The Mac Is Back: The Role of Macrophages in Human Healthy and Complicated Pregnancies
Abstract
1. General Introduction: Development, the Maternal Immune System, and Immune Tolerance
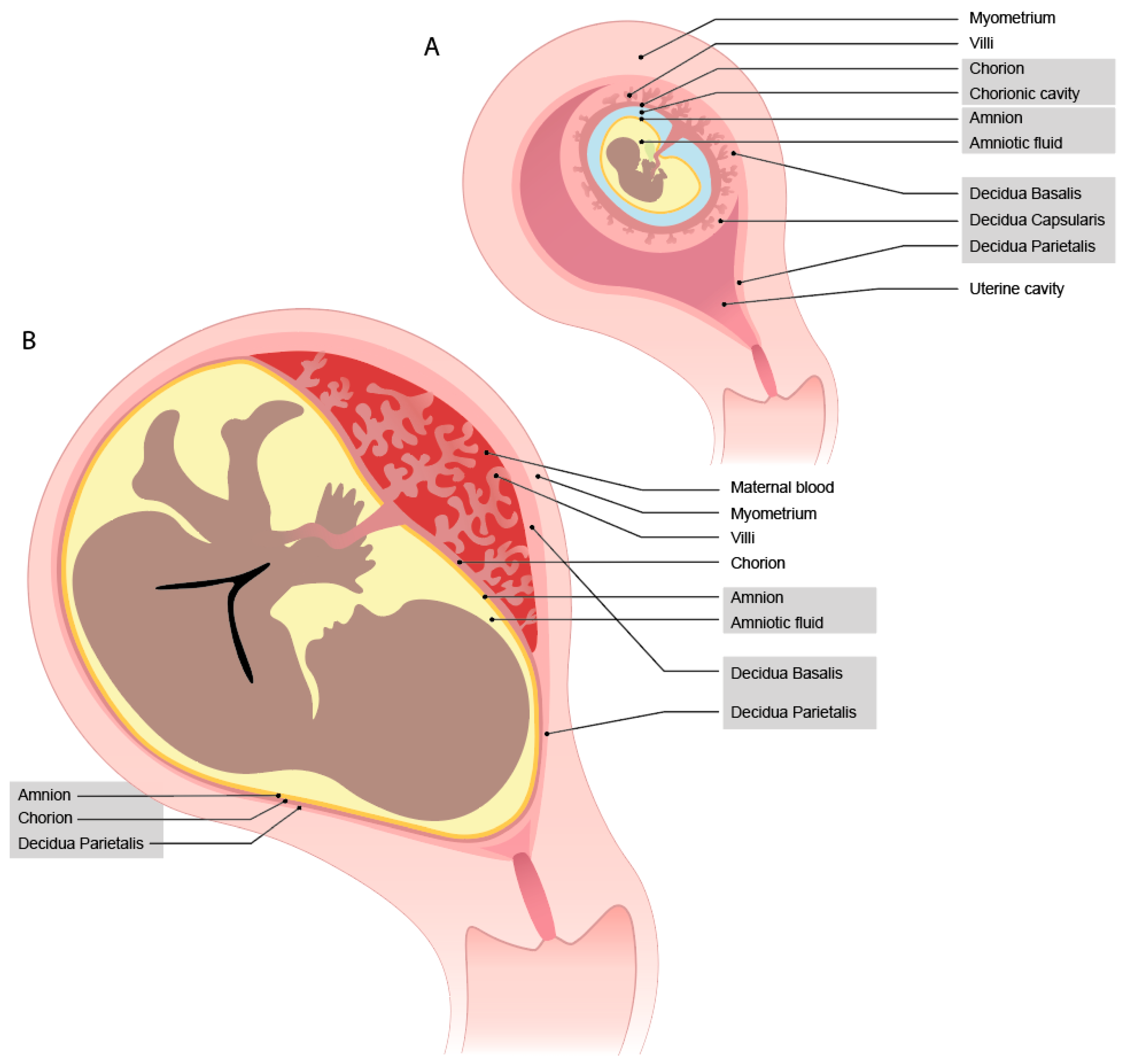
1.1. Development and Trophoblast Invasion
1.2. Interaction Sites and the Concept of Maternal-Fetal Immune Tolerance
1.3. The Immune System at the Maternal-Fetal Interface
1.4. Immune Evasion of Trophoblasts
2. The General Outlook of Macrophages in Pregnancy
3. View on Macrophages at Early Pregnancy
3.1. Immune Cell Frequencies
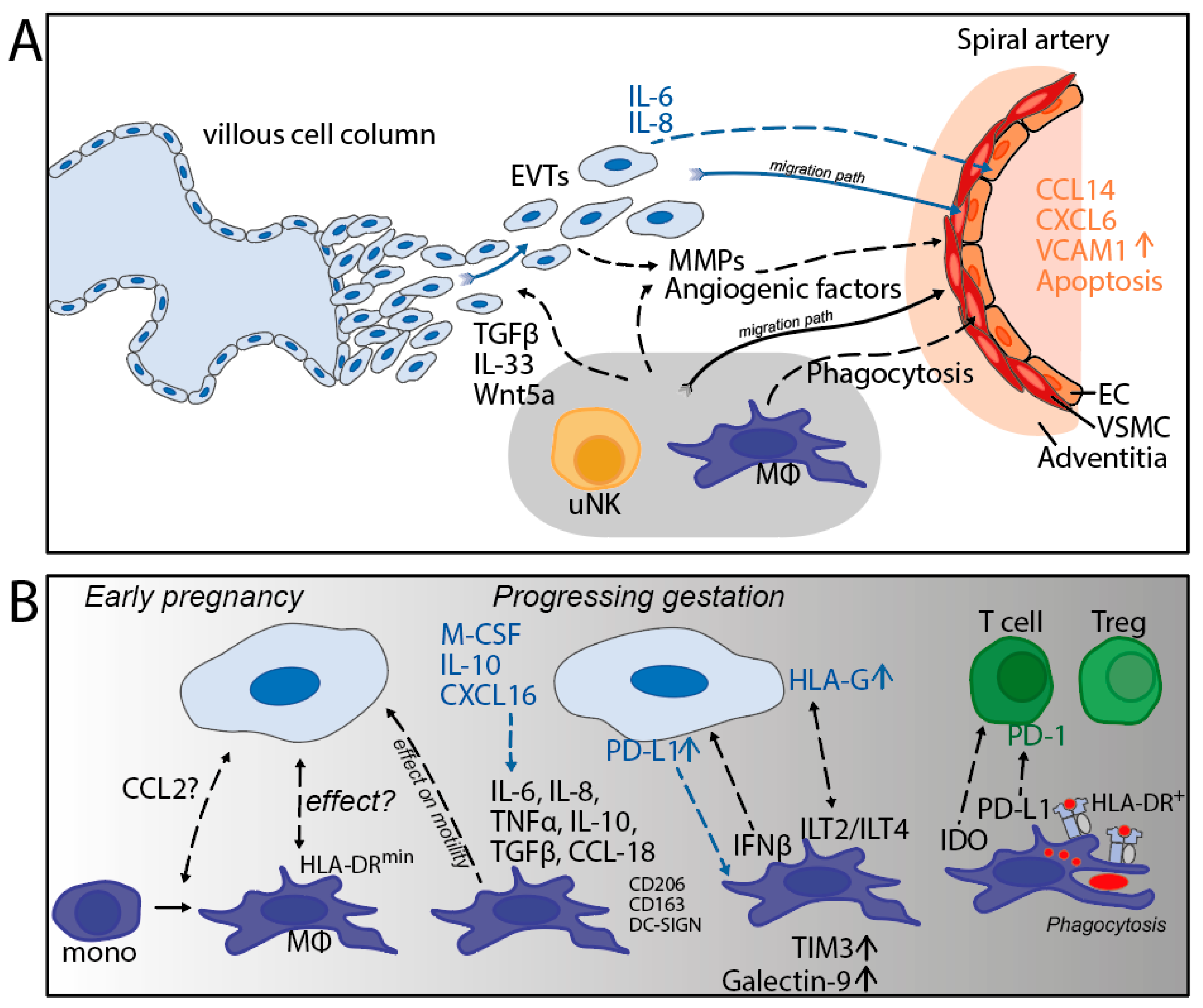
3.2. Spiral Artery Remodeling
3.3. Macrophage Subtypes and Interaction with Trophoblasts
4. View on Macrophages during Late Pregnancy
4.1. Immune Regulatory Molecules
4.2. HLA-DR and Cell-Cell Interactions
4.3. HLA Incompatibility, Immune Parameters and Pregnancy Outcomes
5. Macrophages in Complicated Pregnancies
5.1. Women with Unexplained Recurrent Pregnancy Loss
5.2. Miscarriage Related to Chronic Histiocytic Intervillositis
5.3. Insight on Macrophage Subsets in uRPL from Single-Cell Studies
6. Summary
Author Contributions
Funding
Conflicts of Interest
References
- Turco, M.Y.; Moffett, A. Development of the human placenta. Development 2019, 146, dev163428. [Google Scholar] [CrossRef] [PubMed]
- Hertig, A.T.; Rock, J.; Adams, E.C. A description of 34 human ova within the first 17 days of development. Am. J. Anat. 1956, 98, 435–493. [Google Scholar] [CrossRef] [PubMed]
- Bischof, P.; Irminger-Finger, I. The human cytotrophoblastic cell, a mononuclear chameleon. Int. J. Biochem. Cell Biol. 2005, 37, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Burton, G.J.; Jauniaux, E.; Watson, A.L. Maternal arterial connections to the placental intervillous space during the first trimester of human pregnancy: The Boyd Collection revisited. Am. J. Obstet. Gynecol. 1999, 181, 718–724. [Google Scholar] [CrossRef]
- King, A. Uterine leukocytes and decidualization. Hum. Reprod. Update 2000, 6, 28–36. [Google Scholar] [CrossRef]
- Schlafke, S.; Enders, A.C. Cellular Basis of Interaction Between Trophoblast and Uterus at Implantation. Biol. Reprod. 1975, 12, 41–65. [Google Scholar] [CrossRef]
- van der Zwan, A.; van Unen, V.; Beyrend, G.; Laban, S.; van der Keur, C.; Kapsenberg, H.J.M.; Hollt, T.; Chuva de Sousa Lopes, S.M.; van der Hoorn, M.P.; Koning, F.; et al. Visualizing Dynamic Changes at the Maternal-Fetal Interface Throughout Human Pregnancy by Mass Cytometry. Front. Immunol. 2020, 11, 571300. [Google Scholar] [CrossRef]
- van der Zwan, A.; Bi, K.; Norwitz, E.R.; Crespo, A.C.; Claas, F.H.J.; Strominger, J.L.; Tilburgs, T. Mixed signature of activation and dysfunction allows human decidual CD8(+) T cells to provide both tolerance and immunity. Proc. Natl. Acad. Sci. USA 2018, 115, 385–390. [Google Scholar] [CrossRef]
- Krop, J.; Heidt, S.; Claas, F.H.J.; Eikmans, M. Regulatory T Cells in Pregnancy: It Is Not All About FoxP3. Front. Immunol. 2020, 11, 1182. [Google Scholar] [CrossRef]
- Aluvihare, V.R.; Kallikourdis, M.; Betz, A.G. Regulatory T cells mediate maternal tolerance to the fetus. Nat. Immunol. 2004, 5, 266–271. [Google Scholar] [CrossRef]
- Zenclussen, A.C.; Gerlof, K.; Zenclussen, M.L.; Sollwedel, A.; Bertoja, A.Z.; Ritter, T.; Kotsch, K.; Leber, J.; Volk, H.D. Abnormal T-cell reactivity against paternal antigens in spontaneous abortion: Adoptive transfer of pregnancy-induced CD4+CD25+ T regulatory cells prevents fetal rejection in a murine abortion model. Am. J. Pathol. 2005, 166, 811–822. [Google Scholar] [CrossRef] [PubMed]
- Samstein, R.M.; Josefowicz, S.Z.; Arvey, A.; Treuting, P.M.; Rudensky, A.Y. Extrathymic generation of regulatory T cells in placental mammals mitigates maternal-fetal conflict. Cell 2012, 150, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Taams, L.S.; van Amelsfort, J.M.; Tiemessen, M.M.; Jacobs, K.M.; de Jong, E.C.; Akbar, A.N.; Bijlsma, J.W.; Lafeber, F.P. Modulation of monocyte/macrophage function by human CD4+CD25+ regulatory T cells. Hum. Immunol. 2005, 66, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Tiemessen, M.M.; Jagger, A.L.; Evans, H.G.; van Herwijnen, M.J.; John, S.; Taams, L.S. CD4+CD25+Foxp3+ regulatory T cells induce alternative activation of human monocytes/macrophages. Proc. Natl. Acad. Sci. USA 2007, 104, 19446–19451. [Google Scholar] [CrossRef]
- Ziegler-Heitbrock, L.; Ancuta, P.; Crowe, S.; Dalod, M.; Grau, V.; Hart, D.N.; Leenen, P.J.M.; Liu, Y.-J.; MacPherson, G.; Randolph, G.J.; et al. Nomenclature of monocytes and dendritic cells in blood. Blood 2010, 116, e74–e80. [Google Scholar] [CrossRef]
- Gardner, L.; Moffett, A. Dendritic Cells in the Human Decidua1. Biol. Reprod. 2003, 69, 1438–1446. [Google Scholar] [CrossRef]
- Kunisch, E.; Fuhrmann, R.; Roth, A.; Winter, R.; Lungershausen, W.; Kinne, R.W. Macrophage specificity of three anti-CD68 monoclonal antibodies (KP1, EBM11, and PGM1) widely used for immunohistochemistry and flow cytometry. Ann. Rheum. Dis. 2004, 63, 774–784. [Google Scholar] [CrossRef]
- Ding, J.; Zhang, Y.; Cai, X.; Diao, L.; Yang, C.; Yang, J. Crosstalk Between Trophoblast and Macrophage at the Maternal-Fetal Interface: Current Status and Future Perspectives. Front. Immunol. 2021, 12, 758281. [Google Scholar] [CrossRef]
- Svensson, J.; Jenmalm, M.C.; Matussek, A.; Geffers, R.; Berg, G.; Ernerudh, J. Macrophages at the fetal-maternal interface express markers of alternative activation and are induced by M-CSF and IL-10. J. Immunol. 2011, 187, 3671–3682. [Google Scholar] [CrossRef]
- Svensson-Arvelund, J.; Mehta, R.B.; Lindau, R.; Mirrasekhian, E.; Rodriguez-Martinez, H.; Berg, G.; Lash, G.E.; Jenmalm, M.C.; Ernerudh, J. The human fetal placenta promotes tolerance against the semiallogeneic fetus by inducing regulatory T cells and homeostatic M2 macrophages. J. Immunol. 2015, 194, 1534–1544. [Google Scholar] [CrossRef]
- Brown, M.B.; von Chamier, M.; Allam, A.B.; Reyes, L. M1/M2 macrophage polarity in normal and complicated pregnancy. Front. Immunol. 2014, 5, 606. [Google Scholar] [CrossRef] [PubMed]
- Nagamatsu, T.; Schust, D.J. The contribution of macrophages to normal and pathological pregnancies. Am. J. Reprod. Immunol. 2010, 63, 460–471. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.H.; He, M.; Wang, Y.; Liao, A.H. Modulators of the Balance between M1 and M2 Macrophages during Pregnancy. Front. Immunol. 2017, 8, 120. [Google Scholar] [CrossRef] [PubMed]
- Renaud, S.J.; Graham, C.H. The role of macrophages in utero-placental interactions during normal and pathological pregnancy. Immunol. Investig. 2008, 37, 535–564. [Google Scholar] [CrossRef] [PubMed]
- Ning, F.; Liu, H.; Lash, G.E. The Role of Decidual Macrophages During Normal and Pathological Pregnancy. Am. J. Reprod. Immunol. 2016, 75, 298–309. [Google Scholar] [CrossRef]
- Tang, M.X.; Hu, X.H.; Liu, Z.Z.; Kwak-Kim, J.; Liao, A.H. What are the roles of macrophages and monocytes in human pregnancy? J. Reprod. Immunol. 2015, 112, 73–80. [Google Scholar] [CrossRef]
- Faas, M.M.; de Vos, P. Uterine NK cells and macrophages in pregnancy. Placenta 2017, 56, 44–52. [Google Scholar] [CrossRef]
- Faas, M.M.; Spaans, F.; De Vos, P. Monocytes and macrophages in pregnancy and pre-eclampsia. Front. Immunol. 2014, 5, 298. [Google Scholar] [CrossRef]
- Parasar, P.; Guru, N.; Nayak, N.R. Contribution of macrophages to fetomaternal immunological tolerance. Hum. Immunol. 2021, 82, 325–331. [Google Scholar] [CrossRef]
- Sun, F.; Wang, S.; Du, M. Functional regulation of decidual macrophages during pregnancy. J. Reprod. Immunol. 2021, 143, 103264. [Google Scholar] [CrossRef]
- Sayama, S.; Nagamatsu, T.; Schust, D.J.; Itaoka, N.; Ichikawa, M.; Kawana, K.; Yamashita, T.; Kozuma, S.; Fujii, T. Human decidual macrophages suppress IFN-gamma production by T cells through costimulatory B7-H1:PD-1 signaling in early pregnancy. J. Reprod. Immunol. 2013, 100, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Q.; Zhou, W.J.; Hou, X.X.; Fu, Q.; Li, D.J. Trophoblast-derived CXCL16 induces M2 macrophage polarization that in turn inactivates NK cells at the maternal-fetal interface. Cell. Mol. Immunol. 2018, 15, 1038–1046. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Aldo, P.; You, Y.; Ding, J.; Kaislasuo, J.; Petersen, J.F.; Lokkegaard, E.; Peng, G.; Paidas, M.J.; Simpson, S.; et al. Trophoblast-secreted soluble-PD-L1 modulates macrophage polarization and function. J. Leucoc. Biol. 2020, 108, 983–998. [Google Scholar] [CrossRef]
- Eikmans, M.; van der Keur, C.; Anholts, J.D.H.; Drabbels, J.J.M.; van Beelen, E.; de Sousa Lopes, S.M.C.; van der Hoorn, M.L. Primary Trophoblast Cultures: Characterization of HLA Profiles and Immune Cell Interactions. Front. Immunol. 2022, 13, 814019. [Google Scholar] [CrossRef] [PubMed]
- Shakhawat, A.; Shaikly, V.; Elzatma, E.; Mavrakos, E.; Jabeen, A.; Fernandez, N. Interaction between HLA-G and monocyte/macrophages in human pregnancy. J. Reprod. Immunol. 2010, 85, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.L.; Guo, Y.; So, K.H.; Vijayan, M.; Guo, Y.; Wong, V.H.; Yao, Y.; Lee, K.F.; Chiu, P.C.; Yeung, W.S. Soluble human leukocyte antigen G5 polarizes differentiation of macrophages toward a decidual macrophage-like phenotype. Hum. Reprod. 2015, 30, 2263–2274. [Google Scholar] [CrossRef] [PubMed]
- Petroff, M.G.; Sedlmayr, P.; Azzola, D.; Hunt, J.S. Decidual macrophages are potentially susceptible to inhibition by class Ia and class Ib HLA molecules. J. Reprod. Immunol. 2002, 56, 3–17. [Google Scholar] [CrossRef]
- Abumaree, M.H.; Chamley, L.W.; Badri, M.; El-Muzaini, M.F. Trophoblast debris modulates the expression of immune proteins in macrophages: A key to maternal tolerance of the fetal allograft? J. Reprod. Immunol. 2012, 94, 131–141. [Google Scholar] [CrossRef]
- Vondra, S.; Hobler, A.L.; Lackner, A.I.; Raffetseder, J.; Mihalic, Z.N.; Vogel, A.; Saleh, L.; Kunihs, V.; Haslinger, P.; Wahrmann, M.; et al. The human placenta shapes the phenotype of decidual macrophages. Cell Rep. 2023, 42, 111977. [Google Scholar] [CrossRef]
- Krop, J.; van der Zwan, A.; Ijsselsteijn, M.E.; Kapsenberg, H.; Luk, S.J.; Hendriks, S.H.; van der Keur, C.; Verleng, L.J.; Somarakis, A.; van der Meeren, L.; et al. Imaging mass cytometry reveals the prominent role of myeloid cells at the maternal-fetal interface. iScience 2022, 25, 104648. [Google Scholar] [CrossRef]
- Guo, C.; Cai, P.; Jin, L.; Sha, Q.; Yu, Q.; Zhang, W.; Jiang, C.; Liu, Q.; Zong, D.; Li, K.; et al. Single-cell profiling of the human decidual immune microenvironment in patients with recurrent pregnancy loss. Cell Discov. 2021, 7, 1. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Wang, M.; Liang, G.; Jin, P.; Wang, P.; Xu, Y.; Qian, Y.; Jiang, X.; Qian, J.; Dong, M. Pro-Inflammatory Signature in Decidua of Recurrent Pregnancy Loss Regardless of Embryonic Chromosomal Abnormalities. Front. Immunol. 2021, 12, 772729. [Google Scholar] [CrossRef]
- Greenbaum, S.; Averbukh, I.; Soon, E.; Rizzuto, G.; Baranski, A.; Greenwald, N.; Bosse, M.; Jaswa, E.G.; Khair, Z.; Kwok, S.; et al. Spatio-temporal coordination at the maternal-fetal interface promotes trophoblast invasion and vascular remodeling in the first half of human pregnancy. bioRxiv 2021. [Google Scholar] [CrossRef]
- Harris, L.K.; Benagiano, M.; D’Elios, M.M.; Brosens, I.; Benagiano, G. Placental bed research: II. Functional and immunological investigations of the placental bed. Am. J. Obstet. Gynecol. 2019, 221, 457–469. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.D.; Dunk, C.E.; Aplin, J.D.; Harris, L.K.; Jones, R.L. Evidence for immune cell involvement in decidual spiral arteriole remodeling in early human pregnancy. Am. J. Pathol. 2009, 174, 1959–1971. [Google Scholar] [CrossRef]
- Lash, G.E.; Pitman, H.; Morgan, H.L.; Innes, B.A.; Agwu, C.N.; Bulmer, J.N. Decidual macrophages: Key regulators of vascular remodeling in human pregnancy. J. Leukoc. Biol. 2016, 100, 315–325. [Google Scholar] [CrossRef]
- Choudhury, R.H.; Dunk, C.E.; Lye, S.J.; Aplin, J.D.; Harris, L.K.; Jones, R.L. Extravillous Trophoblast and Endothelial Cell Crosstalk Mediates Leukocyte Infiltration to the Early Remodeling Decidual Spiral Arteriole Wall. J. Immunol. 2017, 198, 4115–4128. [Google Scholar] [CrossRef]
- Choudhury, R.H.; Dunk, C.E.; Lye, S.J.; Harris, L.K.; Aplin, J.D.; Jones, R.L. Decidual leucocytes infiltrating human spiral arterioles are rich source of matrix metalloproteinases and degrade extracellular matrix in vitro and in situ. Am. J. Reprod. Immunol. 2019, 81, e13054. [Google Scholar] [CrossRef]
- Robson, A.; Harris, L.K.; Innes, B.A.; Lash, G.E.; Aljunaidy, M.M.; Aplin, J.D.; Baker, P.N.; Robson, S.C.; Bulmer, J.N. Uterine natural killer cells initiate spiral artery remodeling in human pregnancy. FASEB J. 2012, 26, 4876–4885. [Google Scholar] [CrossRef]
- Iqbal, S.; Kumar, A. Characterization of in vitro generated human polarized macrophages. J. Clin. Cell. Immunol. 2015, 6, 380. [Google Scholar] [CrossRef]
- Abrahams, V.M.; Kim, Y.M.; Straszewski, S.L.; Romero, R.; Mor, G. Macrophages and apoptotic cell clearance during pregnancy. Am. J. Reprod. Immunol. 2004, 51, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Fock, V.; Mairhofer, M.; Otti, G.R.; Hiden, U.; Spittler, A.; Zeisler, H.; Fiala, C.; Knofler, M.; Pollheimer, J. Macrophage-derived IL-33 is a critical factor for placental growth. J. Immunol. 2013, 191, 3734–3743. [Google Scholar] [CrossRef] [PubMed]
- Meinhardt, G.; Saleh, L.; Otti, G.R.; Haider, S.; Velicky, P.; Fiala, C.; Pollheimer, J.; Knofler, M. Wingless ligand 5a is a critical regulator of placental growth and survival. Sci. Rep. 2016, 6, 28127. [Google Scholar] [CrossRef] [PubMed]
- Renaud, S.J.; Macdonald-Goodfellow, S.K.; Graham, C.H. Coordinated regulation of human trophoblast invasiveness by macrophages and interleukin 10. Biol. Reprod. 2007, 76, 448–454. [Google Scholar] [CrossRef]
- Renaud, S.J.; Postovit, L.M.; Macdonald-Goodfellow, S.K.; McDonald, G.T.; Caldwell, J.D.; Graham, C.H. Activated macrophages inhibit human cytotrophoblast invasiveness in vitro. Biol. Reprod. 2005, 73, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Mani, S.; Clair, G.; Olson, H.M.; Paurus, V.L.; Ansong, C.K.; Blundell, C.; Young, R.; Kanter, J.; Gordon, S.; et al. A microphysiological model of human trophoblast invasion during implantation. Nat. Commun. 2022, 13, 1252. [Google Scholar] [CrossRef]
- Gustafsson, C.; Mjosberg, J.; Matussek, A.; Geffers, R.; Matthiesen, L.; Berg, G.; Sharma, S.; Buer, J.; Ernerudh, J. Gene expression profiling of human decidual macrophages: Evidence for immunosuppressive phenotype. PLoS ONE 2008, 3, e2078. [Google Scholar] [CrossRef]
- Houser, B.L.; Tilburgs, T.; Hill, J.; Nicotra, M.L.; Strominger, J.L. Two unique human decidual macrophage populations. J. Immunol. 2011, 186, 2633–2642. [Google Scholar] [CrossRef]
- Jiang, X.; Du, M.R.; Li, M.; Wang, H. Three macrophage subsets are identified in the uterus during early human pregnancy. Cell. Mol. Immunol. 2018, 15, 1027–1037. [Google Scholar] [CrossRef]
- Vento-Tormo, R.; Efremova, M.; Botting, R.A.; Turco, M.Y.; Vento-Tormo, M.; Meyer, K.B.; Park, J.E.; Stephenson, E.; Polanski, K.; Goncalves, A.; et al. Single-cell reconstruction of the early maternal-fetal interface in humans. Nature 2018, 563, 347–353. [Google Scholar] [CrossRef]
- Chen, P.; Zhou, L.; Chen, J.; Lu, Y.; Cao, C.; Lv, S.; Wei, Z.; Wang, L.; Chen, J.; Hu, X.; et al. The Immune Atlas of Human Deciduas With Unexplained Recurrent Pregnancy Loss. Front. Immunol. 2021, 12, 689019. [Google Scholar] [CrossRef]
- Zhang, Y.; Qu, D.; Sun, J.; Zhao, L.; Wang, Q.; Shao, Q.; Kong, B.; Zhang, Y.; Qu, X. Human trophoblast cells induced MDSCs from peripheral blood CD14(+) myelomonocytic cells via elevated levels of CCL2. Cell. Mol. Immunol. 2016, 13, 615–627. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.L.; Robinette, M.L.; Bugatti, M.; Longtine, M.S.; Colvin, B.N.; Lantelme, E.; Vermi, W.; Colonna, M.; Nelson, D.M.; Cella, M. Two Distinct Myeloid Subsets at the Term Human Fetal-Maternal Interface. Front. Immunol. 2017, 8, 1357. [Google Scholar] [CrossRef] [PubMed]
- Heikkinen, J.; Mottonen, M.; Komi, J.; Alanen, A.; Lassila, O. Phenotypic characterization of human decidual macrophages. Clin. Exp. Immunol. 2003, 131, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Tilburgs, T.; Scherjon, S.A.; van der Mast, B.J.; Haasnoot, G.W.; Versteeg, V.D.V.-M.M.; Roelen, D.L.; van Rood, J.J.; Claas, F.H. Fetal-maternal HLA-C mismatch is associated with decidual T cell activation and induction of functional T regulatory cells. J. Reprod. Immunol. 2009, 82, 148–157. [Google Scholar] [CrossRef] [PubMed]
- van ‘t Hof, L.J.; Schotvanger, N.; Haasnoot, G.W.; van der Keur, C.; Roelen, D.L.; Lashley, L.; Claas, F.H.J.; Eikmans, M.; van der Hoorn, M.P. Maternal-Fetal HLA Compatibility in Uncomplicated and Preeclamptic Naturally Conceived Pregnancies. Front. Immunol. 2021, 12, 673131. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Tan, J.; Martino, M.M.; Lui, K.O. Regulatory T-Cells: Potential Regulator of Tissue Repair and Regeneration. Front. Immunol. 2018, 9, 585. [Google Scholar] [CrossRef]
- Liu, W.; Xiao, X.; Demirci, G.; Madsen, J.; Li, X.C. Innate NK cells and macrophages recognize and reject allogeneic nonself in vivo via different mechanisms. J. Immunol. 2012, 188, 2703–2711. [Google Scholar] [CrossRef]
- Lin, C.M.; Gill, R.G. Direct and indirect allograft recognition: Pathways dictating graft rejection mechanisms. Curr. Opin. Organ. Transpl. 2016, 21, 40–44. [Google Scholar] [CrossRef]
- van der Hoorn, M.L.; Lashley, E.E.; Bianchi, D.W.; Claas, F.H.; Schonkeren, C.M.; Scherjon, S.A. Clinical and immunologic aspects of egg donation pregnancies: A systematic review. Hum. Reprod. Update 2010, 16, 704–712. [Google Scholar] [CrossRef]
- van der Hoorn, M.L.; Scherjon, S.A.; Claas, F.H. Egg donation pregnancy as an immunological model for solid organ transplantation. Transpl. Immunol. 2011, 25, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Eikmans, M.; Hoorn, M.V. The Role of Macrophages in Oocyte Donation Pregnancy: A Systematic Review. Int. J. Mol. Sci. 2020, 21, 939. [Google Scholar] [CrossRef] [PubMed]
- Storgaard, M.; Loft, A.; Bergh, C.; Wennerholm, U.B.; Soderstrom-Anttila, V.; Romundstad, L.B.; Aittomaki, K.; Oldereid, N.; Forman, J.; Pinborg, A. Obstetric and neonatal complications in pregnancies conceived after oocyte donation: A systematic review and meta-analysis. BJOG 2017, 124, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Tarlatzi, T.B.; Imbert, R.; Alvaro Mercadal, B.; Demeestere, I.; Venetis, C.A.; Englert, Y.; Delbaere, A. Does oocyte donation compared with autologous oocyte IVF pregnancies have a higher risk of preeclampsia? Reprod. Biomed. Online 2017, 34, 11–18. [Google Scholar] [CrossRef]
- Masoudian, P.; Nasr, A.; de Nanassy, J.; Fung-Kee-Fung, K.; Bainbridge, S.A.; El Demellawy, D. Oocyte donation pregnancies and the risk of preeclampsia or gestational hypertension: A systematic review and metaanalysis. Am. J. Obstet. Gynecol. 2016, 214, 328–339. [Google Scholar] [CrossRef]
- Mascarenhas, M.; Sunkara, S.K.; Antonisamy, B.; Kamath, M.S. Higher risk of preterm birth and low birth weight following oocyte donation: A systematic review and meta-analysis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 218, 60–67. [Google Scholar] [CrossRef]
- Rodriguez-Wallberg, K.A.; Berger, A.S.; Fagerberg, A.; Olofsson, J.I.; Scherman-Pukk, C.; Lindqvist, P.G.; Nasiell, J. Increased incidence of obstetric and perinatal complications in pregnancies achieved using donor oocytes and single embryo transfer in young and healthy women. A prospective hospital-based matched cohort study. Gynecol. Endocrinol. 2019, 35, 314–319. [Google Scholar] [CrossRef]
- van Bentem, K.; Bos, M.; van der Keur, C.; Brand-Schaaf, S.H.; Haasnoot, G.W.; Roelen, D.L.; Eikmans, M.; Heidt, S.; Claas, F.H.J.; Lashley, E.; et al. The development of preeclampsia in oocyte donation pregnancies is related to the number of fetal-maternal HLA class II mismatches. J. Reprod. Immunol. 2019, 137, 103074. [Google Scholar] [CrossRef]
- Schonkeren, D.; Swings, G.; Roberts, D.; Claas, F.; de Heer, E.; Scherjon, S. Pregnancy close to the edge: An immunosuppressive infiltrate in the chorionic plate of placentas from uncomplicated egg cell donation. PLoS ONE 2012, 7, e32347. [Google Scholar] [CrossRef]
- Burk, M.R.; Troeger, C.; Brinkhaus, R.; Holzgreve, W.; Hahn, S. Severely reduced presence of tissue macrophages in the basal plate of pre-eclamptic placentae. Placenta 2001, 22, 309–316. [Google Scholar] [CrossRef]
- van Bentem, K.; Bos, M.; van der Keur, C.; Kapsenberg, H.; Lashley, E.; Eikmans, M.; van der Hoorn, M.L. Different immunoregulatory components at the decidua basalis of oocyte donation pregnancies. Hum. Immunol. 2022, 83, 319–327. [Google Scholar] [CrossRef] [PubMed]
- van der Hoorn, M.P.; van Egmond, A.; Swings, G.; van Beelen, E.; van der Keur, C.; Tirado-Gonzalez, I.; Blois, S.M.; Karumanchi, S.A.; Bianchi, D.W.; Claas, F.H.J.; et al. Differential immunoregulation in successful oocyte donation pregnancies compared with naturally conceived pregnancies. J. Reprod. Immunol. 2014, 101–102, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Aiyer, K.T.S.; Kapsenberg, J.M.; Roelen, D.L.; van der Hoorn, M.L.; Eikmans, M. Uncomplicated oocyte donation pregnancies display an elevated CD163-positive type 2 macrophage load in the decidua, which is associated with fetal-maternal HLA mismatches. Am. J. Reprod. Immunol. 2022, 87, e13511. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Krop, J.; Eikmans, M.; van der Hoorn, M.L. Leiden University Medical Center (LUMC), Albinusdreef 2, 2333 ZA Leiden, The Netherlands. 2023; manuscript in preparation. [Google Scholar]
- Youssef, A.; Lashley, L.; Dieben, S.; Verburg, H.; van der Hoorn, M.-L. Defining recurrent pregnancy loss: Associated factors and prognosis in couples with two versus three or more pregnancy losses. Reprod. Biomed. Online 2020, 41, 679–685. [Google Scholar] [CrossRef] [PubMed]
- RPL, T.E.G.G.o.; Bender Atik, R.; Christiansen, O.B.; Elson, J.; Kolte, A.M.; Lewis, S.; Middeldorp, S.; Nelen, W.; Peramo, B.; Quenby, S.; et al. ESHRE guideline: Recurrent pregnancy loss. Hum. Reprod. Open 2018, 2018, hoy004. [Google Scholar] [CrossRef]
- Keller, C.C.; Eikmans, M.; van der Hoorn, M.-L.P.; Lashley, L.E.E.L.O. Recurrent miscarriages and the association with regulatory T cells; A systematic review. J. Reprod. Immunol. 2020, 139, 103105. [Google Scholar] [CrossRef]
- Wang, W.J.; Hao, C.F.; Lin, Q.D. Dysregulation of macrophage activation by decidual regulatory T cells in unexplained recurrent miscarriage patients. J. Reprod. Immunol. 2011, 92, 97–102. [Google Scholar] [CrossRef]
- Guenther, S.; Vrekoussis, T.; Heublein, S.; Bayer, B.; Anz, D.; Knabl, J.; Navrozoglou, I.; Dian, D.; Friese, K.; Makrigiannakis, A.; et al. Decidual Macrophages Are Significantly Increased in Spontaneous Miscarriages and Over-Express FasL: A Potential Role for Macrophages in Trophoblast Apoptosis. Int. J. Mol. Sci. 2012, 13, 9069–9080. [Google Scholar] [CrossRef]
- Krop, J.; van der Meeren, L.; van der Hoorn, M.L.; Ijsselsteijn, M.; Dijkstra, K.; Kapsenberg, H.; van der Keur, C.; Cornish; Nikkels, P.; Koning, F.; et al. Identification of a unique intervillous cellular signature in chronic histiocytic intervillositis. Leiden University Medical Center (LUMC), Albinusdreef 2, 2333 ZA Leiden, The Netherlands. 2023; manuscript submitted. [Google Scholar]
- Hussein, K.; Stucki-Koch, A.; Müller, A.M.; Arnold, R.; Kreipe, H.; Feist, H. Complement receptor-associated CD163(+)/CD18(+)/CD11c(+)/CD206(-)/CD209(-) expression profile in chronic histiocytic intervillositis of the placenta. Placenta 2019, 78, 23–28. [Google Scholar] [CrossRef]
- Tsao, F.Y.; Wu, M.Y.; Chang, Y.L.; Wu, C.T.; Ho, H.N. M1 macrophages decrease in the deciduae from normal pregnancies but not from spontaneous abortions or unexplained recurrent spontaneous abortions. J. Formos. Med. Assoc. 2018, 117, 204–211. [Google Scholar] [CrossRef]
- Wang, W.; Vilella, F.; Alama, P.; Moreno, I.; Mignardi, M.; Isakova, A.; Pan, W.; Simon, C.; Quake, S.R. Single-cell transcriptomic atlas of the human endometrium during the menstrual cycle. Nat. Med. 2020, 26, 1644–1653. [Google Scholar] [CrossRef] [PubMed]
- Sang, Y.; Li, Y.; Xu, L.; Chen, J.; Li, D.; Du, M. Dysfunction of CCR1(+) decidual macrophages is a potential risk factor in the occurrence of unexplained recurrent pregnancy loss. Front. Immunol. 2022, 13, 1045532. [Google Scholar] [CrossRef] [PubMed]


| Gestation | Main Subtypes | Resident Marker | Other Markers | Possible Function | Reference |
|---|---|---|---|---|---|
| 6–8 week | CD11chiCCR2+ (14%) | TNF pathway; IL1-β; COX2 | phagocytosis; inflammatory-related | [59] | |
| CD11chiCRR2− (6%) | HMOX1 | anti-inflammatory; anti-oxidative | |||
| CD11cloCCR2− (80%) | CD209hi | DC-SIGNhi; HLA-DRhi | |||
| 6–12 week | CD11chi (20%) | CD206lo; CD209lo | HLA-DR; TNFα; IL-6, IL10 | inflammatory processes; lipid metabolism | [58] |
| CD11clo (69%) | CD206hi; CD209hi | HLA-DR; TGFβ | ECM formation; growth regulation | ||
| 6–12 week | CD11chi | IL-10hi; TNF, IL-1, IL-6, VEGF-A | remodeling | [60] | |
| CD11clo | CCL-2, CCL-8, IGF-1, HGF, PDGF | self-renewal; remodeling | |||
| 10–11 week | DC-SIGN/CD209+ | CD163+; CD204+ | M1 (28%): CD11blo; CD11c−; HLA-DR− | close to trophoblasts | [43] |
| M2 (4%): CD11b+; CD11c−; HLA-DR+ | |||||
| M3 (8%): CD11blo; CD11clo; HLA-DR+ | |||||
| DC-SIGN/CD209− | CD163+; CD204+ | M4 (52%): CD11b−; CD11c−; HLA-DR− | close to trophoblasts | ||
| M5 (5%): CD11blo; CD11c−; HLA-DR+ | |||||
| 6–20 week | DC-SIGN/CD209+ | CD163+; CD206+ | M2a (64%): CD11c−; HLA-DR+ | upregulate TIM-3 and GAL-9 with increased GA | [40] |
| M2b (2.7%): CD11c+; HLA-DR+ | upregulate TIM-3 and GAL-9 with increased GA | ||||
| M2c (10.3%): CD11c−; HLA-DR− | |||||
| DC-SIGN/CD209− | CD163+; CD206+ | M1a (19%): CD11c−; HLA-DR+ | |||
| M1b (4%): CD11c−; HLA-DR+ |
| Studied Cell Types | Main Findings | Reference |
|---|---|---|
| M1 macrophages: CD68+IL-10lowiNOS+ M2 macrophages: CD68+IL10+iNOSlow | M1 numbers decrease in healthy pregnancy, but stay the same in uRPL during first trimester M2 numbers increase in healthy pregnancy, but decrease in uRPL during first trimester | [92] |
| CCR1+ macrophages: CD163+CD206+ IL-10+ TGF-β+ CD80low CD86low | CCR1+ macrophages enriched in healthy 1st trimester decidua, but not in uRPL CCR1+ macrophages during uRPL have an attenuated immunosuppressive phenotype | [94] |
| Mac1: M1 polarization characteristics Mac2: enriched with M2 specific genes | Mac1 proportion modestly elevated and mac2 proportion highly decreased in uRPL Mac2 interacts with NK cells during healthy pregnancy, but switches toward interaction with T cells in uRPL | [41] |
| 10 CD14+ monocyte/macrophages clus-ters | cluster 6 macrophages with both M1- (CD163, IL-10) and M2-features (CCL2,-3,-4) enriched in uRPL | [61] |
| M1: CD68+CD11b+CD206lowCD11c+ M2: CD68+CD11bmedCD206+CD11cmed | M1 proportion two-fold higher and M2 proportion slightly decreased in uRPL CD11chighCCR2− macrophage proportions increased and CD11clowCCR2− macrophages decreased in uRPL | [42] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krop, J.; Tian, X.; van der Hoorn, M.-L.; Eikmans, M. The Mac Is Back: The Role of Macrophages in Human Healthy and Complicated Pregnancies. Int. J. Mol. Sci. 2023, 24, 5300. https://doi.org/10.3390/ijms24065300
Krop J, Tian X, van der Hoorn M-L, Eikmans M. The Mac Is Back: The Role of Macrophages in Human Healthy and Complicated Pregnancies. International Journal of Molecular Sciences. 2023; 24(6):5300. https://doi.org/10.3390/ijms24065300
Chicago/Turabian StyleKrop, Juliette, Xuezi Tian, Marie-Louise van der Hoorn, and Michael Eikmans. 2023. "The Mac Is Back: The Role of Macrophages in Human Healthy and Complicated Pregnancies" International Journal of Molecular Sciences 24, no. 6: 5300. https://doi.org/10.3390/ijms24065300
APA StyleKrop, J., Tian, X., van der Hoorn, M.-L., & Eikmans, M. (2023). The Mac Is Back: The Role of Macrophages in Human Healthy and Complicated Pregnancies. International Journal of Molecular Sciences, 24(6), 5300. https://doi.org/10.3390/ijms24065300







