Mitochondrial Transfer into Human Oocytes Improved Embryo Quality and Clinical Outcomes in Recurrent Pregnancy Failure Cases
Abstract
1. Introduction
2. Results
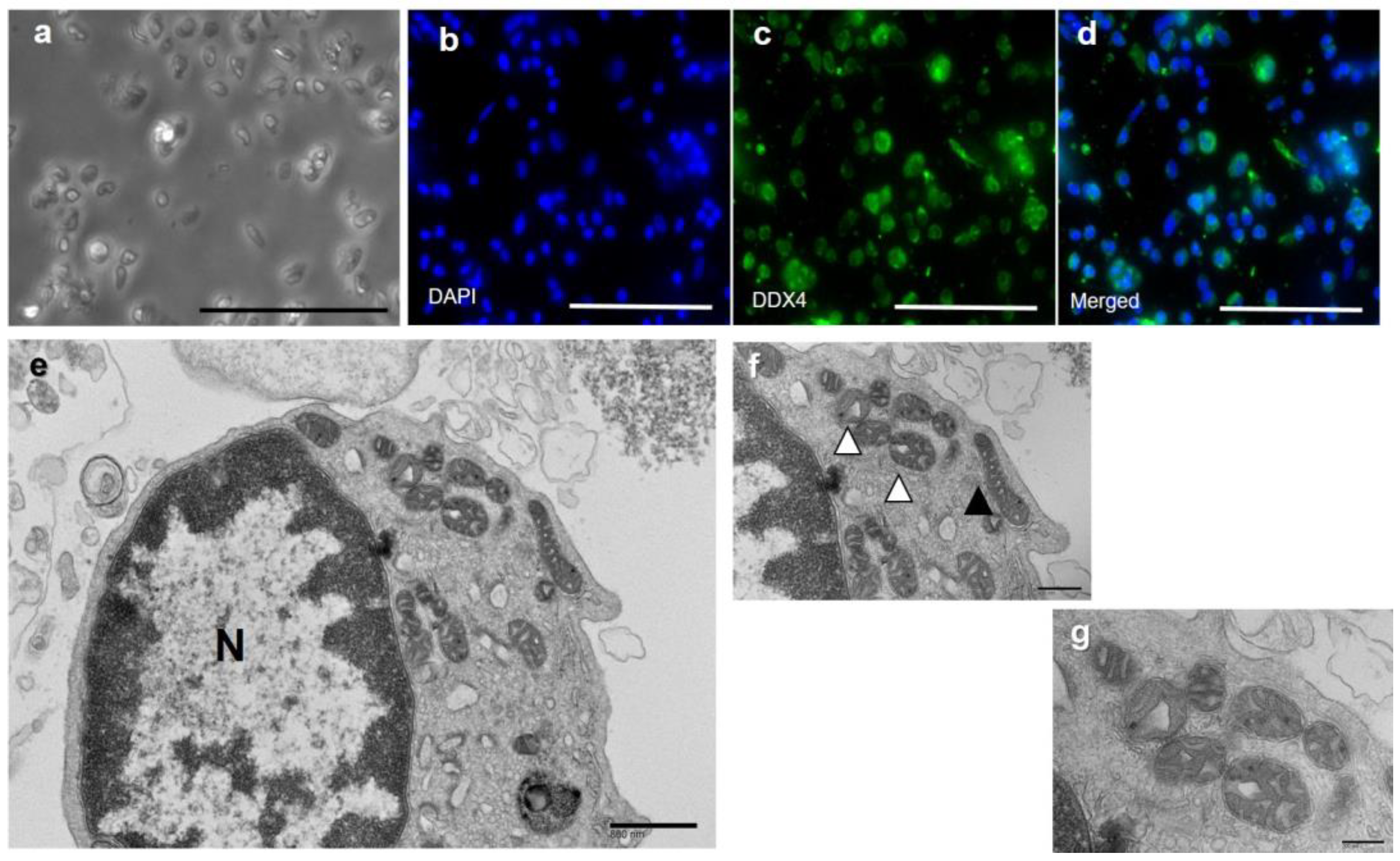
2.1. Cell Preparation: p-OSCs Were Successfully Isolated from Ovarian Cortical Tissues
2.2. Clinical Outcomes
2.3. Analysis of mtDNA Sequences of the Patients (Mothers) and Their Children Born after MT
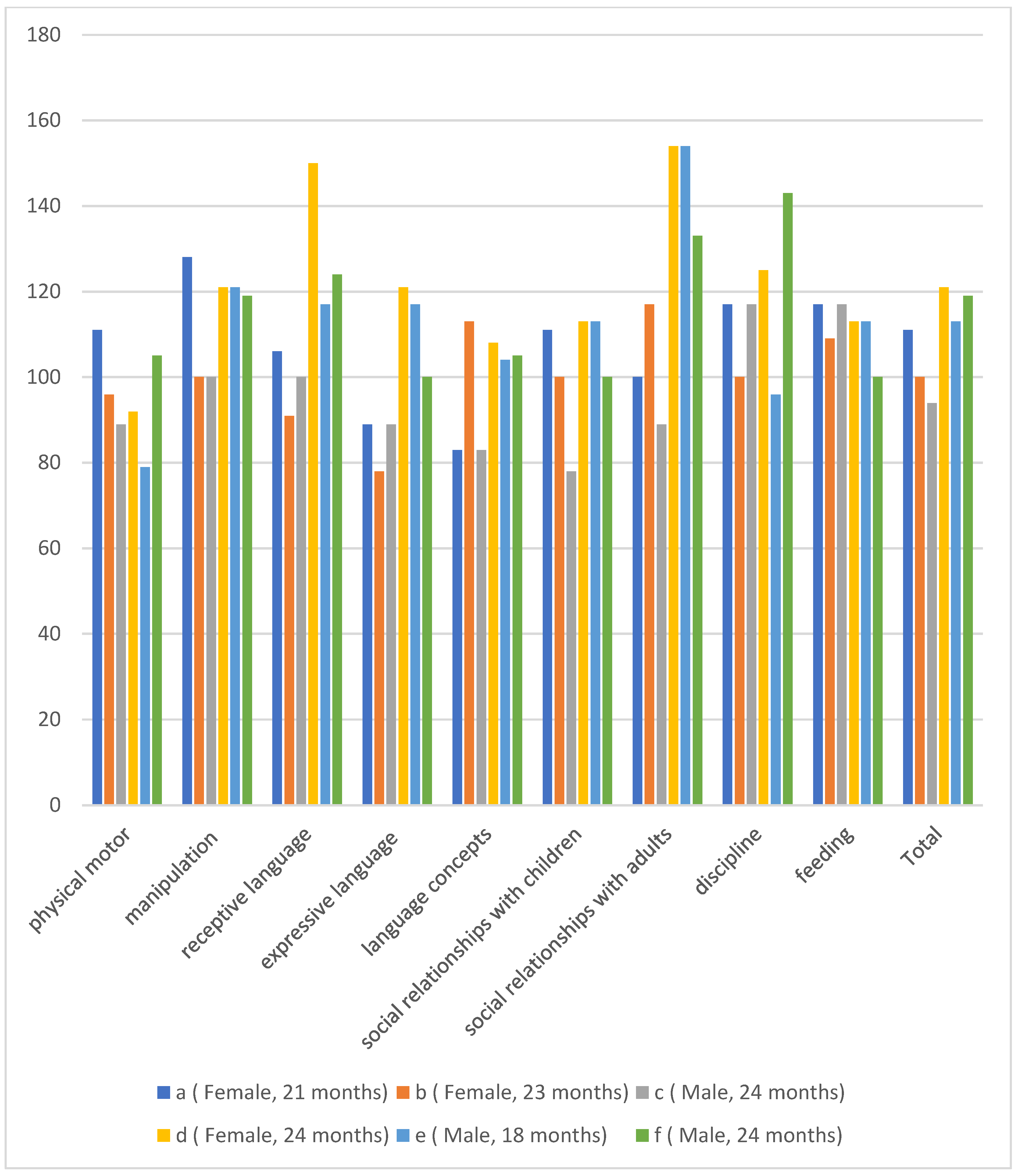
2.4. Follow-Up of Children Born after MT
3. Discussion
4. Materials and Methods
4.1. Study Design and Patient Selection
4.2. Evaluation of the Oxidative Stress Status of Patients
4.3. Isolation of p-OSCs, Extraction of Mitochondria from p-OSCs, and Injection of Mitochondria into Oocytes (Figure 3)
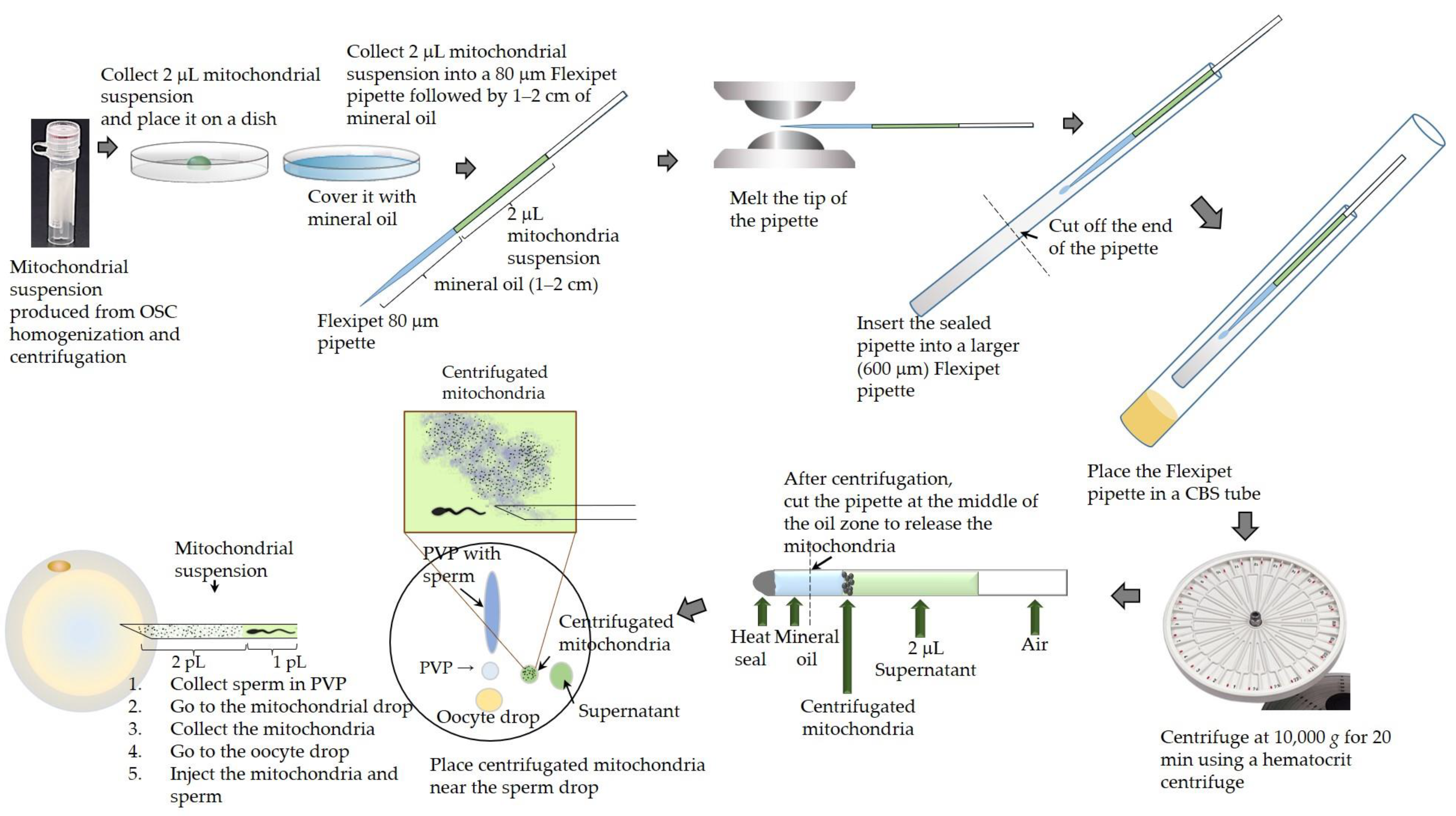
4.4. Ovarian Stimulation and Oocyte Retrieval
4.5. Embryo Transfer and Diagnosis of Pregnancy
4.6. Transmission Electron Microscopic Examination of p-OSCs
4.7. Comparison of mtDNA Sequences of Children and Their Mothers
4.7.1. DNA Extraction and Polymerase Chain Reaction (PCR) Amplification
4.7.2. Library Construction
4.7.3. Template Preparation and Sequencing
4.7.4. Data Analysis
4.8. Follow-Up of Children Born after MT
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Penzias, A.S. Recurrent IVF Failure: Other Factors. Fertil. Steril. 2012, 97, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Van Blerkom, J. Mitochondrial Function in the Human Oocyte and Embryo and Their Role in Developmental Competence. Mitochondrion 2011, 11, 797–813. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, G.; Majumdar, A.; Verma, I.; Upadhyaya, K. Relationship Between Morphology, Euploidy and Implantation Potential of Cleavage and Blastocyst Stage Embryos. J. Hum. Reprod. Sci. 2017, 10, 49–57. [Google Scholar] [CrossRef]
- Liu, L. Ageing-Associated Aberration in Meiosis of Oocytes from Senescence-Accelerated Mice. Hum. Reprod. 2002, 17, 2678–2685. [Google Scholar] [CrossRef]
- Kristensen, S.G.; Pors, S.E.; Andersen, C.Y. Improving Oocyte Quality by Transfer of Autologous Mitochondria from Fully Grown Oocytes. Hum. Reprod. 2017, 32, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Craven, L.; Tuppen, H.A.; Greggains, G.D.; Harbottle, S.J.; Murphy, J.L.; Cree, L.M.; Murdoch, A.P.; Chinnery, P.F.; Taylor, R.W.; Lightowlers, R.N.; et al. Pronuclear Transfer in Human Embryos to Prevent Transmission of Mitochondrial DNA Disease. Nature 2010, 465, 82–85. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, H.; Luo, S.; Lu, Z.; Chávez-Badiola, A.; Liu, Z.; Yang, M.; Merhi, Z.; Silber, S.J.; Munné, S.; et al. Live Birth Derived from Oocyte Spindle Transfer to Prevent Mitochondrial Disease. Reprod. Biomed. Online 2017, 34, 361–368. [Google Scholar] [CrossRef]
- Wang, T.; Sha, H.; Ji, D.; Zhang, H.L.; Chen, D.; Cao, Y.; Zhu, J. Polar Body Genome Transfer for Preventing the Transmission of Inherited Mitochondrial Diseases. Cell 2014, 157, 1591–1604. [Google Scholar] [CrossRef]
- Takeda, K.; Tasai, M.; Akagi, S.; Matsukawa, K.; Takahashi, S.; Iwamoto, M.; Srirattana, K.; Onishi, A.; Tagami, T.; Nirasawa, K.; et al. Microinjection of Serum-Starved Mitochondria Derived from Somatic Cells Affects Parthenogenetic Development of Bovine and Murine Oocytes. Mitochondrion 2010, 10, 137–142. [Google Scholar] [CrossRef]
- Barritt, J.A.; Willadsen, S.; Brenner, C.; Cohen, J. Epigenetic and Experimental Modifications in Early Mammalian Development: Part II. Hum. Reprod. Update 2001, 7, 428–435. [Google Scholar] [CrossRef]
- Igarashi, H.; Takahashi, T.; Abe, H.; Nakano, H.; Nakajima, O.; Nagase, S. Poor Embryo Development in Post-Ovulatory in Vivo-Aged Mouse Oocytes Is Associated with Mitochondrial Dysfunction, but Mitochondrial Transfer from Somatic Cells Is Not Sufficient for Rejuvenation. Hum. Reprod. 2016, 31, 2331–2338. [Google Scholar] [CrossRef] [PubMed]
- Hua, S.; Zhang, Y.; Li, X.C.; Ma, L.B.; Cao, J.W.; Dai, J.P.; Li, R. Effects of Granulosa Cell Mitochondria Transfer on the Early Development of Bovine Embryos in Vitro. Cloning Stem Cells 2007, 9, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Tzeng, C.R.; Hsieh, R.H.; Au, H.K.; Yen, Y.H.; Chang, S.J.; Cheng, Y.F. Mitochondria Transfer (MIT) into Oocyte from Autologous Cumulus Granulosa Cells (CGCs). Fertil. Steril. 2004, 82, S53. [Google Scholar] [CrossRef]
- St. John, J.C. Mitochondria and Female Germline Stem Cells—A Mitochondrial DNA Perspective. Cells 2019, 8, 852. [Google Scholar] [CrossRef] [PubMed]
- Woods, D.C.; Tilly, J. Autologous Germline Mitochondrial Energy Transfer (AUGMENT) in Human Assisted Reproduction. Semin. Reprod. Med. 2015, 33, 410–421. [Google Scholar] [CrossRef]
- Tilly, J.L.; Sinclair, D.A. Germline Energetics, Aging, and Female Infertility. Cell Metab. 2013, 17, 838–850. [Google Scholar] [CrossRef] [PubMed]
- Fakih, M.H.; El Shmoury, M.; Szeptycki, J.; de la Cruz, D.B.; Lux, C.; Verjee, S.; Burgess, C.M.; Cohn, G.M.; Casper, R.F. The AUGMENTSM Treatment: Physician Reported Outcomes of the Initial Global Patient Experience. JFIV Reprod. Med. Genet 2015, 3, 1000154. [Google Scholar] [CrossRef]
- Labarta, E.; de los Santos, M.J.; Herraiz, S.; Escribá, M.J.; Marzal, A.; Buigues, A.; Pellicer, A. Autologous Mitochondrial Transfer as a Complementary Technique to Intracytoplasmic Sperm Injection to Improve Embryo Quality in Patients Undergoing in Vitro Fertilization—a Randomized Pilot Study. Fertil. Steril. 2019, 111, 86–96. [Google Scholar] [CrossRef]
- Oktay, K.; Baltaci, V.; Sonmezer, M.; Turan, V.; Unsal, E.; Baltaci, A.; Aktuna, S.; Moy, F. Oogonial Precursor Cell-Derived Autologous Mitochondria Injection to Improve Outcomes in Women with Multiple IVF Failures Due to Low Oocyte Quality: A Clinical Translation. Reprod. Sci. 2015, 22, 1612–1617. [Google Scholar] [CrossRef]
- Kankanam Gamage, U.S.; Hashimoto, S.; Miyamoto, Y.; Nakano, T.; Yamanaka, M.; Koike, A.; Satoh, M.; Morimoto, Y. Mitochondria Transfer from Adipose Stem Cells Improves the Developmental Potential of Cryopreserved Oocytes. Biomolecules 2022, 12, 1008. [Google Scholar] [CrossRef]
- Veeck, L.L. Oocyte Assessment and Biological Performance. Ann. N. Y. Acad. Sci. 1988, 541, 259–274. [Google Scholar] [CrossRef] [PubMed]
- Gardner, D.K.; Lane, M.; Stevens, J.; Schlenker, T.; Schoolcraft, W.B. Blastocyst Score Affects Implantation and Pregnancy Outcome: Towards a Single Blastocyst Transfer. Fertil. Steril. 2000, 73, 1155–1158. [Google Scholar] [CrossRef]
- Alpha Scientists in Reproductive Medicine. ESHRE Special Interest Group Embryology Istanbul Consensus Workshop on Embryo Assessment: Proceedings of an Expert Meeting. Reprod. Biomed. Online 2011, 22, 632–646. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, S.; Matsumoto, H.; Hashimoto, S.; Brahmajosyula, M.; Ohgaki, A.; Tarui, S.; Matoba, M.; Satoh, M.; Fukuda, A.; Morimoto, Y. A Novel Embryo Quality Scoring System to Compare Groups of Embryos at Different Developmental Stages. J. Assist. Reprod. Genet. 2021, 38, 1123–1132. [Google Scholar] [CrossRef] [PubMed]
- Lawless, C.; Greaves, L.; Reeve, A.K.; Turnbull, D.M.; Vincent, A.E. The Rise and Rise of Mitochondrial DNA Mutations. Open Biol. 2020, 10, 200061. [Google Scholar] [CrossRef] [PubMed]
- Barritt, J.A.; Brenner, C.A.; Willadsen, S.; Cohen, J. Spontaneous and Artificial Changes in Human Ooplasmic Mitochondria. Hum. Reprod. 2000, 15, 207–217. [Google Scholar] [CrossRef]
- Payne, B.A.I.; Chinnery, P.F. Mitochondrial Dysfunction in Aging: Much Progress but Many Unresolved Questions. Biochim. Biophys. Acta Bioenerg. 2015, 1847, 1347–1353. [Google Scholar] [CrossRef]
- Nissanka, N.; Minczuk, M.; Moraes, C.T. Mechanisms of Mitochondrial DNA Deletion Formation. Trends Genet. 2019, 35, 235–244. [Google Scholar] [CrossRef]
- Jansen, R.P.S.; Boer, K.d. The Bottleneck: Mitochondrial Imperatives in Oogenesis and Ovarian Follicular Fate. Mol. Cell. Endocrinol. 1998, 145, 81–88. [Google Scholar] [CrossRef]
- Rugarli, E.I.; Langer, T. Mitochondrial Quality Control: A Matter of Life and Death for Neurons. EMBO J. 2012, 31, 1336–1349. [Google Scholar] [CrossRef]
- Rossignol, R.; Faustin, B.; Rocher, C.; Malgat, M.; Mazat, J.; Letellier, T. Mitochondrial Threshold Effects. Biochem. J. 2003, 370, 751–762. [Google Scholar] [CrossRef]
- Ma, K.; Zhao, X.; Li, H.; Cao, Y.; Li, W.; Ouyang, J.; Xie, L.; Liu, W. Massive Parallel Sequencing of Mitochondrial DNA Genomes from Mother-Child Pairs Using the Ion Torrent Personal Genome Machine (PGM). Forensic. Sci. Int. Genet. 2018, 32, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, K.; Esposito, E.; Wang, X.; Terasaki, Y.; Liu, Y.; Xing, C.; Ji, X.; Lo, E.H. Transfer of Mitochondria from Astrocytes to Neurons after Stroke. Nature 2016, 535, 551–555. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.N.; Das, S.R.; Emin, M.T.; Wei, M.; Sun, L.; Westphalen, K.; Rowlands, D.J.; Quadri, S.K.; Bhattacharya, S.; Bhattacharya, J. Mitochondrial Transfer from Bone-Marrow–Derived Stromal Cells to Pulmonary Alveoli Protects against Acute Lung Injury. Nat. Med. 2012, 18, 759–765. [Google Scholar] [CrossRef] [PubMed]
- Ballinger, S.W. Mitochondrial Dysfunction in Cardiovascular Disease. Free Radic. Biol. Med. 2005, 38, 1278–1295. [Google Scholar] [CrossRef]
- Van Blerkom, J.; Sinclair, J.; Davis, P. Mitochondrial Transfer between Oocytes: Potential Applications of Mitochondrial Donation and the Issue of Heteroplasmy. Hum. Reprod. 1998, 13, 2857–2868. [Google Scholar] [CrossRef]
- Choi, H.W.; Kim, J.H.; Chung, M.K.; Hong, Y.J.; Jang, H.S.; Seo, B.J.; Jung, T.H.; Kim, J.S.; Chung, H.M.; Byun, S.J.; et al. Mitochondrial and Metabolic Remodeling During Reprogramming and Differentiation of the Reprogrammed Cells. Stem Cells Dev. 2015, 24, 1366–1373. [Google Scholar] [CrossRef] [PubMed]
- Suhr, S.T.; Chang, E.A.; Tjong, J.; Alcasid, N.; Perkins, G.A.; Goissis, M.D.; Ellisman, M.H.; Perez, G.I.; Cibelli, J.B. Mitochondrial Rejuvenation After Induced Pluripotency. PLoS ONE 2010, 5, e14095. [Google Scholar] [CrossRef]
- Zhang, C.; Tao, L.; Yue, Y.; Ren, L.; Zhang, Z.; Wang, X.; Tian, J.; An, L. Mitochondrial Transfer from Induced Pluripotent Stem Cells Rescues Developmental Potential of in Vitro Fertilized Embryos from Aging Females. Biol. Reprod. 2021, 104, 1114–1125. [Google Scholar] [CrossRef]
- Tang, S.; Yang, N.; Yu, M.; Wang, S.; Hu, X.; Ni, H.; Cai, W. Noninvasive Autologous Mitochondria Transport Improves the Quality and Developmental Potential of Oocytes from Aged Mice. F&S Sci. 2022, 3, 310–321. [Google Scholar] [CrossRef]
- White, Y.A.R.; Woods, D.C.; Takai, Y.; Ishihara, O.; Seki, H.; Tilly, J.L. Oocyte Formation by Mitotically Active Germ Cells Purified from Ovaries of Reproductive-Age Women. Nat. Med. 2012, 18, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.; Bagley, J.; Skaznik-Wikiel, M.; Lee, H.; Adams, G.B.; Niikura, Y.; Tschudy, K.S.; Tilly, J.C.; Cortes, M.L.; Forkert, R.; et al. Oocyte Generation in Adult Mammalian Ovaries by Putative Germ Cells in Bone Marrow and Peripheral Blood. Cell 2005, 122, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Zou, K.; Yuan, Z.; Yang, Z.; Luo, H.; Sun, K.; Zou, L.; Xiang, J.; Shi, L.; Yu, Q.; Zhang, Y.; et al. Production of Offspring from a Germline Stem Cell Line Derived from Neonatal Ovaries. Nat. Cell Biol. 2009, 11, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Pacchiarotti, J.; Maki, C.; Ramos, T.; Marh, J.; Howerton, K.; Wong, J.; Pham, J.; Anorve, S.; Chow, Y.-C.; Izadyar, F. Differentiation Potential of Germ Line Stem Cells Derived from the Postnatal Mouse Ovary. Differentiation 2010, 79, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zheng, W.; Shen, Y.; Adhikari, D.; Ueno, H.; Liu, K. Experimental Evidence Showing That No Mitotically Active Female Germline Progenitors Exist in Postnatal Mouse Ovaries. Proc. Natl. Acad. Sci. USA 2012, 109, 12580–12585. [Google Scholar] [CrossRef]
- Park, E.S.; Tilly, J.L. Use of DEAD-Box Polypeptide-4 (DdX4) Gene Promoter-Driven Fluorescent Reporter Mice to Identify Mitotically Active Germ Cells in Post-Natal Mouse Ovaries. Mol. Hum. Reprod. 2015, 21, 58–65. [Google Scholar] [CrossRef]
- Woods, D.C.; Telfer, E.E.; Tilly, J.L. Oocyte Family Trees: Old Branches or New Stems? PLoS Genet. 2012, 8, e1002848. [Google Scholar] [CrossRef]
- Woods, D.C.; Tilly, J. An Evolutionary Perspective on Adult Female Germline Stem Cell Function from Flies to Humans. Semin. Reprod. Med. 2013, 31, 24–32. [Google Scholar] [CrossRef]
- Zarate-Garcia, L.; Lane, S.I.R.; Merriman, J.A.; Jones, K.T. FACS-Sorted Putative Oogonial Stem Cells from the Ovary Are Neither DDX4-Positive nor Germ Cells. Sci. Rep. 2016, 6, 27991. [Google Scholar] [CrossRef]
- Wagner, M.; Yoshihara, M.; Douagi, I.; Damdimopoulos, A.; Panula, S.; Petropoulos, S.; Lu, H.; Pettersson, K.; Palm, K.; Katayama, S.; et al. Single-Cell Analysis of Human Ovarian Cortex Identifies Distinct Cell Populations but No Oogonial Stem Cells. Nat. Commun. 2020, 11, 1147. [Google Scholar] [CrossRef]
- Cortopassi, G.A.; Arnheim, N. Detection of a Specific Mitochondrial DNA Deletion in Tissues of Older Humans. Nucleic Acids Res. 1990, 18, 6927–6933. [Google Scholar] [CrossRef] [PubMed]
- Cortopassi, G.A.; Shibata, D.; Soong, N.W.; Arnheim, N. A Pattern of Accumulation of a Somatic Deletion of Mitochondrial DNA in Aging Human Tissues. Proc. Natl. Acad. Sci. USA 1992, 89, 7370–7374. [Google Scholar] [CrossRef] [PubMed]
- Babayev, E.; Wang, T.; Szigeti-Buck, K.; Lowther, K.; Taylor, H.S.; Horvath, T.; Seli, E. Reproductive Aging Is Associated with Changes in Oocyte Mitochondrial Dynamics, Function, and MtDNA Quantity. Maturitas 2016, 93, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Hao, J.; Meng, T.; Guo, L.; Dong, M.; Fan, L.; Ouyang, Y.; Wang, G.; Sun, Q.; Ou, X.; et al. Transfer of Autologous Mitochondria from Adipose Tissue-Derived Stem Cells Rescues Oocyte Quality and Infertility in Aged Mice. Aging 2017, 9, 2480–2488. [Google Scholar] [CrossRef]
- Bhatti, J.S.; Bhatti, G.K.; Reddy, P.H. Mitochondrial Dysfunction and Oxidative Stress in Metabolic Disorders — A Step towards Mitochondria Based Therapeutic Strategies. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 1066–1077. [Google Scholar] [CrossRef]
- Sinha, K.; Das, J.; Pal, P.B.; Sil, P.C. Oxidative Stress: The Mitochondria-Dependent and Mitochondria-Independent Pathways of Apoptosis. Arch. Toxicol. 2013, 87, 1157–1180. [Google Scholar] [CrossRef]
- Woods, D.C.; Tilly, J.L. Isolation, Characterization and Propagation of Mitotically Active Germ Cells from Adult Mouse and Human Ovaries. Nat. Protoc. 2013, 8, 966–988. [Google Scholar] [CrossRef]
- Ravichandran, K.; McCaffrey, C.; Grifo, J.; Morales, A.; Perloe, M.; Munne, S.; Wells, D.; Fragouli, E. Mitochondrial DNA Quantification as a Tool for Embryo Viability Assessment: Retrospective Analysis of Data from Single Euploid Blastocyst Transfers. Hum. Reprod. 2017, 32, 1282–1292. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, R.H.; Au, H.K.; Yeh, T.S.; Chang, S.J.; Cheng, Y.F.; Tzeng, C.R. Decreased Expression of Mitochondrial Genes in Human Unfertilized Oocytes and Arrested Embryos. Fertil. Steril. 2004, 81, 912–918. [Google Scholar] [CrossRef]
- Pacchiarotti, A.; Selman, H.; Valeri, C.; Napoletano, S.; Sbracia, M.; Antonini, G.; Biagiotti, G.; Pacchiarotti, A. Ovarian Stimulation Protocol in IVF: An Up-to-Date Review of the Literature. Curr. Pharm. Biotechnol. 2016, 17, 303–315. [Google Scholar] [CrossRef]
- Haas, J.; Casper, R.F. In Vitro Fertilization Treatments with the Use of Clomiphene Citrate or Letrozole. Fertil. Steril. 2017, 108, 568–571. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.A.; Parikh, S.; Patel, D.R. Understanding Basic Concepts of Developmental Diagnosis in Children. Transl. Pediatr. 2020, 9, S9–S22. [Google Scholar] [CrossRef] [PubMed]
- Aoki, S.; Hashimoto, K.; Ikeda, N.; Takekoh, M.; Fujiwara, T.; Morisaki, N.; Mezawa, H.; Tachibana, Y.; Ohya, Y. Comparison of the Kyoto Scale of Psychological Development 2001 with the Parent-Rated Kinder Infant Development Scale (KIDS). Brain Dev. 2016, 38, 481–490. [Google Scholar] [CrossRef]
- Hashimoto, K.; Matsui, N.; Yakuwa, H.; Miyamura, K. Evaluation of the Family- Rated Kinder Infant Development Scale (KIDS) for Disabled Children. Jikeikai Med. J. 2012, 59, 5–10. [Google Scholar]
| No. of Retrieved Oocytes | 884 | No. of Miscarriages | 4 |
| No. of oocyte retrieval procedures | 105 | % Miscarriage | 26.7 (4/15) |
| No. of mature oocytes | 702 | No. of ectopic pregnancies | 1 |
| Average no. of mature oocytes per oocyte retrieval cycle | 8.4 (884/105) | No. of twin pregnancies | 3 |
| No. of fertilized oocytes | 432 | No. of implantations | 18 |
| % Fertilization | 61.5 (432/702) | % Implantation | 18.6 (18/97) |
| No. of embryo transfers | 63 | No. of Live births | 11 |
| No. of transferred embryos | 97 | % Live birth | 17.5 (11/63) |
| No. of clinical pregnancies | 15 | No. of babies born | 13 |
| % Clinical pregnancies per embryo transfer | 23.8 (15/63) |
| Pre-MT Group | MT Group | p Value | |
|---|---|---|---|
| Number of patients | 33 | 33 | |
| Age | 34.6 ± 3.7 | 36.4 ± 3.9 | n.s. |
| Body mass index (BMI) | 21.1 ± 3.2 | 21.2 ± 3.3 | n.s. |
| Anti-Müllerian hormone (AMH) (ng/mL) | 4.2 ± 4.8 | 1.8 ± 1.5 | n.s. |
| Diacron-Reactive Oxygen Metabolites tests (d-ROM) (CARR U) | 354.9 ± 66.1 | 321.1 ± 81.5 | n.s. |
| Biological Antioxidant Potential (BAP) (μmol/L) | 2222.5 ± 54.9 | 2270.3 ± 207.6 | n.s. |
| Total oocyte No. | 718 | 387 | |
| Average oocyte No. retrieved per patient | 8.4 ± 4.4 | 11.4 ± 6.9 | p < 0.05 |
| Total fertilized oocytes No. | 459 | 208 | |
| Average No. of fertilized oocytes per patient | 5.2 ± 3.1 | 6.2 ± 3.5 | p < 0.05 |
| Average fertilization rate (%) | 61.8 ± 19.9 | 59.3 ± 22.8 | n.s. |
| Total No. of transferable embryos | 163 | 119 | |
| Average transferable embryo rate per zygote (%) | 33.2 ± 23.2 | 62.4 ± 22.3 | p < 0.05 |
| Average good-quality embryo rate per zygote (%) | 6.9 ± 9.3 | 23.7 ± 24.4 | p < 0.05 |
| Average EQS of the transferable embryo on Day 3 | 1.1± 1.0 | 1.9 ± 0.9 | p < 0.05 |
| No. of embryos transferred | 98 | 78 | |
| No. of embryo transfers | 91 | 51 | |
| Clinical pregnancy rate per embryo transfer (%) | 0 | 27.4 (14/51) | p < 0.05 |
| Live birth rate per embryo transfer (%) | 0 | 21.5 (11/51) | p < 0.05 |
| No. of babies born | 0 | 13 | |
| Miscarriage rate (%) | ND | 21.4 (3 /14) |
| Comparison | Concordance Rate | |
|---|---|---|
| Family A | Mother vis-à-vis child | 100% |
| Family B | Mother vis-à-vis child | 100% |
| Family C | Mother vis-à-vis child | 100% |
| Family D * | Mother vis-à-vis child 1 | 99% |
| Family D * | Mother vis-à-vis child 2 | 100% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morimoto, Y.; Gamage, U.S.K.; Yamochi, T.; Saeki, N.; Morimoto, N.; Yamanaka, M.; Koike, A.; Miyamoto, Y.; Tanaka, K.; Fukuda, A.; et al. Mitochondrial Transfer into Human Oocytes Improved Embryo Quality and Clinical Outcomes in Recurrent Pregnancy Failure Cases. Int. J. Mol. Sci. 2023, 24, 2738. https://doi.org/10.3390/ijms24032738
Morimoto Y, Gamage USK, Yamochi T, Saeki N, Morimoto N, Yamanaka M, Koike A, Miyamoto Y, Tanaka K, Fukuda A, et al. Mitochondrial Transfer into Human Oocytes Improved Embryo Quality and Clinical Outcomes in Recurrent Pregnancy Failure Cases. International Journal of Molecular Sciences. 2023; 24(3):2738. https://doi.org/10.3390/ijms24032738
Chicago/Turabian StyleMorimoto, Yoshiharu, Udayanga Sanath Kankanam Gamage, Takayuki Yamochi, Noriatsu Saeki, Naoharu Morimoto, Masaya Yamanaka, Akiko Koike, Yuki Miyamoto, Kumiko Tanaka, Aisaku Fukuda, and et al. 2023. "Mitochondrial Transfer into Human Oocytes Improved Embryo Quality and Clinical Outcomes in Recurrent Pregnancy Failure Cases" International Journal of Molecular Sciences 24, no. 3: 2738. https://doi.org/10.3390/ijms24032738
APA StyleMorimoto, Y., Gamage, U. S. K., Yamochi, T., Saeki, N., Morimoto, N., Yamanaka, M., Koike, A., Miyamoto, Y., Tanaka, K., Fukuda, A., Hashimoto, S., & Yanagimachi, R. (2023). Mitochondrial Transfer into Human Oocytes Improved Embryo Quality and Clinical Outcomes in Recurrent Pregnancy Failure Cases. International Journal of Molecular Sciences, 24(3), 2738. https://doi.org/10.3390/ijms24032738







