1. Introduction
The human placenta is a transitory organ at the interface between the maternal and fetal circulation. This organ ensures essential functions for the maintenance of pregnancy and for fetal development [
1]. The placenta provides nutrient and gaseous transport, elimination of waste products, and intensive endocrine function by producing several hormones (human chorionic gonadotropin (hCG) and steroid hormones) essential for placental development and metabolism. The placenta is also a selective barrier that assures protection of the fetus against the many xenobiotics to which the mother is exposed. However, many substances can accumulate in and/or cross the placenta via different mechanisms and consequently reach the fetus. The placenta is composed of chorionic villi, which are the structural and functional units of the human placenta. These villi consist of a mesenchymal core in which the fetal vessels are embedded and which is bordered by an epithelium. This epithelium is formed by the outer syncytiotrophoblast (ST) layer, a multinucleated syncytium, and the underlying mononuclear villous cytotrophoblasts (VCT), which constantly differentiate and fuse to renew the syncytium. VCT can be purified from human placenta obtained following a term delivery. When cultivated in vitro, these cells retain the ability to aggregate and fuse spontaneously after 24–48 h to form the ST and are, therefore, a better model of physiological conditions than typical trophoblast cell lines, such as the BeWo choriocarcinoma trophoblasts. From 10 gestational weeks, the placental chorionic villi bathe freely in maternal blood in the intervillous space. Therefore, any foreign molecules with a functional activity, such as pollutants that may accumulate within the placenta, can directly impact placental functioning, with repercussions on pregnancy outcome [
2].
Environmental pollutants such as polycyclic aromatic hydrocarbon (PAH) and nanoparticles (particles whose three dimensions are under 100 nm) are widespread contaminants. One of the most represented PAH is benzo(a)pyrene (B
aP), a five-ring benzene that has been classified by the International Agency for Research on Cancer in group I (a carcinogenic, mutagenic, and reprotoxic agent for humans) and which is also an endocrine disruptor [
3]. B
aP is formed by incomplete combustion of organic compounds and is found in cigarette smoke, industrial waste, charbroiled food (e.g., meat, fish, vegetables, bread, etc.), and in car exhaust fumes (diesel). One of the main cellular targets of B
aP is the ligand-activated transcription factor AhR: the aryl hydrocarbon receptor, which is involved in xenobiotic detoxification by inducing phase I and phase II detoxifying enzymes [
4]. The induction of these enzymes allows the formation of B
aP metabolites, the majority of which are eliminated from the body through phase III membrane transporters. However, in the case of PAH and specifically B
aP, such processes may also contribute to B
aP mutagenesis and toxicity. Indeed, the AhR-dependent metabolization of B
aP may lead to reactive electrophilic metabolites (such as benzo-(a)-pyrene-diol epoxides) that form adducts with DNA. Moreover, phase I enzymes may also generate reactive oxygen species (ROS) through decoupling enzymatic events. In human placenta, AhR is involved in biological responses to B
aP (detoxification but also bioactivation). We previously showed that B
aP induces phase I genes (CYP1A1, CYP1B1, and CYP1A2) in human primary trophoblasts [
5]. Several cohort studies found PAH (including B
aP) in maternal blood [
6], placenta, cord, and milk [
7] from both smoking and nonsmoking women. In women that smoked during pregnancy, B
aP was detected in both placenta and in umbilical cord blood, together with B
aP–DNA adducts [
8,
9]. During pregnancy, exposure to B
aP is correlated with an increased risk of early miscarriage [
10] and complications of pregnancy such as prematurity [
11] and lower birth weight [
12].
Cerium (Ce) is a metal of the lanthanide group, which oxidizes rapidly on contact with air. It has two ionizing oxidation states: Ce
3+ and Ce
4+. Cerium dioxide nanoparticles (CeO
2 NP) gained the attention of the scientific community because they have been used in numerous applications since the 2000s [
13], for example, as additives in diesel fuels (EnvirOx) and in cigarettes, and as polishing agents (glass, ceramic). These nanoparticles are also studied as potential medicines (e.g., antitumor drugs) [
14,
15] for their pro- and antioxidant properties, which depend on multiple factors, such as the Ce
3+/Ce
4+ ratio, dose, and exposure time. When Ce is added to diesel fuels, particles released into the environment by cars [
16,
17] have a 6.5% increased Ce content [
14,
18]. In 2010, the Organization of Economic Cooperation and Development included CeO
2 NP in the top priority list of nanomaterials requiring urgent evaluation. There are few data concerning the impact of CeO
2 NP, to which pregnant women may be involuntarily exposed. Recent publications reported the detection of Ce in maternal blood, urine, and breast milk, but these data remain scarce, with significant differences depending on the place of residence of the pregnant women (from 21 to 70 ng/L in serum samples from Spanish women in 2009 and from 10 to 5180 ng/L in serum samples from pregnant women in Shanxi Province, China, between 2010 and 2018) [
19,
20]. An increased concentration of cerium in maternal blood and urine has been associated with a risk of fetal neural tube defects and a reduced level of thyroid-stimulating hormone in newborns [
19,
21]. CeO
2 NP also appear to be responsible for placentation abnormalities [
22], in particular by the excessive activation of autophagy and a decrease in invasive and migratory abilities in first-trimester HTR-8/SVneo trophoblast cell lines [
23,
24]. We previously showed that CeO
2 NP have adverse effects on term human primary trophoblasts, affecting their viability in a dose-dependent manner, their capacity to differentiate to form ST, and the endocrine function of the placenta, with disruption of the secretion of peptides, such as hCG and steroid hormones [
25]. However, humans are rarely exposed to CeO
2 NP alone. As a result of their high surface-to-volume ratio and high reactivity, CeO
2 NP can interact with other pollutants from common sources, including fuel-borne chemicals such as B
aP, the emission of which is increased by 35% in cerium-additive fuels [
17,
18,
26]. Hence, CeO
2 NP and B
aP are encountered in combination (from common combustion processes such as diesel exhaust, cigarette smoke, etc.) and human exposure to both contaminants in a mixture is common. Prior exposure to NP can alter the barrier and defense functions of trophoblasts (metabolism, endocrine function, antioxidant defense, signaling pathways, etc.), which could become more vulnerable to other chemical pollutants (such as B
aP). In addition, the lipophilic properties of B
aP may modify the mechanism and kinetics of NP cellular uptake. Human exposure to these two pollutants is mainly through the respiratory tract. Subsequently, lipophilic B
aP and NP, including CeO
2 NP, are able to cross biological barriers, enter the systemic circulation, and reach blood-irrigated organs, such as the placenta [
27,
28]. As compared with individual exposure, these pollutants in combination may synergistically affect the proper functioning of the placenta by activating different cellular pathways in parallel.
In this study, we investigated the molecular consequences of co-exposure to CeO
2 NP and B
aP vs. their individual exposure in terms of the major actors of cellular stress by using chorionic villi and primary cytotrophoblasts purified from human placentas at term of pregnancy. The ratio of pollutants tested here mimics the airborne levels of PAH found on atmospheric ultrafine particles (UFP) in Paris [
29] (from 0.15 to 2 mg PAH per g of UFP), which we adjusted to 1 µM of B
aP per 10 µg/cm
2 of CeO
2 NP in our study. Here, we show that B
aP activates the AhR signaling pathway (measured by CYP1A1 induction) and DNA damage, with the stabilization of p53, its target p21, and phosphorylated H2AX (ɣ-H2AX), but without the activation of the antioxidant defense system. Both AhR antagonist and p53 inhibitor diminished p21 induction, although p53 inhibition had no effect on DNA damage. Furthermore, co-exposure of trophoblasts to B
aP and CeO
2 did not modify B
aP metabolization and p53 activation, suggesting that B
aP has a dominant effect. However, no DNA damage was observed after co-exposure, potentially due to the antioxidant effect of CeO
2 NP suggested by a decrease in Prx-SO
3.
3. Discussion
In this study, we aimed to determine the major factors in cellular stress, i.e., the factors whose expression is modulated after individual exposure and co-exposure of primary human VCT to two major environmental pollutants: BaP and CeO2 NP. Trophoblastic cell lines (such as BeWo, JEG, JAR) represent one of the most used trophoblastic models in placental toxicology, but they are either modified or derived from choriocarcinoma cells with an indefinite proliferation phenotype. BeWo cells also require the addition of forskolin (an activator of PKA via cyclic AMP) in order to terminally differentiate to form the ST. For all these reasons, we used primary VCT purified from term human placenta because they are a better model of physiological conditions than trophoblast cell lines due to their capacity to spontaneously aggregate and fuse in vitro at 24–48 h of culture to form the ST. Primary VCT are nonproliferating cells in vitro.
Since the exact B
aP/CeO
2 NP ratio in ambient air or in maternal blood is not known and is difficult to measure due to large variations depending on place of residence, lifestyle (diet, smoking), etc., the ratio of pollutants tested here is close to the airborne levels of PAH found on UFP in Paris [
31] (from 0.15 to 2 ng PAH per µg of UFP). We used 0.6 µM of B
aP per 6.3 µg/cm
2 of CeO
2 NP in the Cell Stress Array and we used the same ratio but rounded up to 1 µM per 10 µg/cm
2 in the other studies. Because the molar mass of B
aP is 252.3 g/mol, the ratio of 1 µM of B
aP (252.3 µg/L) per 10 µg/cm
2 of CeO
2 NP corresponds to 6.63 ng of B
aP per µg of CeO
2 NP. We showed in our previous publication that CeO
2 NP concentrations below 80 µg/mL (12.6 µg/cm
2) did not affect the metabolic activity of VCT at the exposure time chosen for our experiments (24 h), but did disrupt both trophoblast morphological and functional differentiation [
25].
We show here that B
aP only affected the metabolic activity of primary trophoblasts at concentrations higher than 2 μM and after prolonged exposure (72 h) (
Figure 1A). The authors and others showed that exposure to B
aP leads to the activation of the AhR pathway and increased expression of CYP1A1 and CYP1B1, which are involved in the bioactivation of B
aP, including in the BeWo cell line [
5,
32]. We showed the activation of the AhR signaling pathways by B
aP via a dose-dependent (0.1 to 1 μM) increase in protein expression of CYP1A1 after 24 h of exposure (
Figure 1C), with the lowest concentration of 0.1 µM approaching those found in maternal blood. Then, we confirmed that lower concentrations of B
aP (0.1, 0.6 μM), which are more representative of the placenta, had cellular effects similar to those of 1 μM of B
aP. Our results for CYP1A1 converge towards the activation of the detoxification pathway after exposure to B
aP. This is emphasized by the use of the AhR antagonist, CH223191, which partially inhibited the induction of CYP1A1 after B
aP incubation (
Supplementary Figure S2).
In order to identify other signaling pathways modulated by exposure to B
aP and/or CeO
2 NP, we used the Cell Stress Array to observe fine changes in the levels of the main cellular stress proteins and factors. This test was carried out at nontoxic doses of pollutants and after 24 h of exposure in order to be sure that the AhR pathway had, indeed, been activated at this stage. However, the chosen dose of NP used here (6.3 µg/cm
2 representing 40 µg/mL) was able to disturb placental functions (morphological and functional differentiation), as reported in our previous publication [
25]. Both the WST-1 cell viability assay and the absence of cleavage of PARP-1, a target of caspase 3, highlight that B
aP does not induce apoptosis at the tested concentration and times (0.6 μM for 30 min, 4 h, and 24 h) (
Figure 2B).
Concerning CeO
2 NP, we found a significant decrease in the superoxidized form of peroxiredoxins (Prx-SO
3) compared to the control after 24 h of individual and co-exposure to CeO
2 NP in the five placentas (
Figure 5). Peroxiredoxins are proteins with intracellular H
2O
2 sensor thiol groups. Their superoxidized form Prx-SO
3 (excessive oxidation) indicates an abundance of hydrogen peroxide. This decrease in Prx-SO
3 after exposure to CeO
2 NP and co-exposure suggests that these nanoparticles have an antioxidant effect.
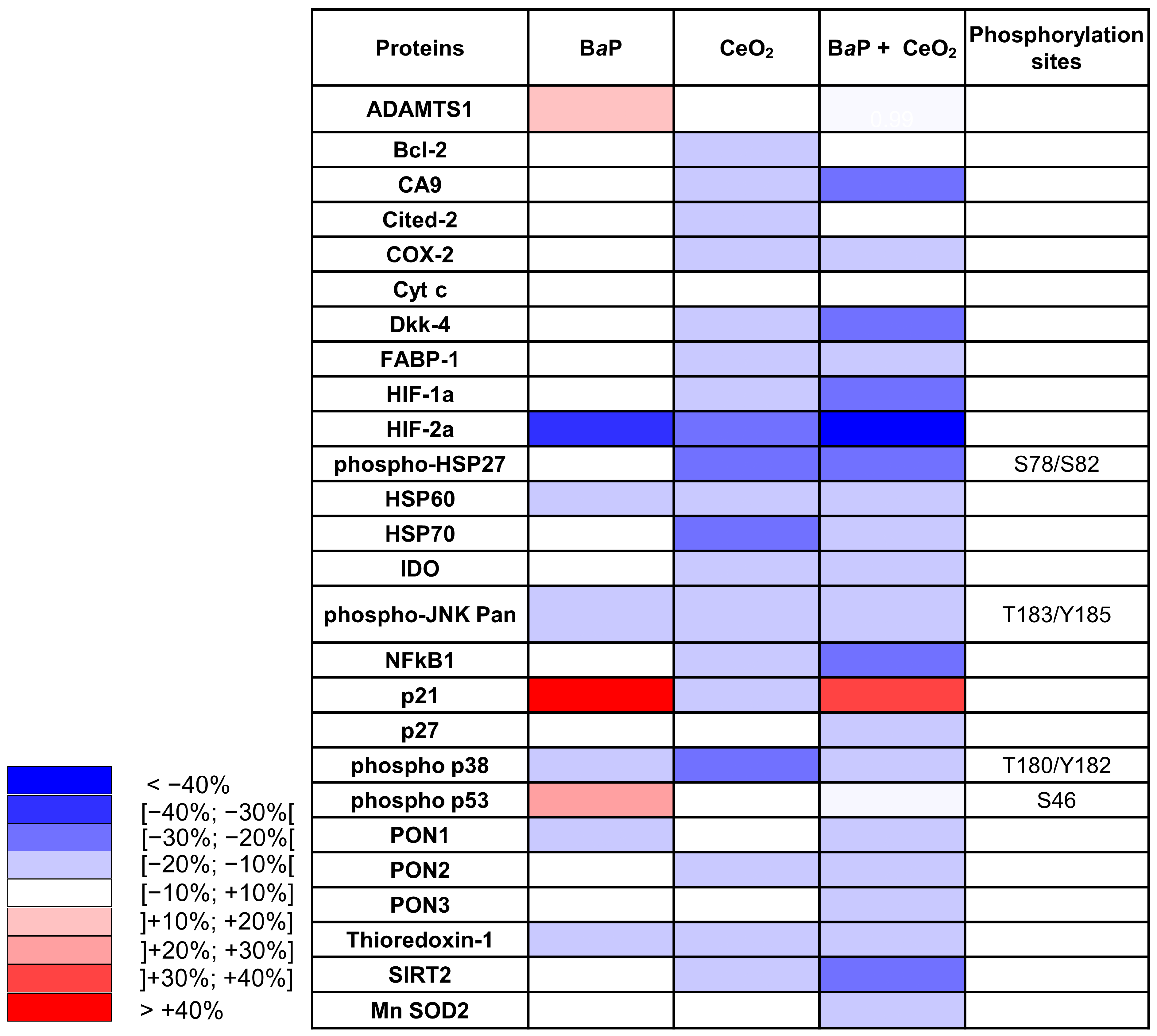
Furthermore, an analysis of the results of the Cell Stress Array also showed a decrease in Mn-SOD2 with the co-exposure to CeO
2 NP and B
aP. CeO
2 NP notably induced a reduction in the proteins involved in the redox-sensitive pathway, such as p38/HSP27 (−21% and −25%, respectively) and those involving HSP70 (−29%). The protein p38, from the MAPK family (mitogen-activated protein kinase), is involved in different signaling pathways, including the cell cycle, differentiation, cell stress, and cell death. After phosphorylation under oxidative stress, p38 activates a MAPKAP-K2 (protein kinase-2 activated by MAP kinase 2) involved in the change in conformation by phosphorylation of the heat shock protein HSP27. Phosphorylated HSP27 is involved in the remodeling of the actin network in particular. Microvilli containing actin are present on the apical membrane of ST and their remodeling processes are essential for trophoblast differentiation and function [
33]. These decreases in p38 and HSP27 protein levels can be correlated with the results obtained previously by our laboratory showing the decrease in the fusion of VCT into the ST after exposure to CeO
2 NP at the same concentration [
25]. The protein expression of HSP70 can be modulated by different mechanisms under conditions of oxidative stress. Caution is needed regarding our interpretations because the results of the Cell Stress Array were obtained at a single exposure time of 24 h. It would, therefore, be interesting to study the p38 pathway at very short exposure times (15 min, 30 min, and 1 h) to CeO
2 NP alone in order to discover the pathways involved in the variation in phosphorylation level.
NFκB in its dimer form (p50/p65) is a transcription factor activated by oxidative stress. Under cellular stress, NFκB is translocated in the nucleus and participates in the process of cytokine secretion, angiogenesis, and the inhibition of apoptosis. However, the Cell Stress Array analysis showed a decrease in NFκB1 subunit (p50) levels and an absence of significant modification of the protein level of the NFκB p65 subunit by Western blot when exposed to CeO
2 NP alone or in co-exposure with B
aP (
Figure 5). However, we know that the activity of NFκB is mainly regulated at the level of its nuclear localization. Studying the translocation of NFκB by IHC could provide more information.
In summary, CeO
2 NP have an antioxidant potential in individual exposure or in co-exposure with B
aP. Our observations of these NP are in agreement with those of our previous report [
25] and with other data from the literature [
34], describing an antioxidant effect probably by acting as a ROS scavenger, with a mimetic activity of catalase at the NP surface created by oxygen vacancies [
35].
Both the Cell Stress Array and IHC revealed that exposure to CeO
2 NP led to a decrease in the protein levels of HIF-2α, with a greater decrease in co-exposure in the Cell Stress Array, which suggests a synergistic effect. However, different results were seen for HIF-1α in the Cell Stress Array and IHC after exposure to B
aP and/or CeO
2 NP. Our attempt at validating this via Western blot was unsuccessful because we did not have quantifiable HIF bands without hypoxic chemical induction (such as cobalt chloride). Nevertheless, HIF-1α stabilization by CeO
2 NP was reported by Das et al., showing that a high Ce
3+/Ce
4+ ratio can activate angiogenesis via HIF-1α stabilization in HUVEC cells [
36]. In addition, the metalloproteinase ADAMTS1, which was increased by exposure to B
aP in the Cell Stress Array (
Figure 4), is induced by hypoxia in HUVEC cells by binding of HIF-1α to the promoter of ADAMTS1. The first stages of placental development in the first trimester take place in a low O
2 environment (1–2%). HIF are stabilized during this critical period and play essential roles in placental morphogenesis, trophoblast proliferation, and angiogenesis [
37]. These roles have been described using gene knockout mouse models. HIF-1α
−/− and HIF-2α
−/− embryos present placental vascularization abnormalities [
38]. However, in the term placenta, the oxygen environment is approximately 8% and HIF are degraded by the proteasome. High levels of HIF-1α in term placentas are associated with placental pathologies such as preeclampsia and intrauterine growth retardation [
39]. A large number of studies show that HIF-1α and HIF-2α do not have the same expression profile during hypoxia. HIF-1α is activated early but for a limited time while HIF-2α is activated later but for longer. Recent transcriptome studies have shown different target gene panels depending on whether HIF-1α or HIF-2α is activated by hypoxia [
40]. HIF targets include VEGF, sFlt-1, and angiotensin 2, which have roles in angiogenesis [
40]. It has been demonstrated that HIF-1α/ARNT dimers compete for the ARNT bond [
41]. However, we noted a temporary increase in AhR after exposure to B
aP for 30 min. AhR could, therefore, compete with HIF-1α. In view of the modifications observed in the levels of HIF in term VCT, additional studies should be carried out in this particular pathway in order to determine whether chronic exposure to B
aP and/or CeO
2 NP in the first trimester of pregnancy induces angiogenesis abnormalities by changes in HIF levels.
Regarding the pro-oxidant effect of B
aP, exposure to 0.6 μM of B
aP for 24 h did not induce an increase in the protein level of Prx-SO
3 according to Western blot (
Figure 5). We studied the second line of defense against oxidative stress by analyzing protein levels of heme oxygenase-1 (HO-1) by Western blot, but we did not observe an increase in HO-1 levels after 24 h of exposure to B
aP (
Figure 5). These results combined with those of the Cell Stress Array for individual exposure to B
aP led to the conclusion that, at a concentration of 0.6 µM and 24 h of exposure, B
aP does not modulate the protein expression of antioxidant defenses.
However, the Cell Stress Array revealed an increase in the phosphorylation of p53 on serine 46 after exposure to 0.6 µM of B
aP (
Figure 4). These results were confirmed by Western blot after 24 h of the 1 µM B
aP treatment (
Figure 6). In the absence of cellular stress, p53 is localized in the cytoplasm and is maintained at a low level of expression by a negative feedback loop mediated by an E3 ubiquitin ligase such as Mdm2, leading to degradation by the proteasome. The p53 protein is stabilized by phosphorylation at multiple sites (such as serine 15 or serine 46) under conditions of cellular stress, such as oxidative stress, DNA damage, and hypoxia [
42]. B
aP at the times and doses tested did not lead to overoxidation of Prx-SO
3. However, Western blotting revealed an increase in γ-H2AX, the phosphorylated form of the histone H2AX, signaling double-stranded DNA damage after B
aP treatment (
Figure 6). The accumulation of phosphorylated p53 in the cytoplasm leads to its nuclear translocation for the activation of target genes, which leads to different cellular fates (cell cycle arrest, quiescence, senescence, or apoptosis). The increase in the protein level of p53 does not seem to be linked to the induction of apoptosis in trophoblasts incubated with B
aP (
Figure 1). This could instead be linked to the genotoxic effect of B
aP with the increase in γ-H2AX after B
aP treatment (
Figure 6). Interestingly, co-exposure did not lead to an increase in γ-H2AX compared to the control. This difference between exposure to B
aP alone and co-exposure could be due to the potential antioxidant effect of CeO
2 NP, counterbalancing the genotoxic effect of B
aP. Indeed, CeO
2 NP are currently being studied for therapeutic applications due to their antioxidant properties and their ability to reduce DNA damage [
43].
In parallel, the Cell Stress Array shows a 41% increase in the p21 protein level after exposure to B
aP and a 30% increase after co-exposure to B
aP and CeO
2 NP compared to the control. In addition, p21, which is also known as cyclin-dependent kinase inhibitor 1, is a transcriptional target of p53 involved in cell cycle regulation and senescence. This result was then confirmed by Western blotting and RT-qPCR (
Figure 6). We confirmed that the induction of p21 after treatment with B
aP was due to the action of the transcription factor p53 using pifithrin-α, an inhibitor of the transactivation of p53-responsive genes. Moreover, pifithrin-α is also a potent agonist of AhR [
44]. The use of this inhibitor with B
aP led to p21 mRNA and protein levels close to control values (
Figure 7C). These observations are in agreement with those reported in the literature for trophoblastic cell lines. For example, in the JEG-3 choriocarcinoma cell line, exposure to B
aP leads to DNA damage with the phosphorylation of p53 on serine 15 and the induction of the expression of p21, which is responsible for cycle cell arrest in the G2/M phase, without leading to cell apoptosis [
45,
46]. In BeWo cell lines, B
aP activates the AhR pathway with the induction of CYP1A1, increases syncytialization and the production of βhCG, and stabilizes p53 by phosphorylation with the induction of its target p21 [
32]. However, it is important to highlight that, unlike trophoblastic cell lines, primary cytotrophoblasts are differentiated nonproliferative cells, which have exited the cell cycle. Thus, in primary VCT, the observed increase in p21 level cannot be correlated with cell cycle arrest. As pregnancy progresses, inner VCT fuse with the outer ST of the chorionic villi. This phenomenon is considered to indicate the terminal stage of CT differentiation and is mediated by the retroviral syncytin-1 protein [
47]. The multinucleated ST layer of the placenta exhibits characteristics of senescent cells (including the expression of the senescence marker p53 and the CDK inhibitor p21, with a lack of proliferation) [
48]. These senescence pathways are dysregulated up or down in placental pathologies, such as intrauterine growth restriction or pre-eclampsia [
49,
50]. The functional significance of senescence in ST for the human placenta is not understood. One hypothesis is that the resistance of senescent cells to apoptosis maintains ST viability throughout pregnancy, also involving upregulation of anti-apoptotic proteins of the Bcl-2 protein family, which is known to maintain senescent cell viability in other cell types [
51]. The induction of p21 after B
aP treatment and the decrease in Bcl-2 observed in the Cell Stress Array (
Figure 4) could affect the syncytium and lead to premature aging of the placenta.
Western blot and RT-qPCR results indicate that the stabilization of p53 with the induction of its target p21 partially depends on the metabolism of BaP by the AhR pathway. Indeed, after treatment with BaP in the presence of the AhR antagonist, the stabilization of p53 and the induction of p21 were reduced, and no increase in γ-H2AX was observed. With the stabilization of p53, the induction of p21 and CYP1A1, and the phosphorylation of H2AX, we can assume that BaP preferentially induces genotoxicity with the absence of major oxidative stress at this concentration of BaP.
Overall, the Cell Stress Array highlights a greater number of actors whose protein levels decrease in co-exposure compared to individual exposure. This, therefore, suggests that other routes could be modulated in relation to individual exposure. However, the overall decreases observed after incubation with CeO
2 NP could be due to their capacity to adsorb proteins on their surface [
52] and form a corona. This could explain the differences in results observed between the Cell Stress Array, Western blot, and IHC. Indeed, for the Cell Stress Array and Western blots, protein extracts were centrifuged beforehand. The sedimented CeO
2 NP may have carried proteins with them. However, the differences in IHC results may be due to the different response of all the villous cells compared to that of the trophoblasts only. In the cytoplasm, NP aggregates can sequestrate intracellular proteins. CeO
2 NP could, thus, modulate the signaling pathways by capturing molecules that can no longer intervene in their pathway. B
aP, being lipophilic and crossing plasma membranes freely, could facilitate the cellular internalization of NP. Conversely, the ability of nanoparticles to adsorb B
aP on their surface could not only modify the internalization of B
aP and CeO
2 NP, but could also change the kinetics of B
aP biotransformation in the cells and thus modify its toxicity. Additional studies are needed to verify these hypotheses.
4. Materials and Methods
4.1. Placenta Collection
The study was performed according to the principles of the Declaration of Helsinki. Placentas were collected from nonsmoking, healthy women with pregnancies delivered by caesarean section between 39 and 41 weeks of amenorrhea (hereafter called “term”). After obtaining written consent from informed patients and approval from our local ethics committee (CPP: 2015-May-13909), placentas were obtained from maternity hospitals in the Île-de-France region: Port-Royal Maternity, the Institut Mutualiste Montsouris, the Private Hospital of Antony, the Béclère Hospital, the Beaujon Hospital and the Diaconesses Hospital.
4.2. Cytotrophoblast Purification and Culture
After collection, placental tissues were washed in Ca
2+- and Mg
2+-free Hank’s balanced salt solution (HBSS, Gibco #14175, Thermo Fisher Scientific, Illkirch, France). Chorionic villi were gently scraped free from vessels and connective tissue, and dissected into approximate 25 mg fragments. The time for placental dissection was kept under 30 min to prevent tissue degradation. The mononucleated villous cytotrophoblasts were isolated, based on the methods of Kliman et al. [
53]. After dissection, the chorionic villi were washed in Ca
2+- and Mg
2+-free HBSS, and then digested in trypsin digestion medium containing HBSS 5 mL/g, 0.1% trypsin (Gibco #27250-018), 0.1 M MgSO
4 (Merck #5886-0500, New York, NY, USA), 0.1 M CaCl
2 (Merck #1-02820-1000), 4% milk, and 50 Kunitz/mL DNAse type IV (Sigma-Aldrich #D5025, Saint Quentin Fallavier, France), for 30 min at 37 °C without stirring. The following digestions of 10 min, with renewal of the trypsin solution at each digestion, were monitored by observation under an optical microscope. Digestions containing a majority of VCT were kept and pooled. The chorionic villi were finally washed with warm HBSS (37 °C). Each time, the supernatant containing VCT was collected after tissue sedimentation, filtered (on 40 µm pore filters), and incubated with 10% fetal calf serum (FCS, vol/vol) to stop the trypsin activity. After purification by Percoll gradient, VCT were resuspended and cultured in Dulbecco′s modified Eagle′s medium (DMEM, containing 1 g/L glucose, pyruvate, without phenol red, Thermo Fisher Scientific, #11880, Illkirch, France) supplemented with 10% FCS (Eurobio #CVFSVF00-01, Les Ulis, France), 2 mM glutamine (Sigma-Aldrich #G7513), 100 IU/mL penicillin, and 100 µg/mL streptomycin (Gibco #15140-122, Thermo Fisher Scientific) at 150,000 cells/cm
2 on 60 mm diameter culture dishes. After around 16 h of culture (here termed “overnight”), VCT were carefully washed to eliminate nonadherent cells. Purified VCT were characterized for each culture to ensure the homogeneity between purifications by microscopic visualization, the ability to aggregate at 24 h of culture and to form ST at 48 h, and by monitoring the production of hCG secreted into the supernatant.
4.3. Treatments
B
aP (Sigma-Aldrich #B1760) was dissolved in DMSO at 10 mM and stored at 4 °C (stock solution). Trophoblasts were incubated with B
aP for 30 min, 4 h, or 24 h at a final concentration of 0.1, 0.6, or 1 μM. CeO
2 NP were obtained from the Joint Research Centre of the European Union (NM-212, IHCP, Ispra, Italy). The physical and chemical characterization of CeO
2 NP had been previously performed [
25]. NP were dispersed in DMEM cell culture media without FCS at a concentration of 3 mg/mL through sonication with a sonifier equipped with a cup horn (450 W and 50/60 Hz, Branson, Danbury, CT, USA) at 70% amplitude, on ice, for 2 min. NP were then sequentially diluted in cell culture medium with FCS immediately before use, to give a final concentration of 6.3 μg/cm
2 (corresponding to 40 μg/mL as in our previous study [
25]) or of 10 μg/cm
2 (64 μg/mL). Trophoblasts were incubated with CeO
2 NP for 30 min, 4 h, or 24 h. The AhR antagonist CH223191 (Sigma-Aldrich #182705) was used at 3 μM in culture medium (3 mM stock solution in DMSO) and p53 inhibitor pifithrin-α (Sigma-Aldrich #P4359) was used at 20 µM in culture medium (10 mM stock solution in DMSO). AhR and p53 inhibitors were incubated with cells for 1 h prior to B
aP incubation. Staurosporin (STS, Sigma-Aldrich #S4400) was used at a final concentration of 0.5 µM for 2 to 6 h and TBHP (Luperox TBH70X solution, Sigma-Aldrich #458139) at 100 µM for 4 h.
4.4. Metabolic Activity by WST-1
Metabolic activity was assessed using the WST-1 assay (Sigma-Aldrich #11644807001)—in which mitochondrial dehydrogenase cleaves the tetrazolium salt 2-(4-iodophenyl)-3-(4-nitrophenyl)5-(2,4-disulfophenyl)-2H-tetrazolium (WST-1) into formazan—according to the manufacturer’s instructions. Trophoblast cells were cultured overnight in 48-well plates, washed with fresh medium and treated accordingly for 24 to 72 h. At the end of the exposure time, cells were rinsed with culture medium and then WST-1 reagent was added (1:100) to each well and incubated for 3 h at 37 °C. Spectrometric absorbance was measured using a microplate reader (EnSpire 2300 Multilabel reader, PerkinElmer, Villebon-sur-Yvette, France) at 440 nm, and using 600 nm as correction wavelength (to remove the nonspecific emission). To measure the background noise from NP, the same experiment was performed in parallel, followed by the addition of Triton to each well 15 min before adding WST-1 reagent, and the corresponding absorbances were subtracted from the results.
4.5. Protein Extraction and Western Blot
Total protein extracts from trophoblast cells from 60 mm dish cultures were obtained by harvesting cells in Laemmli lysis buffer, with added protease inhibitors (1:100, Protease Inhibitor Cocktail Set I, Calbiochem Merck #539131) and phosphatase inhibitors (1:50, Phosphatase Inhibitor Cocktail Set V, Calbiochem Merck # 524629). Protein extracts were centrifuged for 5 min at 14,000× g and stored at −80 °C. Protein concentrations were determined using the Pierce™ BCA Protein Assay Kit (Thermo Fisher Scientific #23235). Equal amounts of proteins (30 µg) were separated on 7.5%, 4–15%, or 8–16% SDS-PAGE mini-PROTEAN® TGX™ precast protein gel (Bio-Rad #4561085 #4561084 #4561103, Marnes-la-Coquette, France) under reducing conditions (dithiothreitol 10%, Sample Reducing Agent 10×, Invitrogen #NP0009, Waltham, MA, USA) and transferred onto a nitrocellulose membrane (Trans-blot Turbo Transfer pack, 0.2 µm nitrocellulose, Bio-Rad #1704159). Blots were incubated overnight with the primary antibody at 4 °C, and then for 1 h with the appropriate DyLight 680 or 800 Fluor-conjugated secondary antibody (Thermo Fisher Scientific #35568 and #35521). The primary antibodies used were mouse anti-AhR (1:500, Sigma-Aldrich #WH0000196-M2), rabbit anti-CYP1A1 (1:1000, Proteintech #3241-1-AP, Rosemont, IL, USA), rabbit anti-HO-1 (1:1000, Enzo Life Sciences #ADI-SPA-895D, Farmingdale, NY, USA), rabbit anti-ɣ-H2AX (1:1000, Cell Signaling, #9718, Danvers, MA, USA), rabbit anti-NFκB p65 (1:2000, Abcam #ab16502, Cambridge, UK), rabbit anti-p21 (1:1000, Cell Signaling, #2947), mouse anti-p53 (1:1000, Cell Signaling #18032), rabbit anti-PARP-1 (1:1000, Cell Signaling #9542), rabbit anti-Prx-SO3 (1:500, Abcam #ab16830), mouse anti-vinculin (1:1000, Sigma-Aldrich 1V9131). Secondary antibodies were DyLight 800-labeled anti-mouse (1:20,000, Thermo Fisher Scientific #35521) or DyLight 680-labeled anti-rabbit (1:20,000, Thermo Fisher Scientific #35568). Primary and secondary antibodies were diluted in the following dilution buffer: 1X TBS, 0.1% Tween-20, 5% nonfat dry milk, 0.1% sodium azide. Blots were scanned with an Odyssey® Imaging System (Li-COR, Bad Homburg, Germany). Quantification was performed using the Li-COR Odyssey software.
4.6. hCG ELISA
The hCG assay was carried out in the centrifuged supernatants (3 min at 500× g) of the VCT exposed to pollutants for 48 h. The assays were performed according to the supplier’s instructions (Abcam® ab100533) by sandwich ELISA (enzyme-linked immunosorbent assay). The calibration range was established with recombinant human hCG and varying from 54.87 pg/mL to 40,000 pg/mL. The range and the samples were deposited in duplicate. The absorbance was measured at 450 nm using a microplate reader (EnSpire 2300 Multilabel reader, PerkinElmer™) from which the absorbance measured at 600 nm was removed in order to exclude nonspecific emissions from the plate. These results were normalized to the quantity of protein present in each culture well.
4.7. Cell Stress Array
The effects of BaP and CeO2 NP on signaling involved in cellular stress were evaluated using the Human Cell Stress Array kit (R&D systems #ARY018) in accordance with the manufacturer’s instructions. Total protein extracts obtained with the lysis buffer from the kit were incubated for 30 min on a rocking platform shaker at 4 °C. Equal amounts of each total protein extract (n = 5) for each condition were pooled to obtain 300 μg of a representative sample for each condition. Membranes were saturated for 1 h with the buffer provided and then incubated overnight at 4 °C with the protein extracts and the detection antibody cocktail. Membranes were washed three times for 10 min and incubated with streptavidin coupled with a fluorochrome (1:2000, LI-COR Biosciences IRDye #926-32230). Blots were scanned with an Odyssey® Imaging System (Li-COR, Bad Homburg, Germany). Quantification was performed using the Li-COR Odyssey software.
4.8. RNA Extraction, Reverse Transcription, and Real-Time Quantitative PCR
The treated VCT were recovered by trypsin-EDTA 0.05% (Gibco™ 25300054). Total RNA extraction was performed on pellets of 3 million cells with the RNeasy
® mini kit (Qiagen
® 74104) according to the supplier’s instructions, with DNase (Qiagen
® 79254). After elution of the RNAs, the RNA concentration (A260) and the purity of the samples (A260/A280 and A260/A230 ratios) were determined by a spectrophotometer (Nanodrop ND-1000). RNAs with an A260/A230 ratio of less than 1.6 were purified with the Qiagen
® RNeasy
® MinElute Cleanup kit (74204). The total RNAs recovered were stored at −80 °C after addition of 1 µL of RNase-OUT (Invitrogen™ 10777019). Reverse transcription was performed using the Transcriptase inverse SuperScript™ III kit (Invitrogen™ 18080-044) using random primer (Invitrogen, Paisley, UK) for 1 µg RNA. RT-qPCR was carried out in a 10 µL reaction volume containing 10 ng of cDNA, 5 µM of each primer, and 5 µL of SYBR™ Green (Master Mix PowerTrack™ Applied Biosystems™ A46111) using the CFX384 Real-Time PCR Detection System (Bio-Rad™). All primers (
Table 1) were validated beforehand [
5]. PCR cycles consisted of the following steps: polymerase activation (2 min, 95 °C), denaturation (15 s, 95 °C), and annealing and extension (1 min, 60 °C), followed by a melting curve to ensure no contamination. The threshold cycle (Ct) was measured as the number of cycles at which the fluorescence emission first exceeds the background. The relative amounts of mRNA were estimated using the ∆∆Ct method and then expressed as fold change [
54]. Primers for the reference genes HPRT, RPLO, and SDHA were used for the normalization of the results. Each gene was normalized to the geometric mean of the 3 reference genes.
4.9. Immunochemistry
Placental explants treated for 24 h with DMSO, 1 µM of B
aP, and/or 10 µg/cm
2 of CeO
2 NP (
n = 3 per condition) were fixed in 4% paraformaldehyde (Electron Microscopy Sciences, Hatfield, PA, USA) overnight at 4 °C, and then dehydrated and embedded in Paraplast
®. Placental tissue sections (5 µm) were deparaffinized, rehydrated in ethanol/water, and processed for heat-induced antigen retrieval in citrate buffer, pH 6.1 or Tris/EDTA buffer pH 9 (Dako, Trappes, France) for 45 min at 90 °C in a water bath. Sections were permeabilized in 0.3% Triton X-100 for 4 min, and then incubated with rabbit anti-hypoxia-inducible factor 1α (HIF-1α) antibody diluted 1:100 (200 µg/mL; Novus Biologicals #NB100-449, Englewood, CO, USA), rabbit anti-hypoxia-inducible factor 2α (HIF-2α) antibody diluted 1:500 (1 mg/mL; Novus Biologicals #NB100-122), rabbit anti-p21 antibody diluted 1:50 (244 µg/mL; Cell Signaling #2947), mouse anti-p53 antibody diluted 1:100 (237 µg/mL; Dako #M7001), or nonspecific rabbit/mouse IgG as controls. Staining was visualized using Novolink™ Polymer Detection Systems (Leica Biosystems). Slides were scanned using a Lamina multilabel slide scanner (Perkin Elmer) using brightfield imaging at a magnification of ×20. Quantification of protein expression was performed on ten random 100 × 100 pixel images per sample (
n = 3 per condition, resulting in a total of 30 images) using the ImageJ 1.52 software, and thresholding analysis was performed as previously described [
55].
4.10. Statistical Analysis
Statistical tests were performed using the GraphPad Prism 7.04 software (La Jolla, CA, USA). The results of the quantitative analysis are presented as means ± standard error of the mean (SEM). The Shapiro–Wilks normality test was performed to determine whether the samples followed a normal distribution. Differences between groups were evaluated with the parametric paired Student’s t-test for samples with a normal distribution and with the nonparametric Wilcoxon–Mann–Whitney test for samples with a non-normal distribution. A p-value lower than 0.05 was considered to be statistically significant, with p < 0.05, p < 0.01, p < 0.001 p < 0.0001 represented as *, **, *** and ****, respectively.