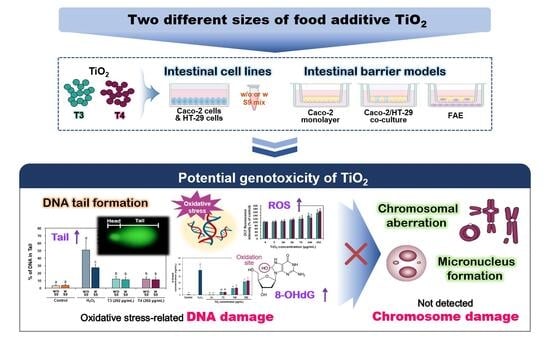
Comprehensive Evaluation of the Genotoxic Potential of Food Additive Titanium Dioxide in Human Intestinal Cell Systems
Abstract
1. Introduction
2. Results and Discussion
2.1. Characterization of TiO2
2.2. Cytotoxicity and ROS Generation of TiO2
2.3. DNA Damage Caused by TiO2 in Human Intestinal Cell Lines
2.4. Effect of a Metabolic Activation on DNA Damage Caused by TiO2
2.5. DNA Damage Caused by TiO2 in Human Intestinal Barrier Models
2.6. 8-Hydroxy-2′-Deoxyguanosine (8-OHdG) Generation Caused by TiO2
2.7. In Vitro Chromosomal Aberration Caused by TiO2
2.8. In Vitro Micronucleus Formation Caused by TiO2
3. Materials and Methods
3.1. Food-Grade TiO2 and Characterization
3.2. Cell Culture and Cytotoxicity
3.3. ROS Generation and Antioxidant Enzyme Activity
3.4. In Vitro Intestinal Barrier Models
3.5. Comet Assay
3.6. 8-OHdG Assay
3.7. In Vitro Chromosomal Aberration Assay
3.8. In Vitro Micronucleus Assay
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CAT | Catalase |
| CPA | Cyclophosphamide |
| DCF | Dichlorofluorescein |
| DDW | Distilled and deionized water |
| EFSA | European Food Safety Authority |
| EU | European Union |
| Excl | Excluding |
| FAE | Follicle-associated epithelium |
| FAO | Food and Agriculture Organization |
| FBS | Fetal Bovine Serum |
| FDA | United States Food and Drug Administration |
| 8-OHdG | 8-Hydroxy-2′-Deoxyguanosine |
| JECFA | Joint Expert Committee on Food Additives |
| LDH | Lactate dehydrogenase |
| MMC | Mitomycin C |
| MMS | Methyl methanesulfonate |
| MNBN | Micronucleated binucleated cell |
| ND | Not detectable |
| NP | Nanoparticle |
| PBS | Phosphate-buffered saline |
| ROS | Reactive oxygen species |
| SEM | Scanning electron microscopy |
| SOD | Superoxide dismutase |
| TEER | Transepithelial electrical resistance |
| WHO | World Health Organization |
References
- Chandoliya, R.; Sharma, S.; Sharma, V.; Joshi, R.; Sivanesan, I. Titanium dioxide nanoparticle: A comprehensive review on synthesis, applications and toxicity. Plants 2024, 13, 2964. [Google Scholar] [CrossRef]
- Joshi, N.C.; Negi, P.B.; Gururani, P. A review on metal/metal oxide nanoparticles in food processing and packaging. Food Sci. Biotechnol. 2024, 33, 1307–1322. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration (FDA). CFR-Code of Federal Regulations Title 21 CRF 73.575. Available online: https://www.ecfr.gov/current/title-21/chapter-I/subchapter-A/part-73#se21.1.73_1575 (accessed on 20 November 2025).
- European Commission. Regulation (EC) No 1333/2008 of the European Parliament and of the Council of 16 December 2008 on Food Additives (Text with EEA Relevance). 2008. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32008R1333&qid=1763799460381 (accessed on 22 November 2025).
- EFSA Panel on Food Additives and Flavourings (FAF); Younes, M.; Aquilina, G.; Castle, L.; Engel, K.H.; Fowler, P.; Frutos Fernandez, M.J.; Fürst, P.; Gundert-Remy, U.; Gürtler, R.; et al. Safety assessment of titanium dioxide (E171) as a food additive. EFSA J. 2021, 19, e06585. [Google Scholar] [CrossRef]
- Bettini, S.; Boutet-Robinet, E.; Cartier, C.; Coméra, C.; Gaultier, E.; Dupuy, J.; Naud, N.; Taché, S.; Grysan, P.; Reguer, S.; et al. Food-grade TiO2 impairs intestinal and systemic immune homeostasis, initiates preneoplastic lesions and promotes aberrant crypt development in the rat colon. Sci. Rep. 2017, 7, 40373. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Lee, H.-J.; Son, Y.; Bae, M.J.; Jo, W.S.; Park, J.H.; Jeong, S.; Moon, C.; Shin, I.-S.; Lee, C.G.; et al. Low-dose-rate gamma radiation aggravates titanium dioxide nanoparticle-induced lung injury in mice. Mol. Cell. Toxicol. 2024, 20, 389–398. [Google Scholar] [CrossRef]
- Dorier, M.; Béal, D.; Marie-Desvergne, C.; Dubosson, M.; Barreau, F.; Houdeau, E.; Herlin-Boime, N.; Carriere, M. Continuous in vitro exposure of intestinal epithelial cells to E171 food additive causes oxidative stress, inducing oxidation of DNA bases but no endoplasmic reticulum stress. Nanotoxicology 2017, 11, 751–761. [Google Scholar]
- Dorier, M.; Béal, D.; Tisseyre, C.; Marie-Desvergne, C.; Dubosson, M.; Barreau, F.; Houdeau, E.; Herlin-Boime, N.; Rabilloud, T.; Carriere, M. The food additive E171 and titanium dioxide nanoparticles indirectly alter the homeostasis of human intestinal epithelial cells in vitro. Environ. Sci. Nano 2019, 6, 1549–1561. [Google Scholar] [CrossRef]
- World Health Organization. Compendium of Food Additive Specifications: Joint FAO/WHO Expert Committee on Food Additives, 97th Meeting, Rome, 31 October–9 November 2023; Food & Agriculture Organization: Rome, Italy, 2024. [Google Scholar]
- Additives, J. Thirteenth Report of the Joint Fao/Who Expert Committee on Food Additives. Specifcations for the Identity and Purity of Food Additives and Their Toxicological Evaluation; World Health Organization: Geneva, Switzerland, 1970. [Google Scholar]
- National Toxicology Program. Bioassay of titanium dioxide for possible carcinogenicity. Natl. Cancer Inst. Carcinog. Tech. Rep. Ser. 1979, 97, 1–123. [Google Scholar]
- United Kingdom’s Food Standards Authority (FSA). Annual FSA Science Update. 2025. Available online: https://www.food.gov.uk/print/pdf/node/27166 (accessed on 22 November 2025).
- Health Canada. State of the Science of Titanium Dioxide (TiO2) as a Food Additive. 2022. Available online: https://publications.gc.ca/collections/collection_2022/sc-hc/H164-341-2022-eng.pdf (accessed on 22 November 2025).
- Food Standards Australia New Zealand (FSANZ). Review of Titanium Dioxide as a Food Additive. 2022. Available online: https://www.foodstandards.gov.au/sites/default/files/consumer/foodtech/Documents/FSANZ_TiO2_Assessment_report.pdf (accessed on 22 November 2025).
- Blevins, L.K.; Crawford, R.B.; Bach, A.; Rizzo, M.D.; Zhou, J.; Henriquez, J.E.; Khan, D.M.I.O.; Sermet, S.; Arnold, L.L.; Pennington, K.L.; et al. Evaluation of immunologic and intestinal effects in rats administered an E 171-containing diet, a food grade titanium dioxide (TiO2). Food Chem. Toxicol. 2019, 133, 110793. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.-M.; Nam, H.-N.; Choi, S.-J. Effects of the Interactions Between Food Additive Titanium Dioxide and Matrices on Genotoxicity. Int. J. Mol. Sci. 2025, 26, 617. [Google Scholar] [CrossRef]
- Hwang, J.-S.; Yu, J.; Kim, H.-M.; Oh, J.-M.; Choi, S.-J. Food additive titanium dioxide and its fate in commercial foods. Nanomaterials 2019, 9, 1175. [Google Scholar] [CrossRef]
- Korea Health Industry Development Institute (KHIDI). National Food & Nutrition Statistics. 2017. Available online: https://www.khidi.or.kr/kps/dhraStat/result2?menuId=MENU01653&gubun=&year=2017 (accessed on 19 November 2020).
- Yu, J.; Kim, Y.-H.; Choi, S.-J. Usage- and daily intake-based cytotoxicity study of frequently used natural food additives in South Korea. Korean J. Food Sci. Technol. 2020, 52, 546–554. [Google Scholar]
- Bhattacharya, K.; Davoren, M.; Boertz, J.; Schins, R.P.; Hoffmann, E.; Dopp, E. Titanium dioxide nanoparticles induce oxidative stress and DNA-adduct formation but not DNA-breakage in human lung cells. Part. Fibre Toxicol. 2009, 6, 17. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.Y.; Lee, T.G.; Reipa, V.; Heo, M.B. Titanium dioxide induces apoptosis under UVA irradiation via the generation of lysosomal membrane permeabilization-dependent reactive oxygen species in HaCaT cells. Nanomaterials 2021, 11, 1943. [Google Scholar] [CrossRef] [PubMed]
- Fattori, A.C.M.; Brassolatti, P.; Feitosa, K.A.; Pedrino, M.; de O Correia, R.; Albuquerque, Y.R.; de A Rodolpho, J.M.; Luna, G.L.F.; Cancino-Bernardi, J.; Zucolotto, V.; et al. Titanium dioxide nanoparticle (TiO2 NP) induces toxic effects on la-9 mouse fibroblast cell line. Cell. Physiol. Biochem. 2023, 57, 63–81. [Google Scholar] [CrossRef]
- Freire, K.; Ordoñez Ramos, F.; Soria, D.B.; Pabón Gelves, E.; Di Virgilio, A.L. Cytotoxicity and DNA damage evaluation of TiO2 and ZnO nanoparticles. Uptake in lung cells in culture. Toxicol. Res. 2021, 10, 192–202. [Google Scholar] [CrossRef]
- Guichard, Y.; Schmit, J.; Darne, C.; Gaté, L.; Goutet, M.; Rousset, D.; Rastoix, O.; Wrobel, R.; Witschger, O.; Martin, A.; et al. Cytotoxicity and genotoxicity of nanosized and microsized titanium dioxide and iron oxide particles in Syrian hamster embryo cells. Ann. Occup. Hyg. 2012, 56, 631–644. [Google Scholar] [CrossRef]
- Wang, Y.; Cui, H.; Zhou, J.; Li, F.; Wang, J.; Chen, M.; Liu, Q. Cytotoxicity, DNA damage, and apoptosis induced by titanium dioxide nanoparticles in human non-small cell lung cancer A549 cells. Environ. Sci. Pollut. Res. 2015, 22, 5519–5530. [Google Scholar] [CrossRef]
- Woodruff, R.S.; Li, Y.; Yan, J.; Bishop, M.; Jones, M.Y.; Watanabe, F.; Biris, A.S.; Rice, P.; Zhou, T.; Chen, T. Genotoxicity evaluation of titanium dioxide nanoparticles using the Ames test and Comet assay. J. Appl. Toxicol. 2012, 32, 934–943. [Google Scholar] [CrossRef]
- Hackenberg, S.; Friehs, G.; Kessler, M.; Froelich, K.; Ginzkey, C.; Koehler, C.; Scherzed, A.; Burghartz, M.; Kleinsasser, N. Nanosized titanium dioxide particles do not induce DNA damage in human peripheral blood lymphocytes. Environ. Mol. Mutagen. 2011, 52, 264–268. [Google Scholar] [CrossRef]
- Shukla, R.K.; Kumar, A.; Gurbani, D.; Pandey, A.K.; Singh, S.; Dhawan, A. TiO2 nanoparticles induce oxidative DNA damage and apoptosis in human liver cells. Nanotoxicology 2013, 7, 48–60. [Google Scholar] [CrossRef]
- Anderson, D.; Yu, T.W.; Schmezer, P. An investigation of the DNA-damaging ability of benzene and its metabolites in human lymphocytes, using the comet assay. Environ. Mol. Mutagen. 1995, 26, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Fröhlich, E. Comparison of conventional and advanced in vitro models in the toxicity testing of nanoparticles. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1091–1107. [Google Scholar] [CrossRef] [PubMed]
- Kämpfer, A.A.M.; Busch, M.; Büttner, V.; Bredeck, G.; Stahlmecke, B.; Hellack, B.; Masson, I.; Sofranko, A.; Albrecht, C.; Schins, R.P.F. Model Complexity as Determining Factor for In Vitro Nanosafety Studies: Effects of Silver and Titanium Dioxide Nanomaterials in Intestinal Models. Small 2021, 17, e2004223. [Google Scholar] [CrossRef]
- Brandão, F.; Fernández-Bertólez, N.; Rosário, F.; Bessa, M.J.; Fraga, S.; Pásaro, E.; Teixeira, J.P.; Laffon, B.; Valdiglesias, V.; Costa, C. Genotoxicity of TiO2 nanoparticles in four different human cell lines (A549, HEPG2, A172 and SH-SY5Y). Nanomaterials 2020, 10, 412. [Google Scholar] [CrossRef]
- Shukla, R.K.; Sharma, V.; Pandey, A.K.; Singh, S.; Sultana, S.; Dhawan, A. ROS-mediated genotoxicity induced by titanium dioxide nanoparticles in human epidermal cells. Toxicol. In Vitro 2011, 25, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Jeong, M.H.; Seo, H.S.; Park, M.K.; Park, H.J.; Nah, S.S. In vitro and in vivo evaluation of the genotoxicity of titanium dioxide, GST. Environ. Anal. Health Toxicol. 2023, 38, e2023008. [Google Scholar] [CrossRef]
- Kim, Y.E.; Park, H.J. Mutagenicity and Genotoxicity of Calcium from Jeju Lava Seawater. Food Eng. Prog. 2023, 27, 303–311. [Google Scholar] [CrossRef]
- OECD. Test No. 473: In Vitro Mammalian Chromosomal Aberration Test. In OECD Guidelines for the Testing of Chemicals; Section 4; OECD Publishing: Paris, France, 2016. [Google Scholar]
- Patel, S.; Patel, P.; Bakshi, S.R. Titanium dioxide nanoparticles: An in vitro study of DNA binding, chromosome aberration assay, and comet assay. Cytotechnology 2017, 69, 245–263. [Google Scholar] [CrossRef]
- Ünal, F.; Korkmaz, F.D.; Suludere, Z.; Erol, Ö.; Yüzbaşıoğlu, D. Genotoxicity of two nanoparticles: Titanium dioxide and zinc oxide. Gazi Univ. J. Sci. 2021, 34, 948–958. [Google Scholar] [CrossRef]
- Warheit, D.B.; Hoke, R.A.; Finlay, C.; Donner, E.M.; Reed, K.L.; Sayes, C.M. Development of a base set of toxicity tests using ultrafine TiO2 particles as a component of nanoparticle risk management. Toxicol. Lett. 2007, 171, 99–110. [Google Scholar] [CrossRef]
- Kirkland, D.; Burzlaff, A.; Czich, A.; Doak, S.H.; Fowler, P.; Pfuhler, S.; Stankowski, L.F. Updated assessment of the genotoxic potential of titanium dioxide based on reviews of in vitro comet, mode of action and cellular uptake studies, and recent publications. Regul. Toxicol. Pharmacol. 2024, 154, 105734. [Google Scholar] [CrossRef]
- Bazin, E.; Mourot, A.; Humpage, A.R.; Fessard, V. Genotoxicity of a freshwater cyanotoxin, cylindrospermopsin, in two human cell lines: Caco-2 and HepaRG. Environ. Mol. Mutagen. 2010, 51, 251–259. [Google Scholar] [CrossRef]
- Díez-Quijada, L.; Prieto, A.I.; Puerto, M.; Jos, Á.; Cameán, A.M. In vitro mutagenic and genotoxic assessment of a mixture of the cyanotoxins microcystin-LR and cylindrospermopsin. Toxins 2019, 11, 318. [Google Scholar] [CrossRef] [PubMed]
- Proquin, H.; Rodríguez-Ibarra, C.; Moonen, C.G.; Urrutia Ortega, I.M.; Briedé, J.J.; de Kok, T.M.; van Loveren, H.; Chirino, Y.I. Titanium dioxide food additive (E171) induces ROS formation and genotoxicity: Contribution of micro and nano-sized fractions. Mutagenesis 2017, 32, 139–149, Erratum in Mutagenesis 2018, 33, 267–268. https://doi.org/10.1093/mutage/gey011. [Google Scholar] [CrossRef]
- Vignard, J.; Pettes-Duler, A.; Gaultier, E.; Cartier, C.; Weingarten, L.; Biesemeier, A.; Taubitz, T.; Pinton, P.; Bebeacua, C.; Devoille, L.; et al. Food-grade titanium dioxide translocates across the buccal mucosa in pigs and induces genotoxicity in an in vitro model of human oral epithelium. Nanotoxicology 2023, 17, 289–309. [Google Scholar] [CrossRef]
- Liang, C.; Zhang, X.; Fang, J.; Sun, N.; Liu, H.; Feng, Y.; Wang, H.; Yu, Z.; Jia, X. Genotoxicity evaluation of food additive titanium dioxide using a battery of standard in vivo tests. Regul. Toxicol. Pharmacol. 2024, 148, 105586. [Google Scholar] [CrossRef] [PubMed]
- Ling, M.; Liang, J.; Lyu, Z.; Yu, P.; Yang, M.; Wu, X.; Zhang, W.; Lu, L.; Zhao, Y.; Bian, Q. Genotoxicity assessment of food-grade titanium dioxide. Food Chem. Toxicol. 2025, 201, 115476. [Google Scholar] [CrossRef] [PubMed]
- Warheit, D.B. Safety of titanium dioxide (E171) as a food additive for humans. Front. Toxicol. 2024, 6, 1333746. [Google Scholar] [CrossRef]
- EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS). Re-evaluation of titanium dioxide (E 171) as a food additive. EFSA J. 2016, 14, e04545. [CrossRef]
- Jo, M.-R.; Yu, J.; Kim, H.-J.; Song, J.H.; Kim, K.-M.; Oh, J.-M.; Choi, S.-J. Titanium dioxide nanoparticle-biomolecule interactions influence oral absorption. Nanomaterials 2016, 6, 225. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-I.; Kwon, R.-Y.; Choi, S.-J. Food Additive Solvents Increase the Dispersion, Solubility, and Cytotoxicity of ZnO Nanoparticles. Nanomaterials 2023, 13, 2573. [Google Scholar] [CrossRef] [PubMed]
- Lian, P.; Braber, S.; Varasteh, S.; Wichers, H.J.; Folkerts, G. Hypoxia and heat stress affect epithelial integrity in a Caco-2/HT-29 co-culture. Sci. Rep. 2021, 11, 13186. [Google Scholar] [CrossRef] [PubMed]






| Sample | Average Size (nm) | Distribution (Number %) | ||
|---|---|---|---|---|
| <100 nm | 100–200 nm | >200 nm | ||
| T3 | 161.1 ± 28.0 | ND | 89 | 11 |
| T4 | 123.0 ± 26.7 | 18 | 82 | ND |
| Treatment | Conc. (μg/mL) | S9 Mix | No. of Structural Aberrations | Gap | Total Aberration Cells (Excl Gaps) | Total Aberration Frequency (%) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Chromatid Type | Chromosome Type | Fragmentation | ||||||||
| Break | Exchange | Break | Exchange | |||||||
| Control | 0 | − | 1 | 0 | 0 | 1 | 2 | 0 | 4 | 2.0 |
| Positive control (MMC) | 0.2 | 12 | 0 | 1 | 4 | 14 | 1 | 31 | 15.5 | |
| T3 | 292 | 3 | 0 | 0 | 2 | 0 | 0 | 5 | 2.5 | |
| T4 | 292 | 3 | 0 | 0 | 1 | 0 | 1 | 4 | 2.0 | |
| Control | 0 | + | 3 | 0 | 0 | 0 | 2 | 1 | 5 | 2.5 |
| Positive control (CPA) | 10 | 8 | 0 | 0 | 2 | 23 | 2 | 33 | 16.5 | |
| T3 | 292 | 5 | 0 | 0 | 1 | 0 | 2 | 6 | 3.0 | |
| T4 | 292 | 1 | 0 | 0 | 2 | 3 | 2 | 6 | 3.0 | |
| Treatment | Conc. (μg/mL) | S9 Mix | MNBN (%) |
|---|---|---|---|
| Control | 0 | − | 3.0 |
| Positive control (MMS) | 25 | 8.5 | |
| T3 | 292 | 2.5 | |
| T4 | 292 | 2.5 | |
| Control | 0 | + | 2.5 |
| Positive control (CPA) | 10 | 9.0 | |
| T3 | 292 | 2.5 | |
| T4 | 292 | 3.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nam, H.-N.; Jeong, S.-M.; Kim, S.-B.; Choi, S.-J. Comprehensive Evaluation of the Genotoxic Potential of Food Additive Titanium Dioxide in Human Intestinal Cell Systems. Int. J. Mol. Sci. 2025, 26, 12026. https://doi.org/10.3390/ijms262412026
Nam H-N, Jeong S-M, Kim S-B, Choi S-J. Comprehensive Evaluation of the Genotoxic Potential of Food Additive Titanium Dioxide in Human Intestinal Cell Systems. International Journal of Molecular Sciences. 2025; 26(24):12026. https://doi.org/10.3390/ijms262412026
Chicago/Turabian StyleNam, Han-Na, Su-Min Jeong, Su-Bin Kim, and Soo-Jin Choi. 2025. "Comprehensive Evaluation of the Genotoxic Potential of Food Additive Titanium Dioxide in Human Intestinal Cell Systems" International Journal of Molecular Sciences 26, no. 24: 12026. https://doi.org/10.3390/ijms262412026
APA StyleNam, H.-N., Jeong, S.-M., Kim, S.-B., & Choi, S.-J. (2025). Comprehensive Evaluation of the Genotoxic Potential of Food Additive Titanium Dioxide in Human Intestinal Cell Systems. International Journal of Molecular Sciences, 26(24), 12026. https://doi.org/10.3390/ijms262412026