Levels of Exhaled Fraction of Nitric Oxide (FeNO) and Type 2 Biomarkers in Individuals Naturally Exposed to Helminth Parasites in a Tropical Region
Abstract
1. Introduction
2. Results
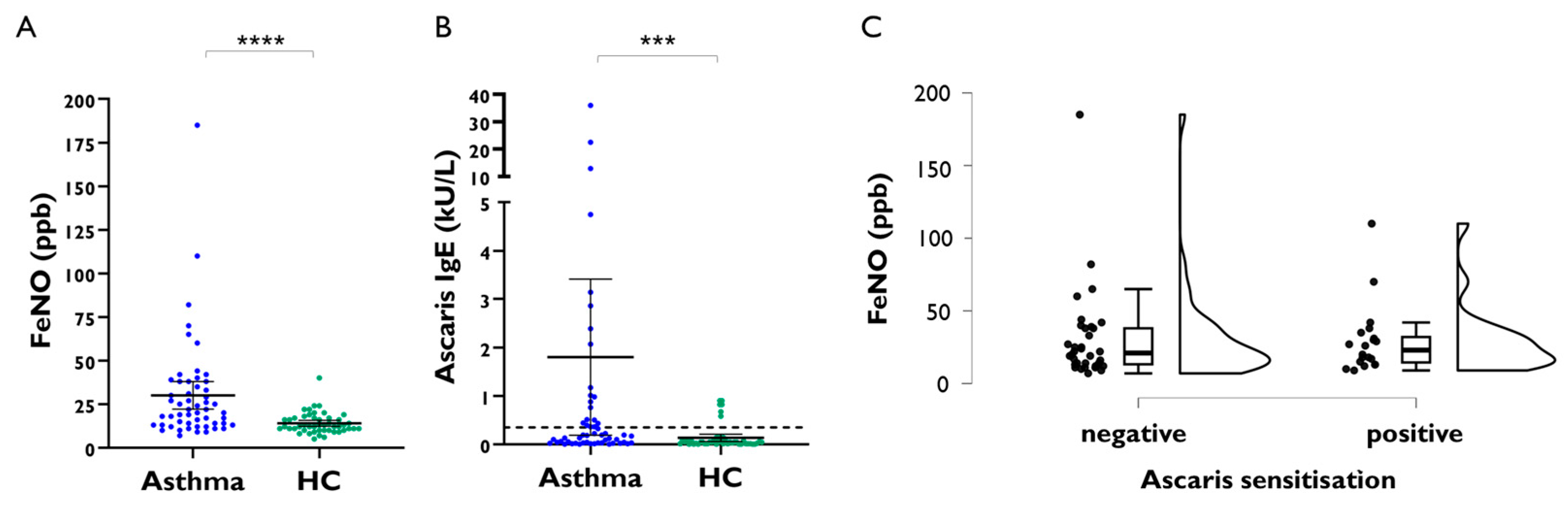
2.1. FeNO Levels in Asthmatics with Positive Specific IgE to Ascaris
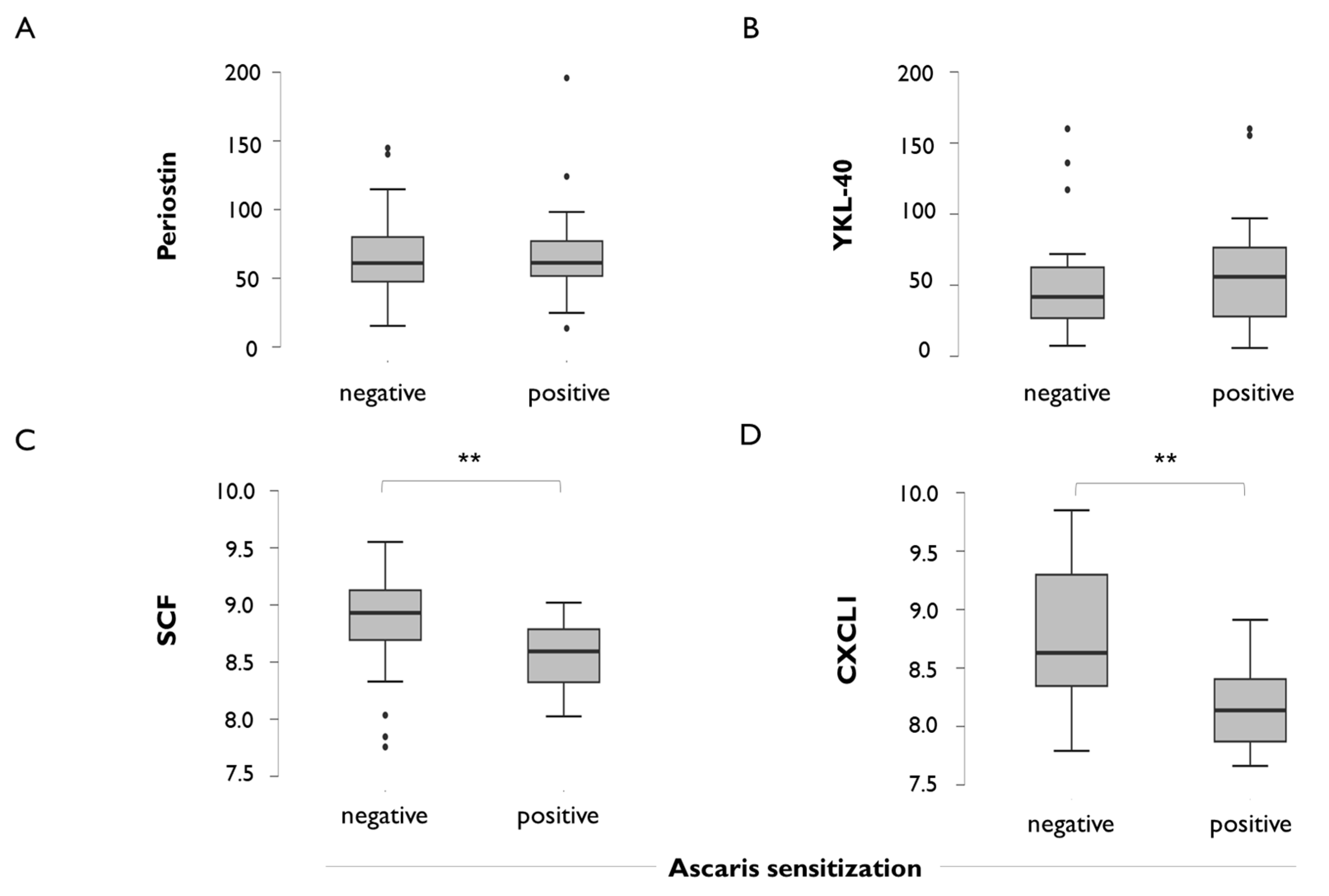
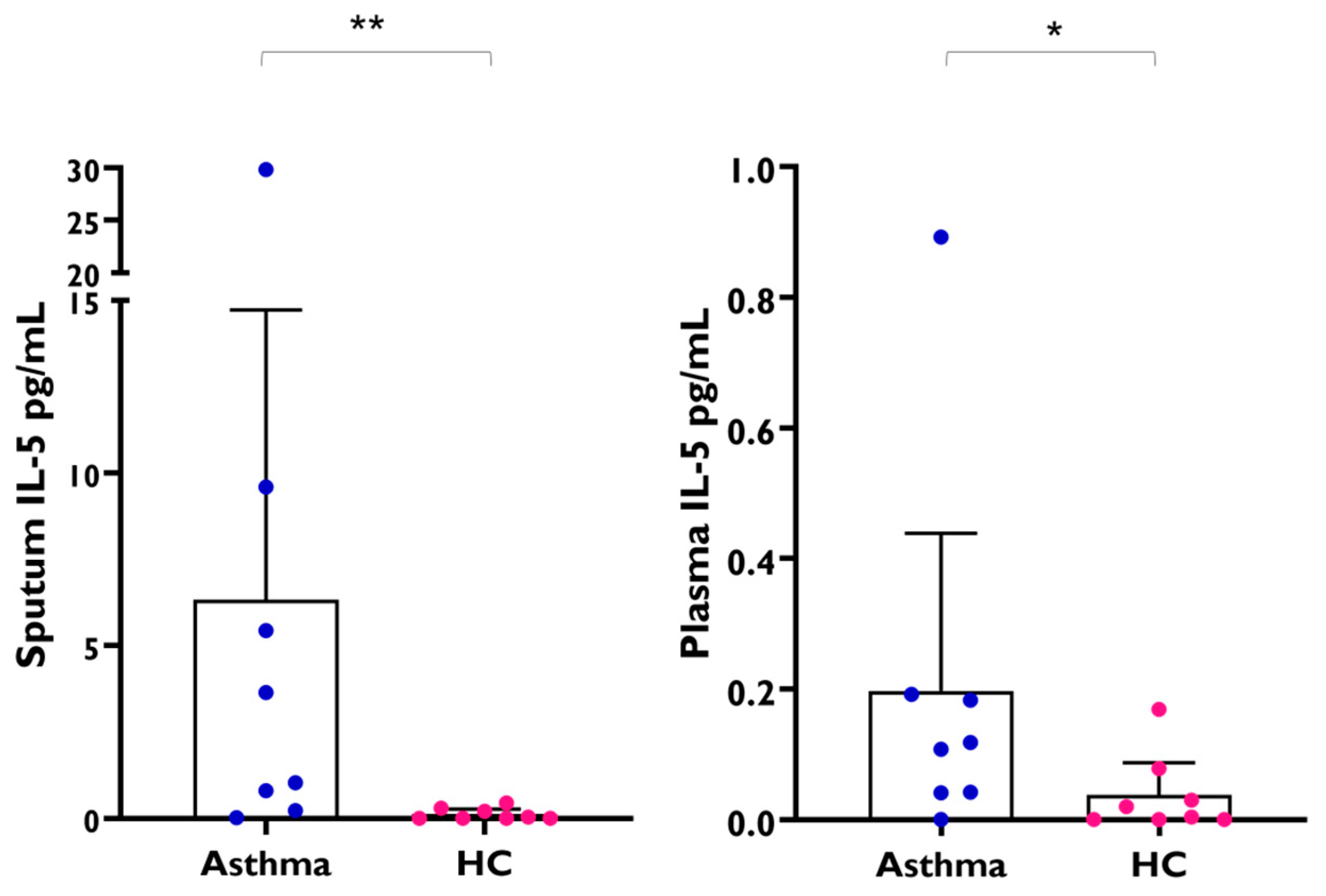
2.2. The Relationship of FeNO Levels and Specific IgE to Ascaris with Plasma Mediators in Asthmatics
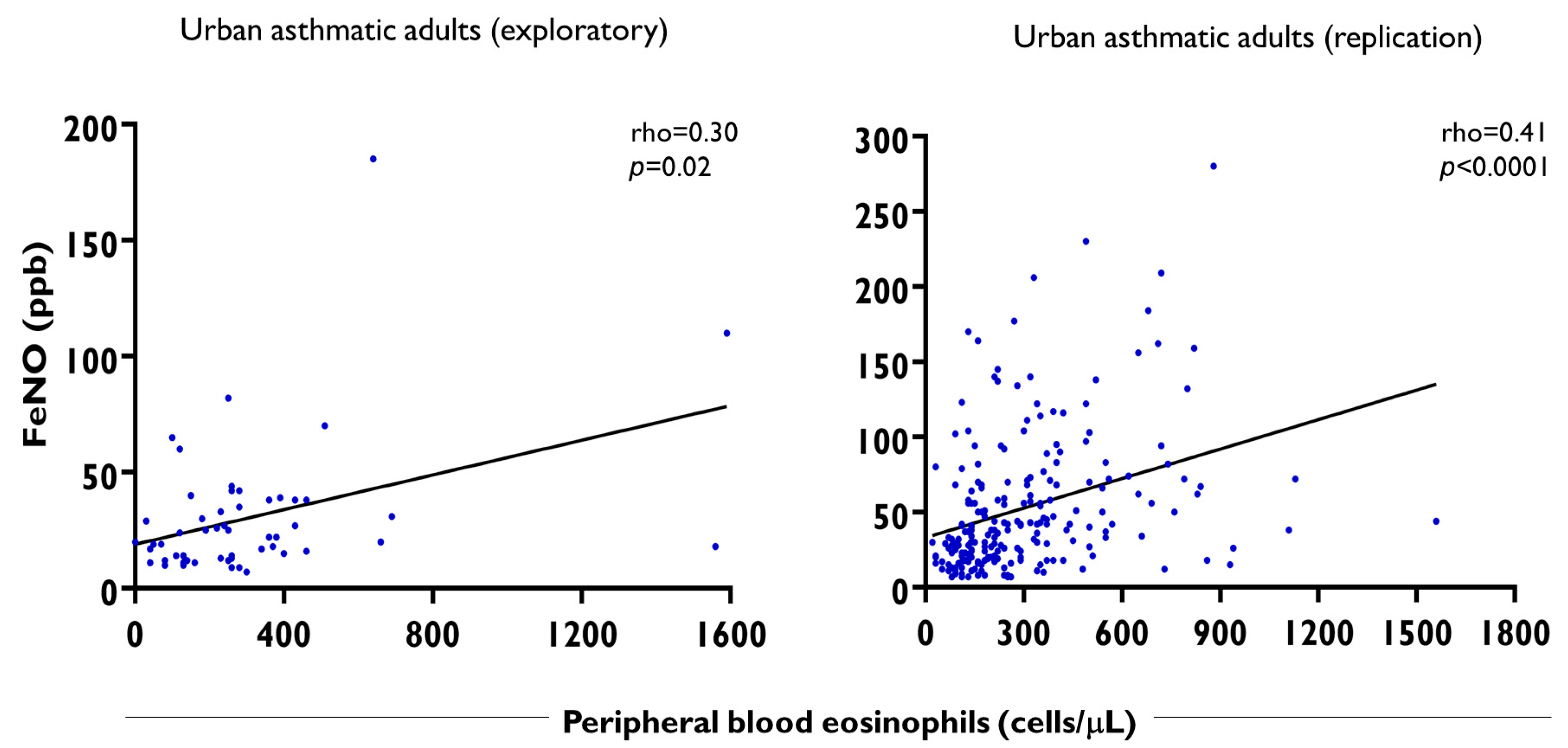
2.3. FeNO Levels in Adult Asthmatics with Positive Specific IgE to Ascaris lumbricoides and in the Context of Active Infection
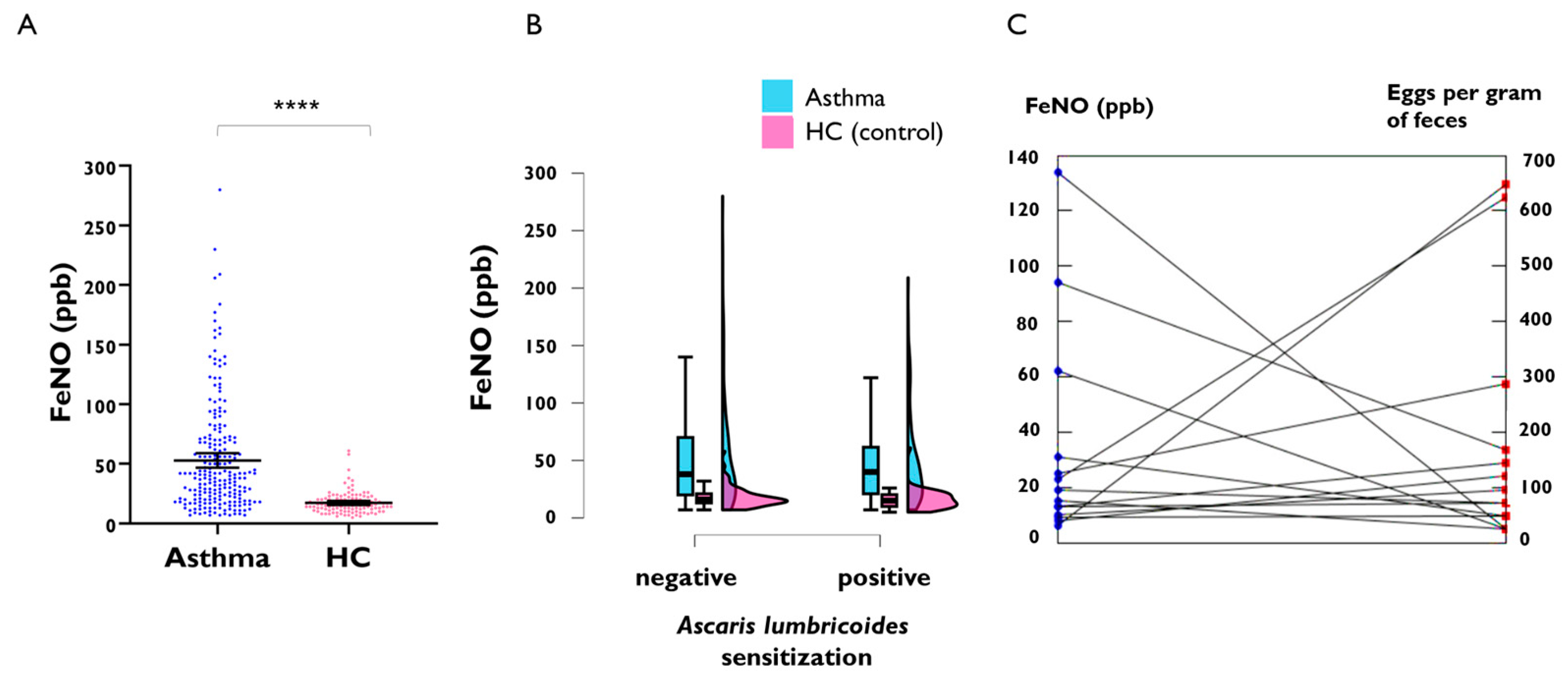
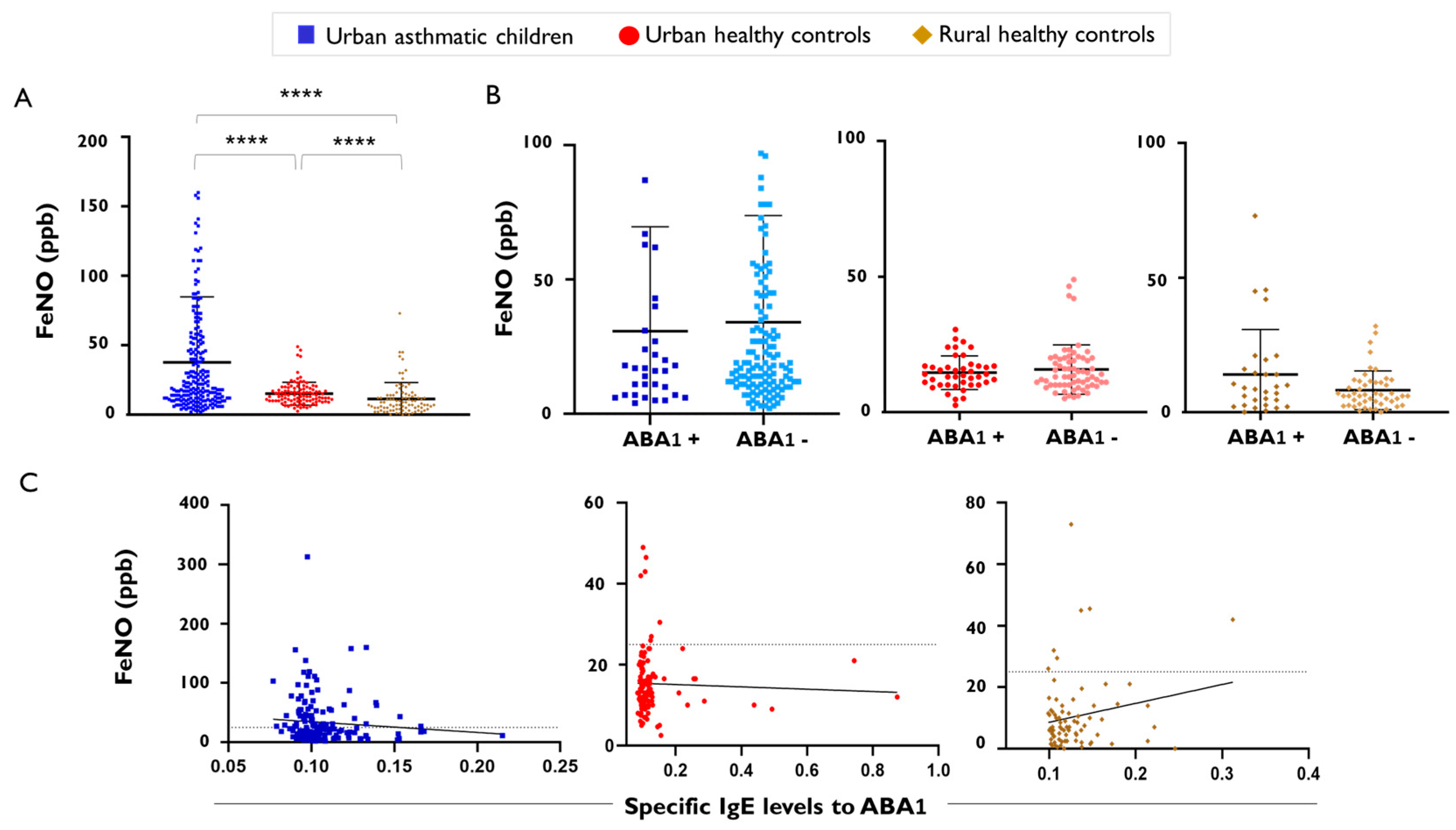
2.4. FeNO Levels in Asthmatics and Healthy Children with Active Ascaris lumbricoides Infection
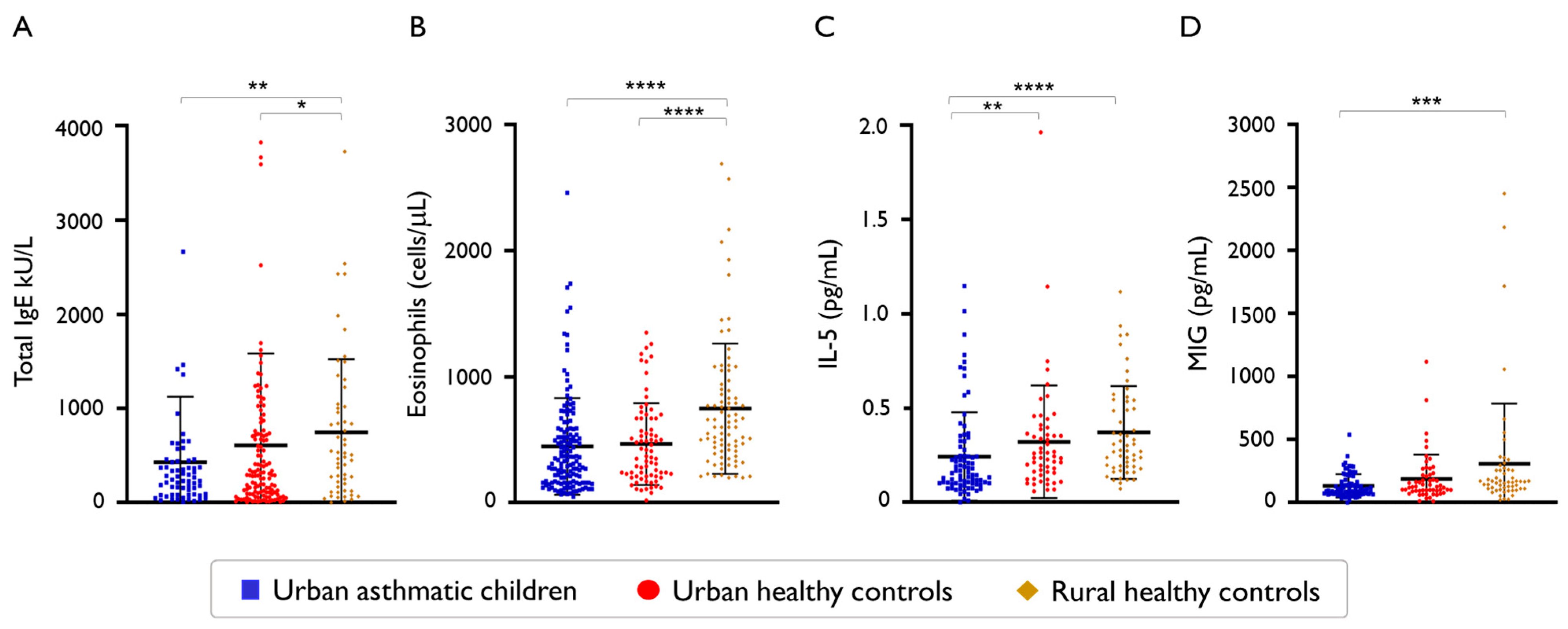
2.5. Other Type 2 Biomarkers and Chemokines in Healthy Rural Children with Low FeNO Levels
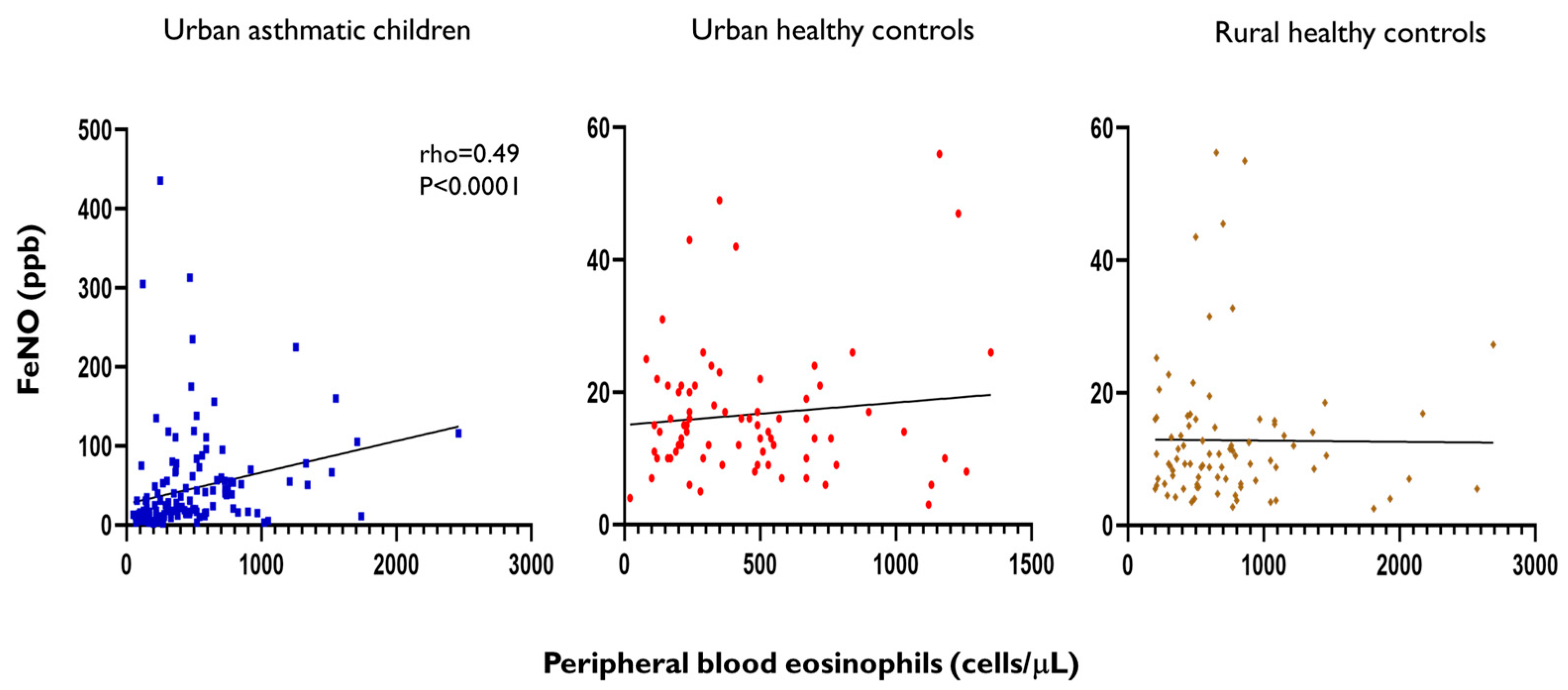
2.6. The Relationship of FeNO Levels with Blood Eosinophil Counts
3. Discussion
4. Materials and Methods
4.1. Adult Cohorts
4.2. Children Cohort
4.2.1. Urban Children
4.2.2. Rural Children
4.3. Fractional Exhaled Nitric Oxide (FeNO)
4.4. Blood Sample Collection
4.5. Stool Samples
4.6. Induced Sputum Samples
4.7. IgE Measurements
4.8. Periostin and YKL-40 Measurements
4.9. Proximity Extension Assay for Plasma Protein Levels
4.10. Cytokine Bead Array
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CI | Confidence interval |
| CXCL1 | C-X-C motif chemokine ligand 1 |
| CXCL9 | C-X-C motif chemokine ligand 9 |
| epg | Eggs per gram |
| FeNO | Exhaled fraction of nitric oxide |
| GINA | Global Initiative for Asthma |
| HC | Healthy control |
| IL-5 | Interleukin 5 |
| iNOS | Inducible nitric oxide synthase |
| IQR | Interquartile range |
| ppb | Parts per billion |
| SCF | Stem cell factor |
References
- Vacca, F.; Le Gros, G. Tissue-specific immunity in helminth infections. Mucosal Immunol. 2022, 15, 1212–1223. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.D.; Wang, Z.J.; Ogulur, I.; Li, S.J.; Yazici, D.; Li, X.H.; Pat, Y.; Zheng, Y.; Babayev, H.; Zeyneloglu, C.; et al. Type 2 Immunity and Its Role in Allergic Disorders. Allergy 2025, 80, 1848–1877. [Google Scholar] [CrossRef]
- Maizels, R.M.; Gause, W.C. Targeting helminths: The expanding world of type 2 immune effector mechanisms. J. Exp. Med. 2023, 220, e20221381. [Google Scholar] [CrossRef]
- Can, U.I.; Stenske, S.E.; Rosenbaum, M.D.; Reinhardt, R.L. Rapid group-2 innate lymphoid cell mobilization from the intestine aids in early lung defense and repair. Cell Rep. 2025, 44, 115868. [Google Scholar] [CrossRef] [PubMed]
- Henry, E.K.; Inclan-Rico, J.M.; Siracusa, M.C. Type 2 cytokine responses: Regulating immunity to helminth parasites and allergic inflammation. Curr. Pharmacol. Rep. 2017, 3, 346–359. [Google Scholar] [CrossRef]
- Loser, S.; Smith, K.A.; Maizels, R.M. Innate Lymphoid Cells in Helminth Infections-Obligatory or Accessory? Front. Immunol. 2019, 10, 620. [Google Scholar] [CrossRef]
- Gazzinelli-Guimaraes, P.H.; Golec, D.P.; Karmele, E.P.; Sciurba, J.; Bara-Garcia, P.; Hill, T.; Kang, B.; Bennuru, S.; Schwartzberg, P.L.; Nutman, T.B. Eosinophil trafficking in allergen-mediated pulmonary inflammation relies on IL-13-driven CCL-11 and CCL-24 production by tissue fibroblasts and myeloid cells. J. Allergy Clin. Immunol. Glob. 2023, 2, 100131. [Google Scholar] [CrossRef]
- Zakzuk, J.; Lopez, J.F.; Akdis, C.; Caraballo, L.; Akdis, M.; van de Veen, W. Human Ascaris infection is associated with higher frequencies of IL-10 producing B cells. PLoS Neglected Trop. Dis. 2024, 18, e0012520. [Google Scholar] [CrossRef]
- Cooper, P.J. Can intestinal helminth infections (geohelminths) affect the development and expression of asthma and allergic disease? Clin. Exp. Immunol. 2002, 128, 398–404. [Google Scholar] [CrossRef] [PubMed]
- Bohnacker, S.; Troisi, F.; de Los Reyes Jimenez, M.; Esser-von Bieren, J. What Can Parasites Tell Us About the Pathogenesis and Treatment of Asthma and Allergic Diseases. Front. Immunol. 2020, 11, 2106. [Google Scholar] [CrossRef] [PubMed]
- Maizels, R.M. Infections and allergy-helminths, hygiene and host immune regulation. Curr. Opin. Immunol. 2005, 17, 656–661. [Google Scholar] [CrossRef]
- Smits, H.H.; Everts, B.; Hartgers, F.C.; Yazdanbakhsh, M. Chronic helminth infections protect against allergic diseases by active regulatory processes. Curr. Allergy Asthma Rep. 2010, 10, 3–12. [Google Scholar] [CrossRef]
- Danilowicz-Luebert, E.; O’Regan, N.L.; Steinfelder, S.; Hartmann, S. Modulation of specific and allergy-related immune responses by helminths. J. Biomed. Biotechnol. 2011, 2011, 821578. [Google Scholar] [CrossRef]
- Caraballo, L.; Llinas-Caballero, K. The Relationship of Parasite Allergens to Allergic Diseases. Curr. Allergy Asthma Rep. 2023, 23, 363–373. [Google Scholar] [CrossRef]
- Avokpaho, E.; Gineau, L.; Sabbagh, A.; Atindegla, E.; Fiogbe, A.; Galagan, S.; Ibikounle, M.; Massougbodji, A.; Walson, J.L.; Luty, A.J.F.; et al. Multiple overlapping risk factors for childhood wheeze among children in Benin. Eur. J. Med. Res. 2022, 27, 304. [Google Scholar] [CrossRef]
- Arrais, M.; Maricoto, T.; Nwaru, B.I.; Cooper, P.J.; Gama, J.M.R.; Brito, M.; Taborda-Barata, L. Helminth infections and allergic diseases: Systematic review and meta-analysis of the global literature. J. Allergy Clin. Immunol. 2022, 149, 2139–2152. [Google Scholar] [CrossRef]
- Mkhize-Kwitshana, Z.L.; Naidoo, P.; Nkwanyana, N.M.; Mabaso, M.L.H. Concurrent allergy and helminthiasis in underprivileged urban South African adults previously residing in rural areas. Parasite Immunol. 2022, 44, e12913. [Google Scholar] [CrossRef]
- Takeuchi, H.; Takanashi, S.; Hasan, S.M.T.; Hore, S.K.; Yeasmin, S.; Ahmad, S.M.; Alam, M.J.; Jimba, M.; Khan, M.A.; Iwata, T. Anti-Ascaris IgE as a Risk Factor for Asthma Symptoms among 5-Year-Old Children in Rural Bangladesh with Even Decreased Ascaris Infection Prevalence. Int. Arch. Allergy Immunol. 2022, 183, 662–672. [Google Scholar] [CrossRef] [PubMed]
- Santiago Hda, C.; Ribeiro-Gomes, F.L.; Bennuru, S.; Nutman, T.B. Helminth infection alters IgE responses to allergens structurally related to parasite proteins. J. Immunol. 2015, 194, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Santiago Hda, C.; Nutman, T.B. Role in Allergic Diseases of Immunological Cross-Reactivity between Allergens and Homologues of Parasite Proteins. Crit. Rev. Immunol. 2016, 36, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zakzuk, J.; Acevedo, N.; Harb, H.; Eick, L.; Renz, H.; Potaczek, D.P.; Caraballo, L. IgE Levels to Ascaris and House Dust Mite Allergens Are Associated With Increased Histone Acetylation at Key Type-2 Immune Genes. Front. Immunol. 2020, 11, 756. [Google Scholar] [CrossRef]
- Matsunaga, K.; Koarai, A.; Koto, H.; Shirai, T.; Muraki, M.; Yamaguchi, M.; Hanaoka, M.; Japanese Respiratory Society Assembly on Pulmonary, P. Guidance for type 2 inflammatory biomarkers. Respir. Investig. 2025, 63, 273–288. [Google Scholar] [CrossRef]
- Escamilla-Gil, J.M.; Fernandez-Nieto, M.; Acevedo, N. Understanding the Cellular Sources of the Fractional Exhaled Nitric Oxide (FeNO) and Its Role as a Biomarker of Type 2 Inflammation in Asthma. Biomed. Res. Int. 2022, 2022, 5753524. [Google Scholar] [CrossRef]
- Dweik, R.A.; Boggs, P.B.; Erzurum, S.C.; Irvin, C.G.; Leigh, M.W.; Lundberg, J.O.; Olin, A.C.; Plummer, A.L.; Taylor, D.R.; American Thoracic Society Committee on Interpretation of Exhaled Nitric Oxide Levels for Clinical Applications. An official ATS clinical practice guideline: Interpretation of exhaled nitric oxide levels (FENO) for clinical applications. Am. J. Respir. Crit. Care Med. 2011, 184, 602–615. [Google Scholar] [CrossRef]
- Plaza Moral, V.; Alobid, I.; Alvarez Rodriguez, C.; Blanco Aparicio, M.; Ferreira, J.; Garcia, G.; Gomez-Outes, A.; Garin Escriva, N.; Gomez Ruiz, F.; Hidalgo Requena, A.; et al. GEMA 5.3. Spanish Guideline on the Management of Asthma. Open Respir. Arch. 2023, 5, 100277. [Google Scholar] [CrossRef]
- Cooper, P.J.; Chis Ster, I.; Chico, M.E.; Vaca, M.; Oviedo, Y.; Maldonado, A.; Barreto, M.L.; Platts-Mills, T.A.E.; Strachan, D.P. Impact of early life geohelminths on wheeze, asthma and atopy in Ecuadorian children at 8 years. Allergy 2021, 76, 2765–2775. [Google Scholar] [CrossRef] [PubMed]
- Hon, K.L.; Leung, A.K.C. An update on the current and emerging pharmacotherapy for the treatment of human ascariasis. Expert Opin. Pharmacother. 2024. [Google Scholar] [CrossRef] [PubMed]
- Weatherhead, J.E.; Gazzinelli-Guimaraes, P.; Knight, J.M.; Fujiwara, R.; Hotez, P.J.; Bottazzi, M.E.; Corry, D.B. Host Immunity and Inflammation to Pulmonary Helminth Infections. Front. Immunol. 2020, 11, 594520. [Google Scholar] [CrossRef] [PubMed]
- Ozdemir, O. Loeffler’s syndrome: A type of eosinophilic pneumonia mimicking community-acquired pneumonia and asthma that arises from Ascaris lumbricoides in a child. North Clin. Istanb. 2020, 7, 506–507. [Google Scholar] [CrossRef]
- Lopez, J.F.; Zakzuk, J.; Satitsuksanoa, P.; Lozano, A.; Buergi, L.; Heider, A.; Alvarado-Gonzalez, J.C.; Babayev, H.; Akdis, C.; van de Veen, W.; et al. Elevated circulating group-2 innate lymphoid cells expressing activation markers and correlated tryptase AB1 levels in active ascariasis. Front. Immunol. 2024, 15, 1459961. [Google Scholar] [CrossRef]
- Mugob, B.B.; Ngum, N.H.; Boubga, C.; Ngwenah, F.E.; Mahamat, O. Analysis of oxidative status, inflammatory cytokines, and Ascaris lumbricoides infection in women at a health district in Bamenda, Northwest, Cameroon. Egypt. J. Intern. Med. 2024, 36, 42. [Google Scholar] [CrossRef]
- Specjalski, K.; Chelminska, M.; Jassem, E. YKL-40 protein correlates with the phenotype of asthma. Lung 2015, 193, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H. Role of serum periostin in the management of asthma and its comorbidities. Respir. Investig. 2020, 58, 144–154. [Google Scholar] [CrossRef]
- Izuhara, K.; Nunomura, S.; Nanri, Y.; Ono, J.; Takai, M.; Kawaguchi, A. Periostin: An emerging biomarker for allergic diseases. Allergy 2019, 74, 2116–2128. [Google Scholar] [CrossRef] [PubMed]
- Hanibuchi, M.; Mitsuhashi, A.; Kajimoto, T.; Saijo, A.; Sato, S.; Kitagawa, T.; Nishioka, Y. Clinical significance of fractional exhaled nitric oxide and periostin as potential markers to assess therapeutic efficacy in patients with cough variant asthma. Respir. Investig. 2023, 61, 16–22. [Google Scholar] [CrossRef]
- Isomura, Y.; Hanibuchi, M.; Sato, S.; Mitsuhashi, A.; Kajimoto, T.; Saijo, A.; Nishioka, Y. Serial Changes of Fractional Exhaled Nitric Oxide and Periostin in the Treatment Course of Cough Variant Asthma. Am. J. Respir. Crit. Care Med. 2025, 211, A3460. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, X.; Liu, Y.; Zhang, L.; Zheng, J.; Wang, J.; Hansbro, P.M.; Wang, L.; Wang, G.; Hsu, A.C. Chitinase-like protein YKL-40 correlates with inflammatory phenotypes, anti-asthma responsiveness and future exacerbations. Respir. Res. 2019, 20, 95. [Google Scholar] [CrossRef]
- Chimponda, T.N.; Mduluza, T. Inflammation during Schistosoma haematobium infection and anti-allergy in pre-school-aged children living in a rural endemic area in Zimbabwe. Trop. Med. Int. Health 2020, 25, 618–623. [Google Scholar] [CrossRef]
- Zakzuk, J.; Casadiego, S.; Mercado, A.; Alvis-Guzman, N.; Caraballo, L. Ascaris lumbricoides infection induces both, reduction and increase of asthma symptoms in a rural community. Acta Trop. 2018, 187, 1–4. [Google Scholar] [CrossRef]
- Tsai, M.; Valent, P.; Galli, S.J. KIT as a master regulator of the mast cell lineage. J. Allergy Clin. Immunol. 2022, 149, 1845–1854. [Google Scholar] [CrossRef] [PubMed]
- Al-Muhsen, S.; Shablovsky, G.; Olivenstein, R.; Mazer, B.; Hamid, Q. The expression of stem cell factor and c-kit receptor in human asthmatic airways. Clin. Exp. Allergy 2004, 34, 911–916. [Google Scholar] [CrossRef] [PubMed]
- Makowska, J.S.; Cieslak, M.; Kowalski, M.L. Stem cell factor and its soluble receptor (c-kit) in serum of asthmatic patients-correlation with disease severity. BMC Pulm. Med. 2009, 9, 27. [Google Scholar] [CrossRef]
- Da Silva, C.A.; Reber, L.; Frossard, N. Stem cell factor expression, mast cells and inflammation in asthma. Fundam. Clin. Pharmacol. 2006, 20, 21–39. [Google Scholar] [CrossRef]
- Oliveira, S.H.; Taub, D.D.; Nagel, J.; Smith, R.; Hogaboam, C.M.; Berlin, A.; Lukacs, N.W. Stem cell factor induces eosinophil activation and degranulation: Mediator release and gene array analysis. Blood 2002, 100, 4291–4297. [Google Scholar] [CrossRef]
- Donaldson, L.E.; Schmitt, E.; Huntley, J.F.; Newlands, G.F.; Grencis, R.K. A critical role for stem cell factor and c-kit in host protective immunity to an intestinal helminth. Int. Immunol. 1996, 8, 559–567. [Google Scholar] [CrossRef]
- Newlands, G.F.; Miller, H.R.; MacKellar, A.; Galli, S.J. Stem cell factor contributes to intestinal mucosal mast cell hyperplasia in rats infected with Nippostrongylus brasiliensis or Trichinella spiralis, but anti-stem cell factor treatment decreases parasite egg production during N brasiliensis infection. Blood 1995, 86, 1968–1976. [Google Scholar] [CrossRef]
- Grencis, R.K.; Else, K.J.; Huntley, J.F.; Nishikawa, S.I. The in vivo role of stem cell factor (c-kit ligand) on mastocytosis and host protective immunity to the intestinal nematode Trichinella spiralis in mice. Parasite Immunol. 1993, 15, 55–59. [Google Scholar] [CrossRef]
- Murakami, M.; Austen, K.F.; Arm, J.P. The immediate phase of c-kit ligand stimulation of mouse bone marrow-derived mast cells elicits rapid leukotriene C4 generation through posttranslational activation of cytosolic phospholipase A2 and 5-lipoxygenase. J. Exp. Med. 1995, 182, 197–206. [Google Scholar] [CrossRef]
- Ptaschinski, C.; Zhu, D.; Fonseca, W.; Lukacs, N.W. Stem cell factor inhibition reduces Th2 inflammation and cellular infiltration in a mouse model of eosinophilic esophagitis. Mucosal Immunol. 2023, 16, 727–739. [Google Scholar] [CrossRef] [PubMed]
- Ogulur, I.; Mitamura, Y.; Yazici, D.; Pat, Y.; Ardicli, S.; Li, M.; D’Avino, P.; Beha, C.; Babayev, H.; Zhao, B.; et al. Type 2 immunity in allergic diseases. Cell. Mol. Immunol. 2025, 22, 211–242. [Google Scholar] [CrossRef] [PubMed]
- Radtke, D.; Voehringer, D. Granulocyte development, tissue recruitment, and function during allergic inflammation. Eur. J. Immunol. 2023, 53, e2249977. [Google Scholar] [CrossRef] [PubMed]
- Willis, C.R.; Siegel, L.; Leith, A.; Mohn, D.; Escobar, S.; Wannberg, S.; Misura, K.; Rickel, E.; Rottman, J.B.; Comeau, M.R.; et al. IL-17RA Signaling in Airway Inflammation and Bronchial Hyperreactivity in Allergic Asthma. Am. J. Respir. Cell Mol. Biol. 2015, 53, 810–821. [Google Scholar] [CrossRef]
- Xu, L.; Wei, Z.; Wu, R.; Kong, S.; Bin, J.; Gao, Y.; Fang, L. Acute airway eosinophilic inflammation model in mice induced by ovalbumin, house dust mite, or shrimp tropomyosin: A comparative study. Front. Allergy 2025, 6, 1594028. [Google Scholar] [CrossRef]
- Sawant, K.V.; Poluri, K.M.; Dutta, A.K.; Sepuru, K.M.; Troshkina, A.; Garofalo, R.P.; Rajarathnam, K. Chemokine CXCL1 mediated neutrophil recruitment: Role of glycosaminoglycan interactions. Sci. Rep. 2016, 6, 33123. [Google Scholar] [CrossRef]
- Alkhouri, H.; Moir, L.M.; Armour, C.L.; Hughes, J.M. CXCL1 is a negative regulator of mast cell chemotaxis to airway smooth muscle cell products in vitro. Clin. Exp. Allergy 2014, 44, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Rahmawati, S.F.; Te Velde, M.; Kerstjens, H.A.M.; Domling, A.S.S.; Groves, M.R.; Gosens, R. Pharmacological Rationale for Targeting IL-17 in Asthma. Front. Allergy 2021, 2, 694514. [Google Scholar] [CrossRef]
- Else, K.J.; Keiser, J.; Holland, C.V.; Grencis, R.K.; Sattelle, D.B.; Fujiwara, R.T.; Bueno, L.L.; Asaolu, S.O.; Sowemimo, O.A.; Cooper, P.J. Whipworm and roundworm infections. Nat. Rev. Dis. Primers 2020, 6, 44. [Google Scholar] [CrossRef] [PubMed]
- Kiflie, A.; Bewket, G.; Tajebe, F.; Abate, E.; Schön, T.; Blomgran, R. Helminth species-specific effects on IFN-gamma producing T cells during active and latent tuberculosis. PLoS Neglected Trop. Dis. 2023, 17, e0011094. [Google Scholar] [CrossRef]
- Sharma, M.; Khurana, S. Immunomodulation by helminthic parasites and worm therapy. Trop. Parasitol. 2025, 15, 2–7. [Google Scholar] [CrossRef]
- Maizels, R.M.; McSorley, H.J.; Smits, H.H.; Ten Dijke, P.; Hinck, A.P. Cytokines from parasites: Manipulating host responses by molecular mimicry. Biochem. J. 2025, 482, 433–449. [Google Scholar] [CrossRef]
- Stear, M.; Maruszewska-Cheruiyot, M.; Donskow-Lysoniewska, K. Modulation of the Immune Response by Nematode Derived Molecules. Int. J. Mol. Sci. 2025, 26, 5600. [Google Scholar] [CrossRef]
- Arora, P.; Moll, J.M.; Andersen, D.; Workman, C.T.; Williams, A.R.; Kristiansen, K.; Brix, S. Body fluid from the parasitic worm Ascaris suum inhibits broad-acting pro-inflammatory programs in dendritic cells. Immunology 2020, 159, 322–334. [Google Scholar] [CrossRef] [PubMed]
- Antunes, M.; Titz, T.D.O.; Batista, I.D.F.C.; Marques-Porto, R.; Oliveira, C.F.D.; De Araujo, C.A.; Macedo-Soares, M.F.D. Immunosuppressive PAS-1 is an excretory/secretory protein released by larval and adult worms of the ascarid nematode Ascaris suum. J. Helminthol. 2015, 89, 367–374. [Google Scholar] [CrossRef]
- Caraballo, L.; Zakzuk, J.; Acevedo, N. Helminth-derived cystatins: The immunomodulatory properties of an Ascaris lumbricoides cystatin. Parasitology 2021, 148, 1744–1756. [Google Scholar] [CrossRef] [PubMed]
- Acevedo, N.; Lozano, A.; Zakzuk, J.; Llinas-Caballero, K.; Brodin, D.; Nejsum, P.; Williams, A.R.; Caraballo, L. Cystatin from the helminth Ascaris lumbricoides upregulates mevalonate and cholesterol biosynthesis pathways and immunomodulatory genes in human monocyte-derived dendritic cells. Front. Immunol. 2024, 15, 1328401. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, L.C.; Newcombe, P.J.; Cunha, S.S.; Alcantara-Neves, N.M.; Genser, B.; Cruz, A.A.; Simoes, S.M.; Fiaccone, R.; Amorim, L.; Cooper, P.J.; et al. Early infection with Trichuris trichiura and allergen skin test reactivity in later childhood. Clin. Exp. Allergy 2008, 38, 1769–1777. [Google Scholar] [CrossRef]
- Kang, S.A.; Park, M.K.; Cho, M.K.; Park, S.K.; Jang, M.S.; Yang, B.G.; Jang, M.H.; Kim, D.H.; Yu, H.S. Parasitic nematode-induced CD4+Foxp3+T cells can ameliorate allergic airway inflammation. PLoS Neglected Trop. Dis. 2014, 8, e3410. [Google Scholar] [CrossRef]
- Araujo, C.A.; Perini, A.; Martins, M.A.; Macedo, M.S.; Macedo-Soares, M.F. PAS-1, a protein from Ascaris suum, modulates allergic inflammation via IL-10 and IFN-gamma, but not IL-12. Cytokine 2008, 44, 335–341. [Google Scholar] [CrossRef]
- Perez, M.G.; Gillan, V.; Anderson, W.M.; Gerbe, F.; Herbert, F.; McNeilly, T.N.; Maizels, R.M.; Jay, P.; Devaney, E.; Britton, C. A secreted helminth microRNA suppresses gastrointestinal cell differentiation required for innate immunity. Front. Immunol. 2025, 16, 1558132. [Google Scholar] [CrossRef]
- Getaneh, A.; Tamrat, A.; Tadesse, K. Arginase activity in peripheral blood of patients with intestinal schistosomiasis, Wonji, Central Ethiopia. Parasite Immunol. 2015, 37, 380–383. [Google Scholar] [CrossRef]
- Kouro, T.; Takatsu, K. IL-5- and eosinophil-mediated inflammation: From discovery to therapy. Int. Immunol. 2009, 21, 1303–1309. [Google Scholar] [CrossRef]
- Jackson, D.J.; Wechsler, M.E.; Brusselle, G.; Buhl, R. Targeting the IL-5 pathway in eosinophilic asthma: A comparison of anti-IL-5 versus anti-IL-5 receptor agents. Allergy 2024, 79, 2943–2952. [Google Scholar] [CrossRef]
- Noble, S.L.; Vacca, F.; Hilligan, K.L.; Mules, T.C.; Le Gros, G.; Inns, S. Helminth infection induces a distinct subset of CD101(hi) lung tissue-infiltrating eosinophils that are differentially regulated by type 2 cytokines. Immunol. Cell Biol. 2024, 102, 734–746. [Google Scholar] [CrossRef]
- Agache, I.; Salazar, J.; Rodriguez-Tanta, Y.; Saenz, F.K.F.; Haahtela, T.; Traidl-Hoffmann, C.; Damialis, A.; Vecillas, L.; Giovannini, M.; Nadeau, K.C.; et al. The Impact of Rhinovirus, Syncytial Respiratory Virus and Helminth Infection on the Risk of New-Onset Asthma and Other Allergic Conditions-A Systematic Review for the EAACI Guidelines on Environmental Science for Allergic Diseases and Asthma. Allergy 2025, 80, 1878–1898. [Google Scholar] [CrossRef] [PubMed]
- Escamilla-Gil, J.M.; Torres-Duque, C.A.; Llinas-Caballero, K.; Proanos-Jurado, N.J.; De Vivero, M.M.; Ramirez, J.C.; Regino, R.; Florez de Arco, L.T.; Dennis, R.; Gonzalez-Garcia, M.; et al. Plasma Levels of CXCL9 and MCP-3 are Increased in Asthma-COPD Overlap (ACO) Patients. Int. J. Chron. Obs. Pulmon Dis. 2025, 20, 1161–1174. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Helminth control in school-age children: A guide for managers of control programmes. In Helminth Control in School-Age Children: A Guide for Managers of Control Programmes; World Health Organization: Geneva, Switzerland, 2011; p. 87. [Google Scholar]
- Dragonieri, S.; Bikov, A.; Capuano, A.; Scarlata, S.; Carpagnano, G.E. Methodological Aspects of Induced Sputum. Adv. Respir. Med. 2023, 91, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Guiot, J.; Demarche, S.; Henket, M.; Paulus, V.; Graff, S.; Schleich, F.; Corhay, J.L.; Louis, R.; Moermans, C. Methodology for Sputum Induction and Laboratory Processing. J. Vis. Exp. 2017, 130, e56612. [Google Scholar] [CrossRef] [PubMed]
| Code | Group | Age | Sex | FeNO (ppb) | Ascaris Epg of Faeces |
|---|---|---|---|---|---|
| Cs013 | HC | 7 | M | 10 | 72 |
| Cs024 | HC | 5 | F | 12 | 322 |
| Cs029 | HC | 10 | F | 13 | 2783 |
| Cs031 | HC | 10 | M | 14 | 3657 |
| Cs062 | HC | 11 | F | 11 | 4 |
| Cs086 | HC | 6 | F | 15 | 192 |
| Cs094 | HC | 8 | F | 20 | 120 |
| Cs108 | HC | 8 | M | 22 | 1416 |
| Cs142 | HC | 14 | F | 10 | 24 |
| AN022 | Asthma | 12 | F | 18 | 96 |
| AN047 | Asthma | 7 | M | 16 | 2277 |
| AN057 | Asthma | 12 | M | 7 | 7636 |
| AN223 | Asthma | 8 | M | 29 | 288 |
| AN279 | Asthma | 9 | M | 5 | 96 |
| AN338 | Asthma | 10 | M | 18 | 24 |
| Code | Age | Sex | FeNO (ppb) | Trichuris Trichiura | Ascaris Lumbricoides | Ancylostoma Duodenale | Hymenolepis Nana |
|---|---|---|---|---|---|---|---|
| LA012 | 7 | M | 1 | 1872 | 72 | - | - |
| LA019 | 7 | M | 16 | 120 | - | - | - |
| LA021 | 8 | M | 45 | 72 | - | - | - |
| LA028 | 8 | F | 4.5 | 600 | - | - | - |
| LA034 | 8 | M | 0 | 48 | - | 168 | - |
| LA041 | 9 | F | 2.5 | 2592 | - | - | - |
| LA053 | 9 | M | 21 | 336 | - | - | - |
| LA056 | 6 | F | 2 | 6480 | - | - | - |
| LA059 | 11 | M | 3 | 480 | - | - | - |
| LA060 | 7 | M | 2 | 17,520 | - | - | - |
| LA061 | 7 | M | 5 | 360 | - | - | - |
| ST050 | 6 | M | 7.5 | 216 | - | - | - |
| ST085 | 13 | F | 6.5 | 96 | - | - | - |
| ST093 | 9 | F | 4 | 120 | - | - | 72 |
| ST094 | 7 | M | 12 | 48 | - | 312 | 72 |
| ST095 | 8 | F | 19.5 | 288 | 1824 | - | - |
| ST097 | 10 | F | 22.3 | 9432 | - | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Vivero, M.M.; Reina, R.; Ramírez, J.; Zakzuk, J.; Escamilla Gil, J.M.; Zelaya, B.; Florez de Arco, L.T.; Potaczek, D.P.; Caraballo, L.; Acevedo, N. Levels of Exhaled Fraction of Nitric Oxide (FeNO) and Type 2 Biomarkers in Individuals Naturally Exposed to Helminth Parasites in a Tropical Region. Int. J. Mol. Sci. 2025, 26, 8344. https://doi.org/10.3390/ijms26178344
De Vivero MM, Reina R, Ramírez J, Zakzuk J, Escamilla Gil JM, Zelaya B, Florez de Arco LT, Potaczek DP, Caraballo L, Acevedo N. Levels of Exhaled Fraction of Nitric Oxide (FeNO) and Type 2 Biomarkers in Individuals Naturally Exposed to Helminth Parasites in a Tropical Region. International Journal of Molecular Sciences. 2025; 26(17):8344. https://doi.org/10.3390/ijms26178344
Chicago/Turabian StyleDe Vivero, Maria M., Randy Reina, Jonathan Ramírez, Josefina Zakzuk, Jose Miguel Escamilla Gil, Bayron Zelaya, Lucila Teresa Florez de Arco, Daniel P. Potaczek, Luis Caraballo, and Nathalie Acevedo. 2025. "Levels of Exhaled Fraction of Nitric Oxide (FeNO) and Type 2 Biomarkers in Individuals Naturally Exposed to Helminth Parasites in a Tropical Region" International Journal of Molecular Sciences 26, no. 17: 8344. https://doi.org/10.3390/ijms26178344
APA StyleDe Vivero, M. M., Reina, R., Ramírez, J., Zakzuk, J., Escamilla Gil, J. M., Zelaya, B., Florez de Arco, L. T., Potaczek, D. P., Caraballo, L., & Acevedo, N. (2025). Levels of Exhaled Fraction of Nitric Oxide (FeNO) and Type 2 Biomarkers in Individuals Naturally Exposed to Helminth Parasites in a Tropical Region. International Journal of Molecular Sciences, 26(17), 8344. https://doi.org/10.3390/ijms26178344









