Zebrafish: A Relevant Genetic Model for Human Primary Immunodeficiency (PID) Disorders?
Abstract
1. Introduction
2. The Zebrafish Immune System in Comparison to That of Humans
3. Zebrafish Models of Lymphoid Disorders
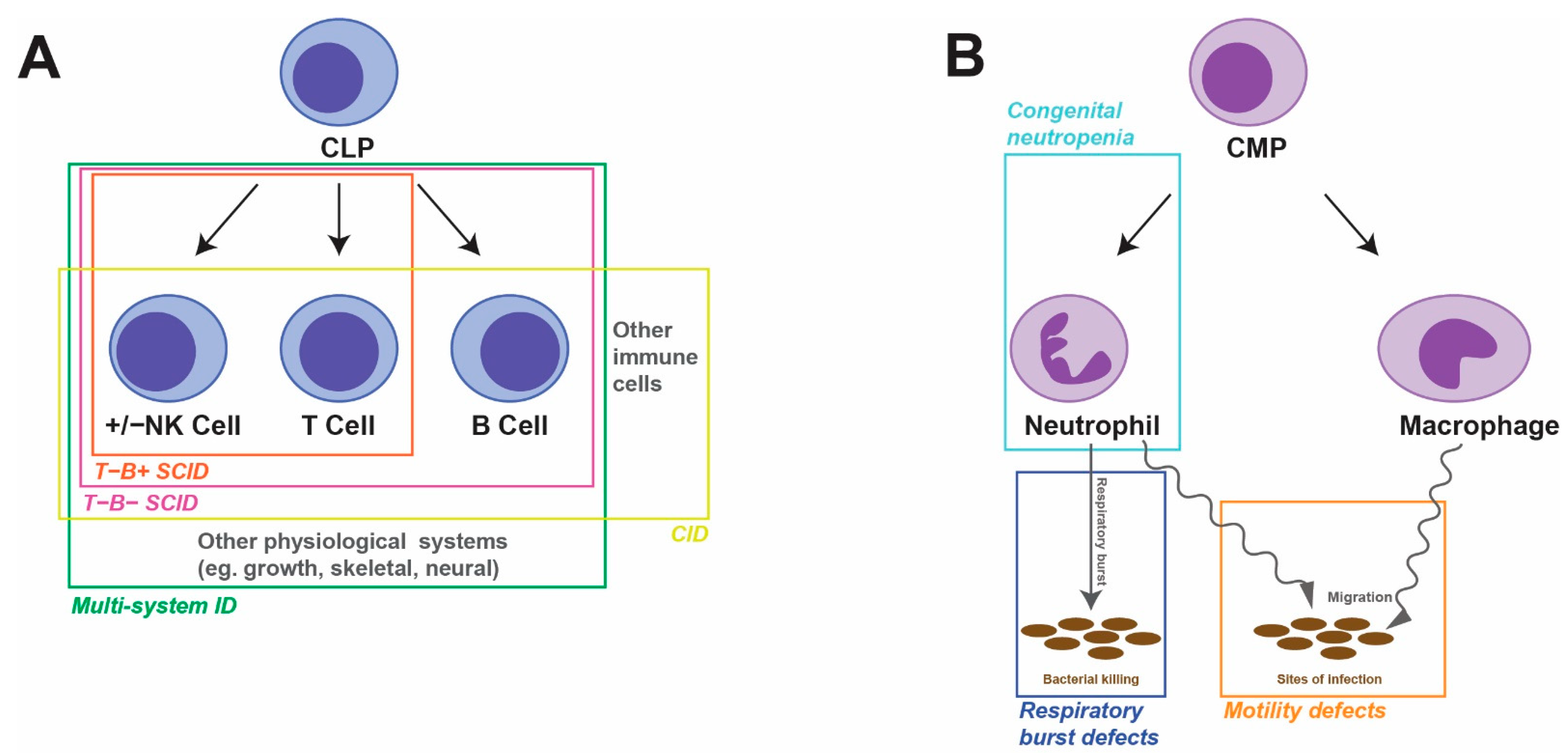
3.1. Severe Combined Immunodeficiency
3.1.1. T−B− SCID
3.1.2. T−B+ SCID
3.2. Combined Immunodeficiency
3.3. Multi-System Immunodeficiency
3.4. Auto-Inflammatory Disorders
4. Zebrafish Models of Myeloid Disorders
4.1. Congenital Neutropenia
4.2. Motility Defects
4.3. Respiratory Burst Defects
5. Other Zebrafish Immunodeficiency Mutants
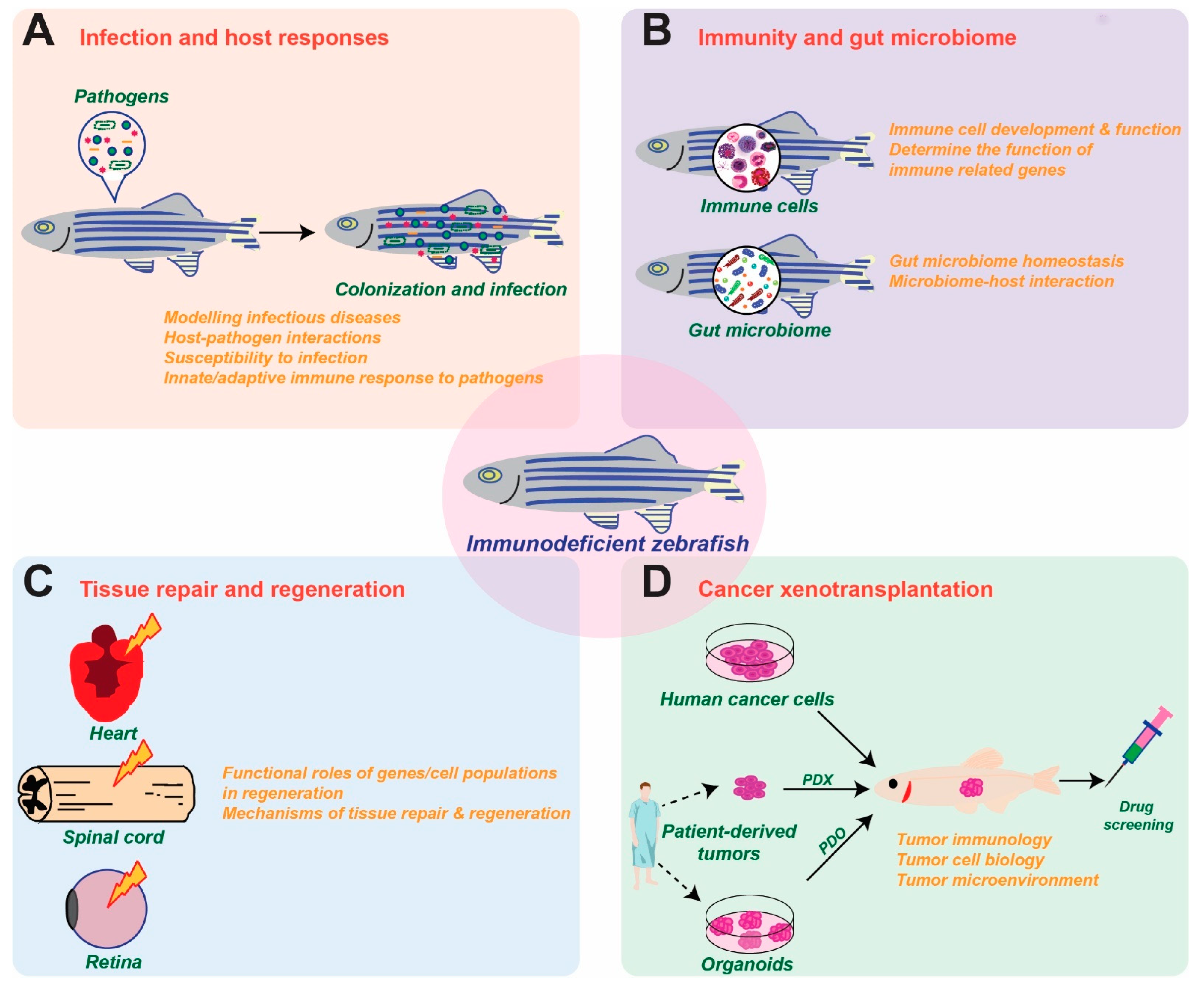
6. Applications of Zebrafish Immunodeficiency Models
6.1. Infection and Host Responses
6.2. Immunity and Gut Microbiome
6.3. Tissue Repair and Regeneration
6.4. Cancer Xenotransplantation
6.5. Other Applications
7. Limitations and Future Prospects
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kraus, R.F.; Gruber, M.A. Neutrophils—From bone marrow to first-line defense of the innate immune system. Front. Immunol. 2021, 12, 767175. [Google Scholar] [CrossRef]
- Gharavi, A.T.; Hanjani, N.A.; Movahed, E.; Doroudian, M. The role of macrophage subtypes and exosomes in immunomodulation. Cell. Mol. Biol. Lett. 2022, 27, 83. [Google Scholar] [CrossRef]
- Marshall, J.S.; Warrington, R.; Watson, W.; Kim, H.L. An introduction to immunology and immunopathology. Allergy Asthma Clin. Immunol. 2018, 14, 49. [Google Scholar] [CrossRef]
- Tangye, S.G.; Al-Herz, W.; Bousfiha, A.; Cunningham-Rundles, C.; Franco, J.L.; Holland, S.M.; Klein, C.; Morio, T.; Oksenhendler, E.; Picard, C.; et al. Human inborn errors of immunity: 2022 update on the classification from the International Union of Immunological Societies Expert Committee. J. Clin. Immunol. 2022, 42, 1473–1507. [Google Scholar] [CrossRef] [PubMed]
- McCusker, C.; Upton, J.; Warrington, R. Primary immunodeficiency. Allergy Asthma Clin. Immunol. 2018, 14, 61. [Google Scholar] [CrossRef]
- Rasighaemi, P.; Basheer, F.; Liongue, C.; Ward, A.C. Zebrafish as a model for leukemia and other hematopoietic disorders. J. Hematol. Oncol. 2015, 8, 29. [Google Scholar] [CrossRef]
- Iwanami, N. Zebrafish as a model for understanding the evolution of the vertebrate immune system and human primary immunodeficiency. Exp. Hematol. 2014, 42, 697–706. [Google Scholar] [CrossRef] [PubMed]
- Rafferty, S.A.; Quinn, T.A. A beginner’s guide to understanding and implementing the genetic modification of zebrafish. Prog. Biophys. Mol. Biol. 2018, 138, 3–19. [Google Scholar] [CrossRef]
- Otterstrom, J.J.; Lubin, A.; Payne, E.M.; Paran, Y. Technologies bringing young Zebrafish from a niche field to the limelight. SLAS Technol. 2022, 27, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Rissone, A.; Burgess, S.M. Rare genetic blood disease modeling in zebrafish. Front. Genet. 2018, 9, 348. [Google Scholar] [CrossRef]
- Konantz, M.; Schurch, C.; Hanns, P.; Muller, J.S.; Sauteur, L.; Lengerke, C. Modeling hematopoietic disorders in zebrafish. Dis. Model. Mech. 2019, 12, dmm040360. [Google Scholar] [CrossRef] [PubMed]
- Stosik, M.; Tokarz-Deptula, B.; Deptula, W. Haematopoiesis in zebrafish (Danio rerio). Front. Immunol. 2022, 13, 902941. [Google Scholar] [CrossRef]
- Hu, Y.X.; Jing, Q. Zebrafish: A convenient tool for myelopoiesis research. Cell Regen. 2023, 12, 2. [Google Scholar] [CrossRef]
- Faisal, M.; Hassan, M.; Kumar, A.; Zubair, M.; Jamal, M.; Menghwar, H.; Saad, M.; Kloczkowski, A. Hematopoietic stem and progenitor cells (HSPCs) and hematopoietic microenvironment: Molecular and bioinformatic studies of the zebrafish models. Int. J. Mol. Sci. 2022, 23, 7285. [Google Scholar] [CrossRef]
- Bajoghli, B.; Dick, A.M.; Claasen, A.; Doll, L.; Aghaallaei, N. Zebrafish and medaka: Two teleost models of T-cell and thymic development. Int. J. Mol. Sci. 2019, 20, 4179. [Google Scholar] [CrossRef] [PubMed]
- Moore, F.E.; Garcia, E.G.; Lobbardi, R.; Jain, E.; Tang, Q.; Moore, J.C.; Cortes, M.; Molodtsov, A.; Kasheta, M.; Luo, C.C. Single-cell transcriptional analysis of normal, aberrant, and malignant hematopoiesis in zebrafish. J. Exp. Med. 2016, 213, 979–992. [Google Scholar] [CrossRef] [PubMed]
- Lieschke, G.J.; Oates, A.C.; Crowhurst, M.O.; Ward, A.C.; Layton, J.E. Morphologic and functional characterization of granulocytes and macrophages in embryonic and adult zebrafish. Blood 2001, 98, 3087–3096. [Google Scholar] [CrossRef] [PubMed]
- Yoong, S.; O’Connell, B.; Soanes, A.; Crowhurst, M.O.; Lieschke, G.J.; Ward, A.C. Characterization of the zebrafish matrix metalloproteinase 9 gene and its developmental expression pattern. Gene Expr. Patterns 2007, 7, 39–46. [Google Scholar] [CrossRef]
- Rosowski, E.E. Determining macrophage versus neutrophil contributions to innate immunity using larval zebrafish. Dis. Model. Mech. 2020, 13, dmm041889. [Google Scholar] [CrossRef]
- Herbomel, P.; Thisse, B.; Thisse, C. Ontogeny and behaviour of early macrophages in the zebrafish embryo. Development 1999, 126, 3735–3745. [Google Scholar] [CrossRef]
- Lewis, K.L.; Del Cid, N.; Traver, D. Perspectives on antigen presenting cells in zebrafish. Dev. Comp. Immunol. 2014, 46, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Balla, K.M.; Lugo-Villarino, G.; Spitsbergen, J.M.; Stachura, D.L.; Hu, Y.; Bañuelos, K.; Romo-Fewell, O.; Aroian, R.V.; Traver, D. Eosinophils in the zebrafish: Prospective isolation, characterization, and eosinophilia induction by helminth determinants. Blood 2010, 116, 3944–3954. [Google Scholar] [CrossRef] [PubMed]
- Da’as, S.; Teh, E.M.; Dobson, J.T.; Nasrallah, G.K.; McBride, E.R.; Wang, H.; Neuberg, D.S.; Marshall, J.S.; Lin, T.-J.; Berman, J.N. Zebrafish mast cells possess an FcɛRI-like receptor and participate in innate and adaptive immune responses. Dev. Comp. Immunol. 2011, 35, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Wan, F.; Hu, C.-B.; Ma, J.-X.; Gao, K.; Xiang, L.-X.; Shao, J.-Z. Characterization of γδ T cells from zebrafish provides insights into their important role in adaptive humoral immunity. Front. Immunol. 2017, 7, 675. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, A.M.; Moustafa, F.M.; Romanowski, K.E.; Steiner, L.A. Zebrafish immunoglobulin IgD: Unusual exon usage and quantitative expression profiles with IgM and IgZ/T heavy chain isotypes. Mol. Immunol. 2011, 48, 2220–2223. [Google Scholar] [CrossRef]
- Tang, Q.; Iyer, S.; Lobbardi, R.; Moore, J.C.; Chen, H.; Lareau, C.; Hebert, C.; Shaw, M.L.; Neftel, C.; Suva, M.L. Dissecting hematopoietic and renal cell heterogeneity in adult zebrafish at single-cell resolution using RNA sequencing. J. Exp. Med. 2017, 214, 2875–2887. [Google Scholar] [CrossRef]
- Huang, Y.; Lu, Y.; He, Y.; Feng, Z.; Zhan, Y.; Huang, X.; Liu, Q.; Zhang, J.; Li, H.; Huang, H. Ikzf1 regulates embryonic T lymphopoiesis via Ccr9 and Irf4 in zebrafish. J. Biol. Chem. 2019, 294, 16152–16163. [Google Scholar] [CrossRef]
- Liongue, C.; Taznin, T.; Ward, A.C. Signaling via the CytoR/JAK/STAT/SOCS pathway: Emergence during evolution. Mol. Immunol. 2016, 71, 166–175. [Google Scholar] [CrossRef]
- Sommer, F.; Torraca, V.; Meijer, A.H. Chemokine receptors and phagocyte biology in zebrafish. Front. Immunol. 2020, 11, 325. [Google Scholar] [CrossRef]
- Wienholds, E.; Schulte-Merker, S.; Walderich, B.; Plasterk, R.H. Target-selected inactivation of the zebrafish rag1 gene. Science 2002, 297, 99–102. [Google Scholar] [CrossRef]
- Petrie-Hanson, L.; Hohn, C.; Hanson, L. Characterization of rag 1 mutant zebrafish leukocytes. BMC Immunol. 2009, 10, 8. [Google Scholar] [CrossRef]
- Tang, Q.; Abdelfattah, N.S.; Blackburn, J.S.; Moore, J.C.; Martinez, S.A.; Moore, F.E.; Lobbardi, R.; Tenente, I.M.; Ignatius, M.S.; Berman, J.N. Optimized cell transplantation using adult rag2 mutant zebrafish. Nat. Methods 2014, 11, 821–824. [Google Scholar] [CrossRef] [PubMed]
- Jung, I.H.; Chung, Y.-Y.; Jung, D.E.; Kim, Y.J.; Kim, K.-S.; Park, S.W. Impaired lymphocytes development and xenotransplantation of gastrointestinal tumor cells in prkdc-null SCID zebrafish model. Neoplasia 2016, 18, 468–479. [Google Scholar] [CrossRef] [PubMed]
- Rissone, A.; Weinacht, K.G.; la Marca, G.; Bishop, K.; Giocaliere, E.; Jagadeesh, J.; Felgentreff, K.; Dobbs, K.; Al-Herz, W.; Jones, M.; et al. Reticular dysgenesis-associated AK2 protects hematopoietic stem and progenitor cell development from oxidative stress. J. Exp. Med. 2015, 212, 1185–1202. [Google Scholar] [CrossRef] [PubMed]
- Sertori, R.; Jones, R.; Basheer, F.; Rivera, L.; Dawson, S.; Loke, S.; Heidary, S.; Dhillon, A.; Liongue, C.; Ward, A.C. Generation and characterization of a zebrafish IL-2Rgc SCID model. Int. J. Mol. Sci. 2022, 23, 2385. [Google Scholar] [CrossRef] [PubMed]
- Iwanami, N.; Mateos, F.; Hess, I.; Riffel, N.; Soza-Ried, C.; Schorpp, M.; Boehm, T. Genetic evidence for an evolutionarily conserved role of IL-7 signaling in T cell development of zebrafish. J. Immunol. 2011, 186, 7060–7066. [Google Scholar] [CrossRef]
- Basheer, F.; Lee, E.; Liongue, C.; Ward, A.C. Zebrafish model of severe combined immunodeficiency (SCID) due to JAK3 mutation. Biomolecules 2022, 12, 1521. [Google Scholar] [CrossRef]
- Moore, J.C.; Mulligan, T.S.; Yordán, N.T.; Castranova, D.; Pham, V.N.; Tang, Q.; Lobbardi, R.; Anselmo, A.; Liwski, R.S.; Berman, J.N. T cell immune deficiency in zap70 mutant zebrafish. Mol. Cell. Biol. 2016, 36, 2868–2876. [Google Scholar] [CrossRef]
- Schorpp, M.; Bialecki, M.; Diekhoff, D.; Walderich, B.; Odenthal, J.; Maischein, H.-M.; Zapata, A.G.; Boehm, T. Conserved functions of Ikaros in vertebrate lymphocyte development: Genetic evidence for distinct larval and adult phases of T cell development and two lineages of B cells in zebrafish. J. Immunol. 2006, 177, 2463–2476. [Google Scholar] [CrossRef]
- Heidary, S.; Awasthi, N.; Page, N.; Lewis, R.S.; Liongue, C.; Ward, A.C. A zebrafish model of growth hormone insensitivity syndrome with immune dysregulation 1 (GHISID1). Cell. Mol. Life Sci. 2023, in press. [Google Scholar]
- Volpi, S.; Yamazaki, Y.; Brauer, P.M.; van Rooijen, E.; Hayashida, A.; Slavotinek, A.; Sun Kuehn, H.; Di Rocco, M.; Rivolta, C.; Bortolomai, I.; et al. EXTL3 mutations cause skeletal dysplasia, immune deficiency, and developmental delay. J. Exp. Med. 2017, 214, 623–637. [Google Scholar] [CrossRef]
- Somech, R.; Lev, A.; Lee, Y.N.; Simon, A.J.; Barel, O.; Schiby, G.; Avivi, C.; Barshack, I.; Rhodes, M.; Yin, J.; et al. Disruption of thrombocyte and T lymphocyte development by a mutation in ARPC1B. J. Immunol. 2017, 199, 4036–4045. [Google Scholar] [CrossRef]
- Putti, S.; Giovinazzo, A.; Merolle, M.; Falchetti, M.L.; Pellegrini, M. ATM kinase dead: From ataxia telangiectasia syndrome to cancer. Cancers 2021, 13, 5498. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.Y.; Sertori, R.; Contreras, A.V.; Hamer, M.; Messing, M.; Del Bel, K.L.; Lopez-Rangel, E.; Chan, E.S.; Rehmus, W.; Milner, J.D.; et al. A novel germline heterozygous BCL11B variant causing severe atopic disease and immune dysregulation. Front. Immunol. 2021, 12, 788278. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Wang, L.; Wang, S.; Gao, Y.; Wei, Y.; Liu, F. Foxn1 maintains thymic epithelial cells to support T-cell development via mcm2 in zebrafish. Proc. Natl. Acad. Sci. USA 2012, 109, 21040–21045. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.-Z.; Wang, Z.-L.; Choi, T.-I.; Huang, W.-T.; Wang, H.-T.; Han, Y.-Y.; Zhu, L.-Y.; Kim, H.-T.; Choi, J.-H.; Lee, J.-S. Chd7 is critical for early T-cell development and thymus organogenesis in zebrafish. Am. J. Pathol. 2018, 188, 1043–1058. [Google Scholar] [CrossRef]
- Piotrowski, T.; Ahn, D.G.; Schilling, T.F.; Nair, S.; Ruvinsky, I.; Geisler, R.; Rauch, G.J.; Haffter, P.; Zon, L.I.; Zhou, Y.; et al. The zebrafish van gogh mutation disrupts tbx1, which is involved in the DiGeorge deletion syndrome in humans. Development 2003, 130, 5043–5052. [Google Scholar] [CrossRef] [PubMed]
- Castro, C.N.; Rosenzwajg, M.; Carapito, R.; Shahrooei, M.; Konantz, M.; Khan, A.; Miao, Z.; Gross, M.; Tranchant, T.; Radosavljevic, M.; et al. NCKAP1L defects lead to a novel syndrome combining immunodeficiency, lymphoproliferation, and hyperinflammation. J. Exp. Med. 2020, 217, e20192275. [Google Scholar] [CrossRef]
- Basheer, F.; Rasighaemi, P.; Liongue, C.; Ward, A.C. Zebrafish granulocyte colony-stimulating factor receptor maintains neutrophil number and function throughout the life span. Infect. Immun. 2019, 87, e00793-18. [Google Scholar] [CrossRef]
- Witzel, M.; Petersheim, D.; Fan, Y.; Bahrami, E.; Racek, T.; Rohlfs, M.; Puchalka, J.; Mertes, C.; Gagneur, J.; Ziegenhain, C.; et al. Chromatin-remodeling factor SMARCD2 regulates transcriptional networks controlling differentiation of neutrophil granulocytes. Nat. Genet. 2017, 49, 742–752. [Google Scholar] [CrossRef]
- Jones, R.A.; Feng, Y.; Worth, A.J.; Thrasher, A.J.; Burns, S.O.; Martin, P. Modelling of human Wiskott-Aldrich syndrome protein mutants in zebrafish larvae using in vivo live imaging. J. Cell Sci. 2013, 126, 4077–4084. [Google Scholar] [CrossRef]
- Doll, L.; Aghaallaei, N.; Dick, A.M.; Welte, K.; Skokowa, J.; Bajoghli, B. A zebrafish model for HAX1-associated congenital neutropenia. Haematologica 2021, 106, 1311–1320. [Google Scholar] [CrossRef] [PubMed]
- Vilboux, T.; Lev, A.; Malicdan, M.C.; Simon, A.J.; Jarvinen, P.; Racek, T.; Puchalka, J.; Sood, R.; Carrington, B.; Bishop, K.; et al. A congenital neutrophil defect syndrome associated with mutations in VPS45. N. Engl. J. Med. 2013, 369, 54–65. [Google Scholar] [CrossRef] [PubMed]
- Oyarbide, U.; Shah, A.N.; Amaya-Mejia, W.; Snyderman, M.; Kell, M.J.; Allende, D.S.; Calo, E.; Topczewski, J.; Corey, S.J. Loss of Sbds in zebrafish leads to neutropenia and pancreas and liver atrophy. JCI Insight 2020, 5, e134309. [Google Scholar] [CrossRef] [PubMed]
- Bader, A.; Gao, J.; Riviere, T.; Schmid, B.; Walzog, B.; Maier-Begandt, D. Molecular insights into neutrophil biology from the zebrafish perspective: Lessons from CD18 deficiency. Front. Immunol. 2021, 12, 677994. [Google Scholar] [CrossRef]
- Deng, Q.; Yoo, S.K.; Cavnar, P.J.; Green, J.M.; Huttenlocher, A. Dual roles for Rac2 in neutrophil motility and active retention in zebrafish hematopoietic tissue. Dev. Cell 2011, 21, 735–745. [Google Scholar] [CrossRef]
- Rosowski, E.E.; Deng, Q.; Keller, N.P.; Huttenlocher, A. Rac2 functions in both neutrophils and macrophages to mediate motility and host defense in larval zebrafish. J. Immunol. 2016, 197, 4780–4790. [Google Scholar] [CrossRef]
- Walters, K.B.; Green, J.M.; Surfus, J.C.; Yoo, S.K.; Huttenlocher, A. Live imaging of neutrophil motility in a zebrafish model of WHIM syndrome. Blood 2010, 116, 2803–2811. [Google Scholar] [CrossRef]
- Tauzin, S.; Starnes, T.W.; Becker, F.B.; Lam, P.Y.; Huttenlocher, A. Redox and Src family kinase signaling control leukocyte wound attraction and neutrophil reverse migration. J. Cell Biol. 2014, 207, 589–598. [Google Scholar] [CrossRef]
- Schoen, T.J.; Rosowski, E.E.; Knox, B.P.; Bennin, D.; Keller, N.P.; Huttenlocher, A. Neutrophil phagocyte oxidase activity controls invasive fungal growth and inflammation in zebrafish. J. Cell Sci. 2019, 133, jcs236539. [Google Scholar] [CrossRef]
- Pase, L.; Layton, J.E.; Wittmann, C.; Ellett, F.; Nowell, C.J.; Reyes-Aldasoro, C.C.; Varma, S.; Rogers, K.L.; Hall, C.J.; Keightley, M.C. Neutrophil-delivered myeloperoxidase dampens the hydrogen peroxide burst after tissue wounding in zebrafish. Curr. Biol. 2012, 22, 1818–1824. [Google Scholar] [CrossRef]
- Wang, K.; Fang, X.; Ma, N.; Lin, Q.; Huang, Z.; Liu, W.; Xu, M.; Chen, X.; Zhang, W.; Zhang, Y. Myeloperoxidase-deficient zebrafish show an augmented inflammatory response to challenge with Candida albicans. Fish Shellfish. Immunol. 2015, 44, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Fischer, A. Severe combined immunodeficiencies (SCID). Clin. Exp. Immunol. 2000, 122, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Michniacki, T.F.; Seth, D.; Secord, E. Severe Combined Immunodeficiency: A review for neonatal clinicians. Neoreviews 2019, 20, e326–e335. [Google Scholar] [CrossRef] [PubMed]
- Bosticardo, M.; Pala, F.; Notarangelo, L.D. RAG deficiencies: Recent advances in disease pathogenesis and novel therapeutic approaches. Eur. J. Immunol. 2021, 51, 1028–1038. [Google Scholar] [CrossRef]
- Hoenig, M.; Pannicke, U.; Gaspar, H.B.; Schwarz, K. Recent advances in understanding the pathogenesis and management of reticular dysgenesis. Br. J. Haematol. 2018, 180, 644–653. [Google Scholar] [CrossRef]
- Hernandez, J.D.; Hsieh, E.W.Y. A great disturbance in the force: IL-2 receptor defects disrupt immune homeostasis. Curr. Opin. Pediatr. 2022, 34, 580–588. [Google Scholar] [CrossRef]
- Sertori, R.; Liongue, C.; Basheer, F.; Lewis, K.L.; Rasighaemi, P.; de Coninck, D.; Traver, D.; Ward, A.C. Conserved IL-2Rgc signaling mediates lymphopoiesis in zebrafish. J. Immunol. 2016, 196, 135–143. [Google Scholar] [CrossRef]
- Ashouri, J.F.; Lo, W.L.; Nguyen, T.T.T.; Shen, L.; Weiss, A. ZAP70, too little, too much can lead to autoimmunity. Immunol. Rev. 2022, 307, 145–160. [Google Scholar] [CrossRef]
- Boast, B.; Nunes-Santos, C.J.; Kuehn, H.S.; Rosenzweig, S.D. Ikaros-associated diseases: From mice to humans and back again. Front. Pediatr. 2021, 9, 705497. [Google Scholar] [CrossRef]
- Smith, M.R.; Satter, L.R.F.; Vargas-Hernandez, A. STAT5b: A master regulator of key biological pathways. Front. Immunol. 2022, 13, 1025373. [Google Scholar] [CrossRef]
- Xiong, S.; Mei, J.; Huang, P.; Jing, J.; Li, Z.; Kang, J.; Gui, J.F. Essential roles of stat5.1/stat5b in controlling fish somatic growth. J. Genet. Genom. 2017, 44, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Piotrowski, T.; Schilling, T.F.; Brand, M.; Jiang, Y.J.; Heisenberg, C.P.; Beuchle, D.; Grandel, H.; van Eeden, F.J.; Furutani-Seiki, M.; Granato, M.; et al. Jaw and branchial arch mutants in zebrafish II: Anterior arches and cartilage differentiation. Development 1996, 123, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Dupre, L.; Prunier, G. Deciphering actin remodelling in immune cells through the prism of actin-related inborn errors of immunity. Eur. J. Cell Biol. 2023, 102, 151283. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Wang, P.; Chen, J.; Ying, Y.; Chen, Y.; Gilson, E.; Lu, Y.; Ye, J. Loss of atm in zebrafish as a model of ataxia-telangiectasia syndrome. Biomedicines 2022, 10, 392. [Google Scholar] [CrossRef]
- Punwani, D.; Zhang, Y.; Yu, J.; Cowan, M.J.; Rana, S.; Kwan, A.; Adhikari, A.N.; Lizama, C.O.; Mendelsohn, B.A.; Fahl, S.P. Multisystem anomalies in severe combined immunodeficiency with mutant BCL11B. N. Engl. J. Med. 2016, 375, 2165–2176. [Google Scholar] [CrossRef] [PubMed]
- Collins, C.; Sharpe, E.; Silber, A.; Kulke, S.; Hsieh, E.W.Y. Congenital athymia: Genetic etiologies, clinical manifestations, diagnosis, and treatment. J. Clin. Immunol. 2021, 41, 881–895. [Google Scholar] [CrossRef] [PubMed]
- Lv, P.; Ma, D.; Gao, S.; Zhang, Y.; Bae, Y.-K.; Liang, G.; Gao, S.; Choi, J.-H.; Kim, C.-H.; Wang, L. Generation of foxn1/casper mutant zebrafish for allograft and xenograft of normal and malignant cells. Stem Cell Rep. 2020, 15, 749–760. [Google Scholar] [CrossRef]
- Liongue, C.; Ward, A.C. Granulocyte colony-stimulating factor receptor mutations in myeloid malignancy. Front. Oncol. 2014, 4, 93. [Google Scholar] [CrossRef]
- Pazhakh, V.; Clark, S.; Keightley, M.C.; Lieschke, G.J. A GCSFR/CSF3R zebrafish mutant models the persistent basal neutrophil deficiency of severe congenital neutropenia. Sci. Rep. 2017, 7, 44455. [Google Scholar] [CrossRef]
- Klein, C. Kostmann’s disease and HCLS1-associated protein X-1 (HAX1). J. Clin. Immunol. 2017, 37, 117–122. [Google Scholar] [CrossRef]
- Bellanne-Chantelot, C.; Schmaltz-Panneau, B.; Marty, C.; Fenneteau, O.; Callebaut, I.; Clauin, S.; Docet, A.; Damaj, G.L.; Leblanc, T.; Pellier, I.; et al. Mutations in the SRP54 gene cause severe congenital neutropenia as well as Shwachman-Diamond-like syndrome. Blood 2018, 132, 1318–1331. [Google Scholar] [CrossRef] [PubMed]
- Fekadu, J.; Modlich, U.; Bader, P.; Bakhtiar, S. Understanding the role of LFA-1 in leukocyte adhesion deficiency type I (LAD I): Moving towards inflammation? Int. J. Mol. Sci. 2022, 23, 3578. [Google Scholar] [CrossRef] [PubMed]
- Lougaris, V.; Baronio, M.; Gazzurelli, L.; Benvenuto, A.; Plebani, A. RAC2 and primary human immune deficiencies. J. Leukoc. Biol. 2020, 108, 687–696. [Google Scholar] [CrossRef] [PubMed]
- McDermott, D.H.; Murphy, P.M. WHIM syndrome: Immunopathogenesis, treatment and cure strategies. Immunol. Rev. 2019, 287, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.H.; Yang, Y.H.; Chiang, B.L. Chronic granulomatous disease: A comprehensive review. Clin. Rev. Allergy Immunol. 2021, 61, 101–113. [Google Scholar] [CrossRef]
- Van der Veen, B.S.; de Winther, M.P.; Heeringa, P. Myeloperoxidase: Molecular mechanisms of action and their relevance to human health and disease. Antioxid. Redox Signal. 2009, 11, 2899–2937. [Google Scholar] [CrossRef]
- Trede, N.S.; Medenbach, J.; Damianov, A.; Hung, L.H.; Weber, G.J.; Paw, B.H.; Zhou, Y.; Hersey, C.; Zapata, A.; Keefe, M.; et al. Network of coregulated spliceosome components revealed by zebrafish mutant in recycling factor p110. Proc. Natl. Acad. Sci. USA 2007, 104, 6608–6613. [Google Scholar] [CrossRef]
- Chi, Y.; Huang, Z.; Chen, Q.; Xiong, X.; Chen, K.; Xu, J.; Zhang, Y.; Zhang, W. Loss of runx1 function results in B cell immunodeficiency but not T cell in adult zebrafish. Open Biol. 2018, 8, 2046–2441. [Google Scholar] [CrossRef]
- Xie, L.; Tao, Y.; Wu, R.; Ye, Q.; Xu, H.; Li, Y. Congenital asplenia due to a tlx1 mutation reduces resistance to Aeromonas hydrophila infection in zebrafish. Fish Shellfish. Immunol. 2019, 95, 538–545. [Google Scholar] [CrossRef]
- Lawir, D.-F.; Iwanami, N.; Schorpp, M.; Boehm, T. A missense mutation in zbtb17 blocks the earliest steps of T cell differentiation in zebrafish. Sci. Rep. 2017, 7, 44145. [Google Scholar] [CrossRef]
- Hess, I.; Iwanami, N.; Schorpp, M.; Boehm, T. Zebrafish model for allogeneic hematopoietic cell transplantation not requiring preconditioning. Proc. Natl. Acad. Sci. USA 2013, 110, 4327–4332. [Google Scholar] [CrossRef] [PubMed]
- Cai, P.; Mao, X.; Zhao, J.; Luo, L. Ribosome biogenesis protein Urb2 regulates hematopoietic stem cells development via P53 pathway in zebrafish. Biochem. Biophys. Res. Commun. 2018, 497, 776–782. [Google Scholar] [CrossRef] [PubMed]
- Simon, A.J.; Lev, A.; Zhang, Y.; Weiss, B.; Rylova, A.; Eyal, E.; Kol, N.; Barel, O.; Cesarkas, K.; Soudack, M. Mutations in STN1 cause Coats plus syndrome and are associated with genomic and telomere defects. J. Exp. Med. 2016, 213, 1429–1440. [Google Scholar] [CrossRef]
- Wong, H.H.; Seet, S.H.; Maier, M.; Gurel, A.; Traspas, R.M.; Lee, C.; Zhang, S.; Talim, B.; Loh, A.Y.T.; Chia, C.Y.; et al. Loss of C2orf69 defines a fatal autoinflammatory syndrome in humans and zebrafish that evokes a glycogen-storage-associated mitochondriopathy. Am. J. Hum. Genet. 2021, 108, 1301–1317. [Google Scholar] [CrossRef]
- Linder, M.I.; Mizoguchi, Y.; Hesse, S.; Csaba, G.; Tatematsu, M.; Lyszkiewicz, M.; Zietara, N.; Jeske, T.; Hastreiter, M.; Rohlfs, M.; et al. Human genetic defects in SRP19 and SRPRA cause severe congenital neutropenia with distinctive proteome changes. Blood 2023, 141, 645–658. [Google Scholar] [CrossRef] [PubMed]
- Yue, Z.; Fan, Z.; Zhang, H.; Feng, B.; Wu, C.; Chen, S.; Ouyang, J.; Fan, H.; Weng, P.; Feng, H.; et al. Differential roles of the fish chitinous membrane in gut barrier immunity and digestive compartments. EMBO Rep. 2023, e56645. [Google Scholar] [CrossRef]
- Loes, A.N.; Hinman, M.N.; Farnsworth, D.R.; Miller, A.C.; Guillemin, K.; Harms, M.J. Identification and characterization of zebrafish Tlr4 coreceptor Md-2. J. Immunol. 2021, 206, 1046–1057. [Google Scholar] [CrossRef]
- Myllymaki, H.; Yu, P.P.; Feng, Y. Opportunities presented by zebrafish larval models to study neutrophil function in tissues. Int. J. Biochem. Cell Biol. 2022, 148, 106234. [Google Scholar] [CrossRef]
- Swaim, L.E.; Connolly, L.E.; Volkman, H.E.; Humbert, O.; Born, D.E.; Ramakrishnan, L. Mycobacterium marinum infection of adult zebrafish causes caseating granulomatous tuberculosis and is moderated by adaptive immunity. Infect. Immun. 2006, 74, 6108–6117. [Google Scholar] [CrossRef]
- Hohn, C.; Petrie-Hanson, L. Rag1−/− mutant zebrafish demonstrate specific protection following bacterial re-exposure. PLoS ONE 2012, 7, e44451. [Google Scholar] [CrossRef]
- van der Vaart, M.; van Soest, J.J.; Spaink, H.P.; Meijer, A.H. Functional analysis of a zebrafish myd88 mutant identifies key transcriptional components of the innate immune system. Dis. Model. Mech. 2013, 6, 841–854. [Google Scholar] [PubMed]
- Gomes, M.C.; Mostowy, S. The case for modeling human infection in zebrafish. Trends Microbiol. 2020, 28, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Kam, J.Y.; Hortle, E.; Krogman, E.; Warner, S.E.; Wright, K.; Luo, K.; Cheng, T.; Manuneedhi Cholan, P.; Kikuchi, K.; Triccas, J.A. Rough and smooth variants of Mycobacterium abscessus are differentially controlled by host immunity during chronic infection of adult zebrafish. Nat. Commun. 2022, 13, 952. [Google Scholar] [CrossRef]
- Farr, D.; Nag, D.; Chazin, W.J.; Harrison, S.; Thummel, R.; Luo, X.; Raychaudhuri, S.; Withey, J.H. Neutrophil-associated responses to Vibrio cholerae infection in a natural host model. Infect. Immun. 2022, 90, e0046621. [Google Scholar] [CrossRef] [PubMed]
- Robertson, T.F.; Huttenlocher, A. Real-time imaging of inflammation and its resolution: It’s apparent because it’s transparent. Immunol. Rev. 2022, 306, 258–270. [Google Scholar] [CrossRef] [PubMed]
- Linnerz, T.; Hall, C.J. The diverse roles of phagocytes during bacterial and fungal infections and sterile inflammation: Lessons from zebrafish. Front. Immunol. 2020, 11, 1094. [Google Scholar] [CrossRef]
- Stagaman, K.; Sharpton, T.J.; Guillemin, K. Zebrafish microbiome studies make waves. Lab. Anim. 2020, 49, 201–207. [Google Scholar] [CrossRef]
- Cornuault, J.K.; Byatt, G.; Paquet, M.E.; De Koninck, P.; Moineau, S. Zebrafish: A big fish in the study of the gut microbiota. Curr. Opin. Biotechnol. 2022, 73, 308–313. [Google Scholar] [CrossRef]
- Lane, J.P.; Stewart, C.J.; Cummings, S.P.; Gennery, A.R. Gut microbiome variations during hematopoietic stem cell transplant in severe combined immunodeficiency. J. Allergy Clin. Immunol. 2015, 135, 1654–1656.e1652. [Google Scholar] [CrossRef]
- Brugman, S.; Schneeberger, K.; Witte, M.; Klein, M.R.; van den Bogert, B.; Boekhorst, J.; Timmerman, H.M.; Boes, M.L.; Kleerebezem, M.; Nieuwenhuis, E.E. T lymphocytes control microbial composition by regulating the abundance of Vibrio in the zebrafish gut. Gut Microbes 2014, 5, 737–747. [Google Scholar] [CrossRef]
- Burns, A.R.; Miller, E.; Agarwal, M.; Rolig, A.S.; Milligan-Myhre, K.; Seredick, S.; Guillemin, K.; Bohannan, B.J. Interhost dispersal alters microbiome assembly and can overwhelm host innate immunity in an experimental zebrafish model. Proc. Natl. Acad. Sci. USA 2017, 114, 11181–11186. [Google Scholar] [CrossRef] [PubMed]
- Earley, A.M.; Graves, C.L.; Shiau, C.E. Critical role for a subset of intestinal macrophages in shaping gut microbiota in adult zebrafish. Cell Rep. 2018, 25, 424–436. [Google Scholar] [CrossRef] [PubMed]
- Marques, I.J.; Lupi, E.; Mercader, N. Model systems for regeneration: Zebrafish. Development 2019, 146, dev167692. [Google Scholar] [CrossRef] [PubMed]
- Francoeur, N.; Sen, R. Advances in cardiac development and regeneration using zebrafish as a model system for high-throughput research. J. Dev. Biol. 2021, 9, 40. [Google Scholar] [CrossRef]
- Henke, K.; Farmer, D.T.; Niu, X.; Kraus, J.M.; Galloway, J.L.; Youngstrom, D.W. Genetically engineered zebrafish as models of skeletal development and regeneration. Bone 2023, 167, 116611. [Google Scholar] [CrossRef]
- Kikuchi, K. New function of zebrafish regulatory T cells in organ regeneration. Curr. Opin. Immunol. 2020, 63, 7–13. [Google Scholar] [CrossRef]
- Li, L.; Yan, B.; Shi, Y.-Q.; Zhang, W.-Q.; Wen, Z.-L. Live imaging reveals differing roles of macrophages and neutrophils during zebrafish tail fin regeneration. J. Biol. Chem. 2012, 287, 25353–25360. [Google Scholar] [CrossRef]
- Var, S.R.; Byrd-Jacobs, C.A. Role of macrophages and microglia in zebrafish regeneration. Int. J. Mol. Sci. 2020, 21, 4768. [Google Scholar] [CrossRef]
- Campbell, C.A.; Fursova, O.; Cheng, X.; Snella, E.; McCune, A.; Li, L.; Solchenberger, B.; Schmid, B.; Sahoo, D.; Morton, M. A zebrafish model of granulin deficiency reveals essential roles in myeloid cell differentiation. Blood Adv. 2021, 5, 796–811. [Google Scholar] [CrossRef]
- Bernut, A.; Loynes, C.A.; Floto, R.A.; Renshaw, S.A. Deletion of cftr leads to an excessive neutrophilic response and defective tissue repair in a zebrafish model of sterile inflammation. Front. Immunol. 2020, 11, 1733. [Google Scholar] [CrossRef]
- Kwiatkowska, I.; Hermanowicz, J.M.; Iwinska, Z.; Kowalczuk, K.; Iwanowska, J.; Pawlak, D. Zebrafish—An optimal model in experimental oncology. Molecules 2022, 27, 4223. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Wang, X.; Zhao, Y.; Li, Z.; Lin, S.; Wei, Y.; Yang, H. A novel xenograft model in zebrafish for high-resolution investigating dynamics of neovascularization in tumors. PLoS ONE 2011, 6, e21768. [Google Scholar] [CrossRef] [PubMed]
- Jung, D.-W.; Oh, E.-S.; Park, S.-H.; Chang, Y.-T.; Kim, C.-H.; Choi, S.-Y.; Williams, D.R. A novel zebrafish human tumor xenograft model validated for anti-cancer drug screening. Mol. BioSystems 2012, 8, 1930–1939. [Google Scholar] [CrossRef]
- Drabsch, Y.; He, S.; Zhang, L.; Snaar-Jagalska, B.E.; ten Dijke, P. Transforming growth factor-β signalling controls human breast cancer metastasis in a zebrafish xenograft model. Breast Cancer Res. 2013, 15, R106. [Google Scholar] [CrossRef]
- Pudelko, L.; Edwards, S.; Balan, M.; Nyqvist, D.; Al-Saadi, J.; Dittmer, J.; Almlöf, I.; Helleday, T.; Bräutigam, L. An orthotopic glioblastoma animal model suitable for high-throughput screenings. Neuro. Oncol. 2018, 20, 1475–1484. [Google Scholar] [CrossRef]
- Vargas-Patron, L.A.; Agudelo-Dueñas, N.; Madrid-Wolff, J.; Venegas, J.A.; González, J.M.; Forero-Shelton, M.; Akle, V. Xenotransplantation of human glioblastoma in zebrafish larvae: In vivo imaging and proliferation assessment. Biol. Open 2019, 8, bio043257. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.C.; Tang, Q.; Yordán, N.T.; Moore, F.E.; Garcia, E.G.; Lobbardi, R.; Ramakrishnan, A.; Marvin, D.L.; Anselmo, A.; Sadreyev, R.I. Single-cell imaging of normal and malignant cell engraftment into optically clear prkdc-null SCID zebrafish. J. Exp. Med. 2016, 213, 2575–2589. [Google Scholar] [CrossRef]
- Weiss, J.M.; Lumaquin-Yin, D.; Montal, E.; Suresh, S.; Leonhardt, C.S.; White, R.M. Shifting the focus of zebrafish toward a model of the tumor microenvironment. Elife 2022, 11, 69703. [Google Scholar] [CrossRef]
- Somasagara, R.R.; Huang, X.; Xu, C.; Haider, J.; Serody, J.S.; Armistead, P.M.; Leung, T. Targeted therapy of human leukemia xenografts in immunodeficient zebrafish. Sci. Rep. 2021, 11, 5715. [Google Scholar] [CrossRef]
- Yan, C.; Brunson, D.C.; Tang, Q.; Do, D.; Iftimia, N.A.; Moore, J.C.; Hayes, M.N.; Welker, A.M.; Garcia, E.G.; Dubash, T.D. Visualizing engrafted human cancer and therapy responses in immunodeficient zebrafish. Cell 2019, 177, 1903–1914.e14. [Google Scholar] [CrossRef]
- Yan, C.; Yang, Q.; Zhang, S.; Millar, D.G.; Alpert, E.J.; Do, D.; Veloso, A.; Brunson, D.C.; Drapkin, B.J.; Stanzione, M. Single-cell imaging of T cell immunotherapy responses in vivo. J. Exp. Med. 2021, 218, e20210314. [Google Scholar] [CrossRef] [PubMed]
- Zeng, M.; Pi, C.; Li, K.; Sheng, L.; Zuo, Y.; Yuan, J.; Zou, Y.; Zhang, X.; Zhao, W.; Lee, R.J. Patient-derived xenograft: A more standard “avatar” model in preclinical studies of gastric cancer. Front. Oncol. 2022, 12, 898563. [Google Scholar] [CrossRef] [PubMed]
- Gaudenzi, G.; Albertelli, M.; Dicitore, A.; Würth, R.; Gatto, F.; Barbieri, F.; Cotelli, F.; Florio, T.; Ferone, D.; Persani, L. Patient-derived xenograft in zebrafish embryos: A new platform for translational research in neuroendocrine tumors. Endocrine 2017, 57, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Tang, Q.; Moore, J.C.; Ignatius, M.S.; Tenente, I.M.; Hayes, M.N.; Garcia, E.G.; Torres Yordán, N.; Bourque, C.; He, S.; Blackburn, J.S. Imaging tumour cell heterogeneity following cell transplantation into optically clear immune-deficient zebrafish. Nat. Commun. 2016, 7, 10358. [Google Scholar] [CrossRef]
- Fior, R.; Póvoa, V.; Mendes, R.V.; Carvalho, T.; Gomes, A.; Figueiredo, N.; Ferreira, M.G. Single-cell functional and chemosensitive profiling of combinatorial colorectal therapy in zebrafish xenografts. Proc. Natl. Acad. Sci. USA 2017, 114, E8234–E8243. [Google Scholar] [CrossRef]
- Costa, B.; Ferreira, S.; Póvoa, V.; Cardoso, M.J.; Vieira, S.; Stroom, J.; Fidalgo, P.; Rio-Tinto, R.; Figueiredo, N.; Parés, O. Developments in zebrafish avatars as radiotherapy sensitivity reporters—Towards personalized medicine. EBioMedicine 2020, 51, 102578. [Google Scholar] [CrossRef]
- Rebelo de Almeida, C.; Mendes, R.V.; Pezzarossa, A.; Gago, J.; Carvalho, C.; Alves, A.; Nunes, V.; Brito, M.J.; Cardoso, M.J.; Ribeiro, J. Zebrafish xenografts as a fast screening platform for bevacizumab cancer therapy. Commun. Biol. 2020, 3, 299. [Google Scholar] [CrossRef]
- O’Meara, C.P.; Guerri, L.; Lawir, D.F.; Mateos, F.; Iconomou, M.; Iwanami, N.; Soza-Ried, C.; Sikora, K.; Siamishi, I.; Giorgetti, O.; et al. Genetic landscape of T cells identifies synthetic lethality for T-ALL. Commun. Biol. 2021, 4, 1201. [Google Scholar] [CrossRef]
- Xia, H.; Chen, H.; Cheng, X.; Yin, M.; Yao, X.; Ma, J.; Huang, M.; Chen, G.; Liu, H. Zebrafish: An efficient vertebrate model for understanding role of gut microbiota. Mol. Med. 2022, 28, 161. [Google Scholar] [CrossRef]
- Murdoch, C.C.; Rawls, J.F. Commensal microbiota regulate vertebrate innate immunity-insights from the zebrafish. Front. Immunol. 2019, 10, 2100. [Google Scholar] [CrossRef]
- Chen, J.; Liao, S.; Xiao, Z.; Pan, Q.; Wang, X.; Shen, K.; Wang, S.; Yang, L.; Guo, F.; Liu, H.F.; et al. The development and improvement of immunodeficient mice and humanized immune system mouse models. Front. Immunol. 2022, 13, 1007579. [Google Scholar] [CrossRef] [PubMed]
- Al-Hamaly, M.A.; Turner, L.T.; Rivera-Martinez, A.; Rodriguez, A.; Blackburn, J.S. Zebrafish cancer avatars: A translational platform for analyzing tumor heterogeneity and predicting patient outcomes. Int. J. Mol. Sci. 2023, 24, 2288. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, M. The application of zebrafish patient-derived xenograft tumor models in the development of antitumor agents. Med. Res. Rev. 2023, 43, 212–236. [Google Scholar] [CrossRef] [PubMed]
| Human Defect † | Inheritance | OMIM | Zebrafish Model * | Immune Phenotype # | References |
|---|---|---|---|---|---|
| Lymphoid deficiencies | |||||
| T−B− severe combined immunodeficiency (SCID) | |||||
| RAG1 | AR | 179615 | rag1 LOF | ↓ T and B cells | [30,31] |
| RAG2 | AR | 179616 | rag2 hypomorph | ↓ T cells, variable B cell deficiency | [16,32] |
| PRKDC | AR | 615966 | prkdc LOF | ↓ T, B and NK (but not NK-like) cells | [33] |
| AK2 | AR | 103020 | ak2 LOF + KD | ↓ embryonic T cells (and neutrophils) | [34] |
| T−B+ severe combined immunodeficiency (SCID) | |||||
| IL2RG | XL | 308380 | il2rga LOF | ↓ T and NK cells | [26,35] |
| JAK3 | AR | 600173 | jak3 LOF | ↓ T and NK cells, minor defects in mature B cells and neutrophils | [16,36,37] |
| IL7R | AR | 146661 | il7r LOF | ↓ embryonic T cells | [36] |
| Combined immunodeficiencies (CID) | |||||
| ZAP70 | AR | 269840 | zap70 LOF | ↓ thymic and mature kidney T cells | [38] |
| IKZF1 | AD | 603023 | ikzf1 LOF | ↓ embryonic T cells, less severe defect in juveniles and adults | [39] |
| Multi-system immunodeficiencies | |||||
| STAT5B | AR | 245590 | stat5.1 LOF | ↓ T cells throughout lifespan, ↑ T cell activation | [40] |
| EXTL3 | AR | 617425 | extl3 LOF | ↓ embryonic T cells | [41] |
| ARPC1B | AR | 604223 | arpc1b KD | ↓ embryonic T cells | [42] |
| ATM | AR | 607585 | atm LOF | ↓ embryonic T cell and neutrophils, ↓ lymphocytes and ↑ precursors in adults | [43] |
| BCL11B | AD | 617237 | BCL11B GOF TG | ↓ embryonic T cells | [44] |
| FOXN1 | AR | 601705 | foxn1 LOF | ↓ embryonic T cells | [45] |
| CHD7 | AD | 608892 | chd7 LOF + KD | ↓ embryonic T cells | [46] |
| TBX1 | AD | 602054 | tbx1 LOF | athymic | [47] |
| Autoinflammatory disorders | |||||
| NCKAP1L | AR | 618982 | nckap1l LOF | defective neutrophil migration | [48] |
| Myeloid deficiencies | |||||
| Congenital neutropenias | |||||
| CSF3R | AR | 138971 | csf3r LOF | ↓ neutrophils throughout lifespan | [49] |
| SMARCD2 | AR | 601736 | smarcd2 LOF + KD | ↓ embryonic neutrophils | [50] |
| WAS | XL | 300392 | was LOF | defective neutrophil and macrophage migration | [51] |
| HAX1 | AR | 605998 | hax1 KD | ↓ embryonic neutrophils | [52] |
| VPS45 | AR | 615288 | vps45 KD | ↓ embryonic neutrophils | [53] |
| SRP54 | AD | 604857 | srp45 LOF | ↓ embryonic neutrophils | [54] |
| Motility defects | |||||
| ITGB2 | AR | 600065 | itgb2 LOF | defective neutrophil trafficking to inflammatory sites | [55] |
| RAC2 | AD | 608203 | rac2 LOF + KD | defective neutrophil and macrophage mobility | [56,57] |
| CXCR4 | AD | 162643 | cxcr4 GOF TG | ↓ embryonic neutrophils, defective neutrophil mobility and wound recruitment | [58] |
| Respiratory burst defects | |||||
| CYBA | AR | 608508 | cyba LOF | defective neutrophil reverse migration and macrophage wound attraction | [59,60] |
| MPO | AR | 254600 | mpo LOF | sustained hydrogen peroxidase burst | [61,62] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basheer, F.; Sertori, R.; Liongue, C.; Ward, A.C. Zebrafish: A Relevant Genetic Model for Human Primary Immunodeficiency (PID) Disorders? Int. J. Mol. Sci. 2023, 24, 6468. https://doi.org/10.3390/ijms24076468
Basheer F, Sertori R, Liongue C, Ward AC. Zebrafish: A Relevant Genetic Model for Human Primary Immunodeficiency (PID) Disorders? International Journal of Molecular Sciences. 2023; 24(7):6468. https://doi.org/10.3390/ijms24076468
Chicago/Turabian StyleBasheer, Faiza, Robert Sertori, Clifford Liongue, and Alister C. Ward. 2023. "Zebrafish: A Relevant Genetic Model for Human Primary Immunodeficiency (PID) Disorders?" International Journal of Molecular Sciences 24, no. 7: 6468. https://doi.org/10.3390/ijms24076468
APA StyleBasheer, F., Sertori, R., Liongue, C., & Ward, A. C. (2023). Zebrafish: A Relevant Genetic Model for Human Primary Immunodeficiency (PID) Disorders? International Journal of Molecular Sciences, 24(7), 6468. https://doi.org/10.3390/ijms24076468








