The Utility of Amino Acid Metabolites in the Diagnosis of Major Depressive Disorder and Correlations with Depression Severity
Abstract
1. Introduction
2. Results
2.1. Demographic and Clinical Characteristics of HCs and MDD Patients
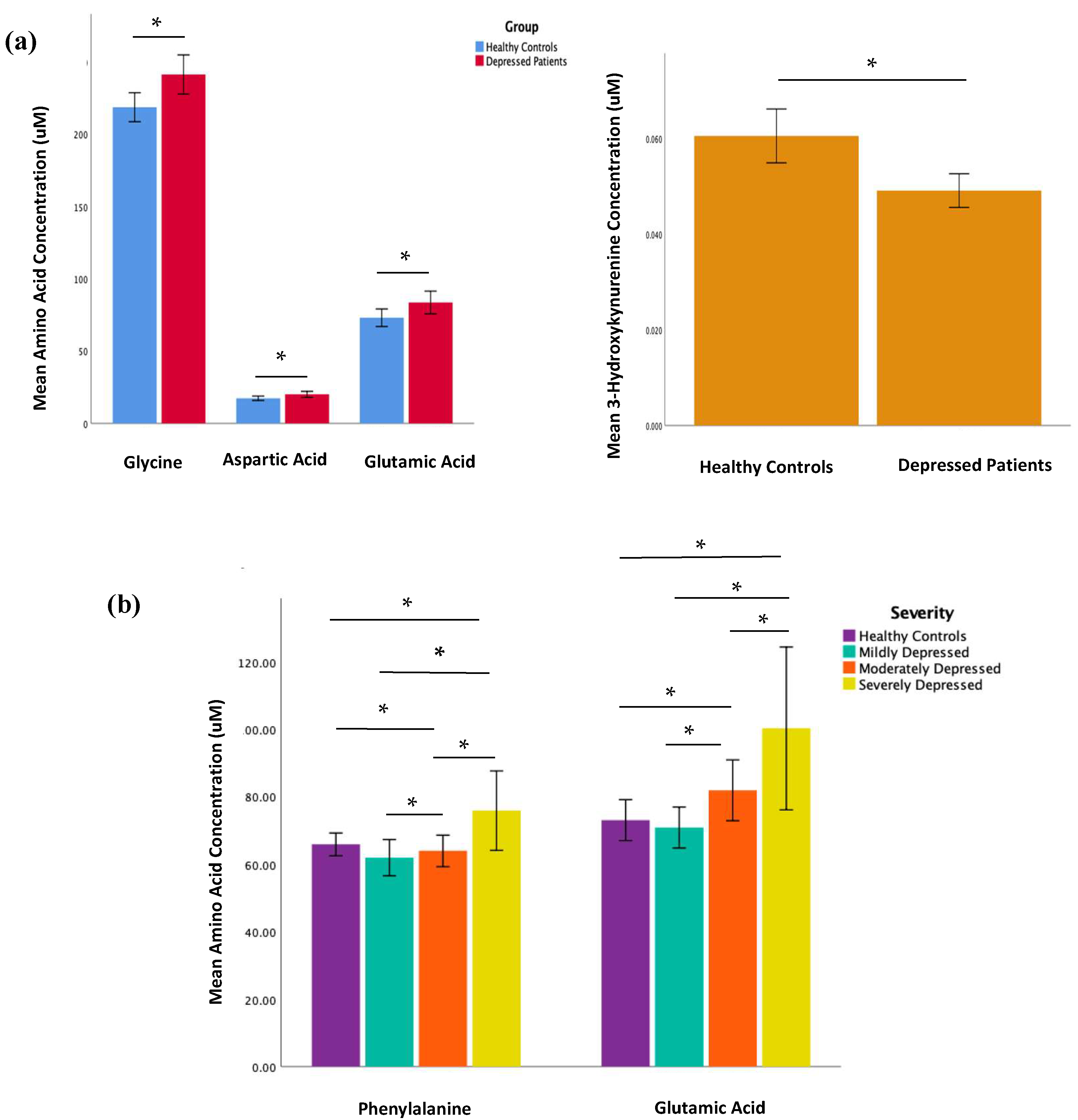
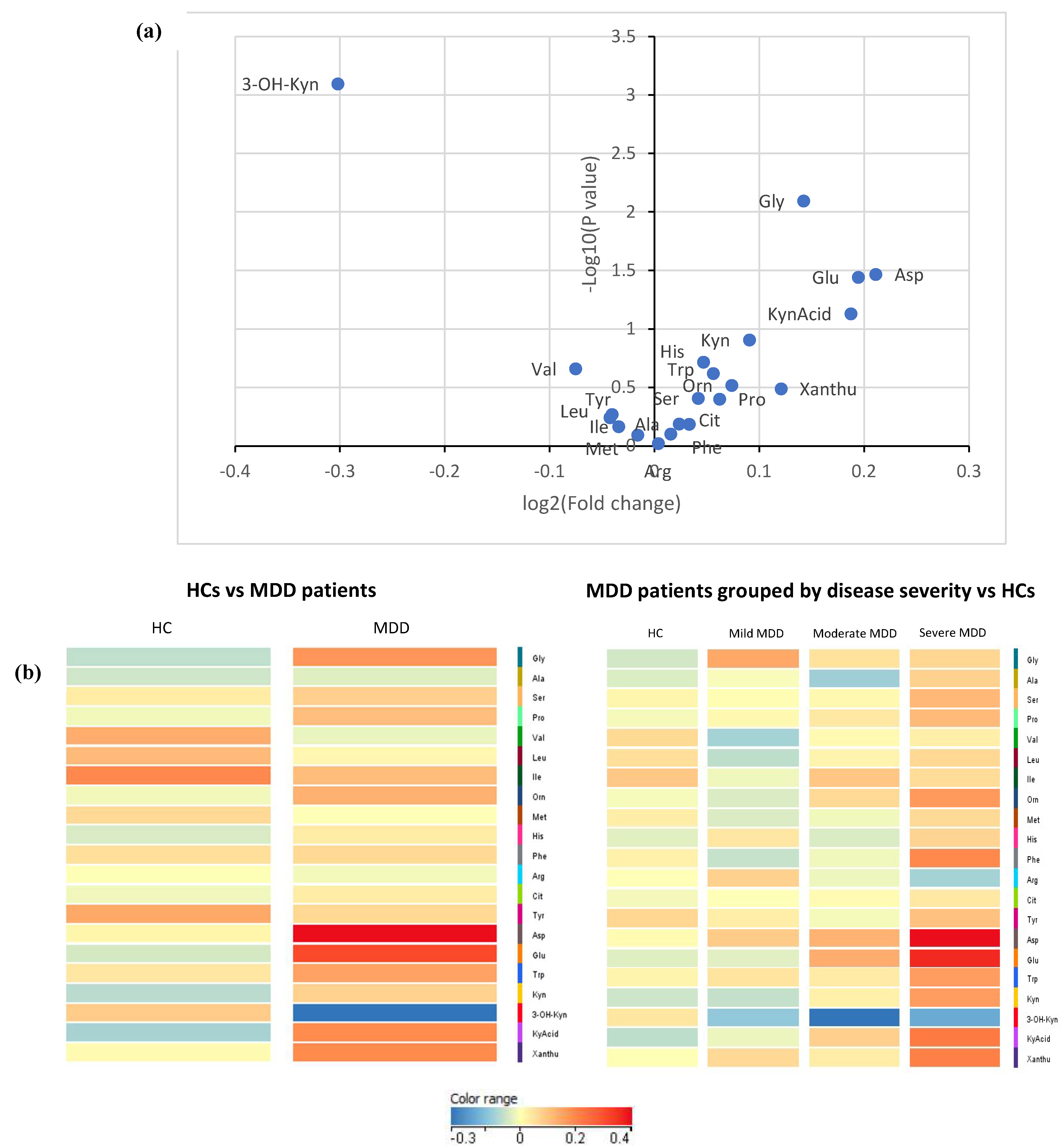
2.2. Amino Acid and Kynurenine Pathway Metabolite Concentrations
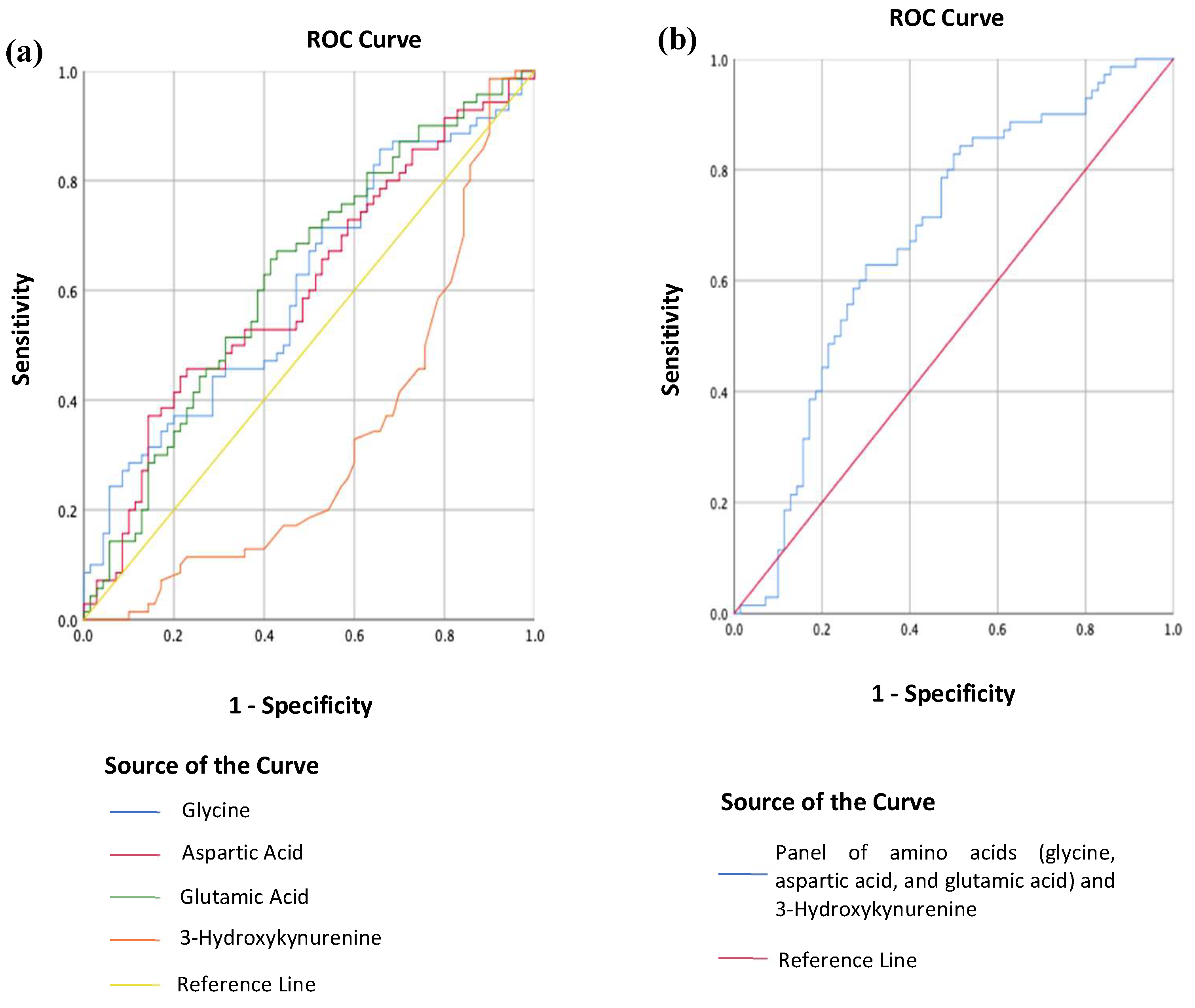
2.3. Differentiating MDD from HC Using Metabolites
3. Discussion
3.1. Glutamic Acid, Aspartic Acid, Glycine, and Phenylalanine in Depression
3.2. 3-Hydroxykynurenine in Depression
3.3. Using Metabolites to Differentiate MDD Patients from HCs
3.4. Strengths and Limitations
4. Materials and Methods
4.1. Sample Size
4.2. Participants
4.3. Blood Collection and Metabolite Analysis
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Erjavec, G.N.; Konjevod, M.; Perkovic, M.N.; Strac, D.S.; Tudor, L.; Barbas, C.; Grune, T.; Zarkovic, N.; Pivac, N. Short overview on metabolomic approach and redox changes in psychiatric disorders. Redox Biol. 2018, 14, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Guijas, C.; Montenegro-Burke, J.R.; Warth, B.; E Spilker, M.; Siuzdak, G. Metabolomics activity screening for identifying metabolites that modulate phenotype. Nat. Biotechnol. 2018, 36, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.H.; Ivanisevic, J.; Siuzdak, G. Metabolomics: Beyond biomarkers and towards mechanisms. Nat. Rev. Mol. Cell Biol. 2016, 17, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Anthony, T.G. Homeostatic responses to amino acid insufficiency. Anim. Nutr. 2015, 1, 135–137. [Google Scholar] [CrossRef]
- Woo, H.-I.; Chun, M.-R.; Yang, J.-S.; Lim, S.-W.; Kim, M.-J.; Kim, S.-W.; Myung, W.-J.; Kim, D.-K.; Lee, S.-Y. Plasma Amino Acid Profiling in Major Depressive Disorder Treated with Selective Serotonin Reuptake Inhibitors. CNS Neurosci. Ther. 2015, 21, 417–424. [Google Scholar] [CrossRef]
- Morilak, D.A.; Frazer, A. Antidepressants and brain monoaminergic systems: A dimensional approach to understanding their behavioural effects in depression and anxiety disorders. Int. J. Neuropsychopharmacol. 2004, 7, 193–218. [Google Scholar] [CrossRef]
- Jenkins, T.A.; Nguyen, J.C.D.; Polglaze, K.E.; Bertrand, P.P. Influence of Tryptophan and Serotonin on Mood and Cognition with a Possible Role of the Gut-Brain Axis. Nutrients 2016, 8, 56. [Google Scholar] [CrossRef]
- E Lakhan, S.; Vieira, K.F. Nutritional therapies for mental disorders. Nutr. J. 2008, 7, 2. [Google Scholar] [CrossRef]
- Ogawa, S.; Koga, N.; Hattori, K.; Matsuo, J.; Ota, M.; Hori, H.; Sasayama, D.; Teraishi, T.; Ishida, I.; Yoshida, F.; et al. Plasma amino acid profile in major depressive disorder: Analyses in two independent case-control sample sets. J. Psychiatr. Res. 2018, 96, 23–32. [Google Scholar] [CrossRef]
- Pan, J.X.; Xia, J.J.; Deng, F.L.; Liang, W.W.; Wu, J.; Yin, B.M.; Dong, M.X.; Chen, J.J.; Ye, F.; Wang, H.Y.; et al. Diagnosis of major depressive disorder based on changes in multiple plasma neurotransmitters: A targeted metabolomics study. Transl. Psychiatry 2018, 8, 130. [Google Scholar] [CrossRef]
- Baranyi, A.; Amouzadeh-Ghadikolai, O.; Von Lewinski, D.; Rothenhäusler, H.-B.; Theokas, S.; Robier, C.; Mangge, H.; Reicht, G.; Hlade, P.; Meinitzer, A. Branched-Chain Amino Acids as New Biomarkers of Major Depression—A Novel Neurobiology of Mood Disorder. PLoS ONE 2016, 11, e0160542. [Google Scholar] [CrossRef]
- Setoyama, D.; Kato, T.A.; Hashimoto, R.; Kunugi, H.; Hattori, K.; Hayakawa, K.; Sato-Kasai, M.; Shimokawa, N.; Kaneko, S.; Yoshida, S.; et al. Plasma Metabolites Predict Severity of Depression and Suicidal Ideation in Psychiatric Patients—A Multicenter Pilot Analysis. PLoS ONE 2016, 11, e0165267. [Google Scholar] [CrossRef]
- Mauri, M.C.; Boscati, L.; Volonteri, L.S.; Scalvini, M.E.; Steinhilber, C.P.; Laini, V.; Zamberlan, F. Predictive Value of Amino Acids in the Treatment of Major Depression with Fluvoxamine. Neuropsychobiology 2001, 44, 134–138. [Google Scholar] [CrossRef] [PubMed]
- Tuck, M.K.; Chan, D.W.; Chia, D.; Godwin, A.K.; Grizzle, W.E.; Krueger, K.E.; Rom, W.; Sanda, M.; Sorbara, L.; Stass, S.; et al. Standard Operating Procedures for Serum and Plasma Collection: Early Detection Research Network Consensus Statement Standard Operating Procedure Integration Working Group. J. Proteome Res. 2009, 8, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Ranger, M.; Johnston, C.C.; Limperopoulos, C.; Rennick, J.E.; du Plessis, A.J. Cerebral near-infrared spectroscopy as a measure of nociceptive evoked activity in critically ill infants. Pain Res. Manag. 2011, 16, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Drevets, W.C. Orbitofrontal Cortex Function and Structure in Depression. Ann. N. Y. Acad. Sci. 2007, 1121, 499–527. [Google Scholar] [CrossRef]
- Mauri, M.C.; Ferrara, A.; Boscati, L.; Bravin, S.; Zamberlan, F.; Alecci, M.; Invernizzi, G. Plasma and Platelet Amino Acid Concentrations in Patients Affected by Major Depression and under Fluvoxamine Treatment. Neuropsychobiology 1998, 37, 124–129. [Google Scholar] [CrossRef]
- Mitani, H.; Shirayama, Y.; Yamada, T.; Maeda, K.; Ashby, C.R.; Kawahara, R. Correlation between plasma levels of glutamate, alanine and serine with severity of depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 2006, 30, 1155–1158. [Google Scholar] [CrossRef]
- Umehara, H.; Numata, S.; Watanabe, S.Y.; Hatakeyama, Y.; Kinoshita, M.; Tomioka, Y.; Nakahara, K.; Nikawa, T.; Ohmori, T. Altered KYN/TRP, Gln/Glu, and Met/methionine sulfoxide ratios in the blood plasma of medication-free patients with major depressive disorder. Sci. Rep. 2017, 7, 4855. [Google Scholar] [CrossRef]
- Hashimoto, K.; Sawa, A.; Iyo, M. Increased Levels of Glutamate in Brains from Patients with Mood Disorders. Biol. Psychiatry 2007, 62, 1310–1316. [Google Scholar] [CrossRef]
- Moriguchi, S.; Takamiya, A.; Noda, Y.; Horita, N.; Wada, M.; Tsugawa, S.; Plitman, E.; Sano, Y.; Tarumi, R.; ElSalhy, M.; et al. Glutamatergic neurometabolite levels in major depressive disorder: A systematic review and meta-analysis of proton magnetic resonance spectroscopy studies. Mol. Psychiatry 2019, 24, 952–964. [Google Scholar] [CrossRef] [PubMed]
- Inoshita, M.; Umehara, H.; Watanabe, S.-Y.; Nakataki, M.; Kinoshita, M.; Tomioka, Y.; Tajima, A.; Numata, S.; Ohmori, T. Elevated peripheral blood glutamate levels in major depressive disorder. Neuropsychiatr. Dis. Treat. 2018, 14, 945–953. [Google Scholar] [CrossRef] [PubMed]
- Szabo, S.T.; Kilts, J.D.; Naylor, J.C.; Youssef, N.; Strauss, J.L.; Morey, R.A.; Brancu, M.; Hamer, R.M.; Bradford, D.W.; Marx, C.E.; et al. Amino Acids as Biomarker Candidates for Suicidality in Male OEF/OIF Veterans: Relevance to NMDA Receptor Modulation and Nitric Oxide Signaling. Mil. Med. 2014, 179, 486–491. [Google Scholar] [CrossRef] [PubMed]
- Blanke, M.L.; Van Dongen, A.M.J. Frontiers in Neuroscience Activation Mechanisms of the NMDA Receptor. In Biology of the NMDA Receptor; Van Dongen, A.M., Ed.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2009; pp. 283–312. [Google Scholar] [CrossRef]
- Smith, Q.R. Transport of Glutamate and Other Amino Acids at the Blood-Brain Barrier. J. Nutr. 2000, 130, 1016S–1022S. [Google Scholar] [CrossRef] [PubMed]
- Newcomer, J.W.; Farber, N.B.; Olney, J.W. NMDA receptor function, memory, and brain aging. Dialogues Clin. Neurosci. 2000, 2, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Cheng, Q.; Chen, C.; Luo, Z.; Feng, D. Excessive Activation of NMDA Receptors in the Pathogenesis of Multiple Peripheral Organs via Mitochondrial Dysfunction, Oxidative Stress, and Inflammation. SN Compr. Clin. Med. 2020, 2, 551–569. [Google Scholar] [CrossRef]
- Bahji, A.; Vazquez, G.H.; Zarate, C.A., Jr. Comparative efficacy of racemic ketamine and esketamine for depression: A systematic review and meta-analysis. J. Affect. Disord. 2021, 278, 542–555. [Google Scholar] [CrossRef]
- Flight, M.H. Trial watch: Phase II boost for glutamate-targeted antidepressants. Nat. Rev. Drug Discov. 2013, 12, 897. [Google Scholar] [CrossRef]
- Krystal, J.H.; Sanacora, G.; Duman, R.S. Rapid-Acting Glutamatergic Antidepressants: The Path to Ketamine and beyond. Biol. Psychiatry 2013, 73, 1133–1141. [Google Scholar] [CrossRef]
- Li, N.; Lee, B.; Liu, R.J.; Banasr, M.; Dwyer, J.M.; Iwata, M.; Li, X.Y. mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science 2010, 329, 959–964. [Google Scholar] [CrossRef]
- Tarrés-Gatius, M.; Miquel-Rio, L.; Campa, L.; Artigas, F.; Castañé, A. Involvement of NMDA receptors containing the GluN2C subunit in the -psychotomimetic and antidepressantlike effects of ketamine. Transl. Psychiatry 2020, 10, 427. [Google Scholar] [CrossRef] [PubMed]
- Lazarevic, V.; Yang, Y.; Flais, I.; Svenningsson, P. Ketamine decreases neuronally released glutamate via retrograde stimulation of presynaptic adenosine A1 receptors. Mol. Psychiatry 2021, 26, 7425–7435. [Google Scholar] [CrossRef] [PubMed]
- Onaolapo, A.Y.; Onaolapo, O.J. Glutamate and depression: Reflecting a deepening knowledge of the gut and brain effects of a ubiquitous molecule. World J. Psychiatry 2021, 11, 297–315. [Google Scholar] [CrossRef] [PubMed]
- Al Hafid, N.; Christodoulou, J. Phenylketonuria: A review of current and future treatments. Transl. Pediatr. 2015, 4, 304–317. [Google Scholar] [CrossRef]
- Ashe, K.; Kelso, W.; Farrand, S.; Panetta, J.; Fazio, T.; De Jong, G.; Walterfang, M. Psychiatric and Cognitive Aspects of Phenylketonuria: The Limitations of Diet and Promise of New Treatments. Front. Psychiatry 2019, 10, 561. [Google Scholar] [CrossRef]
- Bilder, D.A.; Kobori, J.A.; Cohen-Pfeffer, J.L.; Johnson, E.M.; Jurecki, E.R.; Grant, M.L. Neuropsychiatric comorbidities in adults with phenylketonuria: A retrospective cohort study. Mol. Genet. Metab. 2017, 121, 1–8. [Google Scholar] [CrossRef]
- Hörster, F.; A Schwab, M.; Sauer, S.W.; Pietz, J.; Hoffmann, G.F.; Okun, J.G.; Kölker, S.; Kins, S. Phenylalanine Reduces Synaptic Density in Mixed Cortical Cultures from Mice. Transl. Investig. 2006, 59, 544–548. [Google Scholar] [CrossRef]
- Sirtori, L.; Dutra-Filho, C.; Fitarelli, D.; Sitta, A.; Haeser, A.; Barschak, A.; Wajner, M.; Coelho, D.; Llesuy, S.F.; Belló-Klein, A.; et al. Oxidative stress in patients with phenylketonuria. Biochim. Et Biophys. Acta (BBA) Mol. Basis Dis. 2005, 1740, 68–73. [Google Scholar] [CrossRef]
- Fernandes, C.G.; Leipnitz, G.; Seminotti, B.; Amaral, A.U.; Zanatta, A.; Vargas, C.R.; Dutra Filho, C.S.; Wajner, M. Experimental evidence that phenylalanine provokes oxidative stress in hippocampus and cerebral cortex of developing rats. Cell. Mol. Neurobiol. 2010, 30, 317–326. [Google Scholar] [CrossRef]
- Rosa, A.P.; Jacques, C.E.D.; Moraes, T.B.; Wannmacher, C.M.D.; Dutra, D.M.; Dutra-Filho, C.S. Phenylpyruvic Acid Decreases Glucose-6-Phosphate Dehydrogenase Activity in Rat Brain. Cell. Mol. Neurobiol. 2012, 32, 1113–1118. [Google Scholar] [CrossRef]
- Anderson, D.N.; Wilkinson, A.M.; Abou-Saleh, M.T.; Blair, J.A. Recovery from depression after electroconvulsive therapy is accompanied by evidence of increased tetrahydrobiopterin-dependent hydroxylation. Acta Psychiatr. Scand. 1994, 90, 10–13. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Ding, L.; Zhang, H.; Mellor, D.; Wu, H.; Zhao, D.; Wu, C.; Lin, Z.; Yuan, J.; Peng, D. The Metabolic Factor Kynurenic Acid of Kynurenine Pathway Predicts Major Depressive Disorder. Front. Psychiatry 2018, 9, 552. [Google Scholar] [CrossRef] [PubMed]
- Okuda, S.; Nishiyama, N.; Saito, H.; Katsuki, H. 3-Hydroxykynurenine, an Endogenous Oxidative Stress Generator, Causes Neuronal Cell Death with Apoptotic Features and Region Selectivity. J. Neurochem. 1998, 70, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Colín-González, A.L.; Maldonado, P.D.; Santamaria, A. 3-Hydroxykynurenine: An intriguing molecule exerting dual actions in the central nervous system. Neurotoxicology 2013, 34, 189–204. [Google Scholar] [CrossRef]
- Colín-González, A.L.; Maya-López, M.; Pedraza-Chaverrí, J.; Ali, S.F.; Chavarría, A.; Santamaría, A. The Janus faces of 3-hydroxykynurenine: Dual redox modulatory activity and lack of neurotoxicity in the rat striatum. Brain Res. 2014, 1589, 1–14. [Google Scholar] [CrossRef]
- Marx, W.; McGuinness, A.J.; Rocks, T.; Ruusunen, A.; Cleminson, J.; Walker, A.J.; Gomes-Da-Costa, S.; Lane, M.; Sanches, M.; Diaz, A.P.; et al. The kynurenine pathway in major depressive disorder, bipolar disorder, and schizophrenia: A meta-analysis of 101 studies. Mol. Psychiatry 2021, 26, 4158–4178. [Google Scholar] [CrossRef]
- Curzon, G.; Bridges, P.K. Tryptophan metabolism in depression. J. Neurol. Neurosurg. Psychiatry 1970, 33, 698–704. [Google Scholar] [CrossRef]
- Erabi, H.; Okada, G.; Shibasaki, C.; Setoyama, D.; Kang, D.; Takamura, M.; Yoshino, A.; Fuchikami, M.; Kurata, A.; Kato, T.A.; et al. Kynurenic acid is a potential overlapped biomarker between diagnosis and treatment response for depression from metabolome analysis. Sci. Rep. 2020, 10, 16822. [Google Scholar] [CrossRef]
- Sun, Y.; Drevets, W.; Turecki, G.; Li, Q.S. The relationship between plasma serotonin and kynurenine pathway metabolite levels and the treatment response to escitalopram and desvenlafaxine. Brain Behav. Immun. 2020, 87, 404–412. [Google Scholar] [CrossRef]
- Mandrekar, J.N. Receiver Operating Characteristic Curve in Diagnostic Test Assessment. J. Thorac. Oncol. 2010, 5, 1315–1316. [Google Scholar] [CrossRef] [PubMed]
- Hardikar, S.; Albrechtsen, R.D.; Achaintre, D.; Lin, T.; Pauleck, S.; Playdon, M.; Holowatyj, A.N.; Gigic, B.; Schrotz-King, P.; Boehm, J.; et al. Impact of Pre-Blood Collection Factors on Plasma Metabolomic Profiles. Metabolites 2020, 10, 213. [Google Scholar] [CrossRef] [PubMed]
- Kovalik, J.P.; Zhao, X.; Gao, F.; Leng, S.; Chow, V.; Chew, H.; Teo, L.L.Y.; Tan, R.S.; Ewe, S.H.; Tan, H.C.; et al. Amino acid differences between diabetic older adults and non-diabetic older adults and their associations with cardiovascular function. J. Mol. Cell. Cardiol. 2021, 158, 63–71. [Google Scholar] [CrossRef] [PubMed]
| HC (n = 70) | MDD (n = 70) | p Value | |
|---|---|---|---|
| Age (years) | 28.2 (SD 7.3) | 28.3 (SD 7.2) | 0.926 |
| Sex | 1.000 | ||
| Male | 16 (22.9%) | 16 (22.9%) | |
| Female | 54 (77.1%) | 54 (77.1%) | |
| Ethnicity | 1.000 | ||
| Chinese | 45 (64.3%) | 45 (64.3%) | |
| Malay | 15 (21.4%) | 15 (21.4%) | |
| Indian | 9 (12.9%) | 9 (12.9%) | |
| Eurasian | 1 (1.4%) | 1 (1.4%) | |
| Education (years) | 15.6 (SD 1.2) | 14.5 (SD 1.8) | <0.001 |
| HAM-D | 1.9 (SD 2.5) | 19.8 (SD 5.4) | <0.001 |
| Mild (8–16) | 4 (5.7%) | 21 (30.0%) | |
| Moderate (17–23) | 0 | 30 (42.9%) | |
| Severe (≥24) | 0 | 19 (27.1%) | |
| Family psychiatric history | 17 (24.3%) | 30 (42.9%) | 0.032 |
| History of trauma | 14 (20.0%) | 35 (50%) | <0.001 |
| Age at MDD onset (years) | 20.7 (SD 7.5) | ||
| Duration of illness (years) | 7.9 (SD 6.5) | ||
| Past admission to psychiatric ward | 16 (22.9%) | ||
| Past suicide attempt | 32 (45.7%) | ||
| Perceived social support | <0.001 | ||
| Poor | 0 | 17 (24.3%) | |
| Average | 18 (25.7%) | 44 (62.9%) | |
| Good | 52 (74.3%) | 9 (12.9%) | |
| Pharmacotherapy | 60 (85.7%) | ||
| Antidepressants | 58 (96.7%) | ||
| Antipsychotics | 10 (16.7%) | ||
| Anxiolytics and sedatives | 8 (13.3%) | ||
| Mood stabilisers | 4 (6.7%) | ||
| Fluoxetine equivalent dose (mg/day) | 32.79 (SD 2.5) | ||
| Diazepam equivalent dose (mg/day) | 8.75 (SD 1.8) | ||
| Chlorpromazine equivalent dose (mg/day) | 91.21 (SD 20.6) |
| Metabolite | Concentration (uM) | ||||
|---|---|---|---|---|---|
| HCs (n =70) | MDD (n = 70) | t statistic | p Value | Corrected p Value (Bonferroni) | |
| Glycine | 218.5 (SD 42.1) | 241.2 (SD 56.7) | −2.68 | 0.008 | 0.168 |
| Alanine | 427.6 (SD 81.9) | 434.6 (SD 99.1) | −0.455 | 0.650 | 1.000 |
| Serine | 130.6 (SD 19.8) | 134.4 (SD 31.2) | −0.862 | 0.391 | 1.000 |
| Proline | 193.3 (SD 59.2) | 201.8 (SD 59.1) | −0.849 | 0.397 | 1.000 |
| Valine | 253.1 (SD 61.7) | 240.2 (SD 61.7) | 1.24 | 0.219 | 1.000 |
| Leucine | 123.7 (SD 38.5) | 120.1 (SD 36.8) | 0.567 | 0.572 | 1.000 |
| Isoleucine | 69.4 (SD 24.2) | 67.7 (SD 22.8) | 0.408 | 0.684 | 1.000 |
| Ornithine | 69.1 (SD 20.0) | 72.7 (SD 21.4) | −1.03 | 0.303 | 1.000 |
| Methionine | 25.8 (SD 6.6) | 25.5 (SD 7.4) | 0.242 | 0.809 | 1.000 |
| Histidine | 77.4 (SD 11.1) | 80.0 (SD 11.9) | −1.31 | 0.192 | 1.000 |
| Phenylalanine | 65.9 (SD 14.0) | 66.6 (SD 17.1) | −0.27 | 0.789 | 1.000 |
| Arginine | 101.5 (SD 23.2) | 101.7 (SD 27.3) | −0.057 | 0.954 | 1.000 |
| Citrulline | 27.2 (SD 7.5) | 27.8 (SD 9.0) | −0.451 | 0.653 | 1.000 |
| Tyrosine | 65.4 (SD 18.2) | 63.6 (SD 16.6) | 0.615 | 0.539 | 1.000 |
| Aspartic Acid | 17.4 (SD 6.4) | 20.1 (SD 8.6) | −2.14 | 0.034 | 0.714 |
| Glutamic Acid | 73.0 (SD 25.5) | 83.6 (SD 32.9) | −2.12 | 0.036 | 0.756 |
| Tryptophan | 50.9 (SD 9.8) | 52.9 (SD 10.4) | −1.18 | 0.240 | 1.000 |
| Kynurenine | 1.27 (SD 0.294) | 1.35 (SD 0.330) | −1.55 | 0.124 | 1.000 |
| 3-Hydroxykynurenine | 0.061 (SD 0.024) | 0.049 (SD 0.014) | 3.43 | <0.01 | 0.210 |
| Kynurenic acid | 0.037 (SD 0.014) | 0.042 (SD 0.019) | −1.80 | 0.074 | 1.000 |
| Xanthurenic acid | 0.017 (SD 0.008) | 0.018 (SD 0.009) | −0.99 | 0.324 | 1.000 |
| Concentration (uM) | ||||||
|---|---|---|---|---|---|---|
| Metabolite | Mild MDD (n = 21) | Moderate MDD (n = 30) | Severe MDD (n = 19) | F Statistic | p Value | Corrected p Value (Bonferroni) |
| Glycine | 249.9 (SD 49.8) | 235.3 (SD 55.0) | 240.8 (SD 67.3) | 0.399 | 0.672 | 1.00 |
| Alanine | 438.3 (SD 100.5) | 414.6 (SD 105.9) | 462.2 (SD 83.0) | 1.38 | 0.259 | 0.313 |
| Serine | 129.0 (SD 16.3) | 132.3 (SD 31.4) | 143.8 (SD 41.5) | 1.24 | 0.295 | 0.419 |
| Proline | 199.8 (SD 76.6) | 199.2 (SD 53.3) | 208.1 (SD 46.9) | 0.148 | 0.863 | 1.00 |
| Valine | 225.3 (SD 45.3) | 247.8 (SD 77.1) | 244.8 (SD 48.6) | 0.887 | 0.417 | 1.00 |
| Leucine | 113.3 (SD 32.4) | 121.4 (SD 38.1) | 125.4 (SD 40.0) | 0.564 | 0.571 | 1.00 |
| Isoleucine | 64.1 (SD 20.8) | 70.6 (SD 26.2) | 67.2 (SD 19.6) | 0.506 | 0.605 | 1.00 |
| Ornithine | 67.1 (SD 17.3) | 73.0 (SD 22.0) | 78.5 (SD 24.0) | 1.42 | 0.249 | 0.292 |
| Methionine | 24.7 (SD 7.0) | 25.5 (SD 8.4) | 26.5 (SD 6.6) | 0.261 | 0.771 | 1.00 |
| Histidine | 81.1 (SD 9.5) | 77.1 (SD 11.9) | 83.3 (SD 13.5) | 1.79 | 0.175 | 0.222 |
| Phenylalanine | 62.0 (SD 11.7) | 64.0 (SD 12.4) | 75.9 (SD 24.5) | 4.28 | 0.018 | 0.134 |
| Arginine | 106.2 (SD 19.2) | 101.2 (SD 28.1) | 97.6 (SD 33.7) | 0.493 | 0.613 | 1.00 |
| Citrulline | 27.1 (SD 6.14) | 28.3 (SD 11.8) | 27.9 (SD 6.5) | 0.096 | 0.908 | 1.00 |
| Tyrosine | 63.2 (SD 15.1) | 61.6 (SD 15.9) | 67.2 (SD 19.6) | 0.663 | 0.519 | 1.00 |
| Aspartic Acid | 18.1 (SD 5.1) | 19.0 (SD 6.9) | 24.1 (SD 12.5) | 2.99 | 0.057 | 0.131 |
| Glutamic Acid | 70.9 (SD 13.4) | 81.9 (SD 24.2) | 100.3 (SD 50.2) | 4.47 | 0.015 | 0.013 |
| Tryptophan | 52.0 (SD 11.1) | 51.5 (SD 9.9) | 56.3 (SD 10.3) | 1.37 | 0.261 | 1.00 |
| Kynurenine | 1.28 (SD 0.419) | 1.33 (SD 0.252) | 1.46 (SD 0.316) | 0.867 | 0.178 | 0.227 |
| 3-Hydroxykynurenine | 0.052 (SD 0.018) | 0.047 (SD 0.013) | 0.050 (SD 0.014) | 1.77 | 0.425 | 1.00 |
| Kynurenic acid | 0.040 (SD 0.025) | 0.041 (SD 0.016) | 0.045 (SD 0.016) | 0.355 | 0.703 | 1.00 |
| Xanthurenic acid | 0.018 (SD 0.010) | 0.018 (SD 0.010) | 0.020 (SD 0.010) | 0.273 | 0.863 | 1.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ho, C.S.H.; Tay, G.W.N.; Wee, H.N.; Ching, J. The Utility of Amino Acid Metabolites in the Diagnosis of Major Depressive Disorder and Correlations with Depression Severity. Int. J. Mol. Sci. 2023, 24, 2231. https://doi.org/10.3390/ijms24032231
Ho CSH, Tay GWN, Wee HN, Ching J. The Utility of Amino Acid Metabolites in the Diagnosis of Major Depressive Disorder and Correlations with Depression Severity. International Journal of Molecular Sciences. 2023; 24(3):2231. https://doi.org/10.3390/ijms24032231
Chicago/Turabian StyleHo, Cyrus Su Hui, Gabrielle Wann Nii Tay, Hai Ning Wee, and Jianhong Ching. 2023. "The Utility of Amino Acid Metabolites in the Diagnosis of Major Depressive Disorder and Correlations with Depression Severity" International Journal of Molecular Sciences 24, no. 3: 2231. https://doi.org/10.3390/ijms24032231
APA StyleHo, C. S. H., Tay, G. W. N., Wee, H. N., & Ching, J. (2023). The Utility of Amino Acid Metabolites in the Diagnosis of Major Depressive Disorder and Correlations with Depression Severity. International Journal of Molecular Sciences, 24(3), 2231. https://doi.org/10.3390/ijms24032231








