Ginsenosides from Panax ginseng as Key Modulators of NF-κB Signaling Are Powerful Anti-Inflammatory and Anticancer Agents
Abstract
1. Introduction
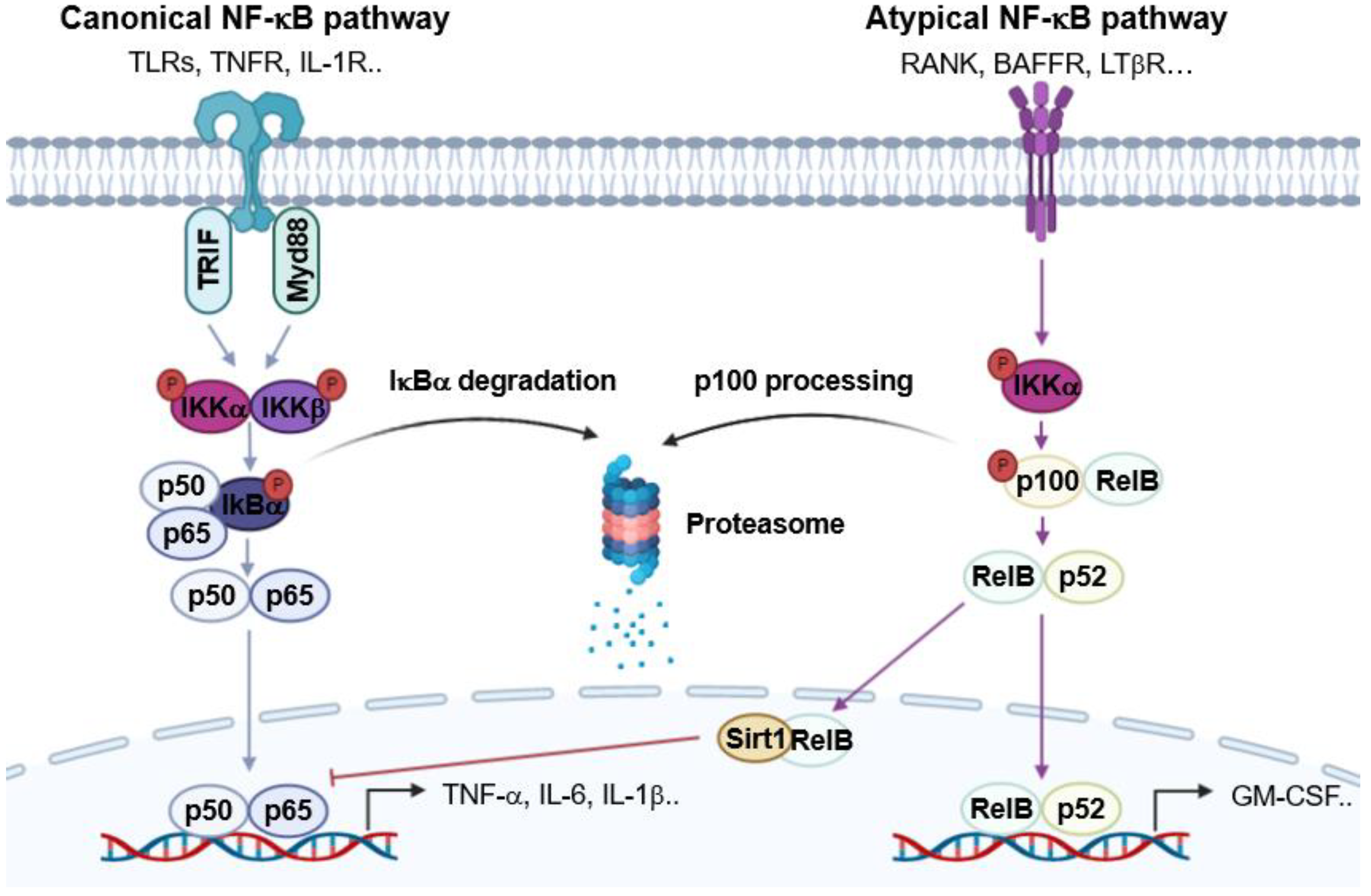
2. NF-κB and Immune Responses
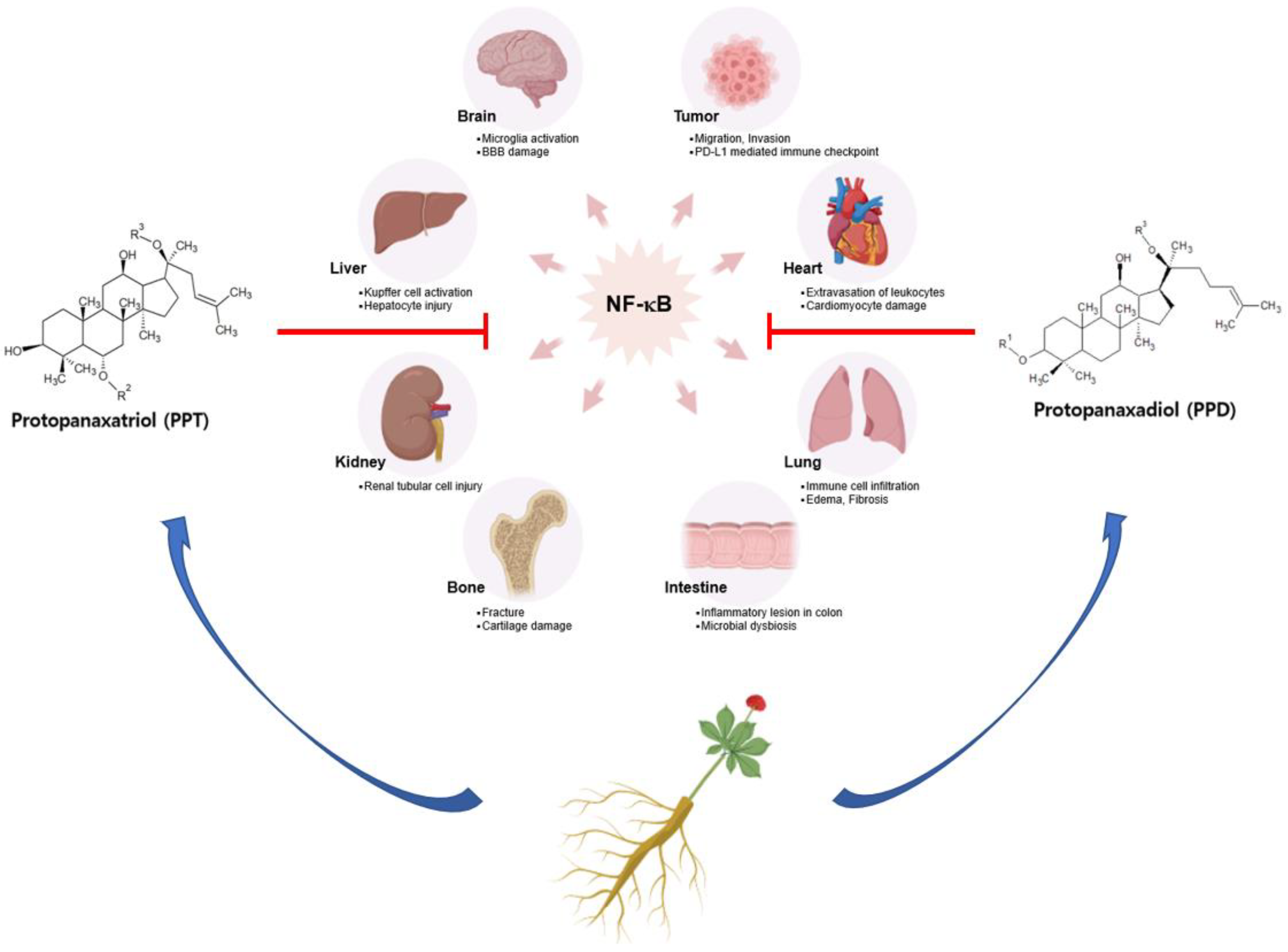
3. Pharmacological Activities of Ginsenosides on Inflammatory Diseases
3.1. Neural Inflammation and Neurodegenerative Diseases
3.2. Myocardial Inflammation and Heart Failure
3.3. Chronic Liver Diseases and Metabolic Disorder
3.4. Lung Inflammation and Pulmonary Disorder
3.5. Kidney Inflammation and Nephropathy
3.6. Colitis and Digestive Disorder
3.7. Bone Disorders
4. NF-κB and Cancer
5. Anticancer Effects of Ginsenosides via NF-κB Modulation
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| COPD | Chronic obstructive pulmonary disease. |
| NF-κB | Nuclear factor kappa B. |
| RelA | p65. |
| PRRs | Pattern recognition receptors. |
| PAMPs | Pathogen-associated molecular patterns. |
| DAMPs | Damage-associated molecular patterns. |
| TLRs | Toll-like receptors. |
| MyD88 | Myeloid differentiation primary response 88. |
| TRIF | TIR-domain containing adapter-inducing interferon-beta. |
| IKK | Inhibitor of NF-κB kinase. |
| IκBα | Inhibitor of NF-κB. |
| COX-2 | Cyclooxygenase-2. |
| iNOS | Inducible NO synthase. |
| IL | Interleukin. |
| TNF-α | Tumor necrosis factor-alpha. |
| NLPR3 | NLR family pyrin domain containing 3. |
| NSAIDs | Nonsteroidal anti-inflammatory drugs. |
| PPD | Protopanaxadiol. |
| PPT | Protopanaxatriol. |
| OA | Oleanolic acid. |
| OT | Ocotillol. |
| IBD | Inflammatory bowel disease. |
| TNFRs | Tumor necrosis factor receptors. |
| IL-1Rs | Interleukin-1 receptors. |
| RANK | Receptor activator of NF-κB. |
| BAFFR | B cell activating factor receptor. |
| LTβR | Lymphotoxin beta receptor. |
| RANKL | Receptor activator of NF-κB ligand. |
| BAF | B cell activating factor. |
| LTα1β2 | Lymphotoxin alpha/beta. |
| GM-CSF | Granulocyte-macrophage colony-stimulating factor. |
| MAPK | Mitogen-activated protein kinase. |
| JAK | Janus kinase. |
| STAT | Signal transduction and activator of transcription. |
| LPS | Lipopolysaccharide. |
| CNS | Central nervous system. |
| BBB | Blood–brain barrier. |
| ZO-1 | Zonula occludens-1. |
| PPARγ | Proliferator-activated receptor gamma. |
| MMP | Matrix metalloproteinase. |
| CUMS | Chronic unpredictable mild stress. |
| CPZ | Cuprizone. |
| Iba-1 | Ionized calcium-binding adapter molecule-1. |
| CXCL10 | Chemokine ligand 10. |
| 3-NP | 3-nitropropionic acid. |
| HUVECs | Human umbilical vascular endothelial cells. |
| Ang-II | Angiotensin-II. |
| HMGB1 | High mobility group box 1 protein. |
| RAGE | Receptor for advanced glycation end products. |
| ALD | Alcoholic liver disease. |
| NAFLD | Non-alcoholic fatty liver disease. |
| ALT | Alanine transaminase. |
| AST | Aspartate transaminase. |
| LDH | Lactate dehydrogenase. |
| AKP | Alkaline phosphatase. |
| TGF-β1 | Transforming growth factor-beta1. |
| NASH | Nonalcoholic steatohepatitis. |
| FoxO1 | Forkhead box O1. |
| HO-1 | Heme oxygenase-1. |
| Nrf2 | Nuclear factor erythroid-2-related factor 2. |
| ARDS | Acute respiratory syndrome. |
| EMT | Endothelial–mesenchymal transition. |
| LC3 | Microtubule-associated protein light chain 3. |
| Snail | Zinc finger protein SNAI1. |
| Slug | Zinc finger protein SNAI2. |
| TWIST1 | Twist-related protein 1. |
| EGFR | Epidermal growth factor. |
| Akt | Protein kinase B. |
| PD-L1 | Programmed death-ligand 1. |
| PD-1 | Programmed cell death protein 1. |
| IFN-γ | Interferon-gamma. |
| Anxa2 | Annexin A2. |
| PI3K | Phosphatidylinositol 3-kinase. |
| mTOR | Mechanistic target of rapamycin. |
References
- Chovatiya, R.; Medzhitov, R. Stress, inflammation, and defense of homeostasis. Mol. Cell 2014, 54, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Louati, K.; Berenbaum, F. Fatigue in chronic inflammation—A link to pain pathways. Arthritis Res. Ther. 2015, 17, 254. [Google Scholar] [CrossRef] [PubMed]
- Delano, M.J.; Ward, P.A. The immune system’s role in sepsis progression, resolution, and long-term outcome. Immunol. Rev. 2016, 274, 330–353. [Google Scholar] [CrossRef] [PubMed]
- Bosmann, M.; Ward, P.A. The inflammatory response in sepsis. Trends Immunol. 2013, 34, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Maspero, J.; Adir, Y.; Al-Ahmad, M.; Celis-Preciado, C.A.; Colodenco, F.D.; Giavina-Bianchi, P.; Lababidi, H.; Ledanois, O.; Mahoub, B.; Perng, D.W.; et al. Type 2 inflammation in asthma and other airway diseases. ERJ Open Res. 2022, 8. [Google Scholar] [CrossRef]
- Sen, R.; Baltimore, D. Multiple nuclear factors interact with the immunoglobulin enhancer sequences. Cell 1986, 46, 705–716. [Google Scholar] [CrossRef]
- Sen, R. NF-kappaB and the immunoglobulin kappa gene enhancer. J. Exp. Med. 2004, 200, 1099–1102. [Google Scholar] [CrossRef]
- Guttridge, D.C.; Albanese, C.; Reuther, J.Y.; Pestell, R.G.; Baldwin, A.S., Jr. NF-kappaB controls cell growth and differentiation through transcriptional regulation of cyclin D1. Mol. Cell. Biol. 1999, 19, 5785–5799. [Google Scholar] [CrossRef]
- Oeckinghaus, A.; Ghosh, S. The NF-kappaB family of transcription factors and its regulation. Cold Spring Harb. Perspect. Biol. 2009, 1, a000034. [Google Scholar] [CrossRef]
- Florio, T.J.; Lokareddy, R.K.; Yeggoni, D.P.; Sankhala, R.S.; Ott, C.A.; Gillilan, R.E.; Cingolani, G. Differential recognition of canonical NF-κB dimers by Importin α3. Nat. Commun. 2022, 13, 1207. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [PubMed]
- Kumar, H.; Kawai, T.; Akira, S. Pathogen recognition by the innate immune system. Int. Rev. Immunol. 2011, 30, 16–34. [Google Scholar] [CrossRef] [PubMed]
- Hennessy, E.J.; Parker, A.E.; O’Neill, L.A. Targeting Toll-like receptors: Emerging therapeutics? Nat. Rev. Drug Discov. 2010, 9, 293–307. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Sato, S.; Hemmi, H.; Hoshino, K.; Kaisho, T.; Sanjo, H.; Takeuchi, O.; Sugiyama, M.; Okabe, M.; Takeda, K.; et al. Role of adaptor TRIF in the MyD88-independent toll-like receptor signaling pathway. Science 2003, 301, 640–643. [Google Scholar] [CrossRef] [PubMed]
- Piras, V.; Selvarajoo, K. Beyond MyD88 and TRIF Pathways in Toll-Like Receptor Signaling. Front. Immunol. 2014, 5, 70. [Google Scholar] [CrossRef]
- Dorrington, M.G.; Fraser, I.D.C. NF-κB Signaling in Macrophages: Dynamics, Crosstalk, and Signal Integration. Front. Immunol. 2019, 10, 705. [Google Scholar] [CrossRef]
- Wu, X.; Wang, Z.; Shi, J.; Yu, X.; Li, C.; Liu, J.; Zhang, F.; Chen, H.; Zheng, W. Macrophage polarization toward M1 phenotype through NF-κB signaling in patients with Behçet’s disease. Arthritis Res. Ther. 2022, 24, 249. [Google Scholar] [CrossRef]
- Brasier, A.R. The nuclear factor-kappaB-interleukin-6 signalling pathway mediating vascular inflammation. Cardiovasc. Res. 2010, 86, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Akira, S.; Hirano, T.; Taga, T.; Kishimoto, T. Biology of multifunctional cytokines: IL 6 and related molecules (IL 1 and TNF). FASEB J. 1990, 4, 2860–2867. [Google Scholar] [CrossRef]
- Boaru, S.G.; Borkham-Kamphorst, E.; Van de Leur, E.; Lehnen, E.; Liedtke, C.; Weiskirchen, R. NLRP3 inflammasome expression is driven by NF-κB in cultured hepatocytes. Biochem. Biophys. Res. Commun. 2015, 458, 700–706. [Google Scholar] [CrossRef]
- Arbore, G.; West, E.E.; Spolski, R.; Robertson, A.A.B.; Klos, A.; Rheinheimer, C.; Dutow, P.; Woodruff, T.M.; Yu, Z.X.; O’Neill, L.A.; et al. T helper 1 immunity requires complement-driven NLRP3 inflammasome activity in CD4+ T cells. Science 2016, 352, aad1210. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Gaynor, R.B. Therapeutic potential of inhibition of the NF-kappaB pathway in the treatment of inflammation and cancer. J. Clin. Investig. 2001, 107, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Auphan, N.; DiDonato, J.A.; Rosette, C.; Helmberg, A.; Karin, M. Immunosuppression by glucocorticoids: Inhibition of NF-kappa B activity through induction of I kappa B synthesis. Science 1995, 270, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Hawkey, C.J. COX-1 and COX-2 inhibitors. Best Pract. Res. Clin. Gastroenterol. 2001, 15, 801–820. [Google Scholar] [CrossRef] [PubMed]
- Cea, L.A.; Balboa, E.; Puebla, C.; Vargas, A.A.; Cisterna, B.A.; Escamilla, R.; Regueira, T.; Sáez, J.C. Dexamethasone-induced muscular atrophy is mediated by functional expression of connexin-based hemichannels. Biochim. Biophys. Acta 2016, 1862, 1891–1899. [Google Scholar] [CrossRef] [PubMed]
- Page, T.H.; Turner, J.J.; Brown, A.C.; Timms, E.M.; Inglis, J.J.; Brennan, F.M.; Foxwell, B.M.; Ray, K.P.; Feldmann, M. Nonsteroidal anti-inflammatory drugs increase TNF production in rheumatoid synovial membrane cultures and whole blood. J. Immunol. 2010, 185, 3694–3701. [Google Scholar] [CrossRef]
- Helms, S. Cancer prevention and therapeutics: Panax ginseng. Altern. Med. Rev. 2004, 9, 259–274. [Google Scholar]
- Lee, S.; Rhee, D.K. Effects of ginseng on stress-related depression, anxiety, and the hypothalamic-pituitary-adrenal axis. J. Ginseng Res. 2017, 41, 589–594. [Google Scholar] [CrossRef]
- Hong, B.N.; Ji, M.G.; Kang, T.H. The efficacy of red ginseng in type 1 and type 2 diabetes in animals. Evid.-Based Complement. Alternat. Med. 2013, 2013, 593181. [Google Scholar] [CrossRef]
- Kim, J.H.; Yi, Y.S.; Kim, M.Y.; Cho, J.Y. Role of ginsenosides, the main active components of Panax ginseng, in inflammatory responses and diseases. J. Ginseng Res. 2017, 41, 435–443. [Google Scholar] [CrossRef]
- You, L.; Cho, J.Y. The regulatory role of Korean ginseng in skin cells. J. Ginseng Res. 2021, 45, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Xue, Q.; He, N.; Wang, Z.; Fu, X.; Aung, L.H.H.; Liu, Y.; Li, M.; Cho, J.Y.; Yang, Y.; Yu, T. Functional roles and mechanisms of ginsenosides from Panax ginseng in atherosclerosis. J. Ginseng Res. 2021, 45, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Ratan, Z.A.; Haidere, M.F.; Hong, Y.H.; Park, S.H.; Lee, J.O.; Lee, J.; Cho, J.Y. Pharmacological potential of ginseng and its major component ginsenosides. J. Ginseng Res. 2021, 45, 199–210. [Google Scholar] [CrossRef]
- Lee, S.M.; Bae, B.S.; Park, H.W.; Ahn, N.G.; Cho, B.G.; Cho, Y.L.; Kwak, Y.S. Characterization of Korean Red Ginseng (Panax ginseng Meyer): History, preparation method, and chemical composition. J. Ginseng Res. 2015, 39, 384–391. [Google Scholar] [CrossRef]
- Wan, Y.; Wang, J.; Xu, J.F.; Tang, F.; Chen, L.; Tan, Y.Z.; Rao, C.L.; Ao, H.; Peng, C. Panax ginseng and its ginsenosides: Potential candidates for the prevention and treatment of chemotherapy-induced side effects. J. Ginseng Res. 2021, 45, 617–630. [Google Scholar] [CrossRef]
- Truong, V.L.; Keum, Y.S.; Jeong, W.S. Red ginseng oil promotes hair growth and protects skin against UVC radiation. J. Ginseng Res. 2021, 45, 498–509. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.J.; Jin, H.; Nah, S.Y.; Lee, B.Y. Gintonin-enriched fraction improves sarcopenia by maintaining immune homeostasis in 20- to 24-month-old C57BL/6J mice. J. Ginseng Res. 2021, 45, 744–753. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.J.; Jin, H.; Lee, B.Y. The non-saponin fraction of Korean Red Ginseng ameliorates sarcopenia by regulating immune homeostasis in 22-26-month-old C57BL/6J mice. J. Ginseng Res. 2022, 46, 809–818. [Google Scholar] [CrossRef]
- Nam, S.M.; Choi, J.H.; Choi, S.H.; Cho, H.J.; Cho, Y.J.; Rhim, H.; Kim, H.C.; Cho, I.H.; Kim, D.G.; Nah, S.Y. Ginseng gintonin alleviates neurological symptoms in the G93A-SOD1 transgenic mouse model of amyotrophic lateral sclerosis through lysophosphatidic acid 1 receptor. J. Ginseng Res. 2021, 45, 390–400. [Google Scholar] [CrossRef]
- Choi, W.; Kim, H.S.; Park, S.H.; Kim, D.; Hong, Y.D.; Kim, J.H.; Cho, J.Y. Syringaresinol derived from Panax ginseng berry attenuates oxidative stress-induced skin aging via autophagy. J. Ginseng Res. 2022, 46, 536–542. [Google Scholar] [CrossRef]
- Gao, Y.; Yuan, D.; Gai, L.; Wu, X.; Shi, Y.; He, Y.; Liu, C.; Zhang, C.; Zhou, G.; Yuan, C. Saponins from Panax japonicus ameliorate age-related renal fibrosis by inhibition of inflammation mediated by NF-kappaB and TGF-beta1/Smad signaling and suppression of oxidative stress via activation of Nrf2-ARE signaling. J. Ginseng Res. 2021, 45, 408–419. [Google Scholar] [CrossRef] [PubMed]
- Yao, F.; Li, X.; Sun, J.; Cao, X.; Liu, M.; Li, Y.; Liu, Y. Thermal transformation of polar into less-polar ginsenosides through demalonylation and deglycosylation in extracts from ginseng pulp. Sci. Rep. 2021, 11, 1513. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.Y.; Zeng, J.Z.; Wong, A.S.T. Chemical Structures and Pharmacological Profiles of Ginseng Saponins. Molecules 2019, 24, 2443. [Google Scholar] [CrossRef]
- Niu, X.N.; Luo, W.; Lv, C.N.; Lu, J.C. Research progress on naturally-occurring and semi-synthetic ocotillol-type ginsenosides in the genus Panax L. (Araliaceae). Chin. J. Nat. Med. 2021, 19, 648–655. [Google Scholar] [CrossRef]
- Hou, M.; Wang, R.; Zhao, S.; Wang, Z. Ginsenosides in Panax genus and their biosynthesis. Acta Pharm. Sin. B 2021, 11, 1813–1834. [Google Scholar] [CrossRef] [PubMed]
- Shu, P.P.; Li, L.X.; He, Q.M.; Pan, J.; Li, X.L.; Zhu, M.; Yang, Y.; Qu, Y. Identification and quantification of oleanane triterpenoid saponins and potential analgesic and anti-inflammatory activities from the roots and rhizomes of Panax stipuleanatus. J. Ginseng Res. 2021, 45, 305–315. [Google Scholar] [CrossRef]
- Lin, K.; Sze, S.C.; Liu, B.; Zhang, Z.; Zhang, Z.; Zhu, P.; Wang, Y.; Deng, Q.; Yung, K.K.; Zhang, S. 20(S)-protopanaxadiol and oleanolic acid ameliorate cognitive deficits in APP/PS1 transgenic mice by enhancing hippocampal neurogenesis. J. Ginseng Res. 2021, 45, 325–333. [Google Scholar] [CrossRef]
- Lee, M.J.; Choi, J.H.; Oh, J.; Lee, Y.H.; In, J.G.; Chang, B.J.; Nah, S.Y.; Cho, I.H. Rg3-enriched Korean Red Ginseng extract inhibits blood-brain barrier disruption in an animal model of multiple sclerosis by modulating expression of NADPH oxidase 2 and 4. J. Ginseng Res. 2021, 45, 433–441. [Google Scholar] [CrossRef]
- Lee, J.O.; Hwang, S.H.; Shen, T.; Kim, J.H.; You, L.; Hu, W.; Cho, J.Y. Enhancement of skin barrier and hydration-related molecules by protopanaxatriol in human keratinocytes. J. Ginseng Res. 2021, 45, 354–360. [Google Scholar] [CrossRef]
- Yu, Y.; Wang, M.; Chen, R.; Sun, X.; Sun, G.; Sun, X. Gypenoside XVII protects against myocardial ischemia and reperfusion injury by inhibiting ER stress-induced mitochondrial injury. J. Ginseng Res. 2021, 45, 642–653. [Google Scholar] [CrossRef]
- Liu, C.X.; Xiao, P.G. Recent advances on ginseng research in China. J. Ethnopharmacol. 1992, 36, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Shibata, S. Chemistry and cancer preventing activities of ginseng saponins and some related triterpenoid compounds. J. Korean Med. Sci. 2001, 16, S28–S37. [Google Scholar] [CrossRef]
- Lu, C.; Wang, Y.; Lv, J.; Jiang, N.; Fan, B.; Qu, L.; Li, Y.; Chen, S.; Wang, F.; Liu, X. Ginsenoside Rh2 reverses sleep deprivation-induced cognitive deficit in mice. Behav. Brain Res. 2018, 349, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Park, M.W.; Ha, J.; Chung, S.H. 20(S)-ginsenoside Rg3 enhances glucose-stimulated insulin secretion and activates AMPK. Biol. Pharm. Bull. 2008, 31, 748–751. [Google Scholar] [CrossRef] [PubMed]
- Li, X.F.; Lui, C.N.; Jiang, Z.H.; Ken, Y.K. Neuroprotective effects of ginsenosides Rh1 and Rg2 on neuronal cells. Chin. Med. 2011, 6, 19. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Shin, K.K.; Kim, H.; Hong, Y.H.; Choi, W.; Kwak, Y.S.; Han, C.K.; Hyun, S.H.; Cho, J.Y. Korean Red Ginseng exerts anti-inflammatory and autophagy-promoting activities in aged mice. J. Ginseng Res. 2021, 45, 717–725. [Google Scholar] [CrossRef]
- Ahn, H.; Han, B.C.; Hong, E.J.; An, B.S.; Lee, E.; Lee, S.H.; Lee, G.S. Korean Red Ginseng attenuates ultraviolet-mediated inflammasome activation in keratinocytes. J. Ginseng Res. 2021, 45, 456–463. [Google Scholar] [CrossRef]
- Alam, M.J.; Hossain, M.A.; Bhilare, K.D.; Kang, C.W.; Kim, J.H. Korean Red Ginseng modulates immune function by upregulating CD4(+)CD8(+) T cells and NK cell activities on porcine. J. Ginseng Res. 2023, 47, 155–158. [Google Scholar] [CrossRef]
- Aravinthan, A.; Hossain, M.A.; Kim, B.; Kang, C.W.; Kim, N.S.; Hwang, K.C.; Kim, J.H. Ginsenoside Rb(1) inhibits monoiodoacetate-induced osteoarthritis in postmenopausal rats through prevention of cartilage degradation. J. Ginseng Res. 2021, 45, 287–294. [Google Scholar] [CrossRef]
- Park, M.H.; Hong, J.T. Roles of NF-κB in Cancer and Inflammatory Diseases and Their Therapeutic Approaches. Cells 2016, 5, 15. [Google Scholar] [CrossRef]
- Schuliga, M. NF-kappaB Signaling in Chronic Inflammatory Airway Disease. Biomolecules 2015, 5, 1266–1283. [Google Scholar] [CrossRef] [PubMed]
- Tak, P.P.; Firestein, G.S. NF-kappaB: A key role in inflammatory diseases. J. Clin. Investig. 2001, 107, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Rathnasamy, G.; Sivakumar, V.; Rangarajan, P.; Foulds, W.S.; Ling, E.A.; Kaur, C. NF-κB-mediated nitric oxide production and activation of caspase-3 cause retinal ganglion cell death in the hypoxic neonatal retina. Investig. Ophthalmol. Vis. Sci. 2014, 55, 5878–5889. [Google Scholar] [CrossRef] [PubMed]
- Mizgerd, J.P.; Lupa, M.M.; Spieker, M.S. NF-kappaB p50 facilitates neutrophil accumulation during LPS-induced pulmonary inflammation. BMC Immunol. 2004, 5, 10. [Google Scholar] [CrossRef]
- Zhou, F.; Zhu, Z.; Gao, J.; Yang, C.; Wen, L.; Liu, L.; Zuo, X.; Zheng, X.; Shi, Y.; Zhu, C.; et al. NFKB1 mediates Th1/Th17 activation in the pathogenesis of psoriasis. Cell. Immunol. 2018, 331, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.C. The non-canonical NF-κB pathway in immunity and inflammation. Nat. Rev. Immunol. 2017, 17, 545–558. [Google Scholar] [CrossRef] [PubMed]
- Alharbi, K.S.; Fuloria, N.K.; Fuloria, S.; Rahman, S.B.; Al-Malki, W.H.; Javed Shaikh, M.A.; Thangavelu, L.; Singh, S.K.; Rama Raju Allam, V.S.; Jha, N.K.; et al. Nuclear factor-kappa B and its role in inflammatory lung disease. Chem. Biol. Interact. 2021, 345, 109568. [Google Scholar] [CrossRef]
- Codarri, L.; Gyülvészi, G.; Tosevski, V.; Hesske, L.; Fontana, A.; Magnenat, L.; Suter, T.; Becher, B. RORγt drives production of the cytokine GM-CSF in helper T cells, which is essential for the effector phase of autoimmune neuroinflammation. Nat. Immunol. 2011, 12, 560–567. [Google Scholar] [CrossRef]
- El-Behi, M.; Ciric, B.; Dai, H.; Yan, Y.; Cullimore, M.; Safavi, F.; Zhang, G.X.; Dittel, B.N.; Rostami, A. The encephalitogenicity of T(H)17 cells is dependent on IL-1- and IL-23-induced production of the cytokine GM-CSF. Nat. Immunol. 2011, 12, 568–575. [Google Scholar] [CrossRef]
- Mockenhaupt, K.; Gonsiewski, A.; Kordula, T. RelB and Neuroinflammation. Cells 2021, 10, 1609. [Google Scholar] [CrossRef]
- Saccani, S.; Pantano, S.; Natoli, G. Modulation of NF-kappaB activity by exchange of dimers. Mol. Cell 2003, 11, 1563–1574. [Google Scholar] [CrossRef]
- Chen, X.; El Gazzar, M.; Yoza, B.K.; McCall, C.E. The NF-kappaB factor RelB and histone H3 lysine methyltransferase G9a directly interact to generate epigenetic silencing in endotoxin tolerance. J. Biol. Chem. 2009, 284, 27857–27865. [Google Scholar] [CrossRef]
- Huang, P.; Han, J.; Hui, L. MAPK signaling in inflammation-associated cancer development. Protein Cell 2010, 1, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Biehl, A.; Gadina, M.; Hasni, S.; Schwartz, D.M. JAK-STAT Signaling as a Target for Inflammatory and Autoimmune Diseases: Current and Future Prospects. Drugs 2017, 77, 521–546. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.O.; Yang, Y.; Tao, Y.; Yi, Y.S.; Cho, J.Y. Korean Red Ginseng saponin fraction exerts anti-inflammatory effects by targeting the NF-κB and AP-1 pathways. J. Ginseng Res. 2022, 46, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Kang, N.; Hu, C.; Zhang, Z.; Xu, Q.; Liu, Y.; Yang, S. Ginsenoside Rb1 exerts anti-inflammatory effects in vitro and in vivo by modulating toll-like receptor 4 dimerization and NF-kB/MAPKs signaling pathways. Phytomedicine 2020, 69, 153197. [Google Scholar] [CrossRef]
- Yang, G.; Gao, M.; Sun, Y.; Wang, C.; Fang, X.; Gao, H.; Diao, W.; Yu, H. Design, synthesis and anti-inflammatory activity of 3-amino acid derivatives of ocotillol-type sapogenins. Eur. J. Med. Chem. 2020, 202, 112507. [Google Scholar] [CrossRef]
- Shao, F.; Wang, X.; Wu, H.; Wu, Q.; Zhang, J. Microglia and Neuroinflammation: Crucial Pathological Mechanisms in Traumatic Brain Injury-Induced Neurodegeneration. Front. Aging Neurosci. 2022, 14, 825086. [Google Scholar] [CrossRef]
- Castro, P.; Azevedo, E.; Serrador, J.; Rocha, I.; Sorond, F. Hemorrhagic transformation and cerebral edema in acute ischemic stroke: Link to cerebral autoregulation. J. Neurol. Sci. 2017, 372, 256–261. [Google Scholar] [CrossRef]
- Frakes, A.E.; Ferraiuolo, L.; Haidet-Phillips, A.M.; Schmelzer, L.; Braun, L.; Miranda, C.J.; Ladner, K.J.; Bevan, A.K.; Foust, K.D.; Godbout, J.P.; et al. Microglia induce motor neuron death via the classical NF-κB pathway in amyotrophic lateral sclerosis. Neuron 2014, 81, 1009–1023. [Google Scholar] [CrossRef]
- Madhi, I.; Kim, J.H.; Shin, J.E.; Kim, Y. Ginsenoside Re exhibits neuroprotective effects by inhibiting neuroinflammation via CAMK/MAPK/NF-κB signaling in microglia. Mol. Med. Rep. 2021, 24, 698. [Google Scholar] [CrossRef]
- Saini, V.; Guada, L.; Yavagal, D.R. Global Epidemiology of Stroke and Access to Acute Ischemic Stroke Interventions. Neurology 2021, 97, S6–S16. [Google Scholar] [CrossRef] [PubMed]
- Su, L.J.; Ren, Y.C.; Chen, Z.; Ma, H.F.; Zheng, F.; Li, F.; Zhang, Y.Y.; Gong, S.S.; Kou, J.P. Ginsenoside Rb1 improves brain, lung, and intestinal barrier damage in middle cerebral artery occlusion/reperfusion (MCAO/R) micevia the PPARγ signaling pathway. Chin. J. Nat. Med. 2022, 20, 561–571. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, X.; Hu, G.; Zhang, G.; Zhao, G.; Shi, M. Ginsenoside Rd attenuates blood-brain barrier damage by suppressing proteasome-mediated signaling after transient forebrain ischemia. Neuroreport 2020, 31, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Jiang, N.; Jingwei, L.; Wang, H.; Huang, H.; Wang, Q.; Zeng, G.; Li, S.; Liu, X. Ginsenoside 20(S)-protopanaxadiol attenuates depressive-like behaviour and neuroinflammation in chronic unpredictable mild stress-induced depressive rats. Behav. Brain Res. 2020, 393, 112710. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.X.; Chu, S.F.; Wang, S.S.; Tian, Y.J.; He, W.B.; Du, Y.S.; Wang, Z.Z.; Yan, X.; Zhang, Z.; Chen, N.H. Rg1 exerts protective effect in CPZ-induced demyelination mouse model via inhibiting CXCL10-mediated glial response. Acta Pharmacol. Sin. 2022, 43, 563–576. [Google Scholar] [CrossRef]
- Yang, X.; Chu, S.F.; Wang, Z.Z.; Li, F.F.; Yuan, Y.H.; Chen, N.H. Ginsenoside Rg1 exerts neuroprotective effects in 3-nitropronpionic acid-induced mouse model of Huntington’s disease via suppressing MAPKs and NF-κB pathways in the striatum. Acta Pharmacol. Sin. 2021, 42, 1409–1421. [Google Scholar] [CrossRef] [PubMed]
- Heidenreich, P.A.; Albert, N.M.; Allen, L.A.; Bluemke, D.A.; Butler, J.; Fonarow, G.C.; Ikonomidis, J.S.; Khavjou, O.; Konstam, M.A.; Maddox, T.M.; et al. Forecasting the impact of heart failure in the United States: A policy statement from the American Heart Association. Circ. Heart Fail. 2013, 6, 606–619. [Google Scholar] [CrossRef]
- GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar] [CrossRef]
- Frangogiannis, N.G. The inflammatory response in myocardial injury, repair, and remodelling. Nat. Rev. Cardiol. 2014, 11, 255–265. [Google Scholar] [CrossRef]
- Castillo, E.C.; Vázquez-Garza, E.; Yee-Trejo, D.; García-Rivas, G.; Torre-Amione, G. What Is the Role of the Inflammation in the Pathogenesis of Heart Failure? Curr. Cardiol. Rep. 2020, 22, 139. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Nguyen, T.L.L.; Myung, C.S.; Heo, K.S. Ginsenoside Rh1 protects human endothelial cells against lipopolysaccharide-induced inflammatory injury through inhibiting TLR2/4-mediated STAT3, NF-κB, and ER stress signaling pathways. Life Sci. 2022, 309, 120973. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Hu, X.; Wang, H.; Wang, R.; Sun, Z.; Tan, X.; Liu, S.; Wang, H. Effects of a dammarane-type saponin, ginsenoside Rd, in nicotine-induced vascular endothelial injury. Phytomedicine 2020, 79, 153325. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Yan, D.; Sun, Q.; Tao, J.; Xu, L.; Sun, H.; Zhao, H. Ginsenoside Rg1 attenuates cardiomyocyte apoptosis and inflammation via the TLR4/NF-kB/NLRP3 pathway. J. Cell. Biochem. 2020, 121, 2994–3004. [Google Scholar] [CrossRef] [PubMed]
- Ren, B.; Feng, J.; Yang, N.; Guo, Y.; Chen, C.; Qin, Q. Ginsenoside Rg3 attenuates angiotensin II-induced myocardial hypertrophy through repressing NLRP3 inflammasome and oxidative stress via modulating SIRT1/NF-κB pathway. Int. Immunopharmacol. 2021, 98, 107841. [Google Scholar] [CrossRef]
- Tu, C.; Wan, B.; Zeng, Y. Ginsenoside Rg3 alleviates inflammation in a rat model of myocardial infarction via the SIRT1/NF-κB pathway. Exp. Ther. Med. 2020, 20, 238. [Google Scholar] [CrossRef]
- Yang, H.; Wang, H.; Andersson, U. Targeting Inflammation Driven by HMGB1. Front. Immunol. 2020, 11, 484. [Google Scholar] [CrossRef]
- Paudel, Y.N.; Angelopoulou, E.; Piperi, C.; Othman, I.; Aamir, K.; Shaikh, M.F. Impact of HMGB1, RAGE, and TLR4 in Alzheimer’s Disease (AD): From Risk Factors to Therapeutic Targeting. Cells 2020, 9, 383. [Google Scholar] [CrossRef]
- Zhong, H.; Li, X.; Zhou, S.; Jiang, P.; Liu, X.; Ouyang, M.; Nie, Y.; Chen, X.; Zhang, L.; Liu, Y.; et al. Interplay between RAGE and TLR4 Regulates HMGB1-Induced Inflammation by Promoting Cell Surface Expression of RAGE and TLR4. J. Immunol. 2020, 205, 767–775. [Google Scholar] [CrossRef] [PubMed]
- Qi, Z.; Yan, Z.; Wang, Y.; Ji, N.; Yang, X.; Zhang, A.; Li, M.; Xu, F.; Zhang, J. Ginsenoside Rh2 Inhibits NLRP3 Inflammasome Activation and Improves Exosomes to Alleviate Hypoxia-Induced Myocardial Injury. Front. Immunol. 2022, 13, 883946. [Google Scholar] [CrossRef]
- Sharma, A.; Nagalli, S. Chronic Liver Disease. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Cheemerla, S.; Balakrishnan, M. Global Epidemiology of Chronic Liver Disease. Clin. Liver Dis. 2021, 17, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Dixon, L.J.; Barnes, M.; Tang, H.; Pritchard, M.T.; Nagy, L.E. Kupffer cells in the liver. Compr. Physiol. 2013, 3, 785–797. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Deng, X.; Liu, Y.; Tan, Q.; Huang, G.; Che, Q.; Guo, J.; Su, Z. Kupffer Cells in Non-alcoholic Fatty Liver Disease: Friend or Foe? Int. J. Biol. Sci. 2020, 16, 2367–2378. [Google Scholar] [CrossRef] [PubMed]
- Suraweera, D.B.; Weeratunga, A.N.; Hu, R.W.; Pandol, S.J.; Hu, R. Alcoholic hepatitis: The pivotal role of Kupffer cells. World J. Gastrointest. Pathophysiol. 2015, 6, 90–98. [Google Scholar] [CrossRef]
- Xia, T.; Fang, B.; Kang, C.; Zhao, Y.; Qiang, X.; Zhang, X.; Wang, Y.; Zhong, T.; Xiao, J.; Wang, M. Hepatoprotective Mechanism of Ginsenoside Rg1 against Alcoholic Liver Damage Based on Gut Microbiota and Network Pharmacology. Oxid. Med. Cell. Longev. 2022, 2022, 5025237. [Google Scholar] [CrossRef]
- Cignarelli, A.; Genchi, V.A.; Perrini, S.; Natalicchio, A.; Laviola, L.; Giorgino, F. Insulin and Insulin Receptors in Adipose Tissue Development. Int. J. Mol. Sci. 2019, 20, 759. [Google Scholar] [CrossRef]
- Kitade, H.; Chen, G.; Ni, Y.; Ota, T. Nonalcoholic Fatty Liver Disease and Insulin Resistance: New Insights and Potential New Treatments. Nutrients 2017, 9, 387. [Google Scholar] [CrossRef]
- Rinaldi, L.; Pafundi, P.C.; Galiero, R.; Caturano, A.; Morone, M.V.; Silvestri, C.; Giordano, M.; Salvatore, T.; Sasso, F.C. Mechanisms of Non-Alcoholic Fatty Liver Disease in the Metabolic Syndrome. A Narrative Review. Antioxidants 2021, 10, 270. [Google Scholar] [CrossRef]
- Gao, B.; Tsukamoto, H. Inflammation in Alcoholic and Nonalcoholic Fatty Liver Disease: Friend or Foe? Gastroenterology 2016, 150, 1704–1709. [Google Scholar] [CrossRef]
- Su, W.Y.; Fan, M.L.; Li, Y.; Hu, J.N.; Cai, E.B.; Zhu, H.Y.; Song, M.J.; Li, W. 20(S)-ginsenoside Rh1 alleviates T2DM induced liver injury via the Akt/FOXO1 pathway. Chin. J. Nat. Med. 2022, 20, 669–678. [Google Scholar] [CrossRef]
- Liu, Q.; Bengmark, S.; Qu, S. The role of hepatic fat accumulation in pathogenesis of non-alcoholic fatty liver disease (NAFLD). Lipids Health Dis. 2010, 9, 42. [Google Scholar] [CrossRef]
- Lin, Y.; Hu, Y.; Hu, X.; Yang, L.; Chen, X.; Li, Q.; Gu, X. Ginsenoside Rb2 improves insulin resistance by inhibiting adipocyte pyroptosis. Adipocyte 2020, 9, 302–312. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Cai, X.; Le, R.; Zhang, M.; Gu, X.; Shen, F.; Hong, G.; Chen, Z. Isoliquiritigenin protects against sepsis-induced lung and liver injury by reducing inflammatory responses. Biochem. Biophys. Res. Commun. 2018, 496, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Hamacher, J.; Gorshkov, B.; White, R.; Sridhar, S.; Verin, A.; Chakraborty, T.; Lucas, R. The Dual Role of TNF in Pulmonary Edema. J. Cardiovasc. Dis. Res. 2010, 1, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Jang, W.Y.; Lee, H.P.; Kim, S.A.; Huang, L.; Yoon, J.H.; Shin, C.Y.; Mitra, A.; Kim, H.G.; Cho, J.Y. Angiopteris cochinchinensis de Vriese Ameliorates LPS-Induced Acute Lung Injury via Src Inhibition. Plants 2022, 11, 1306. [Google Scholar] [CrossRef] [PubMed]
- Imanifooladi, A.A.; Yazdani, S.; Nourani, M.R. The role of nuclear factor-kappaB in inflammatory lung disease. Inflamm. Allergy Drug Targets 2010, 9, 197–205. [Google Scholar] [CrossRef]
- Li, H.; Cui, L.; Liu, Q.; Dou, S.; Wang, W.; Xie, M.; Xu, X.; Zheng, C.; Li, T.; Huang, S.; et al. Ginsenoside Rb3 Alleviates CSE-induced TROP2 Upregulation through p38 MAPK and NF-κB Pathways in Basal Cells. Am. J. Respir. Cell Mol. Biol. 2021, 64, 747–759. [Google Scholar] [CrossRef]
- Liu, J.; Fan, G.; Tao, N.; Feng, F.; Meng, C.; Sun, T. Ginsenoside Rb1 Alleviates Bleomycin-Induced Pulmonary Inflammation and Fibrosis by Suppressing Central Nucleotide-Binding Oligomerization-, Leucine-Rich Repeat-, and Pyrin Domains-Containing Protein Three Inflammasome Activation and the NF-κB Pathway. Drug Des. Dev. Ther. 2022, 16, 1793–1809. [Google Scholar] [CrossRef]
- Wei, E.; Fang, X.; Jia, P.; Li, M.; Jin, P.; Li, F.; Wang, H.; Gao, D. Ginsenoside Rb1 Alleviates Lipopolysaccharide-Induced Inflammatory Injury by Downregulating miR-222 in WI-38 Cells. Cell Transplant. 2021, 30, 9636897211002787. [Google Scholar] [CrossRef]
- Ding, J.; Xu, Z.; Zhang, Y.; Tan, C.; Hu, W.; Wang, M.; Xu, Y.; Tang, J. Exosome-mediated miR-222 transferring: An insight into NF-κB-mediated breast cancer metastasis. Exp. Cell Res. 2018, 369, 129–138. [Google Scholar] [CrossRef]
- Ji, Q.; Sun, Z.; Yang, Z.; Zhang, W.; Ren, Y.; Chen, W.; Yao, M.; Nie, S. Protective effect of ginsenoside Rg1 on LPS-induced apoptosis of lung epithelial cells. Mol. Immunol. 2021, 136, 168–174. [Google Scholar] [CrossRef]
- Wardyn, J.D.; Ponsford, A.H.; Sanderson, C.M. Dissecting molecular cross-talk between Nrf2 and NF-κB response pathways. Biochem. Soc. Trans. 2015, 43, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, K.; Nakano, Y.; Imai, T.; Takagi, T.; Tsuruma, K.; Shimazawa, M.; Iwama, T.; Hara, H. A novel nuclear factor erythroid 2-related factor 2 (Nrf2) activator RS9 attenuates brain injury after ischemia reperfusion in mice. Neuroscience 2016, 333, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.U.; Lin, J.; Liu, X.; Li, H.; Lu, W.; Zhong, Z.; Zhang, H.; Waqas, M.; Shen, L. Insights into predicting diabetic nephropathy using urinary biomarkers. Biochim. Biophys. Acta. Proteins Proteom. 2020, 1868, 140475. [Google Scholar] [CrossRef] [PubMed]
- Samsu, N. Diabetic Nephropathy: Challenges in Pathogenesis, Diagnosis, and Treatment. BioMed Res. Int. 2021, 2021, 1497449. [Google Scholar] [CrossRef]
- Li, Y.; Hou, J.G.; Liu, Z.; Gong, X.J.; Hu, J.N.; Wang, Y.P.; Liu, W.C.; Lin, X.H.; Wang, Z.; Li, W. Alleviative effects of 20(R)-Rg3 on HFD/STZ-induced diabetic nephropathy via MAPK/NF-κB signaling pathways in C57BL/6 mice. J. Ethnopharmacol. 2021, 267, 113500. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhu, C.; Yang, H.; Deng, J.; Fan, D. Protective effect of ginsenoside Rg5 against kidney injury via inhibition of NLRP3 inflammasome activation and the MAPK signaling pathway in high-fat diet/streptozotocin-induced diabetic mice. Pharmacol. Res. 2020, 155, 104746. [Google Scholar] [CrossRef]
- Liang, W.J.; Yang, H.W.; Liu, H.N.; Qian, W.; Chen, X.L. HMGB1 upregulates NF-kB by inhibiting IKB-α and associates with diabetic retinopathy. Life Sci. 2020, 241, 117146. [Google Scholar] [CrossRef]
- Zhou, S.; Lu, S.; Guo, S.; Zhao, L.; Han, Z.; Li, Z. Protective Effect of Ginsenoside Rb1 Nanoparticles Against Contrast-Induced Nephropathy by Inhibiting High Mobility Group Box 1 Gene/Toll-Like Receptor 4/NF-κB Signaling Pathway. J. Biomed. Nanotechnol. 2021, 17, 2085–2098. [Google Scholar] [CrossRef]
- Dong, L.; Xie, J.; Wang, Y.; Jiang, H.; Chen, K.; Li, D.; Wang, J.; Liu, Y.; He, J.; Zhou, J.; et al. Mannose ameliorates experimental colitis by protecting intestinal barrier integrity. Nat. Commun. 2022, 13, 4804. [Google Scholar] [CrossRef]
- Wunderlich, C.M.; Ackermann, P.J.; Ostermann, A.L.; Adams-Quack, P.; Vogt, M.C.; Tran, M.L.; Nikolajev, A.; Waisman, A.; Garbers, C.; Theurich, S.; et al. Obesity exacerbates colitis-associated cancer via IL-6-regulated macrophage polarisation and CCL-20/CCR-6-mediated lymphocyte recruitment. Nat. Commun. 2018, 9, 1646. [Google Scholar] [CrossRef] [PubMed]
- He, R.; Li, Y.; Han, C.; Lin, R.; Qian, W.; Hou, X. L-Fucose ameliorates DSS-induced acute colitis via inhibiting macrophage M1 polarization and inhibiting NLRP3 inflammasome and NF-kB activation. Int. Immunopharmacol. 2019, 73, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Cerf-Bensussan, N.; Gaboriau-Routhiau, V. The immune system and the gut microbiota: Friends or foes? Nat. Rev. Immunol. 2010, 10, 735–744. [Google Scholar] [CrossRef]
- Jang, Y.J.; Kim, W.K.; Han, D.H.; Lee, K.; Ko, G. Lactobacillus fermentum species ameliorate dextran sulfate sodium-induced colitis by regulating the immune response and altering gut microbiota. Gut Microbes 2019, 10, 696–711. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Paul, S.; Kundu, P. NF-κB Regulation by Gut Microbiota Decides Homeostasis or Disease Outcome During Ageing. Front. Cell. Dev. Biol. 2022, 10, 874940. [Google Scholar] [CrossRef]
- Chen, H.; Yang, H.; Deng, J.; Fan, D. Ginsenoside Rk3 Ameliorates Obesity-Induced Colitis by Regulating of Intestinal Flora and the TLR4/NF-κB Signaling Pathway in C57BL/6 Mice. J. Agric. Food Chem. 2021, 69, 3082–3093. [Google Scholar] [CrossRef]
- Fu, T.; Li, Y.; Oh, T.G.; Cayabyab, F.; He, N.; Tang, Q.; Coulter, S.; Truitt, M.; Medina, P.; He, M.; et al. FXR mediates ILC-intrinsic responses to intestinal inflammation. Proc. Natl. Acad. Sci. USA 2022, 119, e2213041119. [Google Scholar] [CrossRef]
- Tang, K.; Kong, D.; Peng, Y.; Guo, J.; Zhong, Y.; Yu, H.; Mai, Z.; Chen, Y.; Chen, Y.; Cui, T.; et al. Ginsenoside Rc attenuates DSS-induced ulcerative colitis, intestinal inflammatory, and barrier function by activating the farnesoid X receptor. Front. Pharmacol. 2022, 13, 1000444. [Google Scholar] [CrossRef]
- Duan, X.; Cai, H.; Hu, T.; Lin, L.; Zeng, L.; Wang, H.; Cao, L.; Li, X. Ginsenoside Rg3 treats acute radiation proctitis through the TLR4/MyD88/NF-κB pathway and regulation of intestinal flora. Front. Cell. Infect. Microbiol. 2022, 12, 1028576. [Google Scholar] [CrossRef]
- Weitzmann, M.N. Bone and the Immune System. Toxicol. Pathol. 2017, 45, 911–924. [Google Scholar] [CrossRef]
- Tao, H.; Li, W.; Zhang, W.; Yang, C.; Zhang, C.; Liang, X.; Yin, J.; Bai, J.; Ge, G.; Zhang, H.; et al. Urolithin A suppresses RANKL-induced osteoclastogenesis and postmenopausal osteoporosis by, suppresses inflammation and downstream NF-κB activated pyroptosis pathways. Pharmacol. Res. 2021, 174, 105967. [Google Scholar] [CrossRef] [PubMed]
- Palazzo, C.; Nguyen, C.; Lefevre-Colau, M.M.; Rannou, F.; Poiraudeau, S. Risk factors and burden of osteoarthritis. Ann. Phys. Rehabil. Med. 2016, 59, 134–138. [Google Scholar] [CrossRef] [PubMed]
- Philpott, H.T.; O’Brien, M.; McDougall, J.J. Attenuation of early phase inflammation by cannabidiol prevents pain and nerve damage in rat osteoarthritis. Pain 2017, 158, 2442–2451. [Google Scholar] [CrossRef] [PubMed]
- Berenbaum, F.; Walker, C. Osteoarthritis and inflammation: A serious disease with overlapping phenotypic patterns. Postgrad. Med. 2020, 132, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Rucci, N.; Rufo, A.; Alamanou, M.; Capulli, M.; Del Fattore, A.; Ahrman, E.; Capece, D.; Iansante, V.; Zazzeroni, F.; Alesse, E.; et al. The glycosaminoglycan-binding domain of PRELP acts as a cell type-specific NF-kappaB inhibitor that impairs osteoclastogenesis. J. Cell Biol. 2009, 187, 669–683. [Google Scholar] [CrossRef]
- Zhang, D.; Du, J.; Yu, M.; Suo, L. Ginsenoside Rb1 prevents osteoporosis via the AHR/PRELP/NF-κB signaling axis. Phytomed. Int. J. Phytother. Phytopharm. 2022, 104, 154205. [Google Scholar] [CrossRef]
- Hossain, M.A.; Alam, M.J.; Kim, B.; Kang, C.W.; Kim, J.H. Ginsenoside-Rb1 prevents bone cartilage destruction through down-regulation of p-Akt, p-P38, and p-P65 signaling in rabbit. Phytomed. Int. J. Phytother. Phytopharm. 2022, 100, 154039. [Google Scholar] [CrossRef]
- Sun, M.; Ji, Y.; Li, Z.; Chen, R.; Zhou, S.; Liu, C.; Du, M. Ginsenoside Rb3 Inhibits Pro-Inflammatory Cytokines via MAPK/AKT/NF-κB Pathways and Attenuates Rat Alveolar Bone Resorption in Response to Porphyromonas gingivalis LPS. Molecules 2020, 25, 4815. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef]
- Smith, B.N.; Bhowmick, N.A. Role of EMT in Metastasis and Therapy Resistance. J. Clin. Med. 2016, 5, 17. [Google Scholar] [CrossRef]
- Jeon, J.H.; Kim, D.K.; Shin, Y.; Kim, H.Y.; Song, B.; Lee, E.Y.; Kim, J.K.; You, H.J.; Cheong, H.; Shin, D.H.; et al. Migration and invasion of drug-resistant lung adenocarcinoma cells are dependent on mitochondrial activity. Exp. Mol. Med. 2016, 48, e277. [Google Scholar] [CrossRef] [PubMed]
- Fanfone, D.; Wu, Z.; Mammi, J.; Berthenet, K.; Neves, D.; Weber, K.; Halaburkova, A.; Virard, F.; Bunel, F.; Jamard, C.; et al. Confined migration promotes cancer metastasis through resistance to anoikis and increased invasiveness. Elife 2022, 11, e73150. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Lin, L.; Zhang, Z.; Zhang, H.; Hu, H. Targeting NF-κB pathway for the therapy of diseases: Mechanism and clinical study. Signal Transduct. Target. Ther. 2020, 5, 209. [Google Scholar] [CrossRef]
- Rasmi, R.R.; Sakthivel, K.M.; Guruvayoorappan, C. NF-κB inhibitors in treatment and prevention of lung cancer. Biomed. Pharmacother. 2020, 130, 110569. [Google Scholar] [CrossRef] [PubMed]
- Fares, J.; Fares, M.Y.; Khachfe, H.H.; Salhab, H.A.; Fares, Y. Molecular principles of metastasis: A hallmark of cancer revisited. Signal Transduct. Target. Ther. 2020, 5, 28. [Google Scholar] [CrossRef]
- Nishida, N.; Yano, H.; Nishida, T.; Kamura, T.; Kojiro, M. Angiogenesis in cancer. Vasc. Health Risk Manag. 2006, 2, 213–219. Available online: https://www.dovepress.com/angiogenesis-in-cancer-peer-reviewed-fulltext-article-VHRM (accessed on 24 February 2023). [CrossRef]
- Kessenbrock, K.; Plaks, V.; Werb, Z. Matrix metalloproteinases: Regulators of the tumor microenvironment. Cell 2010, 141, 52–67. [Google Scholar] [CrossRef]
- Webb, A.H.; Gao, B.T.; Goldsmith, Z.K.; Irvine, A.S.; Saleh, N.; Lee, R.P.; Lendermon, J.B.; Bheemreddy, R.; Zhang, Q.; Brennan, R.C.; et al. Inhibition of MMP-2 and MMP-9 decreases cellular migration, and angiogenesis in in vitro models of retinoblastoma. BMC Cancer 2017, 17, 434. [Google Scholar] [CrossRef]
- Børretzen, A.; Gravdal, K.; Haukaas, S.A.; Mannelqvist, M.; Beisland, C.; Akslen, L.A.; Halvorsen, O.J. The epithelial-mesenchymal transition regulators Twist, Slug, and Snail are associated with aggressive tumour features and poor outcome in prostate cancer patients. J. Pathol. Clin. Res. 2021, 7, 253–270. [Google Scholar] [CrossRef]
- Pires, B.R.; Mencalha, A.L.; Ferreira, G.M.; de Souza, W.F.; Morgado-Díaz, J.A.; Maia, A.M.; Corrêa, S.; Abdelhay, E.S. NF-kappaB Is Involved in the Regulation of EMT Genes in Breast Cancer Cells. PLoS ONE 2017, 12, e0169622. [Google Scholar] [CrossRef]
- Rhee, J.W.; Lee, K.W.; Kim, D.; Lee, Y.; Jeon, O.H.; Kwon, H.J.; Kim, D.S. NF-kappaB-dependent regulation of matrix metalloproteinase-9 gene expression by lipopolysaccharide in a macrophage cell line RAW 264.7. J. Biochem. Mol. Biol. 2007, 40, 88–94. [Google Scholar] [CrossRef] [PubMed]
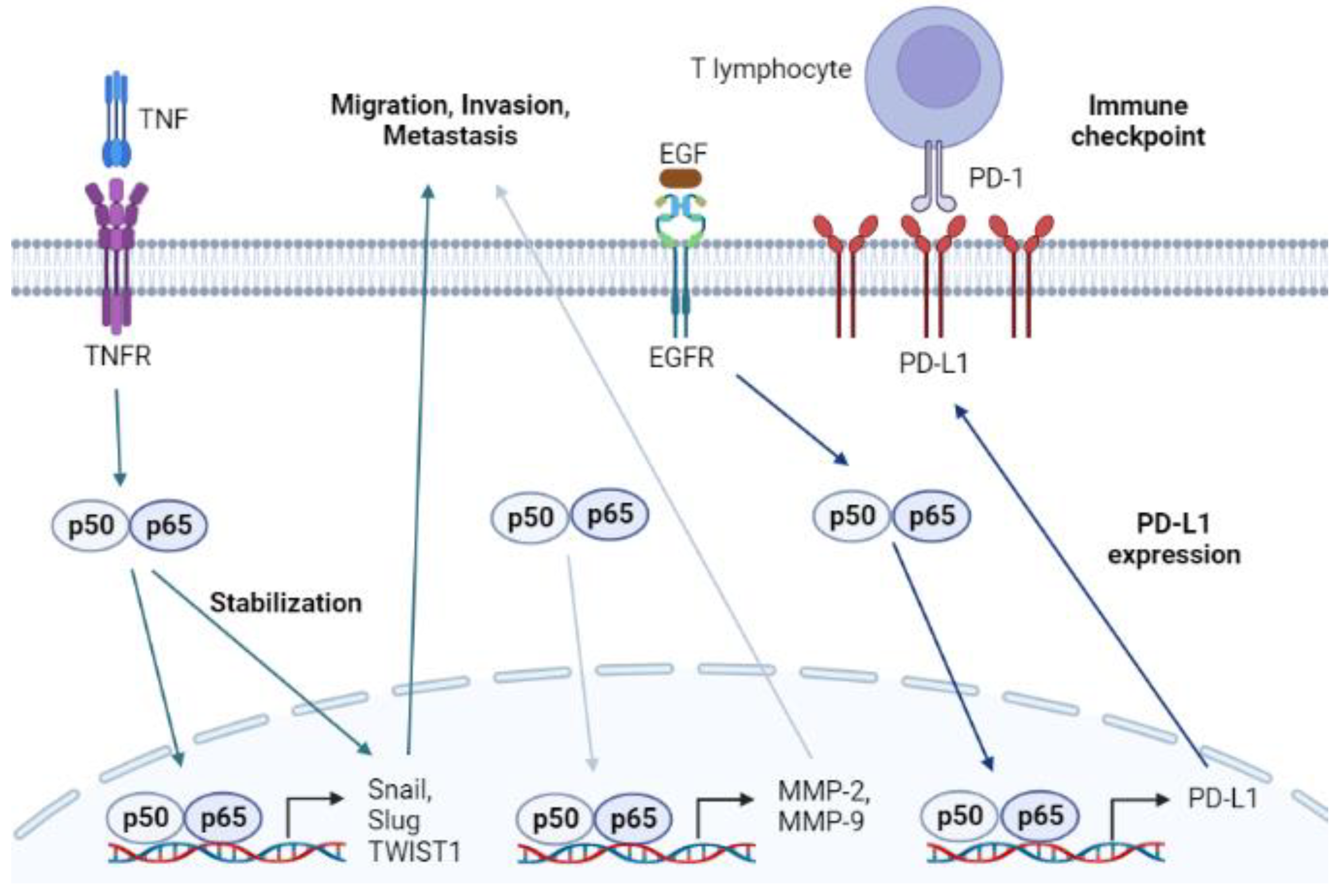
- Wu, Y.; Zhou, B.P. TNF-alpha/NF-kappaB/Snail pathway in cancer cell migration and invasion. Br. J. Cancer 2010, 102, 639–644. [Google Scholar] [CrossRef]
- Nishimura, Y.; Takiguchi, S.; Ito, S.; Itoh, K. EGF-stimulated AKT activation is mediated by EGFR recycling via an early endocytic pathway in a gefitinib-resistant human lung cancer cell line. Int. J. Oncol. 2015, 46, 1721–1729. [Google Scholar] [CrossRef]
- Bai, D.; Ueno, L.; Vogt, P.K. Akt-mediated regulation of NFkappaB and the essentialness of NFkappaB for the oncogenicity of PI3K and Akt. Int. J. Cancer 2009, 125, 2863–2870. [Google Scholar] [CrossRef] [PubMed]
- Antonangeli, F.; Natalini, A.; Garassino, M.C.; Sica, A.; Santoni, A.; Di Rosa, F. Regulation of PD-L1 Expression by NF-κB in Cancer. Front. Immunol. 2020, 11, 584626. [Google Scholar] [CrossRef]
- Wang, X.; Yang, L.; Huang, F.; Zhang, Q.; Liu, S.; Ma, L.; You, Z. Inflammatory cytokines IL-17 and TNF-α up-regulate PD-L1 expression in human prostate and colon cancer cells. Immunol. Lett. 2017, 184, 7–14. [Google Scholar] [CrossRef] [PubMed]
- He, X.L.; Xu, X.H.; Shi, J.J.; Huang, M.; Wang, Y.; Chen, X.; Lu, J.J. Anticancer Effects of Ginsenoside Rh2: A Systematic Review. Curr. Mol. Pharmacol. 2022, 15, 179–189. [Google Scholar] [CrossRef]
- Hong, J.; Gwon, D.; Jang, C.Y. Ginsenoside Rg1 suppresses cancer cell proliferation through perturbing mitotic progression. J. Ginseng Res. 2022, 46, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Ye, Y.; Xiao, L.; Duan, X.; Zhang, Y.; Zhang, H. Anticancer effects of ginsenoside Rg3 (Review). Int. J. Mol. Med. 2017, 39, 507–518. [Google Scholar] [CrossRef]
- Ahuja, A.; Kim, J.H.; Kim, J.H.; Yi, Y.S.; Cho, J.Y. Functional role of ginseng-derived compounds in cancer. J. Ginseng Res. 2018, 42, 248–254. [Google Scholar] [CrossRef]
- Jin, Y.; Huynh, D.T.N.; Myung, C.S.; Heo, K.S. Ginsenoside Rh1 Prevents Migration and Invasion through Mitochondrial ROS-Mediated Inhibition of STAT3/NF-κB Signaling in MDA-MB-231 Cells. Int. J. Mol. Sci. 2021, 22, 458. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Choi, P.; Kim, T.; Kim, Y.; Song, B.G.; Park, Y.T.; Choi, S.J.; Yoon, C.H.; Lim, W.C.; Ko, H.; et al. Ginsenosides Rk1 and Rg5 inhibit transforming growth factor-β1-induced epithelial-mesenchymal transition and suppress migration, invasion, anoikis resistance, and development of stem-like features in lung cancer. J. Ginseng Res. 2021, 45, 134–148. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Yang, J.; Qu, L.; Deng, X.; Duan, Z.; Fu, R.; Liang, L.; Fan, D. Ginsenoside Rk1 induces apoptosis and downregulates the expression of PD-L1 by targeting the NF-κB pathway in lung adenocarcinoma. Food Funct. 2020, 11, 456–471. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chu, S.; Lin, M.; Gao, Y.; Liu, Y.; Yang, S.; Zhou, X.; Zhang, Y.; Hu, Y.; Wang, H.; et al. Anticancer property of ginsenoside Rh2 from ginseng. Eur. J. Med. Chem. 2020, 203, 112627. [Google Scholar] [CrossRef]
- Wang, Y.S.; Li, H.; Li, Y.; Zhang, S.; Jin, Y.H. (20S)G-Rh2 Inhibits NF-κB Regulated Epithelial-Mesenchymal Transition by Targeting Annexin A2. Biomolecules 2020, 10, 528. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Gao, H.; Feng, X.; Bi, C.; Zhang, J.; Yin, J. Ginsenoside Rh2 impedes proliferation and migration and induces apoptosis by regulating NF-κB, MAPK, and PI3K/Akt/mTOR signaling pathways in osteosarcoma cells. J. Biochem. Mol. Toxicol. 2020, 34, e22597. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yang, L.; Dong, L.; Yang, Z.W.; Zhang, J.; Zhang, S.L.; Niu, M.J.; Xia, J.W.; Gong, Y.; Zhu, N.; et al. Crosstalk between the Akt/mTORC1 and NF-κB signaling pathways promotes hypoxia-induced pulmonary hypertension by increasing DPP4 expression in PASMCs. Acta Pharmacol. Sin. 2019, 40, 1322–1333. [Google Scholar] [CrossRef]
- Surh, Y.J.; Na, H.K.; Lee, J.Y.; Keum, Y.S. Molecular mechanisms underlying anti-tumor promoting activities of heat-processed Panax ginseng C.A. Meyer. J. Korean Med. Sci. 2001, 16, S38–S41. [Google Scholar] [CrossRef]
- Wei, W.; Liu, L.; Liu, X.; Tao, Y.; Gong, J.; Wang, Y.; Liu, S. Black ginseng protects against Western diet-induced nonalcoholic steatohepatitis by modulating the TLR4/NF-κB signaling pathway in mice. J. Food Biochem. 2022, 46, e14432. [Google Scholar] [CrossRef]
- Pei, H.; Liu, S.; Zeng, J.; Liu, J.; Wu, H.; Chen, W.; He, Z.; Du, R. Ros-mediated mitochondrial oxidative stress is involved in the ameliorating effect of ginsenoside GSLS on chlorpyrifos induced hepatotoxicity in mice. Aging 2022, 15, 675–688. [Google Scholar] [CrossRef]
- Saisho, Y. Metformin and Inflammation: Its Potential Beyond Glucose-lowering Effect. Endocr. Metab. Immune Disord.-Drug Targets 2015, 15, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Choi, H.K.; Huang, L. State of Panax ginseng Research: A Global Analysis. Molecules 2017, 22, 1518. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.E.; Mwesige, B.; Yi, Y.S.; Yoo, B.C. Ginsenosides: The need to move forward from bench to clinical trials. J. Ginseng Res. 2019, 43, 361–367. [Google Scholar] [CrossRef]
- Lu, P.; Su, W.; Miao, Z.H.; Niu, H.R.; Liu, J.; Hua, Q.L. Effect and mechanism of ginsenoside Rg3 on postoperative life span of patients with non-small cell lung cancer. Chin. J. Integr. Med. 2008, 14, 33–36. [Google Scholar] [CrossRef] [PubMed]
- Miao, L.; Yang, Y.; Li, Z.; Fang, Z.; Zhang, Y.; Han, C.C. Ginsenoside Rb2: A review of pharmacokinetics and pharmacological effects. J. Ginseng Res. 2022, 46, 206–213. [Google Scholar] [CrossRef]
- Won, H.J.; Kim, H.I.; Park, T.; Kim, H.; Jo, K.; Jeon, H.; Ha, S.J.; Hyun, J.M.; Jeong, A.; Kim, J.S.; et al. Non-clinical pharmacokinetic behavior of ginsenosides. J. Ginseng Res. 2019, 43, 354–360. [Google Scholar] [CrossRef]
- Sun, M.; Zhu, C.; Long, J.; Lu, C.; Pan, X.; Wu, C. PLGA microsphere-based composite hydrogel for dual delivery of ciprofloxacin and ginsenoside Rh2 to treat Staphylococcus aureus-induced skin infections. Drug Deliv. 2020, 27, 632–641. [Google Scholar] [CrossRef]
- Mathiyalagan, R.; Wang, C.; Kim, Y.J.; Castro-Aceituno, V.; Ahn, S.; Subramaniyam, S.; Simu, S.Y.; Jiménez-Pérez, Z.E.; Yang, D.C.; Jung, S.K. Preparation of Polyethylene Glycol-Ginsenoside Rh1 and Rh2 Conjugates and Their Efficacy against Lung Cancer and Inflammation. Molecules 2019, 24, 4367. [Google Scholar] [CrossRef]
- Jin, L.; Liu, J.; Wang, S.; Zhao, L.; Li, J. Evaluation of 20(S)-ginsenoside Rg3 loaded hydrogel for the treatment of perianal ulcer in a rat model. J. Ginseng Res. 2022, 46, 771–779. [Google Scholar] [CrossRef]
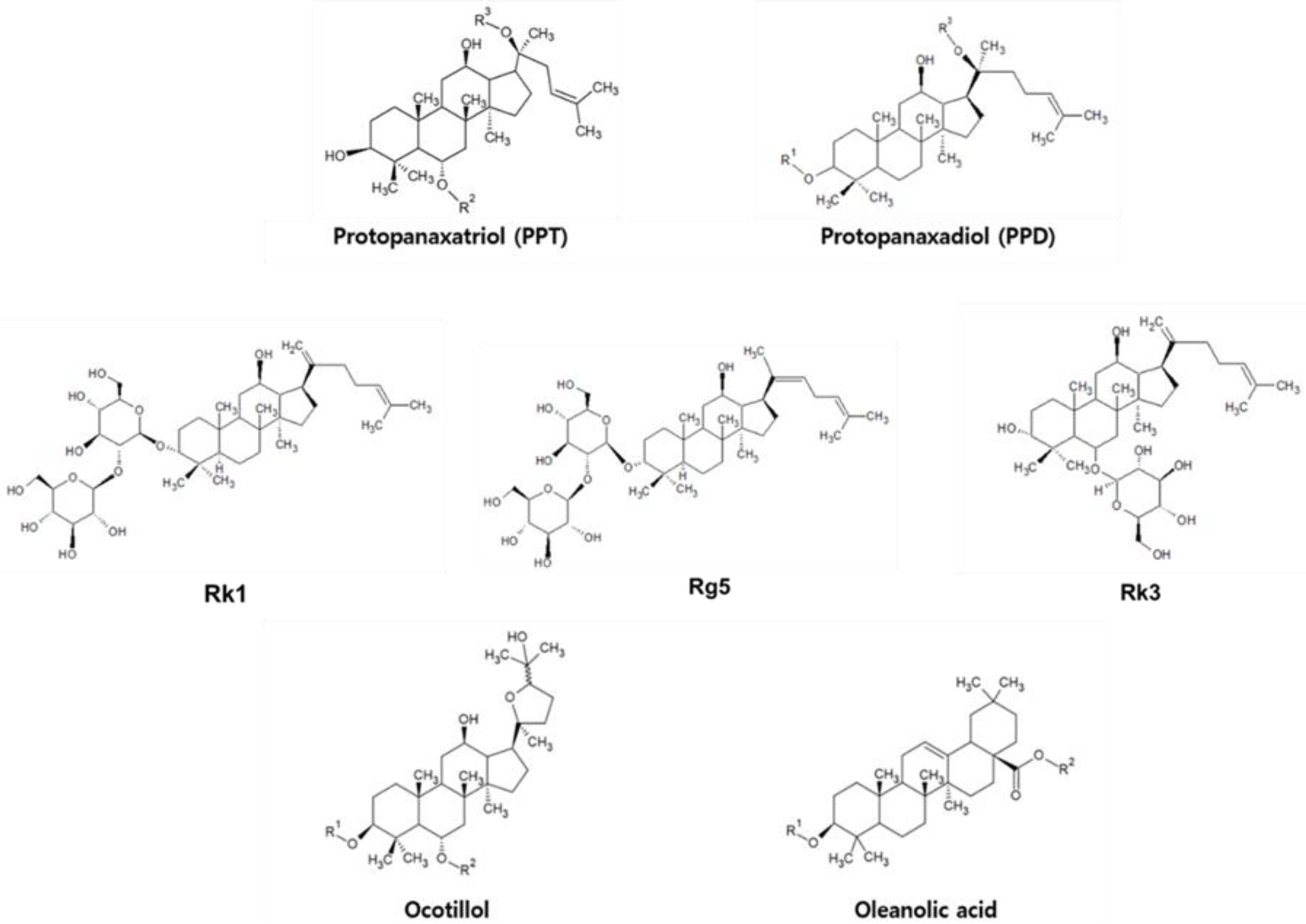
| PPD Type | PPT Type |
|---|---|
| Rb1, Rd, Rg3, 20(R)-Rg3, Rh2, Rb3, 20(S)-Rh2, Rk1, Rg5, and Rc | Rg1, Rh1, Rk3, R1 |
| PPT | Ginsenoside | R2 | R3 |
| Rg1 | OGlc | Glc | |
| Rh1 | OGlc | H | |
| R1 | Glc(2 → 1)Xyl | Glc | |
| PPD | Ginsenoside | R1 | R3 |
| Rb1 | Glc(2 → 1)Glc | Glc(6 → 1)Glc | |
| Rd | Glc(2 → 1)Glc | Glc | |
| Rg3 | Glc(2 → 1)Glc | H | |
| 20(R)-Rg3 | Glc(2 → 1)Glc | H | |
| Rh2 | Glc | H | |
| 20(S)-Rh2 | Glc | H | |
| Rb3 | Glc(2 → 1)Glc | Glc(6 → 1)Xyl | |
| Rc | Glc(2 → 1)Glc | Glc(6 → 1)Araf |
| Inflammation Site | Test Type | Dose | Type | Molecular Mechanism | Ref. |
|---|---|---|---|---|---|
| Neurodegenerative disorder | In vivo (Middle cerebral artery occlusion-/reperfusion-induced mouse ischemic stroke model.) (Strain not mentioned.) | Not mentioned | Rb1 |
| [83] |
| In vivo (Middle cerebral artery occlusion-/reperfusion-induced Sprague Dawley rat ischemic stroke model.) | 30 mg/kg | Rd |
| [84] | |
| In vivo (CUMS-induced Sprague Dawley rat depression model.) | 20, 40 μM/kg | 20(S)-protopanaxadiol |
| [85] | |
| In vitro (LPS-treated CTX TNA2 cell line.) In vivo (CPZ-induced C57BL/6N mouse demyelination model.) | 40 μM 5, 10, and 20 mg/kg | Rg1 |
| [86] | |
| In vivo (3-NP-induced C57BL/6 mouse Huntington’s disease model.) | 10, 20, and 40 mg/kg | Rg1 |
| [87] | |
| Myocardial injury | In vitro (LPS-treated HUVEC cell line.) In vivo (LPS-induced C57BL/6 mouse endothelial cell injury model.) | 25, 50 μM 5 mg/kg | Rh1 |
| [92] |
| In vitro (Nicotine-treated HUVEC cell line.) In vivo (Nicotine-induced Sprague Dawley rat endothelial cell injury model.) | 7.5, 15, and 30 μM 25, 50 mg/kg | Rd |
| [93] | |
| Ex vivo (LPS-treated neonatal rat cardiomyocytes.) In vivo (LPS-induced C57BL/6J mouse septic cardiac dysfunction model.) | 20 μM Not mentioned | Rg1 |
| [94] | |
| In vitro (Angiotensin-II-treated AC16 and HCM cell line.) In vivo (Transverse aortic constriction-induced Sprague Dawley rat myocardial hypertrophy model.) | 10, 20, and 40 μM 30 mg/kg | Rg3 |
| [95] | |
| In vivo (Coronary-artery-ligation-induced Sprague Dawley rat myocardial infarction model.) | 30 mg/kg | Rg3 |
| [96] | |
| In vitro (Hypoxia-treated H9C2 cell line.) | 2 μM | Rh2 |
| [100] | |
| Metabolic disorder | In vivo (ICR mouse alcoholic liver damage model.) | 10, 40 mg/kg | Rg1 |
| [106] |
| In vivo (STZ-induced C57BL/6 mouse liver injury model.) | 5, 10 mg/kg | 20(S)-Rh1 |
| [111] | |
| In vitro (TNF-α-treated 3T3-L1 preadipocyte cell line.) In vivo (HFD-induced C57BL/6 mouse obesity model.) | 5 ng/mL 40 mg/kg | Rb2 |
| [113] | |
| Pulmonary inflammation | Ex vivo (CSE-treated human airway basal cells.) | 10, 20, 50 μM | Rb3 |
| [118] |
| In vitro (LPS-treated THP-1 and MRC-5 cell line.) In vivo (BLM-induced C57BL/6 mouse pulmonary fibrosis model.) | 20 μM 20 mg/kg | Rb1 |
| [119] | |
| In vitro (LPS-treated WI-38 cell line.) | 20, 30 μM | Rb1 |
| [120] | |
| In vitro (LPS-treated MLE-12 cell line.) In vivo (LPS-induced C57BL/6 mouse ALI model.) | Not mentioned 30 mg/kg | Rg1 |
| [122] | |
| Kidney inflammation | In vivo (HFD-induced C57BL/6 mouse T2D model.) | 30, 60 mg/kg | 20(R)-Rg3 |
| [127] |
| In vitro (Iopromide-treated NRK-52E cell line.) In vivo (CIN C57BL/6 mouse nepropathy model.) | Not mentioned 20, 70 mg/kg | Rb1-nanoparticle (PEG/PLGA) |
| [130] | |
| In vivo (HFD/STZ-induced C57BL/6 mouse diabetes model.) | 30, 60 mg/kg | Rg5 |
| [128] | |
| Colitis | In vivo (Obesity-induced C57BL/6 mouse colitis model.) | 30, 60 mg/kg | Rk3 |
| [137] |
| In vitro (LPS-induced LS174T cell line.) In vivo (DSS-induced C57BL/6 mouse colitis model.) | 6.25, 12.5, and 25 μM 5, 10, and 20 mg/kg | Rc |
| [139] | |
| In vivo (Acute-radiation-induced Wistar rat proctitis model.) | 20, 40, and 80 mg/kg | Rg3 |
| [140] | |
| Bone disorder | In vivo (DEX-induced Sprague Dawley rat osteoporosis model.) | 1, 6 mg/kg | Rb1 |
| [147] |
| In vivo (Hollow trephine-induced New Zealand rabbit knee OA model.) | 30, 100 μg/kg | Rb1 |
| [148] | |
| In vitro (Porphyromonas gingivalis LPS-stimulated human periodontal ligament cells.) In vivo (P. gingivalis LPS-induced Sprague Dawley rat periodontitis model.) | 25, 50, and 100 μM 100 μM | Rb3 |
| [149] |
| Inflammation Site | Test Type | Dose | Type | Molecular Mechanism | Ref. |
|---|---|---|---|---|---|
| Breast cancer | In vitro (MDA-MB-231 breast cancer cell line.) | 25, 50 μM | Rh1 |
| [172] |
| In vitro (MDA-MB-231 and MCF-7 breast cancer cell lines.) | 2, 4, and 6 μM | 20(S)-Rh2 |
| [176] | |
| Lung cancer | In vitro (A549 and PC9 lung adenocarcinoma cell lines.) In vivo (A549 xenograft nude mouse model.) | 75, 100, and 125 μM 10, 20 mg/kg | Rk1 |
| [174] |
| In vitro (TGF-β1-treated A549 lung adenocarcinoma cell line.) | Rk1 (45, 50 μM) Rg5 (200, 250 μM) | Rk1, Rg5 |
| [173] | |
| Osteosarcoma | In vitro (U20S osteosarcoma cell line.) | 8, 80 μM | Rh2 |
| [177] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jang, W.Y.; Hwang, J.Y.; Cho, J.Y. Ginsenosides from Panax ginseng as Key Modulators of NF-κB Signaling Are Powerful Anti-Inflammatory and Anticancer Agents. Int. J. Mol. Sci. 2023, 24, 6119. https://doi.org/10.3390/ijms24076119
Jang WY, Hwang JY, Cho JY. Ginsenosides from Panax ginseng as Key Modulators of NF-κB Signaling Are Powerful Anti-Inflammatory and Anticancer Agents. International Journal of Molecular Sciences. 2023; 24(7):6119. https://doi.org/10.3390/ijms24076119
Chicago/Turabian StyleJang, Won Young, Ji Yeon Hwang, and Jae Youl Cho. 2023. "Ginsenosides from Panax ginseng as Key Modulators of NF-κB Signaling Are Powerful Anti-Inflammatory and Anticancer Agents" International Journal of Molecular Sciences 24, no. 7: 6119. https://doi.org/10.3390/ijms24076119
APA StyleJang, W. Y., Hwang, J. Y., & Cho, J. Y. (2023). Ginsenosides from Panax ginseng as Key Modulators of NF-κB Signaling Are Powerful Anti-Inflammatory and Anticancer Agents. International Journal of Molecular Sciences, 24(7), 6119. https://doi.org/10.3390/ijms24076119







