Hydrogel-Based Vitreous Substitutes
Abstract
1. Introduction
| Type | Examples | Advantages | Limitations | Applications | Ref. |
|---|---|---|---|---|---|
| Gases | SF6, C3F8 |
|
| Retinal detachment, macular hole surgery | [11,12,13] |
| Silicone Oils | 1000 cSt, 5000 cSt silicone oil |
|
| Complicated retinal detachment, proliferative vitreoretinopathy | [14,15,16] |
| Heavy Silicone Oils | Densiron-68, Oxane HD |
|
| Inferior retinal detachment | [17] |
| Perfluorocarbon Liquids (PFCLs) | PFO, perfluorodecalin |
|
| Retinal reattachment | [18,19,20] |
| Hydrogels (Natural) | Hyaluronic-acid-based, collagen |
|
| Experimental vitreous substitute | [21] |
| Hydrogels (Synthetic) | PEG-based, PVA, polyacrylamide |
|
| Vitreous replacement in preclinical studies | [8,22] |
| Smart Hydrogels | Thermo-responsive, pH-sensitive |
|
| Targeted drug delivery, long-acting tamponade | [23,24] |
| Hydrophilic Polymers | PVA, polyHEMA |
|
| Vitreous substitute | [25] |
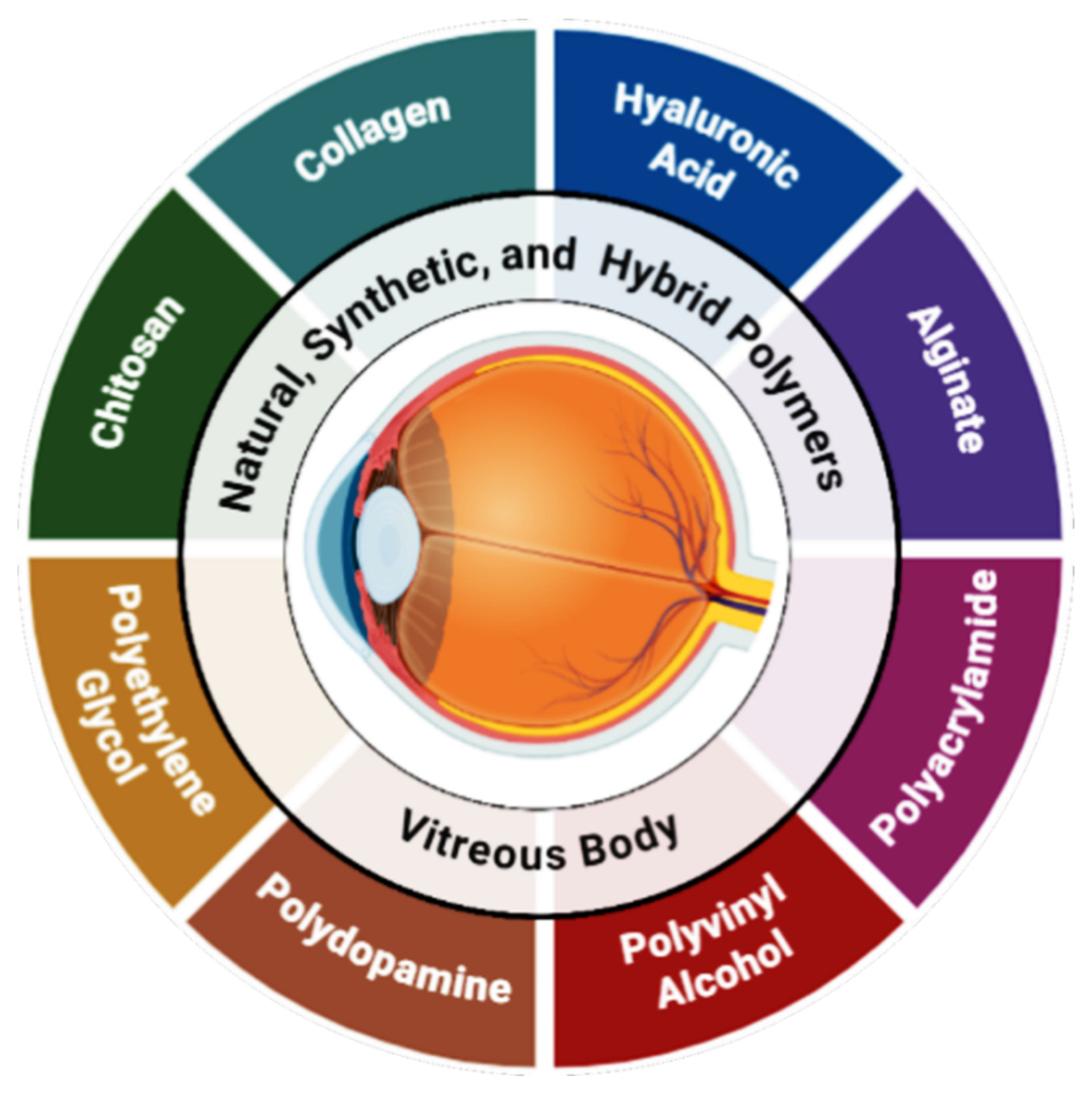
2. Types of Polymers
2.1. Natural Polymers
2.2. Synthetic Polymers
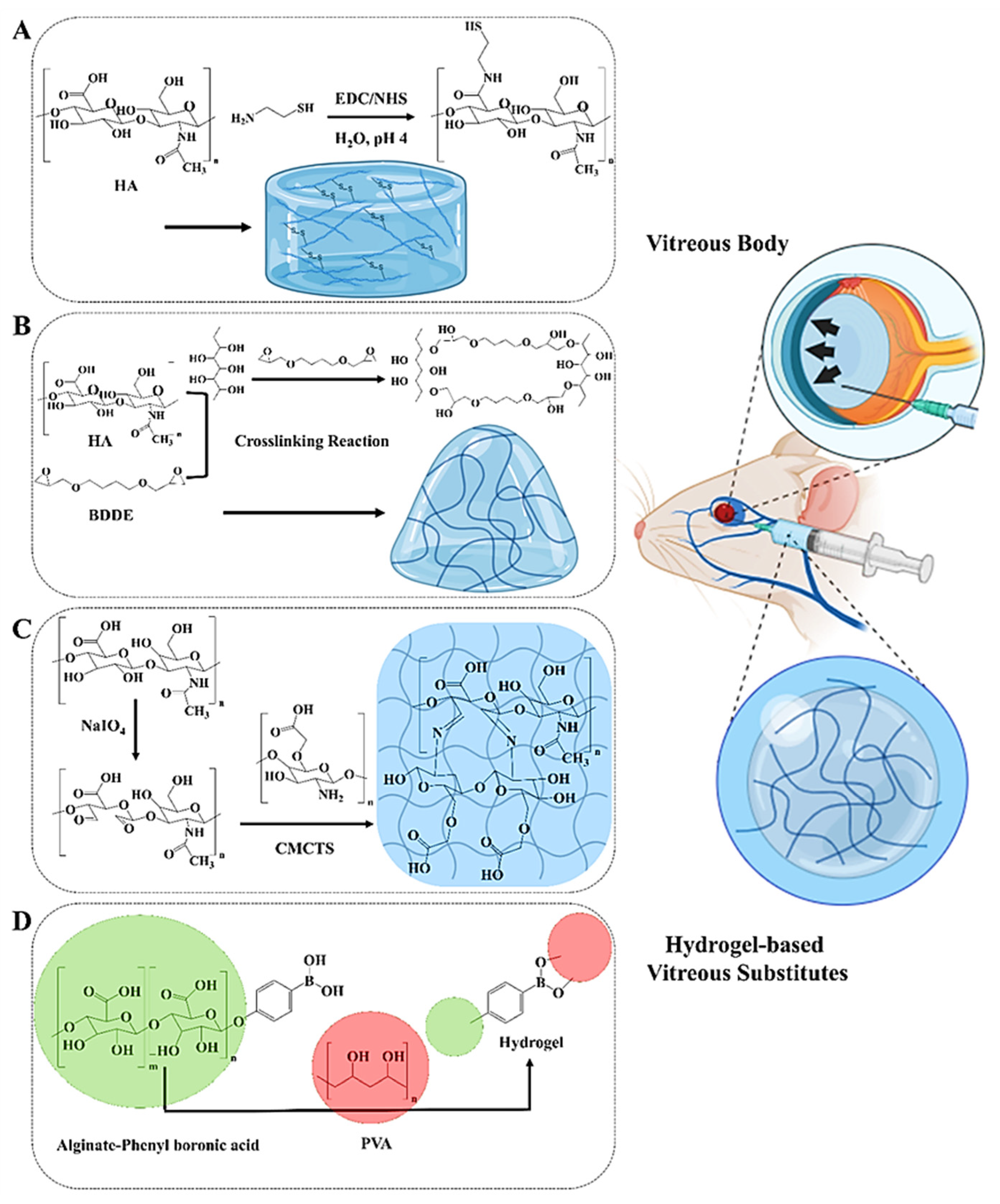
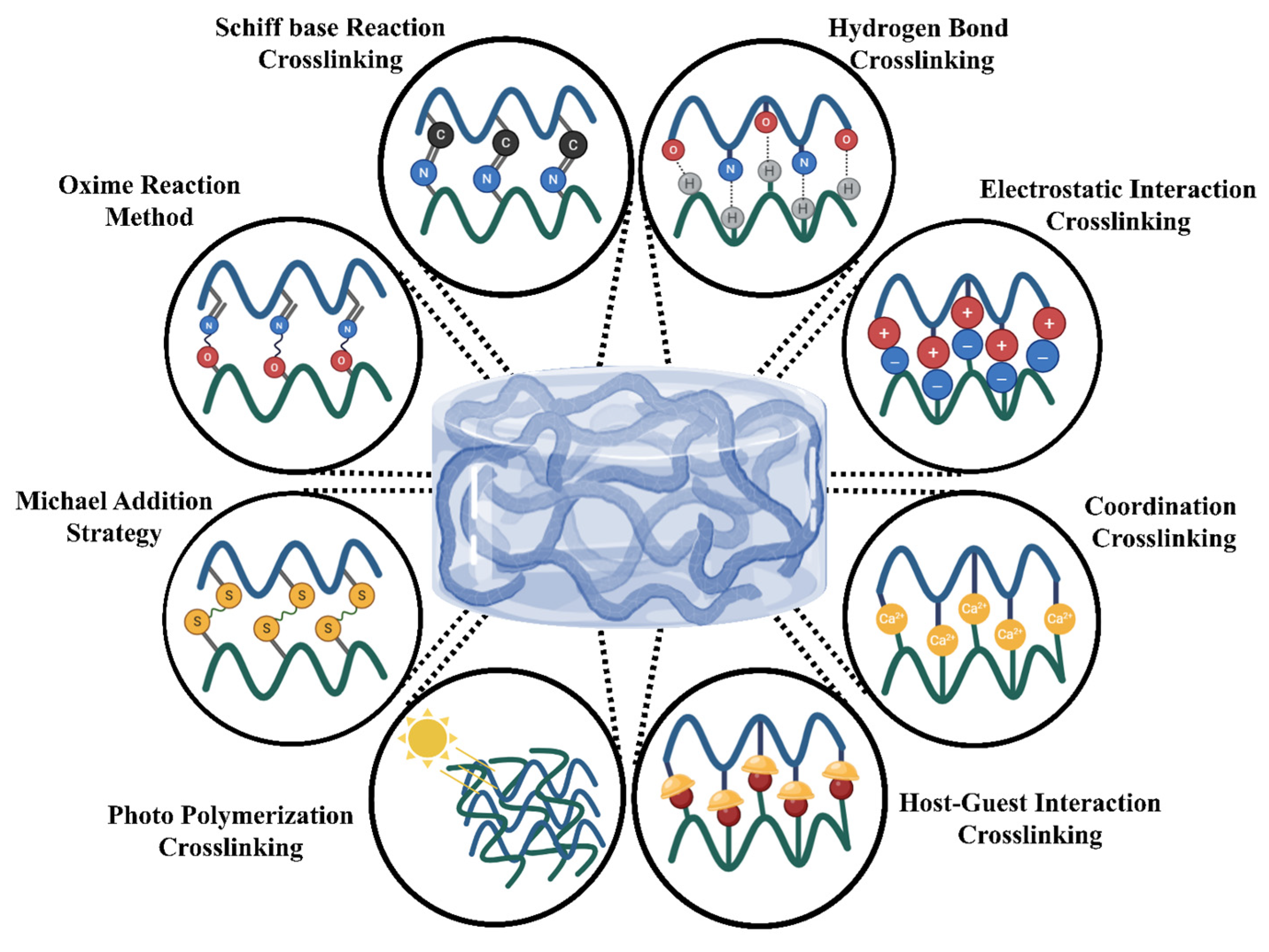
3. Crosslinking Strategies in Hydrogel Design
3.1. Physical Crosslinking Methods
3.2. Chemical Crosslinking Methods
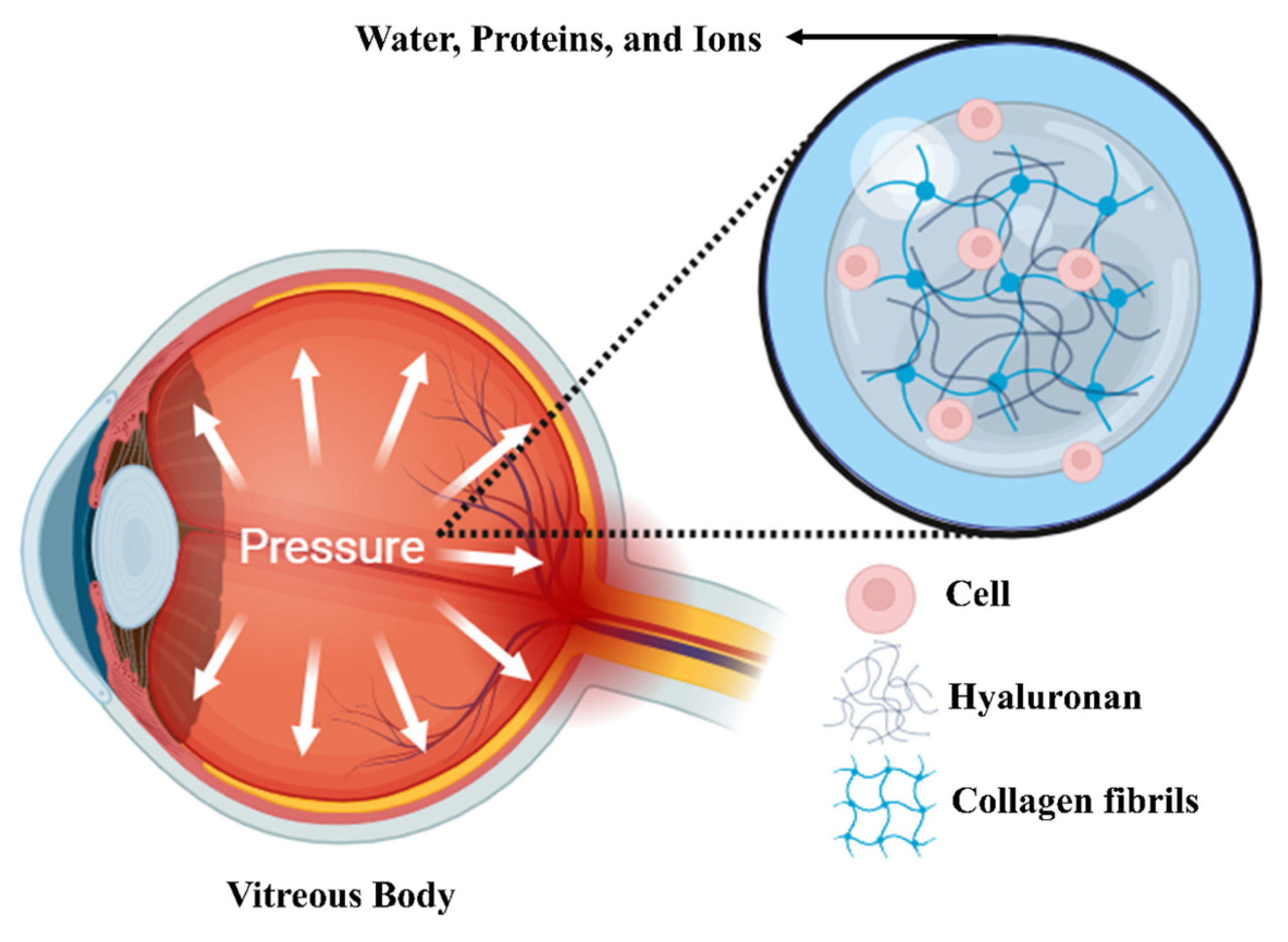
4. Mechanical Properties of Hydrogel Vitreous Substitutes
4.1. Mechanical Characterization of Hydrogel-Based Vitreous Substitutes
4.2. Aging and Dehydration Effects on Mechanical Stability
5. Recent Advances in Smart Hydrogel Systems
6. Evolution of Intraocular Tamponade Agents in Clinical Practice
7. Challenges and Future Directions in Hydrogel-Based Vitreous Substitutes
8. Summary and Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Lin, Q.; Lim, J.Y.; Xue, K.; Su, X.; Loh, X.J. Polymeric hydrogels as a vitreous replacement strategy in the eye. Biomaterials 2021, 268, 120547. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Yang, J.; Liu, Y.; Xie, X.; Hao, H.; Xing, X.; Liu, W. An in situ-forming polyzwitterion hydrogel: Towards vitreous substitute application. Bioact. Mater. 2021, 6, 3085–3096. [Google Scholar] [CrossRef] [PubMed]
- Issa, R.; Xia, T.; Zarbin, M.A.; Bhagat, N. Silicone oil removal: Post-operative complications. Eye 2020, 34, 537–543. [Google Scholar] [CrossRef]
- Gorimanipalli, B.; Muthu, S.; Shetty, R.; Ranade, R.R.; Khamar, P. Nexus of ocular motility and dry eye. Indian J. Ophthalmol. 2023, 71, 1546–1550. [Google Scholar] [CrossRef]
- Yadav, I.; Purohit, S.D.; Singh, H.; Bhushan, S.; Yadav, M.K.; Velpandian, T.; Chawla, R.; Hazra, S.; Mishra, N.C. Vitreous substitutes: An overview of the properties, importance, and development. J. Biomed. Mater. Res. Part B Appl. Biomater. 2021, 109, 1156–1176. [Google Scholar] [CrossRef]
- Kodavaty, J. Poly (vinyl alcohol) and hyaluronic acid hydrogels as potential biomaterial systems-A comprehensive review. J. Drug Deliv. Sci. Technol. 2022, 71, 103298. [Google Scholar] [CrossRef]
- Mishra, D.; Gade, S.; Glover, K.; Sheshala, R.; Singh, T.R.R. Vitreous humor: Composition, characteristics and implication on intravitreal drug delivery. Curr. Eye Res. 2023, 48, 208–218. [Google Scholar] [CrossRef]
- Yu, S.; Wang, S.; Xia, L.; Hu, H.; Zou, M.; Jiang, Z.; Chi, J.; Zhang, Y.; Li, C.; Yang, W.; et al. Injectable self-crosslinking hydrogels based on hyaluronic acid as vitreous substitutes. Int. J. Biol. Macromol. 2022, 208, 159–171. [Google Scholar] [CrossRef]
- Donati, S.; Caprani, S.M.; Airaghi, G.; Vinciguerra, R.; Bartalena, L.; Testa, F.; Mariotti, C.; Porta, G.; Simonelli, F.; Azzolini, C. Vitreous substitutes: The present and the future. Biomed. Res. Int. 2014, 2014, 351804. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Ran, R.; Ma, Y.; Zhang, M. Polymeric hydrogel as a vitreous substitute: Current research, challenges, and future directions. Biomed. Mater. 2021, 16, 042012. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.S.; Smiddy, W.E.; Feuer, W.J.; Shi, W. Outcomes of sulfur hexafluoride (SF6) versus perfluoropropane (C3F8) gas tamponade for macular hole surgery. Retina 2008, 28, 1408–1415. [Google Scholar] [CrossRef]
- Casini, G.; Loiudice, P.; De Cillà, S.; Radice, P.; Nardi, M. Sulfur hexafluoride (SF6) versus perfluoropropane (C3F8) tamponade and short term face-down position for macular hole repair: A randomized prospective study. Int. J. Retin. Vitr. 2016, 2, 10. [Google Scholar] [CrossRef]
- Gurunadh, V.; Banarji, A.; Patyal, S.; Upadhyay, A.; Ahluwalia, T.; Gupta, R.; Bhaduria, M. Evaluation of vitreous substitutes in managing complicated vitreo-retinal surgeries. Med. J. Armed Forces India 2010, 66, 125–128. [Google Scholar] [CrossRef]
- Cibis, P.A.; Becker, B.; Okun, E.; Canaan, S. The use of liquid silicone in retinal detachment surgery. Arch. Ophthalmol. 1962, 68, 590–599. [Google Scholar] [CrossRef]
- Franks, W.; Leaver, P. Removal of silicone oil—Rewards and penalties. Eye 1991, 5, 333–337. [Google Scholar] [CrossRef]
- Zafar, S.; Shakir, M.; Mahmood, S.A.; Amin, S.; Iqbal, Z. Comparison of 1000-centistoke versus 5000-centistoke silicone oil in complex retinal detachment surgery. J. Coll. Physicians Surg. Pak. 2016, 26, 36–40. [Google Scholar] [PubMed]
- Shettigar, M.P.; Dave, V.P.; Chou, H.-D.; Fung, A.; Iguban, E.; de Ribot, F.M.; Zabala, C.; Hsieh, Y.-T.; Lalwani, G. Vitreous substitutes and tamponades–A review of types, applications, and future directions. Indian J. Ophthalmol. 2024, 72, 1102–1111. [Google Scholar] [CrossRef] [PubMed]
- Vaziri, K.; Schwartz, S.; Leffler, C.; Flynn, H. Surgical tamponade in the treatment of retinal detachment. Retin Physician 2016, 12, 32–37. [Google Scholar]
- Yu, Q.; Liu, K.; Su, L.; Xia, X.; Xu, X. Perfluorocarbon liquid: Its application in vitreoretinal surgery and related ocular inflammation. BioMed Res. Int. 2014, 2014, 250323. [Google Scholar] [CrossRef]
- Bhurayanontachai, P.; Seepongphun, U. Outcomes of a postoperative perfluorocarbon liquid tamponade for complex retinal detachments: 12 years of experience in southern Thailand. BMC Ophthalmol. 2020, 20, 358. [Google Scholar] [CrossRef] [PubMed]
- Laradji, A.; Shui, Y.-B.; Karakocak, B.B.; Evans, L.; Hamilton, P.; Ravi, N. Bioinspired thermosensitive hydrogel as a vitreous substitute: Synthesis, properties, and progress of animal studies. Materials 2020, 13, 1337. [Google Scholar] [CrossRef]
- Pritchard, C.D.; Crafoord, S.; Andréasson, S.; Arnér, K.M.; O’Shea, T.M.; Langer, R.; Ghosh, F.K. Evaluation of viscoelastic poly (ethylene glycol) sols as vitreous substitutes in an experimental vitrectomy model in rabbits. Acta Biomater. 2011, 7, 936–943. [Google Scholar] [CrossRef]
- Zhang, K.; Xue, K.; Loh, X.J. Thermo-responsive hydrogels: From recent progress to biomedical applications. Gels 2021, 7, 77. [Google Scholar] [CrossRef] [PubMed]
- Thacker, M.; Tseng, C.-L.; Lin, F.-H. Substitutes and colloidal system for vitreous replacement and drug delivery: Recent progress and future prospective. Polymers 2020, 13, 121. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Chen, H.; Liu, Y.; Huang, Z.; Sun, X.; Zhou, L.; Lu, X.; Gao, Q. A novel vitreous substitute of using a foldable capsular vitreous body injected with polyvinylalcohol hydrogel. Sci. Rep. 2013, 3, 1838. [Google Scholar] [CrossRef]
- Su, X.; Tan, M.J.; Li, Z.; Wong, M.; Rajamani, L.; Lingam, G.; Loh, X.J. Recent progress in using biomaterials as vitreous substitutes. Biomacromolecules 2015, 16, 3093–3102. [Google Scholar] [CrossRef]
- Schulz, A.; Wakili, P.; Januschowski, K.; Heinz, W.R.; Engelhard, M.; Menz, H.; Szurman, P. Safety and performance assessment of hyaluronic acid-based vitreous substitutes in patients with phthisis bulbi. Acta Ophthalmol. 2023, 101, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-A.; Tai, Y.-N.; Hsieh, E.-H.; Thacker, M.; Lin, I.-C.; Tseng, C.-L.; Lin, F.-H. Injectable cross-linked hyaluronic acid hydrogels with epigallocatechin gallate loading as vitreous substitutes. Int. J. Biol. Macromol. 2024, 275, 133467. [Google Scholar] [CrossRef]
- Lorenzo Lopez, M.; Kearns, V.R.; Curran, J.M.; Patterson, E.A. Diffusion of nanoparticles in heterogeneous hydrogels as vitreous humour in vitro substitutes. Sci. Rep. 2024, 14, 17441. [Google Scholar] [CrossRef]
- Schramm, C.; Spitzer, M.S.; Henke-Fahle, S.; Steinmetz, G.; Januschowski, K.; Heiduschka, P.; Geis-Gerstorfer, J.; Biedermann, T.; Bartz-Schmidt, K.U.; Szurman, P. The cross-linked biopolymer hyaluronic acid as an artificial vitreous substitute. Investig. Ophthalmol. Vis. Sci. 2012, 53, 613–621. [Google Scholar] [CrossRef]
- Barth, H.; Crafoord, S.; Andréasson, S.; Ghosh, F. A cross-linked hyaluronic acid hydrogel (Healaflow®) as a novel vitreous substitute. Graefe’s Arch. Clin. Exp. Ophthalmol. 2016, 254, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Suri, S.; Banerjee, R. In vitro evaluation of in situ gels as short term vitreous substitutes. J. Biomed. Mater. Res. Part A Off. J. Soc. Biomater. Jpn. Soc. Biomater. Aust. Soc. Biomater. Korean Soc. Biomater. 2006, 79, 650–664. [Google Scholar] [CrossRef]
- Pellá, M.C.; Lima-Tenório, M.K.; Tenório-Neto, E.T.; Guilherme, M.R.; Muniz, E.C.; Rubira, A.F. Chitosan-based hydrogels: From preparation to biomedical applications. Carbohydr. Polym. 2018, 196, 233–245. [Google Scholar] [CrossRef]
- Wang, S.; Chi, J.; Jiang, Z.; Hu, H.; Yang, C.; Liu, W.; Han, B. A self-healing and injectable hydrogel based on water-soluble chitosan and hyaluronic acid for vitreous substitute. Carbohydr. Polym. 2021, 256, 117519. [Google Scholar] [CrossRef]
- Aminabhavi, T.; Dharupaneedi, S. Production of chitosan-based hydrogels for biomedical applications. In Chitosan Based Biomaterials; Elsevier: Amsterdam, The Netherlands, 2017; Volume 1, pp. 295–319. [Google Scholar]
- Gombos, G.; Berman, E. Chemical and clinical observations on the fate of various vitreous substitutes. Acta Ophthalmol. 1967, 45, 794–806. [Google Scholar] [CrossRef]
- Oosterhuis, J.; Van Haeringen, N.; Jeltes, I.; Glasius, E. Polygeline as a vitreous substitute: I. Observations in rabbits. Arch. Ophthalmol. 1966, 76, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, M.; Tanaka, M.; Miyata, T. Evaluation of collagen gel and hyaluronic acid as vitreous substitutes. Ophthalmic Res. 1997, 29, 409–420. [Google Scholar] [CrossRef]
- Liang, C.; Peyman, G.A.; Serracarbassa, P.; Calixto, N.; Chow, A.A.; Rao, P. An evaluation of methylated collagen as a substitute for vitreous and aqueous humor. Int. Ophthalmol. 1998, 22, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Avitabile, T.; Marano, F.; Castiglione, F.; Bucolo, C.; Cro, M.; Ambrosio, L.; Ferrauto, C.; Reibaldi, A. Biocompatibility and biodegradation of intravitreal hyaluronan implants in rabbits. Biomaterials 2001, 22, 195–200. [Google Scholar] [CrossRef]
- Naik, K.; du Toit, L.C.; Ally, N.; Choonara, Y.E. In vivo evaluation of a Nano-enabled therapeutic vitreous substitute for the precise delivery of triamcinolone to the posterior segment of the eye. Drug Deliv. Transl. Res. 2024, 14, 2668–2694. [Google Scholar] [CrossRef]
- Tarsitano, M.; Cristiano, M.C.; Fresta, M.; Paolino, D.; Rafaniello, C. Alginate-based composites for corneal regeneration: The optimization of a biomaterial to overcome its limits. Gels 2022, 8, 431. [Google Scholar] [CrossRef]
- Choi, G.; An, S.H.; Choi, J.-W.; Rho, M.S.; Park, W.C.; Jeong, W.J.; Cha, H.J. Injectable alginate-based in situ self-healable transparent hydrogel as a vitreous substitute with a tamponading function. Biomaterials 2024, 305, 122459. [Google Scholar] [CrossRef]
- Wright, B.; De Bank, P.A.; Luetchford, K.A.; Acosta, F.R.; Connon, C.J. Oxidized alginate hydrogels as niche environments for corneal epithelial cells. J. Biomed. Mater. Res. Part A 2014, 102, 3393–3400. [Google Scholar] [CrossRef]
- Kopeček, J. Hydrogel biomaterials: A smart future? Biomaterials 2007, 28, 5185–5192. [Google Scholar] [CrossRef] [PubMed]
- Vijayasekaran, S.; Chirila, T.V.; Hong, Y.; Tahija, S.G.; Dalton, P.D.; Constable, I.J.; McAllister, I.L. Poly (I-vinyl-2-pyrrolidinone) hydrogels as vitreous substitutes: Histopathological evaluation in the animal eye. J. Biomater. Sci. Polym. Ed. 1996, 7, 685–696. [Google Scholar] [CrossRef] [PubMed]
- Hogen-Esch, T.; Shah, K.; Fitzgerald, C. Development of injectable poly (glyceryl methacrylate) hydrogels for vitreous prosthesis. J. Biomed. Mater. Res. 1976, 10, 975–976. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Vigo, J.; Refojo, M.F.; Verstraeten, T. Evaluation of a viscoelastic solution of hydroxypropyl methylcellulose as a potential vitreous substitute. Retina 1990, 10, 148–152. [Google Scholar] [CrossRef]
- Maruoka, S.; Matsuura, T.; Kawasaki, K.; Okamoto, M.; Yoshiaki, H.; Kodama, M.; Sugiyama, M.; Annaka, M. Biocompatibility of polyvinylalcohol gel as a vitreous substitute. Curr. Eye Res. 2006, 31, 599–606. [Google Scholar] [CrossRef]
- Swindle, K.E.; Hamilton, P.D.; Ravi, N. Advancements in the development of artificial vitreous humor utilizing polyacrylamide copolymers with disulfide crosslinkers. Polym Prep 2006, 47, 56–60. [Google Scholar]
- Cavalieri, F.; Miano, F.; D’Antona, P.; Paradossi, G. Study of gelling behavior of poly (vinyl alcohol)-methacrylate for potential utilizations in tissue replacement and drug delivery. Biomacromolecules 2004, 5, 2439–2446. [Google Scholar] [CrossRef]
- Refojo, M.; Leong, F.L. Poly (methyl acrylate-co-hydroxyethyl acrylate) hydrogel implant material of strength and softness. J. Biomed. Mater. Res. 1981, 15, 497–509. [Google Scholar] [CrossRef]
- Chaterji, S.; Kwon, I.K.; Park, K. Smart polymeric gels: Redefining the limits of biomedical devices. Prog. Polym. Sci. 2007, 32, 1083–1122. [Google Scholar] [CrossRef]
- Plant, G.W.; Chirila, T.V.; Harvey, A.R. Implantation of collagen IV/poly (2-hydroxyethyl methacrylate) hydrogels containing Schwann cells into the lesioned rat optic tract. Cell Transplant. 1998, 7, 381–391. [Google Scholar] [CrossRef]
- Aliyar, H.A.; Hamilton, P.D.; Ravi, N. Refilling of ocular lens capsule with copolymeric hydrogel containing reversible disulfide. Biomacromolecules 2005, 6, 204–211. [Google Scholar] [CrossRef]
- Refojo, M.F.; Zauberman, H. Optical properties of gels designed for vitreous implantation. Investig. Ophthalmol. Vis. Sci. 1973, 12, 465–467. [Google Scholar]
- Annaka, M.; Mortensen, K.; Vigild, M.E.; Matsuura, T.; Tsuji, S.; Ueda, T.; Tsujinaka, H. Design of an injectable in situ gelation biomaterials for vitreous substitute. Biomacromolecules 2011, 12, 4011–4021. [Google Scholar] [CrossRef] [PubMed]
- Kinder, F.; Giannoudis, P.V.; Boddice, T.; Howard, A. The effect of an abnormal BMI on orthopaedic trauma patients: A systematic review and meta-analysis. J. Clin. Med. 2020, 9, 1302. [Google Scholar] [CrossRef]
- Shaer, C.; Oppenheimer, L.; Lin, A.; Ishida, H. Advanced carbon materials derived from polybenzoxazines: A review. Polymers 2021, 13, 3775. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Yang, J.; Xiao, S.; Hu, R.; Bhaway, S.M.; Vogt, B.D.; Zhang, M.; Chen, Q.; Ma, J.; Chang, Y. Salt-responsive polyzwitterionic materials for surface regeneration between switchable fouling and antifouling properties. Acta Biomater. 2016, 40, 62–69. [Google Scholar] [CrossRef]
- Wang, X.; Li, F.; Liu, X.; Zhang, H. Applications and recent developments of hydrogels in ophthalmology. ACS Biomater. Sci. Eng. 2023, 9, 5968–5984. [Google Scholar] [CrossRef]
- Wang, T.; Deng, J.; Ran, R.; Shi, W.; Gao, Y.; Ren, X.; Cao, J.; Zhang, M. In-situ forming PEG-engineering hydrogels with anti-fouling characteristics as an artificial vitreous body. Chem. Eng. J. 2022, 449, 137486. [Google Scholar] [CrossRef]
- Lamponi, S.; Leone, G.; Consumi, M.; Greco, G.; Magnani, A. In vitro biocompatibility of new PVA-based hydrogels as vitreous body substitutes. J. Biomater. Sci. Polym. Ed. 2012, 23, 555–575. [Google Scholar] [CrossRef] [PubMed]
- Mueller-Jensen, K. Polyacrylamide as an alloplastic vitreous implant. Albrecht Von Graefes Arch. Für Klin. Und Exp. Ophthalmol. 1973, 189, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Dhyani, A.; Kumar, G. Fabrication, Characterization, and in vitro Evaluation of Atenolol Loaded Microsponges for Ocular Delivery. Indian J. Pharm. Educ. Res. 2024, 58, s149–s157. [Google Scholar] [CrossRef]
- Lima, L.H.; Morales, Y.; Cabral, T. Ocular Biocompatibility of Poly-N-Isopropylacrylamide (pNIPAM). J. Ophthalmol. 2016, 2016, 5356371. [Google Scholar] [CrossRef]
- Mieler, W.; Drapala, P.; Benac, S.; Brey, E.; Perez-Luna, V.; Derwent, J.K. Thermo-responsive hydrogel drug delivery system for the posterior segment of the eye. Investig. Ophthalmol. Vis. Sci. 2008, 49, 3873. [Google Scholar]
- Chelu, M.; Musuc, A.M. Biomaterials-based hydrogels for therapeutic applications. In Biomaterials in Microencapsulation; IntechOpen: London, UK, 2024. [Google Scholar]
- Hu, W.; Wang, Z.; Xiao, Y.; Zhang, S.; Wang, J. Advances in crosslinking strategies of biomedical hydrogels. Biomater. Sci. 2019, 7, 843–855. [Google Scholar] [CrossRef]
- Maitra, J.; Shukla, V.K. Cross-linking in hydrogels-a review. Am. J. Polym. Sci 2014, 4, 25–31. [Google Scholar]
- Song, P.; Wang, H. High-performance polymeric materials through hydrogen-bond cross-linking. Adv. Mater. 2020, 32, 1901244. [Google Scholar] [CrossRef]
- Uesugi, K.; Sakaguchi, H.; Hayashida, Y.; Hayashi, R.; Baba, K.; Suganuma, Y.; Yokoi, H.; Tsujikawa, M.; Nishida, K. A self-assembling peptide gel as a vitreous substitute: A rabbit study. Investig. Ophthalmol. Vis. Sci. 2017, 58, 4068–4075. [Google Scholar] [CrossRef]
- Wang, H.; Wu, Y.; Cui, C.; Yang, J.; Liu, W. Antifouling super water absorbent supramolecular polymer hydrogel as an artificial vitreous body. Adv. Sci. 2018, 5, 1800711. [Google Scholar] [CrossRef]
- Yang, J.; Chen, Y.; Zhao, L.; Zhang, J.; Luo, H. Constructions and properties of physically cross-linked hydrogels based on natural polymers. Polym. Rev. 2023, 63, 574–612. [Google Scholar] [CrossRef]
- Kim, G.O.; Kim, N.; Kim, D.Y.; Kwon, J.S.; Min, B.-H. An electrostatically crosslinked chitosan hydrogel as a drug carrier. Molecules 2012, 17, 13704–13711. [Google Scholar] [CrossRef]
- Liu, X.; He, X.; Yang, B.; Lai, L.; Chen, N.; Hu, J.; Lu, Q. Dual physically cross-linked hydrogels incorporating hydrophobic interactions with promising repairability and ultrahigh elongation. Adv. Funct. Mater. 2021, 31, 2008187. [Google Scholar] [CrossRef]
- de Groot, J.H.; Spaans, C.J.; van Calck, R.V.; van Beijma, F.J.; Norrby, S.; Pennings, A.J. Hydrogels for an accommodating intraocular lens. An explorative study. Biomacromolecules 2003, 4, 608–616. [Google Scholar] [CrossRef]
- Raia, N.R.; Jia, D.; Ghezzi, C.E.; Muthukumar, M.; Kaplan, D.L. Characterization of silk-hyaluronic acid composite hydrogels towards vitreous humor substitutes. Biomaterials 2020, 233, 119729. [Google Scholar] [CrossRef]
- Mantooth, S.M.; Munoz-Robles, B.G.; Webber, M.J. Dynamic hydrogels from host–guest supramolecular interactions. Macromol. Biosci. 2019, 19, 1800281. [Google Scholar] [CrossRef] [PubMed]
- Sinawang, G.; Osaki, M.; Takashima, Y.; Yamaguchi, H.; Harada, A. Biofunctional hydrogels based on host–guest interactions. Polym. J. 2020, 52, 839–859. [Google Scholar] [CrossRef]
- Fang, G.; Zhao, R.; Zhu, L.; Wang, Q.; Peng, S.; Kang, L.; Lu, H.; Zhang, G.; Tang, B. Nanoemulsion-based pseudopolyrotaxane hydrogel for enhanced corneal bioavailability and treatment of corneal inflammation. J. Control. Release 2025, 379, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Cass, P.; Knower, W.; Pereeia, E.; Holmes, N.P.; Hughes, T. Preparation of hydrogels via ultrasonic polymerization. Ultrason. Sonochemistry 2010, 17, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Yuan, S.; Wang, J.; Shen, Y.; Deng, S.; Xie, L.; Yang, Q. The formation mechanism of hydrogels. Curr. Stem Cell Res. Ther. 2018, 13, 490–496. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, R.; Ouyang, C.; Wang, S.; Li, S.; Yin, X.; Deng, Z.; Han, B.; Chi, J. Photocurable Dual-Network Hydrogels Based on Natural Polymers for Sutureless Repair of Large Corneal Defects. Small 2025, 21, 2500150. [Google Scholar] [CrossRef]
- Liu, Y.; Weng, R.; Wang, W.; Wei, X.; Li, J.; Chen, X.; Liu, Y.; Lu, F.; Li, Y. Tunable physical and mechanical properties of gelatin hydrogel after transglutaminase crosslinking on two gelatin types. Int. J. Biol. Macromol. 2020, 162, 405–413. [Google Scholar] [CrossRef]
- Li, Z.; Lu, F.; Liu, Y. A review of the mechanism, properties, and applications of hydrogels prepared by enzymatic cross-linking. J. Agric. Food Chem. 2023, 71, 10238–10249. [Google Scholar] [CrossRef]
- Le Thi, P.; Lee, Y.; Nguyen, D.H.; Park, K.D. In situ forming gelatin hydrogels by dual-enzymatic cross-linking for enhanced tissue adhesiveness. J. Mater. Chem. B 2017, 5, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Hou, S.; Lake, R.; Park, S.; Edwards, S.; Jones, C.; Jeong, K.J. Injectable macroporous hydrogel formed by enzymatic cross-linking of gelatin microgels. ACS Appl. Bio Mater. 2018, 1, 1430–1439. [Google Scholar] [CrossRef] [PubMed]
- Tortora, M.; Cavalieri, F.; Chiessi, E.; Paradossi, G. Michael-type addition reactions for the in situ formation of poly (vinyl alcohol)-based hydrogels. Biomacromolecules 2007, 8, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Censi, R.; Fieten, P.J.; di Martino, P.; Hennink, W.E.; Vermonden, T. In situ forming hydrogels by tandem thermal gelling and Michael addition reaction between thermosensitive triblock copolymers and thiolated hyaluronan. Macromolecules 2010, 43, 5771–5778. [Google Scholar] [CrossRef]
- Chang, J.; Tao, Y.; Wang, B.; Guo, B.-H.; Xu, H.; Jiang, Y.-R.; Huang, Y. An in situ-forming zwitterionic hydrogel as vitreous substitute. J. Mater. Chem. B 2015, 3, 1097–1105. [Google Scholar] [CrossRef]
- Collins, J.; Xiao, Z.; Müllner, M.; Connal, L.A. The emergence of oxime click chemistry and its utility in polymer science. Polym. Chem. 2016, 7, 3812–3826. [Google Scholar] [CrossRef]
- Grover, G.N.; Lam, J.; Nguyen, T.H.; Segura, T.; Maynard, H.D. Biocompatible hydrogels by oxime click chemistry. Biomacromolecules 2012, 13, 3013–3017. [Google Scholar] [CrossRef]
- Baker, A.E.; Cui, H.; Ballios, B.G.; Ing, S.; Yan, P.; Wolfer, J.; Wright, T.; Dang, M.; Gan, N.Y.; Cooke, M.J. Stable oxime-crosslinked hyaluronan-based hydrogel as a biomimetic vitreous substitute. Biomaterials 2021, 271, 120750. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Liu, Y.; Hsu, S.-h. Hydrogels based on Schiff base linkages for biomedical applications. Molecules 2019, 24, 3005. [Google Scholar] [CrossRef]
- Pappalardo, R.; Boffito, M.; Cassino, C.; Caccamo, V.; Chiono, V.; Ciardelli, G. Schiff-base cross-linked hydrogels based on properly synthesized poly (ether urethane) s as potential drug delivery vehicles in the biomedical field: Design and characterization. ACS Omega 2024, 9, 45774–45788. [Google Scholar] [CrossRef] [PubMed]
- Chand, R.; Janarthanan, G.; Elkhoury, K.; Vijayavenkataraman, S. Digital light processing 3D bioprinting of biomimetic corneal stroma equivalent using gelatin methacryloyl and oxidized carboxymethylcellulose interpenetrating network hydrogel. Biofabrication 2025, 17, 025011. [Google Scholar] [CrossRef]
- Zhang, Y.; Xiong, Y.; Li, X.; Zhang, S.; Xu, W.; Chen, H.; Xu, L. Oxidization and Salting Out Synergistically Induced Highly Elastic, Conductive, and Sensitive Polyvinyl Alcohol Hydrogels. Adv. Funct. Mater. 2025, 35, 2415207. [Google Scholar] [CrossRef]
- Tram, N.K.; Jiang, P.; Torres-Flores, T.C.; Jacobs, K.M.; Chandler, H.L.; Swindle-Reilly, K.E. A hydrogel vitreous substitute that releases antioxidant. Macromol. Biosci. 2020, 20, 1900305. [Google Scholar] [CrossRef]
- Hammer, M.; Muuss, M.; Herbster, L.; Herth, J.; Scheuerle, A.; Khoramnia, R.; Łabuz, G.; Uhl, P.; Auffarth, G.U. Viscoelastic, optical, and surgical properties of vitreous body replacement hydrogels after aging compared to porcine vitreous bodies and silicone oils. Transl. Vis. Sci. Technol. 2024, 13, 5. [Google Scholar] [CrossRef]
- Xue, K.; Liu, Z.; Lin, Q.; Lim, J.Y.; Tang, K.Y.; Wong, S.L.; Parikh, B.H.; Su, X.; Loh, X.J. PCL-based thermogelling polymer: Molecular weight effects on its suitability as vitreous tamponade. ACS Appl. Bio Mater. 2020, 3, 9043–9053. [Google Scholar] [CrossRef]
- Tram, N.K.; Swindle-Reilly, K.E. Erratum in Rheological properties and age-related changes of the human vitreous humor. Front. Bioeng. Biotechnol. 2018, 6, 199. [Google Scholar] [CrossRef] [PubMed]
- Schulz, A.; Wahl, S.; Rickmann, A.; Ludwig, J.; Stanzel, B.V.; von Briesen, H.; Szurman, P. Age-related loss of human vitreal viscoelasticity. Transl. Vis. Sci. Technol. 2019, 8, 56. [Google Scholar] [CrossRef]
- Luo, R.H.; Tram, N.K.; Parekh, A.M.; Puri, R.; Reilly, M.A.; Swindle-Reilly, K.E. The roles of vitreous biomechanics in ocular disease, biomolecule transport, and pharmacokinetics. Curr. Eye Res. 2023, 48, 195–207. [Google Scholar] [CrossRef] [PubMed]
- Schulz, A.; Germann, A.; Heinz, W.R.; Engelhard, M.; Menz, H.; Rickmann, A.; Meiser, I.; Wien, S.; Wagner, S.; Januschowski, K. Translation of hyaluronic acid–based vitreous substitutes towards current regulations for medical devices. Acta Ophthalmol. 2023, 101, 422–432. [Google Scholar] [CrossRef]
- Stryjewski, T.P.; Stefater, J.A., III; Roth, L.; Li, J.; Awh, C.C.; Moshfeghi, D.M.; Kim, L.A.; Eliott, D. PYK-1105: Preclinical evaluation of a novel biodegradable vitreous substitute for retinal tamponade. J. Vitreoretin. Dis. 2021, 5, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Ma, S.; Wu, M.; Cui, H.; Wu, R.; Chen, S.; Xu, C.; Lu, X.; Feng, S. Self-assembling hydrogel loaded with 5-FU PLGA microspheres as a novel vitreous substitute for proliferative vitreoretinopathy. J. Biomed. Mater. Res. Part A 2020, 108, 2435–2446. [Google Scholar] [CrossRef] [PubMed]
- Leon-Cecilla, A.; Vazquez-Perez, F.J.; Gila-Vilchez, C.; Álvarez de Cienfuegos, L.; Lopez-Lopez, M.T. Alginate hydrogels reinforced by dehydration under stress—Application to a soft magnetic actuator. Gels 2023, 9, 39. [Google Scholar] [CrossRef]
- He, X.; Liu, R.; Liu, H.; Wang, R.; Xi, Z.; Lin, Y.; Wang, J. Facile preparation of tunicate-inspired chitosan hydrogel adhesive with self-healing and antibacterial properties. Polymers 2021, 13, 4322. [Google Scholar] [CrossRef]
- Lachance-Brais, C.; Yao, C.; Reyes-Valenzuela, A.; Asohan, J.; Guettler, E.; Sleiman, H.F. Exceptional nuclease resistance of DNA and RNA with the addition of small-molecule nucleobase mimics. J. Am. Chem. Soc. 2024, 146, 5811–5822. [Google Scholar] [CrossRef]
- Heimann, H.; Zou, X.; Jandeck, C.; Kellner, U.; Bechrakis, N.E.; Kreusel, K.-M.; Helbig, H.; Krause, L.; Schüler, A.; Bornfeld, N.; et al. Primary vitrectomy for rhegmatogenous retinal detachment: An analysis of 512 cases. Graefe’s Arch. Clin. Exp. Ophthalmol. 2006, 244, 69–78. [Google Scholar] [CrossRef]
- Novak, M.A.; Rice, T.A.; Michels, R.G.; Auer, C. Vitreous hemorrhage after vitrectomy for diabetic retinopathy. Ophthalmology 1984, 91, 1485–1489. [Google Scholar] [CrossRef]
- Han, D.P.; Pulido, J.S.; Mieler, W.F.; Johnson, M.W. Vitrectomy for proliferative diabetic retinopathy with severe equatorial fibrovascular proliferation. Am. J. Ophthalmol. 1995, 119, 563–570. [Google Scholar] [CrossRef]
- Ohm, J. Über die Behandlung der Netzhautablösung durch operative Entleerung der subretinalen Flüssigkeit und Einspritzung von Luft in den Glaskörper. Albrecht Von Graefes Arch. Für Ophthalmol. 1911, 79, 442–450. [Google Scholar] [CrossRef]
- Arruga, H.; Rhode, S.L. Present status of treatment of detachment of the retina. Arch. Ophthalmol. 1935, 13, 523–537. [Google Scholar] [CrossRef]
- Norton, E.; Aaberg, T.; Fung, W.; Curtin, V.T. Giant retinal tears. I. Clinical management with intravitreal air. Trans. Am. Ophthalmol. Soc. 1969, 67, 374. [Google Scholar]
- Chang, S.; Coleman, D.J.; Lincoff, H.; Wilcox, L.M., Jr.; Maisel, J.M.; Braunstein, R. Perfluoropropane gas in the management of proliferative vitreoretinopathy. Am. J. Ophthalmol. 1984, 98, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Modi, A.; Giridhar, A.; Gopalakrishnan, M. Sulfurhexafluoride (SF6) versus perfluoropropane (C3F8) gas as tamponade in macular hole surgery. Retina 2017, 37, 283–290. [Google Scholar] [CrossRef]
- Sayin, O.; Altinkaynak, H. Intravitreal C3F8 versus SF6: Effects on corneal tomography parameters and intraocular pressure in vitreomacular traction treatment. Int. Ophthalmol. 2025, 45, 118. [Google Scholar] [CrossRef]
- Wilson, D.I.; te Water Naude, A.D.; Snead, M.P. Refinements in the use of silicone oil as an intraocular tamponade. Eye 2024, 38, 1810–1815. [Google Scholar] [CrossRef]
- Barca, F.; Caporossi, T.; Rizzo, S. Silicone oil: Different physical proprieties and clinical applications. BioMed Res. Int. 2014, 2014, 502143. [Google Scholar] [CrossRef] [PubMed]
- Rush, R.B.; Penella, A.D.V.; Reinauer, R.M.; Rush, S.W.; Bastar, P.G. Silicone oil versus perfluoropropane gas tamponade during vitrectomy for tractional retinal detachment or fibrous proliferation: A randomized clinical trial. Retina 2021, 41, 1407–1415. [Google Scholar] [CrossRef] [PubMed]
- Valentín-Bravo, F.J.; García-Onrubia, L.; Andrés-Iglesias, C.; Valentín-Bravo, E.; Martín-Vallejo, J.; Pastor, J.C.; Usategui-Martín, R.; Pastor-Idoate, S. Complications associated with the use of silicone oil in vitreoretinal surgery: A systemic review and meta-analysis. Acta Ophthalmol. 2022, 100, e864–e880. [Google Scholar] [CrossRef]
- Lucke, K.H.; Foerster, M.H.; Laqua, H. Long-term results of vitrectomy and silicone oil in 500 cases of complicated retinal detachments. Am. J. Ophthalmol. 1987, 104, 624–633. [Google Scholar] [CrossRef]
- Lei, B.; Chen, X.; Zhou, M.; Zhang, T.; Gu, R.; Shu, Q.; Chang, Q.; Xu, G.; Jiang, R. Outcomes after silicone oil removal in patients with acute retinal necrosis who underwent vitrectomy. Ocul. Immunol. Inflamm. 2025, 33, 751–758. [Google Scholar] [CrossRef]
- Chen, Y.; Kearns, V.R.; Zhou, L.; Sandinha, T.; Lam, W.C.; Steel, D.H.; Chan, Y.K. Silicone oil in vitreoretinal surgery: Indications, complications, new developments and alternative long-term tamponade agents. Acta Ophthalmol. 2021, 99, 240–250. [Google Scholar] [CrossRef]
- Benchimol, A.; Denys, P.; Lejoyeux, R.; Bruneau, S.; Bonnin, S.; Couturier, A. Surgical outcomes of vitrectomy with gas or silicone oil tamponade for giant retinal tears. Ophthalmologica 2025, 248, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Al-Shehri, A.M.; Aljohani, S.; Aldihan, K.A.; Alrashedi, M.J.; Alrasheed, S.; Schatz, P. Effect of silicone oil versus gas tamponade on macular layer microstructure after pars plana vitrectomy for macula on rhegmatogenous retinal detachment. BMC Ophthalmol. 2024, 24, 119. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.S.; Mihalache, A.; Lau, T.H.A.; Popovic, M.M.; Kertes, P.J.; Muni, R.H. Pars plana vitrectomy with silicone oil or gas tamponade for uncomplicated retinal detachment: A systematic review and meta-analysis. Am. J. Ophthalmol. 2024, 266, 144–155. [Google Scholar] [CrossRef]
- Chang, S. Low viscosity liquid fluorochemicals in vitreous surgery. Am. J. Ophthalmol. 1987, 103, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Sun, X.; Su, M.; Wang, X.; Jiang, F. Perfluorocarbon liquid as a short-term tamponade for managing severe open globe injuries. Int. J. Retin. Vitr. 2025, 11, 35. [Google Scholar] [CrossRef]
- Mikhail, M.; Mangioris, G.; Best, R.; McGimpsey, S.; Chan, W. Management of giant retinal tears with vitrectomy and perfluorocarbon liquid postoperatively as a short-term tamponade. Eye 2017, 31, 1290–1295. [Google Scholar] [CrossRef]
- Ciardella, A.P.; Langton, K.; Chang, S. Intraocular dispersion of perfluorocarbon liquids insilicone oil. Am. J. Ophthalmol. 2003, 136, 365–367. [Google Scholar] [CrossRef]
- Kannan, N.B.; Vallinayagam, M.; Balakrishnan, T.; Dey Sarkar, A.; Rajan, R.P.; Ramasamy, K. Short-term endotamponade with perfluorocarbon liquids for giant retinal tear–associated retinal detachment. J. Vitreoretin. Dis. 2025, 9, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Pérez, S.M.P.; Arias, V.C.; Ramírez, D.J.; Martínez, C.E.; Tamayo Martínez, N. Efficacy of the use of perfluorocarbon as a temporary tamponade agent in severe ocular trauma and/or complex retinopexy: A scoping review. Int. J. Retin. Vitr. 2024, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Lincoff, H.; Mardirossian, J.; Lincoff, A.; Liggett, P.; Iwamoto, T.; Jakobiec, F. Intravitreal longevity of three perfluorocarbon gases. Arch. Ophthalmol. 1980, 98, 1610–1611. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, P.; Luff, A.; Aylward, G. Results of primary retinal reattachment surgery: A prospective audit. Eye 1997, 11, 869–871. [Google Scholar] [CrossRef]
- Jin, Y.; Li, Y.; Song, S.; Ding, Y.; Dong, Y.; Lu, Y.; Liu, D.; Zhang, C. DNA supramolecular hydrogel as a biocompatible artificial vitreous substitute. Adv. Mater. Interfaces 2022, 9, 2101321. [Google Scholar] [CrossRef]
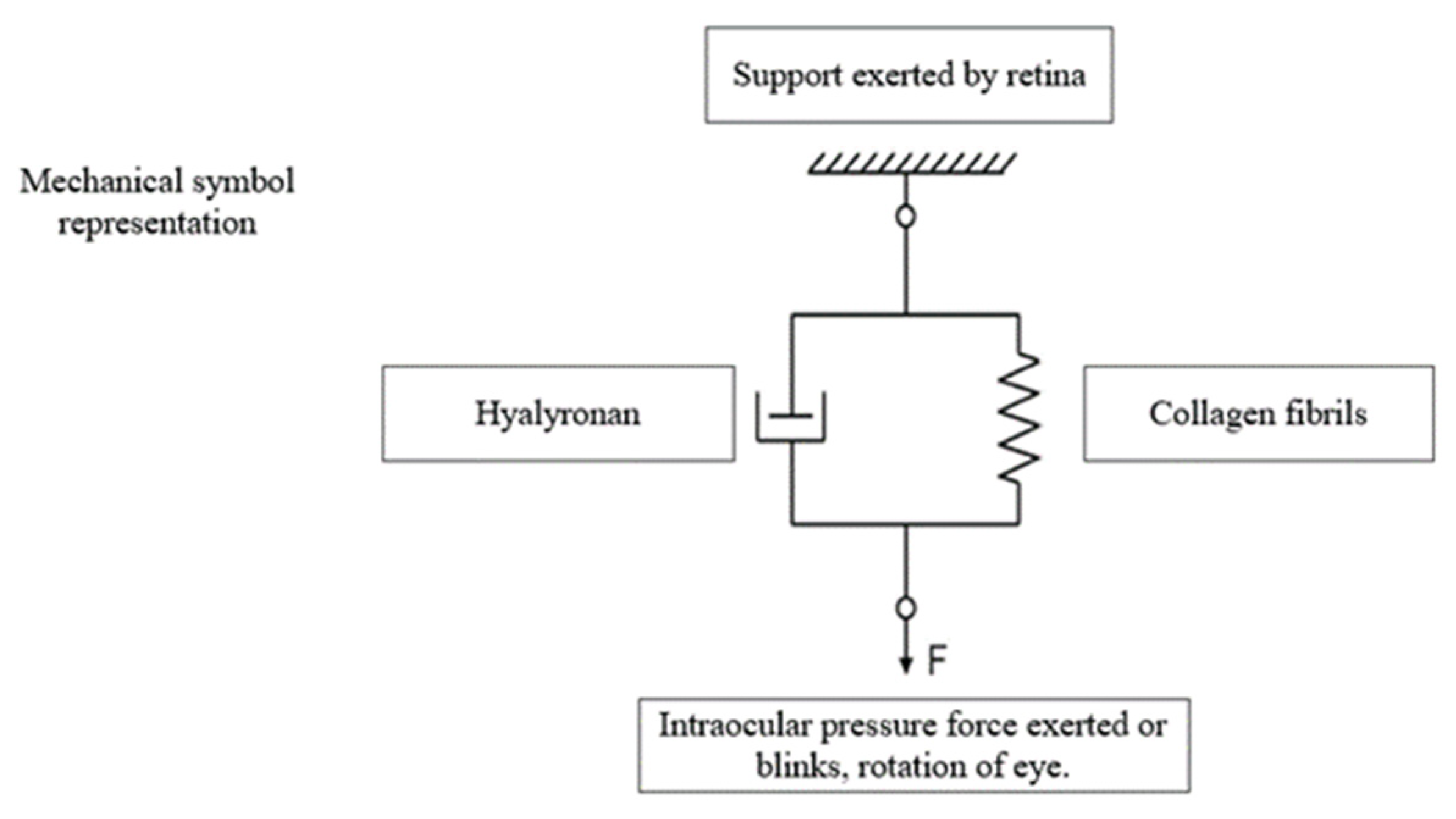
| Type | Polymer | Hydrogel Properties | Advantages | Limitations | Ref. |
|---|---|---|---|---|---|
| Natural | Hyaluronic Acid | Viscoelastic, biodegradable, transparent |
|
| [58] |
| Collagen | Fibrillar structure, biocompatible |
|
| [59] | |
| Gelatin | Thermo-sensitive, porous |
|
| [60] | |
| Alginate | Ionically crosslinked, gel-like |
|
| [43] | |
| Chitosan | Cationic, bioadhesive |
|
| [61] | |
| Synthetic | PEG | Tunable, hydrophilic, crosslinkable |
|
| [62] |
| PVA | Elastic, transparent, water-retentive |
|
| [63] | |
| Poly(acrylamide) (PAAm) | Soft, transparent gel |
|
| [50,64] | |
| Pluronic (Poloxamer) | Thermo-responsive triblock copolymer |
|
| [65] | |
| Poly(N-isopropylacrylamide) (PNIPAM) | Thermo-responsive hydrogel |
|
| [66,67] |
| Property | Target Range | Description | Functional Importance | Ref. |
|---|---|---|---|---|
| Viscosity | 300–2000 centipoise (cP) | Resistance to flow or thickness of the fluid | Allows for injection through small-bore needles while maintaining structure | [9] |
| Density | 1.005–1.008 g/cm3 | Mass per unit volume, matched to native vitreous | Prevents buoyancy-related displacement within the vitreous cavity | [102] |
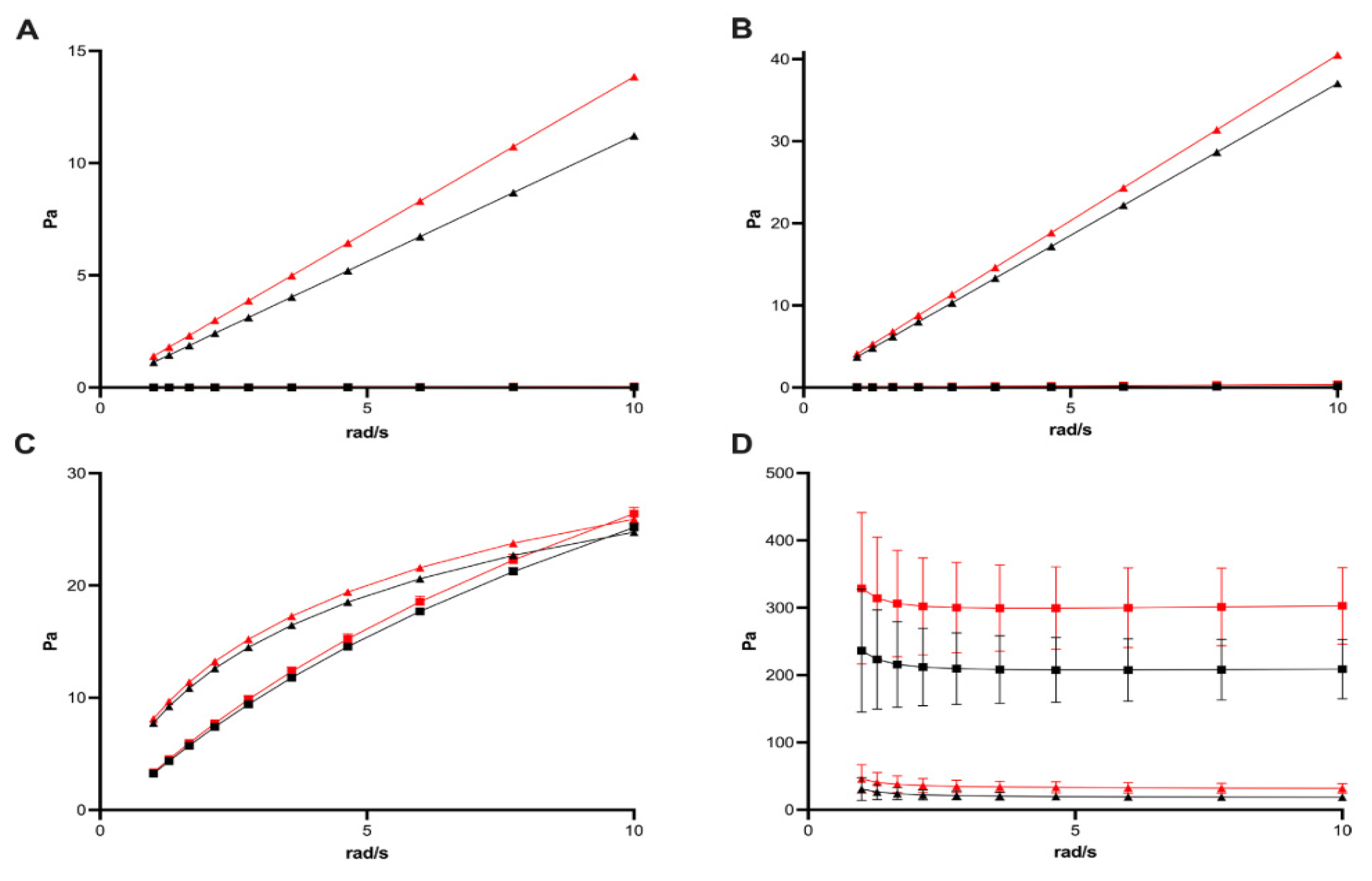
| Viscoelasticity (Storage Modulus, G′) | ~5–10 Pa | Gel-like elasticity and stress-response under deformation | Provides mechanical support and mimics the cushioning effect of natural vitreous | [103] |
| Buoyancy/Surface Tension | Sufficient interfacial tension | Floatation capability and sealing ability against retinal breaks | Facilitates tamponade of retinal breaks and stabilizes retina post-surgery | [17] |
| Injectability | Through 22–33-gauge needles | Compatibility with fine surgical needles | Enables minimally invasive delivery into the vitreous cavity | [34] |
| Shear-Thinning Behavior | Viscosity decreases under shear, recovers post-injection | Reduction in viscosity during injection with rapid gel recovery afterward | Facilitates easy injection while regaining mechanical strength in situ | [43] |
| Product | Type | Clinical Use | Comments | Ref. |
|---|---|---|---|---|
| Air | Gas tamponade | Widely used for short-term tamponade | Oldest tamponade: short duration limits long-term use | [114,115] |
| Sulfur Hexafluoride (SF6) | Gas tamponade | Common primary tamponade for retinal detachment | Safer than C3F8 for IOP spikes; faster absorption | [116,119] |
| Perfluoropropane (C3F8) | Gas tamponade | Used for complex/recurrent retinal detachments | Longer tamponade effect but higher risk of IOP elevation and cataracts | [117,118,119] |
| Silicone Oil | Liquid tamponade | Long-term tamponade for complicated detachments | FDA-approved; associated with cataracts, glaucoma, keratopathy, and visual loss | [120,121,122,123,124,125,126] |
| Perfluorocarbon Liquids (PFCLs) | Liquid tamponade (intraoperative) | Short-term intraoperative tamponade | Used temporarily during surgery; must be removed to avoid toxicity | [130,131,132,133,134,135] |
| Vitargus® (ABV-1701 Hydrogel) | Hydrogel-based substitute (investigational) | Under clinical trial for temporary vitreous substitution | Emerging hydrogel alternative with promising early clinical results | [14,136,137] |
| Remaining Issues | Description | Potential Solution |
|---|---|---|
| Long-term stability | Hydrogels tend to degrade, shrink, or lose transparency over time inside the ocular environment | Designing interpenetrating polymer networks (IPNs); incorporation of more stable synthetic polymers (e.g., PEG and PVA); optimizing crosslinking density |
| Biocompatibility and tissue integration | Risk of inflammation, fibrosis, or poor adhesion to surrounding ocular tissues | Surface functionalization with bioactive peptides; incorporation of natural polymers (e.g., HA and collagen); anti-inflammatory modifications |
| Mechanical mismatch | Inability to fully replicate native vitreous viscoelasticity and support for retina | Fine-tuning crosslinking methods; using dual-crosslinking systems; tailoring polymer molecular weight and concentration |
| Controlled biodegradability | Premature or uncontrolled degradation affects long-term performance | Development of stimuli-responsive hydrogels; balancing enzymatic vs. hydrolytic degradability; hybrid natural-synthetic systems |
| Drug delivery limitations | Current hydrogels may have burst release or insufficient loading capacity | Incorporation of nanoparticles or liposomes; covalent drug–polymer conjugates; design of responsive hydrogels (pH, ROS, and enzymes) |
| Functionalization for tissue repair | Limited regenerative ability and lack of bioactivity | Incorporation of growth factors, stem-cell-supportive motifs, and ECM-mimetic peptides |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sojdeh, S.; Panjipour, A.; Bejandi, Z.B.; Salehi, M.; Yaghmour, A.; Arabpour, Z.; Djalilian, A.R.; Chan, R.V.P. Hydrogel-Based Vitreous Substitutes. Int. J. Mol. Sci. 2025, 26, 8406. https://doi.org/10.3390/ijms26178406
Sojdeh S, Panjipour A, Bejandi ZB, Salehi M, Yaghmour A, Arabpour Z, Djalilian AR, Chan RVP. Hydrogel-Based Vitreous Substitutes. International Journal of Molecular Sciences. 2025; 26(17):8406. https://doi.org/10.3390/ijms26178406
Chicago/Turabian StyleSojdeh, Soheil, Amirhosein Panjipour, Zahra Bibak Bejandi, Majid Salehi, Amal Yaghmour, Zohreh Arabpour, Ali R. Djalilian, and R. V. Paul Chan. 2025. "Hydrogel-Based Vitreous Substitutes" International Journal of Molecular Sciences 26, no. 17: 8406. https://doi.org/10.3390/ijms26178406
APA StyleSojdeh, S., Panjipour, A., Bejandi, Z. B., Salehi, M., Yaghmour, A., Arabpour, Z., Djalilian, A. R., & Chan, R. V. P. (2025). Hydrogel-Based Vitreous Substitutes. International Journal of Molecular Sciences, 26(17), 8406. https://doi.org/10.3390/ijms26178406







