Phytochemicals from Purwoceng (Pimpinella pruatjan) and Their Potential in Chronic Disease Prevention: Focus on Kidney Health
Abstract
1. Introduction
2. Results
2.1. Purwoceng Root Extract
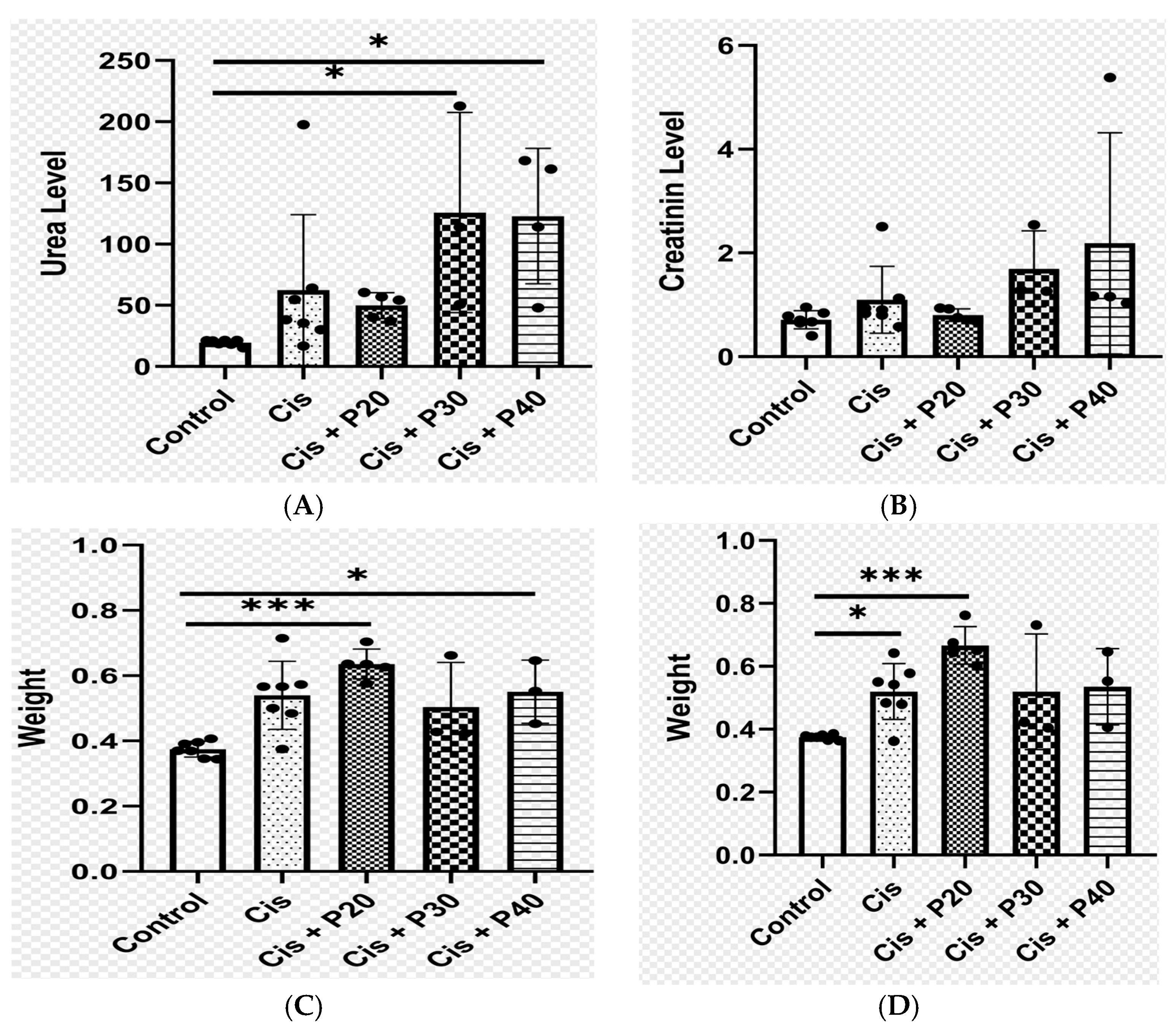
2.2. Baseline and Post-Treatment Characteristics of the Rats
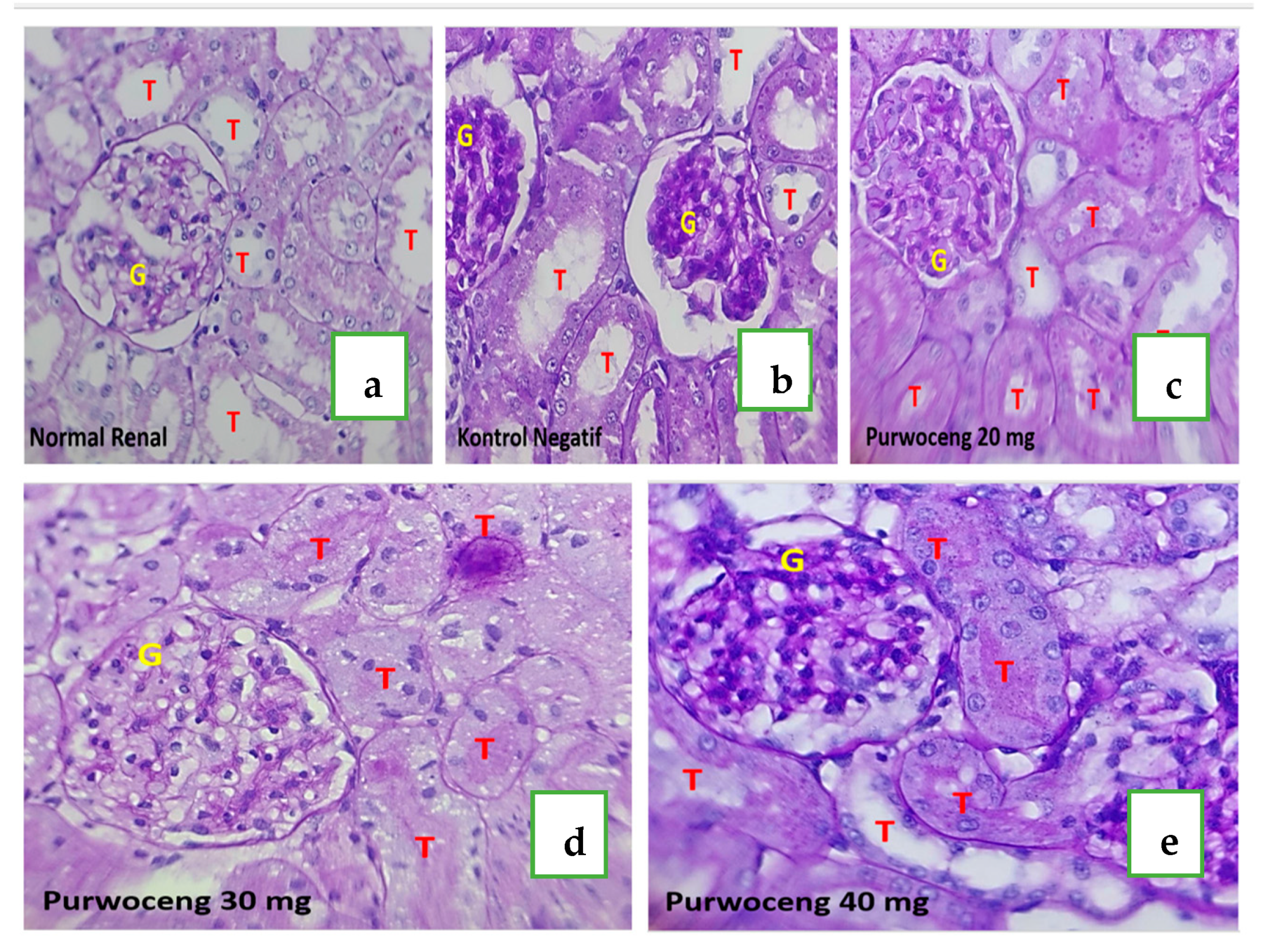
2.3. Histopathological Renal Appearance
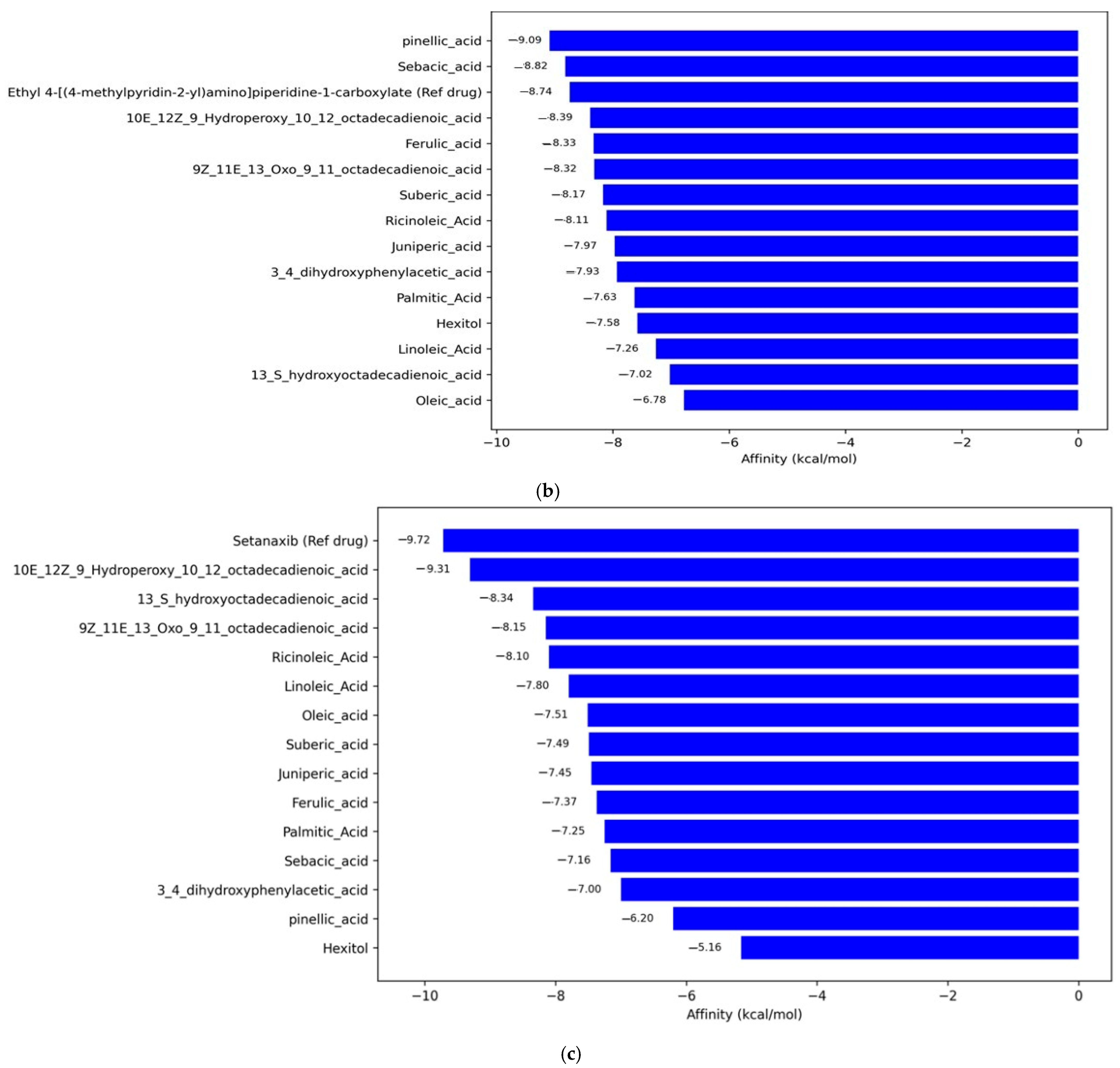
2.4. In Silico Prediction Target Radical Enzyme
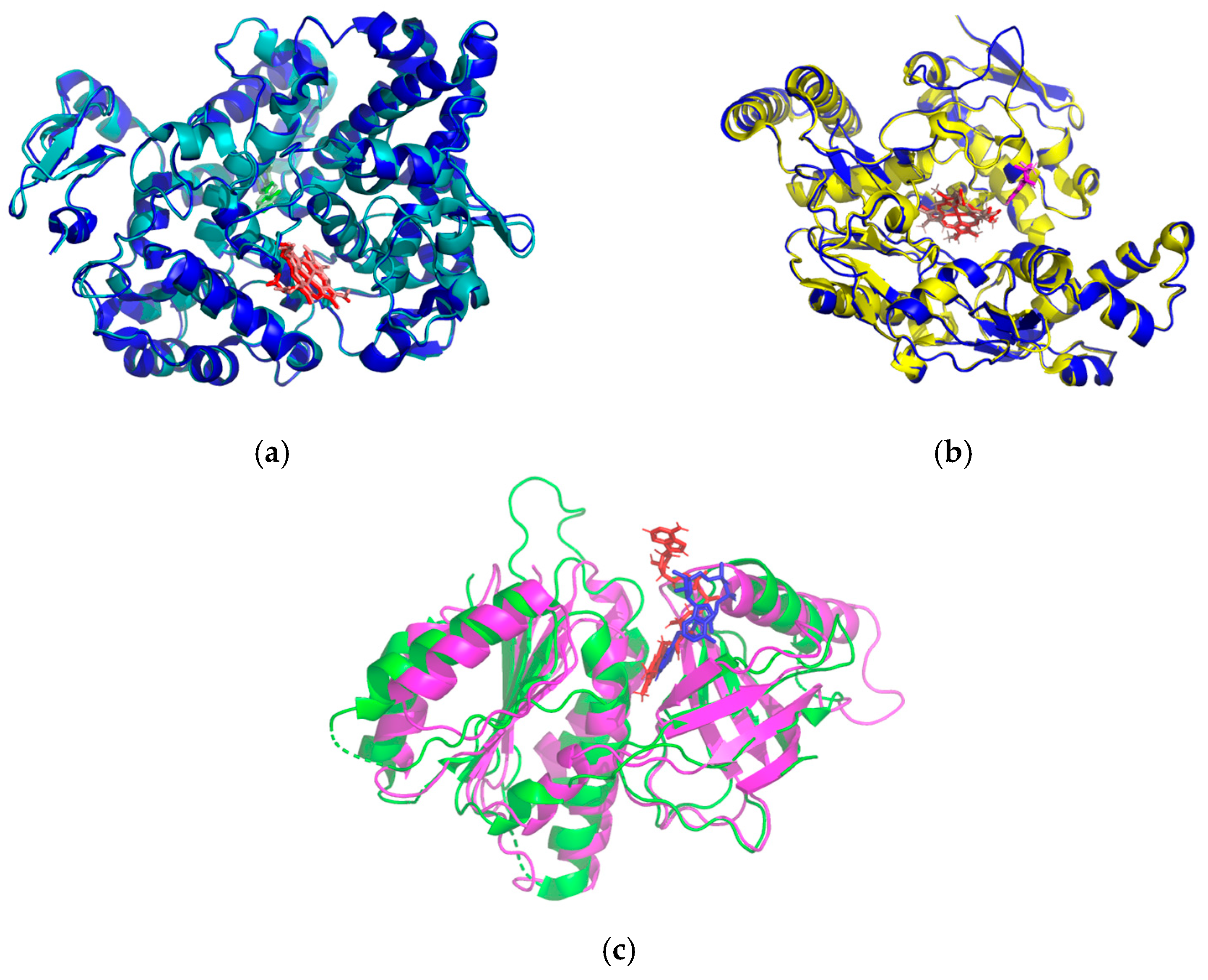
2.4.1. Structure Align
2.4.2. Docking Validation
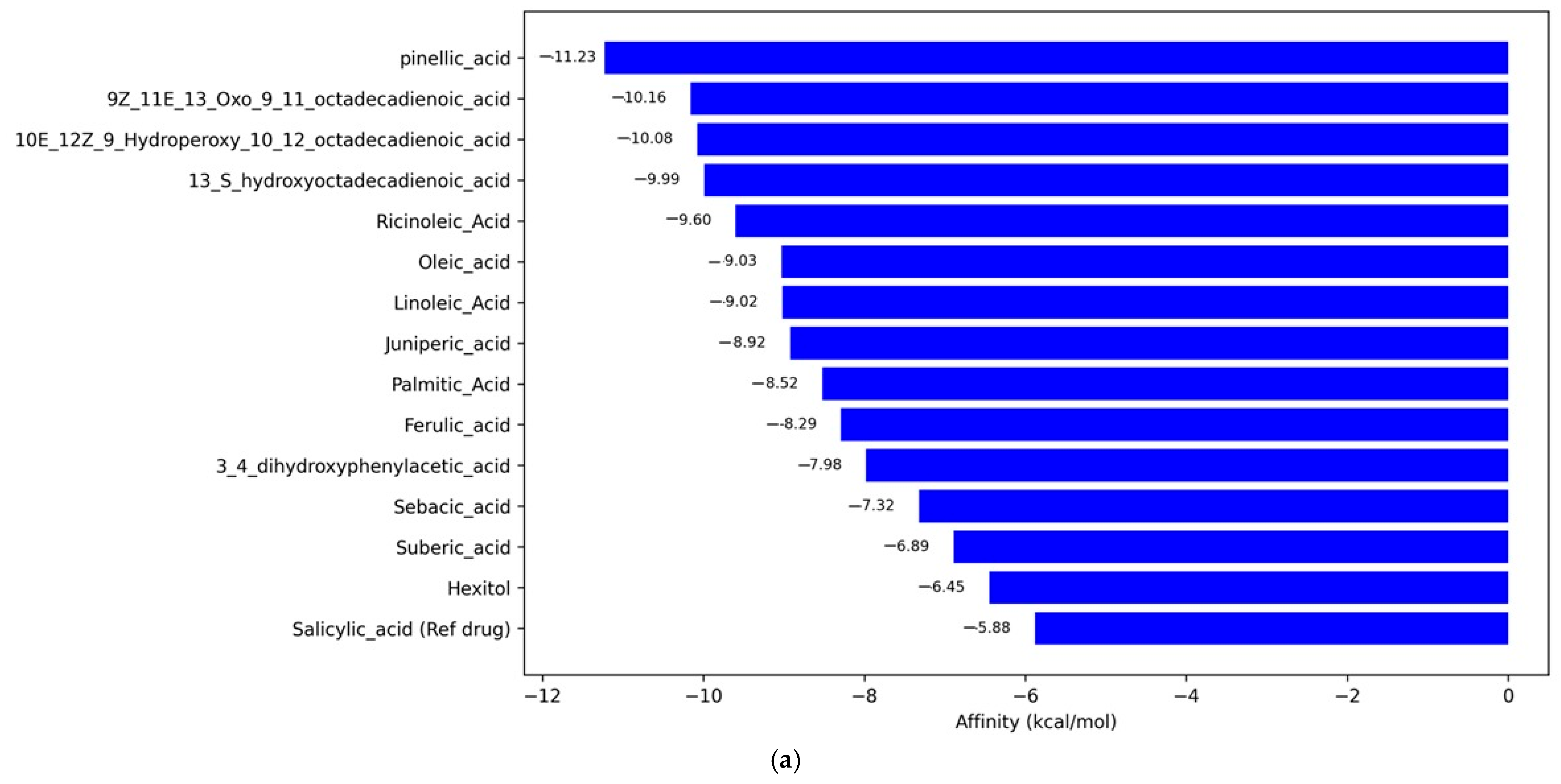
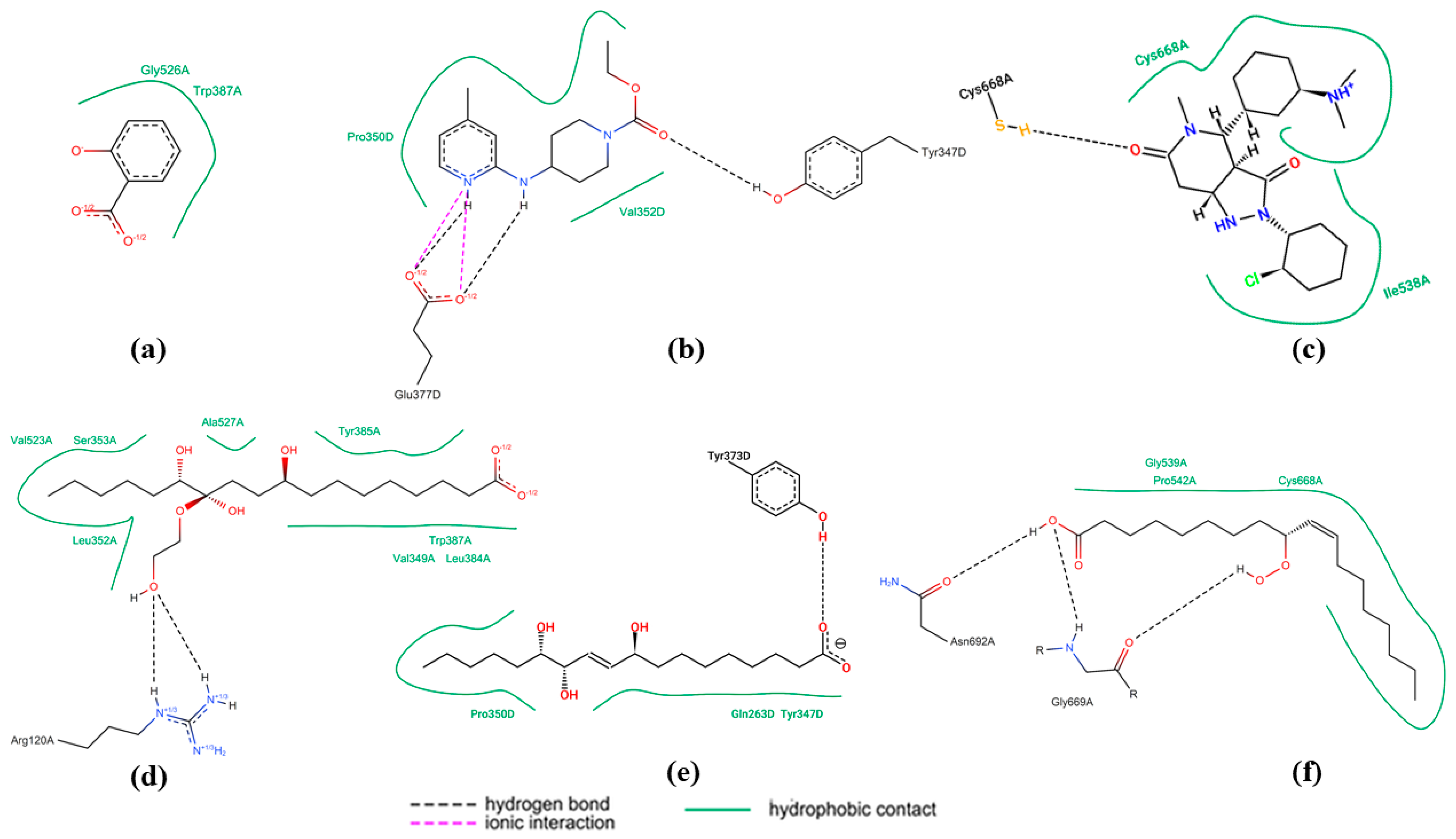
2.4.3. Molecular Docking
3. Discussion
4. Materials and Methods
4.1. Study Design and Setting
4.2. Sample Preparation and Extraction
4.3. Phytochemical Content
4.4. Antioxidant Capacity
4.5. Compound Identification in Purwoceng Root Extract
4.6. In Silico Prediction
4.7. Inclusion and Exclusion Criteria
4.8. Experimental Protocol
4.9. Data Collections
4.10. Data Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABTS | 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) |
| ANOVA | analysis of variance |
| BUN | blood urea nitrogen |
| CKD | chronic kidney disease |
| CPU | colony-forming unit |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| GAE | gallic acid equivalent |
| LC-MS | liquid chromatography–mass spectrometry |
| QE | quercetin equivalent |
| TE | Trolox equivalent |
| TPC | total plate count |
| UHPLC | ultra-high performance liquid chromatography |
References
- Shim, H.W.; Lee, W.-Y.; Ham, Y.-K.; Lim, S.D.; Hwang, S.-G.; Park, H.-J. Catechin Hydrate Prevents Cisplatin-Induced Spermatogonia GC-1 Spg Cellular Damage. J. Anim. Reprod. Biotechnol. 2024, 39, 145–152. [Google Scholar] [CrossRef]
- Syska, K.; Nurhayati, A.D. Ropiudin Characteristics and Antioxidant Activity of Dried Purwoceng (Pimpinella alpina Molk) as Functional Food to Increase Body Immune. J. Basic Sci. Technol. 2023, 12, 1–11. [Google Scholar]
- Koc, K.; Geyikoglu, F.; Cakmak, O.; Koca, A.; Kutlu, Z.; Aysin, F.; Yilmaz, A.; Aşkın, H. The Targets of β-Sitosterol as a Novel Therapeutic against Cardio-Renal Complications in Acute Renal Ischemia/Reperfusion Damage. Naunyn Schmiedebergs Arch. Pharmacol. 2020, 394, 469–479. [Google Scholar] [CrossRef]
- Nematbakhsh, M.; Ashrafi, F.; Pezeshki, Z.; Fatahi, Z.; Kianpoor, F.; Sanei, M.H.; Talebi, A. A Histopathological Study of Nephrotoxicity, Hepatoxicity or Testicular Toxicity: Which One Is the First Observation as Side Effect of Cisplatin-Induced Toxicity in Animal Model? J. Nephropathol. 2012, 1, 190. [Google Scholar] [CrossRef]
- Yao, X.; Panichpisal, K.; Kurtzman, N.; Nugent, K. Cisplatin Nephrotoxicity: A Review. Am. J. Med. Sci. 2007, 334, 115–124. [Google Scholar] [CrossRef]
- Norozi Sarkarabad, M.; Zare, S. Evaluation of the Effect of Taurine on Cisplatin-Induced Hepatic Injury and Oxidative Stress in Male Rats. Physiol. Pharmacol. 2011, 15, 427–434. [Google Scholar]
- Chirino, Y.I.; Pedraza-Chaverri, J. Role of Oxidative and Nitrosative Stress in Cisplatin-Induced Nephrotoxicity. Exp. Toxicol. Pathol. 2009, 61, 223–242. [Google Scholar] [CrossRef]
- Miller, R.P.; Tadagavadi, R.K.; Ramesh, G.; Reeves, W.B. Mechanisms of Cisplatin Nephrotoxicity. Toxins 2010, 2, 2490–2518. [Google Scholar] [CrossRef]
- Lieberthal, W.; Triaca, V.; Levine, J. Mechanisms of Death Induced by Cisplatin in Proximal Tubular Epithelial Cells: Apoptosis vs. Necrosis. Am. J. Physiol. 1996, 270, F700–F708. [Google Scholar] [CrossRef]
- Okuda, M.; Masaki, K.; Fukatsu, S.; Hashimoto, Y.; Inui, K.I. Role of Apoptosis in Cisplatin-Induced Toxicity in the Renal Epithelial Cell Line LLC-PK1: Implication of the Functions of Apical Membranes. Biochem. Pharmacol. 2000, 59, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Jeong, H.; Choo, Y.H.; Kim, M.; Ha, E.J.; Oh, J.; Shim, Y.; Kim, S.B.; Jung, H.G.; Park, S.H.; et al. Optimizing Mannitol Use in Managing Increased Intracranial Pressure: A Comprehensive Review of Recent Research and Clinical Experiences. Korean J. Neurotrauma 2023, 19, 162. [Google Scholar] [CrossRef]
- Lu, Y.; Wahl, L.M. Oxidative Stress Augments the Production of Matrix Metalloproteinase-1, Cyclooxygenase-2, and Prostaglandin E2 through Enhancement of NF-Kappa B Activity in Lipopolysaccharide-Activated Human Primary Monocytes. J. Immunol. 2005, 175, 5423–5429. [Google Scholar] [CrossRef]
- Murakami, A.; Ohigashi, H. Targeting NOX, INOS and COX2 in Inflammatory Cells: Chemoprevention Using Food Phytochemicals. Int. J. Cancer 2007, 121, 2357–2363. [Google Scholar] [CrossRef]
- Morris, G.; Gevezova, M.; Sarafian, V.; Maes, M. Redox Regulation of the Immune Response. Cell Mol. Immunol. 2022, 19, 1079–1101. [Google Scholar] [CrossRef]
- Bhol, N.K.; Bhanjadeo, M.M.; Singh, A.K.; Dash, U.C.; Ojha, R.R.; Majhi, S.; Duttaroy, A.K.; Jena, A.B. The Interplay between Cytokines, Inflammation, and Antioxidants: Mechanistic Insights and Therapeutic Potentials of Various Antioxidants and Anti-Cytokine Compounds. Biomed. Pharmacother. 2024, 178, 117177. [Google Scholar] [CrossRef] [PubMed]
- Peraturan BPOM No. 32 Tahun 2019. Available online: https://peraturan.bpk.go.id/Details/285067/peraturan-bpom-no-32-tahun-2019 (accessed on 7 April 2025).
- WHO Guideline for Assessing Quality of Herbal Medicines with Reference to Contaminants and Residues, 3rd ed.; Recommendations; World Health Organization: Geneva, Switzerland, 2007; pp. 130–143.
- Sumarni, E.; Batubara, I.; Suhardiyanto, H.; Widodo, S.; Solahudin, M.; Rohaeti, E.; Laumal, F. Application of Hydroponic Technology to the Yield and Quality of Purwoceng “Viagra from Asia”. Inmateh Agric. Eng. 2023, 69, 46–54. [Google Scholar] [CrossRef]
- Batubara, I.; Asoka, S.F.; Sumarni, E.; Suhardiyanto, H.; Solahudin, M.; Widodo, S.; Supriyanto, S.; Rohaeti, E.; Wahyu, Y.; Laumal, F.; et al. Secondary Metabolites and Antioxidant Activity of Purwoceng (Pimpinella pruatjan) Root Extracts from Various Hydroponic Planting Techniques. J. Tek. Pertan. Lampung 2023, 12, 641. [Google Scholar] [CrossRef]
- Wu, J.; Xiong, W.; Liu, W.; Wu, J.; Ruan, R.; Fu, P.; Wang, Y.; Liu, Y.; Leng, X.; Li, P.; et al. The Effects of Dietary N-3 Highly Unsaturated Fatty Acids on Growth, Antioxidant Capacity, Immunity, and Oxylipin Profiles in Acipenser Dabryanus. Antioxidants 2024, 13, 421. [Google Scholar] [CrossRef]
- Al-Attar, A.M.; Alrobai, A.A.; Almalki, D.A. Protective Effect of Olive and Juniper Leaves Extracts on Nephrotoxicity Induced by Thioacetamide in Male Mice. Saudi. J. Biol. Sci. 2017, 24, 15–22. [Google Scholar] [CrossRef]
- Mascolo, N.; Autore, G.; Capasso, F.; Menghini, A.; Fasulo, M.P. Biological Screening of Italian Medicinal Plants for Anti-inflammatory Activity. Phytother. Res. 1987, 1, 28–31. [Google Scholar] [CrossRef]
- Cselőtey, A.; Baglyas, M.; Király, N.; Ott, P.G.; Glavnik, V.; Vovk, I.; Móricz, Á.M. Bioassay-Guided Isolation and Identification of Antibacterial Compounds from Invasive Tree of Heaven Stem and Trunk Bark. Molecules 2024, 29, 5846. [Google Scholar] [CrossRef]
- Sun, W.; Shahrajabian, M.H.; Cheng, Q. Anise (Pimpinella anisum L.), a Dominant Spice and Traditional Medicinal Herb for Both Food and Medicinal Purposes. Cogent Biol. 2019, 5, 1673688. [Google Scholar] [CrossRef]
- Jana, S.; Mitra, P.; Dutta, A.; Khatun, A.; Kumar Das, T.; Pradhan, S.; Kumar Nandi, D.; Roy, S. Early Diagnostic Biomarkers for Acute Kidney Injury Using Cisplatin-Induced Nephrotoxicity in Rat Model. Curr. Res. Toxicol. 2023, 5, 100135. [Google Scholar] [CrossRef] [PubMed]
- Pezeshki, Z.; Khosravi, A.; Nekuei, M.; Khoshnood, S.; Zandi, E.; Eslamian, M.; Talebi, A.; Emami, S.N.E.D.; Nematbakhsh, M. Time Course of Cisplatin-Induced Nephrotoxicity and Hepatotoxicity. J. Nephropathol. 2017, 6, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Perše, M.; Večerić-Haler, Ž. Cisplatin-Induced Rodent Model of Kidney Injury: Characteristics and Challenges. Biomed. Res. Int. 2018, 2018, 1462802. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Huang, Z.; Cao, X.; Chen, X.; Zou, T.; You, J. Plant-Derived Polyphenols as Nrf2 Activators to Counteract Oxidative Stress and Intestinal Toxicity Induced by Deoxynivalenol in Swine: An Emerging Research Direction. Antioxidants 2022, 11, 2379. [Google Scholar] [CrossRef]
- Schaich, K.M.; Tian, X.; Xie, J. Hurdles and Pitfalls in Measuring Antioxidant Efficacy: A Critical Evaluation of ABTS, DPPH, and ORAC Assays. J. Funct. Foods 2015, 14, 111–125. [Google Scholar] [CrossRef]
- Garcia, E.J.; Cadorin Oldoni, T.L.; de Alencar, S.M.; Reis, A.; Loguercio, A.D.; Miranda Grande, R.H. Antioxidant Activity by DPPH Assay of Potential Solutions to Be Applied on Bleached Teeth. Braz. Dent. J. 2012, 23, 22–27. [Google Scholar] [CrossRef]
- Coniglio, S.; Shumskaya, M.; Vassiliou, E. Unsaturated Fatty Acids and Their Immunomodulatory Properties. Biology 2023, 12, 279. [Google Scholar] [CrossRef]
- Palomer, X.; Pizarro-Delgado, J.; Barroso, E.; Vázquez-Carrera, M. Palmitic and Oleic Acid: The Yin and Yang of Fatty Acids in Type 2 Diabetes Mellitus. Trends Endocrinol. Metab. 2018, 29, 178–190. [Google Scholar] [CrossRef]
- Pan, F.G.; Yang, E.Q.; Xu, J.Y.; Lu, Y.F.; Yang, C.X.; Zhang, Y.D.; Li, S.; Liu, B.Q. Review on Preparation Methods, Mechanisms and Applications for Antioxidant Peptides in Oil. Grasas Y Aceites 2022, 73, e478. [Google Scholar] [CrossRef]
- Vieira, C.; Evangelista, S.; Cirillo, R.; Lippi, A.; Maggi, C.A.; Manzini, S. Effect of Ricinoleic Acid in Acute and Subchronic Experimental Models of Inflammation. Mediat. Inflamm. 2000, 9, 223–228. [Google Scholar] [CrossRef]
- Setiawan, S.; Rahardjo, M. Respon Pemupukan Terhadap Pertumbuhan, Produksi Dan Mutu Herba Meniran (Phyllantus niruri). Bul. Penelit. Tanam. Rempah Dan Obat 2016, 26, 25. [Google Scholar] [CrossRef]
- Batubara, I.; Asoka, S.F.; Sumarni, E.; Suhardiyanto, H.; Solahudin, M.; Widodo, S.; Supriyanto; Rohaeti, E.; Yudiwanti; Laumal, F.; et al. The Effect of Different Hydroponic Types and Nutrient Concentrations on the Chemical Composition and Antioxidant Capacity of Purwoceng (Pimpinella pruatjan) Extract. Hayati J. Biosci. 2023, 30, 985–994. [Google Scholar] [CrossRef]
- Batubara, I.; Komariah, K.; Sandrawati, A.; Nurcholis, W. Genotype Selection for Phytochemical Content and Pharmacological Activities in Ethanol Extracts of Fifteen Types of Orthosiphon aristatus (Blume) Miq. Leaves Using Chemometric Analysis. Sci. Rep. 2020, 10, 20945. [Google Scholar] [CrossRef]
- Prayogo, Y.H.; Batubara, I.; Wahyudi, S.T.; Sari, R.K.; Syafii, W. Metabolite Profile, Antiproliferative, and Antioxidant Activity of Acacia auriculiformis and Acacia crassicarpa Extracts. J. Saudi Chem. Soc. 2023, 27, 101742. [Google Scholar] [CrossRef]
- MzCloud—Advanced Mass Spectral Database. Available online: https://www.mzcloud.org/ (accessed on 18 August 2025).
- ChemSpider: Search and Share Chemistry—Homepage. Available online: https://www.chemspider.com/ (accessed on 18 August 2025).
- Santos-Martins, D.; Solis-Vasquez, L.; Tillack, A.F.; Sanner, M.F.; Koch, A.; Forli, S. Accelerating AutoDock4 with GPUs and Gradient-Based Local Search. J. Chem. Theory Comput. 2021, 17, 1060–1073. [Google Scholar] [CrossRef] [PubMed]
- Arjadi, F.; Kurniawan, D.W.; Wibowo, Y.; Siswandari, W.; Rujito, L. No Acute Toxicity Tests of Purwoceng (Pimpinella pruatjan Molk.) Ethanolic Extract on Male Albino Rat by Determined Hepatorenal Function Test and Histopathology. Molekul 2019, 14, 117–125. [Google Scholar] [CrossRef]
- Experimental Design: Theory and Application—W. T. Federer—Google Books. Available online: https://books.google.co.id/books/about/Experimental_Design.html?id=IqfCugAACAAJ&redir_esc=y (accessed on 7 April 2025).
- Arfian, N.; Budiharjo, S.; Wibisono, D.P.; Setyaningsih, W.A.W.; Romi, M.M.; Saputri, R.L.A.A.-N.W.; Rofiah, E.K.; Rahmanti, T.; Agustin, M.; Sari, D.C.R. Vitamin D Ameliorates Kidney Ischemia Reperfusion Injury via Reduction of Inflammation and Myofibroblast Expansion. Kobe J. Med. Sci. 2020, 65, E138. [Google Scholar]

| Safety Parameter | Standards Permitted by | Result | |||||
|---|---|---|---|---|---|---|---|
| BPOM No. 13 of 2019 | WHO 2007 | ||||||
| Malaysia | Canada | Singapore | Thailand | Recommendation | |||
| Heavy metal content safety of purwoceng | |||||||
| Cd Maximum (mg/Kg) | 0.3 | - | 0.3 | - | 0.3 | 0.3 | <detection limit (LOD 0.03 mg/kg) |
| Pb Maximum (mg/Kg) | 10 | 10 | 10 | 20 | 10 | 10 | <detection limit (LOD 0.033 mg/kg) |
| As Maximum (mg/Kg) | 5 | 5 | 5 | 5 | 4 | - | <detection limit (LOD 0.008 mg/kg) |
| Safety Parameter | Standards Permitted by | Result | |
|---|---|---|---|
| BPOM No. 13 of 2019 | WHO 2007 | ||
| Recommendation | |||
| Total plate count (TPC) maximum (CFU/g) | 5 × 107 | 107 | Negative |
| Coliform maximum (CFU/g) | 103 | 102 | Negative |
| mold/yeast maximum (CFU/g) | 5 × 105 | 104 | Negative |
| Compound | Content | Method of Determination |
|---|---|---|
| Total phenolic content (% gallic acid equivalent/extract) | 2.73 | Semi-qualitative using a spectrophotometer UV-Vis |
| Total flavonoid content (% quercetin equivalent/extract) | 0.75 | Semi-qualitative using a spectrophotometer UV-Vis |
| (E)-Ferulic acid (%) | 1.26 | LC-HR MS |
| 3,4-dihydroxyphenylacetic acid (%) | 4.70 | LC-HR MS |
| (−)-pinellic acid (%) | 3.32 | LC-HR MS |
| 13S-hydroxyoctadecadienoic acid (%) | 2.97 | LC HR MS |
| Juniperic acid (%) | 7.28 | LC HR MS |
| Linoleic acid (%) | 1.99 | LC-HR MS |
| Sample | IC50 (mg/L) | |
|---|---|---|
| DPPH | ABTS | |
| Purwoceng extract | 376.88 | 1269.77 |
| Vitamin C | 4.70 | 36.90 |
| Enzyme | Organism | Accession | Query Cover (%) | E-Value | Per. Ident (%) |
|---|---|---|---|---|---|
| NOX5 | Cylindrospermum stagnale | K9WT99 | 95 | 7 × 10−49 | 34.86 |
| Mus musculus | AAI72138.1 | ||||
| iNOS | Homo sapiens | P35228 | 100 | 0 | 87.26 |
| Mus musculus | AAC52356.1 | ||||
| COX2 | Homo sapiens | P35354 | 100 | 0 | 88.22 |
| Mus musculus | NP_035328.2 |
| Enzyme | PDB_ID | Organism | Superpose (Å) |
|---|---|---|---|
| NOX5 | 8CAO | Cylindrospermum stagnale | 2.391 |
| 6WXR | Mus musculus | ||
| iNOS | 3E7G | Homo sapiens | 0.385 |
| 4UX6 | Mus musculus | ||
| COX2 | 5F1A | Homo sapiens | 0.39 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dewi, T.U.; Rukmi, K.W.; Mahendra, F.R.; Munawaroh, F.; Ryadi, Y.; Widi, L.N.; Nurdin, N.M.; Batubara, I. Phytochemicals from Purwoceng (Pimpinella pruatjan) and Their Potential in Chronic Disease Prevention: Focus on Kidney Health. Int. J. Mol. Sci. 2025, 26, 8404. https://doi.org/10.3390/ijms26178404
Dewi TU, Rukmi KW, Mahendra FR, Munawaroh F, Ryadi Y, Widi LN, Nurdin NM, Batubara I. Phytochemicals from Purwoceng (Pimpinella pruatjan) and Their Potential in Chronic Disease Prevention: Focus on Kidney Health. International Journal of Molecular Sciences. 2025; 26(17):8404. https://doi.org/10.3390/ijms26178404
Chicago/Turabian StyleDewi, Trisni U., Kartika W. Rukmi, Fachrur R. Mahendra, Fauziyatul Munawaroh, Yusuf Ryadi, Leliana N. Widi, Naufal M. Nurdin, and Irmanida Batubara. 2025. "Phytochemicals from Purwoceng (Pimpinella pruatjan) and Their Potential in Chronic Disease Prevention: Focus on Kidney Health" International Journal of Molecular Sciences 26, no. 17: 8404. https://doi.org/10.3390/ijms26178404
APA StyleDewi, T. U., Rukmi, K. W., Mahendra, F. R., Munawaroh, F., Ryadi, Y., Widi, L. N., Nurdin, N. M., & Batubara, I. (2025). Phytochemicals from Purwoceng (Pimpinella pruatjan) and Their Potential in Chronic Disease Prevention: Focus on Kidney Health. International Journal of Molecular Sciences, 26(17), 8404. https://doi.org/10.3390/ijms26178404








