Simulated Microgravity Alters P-Glycoprotein Efflux Function and Expression via the Wnt/β-Catenin Signaling Pathway in Rat Intestine and Brain
Abstract
1. Introduction
2. Results
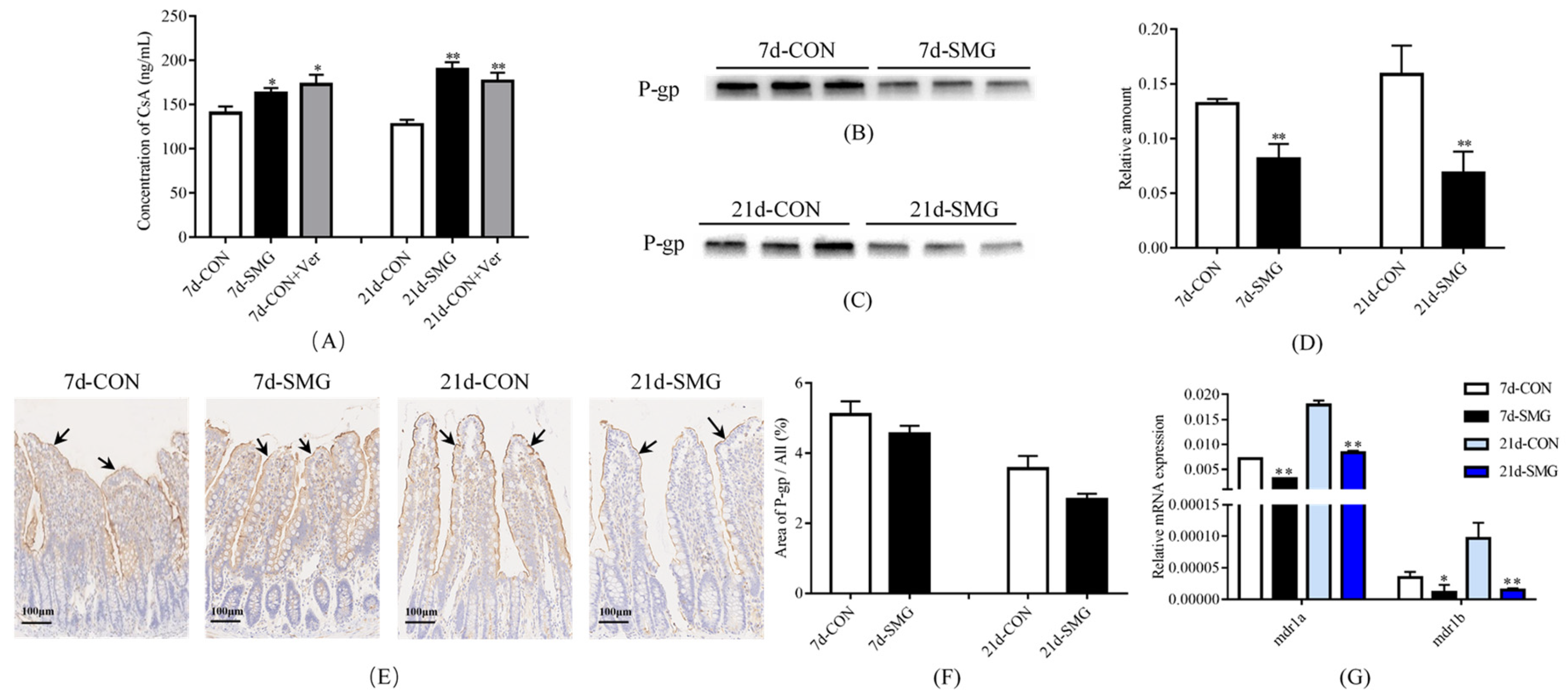
2.1. Smg Inhibited the Efflux Function and Expression of P-Gp in Rat Intestine and Caco-2 Cells
2.1.1. SMG Inhibited the Efflux Function and Expression of P-Gp in Rat Intestine
2.1.2. 72 h SMG Treatment Inhibited the Efflux Function and Expression of P-Gp in Caco-2 Cells
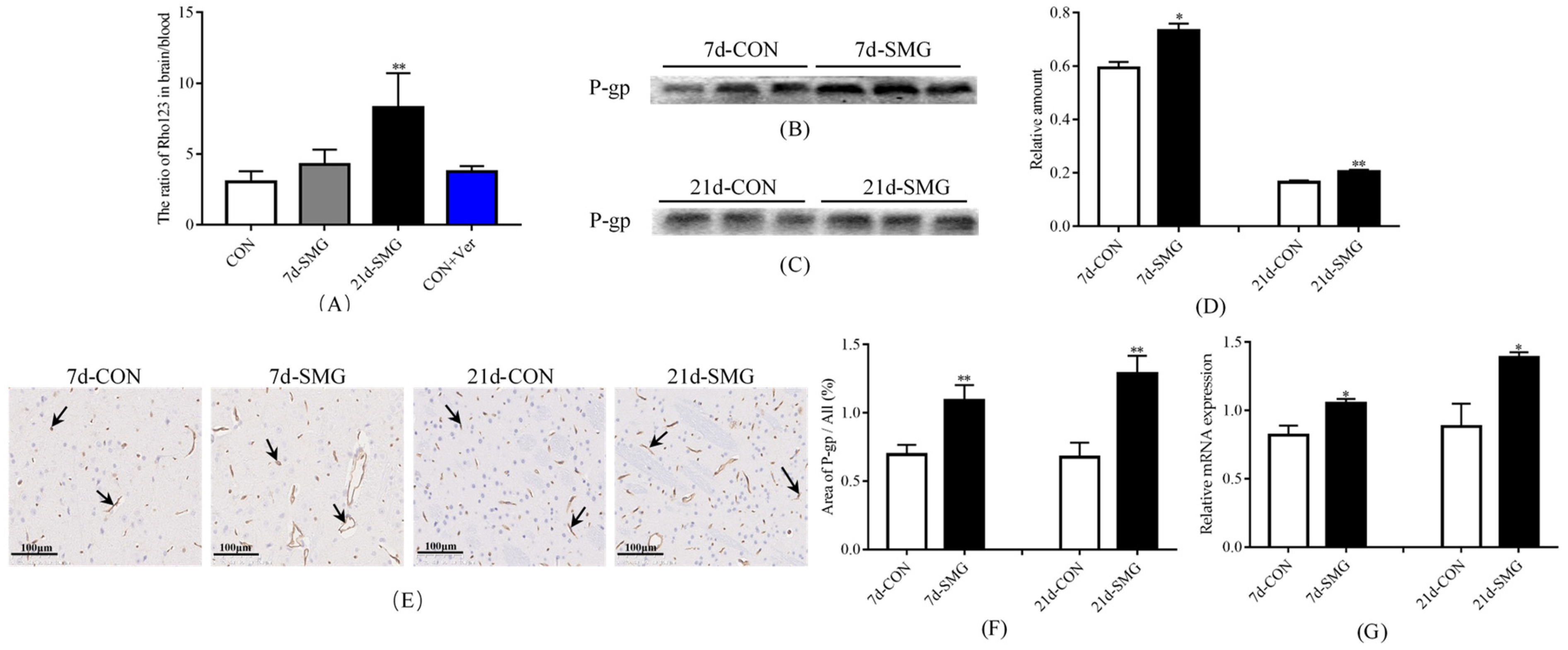
2.2. SMG Inhibited the Efflux Function and Activated the Expression of P-Gp in Rat Brain and hCMEC/D3 Cells
2.2.1. SMG Inhibited the Efflux Function and Activated the Expression of P-Gp in Rat Brain
2.2.2. 48 and 72 h SMG Treatment Inhibited the Efflux Function and Activated the Expression of P-Gp in hCMEC/D3 Cells
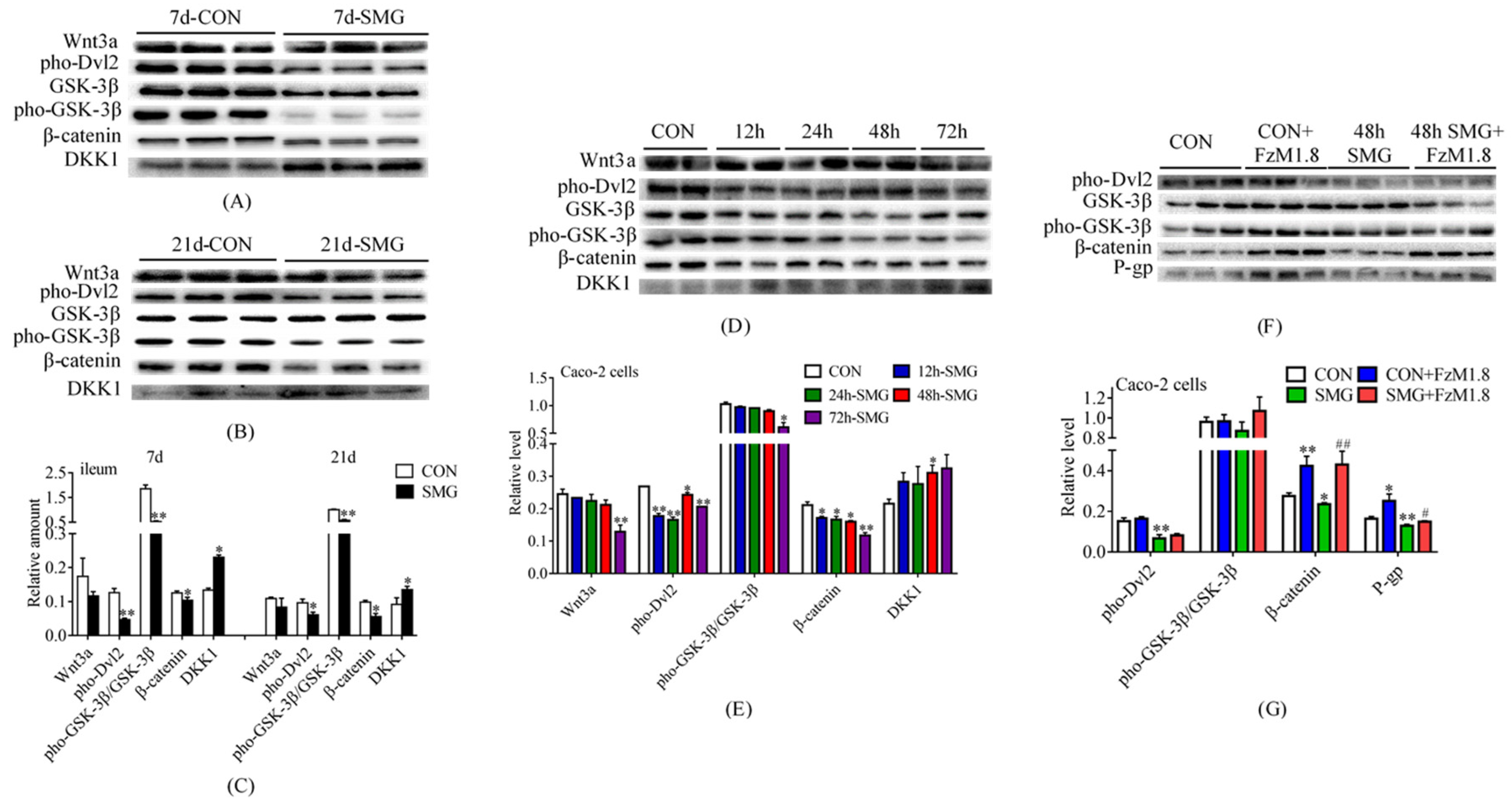
2.3. SMG Down-Regulated P-Gp Expression by Inhibiting the Wnt/β-Catenin Signaling Pathway in Rat Intestine and Caco-2 Cells
2.4. SMG Up-Regulated P-Gp Expression by Activating the Wnt/β-Catenin Signaling Pathway in Rat Brain and hCMEC/D3 Cells
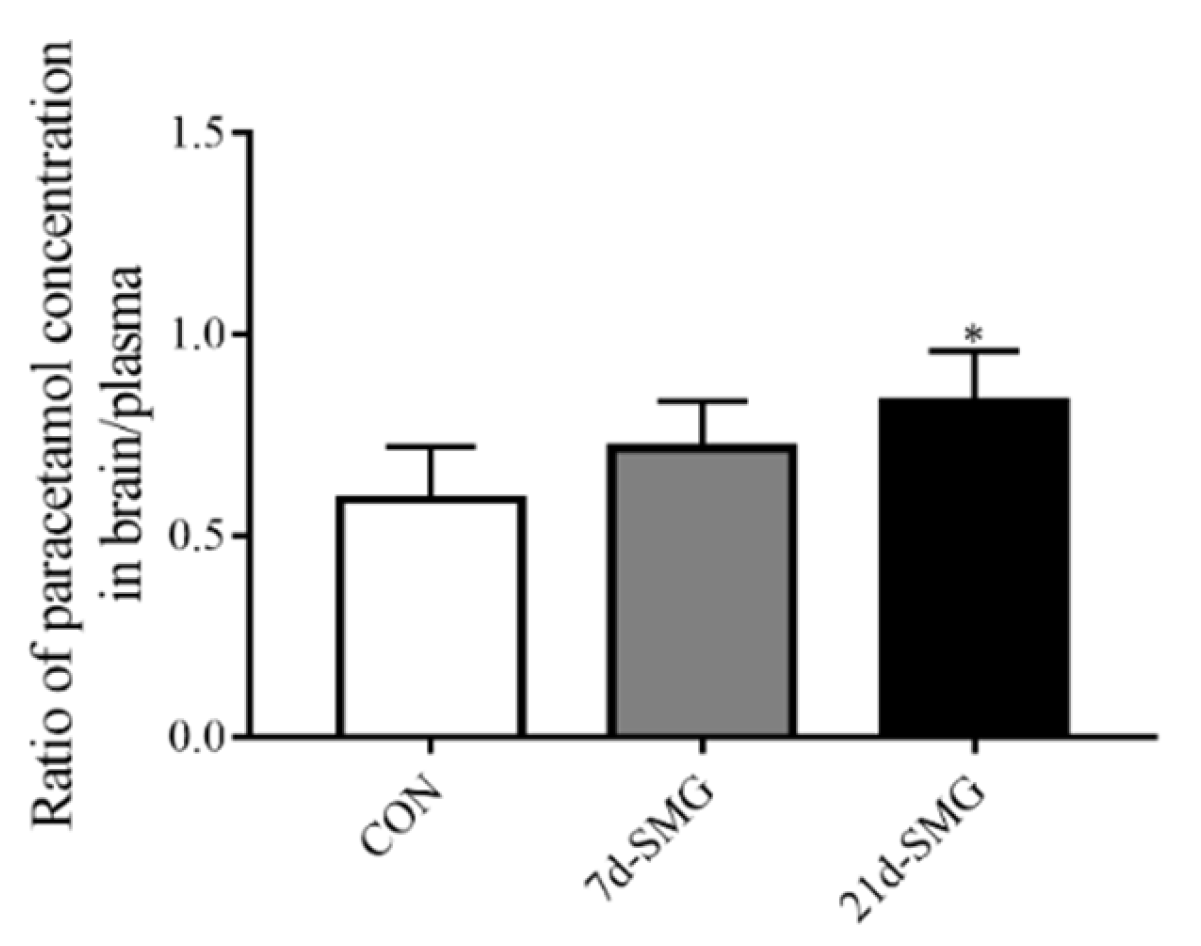
2.5. SMG Promoted the In Vivo Intestinal Absorption and Brain Distribution of AP
3. Discussion
4. Materials and Methods
4.1. Animal Treatment and Sample Collection
4.2. Efflux Function Analysis of P-Gp in Rat Intestine and Brain
4.3. Immunohistochemical Staining
4.4. Cell Culture and SMG Treatment
4.5. Efflux Function Analysis of P-Gp in Caco-2 and hCMEC/D3 Cells
4.6. Western-Blot
4.7. qPCR
4.8. Immunofluorescence
4.9. Rat Single-Pass Intestinal Perfusion of Acetaminophen
4.10. Rat Brain Distribution of Acetaminophen
4.11. Acetaminophen Determination Using the HPLC-UV Method
4.12. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| P-gp | permeability glycoprotein |
| MG | microgravity |
| SMG | simulated microgravity |
| CON | Control |
| IECs | intestinal epithelial cells |
| BBB | blood-brain barrier |
| AP | acetaminophen |
| Caco-2 | human colon adenocarcinoma cell |
| hCMEC/D3 | human cerebral microvascular endothelial cell |
| CsA | cyclosporine A |
| Ver | Verapamil |
| IHC | immunohistochemical |
| qPCR | Quantitative Polymerase Chain Reaction |
| rhodamine 123 | Rho123 |
| pho-Dvl2 | phosphorylated disheveled 2 |
| GSK-3β | glycogen synthase kinase-3β |
| DKK1 | Dickkopf 1 |
| SPIP | single-pass intestinal perfusion |
| Peff | effective permeability |
| HPLC | high-performance liquid chromatography |
| IS | internal standard |
| Ka | absorption constant |
| Kp brain | brain-to-plasma concentration ratio |
| Axin | axis inhibitor |
| APC | adenomatous polyposis coli protein |
| CK1 | casein kinase 1 |
| Fzd | Frizzled |
| LRP5/6 | lipoprotein receptor-related proteins 5/6 |
| TCF | transcription factor |
| BSA | bovine serum albumin |
| TBST | Tris-buffered saline tween |
| DMSO | dimethyl sulfoxide |
| PBS | phosphate buffer saline |
| GAPDH | glyceraldehyde-3-phosphate dehydrogenase |
| KR solution | Krebs–Ringer’s solution |
References
- Kandarpa, K.; Schneider, V.; Ganapathy, K. Human health during space travel: An overview. Neurol. India 2019, 67, S176–S181. [Google Scholar] [PubMed]
- Shen, M.; Frishman, W.H. Effects of spaceflight on cardiovascular physiology and health. Cardiol. Rev. 2019, 27, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Yuan, M.; Cheng, C.; Xia, D.H.; Wu, S.W. Chinese herbal medicine effects on muscle atrophy induced by simulated microgravity. Aerosp. Med. Hum. Perform. 2018, 89, 883–888. [Google Scholar] [CrossRef] [PubMed]
- Nagaraja, M.P.; Risin, D. The current state of bone loss research: Data from spaceflight and microgravity simulators. J. Cell. Biochem. 2013, 114, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Russomano, T.; Da Rosa, M.; Dos Santos, M.A. Space motion sickness: A common neurovestibular dysfunction in microgravity. Neurol. India 2019, 67, 214. [Google Scholar] [CrossRef] [PubMed]
- Borchers, A.T.; Keen, C.L.; Gershwin, M.E. Microgravity and immune responsiveness: Implications for space travel. Nutrition 2002, 18, 889–898. [Google Scholar] [CrossRef]
- Alvarez, R.; Stork, C.A.; Sayoc-Becerra, A.; Marchelletta, R.R.; Prisk, G.K.; Mccole, D.F. A simulated microgravity environment causes a sustained defect in epithelial barrier function. Sci. Rep. 2019, 9, 17531. [Google Scholar] [CrossRef]
- Kast, J.; Yu, Y.; Seubert, C.N.; Wotring, V.E.; Derendorf, H. Drugs in space: Pharmacokinetics and pharmacodynamics in astronauts. Eur. J. Pharm. Sci. 2017, 109, S2–S8. [Google Scholar] [CrossRef]
- Stepanek, J.; Blue, R.S.; Parazynski, S. Space medicine in the era of civilian spaceflight. N. Engl. J. Med. 2019, 380, 1053–1060. [Google Scholar] [CrossRef]
- Choi, Y.H.; Yu, A.M. ABC transporters in multidrug resistance and pharmacokinetics, and strategies for drug development. Curr. Pharm. Des. 2014, 20, 793–807. [Google Scholar] [CrossRef]
- Vilar, S.; Sobarzo-Sánchez, E.; Uriarte, E. In silico prediction of P-glycoprotein binding: Insights from molecular docking studies. Curr. Med. Chem. 2019, 26, 1746–1760. [Google Scholar] [CrossRef] [PubMed]
- Lavie, Y.; Fiuccii, G.; Liscovitch, M. Upregulation of caveolae in multidrug resistant cancer cells: Functional implications. Adv. Drug Deliv. Rev. 2001, 49, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, C.; Buyse, M.; German-Fattal, M.; Gimenez, F. Influence of the pro-inflammatory cytokines on P-glycoprotein expression and functionality. J. Pharm. Pharm. Sci. 2004, 7, 359–371. [Google Scholar] [PubMed]
- Shen, D.Y.; Zhang, W.; Zeng, X.; Liu, C.Q. Inhibition of Wnt/β-catenin signaling downregulates P-glycoprotein and reverses multi-drug resistance of cholangiocarcinoma. Cancer Sci. 2013, 104, 1303–1308. [Google Scholar] [CrossRef]
- Wijesuriya, H.C.; Bullock, J.Y.; Faull, R.L.; Hladky, S.B.; Barrand, M.A. ABC efflux transporters in brain vasculature of Alzheimer’s subjects. Brain Res. 2010, 1358, 228–238. [Google Scholar] [CrossRef]
- Kim, D.Y.; Jung, S.Y.; Kim, K.; Kim, C.J. Treadmill exercise ameliorates Alzheimer disease-associated memory loss through the Wnt signaling pathway in the streptozotocin-induced diabetic rats. J. Exerc. Rehabil. 2016, 12, 276–283. [Google Scholar] [CrossRef]
- Wessler, J.D.; Grip, L.T.; Mendell, J.; Giugliano, R.P. The P-glycoprotein transport system and cardiovascular drugs. J. Am. Coll. Cardiol. 2013, 61, 2495–2502. [Google Scholar] [CrossRef]
- Lin, J.H.; Yamazaki, M. Role of P-glycoprotein in pharmacokinetics: Clinical implications. Clin. Pharmacokinet. 2003, 42, 59–98. [Google Scholar] [CrossRef]
- Arellano, C.; Philibert, C.; Vachoux, C.; Woodley, J.; Houin, G. The metabolism of midazolam and comparison with other CYP enzyme substrates during intestinal absorption: In vitro studies with rat everted gut sacs. J. Pharm. Pharm. Sci. 2007, 10, 26–36. [Google Scholar]
- Zhang, Y.; Huang, J.; Liu, Y.; Guo, T.; Wang, L. Using the lentiviral vector system to stably express chicken P-gp and BCRP in MDCK cells for screening the substrates and studying the interplay of both transporters. Arch. Toxicol. 2018, 92, 2027–2042. [Google Scholar] [CrossRef]
- Geng, J.; Luan, L.B. Transport characteristics of ibuprofen in Caco-2 cell monolayers. Chin. J. Pharm. 2010, 41, 583–586. [Google Scholar]
- Li, Y.J.; Liu, S.; Liu, H.Y.; Cui, Y.Y.; Deng, Y.L. Dragon’s blood regulates Rac1-WAVE2-Arp2/3 signaling pathway to protect rat intestinal epithelial barrier dysfunction induced by simulated microgravity. Int. J. Mol. Sci. 2021, 22, 2722. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.J.; Huang, L.L.; Iqbal, J.; Deng, Y.L. Investigation on P-glycoprotein function and its interacting proteins under simulated microgravity. Space Sci. Technol. 2021, 2021, 9835728. [Google Scholar] [CrossRef]
- Lu, S.K.; Bai, S.; Javeri, K.; Brunner, L.J. Altered cytochrome P450 and P-glycoprotein levels in rats during simulated weightlessness. Aviat. Space Environ. Med. 2002, 73, 112–118. [Google Scholar] [PubMed]
- Wagner, J.; Abdel-Rahman, S.M. Pediatric pharmacokinetics. Pediatr. Rev. 2013, 34, 258–269. [Google Scholar] [CrossRef]
- Xiang, Q.; Xiao, C.; Xu, M.; Xu, B.; Li, H.Y.; Geng, C.Y.; Fang, Q.; Tang, J.T. Effects of space environment on gene expression profile of melanoma cells. Chin. J. Aerospace. Med. 2006, 17, 165–168. [Google Scholar]
- Yan, R.R.; Liu, H.Y.; Lv, F.; Deng, Y.L.; Li, Y.J. Rac1/Wave2/Arp3 Pathway Mediates Rat Blood-Brain Barrier Dysfunction under Simulated Microgravity Based on Proteomics Strategy. Int. J. Mol. Sci. 2021, 22, 5165. [Google Scholar] [CrossRef]
- Liu, X.L.; Tang, J.; Song, J.; He, J.; Xu, P.; Peng, W.X. The effect of tetramethylpyrazine on the pharmacokinetics of intragastrically administered cyclosporine A in rats. Acta Pharm. Sin. 2006, 41, 882–887. [Google Scholar]
- Riccio, G.; Bottone, S.; La Regina, G.; Badolati, N.; Passacantilli, S.; Rossi, G.B.; Accardo, A.; Dentice, M.; Silvestri, R.; Novellino, E.; et al. A negative allosteric modulator of WNT receptor Frizzled 4 switches into an allosteric agonist. Biochemistry 2018, 57, 839–851. [Google Scholar] [CrossRef]
- You, L.; Zhang, C.; Yarravarapu, N.; Morlock, L.; Wang, X.; Zhang, L.; Williams, N.S.; Lum, L.; Chen, C. Development of a triazole class of highly potent Porcn inhibitors. Bioorg. Med. Chem. Lett. 2016, 26, 5891–5895. [Google Scholar] [CrossRef]
- Strindberg, S.; Plum, J.; Stie, M.B.; Christiansen, M.L.; Hagner Nielsen, L.; Rades, T.; Müllertz, A. Effect of supersaturation on absorption of indomethacin and tadalafil in a single pass intestinal perfusion rat model, in the absence and presence of a precipitation inhibitor. Eur. J. Pharm. Biopharm. 2020, 151, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Stappaerts, J.; Brouwers, J.; Annaert, P.; Augustijns, P. In situ perfusion in rodents to explore intestinal drug absorption: Challenges and opportunities. Int. J. Pharm. 2015, 478, 665–681. [Google Scholar] [CrossRef] [PubMed]
- Komorowski, M.; Fleming, S.; Kirkpatrick, A.W. Fundamentals of Anesthesiology for Spaceflight. J. Cardiothorac. Vasc. Anesth. 2016, 30, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Dello Russo, C.; Bandiera, T.; Monici, M.; Surdo, L.; Yip, V.L.M.; Wotring, V.E.; Morbidelli, L. Physiological adaptations affecting drug pharmacokinetics in space: What do we really know? A critical review of the literature. Br. J. Pharmacol. 2022, 179, 2538–2557. [Google Scholar] [CrossRef] [PubMed]
- Lown, K.S.; Mayo, R.R.; Leichtman, A.B.; Hsiao, H.L.; Turgeon, D.K.; Schmiedlin-Ren, P.; Brown, M.B.; Guo, W.; Rossi, S.J.; Benet, L.Z.; et al. Role of intestinal P-glycoprotein (mdr1) in interpatient variation in the oral bioavailability of cyclosporine. Clin. Pharmacol. Ther. 1997, 62, 248–260. [Google Scholar] [CrossRef]
- Hashida, T.; Masuda, S.; Uemoto, S.; Saito, H.; Tanaka, K.; Inui, K. Pharmacokinetic and prognostic significance of intestinal MDR1 expression in recipients of living-donor liver transplantation. Clin. Pharmacol. Ther. 2001, 69, 308–316. [Google Scholar] [CrossRef]
- Terada, T.; Hira, D. Intestinal and hepatic drug transporters: Pharmacokinetic, pathophysiological, and pharmacogenetic roles. J. Gastroenterol. 2015, 50, 508–519. [Google Scholar] [CrossRef]
- Zhao, J. Study on the Pharmacokinetics of Vitamin B6 and Acetaminophen in Simulated Weightlessness SD Rats. Master’s Thesis, Fourth Military Medical University, Xi’an, China, 2018. [Google Scholar]
- Bartels, A.L.; de Klerk, O.L.; Kortekaas, R.; de Vries, J.J.; Leenders, K.L. 11C-verapamil to assess P-gp function in human brain during aging, depression and neurodegenerative disease. Curr. Top. Med. Chem. 2010, 10, 1775–1784. [Google Scholar] [CrossRef]
- Ding, Y.; Wang, R.; Zhang, J.; Zhao, A.; Lu, H.; Li, W.; Wang, C.; Yuan, X. Potential Regulation Mechanisms of P-gp in the Blood-Brain Barrier in Hypoxia. Curr. Pharm. Des. 2019, 25, 1041–1051. [Google Scholar] [CrossRef]
- Ejsing, T.B.; Linnet, K. Influence of P-glycoprotein inhibition on the distribution of the tricyclic antidepressant nortriptyline over the blood-brain barrier. Hum. Psychopharmacol. Clin. Exp. 2005, 20, 149–153. [Google Scholar] [CrossRef]
- Marier, J.F.; Deschênes, J.L.; Hage, A.; Seliniotakis, E.; Gritsas, A.; Flarakos, T.; Beaudry, F.; Vachon, P. Enhancing the uptake of dextromethorphan in the CNS of rats by concomitant administration of the P-gp inhibitor verapamil. Life Sci. 2005, 77, 2911–2926. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Yang, Z.; Yang, J.; Yang, H. Increased P-glycoprotein expression and decreased phenobarbital distribution in the brain of pentylenetetrazole-kindled rats. Neuropharmacology 2007, 53, 657–663. [Google Scholar] [CrossRef] [PubMed]
- van Vliet, E.A.; van Schaik, R.; Edelbroek, P.M.; Voskuyl, R.A.; Redeker, S.; Aronica, E.; Wadman, W.J.; Gorter, J.A. Region-specific overexpression of P-glycoprotein at the blood-brain barrier affects brain uptake of phenytoin in epileptic rats. J. Pharmacol. Exp. Ther. 2007, 322, 141–147. [Google Scholar] [CrossRef]
- van Vliet, E.A.; van Schaik, R.; Edelbroek, P.M.; Redeker, S.; Aronica, E.; Wadman, W.J.; Marchi, N.; Vezzani, A.; Gorter, J.A. Inhibition of the multidrug transporter P-glycoprotein improves seizure control in phenytoin-treated chronic epileptic rats. Epilepsia 2006, 47, 672–680. [Google Scholar] [CrossRef] [PubMed]
- Watari, R.; Matsuda, A.; Ohnishi, S.; Hasegawa, H. Minimal contribution of P-gp on the low brain distribution of naldemedine, a peripherally acting μ-opioid receptor antagonist. Drug Metab. Pharmacokinet. 2019, 34, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Uchida, Y.; Ohtsuki, S.; Kamiie, J.; Terasaki, T. Blood-brain barrier (BBB) pharmacoproteomics: Reconstruction of in vivo brain distribution of 11 P-glycoprotein substrates based on the BBB transporter protein concentration, in vitro intrinsic transport activity, and unbound fraction in plasma and brain in mice. J. Pharmacol. Exp. Ther. 2011, 339, 579–588. [Google Scholar] [PubMed]
- Shen, Y.; Li, H.D. P-glycoprotein mediated drug-drug interaction of central nervous system drugs. Chin. J. Clin. Pharmacol. 2008, 24, 533–538. [Google Scholar]
- Tsujikawa, K.; Dan, Y.; Nogawa, K.; Sato, H.; Yamada, Y.; Murakami, H.; Ohtani, H.; Sawada, Y.; Iga, T. Potentiation of domperidone-induced catalepsy by a P-glycoprotein inhibitor, cyclosporin A. Biopharm. Drug Dispos. 2003, 24, 105–114. [Google Scholar] [CrossRef]
- Dan, Y.; Murakami, H.; Koyabu, N.; Ohtani, H.; Sawada, Y. Distribution of domperidone into the rat brain is increased by brain ischaemia or treatment with the P-glycoprotein inhibitor verapamil. J. Pharm. Pharmacol. 2002, 54, 729–733. [Google Scholar] [CrossRef]
- Eyal, S.; Derendorf, H. Medications in Space: In Search of a Pharmacologist’s Guide to the Galaxy. Pharm. Res. 2019, 36, 148. [Google Scholar] [CrossRef]
- Pool, S.L.; Nicogossian, A. Biomedical results of the Space Shuttle orbital flight test program. Aviat. Space Environ. Med. 1983, 54, S41–S49. [Google Scholar] [PubMed]
- Caiani, E.G.; Martin-Yebra, A.; Landreani, F.; Bolea, J.; Laguna, P.; Vaïda, P. Weightlessness and cardiac rhythm disorders: Current knowledge from space flight and bed-rest studies. Front. Astron. Space Sci. 2016, 3, 27. [Google Scholar] [CrossRef]
- Wotring, V.E. Medication use by U.S. crewmembers on the International Space Station. FASEB J. 2015, 29, 4417–4423. [Google Scholar] [CrossRef] [PubMed]
- Wotring, V.E.; Smith, L.K. Dose Tracker Application for Collecting Medication Use Data from International Space Station Crew. Aerosp. Med. Hum. Perform. 2020, 91, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Eyal, S. How do the pharmacokinetics of drugs change in astronauts in space? Expert. Opin. Drug Metab. Toxicol. 2020, 16, 353–356. [Google Scholar] [CrossRef]
- Cintron, N.M.; Putcha, L.; Chen, Y.M.; Vanderploeg, J.M. Inflight salivary pharmacokinetics of acetaminophen in saliva. In Results of the Life Science DSOs Conducted Aboard the Space Shuttle 1981–1986; NASA: Washington, DC, USA, 1987; pp. 19–23. [Google Scholar]
- Gandia, P.; Bareille, M.P.; Saivin, S.; Le-Traon, A.P.; Lavit, M.; Guell, A.; Houin, G. Influence of simulated weightlessness on the oral pharmacokinetics of acetaminophen as a gastric emptying probe in man: A plasma and a saliva study. J. Clin. Pharmacol. 2003, 43, 1235–1243. [Google Scholar] [CrossRef]
- Idkaidek, N.; Arafat, T. Effect of microgravity on the pharmacokinetics of Ibuprofen in humans. J. Clin. Pharmacol. 2011, 51, 1685–1689. [Google Scholar] [CrossRef]
- Polyakov, A.V.; Svistunov, A.A.; Kondratenko, S.N.; Kovachevich, I.V.; Repenkova, L.G.; Savelyeva, M.I.; Kukes, V.G. Peculiarities of Pharmacokinetics and Bioavailability of Some Cardiovascular Drugs under Conditions of Antiorthostatic Hypokinesia. Bull. Exp. Biol. Med. 2020, 168, 465–469. [Google Scholar] [CrossRef]
- Gandia, P.; Saivin, S.; Le-Traon, A.P.; Guell, A.; Houin, G. Influence of simulated weightlessness on the intramuscular and oral pharmacokinetics of promethazine in 12 human volunteers. J. Clin. Pharmacol. 2006, 46, 1008–1016. [Google Scholar] [CrossRef]
- Zhao, Y.; Dong, S.; Kong, Y.; Rui, Q.; Wang, D. Molecular basis of intestinal canonical Wnt/β-catenin BAR-1 in response to simulated microgravity in Caenorhabditis elegans. Biochem. Biophys. Res. Commun. 2020, 522, 198–204. [Google Scholar] [CrossRef]
- Guo, Y.; Xiao, L.; Sun, L.; Liu, F. Wnt/beta-catenin signaling: A promising new target for fibrosis diseases. Physiol. Res. 2012, 61, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Sebio, A.; Kahn, M.; Lenz, H.J. The potential of targeting Wnt/β-catenin in colon cancer. Expert Opin. Ther. Targets 2014, 18, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Lorenzon, A.; Calore, M.; Poloni, G.; De Windt, L.J.; Braghetta, P.; Rampazzo, A. Wnt/β-catenin pathway in arrhythmogenic cardiomyopathy. Oncotarget 2017, 8, 60640–60655. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Xu, C.; Lu, M.; Wu, X.; Tang, L.; Wu, X. Wnt/β-catenin signaling links embryonic lung development and asthmatic airway remodeling. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 3226–3242. [Google Scholar] [CrossRef] [PubMed]
- Laksitorini, M.D.; Yathindranath, V.; Xiong, W.; Hombach-Klonisch, S.; Miller, D.W. Modulation of Wnt/β-catenin signaling promotes blood-brain barrier phenotype in cultured brain endothelial cells. Sci. Rep. 2019, 9, 19718. [Google Scholar] [CrossRef]
- Kang, Y.; Zhang, S.; Cao, W.; Wan, D.; Sun, L. Knockdown of LncRNA CRNDE suppresses proliferation and P-glycoprotein-mediated multidrug resistance in acute myelocytic leukemia through the Wnt/β-catenin pathway. Biosci. Rep. 2020, 40, BSR20193450. [Google Scholar] [CrossRef]
- Xia, Z.; Guo, M.; Liu, H.; Jiang, L.; Li, Q.; Peng, J.; Li, J.D.; Shan, B.; Feng, P.; Ma, H. CBP-dependent Wnt/β-catenin signaling is crucial in regulation of MDR1 transcription. Curr. Cancer Drug Targets 2015, 15, 519–532. [Google Scholar] [CrossRef]
- Li, W.; Xue, H.; Li, Y.; Li, P.; Ma, F.; Liu, M.; Kong, S. ATDC promotes the growth and invasion of hepatocellular carcinoma cells by modulating GSK-3β/Wnt/β-catenin signalling. Clin. Exp. Pharmacol. Physiol. 2019, 46, 845–853. [Google Scholar] [CrossRef]
- Yu, X.; Yan, N.; Li, Z.; Hua, Y.; Chen, W. FGF19 sustains the high proliferative ability of keratinocytes in psoriasis through the regulation of Wnt/GSK-3β/β-catenin signalling via FGFR4. Clin. Exp. Pharmacol. Physiol. 2019, 46, 761–769. [Google Scholar] [CrossRef]
- Wang, X.; Hawkins, B.T.; Miller, D.S. Activating PKC-β1 at the blood-brain barrier reverses induction of P-glycoprotein activity by dioxin and restores drug delivery to the CNS. J. Cereb. Blood Flow Metab. 2011, 31, 1371–1375. [Google Scholar] [CrossRef]
- Bentires-Alj, M.; Barbu, V.; Fillet, M.; Chariot, A.; Relic, B.; Jacobs, N.; Gielen, J.; Merville, M.P.; Bours, V. NF-kappaB transcription factor induces drug resistance through MDR1 expression in cancer cells. Oncogene 2003, 22, 90–97. [Google Scholar] [CrossRef] [PubMed]
- McCubrey, J.A.; Steelman, L.S.; Abrams, S.L.; Lee, J.T.; Chang, F.; Bertrand, F.E.; Navolanic, P.M.; Terrian, D.M.; Franklin, R.A.; D’Assoro, A.B.; et al. Roles of the RAF/MEK/ERK and PI3K/PTEN/AKT pathways in malignant transformation and drug resistance. Adv. Enzyme Regul. 2006, 46, 249–279. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.H.; Kim, C.G.; Lim, Y.; Shin, S.Y.; Lee, Y.H. Curcumin down-regulates the multidrug-resistance mdr1b gene by inhibiting the PI3K/Akt/NFkB pathway. Cancer Lett. 2008, 259, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Morey-Holton, E.; Wronski, T.J. Animal models for simulating weightlessness. Physiologist 1982, 24, 45–48. [Google Scholar]
- Scanziani, E. Immunohistochemical staining of fixed tissues. Methods Mol. Biol. 1998, 104, 133–140. [Google Scholar]
- Ko, Y.J.; Zaharias, R.S.; Seabold, D.A.; Lafoon, J.; Schneider, G.B. Osteoblast differentiation is enhanced in rotary cell culture simulated microgravity environments. J. Prosthodont. 2007, 16, 431–438. [Google Scholar] [CrossRef]
- Kararli, T.T. Comparison of the gastrointestinal anatomy, physiology, and biochemistry of humans and commonly used laboratory animals. Biopharm. Drug Dispos. 1995, 16, 351–380. [Google Scholar] [CrossRef]



| Concentration (mg/mL) | Ka (10−2, min−1) | Peff (10−3, cm/min) | ||
|---|---|---|---|---|
| 7 days | CON | 1.684 ± 0.015 | 7.75 ± 0.11 | 8.86 ± 0.16 |
| CON + Ver | 1.516 ± 0.052 * | 8.93 ± 0.37 * | 10.77 ± 0.61 * | |
| SMG | 1.571 ± 0.029 ** | 8.55 ± 0.21 ** | 10.11 ± 0.33 ** | |
| 21 days | CON | 1.965 ± 0.033 | 5.66 ± 0.19 | 6.01 ± 0.23 |
| CON + Ver | 1.727 ± 0.025 ** | 7.44 ± 0.18 ** | 8.41 ± 0.25 ** | |
| SMG | 1.54 ± 0.066 ** | 8.76 ± 0.47 ** | 10.47 ± 0.76 ** | |
| Gene | Primer | Sequences |
|---|---|---|
| Rat mdr1a | Forward | 5′-GGTTCGGTGCCTACTTGGTG-3′ |
| Reverse | 5′-GATGTGGGATGCTGAGACTTTG-3′ | |
| Rat mdr1b | Forward | 5′-GAAATAATGCTTATGAATCCCAAA-3′ |
| Reverse | 5′-GGTTTCATGGTCGTCGTCTCTTGA-3′ | |
| Rat GAPDH | Forward | 5′-TCTCTTGTGACAAAGTGGACAT-3′ |
| Reverse | 5′-GGTGATGGGTTTCCCGTTGA-3′ | |
| Human MDR1 | Forward | 5′-TTGCTGCTTACATTCAGGTTTCA-3′ |
| Reverse | 5′-AGCCTATCTCCTGTCGCATTA-3′ | |
| Human GAPDH | Forward | 5′-ACAACTTTGGTATCGTGGAAGG-3′ |
| Reverse | 5′-GCCATCACGCCACAGTTTC-3′ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, H.; Liang, M.; Deng, Y.; Li, Y. Simulated Microgravity Alters P-Glycoprotein Efflux Function and Expression via the Wnt/β-Catenin Signaling Pathway in Rat Intestine and Brain. Int. J. Mol. Sci. 2023, 24, 5438. https://doi.org/10.3390/ijms24065438
Liu H, Liang M, Deng Y, Li Y. Simulated Microgravity Alters P-Glycoprotein Efflux Function and Expression via the Wnt/β-Catenin Signaling Pathway in Rat Intestine and Brain. International Journal of Molecular Sciences. 2023; 24(6):5438. https://doi.org/10.3390/ijms24065438
Chicago/Turabian StyleLiu, Huayan, Min Liang, Yulin Deng, and Yujuan Li. 2023. "Simulated Microgravity Alters P-Glycoprotein Efflux Function and Expression via the Wnt/β-Catenin Signaling Pathway in Rat Intestine and Brain" International Journal of Molecular Sciences 24, no. 6: 5438. https://doi.org/10.3390/ijms24065438
APA StyleLiu, H., Liang, M., Deng, Y., & Li, Y. (2023). Simulated Microgravity Alters P-Glycoprotein Efflux Function and Expression via the Wnt/β-Catenin Signaling Pathway in Rat Intestine and Brain. International Journal of Molecular Sciences, 24(6), 5438. https://doi.org/10.3390/ijms24065438






