The Potential Chemopreventive Effect of Andrographis paniculata on 1,2-Dimethylhydrazine and High-Fat-Diet-Induced Colorectal Cancer in Sprague Dawley Rats
Abstract
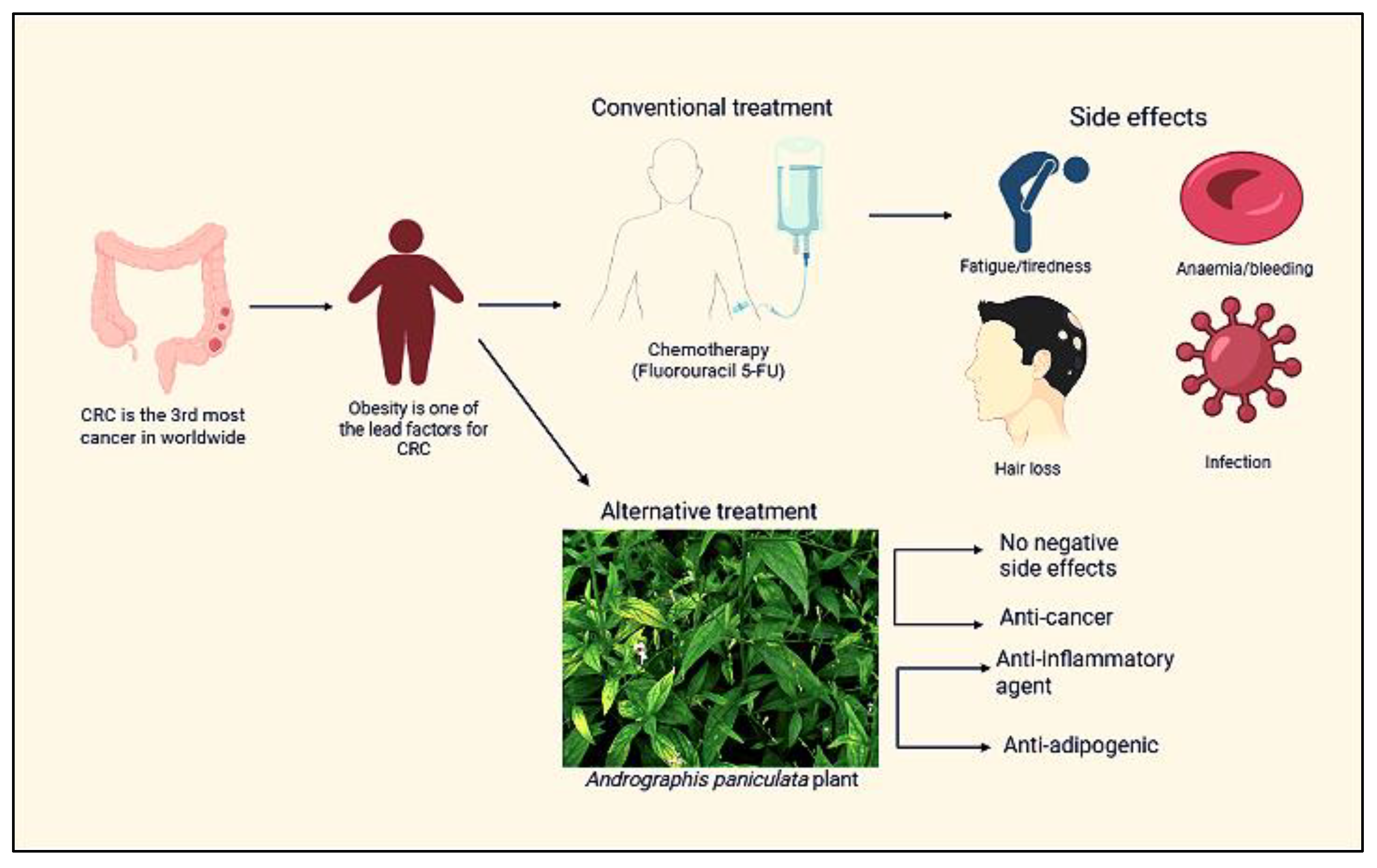
1. Introduction
2. Results
2.1. Phytochemical Screening and Identification of A. paniculata Ethanol Extract
2.2. Impact of A. paniculata on Food Intake, Body Weight, Retroperitoneal White Adipose Tissue (RpWAT), Colon Weight, and Colon Length
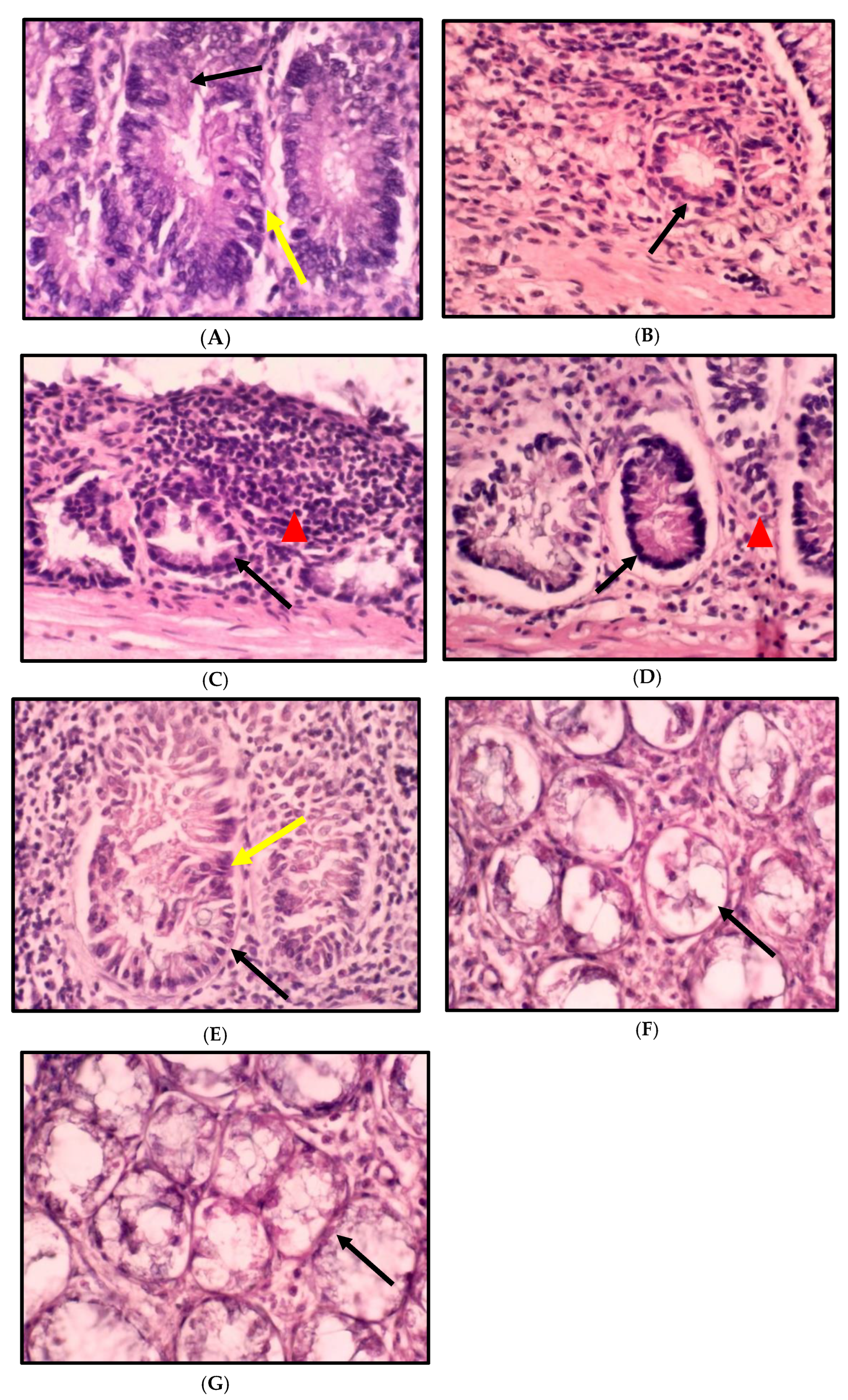
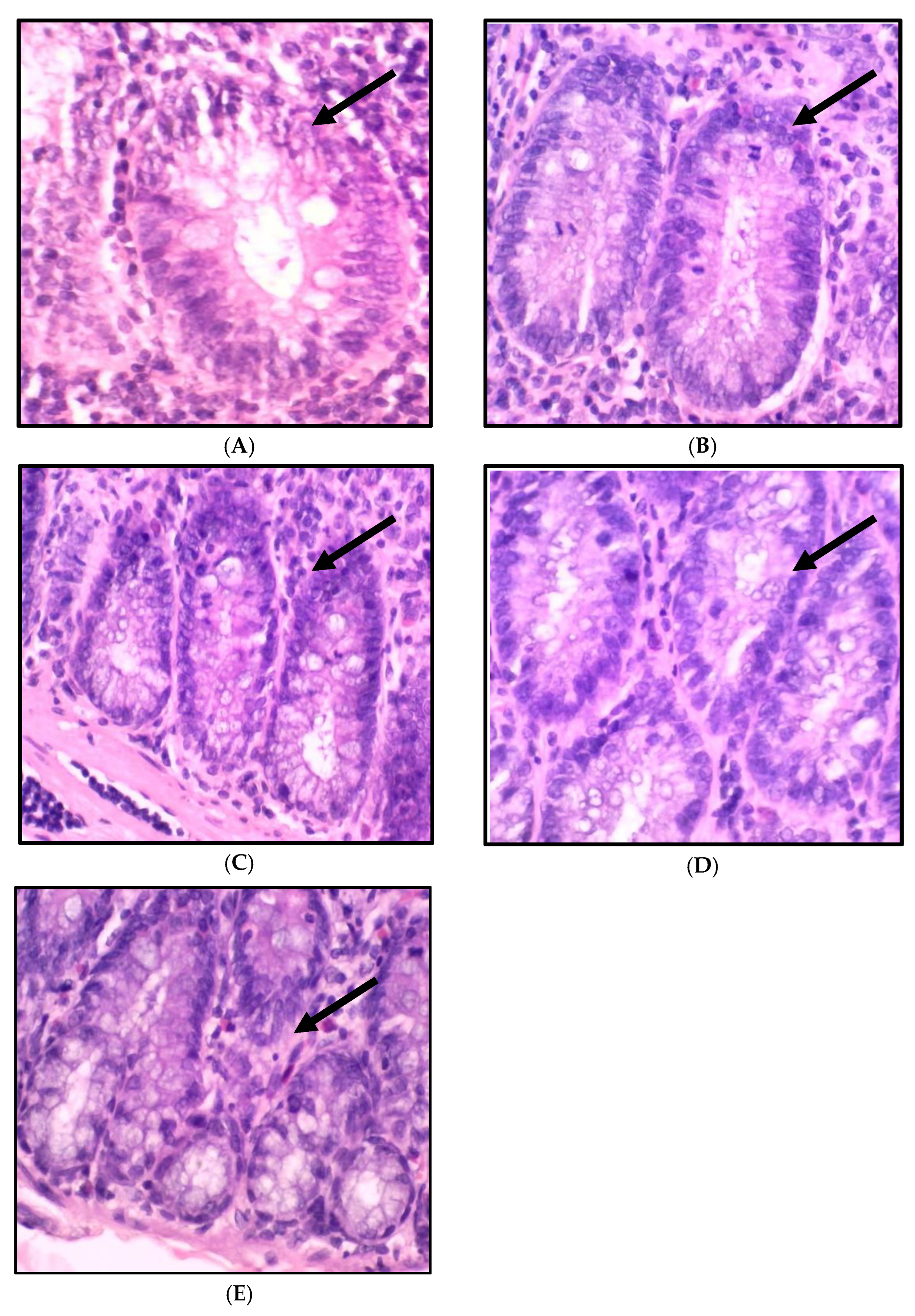
2.3. Impact of A. paniculata on the Histopathological Finding of the Colon in Treated Rats
2.4. Impact of A. paniculata on the Overall Number of ACF in the Colon
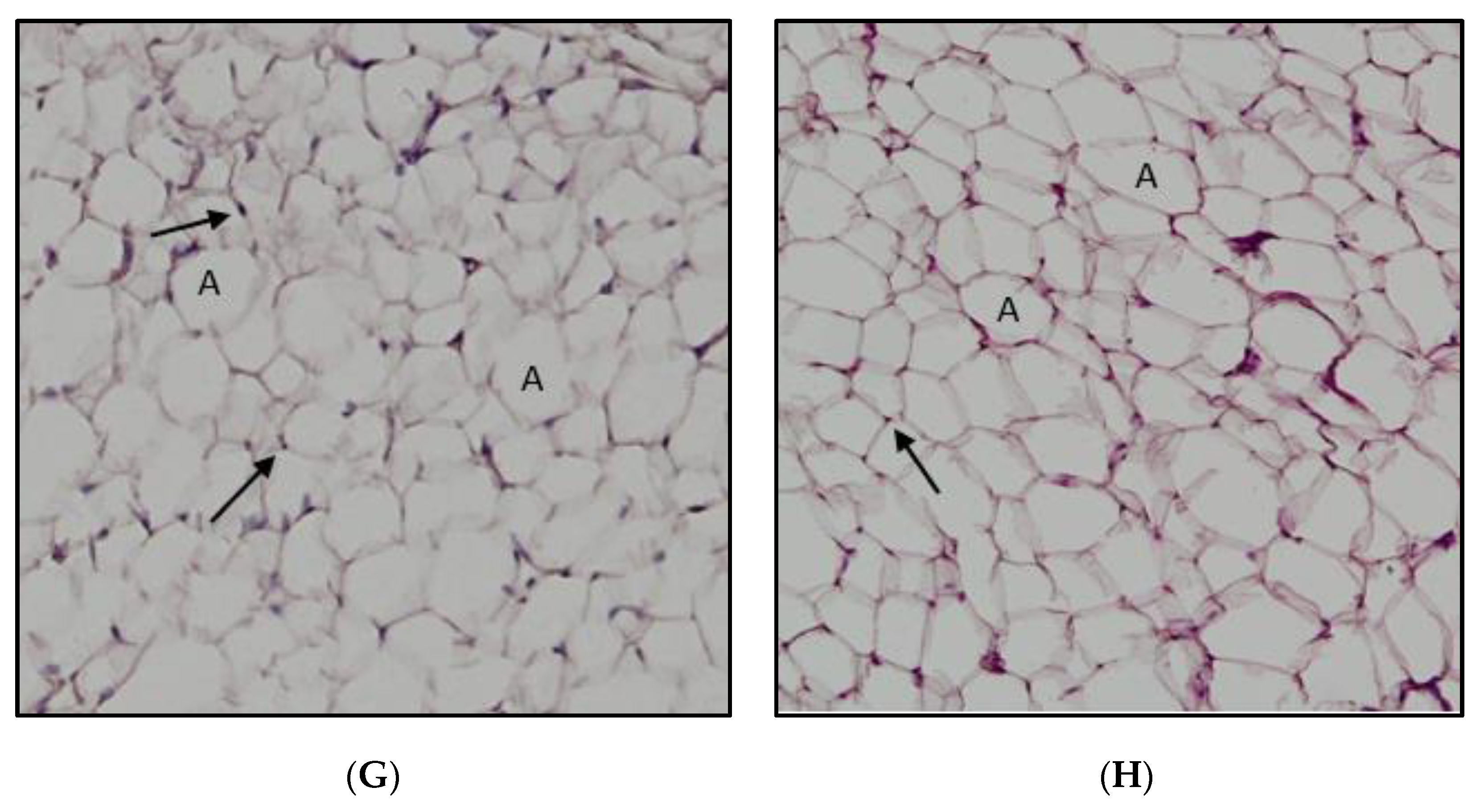
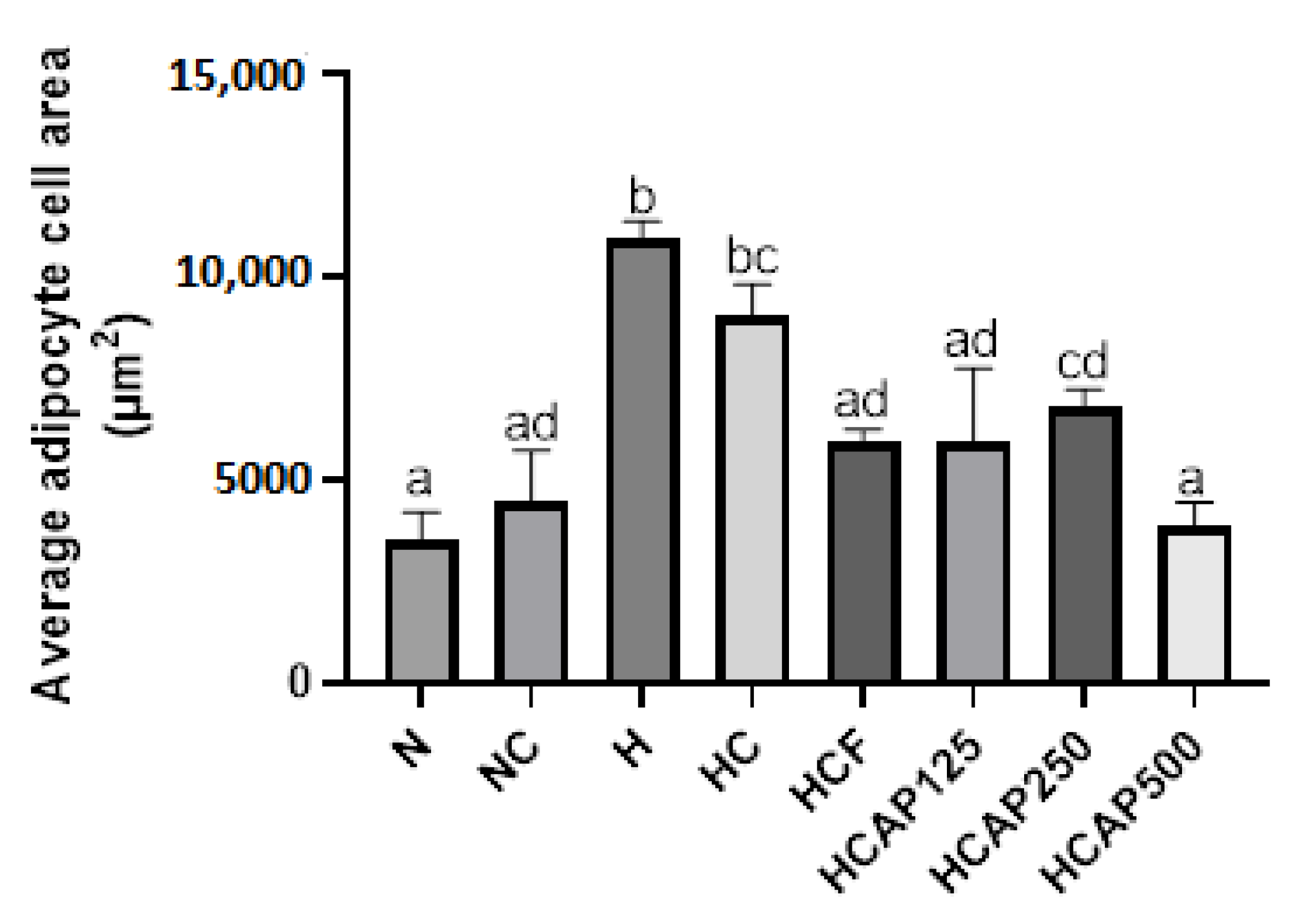
2.5. Impact of A. paniculata on the Retroperitoneal White Adipose Tissue (RpWAT)
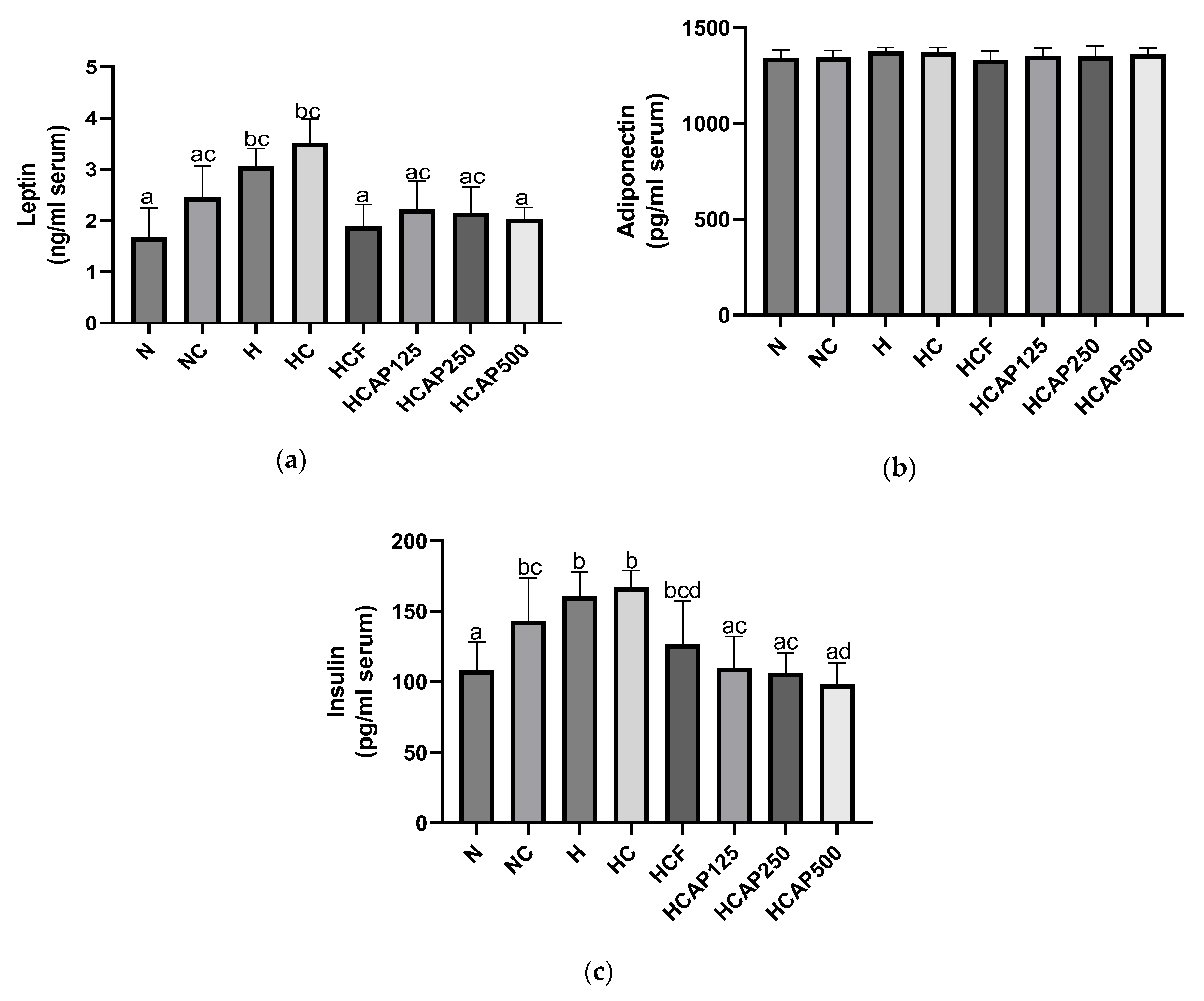
2.6. Impact of A. paniculata on the Serum Leptin, Adiponectin, and Insulin Concentration
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. High-Fat Diet Preparation
4.3. Animal Study
4.4. Chemicals
4.5. Phytochemical Screening
4.6. Biochemical Test
4.7. Histopathological Examination
4.8. ACF Counting
4.9. Adipocyte Area Counting
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.R.A.; Ismail, I.; Suan, M.A.M.; Ahmad, F.; Khazim, W.K.W.; Othman, Z.; Said, R.M.; Tan, W.L.; Mohammed, S.R.N.S.; Soelar, S.A.; et al. Incidence and Mortality Rates of Colorectal Cancer in Malaysia. Epidemiol. Health 2016, 38, e2016007. [Google Scholar] [CrossRef] [PubMed]
- Mariotto, A.B.; Robin Yabroff, K.; Shao, Y.; Feuer, E.J.; Brown, M.L. Projections of the Cost of Cancer Care in the United States: 2010–2020. JNCI J. Natl. Cancer Inst. 2011, 103, 117. [Google Scholar] [CrossRef]
- Sninsky, J.A.; Shore, B.M.; Lupu, G.V.; Crockett, S.D. Risk Factors for Colorectal Polyps and Cancer. Gastrointest. Endosc. Clin. N. Am. 2022, 32, 195–213. [Google Scholar] [CrossRef]
- Vandevijvere, S.; Chow, C.C.; Hall, K.D.; Umali, E.; Swinburn, B.A. Increased Food Energy Supply as a Major Driver of the Obesity Epidemic: A Global Analysis. Bull. World Health Organ. 2015, 93, 446. [Google Scholar] [CrossRef]
- Moghaddam, A.A.; Woodward, M.; Huxley, R. Obesity and Risk of Colorectal Cancer: A Meta-Analysis of 31 Studies with 70,000 Events. Cancer Epidemiol. Biomark. Prev. 2007, 16, 2533–2547. [Google Scholar] [CrossRef]
- Riondino, S.; Roselli, M.; Palmirotta, R.; Della-Morte, D.; Ferroni, P.; Guadagni, F. Obesity and Colorectal Cancer: Role of Adipokines in Tumor Initiation and Progression. World J. Gastroenterol. 2014, 20, 5177–5190. [Google Scholar] [CrossRef]
- Zhang, J.; Guo, S.; Li, J.; Bao, W.; Zhang, P.; Huang, Y.; Ling, P.; Wang, Y.; Zhao, Q. Effects of High-Fat Diet-Induced Adipokines and Cytokines on Colorectal Cancer Development. FEBS Open Bio 2019, 9, 2117–2125. [Google Scholar] [CrossRef]
- Schmoll, H.J.; Van Cutsem, E.; Stein, A.; Valentini, V.; Glimelius, B.; Haustermans, K.; Nordlinger, B.; Van de Velde, C.J.; Balmana, J.; Regula, J.; et al. ESMO Consensus Guidelines for Management of Patients with Colon and Rectal Cancer. A Personalized Approach to Clinical Decision Making. Ann. Oncol. 2012, 23, 2479–2516. [Google Scholar] [CrossRef]
- Vodenkova, S.; Buchler, T.; Cervena, K.; Veskrnova, V.; Vodicka, P.; Vymetalkova, V. 5-Fluorouracil and Other Fluoropyrimidines in Colorectal Cancer: Past, Present and Future. Pharmacol. Ther. 2020, 206, 107–447. [Google Scholar] [CrossRef]
- Garg, M.B.; Lincz, L.F.; Adler, K.; Scorgie, F.E.; Ackland, S.P.; Sakoff, J.A. Predicting 5-Fluorouracil Toxicity in Colorectal Cancer Patients from Peripheral Blood Cell Telomere Length: A Multivariate Analysis. Br. J. Cancer 2012, 107, 1525–1533. [Google Scholar] [CrossRef] [PubMed]
- Wigmore, P.M.; Mustafa, S.; El-Beltagy, M.; Lyons, L.; Umka, J.; Bennett, G. Effects of 5-FU. Adv. Exp. Med. Biol. 2010, 678, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Al-Henhena, N.; Ying, R.P.Y.; Ismail, S.; Najm, W.; Khalifa, S.A.M.; El-Seedi, H.; Abdulla, M.A. Chemopreventive Efficacy of Andrographis Paniculata on Azoxymethane-Induced Aberrant Colon Crypt Foci In Vivo. PLoS ONE 2014, 9, 111118. [Google Scholar] [CrossRef]
- Aiello, P.; Sharghi, M.; Mansourkhani, S.M.; Ardekan, A.P.; Jouybari, L.; Daraei, N.; Peiro, K.; Mohamadian, S.; Rezaei, M.; Heidari, M.; et al. Medicinal Plants in the Prevention and Treatment of Colon Cancer. Oxid. Med. Cell. Longev. 2019, 2019, 2075614. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, T.; Hsieh, C.Y.; Lee, J.J.; Sheu, J.R. Experimental and Clinical Pharmacology of Andrographis Paniculata and Its Major Bioactive Phytoconstituent Andrographolide. Evid. Based. Complement. Alternat. Med. 2013, 2013, 846740. [Google Scholar] [CrossRef] [PubMed]
- Malik, Z.; Parveen, R.; Parveen, B.; Zahiruddin, S.; Aasif Khan, M.; Khan, A.; Massey, S.; Ahmad, S.; Husain, S.A. Anticancer Potential of Andrographolide from Andrographis Paniculata (Burm.f.) Nees and Its Mechanisms of Action. J. Ethnopharmacol. 2021, 272, 1–16. [Google Scholar] [CrossRef]
- Samy, R.P.; Thwin, M.M.; Gopalakrishnakone, P.; Ignacimuthu, S. Ethnobotanical Survey of Folk Plants for the Treatment of Snakebites in Southern Part of Tamilnadu, India. J. Ethnopharmacol. 2008, 115, 302–312. [Google Scholar] [CrossRef]
- Zhu, Q.C.; Gao, R.Y.; Wu, W.; Guo, B.M.; Peng, J.Y.; Qin, H.L. Effect of a High-Fat Diet in Development of Colonic Adenoma in an Animal Model. World J. Gastroenterol. 2014, 20, 8119. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, J.; Zhou, Y.; Qiao, L. Obesity and Gastric Cancer. Front. Biosci. 2012, 17, 2383–2390. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, Z.; Li, J. The Current Status of Treatment for Colorectal Cancer in China: A Systematic Review. Medicine 2017, 96, e8242. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Chen, S.R.; Chai, L.; Zhao, J.; Wang, Y.; Wang, Y. Overview of Pharmacological Activities of Andrographis Paniculata and Its Major Compound Andrographolide. Crit. Rev. Food Sci. Nutr. 2019, 59 (Suppl. S1), S17–S29. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, N.; Varma, A.; Padh, H. Andrographolide: A New Plant-Derived Antineoplastic Entity on Horizon. Evid.-Based Complement. Altern. Med. 2011, 2011, 815390. [Google Scholar] [CrossRef]
- Adedapo, A.A.; Adeoye, B.O.; Sofidiya, M.O.; Oyagbemi, A.A. Antioxidant, Antinociceptive and Anti-Inflammatory Properties of the Aqueous and Ethanolic Leaf Extracts of Andrographis Paniculata in Some Laboratory Animals. J. Basic Clin. Physiol. Pharmacol. 2015, 26, 327–334. [Google Scholar] [CrossRef]
- Chao, W.W.; Lin, B.F. Isolation and Identification of Bioactive Compounds in Andrographis Paniculata (Chuanxinlian). Chin. Med. 2010, 5, 17. [Google Scholar] [CrossRef]
- Cheung, H.Y.; Cheung, C.S.; Kong, C.K. Determination of Bioactive Diterpenoids from Andrographis Paniculata by Micellar Electrokinetic Chromatography. J. Chromatogr. A 2001, 930, 171–176. [Google Scholar] [CrossRef]
- Reddy, M.V.B.; Kishore, P.H.; Rao, C.V.; Gunasekar, D.; Caux, C.; Bodo, B. New 2‘-Oxygenated Flavonoids from Andrographis Affinis. J. Nat. Prod. 2003, 66, 295–297. [Google Scholar] [CrossRef]
- Suriyo, T.; Pholphana, N.; Rangkadilok, N.; Thiantanawat, A.; Watcharasit, P.; Satayavivad, J. Andrographis Paniculata Extracts and Major Constituent Diterpenoids Inhibit Growth of Intrahepatic Cholangiocarcinoma Cells by Inducing Cell Cycle Arrest and Apoptosis. Planta Med. 2014, 80, 533–543. [Google Scholar] [CrossRef]
- Sharma, V.; Qayum, A.; Kaul, S.; Singh, A.; Kapoor, K.K.; Mukherjee, D.; Singh, S.K.; Dhar, M.K. Carbohydrate Modifications of Neoandrographolide for Improved Reactive Oxygen Species-Mediated Apoptosis through Mitochondrial Pathway in Colon Cancer. ACS Omega 2019, 4, 20435–20442. [Google Scholar] [CrossRef]
- Rajendran, P.; Rengarajan, T.; Nandakumar, N.; Palaniswami, R.; Nishigaki, Y.; Nishigaki, I. Kaempferol, a Potential Cytostatic and Cure for Inflammatory Disorders. Eur. J. Med. Chem. 2014, 86, 103–112. [Google Scholar] [CrossRef]
- Lim, J.C.W.; Chan, T.K.; Ng, D.S.; Sagineedu, S.R.; Stanslas, J.; Wong, W.F. Andrographolide and Its Analogues: Versatile Bioactive Molecules for Combating Inflammation and Cancer. Clin. Exp. Pharmacol. Physiol. 2012, 39, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Low, M.; Khoo, C.S.; Münch, G.; Govindaraghavan, S.; Sucher, N.J. An in Vitro Study of Anti-Inflammatory Activity of Standardised Andrographis Paniculata Extracts and Pure Andrographolide. BMC Complement. Altern. Med. 2015, 15, 18. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhu, H.; Wang, R.; Zhou, K.; Jing, Y.; Qiu, F. Ent-Labdane Diterpenoid Lactone Stereoisomers from Andrographis Paniculata. J. Nat. Prod. 2008, 71, 852–855. [Google Scholar] [CrossRef] [PubMed]
- Baracos, V.E.; Martin, L.; Korc, M.; Guttridge, D.C.; Fearon, K.C.H. Cancer-Associated Cachexia. Nat. Rev. Dis. Prim. 2018, 4, 17105. [Google Scholar] [CrossRef]
- Darband, S.G.; Sadighparvar, S.; Yousefi, B.; Kaviani, M.; Ghaderi-Pakdel, F.; Mihanfar, A.; Rahimi, Y.; Mobaraki, K.; Majidinia, M. Quercetin Attenuated Oxidative DNA Damage through NRF2 Signaling Pathway in Rats with DMH Induced Colon Carcinogenesis. Life Sci. 2020, 253, 117584. [Google Scholar] [CrossRef]
- Venkatachalam, K.; Vinayagam, R.; Anand, M.A.V.; Isa, N.M.; Ponnaiyan, R. Biochemical and Molecular Aspects of 1,2-Dimethylhydrazine (DMH)-Induced Colon Carcinogenesis: A Review. Toxicol. Res. 2020, 9, 2–18. [Google Scholar] [CrossRef]
- Sherif, D.A.; Makled, M.N.; Suddek, G.M. The HIV Reverse Transcriptase Inhibitor Tenofovir Suppressed DMH/HFD-Induced Colorectal Cancer in Wistar Rats. Fundam. Clin. Pharmacol. 2021, 35, 940–954. [Google Scholar] [CrossRef]
- Qi, G.; Tang, B.; Zhou, L.; Jikihara, H.; Kiwata, A.; Sakamoto, Y.; Tang, F.; Xiao, S.; Wang, Z.; Wu, Q.; et al. Effects of High-Fat Diet on 1,2-Dimethylhydrazine-Induced Aberrant Crypt Foci and Colorectal Tumours in Rats. Biomed. Rep. 2015, 3, 289. [Google Scholar] [CrossRef]
- Hassan, H.F.H.; Mansour, A.M.; Salama, S.A.; El-Sayed, E.S.M. The Chemopreventive Effect of Thymol against Dimethylhydrazine and/or High Fat Diet-Induced Colon Cancer in Rats: Relevance to NF-ΚB. Life Sci. 2021, 274, 119335. [Google Scholar] [CrossRef]
- Poret, J.M.; Souza-Smith, F.; Marcell, S.J.; Gaudet, D.A.; Tzeng, T.H.; Braymer, H.D.; Harrison-Bernard, L.M.; Primeaux, S.D. High Fat Diet Consumption Differentially Affects Adipose Tissue Inflammation and Adipocyte Size in Obesity-Prone and Obesity-Resistant Rats. Int. J. Obes. 2018, 42, 535–541. [Google Scholar] [CrossRef]
- Mopuri, R.; Ganjayi, M.; Banavathy, K.S.; Parim, B.N.; Meriga, B. Evaluation of Anti-Obesity Activities of Ethanolic Extract of Terminalia Paniculata Bark on High Fat Diet-Induced Obese Rats. BMC Complement. Altern. Med. 2015, 15, 76. [Google Scholar] [CrossRef] [PubMed]
- Sabaratnam, R.; Svenningsen, P. Adipocyte-Endothelium Crosstalk in Obesity. Front. Endocrinol. 2021, 12, 681290. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, A.; Spiers, J.P.; Crowley, V.; Williams, E.; Lithander, F.E. Postprandial Adiponectin and Gelatinase Response to a High-Fat versus an Isoenergetic Low-Fat Meal in Lean, Healthy Men. Nutrition 2015, 31, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-C.; Chien, C.-Y.; Hsu, C.-C.; Lee, C.-H.; Chou, Y.-T.; Shiah, S.-G.; Liu, S.-Y.; Yen, C.-Y.; Hsieh, A.C.-T.; Wabitsch, M.; et al. Obesity-Associated Leptin Promotes Chemoresistance in Colorectal Cancer through YAP-Dependent AXL Upregulation. Am. J. Cancer Res. 2021, 11, 4220–4240. [Google Scholar]
- Arita, Y.; Kihara, S.; Ouchi, N.; Takahashi, M.; Maeda, K.; Miyagawa, J.I.; Hotta, K.; Shimomura, I.; Nakamura, T.; Miyaoka, K.; et al. Paradoxical Decrease of an Adipose-Specific Protein, Adiponectin, in Obesity. Biochem. Biophys. Res. Commun. 1999, 257, 79–83. [Google Scholar] [CrossRef]
- Weyer, C.; Funahashi, T.; Tanaka, S.; Hotta, K.; Matsuzawa, Y.; Pratley, R.E.; Tataranni, P.A. Hypoadiponectinemia in Obesity and Type 2 Diabetes: Close Association with Insulin Resistance and Hyperinsulinemia. J. Clin. Endocrinol. Metab. 2001, 86, 1930–1935. [Google Scholar] [CrossRef]
- Kizer, J.R. A Tangled Threesome: Adiponectin, Insulin Sensitivity, and Adiposity: Can Mendelian Randomization Sort Out Causality? Diabetes 2013, 62, 1007. [Google Scholar] [CrossRef]
- Otake, S.; Takeda, H.; Fujishima, S.; Fukui, T.; Orii, T.; Sato, T.; Sasaki, Y.; Nishise, S.; Kawata, S. Decreased Levels of Plasma Adiponectin Associated with Increased Risk of Colorectal Cancer. World J. Gastroenterol. 2010, 16, 1252. [Google Scholar] [CrossRef]
- Chen, C.C.; Lii, C.K.; Lin, Y.H.; Shie, P.H.; Yang, Y.C.; Huang, C.S.; Chen, H.W. Andrographis Paniculata Improves Insulin Resistance in High-Fat Diet-Induced Obese Mice and TNFα-Treated 3T3-L1 Adipocytes. Am. J. Chin. Med. 2020, 48, 1073–1090. [Google Scholar] [CrossRef]
- Ding, L.; Li, J.; Song, B.; Xiao, X.; Huang, W.; Zhang, B.; Tang, X.; Qi, M.; Yang, Q.; Yang, Q.; et al. Andrographolide Prevents High-Fat Diet-Induced Obesity in C57BL/6 Mice by Suppressing the Sterol Regulatory Element-Binding Protein Pathway. J. Pharmacol. Exp. Ther. 2014, 351, 474–483. [Google Scholar] [CrossRef]
- Bahari, H.; Abidin, A.Z.; Balan, S.S.; Perumal, K.V.; Rosli, N.S.; Lotafi, A.H.A.; Danabala, S.; Manimaran, M.; Shafie, N.H.; Abdullah, M.A.; et al. The Effects of Elateriospermum Tapos against Obese Maternal Rat in Mitigating Obesity Development among Their Adult Female Offspring. Pharmacogn. Mag. 2020, 16, 706. [Google Scholar] [CrossRef]
- Sharma, S.; Joshi, R.; Kumar, D. Quantitative Analysis of Flavonols, Flavonol Glycoside and Homoisoflavonoids in Polygonatum Verticillatum Using UHPLC-DAD-QTOF-IMS and Evaluation of Their Antioxidant Potential. Phytochem. Anal. 2020, 31, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.S.; Liu, X.; Nagel, J.M.; Chamberland, J.P.; Diakopoulos, K.N.; Brinkoetter, M.T.; Hatziapostolou, M.; Wu, Y.; Robson, S.C.; Iliopoulos, D.; et al. Salutary Effects of Adiponectin on Colon Cancer: In Vivo and in Vitro Studies in Mice. Gut 2013, 62, 561–570. [Google Scholar] [CrossRef] [PubMed]
- Fujii, T.; Sakai, E.; Takahashi, H.; Yamada, E.; Ohkubo, H.; Higurashi, T.; Nakajima, A. The Distribution of Human Rectal Aberrant Crypt Foci and Criteria for Defining the Counting Area. Oncol. Lett. 2017, 13, 4501–4504. [Google Scholar] [CrossRef]
- Szymlek-Gay, E.A.; Gray, A.R.; Heath, A.-L.M.; Ferguson, E.L.; Skeaff, C.M. Red Meat Consumption and Serum Lipids and Fatty Acids in Toddlers: Secondary Outcomes of a Randomized Controlled Trial. J. Pediatr. Gastroenterol. Nutr. 2018, 67, 395–400. [Google Scholar] [CrossRef]
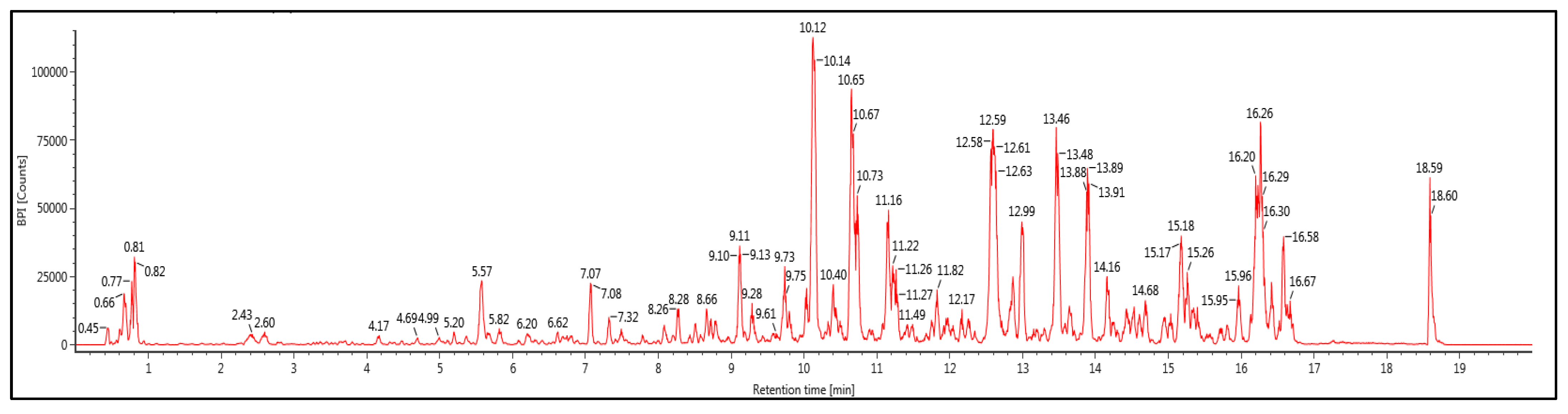

| Compounds | Molecular Formula | RT (min) | Molecular Mass | Observed (m/z) | Mass Error (ppm) |
|---|---|---|---|---|---|
| 19β-Glucosyl-14-deoxy-11,12-didehydroand-rographoside | C20H28O4 | 12.6 | 332.1983 | 331.191 | −1.3 |
| 3,4-O-Dicaffeoylquinic acid | C25H24O12 | 10.65 | 516.1273 | 515.12 | 1 |
| Genistein-7,4′-di-O-β-D-glucoside | C27H30O15 | 9.12 | 594.1594 | 593.1521 | 1.6 |
| 12S-Hydroxyandrographolide | C20H32O6 | 10.73 | 368.2198 | 367.2125 | −0.3 |
| 10-Hydroxyligustroside | C25H32O13 | 12.98 | 540.1845 | 539.1772 | 0.3 |
| 19β-Glucosyl-14-deoxyandrographoside | C26H40O9 | 13.47 | 496.2672 | 495.26 | 0 |
| Group | Total Food Intake (kJ) | Body Weight (g) | RpWAT Weight | Colon Weight | Colon Length (cm) | |||
|---|---|---|---|---|---|---|---|---|
| Initial | Final | (g) | (%BW) | (g) | (%BW) | |||
| N | 7477.40 ± 873.70 a | 227.50 ± 10.9 | 440.33 ± 12.01 a | 6.48 ± 0.71 a | 1.47 | 1.48 ± 0.22 a | 0.34 | 17.53 ± 0.58 a |
| NC | 7917.40 ± 1515.19 a | 226.50 ± 6.4 | 448.17 ± 16.39 a | 9.16 ± 1.60 ab | 2.00 | 1.26 ± 0.10 a | 0.28 | 14.08 ± 0.68 a |
| H | 12342.26 ± 237.56 b | 240.00 ± 5.6 | 571.83 ± 20.20 b | 22.85 ± 3.52 c | 3.94 | 1.67 ± 0.22 a | 0.30 | 15.83 ± 0.94 a |
| HC | 12319.72 ± 991.74 b | 202.20 ± 19.8 | 453.33 ± 19.13 a | 9.17 ± 2.54 ab | 1.95 | 1.43 ± 0.12 a | 0.32 | 16.07 ± 1.37 a |
| HCF | 12586.98 ± 502.32 b | 239.2 ± 7.1 | 461.17 ± 6.09 a | 13.07 ± 3.18 ac | 2.83 | 1.64 ± 0.23 a | 0.35 | 16.07 ± 1.37 a |
| HCAP125 | 12422.76 ± 705.18 b | 238.30 ± 3.3 | 503.67 ± 18.07 ab | 10.90 ± 1.56 ac | 2.13 | 1.70 ± 0.13 a | 0.34 | 15.75 ± 1.11 a |
| HCAP250 | 12525.80 ± 75.72 b | 247.50 ± 8.8 | 512.50 ± 24.24 ab | 14.83 ± 4.32 bc | 2.81 | 1.54 ± 0.11 a | 0.30 | 15.30 ± 0.80 a |
| HCAP500 | 12477.50 ± 271.30 b | 235.8 ± 6.9 | 474.17 ± 7.45 a | 7.72 ± 1.07 ab | 1.64 | 1.70 ± 0.22 a | 0.36 | 16.50 ± 1.65 a |
| Group | No. of Crypts per ACF | Total No. of ACF/Colon | ||||
|---|---|---|---|---|---|---|
| One Crypt | Two Crypts | Three Crypts | Four Crypts | Five or More Crypts | ||
| N | 0 | 0 | 0 | 0 | 0 | 0 |
| NC | 5.25 ± 1.60 ab | 3.50 ± 0.90 a | 1.50 ± 0.70 a | 1.62 ± 0.56 ab | 3.25 ± 1.08 a | 15.37 ± 0.26 ab |
| H | 1.37 ± 0.73 b | 2.00 ± 0.94 a | 3.50 ± 0.56 a | 2.25 ± 0.45 ab | 6.12 ± 1.67 ab | 15.12 ± 0.12 ab |
| HC | 7.50 ± 1.42 a | 3.87 ± 0.63 a | 1.62 ± 0.46 a | 0.87 ± 0.35 b | 6.50 ± 1.50 ab | 20.37 ± 0.80 c |
| HCF | 5.87 ± 1.41 ab | 1.87 ± 0.63 a | 2.00 ± 0.82 a | 0.37 ± 0.18 b | 7.25 ± 1.94 ab | 17.62 ± 0.37 d |
| HCAP125 | 2.80 ± 1.15 ab | 1.80 ± 0.58 a | 1.00 ± 0.31 a | 0.60 ± 0.40 b | 10.80 ± 0.86 b | 17.00 ± 0.83 ad |
| HCAP250 | 1.16 ± 0.47 b | 2.66 ± 0.71 a | 3.83 ± 0.70 a | 3.50 ± 0.76 a | 5.16 ± 0.83 ab | 16.33 ± 0.33 abd |
| HCAP500 | 5.00 ± 1.69 ab | 1.66 ± 0.33 a | 1.66 ± 0.33 a | 1.16 ± 0.98 ab | 4.33 ± 1.72 ab | 13.83 ± 0.87 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Subarmaniam, T.; Mahmad Rusli, R.N.; Perumal, K.V.; Yong, Y.K.; Hadizah, S.; Othman, F.; Salem, K.; Shafie, N.H.; Hasham, R.; Yin, K.B.; et al. The Potential Chemopreventive Effect of Andrographis paniculata on 1,2-Dimethylhydrazine and High-Fat-Diet-Induced Colorectal Cancer in Sprague Dawley Rats. Int. J. Mol. Sci. 2023, 24, 5224. https://doi.org/10.3390/ijms24065224
Subarmaniam T, Mahmad Rusli RN, Perumal KV, Yong YK, Hadizah S, Othman F, Salem K, Shafie NH, Hasham R, Yin KB, et al. The Potential Chemopreventive Effect of Andrographis paniculata on 1,2-Dimethylhydrazine and High-Fat-Diet-Induced Colorectal Cancer in Sprague Dawley Rats. International Journal of Molecular Sciences. 2023; 24(6):5224. https://doi.org/10.3390/ijms24065224
Chicago/Turabian StyleSubarmaniam, Tharani, Rusydatul Nabila Mahmad Rusli, Kokila Vani Perumal, Yoke Keong Yong, Siti Hadizah, Fezah Othman, Khaled Salem, Nurul Husna Shafie, Rosnani Hasham, Khoo Boon Yin, and et al. 2023. "The Potential Chemopreventive Effect of Andrographis paniculata on 1,2-Dimethylhydrazine and High-Fat-Diet-Induced Colorectal Cancer in Sprague Dawley Rats" International Journal of Molecular Sciences 24, no. 6: 5224. https://doi.org/10.3390/ijms24065224
APA StyleSubarmaniam, T., Mahmad Rusli, R. N., Perumal, K. V., Yong, Y. K., Hadizah, S., Othman, F., Salem, K., Shafie, N. H., Hasham, R., Yin, K. B., Abdul Kadir, K. K., Bahari, H., & Zakaria, Z. A. (2023). The Potential Chemopreventive Effect of Andrographis paniculata on 1,2-Dimethylhydrazine and High-Fat-Diet-Induced Colorectal Cancer in Sprague Dawley Rats. International Journal of Molecular Sciences, 24(6), 5224. https://doi.org/10.3390/ijms24065224







