Beyond Joints: Neuropsychiatric Benefits of TNF-α and IL-6 Inhibitors in Rheumatoid Arthritis—Narrative Review
Abstract
1. Introduction
2. Methods
3. Results
3.1. Assessment Tools Used to Evaluate Neuropsychiatric Outcomes
3.2. TNF-α Inhibitors
3.3. IL-6/IL-6R Inhibitors
4. Discussion
4.1. TNF-α Inhibitors and Psychiatric Outcomes
4.2. IL-6/IL-6R Inhibitors and Psychiatric Outcomes
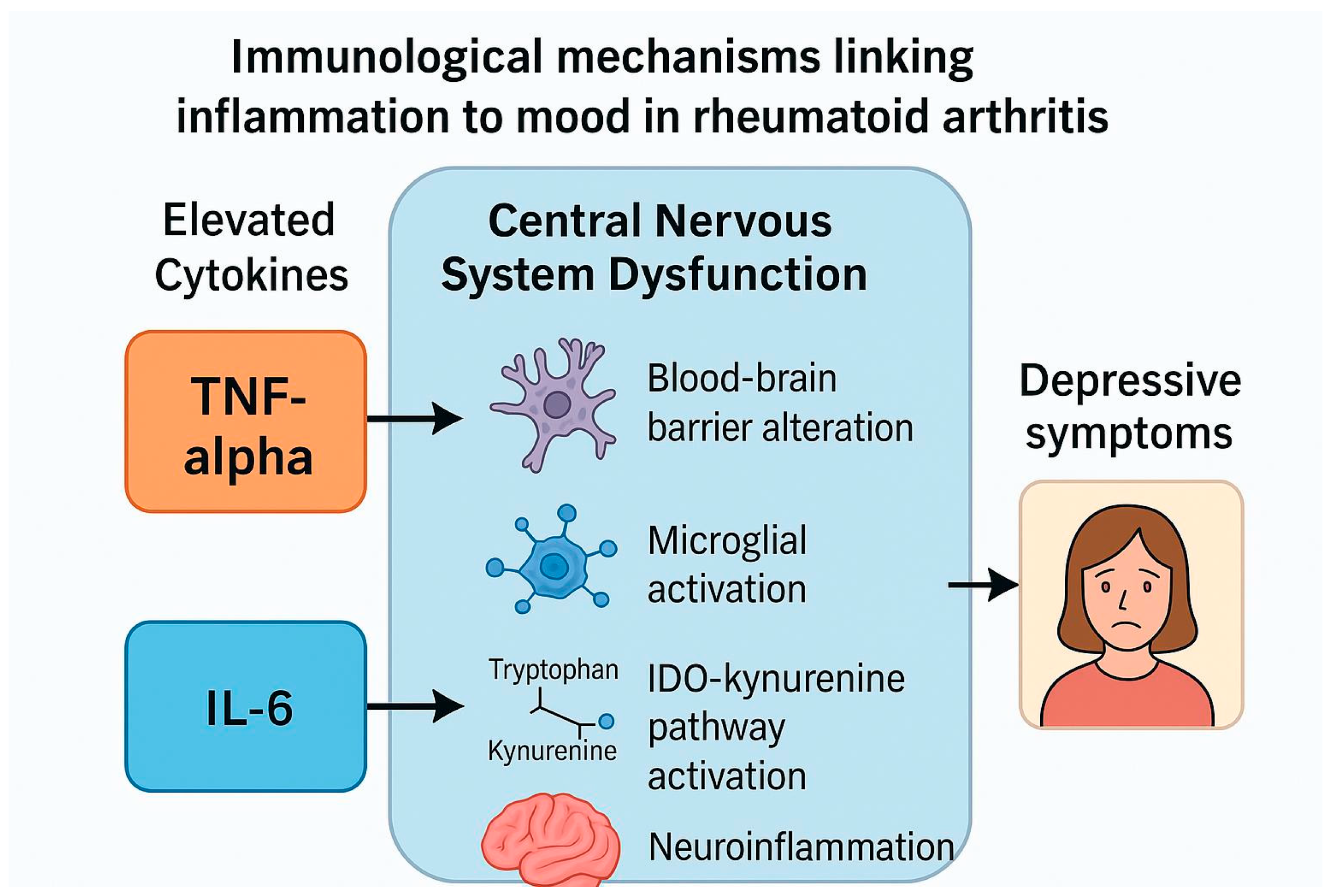
4.3. Proposed Mechanisms Linking Biological Therapies to Psychiatric Outcomes
4.3.1. Blood–Brain Barrier (BBB) Dysfunction
4.3.2. Microglial Activation and Neuroinflammation
4.3.3. Hypothalamic–Pituitary–Adrenal (HPA) Axis Dysregulation
4.3.4. Kynurenine Pathway Activation
4.3.5. Direct Actions of IL-6
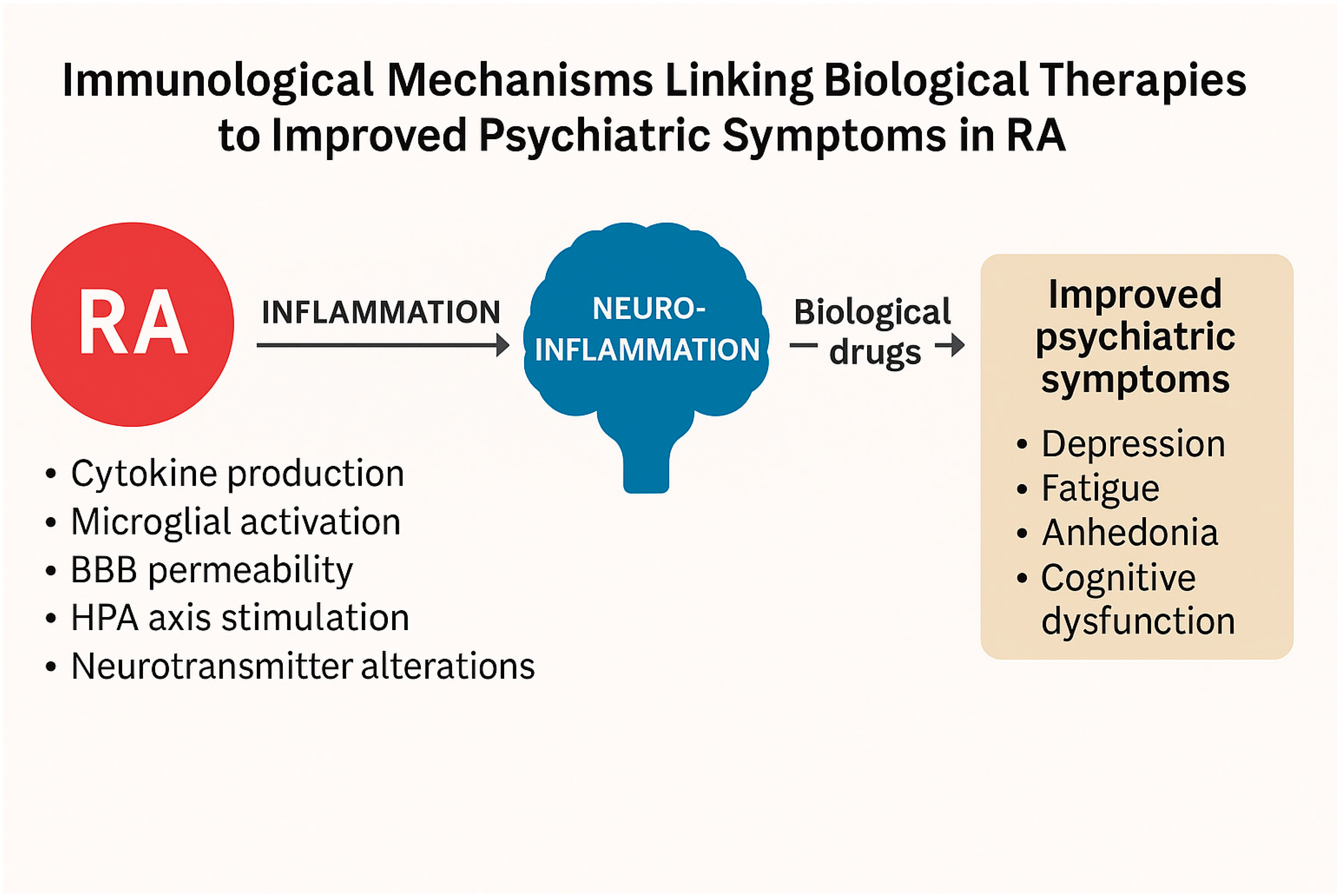
5. Role of Biological Therapies
5.1. Limitations
5.2. Conclusions
5.3. Clinical Implications
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AA | Alopecia Areata |
| ADA/ADL | Adalimumab |
| AE | Adverse Events |
| AM | Amplitude Modulation |
| AS | Ankylosing Spondylitis |
| BDI | Beck Depression Inventory |
| BBB | Blood–Brain Barrier |
| CD | Crohn’s Disease |
| CNS | Central Nervous System |
| CRP | C-Reactive Protein |
| CT | Computed Tomography |
| CZP | Certolizumab Pegol |
| DAS28 | Disease Activity Score 28 |
| ETA | Etanercept |
| EQ-5D | EuroQol 5 Dimensions Questionnaire |
| FACIT-F | Functional Assessment of Chronic Illness Therapy-Fatigue |
| FDA | Food and Drug Administration |
| GLM | Golimumab |
| HAQ | Health Assessment Questionnaire |
| HADS | Hospital Anxiety and Depression Scale |
| HPA | Hypothalamic–Pituitary–Adrenal |
| IBD | Inflammatory Bowel Disease |
| IDO | Indoleamine 2,3-Dioxygenase |
| IgG | Immunoglobulin G |
| IL | Interleukin |
| IL-6 | Interleukin-6 |
| IL-6R | Interleukin-6 Receptor |
| mAb | Monoclonal Antibody |
| MRI | Magnetic Resonance Imaging |
| MTX | Methotrexate |
| PCS | Physical Component Summary |
| PHQ-9 | Patient Health Questionnaire-9 |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| PROs | Patient-Reported Outcomes |
| QoL | Quality of Life |
| RA | Rheumatoid Arthritis |
| SARI | Sarilumab |
| SF-36 | Short Form Health Survey 36 |
| SRK | Sirukumab |
| TCZ | Tocilizumab |
| TNF | Tumor Necrosis Factor |
| TNF-α | Tumor Necrosis Factor-alpha |
| UC | Ulcerative Colitis |
| VAS | Visual Analog Scale |
| WOMAC | Western Ontario and McMaster Universities Osteoarthritis Index |
References
- Alamanos, Y.; Voulgari, P.V.; Drosos, A.A. Incidence and prevalence of rheumatoid arthritis, based on the 1987 American College of Rheumatology criteria: A systematic review. Semin. Arthritis Rheum. 2006, 36, 182–188. [Google Scholar] [CrossRef]
- Smolen, J.S.; Aletaha, D.; McInnes, I.B. Rheumatoid arthritis. Lancet 2016, 388, 2023–2038, Correction in: Lancet 2016, 388, 1984. [Google Scholar] [CrossRef] [PubMed]
- Meade, T.; Manolios, N.; Cumming, R.; Conaghan, P.G.; Katz, P. Cognitive Impairment in Rheumatoid Arthritis: A Systematic Review. Arthritis Care Res. 2018, 70, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Panjrattan, C.; Chauhan, V.S.; Nath, S.; Singh, K.R.; Banyal, N.; Bajwa, M.K. Depression among rheumatoid arthritis patients and barriers to seeking professional help: An observational study. Ind. Psychiatry J. 2023, 32, S136–S140. [Google Scholar] [CrossRef] [PubMed]
- Katz, P.; Pedro, S.; Michaud, K. Sleep Disorders Among Individuals With Rheumatoid Arthritis. Arthritis Care Res. 2023, 75, 1250–1260. [Google Scholar] [CrossRef] [PubMed]
- Maloley, P.M.; England, B.R.; Sayles, H.; Thiele, G.M.; Michaud, K.; Sokolove, J.; Cannon, G.W.; Reimold, A.M.; Kerr, G.S.; Baker, J.F.; et al. Post-traumatic stress disorder and serum cytokine and chemokine concentrations in patients with rheumatoid arthritis. Semin. Arthritis Rheum. 2019, 49, 229–235. [Google Scholar] [CrossRef]
- Albeltagy, E.S.; Elaziz, S.Y.A.; Abozaid, S.Y.; El Zomor, H.M.; Elhamed, S.S.A. Interleukin 6, interleukin 17, disease-related and contextual factor association with depression, and its severity in patients with rheumatoid arthritis. Clin. Rheumatol. 2021, 40, 895–904. [Google Scholar] [CrossRef]
- Liu, Y.; Ho, R.C.; Mak, A. The role of interleukin (IL)-17 in anxiety and depression of patients with rheumatoid arthritis. Int. J. Rheum. Dis. 2012, 15, 183–187. [Google Scholar] [CrossRef]
- El-Tantawy, A.M.; El-Sayed, A.E.; Kora, B.A.; Amin, R.T. Psychiatric morbidity associated with some cytokines (IL-1beta, IL-12, IL-18 and TNF-alpha) among rheumatoid arthritis patients. Egypt. J. Immunol. 2008, 15, 1–11. [Google Scholar]
- Li, Y.C.; Chou, Y.C.; Chen, H.C.; Lu, C.C.; Chang, D.M. Interleukin-6 and interleukin-17 are related to depression in patients with rheumatoid arthritis. Int. J. Rheum. Dis. 2019, 22, 980–985. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Desse, S.; Martinez, A.; Worthen, R.J.; Jope, R.S.; Beurel, E. TNFα disrupts blood brain barrier integrity to maintain prolonged depressive-like behavior in mice. Brain Behav. Immun. 2018, 69, 556–567. [Google Scholar] [CrossRef]
- Xu, D.; Xu, Y.; Gao, X.; Yan, M.; Zhang, C.; Wu, X.; Xia, Q.; Ge, J. Potential value of Interleukin-6 as a diagnostic biomarker in human MDD and the antidepressant effect of its receptor antagonist tocilizumab in lipopolysaccharide-challenged rats. Int. Immunopharmacol. 2023, 124, 110903. [Google Scholar] [CrossRef]
- Cathomas, F.; Fuertig, R.; Sigrist, H.; Hengerer, B.; Newman, G.N.; Hoop, V.; Bizzozzero, M.; Mueller, A.; Luippold, A.; Ceci, A.; et al. CD40-TNF activation in mice induces extended sickness behavior syndrome co-incident with but not dependent on activation of the kynurenine pathway. Brain Behav. Immun. 2015, 50, 125–140. [Google Scholar] [CrossRef]
- Brown, E.; Mc Veigh, C.J.; Santos, L.; Gogarty, M.; Müller, H.K.; Elfving, B.; Brayden, D.J.; Haase, J. TNFα-dependent anhedonia and upregulation of hippocampal serotonin transporter activity in a mouse model of collagen-inducedarthritis. Neuropharmacology 2018, 137, 211220. [Google Scholar] [CrossRef]
- Bingham, C.O., III; Black, S.; Shiff, N.J.; Xu, S.; Langholff, W.; Curtis, J.R. Response to Treatment with Intravenous Golimumab or Infliximab in Rheumatoid Arthritis Patients: PROMIS Results from the Real-World Observational Phase 4 AWARE Study. Rheumatol. Ther. 2023, 10, 659–678. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, S.C.; Tsai, P.H.; Kuo, C.F.; Cheng, T.T.; Lai, N.S.; Lin, J.C.; Lin, L.H.; Tsai, C.Y. Health-related quality of life improvement by adalimumab therapy in patients with rheumatoid arthritis in Taiwan: A nationwide prospective study. J. Chin. Med. Assoc. 2023, 86, 366–374. [Google Scholar] [CrossRef]
- Tiosano, S.; Yavne, Y.; Watad, A.; Langevitz, P.; Lidar, M.; Feld, J.; Tishler, M.; Aamar, S.; Elkayam, O.; Balbir-Gurman, A. The impact of tocilizumab on anxiety and depression in patients with rheumatoid arthritis. Eur. J. Clin. Investig. 2020, 50, e13268. [Google Scholar] [CrossRef]
- Strand, V.; Boklage, S.H.; Kimura, T.; Joly, F.; Boyapati, A.; Msihid, J. High levels of interleukin-6 in patients with rheumatoid arthritis are associated with greater improvements in health-related quality of life for sarilumab compared with adalimumab. Arthritis Res. Ther. 2020, 22, 250. [Google Scholar] [CrossRef] [PubMed]
- Edwards, J.C.; Leandro, M.J.; Cambridge, G.B. Lymphocyte depletion therapy with rituximab in rheumatoid arthritis. Rheum. Dis. Clin. N. Am. 2004, 30, 393–403. [Google Scholar] [CrossRef]
- Genovese, M.C.; Becker, J.C.; Schiff, M.; Luggen, M.; Sherrer, Y.; Kremer, J.; Birbara, C.; Box, J.; Natarajan, K.; Nuamah, I.; et al. Abatacept for rheumatoid arthritis refractory to tumor necrosis factor alpha inhibition. N. Engl. J. Med. 2005, 353, 1114–1123, Correction in: N. Engl. J. Med. 2005, 353, 2311. [Google Scholar] [CrossRef] [PubMed]
- Yirmiya, R.; Goshen, I. Immune modulation of learning, memory, neural plasticity and neurogenesis. Brain Behav. Immun. 2011, 25, 181–213. [Google Scholar] [CrossRef]
- Dantzer, R.; O’Connor, J.C.; Freund, G.G.; Johnson, R.W.; Kelley, K.W. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Miwa, Y.; Nishimi, A.; Nishimi, S.; Saito, M.; Tokunaga, T.; Yanai, R.; Takahashi, R.; Wakabayashi, K.; Kasama, T.; Hosaka, M. Combined infliximab and methotrexate treatment improves the depressive state in rheumatoid arthritis patients more effectively than methotrexate alone. Eur. J. Rheumatol. 2014, 1, 147–149. [Google Scholar] [CrossRef] [PubMed]
- Miwa, Y.; Isojima, S.; Saito, M.; Ikari, Y.; Kobuna, M.; Hayashi, T.; Takahashi, R.; Kasama, T.; Hosaka, M.; Sanada, K. Comparative Study of Infliximab Therapy and Methotrexate Monotherapy to Improve the Clinical Effect in Rheumatoid Arthritis Patients. Intern. Med. 2016, 55, 2581–2585. [Google Scholar] [CrossRef] [PubMed]
- Mathias, S.D.; Colwell, H.H.; Miller, D.P.; Moreland, L.W.; Buatti, M.; Wanke, L. Health-related quality of life and functional status of patients with rheumatoid arthritis randomly assigned to receive etanercept or placebo. Clin. Ther. 2000, 22, 128–139. [Google Scholar] [CrossRef]
- Bae, S.C.; Gun, S.C.; Mok, C.C.; Khandker, R.; Nab, H.W.; Koenig, A.S.; Vlahos, B.; Pedersen, R.; Singh, A. Improved health outcomes with etanercept versus usual DMARD therapy in an Asian population with established rheumatoid arthritis. BMC Musculoskelet. Disord. 2013, 14, 13–21. [Google Scholar] [CrossRef]
- Machado, D.A.; Guzman, R.M.; Xavier, R.M.; Simon, J.A.; Mele, L.; Pedersen, R.; Ferdousi, T.; Koenig, A.S.; Kotak, S.; Vlahos, B. Open-label observation of addition of etanercept versus a conventional disease-modifying antirheumatic drug in subjects with active rheumatoid arthritis despite methotrexate therapy in the Latin American region. J. Clin. Rheumatol. 2014, 20, 25–33. [Google Scholar] [CrossRef]
- Kekow, J.; Moots, R.J.; Emery, P.; Durez, P.; Koenig, A.; Singh, A.; Pedersen, R.; Robertson, D.; Freundlich, B.; Sato, R. Patient-reported outcomes improve with etanercept plus methotrexate in active early rheumatoid arthritis and the improvement is strongly associated with remission: The COMET trial. Ann. Rheum. Dis. 2010, 69, 222–225, Erratum in: Ann. Rheum. Dis. 2011, 70, 1519. [Google Scholar] [CrossRef]
- Kekow, J.; Moots, R.; Khandker, R.; Melin, J.; Freundlich, B.; Singh, A. Improvements in patient-reported outcomes, symptoms of depression and anxiety, and their association with clinical remission among patients with moderate-to-severe active early rheumatoid arthritis. Rheumatology 2011, 50, 401–409. [Google Scholar] [CrossRef]
- Curtis, J.R.; Herrem, C.; Ndlovu, N.; O’Brien, C.; Yazici, Y. A somatization comorbidity phenotype impacts response to therapy in rheumatoid arthritis: Post-hoc results from the certolizumab pegol phase 4 PREDICT trial. Arthritis Res. Ther. 2017, 19, 215. [Google Scholar] [CrossRef]
- Harrold, L.R.; John, A.; Reed, G.W.; Haselkorn, T.; Haselkorn, T.; Karki, C.; Li, Y.; Best, J.; Zlotnick, S.; Kremer, J.M.; et al. Impact of Tocilizumab Monotherapy on Clinical and Patient-Reported Quality-of-Life Outcomes in Patients with Rheumatoid Arthritis. Rheumatol. Ther. 2017, 4, 405–417. [Google Scholar] [CrossRef]
- Manning-Bennett, A.T.; Hopkins, A.M.; Sorich, M.J.; Proudman, S.M.; Foster, D.J.R.; Abuhelwa, A.Y.; Wiese, M.D. The association of depression and anxiety with treatment outcomes in patients with rheumatoid arthritis-a pooled analysis of five randomised controlled trials. Ther. Adv. Musculoskelet. Dis. 2022, 14, 1759720X221111613. [Google Scholar] [CrossRef]
- Behrens, F.; Burmester, G.R.; Hofmann, M.W.; Aringer, M.; Kellner, H.; Liebhaber, A.; Wassenberg, S.; Peters, M.A.; Zortel, M.; Amberger, C. Sustained effectiveness and safety of subcutaneous tocilizumab over two years in the ARATA observational study. Clin. Exp. Rheumatol. 2023, 41, 1463–1472. [Google Scholar] [CrossRef]
- Sun, Y.U.; Wang, D.; Salvadore, G.; Hsu, B.; Curran, M.; Casper, C.; Vermeulen, J.; Kent, J.M.; Singh, J.; Drevets, W.C.; et al. The effects of interleukin-6 neutralizing antibodies on symptoms of depressed mood and anhedonia in patients with rheumatoid arthritis and multicentric Castleman’s disease. Brain Behav. Immun. 2017, 66, 156–164. [Google Scholar] [CrossRef]
- Dhillon, S. Intravenous tocilizumab: A review of its use in adults with rheumatoid arthritis. BioDrugs 2014, 28, 75–106. [Google Scholar] [CrossRef]
- Persoons, P.; Vermeire, S.; Demyttenaere, K.; Fischler, B.; Vandenberghe, J.; Van Oudenhove, L.; Pierik, M.; Hlavaty, T.; Van Assche, G.; Noman, M.; et al. The impact of major depressive disorder on the short-and long-term outcome of Crohn’s disease treatment with infliximab. Aliment. Pharmacol. Ther. 2005, 22, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Minderhoud, I.M.; Samsom, M.; Oldenburg, B. Crohn’s disease, fatigue, and infliximab: Is there a role for cytokines in the pathogenesis of fatigue? World J. Gastroenterol. 2007, 13, 2089–2093. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, R.; Cella, D.; Leonardi, C.; Papp, K.; Gottlieb, A.B.; Dunn, M.; Chiou, C.F.; Patel, V.; Jahreis, A. Effects of etanercept therapy on fatigue and symptoms of depression in subjects treated for moderate to severe plaque psoriasis for up to 96 weeks. Br. J. Dermatol. 2007, 157, 1275–1277. [Google Scholar] [CrossRef] [PubMed]
- Tyring, S.; Gottlieb, A.; Papp, K.; Gordon, K.; Leonardi, C.; Wang, A.; Lalla, D.; Woolley, M.; Jahreis, A.; Zitnik, R.; et al. Etanercept and clinical outcomes, fatigue, and depression in psoriasis: Double-blind placebo-controlled randomised phase III trial. Lancet 2006, 367, 29–35. [Google Scholar] [CrossRef]
- Uzzan, S.; Azab, A.N. Anti-TNF-α Compounds as a Treatment for Depression. Molecules. 2021, 26, 2368. [Google Scholar] [CrossRef]
- Ertenli, I.; Ozer, S.; Kiraz, S.; Apras, S.B.; Akdogan, A.; Karadag, O.; Calguneri, M.; Kalyoncu, U. Infliximab, a TNF-alpha antagonist treatment in patients with ankylosing spondylitis: The impact on depression, anxiety and quality of life level. Rheumatol. Int. 2012, 32, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Webers, C.; Stolwijk, C.; Schiepers, O.; Schoonbrood, T.; van Tubergen, A.; Landewé, R.; van der Heijde, D.; Boonen, A. Infliximab treatment reduces depressive symptoms in patients with ankylosing spondylitis: An ancillary study to a randomized controlled trial (ASSERT). Arthritis Res. Ther. 2020, 22, 225. [Google Scholar] [CrossRef] [PubMed]
- Karson, A.; Demirtaş, T.; Bayramgürler, D.; Balci, F.; Utkan, T. Chronic administration of infliximab (TNF-α inhibitor) decreases depression and anxiety-like behaviour in rat model of chronic mild stress. Basic Clin. Pharmacol. Toxicol. 2013, 112, 335–340. [Google Scholar] [CrossRef]
- Bayramgürler, D.; Karson, A.; Özer, C.; Utkan, T. Effects of long-term etanercept treatment on anxiety- and depression-like neurobehaviors in rats. Physiol. Behav. 2013, 119, 145–148. [Google Scholar] [CrossRef]
- Mansur, R.B.; Delgado-Peraza, F.; Subramaniapillai, M.; Lee, Y.; Iacobucci, M.; Rodrigues, N.; Rosenblat, J.D.; Brietzke, E.; Cosgrove, V.E.; Kramer, N.E.; et al. Extracellular Vesicle Biomarkers Reveal Inhibition of Neuroinflammation by Infliximab in Association with Antidepressant Response in Adults with Bipolar Depression. Cells 2020, 9, 895. [Google Scholar] [CrossRef]
- Mansur, R.B.; Subramaniapillai, M.; Lee, Y.; Pan, Z.; Carmona, N.E.; Shekotikhina, M.; Iacobucci, M.; Rodrigues, N.; Nasri, F.; Rashidian, H.; et al. Leptin mediates improvements in cognitive function following treatment with infliximab in adults with bipolar depression. Psychoneuroendocrinology 2020, 120, 104779. [Google Scholar] [CrossRef] [PubMed]
- Mansur, R.B.; Subramaniapillai, M.; Lee, Y.; Pan, Z.; Carmona, N.E.; Shekotikhina, M.; Iacobucci, M.; Rodrigues, N.; Nasri, F.; Rosenblat, J.D.; et al. Effects of infliximab on brain neurochemistry of adults with bipolar depression. J. Affect. Disord. 2021, 281, 61–66. [Google Scholar] [CrossRef]
- Lwin, M.N.; Serhal, L.; Holroyd, C.; Edwards, C.J. Rheumatoid Arthritis: The Impact of Mental Health on Disease: A Narrative Review. Rheumatol. Ther. 2020, 7, 457–471. [Google Scholar] [CrossRef]

| Generic Name | Trade Name Original, Biosimilars | Group | Molecule Type | Mechanism of Action | |
|---|---|---|---|---|---|
| ADL | Adalimumab | Humira, Amgevita, Hyrimoz, Idacio, Hulio, Amsparity, Imraldi | anti-TNF | Fully human monoclonal IgG1 antibody | Neutralizes TNF-α (soluble and membrane-bound forms); blocks p55/p75 receptors |
| CZP | Certolizumab Pegol | Cimzia | anti-TNF | Humanized Fab’ fragment conjugated with PEG | Neutralizes TNF-α; lacks Fc-mediated effects |
| IFX | Infliximab | Remicade, Inflectra, Remsima, Flixabi, Zessly | anti-TNF | Chimeric IgG1 monoclonal antibody (human-mouse) | Blocks TNF-α; inhibits inflammatory pathways |
| ETN | Etanercept | Enbrel, Benepali, Erelzi | anti-TNF | Fusion protein: p75 TNF receptor + Fc IgG1 | Binds TNF-α and TNF-β; acts as a decoy receptor |
| GLM | Golimumab | Simponi (s.c.), Simponi Aria (i.v.) | anti-TNF | Fully human monoclonal IgG1 antibody | Blocks TNF-α; inhibits inflammatory pathways |
| SRK | Sirukumab | None (discontinued, never approved) | anti-IL-6 | Fully human monoclonal IgG1 antibody | Neutralizes IL-6 (the cytokine, not the receptor) |
| SARI | Sarilumab | Kevzara | anti-IL-6R | Fully human monoclonal IgG1 antibody | Blocks IL-6 receptor (soluble and membrane-bound) |
| TCZ | Tocilizumab | RoActemra (EU), Actemra (USA) | anti-IL-6R | Humanized monoclonal IgG1 antibody | Blocks IL-6 receptor (soluble and membrane-bound) |
| Study | Validated Psychiatric Tools | PROMIS Domains | Generic QoL Measures | Notes |
|---|---|---|---|---|
| Miwa et al., 2014 [23] | SDS | - | - | |
| Miwa et al., 2016 [24] | HAM-D | - | - | |
| Mathias et al., 2000 [25] | - | - | SF-36 | |
| Bae et al., 2013 [26]; Machado et al., 2014 [27]; Kekow et al., 2010/2011 [28,29] | HADS | - | - | |
| Bingham et al., 2023 [15] | - | Depression; Fatigue; Pain Interference; Physical Function | - | |
| Hsieh et al., 2023 [16] | - | - | EQ-5D-3L | |
| Curtis et al., 2017 [30] | - | - | - | Clinical analysis: SCP affected response |
| Tiosano et al., 2020 [17] | HDRS | - | - | |
| Harrold et al., 2017 [31] | - | - | EQ-5D | |
| Manning-Bennett et al., 2022 [32] | - | - | HAQ (functional measure) | Diagnosis from medical history. Pain VAS; remission via CDAI/SDAI; antidepressant use as covariate |
| Behrens et al., 2023 [33] | BDI-II | - | - | |
| Sun et al., 2017 [34] | PDMA | - | SF-36 | |
| Strand et al., 2020 [18] | - | - | SF-36 |
| Study | Study Design (Treatment Length) | Biologic Agent | Measure | Outcome |
|---|---|---|---|---|
| Miwa et al., 2014 [23] | Pilot study (30 weeks) | IFX (n = 34) vs. MTX (n = 42) | SDS | IFX significantly improved depression vs. MTX |
| Miwa et al., 2016 [24] | Open-label cohort (6 months) | IFX (n = 60) vs. MTX (n = 53) | HAM-D | No significant difference between IFX and MTX |
| Bringham et al., 2023 [15] | Observational Phase 4 AWARE (52 weeks) | GAL (n = 685) vs. IFX (n= 585) | PROMIS | Improvement in all PROMIS domains incl. depression |
| Curtis et al., 2017 [30] | RCT Phase 4 PREDICT (52 weeks) | CZP (n = 733) | Clinical data | SCP: lower treatment response, more AEs |
| Mathias et al., 2000 [25] | RCT Phase 3, double-blind (6 months) | ETN (n= 76) vs. placebo (n = 80) | SF-36 MOS | ETN > placebo in improving depressive symptoms |
| Bae et al., 2013 [26] | Open-label, multicentre (16 weeks) | ETN + MTX (n = 197) vs. DMARDs +MTX (n = 103) | HADS | Greater improvements in ETN + MTX group (HADS) |
| Machado et al., 2014 [27] | Open-label, randomized (24 weeks) | ETN + MTX (n = 281) vs. DMARDs + MTX (n = 142) | HADS | Improvements observed in ETN + MTX depressive domains |
| Kekow et al., 2010 [28] | RCT double-blind, COMET 104 weeks | ETN + MTX (n= 274) vs. MTX (n= 268) | HADS | ETN + MTX better than MTX alone in PROs |
| Kekow et al., 2011 [29] | RCT double-blind, COMET * 104 weeks | ETN + MTX vs. MTX | HADS | Clinical remission reduced depressive symptoms |
| Hsieh et al., 2023 [16] | Observational 24 weeks | ADL (n = 100) | EQ-5D-3L | Improvements from baseline to weeks 12, 24 |
| Study | Study Design/Duration | Biologic Agent (n) | Measures | Outcome |
|---|---|---|---|---|
| Tiosano et al., 2020 [17] | Observational 24 weeks | TCZ (n = 91) | HDRS | 66% of patients achieved improvements in depressive domains. |
| Harrold et al., 2017 [31] | Observational cohort study 1 year | TCZ (n = 255) | EQ-5D | 20% to 36% of patients achieved improvements in depressive state. |
| Manning-Bennett et al., 2022 [32] | 5 RCT | TCZ vs. DMARDs (n = 5502) | Clinical | Comorbid depression was associated with less frequent remission (CDAI and SDAI) |
| Behrens et al., 2021 [33] | Observational ARATA 52 weeks | TCZ (n = 1300) | BDI-II | Patients achieved improvements in DAS-28 and PROs; however, patients with depression presented lower response and higher adverse event rates. |
| Sun et al., 2017 [34] | Post hoc analysis RCT 24 weeks | sirukumab vs. siltuximab (n = 176) | PDMA includinngSF-36 | Baseline solute IL-6 receptor levels predicted mental health benefit. The improvement in depressive state by sirukumab correlated positively with the baseline solute IL-6R levels. |
| Strand et al., 2020 [18] | Post hoc analysis RCT MONARCH phase 3, and they were treated for 24 weeks | SARI or ADL (n = 148) | SF-36 | IL-6 blockade > TNF-α in QoL gains no difference in the mental state. High baseline IL-6 levels better improvements in physical domains with SARI compared to ADL. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siuchnińska, H.; Minarowska, A.; Wasilewska, E. Beyond Joints: Neuropsychiatric Benefits of TNF-α and IL-6 Inhibitors in Rheumatoid Arthritis—Narrative Review. Int. J. Mol. Sci. 2025, 26, 8361. https://doi.org/10.3390/ijms26178361
Siuchnińska H, Minarowska A, Wasilewska E. Beyond Joints: Neuropsychiatric Benefits of TNF-α and IL-6 Inhibitors in Rheumatoid Arthritis—Narrative Review. International Journal of Molecular Sciences. 2025; 26(17):8361. https://doi.org/10.3390/ijms26178361
Chicago/Turabian StyleSiuchnińska, Hanna, Alina Minarowska, and Eliza Wasilewska. 2025. "Beyond Joints: Neuropsychiatric Benefits of TNF-α and IL-6 Inhibitors in Rheumatoid Arthritis—Narrative Review" International Journal of Molecular Sciences 26, no. 17: 8361. https://doi.org/10.3390/ijms26178361
APA StyleSiuchnińska, H., Minarowska, A., & Wasilewska, E. (2025). Beyond Joints: Neuropsychiatric Benefits of TNF-α and IL-6 Inhibitors in Rheumatoid Arthritis—Narrative Review. International Journal of Molecular Sciences, 26(17), 8361. https://doi.org/10.3390/ijms26178361






