The Role of Lubricin, Irisin and Exercise in the Prevention and Treatment of Osteoarthritis
Abstract
1. Introduction
2. Molecular Mechanism of Lubricin and Cartilage
2.1. Lubricin
2.2. Lubricin and Cartilage
3. Molecular Mechanism of Irisin and Cartilage
3.1. Irisin and the Bone–Muscle Cross-Talk
3.2. Irisin and Cartilage
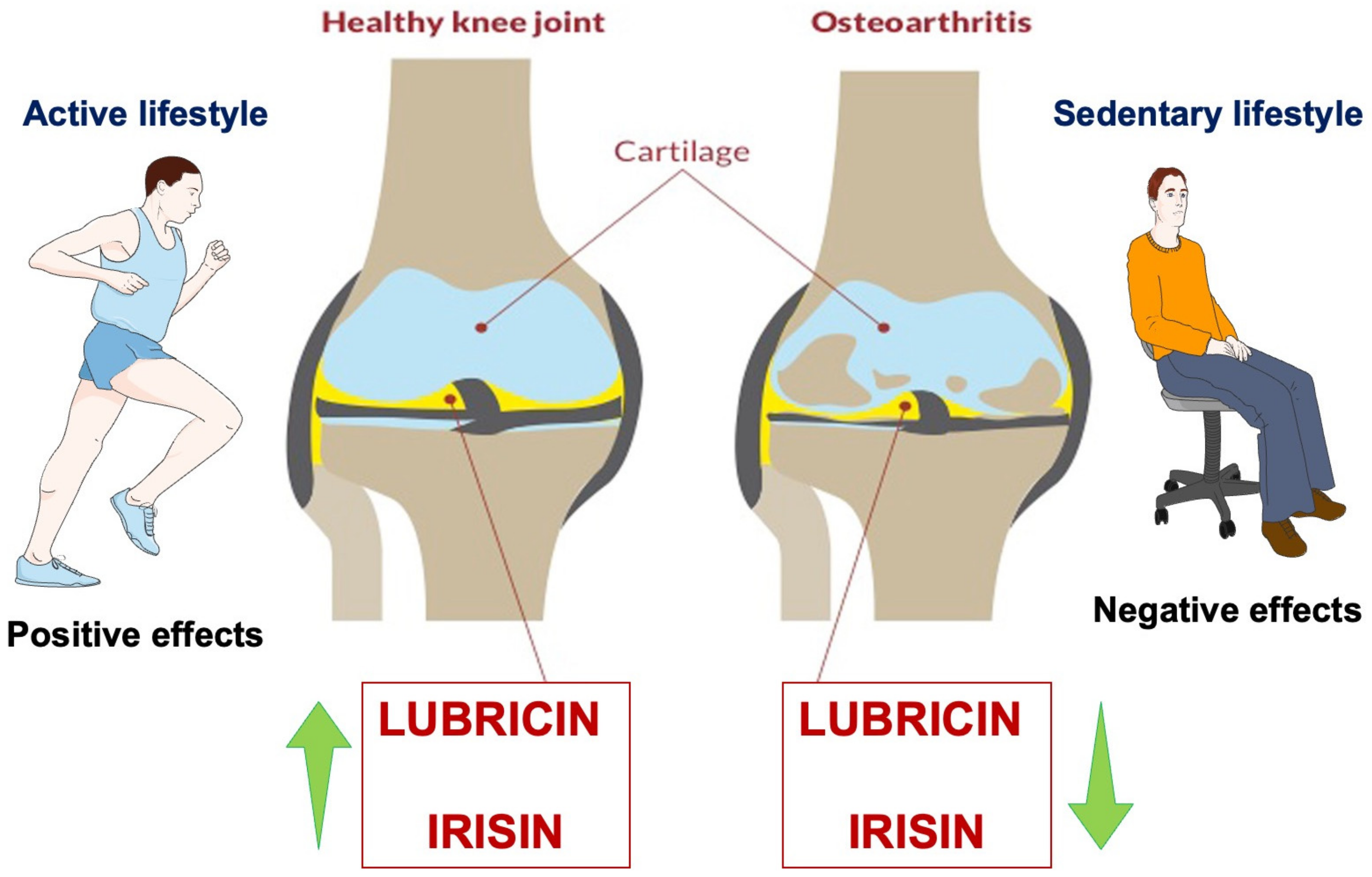
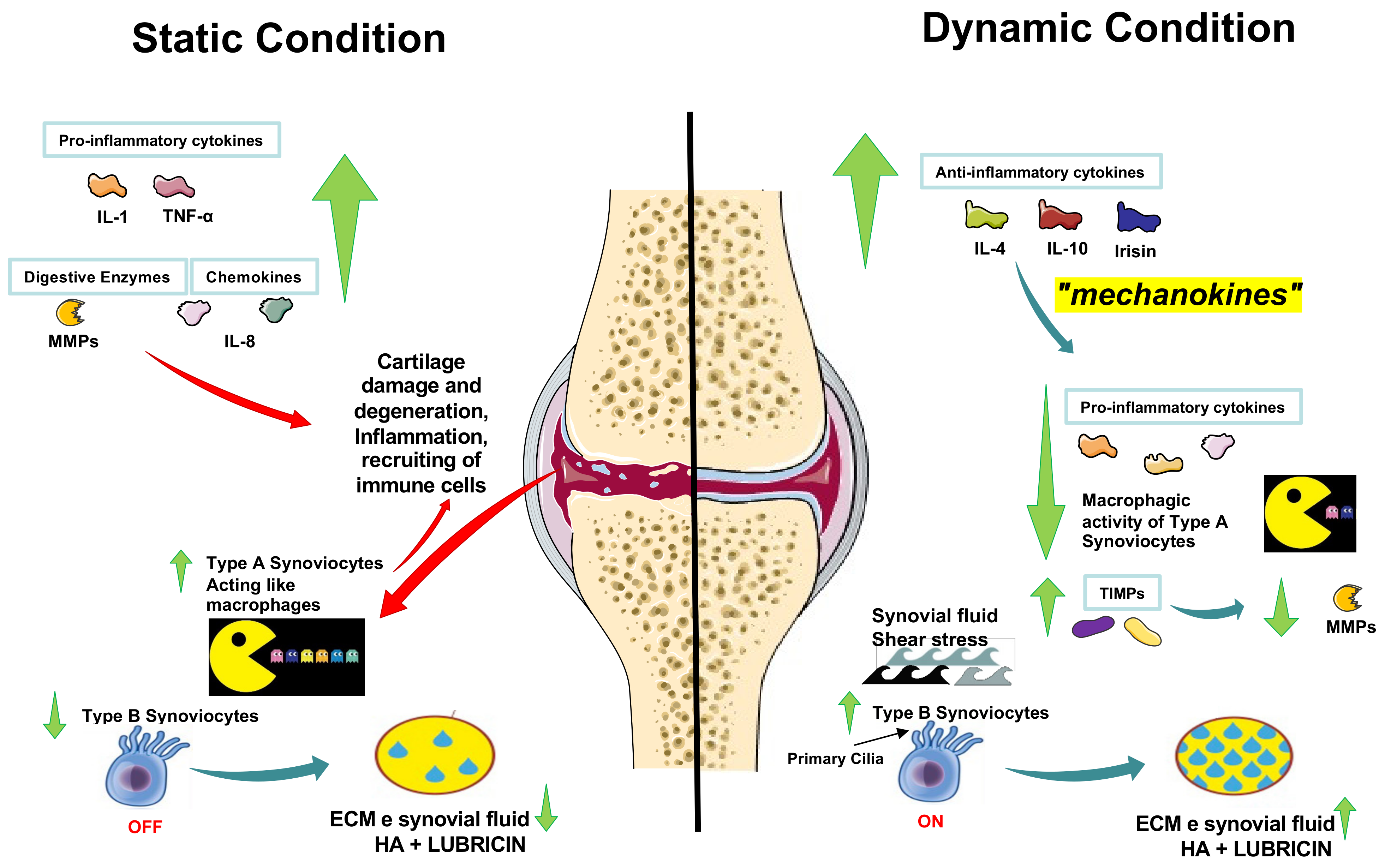
4. Exercise, Lubricin, Irisin, and Cartilage in Osteoarthritis
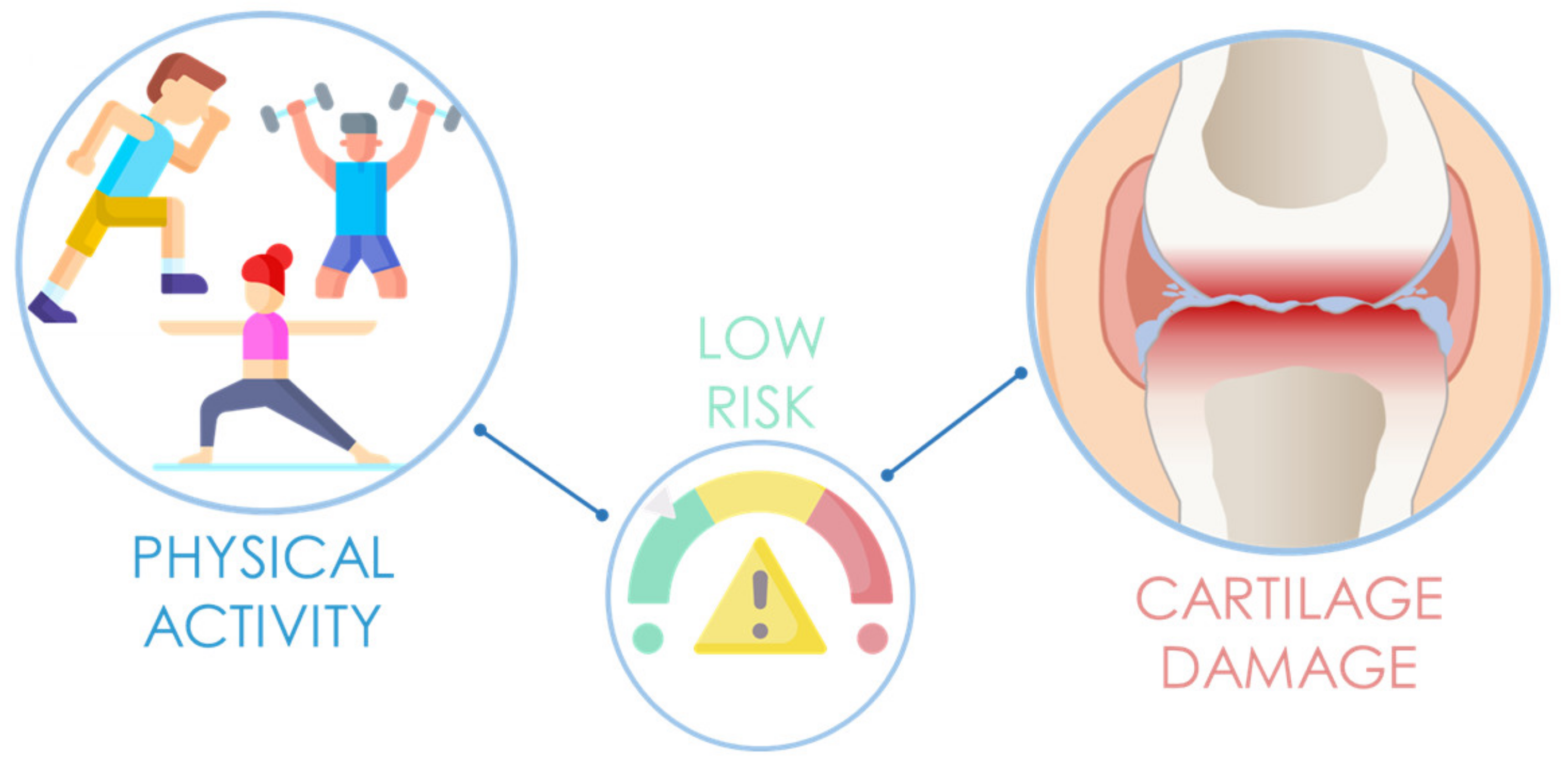
4.1. The Role of Physical Activity in Cartilage Degeneration
4.2. Lubricin and Exercise in Healthy Cartilage and OA
4.3. Irisin and Exercise in Healthy Cartilage and OA
5. Perspectives
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Musumeci, G. The Effect of Mechanical Loading on Articular Cartilage. J. Funct. Morphol. Kinesiol. 2016, 1, 154–161. [Google Scholar] [CrossRef]
- Szychlinska, M.A.; Di Rosa, M.; Castorina, A.; Mobasheri, A.; Musumeci, G. A correlation between intestinal microbiota dysbiosis and osteoarthritis. Heliyon 2019, 5, e01134. [Google Scholar] [CrossRef] [PubMed]
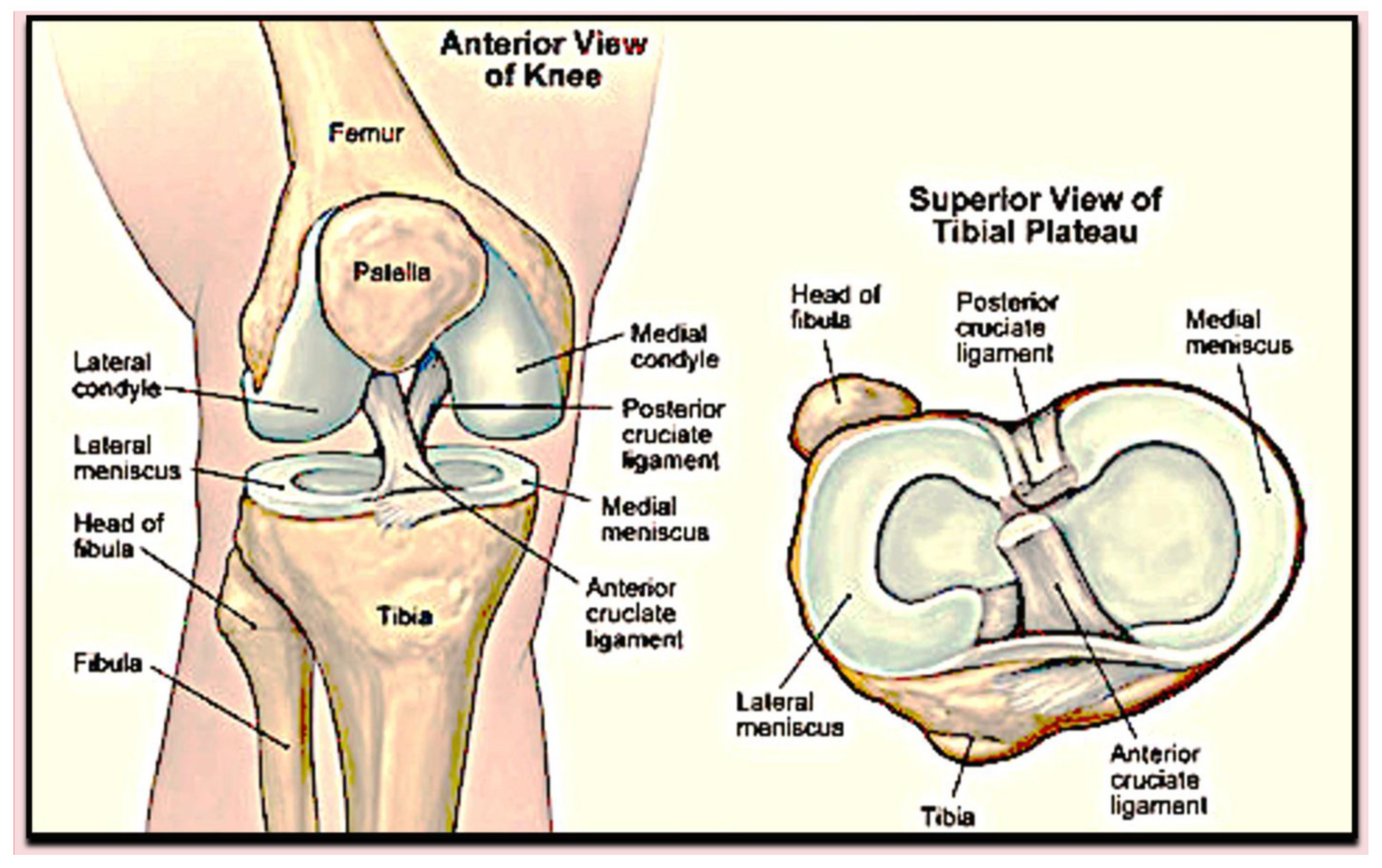
- Blackburn, T.A.; Craig, E. Knee anatomy: A brief review. Phys. Ther. 1980, 60, 1556–1560. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, H.; Wenz, W.; Ivancic, M.; Steck, E.; Richter, W. Early and stable upregulation of collagen type II, collagen type I and YKL40 expression levels in cartilage during early experimental osteoarthritis occurs independent of joint location and histological grading. Arthritis Res. Ther. 2005, 7, R156–R165. [Google Scholar] [CrossRef]
- Lefebvre, V.; Smits, P. Transcriptional control of chondrocyte fate and differentiation. Birth Defects Res. Part C Embryo Today Rev. 2005, 75, 200–212. [Google Scholar] [CrossRef]
- Schumacher, B.L.; Block, J.A.; Schmid, T.M.; Aydelotte, M.B.; Kuettner, K.E. A novel proteoglycan synthesized and secreted by chondrocytes of the superficial zone of articular cartilage. Arch. Biochem. Biophys. 1994, 311, 144–152. [Google Scholar] [CrossRef]
- Iwanaga, T.; Shikichi, M.; Kitamura, H.; Yanase, H.; Nozawa-Inoue, K. Morphology and functional roles of synoviocytes in the joint. Arch. Histol. Cytol. 2000, 63, 17–31. [Google Scholar] [CrossRef]
- Shikichi, M.; Kitamura, H.P.; Yanase, H.; Konno, A.; Takahashi-Iwanaga, H.; Iwanaga, T. Three-dimensional ultrastructure of synoviocytes in the horse joint as revealed by the scanning electron microscope. Arch. Histol. Cytol. 1999, 62, 219–229. [Google Scholar] [CrossRef]
- Ingram, K.R.; Wann, A.K.; Angel, C.K.; Coleman, P.J.; Levick, J.R. Cyclic movement stimulates hyaluronan secretion into the synovial cavity of rabbit joints. J. Physiol. 2008, 586, 1715–1729. [Google Scholar] [CrossRef]
- Castrogiovanni, P.; Di Rosa, M.; Ravalli, S.; Castorina, A.; Guglielmino, C.; Imbesi, R.; Vecchio, M.; Drago, F.; Szychlinska, M.A.; Musumeci, G. Moderate Physical Activity as a Prevention Method for Knee Osteoarthritis and the Role of Synoviocytes as Biological Key. Int. J. Mol. Sci. 2019, 20, 511. [Google Scholar] [CrossRef]
- Han, S. Osteoarthritis year in review 2022: Biology. Osteoarthr. Cartil. 2022, 30, 1575–1582. [Google Scholar] [CrossRef] [PubMed]
- Loreto, C.; Musumeci, G.; Leonardi, R. Chondrocyte-like apoptosis in temporomandibular joint disc internal derangement as a repair-limiting mechanism. An in vivo study. Histol. Histopathol. 2009, 24, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Abramson, S.B.; Attur, M. Developments in the scientific understanding of osteoarthritis. Arthritis Res. Ther. 2009, 11, 227. [Google Scholar] [CrossRef] [PubMed]
- Rattner, J.B.; Sciore, P.; Ou, Y.; van der Hoorn, F.A.; Lo, I.K. Primary cilia in fibroblast-like type B synoviocytes lie within a cilium pit: A site of endocytosis. Histol. Histopathol. 2010, 25, 865–875. [Google Scholar] [CrossRef] [PubMed]
- Szychlinska, M.A.; Trovato, F.M.; Di Rosa, M.; Malaguarnera, L.; Puzzo, L.; Leonardi, R.; Castrogiovanni, P.; Musumeci, G. Co-Expression and Co-Localization of Cartilage Glycoproteins CHI3L1 and Lubricin in Osteoarthritic Cartilage: Morphological, Immunohistochemical and Gene Expression Profiles. Int. J. Mol. Sci. 2016, 17, 359. [Google Scholar] [CrossRef]
- Jones, A.R.; Flannery, C.R. Bioregulation of lubricin expression by growth factors and cytokines. Eur. Cell Mater. 2007, 13, 40–45, discussion 45. [Google Scholar] [CrossRef]
- Lee, I.M.; Shiroma, E.J.; Lobelo, F.; Puska, P.; Blair, S.N.; Katzmarzyk, P.T. Effect of physical inactivity on major non-communicable diseases worldwide: An analysis of burden of disease and life expectancy. Lancet 2012, 380, 219–229. [Google Scholar] [CrossRef]
- Zhang, J.; Valverde, P.; Zhu, X.; Murray, D.; Wu, Y.; Yu, L.; Jiang, H.; Dard, M.M.; Huang, J.; Xu, Z.; et al. Exercise-induced irisin in bone and systemic irisin administration reveal new regulatory mechanisms of bone metabolism. Bone Res. 2017, 5, 16056. [Google Scholar] [CrossRef]
- Li, X.; Zhu, X.; Wu, H.; Van Dyke, T.E.; Xu, X.; Morgan, E.F.; Fu, W.; Liu, C.; Tu, Q.; Huang, D.; et al. Roles and Mechanisms of Irisin in Attenuating Pathological Features of Osteoarthritis. Front. Cell Dev. Biol. 2021, 9, 703670. [Google Scholar] [CrossRef]
- Jay, G.D.; Tantravahi, U.; Britt, D.E.; Barrach, H.J.; Cha, C.J. Homology of lubricin and superficial zone protein (SZP): Products of megakaryocyte stimulating factor (MSF) gene expression by human synovial fibroblasts and articular chondrocytes localized to chromosome 1q25. J. Orthop. Res. 2001, 19, 677–687. [Google Scholar] [CrossRef]
- Nugent-Derfus, G.E.; Chan, A.H.; Schumacher, B.L.; Sah, R.L. PRG4 exchange between the articular cartilage surface and synovial fluid. J. Orthop. Res. 2007, 25, 1269–1276. [Google Scholar] [CrossRef]
- Jones, A.R.; Gleghorn, J.P.; Hughes, C.E.; Fitz, L.J.; Zollner, R.; Wainwright, S.D.; Caterson, B.; Morris, E.A.; Bonassar, L.J.; Flannery, C.R. Binding and localization of recombinant lubricin to articular cartilage surfaces. J. Orthop. Res. 2007, 25, 283–292. [Google Scholar] [CrossRef]
- Rhee, D.K.; Marcelino, J.; Baker, M.; Gong, Y.; Smits, P.; Lefebvre, V.; Jay, G.D.; Stewart, M.; Wang, H.; Warman, M.L.; et al. The secreted glycoprotein lubricin protects cartilage surfaces and inhibits synovial cell overgrowth. J. Clin. Investig. 2005, 115, 622–631. [Google Scholar] [CrossRef]
- Hui, A.Y.; McCarty, W.J.; Masuda, K.; Firestein, G.S.; Sah, R.L. A systems biology approach to synovial joint lubrication in health, injury, and disease. Wiley Interdiscip. Rev. Syst. Biol. Med. 2012, 4, 15–37. [Google Scholar] [CrossRef]
- Jay, G.D.; Fleming, B.C.; Watkins, B.A.; McHugh, K.A.; Anderson, S.C.; Zhang, L.X.; Teeple, E.; Waller, K.A.; Elsaid, K.A. Prevention of cartilage degeneration and restoration of chondroprotection by lubricin tribosupplementation in the rat following anterior cruciate ligament transection. Arthritis Rheum. 2010, 62, 2382–2391. [Google Scholar] [CrossRef]
- Teeple, E.; Elsaid, K.A.; Jay, G.D.; Zhang, L.; Badger, G.J.; Akelman, M.; Bliss, T.F.; Fleming, B.C. Effects of supplemental intra-articular lubricin and hyaluronic acid on the progression of posttraumatic arthritis in the anterior cruciate ligament-deficient rat knee. Am. J. Sports Med. 2011, 39, 164–172. [Google Scholar] [CrossRef]
- Shekhawat, V.K.; Hamilton, J.L.; Pacione, C.A.; Schmid, T.M.; Wimmer, M.A. A Moving Contact Of Articulation Enhances The Biosynthetic And Functional Responses Of Articular Cartilage. Biotribology 2021, 26, 100180. [Google Scholar] [CrossRef]
- Catalán, V.; Frühbeck, G.; Gómez-Ambrosi, J. Chapter 8—Inflammatory and Oxidative Stress Markers in Skeletal Muscle of Obese Subjects. In Obesity; del Moral, A.M., Aguilera García, C.M., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 163–189. [Google Scholar]
- Flori, L.; Testai, L.; Calderone, V. The “irisin system”: From biological roles to pharmacological and nutraceutical perspectives. Life Sci. 2021, 267, 118954. [Google Scholar] [CrossRef]
- Ohtaki, H. Chapter 37—Irisin. In Handbook of Hormones; Takei, Y., Ando, H., Tsutsui, K., Eds.; Academic Press: San Diego, CA, USA, 2016. [Google Scholar]
- Boström, P.; Wu, J.; Jedrychowski, M.P.; Korde, A.; Ye, L.; Lo, J.C.; Rasbach, K.A.; Boström, E.A.; Choi, J.H.; Long, J.Z.; et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 2012, 481, 463–468. [Google Scholar] [CrossRef]
- Colaianni, G.; Cinti, S.; Colucci, S.; Grano, M. Irisin and musculoskeletal health. Ann. N. Y. Acad. Sci. 2017, 1402, 5–9. [Google Scholar] [CrossRef]
- Colaianni, G.; Cuscito, C.; Mongelli, T.; Oranger, A.; Mori, G.; Brunetti, G.; Colucci, S.; Cinti, S.; Grano, M. Irisin enhances osteoblast differentiation in vitro. Int. J. Endocrinol. 2014, 2014, 902186. [Google Scholar] [CrossRef] [PubMed]
- Kawao, N.; Kaji, H. Interactions between muscle tissues and bone metabolism. J. Cell. Biochem. 2015, 116, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.; Lee, K.; Ju, J.H. Recent Updates of Diagnosis, Pathophysiology, and Treatment on Osteoarthritis of the Knee. Int. J. Mol. Sci. 2021, 22, 2619. [Google Scholar] [CrossRef] [PubMed]
- Kalichman, L.; Kobyliansky, E. Radiographic hand osteoarthritis and serum levels of osteocalcin: Cross-sectional study. Rheumatol. Int. 2010, 30, 1131–1135. [Google Scholar] [CrossRef]
- Katz, J.N.; Arant, K.R.; Loeser, R.F. Diagnosis and Treatment of Hip and Knee Osteoarthritis: A Review. JAMA 2021, 325, 568–578. [Google Scholar] [CrossRef]
- Mao, Y.; Xu, W.; Xie, Z.; Dong, Q. Association of Irisin and CRP Levels with the Radiographic Severity of Knee Osteoarthritis. Genet. Test. Mol. Biomark. 2016, 20, 86–89. [Google Scholar] [CrossRef]
- Löffler, D.; Müller, U.; Scheuermann, K.; Friebe, D.; Gesing, J.; Bielitz, J.; Erbs, S.; Landgraf, K.; Wagner, I.V.; Kiess, W.; et al. Serum irisin levels are regulated by acute strenuous exercise. J. Clin. Endocrinol. Metab. 2015, 100, 1289–1299. [Google Scholar] [CrossRef]
- Vadalà, G.; Di Giacomo, G.; Ambrosio, L.; Cannata, F.; Cicione, C.; Papalia, R.; Denaro, V. Irisin Recovers Osteoarthritic Chondrocytes In Vitro. Cells 2020, 9, 1478. [Google Scholar] [CrossRef]
- Wang, F.S.; Kuo, C.W.; Ko, J.Y.; Chen, Y.S.; Wang, S.Y.; Ke, H.J.; Kuo, P.C.; Lee, C.H.; Wu, J.C.; Lu, W.B.; et al. Irisin Mitigates Oxidative Stress, Chondrocyte Dysfunction and Osteoarthritis Development through Regulating Mitochondrial Integrity and Autophagy. Antioxidants 2020, 9, 810. [Google Scholar] [CrossRef]
- He, Z.; Li, H.; Han, X.; Zhou, F.; Du, J.; Yang, Y.; Xu, Q.; Zhang, S.; Zhang, S.; Zhao, N.; et al. Irisin inhibits osteocyte apoptosis by activating the Erk signaling pathway in vitro and attenuates ALCT-induced osteoarthritis in mice. Bone 2020, 141, 115573. [Google Scholar] [CrossRef]
- Messina, O.D.; Vidal Wilman, M.; Vidal Neira, L.F. Nutrition, osteoarthritis and cartilage metabolism. Aging Clin. Exp. Res. 2019, 31, 807–813. [Google Scholar] [CrossRef]
- Oliviero, F.; Ramonda, R. Cartilage-derived biomarkers in osteoarthritis. Indian J. Med. Res. 2021, 153, 413–415. [Google Scholar] [CrossRef]
- Li, X.; Liu, Y.; Liu, Q.; Wang, S.; Ma, Y.; Jin, Q. Recombinant human irisin regulated collagen II, matrix metalloproteinase-13 and the Wnt/β-catenin and NF-κB signaling pathways in interleukin-1β-induced human SW1353 cells. Exp. Ther. Med. 2020, 19, 2879–2886. [Google Scholar] [CrossRef]
- Jia, S.; Yang, Y.; Bai, Y.; Wei, Y.; Zhang, H.; Tian, Y.; Liu, J.; Bai, L. Mechanical Stimulation Protects Against Chondrocyte Pyroptosis Through Irisin-Induced Suppression of PI3K/Akt/NF-κB Signal Pathway in Osteoarthritis. Front. Cell Dev. Biol. 2022, 10, 797855. [Google Scholar] [CrossRef]
- Ercan, Z.; Deniz, G.; Yentur, S.B.; Arikan, F.B.; Karatas, A.; Alkan, G.; Koca, S.S. Effects of acute aerobic exercise on cytokines, klotho, irisin, and vascular endothelial growth factor responses in rheumatoid arthritis patients. Ir. J. Med. Sci. 2022, 192, 491–497. [Google Scholar] [CrossRef]
- Musumeci, G.; Castrogiovanni, P.; Trovato, F.M.; Imbesi, R.; Giunta, S.; Szychlinska, M.A.; Loreto, C.; Castorina, S.; Mobasheri, A. Physical activity ameliorates cartilage degeneration in a rat model of aging: A study on lubricin expression. Scand. J. Med. Sci. Sports 2015, 25, e222–e230. [Google Scholar] [CrossRef]
- Musumeci, G.; Loreto, C.; Leonardi, R.; Castorina, S.; Giunta, S.; Carnazza, M.L.; Trovato, F.M.; Pichler, K.; Weinberg, A.M. The effects of physical activity on apoptosis and lubricin expression in articular cartilage in rats with glucocorticoid-induced osteoporosis. J. Bone Miner. Metab. 2013, 31, 274–284. [Google Scholar] [CrossRef]
- Gay, C.; Chabaud, A.; Guilley, E.; Coudeyre, E. Educating patients about the benefits of physical activity and exercise for their hip and knee osteoarthritis. Systematic literature review. Ann. Phys. Rehabil. Med. 2016, 59, 174–183. [Google Scholar] [CrossRef]
- Ravalli, S.; Roggio, F.; Lauretta, G.; Di Rosa, M.; D’Amico, A.G.; D’Agata, V.; Maugeri, G.; Musumeci, G. Exploiting real-world data to monitor physical activity in patients with osteoarthritis: The opportunity of digital epidemiology. Heliyon 2022, 8, e08991. [Google Scholar] [CrossRef]
- Roberts, H.M.; Moore, J.P.; Griffith-McGeever, C.L.; Fortes, M.B.; Thom, J.M. The effect of vigorous running and cycling on serum COMP, lubricin, and femoral cartilage thickness: A pilot study. Eur. J. Appl. Physiol. 2016, 116, 1467–1477. [Google Scholar] [CrossRef]
- Blaney Davidson, E.N.; van Caam, A.P.; van der Kraan, P.M. Osteoarthritis year in review 2016: Biology. Osteoarthr. Cartil. 2017, 25, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Ni, G.X.; Lei, L.; Zhou, Y.Z. Intensity-dependent effect of treadmill running on lubricin metabolism of rat articular cartilage. Arthritis Res. Ther. 2012, 14, R256. [Google Scholar] [CrossRef] [PubMed]
- Elsaid, K.A.; Zhang, L.; Waller, K.; Tofte, J.; Teeple, E.; Fleming, B.C.; Jay, G.D. The impact of forced joint exercise on lubricin biosynthesis from articular cartilage following ACL transection and intra-articular lubricin’s effect in exercised joints following ACL transection. Osteoarthr. Cartil. 2012, 20, 940–948. [Google Scholar] [CrossRef] [PubMed]
- Ning, K.; Wang, Z.; Zhang, X.A. Exercise-induced modulation of myokine irisin in bone and cartilage tissue-Positive effects on osteoarthritis: A narrative review. Front. Aging Neurosci. 2022, 14, 934406. [Google Scholar] [CrossRef]
- Pesce, M.; La Fratta, I.; Paolucci, T.; Grilli, A.; Patruno, A.; Agostini, F.; Bernetti, A.; Mangone, M.; Paoloni, M.; Invernizzi, M.; et al. From Exercise to Cognitive Performance: Role of Irisin. Appl. Sci. 2021, 11, 7120. [Google Scholar] [CrossRef]
- Tsuchiya, Y.; Ando, D.; Goto, K.; Kiuchi, M.; Yamakita, M.; Koyama, K. High-intensity exercise causes greater irisin response compared with low-intensity exercise under similar energy consumption. Tohoku J. Exp. Med. 2014, 233, 135–140. [Google Scholar] [CrossRef]
- Park, J.; Bae, J.; Lee, J. Complex Exercise Improves Anti-Inflammatory and Anabolic Effects in Osteoarthritis-Induced Sarcopenia in Elderly Women. Healthcare 2021, 9, 711. [Google Scholar] [CrossRef]
- Jürimäe, J.; Remmel, L.; Tamm, A.L.; Purge, P.; Maasalu, K.; Tillmann, V. Associations of Circulating Irisin and Fibroblast Growth Factor-21 Levels with Measures of Energy Homeostasis in Highly Trained Adolescent Rhythmic Gymnasts. J. Clin Med. 2022, 11, 7450. [Google Scholar] [CrossRef]
- Ravalli, S.; Roggio, F.; Magrì, B.; Lauretta, G.; Broggi, G.; Caltabiano, R.; Vecchio, G.M.; Magro, G.; Loreto, C.; Castorina, A.; et al. Immunohistochemical evaluation of autotaxin and lubricin in mild osteoarthritic rat model performing moderate physical activity. Acta Histochem. 2022, 124, 151936. [Google Scholar] [CrossRef]
- Orellana, C.; Calvet, J.; Navarro, N.; Galisteo, C.; Gratacós, J.; Larrosa, M. AB0962 Irisin levels are associated with exercise, pain and function in patients with knee osteoarthritis. Ann. Rheum. Dis. 2018, 77, 1605. [Google Scholar]
- Liu, Y.; Guo, C.; Liu, S.; Zhang, S.; Mao, Y.; Fang, L. Eight Weeks of High-Intensity Interval Static Strength Training Improves Skeletal Muscle Atrophy and Motor Function in Aged Rats via the PGC-1α/FNDC5/UCP1 Pathway. Clin. Interv. Aging 2021, 16, 811–821. [Google Scholar] [CrossRef]
| Study | Sample | Investigation | Main Markers Evaluated | Conclusions |
|---|---|---|---|---|
| Castrogiovanni et al., 2019 [10] | Male Wistar rats divided into groups: control, moderate exercise, ACLT and early OA, ACLT early OA and moderate exercise | Effects of 2 weeks post ACLT, 25 min of moderate running 3 times per week, for 12 weeks, on cartilage-related biomarkers | Synovium, IL-1β, IL-4, IL-6, IL-10, TNF-α, MMP-13, lubricin | Moderate physical activity supported joint tribology, decreased OA-related biomarkers and increased the chondroprotective ones. |
| Szychlinska et al., 2016 [15] | Wistar outbred rats divided into groups: control non-operated group, control operated without ACTL, OA rats with ACTL | Investigation of CHI3L and lubricin and their association with OA | Sections of articular cartilage, radiographs, CHI3L and lubricin | Dysregulation of CHI3L and lubricin could be related to OA development, especially when a KL grade of 2/3 is present |
| Zhang et al., 2017 [18] | Wild-type C57BL/6J and APN-KO mice divided into groups: injected with irisin, injected with saline, injected with lentiviral FNDC5, control EGFP | Effects of 2 weeks of voluntary wheel running on cartilage | FNDC5/irisin, PGC1α, UCP1 | FNDC5/irisin expression is enhanced by 2 weeks of voluntary wheel running at mRNA and protein levels in mice |
| Li et al., 2021 [19] | Irisin over-expressing mice divided into groups and tissue from humans: mice ACLT with PBS, mice ACLT with irisin, deceased human donors | In vitro mechanistic experiment to understand the functioning of irisin | Irisin, IL-1B, IL-6, TNF-α, COX2, MMP-13 | Irisin is important in the development of cartilage and in OA pathogenesis; moreover, irisin could represent a promising therapeutic target in OA |
| Rhee et al., 2005 [23] | Wild-type and lubricin mutant mice | Clinical, radiological and histological characterization and immunohistochemical analysis of lubricin mutant mice to understand the multiple functioning of lubricin during development | Lubricin, synoviocites | Lubricin is fundamental to prevent the deposition of protein onto cartilage from SF, and to inhibit the adhesion of synovial cell to cartilage |
| Jay et al., 2010 [25] | Lewis rats divided into groups: unilateral ACLT, control | Rats were injected with PBS, HSL, rhPRG4 or HSFL twice a week, 7 days after injury, to investigate the degeneration of cartilage | Safranin O stained tibial plateau cartilage, PBS, HSL, rhPRG4 or HSFL, urinary C-terminal cross-linked telopeptide | Regardless of the type of lubricin injection performed, the cartilage degeneration after ACTL was reduced. Intraarticular lubricin injection can delay the degeneration of cartilage |
| Teeple et al., 2011 [26] | Male Lewis rats with ACLT divided into groups based on injection type: LUB, PBS, HA, LUB + HA | Injection of LUB, PBS, HA, LUB+HA to understand if lubricin and/or hyaluronic acid supplementation can reduce cartilage damage in mice with ACLT | CTXII, IL-1β, TNF-α, lubricin, and sGAG, KL grade | LUB+HA supplementation had positive effects on the cartilage of ACLT rats; conversely, HA alone was not effective |
| Mao et al., 2016 [38] | Humans with and without knee OA | Investigation of the levels of irisin and its correlation with OA through enzyme-linked immunosorbent assay | C-reactive protein, irisin | Authors found a negative correlation between the levels of irisin in SF and OA classified with KL grade |
| Löffler et al., 2015 [39] | Humans divided into groups depending on the type of exercise: acute maximal (15 min), short-term acute (30 min), mid-term moderate (6 weeks), long-term low-grade (1 year follow-up) | People were tested for serum irisin levels after performing physical activity of different types and for different times (from 15 min to 1 year) to understand how irisin is influenced by exercise | Irisin, glucose, insulin, triglycerides, cholesterol, testosterone | Irisin’s serum levels are positively correlated with muscle mass. Interestingly, serum irisin levels rose only in the short-term exercise group |
| Vadalà et al., 2020 [40] | Tissue from human patients with OA was divided into groups: cells exposed to PBS and cells exposed to human recombinant irisin | Investigation of the effects of irisin on hOAC through RNA extraction and gene expression analysis, protein extraction and Western blot analysis | Irisin, glycosaminoglycans, collagen type II, ERK, MAPK, Akt, JNK, NF-κB, IL-1, IL-6, MMP-1, MMP-13, TIMP-1, TIMP-3 | The cells exposed to irisin showed increased hOAC content, collagen II protein and gene expression and decreased type X collagen expression. In addition, irisin decreased the levels of IL-1, IL-6, MMP-1, MMP-13 gene expression; conversely, TIMP-1 and TIMP-3 levels increased |
| Wang et al., 2020 [41] | Tissue from human patients with OA and C57L/B6 mice DMM | Investigation of knee OA in humans an in DMM mice to understand the signaling pathway of FNDC5 (the precursor of irisin) | FNDC5, IL-1β, irisin | Irisin is protective against chondrocyte dysfunction |
| He et al., 2020 [42] | C57BL/6J mice divided into groups: sham operated, ACLT with saline, ACLT with irisin | Investigation of the possibility of using irisin in OA as a therapy | Irisin, ERK | Irisin may attenuate cartilage degeneration in mice by the inhibition of osteocyte apoptosis, activating the ERK pathway |
| Li et al., 2020 [45] | Chondrosarcoma cell line SW1353 from human | Western blotting and immunofluorescence analysis of SW1353 cells to understand the interaction of irisin, Wnt/β-catenin and NF-κB pathways | Irisin, IL-1β, MMP-13, NF-κB, collagen II | Irisin can reverse the interleukins’ effects in SW1353 cell line, suggesting that irisin may be protective for cartilage |
| Jia et al., 2022 [46] | Sprague-Dawley rats divided into groups: control, OA, OA + exercise | Investigation of the effects of physical activity on levels of irisin | Radiographs, irisin, IL-1β, MMP-13, ADAMTS-5 | The mechanical stimulation given by moderate exercise protects against OA thanks to irisin. Irisin counterbalances the negative inflammatory effects of IL-1β by suppressing it |
| Musumeci et al., 2015 [48] | Wistar rats divided into groups: sedentary and physically active | 8 weeks of physical activity in total, 2 weeks on treadmill with no inclination for 10 min 5 times per week, 6 weeks on treadmill with 3° slope for 20 min 5 times per week, to analyze the expression of lubricin as a marker of chondrocyte senescence | Lubricin | Moderate physical activity improved lubrication and prevented cartilage degeneration in old rats |
| Musumeci et al., 2013 [49] | Albino Wistar rats divided into: control, OA + prednisolone, prednisolone + treadmill, prednisolone + vibration exercise, prednisolone + vibration exercise + treadmill | Evaluating the effects of different types of physical activity on the expression of lubricin and caspase-3 after prednisolone injection | Lubricin, caspase-3, β tubulin, prednisolone | Exercise was beneficial for cartilage, probably through the expression of lubricin, which inhibits caspase-3, avoiding chondrocyte death |
| Roberts et al., 2016 [52] | Humans divided into groups: runners and cyclists | ELISA analysis to investigate serum lubricin and COMP, ultrasonography of femoral cartilage and how they respond to running and cycling | Lubricin, COMP, ultrasonographic assessment | Acute bouts of running or cycling elicit an increment in cartilage metabolism; moreover, COMP and lubricin levels significantly increased after exercise |
| Ni et al., 2012 [54] | Male Wistar rats divided into groups: control, low-intensity running, moderate-intensity running, high-intensity running | 8 weeks of different types of treadmill training to highlight the differences in cartilage lubricin expression | Lubricin, TGF-β, PRG4 | Low- or moderate-intensity running upregulates PRG4, causing an increase in lubricin. Conversely, excessive running led to a decrement in lubricin levels |
| Elsaid et al., 2012 [55] | Lewis rats divided into groups: 3-week post-ACLT, 3-week post-ACLT + exercise, 5-week post-ACLT + exercise + one intra-articular sham injection of PBS, 5-week post-ACLT + exercise + one intra-articular injection of purified human lubricin | Investigation of the effects of lubricin injection and physical activity in relationship with cartilage degeneration | Lubricin, uCTX II | Intraarticular lubricin injection ameliorated cartilage damage thanks to exercise and preserved the chondrocytes |
| Ravalli et al., 2022 [61] | Wistar outbred rats divided into groups: control, OA, OA + exercise, control + exercise | Rats subjected to physical activity on treadmill 3 times per week for 12 weeks with a progressive duration from 5 to 25 min, to evaluate ATX and lubricin in the process of cartilage homeostasis | ATX, lubricin | ATX may be involved in mild OA and moderate exercise reduced ATX in the femur; lubricin was found in significantly lower concentrations in OA, suggesting its use as a biochemical biomarker |
| Tsuchiya et al., 2014 [58] | Humans divided into groups: treadmill running at 40% of VO2max for 40 min, treadmill running at 80% of VO2max for 20 min | Blood sample collection before and post-exercise to investigate the fluctuations in irisin levels before and after exercise | VO2max, irisin, LDL, HDL | In this preliminary study, it is suggested that a single bout of acute running enhances irisin levels more than low-intensity exercise |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roggio, F.; Petrigna, L.; Trovato, B.; Di Rosa, M.; Musumeci, G. The Role of Lubricin, Irisin and Exercise in the Prevention and Treatment of Osteoarthritis. Int. J. Mol. Sci. 2023, 24, 5126. https://doi.org/10.3390/ijms24065126
Roggio F, Petrigna L, Trovato B, Di Rosa M, Musumeci G. The Role of Lubricin, Irisin and Exercise in the Prevention and Treatment of Osteoarthritis. International Journal of Molecular Sciences. 2023; 24(6):5126. https://doi.org/10.3390/ijms24065126
Chicago/Turabian StyleRoggio, Federico, Luca Petrigna, Bruno Trovato, Michelino Di Rosa, and Giuseppe Musumeci. 2023. "The Role of Lubricin, Irisin and Exercise in the Prevention and Treatment of Osteoarthritis" International Journal of Molecular Sciences 24, no. 6: 5126. https://doi.org/10.3390/ijms24065126
APA StyleRoggio, F., Petrigna, L., Trovato, B., Di Rosa, M., & Musumeci, G. (2023). The Role of Lubricin, Irisin and Exercise in the Prevention and Treatment of Osteoarthritis. International Journal of Molecular Sciences, 24(6), 5126. https://doi.org/10.3390/ijms24065126










