Deaminase Modulation Driving a New Era in Drug Development
Abstract
1. Aim of This Review
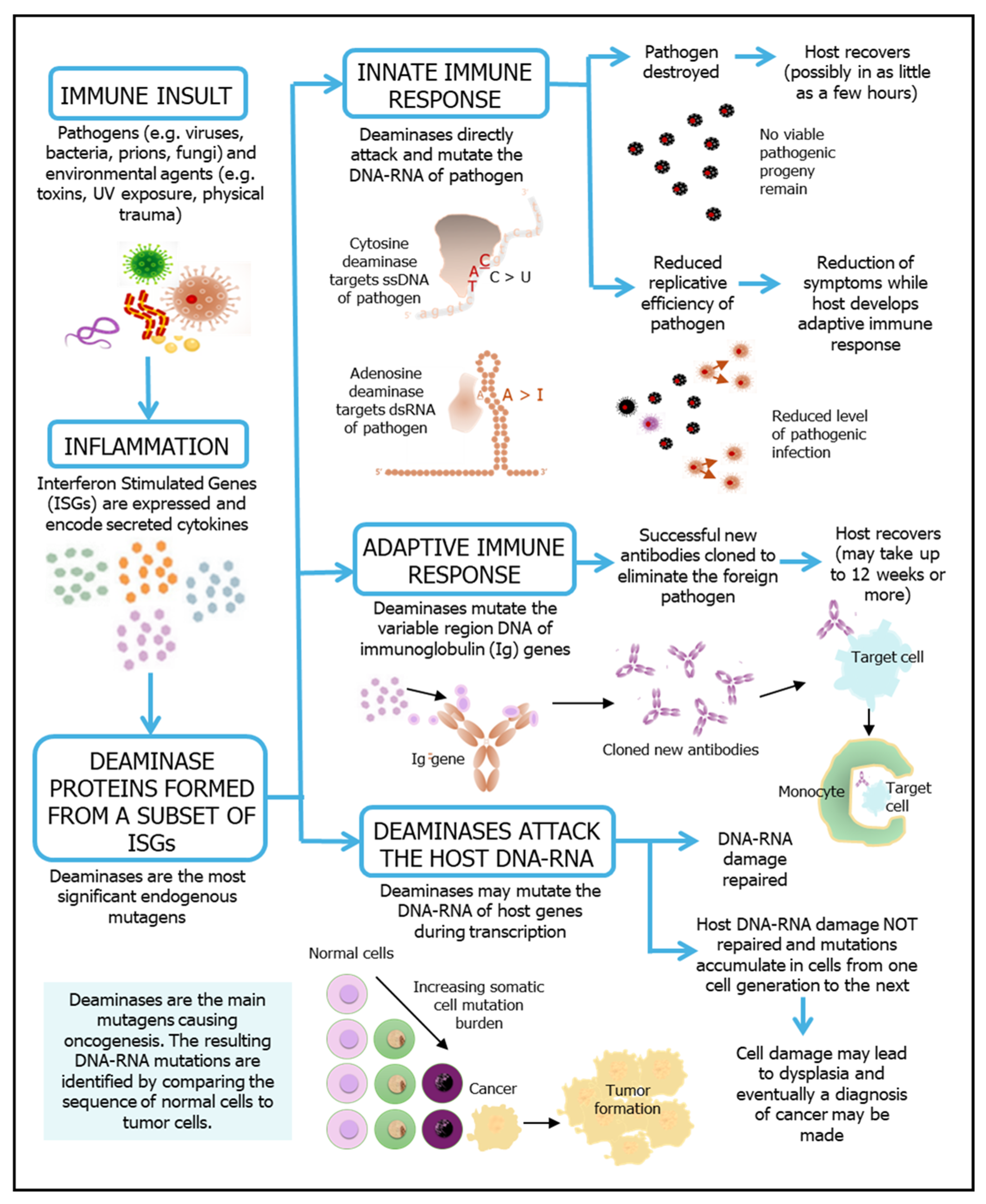
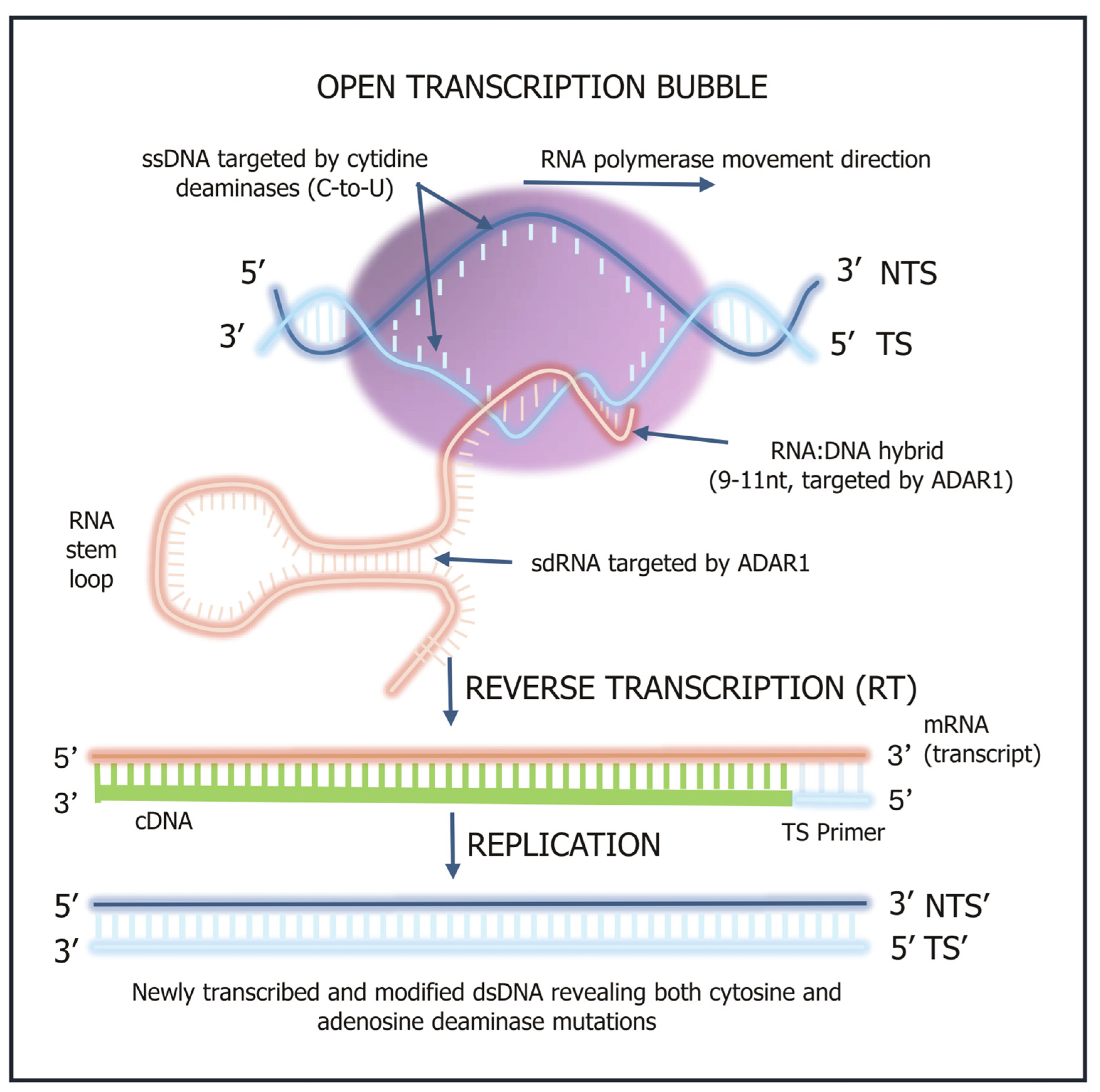
2. Deaminase Action in Inflammation, Immunity and Disease
3. Innate Immune Response
3.1. APOBEC3 Deaminases as ‘Viral Smashers’
3.2. Using Deaminases to Develop New Antimicrobial Drugs
4. Adaptive Immune Response
4.1. Therapeutic Inhibition of AID in Lymphoid Tissue
4.2. Can AID Be Used to Improve Vaccine Efficacy?
5. Cytidine Deaminase Modulation and Non-Lymphoid Cancer
5.1. AID Modulation in Non-Lymphoid Cancers
5.2. APOBEC3 Modulation in Oncology
5.2.1. Promoting Immune Activation in the Tumor Microenvironment
5.2.2. Increasing Response to Cancer Therapeutics
5.2.3. Overcoming Drug Resistance
5.2.4. APOBEC3s and Cancer Progression
5.2.5. APOBEC3 Modulation Drug Development Approaches
6. Adenosine Deaminase Modulation
6.1. ADAR1 Tumor Promotion
6.2. ADAR2 Immunomodulation
7. Harnessing the Power of Deaminase Modulation
| Organization | Deaminase(s) | Modulation Type | Disease Indication(s) | Phase | Notes; Source/[References] |
|---|---|---|---|---|---|
| Accent Therapeutics with AstraZeneca, Lexington, MA, USA | ADAR1 | Inhibition | Lung, breast, ovarian, head & neck cancers | Pre-clinical | Developing small-molecule ADAR1 inhibitors. Citations: [127]; company pipeline. |
| AIRNA Inc., Cambridge, MA, USA | ADAR2 | RNA editing | Genetic diseases | Pre-clinical | ADAR-recruiting oligonucleotides. Source: AIRNA pipeline. |
| ApoGen Biotechnologies, Seattle, WA, USA | APOBEC3 family | Inhibition | APOBEC-driven mutagenic cancers | Pre-clinical | APOBEC3 inhibitors for reducing tumor mutational burden. Source: ApoGen investor materials. |
| Arsenal Biosciences, South San Francisco, CA, USA | Cytidine/adenosine deaminases | Base editing for T-cell engineering | Solid tumors | Pre-clinical | T-cell engineering using base editors. https://arsenalbio.com/2022/01/27/computational-biologys-effect-on-solid-tumors/ (accessed on 19 November 2025) |
| Aspera Biomedicines, La Jolla, CA, USA | ADAR1 (p150) | Inhibition | Solid tumors with ADAR1-mediated immune evasion | Pre-clinical/IND-enabling | Rebecsinib ADAR1p150 antagonist. Citation: [127]. |
| Astex Pharmaceuticals, Pleasanton, CA, USA | Cytidine deaminase | Inhibition | MDS, CMML, AML | Approved | Cedazuridine. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-oral-combination-decitabine-and-cedazuridine-myelodysplastic-syndromes? (accessed on 19 November 2025) |
| Beam Therapeutics, Cambridge, MA, USA | APOBEC-based CBEs, ABEs | Base editing | Sickle cell, FA, oncology | Clinical + Pre-clinical | CBE/ABE programs. Citation: [126]. |
| Bio-Techne (Tocris), Minneapolis, MN, USA | Adenosine deaminase (ADA) | Inhibition | Immunomodulation/research use | Pre-clinical | EHNA hydrochloride ADA inhibitor. Source: Tocris datasheet. |
| Broad Institute/MIT/Harvard, MA, USA | Engineered cytidine deaminase | RNA C→U editing | RNA diseases | Pre-clinical | RESCUE C→U RNA editing system. |
| Covant Therapeutics with Boehringer Ingelheim, Boston, MA, USA | ADAR1 | Inhibition | Immunotherapy-enhanced cancers | Pre-clinical | Covalent ADAR1 inhibitors. Source: company pipeline releases. |
| Editas Medicine, Cambridge, MA, USA | Multiple deaminases | Base editing | Genetic diseases | Pre-clinical | Deaminase-Cas fusion editors. |
| GMDx Genomics, Melbourne Australia | Multiple deaminases | Bioinformatic mapping | Precision oncology | Research | Deaminase binding domain definition. Citation: [1]. |
| Halozyme Therapeutics, San Diego, CA, USA | ADA2 | Enzyme supplementation | Colon, lung, pancreatic cancers | Pre-clinical | PEG-ADA2 reduces tumor adenosine. Source: Halozyme preclinical publications. |
| HuidaGene Therapeutics, Shanghai, China | Novel guanine deaminase/synthetic editors | G→C or G→T editing | Genetic disorders | Pre-clinical | World’s first G→Y base editor. Citation: [133]. |
| Korro Bio, Cambridge, MA, USA | ADAR2 | RNA editing | AATD | Clinical (Phase 1/2a) | KRRO-110 (REWRITE trial). |
| Life Edit Therapeutics, Morrisville, NC, USA | Multiple deaminases | Base editing | Genetic diseases | Pre-clinical | In vivo mRNA base editing. Company platform sources. |
| Mammoth Biosciences, Brisbane, CA, USA | Engineered deaminases | Base editing | Genetic & infectious disease | Pre-clinical | Compact Cas-based deaminase editors. |
| Pfizer Inc., Detroit, MI, USA | Adenosine deaminase (ADA) | Inhibition | Hairy cell leukemia | Approved | Pentostatin (also known as deoxycoformycin). Source Pfizer product information. |
| Scribe Therapeutics, Alameda, CA, USA | Engineered deaminases | Base editing | Neurological & genetic disease | Pre-clinical | CRISPR-based precision deaminases. Citation: [126]. |
| Shape Therapeutics, San Francisco Bay, CA, USA | ADAR2 | RNA editing | Genetic disease | Pre-clinical | ADAR2-based RNA editing via AAV. Source: ShapeTx platform. |
| UCSF RBVI, San Francisco Bay, CA, USA | Multiple deaminases | Molecular modeling | Research | Research tools | Chimera/ChimeraX platform. |
| University of Reading, UK | Multiple deaminases | Protein modeling | Research | Research tools | IntFOLD protein prediction tools. |
| Wave Life Sciences, Cambridge, MA, USA | ADAR | A→I RNA editing | AATD | Clinical (Phase 1/2a) | WVE-006 clinical trials NCT06186492, NCT06405633. Source: News Release 16 October 2024. |
| WHAT IF Foundation, Nijmegen, The Netherlands | Multiple deaminases | Structural modeling | Research | Research tools | 3D enzyme–motif modeling. Source: WHAT IF documentation. |
8. Closing Remarks
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lindley, R.A. A review of the mutational role of deaminases and the generation of a cognate molecular model to explain cancer mutation spectra. Med. Res. Arch. 2020, 8, 1–15. [Google Scholar] [CrossRef]
- Cervantes-Gracia, K.; Gramalla-Schmitz, A.; Weischedel, J.; Chahwan, R. APOBECs orchestrate genomic and epigenomic editing across health and disease. Trends Genet. 2021, 37, 1028–1043. [Google Scholar] [CrossRef]
- Martin, P.; Pardo-Pastor, C.; Jenkins, R.G.; Rosenblatt, J. Imperfect wound healing sets the stage for chronic diseases. Science 2024, 386, eadp2974. [Google Scholar] [CrossRef]
- Schneider, W.M.; Chevillotte, M.D.; Rice, C.M. Interferon-stimulated genes: A complex web of host defenses. Annu. Rev. Immunol. 2014, 32, 513–545. [Google Scholar] [CrossRef]
- Steele, E.J.; Lindley, R.A. Somatic mutation patterns in non-lymphoid cancers resemble the strand-biased somatic hypermutation spectra of antibody genes. DNA Repair 2010, 9, 600–603. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Steele, E.J.; Lindley, R.A. Deaminase-driven reverse transcription mutagenesis in oncogenesis: Critical analysis of transcriptional strand asymmetries of single base substitution signatures. Int. J. Mol. Sci. 2025, 26, 989. [Google Scholar] [CrossRef] [PubMed]
- Lindley, R.A.; Hall, N.E. APOBEC and ADAR deaminases may cause many single nucleotide polymorphisms curated in the OMIM database. Mutat. Res. 2018, 810, 33–38. [Google Scholar] [CrossRef]
- Bayona-Feliu, A.; Herrera-Moyano, E.; Badra-Fajardo, N.; Galvan-Femenia, I.; Soler-Oliva, E.; Aguilera, A. The chromatin network helps prevent cancer-associated mutagenesis at transcription-replication conflicts. Nat. Commun. 2023, 14, 6890. [Google Scholar] [CrossRef]
- Stoy, H.; Zwicky, K.; Kuster, D.; Lang, K.S.; Krietsch, J.; Crossley, M.P.; Schmid, J.A.; Cimprich, K.A.; Merrikh, H.; Lopes, M. Direct visualization of transcription-replication conflicts reveals post-replicative DNA:RNA hybrids. Nat. Struct. Mol. Biol. 2023, 30, 348–359. [Google Scholar] [CrossRef] [PubMed]
- Lindley, R.A.; Steele, E.J. ADAR and APOBEC editing signatures in viral RNA during acute-phase innate immune responses of the host-parasite relationship to Flaviviruses. Res. Rep. 2018, 2, e1–e22. [Google Scholar]
- Meshcheryakova, A.; Pietschmann, P.; Zimmermann, P.; Rogozin, I.B.; Mechtcheriakova, D. AID and APOBECs as multifaceted intrinsic virus-restricting factors: Emerging concepts in the light of COVID-19. Front. Immunol. 2021, 12, 690416. [Google Scholar] [CrossRef] [PubMed]
- Burke, A.J.; Birmingham, W.R.; Zhuo, Y.; Thorpe, T.W.; da Costa, B.Z.; Crawshaw, R.; Rowles, I.; Finnigan, J.D.; Young, C.; Holgate, G.M.; et al. An engineered cytidine deaminase for biocatalytic production of a key intermediate of the COVID-19 antiviral molnupiravir. J. Am. Chem. Soc. 2022, 144, 3761–3765. [Google Scholar] [CrossRef]
- Chen, X.S. Insights into the structures and multimeric status of APOBEC proteins involved in viral restriction and other cellular functions. Viruses 2021, 13, 497. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). 2024 Global Hepatitis Report: Action for Access in Low- and Middle-Income Countries; World Health Organization: Geneva, Switzerland, 2024.
- Baecker, A.; Liu, X.; La Vecchia, C.; Zhang, Z.F. Worldwide incidence of hepatocellular carcinoma cases attributable to major risk factors. Eur. J. Cancer Prev. 2018, 27, 205–212. [Google Scholar] [CrossRef]
- Lucifora, J.; Xia, Y.; Reisinger, F.; Zhang, K.; Stadler, D.; Cheng, X.; Sprinzl, M.F.; Koppensteiner, H.; Makowska, Z.; Volz, T.; et al. Specific and nonhepatotoxic degradation of nuclear hepatitis B virus cccDNA. Science 2014, 343, 1221–1228. [Google Scholar] [CrossRef]
- Kostyushev, D.; Brezgin, S.; Kostyusheva, A.; Ponomareva, N.; Bayurova, E.; Zakirova, N.; Kondrashova, A.; Goptar, I.; Nikiforova, A.; Sudina, A.; et al. Transient and tunable CRISPRa regulation of APOBEC/AID genes for targeting hepatitis B virus. Mol. Ther. Nucleic Acids 2023, 32, 478–493. [Google Scholar] [CrossRef]
- Bonvin, M.; Achermann, F.; Greeve, I.; Stroka, D.; Keogh, A.; Inderbitzin, D.; Candinas, D.; Sommer, P.; Wain-Hobson, S.; Vartanian, J.P.; et al. Interferon-inducible expression of APOBEC3 editing enzymes in human hepatocytes and inhibition of hepatitis B virus replication. Hepatology 2006, 43, 1364–1374. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, X.; Cao, Y.; Yang, Z. Roles of APOBEC3 in hepatitis B virus (HBV) infection and hepatocarcinogenesis. Bioengineered 2021, 12, 2074–2086. [Google Scholar] [CrossRef]
- Budzko, L.; Hoffa-Sobiech, K.; Jackowiak, P.; Figlerowicz, M. Engineered deaminases as a key component of DNA and RNA editing tools. Mol. Ther. Nucleic Acids 2023, 34, 102062. [Google Scholar] [CrossRef]
- Filipić, B.; Ušjak, D.; Rambaher, M.H.; Oljacic, S.; Milenković, M.T. Evaluation of novel compounds as antibacterial or antivirulence agents. Front. Cell. Infect. Microbiol. 2024, 14, 1370062. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Zeng, T.; Duan, L.; Wang, Q.; Zhang, Y.; Xie, C.; Liu, M.; Hu, H.; Huang, J.; Zhang, X.; et al. Optimized TadA variants improve cytidine-to-thymidine base editing efficiency and fidelity. Nat. Commun. 2022, 13, 4714. [Google Scholar]
- Lam, C.H.; Yeh, W.H.; Yeh, C.Y.; Lansky, S.M.; Qi, L.; Muneeruddin, K.; Park, J.; Soto, J.; Cadiñanos, J.; Ding, X.; et al. Enhanced cytosine base editing by engineered TadA deaminases with minimized off-target effects. Nat. Biotechnol. 2023, 41, 621–631. [Google Scholar]
- Feng, Y.; Seija, N.; Di Noia, J.M.; Martin, A. AID in antibody diversification: There and back again. Trends Immunol. 2020, 41, 586–600. [Google Scholar] [CrossRef] [PubMed]
- Muramatsu, M.; Kinoshita, K.; Fagarasan, S.; Yamada, S.; Shinkai, Y.; Honjo, T. Class switch recombination and hypermutation require activation-induced cytidine deaminase (AID), a potential RNA editing enzyme. Nature 2000, 408, 488–492. [Google Scholar] [CrossRef] [PubMed]
- Revy, P.; Muto, T.; Levy, Y.; Geissmann, F.; Plebani, A.; Sanal, O.; Catalan, N.; Forveille, M.; Dufourcq-Labelouse, R.; Gennery, A.; et al. Activation-induced cytidine deaminase (AID) deficiency causes the autosomal recessive form of the hyper-IgM syndrome (HIGM2). Cell 2000, 102, 565–575. [Google Scholar] [CrossRef]
- Methot, S.P.; Di Noia, J.M. Molecular mechanisms of somatic hypermutation and class switch recombination. Adv. Immunol. 2017, 133, 37–87. [Google Scholar]
- Hwang, J.K.; Alt, F.W.; Yeap, L.S. Related mechanisms of antibody somatic hypermutation and class switch recombination. Microbiol. Spectr. 2015, 3, 325–348. [Google Scholar] [CrossRef]
- Steele, E.J.; Lindley, R.A.; Wen, J.; Weiller, G.F. Computational analyses show A-to-G mutations correlate with nascent mRNA hairpins at somatic hypermutation hotspots. DNA Repair 2006, 5, 1346–1363. [Google Scholar] [CrossRef]
- Steele, E.J.; Franklin, A.; Lindley, R.A. Somatic mutation patterns at Ig and non-Ig loci. DNA Repair 2024, 133, 103607. [Google Scholar] [CrossRef] [PubMed]
- Jiao, J.; Lv, Z.; Wang, Y.; Fan, L.; Yang, A. The off-target effects of AID in carcinogenesis. Front. Immunol. 2023, 14, 1221528. [Google Scholar] [CrossRef]
- Rios, L.A.S.; Cloete, B.; Mowla, S. Activation-induced cytidine deaminase: In sickness and in health. J. Cancer Res. Clin. Oncol. 2020, 146, 2721–2730. [Google Scholar] [CrossRef]
- Liu, D.; Hsieh, C.-L.; Lieber, M.R. The RNA tether model for human chromosomal translocation fragile zones. Trends Biochem. Sci. 2024, 49, 391–400. [Google Scholar] [CrossRef]
- Robbiani, D.F.; Nussenzweig, M.C. Chromosome translocation, B cell lymphoma, and activation-induced cytidine deaminase. Annu. Rev. Pathol. Mech. Dis. 2013, 8, 79–103. [Google Scholar] [CrossRef] [PubMed]
- Alt, F.W.; Zhang, Y.; Meng, F.L.; Guo, C.; Schwer, B. Mechanisms of programmed DNA lesions and genomic instability in the immune system. Cell 2013, 152, 417–429. [Google Scholar] [CrossRef] [PubMed]
- Montamat-Sicotte, D.; Litzler, L.C.; Abreu, C.; Safavi, S.; Zahn, A.; Orthwein, A.; Müschen, M.; Oppezzo, P.; Muñoz, D.P.; Di Noia, J.M. HSP90 inhibitors decrease AID levels and activity in mice and in human cells. Eur. J. Immunol. 2015, 45, 2365–2376. [Google Scholar] [CrossRef]
- Tepper, S.; Mortusewicz, O.; Członka, E.; Freire, J.; Gütgemann, D.; Schuetz, M.L.; Dörk, B.; Leonhardt, H.; Barroso, V.R.; Niehrs, N.H. Restriction of AID activity and somatic hypermutation by PARP-1. Nucleic Acids Res. 2019, 47, 7418–7429. [Google Scholar] [CrossRef]
- Klemm, L.; Duy, C.; Iacobucci, I.; Kuchen, S.; von Levetzow, G.; Feldhahn, N.; Henke, N.; Li, Z.; Hoffmann, T.K.; Kim, Y.M.; et al. The B cell mutator AID promotes B lymphoid blast crisis and drug resistance in chronic myeloid leukemia. Cancer Cell 2009, 16, 232–245. [Google Scholar] [CrossRef] [PubMed]
- King, J.J.; Manuel, C.A.; Barrett, C.V.; Raber, S.; Lucas, H.; Sutter, P.; Larijani, M. Catalytic pocket inaccessibility of activation-induced cytidine deaminase is a safeguard against excessive mutagenic activity. Structure 2015, 23, 615–627. [Google Scholar] [CrossRef]
- Alvarez-Gonzalez, J.; Yasgar, A.; Maul, R.W.; Rieffer, A.E.; Crawford, D.J.; Salamango, D.J.; Dorjsuren, D.; Zakharov, A.V.; Jansen, D.J.; Rai, G.; et al. Small molecule inhibitors of activation-induced deaminase decrease class switch recombination in B cells. ACS Pharmacol. Transl. Sci. 2021, 4, 1214–1226. [Google Scholar] [CrossRef]
- Zhao, T.; Cai, Y.; Jiang, Y.; Wang, L.; Zhang, Q.; Liu, H.; Chen, S.; Wang, H.; Wu, W.; Li, X. Vaccine adjuvants: Mechanisms and platforms. Signal Transduct. Target Ther. 2023, 8, 283. [Google Scholar] [CrossRef]
- Li, L.; Honda-Okubo, Y.; Li, C.; Sajkov, D.; Petrovsky, N. Delta inulin adjuvant enhances plasmablast generation, expression of activation-induced cytidine deaminase and B cell affinity maturation in human subjects receiving seasonal influenza vaccine. PLoS ONE 2015, 10, e0130089. [Google Scholar] [CrossRef] [PubMed]
- Toporovski, R.; Morrow, M.P.; Weiner, D.B. Interferons as potential adjuvants in prophylactic vaccines. Expert Opin. Biol. Ther. 2010, 10, 1489–1500. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Li, Y.; Zhang, S.; Chen, Y.; Su, W.; Sanchez, D.J.; Mai, J.D.H.; Zhi, X.; Chen, H.; Ding, X. A self-assembled graphene oxide adjuvant induces both enhanced humoral and cellular immune responses in influenza vaccine. J. Control Release 2024, 365, 716–728. [Google Scholar] [CrossRef]
- Vakili, B.; Karami-Darehnaranji, M.; Mirzaei, E.; Karami, A.R.; Amirkhani, M.H.; Rahimi, A.R.; Nasab, M.J. Graphene oxide as a novel vaccine adjuvant. Int. Immunopharmacol. 2023, 125, 111062. [Google Scholar] [CrossRef] [PubMed]
- Gogoi, H.; Mani, R.; Bhatnagar, R. Re-inventing traditional aluminum-based adjuvants: Insight into a century of advancements. Int. Rev. Immunol. 2024, 44, 58–81. [Google Scholar] [CrossRef]
- Petljak, M.; Alexandrov, L.B.; Brammeld, J.S.; Price, S.; Wedge, D.C.; Grossmann, S.; Dawson, K.J.; Ju, Y.S.; Iorio, F.; Tubio, J.M.C.; et al. Characterizing mutational signatures in human cancer cell lines reveals episodic APOBEC mutagenesis. Cell 2019, 176, 1282–1294.e20. [Google Scholar] [CrossRef]
- Lieber, M.R. Mechanisms of human lymphoid chromosomal translocations. Nat. Rev. Cancer 2016, 16, 387–398. [Google Scholar] [CrossRef]
- Reynaud, C.A.; Weill, J.C. Predicting AID off-targets: A step forward. J. Exp. Med. 2018, 215, 721–722. [Google Scholar] [CrossRef]
- Deng, W. AID in reprogramming: Quick and efficient. Identification of a key enzyme called AID, and its activity in DNA demethylation, may help to overcome a pivotal epigenetic barrier in reprogramming somatic cells toward pluripotency. Bioessays 2010, 32, 385–387. [Google Scholar] [CrossRef]
- Fritz, E.L.; Papavasiliou, F.N. Cytidine deaminases: AIDing DNA demethylation? Genes Dev. 2010, 24, 2107–2114. [Google Scholar] [CrossRef]
- Dominguez, P.M.; Shaknovich, R. Epigenetic function of activation-induced cytidine deaminase and its link to lymphomagenesis. Front. Immunol. 2014, 5, 642. [Google Scholar] [CrossRef]
- Jiao, J.; Jin, Y.; Zheng, M.; Zhang, H.; Yuan, M.; Lv, Z.; Odhiambo, W.; Yu, X.; Zhang, P.; Li, C.; et al. AID and TET2 co-operation modulates FANCA expression by active demethylation in diffuse large B cell lymphoma. Clin. Exp. Immunol. 2019, 195, 190–201. [Google Scholar] [CrossRef]
- Mohri, T.; Nagata, K.; Kuwamoto, S.; Matsushita, M.; Sugihara, H.; Kato, M.; Horie, Y.; Murakami, I.; Hayashi, K. Aberrant expression of AID and AID activators of NF-κB and PAX5 is irrelevant to EBV-associated gastric cancers, but is associated with carcinogenesis in certain EBV-non-associated gastric cancers. Oncol. Lett. 2017, 13, 4133–4140. [Google Scholar] [CrossRef]
- Araki, A.; Jin, L.; Nara, H.; Takeda, Y.; Nemoto, N.; Gazi, M.Y.; Asao, H. IL-21 enhances the development of colitis-associated colon cancer: Possible involvement of activation-induced cytidine deaminase expression. J. Immunol. 2019, 202, 3326–3333. [Google Scholar] [CrossRef]
- Cui, M.; Huang, J.; Zhang, S.; Liu, Q.; Liao, Q.; Qiu, X. Immunoglobulin expression in cancer cells and its critical roles in tumorigenesis. Front. Immunol. 2021, 12, 613530. [Google Scholar] [CrossRef]
- Petljak, M.; Dananberg, A.; Chu, K.; Bergstrom, E.N.; Striepen, J.; von Morgen, P.; Chen, Y.; Shah, H.; Sale, J.E.; Alexandrov, L.B.; et al. Mechanisms of APOBEC3 mutagenesis in human cancer cells. Nature 2022, 607, 799–807. [Google Scholar] [CrossRef]
- Vile, R.G.; Melcher, A.; Pandha, H.; Harrington, K.; Gunn, G.; Fruehauf, S.; Rampling, S.; Kaufman, J.; Gough, M.; Russell, S.J. APOBEC and cancer viroimmunotherapy: Thinking the unthinkable. Clin. Cancer Res. 2021, 27, 3280–3290. [Google Scholar] [CrossRef]
- Butler, K.; Banday, A.R. APOBEC3-mediated mutagenesis in cancer: Causes, clinical significance and therapeutic potential. J. Hematol. Oncol. 2023, 16, 31. [Google Scholar] [CrossRef]
- Guo, H.; Zhu, L.; Huang, L.; Sun, Z.; Zhang, H.; Nong, B.; Xiong, Y. APOBEC alteration contributes to tumor growth and immune escape in pan-cancer. Cancers 2022, 14, 2827. [Google Scholar] [CrossRef]
- Mao, Y.; Lv, M.; Zhang, Y.; Nie, G.; Cui, J.; Wang, Y.; Wang, Y.; Cao, W.; Liu, X.; Wang, X.; et al. APOBEC3B expression and its prognostic potential in breast cancer. Oncol. Lett. 2020, 19, 3205–3214. [Google Scholar] [CrossRef]
- DiMarco, A.V.; Qin, X.; McKinney, B.J.; Garcia, N.M.G.; Van Alsten, S.C.; Mendes, E.A.; Force, J.; Hanks, B.A.; Troester, M.A.; Owzar, K.; et al. APOBEC mutagenesis inhibits breast cancer growth through induction of T cell-mediated antitumor immune responses. Cancer Immunol. Res. 2022, 10, 70–86. [Google Scholar] [CrossRef]
- Asaoka, M.; Patnaik, S.K.; Ishikawa, T.; Takabe, K. Different members of the APOBEC3 family of DNA mutators have opposing associations with the landscape of breast cancer. Am. J. Cancer Res. 2021, 11, 5111. [Google Scholar]
- Glaser, A.P.; Fantini, D.; Wang, Y.; Yu, Y.; Rimar, K.J.; Podojil, J.R.; Miller, S.D.; Meeks, J.J. APOBEC-mediated mutagenesis in urothelial carcinoma is associated with improved survival, mutations in DNA damage response genes, and immune response. Oncotarget 2017, 9, 4537–4548. [Google Scholar] [CrossRef]
- Shi, R.; Wang, X.; Wu, Y.; Xu, B.; Zhao, T.; Trapp, C.; Wang, X.; Unger, K.; Zhou, C.; Lu, S.; et al. APOBEC-mediated mutagenesis is a favorable predictor of prognosis and immunotherapy for bladder cancer patients: Evidence from pan-cancer analysis and multiple databases. Theranostics 2022, 12, 4181–4199. [Google Scholar] [CrossRef]
- Wang, S.; Jia, M.; He, Z.; Liu, X.S. APOBEC3B and APOBEC mutational signature as potential predictive markers for immunotherapy response in non-small cell lung cancer. Oncogene 2018, 37, 3924–3936. [Google Scholar] [CrossRef]
- Chen, H.; Chong, W.; Teng, C.; Yao, Y.; Wang, X.; Li, X. The immune response related mutational signatures and driver genes in non-small-cell lung cancer. Cancer Sci. 2019, 110, 2348–2356. [Google Scholar] [CrossRef]
- Rüder, U.; Denkert, C.; Kunze, C.A.; Jank, P. APOBEC3B protein expression and mRNA analyses in patients with high-grade serous ovarian carcinoma. Histol. Histopathol. 2019, 34, 405–417. [Google Scholar]
- Xia, S.; Gu, Y.; Zhang, H.; Zhang, C.L.; Wang, Y.; Gao, L.; Zhang, W.; Zhang, J.Y.; Cheng, X.X.; Li, J.; et al. Immune inactivation by APOBEC3B enrichment predicts response to chemotherapy and survival in gastric cancer. Oncoimmunology 2021, 10, 1975386. [Google Scholar] [CrossRef]
- Sharma, S.; Patnaik, S.K.; Kemer, Z.; Baysal, B.E. Transient overexpression of exogenous APOBEC3A causes C-to-U RNA editing of thousands of genes. RNA Biol. 2017, 14, 603–610. [Google Scholar] [CrossRef]
- Mamrot, J.; Balachandran, S.; Steele, E.J.; Lindley, R.A. Molecular model linking Th2 polarized M2 tumour-associated macrophages with deaminase-mediated cancer progression mutation signatures. Scand. J. Immunol. 2019, 89, e12760. [Google Scholar] [CrossRef]
- Alqassim, E.Y.; Sharma, S.; Khan, A.N.M.N.H.; Emmons, T.R.; Cortes Gomez, E.; Alahmari, A.; Singel, K.L.; Mark, J.; Davidson, B.A.; McGray, A.J.R.; et al. RNA editing enzyme APOBEC3A promotes pro-inflammatory M1 macrophage polarization. Commun. Biol. 2021, 4, 102. [Google Scholar] [CrossRef]
- Tan, X.; Zheng, S.; Liu, W.; Wang, Q.; Li, Y.; Chen, J.; Huang, M. Effect of APOBEC3A functional polymorphism on renal cell carcinoma is influenced by tumor necrosis factor-α and transcriptional repressor ETS1. Am. J. Cancer Res. 2021, 11, 4347–4363. [Google Scholar]
- Liu, J.; Wang, F.; Zhang, Y.; Liu, J.; Zhao, B. ADAR1-mediated RNA editing and its role in cancer. Front. Cell Dev. Biol. 2022, 10, 956649. [Google Scholar] [CrossRef]
- Bader, S.B.; Ma, T.S.; Simpson, C.J.; Liang, J.; Maezono, S.E.B.; Olcina, M.M.; Buffa, F.M.; Hammond, E.M. Replication catastrophe induced by cyclic hypoxia leads to increased APOBEC3B activity. Nucleic Acids Res. 2021, 49, 7492–7506. [Google Scholar] [CrossRef]
- Grillo, M.J.; Jones, K.F.M.; Carpenter, A.M.; Harris, R.S.; Harki, D.A. The current toolbox for APOBEC drug discovery. Trends Pharmacol. Sci. 2022, 43, 362–377. [Google Scholar] [CrossRef]
- Green, M.; Budagyan, K.; Hayer, K.E.; Reed, M.A.; Savani, M.R.; Wertheim, G.B.; Weitzman, M.D. Cytosine deaminase APOBEC3A sensitizes leukemia cells to inhibition of the DNA replication checkpoint. Cancer Res. 2017, 77, 4579–4588. [Google Scholar] [CrossRef]
- Buisson, R.; Lawrence, M.S.; Benes, C.H.; Zou, L. APOBEC3A and APOBEC3B activities render cancer cells susceptible to ATR inhibition. Cancer Res. 2017, 77, 4567–4578. [Google Scholar] [CrossRef]
- Nikkilä, J.; Kumar, R.; Campbell, J.; Brandsma, I.; Pemberton, H.N.; Wallberg, F.; Nagy, K.; Scheer, I.; Vertessy, B.G.; Serebrenik, A.A.; et al. Elevated APOBEC3B expression drives a kataegic-like mutation signature and replication stress-related therapeutic vulnerabilities in p53-defective cells. Br. J. Cancer 2017, 117, 113–123. [Google Scholar] [CrossRef]
- Wörmann, S.M.; Zhang, A.; Thege, F.I.; Specht, C.D.; Gordon, J.A.; Wallace, M.N.; Ocañas, M.G.; Schenk, M.S.; Chung, D.H.; Morton, J.P.; et al. APOBEC3A drives deaminase domain-independent chromosomal instability to promote pancreatic cancer metastasis. Nat. Cancer 2021, 2, 1338–1356. [Google Scholar] [CrossRef]
- Fujiki, Y.; Yamamoto, Y.; Sueta, A.; Yamamoto-Ibusuki, M.; Goto-Yamaguchi, L.; Tomiguchi, M.; Takeshita, T.; Iwase, H. APOBEC3B gene expression as a novel predictive factor for pathological complete response to neoadjuvant chemotherapy in breast cancer. Oncotarget 2018, 9, 30513–30526. [Google Scholar] [CrossRef]
- Wang, F.; Dong, X.; Yang, F.; Li, X.; Zhang, Y.; Liu, J.; Chen, H.; Zhou, L. Comparative analysis of differentially mutated genes in non-muscle and muscle-invasive bladder cancer in the Chinese population by whole exome sequencing. Front. Genet. 2022, 13, 831146. [Google Scholar] [CrossRef]
- Jafarpour, S.; Yazdi, M.; Nedaeinia, R.; Ghobakhloo, S.; Salehi, R. Unfavorable prognosis and clinical consequences of APOBEC3B expression in breast and other cancers: A systematic review and meta-analysis. Tumour Biol. 2022, 44, 153–169. [Google Scholar] [CrossRef]
- Ma, C.; Liu, P.; Cui, S.; Gao, C.; Tan, X.; Liu, Z.; Xu, R. The identification of APOBEC3G as a potential prognostic biomarker in acute myeloid leukemia and a possible drug target for crotonoside. Molecules 2022, 27, 5804. [Google Scholar] [CrossRef]
- Isozaki, H.; Sakhtemani, R.; Abbasi, A.; Nikpour, N.; Stanzione, M.; Oh, S.; Langenbucher, A.; Monroe, S.; Su, W.; Cabanos, H.F.; et al. Therapy-induced APOBEC3A drives evolution of persistent cancer cells. Nature 2023, 620, 393–401. [Google Scholar] [CrossRef]
- Caswell, D.R.; Gui, P.; Mayekar, M.K.; Law, E.K.; Pich, O.; Bailey, C.; Boumelha, J.; Kerr, D.L.; Blakely, C.M.; Manabe, T.; et al. The role of APOBEC3B in lung tumor evolution and targeted cancer therapy resistance. Nat. Genet. 2024, 56, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Law, E.K.; Sieuwerts, A.M.; LaPara, K.; Leonard, B.; Starrett, G.J.; Molan, A.M.; Temiz, N.A.; Vogel, R.I.; Meijer-van Gelder, M.E.; Sweep, F.C.G.J.; et al. The DNA cytosine deaminase APOBEC3B promotes tamoxifen resistance in ER-positive breast cancer. Sci. Adv. 2016, 2, e1601737. [Google Scholar] [CrossRef]
- Lindley, R.A.; Humbert, P.; Larner, C.; Akmeemana, E.H.; Pendlebury, C.R. Association between targeted somatic mutation (TSM) signatures and HGS-OvCa progression. Cancer Med. 2016, 5, 2629–2640. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Wang, C.; Ma, X.; Sun, Y.; Yan, H.; Xie, L.; Zhang, Z. APOBEC3B, a molecular driver of mutagenesis in human cancers. Cell Biosci. 2017, 7, 29. [Google Scholar] [CrossRef]
- Olson, M.E.; Li, M.; Harris, R.S.; Harki, D.A. Small-molecule APOBEC3G DNA cytosine deaminase inhibitors based on a 4-amino-1,2,4-triazole-3-thiol scaffold. ChemMedChem 2013, 8, 112–117. [Google Scholar] [CrossRef]
- King, J.J.; Borzooee, F.; Im, J.; Asgharpour, M.; Ghorbani, A.; Diamond, C.P.; Fifield, H.; Berghuis, L.; Larijani, M. Structure-based design of first-generation small molecule inhibitors targeting the catalytic pockets of AID, APOBEC3A, and APOBEC3B. ACS Pharmacol. Transl. Sci. 2021, 4, 1390–1407. [Google Scholar] [CrossRef]
- Kvach, M.V.; Barzak, F.M.; Harjes, S.; Schares, H.A.M.; Jameson, G.B.; Ayoub, A.M.; Moorthy, R.; Aihara, H.; Harris, R.S.; Filichev, V.V.; et al. Inhibiting APOBEC3 activity with single-stranded DNA containing 2′-deoxyzebularine analogues. Biochemistry 2019, 58, 391–400. [Google Scholar] [CrossRef]
- Serrano, J.C.; VonTrentini, D.; Berrios, K.N.; Barka, A.; Dmochowski, I.J.; Kohli, R.M. Structure-guided design of a potent and specific inhibitor against the genomic mutator APOBEC3A. ACS Chem. Biol. 2022, 17, 3379–3388. [Google Scholar] [CrossRef]
- Driscoll, C.B.; Schuelke, M.R.; Kottke, T.; Thompson, J.M.; Wongthida, P.; Tonne, J.M.; Huff, A.L.; Miller, A.; Shim, K.G.; Molan, A.; et al. APOBEC3B-mediated corruption of the tumor cell immunopeptidome induces heteroclitic neoepitopes for cancer immunotherapy. Nat. Commun. 2020, 11, 790. [Google Scholar] [CrossRef]
- Yang, Y.; Okada, S.; Sakurai, M. Adenosine-to-inosine RNA editing in neurological development and disease. RNA Biol. 2021, 18, 999–1013. [Google Scholar] [CrossRef]
- Song, B.; Shiromoto, Y.; Minakuchi, M.; Nishikura, K. The role of RNA editing enzyme ADAR1 in human disease. Wiley Interdiscip. Rev. RNA 2022, 13, e1665. [Google Scholar] [CrossRef]
- Khermesh, K.; D’Erchia, A.M.; Barak, M.; Annese, A.; Wachtel, C.; Levanon, E.Y.; Picardi, E.; Eisenberg, E. Reduced levels of protein recoding by A-to-I RNA editing in Alzheimer’s disease. RNA 2016, 22, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, H.G.; Matos, V.J.; Park, S.; Pham, K.M.; Beal, P.A. Selective inhibition of ADAR1 using 8-azanebularine-modified RNA duplexes. Biochemistry 2023, 62, 1376–1387. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ji, W.; Zhao, J.; Song, J.; Zheng, S.; Chen, L.; Li, P.; Tan, X.; Ding, Y.; Pu, R.; et al. Transcriptional repression and apoptosis influence the effect of APOBEC3A/3B functional polymorphisms on biliary tract cancer risk. Int. J. Cancer 2022, 150, 1825–1837. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.R.; Slack, F.J. ADAR1 and its implications in cancer development and treatment. Trends Genet. 2022, 38, 821–830. [Google Scholar] [CrossRef]
- Ramirez-Moya, J.; Baker, A.R.; Slack, F.J.; Santiseban, P. ADAR1-mediated RNA editing is a novel oncogenic process in thyroid cancer and regulates miR-200 activity. Oncogene 2020, 39, 3738–3753. [Google Scholar] [CrossRef]
- Fumagalli, D.; Gacquer, D.; Rothe, F.; Lefort, A.; Libert, F.; Brown, D.; Kheddoumi, N.; Shlien, A.; Konopka, T.; Salgado, R.; et al. Principles governing A-to-I RNA editing in the breast cancer transcriptome. Cell Rep. 2015, 13, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Cottrell, K.A.; Torres, L.S.; Dizon, M.G.; Weber, J.D. 8-Azaadenosine and 8-chloroadenosine are not selective inhibitors of ADAR. Cancer Res. Commun. 2021, 1, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, J.; Zhu, Y.; Chen, L.; Fang, M.; Zhao, H.; Xu, W.; Liu, Z. Targeting ADAR1 with a small molecule for the treatment of prostate cancer. Nat. Cancer 2025, 6, 474–492. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.R.; Miliotis, C.; Ramírez-Moya, J.; Marc, T.; Vlachos, I.S.; Santisteban, P.; Slack, F.J. Transcriptome profiling of ADAR1 targets in triple-negative breast cancer cells reveals mechanisms for regulating growth and invasion. Mol. Cancer Res. 2022, 20, 960–970. [Google Scholar] [CrossRef]
- Binothman, N.; Aljadani, M.; Alghanem, B.; Refai, M.Y.; Rashid, M.; Al Tuwaijri, A.; Alsubhi, N.H.; Alrefaei, G.I.; Khan, M.Y.; Sonbul, S.N.; et al. Identification of novel interact partners of ADAR1 enzyme mediating the oncogenic process in aggressive breast cancer. Sci. Rep. 2023, 13, 8341. [Google Scholar] [CrossRef]
- Ishizuka, J.J.; Manguso, R.T.; Cheruiyot, C.K.; Ki, B.; Panda, A.; Iracheta-Vellve, A.; Miller, B.C.; Du, P.P.; Yates, K.B.; Dubrot, J.; et al. Loss of ADAR1 in tumours overcomes resistance to immune checkpoint blockade. Nature 2019, 565, 43–48. [Google Scholar] [CrossRef]
- Shiromoto, Y.; Sakurai, M.; Minakuchi, M.; Ariyoshi, K.; Nishikura, K. ADAR1 RNA editing enzyme regulates R-loop formation and genome stability at telomeres in cancer cells. Nat. Commun. 2021, 12, 1654. [Google Scholar] [CrossRef]
- Nakahama, T.; Kawahara, Y. Adenosine-to-inosine RNA editing in the immune system: Friend or foe? Cell Mol. Life Sci. 2020, 77, 2931–2948. [Google Scholar] [CrossRef]
- Tossberg, J.T.; Heinrich, R.M.; Farley, V.M.; Alexander, E.J.; Meeks, C.S.; Levin, A.S.; Cooper, T.R. Adenosine-to-inosine RNA editing of Alu double-stranded RNAs is markedly decreased in multiple sclerosis and unedited Alu dsRNAs are potent activators of proinflammatory transcriptional responses. J. Immunol. 2020, 205, 2606–2617. [Google Scholar] [CrossRef]
- Vlachogiannis, N.I.; Gatsiou, A.; Silvestris, D.A.; Sachse, E.; Stellos, T.; Bochenek, J.; Ayers, J.R.; Smith, J.L.; Dimmeler, J.M.; Engelhardt, J.F. RNA modifications in autoimmunity: The epitranscriptome in systemic lupus erythematosus and rheumatoid arthritis. J. Autoimmun. 2020, 106, 102329. [Google Scholar] [CrossRef]
- Chan, C.H.; Lin, C.H.; Qi, L.; Fei, J.; Li, Y.; Yong, Y.K.J.; Liu, M.; Song, Y.; Chow, R.K.; Ng, V.H.; et al. A disrupted RNA editing balance mediated by ADARs (adenosine deaminases that act on RNA) in human hepatocellular carcinoma. Gut 2014, 63, 832–843. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.H.; Qamra, A.; Tan, K.T.; Guo, J.; Yang, H.; Qi, L.; Lin, J.S.; Ng, V.H.; Song, Y.; Hong, H.; et al. ADAR-mediated RNA editing predicts progression and prognosis of gastric cancer. Gastroenterology 2016, 151, 637–650.e10. [Google Scholar] [CrossRef] [PubMed]
- Amin, E.M.; Liu, Y.; Deng, S.; Tan, K.S.; Chudgar, N.; Mayo, M.W.; Sanchez-Vega, F.; Adusumilli, P.S.; Schultz, N.; Jones, D.R. The RNA-editing enzyme ADAR promotes lung adenocarcinoma migration and invasion by stabilizing FAK. Sci. Signal. 2017, 10, eaah3941. [Google Scholar] [CrossRef] [PubMed]
- Shimokawa, T.; Rahman, M.F.-U.; Tostar, U.; Sonkoly, E.; Ståhle, M.; Pivarcsi, A.; Palaniswamy, R.; Zaphiropoulos, P. RNA editing of the GLI1 transcription factor modulates the output of Hedgehog signaling. RNA Biol. 2013, 10, 321–333. [Google Scholar] [CrossRef]
- Galeano, F.; Rossetti, C.; Tomaselli, S.; Cifaldi, L.; Lezzerini, M.; Pezzullo, M.; Boldrini, R.; Massimi, L.; Di Rocco, C.M.; Locatelli, F.; et al. ADAR2-editing activity inhibits glioblastoma growth through the modulation of the CDC14B/Skp2/p21/p27 axis. Oncogene 2013, 32, 998–1009. [Google Scholar] [CrossRef]
- Tomaselli, S.; Galeano, F.; Alon, S.; Raho, R.; Galardi, E.; Polito, F.; de Santis, R.; Gallo, C.A.; Picardi, F.L.; Avolio, M.A.; et al. Modulation of microRNA editing, expression, and processing by ADAR2 deaminase in glioblastoma. Genome Biol. 2015, 16, 5. [Google Scholar] [CrossRef]
- Garcia-Manero, G.; McCloskey, J.; Griffiths, E.A.; Yee, K.W.L.; Zeidan, A.M.; Al-Kali, A.; Deeg, H.J.; Patel, P.A.; Sabloff, M.; Keating, M.M.; et al. Oral decitabine-cedazuridine versus intravenous decitabine for myelodysplastic syndromes and chronic myelomonocytic leukaemia (ASCERTAIN): A registrational, randomised, crossover, pharmacokinetics, phase 3 study. Lancet Haematol. 2024, 11, e15–e26. [Google Scholar] [CrossRef]
- Yanai, M.; Kojima, S.; Sakai, M.; Asano, S.; Nakamura, T.; Takeda, M.; Ikeda, Y.; Yokota, K.; Shirogane, T. ADAR2 is involved in self and nonself recognition of Borna disease virus genomic RNA in the nucleus. J. Virol. 2020, 94, e01513-19. [Google Scholar] [CrossRef]
- Aizawa, H.; Hideyama, T.; Yamashita, T.; Kimura, T.; Suzuki, N.; Aoki, M.; Kwak, S. Deficient RNA-editing enzyme ADAR2 in an amyotrophic lateral sclerosis patient with a FUS(P525L) mutation. J. Clin. Neurosci. 2016, 32, 128–129. [Google Scholar] [CrossRef]
- Wright, A.; Vissel, B. The essential role of AMPA receptor GluR2 subunit RNA editing in the normal and diseased brain. Front. Mol. Neurosci. 2012, 5, 34. [Google Scholar] [CrossRef]
- Katrekar, D.; Xiang, Y.; Palmer, N.; Saha, A.; Meluzzi, D.; Mali, P. Comprehensive interrogation of the ADAR2 deami-nase domain for engineering enhanced RNA editing activity and specificity. eLife 2022, 11, e75555. [Google Scholar] [CrossRef]
- Wettengel, J.; Reautschnig, P.; Geisler, S.; Kahle, P.J.; Stafforst, T. Harnessing human ADAR2 for RNA re-pair—Recoding a PINK1 mutation rescues mitophagy. Nucleic Acids Res. 2017, 45, 2797–2808. [Google Scholar]
- Cesarini, V.; Silvestris, D.A.; Galeano, F.; Tassinari, V.; Martini, M.; Locatelli, F.; Gallo, A. ADAR2 protein is associated with overall survival in GBM patients and its decrease triggers the anchorage-independent cell growth signature. Biomolecules 2022, 12, 1142. [Google Scholar] [CrossRef]
- Dhillon, S. Decitabine/Cedazuridine: First Approval. Drugs 2021, 81, 1623–1630, Erratum in Drugs 2021, 81, 179. [Google Scholar] [CrossRef]
- Yu, Y.; Leete, T.C.; Born, D.A.; Young, L.; Barrera, L.A.; Bertolini, E.; Wang, S.; Zhang, X.; Rees, H.A.; Gaudelli, N.M.; et al. Cytosine base editors with minimized unguided DNA and RNA off-target events and high on-target activity. Nat. Biotechnol. 2020, 38, 883–892. [Google Scholar] [CrossRef]
- Crews, L.A.; Ma, W.; Ladel, L.; Pham, J.; Balaian, L.; Steel, S.K.; Mondala, P.K.; Diep, R.H.; Wu, C.N.; Mason, C.N.; et al. Reversal of malignant ADAR1 splice isoform switching with Rebecsinib. Cell Stem Cell 2023, 30, 250–263.e6. [Google Scholar] [CrossRef] [PubMed]
- Wave Life Sciences. Wave Life Sciences Announces First-Ever Therapeutic RNA Editing in Humans Achieved in RestorAATion-2 Trial of WVE-006 in Alpha-1 Antitrypsin Deficiency. Globe Newswire 2024. Available online: https://www.globenewswire.com/news-release/2024/10/16/2964053/0/en/Wave-Life-Sciences-Announces-First-Ever-Therapeutic-RNA-Editing-in-Humans-Achieved-in-RestorAATion-2-Trial-of-WVE-006-in-Alpha-1-Antitrypsin-Deficiency.html (accessed on 16 November 2025).
- Slesarenko, Y.S.; Shmakov, S.A.; Makarova, K.S.; Koonin, E.V. Off-target effects of base editors: What we know and how we can reduce it. Genes 2022, 13, 1234. [Google Scholar] [CrossRef] [PubMed]
- Pecori, R.; Di Giorgio, S.; Roper, N.; Marangio, A.M.; Cavalieri, V.; Conticello, S.G. Functions and consequences of AID/APOBEC-mediated DNA and RNA deamination. Nat. Rev. Genet. 2022, 23, 505–518. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhao, W.; Li, S.; Luo, X.; Wei, Y.; Zhang, Y.; Ye, Z.; Li, S.; Wang, M.; Tan, J.; et al. Engineering of high-precision C-to-G base editors with expanded site selectivity and target compatibility. Nucleic Acids Res. 2025, 53, gkaf717. [Google Scholar] [CrossRef]
- Xu, W.; Yang, J.; Li, H.; Zhang, X.; Chen, Y.; Sun, L.; Wang, Y. Cutting-edge applications of base editing and prime editing in therapeutics. J. Transl. Med. 2024, 22, 100–112. [Google Scholar] [CrossRef]
- Tong, H.; Liu, N.; Wei, Y.; Zhou, Y.; Li, Y.; Wu, D.; Jin, M.; Cui, S.; Li, H.; Li, G.; et al. Programmable deaminase-free base editors for G-to-Y conversion by engineered glycosylase. Natl Sci Rev. 2023, 10, nwad143. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lindley, R.A. Deaminase Modulation Driving a New Era in Drug Development. Int. J. Mol. Sci. 2025, 26, 11532. https://doi.org/10.3390/ijms262311532
Lindley RA. Deaminase Modulation Driving a New Era in Drug Development. International Journal of Molecular Sciences. 2025; 26(23):11532. https://doi.org/10.3390/ijms262311532
Chicago/Turabian StyleLindley, Robyn A. 2025. "Deaminase Modulation Driving a New Era in Drug Development" International Journal of Molecular Sciences 26, no. 23: 11532. https://doi.org/10.3390/ijms262311532
APA StyleLindley, R. A. (2025). Deaminase Modulation Driving a New Era in Drug Development. International Journal of Molecular Sciences, 26(23), 11532. https://doi.org/10.3390/ijms262311532






