Larches of Kuzhanovo Have a Unique Mutation in the atpF–atpH Intergenic Spacer
Abstract
1. Introduction
2. Results
2.1. Flow Cytometry
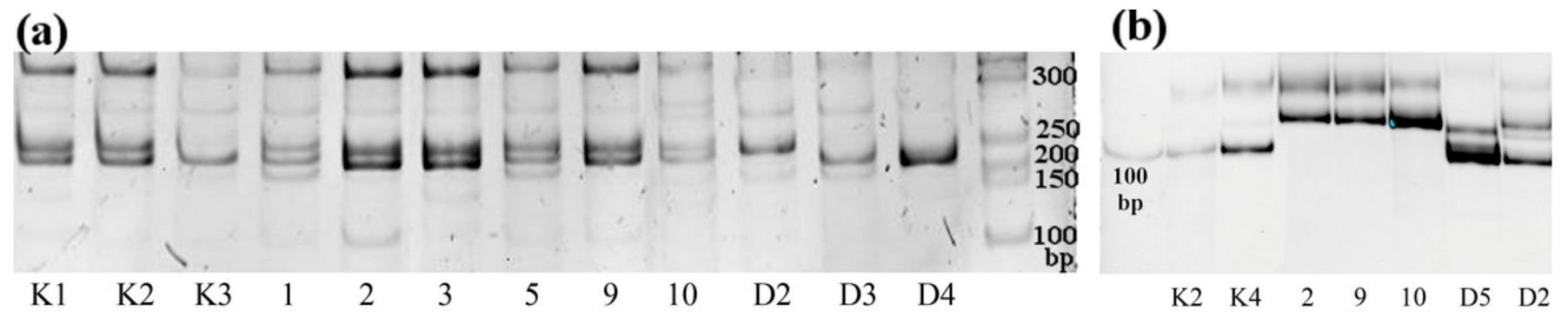
2.2. SSR and ISSR Analyses
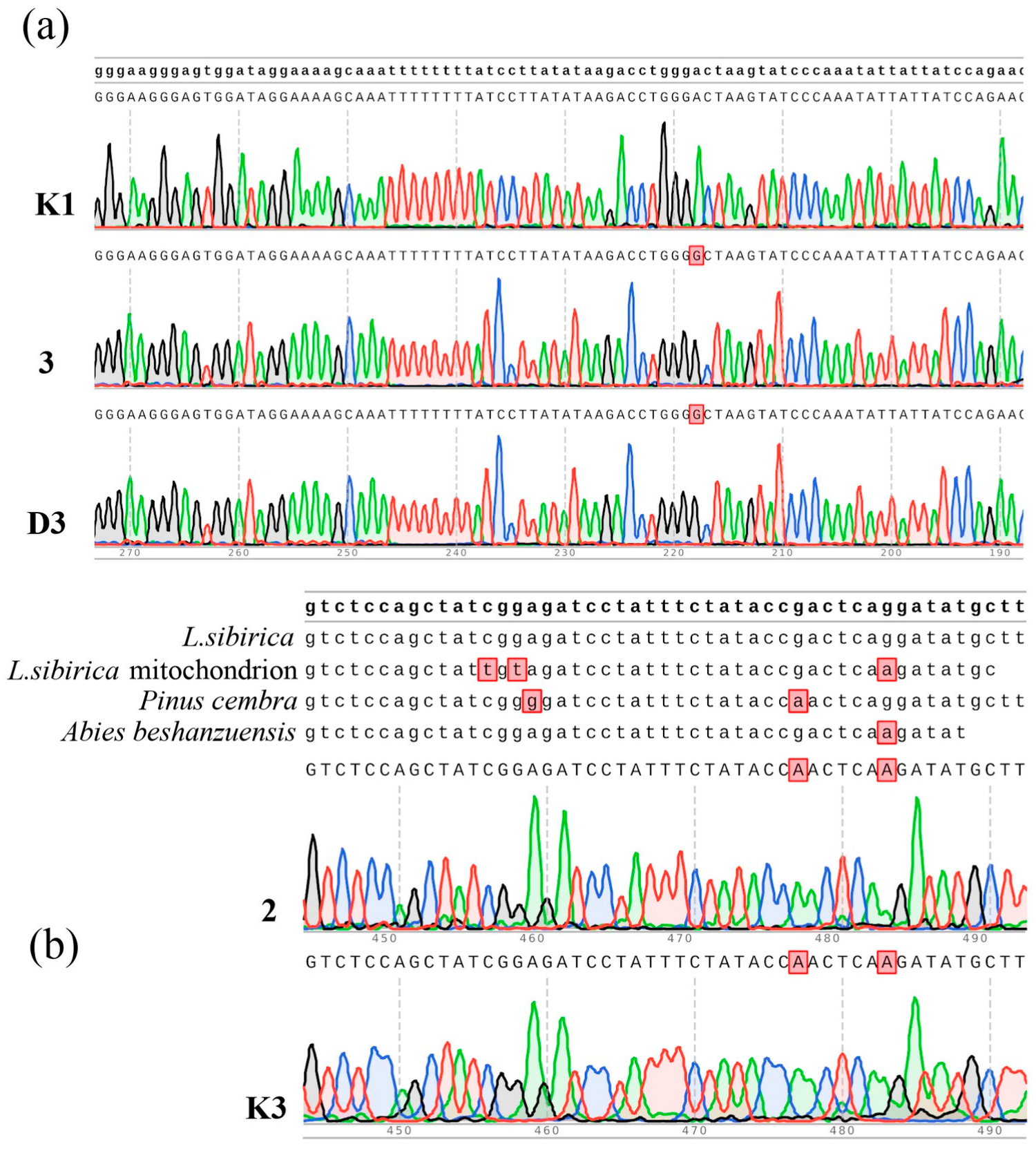
2.3. Analyses of Genetic Markers
2.4. Analyses of Candidate Genes
3. Discussion
4. Materials and methods
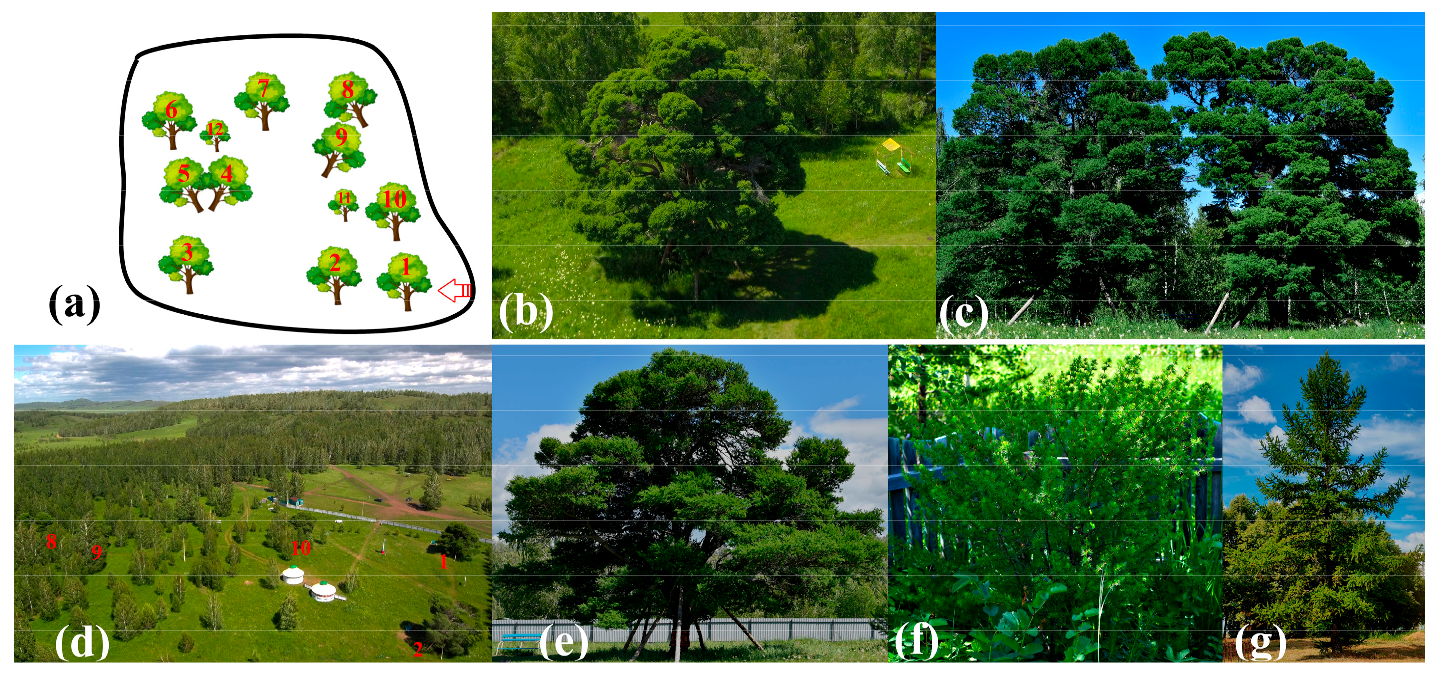
4.1. Plant Material
4.2. Ploidy Analysis
4.3. Genetic Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pâques, L.E.; Foffová, E.; Heinze, B.; Lelu-Walter, M.-A.; Liesebach, M.; Philippe, G. Larches (Larix Sp.). In Forest Tree Breeding in Europe; Pâques, L.E., Ed.; Managing Forest Ecosystems; Springer: Dordrecht, The Netherlands, 2013; Volume 25, pp. 13–122. ISBN 978-94-007-6145-2. [Google Scholar]
- Suyundukov, I.; Gerasimov, S.; Krivosheev, M.; Khabirov, I.; Ishbirdin, A.; Ishmuratova, M. Kuzhanovskie larches: Development of a protection strategy. In Proceedings of the Conference Humans and Nature-Interaction in Specially Protected Natural Areas, Novokuznetsk, Russia, 23–25 September 2021; pp. 178–180. [Google Scholar]
- Meirmans, P.G.; Gros-Louis, M.-C.; Lamothe, M.; Perron, M.; Bousquet, J.; Isabel, N. Rates of Spontaneous Hybridization and Hybrid Recruitment in Co-Existing Exotic and Native Mature Larch Populations. Tree Genet. Genomes 2014, 10, 965–975. [Google Scholar] [CrossRef]
- Sedel’nikova, T.; Muratova, E.; Pimenov, A. Variability of chromosome numbers in gymnosperms. Biol. Bull. Rev. 2011, 1, 100–109. [Google Scholar] [CrossRef]
- Dong, H.; Zhao, Z.; Yuan, Z. Genetic Mapping of Prince Rupprecht’s Larch (Larix Principis-Rupprechtii Mayr) by Specific-Locus Amplified Fragment Sequencing. Genes 2019, 10, 583. [Google Scholar] [CrossRef] [PubMed]
- Pâques, L.E.; Millier, F.; Rozenberg, P. Selection Perspectives for Genetic Improvement of Wood Stiffness in Hybrid Larch (Larix x Eurolepis Henry). Tree Genet. Genomes 2010, 6, 83–92. [Google Scholar] [CrossRef]
- Guo, Q.; Li, H.; Qian, Z.; Lu, J.; Zheng, W. Comparative Study on the Chloroplast Genomes of Five Larix Species from the Qinghai-Tibet Plateau and the Screening of Candidate DNA Markers. J. For. Res. 2021, 32, 2219–2226. [Google Scholar] [CrossRef]
- Wei, X.-X.; Wang, X.-Q. Phylogenetic Split of Larix: Evidence from Paternally Inherited CpDNA Trn T-Trn F Region. Plant Syst. Evol. 2003, 239, 67–77. [Google Scholar] [CrossRef]
- Araki, N.H.T.; Khatab, I.A.; Hemamali, K.K.G.U.; Inomata, N.; Wang, X.-R.; Szmidt, A.E. Phylogeography of Larix Sukaczewii Dyl. and Larix Sibirica L. Inferred from Nucleotide Variation of Nuclear Genes. Tree Genet. Genomes 2008, 4, 611–623. [Google Scholar] [CrossRef]
- Dong, M.; Wang, Z.; He, Q.; Zhao, J.; Fan, Z.; Zhang, J. Development of EST-SSR Markers in Larix Principis-Rupprechtii Mayr and Evaluation of Their Polymorphism and Cross-Species Amplification. Trees 2018, 32, 1559–1571. [Google Scholar] [CrossRef]
- Kulakov, E.; Sivolapov, V. Genetic variability of Sukachev’s larch (Larix sukaczewii djil.) in geographical cultures under Voronezh. For. Eng. J. 2018, 8, 37–44. [Google Scholar] [CrossRef]
- Caré, O.; Gailing, O.; Müller, M.; Krutovsky, K.V.; Leinemann, L. Crown Morphology in Norway Spruce (Picea Abies [Karst.] L.) as Adaptation to Mountainous Environments Is Associated with Single Nucleotide Polymorphisms (SNPs) in Genes Regulating Seasonal Growth Rhythm. Tree Genet. Genomes 2020, 16, 4. [Google Scholar] [CrossRef]
- Kuzmin, D.A.; Feranchuk, S.I.; Sharov, V.V.; Cybin, A.N.; Makolov, S.V.; Putintseva, Y.A.; Oreshkova, N.V.; Krutovsky, K.V. Stepwise Large Genome Assembly Approach: A Case of Siberian Larch (Larix Sibirica Ledeb). BMC Bioinform. 2019, 20, 37. [Google Scholar] [CrossRef]
- Sun, C.; Xie, Y.; Li, Z.; Liu, Y.; Sun, X.; Li, J.; Quan, W.; Zeng, Q.; Van de Peer, Y.; Zhang, S. The Larix Kaempferi Genome Reveals New Insights into Wood Properties. JIPB 2022, 64, 1364–1373. [Google Scholar] [CrossRef]
- Bondar, E.I.; Feranchuk, S.I.; Miroshnikova, K.A.; Sharov, V.V.; Kuzmin, D.A.; Oreshkova, N.V.; Krutovsky, K.V. Annotation of Siberian Larch (Larix Sibirica Ledeb.) Nuclear Genome—One of the Most Cold-Resistant Tree Species in the Only Deciduous GENUS in Pinaceae. Plants 2022, 11, 2062. [Google Scholar] [CrossRef]
- Sboeva, Y.; Vasilieva, U.; Chertov, N.; Pystogova, N.; Boronnikova, S.; Kalendar, R.; Martynenko, N. Molecular genetic identification of Scots pine and Siberian larch populations in Perm krai based on polymorphism of ISSR-PCR markers. SJFS 2020, 4, 35–44. [Google Scholar] [CrossRef]
- Zhou, Z.; Miwa, M.; Hogetsu, T. Analysis of Genetic Structure of a Suillus Grevillei Population in a Larix Kaempferi Stand by Polymorphism of Inter-Simple Sequence Repeat (ISSR). New Phytol. 1999, 144, 55–63. [Google Scholar] [CrossRef]
- Putintseva, Y.A.; Bondar, E.I.; Simonov, E.P.; Sharov, V.V.; Oreshkova, N.V.; Kuzmin, D.A.; Konstantinov, Y.M.; Shmakov, V.N.; Belkov, V.I.; Sadovsky, M.G.; et al. Siberian Larch (Larix Sibirica Ledeb.) Mitochondrial Genome Assembled Using Both Short and Long Nucleotide Sequence Reads Is Currently the Largest Known Mitogenome. BMC Genom. 2020, 21, 654. [Google Scholar] [CrossRef] [PubMed]
- Dyer, A.T.; Leonard, K.J. Contamination, Error, and Nonspecific Molecular Tools. Phytopathology 2000, 90, 565–567. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Ishizuka, W.; Hara, T.; Goto, S. Complete Chloroplast Genome of Japanese Larch (Larix Kaempferi): Insights into Intraspecific Variation with an Isolated Northern Limit Population. Forests 2020, 11, 884. [Google Scholar] [CrossRef]
- Bashalkhanov, S.I.; Konstantinov, Y.M.; Verbitskii, D.S.; Kobzev, V.F. Reconstruction of Phylogenetic Relationships of Larch Larix sukaczewii Dyl. Based on Chloroplast DNAtrnK Intron Sequences. Russ. J. Genet. 2003, 39, 1116–1120. [Google Scholar] [CrossRef]
- O’Brien, I.E.W.; Smith, D.R.; Gardner, R.C.; Murray, B.G. Flow Cytometric Determination of Genome Size in Pinus. Plant Sci. 1996, 115, 91–99. [Google Scholar] [CrossRef]
- Doležel, J.; Greilhuber, J.; Suda, J. Estimation of Nuclear DNA Content in Plants Using Flow Cytometry. Nat. Protoc. 2007, 2, 2233–2244. [Google Scholar] [CrossRef] [PubMed]
- Rogers, S.O.; Bendich, A.J. Extraction of DNA from Milligram Amounts of Fresh, Herbarium and Mummified Plant Tissues. Plant Mol Biol 1985, 5, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Matveeva, T.V.; Pavlova, O.A.; Bogomaz, D.I.; Demkovich, A.E.; Lutova, L.A. Molecular Markers for Plant Species Identification and Phylogenetics. Ecol. Genet. 2011, 9, 32–43. [Google Scholar] [CrossRef]

| Band Size, bp | Control Plants | Larches of Kuzhanovo | Distanced Larches | |||||
|---|---|---|---|---|---|---|---|---|
| 1000 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 0 |
| 900 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 800 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 0 |
| 760 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 0 |
| 600 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 |
| 550 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 1 |
| 510 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 1 |
| 450 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 0 |
| 400 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 1 |
| Primer Name | Forward Primer | Reverse Primer |
|---|---|---|
| Lar_eSSR11 | 5′-AATCCAAATTTCTGGACCCC-3′ | 5′-CCTGCAAAAAGAGGATAGCG-3′ |
| Lar_eSSR54 | 5′-GCGCGCTCTTCTTTTCTCT-3′ | 5′-CGCCGTCGACTGTATAACCT-3′ |
| Lar_eSSR69 | 5′-CAGCTGTAATGAATTCCGCA-3′ | 5′-GAAATGATGCAGGCAGAGGT-3′ |
| Lar_eSSR78F | 5′-CAATCCGATAAAACGCCATC-3′ | 5′-CAGTAACACTCCCGCCTAGC-3′ |
| Lar_eSSR96 | 5′-GCCTTCGCTGATCTGTTTTC-3′ | 5′-TGCTGGTCTCTGTTGTCGTC-3′ |
| Lar_eSSR111 | 5′-GATATCAACTCCCTGCGGAA-3′ | 5′-AGCTGTGAGCGAGAGAGAGG-3′ |
| Lar_eSSR115 | 5′-TTGTGATGCTTCTTTGACCG-3′ | 5′-TTGTGATGCTTCTTTGACCG-3′ |
| Lar_eSSR228 | 5′-CTCTCGTCCATTAAGCTGCC-3′ | 5′-GAGGATTGTGCACACCTTGA-3′ |
| bcLK056 | 5′-ATGGGCTAAGGTATGTTTTACG-3′ | 5′-TGCCAACATCTATACCAAGTCT-3′ |
| bcLK224 | 5′-GAGAGGCCACTACTATTATTAC-3′ | 5′-ATGCGTTCCTTCATTCCTCT-3′ |
| bcLK232 | 5′-TGTTGCTGGGTTGTTGTTAGA-3′ | 5′-GGGTAATAGTTCCAGTCTTTG-3′ |
| bcLK260 | 5′-CTCCATAAGGGGCATCACAT-3′ | 5′-TGGGCTCAAGTTTGGACATTA-3′ |
| bcLK235 | 5′-TTCACTTGTGATCCTAGAGTTAGA-3′ | 5′-AACCCCTAACCATATAATATCCA-3′ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Artyukhin, A.; Sharifyanova, Y.; Krivosheev, M.M.; Mikhaylova, E.V. Larches of Kuzhanovo Have a Unique Mutation in the atpF–atpH Intergenic Spacer. Int. J. Mol. Sci. 2023, 24, 3939. https://doi.org/10.3390/ijms24043939
Artyukhin A, Sharifyanova Y, Krivosheev MM, Mikhaylova EV. Larches of Kuzhanovo Have a Unique Mutation in the atpF–atpH Intergenic Spacer. International Journal of Molecular Sciences. 2023; 24(4):3939. https://doi.org/10.3390/ijms24043939
Chicago/Turabian StyleArtyukhin, Alexander, Yuliya Sharifyanova, Mikhail M. Krivosheev, and Elena V. Mikhaylova. 2023. "Larches of Kuzhanovo Have a Unique Mutation in the atpF–atpH Intergenic Spacer" International Journal of Molecular Sciences 24, no. 4: 3939. https://doi.org/10.3390/ijms24043939
APA StyleArtyukhin, A., Sharifyanova, Y., Krivosheev, M. M., & Mikhaylova, E. V. (2023). Larches of Kuzhanovo Have a Unique Mutation in the atpF–atpH Intergenic Spacer. International Journal of Molecular Sciences, 24(4), 3939. https://doi.org/10.3390/ijms24043939










