Betulinic Acid for Cancer Treatment and Prevention
Abstract
1. Introduction
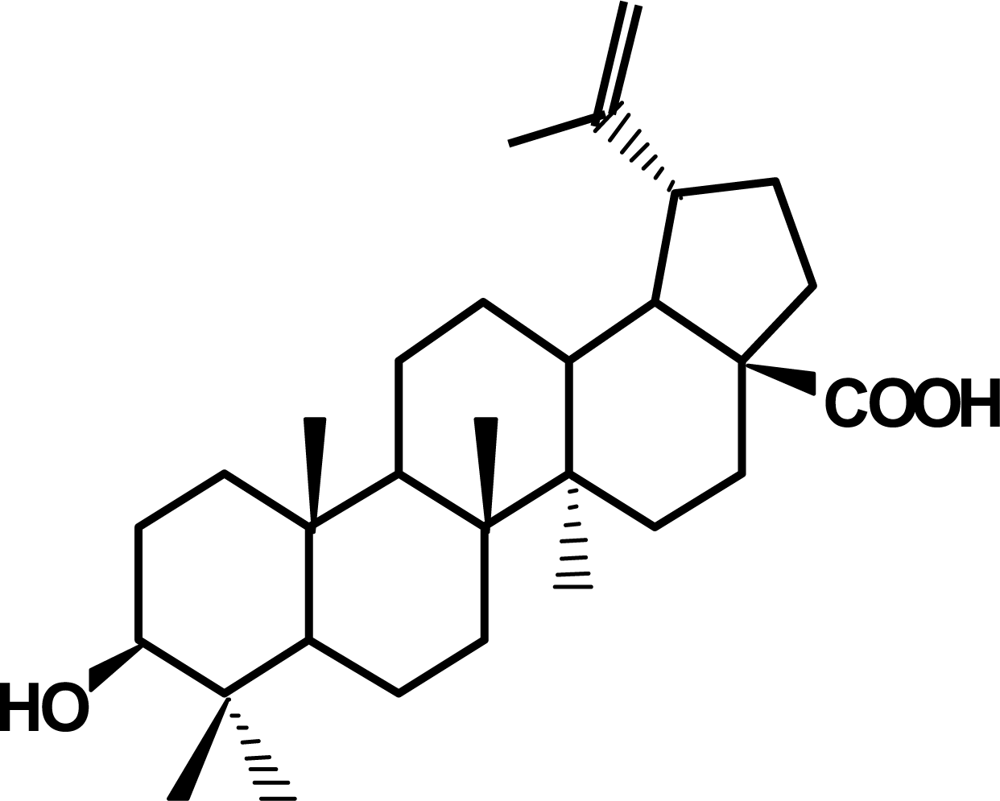
2. Betulinic acid, a phytochemical with antitumor activity
3. Mechanisms of action of betulinic acid
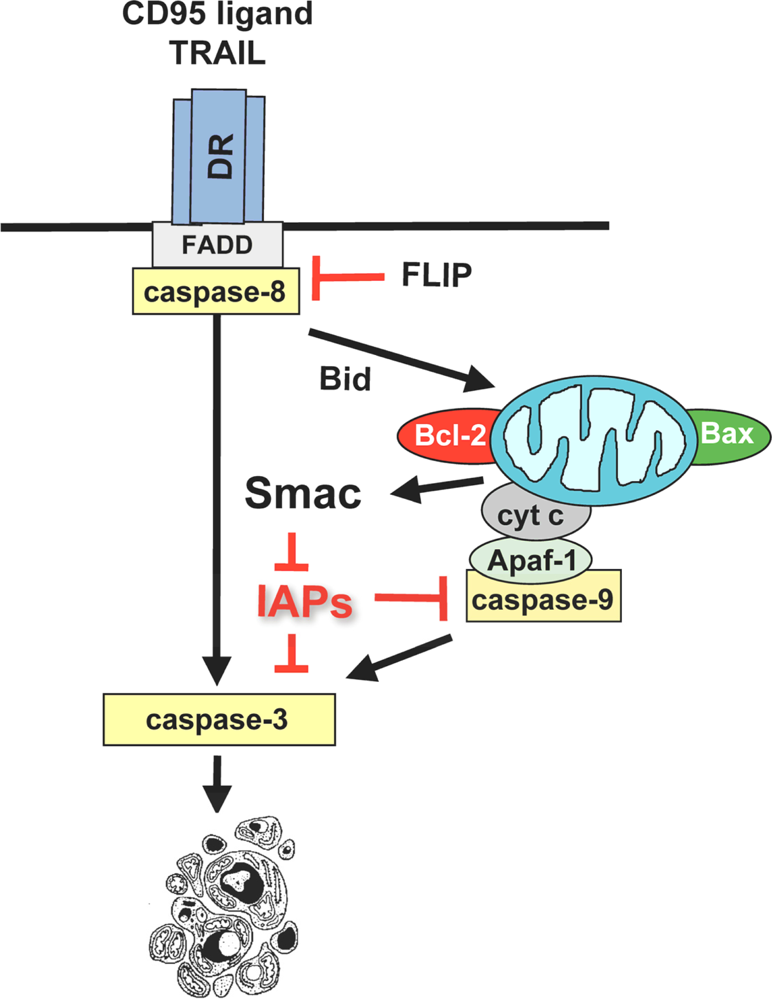
3.1. Activation of the mitochondrial pathway by anticancer therapeutics
3.2. Induction of mitochondrial outer membrane permeabilization by betulinic acid
3.3. Regulation of betulinic acid-induced apoptosis by Bcl-2 family proteins
3.4. Modulation of NF-κB activity by betulinic acid
4. Additional anticancer effects of betulinic acid
5. Anticancer activity of betulinic acid
6. Conclusions
Acknowledgements
References
- Newman, DJ; Cragg, GM; Snader, KM. Natural products as sources of new drugs over the period 1981–2002. J. Nat. Prod. 2003, 66, 1022–1037. [Google Scholar]
- Evan, GI; Vousden, KH. Proliferation, cell cycle and apoptosis in cancer. Nature 2001, 411, 342–348. [Google Scholar]
- Hanahan, D; Weinberg, RA. The hallmarks of cancer. Cell 2000, 100, 57–70. [Google Scholar]
- Galluzzi, L; Larochette, N; Zamzami, N; Kroemer, G. Mitochondria as therapeutic targets for cancer chemotherapy. Oncogene 2006, 25, 4812–4830. [Google Scholar]
- Cichewicz, RH; Kouzi, SA. Chemistry, biological activity, and chemotherapeutic potential of betulinic acid for the prevention and treatment of cancer and HIV infection. Med. Res. Rev. 2004, 24, 90–114. [Google Scholar]
- Alakurtti, S; Makela, T; Koskimies, S; Yli-Kauhaluoma, J. Pharmacological properties of the ubiquitous natural product betulin. Eur. J. Pharm. Sci. 2006, 29, 1–13. [Google Scholar]
- Green, DR; Kroemer, G. The pathophysiology of mitochondrial cell death. Science 2004, 305, 626–629. [Google Scholar]
- Galluzzi, L; Larochette, N; Zamzami, N; Kroemer, G. Mitochondria as therapeutic targets for cancer chemotherapy. Oncogene 2006, 25, 4812–4830. [Google Scholar]
- Fulda, S; Scaffidi, C; Susin, SA; Krammer, PH; Kroemer, G; Peter, ME; Debatin, KM. Activation of mitochondria and release of mitochondrial apoptogenic factors by betulinic acid. J. Biol. Chem. 1998, 273, 33942–33948. [Google Scholar]
- Andre, N; Carre, M; Brasseur, G; Pourroy, B; Kovacic, H; Briand, C; Braguer, D. Paclitaxel targets mitochondria upstream of caspase activation in intact human neuroblastoma cells. FEBS Lett. 2002, 532, 256–260. [Google Scholar]
- Fulda, S; Susin, SA; Kroemer, G; Debatin, KM. Molecular ordering of apoptosis induced by anticancer drugs in neuroblastoma cells. Cancer Res. 1998, 58, 4453–4460. [Google Scholar]
- Wick, W; Grimmel, C; Wagenknecht, B; Dichgans, J; Weller, M. Betulinic acid-induced apoptosis in glioma cells: A sequential requirement for new protein synthesis, formation of reactive oxygen species, and caspase processing. J. Pharmacol. Exp. Ther. 1999, 289, 1306–1312. [Google Scholar]
- Tan, Y; Yu, R; Pezzuto, JM. Betulinic acid-induced programmed cell death in human melanoma cells involves mitogen-activated protein kinase activation. Clin. Cancer Res. 2003, 9, 2866–2875. [Google Scholar]
- Fulda, S; Friesen, C; Los, M; Scaffidi, C; Mier, W; Benedict, M; Nunez, G; Krammer, PH; Peter, ME; Debatin, KM. Betulinic acid triggers CD95 (APO-1/Fas)- and p53-independent apoptosis via activation of caspases in neuroectodermal tumors. Cancer Res. 1997, 57, 4956–4964. [Google Scholar]
- Adams, JM; Cory, S. The Bcl-2 apoptotic switch in cancer development and therapy. Oncogene 2007, 26, 1324–1337. [Google Scholar]
- Selzer, E; Pimentel, E; Wacheck, V; Schlegel, W; Pehamberger, H; Jansen, B; Kodym, R. Effects of betulinic acid alone and in combination with irradiation in human melanoma cells. J. Invest. Dermatol. 2000, 114, 935–940. [Google Scholar]
- Selzer, E; Thallinger, C; Hoeller, C; Oberkleiner, P; Wacheck, V; Pehamberger, H; Jansen, B. Betulinic acid-induced Mcl-1 expression in human melanoma--mode of action and functional significance. Mol. Med. 2002, 8, 877–884. [Google Scholar]
- Thurnher, D; Turhani, D; Pelzmann, M; Wannemacher, B; Knerer, B; Formanek, M; Wacheck, V; Selzer, E. Betulinic acid: a new cytotoxic compound against malignant head and neck cancer cells. Head Neck 2003, 25, 732–740. [Google Scholar]
- Fulda, S; Debatin, KM. Betulinic acid induces apoptosis through a direct effect on mitochondria in neuroectodermal tumors. Med. Pediatr. Oncol. 2000, 35, 616–618. [Google Scholar]
- Meng, RD; El-Deiry, WS. p53-independent upregulation of KILLER/DR5 TRAIL receptor expression by glucocorticoids and interferon-gamma. Exp. Cell Res. 2001, 262, 154–169. [Google Scholar]
- Salti, GI; Kichina, JV; Das Gupta, TK; Uddin, S; Bratescu, L; Pezzuto, JM; Mehta, RG; Constantinou, AI. Betulinic acid reduces ultraviolet-C-induced DNA breakage in congenital melanocytic naeval cells: evidence for a potential role as a chemopreventive agent. Melanoma Res. 2001, 11, 99–104. [Google Scholar]
- Zuco, V; Supino, R; Righetti, SC; Cleris, L; Marchesi, E; Gambacorti-Passerini, C; Formelli, F. Selective cytotoxicity of betulinic acid on tumor cell lines, but not on normal cells. Cancer Lett. 2002, 175, 17–25. [Google Scholar]
- Ehrhardt, H; Fulda, S; Fuhrer, M; Debatin, KM; Jeremias, I. Betulinic acid-induced apoptosis in leukemia cells. Leukemia 2004, 18, 1406–1412. [Google Scholar]
- Kasperczyk, H; La Ferla-Bruhl, K; Westhoff, MA; Behrend, L; Zwacka, RM; Debatin, KM; Fulda, S. Betulinic acid as new activator of NF-kappaB: molecular mechanisms and implications for cancer therapy. Oncogene 2005, 24, 6945–6956. [Google Scholar]
- Takada, Y; Aggarwal, BB. Betulinic acid suppresses carcinogen-induced NF-kappa B activation through inhibition of I kappa B alpha kinase and p65 phosphorylation: abrogation of cyclooxygenase-2 and matrix metalloprotease-9. J. Immunol. 2003, 171, 3278–3286. [Google Scholar]
- Kwon, HJ; Shim, JS; Kim, JH; Cho, HY; Yum, YN; Kim, SH; Yu, J. Betulinic acid inhibits growth factor-induced in vitro angiogenesis via the modulation of mitochondrial function in endothelial cells. Jpn. J. Cancer Res. 2002, 93, 417–425. [Google Scholar]
- Melzig, MF; Bormann, H. Betulinic acid inhibits aminopeptidase N activity. Planta Med. 1998, 64, 655–657. [Google Scholar]
- Sjostrom, H; Noren, O; Olsen, J. Structure and function of aminopeptidase N. Adv. Exp. Med. Biol. 2000, 477, 25–34. [Google Scholar]
- Chintharlapalli, S; Papineni, S; Ramaiah, SK; Safe, S. Betulinic acid inhibits prostate cancer growth through inhibition of specificity protein transcription factors. Cancer Res. 2007, 67, 2816–2823. [Google Scholar]
- Mukherjee, R; Jaggi, M; Rajendran, P; Srivastava, SK; Siddiqui, MJA; Vardhan, A; Burman, AC. Synthesis of 3-O-acyl/3-benzylidene/3-hydrazone/3-hydrazine/17-carboxyacryloyl ester derivatives of betulinic acid as anti-angiogenic agents. Bioorg. Med. Chem. Lett. 2004, 14, 3169–3172. [Google Scholar]
- Chowdhury, AR; Mandal, S; Mittra, B; Sharma, S; Mukhopadhyay, S; Majumder, HK. Betulinic acid, a potent inhibitor of eukaryotic topoisomerase I: identification of the inhibitory step, the major functional group responsible and development of more potent derivatives. Med. Sci. Monit. 2002, 8, BR254–265. [Google Scholar]
- Rieber, M; Strasberg Rieber, M. Induction of p53 without increase in p21WAF1 in betulinic acid-mediated cell death is preferential for human metastatic melanoma. DNA Cell Biol. 1998, 17, 399–406. [Google Scholar]
- Pisha, E; Chai, H; Lee, IS; Chagwedera, TE; Farnsworth, NR; Cordell, GA; Beecher, CW; Fong, HH; Kinghorn, AD; Brown, DM; et al. Discovery of betulinic acid as a selective inhibitor of human melanoma that functions by induction of apoptosis. Nat. Med. 1995, 1, 1046–1051. [Google Scholar]
- Schmidt, ML; Kuzmanoff, KL; Ling-Indeck, L; Pezzuto, JM. Betulinic acid induces apoptosis in human neuroblastoma cell lines. Eur. J. Cancer 1997, 33, 2007–2010. [Google Scholar]
- Sarek, J; Klinot, J; Dzubak, P; Klinotova, E; Noskova, V; Krecek, V; Korinkova, G; Thomson, JO; Janost’akova, A; Wang, S; Parsons, S; Fischer, PM; Zhelev, NZ; Hajduch, M. New lupane derived compounds with pro-apoptotic activity in cancer cells: synthesis and structure-activity relationships. J. Med. Chem. 2003, 46, 5402–5415. [Google Scholar]
- Kim, JY; Koo, HM; Kim, DS. Development of C-20 modified betulinic acid derivatives as antitumor agents. Bioorg. Med. Chem. Lett. 2001, 11, 2405–2408. [Google Scholar]
- You, Y-J; Kim, Y; Nam, N-H; Ahn, B-Z. Synthesis and cytotoxic activity of A-ring modified betulinic acid derivatives. Bioorg. Med. Chem. Lett. 2003, 13, 3137–3140. [Google Scholar]
- Fulda, S; Jeremias, I; Pietsch, T; Debatin, KM. Betulinic acid: a new chemotherapeutic agent in the treatment of neuroectodermal tumors. Klin. Padiatr. 1999, 211, 319–322. [Google Scholar]
- Fulda, S; Jeremias, I; Steiner, HH; Pietsch, T; Debatin, KM. Betulinic acid: a new cytotoxic agent against malignant brain-tumor cells. Int. J. Cancer 1999, 82, 435–441. [Google Scholar]
- Fulda, S; Debatin, K-M. Sensitization for anticancer drug-induced apoptosis by betulinic Acid. Neoplasia 2005, 7, 162–170. [Google Scholar]
- Sawada, N; Kataoka, K; Kondo, K; Arimochi, H; Fujino, H; Takahashi, Y; Miyoshi, T; Kuwahara, T; Monden, Y; Ohnishi, Y. Betulinic acid augments the inhibitory effects of vincristine on growth and lung metastasis of B16F10 melanoma cells in mice. Br. J. Cancer 2004, 90, 1672–1678. [Google Scholar]
- Fulda, S; Jeremias, I; Debatin, K-M. Cooperation of betulinic acid and TRAIL to induce apoptosis in tumor cells. Oncogene 2004, 23, 7611–7620. [Google Scholar]
- Shin, YG; Cho, KH; Chung, SM; Graham, J; Das Gupta, TK; Pezzuto, JM. Determination of betulinic acid in mouse blood, tumor and tissue homogenates by liquid chromatography-electrospray mass spectrometry. J. Chromatogr. B Biomed. Sci. Appl. 1999, 732, 331–336. [Google Scholar]
- Udeani, GO; Zhao, GM; Geun Shin, Y; Cooke, BP; Graham, J; Beecher, CW; Kinghorn, AD; Pezzuto, JM. Pharmacokinetics and tissue distribution of betulinic acid in CD-1 mice. Biopharm. Drug Dispos. 1999, 20, 379–383. [Google Scholar]
- Smith, PF; Ogundele, A; Forrest, A; Wilton, J; Salzwedel, K; Doto, J; Allaway, GP; Martin, DE. Phase I and II study of the safety, virologic effect, and pharmacokinetics/pharmacodynamics of single-dose 3-o-(3′,3′-dimethylsuccinyl)betulinic acid (bevirimat) against human immunodeficiency virus infection. Antimicrob. Agents Chemother. 2007, 51, 3574–3581. [Google Scholar]
- Martin, DE; Blum, R; Doto, J; Galbraith, H; Ballow, C. Multiple-dose pharmacokinetics and safety of bevirimat, a novel inhibitor of HIV maturation, in healthy volunteers. Clin. Pharmacokinet. 2007, 46, 589–598. [Google Scholar]
- Yasukawa, K; Takido, M; Matsumoto, T; Takeuchi, M; Nakagawa, S. Sterol and triterpene derivatives from plants inhibit the effects of a tumor promoter, and sitosterol and betulinic acid inhibit tumor formation in mouse skin two-stage carcinogenesis. Oncology 1991, 48, 72–76. [Google Scholar]
- Liby, K; Honda, T; Williams, CR; Risingsong, R; Royce, DB; Suh, N; Dinkova-Kostova, AT; Stephenson, KK; Talalay, P; Sundararajan, C; Gribble, GW; Sporn, MB. Novel semisynthetic analogues of betulinic acid with diverse cytoprotective, antiproliferative, and proapoptotic activities. Mol. Cancer Ther. 2007, 6, 2113–2119. [Google Scholar]
- Rajendran, P; Jaggi, M; Singh, MK; Mukherjee, R; Burman, AC. Pharmacological evaluation of C-3 modified Betulinic acid derivatives with potent anticancer activity. Invest. New Drugs 2008, 26, 25–34. [Google Scholar]
- Bi, Y; Xu, J; Wu, X; Ye, W; Yuan, S; Zhang, L. Synthesis and cytotoxic activity of 17-carboxylic acid modified 23-hydroxy betulinic acid ester derivatives. Bioorg. Med. Chem. Lett. 2007, 17, 1475–1478. [Google Scholar]
- Saelens, X; Festjens, N; Vande Walle, L; van Gurp, M; van Loo, G; Vandenabeele, P. Toxic proteins released from mitochondria in cell death. Oncogene 2004, 23, 2861–2874. [Google Scholar]
- Walczak, H; Krammer, PH. The CD95 (APO-1/Fas) and the TRAIL (APO-2L) apoptosis systems. Exp. Cell Res. 2000, 256, 58–66. [Google Scholar]
- Fulda, S; Debatin, K-M. Targeting apoptosis pathways in cancer therapy. Curr. Cancer Drug Targets 2004, 4, 569–576. [Google Scholar]
- Degterev, A; Boyce, M; Yuan, J. A decade of caspases. Oncogene 2003, 22, 8543–8567. [Google Scholar]
- Salvesen, GS; Duckett, CS. IAP proteins: blocking the road to death’s door. Nat. Rev. Mol. Cell Biol. 2002, 3, 401–410. [Google Scholar]
- Micheau, O. Cellular FLICE-inhibitory protein: an attractive therapeutic target? Expert Opin. Ther. Targets 2003, 7, 559–573. [Google Scholar]
| Cancer type | ED50 (μg/ml) | References |
|---|---|---|
| melanoma | 1.1–4.8 | [33] |
| neuroblastoma | 2–10 | [14] |
| medulloblastoma | 3–15 | [39] |
| glioblastoma | 5–16 | [39] |
| head & neck cancer | 8 | [18] |
| ovarian carcinoma | 1.8–4.5 | [22] |
| cervix carcinoma | 1.8 | [22] |
| lung carcinoma | 1.5–4.2 | [22] |
| leukemia | 2–15 | [23] |
Share and Cite
Fulda, S. Betulinic Acid for Cancer Treatment and Prevention. Int. J. Mol. Sci. 2008, 9, 1096-1107. https://doi.org/10.3390/ijms9061096
Fulda S. Betulinic Acid for Cancer Treatment and Prevention. International Journal of Molecular Sciences. 2008; 9(6):1096-1107. https://doi.org/10.3390/ijms9061096
Chicago/Turabian StyleFulda, Simone. 2008. "Betulinic Acid for Cancer Treatment and Prevention" International Journal of Molecular Sciences 9, no. 6: 1096-1107. https://doi.org/10.3390/ijms9061096
APA StyleFulda, S. (2008). Betulinic Acid for Cancer Treatment and Prevention. International Journal of Molecular Sciences, 9(6), 1096-1107. https://doi.org/10.3390/ijms9061096




