Nutrient-Enriched Germinated Brown Rice Alters the Intestinal Ecological Network by Regulating Lipid Metabolism in Rats
Abstract
1. Introduction
2. Results
2.1. Nutritional Components of Germinated Brown Rice
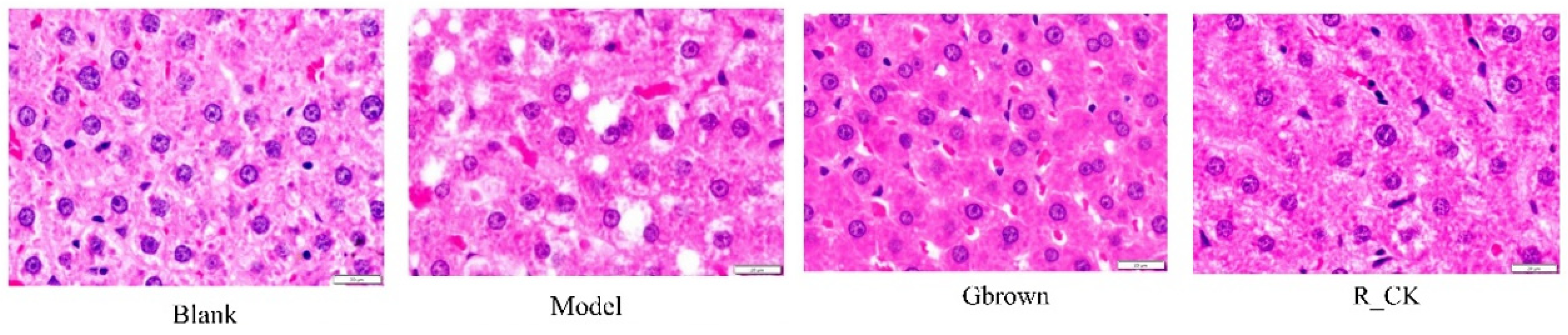
2.2. Rat Liver Morphology
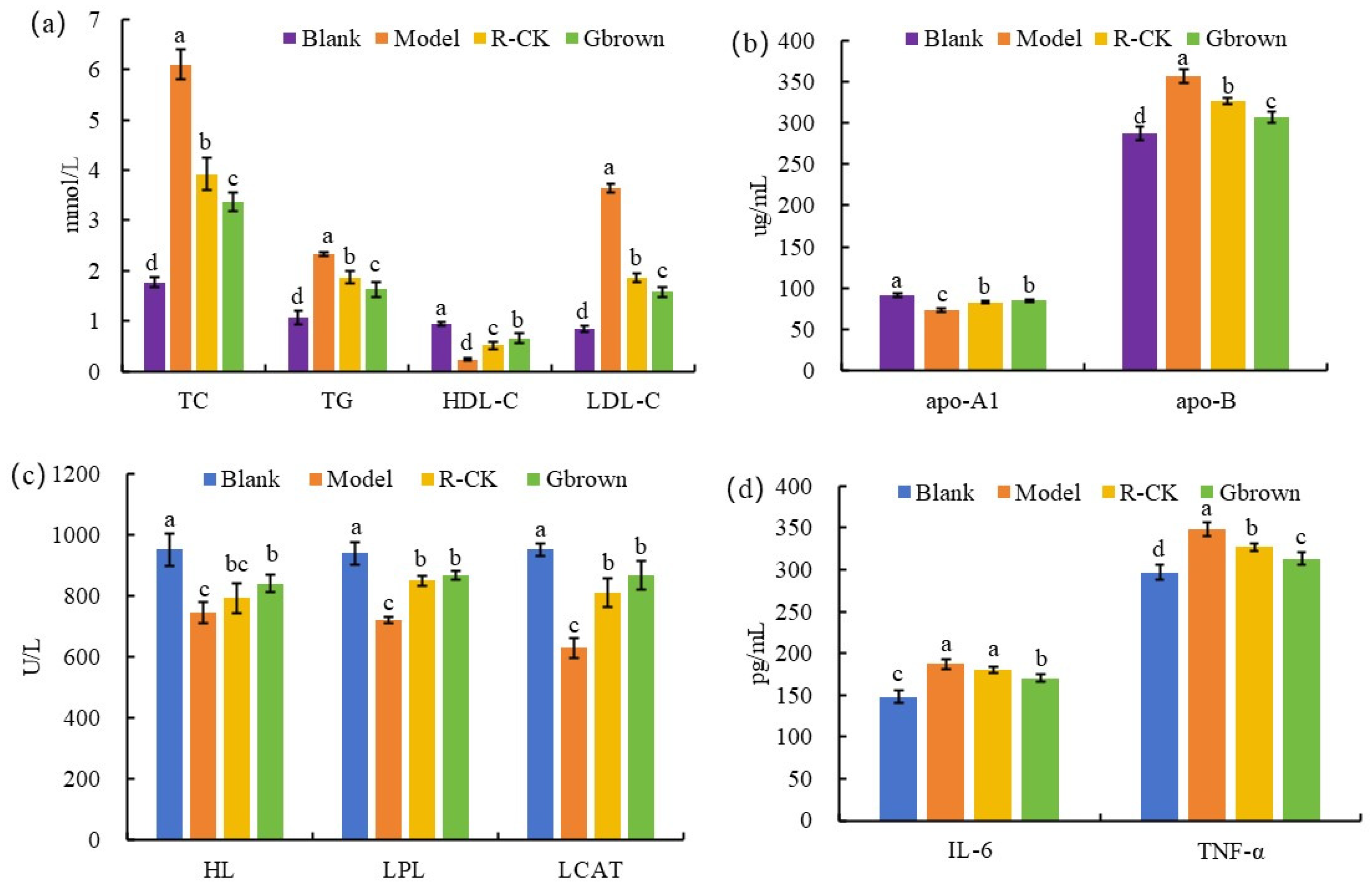
2.3. Blood Lipid Levels and Inflammation in Rats
2.4. Lipid Metabolism-Related Factors in Rats
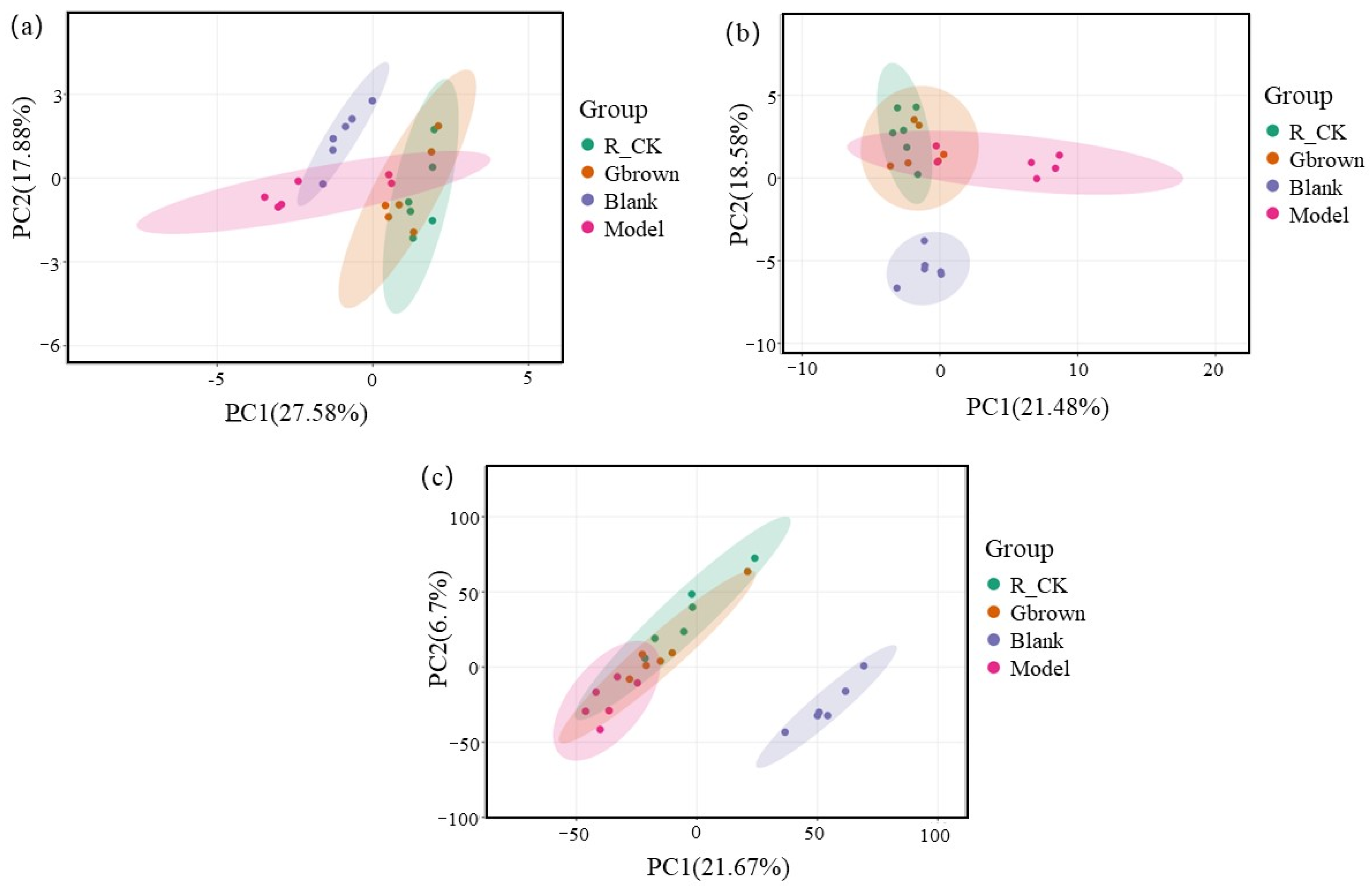
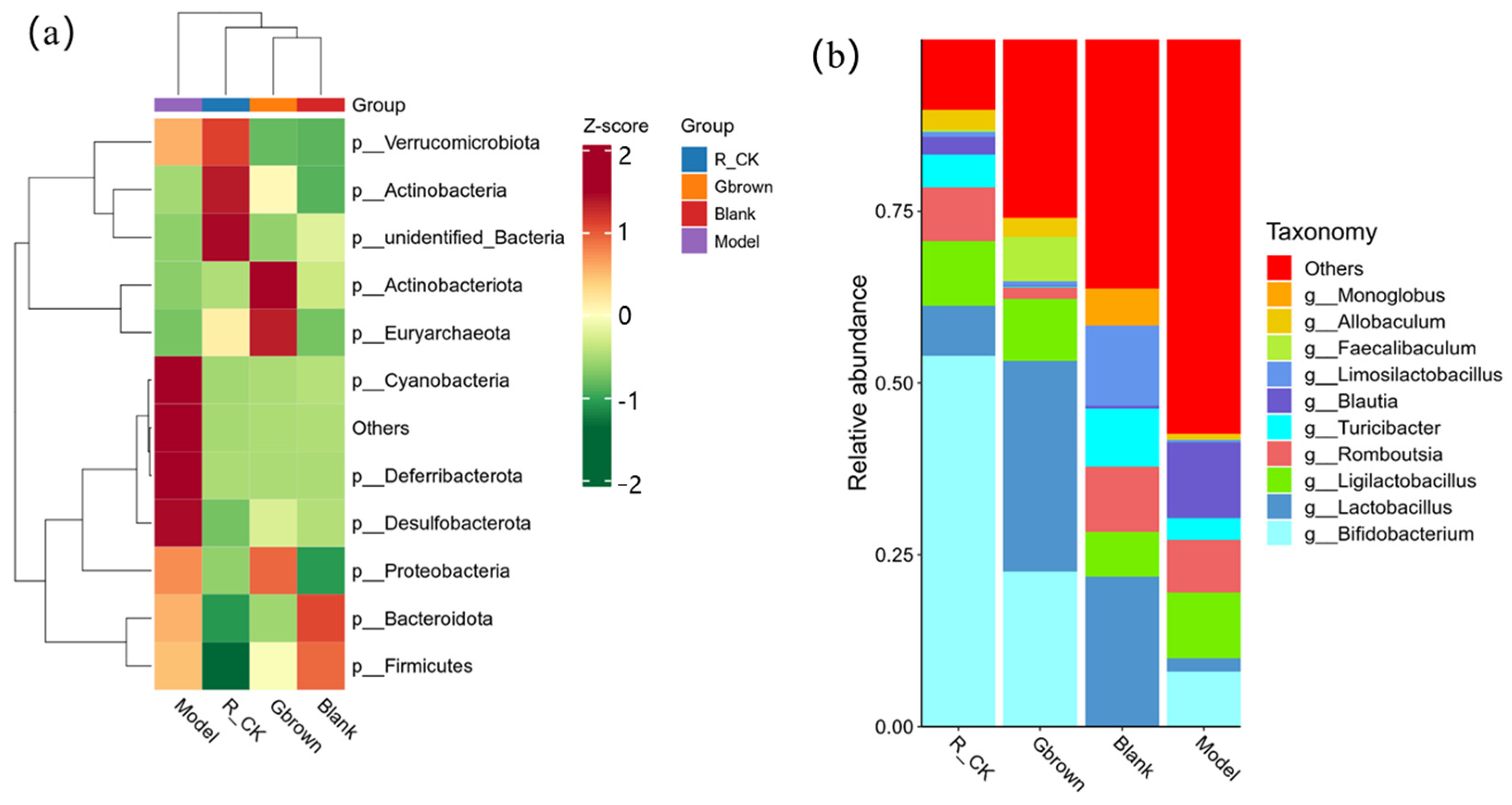
2.5. Rat Intestinal Microbiota
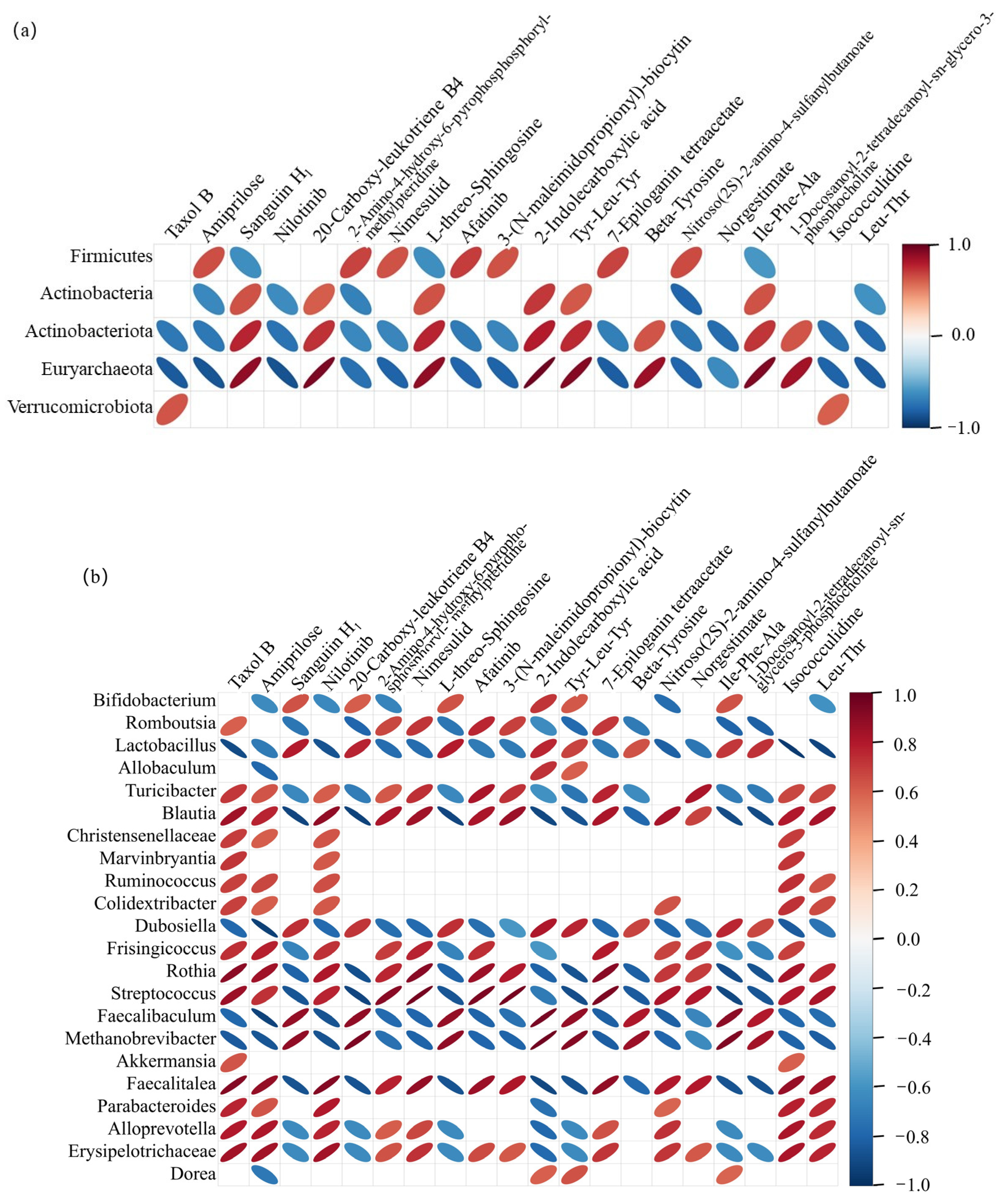
2.6. Correlation Between Gut Microbiota and Metabolite Levels in Rats
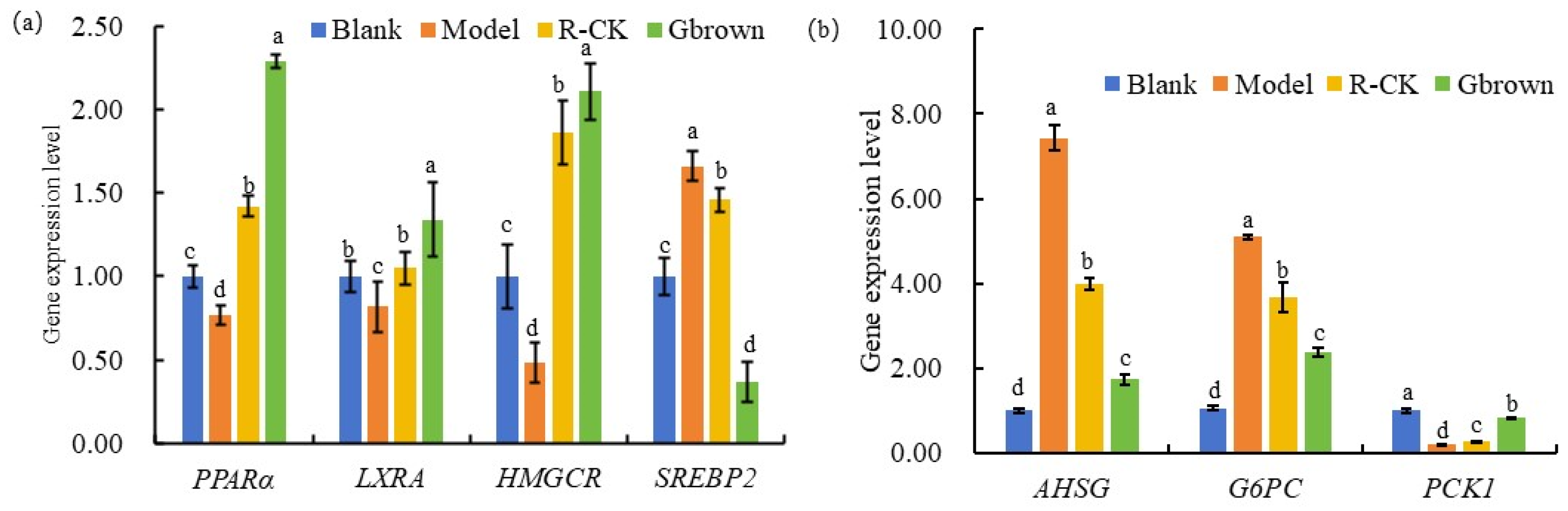
2.7. Lipid Metabolism-Related Genes in Rats
3. Discussion
3.1. Metabolic Regulatory Effects of GABA
3.2. Metabolic Regulation of RS
3.3. Metabolic Regulatory Effects of Flavonoids
3.4. Metabolic Regulatory Effects of Insoluble Dietary Fibers
4. Materials and Methods
4.1. Materials
4.2. Experimental Animals
4.3. Main Reagent
4.4. Main Instruments
4.5. Composition Determination
4.6. Feeding of Experimental Animals
4.7. Sample Collection
4.8. Serum Sample Evaluation
4.9. Hematoxylin and Eosin Staining
4.10. Quantitative Polymerase Chain Reaction (qPCR) Analysis
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Beaulieu, J.C.; Moreau, R.A.; Powell, M.J.; Obando-Ulloa, J.M. Lipid Profiles in Preliminary Germinated Brown Rice Beverages Compared to Non-Germinated Brown and White Rice Beverages. Foods 2022, 11, 220. [Google Scholar] [CrossRef]
- Munarko, H.; Sitanggang, A.B.; Kusnandar, F.; Budijanto, S. Effect of different soaking and germination methods on bioactive compounds of germinated brown rice. Int. J. Food Sci. Technol. 2021, 56, 4540–4548. [Google Scholar] [CrossRef]
- Castanho, A.; Pereira, C.; Lageiro, M.; Oliveira, J.C.; Cunha, L.M.; Brites, C. Improving γ-Oryzanol and γ-Aminobutyric Acid Contents in Rice Beverage Amazake Produced with Brown, Milled and Germinated Rices. Foods 2023, 12, 1476. [Google Scholar] [CrossRef]
- Wei, S.; Wang, N.; Huang, X.; Xu, G.; Xu, X.; Xu, D.; Jin, Y.; Yang, N.; Wu, F. Effect of germination on the quality characteristics and volatile compounds of fermented brown rice cake. Food Biosci. 2022, 50, 102165. [Google Scholar] [CrossRef]
- Ren, C.-Y.; Lu, S.-W.; Guan, L.-J.; Hong, B.; Zhang, Y.-L.; Huang, W.-G.; Li, B.; Liu, W.; Lu, W.-H. The metabolomics variations among rice, brown rice, wet germinated brown rice, and processed wet germinated brown rice. J. Integr. Agric. 2022, 21, 2767–2776. [Google Scholar] [CrossRef]
- Chen, R.; Yan, X.; Cai, M.; Cai, J.; Dai, T.; Liu, Y.; Wu, J. Impact of Germination on the Edible Quality and Nutritional Properties of Brown Rice Noodles. Foods 2024, 13, 2152. [Google Scholar] [CrossRef]
- Kong, L.; Lin, Y.; Liang, J.; Hu, X.; Ashraf, U.; Guo, X.; Bai, S. Dynamic Changes in Vitamin E Biosynthesis during Germination in Brown Rice (Oryza sativa L.). Foods 2022, 11, 3200. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Yang, Y.; Ren, L.; Bian, X.; Liu, X.; Chen, F.; Tan, B.; Fu, Y.; Zhang, X.; Zhang, N. Effects of germination time on the structural, physicochemical and functional properties of brown rice. Int. J. Food Sci. Technol. 2021, 57, 1902–1910. [Google Scholar] [CrossRef]
- He, M.; Guo, T.; Li, D.; Xie, C.; Wang, P.; Yang, R. Effects of roasting on physicochemical characteristics and flavor substances of germinated brown rice. Food Sci. Biotechnol. 2024, 34, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, J.C.; Boue, S.M.; Goufo, P. Health-promoting germinated rice and value-added foods: A comprehensive and systematic review of germination effects on brown rice. Crit. Rev. Food Sci. Nutr. 2023, 63, 11570–11603. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Ma, C.; Bian, X.; Fu, Y.; Zhang, G.; Liu, X.; Zhang, N. Effect of Germination on Mineral Content Changes in Brown Rice (Oryza sativa L.). Biol. Trace Elem. Res. 2024, 203, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Yang, L.; Nie, P.; Zhong, L.; Wu, Y.; Sun, X.; Song, L. Effects of hydrogen-rich water on the nutritional properties, volatile profile and texture of germinated brown rice. Int. J. Food Sci. Technol. 2022, 57, 7666–7680. [Google Scholar] [CrossRef]
- Chao, S.; Mitchell, J.; Prakash, S.; Bhandari, B.; Fukai, S. Effects of variety, early harvest, and germination on pasting properties and cooked grain texture of brown rice. J. Texture Stud. 2022, 53, 503–516. [Google Scholar] [CrossRef]
- Beaulieu, J.C.; Reed, S.S.; Obando-Ulloa, J.M.; Boue, S.M.; Cole, M.R. Green Processing, Germinating and Wet Milling Brown Rice (Oryza sativa) for Beverages: Physicochemical Effects. Foods 2020, 9, 1016. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Yin, Y.; Fang, W.; Yang, Z. Potential of germinated brown rice in beer brewing. J. Cereal Sci. 2023, 114, 103792. [Google Scholar] [CrossRef]
- Pino, A.; Nicosia, F.D.; Agolino, G.; Timpanaro, N.; Barbagallo, I.; Ronsisvalle, S.; Caggia, C.; Randazzo, C.L. Formulation of germinated brown rice fermented products functionalized by probiotics. Innov. Food Sci. Emerg. Technol. 2022, 80, 103076. [Google Scholar] [CrossRef]
- Zhao, R.; Huang, F.; Liu, C.; Asija, V.; Cao, L.; Zhou, M.; Gao, H.; Sun, M.; Weng, X.; Huang, J.; et al. Impact of Germinated Brown Rice and Brown Rice on Metabolism, Inflammation, and Gut Microbiome in High Fat Diet-Induced Insulin Resistant Mice. J. Agric. Food Chem. 2022, 70, 14235–14246. [Google Scholar] [CrossRef]
- Ren, C.; Hong, B.; Zhang, S.; Yuan, D.; Feng, J.; Shan, S.; Zhang, J.; Guan, L.; Zhu, L.; Lu, S. Autoclaving-treated germinated brown rice relieves hyperlipidemia by modulating gut microbiota in humans. Front. Nutr. 2024, 11, 1403200. [Google Scholar] [CrossRef]
- Zhao, F.; Ren, C.; Li, L.; Huang, C.; Zhao, Q.; Zhong, Y.; Hu, Q.; Liao, W.; Xia, H.; Yang, L.; et al. Effects of germinated brown rice and germinated black rice on people with type 2 diabetes mellitus combined with dyslipidaemia. Food Funct. 2024, 15, 6642–6656. [Google Scholar] [CrossRef]
- Shao, Y.; Zhu, H.; Chen, X.; Feng, E.; Chen, C.; Shao, Z.; Li, X.; Liu, L. Associations of ALT, AST and ALT/AST ratio with metabolically unhealthy obesity in the elderly. Front. Nutr. 2025, 12, 1513029. [Google Scholar] [CrossRef] [PubMed]
- Soto-Sánchez, J.; Martínez-Navarro, I.; Mandujano-Lázaro, G.; Rios-Lugo, M.J.; Hernández-Mendoza, H. Serum levels of anti-inflammatory/proinflammatory adipocytokines, and copper levels in overweight and obese women in an adult Mexican population. Hormones 2023, 22, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Pena, A.; Colburn, A.; Nwabichie, E.C.; Sears, D.D.; Lopez, S.V.; Olson, M.; Ayers, S.; Knowler, W.; Shaibi, G. Abstract P100: Changes In Adiponectin But Not Tnfα Predict Changes In Insulin Sensitivity Following Health Behavior Intervention Among Latino Youth. Circulation 2022, 145 (Suppl. S1), AP100. [Google Scholar] [CrossRef]
- Wang, C.; Jiang, T.; Ye, L.; Du, M.; Liu, F.J.B.G. Optimization of the cholesterol gallstone model in C57BL/6 mice and evaluation of Lactobacillus intervention effects. BMC Gastroenterol. 2025, 25, 56. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Calderón, M.C.; Sánchez-Moro, M.D.H.; Rincón, E.O. In vitro Cholesterol Assimilation by Bifidobacterium animalis subsp lactis (BPL1) Probiotic Bacteria Under Intestinal Conditions. Endocr. Metab. Immune Disord.-Drug Targets 2022, 22, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Araie, H.; Hosono, N.; Yamauchi, T. Cellular uptake of CPX-351 by scavenger receptor class B type 1–mediated nonendocytic pathway. Exp. Hematol. 2024, 140, 104651. [Google Scholar] [CrossRef]
- Xiong, L.; Guo, Z.; Wang, H.; Pei, J.; Wu, X.; Yan, P.; Guo, X. The Study on Potential Biomarker in Rat After Withdrawal of Cimaterol Using Untargeted Metabonomics. Chromatographia 2021, 84, 677–686. [Google Scholar] [CrossRef]
- Bao, W.; Cao, C.; Li, S.; Bo, L.; Zhang, M.; Zhao, X.; Liu, Y.; Sun, C. Metabonomic analysis of quercetin against the toxicity of acrylamide in rat urine. Food Funct. 2017, 8, 1204–1214. [Google Scholar] [CrossRef]
- Fang, Q.; Lai, Y.; Zhang, D.; Lei, H.; Wang, F.; Guo, X.; Song, C. Gut microbiota regulation and prebiotic properties of polysaccharides from Oudemansiella raphanipes mushroom. World J. Microbiol. Biotechnol. 2023, 39, 167. [Google Scholar] [CrossRef]
- Ge, Y.-F.; Wei, C.-H.; Wang, W.-H.; Cao, L.-K. The resistant starch from sorghum regulates lipid metabolism in menopausal rats via equol. J. Food Biochem. 2020, 44, e13295. [Google Scholar] [CrossRef]
- Dong, Y.; Dapeng, D.; Juebin, G.; Mingying, C. Alpha-2 Heremans Schmid Glycoprotein (AHSG) promotes the proliferation of bladder cancer cells by regulating the TGF-β signalling pathway. Bioengineered 2022, 13, 14282–14298. [Google Scholar] [CrossRef]
- Zhu, K.; Deng, C.; Du, P.; Liu, T.; Piao, J.; Piao, Y.; Yang, M.; Chen, L. G6PC indicated poor prognosis in cervical cancer and promoted cervical carcinogenesis in vitro and in vivo. Reprod. Biol. Endocrinol. 2022, 20, 50. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.; Wu, J.; Zhang, Y.; Zhuang, W.; Liang, X.F. Role of phosphoenolpyruvate carboxykinase 1 (pck1) in mediating nutrient metabolism in zebrafish. Funct. Integr. Genom. 2023, 23, 67. [Google Scholar] [CrossRef]
- Jin, H.; Han, H.; Song, G.; Oh, H.-J.; Lee, B.-Y. Anti-Obesity Effects of GABA in C57BL/6J Mice with High-Fat Diet-Induced Obesity and 3T3-L1 Adipocytes. Int. J. Mol. Sci. 2024, 25, 995. [Google Scholar] [CrossRef]
- Espes, D.; Liljebäck, H.; Hill, H.; Elksnis, A.; Caballero-Corbalan, J.; Birnir, B.; Carlsson, P.-O. GABA induces a hormonal counter-regulatory response in subjects with long-standing type 1 diabetes. BMJ Open Diabetes Res. Care 2021, 9, e002442. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Devi, S.; Kumar, M.; Dahiya, R.S.; Tarkeshwer. Evaluation of Gamma Amino Butyric Acid (GABA) and Glibenclamide Combination Therapy in Streptozotocin Induced Diabetes. Endocr. Metab. Immune Disord.-Drug Targets 2021, 21, 2005–2016. [Google Scholar] [CrossRef]
- Jiang, X.; Li, Y.; Cao, Z.; Xu, Q.; Zhang, J.; Cao, D.; Chi, X.; Zhang, D. Study on the Mechanism of GABA-Rich Adzuki Bean Regulating Blood Glucose Based on the IRS/PI3K/AKT Pathway. Foods 2024, 13, 2791. [Google Scholar] [CrossRef]
- Lee, S.-J.; Cho, K.H.; Kim, J.C.; Choo, H.J.; Hwang, J.-Y.; Cho, H.C.; Hah, Y.-S. Comparative analysis of anti-obesity effects of green, fermented, and γ-aminobutyric acid teas in a high-fat diet-induced mouse model. Appl. Biol. Chem. 2024, 67, 36. [Google Scholar] [CrossRef]
- Alhamyani, A.; Napit, P.R.; Ali, H.; Ibrahim, M.M.H.; Briski, K.P. Ventrolateral ventromedial hypothalamic nucleus GABA neuron adaptation to recurring Hypoglycemia correlates with up-regulated 5′-AMP-activated protein kinase activity. AIMS Neurosci. 2021, 8, 510–525. [Google Scholar] [CrossRef] [PubMed]
- Lai, M.; Li, N.; Li, Q.; Ge, X.; Huang, Z.; Chen, F.; Liu, B.; Zeng, F. Pueraria lobata Resistant Starch Regulating Lipid Metabolism and Gut Microbiota in High-Fat Diet Mice. Starch-Stärke 2024, 76, 2300123. [Google Scholar] [CrossRef]
- Liu, Y.S.; Zhang, Y.Y.; Xing, T.; Li, J.L.; Wang, X.F.; Zhu, X.D.; Zhang, L.; Gao, F. Glucose and lipid metabolism of broiler chickens fed diets with graded levels of corn resistant starch. Br. Poult. Sci. 2020, 61, 599–607. [Google Scholar] [CrossRef]
- Zeng, X.; Chen, L.; Zheng, B. Extrusion and chlorogenic acid treatment increase the ordered structure and resistant starch levels in rice starch with amelioration of gut lipid metabolism in obese rats. Food Funct. 2024, 15, 5224–5237. [Google Scholar] [CrossRef]
- Zhou, M.; Ma, J.; Kang, M.; Tang, W.; Xia, S.; Yin, J.; Yin, Y. Flavonoids, gut microbiota, and host lipid metabolism. Eng. Life Sci. 2024, 24, 2300065. [Google Scholar] [CrossRef]
- Tan, Z.; Halter, B.; Liu, D.; Gilbert, E.R.; Cline, M.A. Dietary Flavonoids as Modulators of Lipid Metabolism in Poultry. Front. Physiol. 2022, 13, 863860. [Google Scholar] [CrossRef]
- Gilani, S.J.; Bin-Jumah, M.N.; Al-Abbasi, F.A.; Nadeem, M.S.; Imam, S.S.; Alshehri, S.; Ghoneim, M.M.; Afzal, M.; Alzarea, S.I.; Sayyed, N.; et al. Rosinidin Flavonoid Ameliorates Hyperglycemia, Lipid Pathways and Proinflammatory Cytokines in Streptozotocin-Induced Diabetic Rats. Pharmaceutics 2022, 14, 547. [Google Scholar] [CrossRef]
- El-Ouady, F.; Bachir, F.; Eddouks, M. Flavonoids Extracted from Asteriscus graveolens Improve Glucose Metabolism and Lipid Profile in Diabetic Rats. Endocr. Metab. Immune Disord.-Drug Targets 2021, 21, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Kaur, P.; Kaur, H.; Aggarwal, R.; Bains, K.; Mahal, A.K.; Singla, L.D.; Gupta, K. Analysing the Impact of Resistant Starch Formation in Basmati Rice Products: Exploring Associations with Blood Glucose and Lipid Profiles across Various Cooking and Storage Conditions In Vivo. Foods 2024, 13, 1669. [Google Scholar] [CrossRef]
- Hosseini Dastgerdi, A.; Sharifi, M.; Soltani, N. GABA administration improves liver function and insulin resistance in offspring of type 2 diabetic rats. Sci. Rep. 2021, 11, 23155. [Google Scholar] [CrossRef]
- Ge, Y.; Wei, C.; Arumugam, U.R.; Wu, Y.; Cao, L. Quinoa bran insoluble dietary fiber-zinc chelate mediates intestinal flora structure to regulate glucose and lipid metabolism in obese rats. J. Funct. Foods 2023, 108, 105765. [Google Scholar] [CrossRef]
- Zheng, B.; Ao, T.; Zhao, X.; Chen, Y.; Xie, J.; Gao, X.; Liu, L.; Hu, X.; Yu, Q. Comprehensive assessment of the anti-obesity effects of highland barley total, insoluble, and soluble dietary fiber through multi-omics analysis. Food Res. Int. 2024, 189, 114535. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Wang, B.; Li, L.; Zhao, W. Anti-Obesity Effects of Dietary Fibers Extracted from Flaxseed Cake in Diet-Induced Obese Mice. Nutrients 2023, 15, 1718. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wang, J.; Ma, W.; Miao, M.; Sun, G. Effects of Additional Dietary Fiber Supplements on Pregnant Women with Gestational Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Studies. Nutrients 2022, 14, 4626. [Google Scholar] [CrossRef]
- Ishikawa, A.; Oka, H.; Hiemori, M.; Yamashita, H.; Kimoto, M.; Kawasaki, H.; Tsuji, H. Development of a Method for the Determination of gamma-Aminobutyric Acid in Foodstuffs. J. Nutr. Sci. Vitaminol. 2009, 55, 292–295. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.L.; Sun, Z.H.; Shi, Y.C. Improved in vitro assay of resistant starch in cross-linked phosphorylated starch. Carbohydr. Polym. 2019, 210, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wu, Y.; Long, P.P.; Ho, C.T.; Wang, Y.J.; Kan, Z.P.; Cao, L.T.; Zhang, L.; Wan, X.C. LC-MS-Based Metabolomics Reveals the Chemical Changes of Polyphenols during High-Temperature Roasting of Large-Leaf Yellow Tea. J. Agric. Food Chem. 2019, 67, 5405–5412. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wu, Z.; Guo, S.; Meng, X.; Chang, X. Polyphenol-rich extract from wild Lonicera caerulea berry reduces cholesterol accumulation by mediating the expression of hepatic miR-33 and miR-122, HMGCR, and CYP7A1 in rats. J. Funct. Foods 2018, 40, 648–658. [Google Scholar] [CrossRef]

| GABA, mg/100g | Resistant Starch, % | Insoluble Dietary Fiber, % | Flavonoids, mg/100 g | |
|---|---|---|---|---|
| Brown rice | 2.38 ± 0.06 | 0.52 ± 0.02 | 0.78 ± 0.02 | 4.56 ± 0.08 |
| Germinated brown rice | 34.68 ± 0.08 | 2.89 ± 0.04 | 0.96 ± 0.03 | 11.44 ± 0.10 |
| Reagents Name | Company | Location |
|---|---|---|
| Aspartate aminotransferase (AST) test kit | Shanghai Enzyme Linked Biotechnology Co., Ltd. | Shanghai China |
| Alanine aminotransferase (ALT) test kit | ||
| Total Bile Acid (TBA) test kit | ||
| Apolipoprotein A1 (apo-A1) test kit | ||
| Apolipoprotein B (apo-B) test kit | ||
| Glycated Hemoglobin (GHb) test kit | ||
| Leptin (LEP) test kit | ||
| Lipoprotein esterase (LPL) test kit | ||
| Liver lipase (HL) test kit | ||
| Egg Phosphocholesterol Acyltransferase (LCAT) test kit | ||
| Adiponectin (ADPN) test kit | ||
| Endotoxin (ET) test kit | ||
| Interleukin-6 (IL-6) test kit | ||
| Tumor necrosis factor alpha (TNF-α) test kit | ||
| Total cholesterol (TC) test kit | Nanjing Jiancheng Technology Co., Ltd. | Nanjing China |
| Triglyceride (TG) test kit | ||
| Low density lipoprotein cholesterol (LDL-C) test kit | ||
| High density lipoprotein cholesterol (HDL-C) test kit |
| Gene | Primer Sequence 5′-3′ | Length (bp) | |
|---|---|---|---|
| Lipid metabolism-related genes | GAPDH | F: TCTCTGCTCCTCCCTGTTCTA | 121 |
| R: GGTAACCAGGCGTCCGATAC | |||
| HMGCR | F: ACTGAAACACGGGCATTGGGTT | 195 | |
| R: AACACGGCACGGAAAGAACCAT | |||
| SREBP2 | F: GAGGCGGACAACACACAATA | 370 | |
| R: CGGCTCAGAGTCAATGGAATAG | |||
| PPARα | F: TCTGAACATTGGCGTTCGCA | 98 | |
| R: TCCCTCAAGGGGACAACCAG | |||
| LXRA | F: CTGCAACGGAGTTGTGGAAG | 295 | |
| R: TCGCAGCTCAGCACATTGTA | |||
| Glucose metabolism-related genes | GAPDH | F: GGTGGACCTCATGGCCTACAT | 84 |
| R: CTCTCTTGCTCTCAGTATCCTTGCT | |||
| PCK1 | F: GGGTGGAAAGTTGAATGTGTGGGT | 139 | |
| R: TGGCGTTCGGATTTGTCTTC | |||
| G6PC1 | F: AGCCTCTTCAAAAACCTGGGGA | 110 | |
| R: AAACGGAATGGGAGCGACTT | |||
| AHSG | F: ACATCTTCTTCAGGGATTCAGGCA | 116 | |
| R: AGGTTGTCTCCAGCGTGTCAAT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, C.; Lu, S.; Shan, S.; Zhang, S.; Hong, B.; Yuan, D.; Zhang, J.; Gao, S.; Liu, Q.; Fan, X. Nutrient-Enriched Germinated Brown Rice Alters the Intestinal Ecological Network by Regulating Lipid Metabolism in Rats. Int. J. Mol. Sci. 2025, 26, 7693. https://doi.org/10.3390/ijms26167693
Ren C, Lu S, Shan S, Zhang S, Hong B, Yuan D, Zhang J, Gao S, Liu Q, Fan X. Nutrient-Enriched Germinated Brown Rice Alters the Intestinal Ecological Network by Regulating Lipid Metabolism in Rats. International Journal of Molecular Sciences. 2025; 26(16):7693. https://doi.org/10.3390/ijms26167693
Chicago/Turabian StyleRen, Chuanying, Shuwen Lu, Shan Shan, Shan Zhang, Bin Hong, Di Yuan, Jingyi Zhang, Shiwei Gao, Qing Liu, and Xiaobing Fan. 2025. "Nutrient-Enriched Germinated Brown Rice Alters the Intestinal Ecological Network by Regulating Lipid Metabolism in Rats" International Journal of Molecular Sciences 26, no. 16: 7693. https://doi.org/10.3390/ijms26167693
APA StyleRen, C., Lu, S., Shan, S., Zhang, S., Hong, B., Yuan, D., Zhang, J., Gao, S., Liu, Q., & Fan, X. (2025). Nutrient-Enriched Germinated Brown Rice Alters the Intestinal Ecological Network by Regulating Lipid Metabolism in Rats. International Journal of Molecular Sciences, 26(16), 7693. https://doi.org/10.3390/ijms26167693






