Interactions between Guidance Cues and Neuronal Activity: Therapeutic Insights from Mouse Models
Abstract
1. Introduction
2. Ephrins in Sensory Systems
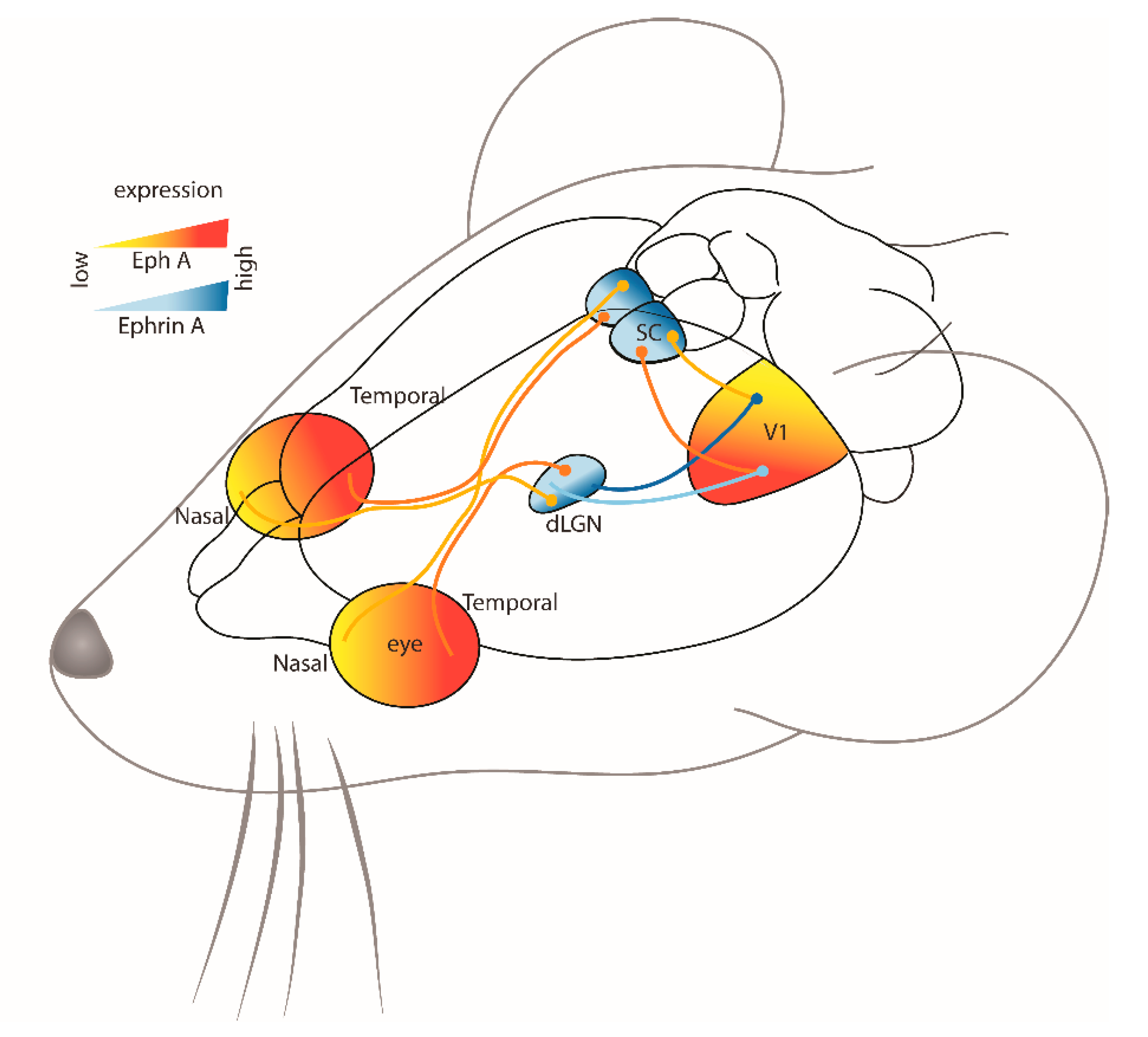
2.1. Visual System
2.1.1. Knockout Animal Models
Collicular Pathways
Thalamic Pathways
The Role of Neuronal Activity
2.1.2. Knock-In Animal Models
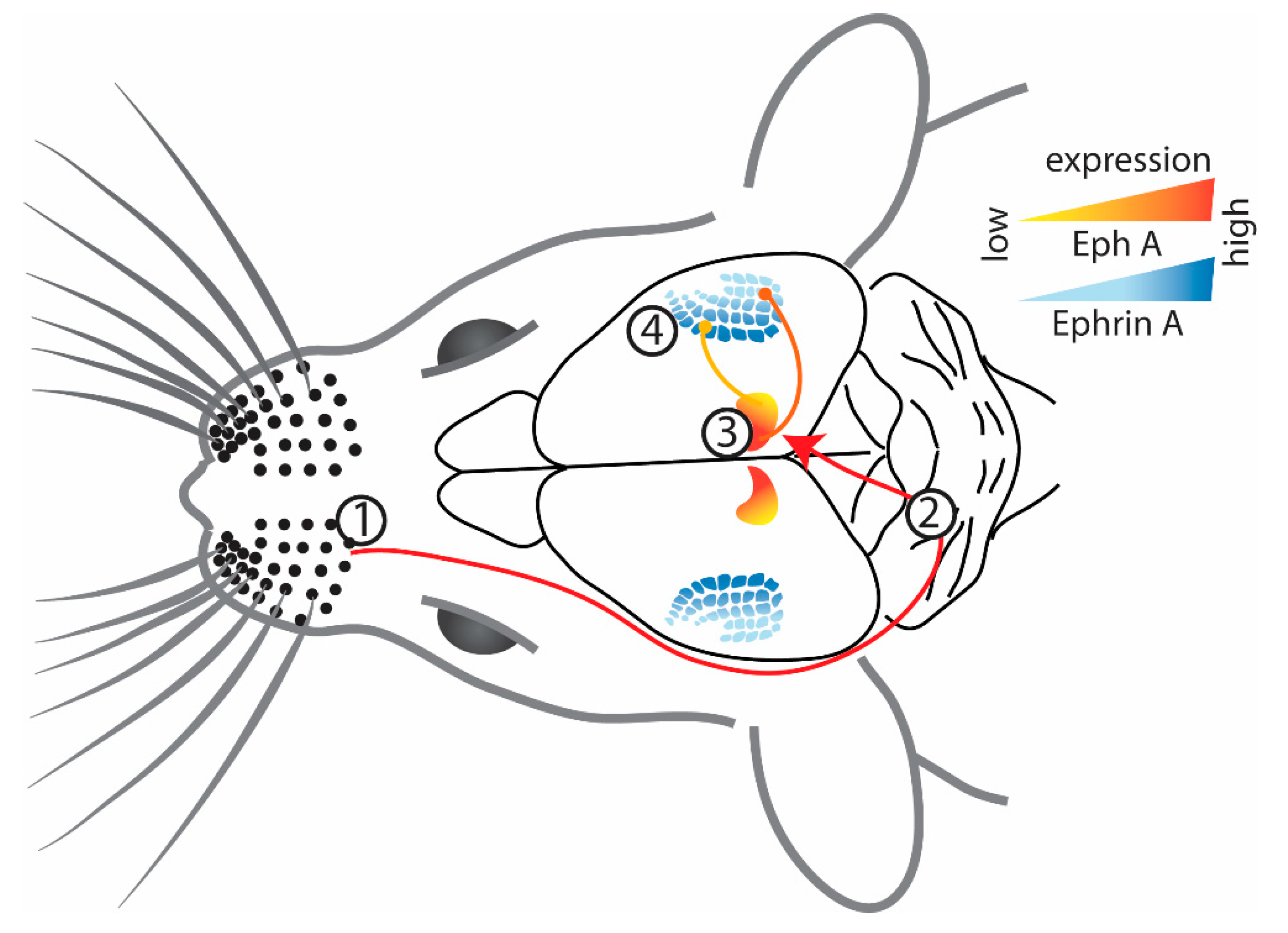
2.2. Somatosensory Cortex
2.3. Auditory System
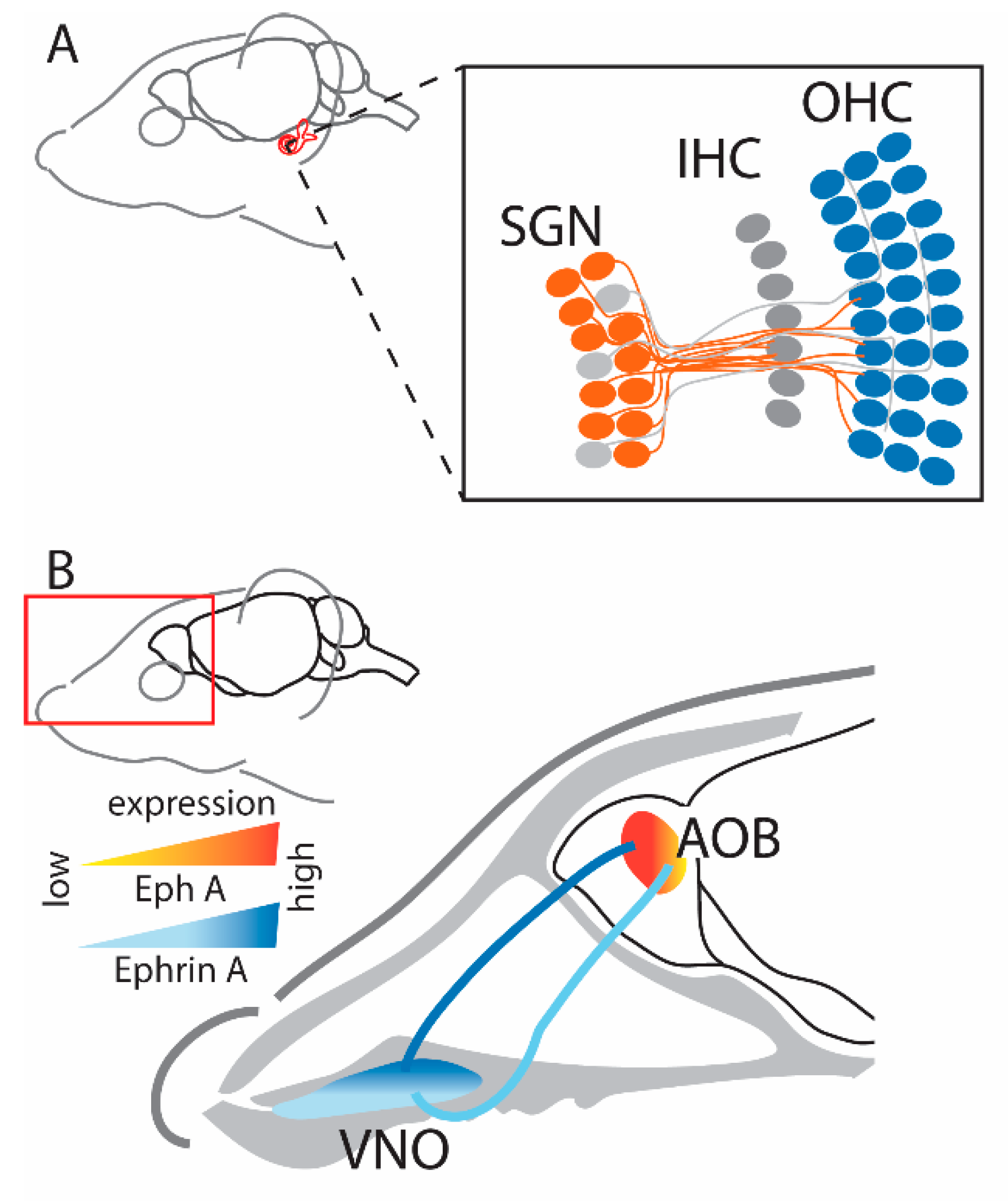
2.4. Olfactory System
2.5. Integration of Disordered Projections in the Superior Colliculus
3. Linking Structure and Function: Abnormal Visual Behavior in Ephrin-KO Mice
4. What Do Ephrin-KO Mice Tell Us about Activity-Dependent Refinement?
5. TMS as a Method to Increase Activity and Compensate for Loss of Ephrins
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thivierge, J.P.; Marcus, G.F. The topographic brain: From neural connectivity to cognition. Trends Neurosci. 2007, 30, 251–259. [Google Scholar] [CrossRef]
- Kaas, J.H. Topographic Maps are Fundamental to Sensory Processing. Brain Res. Bull. 1997, 44, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Cang, J.; Feldheim, D.A. Developmental Mechanisms of Topographic Map Formation and Alignment. Annu. Rev. Neurosci. 2013, 36, 51–77. [Google Scholar] [CrossRef] [PubMed]
- Javitt, D.C.; Freedman, R. Sensory Processing Dysfunction in the Personal Experience and Neuronal Machinery of Schizophrenia. Am. J. Psychiatry 2015, 172, 17–31. [Google Scholar] [CrossRef]
- Sinclair, D.; Oranje, B.; Razak, K.A.; Siegel, S.J.; Schmid, S. Sensory processing in autism spectrum disorders and Fragile X syndrome—From the clinic to animal models. Neurosci. Biobehav. Rev. 2017, 76, 235–253. [Google Scholar] [CrossRef]
- Huber, A.B.; Kolodkin, A.L.; Ginty, D.D.; Cloutier, J.F. Signaling at the growth cone: Ligand-receptor complexes and the control of axon growth and guidance. Annu. Rev. Neurosci. 2003, 26, 509–563. [Google Scholar] [CrossRef] [PubMed]
- Cline, H. Sperry and Hebb: Oil and vinegar? Trends Neurosci. 2003, 26, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Plachez, C.; Richards, L.J. Mechanisms of Axon Guidance in the Developing Nervous System. Curr. Top. Dev. Biol. 2005, 69, 267–346. [Google Scholar] [PubMed]
- Sperry, R.W. Chemoaffinity in the orderly growth of nerve fiber patterns and connections*. Proc. Natl. Acad. Sci. USA 1963, 50, 703–710. [Google Scholar] [CrossRef]
- Cheng, H.J.; Nakamoto, M.; Bergemann, A.D.; Flanagan, J.G. Complementary Gradients in Expression and Binding of ELF-1 and Mek4 in Development of the Topographic Retinotectal Projection Map. Cell 1995, 82, 371–381. [Google Scholar] [CrossRef]
- Wilkinson, D.G. Multiple roles of eph receptors and ephrins in neural development. Nat. Rev. Neurosci. 2001, 2, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.E.; Mann, F.; Erskine, L.; Sakurai, T.; Wei, S.; Rossi, D.J.; Gale, N.W.; Holt, C.E.; Mason, C.A.; Henkemeyer, M. Ephrin-B2 and EphB1 Mediate Retinal Axon Divergence at the Optic Chiasm. Neuron 2003, 39, 919–935. [Google Scholar] [CrossRef] [PubMed]
- Thakar, S.; Chenaux, G.; Henkemeyer, M. Critical roles for EphB and ephrin-B bidirectional signalling in retinocollicular mapping. Nat. Commun. 2011, 2, 431. [Google Scholar] [CrossRef] [PubMed]
- Nikolakopoulou, A.M.; Koeppen, J.; Garcia, M.; Leish, J.; Obenaus, A.; Ethell, I.M. Astrocytic Ephrin-B1 Regulates Synapse Remodeling Following Traumatic Brain Injury. ASN Neuro 2016, 8, 175909141663022. [Google Scholar] [CrossRef]
- Nguyen, A.Q.; Koeppen, J.; Woodruff, S.; Mina, K.; Figueroa, Z.; Ethell, I.M. Astrocytic Ephrin-B1 Controls Synapse Formation in the Hippocampus During Learning and Memory. Front. Synaptic. Neurosci. 2020, 12, 10. [Google Scholar] [CrossRef] [PubMed]
- Talebian, A.; Henkemeyer, M. EphB2 receptor cell-autonomous forward signaling mediates auditory memory recall and learning-driven spinogenesis. Commun. Biol. 2019, 2, 372. [Google Scholar] [CrossRef]
- Trabalza, A.; Colazingari, S.; Sgobio, C.; Bevilacqua, A. Contextual learning increases dendrite complexity and EphrinB2 levels in hippocampal mouse neurons. Behav. Brain Res. 2012, 227, 175–183. [Google Scholar] [CrossRef]
- Abate, G.; Colazingari, S.; Accoto, A.; Conversi, D.; Bevilacqua, A. Dendritic spine density and EphrinB2 levels of hippocampal and anterior cingulate cortex neurons increase sequentially during formation of recent and remote fear memory in the mouse. Behav. Brain Res. 2018, 344, 120–131. [Google Scholar] [CrossRef]
- Godement, P.; Salaun, J.; Imbert, M. Prenatal and Postnatal Development of Retinogeniculate and Retinocollicular Projections in the Mouse. J. Comp. Neurol. 1984, 230, 552–575. [Google Scholar] [CrossRef]
- Kageyama, G.H.; Robertson, R.T. Development of Geniculocortical Projections to Visual Cortex in Rat: Evidence for Early Ingrowth and Synaptogenesis. J. Comp. Neurol. 1993, 335, 123–148. [Google Scholar] [CrossRef]
- Inoue, K.; Terashima, T.; Inoue, Y. Postnatal Development of the Corticotectal Projection from the Visual Cortex of the Mouse. Okajimas Folia Anat. Jpn. 1992, 68, 319–331. [Google Scholar] [CrossRef]
- Sefton, A.J.; Dreher, B.; Harvey, A.R.; Martin, P.R. Visual System. In The Rat Nervous System; Elsevier: Amsterdam, The Netherlands, 2015; pp. 947–983. [Google Scholar]
- Simon, D.K.; O’leary, D.D.M. Development of Topographic Order in the Mammalian Retinocollicular Projection. J. Neurosci. 1992, 72, 1212–1232. [Google Scholar] [CrossRef] [PubMed]
- Osterhout, J.A.; El-Danaf, R.N.; Nguyen, P.L.; Huberman, A.D. Birthdate and Outgrowth Timing Predict Cellular Mechanisms of Axon Target Matching in the Developing Visual Pathway. Cell Rep. 2014, 8, 1006–1017. [Google Scholar] [CrossRef] [PubMed]
- Moses, C.; Wheeler, L.P.G.; LeVaillant, C.J.; Kramer, A.; Ryan, M.; Cozens, G.S.; Sharma, A.; Pollett, M.A.; Rodger, J.; Harvey, A.R. The Acquisition of Target Dependence by Developing Rat Retinal Ganglion Cells. eNeuro 2015, 2, ENEURO.0044-14.2015. [Google Scholar] [CrossRef] [PubMed]
- Beros, J.; Rodger, J.; Harvey, A.R. Age Related Response of Neonatal Rat Retinal Ganglion Cells to Reduced TrkB Signaling in vitro and in vivo. Front. Cell Dev. Biol. 2021, 9, 671087. [Google Scholar] [CrossRef] [PubMed]
- Nicol, X.; Muzerelle, A.; Rio, J.P.; Métin, C.; Gaspar, P. Requirement of adenylate cyclase 1 for the ephrin-A5-dependent retraction of exuberant retinal axons. J. Neurosci. 2006, 26, 862–872. [Google Scholar] [CrossRef]
- Pfeiffenberger, C.; Cutforth, T.; Woods, G.; Yamada, J.; Rentería, R.C.; Copenhagen, D.R.; Flanagan, J.G.; A Feldheim, D. Ephrin-As and neural activity are required for eye-specific patterning during retinogeniculate mapping. Nat. Neurosci. 2005, 8, 1022–1027. [Google Scholar] [CrossRef]
- Frisén, J.; Yates, P.A.; McLaughlin, T.; Friedman, G.C.; O’Leary, D.D.M.; Barbacid, M. Ephrin-A5 (AL-1/RAGS) Is Essential for Proper Retinal Axon Guidance and Topographic Mapping in the Mammalian Visual System. Neuron 1998, 20, 235–243. [Google Scholar] [CrossRef]
- Feldheim, D.A.; Kim, Y.I.; Bergemann, A.D.; Frisén, J.; Barbacid, M.; Flanagan, J.G. Genetic Analysis of Ephrin-A2 and Ephrin-A5 Shows Their Requirement in Multiple Aspects of Retinocollicular Mapping. Neuron 2000, 25, 563–574. [Google Scholar] [CrossRef]
- Sweeney, N.T.; James, K.N.; Sales, E.C.; Feldheim, D.A. Ephrin-as are required for the topographic mapping but not laminar choice of physiologically distinct RGC types. Dev. Neurobiol. 2015, 75, 584–593. [Google Scholar] [CrossRef]
- Wilks, T.A.; Rodger, J.; Harvey, A.R. A role for ephrin-As in maintaining topographic organization in register across interconnected central visual pathways. Eur. J. Neurosci. 2010, 31, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffenberger, C.; Yamada, J.; Feldheim, D.A. Ephrin-As and patterned retinal activity act together in the development of topographic maps in the primary visual system. J. Neurosci. 2006, 26, 12873–12884. [Google Scholar] [CrossRef] [PubMed]
- Cang, J.; Kaneko, M.; Yamada, J.; Woods, G.; Stryker, M.P.; Feldheim, D.A. Ephrin-As guide the formation of functional maps in the visual cortex. Neuron 2005, 48, 577–589. [Google Scholar] [CrossRef] [PubMed]
- Dufour, A.; Seibt, J.; Passante, L.; Depaepe, V.; Ciossek, T.; Frisén, J.; Kullander, K.; Flanagan, J.G.; Polleux, F.; Vanderhaeghen, P. Area Specificity and Topography of Thalamocortical Projections Are Controlled by ephrin/Eph Genes. Neuron 2003, 39, 453–465. [Google Scholar] [CrossRef] [PubMed]
- Uziel, D.; Mü, S.; Zarbalis, K.; Wurst, W.; Levitt, P.; Rgen Bolz, J. Miswiring of Limbic Thalamocortical Projections in the Absence of Ephrin-A5. J. Neurosci. 2002, 22, 9352–9357. [Google Scholar] [CrossRef] [PubMed]
- Kalatsky, V.A.; Stryker, M.P. New Paradigm for Optical Imaging. Neuron 2003, 38, 529–545. [Google Scholar] [CrossRef]
- Bansal, A.; Singer, J.H.; Hwang, B.J.; Xu, W.; Beaudet, A.; Feller, M.B. Mice Lacking Specific Nicotinic Acetylcholine Receptor Subunits Exhibit Dramatically Altered Spontaneous Activity Patterns and Reveal a Limited Role for Retinal Waves in Forming ON and OFF Circuits in the Inner Retina. J. Neurosci. 2000, 20, 7672–7681. [Google Scholar] [CrossRef]
- O’Leary, D.; Fawcett, J.; Cowan, W. Topographic targeting errors in the retinocollicular projection and their elimination by selective ganglion cell death. J. Neurosci. 1986, 6, 3692–3705. [Google Scholar] [CrossRef]
- Fawcett, J.W.; O’Leary, D.D.; Cowan, W.M. Activity and the control of ganglion cell death in the rat retina. Proc. Natl. Acad. Sci. USA 1984, 81, 5589–5593. [Google Scholar] [CrossRef]
- Triplett, J.W.; Owens, M.T.; Yamada, J.; Lemke, G.; Cang, J.; Stryker, M.P.; Feldheim, D.A. Retinal Input Instructs Alignment of Visual Topographic Maps. Cell 2009, 139, 175–185. [Google Scholar] [CrossRef]
- Kerschensteiner, D. Glutamatergic retinal waves. Front. Neural Circuits 2016, 10, 38. [Google Scholar] [CrossRef]
- Cang, J.; Wang, L.; Stryker, M.P.; Feldheim, D.A. Roles of Ephrin-As and Structured Activity in the Development of Functional Maps in the Superior Colliculus. J. Neurosci. 2008, 28, 11015–11023. [Google Scholar] [CrossRef]
- Haustead, D.J.; Lukehurst, S.S.; Clutton, G.T.; Bartlett, C.A.; Dunlop, S.A.; Arrese, C.A.; Sherrard, R.M.; Rodger, J. Functional topography and integration of the contralateral and ipsilateral retinocollicular projections of Ephrin-A−/− mice. J. Neurosci. 2008, 28, 7376–7386. [Google Scholar] [CrossRef]
- Nicol, X.; Voyatzis, S.; Muzerelle, A.; Narboux-Nême, N.; Südhof, T.C.; Miles, R.; Gaspar, P. cAMP oscillations and retinal activity are permissive for ephrin signaling during the establishment of the retinotopic map. Nat. Neurosci. 2007, 10, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.; Yates, P.A.; Burrola, P.; Ortuño, D.; Vaidya, A.; Jessell, T.M.; Pfaff, S.L.; O’Leary, D.D.; Lemke, G. Topographic Mapping from the Retina to the Midbrain Is Controlled by Relative but Not Absolute Levels of EphA Receptor Signaling. Cell 2000, 102, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Woolsey, T.A.; van der Loos, H. The structural organization of layer IV in the somatosensory region (S I) of mouse cerebral cortex. Brain Res. 1970, 17, 205–242. [Google Scholar] [CrossRef] [PubMed]
- Prakash, N.; Vanderhaeghen, P.; Cohen-Cory, S.; Frisé, J.; Flanagan, J.G.; Frostig, R.D. Malformation of the Functional Organization of Somatosensory Cortex in Adult Ephrin-A5 Knock-Out Mice Revealed by In Vivo Functional Imaging. J. Neurosci. 2000, 20, 5841–5847. [Google Scholar] [CrossRef] [PubMed]
- Vanderhaeghen, P.; Lu, Q.; Prakash, N.; Frisén, J.; Walsh, C.A.; Frostig, R.D.; Flanagan, J.G. A mapping Label Required for Normal Scale of Body Representation in the Cortex. Nat. Neurosci. 2000, 3, 358–365. [Google Scholar] [CrossRef]
- Holtmaat, A.J.G.D.; Trachtenberg, J.T.; Wilbrecht, L.; Shepherd, G.M.; Zhang, X.; Knott, G.W.; Svoboda, K. Transient and Persistent Dendritic Spines in the Neocortex In Vivo. Neuron 2005, 45, 279–291. [Google Scholar] [CrossRef]
- Zuo, Y.; Lin, A.; Chang, P.; Gan, W.B. Development of Long-Term Dendritic Spine Stability in Diverse Regions of Cerebral Cortex. Neuron 2005, 46, 181–189. [Google Scholar] [CrossRef]
- Yu, X.; Wang, G.; Gilmore, A.; Yee, A.X.; Li, X.; Xu, T.; Smith, S.J.; Chen, L.; Zuo, Y. Accelerated Experience-Dependent Pruning of Cortical Synapses in Ephrin-A2 Knockout Mice. Neuron 2013, 80, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, L.M.; Liu, H. Comparison of Ephrin-A Ligand and EphA Receptor Distribution in the Developing Inner Ear. Anat. Rec. 1999, 254, 127–134. [Google Scholar] [CrossRef]
- Bianchi, L.M.; Gale, N.W. Distribution of Eph-related molecules in the developing and mature cochlea. Hear. Res. 1998, 117, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Defourny, J.; Poirrier, A.L.; Lallemend, F.; Sánchez, S.M.; Neef, J.; Vanderhaeghen, P.; Soriano, E.; Peuckert, C.; Kullander, K.; Fritzsch, B.; et al. Ephrin-A5/EphA4 signalling controls specific afferent targeting to cochlear hair cells. Nat. Commun. 2013, 4, 1438. [Google Scholar] [CrossRef] [PubMed]
- Pickles, J.O.; Claxton, C.; van Heumen, W.R.A. Complementary and layered expression of Ephs and ephrins in developing mouse inner ear. J. Comp. Neurol. 2002, 449, 207–216. [Google Scholar] [CrossRef]
- Saeger, B.M.; Suhm, M.; Neubüser, A. Ephrin/ephrin receptor expression during early stages of mouse inner ear development. Dev. Dyn. 2011, 240, 1578–1585. [Google Scholar] [CrossRef]
- Zhang, J.H.; Cerretti, D.P.; Yu, T.; Flanagan, J.G.; Zhou, R. Detection of Ligands in Regions Anatomically Connected to Neurons Expressing the Eph Receptor Bsk: Potential Roles in Neuron-Target Interaction. J. Neurosci. 1996, 16, 7182–7192. [Google Scholar] [CrossRef] [PubMed]
- Yates, N.; Robertson, D.; Martin-Iverson, M.; Rodger, J. Auditory brainstem responses of ephrin-A2, Ephrin-A5-/- and Ephrin-A2A5−/− Mice. Audiol. Neurotol. 2014, 19, 115–126. [Google Scholar] [CrossRef]
- Schaette, R.; McAlpine, D. Tinnitus with a Normal Audiogram: Physiological Evidence for Hidden Hearing Loss and Computational Model. J. Neurosci. 2011, 31, 13452–13457. [Google Scholar] [CrossRef] [PubMed]
- Abdul-latif, M.L.; Salazar, J.A.A.; Marshak, S.; Dinh, M.L.; Cramer, K.S. Ephrin-A2 and ephrin-A5 guide contralateral targeting but not topographic mapping of ventral cochlear nucleus axons. Neural Dev. 2015, 10, 27. [Google Scholar] [CrossRef]
- Hoshino, N.; Altarshan, Y.; Alzein, A.; Fernando, A.M.; Nguyen, H.T.; Majewski, E.F.; Chen, V.C.; Rochlin, M.W.; Yu, W. Ephrin-A3 is required for tonotopic map precision and auditory functions in the mouse auditory brainstem. J. Comp. Neurol. 2021, 529, 3633–3654. [Google Scholar] [CrossRef]
- Knoll, B.; Zarbalis, K.; Wurst, W.; Drescher, U. A role for the EphA family in the topographic targeting of vomeronasal axons. Development 2001, 128, 895–906. [Google Scholar] [CrossRef] [PubMed]
- Holmberg, J.; Frisén, J. Ephrins are not only unattractive. Trends Neurosci. 2002, 25, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Drager, U.C.; Hubel, D.H. Topography of visual and somatosensory projections to mouse superior colliculus. J. Neurophysiol. 1976, 39, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Rhoades, R.W.; Mooney, R.D.; Fish, S.E. Subcortical Projections of Area 17 in the Anophthalmic Mouse. Dev. Brain Res. 1985, 17, 171–181. [Google Scholar] [CrossRef]
- Triplett, J.W.; Phan, A.; Yamada, J.; Feldheim, D.A. Alignment of Multimodal Sensory Input in the Superior Colliculus through a Gradient-Matching Mechanism. J. Neurosci. 2012, 32, 5264–5271. [Google Scholar] [CrossRef] [PubMed]
- Champoux, F.; Bacon, B.A.; Lepore, F.; Guillemot, J.P. Effects of early binocular enucleation on auditory and somatosensory coding in the superior colliculus of the rat. Brain Res. 2008, 1191, 84–95. [Google Scholar] [CrossRef]
- Benedetti, F. The development of the somatosensory representation in the superior colliculus of visually deprived mice. Dev. Brain Res. 1992, 65, 173–178. [Google Scholar] [CrossRef]
- Rhoades, R.W. Effects of neonatal enucleation on the functional organization of the superior colliculus in the golden hamster. J. Physiol. 1980, 301, 383–399. [Google Scholar] [CrossRef]
- Hjorth, J.J.J.; Sterratt, D.C.; Cutts, C.S.; Willshaw, D.J.; Eglen, S.J. Quantitative assessment of computational models for retinotopic map formation. Dev. Neurobiol. 2015, 75, 641–666. [Google Scholar] [CrossRef]
- Schneider, G.E. Two Visual Systems. Science 1969, 163, 895–902. [Google Scholar] [CrossRef]
- Gianfranceschi, L.; Fiorentini, A.; Maffei, L. Behavioural visual acuity of wild type and bcl2 transgenic mouse. Vis. Res. 1999, 39, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Prusky, G.T.; West, P.W.R.; Douglas, R.M. Behavioral assessment of visual acuity in mice and rats. Vis. Res. 2000, 40, 2201–2209. [Google Scholar] [CrossRef]
- Avitan, L.; Pujic, Z.; Hughes, N.J.; Scott, E.K.; Goodhill, G.J. Limitations of Neural Map Topography for Decoding Spatial Information. J. Neurosci. 2016, 36, 5385–5396. [Google Scholar] [CrossRef]
- Schneider, G.E. Contrasting visuomotor functions of tectum and cortex in the golden hamster. Psychol. Forsch. 1967, 31, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Balkema, G.; Pinto, L.; Drager, U.; Vanable, J. Characterization of abnormalities in the visual system of the mutant mouse pearl. J. Neurosci. 1981, 1, 1320–1329. [Google Scholar] [CrossRef]
- Arnall, S.; Cheam, L.Y.; Smart, C.; Rengel, A.; Fitzgerald, M.; Thivierge, J.P.; Rodger, J. Abnormal strategies during visual discrimination reversal learning in ephrin-A2−/− mice. Behav. Brain Res. 2010, 209, 109–113. [Google Scholar] [CrossRef]
- Moretti, J.; Poh, E.Z.; Bolland, S.J.; Harvey, A.R.; Albrecht, M.A.; Rodger, J. Concurrent LI-rTMS induces changes in c-Fos expression but not behavior during a progressive ratio task with adult ephrin-A2A5−/− mice. Behav. Brain Res. 2021, 400, 113011. [Google Scholar] [CrossRef] [PubMed]
- Wurzman, R.; Forcelli, P.A.; Griffey, C.J.; Kromer, L.F. Repetitive grooming and sensorimotor abnormalities in an ephrin-A knockout model for Autism Spectrum Disorders. Behav. Brain Res. 2015, 278, 115–128. [Google Scholar] [CrossRef] [PubMed]
- Sheleg, M.; Yochum, C.L.; Richardson, J.R.; Wagner, G.C.; Zhou, R. Ephrin-A5 regulates inter-male aggression in mice. Behav. Brain Res. 2015, 286, 300–307. [Google Scholar] [CrossRef]
- Sheleg, M.; Yu, Q.; Go, C.; Wagner, G.C.; Kusnecov, A.W.; Zhou, R. Decreased maternal behavior and anxiety in ephrin-A5−/− mice. Genes Brain Behav. 2017, 16, 271–284. [Google Scholar] [CrossRef]
- Makowiecki, K.; Garrett, A.; Harvey, A.R.; Rodger, J. Low-intensity repetitive transcranial magnetic stimulation requires concurrent visual system activity to modulate visual evoked potentials in adult mice. Sci. Rep. 2018, 8, 271–284. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, T.; Torborg, C.L.; Feller, M.B.; O’Leary, D.D.M. Retinotopic Map Refinement Requires Spontaneous Retinal Waves during a Brief Critical Period of Development. Neuron 2003, 40, 1147–1160. [Google Scholar] [CrossRef] [PubMed]
- Moretti, J.; Rodger, J. A little goes a long way: Neurobiological effects of low intensity rTMS and implications for mechanisms of rTMS. Curr. Res. Neurobiol. 2022, 3, 100033. [Google Scholar] [CrossRef]
- Ridding, M.C.; Rothwell, J.C. Is there a future for therapeutic use of transcranial magnetic stimulation? Nat. Rev. Neurosci. 2007, 8, 559–567. [Google Scholar] [CrossRef]
- Siebner, H.R.; Funke, K.; Aberra, A.S.; Antal, A.; Bestmann, S.; Chen, R.; Classen, J.; Davare, M.; Di Lazzaro, V.; Fox, P.T.; et al. Transcranial magnetic stimulation of the brain: What is stimulated?—A consensus and critical position paper. Clin. Neurophysiol. 2022, 140, 59–97. [Google Scholar] [CrossRef]
- Huang, Y.Z.; Edwards, M.J.; Rounis, E.; Bhatia, K.P.; Rothwell, J.C. Theta Burst Stimulation of the Human Motor Cortex. Neuron 2005, 45, 201–206. [Google Scholar] [CrossRef]
- Sydnor, V.J.; Cieslak, M.; Duprat, R.; Deluisi, J.; Flounders, M.W.; Long, H.; Scully, M.; Balderston, N.L.; Sheline, Y.I.; Bassett, D.S.; et al. Cortical-subcortical structural connections support transcranial magnetic stimulation engagement of the amygdala. Sci. Adv. 2022, 8, eabn5803. [Google Scholar] [CrossRef] [PubMed]
- Seewoo, B.J.; Feindel, K.W.; Etherington, S.J.; Rodger, J. Resting-state fMRI study of brain activation using low-intensity repetitive transcranial magnetic stimulation in rats. Sci. Rep. 2018, 8, 6706. [Google Scholar] [CrossRef] [PubMed]
- Seewoo, B.J.; Feindel, K.W.; Etherington, S.J.; Rodger, J. Frequency-specific effects of low-intensity rTMS can persist for up to 2 weeks post-stimulation: A longitudinal rs-fMRI/MRS study in rats. Brain Stimul. 2019, 12, 1526–1536. [Google Scholar] [CrossRef]
- Madore, M.R.; Poh, E.; Bollard, S.J.; Rivera, J.; Taylor, J.; Cheng, J.; Booth, E.; Nable, M.; Heath, A.; Yesavage, J.; et al. Moving back in the brain to drive the field forward: Targeting neurostimulation to different brain regions in animal models of depression and neurodegeneration. J. Neurosci. Methods 2021, 360, 109261. [Google Scholar] [CrossRef]
- Tik, M.; Hoffmann, A.; Sladky, R.; Tomova, L.; Hummer, A.; Navarro de Lara, L.; Bukowski, H.; Pripfl, J.; Biswal, B.; Lamm, C.; et al. Towards understanding rTMS mechanism of action: Stimulation of the DLPFC causes network-specific increase in functional connectivity. Neuroimage 2017, 162, 289–296. [Google Scholar] [CrossRef] [PubMed]
- O’Reardon, J.P.; Solvason, H.B.; Janicak, P.G.; Sampson, S.; Isenberg, K.E.; Nahas, Z.; McDonald, W.M.; Avery, D.; Fitzgerald, P.B.; Loo, C.; et al. Efficacy and Safety of Transcranial Magnetic Stimulation in the Acute Treatment of Major Depression: A Multisite Randomized Controlled Trial. Biol. Psychiatry 2007, 62, 1208–1216. [Google Scholar] [CrossRef] [PubMed]
- Carmi, L.; Alyagon, U.; Barnea-Ygael, N.; Zohar, J.; Dar, R.; Zangen, A. Clinical and electrophysiological outcomes of deep TMS over the medial prefrontal and anterior cingulate cortices in OCD patients. Brain Stimul. 2018, 11, 158–165. [Google Scholar] [CrossRef] [PubMed]
- van Praag, H.; Kempermann, G.; Gage, F.H. Neural consequences of enviromental enrichment. Nat. Rev. Neurosci. 2000, 1, 191–198. [Google Scholar] [CrossRef]
- Green, E.J.; Greenough, W.T. Altered synaptic transmission in dentate gyrus of rats reared in complex environments: Evidence from hippocampal slices maintained in vitro. J. Neurophysiol. 1986, 55, 739–750. [Google Scholar] [CrossRef]
- Foster, T.C.; Fugger, H.N.; Cunningham, S.G. Receptor blockade reveals a correspondence between hippocampal-dependent behavior and experience-dependent synaptic enhancement. Brain Res. 2000, 871, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Sharp, P.E.; McNaughton, B.L.; Barnes, C.A. Enhancement of hippocampal field potentials in rats exposed to a novel, complex environment. Brain Res. 1985, 339, 361–365. [Google Scholar] [CrossRef]
- Milighetti, S.; Sterzi, S.; Fregni, F.; Hanlon, C.A.; Hayley, P.; Murphy, M.D.; Bundy, D.T.; Nudo, R.J.; Guggenmos, D.J. Effects of tDCS on spontaneous spike activity in a healthy ambulatory rat model. Brain Stimul. 2020, 13, 1566–1576. [Google Scholar] [CrossRef]
- Tanaka, T.; Isomura, Y.; Kobayashi, K.; Hanakawa, T.; Tanaka, S.; Honda, M. Electrophysiological Effects of Transcranial Direct Current Stimulation on Neural Activity in the Rat Motor Cortex. Front. Neurosci. 2020, 14, 495. [Google Scholar] [CrossRef]
- Tang, A.; Thickbroom, G.; Rodger, J. Repetitive Transcranial Magnetic Stimulation of the Brain. Neuroscientist 2017, 23, 82–94. [Google Scholar] [CrossRef]
- Boyer, M.; Baudin, P.; Stengel, C.; Valero-Cabré, A.; Lohof, A.M.; Charpier, S.; Sherrard, R.M.; Mahon, S. In vivo low-intensity magnetic pulses durably alter neocortical neuron excitability and spontaneous activity. J. Physiol. 2022, 600, 4019–4037. [Google Scholar] [CrossRef] [PubMed]
- Rodger, J.; Mo, C.; Wilks, T.; Dunlop, S.A.; Sherrard, R.M. Transcranial pulsed magnetic field stimulation facilitates reorganization of abnormal neural circuits and corrects behavioral deficits without disrupting normal connectivity. FASEB J. 2012, 26, 1593–1606. [Google Scholar] [CrossRef]
- Martiny, K.; Lunde, M.; Bech, P. Transcranial Low Voltage Pulsed Electromagnetic Fields in Patients with Treatment-Resistant Depression. Biol. Psychiatry 2010, 68, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Makowiecki, K.; Harvey, A.R.; Sherrard, R.M.; Rodger, J. Low-Intensity Repetitive Transcranial Magnetic Stimulation Improves Abnormal Visual Cortical Circuit Topography and Upregulates BDNF in Mice. J. Neurosci. 2014, 34, 10780–10792. [Google Scholar] [CrossRef] [PubMed]
- Poh, E.Z.; Harvey, A.R.; Makowiecki, K.; Rodger, J. Online LI-rTMS during a Visual Learning Task: Differential Impacts on Visual Circuit and Behavioral Plasticity in Adult Ephrin-A2A5−/− Mice. eNeuro 2018, 5, ENEURO.0163-17.2018. [Google Scholar] [CrossRef]
- Poh, E.Z.; Green, C.; Agostinelli, L.; Penrose-Menz, M.; Karl, A.K.; Harvey, A.R.; Rodger, J. Manipulating the Level of Sensorimotor Stimulation during LI-rTMS Can Improve Visual Circuit Reorganisation in Adult Ephrin-A2A5−/− Mice. Int. J. Mol. Sci. 2022, 23, 2418. [Google Scholar] [CrossRef]
- Singh, K.K.; Park, K.J.; Hong, E.J.; Kramer, B.M.; Greenberg, M.E.; Kaplan, D.R.; Miller, F.D. Developmental axon pruning mediated by BDNF-p75NTR-dependent axon degeneration. Nat. Neurosci. 2008, 11, 649–658. [Google Scholar] [CrossRef] [PubMed]
- Ugolini, G.; Cremisi, F.; Maffei, L. TrkA, TrkB and ~75 mRNA expression is developmentally regulated in the rat retina. Brain Res. 1995, 704, 121–124. [Google Scholar] [CrossRef] [PubMed]
- Frost, D.O. BDNF/trkB signaling in the developmental sculpting of visual connections. In Progress in Brain Research; Elsevier: Amsterdam, The Netherlands, 2001; pp. 35–49. [Google Scholar]
- Mu, Y.; Poo, M.M. Spike Timing-Dependent LTP/LTD Mediates Visual Experience-Dependent Plasticity in a Developing Retinotectal System. Neuron 2006, 50, 115–125. [Google Scholar] [CrossRef]
- Davey, F.; Davies, A.M. TrkB signalling inhibits p75-mediated apoptosis induced by nerve growth factor in embryonic proprioceptive neurons. Curr. Biol. 1998, 8, 915–918. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Cory, S.; Kidane, A.H.; Shirkey, N.J.; Marshak, S. Brain-derived neurotrophic factor and the development of structural neuronal connectivity. Dev. Neurobiol. 2010, 70, 271–288. [Google Scholar] [CrossRef]
- Huang, Z.J.; Kirkwood, A.; Pizzorusso, T.; Porciatti, V.; Morales, B.; Bear, M.F.; Maffei, L.; Tonegawa, S. BDNF Regulates the Maturation of Inhibition and the Critical Period of Plasticity in Mouse Visual Cortex. Cell 1999, 98, 739–755. [Google Scholar] [CrossRef] [PubMed]
- Kentner, A.C.; Lambert, K.G.; Hannan, A.J.; Donaldson, S.T. Editorial: Environmental Enrichment: Enhancing Neural Plasticity, Resilience, and Repair. Front. Behav. Neurosci. 2019, 13, 75. [Google Scholar] [CrossRef] [PubMed]
- Vetencourt, J.F.M.; Sale, A.; Viegi, A.; Baroncelli, L.; De Pasquale, R.; O’Leary, O.F.; Castrén, E.; Maffei, L. The Antidepressant Fluoxetine Restores Plasticity in the Adult Visual Cortex. Science 2008, 320, 385–388. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomar, M.; Beros, J.; Meloni, B.; Rodger, J. Interactions between Guidance Cues and Neuronal Activity: Therapeutic Insights from Mouse Models. Int. J. Mol. Sci. 2023, 24, 6966. https://doi.org/10.3390/ijms24086966
Tomar M, Beros J, Meloni B, Rodger J. Interactions between Guidance Cues and Neuronal Activity: Therapeutic Insights from Mouse Models. International Journal of Molecular Sciences. 2023; 24(8):6966. https://doi.org/10.3390/ijms24086966
Chicago/Turabian StyleTomar, Maitri, Jamie Beros, Bruno Meloni, and Jennifer Rodger. 2023. "Interactions between Guidance Cues and Neuronal Activity: Therapeutic Insights from Mouse Models" International Journal of Molecular Sciences 24, no. 8: 6966. https://doi.org/10.3390/ijms24086966
APA StyleTomar, M., Beros, J., Meloni, B., & Rodger, J. (2023). Interactions between Guidance Cues and Neuronal Activity: Therapeutic Insights from Mouse Models. International Journal of Molecular Sciences, 24(8), 6966. https://doi.org/10.3390/ijms24086966






