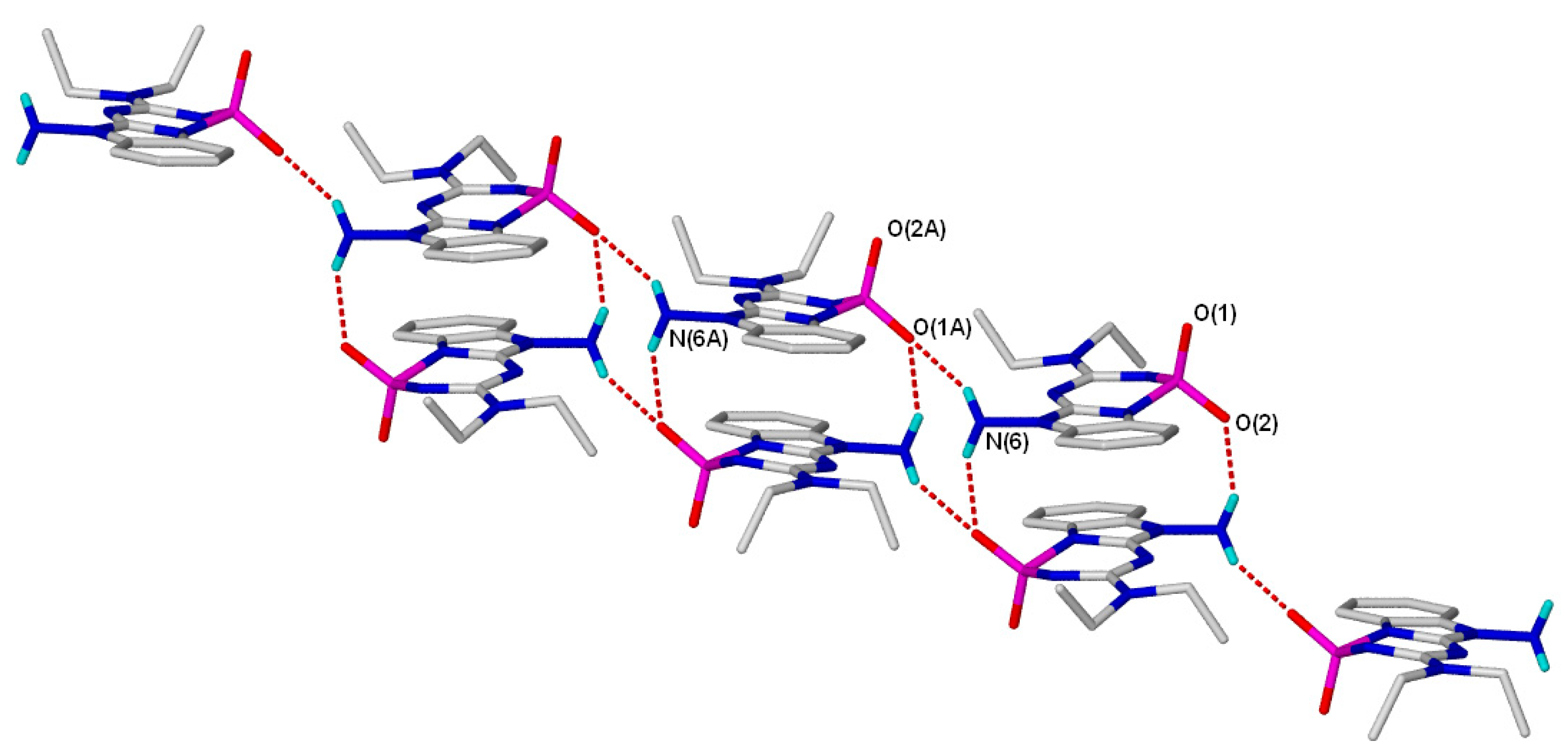
2. Results and Discussion
3. Experimental Section
3.1. Materials and Methods
3.2. 5-Amino-3-(diethylamino)-5H-benzo[4,5]imidazo[1,2-b][1,2,4,6]thiatriazine 1,1-Dioxide (6)
3.3. X-ray Crystallography
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Katritzky, A.R. Introduction: Heterocycles. Chem. Rev. 2004, 104, 2125. [Google Scholar] [CrossRef]
- Francis, C.L. New heteroatom-rich ring systems from N,N-dialkyl-N’-chlorosulfonyl chloroformamidines. Arkivoc 2016, 212–275. [Google Scholar] [CrossRef]
- Cablewski, T.; Francis, C.L.; Liepa, A.J. N,N-Dialkyl-N’-Chlorosulfonyl Chloroformamidines in Heterocyclic Synthesis. V. The Preparation of some Benzimidazo-Fused [1,2,4,6]Thiatriazine Dioxides. Aust. J. Chem. 2008, 61, 59–65. [Google Scholar] [CrossRef]
- Rastogi, R.; Sharma, S. 2-Aminobenzimidazoles in Organic Syntheses. Synthesis 1983, 861–882. [Google Scholar] [CrossRef]
- Vandyshev, D.Y.; Shikhaliev, K.S.; Kokonova, A.V.; Potapov, A.Y.; Kolpakova, M.G.; Sabynin, A.L.; Zubkov, F.I. A novel method for the synthesis of pyrimido[1,2-a]benzimidazoles. Chem. Heterocycl. Compd. 2016, 52, 493–497. [Google Scholar] [CrossRef]
- Vandyshev, D.Y.; Shikhaliyev, H.S.; Potapov, A.Y. Interaction of 1,2-diaminobenzimidazole with N-arylimides. Eur. Chem. Bull. 2015, 4, 424–427. [Google Scholar] [CrossRef]
- Romano, C.; Cuesta, E.; Avendano, C. Reactions of 1,2-diaminobenzimidazoles with α-dielectrophiles: synthesis of pyrimido[1,2-a]benzimidazole derivatives. Heterocycles 1990, 31, 267–276. [Google Scholar] [CrossRef]
- Gnidets, V.P.; Kruglenko, V.P.; Povstyanoi, M.V.; Klyuev, N.A. Condensed imidazo-1,2,4-azines. 31. Synthesis and chemical transformations of substituted 1,2,4-triazepino[2,3-a]benzimidazoles. Chem. Heterocycl. Compd. 2002, 38, 598–606. [Google Scholar] [CrossRef]
- Atmospheric Solids Analysis Probe (ASAP®) Mass Spectrometry. Available online: http://www.asap-ms.com/ (accessed on 10 September 2018).
- Petucci, C.; Diffendal, J. Atmospheric solids analysis probe: A rapid ionization technique for small molecule drugs. J. Mass Spectrom. 2008, 43, 1565–1568. [Google Scholar] [CrossRef] [PubMed]
- Ray, A.D.; Hammond, J.; Major, H. Molecular ions and protonated molecules observed in the atmospheric solids analysis probe analysis of steroids. Eur. J. Mass Spectrom. 2010, 16, 169–174. [Google Scholar] [CrossRef] [PubMed]
- COLLECT: Program for Data Collection; Bruker AXS: Madison, WI, USA, 1997–2004.
- Otwinoski, Z.; Minor, W. [20] Processing of X-ray Diffraction Data Collected in Oscillation Mode. Methods Enzymol. 1997, 276, 307–326. [Google Scholar]
- Blessing, R.H. An empirical correction for absorption anisotropy. Acta Crystallogr. Sect. A 1995, 51, 33–38. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. Sect. C 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tran, V.; Forsyth, C.M.; Francis, C.L. 5-Amino-3-(diethylamino)-5H-benzo[4,5]imidazo[1,2-b][1,2,4,6]thiatriazine 1,1-Dioxide. Molbank 2018, 2018, M1018. https://doi.org/10.3390/M1018
Tran V, Forsyth CM, Francis CL. 5-Amino-3-(diethylamino)-5H-benzo[4,5]imidazo[1,2-b][1,2,4,6]thiatriazine 1,1-Dioxide. Molbank. 2018; 2018(3):M1018. https://doi.org/10.3390/M1018
Chicago/Turabian StyleTran, Victor, Craig M. Forsyth, and Craig L. Francis. 2018. "5-Amino-3-(diethylamino)-5H-benzo[4,5]imidazo[1,2-b][1,2,4,6]thiatriazine 1,1-Dioxide" Molbank 2018, no. 3: M1018. https://doi.org/10.3390/M1018
APA StyleTran, V., Forsyth, C. M., & Francis, C. L. (2018). 5-Amino-3-(diethylamino)-5H-benzo[4,5]imidazo[1,2-b][1,2,4,6]thiatriazine 1,1-Dioxide. Molbank, 2018(3), M1018. https://doi.org/10.3390/M1018




