Identifying SARS-CoV-2 Drugs Binding to the Spike Fatty Acid Binding Pocket Using In Silico Docking and Molecular Dynamics
Abstract
1. Introduction
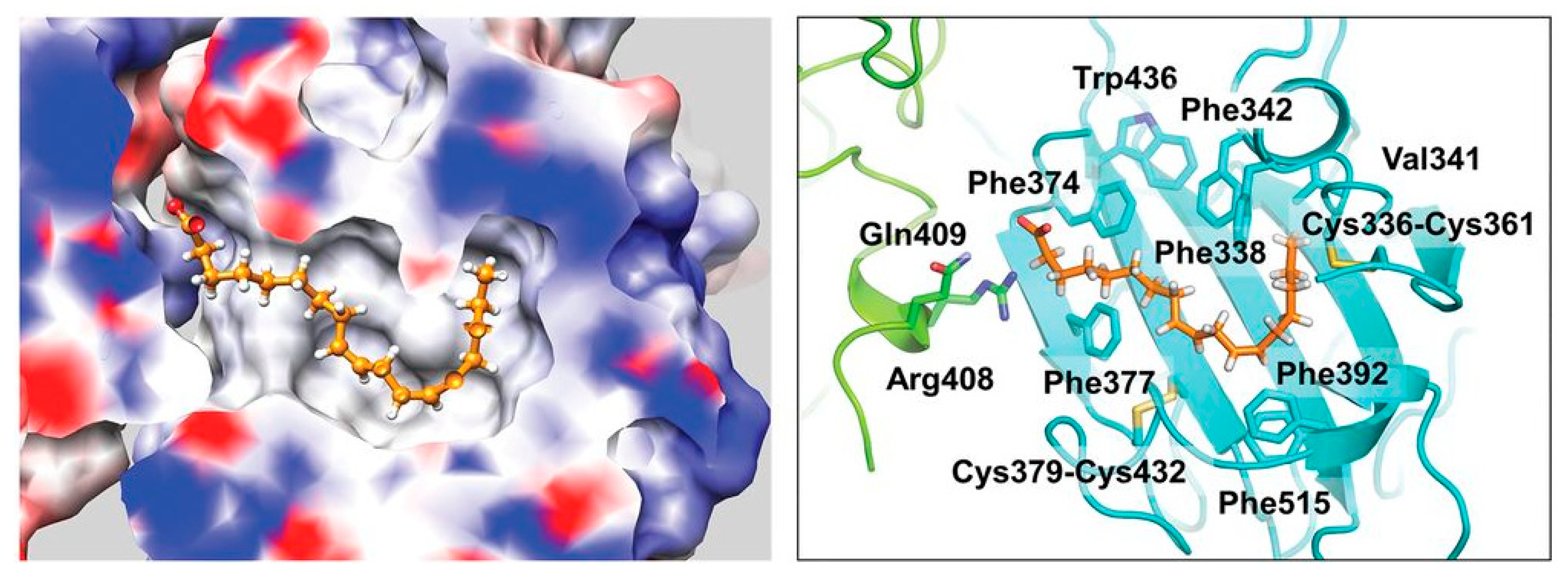
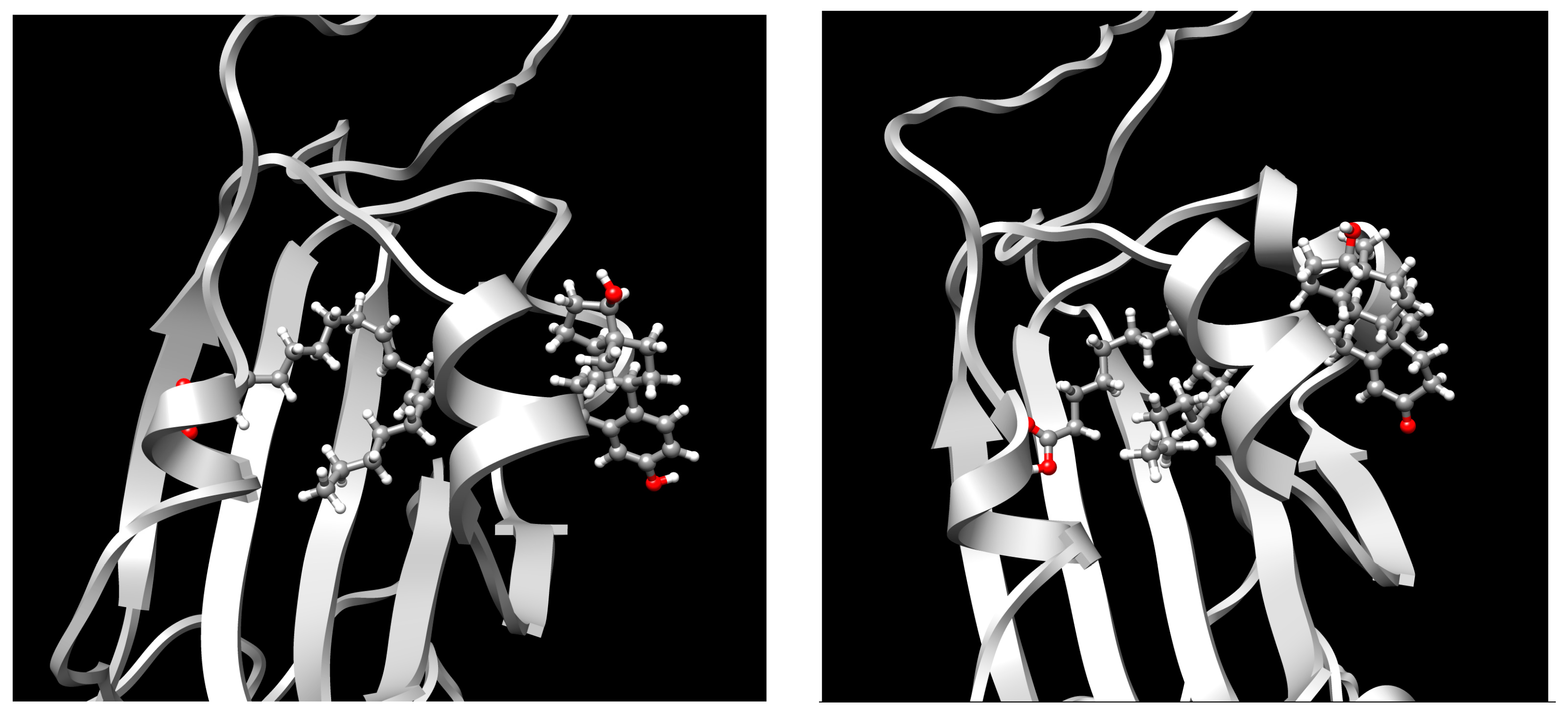
2. Results
3. Discussion
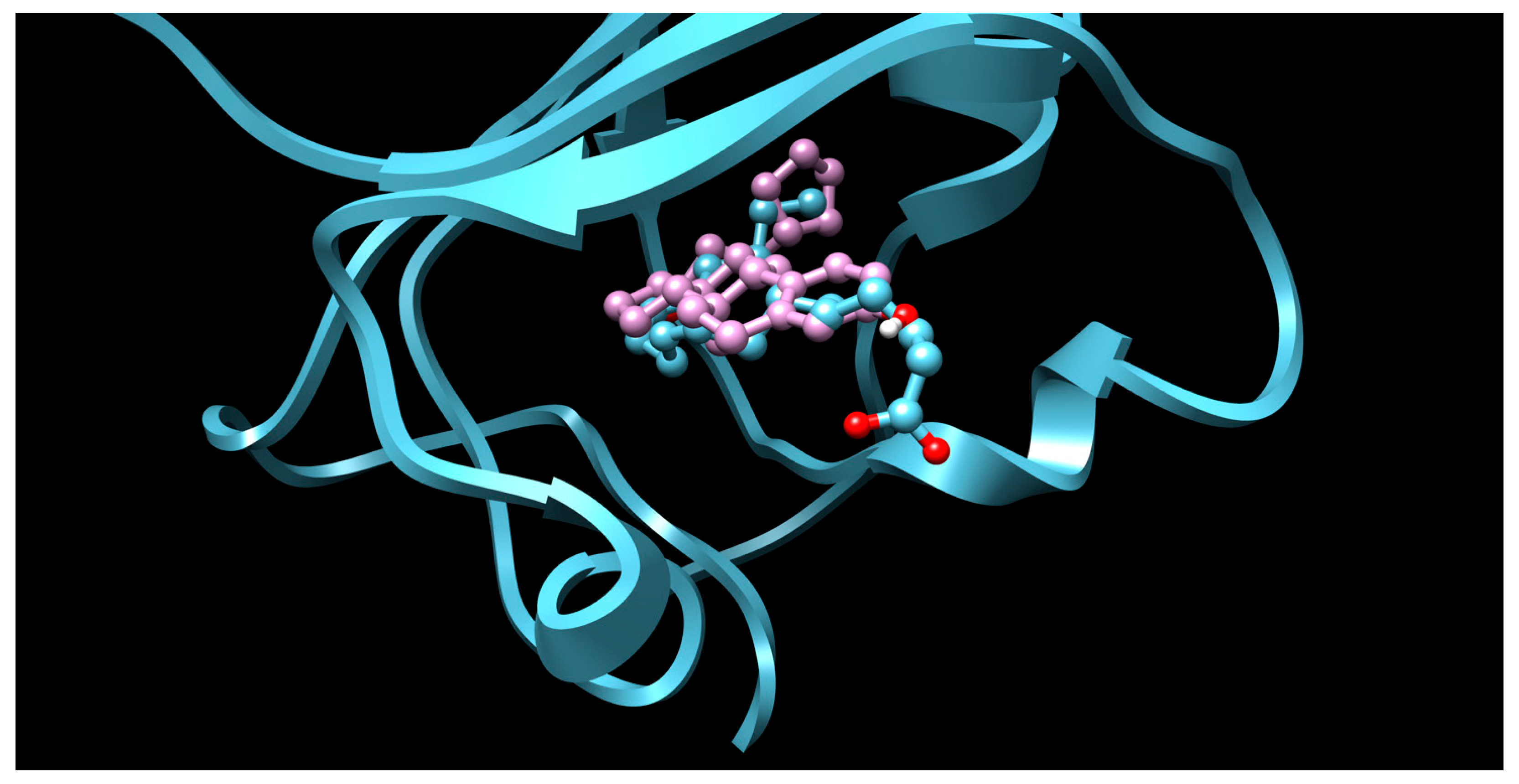
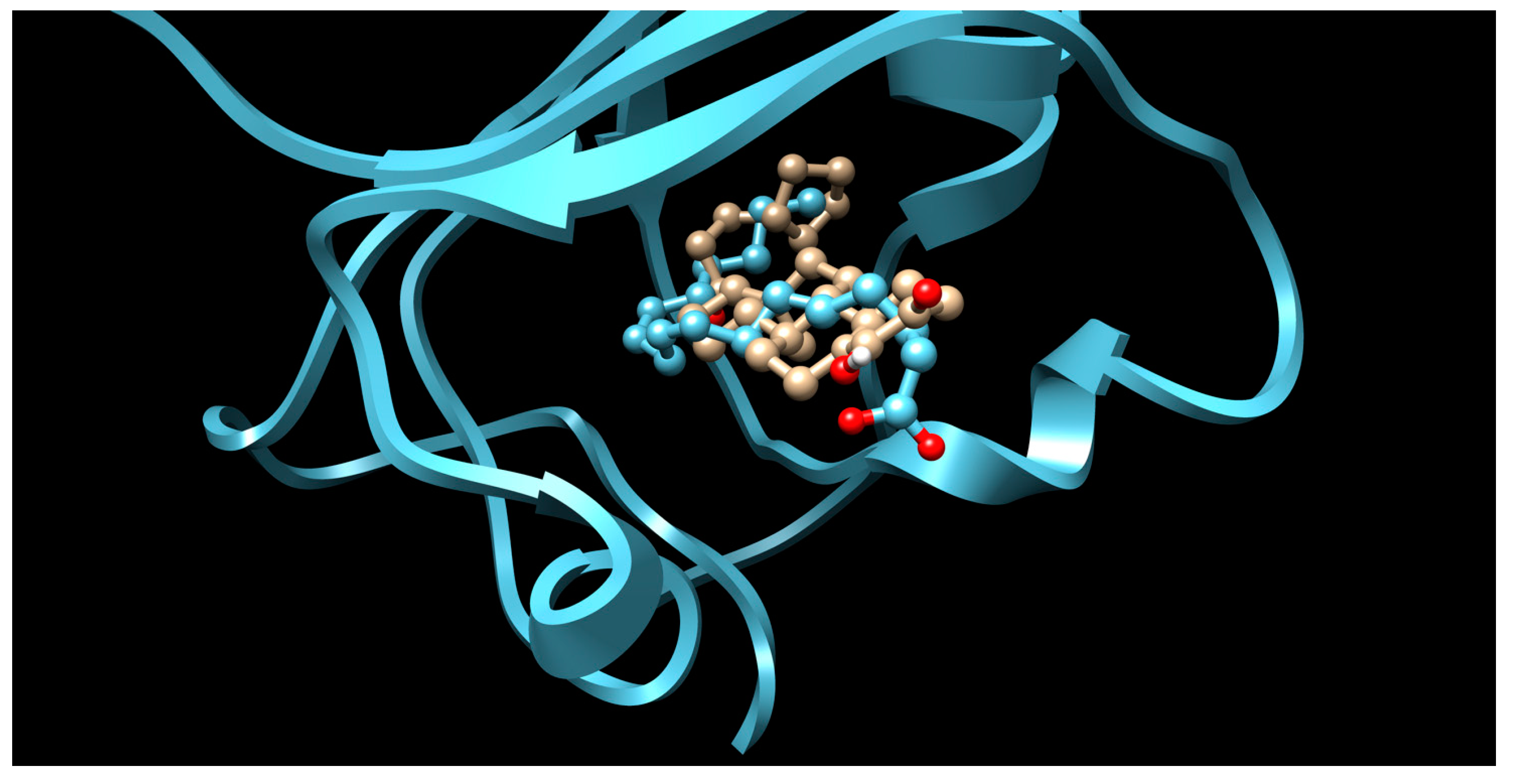
3.1. CNS Drugs
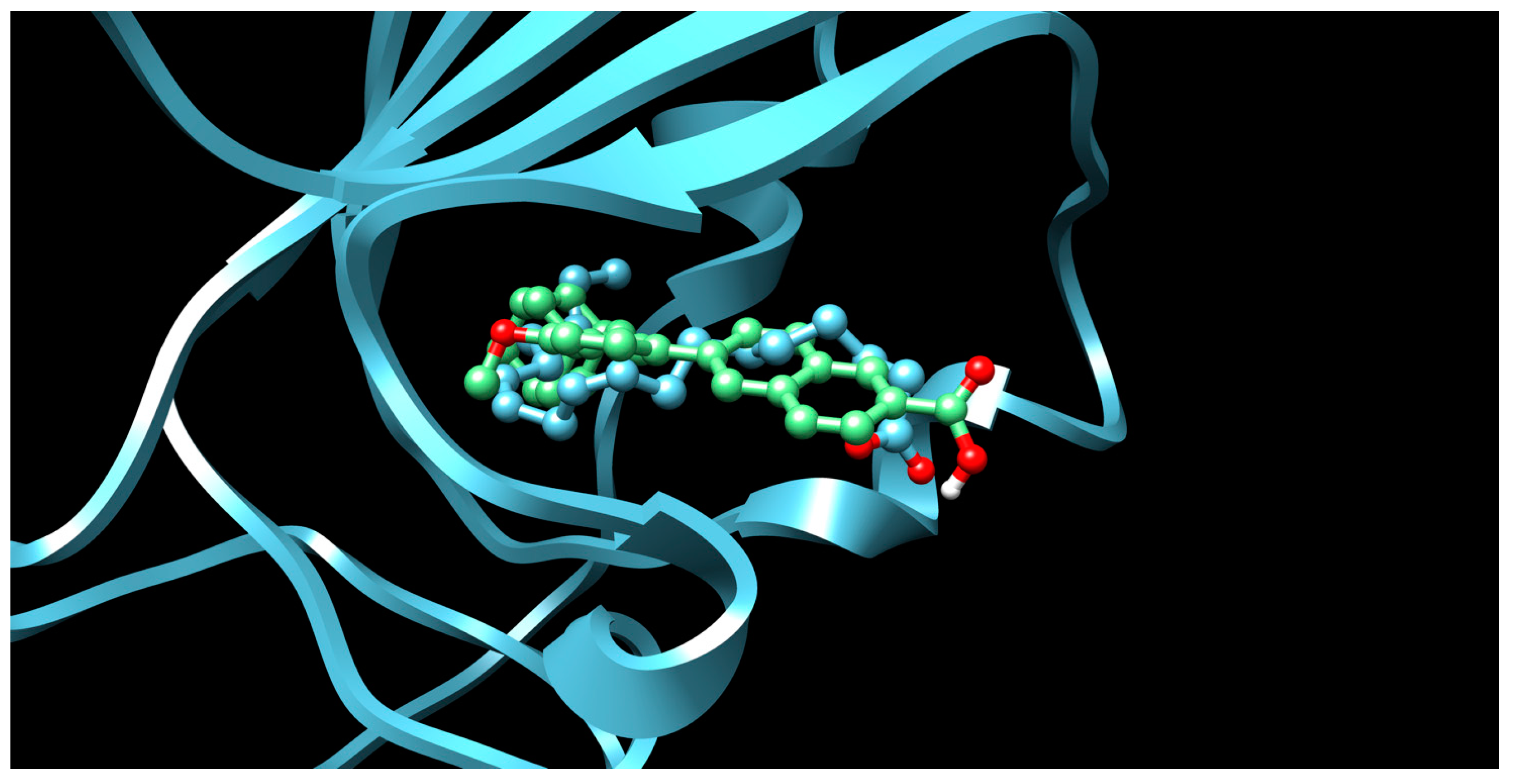
3.2. Sex Hormones and Hormone Analogues
3.3. Corticosteroids
3.4. Vitamin A, D, K, and Analogues
3.5. Cancer Drugs
3.6. Other Drug Classes
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pushpakom, S.; Iorio, F.; Eyers, P.A.; Escott, K.J.; Hopper, S.; Wells, A.; Doig, A.; Guilliams, T.; Latimer, J.; McNamee, C.; et al. Drug repurposing: Progress, challenges and recommendations. Nat. Rev. Drug Disc. 2019, 18, 41–58. [Google Scholar] [CrossRef]
- Du, L.; He, Y.; Zhou, Y.; Liu, S.; Zheng, B.J.; Jiang, S. The spike protein of SARS-CoV--a target for vaccine and therapeutic development. Nat. Rev. Microbiol. 2009, 7, 226–236. [Google Scholar] [CrossRef] [PubMed]
- Toelzer, C.; Gupta, K.; Yadav, S.K.N.; Borucu, U.; Davidson, A.D.; Kavanagh Williamson, M.; Shoemark, D.K.; Garzoni, F.; Staufer, O.; Milligan, R.; et al. Free fatty acid binding pocket in the locked structure of SARS-CoV-2 spike protein. Science 2020, 370, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Goc, A.; Niedzwiecki, A.; Rath, M. Polyunsaturated omega-3 fatty acids inhibit ACE2-controlled SARS-CoV-2 binding and cellular entry. Sci. Rep. 2021, 11, 5207. [Google Scholar] [CrossRef] [PubMed]
- Deganutti, G.; Prischi, F.; Reynolds, C.A. Supervised molecular dynamics for exploring the druggability of the SARS-CoV-2 spike protein. J. Comput. Aided Mol. Des. 2021, 35, 195–207. [Google Scholar] [CrossRef]
- Shoemark, D.K.; Colenso, C.K.; Toelzer, C.; Gupta, K.; Sessions, R.B.; Davidson, A.D.; Berger, I.; Schaffitzel, C.; Spencer, J.; Mulholland, A.J. Molecular simulations suggest vitamins, retinoids and steroids as ligands of the free fatty acid pocket of the SARS-CoV-2 spike protein. Angew. Chem. 2021, 133, 7174–7186. [Google Scholar] [CrossRef]
- Marion, I.U.; Marion, A. Molecular modelling reveals eight novel druggable binding sites in SARS-CoV-2′s spike protein. ChemRxiv 2020. [Google Scholar] [CrossRef]
- Piplani, S.; Singh, P.; Petrovsky, N.; Winkler, D.A. Computational repurposing of drugs and natural products Against SARS-CoV-2 main protease (M(pro)) as potential COVID-19 therapies. Front. Mol. Biosci. 2022, 9, 781039. [Google Scholar] [CrossRef]
- Piplani, S.; Singh, P.; Winkler, D.A.; Petrovsky, N. Potential COVID-19 Therapies from computational repurposing of drugs and natural products against the SARS-CoV-2 helicase. Int. J. Mol. Sci. 2022, 23, 7704. [Google Scholar] [CrossRef]
- Piplani, S.; Singh, P.K.; Winkler, D.A.; Petrovsky, N. In silico comparison of SARS-CoV-2 spike protein-ACE2 binding affinities across species and implications for virus origin. Sci. Rep. 2021, 11, 13063. [Google Scholar] [CrossRef]
- Piplani, S.; Singh, P.K.; Winkler, D.A.; Petrovsky, N. Computationally repurposed drugs and natural products against RNA dependent RNA polymerase as potential COVID-19 therapies. Mol. Biomed. 2021, 2, 28. [Google Scholar] [CrossRef] [PubMed]
- Lecanu, L.; Tillement, L.; Rammouz, G.; Tillement, J.P.; Greeson, J.; Papadopoulos, V. Caprospinol: Moving from a neuroactive steroid to a neurotropic drug. Expert Opin. Investig. Drugs 2009, 18, 265–276. [Google Scholar] [CrossRef]
- Le, B.L.; Andreoletti, G.; Oskotsky, T.; Vallejo-Gracia, A.; Rosales, R.; Yu, K.; Kosti, I.; Leon, K.E.; Bunis, D.G.; Li, C.; et al. Transcriptomics-based drug repositioning pipeline identifies therapeutic candidates for COVID-19. Sci. Rep. 2021, 11, 12310. [Google Scholar] [CrossRef]
- Lu, J.; Hou, Y.; Ge, S.; Wang, X.; Wang, J.; Hu, T.; Lv, Y.; He, H.; Wang, C. Screened antipsychotic drugs inhibit SARS-CoV-2 binding with ACE2 in vitro. Life Sci. 2021, 266, 118889. [Google Scholar] [CrossRef]
- Villoutreix, B.O.; Krishnamoorthy, R.; Tamouza, R.; Leboyer, M.; Beaune, P. Chemoinformatic analysis of psychotropic and antihistaminic drugs in the light of experimental anti-SARS-CoV-2 activities. Adv. Appl. Bioinform. Chem. 2021, 14, 71–85. [Google Scholar] [CrossRef] [PubMed]
- Thurakkal, L.; Singh, S.; Roy, R.; Kar, P.; Sadhukhan, S.; Porel, M. An in-silico study on selected organosulfur compounds as potential drugs for SARS-CoV-2 infection via binding multiple drug targets. Chem. Phys. Lett. 2021, 763, 138193. [Google Scholar] [CrossRef]
- Milani, M.; Donalisio, M.; Bonotto, R.M.; Schneider, E.; Arduino, I.; Boni, F.; Lembo, D.; Marcello, A.; Mastrangelo, E. Combined in silico and in vitro approaches identified the antipsychotic drug lurasidone and the antiviral drug elbasvir as SARS-CoV2 and HCoV-OC43 inhibitors. Antiviral Res. 2021, 189, 105055. [Google Scholar] [CrossRef] [PubMed]
- Jan, J.-T.; Cheng, T.-J.R.; Juang, Y.-P.; Ma, H.-H.; Wu, Y.-T.; Yang, W.-B.; Cheng, C.-W.; Chen, X.; Chou, T.-H.; Shie, J.-J. Identification of existing pharmaceuticals and herbal medicines as inhibitors of SARS-CoV-2 infection. Proc. Natl. Acad. Sci. USA 2021, 118, e2021579118. [Google Scholar] [CrossRef]
- Weston, S.; Coleman, C.M.; Haupt, R.; Logue, J.; Matthews, K.; Li, Y.; Reyes, H.M.; Weiss, S.R.; Frieman, M.B. Broad anti-coronavirus activity of food and drug administration-approved drugs against SARS-CoV-2 in vitro and SARS-CoV in vivo. J. Virol. 2020, 94, e01218–e01220. [Google Scholar] [CrossRef] [PubMed]
- Xiang, R.; Yu, Z.; Wang, Y.; Wang, L.; Huo, S.; Li, Y.; Liang, R.; Hao, Q.; Ying, T.; Gao, Y.; et al. Recent advances in developing small-molecule inhibitors against SARS-CoV-2. Acta Pharmaceut. Sin. B 2022, 12, 1591–1623. [Google Scholar] [CrossRef]
- Dyall, J.; Coleman, C.M.; Hart, B.J.; Venkataraman, T.; Holbrook, M.R.; Kindrachuk, J.; Johnson, R.F.; Olinger, G.G., Jr.; Jahrling, P.B.; Laidlaw, M.; et al. Repurposing of clinically developed drugs for treatment of Middle East respiratory syndrome coronavirus infection. Antimicrob. Agents Chemother. 2014, 58, 4885–4893. [Google Scholar] [CrossRef]
- Plaze, M.; Attali, D.; Petit, A.C.; Blatzer, M.; Simon-Loriere, E.; Vinckier, F.; Cachia, A.; Chrétien, F.; Gaillard, R. Repurposing of chlorpromazine in COVID-19 treatment: The reCoVery study. Encephale 2020, 46, S35–S39. [Google Scholar] [CrossRef]
- Camici, M.; Zuppi, P.; Lorenzini, P.; Scarnecchia, L.; Pinnetti, C.; Cicalini, S.; Nicastri, E.; Petrosillo, N.; Palmieri, F.; D’Offizi, G.; et al. Role of testosterone in SARS-CoV-2 infection: A key pathogenic factor and a biomarker for severe pneumonia. Int. J. Infect. Dis. 2021, 108, 244–251. [Google Scholar] [CrossRef]
- Wu, C.; Chen, X.; Cai, Y.; Xia, J.; Zhou, X.; Xu, S.; Huang, H.; Zhang, L.; Zhou, X.; Du, C.; et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern. Med. 2020, 180, 934–943. [Google Scholar] [CrossRef] [PubMed]
- Rastrelli, G.; Di Stasi, V.; Inglese, F.; Beccaria, M.; Garuti, M.; Di Costanzo, D.; Spreafico, F.; Greco, G.F.; Cervi, G.; Pecoriello, A.; et al. Low testosterone levels predict clinical adverse outcomes in SARS-CoV-2 pneumonia patients. Androl. 2021, 9, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Tsegay, K.B.; Adeyemi, C.M.; Gniffke, E.P.; Sather, D.N.; Walker, J.K.; Smith, S.E.P. A Repurposed drug screen identifies compounds that inhibit the binding of the COVID-19 spike protein to ACE2. Front. Pharmacol. 2021, 12, 685308. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Pan, X.; Huang, Y.; Cheng, C.; Xu, X.; Wu, Y.; Xu, Y.; Shang, W.; Niu, X.; Wan, Y.; et al. Drug repurposing of itraconazole and estradiol benzoate against COVID-19 by blocking SARS-CoV-2 spike protein-mediated membrane fusion. Adv. Therapeut. 2021, 4, 2000224. [Google Scholar] [CrossRef] [PubMed]
- Furnsinn, C.; Nowotny, P.; Brunmair, B.; Gras, F.; Roden, M.; Waldhausl, W.; Vierhapper, H. Thiazolidinediones influence plasma steroids of male obese Zucker rats. Endocrinol. 2002, 143, 327. [Google Scholar] [CrossRef]
- Angus, D.C.; Derde, L.; Al-Beidh, F.; Annane, D.; Arabi, Y.; Beane, A.; van Bentum-Puijk, W.; Berry, L.; Bhimani, Z.; Bonten, M.; et al. Effect of hydrocortisone on mortality and organ support in patients with severe COVID-19: The REMAP-CAP COVID-19 corticosteroid domain randomized clinical trial. JAMA 2020, 324, 1317–1329. [Google Scholar] [CrossRef]
- Pinna, G. Sex and COVID-19: A protective role for reproductive steroids. Trends Endocrinol. Metab. 2021, 32, 3–6. [Google Scholar] [CrossRef]
- Patten, J.J.; Keiser, P.T.; Morselli-Gysi, D.; Menichetti, G.; Mori, H.; Donahue, C.J.; Gan, X.; Valle, I.d.; Geoghegan-Barek, K.; Anantpadma, M.; et al. Identification of potent inhibitors of SARS-CoV-2 infection by combined pharmacological evaluation and cellular network prioritization. iScience 2022, 25, 104925. [Google Scholar] [CrossRef]
- Mansouri, A.; Kowsar, R.; Zakariazadeh, M.; Hakimi, H.; Miyamoto, A. The impact of calcitriol and estradiol on the SARS-CoV-2 biological activity: A molecular modeling approach. Sci. Rep. 2022, 12, 717. [Google Scholar] [CrossRef] [PubMed]
- Mok, C.K.; Ng, Y.L.; Ahidjo, B.A.; Hua Lee, R.C.; Choy Loe, M.W.; Liu, J.; Tan, K.S.; Kaur, P.; Chng, W.J.; Wong, J.E.-L.; et al. Calcitriol, the active form of vitamin D, is a promising candidate for COVID-19 prophylaxis. bioRxiv 2020. [Google Scholar] [CrossRef]
- Ilie, P.C.; Stefanescu, S.; Smith, L. The role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality. Aging Clin. Exp. Res. 2020, 32, 1195–1198. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Lidsky, P.V.; Xiao, Y.; Wu, C.T.; Garcia-Knight, M.; Yang, J.; Nakayama, T.; Nayak, J.V.; Jackson, P.K.; Andino, R.; et al. Ethacridine inhibits SARS-CoV-2 by inactivating viral particles. PLoS Pathog. 2021, 17, e1009898. [Google Scholar] [CrossRef]
- Fidan, O.; Mujwar, S.; Kciuk, M. Discovery of adapalene and dihydrotachysterol as antiviral agents for the Omicron variant of SARS-CoV-2 through computational drug repurposing. Mol. Divers 2022, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Lau, E.Y.; Negrete, O.A.; Bennett, W.F.D.; Bennion, B.J.; Borucki, M.; Bourguet, F.; Epstein, A.; Franco, M.; Harmon, B.; He, S.; et al. Discovery of small-molecule inhibitors of SARS-CoV-2 proteins using a computational and experimental pipeline. Front. Mol. Biosci. 2021, 8, 678701. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.P.; Dirajlal-Fargo, S.; Durieux, J.C.; Tribout, H.; Labbato, D.; McComsey, G.A. Vitamin K & D deficiencies are independently associated with COVID-19 disease severity. Open Forum. Infect. Dis. 2021, 8, ofab408. [Google Scholar] [CrossRef]
- Kaimal, J.M.; Tampere, M.; Le, T.H.; Axelsson, U.; Xu, H.; Axelsson, H.; Bäckström, A.; Marabita, F.; Moussaud-Lamodière, E.; Njenda, D.; et al. Subcellular mapping of the protein landscape of SARS-CoV-2 infected cells for target-centric drug repurposing. bioRxiv 2022. [Google Scholar] [CrossRef]
- Day, C.J.; Bailly, B.; Guillon, P.; Dirr, L.; Jen, F.E.-C.; Spillings, B.L.; Mak, J.; von Itzstein, M.; Haselhorst, T.; Jennings, M.P. Multidisciplinary approaches identify compounds that bind to human ACE2 or SARS-CoV-2 spike protein as candidates to block SARS-CoV-2–ACE2 receptor interactions. MBio 2021, 12, e03620–e03681. [Google Scholar] [CrossRef]
- Mujwar, S. Computational repurposing of tamibarotene against triple mutant variant of SARS-CoV-2. Comp. Biol. Med. 2021, 136, 104748. [Google Scholar] [CrossRef] [PubMed]
- Manickavasagam, P. Spike protein of SARS-CoV-2: Impact of single amino acid mutation and effect of drug binding to the variant-in silico analysis. Preprints 2020, 2020080447. [Google Scholar] [CrossRef]
- Ellinger, B.; Bojkova, D.; Zaliani, A.; Cinatl, J.; Claussen, C.; Westhaus, S.; Reinshagen, J.; Kuzikov, M.; Wolf, M.; Geisslinger, G. Identification of inhibitors of SARS-CoV-2 in-vitro cellular toxicity in human (Caco-2) cells using a large scale drug repurposing collection. 2020, Research Square, s.3.rs-23951/v1. 20 April 2020, PREPRINT (Version 1). Available online: https://www.researchsquare.com/article/rs-23951/v1 (accessed on 24 May 2022).
- Romeo, A.; Iacovelli, F.; Falconi, M. Targeting the SARS-CoV-2 spike glycoprotein prefusion conformation: Virtual screening and molecular dynamics simulations applied to the identification of potential fusion inhibitors. Virus Res. 2020, 286, 198068. [Google Scholar] [CrossRef] [PubMed]
- Cagno, V.; Magliocco, G.; Tapparel, C.; Daali, Y. The tyrosine kinase inhibitor nilotinib inhibits SARS-CoV-2 in vitro. Basic Clin. Pharmacol. Toxicol. 2021, 128, 621–624. [Google Scholar] [CrossRef]
- Dokainish, H.M.; Re, S.; Mori, T.; Kobayashi, C.; Jung, J.; Sugita, Y. The inherent flexibility of receptor binding domains in SARS-CoV-2 spike protein. eLife 2022, 11, e75720. [Google Scholar] [CrossRef]
- Sales-Medina, D.F.; Ferreira, L.R.P.; Romera, L.M.D.; Gonçalves, K.R.; Guido, R.V.C.; Courtemanche, G.; Buckeridge, M.S.; Durigon, É.L.; Moraes, C.B.; Freitas-Junior, L.H. Discovery of clinically approved drugs capable of inhibiting SARS-CoV-2 in vitro infection using a phenotypic screening strategy and network-analysis to predict their potential to treat covid-19. bioRxiv 2020. [Google Scholar] [CrossRef]
- Xiong, R.; Zhang, L.; Li, S.; Sun, Y.; Ding, M.; Wang, Y.; Zhao, Y.; Wu, Y.; Shang, W.; Jiang, X.; et al. Novel and potent inhibitors targeting DHODH are broad-spectrum antivirals against RNA viruses including newly-emerged coronavirus SARS-CoV-2. Protein Cell 2020, 11, 723–739. [Google Scholar] [CrossRef]
- Awad, I.E.; Abu-Saleh, A.A.A.; Sharma, S.; Yadav, A.; Poirier, R.A. High-throughput virtual screening of drug databanks for potential inhibitors of SARS-CoV-2 spike glycoprotein. J. Biomol. Struct. Dyn. 2022, 40, 2099–2112. [Google Scholar] [CrossRef]
- Lazniewski, M.; Dermawan, D.; Hidayat, S.; Muchtaridi, M.; Dawson, W.K.; Plewczynski, D. Drug repurposing for identification of potential spike inhibitors for SARS-CoV-2 using molecular docking and molecular dynamics simulations. Methods 2022, 203, 498–510. [Google Scholar] [CrossRef]
- Singh, A.; Dhar, R. A large-scale computational screen identifies strong potential inhibitors for disrupting SARS-CoV-2 S-protein and human ACE2 interaction. J. Biomol. Struct. Dynam. 2021, 40, 9004–9017. [Google Scholar] [CrossRef] [PubMed]
- Imamura, K.; Sakurai, Y.; Enami, T.; Shibukawa, R.; Nishi, Y.; Ohta, A.; Shu, T.; Kawaguchi, J.; Okada, S.; Hoenen, T.; et al. iPSC screening for drug repurposing identifies anti-RNA virus agents modulating host cell susceptibility. FEBS Open Bio 2021, 11, 1452–1464. [Google Scholar] [CrossRef]
- Kouznetsova, V.L.; Zhang, A.; Miller, M.A.; Tatineni, M.; Greenberg, J.P.; Tsigelny, I.F. Potential SARS-CoV-2 spike protein-ACE2 interface inhibitors: Repurposing FDA-approved drugs. J. Explor. Res. Pharmacol. 2022, 7, 17–29. [Google Scholar] [CrossRef]
- Liu, D.; Wu, P.; Gu, W.; Yang, C.; Yang, X.; Deng, X.; Xu, J.; Jiang, J.; Jiang, C. Potential of angiotensin II receptor blocker telmisartan in reducing mortality among hospitalized patients with COVID-19 compared with recommended drugs. Cell Discov. 2022, 8, 91. [Google Scholar] [CrossRef]
- Duarte, M.; Pelorosso, F.; Nicolosi, L.N.; Salgado, M.V.; Vetulli, H.; Aquieri, A.; Azzato, F.; Castro, M.; Coyle, J.; Davolos, I.; et al. Telmisartan for treatment of Covid-19 patients: An open multicenter randomized clinical trial. eClinicalMedicine 2021, 37, 100962. [Google Scholar] [CrossRef]
- Dhaka, P.; Singh, A.; Choudhary, S.; Kumar, P.; Sharma, G.K.; Tomar, S. Discovery of anti-SARS-CoV-2 molecules using structure-assisted repurposing approach targeting N-protein. bioRxiv 2022. [Google Scholar] [CrossRef]
- Pickard, A.; Calverley, B.C.; Chang, J.; Garva, R.; Gago, S.; Lu, Y.; Kadler, K.E. Discovery of re-purposed drugs that slow SARS-CoV-2 replication in human cells. PLoS Pathog. 2021, 17, e1009840. [Google Scholar] [CrossRef] [PubMed]
- Bakowski, M.A.; Beutler, N.; Wolff, K.C.; Kirkpatrick, M.G.; Chen, E.; Nguyen, T.H.; Riva, L.; Shaabani, N.; Parren, M.; Ricketts, J.; et al. Drug repurposing screens identify chemical entities for the development of COVID-19 interventions. Nat. Commun. 2021, 12, 3309. [Google Scholar] [CrossRef] [PubMed]
- Trezza, A.; Iovinelli, D.; Santucci, A.; Prischi, F.; Spiga, O. An integrated drug repurposing strategy for the rapid identification of potential SARS-CoV-2 viral inhibitors. Sci. Rep. 2020, 10, 13866. [Google Scholar] [CrossRef]
- Arshad, U.; Pertinez, H.; Box, H.; Tatham, L.; Rajoli, R.K.R.; Curley, P.; Neary, M.; Sharp, J.; Liptrott, N.J.; Valentijn, A.; et al. Prioritization of anti-SARS-Cov-2 drug repurposing opportunities based on plasma and target site concentrations derived from their established human pharmacokinetics. Clin. Pharmacol. Ther. 2020, 108, 775–790. [Google Scholar] [CrossRef]
- Behera, S.K.; Mahapatra, N.; Tripathy, C.S.; Pati, S. Drug repurposing for identification of potential inhibitors against SARS-CoV-2 spike receptor-binding domain: An in silico approach. Indian J. Med. Res. 2021, 153, 132–143. [Google Scholar] [CrossRef]
- Carter-Timofte, M.E.; Arulanandam, R.; Kurmasheva, N.; Fu, K.; Laroche, G.; Taha, Z.; van der Horst, D.; Cassin, L.; van der Sluis, R.M.; Palermo, E.; et al. Antiviral Potential of the antimicrobial drug atovaquone against SARS-CoV-2 and emerging variants of concern. ACS Infect. Dis. 2021, 7, 3034–3051. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.; Farag, A.; Wang, P.; Boys, I.N.; Eitson, J.L.; Ohlson, M.B.; Fan, W.; McDougal, M.B.; Schoggins, J.W.; Sadek, H. Identification of Atovaquone and Mebendazole as Repurposed Drugs with Antiviral Activity against SARS-CoV-2; Cambridge Open Engage: Cambridge, UK, 2021. [Google Scholar] [CrossRef]
- Staufer, O.; Gupta, K.; Hernandez Bücher, J.E.; Kohler, F.; Sigl, C.; Singh, G.; Vasileiou, K.; Yagüe Relimpio, A.; Macher, M.; Fabritz, S.; et al. Synthetic virions reveal fatty acid-coupled adaptive immunogenicity of SARS-CoV-2 spike glycoprotein. Nat. Commun. 2022, 13, 868. [Google Scholar] [CrossRef]
- Hardy, E.; Fernandez-Patron, C. Could Endogenous glucocorticoids influence SARS-CoV-2 infectivity? Cells 2022, 11, 2955. [Google Scholar] [CrossRef] [PubMed]
- Sarker, H.; Panigrahi, R.; Hardy, E.; Glover, J.N.M.; Elahi, S.; Fernandez-Patron, C. Glucocorticoids bind to SARS-CoV-2 S1 at multiple sites causing cooperative inhibition of SARS-CoV-2 S1 interaction with ACE2. Front. Immunol. 2022, 13, 906687. [Google Scholar] [CrossRef] [PubMed]
- McNaughton, C.D.; Adams, N.M.; Hirschie Johnson, C.; Ward, M.J.; Schmitz, J.E.; Lasko, T.A. Diurnal variation in SARS-CoV-2 PCR test results: Test accuracy may vary by time of day. J. Biol. Rhythms 2021, 36, 595–601. [Google Scholar] [CrossRef]
- Zhuang, X.; Tsukuda, S.; Wrensch, F.; Wing, P.A.C.; Schilling, M.; Harris, J.M.; Borrmann, H.; Morgan, S.B.; Cane, J.L.; Mailly, L.; et al. The circadian clock component BMAL1 regulates SARS-CoV-2 entry and replication in lung epithelial cells. iScience 2021, 24, 103144. [Google Scholar] [CrossRef]
- Hou, T.; Wang, J.; Li, Y.; Wang, W. Assessing the Performance of the MM/PBSA and MM/GBSA Methods. 1. The accuracy of binding free energy calculations based on molecular dynamics simulations. J. Chem. Inf. Mod. 2011, 51, 69–82. [Google Scholar] [CrossRef]
- Breznik, M.; Ge, Y.; Bluck, J.P.; Briem, H.; Hahn, D.F.; Christ, C.D.; Mortier, J.; Mobley, D.L.; Meier, K. Prioritizing small sets of molecules for synthesis through in-silico tools: A comparison of common ranking methods. ChemMedChem 2023, 18, e202200425. [Google Scholar] [CrossRef]
- El Khoury, L.; Santos-Martins, D.; Sasmal, S.; Eberhardt, J.; Bianco, G.; Ambrosio, F.A.; Solis-Vasquez, L.; Koch, A.; Forli, S.; Mobley, D.L. Comparison of affinity ranking using AutoDock-GPU and MM-GBSA scores for BACE-1 inhibitors in the D3R Grand Challenge 4. J. Comp.-aided Mol. Des. 2019, 33, 1011–1020. [Google Scholar] [CrossRef]
- Muratov, E.N.; Amaro, R.; Andrade, C.H.; Brown, N.; Ekins, S.; Fourches, D.; Isayev, O.; Kozakov, D.; Medina-Franco, J.L.; Merz, K.M.; et al. A critical overview of computational approaches employed for COVID-19 drug discovery. Chem. Soc. Rev. 2021, 50, 9121–9151. [Google Scholar] [CrossRef]
| Drug | ΔG | Drug | ΔG | Drug | ΔG | Drug | ΔG |
|---|---|---|---|---|---|---|---|
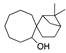 Caprospinol | −38.89 | 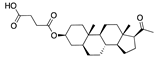 5-α-pregnane−3-β-ol hemisuccinate | −29.60 | 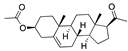 Pregnenolone acetate | −27.75 |  Telmisartan | −23.88 |
 Indacaterol | −35.99 |  Metixene | −29.50 |  Altrenogest | −27.58 | 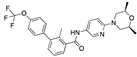 Sonidegib | −23.72 |
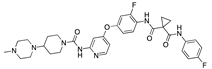 Golvatinib | −35.37 | 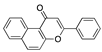 β-Naphthoflavone | −29.32 | 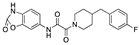 Radiprodil | −27.40 | 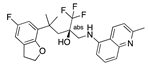 Mapracorat | −23.29 |
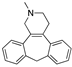 Setiptiline | −35.04 | 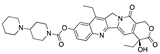 Irinotecan | −29.24 | 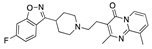 Ocaperidone | −27.36 |  Zanapezil | −23.16 |
 Oxabolone cypionate | −34.78 |  Revefenacin | −29.16 |  Regorafenib | −27.31 | 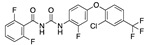 Flufenoxuron | −22.92 |
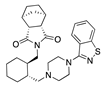 Lurasidone | −33.04 |  Hydroxycortisone (Cetacort) | −29.16 | 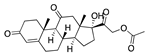 Cortisone acetate | −27.16 | 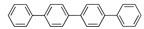 p-quaterphenyl | −22.62 |
 Cinacalcet | −33.02 |  Implitapide | −29.11 |  Tezacaftor | −26.85 |  Fedovapagon | −22.44 |
 Brequinar | −32.94 |  Betulin | −28.83 | 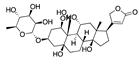 Ouabain | −26.83 | 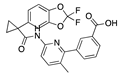 Lumacaftor | −22.33 |
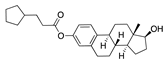 Estradiol cypionate | −32.94 |  Dehydrocholic acid | −28.82 | 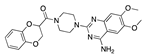 Doxazosin | −26.59 |  Tarazepide | −21.94 |
 Segesterone | −32.80 |  Tecarfarin | −28.80 |  Paliroden | −26.56 |  Xaliproden | −21.92 |
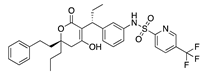 Tipranavir | −32.39 | 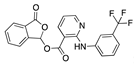 Talniflumate | −28.56 | 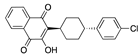 Mepron (atovaquone) | −26.35 | 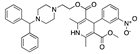 Manidipine | −21.63 |
 Nandrolone decanoate | −32.33 |  Gliquidone | −28.54 |  Aripiprazole | -25.96 |  Ponatinib | −21.19 |
 Arotinoid acid | −32.05 | 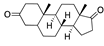 Androstanedione | −28.54 | 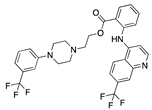 Antrafenine | −25.89 | 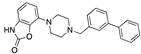 Bifeprunox | −21.16 |
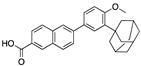 Adapalene | −32.03 |  Troglitazone | −28.53 |  Ezutromid | −25.86 |  Mubritinib | −20.81 |
 Flavoxate 4.3 0 5 6 | −31.70 |  Inecalcitol | −28.26 |  Cabozantinib | −25.81 | 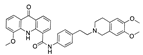 Elacridar | −20.28 |
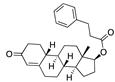 Nandrolone phenpropionate | −31.52 | 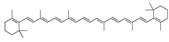 β-carotene | −28.26 | 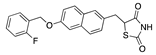 Netoglitazone | −25.53 | 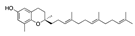 Tocotrienol | −20.11 |
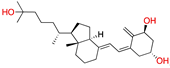 Calcitriol | −31.06 | 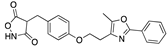 Reglitazar | −28.25 |  Etiocholanolone | −25.44 | 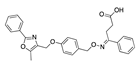 Imiglitazar | −20.09 |
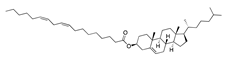 Cholesteryl linoleate | −30.98 | 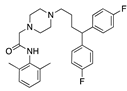 Lidoflazine | −28.11 | 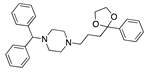 Dotarizine | −25.16 |  Difenpiramide | −19.56 |
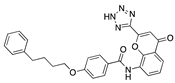 Pranlukast | −30.79 |  Desonide | −28.08 |  Mosapramine | −24.87 |  1-Testosterone | −19.31 |
 Vibegron | −30.72 |  Sorafenib | −28.04 |  Taladegib | −24.73 |  Sarizotan | −18.99 |
 Estradiol valerate | −30.60 |  Menatetrenone | −27.98 | 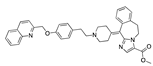 Laniquidar | −24.72 | 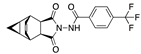 Tecovirimat | −17.77 |
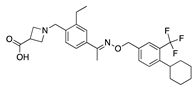 Siponimod | −30.58 |  Clobetasol propionate | −27.95 |  Penfluridol | −24.67 |  Emicerfont | −17.57 |
 (S)-famoxadone | −30.11 | 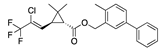 Bifenthrin | −27.78 | 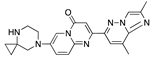 Risdiplam | −24.05 | 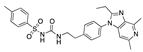 Grapiprant | −17.33 |
 Estradiol benzoate | −29.87 | 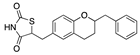 Englitazone | −27.75 | 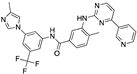 Nilotinib | −24.03 |  Lorpiprazole | −15.96 |
 Ethynodiol diacetate | −29.75 |  Pregnenolone acetate | −27.75 |  Fluspirilene | −23.95 |  Mardepodect | −15.31 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piplani, S.; Singh, P.; Petrovsky, N.; Winkler, D.A. Identifying SARS-CoV-2 Drugs Binding to the Spike Fatty Acid Binding Pocket Using In Silico Docking and Molecular Dynamics. Int. J. Mol. Sci. 2023, 24, 4192. https://doi.org/10.3390/ijms24044192
Piplani S, Singh P, Petrovsky N, Winkler DA. Identifying SARS-CoV-2 Drugs Binding to the Spike Fatty Acid Binding Pocket Using In Silico Docking and Molecular Dynamics. International Journal of Molecular Sciences. 2023; 24(4):4192. https://doi.org/10.3390/ijms24044192
Chicago/Turabian StylePiplani, Sakshi, Puneet Singh, Nikolai Petrovsky, and David A. Winkler. 2023. "Identifying SARS-CoV-2 Drugs Binding to the Spike Fatty Acid Binding Pocket Using In Silico Docking and Molecular Dynamics" International Journal of Molecular Sciences 24, no. 4: 4192. https://doi.org/10.3390/ijms24044192
APA StylePiplani, S., Singh, P., Petrovsky, N., & Winkler, D. A. (2023). Identifying SARS-CoV-2 Drugs Binding to the Spike Fatty Acid Binding Pocket Using In Silico Docking and Molecular Dynamics. International Journal of Molecular Sciences, 24(4), 4192. https://doi.org/10.3390/ijms24044192









