Systematic Review on Working Mechanisms of Signaling Pathways in Fibrosis During Shockwave Therapy
Abstract
1. Introduction
2. Methods
2.1. Eligibility Criteria
2.2. Information Sources
2.3. Search Strategy
2.4. Selection Process
2.5. Data Items and Collection
2.6. Risk of Bias in Individual Studies
3. Results and Discussion
3.1. Study Selection
3.2. Study Characteristics
3.3. Risk of Bias in Studies
3.4. Results of Individual Studies
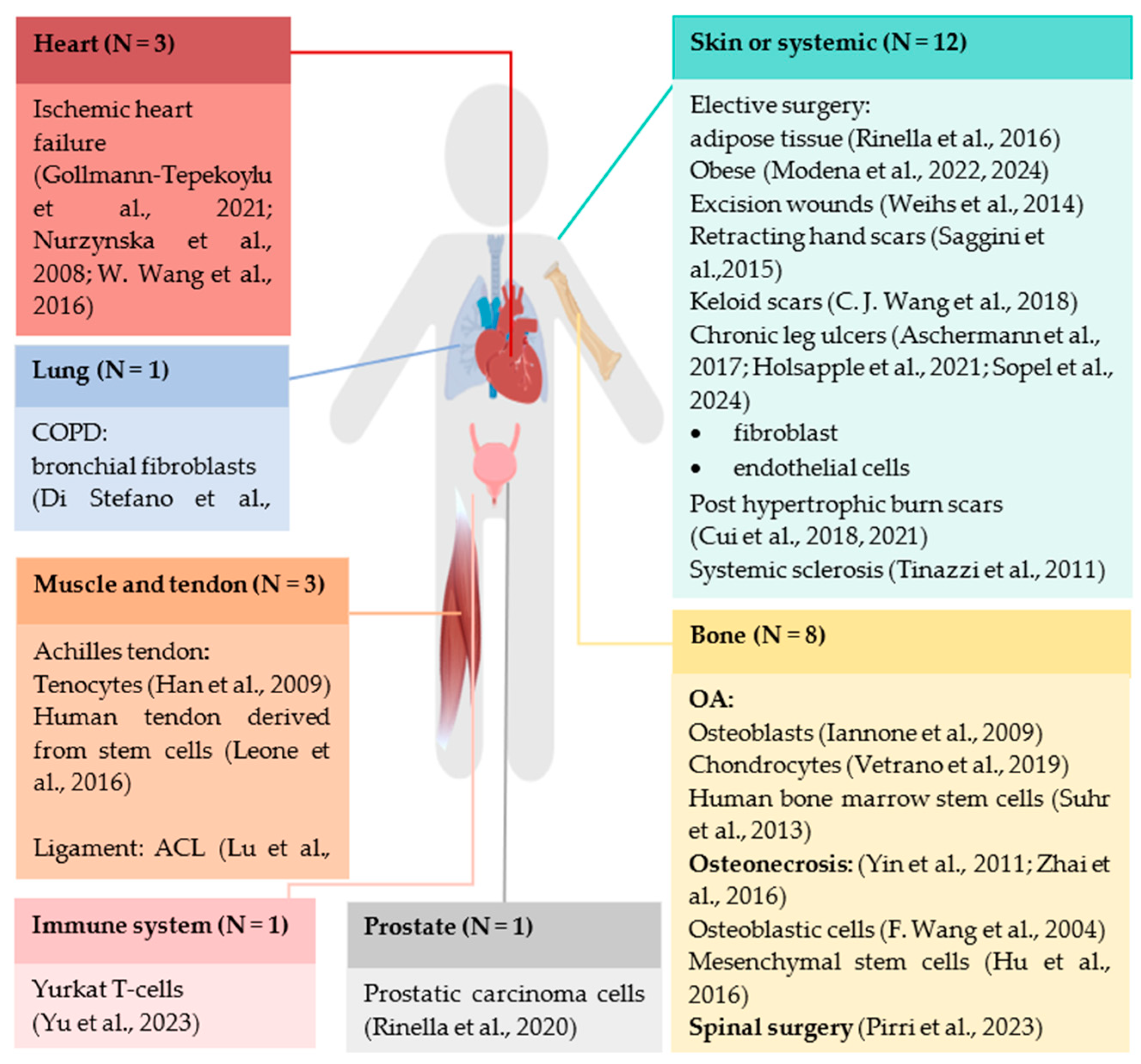
3.4.1. Study Population
3.4.2. Treatment Modalities of Shockwave Therapy
- Inflammation phaseDuring inflammation, treatment modalities in terms of EFD ranged between 0.0024 and 0.30 mJ/mm2 [9,30,31,32]. Only one study described higher energy levels of 180 mJ, but this was in preoperative tissue [17]. This study described higher energy levels and their beneficial effects on natural killer (NK), T, B lymphocytes and proinflammatory macrophages. The lowest EFD (0.0024–0.09 mJ/mm2) and 200 pulses showed positive effects on chemokine ligands 1, 2 and 3 (CXCL1, 2 and 3) [21];On macrophages (M1 and M2) and their associated cytokines, interleukin 1 and 6 (IL-6, IL-1) and tumor necrosis factor α (TNF-α), chemokines and VEGF, studies used EFD between 0.11–0.14 mJ/mm2 during the inflammatory phase. In bronchial fibroblasts, a higher EFD of 0.30 mJ/mm2 was used with the same pulse frequency of 500 was used [30];
- Proliferation phaseDuring the proliferative phase, the EFD reported in the included studies also varied generally from 0.0024 to 0.30, but other cellular changes have been reported. As mentioned above, Wang et al. used low EFDs between 0.0024 and 0.09 with 200 pulses and found effects on mitogen-activated protein kinase 9 (MAPK9). More or less similar results were found by Weihs et al., using EFDs of 0.030–0.19, 3 Hz and 10–300 pulses, also finding an effect on Akt and extracellular-regulated kinase (Akt/Erk) pathway besides p38 MAPK [8]. In another study, an EFD of 0.25 mJ/mm2, 3000 pulses and 3 Hz was used on the Erk pathway. Proliferating cell nuclear antigen (PCNA) and fibronectin were positively affected by an EFD of 0.11 mJ/mm2 [31].In 4 other studies, EFDs between 0.15 to 0.18 and pulses between 250 and 5000 were used. Positive effects were found on cell viability, cell proliferation and cell migration through changes in collagen-I α, TGFβ and VEGF [38]. Similar results were found for differentiation markers (alkaline phosphatase, core binding factorα (Cbfα): glyceraldehyde-3-phosphate dehydrogenase (GADPH), osteocalcin (OCN) β-actin, peroxisome proliferator-activated receptor gamma (PPAR)). In the expression of T cells, Yu et al. found a higher expression of IL-2 with an EFD of 0.18 mJ/mm2 [7]. Three studies demonstrated EFD dependence in cellular changes in p21, p27 and Notch 21, finding significantly higher only by 0.2–0.3 mJ/mm2 EFD [5]. Another study showed that Ki67 varies with EFD. It was only at 0.2 mJ/mm2 that they found a significant increase [37]. In the meantime, Leone et al. found a significant increase in Ki67, but they did not specify the EFD. However, no significant change in Ki67 was found in another study [29];
- Activation and differentiation phaseIn keratinocytes, two studies by Cui et al. used different EFDs and found different outcomes depending on EFD. They were interested in keratin 6.17 and keratin 1.10, and the results showed a significantly higher amount of keratin 6.17 by an EFD 0.2–0.3 and 4 Hz, respectively, while keratin 1 and 10 were not significantly changed at 0.10 mJ/mm2, but at 0.20–0.30 they were significantly higher [4]. Zhai et al. found higher Cbfα1 gene expression at 0.16 mJ/mm2 EFD [26]. An EFD of 0.16 mJ/mm2 also significantly decreased miR138 [21];
- ApoptosisDuring apoptosis, different studies indicate EFD between 0.11 and 0.30, 4 to 5 Hz and 150 to 500 to 1000 pulses [5,9,37]. Most of the apoptosis was described in the caspase pathway, and a significant decrease of 0.2 mJ/mm2 and an increase of 0.3 mJ/mm2 was found [37]. Similar results were found for the proapoptotic factor Bcl-2 associated X-protein (Bax) and the anti-apoptotic factor B-cell lymphoma (Bcl) [5].
3.4.3. Assessment Protocol
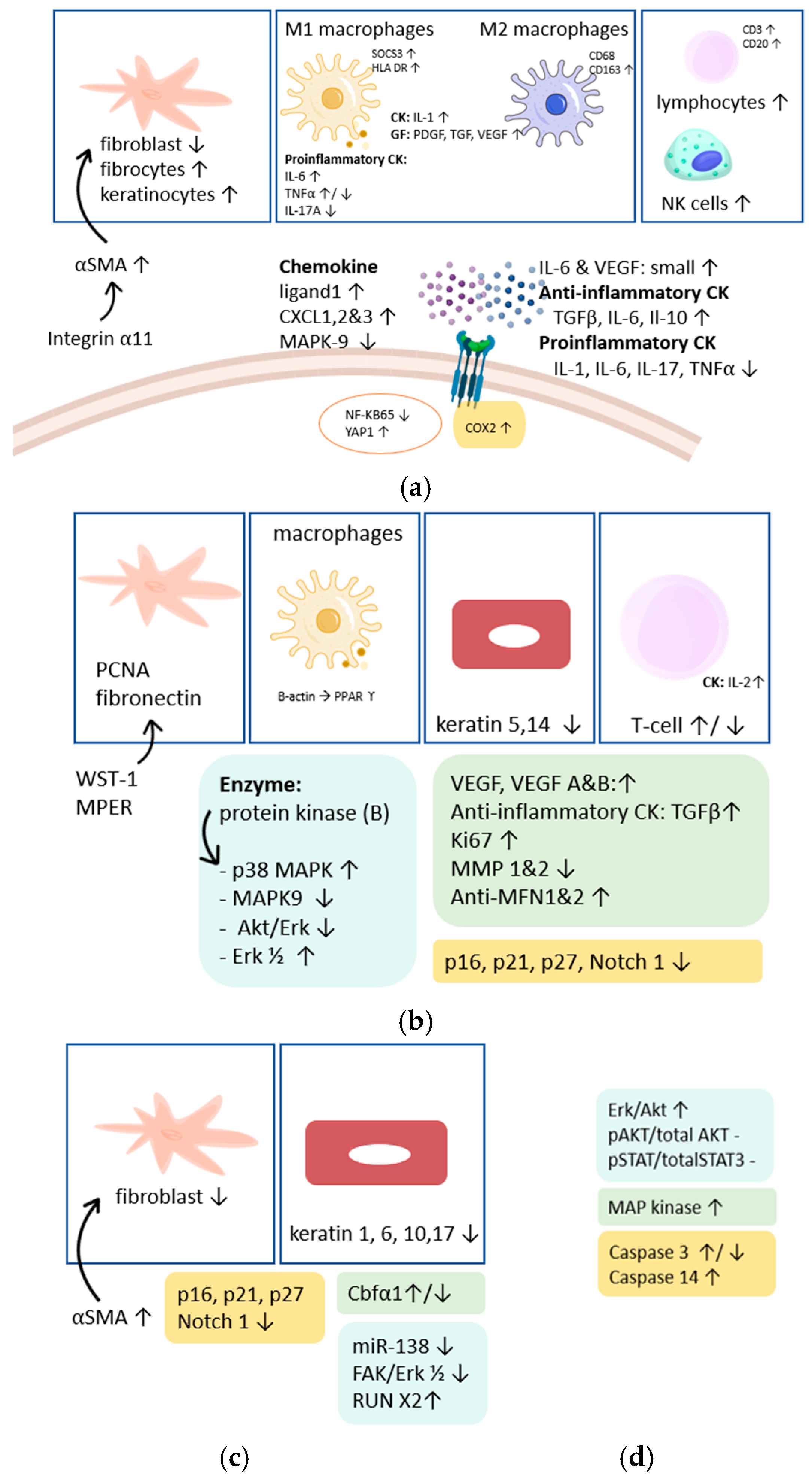
3.4.4. Underlying Mechanisms Caused by SWT
Underlying Mechanisms Caused by SWT in Different Phases of Fibrosis
Effect of Shockwave Therapy on Inflammation
Shockwave Therapy Increases Angiogenesis
Effect of Shockwave Therapy on Proliferation
Effect of Shockwave Therapy on Activation/Differentiation Markers
Effect of Shockwave Therapy on Apoptosis
3.5. Discussion
3.5.1. General Aim
3.5.2. Most Important Findings
3.5.3. Limitations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Zhao, J.C.; Zhang, B.R.; Shi, K.; Wang, J.; Yu, Q.H.; Yu, J.A. Lower energy radial shock wave therapy improves characteristics of hypertrophic scar in a rabbit ear model. Exp. Ther. Med. 2018, 15, 933–939. [Google Scholar] [CrossRef] [PubMed]
- Antar, S.A.; Ashour, N.A.; Marawan, M.E.; Al-Karmalawy, A.A. Fibrosis: Types, Effects, Markers, Mechanisms for Disease Progression, and Its Relation with Oxidative Stress, Immunity, and Inflammation. Int. J. Mol. Sci. 2023, 24, 4004. [Google Scholar] [CrossRef] [PubMed]
- Aschermann, I.; Noor, S.; Venturelli, S.; Sinnberg, T.; Busch, C.; Mnich, C.D. Extracorporal Shock Waves Activate Migration, Proliferation and Inflammatory Pathways in Fibroblasts and Keratinocytes, and Improve Wound Healing in an Open-Label, Single-Arm Study in Patients with Therapy-Refractory Chronic Leg Ulcers. Cell. Physiol. Biochem. 2017, 41, 890–906. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.S.; Joo, S.Y.; Cho, Y.S.; Park, J.H.; Ro, Y.M.; Kim, J.B.; Seo, C.H. Effect of extracorporeal shock wave therapy on keratinocytes derived from human hypertrophic scars. Sci. Rep. 2021, 11, 17296. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.S.; Hong, A.; Kim, J.B.; Yu, J.H.; Cho, Y.S.; Joo, S.Y.; Seo, C.H. Extracorporeal shock wave therapy alters the expression of fibrosis-related molecules in fibroblast derived from human hypertrophic scar. Int. J. Mol. Sci. 2018, 19, 124. [Google Scholar] [CrossRef]
- Gollmann-Tepekoylu, C.; Polzl, L.; Graber, M.; Hirsch, J.; Nagele, F.; Lobenwein, D.; Hess, M.W.; Blumer, M.J.; Kirchmair, E.; Zipperle, J.; et al. miR-19a-3p containing exosomes improve function of ischaemicmyocardiumupon shock wave therapy. Cardiovasc. Res. 2021, 116, 1226–1236. [Google Scholar] [CrossRef]
- Yu, T.; Junger, W.G.; Yuan, C.; Jin, A.; Zhao, Y.; Zheng, X.; Zeng, Y.; Liu, J. Shockwaves increase T-cell proliferation and IL-2 expression through ATP release, P2X7 receptors, and FAK activation. Am. J. Physiol. -Cell Physiol. 2023, 298, C457–C464. [Google Scholar] [CrossRef]
- Weihs, A.M.; Fuchs, C.; Teuschl, A.H.; Hartinger, J.; Slezak, P.; Mittermayr, R.; Redl, H.; Junger, W.G.; Sitte, H.H.; Rünzler, D. Shock wave treatment enhances cell proliferation and improves wound healing by ATP release-coupled Extracellular signal-regulated Kinase (ERK) activation. J. Biol. Chem. 2014, 289, 27090–27104. [Google Scholar] [CrossRef]
- Holsapple, J.S.; Cooper, B.; Berry, S.H.; Staniszewska, A.; Dickson, B.M.; Taylor, J.A.; Bachoo, P.; Wilson, H.M. Low intensity shockwave treatment modulates macrophage functions beneficial to healing chronic wounds. Int. J. Mol. Sci. 2021, 22, 7844. [Google Scholar] [CrossRef] [PubMed]
- Sopel, M.; Kuberka, I.; Szczuka, I.; Taradaj, J.; Rosińczuk, J.; Dymarek, R. Can Shockwave Treatment Elicit a Molecular Response to Enhance Clinical Outcomes in Pressure Ulcers? The SHOck Waves in wouNds Project. Biomedicines 2024, 12, 359. [Google Scholar] [CrossRef]
- Rinella, L.; Marano, F.; Berta, L.; Bosco, O.; Fraccalvieri, M.; Fortunati, N.; Frairia, R.; Catalano, M.G. Extracorporeal shock waves modulate myofibroblast differentiation of adipose-derived stem cells. Wound Repair Regen. 2016, 24, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Modena, D.A.O.; Ferro, A.P.; Cazzo, E.; de Oliveira Guirro, E.C.; Chaim, E.A. Effect of superficial adipose tissue mitochondrial and cellular functionality induced by extracorporeal shock wave therapy (ESWT). Lasers Med. Sci. 2024, 39, 58. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.; Roy, S.; Ghosh, D.; Nandi, S.K. Role of animal models in biomedical research: A review. Lab. Anim. Res. 2022, 38, 18. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Waqar, K.; Shafique, A.; Irfan, R.; Gul, A. In vitro and in vivo animal models: The engineering towards understanding human diseases and therapeutic interventions. In Omics Technologies and Bio-Engineering: Towards Improving Quality of Life; Elsevier Inc.: Amsterdam, The Netherlands, 2018; Volume 1, pp. 431–448. [Google Scholar] [CrossRef]
- Ghallab, A.; Bolt, H.M. In vitro systems: Current limitations and future perspectives. Arch. Toxicol. 2014, 88, 2085–2087. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Modena, D.A.O.; Soares, C.D.; Candido, E.C.; Chaim, F.D.M.; Cazzo, E.; Chaim, E.A. Effect of extracorporeal shock waves on inflammation and angiogenesis of integumentary tissue in obese individuals: Stimulating repair and regeneration. Lasers Med. Sci. 2022, 37, 1289–1297. [Google Scholar] [CrossRef]
- Wang, C.J.; Ko, J.Y.; Chou, W.Y.; Cheng, J.H.; Kuo, Y.R. Extracorporeal shockwave therapy for treatment of keloid scars. Wound Repair Regen. 2018, 26, 69–76. [Google Scholar] [CrossRef]
- Saggini, R.; Saggini, A.; Spagnoli, A.M.; Dodaj, I.; Cigna, E.; Maruccia, M.; Soda, G.; Bellomo, R.G.; Scuderi, N. Etracorporeal shock wave therapy: An emerging treatment modality for retracting scars of the hands. Ultrasound Med. Biol. 2015, 42, 185–195. [Google Scholar] [CrossRef]
- Tinazzi, E.; Amelio, E.; Marangoni, E.; Guerra, C.; Puccetti, A.; Michele, O.; Sara, C.; Elisabetta, S.; Montagnana, M.; Adani, R.; et al. Effects of shock wave therapy in the skin of patients with progressive systemic sclerosis: A pilot study. Rheumatol. Int. 2011, 31, 651–656. [Google Scholar] [CrossRef]
- Hu, J.; Liao, H.; Ma, Z.; Chen, H.; Huang, Z.; Zhang, Y.; Yu, M.; Chen, Y.; Xu, J. Focal Adhesion Kinase Signaling Mediated the Enhancement of Osteogenesis of Human Mesenchymal Stem Cells Induced by Extracorporeal Shockwave. Sci. Rep. 2016, 6, 20875. [Google Scholar] [CrossRef]
- Chen, Y.T.; Yang, C.C.; Sung, P.H.; Lin, K.C.; Chiang, J.Y.; Huang, C.R.; Huang, K.H.; Chuang, F.C.; Chu, Y.C.; Huang, E.Y.; et al. Long-term effect of extracorporeal shock wave therapy on attenuating radiation-induced chronic cystitis in rat. Am. J. Transl. Res. 2020, 12, 999–1015. [Google Scholar] [PubMed]
- Iannone, F.; Moretti, B.; Notarnicola, A.; Moretti, L.; Patella, S.; Patella, V.; Lapadula, G. Extracorporeal shock waves increase interleukin-10 expression by human osteoarthritic and healthy osteoblasts in vitro. Clin. Exp. Rheumatol. 2009, 27, 794–799. [Google Scholar]
- Vetrano, M.; Ranieri, D.; Nanni, M.; Pavan, A.; Malisan, F.; Vulpiani, M.C.; Id, V.V. Hyaluronic Acid (HA), Platelet-Rich Plasm and Extracorporeal Shock Wave Therapy (ESWT) promote human chondrocyte regeneration in vitro and ESWT-mediated increase of CD44 expression enhances their susceptibility to HA treatment. PLoS ONE 2019, 14, e0218740. [Google Scholar] [CrossRef]
- Yin, T.C.; Wang, C.J.; Yang, K.D.; Wang, F.S.; Sun, Y.C. Shockwaves enhance the osteogenetic gene expression in marrow stromal cells from hips with osteonecrosis. Chang. Gung Med. J. 2011, 34, 367–374. [Google Scholar]
- Zhai, L.; Sun, N.; Zhang, B.; Liu, S.T.; Zhao, Z.; Jin, H.C.; Ma, X.L.; Xing, G.Y. Effects of Focused Extracorporeal Shock Waves on Bone Marrow Mesenchymal Stem Cells in Patients with Avascular Necrosis of the Femoral Head. Ultrasound Med. Biol. 2016, 42, 753–762. [Google Scholar] [CrossRef]
- Pirri, C.; Caroccia, B.; Angelini, A.; Piazza, M.; Petrelli, L.; Caputo, I.; Montemurro, C.; Ruggieri, P.; De Caro, R.; Stecco, C. A New Player in the Mechanobiology of Deep Fascia: Yes-Associated Protein (YAP). Int. J. Mol. Sci. 2023, 24, 15389. [Google Scholar] [CrossRef]
- Han, S.H.; Lee, J.W.; Guyton, G.P.; Parks, B.G.; Courneya, J.P.; Schon, L.C. Effect of extracorporeal shock wave therapy on cultured tenocytes. Foot Ankle Int. 2009, 30, 93–98. [Google Scholar] [CrossRef]
- Leone, L.; Raffa, S.; Vetrano, M.; Ranieri, D.; Malisan, F.; Scrofani, C.; Vulpiani, M.C.; Ferretti, A.; Torrisi, M.R.; Visco, V. Extracorporeal Shock Wave Treatment (ESWT) enhances the in vitro-induced differentiation of human tendon-derived stem/progenitor cells (hTSPCs). Oncotarget 2016, 7, 6410–6423. [Google Scholar] [CrossRef]
- Di Stefano, A.; Frairia, R.; Ricciardolo, F.L.M.; Gnemmi, I.; Marino Gammazza, A.; Piraino, A.; Cappello, F.; Balbi, B.; Catalano, M.G. Extracorporeal Shock Waves Increase Markers of Cellular Proliferation in Bronchial Epithelium and in Primary Bronchial Fibroblasts of COPD Patients. Can. Respir. J. 2020, 2020, 1524716. [Google Scholar] [CrossRef]
- Wang, W.; Liu, H.; Song, M.; Fang, W.; Yuan, F. Clinical effect of cardiac shock wave therapy on myocardial ischemia in patients with ischemic heart failure. J. Cardiovasc. Pharmacol. Ther. 2016, 21, 381–387. [Google Scholar] [CrossRef]
- Nurzynska, D.; Di Meglio, F.; Castaldo, C.; Arcucci, A.; Marlinghaus, E.; Russo, S.; Corrado, B.; de Santo, L.; Baldascino, F.; Cotrufo, M.; et al. Shock Waves Activate In Vitro Cultured Progenitors and Precursors Of Cardiac Cell Lineages from the Human Heart. Ultrasound Med. Biol. 2008, 34, 334–342. [Google Scholar] [CrossRef]
- Rinella, L.; Pizzo, B.; Frairia, R.; Delsedime, L.; Calleris, G.; Gontero, P.; Zunino, V.; Fortunati, N.; Arvat, E.; Catalano, M.G. Modulating tumor reactive stroma by extracorporeal shock waves to control prostate cancer progression. Prostate 2020, 80, 1087–1096. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savović, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Sterne, J.A.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef]
- Cuello-Garcia, C.A.; Santesso, N.; Morgan, R.L.; Verbeek, J.; Thayer, K.; Ansari, M.T.; Meerpohl, J.; Schwingshackl, L.; Katikireddi, S.V.; Brozek, J.L.; et al. GRADE guidance 24 optimizing the integration of randomized and non-randomized studies of interventions in evidence syntheses and health guidelines. J. Clin. Epidemiol. 2022, 142, 200–208. [Google Scholar] [CrossRef]
- Suhr, F.; Delhasse, Y.; Bungartz, G.; Schmidt, A.; Pfannkuche, K.; Bloch, W. Cell biological effects of mechanical stimulations generated by focused extracorporeal shock wave applications on cultured human bone marrow stromal cells. Stem Cell Res. 2013, 11, 951–964. [Google Scholar] [CrossRef]
- Lu, C.C.; Chou, S.H.; Shen, P.C.; Chou, P.H.; Ho, M.L.; Tien, Y.C. Extracorporeal shock wave promotes activation of anterior cruciate ligament remnant cells and their paracrine regulation of bone marrow stromal cells’ proliferation, migration, collagen synthesis, and differentiation. Bone Jt. Res. 2020, 9, 457–467. [Google Scholar] [CrossRef]
- Wang, F.; Wang, C.; Chen, Y.; Chang, P.; Huang, Y.; Sun, Y.; Huang, H.; Yang, Y.; Yang, K.D.; Biol, K.D.J. Ras Induction of Superoxide Activates ERK-dependent Angiogenic Transcription Factor HIF-1α and VEGF-A Expression in Shock Wave-stimulated Osteoblasts. J. Biol. Chem. 2004, 279, 10331–10337. [Google Scholar] [CrossRef]
- Koh, S.H.; Park, H.H. Neurogenesis in Stroke Recovery. Transl. Stroke Res. 2017, 8, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.J.; Wang, C.J.; Yang, K.D.; Kuo, Y.R.; Huang, H.C.; Huang, Y.T.; Sun, Y.C.; Wang, F.S. Extracorporeal shock waves promote healing of collagenase-induced Achilles tendinitis and increase TGF-β1 and IGF-I expression. J. Orthop. Res. 2004, 22, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Q.; Shen, T.; Wang, Q.; Yu, X.; Jia, N.; He, Q. Cardiac shock wave therapy protects cardiomyocytes from hypoxia-induced injury by modulating miR-210. Mol. Med. Rep. 2020, 21, 631–640. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.L.; Lu, J.H.; Chueh, K.S.; Juan, T.J.; Wu, B.N.; Chuang, S.M.; Lee, Y.C.; Shen, M.C.; Long, C.Y.; Juan, Y.S. Low-intensity extracorporeal shock wave therapy promotes bladder regeneration and improves overactive bladder induced by ovarian hormone deficiency from rat animal model to human clinical trial. Int. J. Mol. Sci. 2021, 22, 9296. [Google Scholar] [CrossRef]
- Lei, P.P.; Tao, S.M.; Shuai, Q.; Bao, Y.X.; Wang, S.W.; Qu, Y.Q.; Wang, D.H. Extracorporeal cardiac shock wave therapy ameliorates myocardial fibrosis by decreasing the amount of fibrocytes after acute myocardial infarction in pigs. Coron. Artery Dis. 2013, 24, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.J.; Wurtz, T.; Wang, C.J.; Kuo, Y.R.; Yang, K.D.; Huang, H.C.; Wang, F.S. Recruitment of mesenchymal stem cells and expression of TGF-β1 and VEGF in the early stage of shock wave-promoted bone regeneration of segmental defect in rats. J. Orthop. Res. 2004, 22, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.C.; Zhang, B.R.; Hong, L.; Shi, K.; Wu, W.W.; Yu, J.A. Extracorporeal shock wave therapy with low-energy flux density inhibits hypertrophic scar formation in an animal model. Int. J. Mol. Med. 2018, 41, 1931–1938. [Google Scholar] [CrossRef]
- Bonella, F.; Spagnolo, P.; Ryerson, C. Current and Future Treatment Landscape for Idiopathic Pulmonary Fibrosis. Drugs 2023, 83, 1581–1593. [Google Scholar] [CrossRef]
- Zhao, M.; Wang, L.; Wang, M.; Zhou, S.; Lu, Y.; Cui, H.; Racanelli, A.C.; Zhang, L.; Ye, T.; Ding, B.; et al. Targeting fibrosis, mechanisms and cilinical trials. Signal Transduct. Target. Ther. 2022, 7, 206. [Google Scholar] [CrossRef]
- Ku, J.C.; Raiten, J.; Li, Y. Understanding fibrosis: Mechanisms, clinical implications, current therapies, and prospects for future interventions. Biomed. Eng. Adv. 2024, 7, 100118. [Google Scholar] [CrossRef]
- d’Agostino, M.C.; Craig, K.; Tibalt, E.; Respizzi, S. Shock wave as biological therapeutic tool: From mechanical stimulation to recovery and healing, through mechanotransduction. Int. J. Surg. 2015, 24, 147–153. [Google Scholar] [CrossRef]
- Noguchi, S.; Saito, A.; Nagase, T. YAP/TAZ signaling as a molecular link between fibrosis and cancer. Int. J. Mol. Sci. 2018, 19, 3674. [Google Scholar] [CrossRef]
- Wang, D.; Zhao, Y.; Zhou, Y.; Yang, S.; Xiao, X.; Feng, L. Angiogenesis—An Emerging Role in Organ Fibrosis. Int. J. Mol. Sci. 2023, 24, 14123. [Google Scholar] [CrossRef]
- Shebl, F.M.; Pinto, L.A.; García-Piñeres, A.; Lempicki, R.; Williams, M.; Harro, C.; Hildesheim, A. Comparison of mRNA and protein measures of cytokines following vaccination with human papillomavirus-16 L1 virus-like particles. Cancer Epidemiol. Biomarkers Prev. 2010, 19, 978–981. [Google Scholar] [CrossRef]
- Prabahar, A.; Zamora, R.; Barclay, D.; Yin, J.; Ramamoorthy, M.; Bagheri, A.; Johnson, S.A.; Badylak, S.; Vodovotz, Y.; Jiang, P. Unraveling the complex relationship between mRNA and protein abundances: A machine learning-based approach for imputing protein levels from RNA-seq data. NAR Genom. Bioinform. 2024, 6, lqae019. [Google Scholar] [CrossRef]
- Koussounadis, A.; Langdon, S.P.; Um, I.H.; Harrison, D.J.; Smith, V.A. Relationship between differentially expressed mRNA and mRNA-protein correlations in a xenograft model system. Sci. Rep. 2015, 5, 10775. [Google Scholar] [CrossRef]
- Cheng, Z.; Teo, G.; Krueger, S.; Rock, T.M.; Koh, H.W.; Choi, H.; Vogel, C. Differential dynamics of the mammalian mRNA and protein expression response to misfolding stress. Mol. Syst. Biol. 2016, 12, 855. [Google Scholar] [CrossRef]
- Rola, P.; Włodarczak, A.; Barycki, M.; Doroszko, A. Use of the Shock Wave Therapy in Basic Research and Clinical Applications—From Bench to Bedsite. Biomedicines 2022, 10, 568. [Google Scholar] [CrossRef]
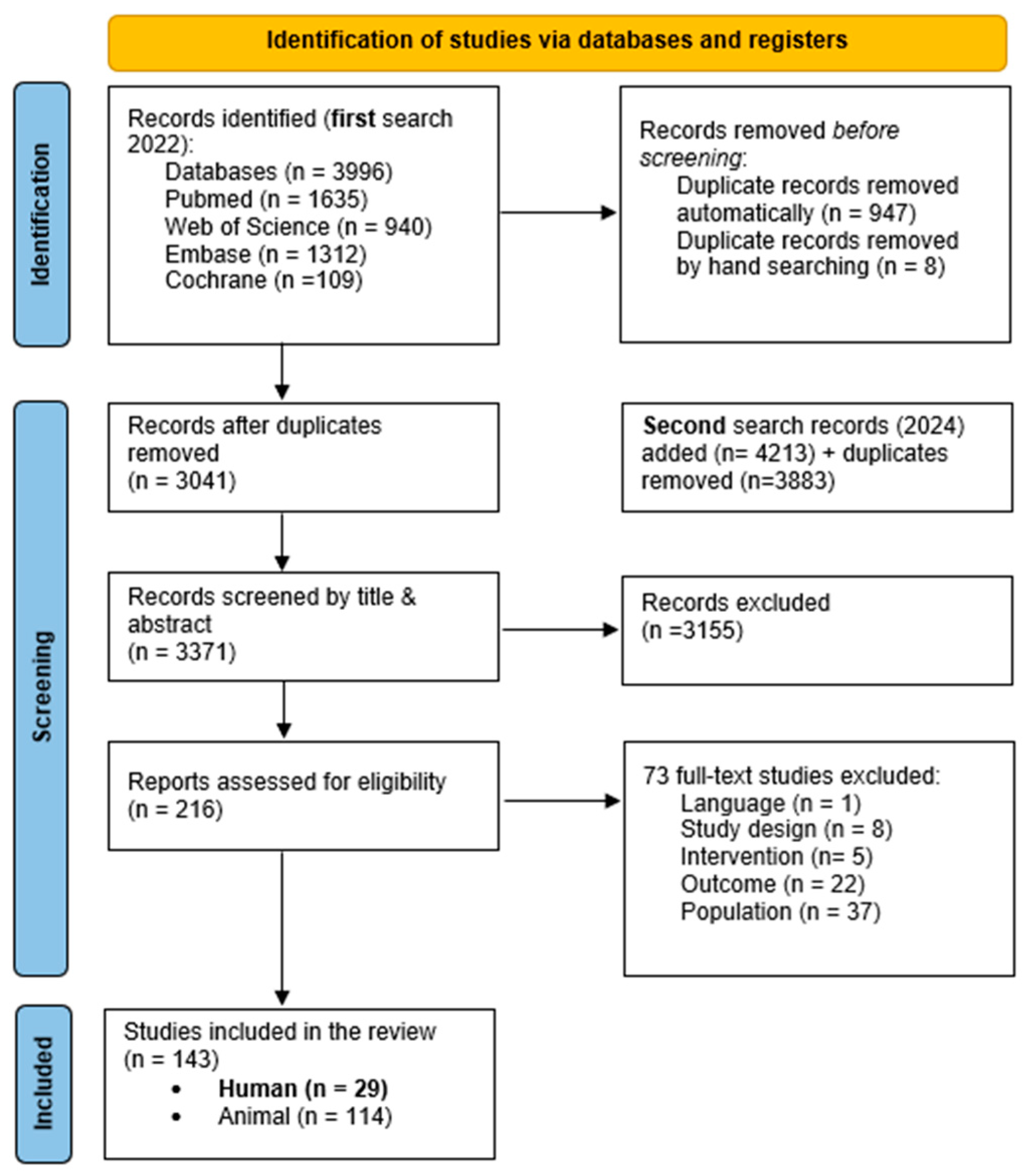

| Inclusion | Exclusion | |
|---|---|---|
| P | Human subjects Animals In vitro, in vivo, ex vivo analysis Fibrosis | No fibrosis, no tissue damage in human or animal subjects Healthy tissue |
| I | Any form of shockwave | No shockwave therapy Effects of other therapy next to shockwave therapy |
| C | / | / |
| O | Cells and molecules Cellular and molecular pathways Mechanotransduction Mechanisms driving fibrogenesis mRNA, DNA | Not underlying mechanism, all ‘visible/external changes’ due to shock wave therapy |
| Study design | Other study designs | Systematic reviews, Meta-analysis |
| Time | / | / |
| Language | Articles written in English, Dutch, German, French | Articles written in any other language |
| Population |
| (Fibrosis[MeSH] OR Fibrosis[Tiab] OR pulmonary fibrosis[MeSH] OR Pulmonary fibrosis[Tiab] OR oral submucous fibrosis[MeSH] OR oral submucous fibrosis[Tiab] OR endomyocardial fibrosis[MeSH] OR endomyocardial fibrosis[Tiab] OR Cystic fibrosis[MeSH] OR cystic fibrosis[Tiab] OR connective tissue cells[MeSH Terms]OR connective tissue cells[Tiab] OR skin[MeSH Terms] OR cutaneous tissue[Tiab] OR skin[Tiab] OR subcutaneous tissue[MeSH Terms] OR subcutaneous tissue[Tiab] OR connective tissue[MeSH] OR connective tissue[Tiab] OR fibroblasts[MeSH] OR fibroblasts[Tiab] OR myofibroblasts[MeSH] OR myofibroblasts[Tiab] OR tissue[Tiab] OR tissues[MeSH]) |
| Intervention |
| (“extracorporeal shockwave therapy”[MeSH] OR extracorporeal shock wave*[Tiab] OR extracorporeal shockwave*[Tiab] OR “Lithotripsy”[MeSH] OR lithotripsy[Tiab] OR shockwave*[Tiab] OR high energy shockwave*[Tiab] OR “High-energy shock waves”[MeSH] OR shock wave*[Tiab] OR radial pressure wave*[Tiab] OR acoustic wave*[Tiab] OR ESWT[Tiab] OR HESW[Tiab] OR SWT[Tiab] OR SW[Tiab] OR RPWT[Tiab] OR AWT[Tiab]) |
| Outcome |
| (Mechanotransduction[Tiab] OR Mechanotransduction, cellular[MeSH] OR Mechanoreceptors[MeSH] OR mechanotransduction, cellular[Tiab] OR Mechanoreceptors[Tiab] OR Mechanical signal transduction[Tiab] OR Mechanosensory transduction[Tiab] OR Target cell[Tiab] OR Receptor, cell surface[Tiab] OR Receptors, cell surface[MeSH] OR Signaling pathways[Tiab] OR Signal transduction[MeSH] OR Signal transduction[Tiab] OR Cell signaling[Tiab] OR Signal transduction system[Tiab] OR Receptor mediated signal transduction[Tiab] OR Signal pathways[Tiab] OR Signal transduction pathways[Tiab] OR Cellular[Tiab] OR Cells[Tiab] OR cells[MeSH Terms] OR Cell[Tiab] OR Cell physiology[Tiab] OR Cell physiological phenomena[MeSH] OR Cell physiological process[Tiab] OR Cell Physiological Phenomenon[Tiab] OR Micro RNA[Tiab] OR Micro RNAs[MeSH] OR Micro RNA[Tiab] OR Micro RNAs[Tiab] OR miRNA[Tiab] OR miRNAs[Tiab] OR Exosomes[MeSH] OR Exosomes[Tiab]) |
| Skin | Treatment | Evaluation | Conclusion | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Underlying Mechanism | Amount | Type of Fibrosis | Reference Study Design | SWT | Other | Timing | Type of Evaluation | Timing | Effect of SWT |
Inflammation
| N = 14 (F) | Obesity: Adipose tissue and skin Pre- operative bariatric surgery | [17] Non-RCT Quasi-experimental design: Post-test design | Left side: frequency: 15 Hz EFD: 180 mJ/4000 shots with 15 mm stainless steel tipEFD: 100 mJ energy/2000 shots with 15-mm plastic tip | Right side: Control | Preop: 2 treatments/ wk | Biopsies: IHC | / | * ↑ expression inflammation (p < 0.001):
Angiogenesis markers (p < 0.001):
|
| Cellular metabolism cAMP Mitochondrial division DRP1 Apoptosis Proliferation
| N = 8 (F) | Obesity grade I and IIPre- operative pre- preparation of bariatric surgery | [12] Non-RCT Quasi-experimental design: Post-test design | 7 SWT sessions EFD: 180 mJ 2000 shots + 15 mm stainless steel tipfrequency: 15 Hz EFD: 100 mJ | / | 2 treatments/ wk | Biopsy: IHC | / | cAMP: morphology was maintained -> no * changes (p < 0.1823) DRP1: no * positivity for the expression of this protein (p < 0.0851) Apoptosis: not reported Anti-MFN1 and anti-MFN2: higher immunopositivity IG → regulation in cell proliferation and MFN2 but not *
|
| Cell markers CD13, CD44, CD90, CD10, CD14Fibroblast marker αSMACol production: I and V Myofibroblast: TGFβ1 expression Integrin a3,5,v,11 | N = 5 | Myo- fibroblast of adipose tissue cells → after elective liposuction | [11] Non-RCT Quasi-experimental design: time-series design | EFD: 0.32 mJ/mm2 PPP of 90 MPa 1000 pulses frequency: 4 Hz EFD: 0.22 mJ/mm2 1000 pulses// 0.32/1000 pulses// 0.59/250 pulses | ControlTGF β1 and αSMA: different modalities | / | Real-time PCR (gene expression) Immunoblotting Immuno- fluorescence microscopy | 0–6–72 h 14 d 21 d | Cell markers:
Myofibroblast: impaired contractility and migration potential TGFβ1 ↑ → αSMA expression Integrina 11 * ↓ (p < 0.001) + effects on wound edges and cell contraction |
| Collagen fibers Angiogenesis marker: CD31 Vessel abundance: SMC Proliferation marker: Ki67 Macrophages: Activation and number: Markers: CD68 M1–M2:
CK and GF: TNF, PDGF, TGF, TNF IL6, VEGF Apoptosis ERK and Akt signaling | N = 9 | Chronic ulcers (venous) → not healing > 8 wks | [9] Non-RCT Quasi-experimental design: 1 group pre-post-test design | EFD: 0.1 mJ/mm2 frequency: 5 Hz 150–500 impulses | / | / | Pre and post-biopsy—Masson’s trichrome staining | / | ↑ but not *
* ↑ TNF (p = 0.036) * ↑ IL-1 (p = 0.029) PDGF (p = 0.029) TGFβ (intensity of 150 pulses)/ (p = 0.048) * ↑ TNF & TGFβ (intensity of 300 pulses) No * change of IL6, VEGF (p > 0.05) Apoptotic cells ↑ BUT no * changes in pAKT/totalAKT ratios or pSTAT3/totalSTAT3 |
| YAP1Myofibroblast activation: αSMAProliferative status: Ki-67Microvascular density: CD31 | N = 10 | Pressure ulcers | [10] Non-RCT Prospective interventional pilot study | Radial ESW: Low-energy: 300 pulses, pressure: 2.5 bars EFD: 0.15 mJ/mm2, frequency 5 Hz | / | 2 sessions, 2 x/wk, 3-day interval in between | Biopsy: IHC assessment and evaluation | baseline and 24 h after first ESW after second ESW | YAP expression: in epidermis and dermis
Myofibroblast activation: αSMA:
After first and second ESW: * ↑ number of cells for the Ki67: in keratinocytes, dermis cells and the papillary layer of the dermis (p < 0.0001) Microvascular density: CD31 After the first and second ESW: the number ↑ (p < 0.0001) |
In vitro:
Pro-inflammatory CK pro-angiogenic activity of endothelial cells in vivoCellular morphology in fibroblasts, keratinocytes and dermal microvascular endothelial cells | In vivo: N = 75 | Chronic leg ulcers: Keratinocytes (HaCaT cells), fibroblasts and endothelial cells in vitro | [3] Non-RCT Quasi-experimental design: time-series design | 0, 375, 750, 1500 shocks at energy level Frequency: 4 impulses per s EFD: 0.136 mJ/mm2 → 0, 50, 100 and 200 impulses per cm2 | No treatment | 4 times 1 x/wk every 3–4 wks | In vivo: wound healing assessment In vitro: FACs, WB, RT-PCR, asymmetric gene expression analyses | 24 h after SW | Cluster formation of fibroblasts dose-dependent Angiogenesis: 375 shocks enough to ↑ capillary formation → cell migration in fibroblasts and cytokines → ↑ wound closure after 24 h due to enhanced migratory capacity → expression of cell cycle regulatory genes and proteinsProliferation: 24 h: no changes in the 72 h: changes in the proliferationAltered expression of cytoskeletal proteins in fibroblasts [reorganization of the cytoskeleton: vimentin]: upregulation after 24 h in WB and dose-dependentActivate immune response factors (in keratinocytes): inflammatory CK: IL-1α upon SWT at 750 impulses and IL-1β at 1500 impulses: * ↑ (p < 0.05) Laminin-322 (via collagen VII): SWT: expression of laminin-332 → promoting wound healing |
| Col I, II, III and X Degrading enzyme: MMP-13 Angiogenesis: CD31, VEGF Anti-inflammatory cytokines: TGFβ1 and IL6 Proliferating and regeneration markers: PCNA and fibronectin Apoptosis markers: Tunel and caspase-3 | N = 39 (22 ESWT/17 steroid group) | Keloids scars (>1 cm) | [18] Non-RCT Prospective open-label randomized case study | EFD: 0.11 mJ/mm2 500 impulses. 4 shocks/s 3 ESWT treatments in 6 wks | Steroid group | / | IHC: Masson Trichome stain | 6, 12, 24 and 48 weeks after the last treatment | * ↓ in col I and III (p < 0.05) * ↑ in MMP-13 (p < 0.05) Small but not * changes in angiogenesis, proliferative and regeneration, anti-inflammatory and apoptosis biomarkers |
| FXII + fibrocytes CD34 of fibroblasts Collagen type I/III CD31 of angiogenesis | N = 70 | Retracting hand scars | [19] RCT | 10 unfocused ESWT 2 x/wk EFD: 0.13 mJ/mm2 Frequency: 6 Hz Duration: 1.5 min (500 pulses for session per 360 impulses/min) | / | / | IHC staining of fibroblasts and angiogenesis | Pre and post-ESWT | * ↑ in dermal fibroblasts, neoangiogenetic response and type I collagen → replacement of type III with type I collagen (p < 0.05) * ↑ in fXIIIa-positive fibrocytes, CD34 dermal expression and CD31-positive small vessels (p < 0.05) |
| Fibrosis markers Endothelial involvement: vWF, VEGF, ICAM-1, MCP-1 Circulating endothelial cells (CECs) Endothelial progenitor cells (EPC) Nitric oxide | N = 30 (F = 29/ M = 1) | Systemic sclerosis (excessive deposition of collagen in the skin and visceral organs) | [20] Pilot studyNon-RCT Quasi-experimental design: pre and post-test design | Defocused ESWT Dorsal and volar forearm: 2000 shots Dorsal side of hand and fingers: 1000 EFD: 0.2–0.25 mJ/mm2 frequency: 4 Hz | / | / | Blood samples: ELISA | Before, 30 and 60 days after ESWT | Fibrosis marker: No * changes vWF, VEGF, ICAM-1, MCP-1: * ↑ on 30 and 60 days after the end of treatment * ↑ CECs/EPC (p < 0.05) NO: ↑ in 2/3 patients (not *) |
| Proliferation: P38 MAPK P44/42 MAPK Total Akt Total Mek ½ | / | Primary human adipose tissue-derived stem cells Full-thickness biopsies from excision wounds | [8] Non-RCT Report Quasi-experimental times series design | In vitro: 10–300 pulses EFD: 0.03–0.19 mJ/mm2 3 Hz In vivo: 100 pulses frequency: 13 Hz Treatment at 0.03, 0.07, 0.13 and 0.19 mJ/mm2 | Control | / | H&E IHC WB | 15 h 24 h 36 h after ESWT | Proliferation and activation: downstream Erk ½, Ki67 activation Mek ½ and P38 MAPK → enhance proliferation
|
| TGFβ1 αSMA col type I fibronectin twist I E cadherin N cadherin | N = 4 | Human fibroblasts from post-burn hypertrophic scars | [5] Non-RCT Quasi-experimental design: times series design | 1000 impulses EFD: 0.1, 0.03 and 0.3 mJ/mm2 | / | / | Real-time PCR WB For mRNA and protein expression | 24 h and 72 h after treatment | TGFβ1, αSMA, vimentin: mRNA and protein: * ↓ after 24 h and 72 h EMT markers: collagen type I mRNA * ↓ after 72 h in all regimens * ↓ after 24 h only in 0.1 and 0.03 mJ/mm2 (p < 0.05) col type I protein * ↓ after both 24 and 72 h (p < 0.05) fibronectin mRNA: 24 h * ↓ in all regimens 72 h: * ↓ only at 0.03 and 0.1 mJ/mm2 BUT fibronectin protein was * ↑ 24 h and 72 h after SW with 0.03 and 0.1 mJ/mm2 (p < 0.05) N and E cadherin (= cell surface markers for EMT) mRNA of N cadherin * ↓ after 24 h and 72 h in all regimens (p < 0.05) E cadherin * ↓ after 24 h and 72 h only in 0.1 and 0.3 mJ/mm2 (p < 0.05) |
| Proliferation marker: keratin 5 and 14 Activation marker: keratin 6 and 6/17 | Keratinocytes form hypertrophic scars (during surgical procedure) Human normal keratinocytes (HNK) Human hypertrophic scar keratinocytes (HTSK) | [4] Non-RCT Quasi-experimental design: times series design | Duolith SD device SD1 Storz medical EFD: 0.1, 0.2, 0.3 mJ/mm2 frequency: 4 Hz and 1000 impulses/cm2 | / | / | mRNA and protein expressionReal-time PCR WB | At 24 h And 72 h | HTSK: Proliferation marker:
| |
| Differentiation marker: keratin 1 and 10 Apoptosis factor: Bax, Bcl2, ASK1, caspase 14 Proliferation and differentiation marker: p21, p27 and Notch1 | Differentiation marker: keratin 1: In HTSKs after 24 h and 72 h * lower in Involucrin expression HTSKs (p < 0.05) 24 h: 0.1 mJ/mm2 not * changed, 24 h: in 0.2 and 0.3 mJ/mm2 * higher (p < 0.05) 72 h: under all regimens * lower (p < 0.05) protein expression of keratin 1 and involucrin in HNKs 24 h and 72 h after SW regimens * higher (p < 0.05) Apoptosis factor: Pro-apoptotic factor Bax/Anti-apoptosis factor Bcl2 24 h and 72 h * higher and lower than that of those in untreated control cells (p < 0.05) ASK1: in HTSKs 24 h and 72 h after SW * higher (p < 0.05) Caspase 14 expression: 24 h after SW was not * different 72 h * ↑ (p < 0.05) Proliferation and differentiation marker: p21, p27 and notch 21 0.1 mJ/mm2: not * changed 0.2–0.3: * higher at 24 h/at 72 h * lower (p < 0.05) | ||||||||
| Bone | Treatment | Evaluation | Conclusion | ||||||
| Underlying Mechanism | Amount | Type of Fibrosis | Reference Study Design | SWT | Other | Timing | Type of Evaluation | Timing | Effect of SWT |
| Osteogenic differentiation: miR-138 differentiation: FAK Erk ½ p38 RUNX2miR138 ERK ½ | N = 3 (all male patients) | MSCs [after surgery: resection]; bone marrow, tendon and adipose tissues | [21] Non-RCT Quasi-experimental design: one group pre and post- test design | EFD: 0.16 mJ/mm2 500 impulses | / | / | Real-time PCR WB analysis | / | TG: SW promotes differentiation of MSCs via FAK
|
| Collagen type II, βactinProliferation: Ki67Apoptosis: caspase-3Migration Cell tracking in healing, migration and wound healing assay | / | hBMSCs [human bone marrow stromal cells] → femoral head from patients after hip joint replacement | [22] Non-RCT Quasi-experimental design: post-test only control group | SW continuous pulse1000 impulsesFrequency: 4 HzEFD:
| control: Sham treatment | / | RT-PCR IHCBoyden chamber assay Cell tracking assay Wound healing assay | 6 h and 12 h after SW 0 min and 30 min after SW 4 h and 8 h | Growth rate: both in treated and untreated hBMSCs Proliferation: Ki67
Migration: F-actin stress fibers Disorganized actin fibers of the cytoskeleton: 0 min no changes/30 min: 0.2 mJ/mm2 * reduced disorganized F-actin (p < 0.05) ↑ wound closure proportion |
| IL-10 TNF-α CD29/β1 integrin CD105/endoglin expression | N = 13 (9 F/4 M): OA/ N = 7 from HD after joint traumatic fracture | Knee OA Human subchondral bone: osteoblasts | [23] Non-RCT Quasi-experimental design: times series design | EFD: (mJ/mm2 and impulses):
| Healthy bone | / | Flowcytometry | / |
|
| VEGF AProtein levels ERK-dependent HIF-1α | / | Fetal preosteoblastic cells | [23] Non-RCT Quasi-experimental design: post-test only control group design | EFD: 0.16 mJ/mm2 frequency: 1 Hz 500 impulses | / | Single treatment | RT-PCT immunoblotting | 15, 30 and 1,3, 6, 12 and 24 h after SW treatment |
→+ effects on angiogenesis |
| Cell morphology: SOX-9, COL1A, COL2AProliferation and repair: TNFα, IL-6 SOX-9, COL1A, COL2A, IL-17A Inflammation: IL-10 | N = 4 | Femoral head (with OA): chondrocytesIn vitro | [24] Non-RCT Quasi-experimental design: post-test only control group design | EFD: 0.14 mJ/mm2 Energy level 1000 impulses | Control HA PRP | / | RT-PCRImmuno-fluorescenceImmunoassay kit | 12 days after SW treatment |
Ki67: not * changed (p < 0.05) Pro-inflammatory cytokines: TNFα, IL-6 * SOX-9, COL1A, COL2A (p < 0.001), IL-17A ↓ Anti-inflammatory cytokines: IL-10 * ↑ (p < 0.05) |
| Angiogenesis: VEGFOsteogenesis gene expression: BMp-2 RUNX2Osteocalcin: mRNA expression | N = 6 patients but 4 samples | Osteonecrosis of the femoral head BMSC for hips | [25] Non-RCT Quasi-experimental design: pre-post-test design | 250 shockwaves In 2 sessions EFD: 0.18 mJ/mm2 | Control | / | RT-PCR | 72 h after SW treatment | VEGF * ↑ expression in comparison to CG (p < 0.05)NOC18 * ↑ the osteogenic markers (BMp-2, RUNX-2) (p < 0.05) osteocalcin mRNA * higher expression (p < 0.05) |
| Cell proliferation: CCK-8 OCN (Β-actin) PPARϒ (Β-actin) Alkaline phosphatase Cbfα (GADPH) | N = 20, but only 6 samples | Bone with avascular necrosis of the femoral head (ANFH) | [26] Non-RCT Quasi-experimental design: post-test only control group design | Intervention: 10 min EFD: 0.16 mJ/mm2 500 impulses frequency: 1 Hz MFL 5000 lithotriptor | Control | / | StainingRT-PCR WB | 3 d 6 d 9 d 12 d 15 d (after SW treatment) | Gene expression on cell proliferation:
|
| Muscle, tendon and ligament | Treatment | Evaluation | Conclusion | ||||||
| Underlying Mechanism | Amount | Type of Fibrosis | Reference Study Design | SWT | Other | Timing | Type of Evaluation | Timing | Effect of SWT |
| In fibroblasts: YAPProliferation rate | N = 8 (F = 4/ M = 4) | Elective spine surgical procedures from fascia thoracolumbalis | [27] Non-RCT Quasi-experimental design: post-test only control group design | SW EFD: 0.25 mJ/mm2 frequency: 2.5 Hz 100 shots | Not treated | / | ImmunoblottingRNA extraction and real-time PCR | 2 h 24 h 48 h (after SW treatment) | YAP activation was induced → * difference in treated cells compared to untreated cells (p < 0.05) YAP activation → upregulation fibrosis-associated gene expression (COL1A1 and HABP2): COL1A1 * enhanced at
|
| Proliferation MMP 1-2-9-13 IL 1-6-13Cell proliferationCell death | / | Diseased and fibrotic Achilles tendon → tenocytes | [28] Non-RCT Quasi-experimental design: post-test only control group design | EFD: 0.17 mJ/mm2 250, 500, 1000 and 2000 times | / | / | qPCR | / | In fibrotic tenocytes: proliferation: * ↓ IL-6, MMP1 and MMP13 (p < 0.05) MMP2 and MMP9 did not * change * ↑ IL-1 (p < 0.05) Cell proliferation: 500 shock group no * diff/1000 2000 shock group: * ↓ Cell death: 500 and 1000 shock group * ↑ (p < 0.05) |
| Differentiation marker: αSMAFibroblast marker: CD 90.2Mesenchymal stem markers: CD44, CD105 and CD146 | N = 5 healthy (ST) N = 5 ruptured (AT) | Human tendon-derived stem/progenitor cells (hTSPC) From Achilles tendon and semitendinosus tendon | [29] Non-RCT Quasi-experimental design: post-test only control group design | / | / | / | Immuno-fluorescenceRT-PCR | / | * ↑αSMA ST (p < 0.05), ↑ in AT but not * ESWT effect: Ki67—analysis of a nuclear marker: after 12 daysUpregulation of COL2A and SOX9 BGLAP and RUNX2 not * different |
| Cell viability, proliferation, migration AND expression of Ki67, COL-I α1, TGFβ, and VEGF | N = 8 (M = 5/ F = 3) | Anterior cruciate ligament | [26] Non-RCT Quasi-experimental design: post-test only control group design | 1000 SW impulses EFD: 0.15 mJ/mm2 | Control group: no SW | / | Real-time PCR Scratch migration assay Transwell migration assay Immuno-fluorescence staining | 0–32 hAfter 12 days: Ki67 | * ↑ cell viability, proliferation and migration (Ki67) (p < 0.01) ↑ COL-I α1, TGFβ, and VEGF expression BMSC proliferation and migration rate * ↑ after coculture [with ACL remnant cells with and without ESW stimulation] (p < 0.01) |
| Lung | Treatment | Evaluation | Conclusion | ||||||
| Underlying Mechanism | Amount | TYPE OF FIBROSIS | Reference Study Design | SWT | Other | Timing | Type of Evaluation | Timing | Effect of SWT |
Bronchial fibroblasts
| N = 6 (N = COPD/ N = 3 control) | COPD Bronchial fibroblastsCOPD patients | [30] Non-RCT Quasi-experimental design: post-test only control group design | Piezoelectric EFD: 0.3 mJ/mm2 frequency: 4 shocks/s 500 pulses | Control: non-treated fibroblasts | / | qRT-PCR ELISA IHC | 0, 4, 24, 48 and 72 h (after SW treatment) | 24 and 48 h: ↑ proliferation mRNA protein levels remodelling markers PCNA mRNA: not ↑ in the ESW group |
Cell proliferation:
| Control smokers with normal lung function | PCNA protein: Tendency to ↑TGFb1 mRNA not * changed/TGFb1 protein * ↑ (p < 0.01: 24 h and p < 0.001: 48 h)Procollagen I: no * difference between TG and CGNF kB-65 * ↓ (p < 0.01: 24 h and p < 0.001: 48 h) WST-1 and M-PER: cell proliferation and * ↑ of specific markers of cell proliferation (p < 0.01: 24 h and p < 0.001: 48 h) | |||||||
| Heart | Treatment | Evaluation | Conclusion | ||||||
| Underlying Mechanism | Amount | Type of Fibrosis | Reference Study Design | SWT | Other | Timing | Type of Evaluation | Timing | Effect of SWT |
| VEFGA VEGFB Chemokine ligan1 CXCL1,2,3 TNF α MAPK-9 | N = 23 (M = 17/ F = 6) | Ischemic heart failure Intervention study | [31] Non-RCT Quasi-experimental design: pre and post-test design | Cardiac shock wave therapy with an electromagnetic shockwave device 9 sessions (200 shocks/session) EFD from 0.0024 to 0.09 mJ/mm2 | / | 3 times per week for 3 weeks | qPCR | 3 days before CSWT (baseline) and 1 week after CSWT | CSWT:
MAPK-9: * ↓ (p < 0.0001) |
| miR-19a-3pCD9, CD63 and CD81 | / | Isolation of HUVECs and myocardial infarctionIn vitro | [6] Non-RCT Quasi-experimental design: post-test only control group design | / | / | day 1 and 3 SWT | Flow cytometry | 2 min, 30 min, 1 h, 4 h and 24 h after SWT | Proliferation and angiogenesis Akt inhibition: by CD9, CD81 and CD63/Erk → enhanced endothelial and proliferation + αSMA CD9, CD63 and CD81 * higher (p < 0.01) miR-19a-3p: induces inhibition of TSP-1 mRNA translation and facilitates the angiogenic effect |
| Neo-angiogenesis: VEGFR-2αSMA α/βMHC, FVIII and GADPH Apoptosis | N = 16 (M = 9/F = 7) healthyN = 8 (M = 5/F = 3) cardiomyopathy patients | In vitro: cardiomyocytes, smooth muscle and endothelial lineage > normal heart > cardiomyopathy | [32] Non-RCT Quasi-experimental design: post-test only control group design | 800 impulses PPP: 18 MPa EFD: 0.10 mJ/mm2 | No SW | Immuno-fluorescenceTUNEL assayWBRT-PCR | 7 d cultured | In neo-angiogenesis: * ↑ in VEGFR-2 expression (p < 0.05) * ↑ αSMA (p < 0.05) Effect on transcriptional activity: mRNA with primers: α/βMHC, FVIII and GADPH housekeeping: * upregulated expression (p < 0.05) Proliferation and differentiation in cardiac primitive cells Apoptosis: no statistically significant difference in both groups | |
| Prostate | Treatment | Evaluation | Conclusion | ||||||
| Underlying Mechanism | Amount | Type of Fibrosis | Reference Study Design | SWT | Other | Timing | Type of Evaluation | Timing | Effect of SWT |
| Cell viability Gene expression: αSMA COL1 | N = 10 tumor tissue samplesIn vitro | Human prostatic cancer cells: Carcinoma associated | [33] Non-RCT: Quasi-experimental design: post-test only control group design | SWT1: CAF E8-1000 EFD: 0.32 mJ/mm2, 1000 pulses frequency = 4 shocks/s | / | / | / | 72 h 48 h (after SW treatment) | ESW effect on: Cell viability: no sign effect/ Small reduction αSMA * ↓: SWT2 (26%↓) > SWT1 (4.3%↓) (p < 0.001) COL 1 * ↓: SWT1 (51%↓) > SWT2 (45%↓) (p < 0.001) |
| Growth of epithelial cells | fibroblasts (CAFs) After prostatectomy | SWT2: CAF E12-250: EFD: 0.59 mJ/mm2 250 pulses frequency: 4 shocks/s PPP: 64 MPa | Protein levelsWB Immuno- fluorescenceMatrigel invasion assay + scratch wound assay | * ↓ αSMA and COL1 (p < 0.01) * ↓ PC3 and DU145: and * ↓ in migration (p < 0.001)Both energy levels: effective in reducing COL 1 and αSMA BUT E12-250: higher ↓ αSMA | |||||
| Immune system | Treatment | Evaluation | Conclusion | ||||||
| Underlying Mechanism | Amount | Type of Fibrosis | Reference Study Design | SWT | Other | Timing | Type of Evaluation | Timing | Effect of SWT |
| Cell stimulation Cell viability ATP release Transfection: siRNA: to block FAK expression T-cell proliferation | N = 3 | Jurkat T-cells | [7] Non-RCT: Quasi-experimental design: post-test only control group design | frequency: 50 Hz PPP: 23+-1.4 MPa EFD: 0.18 mJ/mm2 Exposed to 0, 50, 100, 150, 200, 250, 300, 350, 400, 450, 500 or 600 impulses Time: 10–20 min | Control | / | WB analysis ELISA | phosphorylation of FAK on Tyr397 or Tyr 576/577 P38 MAPK on Thy180/Tyr182: * ↑ with higher impulses till 250 BUT * ↓ with higher impulses + baseline reached with 350 impulses FAK activation -> p38 MAPK
| |
| Outcome | N | Within Study Bias | Reporting Bias | Indirectness | Imprecision | Heterogeneity | Incoherence | Confidence Rating |
|---|---|---|---|---|---|---|---|---|
| Cell contraction | 5 |  |  |  |  |  |  | Very low |
| Fibroblasts | 7 |  |  |  |  |  |  | Very low |
| Myofibroblasts | 3 |  |  |  |  |  |  | Very low |
| αSMA | 4 |  |  |  |  |  |  | Very low |
| Fibrocytes | 1 |  |  |  |  |  |  | Very low |
| ECM component | 2 |  |  |  |  |  |  | Very low |
| Stromal cells | 1 |  |  |  |  |  |  | Very low |
| Collagen I | 7 |  |  |  |  |  |  | Low |
| Collagen II | 3 |  |  |  |  |  |  | Very low |
| Collagen III | 2 |  |  |  |  |  |  | Low |
| Collagen V | 1 |  |  |  |  |  |  | Low |
| Collagen marker | 3 |  |  |  |  |  |  | Low |
| Endothelial cells | 1 |  |  |  |  |  |  | Low |
| Nitric oxide | 1 |  |  |  |  |  |  | Low |
| cAMP | 1 |  |  |  |  |  |  | Very low |
| Keratinocytes | 2 |  |  |  |  |  |  | Low |
| Macrophages | 2 |  |  |  |  |  |  | Low |
| Cytokines | 4 |  |  |  |  |  |  | Low |
| Chemokines | 1 |  |  |  |  |  |  | Low |
| Lymphocytes | 1 |  |  |  |  |  |  | Low |
| NK cells | 1 |  |  |  |  |  |  | Low |
| COX2 | 1 |  |  |  |  |  |  | Low |
| Angiogenesis marker | 3 |  |  |  |  |  |  | Low |
| Growth factor | 7 |  |  |  |  |  |  | Low |
| miR-19a-3p | 1 |  |  |  |  |  |  | Very low |
| Proliferation | 5 |  |  |  |  |  |  | Very low |
| Protein kinase | 3 |  |  |  |  |  |  | Low |
| MAPK, Mek | 2 |  |  |  |  |  |  | Low |
| Cell surface receptors | 2 |  |  |  |  |  |  | Low |
| Alkaline phosphatase | 1 |  |  |  |  |  |  | Low |
| Anti-MFN | 1 |  |  |  |  |  |  | Very low |
| FAK/Erk | 1 |  |  |  |  |  |  | Low |
| Cbfα1 | 1 |  |  |  |  |  |  | Low |
| Erk, Akt, total Akt | 1 |  |  |  |  |  |  | Low |
| pSTAT3 | 1 |  |  |  |  |  |  | Low |
| Caspase 3,14 | 2 |  |  |  |  |  |  | Very low |
| Not Specified in Stage of Fibrosis | ||||
|---|---|---|---|---|
| Cell Type | Cell Category/Marker | Mediator/Gene Expression | Cluster of Differentiation | EFFECT of Shockwave |
| Myofibroblasts | Cell contraction and migration | / | / | impaired contractility and * increased migration potential [11] |
| GF ECM Integrin | TGF β1α11 | ↑ à αSMA [11] * ↓ [11] (p < 0.001) | ||
| Fibroblasts | / | Fibronectin: mRNAFibronectin: protein | / | Amelioration: * ↓ [5] (p < 0.05) Amelioration: * ↑ [5] (p < 0.05) |
| Osteoblasts | / | β integrin | CD29 | not * changed [23] (p < 0.001) |
| Bone marrow stromal cells | / | F-actin | 0.2mJ/mm2 * ↓ disorganized F-actin [22] (p < 0.05) | |
| CollagenEMT marker | I | / | / | ↓ [11]/ * ↓ [18] (p < 0.05)/ ↓ [33] |
| I | mRNA/protein | / | * ↓ [5] (p < 0.05)/↑ [38] | |
| COL-I α1 | / | * ↑ [27] (p < 0.05) | ||
| II | / | SOX9 | no differences in TG and CG [22] upregulation [29] | |
| I/II | / | / | shift from COL 1A à COL 2A (regulated by enhanced SOX9) [24] | |
| III | / | / | * ↓ [18] (p < 0.05) | |
| I/III | / | / | replacement of collagen type III with type I [19] | |
| V | / | / | ↓ [11] | |
| Cell expression | TGFβ1 | / | BMp-2 RUNX2 | * ↑ [25] (p < 0.05) * ↑ [25] (p < 0.05) |
| Circulating endothelial cells | CECs | / | / | * ↑ [20] (p < 0.05) |
| Endothelial progenitor cells | EPC | / | / | * ↑ [20] (p < 0.05) |
| Nitric oxide | NO | / | / | ↑ in 2/3 patients (not * ) [20] |
| Growth factor | GF | VEGF | / | * ↑ [20] (p < 0.05) |
| Fibrosis markers Endothelial involvement | Immunoglobulin | / | vWF, ICAM-1, MCP-1 | * ↑ [20] (p < 0.05) |
| Cellular metabolism | cAMP | / | no * change [12] | |
| Inflammation | ||||
| Cell type | Cell category/marker | Mediator/Gene expression | Cluster of differentiation | EFFECT of shockwave |
| Fibroblasts | / PCNAGF Procollagen I NF-kB-p65 | / mRNA protein TGFβ1
/ | CD117, CD90 | ↑ [30] not ↑ tendency to ↑ not * changed [30] ↑ [30] no * difference [30] ↓ [30] |
| Fibroblasts/myofibroblasts | YAP1 protein | / | / | * expression [10,27] |
| αSMA | / | / | higher values of αSMA after the first and second treatment in comparison to baseline [10] | |
| fibrocytes | / | CD34 | * ↑ [19] (p < 0.05) | |
| Keratinocytes | CK/GF | IL-6/VEGF | / | change but not * [18] |
| Macrophages | / M1-M2 macrophages | / | CD68 M1: SOCS3 expression, HLA DRM2: CD163 | * ↓ [9] (p < 0.05) ↑ expression [9] ↑ expression [9] |
| Macrophages | CK GF CK/GF | IL-1 TNF, PDGF, TGF IL-6/VEGF | / / / | * ↑ [9] (p < 0.05) * ↑ [9] (p < 0.05) no * change [9] |
| / | / | mRNA | CD44 | * ↑ [24] (p < 0.001) |
| / | Pro-inflammatory CK | TNFα IL-6 IL-17A | / / / | * ↓[24] (p < 0.001) [23] (p < 0.05) ↑ [31] * ↓ [24] (p < 0.001) [28] |
| Lymphocytes | T-lymphocytes B-lymphocytes | / / | CD3 CD23 | higher expression [17] |
| NK cells | / | / | / | ↑ expression [17] |
| / | Chemokine pathway | / | ligand 1 CXCL1,2 and 3 | ↑ [31] |
| / | Chemokine | / | MAPK-9 | ↓ [31] |
| Enzymes | / | COX2 | / | ↑ expression [17] |
| Integrin α11 | / | / | / | ↓ overexpression [11] |
| Angiogenesis | ||||
| Cell type | Cell category/marker | Mediator/Gene expression | Cluster of differentiation | EFFECT of shockwave |
| Myofibroblasts | / | / | CD34 | no * difference [17]/no expression [11] |
| / | / | CD14 | lower [11] | |
| Angiogenesis markers | / | / | CD105 CD105/endoglin | not * changed expression [17] |
| / | GF/ / | VEGFVEGFA VEGFR-2 | // / | * higher [3,17,25] p < 0.001 enhanced by Ras-Erk-HIF-α [41] * ↑ [32] |
| / | αSMA | / | / | ↑ [32] ↓ [33] |
| Collagen fibers | / | / | CD31 | * ↑[9,19] (p < 0.05) more ↑ after a first and a second treatment [10] |
| / | / / | miR-19a-3p / | CD9 CD63 CD81 | inhibition of TSP-1 mRNA translation [6] |
| Fibroblasts | / | Laminin/integrin | / | ameliorated due to SWT [3,43] |
| Proliferation | ||||
| Cell type | Cell category/marker | Mediator/Gene expression | Cluster of differentiation | EFFECT of shockwave |
| / | PCNA & fibronectin | / | / | small but not * change [18] |
| / | GF | TGFβ | / | ↑ [38] |
| / | / | VEGF VEGF A and B | / / | ↑ [38] ↑ [31] |
| / | Ki67 | / | / | depending on the EFD: [22] (p < 0.05)
enhanced [8] * ↑ [29] (p < 0.05) more ↑ after a first and second treatment [10] |
| Enzyme | Protein kinase | P38 MAPK | / | enhanced [8] |
| P44/42 MAPK | / | / | ||
| MAPK9 | / | ↓ [31] | ||
| Protein kinase B | Total Akt: Akt/erk Erk1/2 | // | downstream regulation [8] enhanced [43] (through VEGF and VEGF-R) | |
| / | Total Mek 1/2 | / | downstream regulation [8] | |
| Epithelial cells | Keratin5, 14 | / | / | depending on the EFD: 0.1: no effect/but 0.2–0.3: * lower [4] |
| Cell surface receptors | / | / | p21, p27 and Notch1 p16 | depending on the EFD: 0.1: not * changed [4] (p < 0.05) 0.2–0.3: 24 h * higher/72 h * lower [4] (p < 0.05) * ↓ [24] (p < 0.05) |
| Isoenzymes | / | / | Alkaline phosphatase | higher in ESW than the control group [26] |
| GADPH | / | Cbfα | higher in ESW over the whole timeline [26] | |
| Β-actin | / | OCN | detected already in 9 d (not in the control group) [26] | |
| Macrophages | Β-actin | / | PPARϒ | steady for both ESW and control groups at all time points, with significantly weaker levels in the ESW group than in the control group [26] |
| Fibroblasts | / | WST-1 | ↑ [30] | |
| / | M-PER | ↑ [30] | ||
| T-cell | / / | / / | Through ATP, P2X7 receptors FAK activation and MAPK | ↑ with higher impulses till 250 BUT ↓ with higher impulses + baseline reached with 350 impulses [7] |
| CK | IL-2 expression | / | enhance IL-2 expression [7] | |
| ECM remodelling and cell adhesion | Enzyme | / | MMP1 and 2 | * ↓ [28] |
| / | mRNA | α/βMHC, FVIII and GADPH | upregulated expression [32] | |
| Anti-MFN1 Anti-MFN2 | / / | / / | higher scores [12] | |
| Activation/differentation marker | ||||
| Cell type | Cell category/marker | Mediator/Gene expression | Cluster of differentiation | EFFECT of shockwave |
| Keratinocytes | Keratin 6, 17 | / | / | * higher [4] |
| Keratinocytes | Keratin 1, 10 | / | / | depending on the EFD [4] 24 h: 0.1 mJ/mm2 not * changed/in 0.2 and 0.3 mJ/mm2 * higher 72 h: under all regimens * lower |
| / | / | p21, p27 and Notch1 | depending on the EFD: 0.1: not * changed [4] 0.2–0.3: 24 h * higher/72 h * lower [4] (p < 0.05) | |
| / | / | miR-138 differentiation: FAK/Erk ½ pathway RUNX2 | * ↓ [21] (p < 0.01) downstream ↑ | |
| / | / | Cbfα1: Gene expression Protein expression | * ↑ [26] (p < 0.01) * ↓ [26] (p < 0.01) | |
| αSMA | / | / | ↑ [29] | |
| Apoptosis | ||||
| Cell type | Cell category/marker | Mediator/Gene expression | Cluster of differentiation | EFFECT of shockwave |
| Apoptotic cells | Erk Akt signaling | / | / | ↑ [9] |
| / | / | pAKT/totalAKT | no changes [9] | |
| / | / | pSTAT3/totalSTAT3 | no changes [9] | |
| Caspase 3 | / | / | small but not * change [18] | |
| Caspase 3 | / | / | depending on the EFD: 0.2 mJ/mm2: * ↓ after 6 h/transient effect after 12 h 0.3 mJ/mm2: * ↑ (p < 0.05) [22] | |
| Caspase pathway | / | Bax, Bcl2 | * higher (but lower than the untreated cells) [4] | |
| Caspase 14 | / | 24 h not * different/72 h * higher [4] | ||
| Protein kinase | MAP kinase | / | Ask1 | * higher after 24 h and 72 h [4] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Demuynck, L.; Moonen, S.; Thiessen, F.; Vrints, I.; Moortgat, P.; Meirte, J.; van Breda, E.; Van Daele, U. Systematic Review on Working Mechanisms of Signaling Pathways in Fibrosis During Shockwave Therapy. Int. J. Mol. Sci. 2024, 25, 11729. https://doi.org/10.3390/ijms252111729
Demuynck L, Moonen S, Thiessen F, Vrints I, Moortgat P, Meirte J, van Breda E, Van Daele U. Systematic Review on Working Mechanisms of Signaling Pathways in Fibrosis During Shockwave Therapy. International Journal of Molecular Sciences. 2024; 25(21):11729. https://doi.org/10.3390/ijms252111729
Chicago/Turabian StyleDemuynck, Lot, Sarah Moonen, Filip Thiessen, Ina Vrints, Peter Moortgat, Jill Meirte, Eric van Breda, and Ulrike Van Daele. 2024. "Systematic Review on Working Mechanisms of Signaling Pathways in Fibrosis During Shockwave Therapy" International Journal of Molecular Sciences 25, no. 21: 11729. https://doi.org/10.3390/ijms252111729
APA StyleDemuynck, L., Moonen, S., Thiessen, F., Vrints, I., Moortgat, P., Meirte, J., van Breda, E., & Van Daele, U. (2024). Systematic Review on Working Mechanisms of Signaling Pathways in Fibrosis During Shockwave Therapy. International Journal of Molecular Sciences, 25(21), 11729. https://doi.org/10.3390/ijms252111729









