Antifungal Activity of 2-Allylphenol Derivatives on the Botrytis cinerea Strain: Assessment of Possible Action Mechanism
Abstract
1. Introduction
2. Results and Discussion
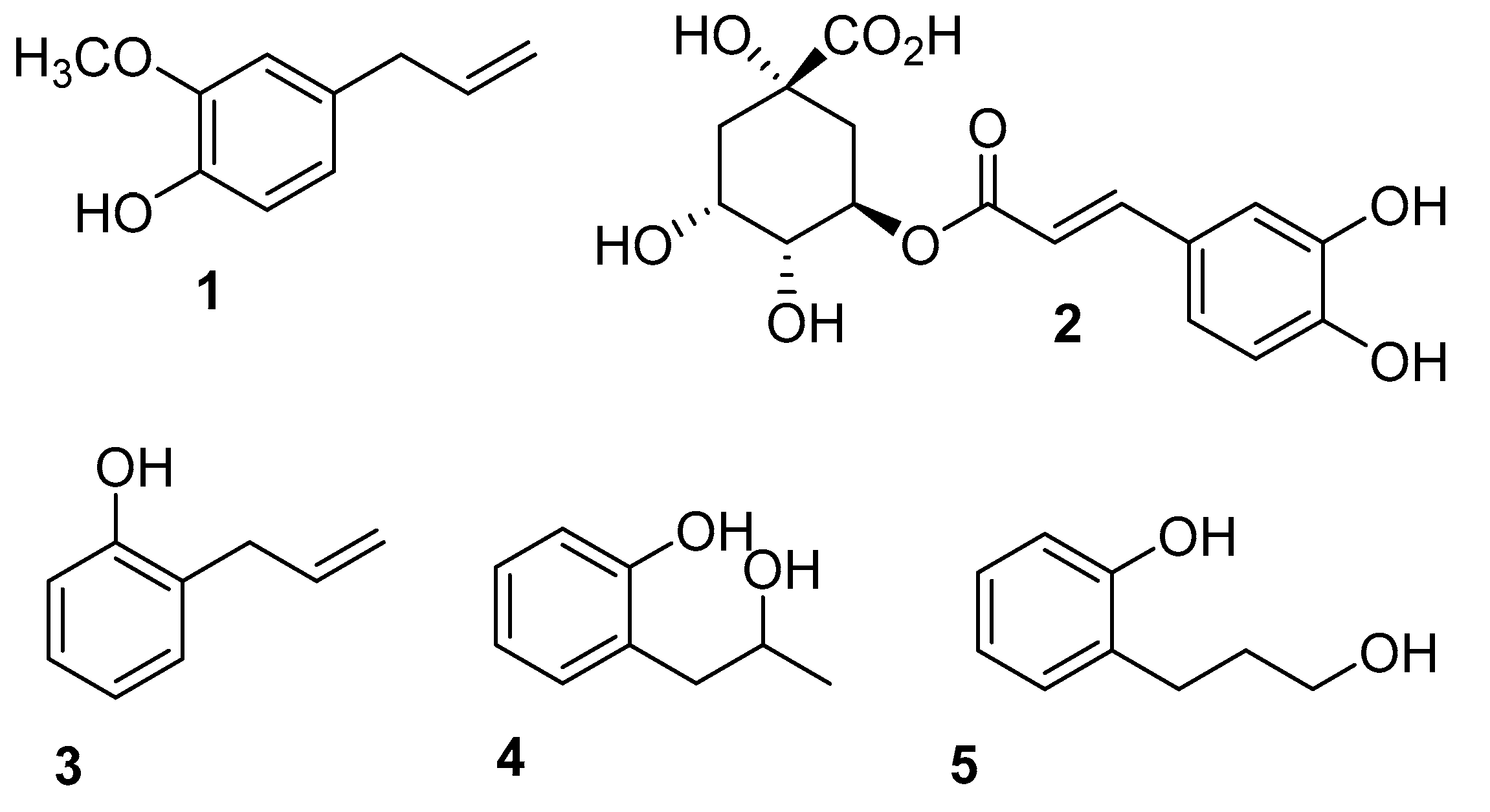
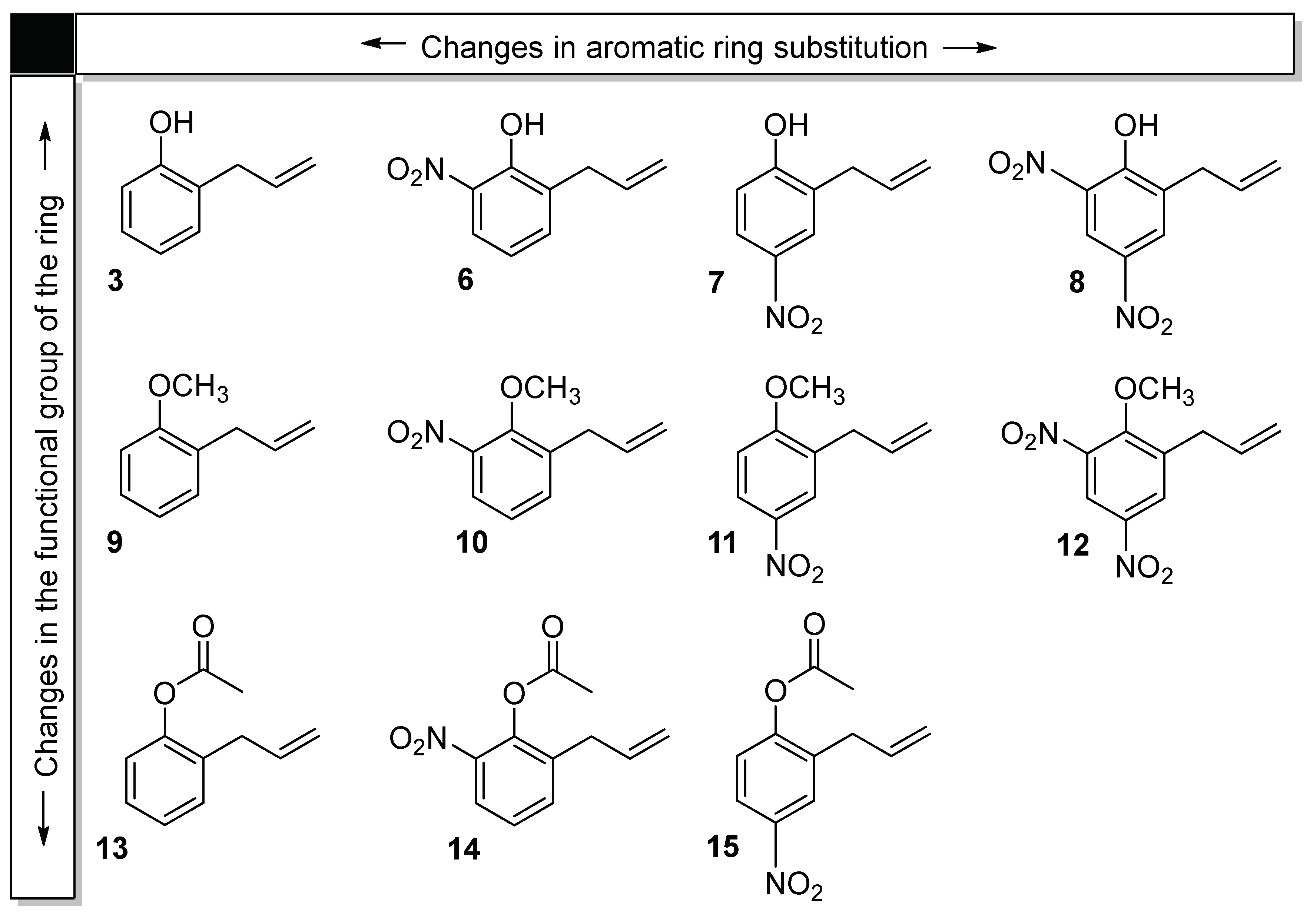
2.1. Synthesis of 2-Allylphenol Derivatives
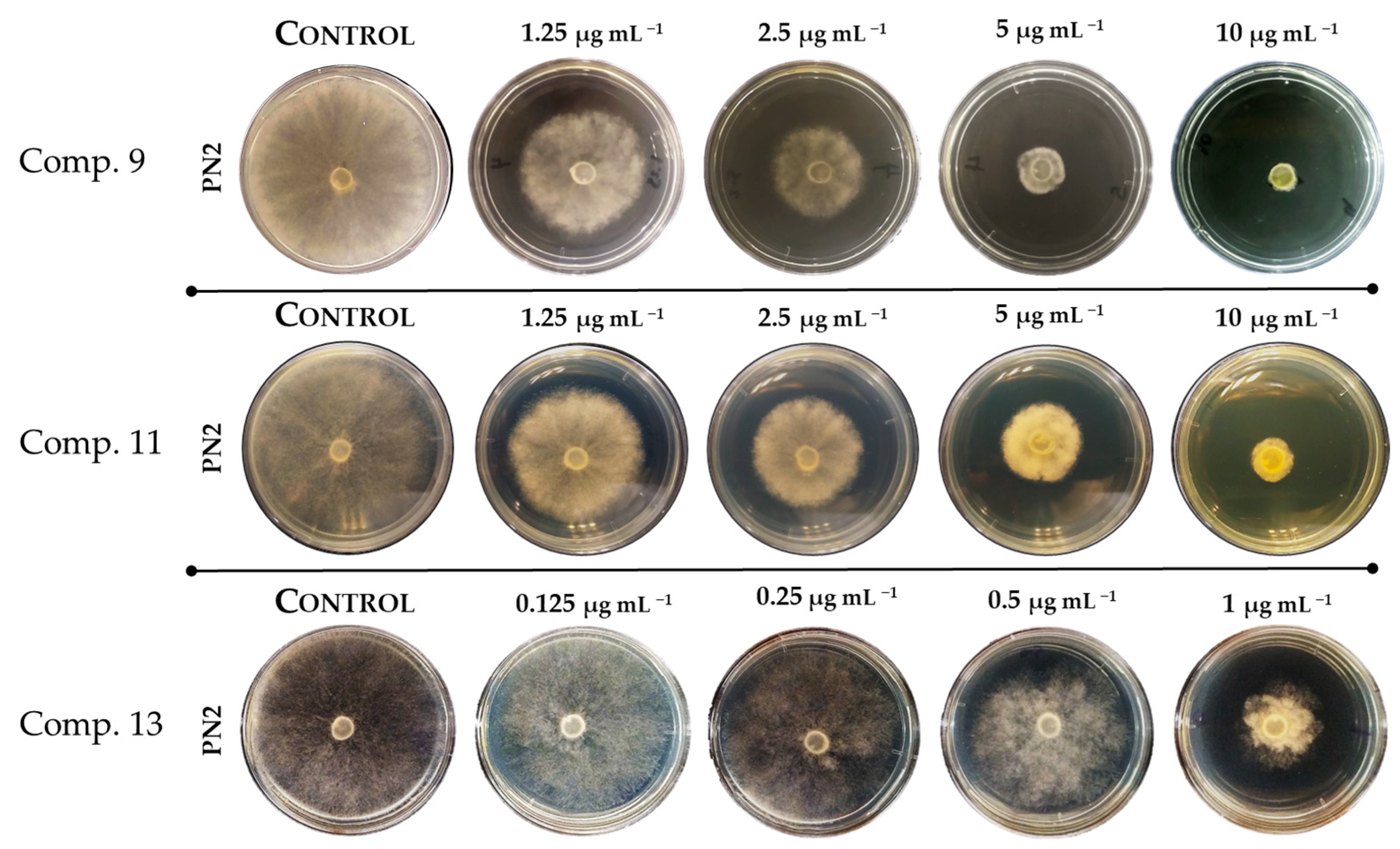
2.2. Evaluation of Antifungal Activity of 2-Allylphenol Derivatives on Botrytis cinerea
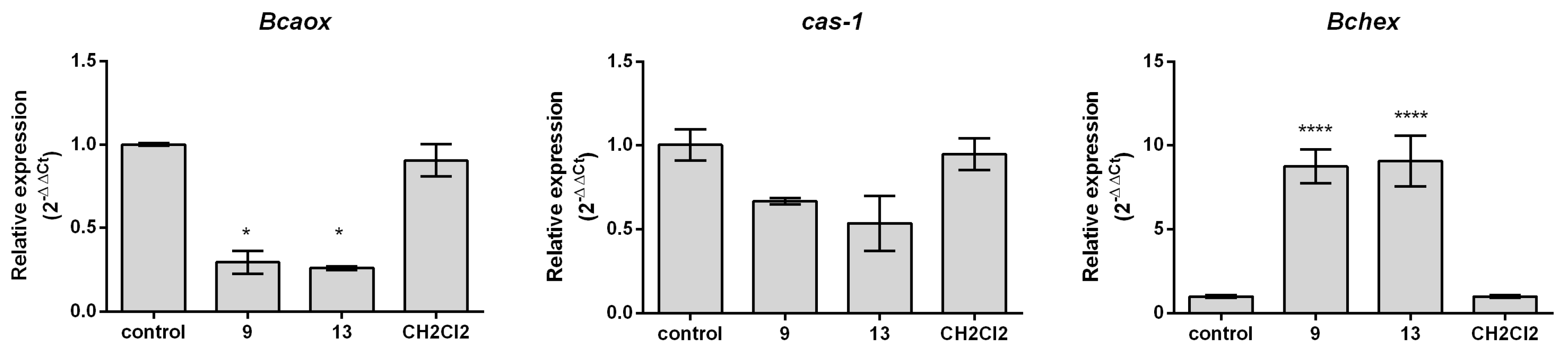
2.3. Effect of 2-Allylphenol Derivatives on Gene Expression
3. Methods and Materials
3.1. General Information
3.2. Chemistry
Synthesis of 1-Allyl-2-methoxy-5-nitrobenzene (11)
3.3. Biological
3.3.1. Fungal Isolate and Culture Conditions
3.3.2. Effect of 2-Allylphenol Derivatives on Mycelial Growth
3.4. Effect on Gene Expression
RNA Extraction and qRT-PCR Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fillinger, S.; Elad, Y. Botrytis—The Fungus, the Pathogen and Its Management in Agricultural Systems; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Fillinger, S.; Walker, A.S. Chemical Control and Resistance Management of Botrytis Diseases. In Botrytis—The Fungus, the Pathogen and Its Management in Agricultural Systems; Fillinger, S., Elad, Y., Eds.; Springer: Cham, Switzerland, 2016; p. 189. [Google Scholar]
- Leroux, P. Chemical control of Botrytis and its resistance to chemical fungicides. In Botrytis: Biology, Pathology and Control; Elad, Y., Williamson, B., Tudzynski, P., Delen, N., Eds.; Kluwer Academic Press: Dordrecht, The Netherlands, 2004; pp. 195–222. [Google Scholar]
- Williamson, B.; Tudzynsk, B.; Tudzynski, P.; van Kan, J.A.L. Botrytis cinerea: The cause of grey mould disease. Mol. Plant Pathol. 2007, 8, 561–580. [Google Scholar] [CrossRef] [PubMed]
- Leroch, M.; Kretschmer, M.; Hahn, M. Fungicide resistance phenotypes of Botrytis cinerea isolates from commercial vineyards in South West Germany. J. Phytopathol. 2011, 159, 63–65. [Google Scholar] [CrossRef]
- Leroux, P.; Fritz, R.; Debieu, D.; Albertini, C.; Lanen, C.; Bach, J.; Gredt, M.; Chapeland, F. Mechanisms of resistance to fungicides in field strains of Botrytis cinerea. Pest Manag. Sci. 2002, 58, 876–888. [Google Scholar] [CrossRef] [PubMed]
- Leroux, P.; Walker, A.S. Activity of fungicides and modulators of membrane drug transporters in field strains of Botrytis cinerea displaying multidrug resistance. Eur. J. Plant Pathol. 2013, 135, 683–693. [Google Scholar] [CrossRef]
- Kretschmer, M.; Leroch, M.; Mosbach, A.; Walker, A.S.; Fillinger, S.; Mernke, D.; Schoonbeek, H.J.; Pradier, J.M.; Leroux, P.; De Waard, M.A.; et al. Fungicide-Driven Evolution and Molecular Basis of Multidrug Resistance in Field Populations of the Grey Mould Fungus Botrytis cinerea. PLoS Pathog. 2009, 5, e1000696. [Google Scholar] [CrossRef]
- Latorre, B.A.; Torres, R. Prevalence of isolates of Botrytis cinerea resistant to multiple fungicides in Chilean vineyards. Crop Prot. 2012, 40, 49–52. [Google Scholar] [CrossRef]
- Fernandez-Ortuno, D.; Grabke, A.; Li, X.; Schnabel, G. Independent Emergence of Resistance to Seven Chemical Classes of Fungicides in Botrytis cinerea. Phytopathology 2015, 105, 424–432. [Google Scholar] [CrossRef]
- Cragg, G.M.; Newman, D.J. Natural products: A continuing source of novel drug leads. Biochim. Biophys. Acta-Gen. Subj. 2013, 1830, 3670–3695. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Tripathi, P.; Dubey, N. Exploitation of natural products as an alternative strategy to control postharvest fungal rotting of fruit and vegetables. Postharvest Biol. Technol. 2004, 32, 235–245. [Google Scholar] [CrossRef]
- Dayan, F.E.; Cantrell, C.L.; Duke, S.O. Natural products in crop protection. Bioorg. Med. Chem. 2009, 17, 4022–4034. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, P.; Dubey, N.K.; Shukla, A.K. Use of some essential oils as post-harvest botanical fungicides in the management of grey mould of grapes caused by Botrytis cinerea. World J. Microbiol. Biotechnol. 2008, 24, 39–46. [Google Scholar] [CrossRef]
- Nxumalo, K.A.; Aremu, A.O.; Fawole, O.A. Potentials of Medicinal Plant Extracts as an Alternative to Synthetic Chemicals in Postharvest Protection and Preservation of Horticultural Crops: A Review. Sustainability 2021, 13, 5897. [Google Scholar] [CrossRef]
- Gahukar, R.T. Evaluation of plant-derived products against pests and diseases of medicinal plants: A review. Crop Prot. 2012, 42, 202–209. [Google Scholar] [CrossRef]
- Castillo, F.; Hernández, D.; Gallegos, G.; Rodríguez, R.; Aguilar, C.N. Antifungal Properties of Bioactive Compounds from Plants. In Fungicides for Plant and Animal Diseases; Dhanasekaran, D., Thajuddin, N., Panneerselvam, A., Eds.; IntechOpen: Rijeka, Croatia, 2012; Chapter 4. [Google Scholar]
- Gurjar, M.S.; Ali, S.; Akthar, M.; Singh, K.S. Efficacy of plant extracts in plant disease management. Agric. Sci. 2012, 3, 425–433. [Google Scholar] [CrossRef]
- Jimenez-Reyes, M.F.; Carrasco, H.; Olea, A.F.; Silva Moreno, E. Natural compounds: A sustainable alternative to the phytopathogens control. J. Chil. Chem. Soc. 2019, 64, 4459–4465. [Google Scholar] [CrossRef]
- Espinoza, L.; Taborga, L.; Diaz, K.; Olea, A.F.; Peña-Cortes, H. Synthesis of linear geranylphenols and their effect on mycelial growth of plant pathogen Botrytis cinerea. Molecules 2014, 19, 1512–1526. [Google Scholar] [CrossRef]
- Taborga, L.; Diaz, K.; Olea, A.F.; Reyes-Bravo, P.; Flores, M.E.; Pena-Cortes, H.; Espinoza, L. Effect of Polymer Micelles on Antifungal Activity of Geranylorcinol Compounds against Botrytis cinerea. J. Agric. Food Chem. 2015, 63, 6890–6896. [Google Scholar] [CrossRef]
- Chavez, M.I.; Soto, M.; Taborga, L.; Diaz, K.; Olea, A.F.; Bay, C.; Pena-Cortes, H.; Espinoza, L. Synthesis and in vitro antifungal activity against Botrytis cinerea of geranylated phenols and their phenyl acetate derivatives. Int. J. Mol. Sci. 2015, 16, 19130–19152. [Google Scholar] [CrossRef]
- Carrasco, H.; Robles-Kelly, C.; Rubio, J.; Olea, A.F.; Martínez, R.; Silva-Moreno, E. Antifungal effect of polygodial on Botrytis cinerea, a fungal pathogen affecting table grapes. Int. J. Mol. Sci. 2017, 18, 2251. [Google Scholar] [CrossRef]
- Robles-Kelly, C.; Rubio, J.; Thomas, M.; Sedan, C.; Martinez, R.; Olea, A.F.; Carrasco, H.; Taborga, L.; Silva-Moreno, E. Effect of drimenol and synthetic derivatives on growth and germination of Botrytis cinerea: Evaluation of possible mechanism of action. Pestic. Biochem. Physiol. 2017, 141, 50–56. [Google Scholar] [CrossRef]
- Cotoras, M.; Folch, C.; Mendoza, L. Characterization of the antifungal activity on Botrytis cinerea of the natural diterpenoids kaurenoic acid and 3 beta-hydroxy-kaurenoic acid. J. Agric. Food Chem. 2004, 52, 2821–2826. [Google Scholar] [CrossRef] [PubMed]
- Melo, R.; Armstrong, V.; Navarro, F.; Castro, P.; Mendoza, L.; Cotoras, M. Characterization of the Fungitoxic Activity on Botrytis cinerea of N-phenyl-driman-9-carboxamides. J. Fungi 2021, 7, 902. [Google Scholar] [CrossRef] [PubMed]
- Dan, W.J.; Tuong, T.M.L.; Wang, D.C.; Li, D.; Zhang, A.L.; Gao, J.M. Natural products as sources of new fungicides (V): Design and synthesis of acetophenone derivatives against phytopathogenic fungi in vitro and in vivo. Bioorg. Med. Chem. Lett. 2018, 28, 2861–2864. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; He, Y.H.; Hu, Y.M.; Chu, Q.R.; Chen, Y.J.; Wu, Z.R.; Zhang, Z.J.; Liu, Y.Q.; Yang, C.J.; Liang, H.J.; et al. Design, Synthesis, and Structure-Activity Relationship Studies of Magnolol Derivatives as Antifungal Agents. J. Agric. Food Chem. 2021, 69, 11781–11793. [Google Scholar] [CrossRef]
- Olea, A.F.; Bravo, A.; Martínez, R.; Thomas, M.; Sedan, C.; Espinoza, L.; Zambrano, E.; Carvajal, D.; Silva-Moreno, E.; Carrasco, H. Antifungal Activity of Eugenol Derivatives against Botrytis cinerea. Molecules 2019, 24, 1239. [Google Scholar] [CrossRef]
- Martínez, G.; Regente, M.; Jacobi, S.; Del Rio, M.; Pinedo, M.; de la Canal, L. Chlorogenic acid is a fungicide active against phytopathogenic fungi. Pestic. Biochem. Physiol. 2017, 140, 30–35. [Google Scholar] [CrossRef]
- Yang, L.; Wen, K.S.; Ruan, X.; Zhao, Y.X.; Wei, F.; Wang, Q. Response of Plant Secondary Metabolites to Environmental Factors. Molecules 2018, 23, 762. [Google Scholar] [CrossRef]
- Carrasco, H.; Espinoza, L.; Cardile, V.; Gallardo, C.; Cardona, W.; Lombardo, L.; Catalan, K.; Russo, A. Eugenol and its synthetic analogues inhibit cell growth of human cancer cells (Part I). J. Braz. Chem. Soc. 2008, 19, 543–548. [Google Scholar] [CrossRef]
- Carrasco, H.; Raimondi, M.; Svetaz, L.; Di Liberto, M.; Rodriguez, M.V.; Espinoza, L.; Madrid, A.; Zacchino, S. Antifungal activity of eugenol analogues. Influence of different substituents and studies on mechanism of action. Molecules 2012, 17, 1002–1024. [Google Scholar]
- Hidalgo, M.E.; de la Rosa, C.; Carrasco, H.; Cardona, W.; Gallardo, C.; Espinoza, L. Antioxidant Capacity of Eugenol Derivatives. Quim. Nova 2009, 32, 1467–1470. [Google Scholar] [CrossRef]
- Tajik, N.; Tajik, M.; Mack, I.; Enck, P. The potential effects of chlorogenic acid, the main phenolic components in coffee, on health: A comprehensive review of the literature. Eur. J. Nutr. 2017, 56, 2215–2244. [Google Scholar] [CrossRef] [PubMed]
- Sung, W.S.; Lee, D.G. Antifungal action of chlorogenic acid against pathogenic fungi, mediated by membrane disruption. Pure Appl. Chem. 2010, 82, 219–226. [Google Scholar] [CrossRef]
- Heitman, E.; Ingram, D.K. Cognitive and neuroprotective effects of chlorogenic acid. Nutr. Neurosci. 2017, 20, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Kobayashi, S.; Yamaguchi, T.; Hibi, M.; Fukuhara, I.; Osaki, N. Coffee Abundant in Chlorogenic Acids Reduces Abdominal Fat in Overweight Adults: A Randomized, Double-Blind, Controlled Trial. Nutrients 2019, 11, 1617. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, J.; Chen, H.; Fan, Y.; Shi, Z. Antifungal activity of eugenol against Botrytis cinerea. Trop. Plant Pathol. 2010, 35, 137–143. [Google Scholar] [CrossRef]
- Ma, L.; He, J.; Liu, H.; Zhou, H. The phenylpropanoid pathway affects apple fruit resistance to Botrytis cinerea. J. Phytopathol. 2018, 166, 206–215. [Google Scholar] [CrossRef]
- Meng, Z.; Wei, Y.; Xu, D.; Hao, S.; Hu, J. Effect of 2-allylphenol against Botrytis cinerea Pers., and its residue in tomato fruit. Crop Prot. 2007, 26, 1711–1715. [Google Scholar] [CrossRef]
- Gong, S.J.; Hao, J.J.; Xia, Y.Y.; Liu, X.L.; Li, J.Q. Inhibitory effect of bionic fungicide 2-allylphenol on Botrytis cinerea (Pers. ex Fr.) in vitro. Pest Manag. Sci. 2009, 65, 1337–1343. [Google Scholar] [CrossRef]
- Qu, T.; Gao, S.; Li, J.; Hao, J.J.; Ji, P. Synthesis and antifungal activity of 2-allylphenol derivatives against fungal plant pathogens. Pestic. Biochem. Physiol. 2017, 135, 47–51. [Google Scholar] [CrossRef]
- Olea, A.F.; Espinoza, L.; Sedan, C.; Thomas, M.; Martinez, R.; Mellado, M.; Carrasco, H.; Diaz, K. Synthesis and In Vitro Growth Inhibition of 2-Allylphenol Derivatives Against Phythopthora cinnamomi Rands. Molecules 2019, 24, 4196. [Google Scholar] [CrossRef]
- Stillwell, W. Chapter 18—Membrane-Associated Processes. In An Introduction to Biological Membranes, 2nd ed.; Stillwell, W., Ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 381–421. [Google Scholar]
- Bartlett, D.W.; Clough, J.M.; Godwin, J.R.; Hall, A.A.; Hamer, M.; Parr-Dobrzanski, B. The strobilurin fungicides. Pest Manag. Sci. 2002, 58, 649–662. [Google Scholar] [CrossRef] [PubMed]
- Inoue, K.; Tsurumi, T.; Ishii, H.; Park, P.; Ikeda, K. Cytological evaluation of the effect of azoxystrobin and alternative oxidase inhibitors in Botrytis cinerea. FEMS Microbiol. Lett. 2012, 326, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Leroux, P.; Gredt, M.; Leroch, M.; Walker, A.S. Exploring mechanisms of resistance to respiratory inhibitors in field strains of Botrytis cinerea, the causal agent of gray mold. Appl. Environ. Microbiol. 2010, 76, 6615–6630. [Google Scholar] [CrossRef] [PubMed]
- Joseph-Horne, T.; Hollomon, D.W.; Wood, P.M. Fungal respiration: A fusion of standard and alternative components. Biochim. Biophys. Acta (BBA)-Bioenerg. 2001, 1504, 179–195. [Google Scholar] [CrossRef]
- Li, Q.; Bai, Z.; O’Donnell, A.; Harvey, L.M.; Hoskisson, P.A.; McNeil, B. Oxidative stress in fungal fermentation processes: The roles of alternative respiration. Biotechnol. Lett. 2011, 33, 457–467. [Google Scholar] [CrossRef]
- Van Aken, O.; Giraud, E.; Clifton, R.; Whelan, J. Alternative oxidase: A target and regulator of stress responses. Physiol. Plant. 2009, 137, 354–361. [Google Scholar] [CrossRef]
- Garmash, E.V.; Belykh, E.S.; Velegzhaninov, I.O. The gene expression profiles of mitochondrial respiratory components in Arabidopsis plants with differing amounts of ALTERNATIVE OXIDASE1a under high intensity light. Plant Signal. Behav. 2021, 16, 1864962. [Google Scholar] [CrossRef]
- Lin, Z.; Wu, J.; Jamieson, P.A.; Zhang, C. Alternative Oxidase Is Involved in the Pathogenicity, Development, and Oxygen Stress Response of Botrytis cinerea. Phytopathology 2019, 109, 1679–1688. [Google Scholar] [CrossRef]
- Shlezinger, N.; Minz, A.; Gur, Y.; Hatam, I.; Dagdas, Y.F.; Talbot, N.J.; Sharon, A. Anti-Apoptotic Machinery Protects the Necrotrophic Fungus Botrytis cinerea from Host-Induced Apoptotic-Like Cell Death during Plant Infection. PLoS Pathog. 2011, 7, e1002185. [Google Scholar] [CrossRef]
- Aguayo, C.; Riquelme, J.; Valenzuela, P.; Hahn, M.; Silva Moreno, E. Bchex virulence gene of Botrytis cinerea: Characterization and functional analysis. J. Gen. Plant Pathol. 2011, 77, 230–238. [Google Scholar] [CrossRef]
- Collinge, A.J.; Markham, P. Woronin Bodies Rapidly Plug Septal Pores of Severed Penicillium-Chrysogenum Hyphae. Exp. Mycol. 1985, 9, 80–85. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Barsottini, M.R.; Pires, B.A.; Vieira, M.L.; Pereira, J.G.; Costa, P.C.; Sanitá, J.; Coradini, A.; Mello, F.; Marschalk, C.; Silva, E.M.; et al. Synthesis and testing of novel alternative oxidase (AOX) inhibitors with antifungal activity against Moniliophthora perniciosa (Stahel), the causal agent of witches’ broom disease of cocoa, and other phytopathogens. Pest Manag. Sci. 2019, 75, 1295–1303. [Google Scholar] [CrossRef]
- Tian, F.; Lee, S.Y.; Woo, S.Y.; Chun, H.S. Alternative Oxidase: A Potential Target for Controlling Aflatoxin Contamination and Propagation of Aspergillus flavus. Front. Microbiol. 2020, 11, 419. [Google Scholar] [CrossRef] [PubMed]
- Wood, P.M.; Hollomon, D.W. A critical evaluation of the role of alternative oxidase in the performance of strobilurin and related fungicides acting at the Qo site of complex III. Pest Manag. Sci. 2003, 59, 499–511. [Google Scholar] [CrossRef]
- Díaz, K.; Espinoza, L.; Carvajal, R.; Silva-Moreno, E.; Olea, A.F.; Rubio, J. Exogenous Application of Brassinosteroid 24-Norcholane 22(S)-23-Dihydroxy Type Analogs to Enhance Water Deficit Stress Tolerance in Arabidopsis thaliana. Int. J. Mol. Sci. 2021, 22, 1158. [Google Scholar] [CrossRef]
- Yamashita, Y.; Ota, M.; Inoue, Y.; Hasebe, Y.; Okamoto, M.; Inukai, T.; Masuta, C.; Sakihama, Y.; Hashidoko, Y.; Kojima, M.; et al. Chemical Promotion of Endogenous Amounts of ABA in Arabidopsis thaliana by a Natural Product, Theobroxide. Plant Cell Physiol. 2016, 57, 986–999. [Google Scholar] [CrossRef]
| 2-Allylphenol Derivatives | PN2 |
|---|---|
| IC50 (μg mL−1) | |
| 3 | 68.0 ± 6.40 |
| 6 | 133 ± 12.8 |
| 7 | 13.0 ± 1.50 |
| 8 | 136 ± 11.5 |
| 9 | 2.00 ± 0.32 |
| 10 | 55.0 ± 4.85 |
| 11 | 3.60 ± 0.28 |
| 12 | 12.0 ± 1.52 |
| 13 | 1.00 ± 0.15 |
| 14 | 17.5 ± 2.30 |
| 15 | 16.0 ± 1.50 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olea, A.F.; Rubio, J.; Sedan, C.; Carvajal, D.; Nuñez, M.; Espinoza, L.; Llovera, L.; Nuñez, G.; Taborga, L.; Carrasco, H. Antifungal Activity of 2-Allylphenol Derivatives on the Botrytis cinerea Strain: Assessment of Possible Action Mechanism. Int. J. Mol. Sci. 2023, 24, 6530. https://doi.org/10.3390/ijms24076530
Olea AF, Rubio J, Sedan C, Carvajal D, Nuñez M, Espinoza L, Llovera L, Nuñez G, Taborga L, Carrasco H. Antifungal Activity of 2-Allylphenol Derivatives on the Botrytis cinerea Strain: Assessment of Possible Action Mechanism. International Journal of Molecular Sciences. 2023; 24(7):6530. https://doi.org/10.3390/ijms24076530
Chicago/Turabian StyleOlea, Andres F., Julia Rubio, Claudia Sedan, Denisse Carvajal, Maria Nuñez, Luis Espinoza, Ligia Llovera, Gerard Nuñez, Lautaro Taborga, and Héctor Carrasco. 2023. "Antifungal Activity of 2-Allylphenol Derivatives on the Botrytis cinerea Strain: Assessment of Possible Action Mechanism" International Journal of Molecular Sciences 24, no. 7: 6530. https://doi.org/10.3390/ijms24076530
APA StyleOlea, A. F., Rubio, J., Sedan, C., Carvajal, D., Nuñez, M., Espinoza, L., Llovera, L., Nuñez, G., Taborga, L., & Carrasco, H. (2023). Antifungal Activity of 2-Allylphenol Derivatives on the Botrytis cinerea Strain: Assessment of Possible Action Mechanism. International Journal of Molecular Sciences, 24(7), 6530. https://doi.org/10.3390/ijms24076530





