Rapid and Nondestructive Evaluation of Wheat Chlorophyll under Drought Stress Using Hyperspectral Imaging
Abstract
1. Introduction
2. Results
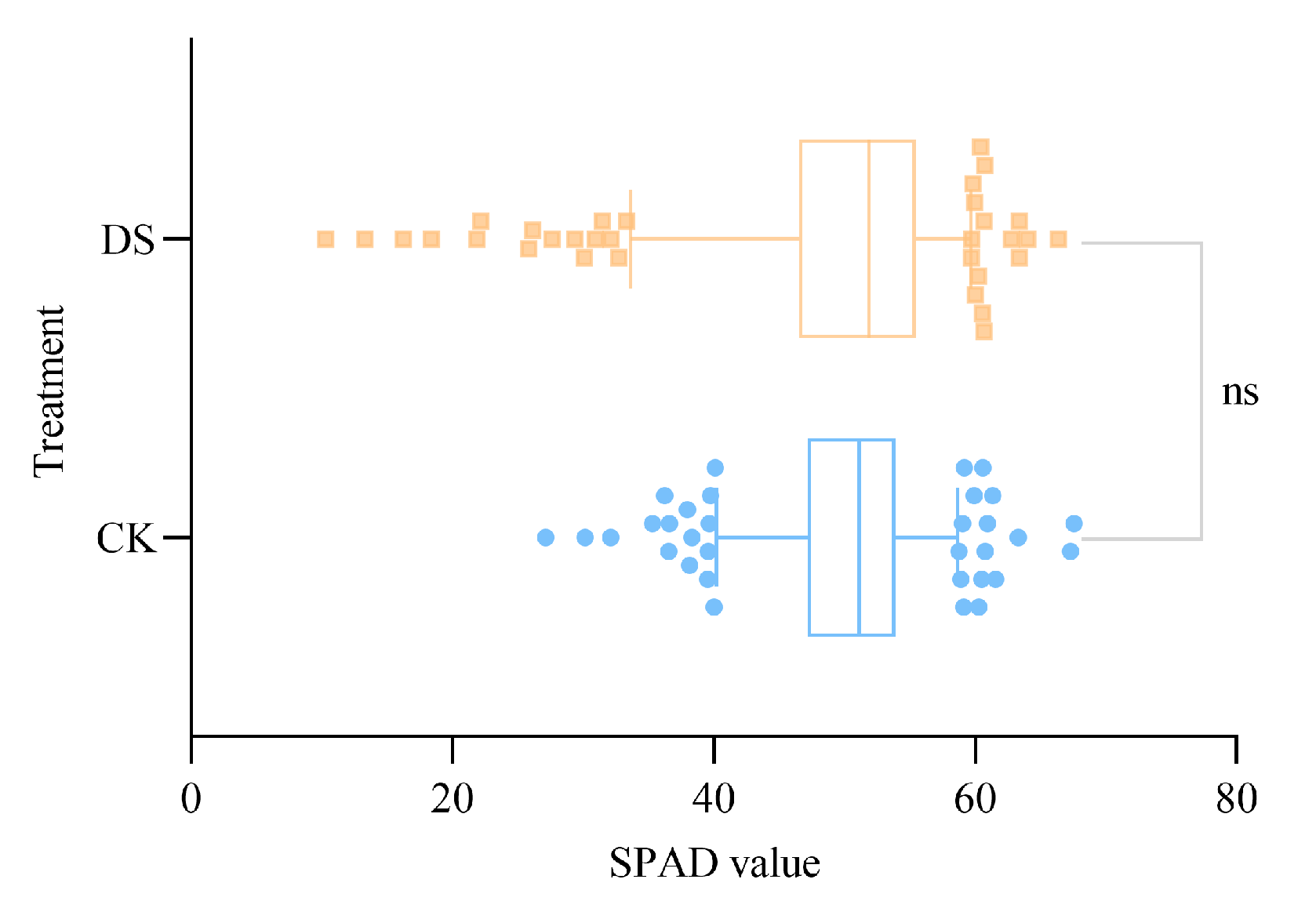
2.1. Chlorophyll Changes in Wheat Leaves under Different Soil Moisture Conditions
2.2. Hyperspectral Characteristics of Wheat Leaves under Different Soil Moisture Conditions
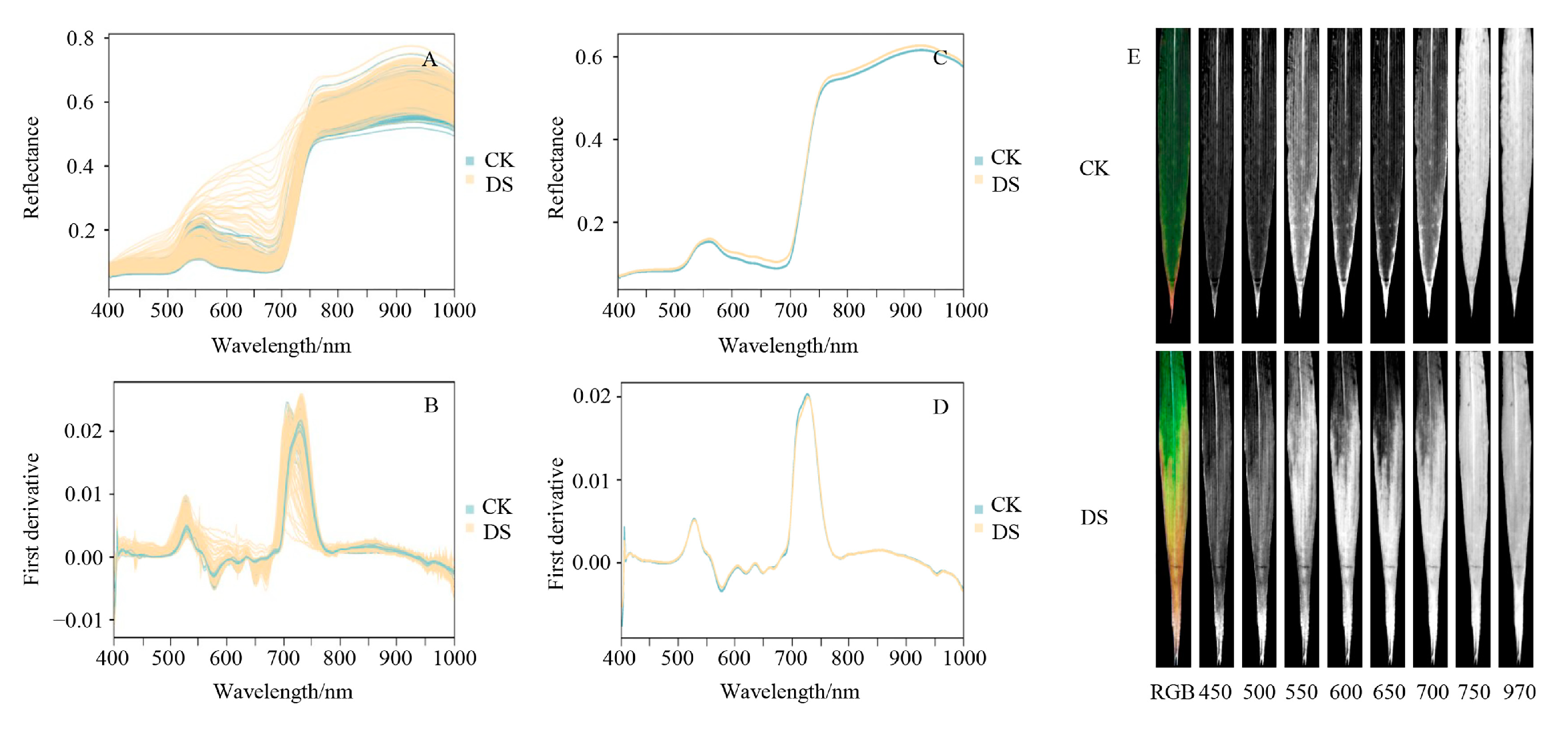
2.2.1. Spectral Reflectance Characteristics under Different Soil Moisture Conditions
2.2.2. Spectral First Derivative Values under Different Soil Moisture Conditions
2.2.3. Spectral Images under Different Soil Moisture Conditions
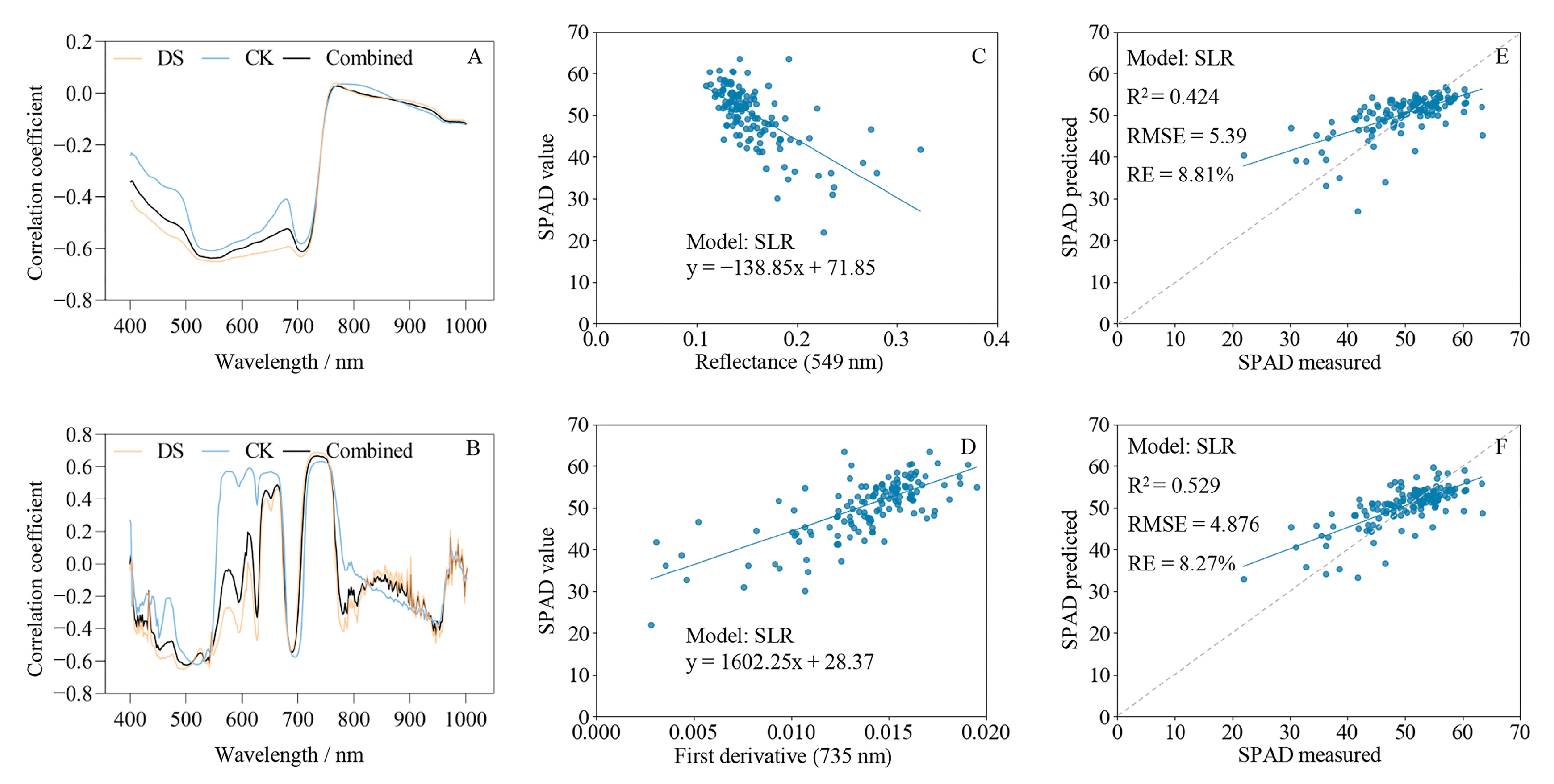
2.3. Correlation Analysis between Hyperspectral Characteristics and SPAD Values
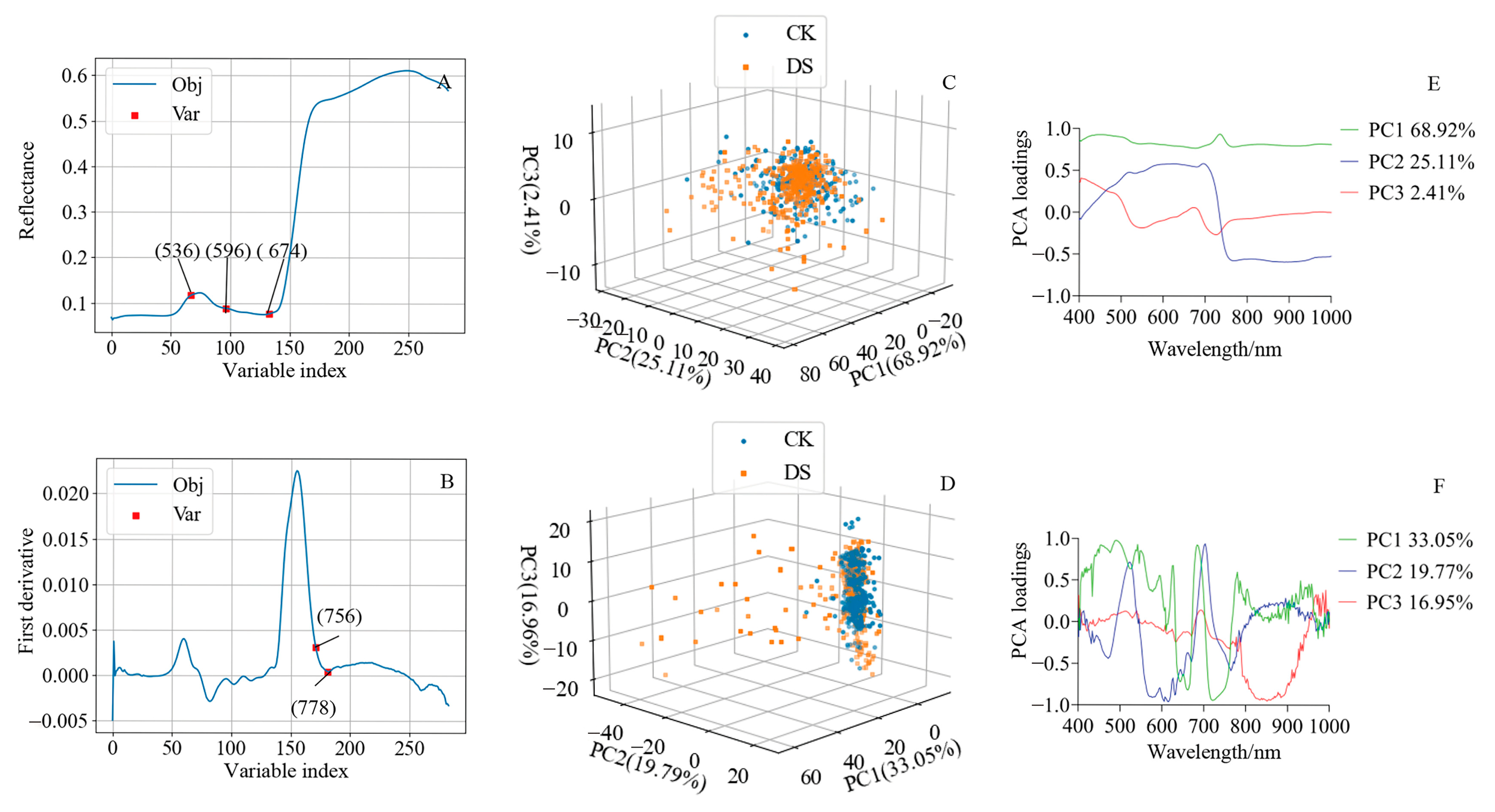
2.4. The Characteristic Bands Identified with the Successive Projections Algorithm for Estimating SPAD Values
2.5. Principal Component Analysis of Hyperspectral Information
2.6. Estimation of SPAD Values Based on Regression Analysis
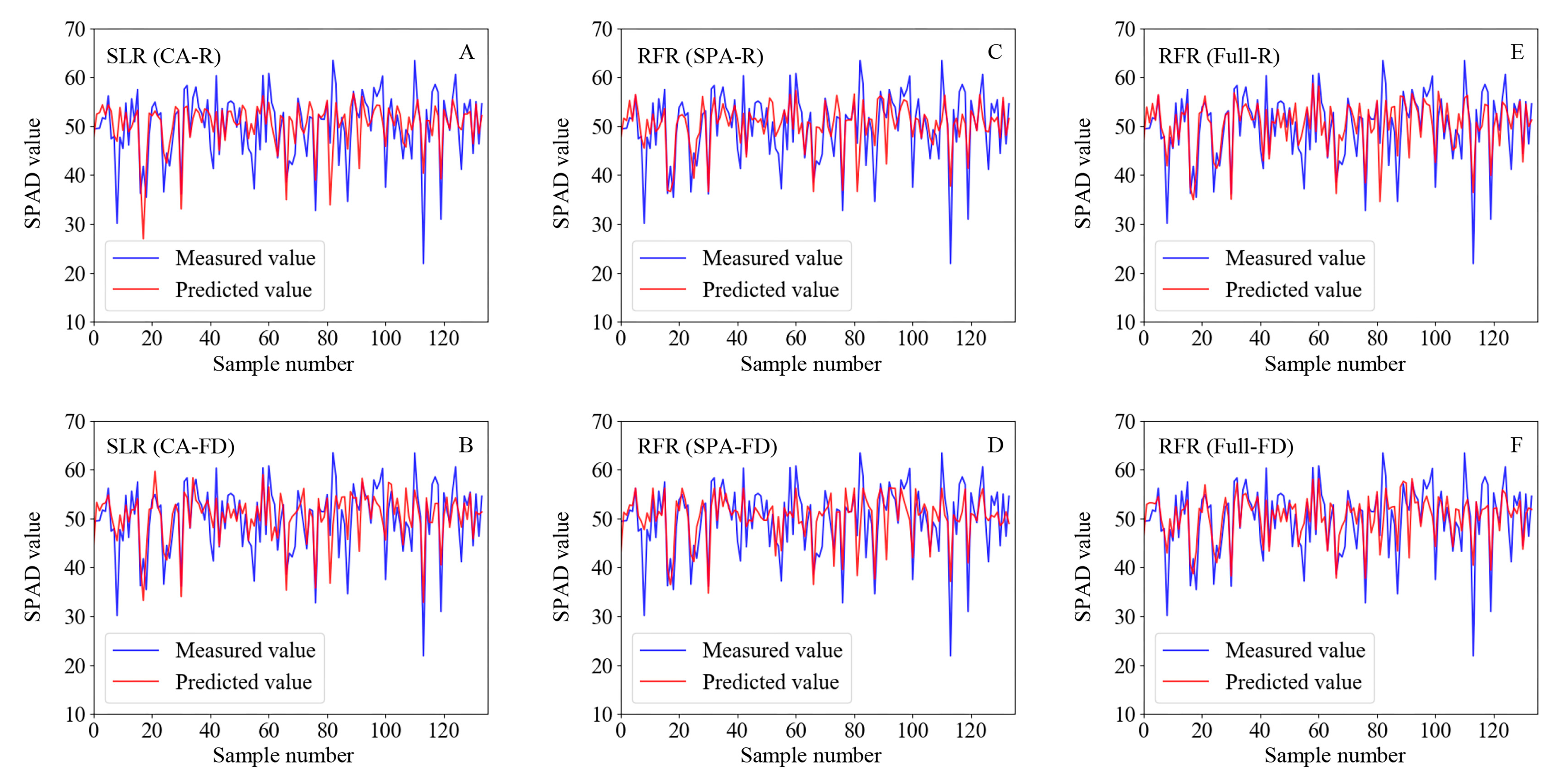
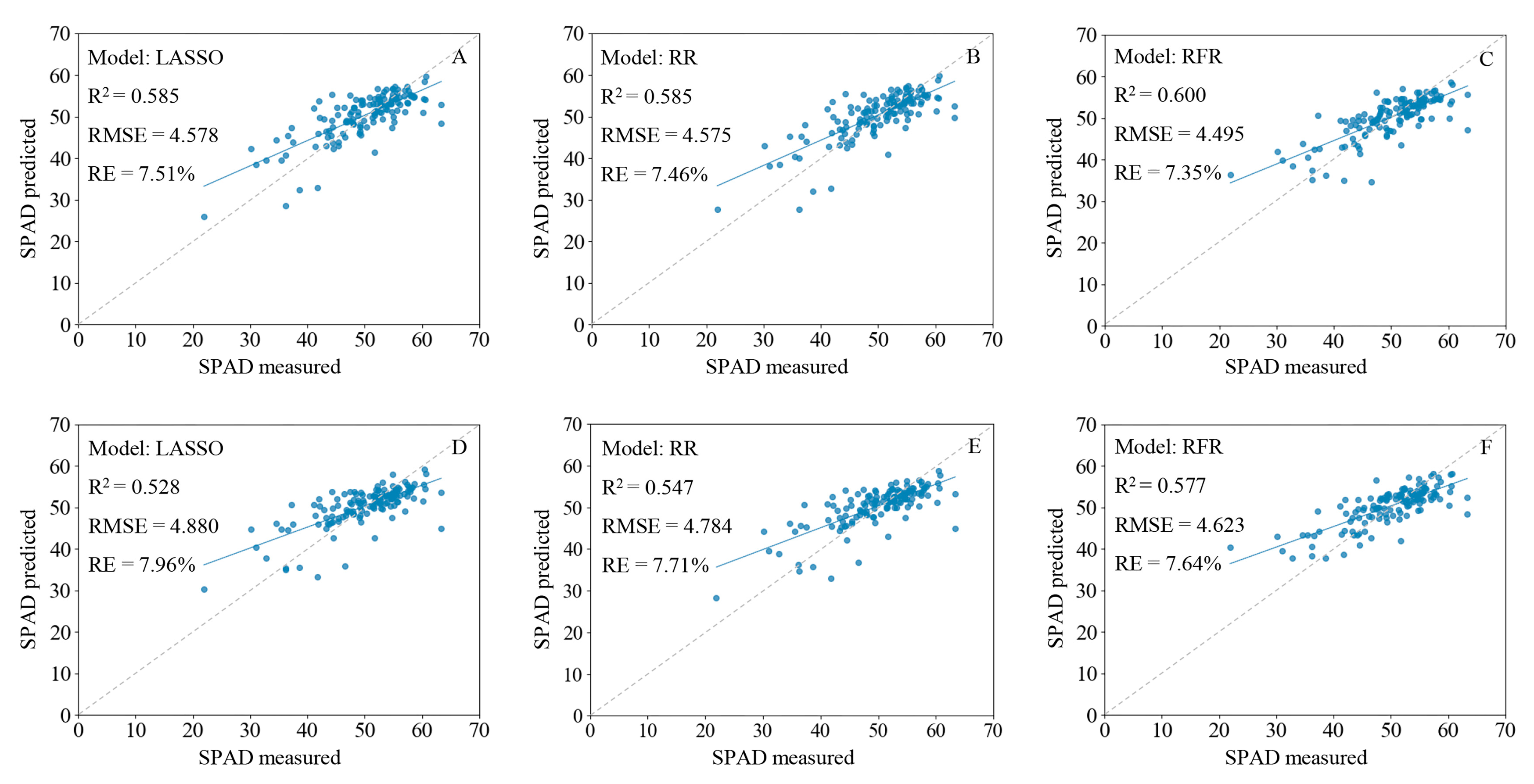
2.6.1. Estimation of SPAD Values Based on Spectral Characteristics
2.6.2. Estimation of SPAD Values Based on Spectral and Image Characteristics
3. Discussion
3.1. Feasibility of Estimating Chlorophyll Content of Wheat Leaves Using Hyperspectral Information
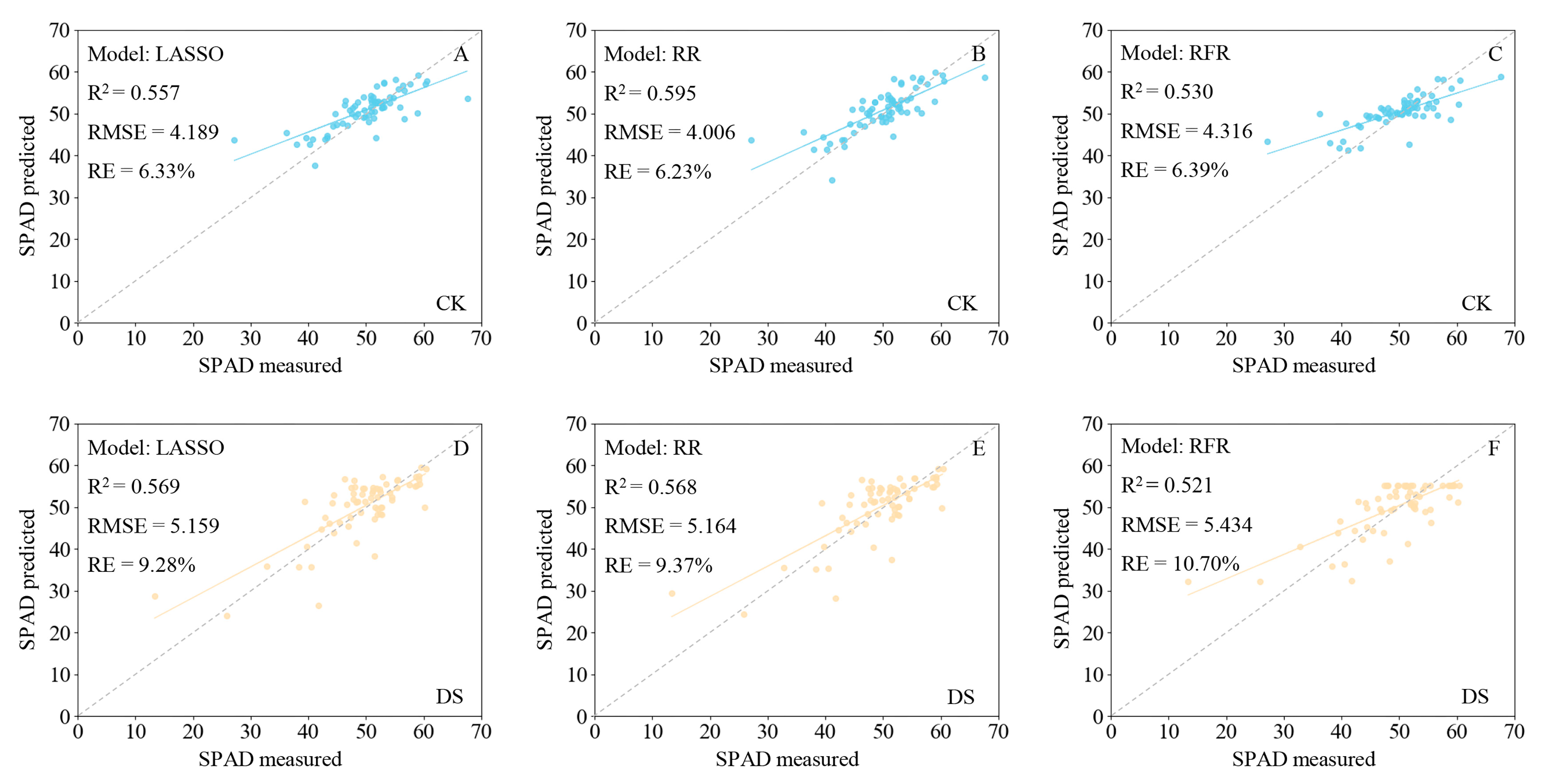
3.2. Models for Estimating SPAD Values in Wheat Leaves
3.3. Utility of Hyperspectral Reflectance for Monitoring Wheat Growth and Evaluating Drought Resistance under Drought Stress
4. Materials and Methods
4.1. Plant Material and Growth Conditions
4.2. Hyperspectral Image Acquisition
4.3. SPAD Values Measurement
4.4. Hyperspectral Image Preprocessing
4.5. Data Processing
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fernie, E.; Tan, D.K.Y.; Liu, S.Y.; Ullah, N.; Khoddami, A. Post-Anthesis Heat Influences Grain Yield, Physical and Nutritional Quality in Wheat: A Review. Agriculture 2022, 12, 886. [Google Scholar] [CrossRef]
- Rane, J.; Singh, A.K.; Kumar, M.; Boraiah, K.M.; Meena, K.K.; Pradhan, A.; Prasad, P.V.V. The Adaptation and Tolerance of Major Cereals and Legumes to Important Abiotic Stresses. Int. J. Mol. Sci. 2021, 22, 12970. [Google Scholar] [CrossRef] [PubMed]
- Daryanto, S.; Wang, L.; Jacinthe, P.-A. Global synthesis of drought effects on cereal, legume, tuber and root crops production: A review. Agric. Water Manag. 2017, 179, 18–33. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, S.; Cheng, M.; Jiang, H.; Zhang, X.; Peng, C.; Lu, X.; Zhang, M.; Jin, J. Effect of Drought on Agronomic Traits of Rice and Wheat: A Meta-Analysis. Int. J. Environ. Res. Public Health 2018, 15, 839. [Google Scholar] [CrossRef]
- Ali, H.E.; Saad, Z.H.; Elsayed, H. The effect of drought on chlorophyll, proline and chemical composition of three varieties of egyptian rice. J. Biol. Chem. 2020, 15, 21–30. [Google Scholar]
- Sharifi, P.; Mohammadkhani, N. Effects of Drought Stress on Photosynthesis Factors in Wheat Genotypes during Anthesis. Cereal Res. Commun. 2016, 44, 229–239. [Google Scholar] [CrossRef]
- Qian, X.; Liu, L.; Croft, H.; Chen, J. Relationship between leaf maximum carboxylation rate and chlorophyll content preserved across 13 species. J. Geophys. Res.-Biogeosciences 2021, 126, e2020JG006076. [Google Scholar] [CrossRef]
- Pandey, A.; Masthigowda, M.H.; Kumar, R.; Pandey, G.C.; Awaji, S.M.; Singh, G.; Singh, G.P. Physio-biochemical characterization of wheat genotypes under temperature stress. Physiol. Mol. Biol. Plants 2023, 29, 131–143. [Google Scholar] [CrossRef]
- Makino, A. Photosynthesis, Grain Yield, and Nitrogen Utilization in Rice and Wheat. Plant Physiol. 2010, 155, 125–129. [Google Scholar] [CrossRef]
- Tanaka, A.; Tanaka, R. Chlorophyll metabolism. Curr. Opin. Plant Biol. 2006, 9, 248–255. [Google Scholar] [CrossRef]
- Wang, G.; Zeng, F.; Song, P.; Sun, B.; Wang, Q.; Wang, J. Effects of reduced chlorophyll content on photosystem functions and photosynthetic electron transport rate in rice leaves. J. Plant Physiol. 2022, 272, 153669. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; He, N.; Hou, J.; Xu, L.; Liu, C.; Zhang, J.; Wang, Q.; Zhang, X.; Wu, X. Factors Influencing Leaf Chlorophyll Content in Natural Forests at the Biome Scale. Front. Ecol. Evol. 2018, 6, 64. [Google Scholar] [CrossRef]
- Hörtensteiner, S.; Kräutler, B. Chlorophyll breakdown in higher plants. Biochim. Et Biophys. Acta (BBA)-Bioenerg. 2011, 1807, 977–988. [Google Scholar] [CrossRef]
- Hashimoto, H.; Uragami, C.; Cogdell, R.J. Carotenoids and Photosynthesis. In Carotenoids in Nature: Biosynthesis, Regulation and Function; Stange, C., Ed.; Springer International Publishing: Cham, Switzerland, 2016; pp. 111–139. [Google Scholar]
- Yang, Y.-Z.; Li, T.; Teng, R.-M.; Han, M.-H.; Zhuang, J. Low temperature effects on carotenoids biosynthesis in the leaves of green and albino tea plant (Camellia sinensis (L.) O. Kuntze). Sci. Hortic. 2021, 285, 110164. [Google Scholar] [CrossRef]
- Nikolaeva, M.K.; Maevskaya, S.N.; Shugaev, A.G.; Bukhov, N.G. Effect of drought on chlorophyll content and antioxidant enzyme activities in leaves of three wheat cultivars varying in productivity. Russ. J. Plant Physiol. 2010, 57, 87–95. [Google Scholar] [CrossRef]
- Chowdhury, M.K.; Hasan, M.A.; Bahadur, M.M.; Islam, M.R.; Hakim, M.A.; Iqbal, M.A.; Javed, T.; Raza, A.; Shabbir, R.; Sorour, S.; et al. Evaluation of drought tolerance of some wheat (Triticum aestivum L.) genotypes through phenology, growth, and physiological Indices. Agron.-Basel 2021, 11, 1792. [Google Scholar] [CrossRef]
- Chen, J.L.; Zhao, X.Y.; Zhang, Y.Q.; Li, Y.Q.; Luo, Y.Q.; Ning, Z.Y.; Wang, R.X.; Wang, P.Y.; Cong, A.Q. Effects of drought and rehydration on the physiological responses of artemisia halodendron. Water 2019, 11, 793. [Google Scholar] [CrossRef]
- Xie, H.Y.; Li, M.R.; Chen, Y.J.; Zhou, Q.P.; Liu, W.H.; Liang, G.L.; Jia, Z.F. Important physiological changes due to drought stress on oat. Front. Ecol. Evol. 2021, 9, 644726. [Google Scholar] [CrossRef]
- Wasaya, A.; Manzoor, S.; Yasir, T.A.; Sarwar, N.; Mubeen, K.; Ismail, I.A.; Raza, A.; Rehman, A.; Hossain, A.; El Sabagh, A. Evaluation of fourteen bread wheat (Triticum aestivum L.) genotypes by observing gas exchange parameters, relative water and chlorophyll content, and yield attributes under drought stress. Sustainability 2021, 13, 4799. [Google Scholar] [CrossRef]
- Saha, S.; Begum, H.H.; Nasrin, S.; Samad, R. Effects of drought stress on pigment and protein contents and antioxidant enzyme activities in five varieties of rice (Oryza sativa L.). Bangladesh J. Bot. 2020, 49, 997–1002. [Google Scholar] [CrossRef]
- Hinge, P.; Kale, A.; Pawar, B.; Jadhav, A.; Chimote, V.; Gadakh, S. Effect of PEG induced water stress on chlorophyll content, membrane injury index, osmoprotectants and antioxidant enzymes activities in sorghum (Sorghum bicolor (L) Moench). Maydica 2015, 60, 1–10. [Google Scholar]
- Liu, S.; Li, S.; Fan, X.Y.; Yuan, G.D.; Hu, T.; Shi, X.M.; Huang, J.B.; Pu, X.Y.; Wu, C.S. Comparison of two noninvasive methods for measuring the pigment content in foliose macrolichens. Photosynth. Res. 2019, 141, 245–257. [Google Scholar] [CrossRef]
- Zhao, Y.; Yan, C.; Lu, S.; Wang, P.; Li, R. Estimation of chlorophyll content in intertidal mangrove leaves with different thicknesses using hyperspectral data. Ecol. Indic. 2019, 106, 105511. [Google Scholar] [CrossRef]
- Asaari, M.S.M.; Mertens, S.; Verbraeken, L.; Dhondt, S.; Inze, D.; Bikram, K.; Scheunders, P. Non-destructive analysis of plant physiological traits using hyperspectral imaging: A case study on drought stress. Comput. Electron. Agric. 2022, 195, 106806. [Google Scholar] [CrossRef]
- Xia, J.; Cao, H.; Yang, Y.; Zhang, W.; Huang, B. Detection of waterlogging stress based on hyperspectral images of oilseed rape leaves (Brassica napus L.). Comput. Electron. Agric. 2019, 159, 59–68. [Google Scholar] [CrossRef]
- Huang, Y.B.; Chen, Z.X.; Yu, T.; Huang, X.Z.; Gu, X.F. Agricultural remote sensing big data: Management and applications. J. Integr. Agric. 2018, 17, 1915–1931. [Google Scholar] [CrossRef]
- Tan, J.Y.; Ker, P.J.; Lau, K.Y.; Hannan, M.A.; Tang, S.G.H. Applications of Photonics in Agriculture Sector: A Review. Molecules 2019, 24, 2025. [Google Scholar] [CrossRef]
- Mahlein, A.K. Plant Disease Detection by Imaging Sensors—Parallels and Specific Demands for Precision Agriculture and Plant Phenotyping. Plant Dis. 2016, 100, 241–251. [Google Scholar] [CrossRef]
- Stewart, E.L.; Lucas, G.B.; Campbell, C.L.; Lucas, L.T. Introduction to plant diseases: Identification and management. Mycologia 1991, 83, 243. [Google Scholar] [CrossRef]
- Mishra, P.; Asaari, M.S.M.; Herrero-Langreo, A.; Lohumi, S.; Diezma, B.; Scheunders, P. Close range hyperspectral imaging of plants: A review. Biosyst. Eng. 2017, 164, 49–67. [Google Scholar] [CrossRef]
- Carter, G.A.; Knapp, A.K. Leaf optical properties in higher plants: Linking spectral characteristics to stress and chlorophyll concentration. Am. J. Bot. 2001, 88, 677–684. [Google Scholar] [CrossRef]
- Cui, B.; Zhao, Q.J.; Huang, W.J.; Song, X.Y.; Ye, H.C.; Zhou, X.F. Leaf chlorophyll content retrieval of wheat by simulated RapidEye, Sentinel-2 and EnMAP data. J. Integr. Agric. 2019, 18, 1230–1245. [Google Scholar] [CrossRef]
- Xie, Q.Y.; Dash, J.; Huang, W.J.; Peng, D.L.; Qin, Q.M.; Mortimer, H.; Casa, R.; Pignatti, S.; Laneve, G.; Pascucci, S.; et al. Vegetation Indices combining the Red and Red-Edge spectral information for leaf area index retrieval. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 2018, 11, 1482–1493. [Google Scholar] [CrossRef]
- Zhao, D.; Raja Reddy, K.; Kakani, V.G.; Read, J.J.; Carter, G.A. Corn (Zea mays L.) growth, leaf pigment concentration, photosynthesis and leaf hyperspectral reflectance properties as affected by nitrogen supply. Plant Soil 2003, 257, 205–218. [Google Scholar] [CrossRef]
- Carter, G.A.; Spiering, B.A. Optical properties of intact leaves for estimating chlorophyll concentration. J. Environ. Qual. 2002, 31, 1424–1432. [Google Scholar] [CrossRef]
- Watt, M.S.; Leonardo, E.M.C.; Estarija, H.J.C.; Massam, P.; de Silva, D.; O’Neill, R.; Lane, D.; McDougal, R.; Buddenbaum, H.; Zarco-Tejada, P.J. Long-term effects of water stress on hyperspectral remote sensing indicators in young radiata pine. For. Ecol. Manage. 2021, 502, 119707. [Google Scholar] [CrossRef]
- Xie, Y.K.; Feng, M.C.; Wang, C.; Yang, W.D.; Sun, H.; Yang, C.B.; Jing, B.H.; Qiao, X.X.; Kubar, M.S.; Song, J.Y. Hyperspectral monitor on chlorophyll density in winter wheat under water stress. Agron. J. 2020, 112, 3667–3676. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; Hornero, A.; Beck, P.; Kattenborn, T.; Kempeneers, P.; Hernández-Clemente, R. Chlorophyll content estimation in an open-canopy conifer forest with Sentinel-2A and hyperspectral imagery in the context of forest decline. Remote Sens. Environ. 2019, 223, 320–335. [Google Scholar] [CrossRef] [PubMed]
- Sonobe, R.; Sano, T.; Horie, H. Using spectral reflectance to estimate leaf chlorophyll content of tea with shading treatments. Biosyst. Eng. 2018, 175, 168–182. [Google Scholar] [CrossRef]
- Zhang, H.C.; Ge, Y.F.; Xie, X.Y.; Atefi, A.; Wijewardane, N.K.; Thapa, S. High throughput analysis of leaf chlorophyll content in sorghum using RGB, hyperspectral, and fluorescence imaging and sensor fusion. Plant Methods 2022, 18, 1–17. [Google Scholar] [CrossRef]
- Peng, Y.; Fan, M.; Wang, Q.H.; Lan, W.J.; Long, Y.T. Best hyperspectral indices for assessing leaf chlorophyll content in a degraded temperate vegetation. Ecol. Evol. 2018, 8, 7068–7078. [Google Scholar] [CrossRef] [PubMed]
- Qi, H.X.; Zhu, B.Y.; Kong, L.X.; Yang, W.G.; Zou, J.; Lan, Y.B.; Zhang, L. Hyperspectral inversion model of chlorophyll content in peanut leaves. Appl. Sci. 2020, 10, 2259. [Google Scholar] [CrossRef]
- Zhang, Y.; Hui, J.; Qin, Q.; Sun, Y.; Zhang, T.; Sun, H.; Li, M. Transfer-learning-based approach for leaf chlorophyll content estimation of winter wheat from hyperspectral data. Remote Sens. Environ. 2021, 267, 112724. [Google Scholar] [CrossRef]
- Colombo, R.; Meroni, M.; Marchesi, A.; Busetto, L.; Rossini, M.; Giardino, C.; Panigada, C. Estimation of leaf and canopy water content in poplar plantations by means of hyperspectral indices and inverse modeling. Remote Sens. Environ. 2008, 112, 1820–1834. [Google Scholar] [CrossRef]
- Eitel, J.U.H.; Gessler, P.E.; Smith, A.M.S.; Robberecht, R. Suitability of existing and novel spectral indices to remotely detect water stress in Populus spp. For. Ecol. Manag. 2006, 229, 170–182. [Google Scholar] [CrossRef]
- Sytar, O.; Brestic, M.; Zivcak, M.; Olsovska, K.; He, X. Applying hyperspectral imaging to explore natural plant diversity towards improving salt stress tolerance. Sci. Total Environ. 2017, 578, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Wahabzada, M.; Mahlein, A.K.; Bauckhage, C.; Steiner, U.; Oerke, E.C.; Kersting, K. Plant phenotyping using probabilistic topic models: Uncovering the hyperspectral language of plants. Sci. Rep. 2016, 6, 22482. [Google Scholar] [CrossRef]
- Liu, B.; Yue, Y.M.; Li, R.; Shen, W.J.; Wang, K.L. Plant leaf chlorophyll content retrieval based on a field imaging spectroscopy system. Sensors 2014, 14, 19910–19925. [Google Scholar] [CrossRef]
- George, R.; Padalia, H.; Sinha, S.K.; Kumar, A.S. Evaluating sensitivity of hyperspectral indices for estimating mangrove chlorophyll in Middle Andaman Island, India. Environ. Monit. Assess. 2020, 191, 785. [Google Scholar] [CrossRef]
- Niu, L.; Gao, C.; Sun, J.; Liu, Y.; Zhang, X.; Wang, F. Study on hyperspectral estimation model of chlorophyll content in grape leaves. Agric. Blotechnol. 2018, 7, 5. [Google Scholar]
- Gang, L.; Wan, S.; Jian, Z.; Yang, Z.; Qin, P. Leaf chlorophyll fluorescence, hyperspectral reflectance, pigments content, malondialdehyde and proline accumulation responses of castor bean (Ricinus communis L.) seedlings to salt stress levels. Ind. Crops Prod. 2010, 31, 13–19. [Google Scholar]
- Sonobe, R.; Hirono, Y.; Oi, A. Non-destructive detection of tea leaf chlorophyll content using hyperspectral reflectance and machine learning algorithms. Plants 2020, 9, 368. [Google Scholar] [CrossRef]
- Niu, L.; Zhang, X.; Sun, J.; Zheng, J.; Wang, F. Research on estimation models of chlorophyll content in apple leaves based on imaging hyperspectral data. Agric. Biotechnol. 2018, 7, 220–223+236. [Google Scholar]
- Xie, Y.K.; Wang, C.; Yang, W.D.; Feng, M.C.; Qiao, X.X.; Song, J.Y. Canopy hyperspectral characteristics and yield estimation of winter wheat (Triticum aestivum) under low temperature injury. Sci. Rep. 2020, 10, 244. [Google Scholar] [CrossRef]
- Li, C.C.; Cui, Y.Q.; Ma, C.Y.; Niu, Q.L.; Li, J.B. Hyperspectral inversion of maize biomass coupled with plant height data. Crop Sci. 2021, 61, 2067–2079. [Google Scholar] [CrossRef]
- Fan, J.H.; Zhou, J.; Wang, B.W.; de Leon, N.; Kaeppler, S.M.; Lima, D.C.; Zhang, Z. Estimation of maize yield and flowering time using multi-temporal UAV-based hyperspectral data. Remote Sens. 2022, 14, 3052. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, Y.H.; Xiao, H.; Gu, X.Z.; Pan, L.Q.; Tu, K. Hyperspectral imaging detection of decayed honey peaches based on their chlorophyll content. Food Chem. 2017, 235, 194–202. [Google Scholar] [CrossRef]
- Wang, H.F.; Huo, Z.G.; Zhou, G.S.; Liao, Q.H.; Feng, H.K.; Wu, L. Estimating leaf SPAD values of freeze-damaged winter wheat using continuous wavelet analysis. Plant Physiol. Biochem. 2016, 98, 39–45. [Google Scholar] [CrossRef]
- Yuan, Z.R.; Ye, Y.; Wei, L.F.; Yang, X.; Huang, C. Study on the optimization of hyperspectral characteristic bands combined with monitoring and visualization of pepper leaf SPAD value. Sensors 2022, 22, 183. [Google Scholar] [CrossRef]
- Jang, S.H.; Hwang, Y.K.; Lee, H.J.; Lee, J.S.; Kim, Y.H. Selecting significant wavelengths to predict chlorophyll content of grafted cucumber seedlings using hyperspectral images. Korean J. Remote Sens. 2018, 34, 681–692. [Google Scholar]
- Rehman, S.U.; Bilal, M.; Rana, R.M.; Tahir, M.N.; Shah, M.K.N.; Ayalew, H.; Yan, G.J. Cell membrane stability and chlorophyll content variation in wheat (Triticum aestivum) genotypes under conditions of heat and drought. Crop Pasture Sci. 2016, 67, 712–718. [Google Scholar] [CrossRef]
- Rolando, J.L.; Ramirez, D.A.; Yactayo, W.; Monneveux, P.; Quiroz, R. Leaf greenness as a drought tolerance related trait in potato (Solanum tuberosum L.). Environ. Exp. Bot. 2015, 110, 27–35. [Google Scholar] [CrossRef]
- Hou, D.; Bi, J.; Ma, L.; Zhang, K.; Li, D.; Rehmani, M.I.A.; Tan, J.; Bi, Q.; Wei, Y.; Liu, G.; et al. Effects of Soil Moisture Content on Germination and Physiological Characteristics of Rice Seeds with Different Specific Gravity. Agronomy 2022, 12, 500. [Google Scholar] [CrossRef]
- Chen, X.Y.; Zhu, Y.; Ding, Y.; Pan, R.M.; Shen, W.Y.; Yu, X.R.; Xiong, F. The relationship between characteristics of root morphology and grain filling in wheat under drought stress. PeerJ 2021, 9, e12015. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.; Hussain, M.; Siddique, K.H.M. Drought Stress in Wheat during Flowering and Grain-filling Periods. Crit. Rev. Plant Sci. 2014, 33, 331–349. [Google Scholar] [CrossRef]
- Du, T.; Meng, P.; Huang, J.; Peng, S.; Xiong, D. Fast photosynthesis measurements for phenotyping photosynthetic capacity of rice. Plant Methods 2020, 16, 6. [Google Scholar] [CrossRef]
- Liu, Z.A.; Yang, J.P.; Yang, Z.C. Using a chlorophyll meter to estimate tea leaf chlorophyll and nitrogen contents. J. Soil Sci. Plant Nutr. 2012, 12, 339–348. [Google Scholar] [CrossRef]
- Wakiyama, Y. The Relationship between SPAD Values and Leaf Blade Chlorophyll Content throughout the Rice Development Cycle. Jarq-Jpn. Agric. Res. Q. 2016, 50, 329–334. [Google Scholar] [CrossRef]
- Shibaeva, T.G.; Mamaev, A.V.; Sherudilo, E.G. Evaluation of a SPAD-502 Plus Chlorophyll Meter to Estimate Chlorophyll Content in Leaves with Interveinal Chlorosis. Russ. J. Plant Physiol. 2020, 67, 690–696. [Google Scholar] [CrossRef]
- Eberly, L.E. Correlation and simple linear regression. Methods Mol. Biol. (Clifton N.J.) 2007, 404, 143–164. [Google Scholar]
- Tibshirani, R. Regression Shrinkage and Selection Via the Lasso. J. R. Stat. Soc. Ser. B (Methodol.) 1996, 58, 267–288. [Google Scholar] [CrossRef]
- Hoerl, A.E.; Kennard, R.W. Ridge regression: Biased estimation for nonorthogonal problems. Technometrics 2000, 42, 80–86. [Google Scholar] [CrossRef]
- Breiman, L. Random Forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]

| Data Set | Model | Training Set | (n = 536) | Testing Set | (n = 134) | ||
|---|---|---|---|---|---|---|---|
| R2 | RMSE | RE | R2 | RMSE | RE | ||
| CA-R (549 nm) | SLR | 0.401 | 5.549 | 9.17% | 0.424 | 5.390 | 8.81% |
| CA-FD (735 nm) | SLR | 0.426 | 5.434 | 9.01% | 0.529 | 4.876 | 8.27% |
| SPA-R | LASSO | 0.405 | 5.532 | 9.12% | 0.417 | 5.430 | 8.86% |
| RR | 0.405 | 5.532 | 9.12% | 0.417 | 5.426 | 8.85% | |
| RFR | 0.478 | 5.183 | 8.60% | 0.478 | 5.134 | 8.40% | |
| SPA-FD | LASSO | 0.407 | 5.523 | 9.25% | 0.518 | 4.934 | 8.49% |
| RR | 0.407 | 5.523 | 9.26% | 0.517 | 4.939 | 8.51% | |
| RFR | 0.510 | 5.023 | 8.57% | 0.510 | 4.974 | 8.47% | |
| PCA-R | LASSO | 0.484 | 5.150 | 8.39% | 0.571 | 4.655 | 7.64% |
| RR | 0.488 | 5.130 | 8.20% | 0.580 | 4.607 | 7.47% | |
| RFR | 0.555 | 4.788 | 7.87% | 0.555 | 4.739 | 7.89% | |
| PCA-FD | LASSO | 0.454 | 5.298 | 8.62% | 0.496 | 5.045 | 7.90% |
| RR | 0.454 | 5.298 | 8.64% | 0.497 | 5.039 | 7.99% | |
| RFR | 0.560 | 4.755 | 7.74% | 0.560 | 4.714 | 7.62% | |
| Full-R | LASSO | 0.587 | 4.609 | 7.43% | 0.585 | 4.578 | 7.51% |
| RR | 0.586 | 4.617 | 7.45% | 0.585 | 4.575 | 7.46% | |
| RFR | 0.600 | 4.535 | 7.40% | 0.600 | 4.495 | 7.35% | |
| Full-FD | LASSO | 0.528 | 4.929 | 8.12% | 0.528 | 4.880 | 7.96% |
| RR | 0.548 | 4.824 | 7.81% | 0.547 | 4.784 | 7.71% | |
| RFR | 0.579 | 4.653 | 7.63% | 0.577 | 4.623 | 7.64% | |
| Variable | SPAD | L* | a* | b* |
|---|---|---|---|---|
| SPAD | 1 | |||
| L* | −0.591 ** | |||
| a* | −0.164 ** | 0.438 ** | ||
| b* | −0.600 ** | 0.912 ** | 0.378 ** | 1 |
| Data Set | Training Set (n = 536) | Testing Set (n = 134) | ||||
|---|---|---|---|---|---|---|
| R2 | RMSE | RE | R2 | RMSE | RE | |
| (CA-R) + L*a*b* | 0.486 | 5.14 | 8.52% | 0.486 | 5.095 | 8.23% |
| (CA-FD) + L*a*b* | 0.519 | 4.973 | 8.19% | 0.519 | 4.925 | 8.18% |
| (SPA-R) + L*a*b* | 0.502 | 5.061 | 8.38% | 0.501 | 5.021 | 8.27% |
| (SPA-FD) + L*a*b* | 0.51 | 5.019 | 8.37% | 0.51 | 4.972 | 8.13% |
| (PCA-R) + L*a*b* | 0.46 | 5.271 | 8.76% | 0.46 | 5.218 | 8.29% |
| (PCA-FD) + L*a*b* | 0.584 | 4.626 | 7.47% | 0.584 | 4.579 | 7.48% |
| (Full-R) + L*a*b* | 0.61 | 4.478 | 7.30% | 0.61 | 4.439 | 7.35% |
| (Full-FD) + L*a*b* | 0.578 | 4.661 | 7.64% | 0.578 | 4.617 | 7.58% |
| L*a*b* | 0.435 | 5.39 | 9.06% | 0.434 | 5.346 | 8.38% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Y.; Nan, R.; Mi, T.; Song, Y.; Shi, F.; Liu, X.; Wang, Y.; Sun, F.; Xi, Y.; Zhang, C. Rapid and Nondestructive Evaluation of Wheat Chlorophyll under Drought Stress Using Hyperspectral Imaging. Int. J. Mol. Sci. 2023, 24, 5825. https://doi.org/10.3390/ijms24065825
Yang Y, Nan R, Mi T, Song Y, Shi F, Liu X, Wang Y, Sun F, Xi Y, Zhang C. Rapid and Nondestructive Evaluation of Wheat Chlorophyll under Drought Stress Using Hyperspectral Imaging. International Journal of Molecular Sciences. 2023; 24(6):5825. https://doi.org/10.3390/ijms24065825
Chicago/Turabian StyleYang, Yucun, Rui Nan, Tongxi Mi, Yingxin Song, Fanghui Shi, Xinran Liu, Yunqi Wang, Fengli Sun, Yajun Xi, and Chao Zhang. 2023. "Rapid and Nondestructive Evaluation of Wheat Chlorophyll under Drought Stress Using Hyperspectral Imaging" International Journal of Molecular Sciences 24, no. 6: 5825. https://doi.org/10.3390/ijms24065825
APA StyleYang, Y., Nan, R., Mi, T., Song, Y., Shi, F., Liu, X., Wang, Y., Sun, F., Xi, Y., & Zhang, C. (2023). Rapid and Nondestructive Evaluation of Wheat Chlorophyll under Drought Stress Using Hyperspectral Imaging. International Journal of Molecular Sciences, 24(6), 5825. https://doi.org/10.3390/ijms24065825






