Kainate Receptor Antagonists: Recent Advances and Therapeutic Perspective
Abstract
1. Introduction
2. Structure and Properties of Kainate Receptors
2.1. Structure of Ionotropic Kainate Receptors
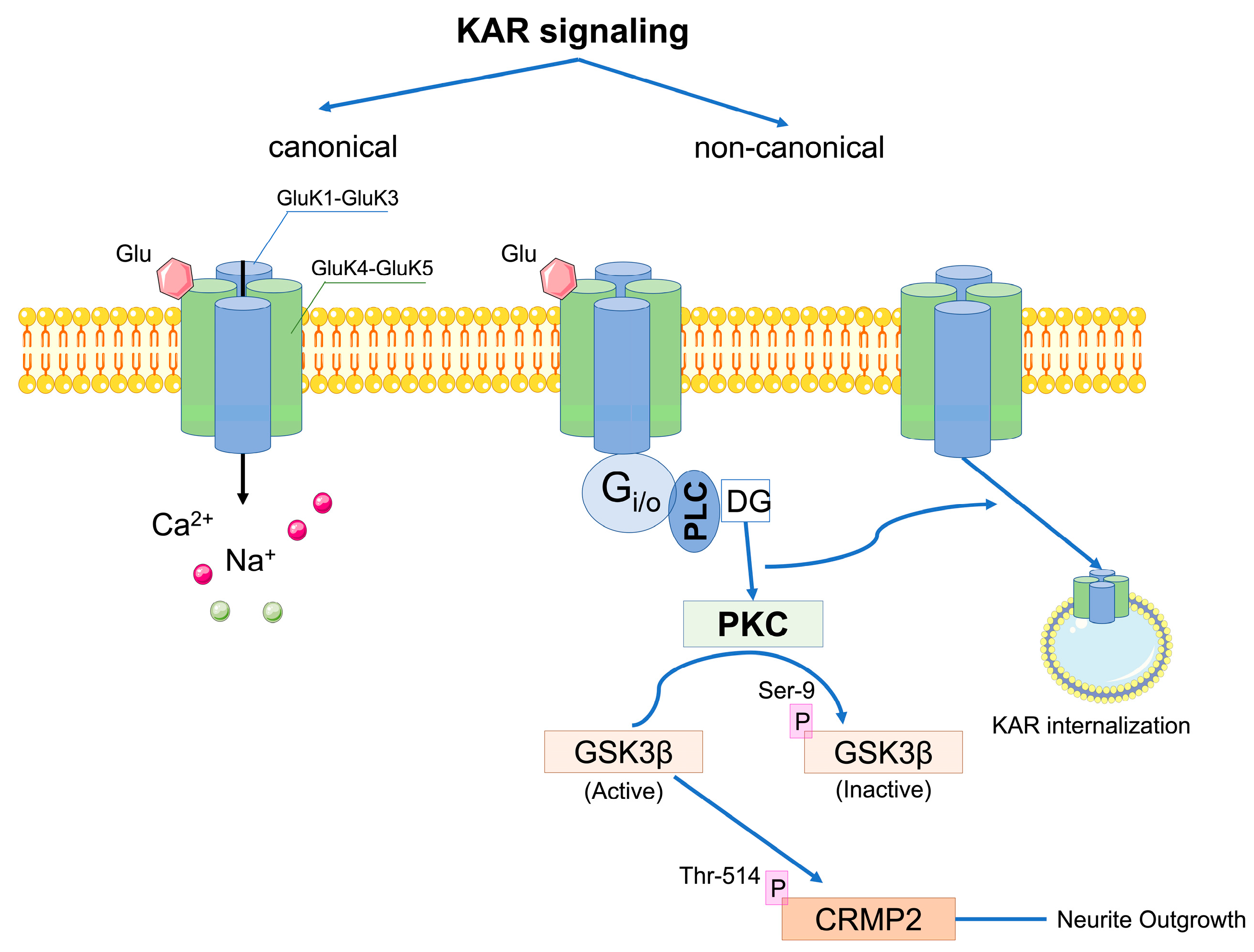
2.2. Activity of Kainate Receptors
2.3. Pathophysiological Role of Kainate Receptors
3. Competitive Antagonists of the Kainate Receptors
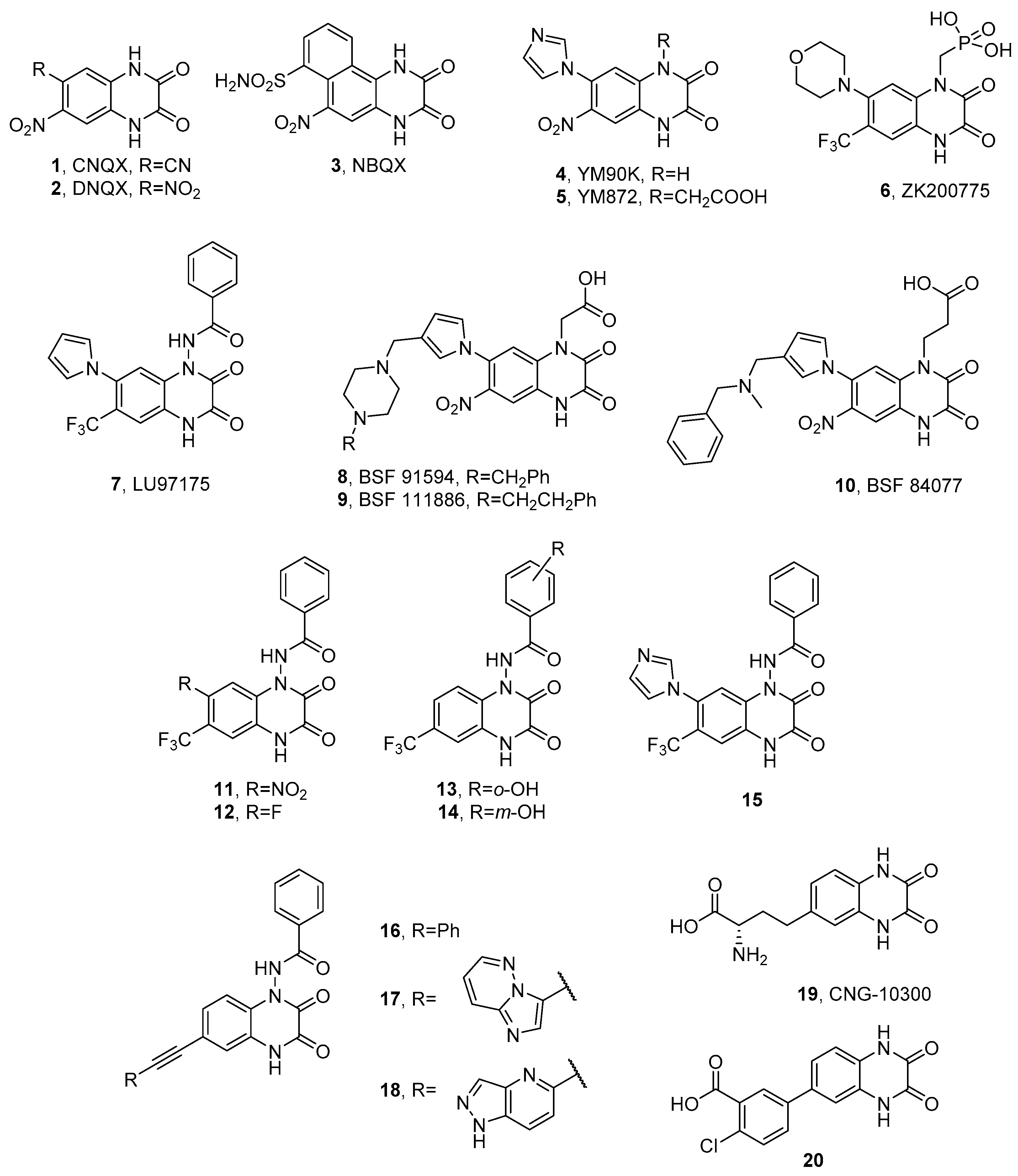
3.1. Quinoxaline-2,3-diones
| cmpd | Native iGluRs | ||
|---|---|---|---|
| AMPA [3H]AMPA | KA [3H]KA | NMDA [3H]CPP or [3H]Glu | |
| CNQX (1) | 0.30 ± 0.15 (IC50) a 0.25 ± 0.01 (Ki) b | 1.5 ± 0.3 (IC50) a 2.7 ± 0.1 (IC50) b | 25 (IC50) a 45 ± 4 (IC50) b |
| DNQX (2) | 0.50 ± 0.10 (IC50) a | 2.0 ± 0.1 (IC50) a | 40 (IC50) a |
| NBQX (3) | 0.046 ± 0.001 (Ki) b 0.15 ± 0.01 (IC50) c 0.12 ± 0.04 (Ki) d | 6.8 ± 0.2 (IC50) b 4.8 ± 0.5 (IC50) c 3.7 ± 1.0 (Ki) d | >100 (IC50) b >90 (IC50) c |
| YM90K (4) | 0.071 ± 0.002 (Ki) b | 4.4 ± 0.1 (IC50) b | >100 (IC50) b |
| YM872 (5) | 0.096 ± 0.002 (Ki) b | 4.6 ± 0.1 (IC50) b | >100 (IC50) b |
| ZK200775 (6) | 0.12 ± 0.09 (IC50) e | 2.5 ± 0.2 (IC50) e | 2.8 ± 0.35 (IC50) e |
| cmpd | Ki [μM] | ||||
|---|---|---|---|---|---|
| Recombinant Homomeric iGluRs | |||||
| GluK1 | GluK2 | GluK3 | GluK5 | GluA2 | |
| CNQX (1) | 1.3 ± 0.3 a | 1.1 ± 0.1 b 1.5 ± 0.01 a | 0.64 ± 0.05 a | 8.4 ± 0.9 c,d | 0.33 ± 0.03 a |
| DNQX (2) | 0.65 ± 0.03 a | 2.1 ± 0.3 a | 0.36 ± 0.03 a | 7.1 ± 0.9 a | 0.25 ± 0.01 a |
| NBQX (3) | 1.4 ± 0.4 e 12 ± 4 f 2.6 ± 0.1 d | 2.3 e 13 ± 2 f 5.4 ± 1.2 d | 3.2 e 24 ± 7 f 18 ± 8 b 3.4 ± 0.6 d | 19 ± 8 e >100 f >100 c 152 ± 23 d | 0.26 ± 0.04 f 0.077 ± 0.010 d |
| LU97175 (7) | 0.088 ± 0.033 e 0.70 ± 0.12 d | 0.31 ± 0.19 e 0.49 ± 0.06 d | 0.022 ± 0.003 e 0.19 ± 0.029 d | 6.9 ± 0.03 e 24 ± 4 d | 1.52 ± 0.25 d |
| 8g | 0.0038 | 4.1 | 0.68 | 20 | nd |
| 9g | 0.012 | 1.4 | 1.4 | 25 | nd |
| 10g | 0.43 | 23 | nd | >30 | nd |
| 11d | 1.1 ± 0.1 | 2.0 ± 0.1 | 0.53 ± 0.15 | 43 ± 11 | 4.9 ± 0.6 |
| 12d | 0.80 ± 0.11 | 0.81 ± 0.08 | 0.28 ± 0.03 | 31 ± 4 | 6.2 ± 1.4 |
| 13d | 1.1 ± 0.1 | 0.91 ± 0.17 | 0.14 ± 0.02 | 26 ± 2 | 4.1 ± 0.6 |
| 14d | 0.80 ± 0.18 | 0.84 ± 0.19 | 0.33 ± 0.01 | 45 ± 4 | 6.0 ± 0.1 |
| 15h | 0.17 ± 0.01 | 0.52 ± 0.14 | 0.08 ± 0.01 | 5.2 ± 0.30 | 5.7 ± 0.3 |
| 16d | >100 | ≈100 | 2.9 ± 0.3 | >100 | 24 ± 6 |
| 17i | ≈100 | >100 | 0.25 ± 0.01 | >100 | >100 |
| 18i | 0.15 ± 0.05 | 0.091 ± 0.01 | 0.13 ± 0.03 | 3.8 ± 0.1 | 0.23 ± 0.02 |
| 19a | 16 ± 1 | 9.5 ± 1.2 | 59 ± 3 | nd | 21 ± 2 |
| 20j | 1.2 ± 0.7 | 33 ± 3 | 37 ± 4 | nd | >100 |
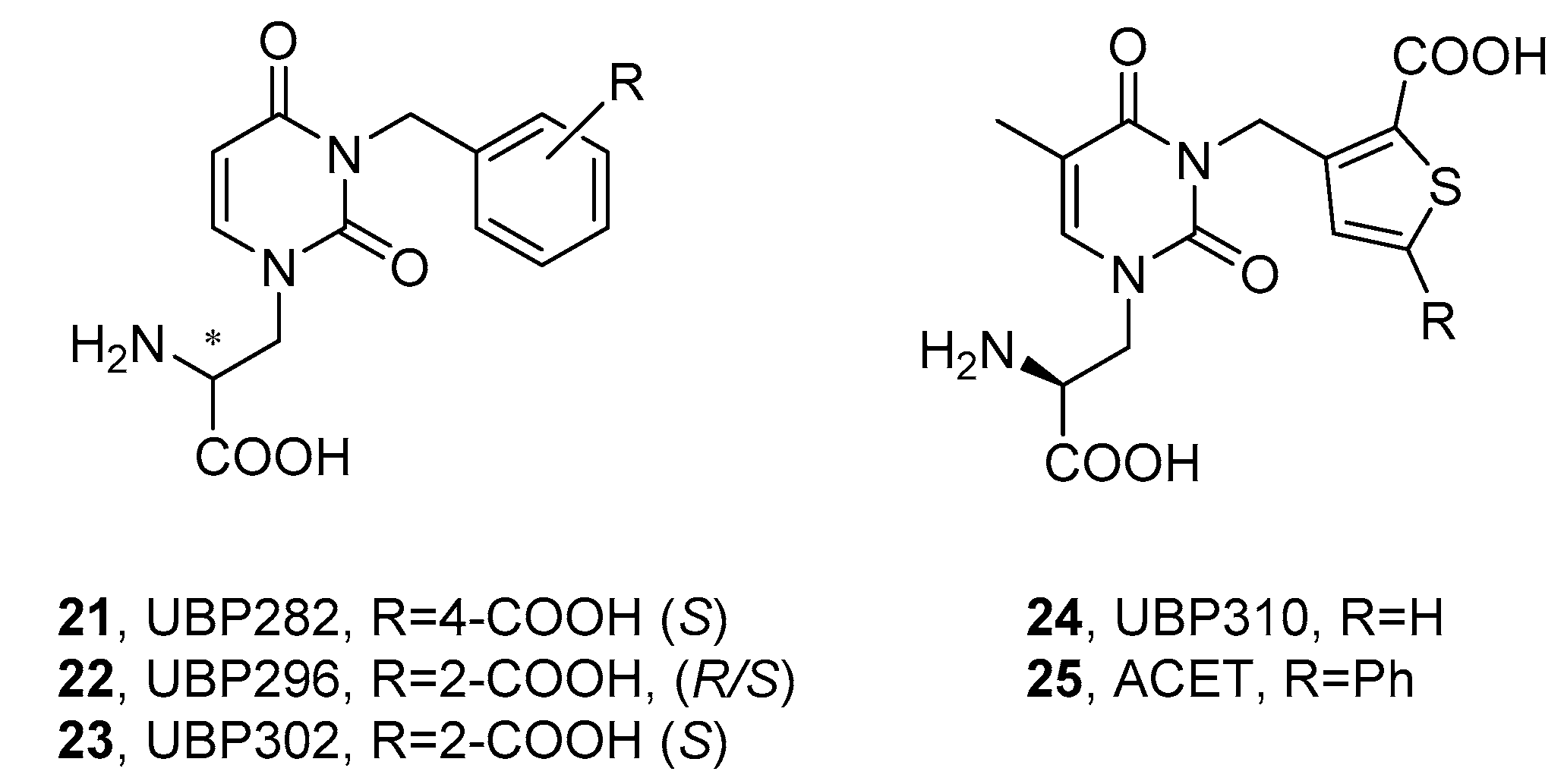
3.2. α-Amino Acid Antagonists
3.2.1. Willardiines
| cmpd | Native iGluRs | |
|---|---|---|
| AMPA IC50 a | GluK1-Containing KARs KD b | |
| UPB282 (21) | 10 ± 2 c | 9.3 ± 0.5 d |
| UBP296 (22) | 98 ± 9 d | 1.1 ± 0.1 d |
| UBP302 (23) | 106 ± 13 d | 0.40 ± 0.05 d |
| UBP310 (24) | 16 ± 5 e | 0.018 ± 0.004 e |
| ACET (UBP316, 25) | 17 ± 1 e | 0.012 ± 0.001 e |
| cmpd | Recombinant Homomeric iGluRs | ||||
|---|---|---|---|---|---|
| GluK1 | GluK2 | GluK3 | GluK5 | GluA2 | |
| UPB282 (21) a | nd | >100 (IC50) | nd | >1000 (IC50) | nd |
| UBP296 (22) | 0.60 ± 0.10 (Kb) b | >1000 (IC50) a | 374 ± 122 (Ki) c | >100 (IC50) a | >300 (Kb) b |
| UBP302 (23) | 0.60 ± 0.10 (Kb) d | >1000 (Ki) c >300 (IC50) d | 4.0 ± 0.2 (IC50) e | nd | >300 (IC50) d |
| UBP310 (24) | 0.022 ± 0.005 (Ki) f 0.010 ± 0.001 (Kb) d | >100 (IC50) d | 0.93 ± 0.12 (Ki) f 0.023 ± 0.002 (IC50) e | >100 (Ki) g | >100 (IC50) d |
| ACET (25) | 0.0070 ± 0.0010 (Kb) d | >100(IC50) d | 0.092 ± 0.009 (IC50) e | nd | >100 (IC50) d |
3.2.2. Decahydroisoquinolines
| cmpd | Ki [μM] | |||
|---|---|---|---|---|
| Recombinant Homomeric iGluRs | ||||
| GluA2 | GluK1 | GluK3 | GluK5 | |
| LY293558 (26) a | 3.2 ± 0.3 | 4.2 ± 0.3 | >100 | >100 |
| LY377770 (27) b | 35 ± 6 | 3.1 ± 1.0 | nd | nd |
| LY382884 (28) c | 553 | 3.6 | nd | nd |
| 29d | 117 ± 16 | 0.16 ± 0.08 | 49 ± 2 | nd |
| LY466195 (31) e | 269 ± 22 | 0.050 ± 0.020 | 8.9 ± 0.2 | 270 ± 31 |
| LY458545 (32) f | 8.3 ± 2.0 | 1.7 ± 0.4 | nd | nd |
| LY457694 (33) f | 5.5 ± 1.8 | 1.6 ± 0.6 | nd | nd |
| LY545694 (34) g | 30 | 0.2 | nd | nd |
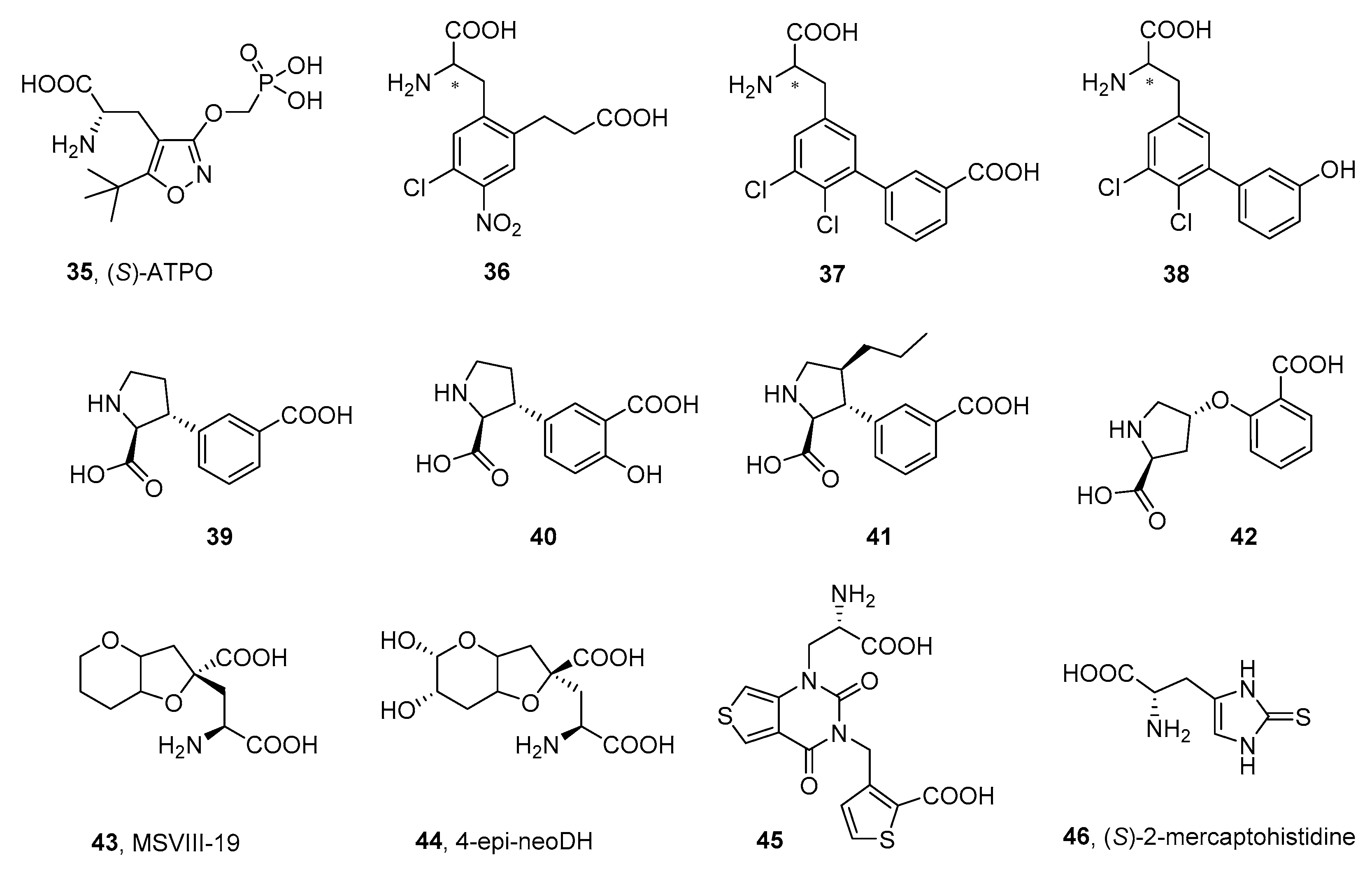
3.2.3. Other α-Amino Acid Antagonists
| cmpd | IC50/Ki [μM] | Ki [μM] | ||||
|---|---|---|---|---|---|---|
| Native iGluRs | Recombinant Homomeric iGluRs | |||||
| AMPA [3H]AMPA | KA [3H]KA | GluK1 | GluK2 | GluK3 | GluA2 | |
| 35 | 16 ± 1 (IC50) a | >100 (IC50) a | 2.2 ± 0.5 b 2.9 ± 0.3 c 2.6 ± 0.4 d | >100 c,d | >100 c >1000 d | 60 ± 5 d |
| 36e | 3.4 ± 0.3 (IC50) | nd | 3.0 ± 0.8 | nd | 14 ± 3 | nd |
| (S)-36 e | nd | nd | 1.5 ± 0.2 | nd | 8.0 ± 1.1 | nd |
| 37c | >100 (IC50) | >100 (IC50) | 2.8 ± 0.8 | >100 | >100 | nd |
| 38 | 4.6 ± 0.2 (IC50) c | >100 (IC50) c | 92 ± 2 f | 78 ± 10 f | 52 ± 3 f | 5.0 ± 0.4 f |
| 39g | 51 ± 10 (IC50) | 22 ± 2 (IC50) | 4.3 ± 0.4 | >100 | 8.1 ± 0.6 | 67 ± 16 |
| 40d | 2.0 (IC50) | 1.4 (IC50) | 4.8 ± 1.5 | 10–100 | 0.87 ± 0.09 | nd |
| 41d | 48 (IC50) | 22 (IC50) | 0.62 ± 0.10 | 81 ± 8 | 2.2 ± 0.4 | nd |
| 42h | >100 (Ki) | >100 (Ki) | 4.0 | >100 | >100 | nd |
| 43 | nd | nd | 0.13 ± 0.02 i | >100 i | >100 i | >100 j |
| 44 | nd | nd | 2.4 j 0.56 k | 7.7 j 6.7 k | >100 k | >100 j >30 k |
| 45l | nd | nd | 0.16 ± 0.02 | 99 ± 22 | 7.5 ± 0.3 | 73 ± 15 |
| 46m | >100 (Ki) | >100 (Ki) | 177 ± 4 | >5000 | 6.4 ± 0.7 | >1000 |
3.3. Structurally Dissimilar AMPAR/KAR Antagonists
4. Non-Competitive Antagonists and Channel Blockers of Kainate Receptors
5. RNA Aptamers
6. The Therapeutic Potential of Non-NMDA Receptors Antagonists
| Disease | Target/Mechanism | Drug | Stage of Development |
|---|---|---|---|
| Neuropathic Pain | Competitive GluK1 antagonist | LY 5,454,694 tosylate | Phase II (compl. 2010) |
| Competitive AMPAR/GluK1 antagonist | NGX426 (tezampanel prodrug) | Phase I (compl. 2008) | |
| Competitive AMPAR/KA antagonist | NS 1209 | Phase II (compl. 2007) | |
| Competitive AMPAR/GluK1 antagonist | Tezampanel (NGX424 LY 293558) | Phase I | |
| Migraine | Competitive AMPAR/GluK1 antagonist | Tezampanel | Phase II (compl. 2009) |
| Competitive AMPAR antagonist | Selurampanel | Phase II (compl. 2012) | |
| Non-competitive KAR antagonist | Topiramate | Phase IV (compl. 2015) | |
| Epilepsy | Competitive AMPAR antagonist | Becampanel | Phase II (compl. 2011) |
| Competitive AMPAR antagonist | Selurampanel | Phase II (compl. 2012) | |
| Competitive AMPAR antagonist | NS 1209 | Phase II | |
| Non-competitive KAR antagonist | Topiramate | Phase IV (compl. 2010) | |
| Non-competitive AMPAR/KAR antagonist | Perampanel | Marketed 2012 | |
| Non-competitive AMPAR antagonist | Talampanel | Phase II (compl. 2006) | |
| Stroke and Head Trauma | Competitive AMPAR antagonist | ZK200775 | Phase II (dis) |
| Amyotrophic Lateral Sclerosis | Non-competitive AMPAR antagonist | Talampanel | Phase II (compl. 2010) |
| Non-competitive AMPAR/KAR antagonist | Perampanel | Phase II (terminated 2021) | |
| Parkinson’s Disease | Non-competitive AMPAR antagonist | Talampanel | Phase II (compl. 2006) |
| Non- competitive AMPAR/KAR antagonist | Perampanel | Phase III (compl. 2012) | |
| Eating Disorder | Non-competitive KAR antagonist | Topiramate | Phase I (compl. 2018) |
| Obesity | Non-competitive KAR antagonist | Topiramate | Phase II (compl. 2017) |
| Chronic Subjective Tinnitus | Competitive AMPAR antagonist | Selurampanel | Phase II (compl. 2012) |
| Tourette Syndrome | Non-competitive KAR antagonist | Topiramate | Phase III (compl. 2008) |
| Glial Brain Tumors | Non-competitive AMPAR/KAR antagonist | Perampanel | Phase IV (compl. 2017) |
| Non-competitive AMPAR antagonist | Talampanel | Phase II (compl. 2011) | |
| Alcohol Dependence | Non-competitive KAR antagonist | Topiramate | Phase III (compl. 2011) |
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Willard, S.S.; Koochekpour, S. Glutamate, glutamate receptors, and downstream signaling pathways. Int. J. Biol. Sci. 2013, 9, 948–959. [Google Scholar] [CrossRef] [PubMed]
- Hansen, K.B.; Wollmuth, L.P.; Bowie, D.; Furukawa, H.; Menniti, F.S.; Sobolevsky, A.I.; Swanson, G.T.; Swanger, S.A.; Greger, I.H.; Nakagawa, T.; et al. Structure, function, and pharmacology of glutamate receptor ion channels. Pharmacol. Rev. 2021, 73, 298–487. [Google Scholar] [CrossRef] [PubMed]
- Traynelis, S.F.; Wollmuth, L.P.; McBain, C.J.; Menniti, F.S.; Vance, K.M.; Ogden, K.K.; Hansen, K.B.; Yuan, H.; Myers, S.J.; Dingledine, R. Glutamate receptor ion channels: Structure, regulation, and function. Pharmacol. Rev. 2010, 62, 405–496. [Google Scholar] [CrossRef] [PubMed]
- Valbuena, S.; Lerma, J. Non-canonical signaling, the hidden life of ligand-gated ion channels. Neuron 2016, 92, 316–329. [Google Scholar] [CrossRef] [PubMed]
- Negrete-Díaz, J.V.; Sihra, T.S.; Flores, G.; Rodríguez-Moreno, A. Non-canonical mechanisms of presynaptic kainate receptors controlling glutamate release. Front. Mol. Neurosci. 2018, 11, 128. [Google Scholar] [CrossRef]
- Collingridge, G.L.; Olsen, R.W.; Peters, J.; Spedding, M. A nomenclature for ligand-gated ion channels. Neuropharmacology 2009, 56, 2–5. [Google Scholar] [CrossRef]
- Schiffer, H.H.; Swanson, G.T.; Heinemann, S.F. Rat GluR7 and a carboxy-terminal splice variant, GluR7b, are functional kainate receptor subunits with a low sensitivity to glutamate. Neuron 1997, 19, 1141–1146. [Google Scholar] [CrossRef]
- Alt, A.; Weiss, B.; Ogden, A.M.; Knauss, J.L.; Oler, J.; Ho, K.; Large, T.H.; Bleakman, D. Pharmacological characterization of glutamatergic agonists and antagonists at recombinant human homomeric and heteromeric kainate receptors in vitro. Neuropharmacology 2004, 46, 793–806. [Google Scholar] [CrossRef]
- Sagot, E.; Pickering, D.S.; Pu, X.; Umberti, M.; Stensbøl, T.B.; Nielsen, B.; Chapelet, M.; Bolte, J.; Gefflaut, T.; Bunch, L. Chemo-enzymatic synthesis of a series of 2,4-syn-functionalized (S)-glutamate analogues: New insight into the structure-activity relation of ionotropic glutamate receptor subtypes 5, 6, and 7. J. Med. Chem. 2008, 51, 4093–4103. [Google Scholar] [CrossRef]
- Mott, D.D.; Rojas, A.; Fisher, J.L.; Dingledine, R.J.; Benveniste, M. Subunit-specific desensitization of heteromeric kainate receptors. J. Physiol. 2010, 588, 683–700. [Google Scholar] [CrossRef]
- Møllerud, S.; Frydenvang, K.; Pickering, D.S.; Kastrup, J.S. Lessons from crystal structures of kainate receptors. Neuropharmacology 2017, 112, 16–28. [Google Scholar] [CrossRef]
- Fernandes, H.B.; Catches, J.S.; Petralia, R.S.; Copits, B.A.; Xu, J.; Russell, T.A.; Swanson, G.T.; Contractor, A. High affinity kainate receptor subunits are necessary for ionotropic but not metabotropic signaling. Neuron 2009, 63, 818–829. [Google Scholar] [CrossRef]
- Møllerud, S.; Kastrup, J.S.; Pickering, D.S. A pharmacological profile of the high-affinity GluK5 kainate receptor. Eur. J. Pharmacol. 2016, 788, 315–320. [Google Scholar] [CrossRef]
- Pinheiro, P.; Mulle, C. Kainate receptors. Cell Tissue Res. 2006, 326, 457–482. [Google Scholar] [CrossRef]
- Evans, A.J.; Gurung, S.; Henley, J.M.; Nakamura, Y.; Wilkinson, K.A. Exciting times: New advances towards understanding the regulation and roles of kainate receptors. Neurochem. Res. 2019, 44, 572–584. [Google Scholar] [CrossRef]
- Lerma, J.; Paternain, A.V.; Rodríguez-Moreno, A.; López-García, J.C. Molecular physiology of kainate receptors. Physiol. Rev. 2001, 81, 971–998. [Google Scholar] [CrossRef]
- Perrais, D.; Coussen, F.; Mulle, C. Atypical functional properties of GluK3-containing kainate receptors. J. Neurosci. 2009, 29, 15499–15510. [Google Scholar] [CrossRef]
- Negrete-Díaz, J.V.; Falcón-Moya, R.; Rodríguez-Moreno, A. Kainate receptors: From synaptic activity to disease. FEBS J. 2021, 289, 5074–5088. [Google Scholar] [CrossRef]
- Jin, X.-T.; Smith, Y. Localization and functions of kainate receptors in the basal ganglia. Adv. Exp. Med. Biol. 2011, 717, 27–37. [Google Scholar] [CrossRef]
- Lerma, J.; Marques, J.M. Kainate receptors in health and disease. Neuron 2013, 80, 292–311. [Google Scholar] [CrossRef]
- Bloss, E.B.; Hunter, R.G. Hippocampal kainate receptors. In Vitamins and Hormones; Academic Press Inc.: Cambridge, MA, USA, 2010; Volume 82, pp. 167–184. [Google Scholar]
- Pinheiro, P.S.; Perrais, D.; Coussen, F.; Barhanin, J.; Bettler, B.; Mann, J.R.; Malva, J.O.; Heinemann, S.F.; Mulle, C. GluR7 is an essential subunit of presynaptic kainate autoreceptors at hippocampal mossy fiber synapses. Proc. Natl. Acad. Sci. USA 2007, 104, 12181–12186. [Google Scholar] [CrossRef] [PubMed]
- Perrais, D.; Pinheiro, P.S.; Jane, D.E.; Mulle, C. Antagonism of recombinant and native GluK3-containing kainate receptors. Neuropharmacology 2009, 56, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Valbuena, S.; Lerma, J. Kainate receptors, homeostatic gatekeepers of synaptic plasticity. Neuroscience 2021, 456, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Wyeth, M.S.; Pelkey, K.A.; Yuan, X.; Vargish, G.; Johnston, A.D.; Hunt, S.; Fang, C.; Abebe, D.; Mahadevan, V.; Fisahn, A.; et al. NETO auxiliary subunits regulate interneuron somatodendritic and presynaptic kainate receptors to control network inhibition. Cell Rep. 2017, 20, 2156–2168. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Sun, J.; Gao, Y.; Li, B.; Wang, Y.; Dong, Y.; An, W.; Li, H.; Yang, B.; Ge, Y.; et al. Kainate receptor modulation by NETO2. Nature 2021, 599, 325–329. [Google Scholar] [CrossRef]
- Vinnakota, R.; Dhingra, S.; Kumari, J.; Ansari, M.Y.; Shukla, E.; Nerkar, M.D.; Kumar, J. Role of Neto1 extracellular domain in modulation of kainate receptors. Int. J. Biol. Macromol. 2021, 192, 525–536. [Google Scholar] [CrossRef]
- Mennesson, M.; Orav, E.; Gigliotta, A.; Kulesskaya, N.; Saarnio, S.; Kirjavainen, A.; Kesaf, S.; Winkel, F.; Pou, M.L.; Umemori, J.; et al. Cognition and behavior kainate receptor auxiliary subunit NETO2-related cued fear conditioning impairments associate with defects in amygdala development and excitability. eNeuro 2020, 7, 1–15. [Google Scholar] [CrossRef]
- Falcón-Moya, R.; Rodríguez-Moreno, A. Metabotropic actions of kainate receptors modulating glutamate release. Neuropharmacology 2021, 197, 108696. [Google Scholar] [CrossRef]
- Sihra, T.S.; Rodríguez-Moreno, A. Presynaptic kainate receptor-mediated bidirectional modulatory actions: Mechanisms. Neurochem. Int. 2013, 62, 982–987. [Google Scholar] [CrossRef]
- Rodríguez-Moreno, A.; Sihra, T.S. Metabotropic actions of kainate receptors in the control of glutamate release in the hippocampus. Adv. Exp. Med. Biol. 2011, 717, 39–48. [Google Scholar] [CrossRef]
- Kesaf, S.; Khirug, S.; Dinh, E.; Saez Garcia, M.; Soni, S.; Orav, E.; Delpire, E.; Taira, T.; Lauri Sari, E.; Rivera, C. The kainate receptor subunit GluK2 interacts with KCC2 to promote maturation of dendritic spines. Front. Cell. Neurosci. 2020, 14, 252. [Google Scholar] [CrossRef]
- Petrovic, M.M.; Viana Da Silva, S.; Clement, J.P.; Vyklicky, L.; Mulle, C.; González-González, I.M.; Henley, J.M. Metabotropic action of postsynaptic kainate receptors triggers hippocampal long-term potentiation. Nat. Neurosci. 2017, 20, 529–539. [Google Scholar] [CrossRef]
- Kumari, J.; Bendre, A.D.; Bhosale, S.; Vinnakota, R.; Burada, A.P.; Tria, G.; Ravelli, R.B.G.; Peters, P.J.; Joshi, M.; Kumar, J. Structural dynamics of the GluK3-kainate receptor neurotransmitter binding domains revealed by cryo-EM. Int. J. Biol. Macromol. 2020, 149, 1051–1058. [Google Scholar] [CrossRef]
- Kumari, J.; Vinnakota, R.; Kumar, J. Structural and functional insights into GluK3-kainate receptor desensitization and recovery. Sci. Rep. 2019, 9, 10254. [Google Scholar] [CrossRef]
- Khanra, N.; Brown, P.M.G.E.; Perozzo, A.M.; Bowie, D.; Meyerson, J.R. Architecture and structural dynamics of the heteromeric GluK2/K5 kainate receptor. eLife 2021, 10, e66097. [Google Scholar] [CrossRef]
- Selvakumar, P.; Lee, J.; Khanra, N.; He, C.; Munguba, H.; Kiese, L.; Broichhagen, J.; Reiner, A.; Levitz, J.; Meyerson, J.R. Structural and compositional diversity in the kainate receptor family. Cell Rep. 2021, 37, 109891. [Google Scholar] [CrossRef]
- Available online: www.clinicaltrials.gov (accessed on 8 December 2022).
- Celli, R.; Fornai, F. Targeting ionotropic glutamate receptors in the treatment of epilepsy. Curr. Neuropharmacol. 2021, 19, 747–765. [Google Scholar] [CrossRef]
- Celesia, G.G.; Chen, R.C.; Bamforth, B.J. Effects of ketamine in epilepsy. Neurology 1975, 25, 169–172. [Google Scholar] [CrossRef]
- Traynor, K. Perampanel approved for epilepsy. Am. J. Health Syst. Pharm. 2012, 69, 2024. [Google Scholar] [CrossRef]
- Villanueva, V.; Montoya, J.; Castillo, A.; Mauri-Llerda, J.; Giner, P.; López-González, F.J.; Piera, A.; Villanueva-Hernández, P.; Bertol, V.; Garcia-Escrivá, A.; et al. Perampanel in routine clinical use in idiopathic generalized epilepsy: The 12-month GENERAL study. Epilepsia 2018, 59, 1740–1752. [Google Scholar] [CrossRef]
- Fritsch, B.; Reis, J.; Gasior, M.; Kaminski, R.M.; Rogawski, M.A. Role of GluK1 kainate receptors in seizures, epileptic discharges, and epileptogenesis. J. Neurosci. 2014, 34, 5765–5775. [Google Scholar] [CrossRef] [PubMed]
- Lique Peret, A.; Christie, L.A.; Ouedraogo, D.W.; Gorlewicz, A.; Rô Me Epsztein, J.; Mulle, C.; Rie Cré Pel, V. Contribution of aberrant GluK2-containing kainate receptors to chronic seizures in temporal lobe epilepsy. Cell Rep. 2014, 8, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Henley, J.M.; Nair, J.D.; Seager, R.; Yucel, B.P.; Woodhall, G.; Henley, B.S.; Talandyte, K.; Needs, H.I.; Wilkinson, K.A. Kainate and AMPA receptors in epilepsy: Cell biology, signalling pathways and possible crosstalk. Neuropharmacology 2021, 195, 108569. [Google Scholar] [CrossRef] [PubMed]
- Falcón-Moya, R.; Sihra, T.S.; Rodríguez-Moreno, A. Kainate receptors: Role in epilepsy. Front. Mol. Neurosci. 2018, 11, 217. [Google Scholar] [CrossRef] [PubMed]
- Crépel, V.; Mulle, C. Physiopathology of kainate receptors in epilepsy. Curr. Opin. Pharmacol. 2015, 20, 83–88. [Google Scholar] [CrossRef]
- Vargas, J.R.; Takahashi, D.K.; Thomson, K.E.; Wilcox, K.S. The expression of kainate receptor subunits in hippocampal astrocytes after experimentally induced status epilepticus. J. Neuropathol. Exp. Neurol. 2013, 72, 919–932. [Google Scholar] [CrossRef]
- Victor Nadler, J. Kainic acid as a tool for the study of temporal lobe epilepsy. Life Sci. 1981, 29, 2031–2042. [Google Scholar] [CrossRef]
- Vincent, P.; Mulle, C. Kainate receptors in epilepsy and excitotoxicity. Neuroscience 2009, 158, 309–323. [Google Scholar] [CrossRef]
- Matute, C. Therapeutic potential of kainate receptors. CNS Neurosci. Ther. 2011, 17, 661–669. [Google Scholar] [CrossRef]
- Zhuo, M. Ionotropic glutamate receptors contribute to pain transmission and chronic pain. Neuropharmacology 2017, 112, 228–234. [Google Scholar] [CrossRef]
- Wu, L.-J.; Ko, S.; Zhuo, M. Kainate receptors and pain: From dorsal root ganglion to the anterior cingulate cortex. Curr. Pharm. Des. 2007, 13, 1597–1605. [Google Scholar] [CrossRef][Green Version]
- Bhangoo, S.K.; Swanson, G.T. Kainate receptor signaling in pain pathways. Mol. Pharmacol. 2013, 83, 307–315. [Google Scholar] [CrossRef]
- Bleakman, D.; Alt, A.; Nisenbaum, E.S. Glutamate receptors and pain. Semin. Cell Dev. Biol. 2006, 17, 592–604. [Google Scholar] [CrossRef]
- Dominguez, E.; Iyengar, S.; Shannon, H.E.; Bleakman, D.; Alt, A.; Arnold, B.M.; Bell, M.G.; Bleisch, T.J.; Buckmaster, J.L.; Castano, A.M.; et al. Two prodrugs of potent and selective GluR5 kainate receptor antagonists actives in three animal models of pain. J. Med. Chem. 2005, 48, 4200–4203. [Google Scholar] [CrossRef]
- Andreou, A.P.; Holland, P.R.; Goadsby, P.J. Activation of iGluR5 kainate receptors inhibits neurogenic dural vasodilatation in an animal model of trigeminovascular activation. Br. J. Pharmacol. 2009, 157, 464. [Google Scholar] [CrossRef]
- Hoffmann, J.; Charles, A. Glutamate and its receptors as therapeutic targets for migraine. Neurotherapeutics 2018, 15, 361–370. [Google Scholar] [CrossRef]
- Samengo, I.; Curro, D.; Navarra, P.; Barrese, V.; Taglialatela, M.; Martire, M. Molecular and pharmacological evidence for a facilitatory functional role of pre-synaptic GLUK2/3 kainate receptors on GABA release in rat trigeminal caudal nucleus. Eur. J. Pain 2012, 16, 1148–1157. [Google Scholar] [CrossRef]
- Valbuena, S.; Lerma, J. Losing balance: Kainate receptors and psychiatric disorders comorbidities. Neuropharmacology 2021, 191, 108558. [Google Scholar] [CrossRef]
- Zhuo, M. Cortical kainate receptors and behavioral anxiety. Mol. Brain 2017, 10, 16. [Google Scholar] [CrossRef]
- Kilic, G.; Ismail Kucukali, C.; Orhan, N.; Ozkok, E.; Zengin, A.; Aydin, M.; Kara, I. Are GRIK3 (T928G) gene variants in schizophrenia patients different from those in their first-degree relatives? Psychiatry Res. 2010, 175, 43–46. [Google Scholar] [CrossRef]
- Ahmad, Y.; Bhatia, M.S.; Mediratta, P.K.; Sharma, K.K.; Negi, H.; Chosdol, K.; Sinha, S. Association between the ionotropic glutamate receptor kainate3 (GRIK3) Ser310Ala polymorphism and schizophrenia in the Indian population. World J. Biol. Psychiatry 2009, 10, 330–333. [Google Scholar] [CrossRef] [PubMed]
- Shibata, H.; Shibata, A.; Ninomiya, H.; Tashiro, N.; Fukumaki, Y. Association study of polymorphisms in the GluR6 kainate receptor gene (GRIK2) with schizophrenia. Psychiatry Res. 2002, 113, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Iida, I.; Konno, K.; Natsume, R.; Abe, M.; Watanabe, M.; Sakimura, K.; Terunuma, M. A comparative analysis of kainate receptor GluK2 and GluK5 knockout mice in a pure genetic background. Behav. Brain Res. 2021, 405, 113194. [Google Scholar] [CrossRef] [PubMed]
- Bunch, L.; Krogsgaard-Larsen, P. Subtype selective kainic acid receptor agonists: Discovery and approaches to rational design. Med. Res. Rev. 2009, 29, 3–28. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.H.; Wang, S.; Chuang, H.H.; Oswald, R.E. Mechanism of AMPA receptor activation by partial agonists: Disulfide trapping of closed lobe conformations. J. Biol. Chem. 2011, 286, 35257–35266. [Google Scholar] [CrossRef]
- Honoré, T.; Davies, S.N.; Drejer, J.; Fletcher, E.J.; Jacobsen, P.; Lodge, D.; Nielsen, F.E. Quinoxalinediones: Potent competitive non-NMDA glutamate receptor antagonists. Science 1988, 241, 701–703. [Google Scholar] [CrossRef]
- Sheardown, M.J.; Nielsen, E.O.; Hansen, A.J.; Jacobsen, P.; Honore, T. 2,3-Dihydroxy-6-nitro-7-sulfamoyl-benzo(F)quinoxaline: A neuroprotectant for cerebral ischemia. Science 1990, 247, 571–574. [Google Scholar] [CrossRef]
- Judge, M.E.; Sheardown, M.J.; Jacobsen, P.; Honoré, T. Protection against post-ischemic behavioral pathology by the α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) antagonist 2,3-dihydroxy-6-nitro-7-sulfamoyl-benzo(f)quinoxaline (NBQX) in the gerbil. Neurosci. Lett. 1991, 133, 291–294. [Google Scholar] [CrossRef]
- Lubisch, W.; Behl, B.; Hofmann, H.P. Amido-Quinoxalinediones, the Preparation and Use Thereof. U.S. Patent 5,773,439, 30 June 1998. [Google Scholar]
- Löscher, W.; Lehmann, H.; Behl, B.; Seemann, D.; Teschendorf, H.J.; Hofmann, H.P.; Lubisch, W.; Höger, T.; Lemaire, H.G.; Gross, G. A new pyrrolyl-quinoxalinedione series of non-NMDA glutamate receptor antagonists: Pharmacological characterization and comparison with NBQX and valproate in the kindling model of epilepsy. Eur. J. Neurosci. 1999, 11, 250–262. [Google Scholar] [CrossRef]
- Twele, F.; Bankstahl, M.; Klein, S.; Römermann, K.; Löscher, W. The AMPA receptor antagonist NBQX exerts anti-seizure but not antiepileptogenic effects in the intrahippocampal kainate mouse model of mesial temporal lobe epilepsy. Neuropharmacology 2015, 95, 234–242. [Google Scholar] [CrossRef]
- Lippman-Bell, J.J.; Rakhade, S.N.; Klein, P.M.; Obeid, M.; Jackson, M.C.; Joseph, A.; Jensen, F.E. AMPA Receptor antagonist NBQX attenuates later-life epileptic seizures and autistic-like social deficits following neonatal seizures. Epilepsia 2013, 54, 1922–1932. [Google Scholar] [CrossRef]
- Kong, L.-L.; Yu, L.-C. It is AMPA receptor, not kainate receptor, that contributes to the NBQX-induced antinociception in the spinal cord of rats. Brain Res. 2006, 1100, 73–77. [Google Scholar] [CrossRef]
- Van Damme, P.; Leyssen, M.; Callewaert, G.; Robberecht, W.; Van Den Bosch, L. The AMPA receptor antagonist NBQX prolongs survival in a transgenic mouse model of amyotrophic lateral sclerosis. Neurosci. Lett. 2003, 343, 81–84. [Google Scholar] [CrossRef]
- Goda, M.; Isono, M.; Fujiki, M.; Kobayashi, H. Both MK801 and NBQX reduce the neuronal damage after impact-acceleration brain injury. J. Neurotrauma 2002, 19, 1445–1456. [Google Scholar] [CrossRef]
- Follett, P.L.; Rosenberg, P.A.; Volpe, J.J.; Jensen, F.E. NBQX attenuates excitotoxic injury in developing white matter. J. Neurosci. 2000, 20, 9235–9241. [Google Scholar] [CrossRef]
- Swedberg, M.D.; Jacobsen, P.; Honoré, T. Anticonvulsant, anxiolytic and discriminative effects of the AMPA antagonist 2,3-dihydroxy-6-nitro-7-sulfamoyl-benzo(f)quinoxaline (NBQX). J. Pharmacol. Exp. Ther. 1995, 274, 1113–1121. [Google Scholar]
- Matsuoka, Y.; Kitamura, Y.; Tsukahara, T.; Terai, K.; Tooyama, I.; Kimura, H.; Taniguchi, T. Neuroprotective effects of NBQX on hypoxia-induced neuronal damage in rat hippocampus. Neuroreport 1995, 6, 2205–2208. [Google Scholar] [CrossRef]
- Löscher, W.; Hönack, D. Effects of the non-NMDA antagonists NBQX and the 2,3-benzodiazepine GYKI 52466 on different seizure types in mice: Comparison with diazepam and interactions with flumazenil. Br. J. Pharmacol. 1994, 113, 1349–1357. [Google Scholar] [CrossRef]
- Catarzi, D.; Colotta, V.; Varano, F. Competitive AMPA receptor antagonists. Med. Res. Rev. 2007, 27, 239–278. [Google Scholar] [CrossRef]
- Nikam, S.S.; Kornberg, B.E. AMPA receptor antagonists. Curr. Med. Chem. 2001, 8, 155–170. [Google Scholar] [CrossRef]
- Larsen, A.M.; Bunch, L. Medicinal chemistry of competitive kainate receptor antagonists. ACS Chem. Neurosci. 2011, 2, 60–74. [Google Scholar] [CrossRef] [PubMed]
- Shimizu-Sasamata, M.; Kawasaki-Yatsugi, S.; Okada, M.; Sakamoto, S.; Yatsugi, S.; Togami, J.; Hatanaka, K.; Ohmori, J.; Koshiya, K.; Usuda, S.; et al. YM90K: Pharmacological characterization as a selective and potent alpha-amino-3-hydroxy-5-methylisoxazole-4-propionate/kainate receptor antagonist. J. Pharmacol. Exp. Ther. 1996, 276, 84–92. [Google Scholar] [PubMed]
- Takahashi, M.; Kohara, A.; Shishikura, J.; Kawasaki-Yatsugi, S.; Ni, J.W.; Yatsugi, S.; Sakamoto, S.; Okada, M.; Shimizu-Sasamata, M.; Yamaguchi, T. YM872: A selective, potent and highly water-soluble alpha-amino-3-hydroxy-5-methylisoxazole-4-propionic acid receptor antagonist. CNS Drug Rev. 2002, 8, 337–352. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, T.; Hoshino, S.; Kobayashi, S.; Asakura, T.; Takahashi, M.; Atsumi, T.; Teramoto, A. The glutamate AMPA receptor antagonist, YM872, attenuates cortical tissue loss, regional cerebral edema, and neurological motor deficits after experimental brain injury in rats. J. Neurotrauma 2003, 20, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Turski, L.; Huth, A.; Sheardown, M.J.; McDonald, F.; Neuhaus, R.; Schneider, H.H.; Dirnagl, U.; Wiegand, F.; Jacobsen, P.; Ottow, E. ZK200775: A phosphonate quinoxalinedione AMPA antagonist for neuroprotection in stroke and trauma. Proc. Natl. Acad. Sci. USA 1998, 95, 10960–10965. [Google Scholar] [CrossRef]
- Terai, K.; Suzuki, M.; Sasamata, M. Therapeutic Agent for Brain Hemorrhage. WO2004002488, 8 January 2004. [Google Scholar]
- Ishiuchi, S. Remedy for Glioblastoma. CA2479495, 9 October 2003. [Google Scholar]
- Kohara, A.; Okada, M.; Tsutsumi, R.; Ohno, K.; Takahashi, M.; Shimizu-Sasamata, M.; Shishikura, J.; Inami, H.; Sakamoto, S.; Yamaguchi, T. In-vitro characterization of YM872, a selective, potent and highly water-soluble alpha-amino-3-hydroxy-5-methylisoxazole-4-propionate receptor antagonist. J. Pharm. Pharmacol. 1998, 50, 795–801. [Google Scholar] [CrossRef]
- Pallesen, J.; Møllerud, S.; Frydenvang, K.; Pickering, D.S.; Bornholdt, J.; Nielsen, B.; Pasini, D.; Han, L.; Marconi, L.; Kastrup, J.S.; et al. N1-substituted quinoxaline-2,3-diones as kainate receptor antagonists: X-ray crystallography, structure−affinity relationships, and in vitro pharmacology. ACS Chem. Neurosci. 2019, 10, 1841–1853. [Google Scholar] [CrossRef]
- Madsen, U.; Stensbol, T.; Krogsgaard-Larsen, P. Inhibitors of ampa and kainate receptors. Curr. Med. Chem. 2012, 8, 1291–1301. [Google Scholar] [CrossRef]
- Lubisch, W.; Behl, B.; Henn, C.; Hofmann, H.P.; Reeb, J.; Regner, F.; Vierling, M. Pyrrolylquinoxalinediones carrying a piperazine residue represent highly potent and selective ligands to the homomeric kainate receptor GluR5. Bioorg. Med. Chem. Lett. 2002, 12, 2113–2116. [Google Scholar] [CrossRef]
- Møllerud, S.; Hansen, R.B.; Pallesen, J.; Temperini, P.; Pasini, D.; Bornholt, J.; Nielsen, B.; Mamedova, E.; Chalupnik, P.; Paternain, A.V.; et al. N-(7-(1H-Imidazol-1-yl)-2,3-dioxo-6-(trifluoromethyl)-3,4-dihydroquinoxalin-1(2H)-yl)benzamide, a new kainate receptor selective antagonist and analgesic: Synthesis, X-ray crystallography, structure-affinity relationships, and in vitro and in vivo pharmacology. ACS Chem. Neurosci. 2019, 10, 4685–4695. [Google Scholar] [CrossRef]
- Pøhlsgaard, J.; Frydenvang, K.; Madsen, U.; Kastrup, J.S. Lessons from more than 80 structures of the GluA2 ligand-binding domain in complex with agonists, antagonists and allosteric modulators. Neuropharmacology 2011, 60, 135–150. [Google Scholar] [CrossRef]
- Chałupnik, P.; Vialko, A.; Pickering, D.S.; Hinkkanen, M.; Donbosco, S.; Møller, T.C.; Jensen, A.A.; Nielsen, B.; Bay, Y.; Kristensen, A.S.; et al. Discovery of the first highly selective antagonist of the GluK3 kainate receptor subtype. Int. J. Mol. Sci. 2022, 23, 8797. [Google Scholar] [CrossRef]
- Demmer, C.S.; Møller, C.; Brown, P.M.; Han, L.; Pickering, D.S.; Nielsen, B.; Bowie, D.; Frydenvang, K.; Kastrup, J.S.; Bunch, L. Binding mode of an α-amino acid-linked quinoxaline-2,3-dione analogue at glutamate receptor subtype GluK1. ACS Chem. Neurosci. 2015, 6, 845–854. [Google Scholar] [CrossRef]
- Dolman, N.P.; Troop, H.M.; More, J.C.; Alt, A.; Knauss, J.L.; Nistico, R.; Jack, S.; Morley, R.M.; Bortolotto, Z.A.; Roberts, P.J.; et al. Synthesis and pharmacology of willardiine derivatives acting as antagonists of kainate receptors. J. Med. Chem. 2005, 48, 7867–7881. [Google Scholar] [CrossRef]
- Bleakman, D.; Ogden, A.-M.; Ornstein, P.L.; Hoo, K. Pharmacological characterization of a GluR6 kainate receptor in cultured hippocampal neurons. Eur. J. Pharmacol. 1999, 378, 331–337. [Google Scholar] [CrossRef]
- Demmer, C.S.; Rombach, D.; Liu, N.; Nielsen, B.; Pickering, D.S.; Bunch, L. Revisiting the quinoxalinedione scaffold in the construction of new ligands for the ionotropic glutamate receptors. ACS Chem. Neurosci. 2017, 8, 2477–2495. [Google Scholar] [CrossRef]
- More, J.C.A.; Troop, H.M.; Dolman, N.P.; Jane, D.E. Structural requirements for novel willardiine derivatives acting as AMPA and kainate receptor antagonists. Br. J. Pharmacol. 2003, 138, 1093–1100. [Google Scholar] [CrossRef]
- More, J.C.A.; Nistico, R.; Dolman, N.P.; Clarke, V.R.J.; Alt, A.J.; Ogden, A.M.; Buelens, F.P.; Troop, H.M.; Kelland, E.E.; Pilato, F.; et al. Characterisation of UBP296: A novel, potent and selective kainate receptor antagonist. Neuropharmacology 2004, 47, 46–64. [Google Scholar] [CrossRef]
- Mayer, M.L.; Ghosal, A.; Dolman, N.P.; Jane, D.E. Crystal structures of the kainate receptor GluR5 ligand binding core dimer with novel GluR5-selective antagonists. J. Neurosci. 2006, 26, 2852–2861. [Google Scholar] [CrossRef]
- Dolman, N.P.; More, J.C.; Alt, A.; Knauss, J.L.; Pentikainen, O.T.; Glasser, C.R.; Bleakman, D.; Mayer, M.L.; Collingridge, G.L.; Jane, D.E. Synthesis and pharmacological characterization of N3-substituted willardiine derivatives: Role of the substituent at the 5-position of the uracil ring in the development of highly potent and selective GLUK5 kainate receptor antagonists. J. Med. Chem. 2007, 50, 1558–1570. [Google Scholar] [CrossRef]
- Jane, D.E.; Lodge, D.; Collingridge, G.L. Kainate receptors: Pharmacology, function and therapeutic potential. Neuropharmacology 2009, 56, 90–113. [Google Scholar] [CrossRef]
- Stayte, S.; Laloli, K.J.; Rentsch, P.; Lowth, A.; Li, K.M.; Pickford, R.; Vissel, B. The kainate receptor antagonist UBP310 but not single deletion of GluK1, GluK2, or GluK3 subunits, inhibits MPTP-induced degeneration in the mouse midbrain. Exp. Neurol. 2020, 323, 113062. [Google Scholar] [CrossRef] [PubMed]
- Pollok, S.; Reiner, A. Subunit-selective iGluR antagonists can potentiate heteromeric receptor responses by blocking desensitization. Proc. Natl. Acad. Sci. USA 2020, 117, 25851–25858. [Google Scholar] [CrossRef] [PubMed]
- Dargan, S.L.; Clarke, V.R.J.; Alushin, G.M.; Sherwood, J.L.; Nisticò, R.; Bortolotto, Z.A.; Ogden, A.M.; Bleakman, D.; Doherty, A.J.; Lodge, D.; et al. ACET is a highly potent and specific kainate receptor antagonist: Characterisation and effects on hippocampal mossy fibre function. Neuropharmacology 2009, 56, 121–130. [Google Scholar] [CrossRef] [PubMed]
- More, J.C.; Troop, H.M.; Jane, D.E. The novel antagonist 3-CBW discriminates between kainate receptors expressed on neonatal rat motoneurones and those on dorsal root C-fibres. Br. J. Pharmacol. 2002, 137, 1125–1133. [Google Scholar] [CrossRef]
- Krogsgaard-Larsen, N.; Storgaard, M.; Møller, C.; Demmer, C.S.; Hansen, J.; Han, L.; Monrad, R.N.; Nielsen, B.; Tapken, D.; Pickering, D.S.; et al. Structure-activity relationship study of ionotropic glutamate receptor antagonist (2S,3R)-3-(3-carboxyphenyl)pyrrolidine-2-carboxylic acid. J. Med. Chem. 2015, 58, 6131–6150. [Google Scholar] [CrossRef]
- Gilron, I.; Max, M.B.; Lee, G.; Booher, S.L.; Sang, C.N.; Chappell, A.S.; Dionne, R.A. Effects of the 2-amino-3-hydroxy-5-methyl-4-isoxazole-proprionic acid/kainate antagonist LY293558 on spontaneous and evoked postoperative pain. Clin. Pharmacol. Ther. 2000, 68, 320–327. [Google Scholar] [CrossRef]
- Sang, C.N.; Hostetter, M.P.; Gracely, R.H.; Chappell, A.S.; Schoepp, D.D.; Lee, G.; Whitcup, S.; Caruso, R.; Max, M.B. AMPA/kainate antagonist LY293558 reduces capsaicin-evoked hyperalgesia but not pain in normal skin in humans. Anesthesiology 1998, 89, 1060–1067. [Google Scholar] [CrossRef]
- Lee, H.J.; Pogatzki-Zahn, E.M.; Brennan, T.J. The effect of the AMPA/kainate receptor antagonist LY293558 in a rat model of postoperative pain. J. Pain Palliat. Care Pharmacother. 2006, 7, 768–777. [Google Scholar] [CrossRef]
- Aroniadou-Anderjaska, V.; Figueiredo, T.H.; Apland, J.P.; Braga, M.F. Targeting the glutamatergic system to counteract organophosphate poisoning: A novel therapeutic strategy. Neurobiol. Dis. 2020, 133, 104406. [Google Scholar] [CrossRef]
- Figueiredo, T.H.; Qashu, F.; Apland, J.P.; Aroniadou-Anderjaska, V.; Souza, A.P.; Braga, M.F.M. The GluK1 (GluR5) kainate/ -amino-3-hydroxy-5-methyl-4- isoxazolepropionic acid receptor antagonist LY293558 reduces soman-induced seizures and neuropathology. J. Pharmacol. Exp. Ther. 2011, 336, 303–312. [Google Scholar] [CrossRef]
- Martinez-Perez, J.A.; Iyengar, S.; Shannon, H.E.; Bleakman, D.; Alt, A.; Arnold, B.M.; Bell, M.G.; Bleisch, T.J.; Castaño, A.M.; Del Prado, M.; et al. GluK1 antagonists from 6-(carboxy)phenyl decahydroisoquinoline derivatives. SAR and evaluation of a prodrug strategy for oral efficacy in pain models. Bioorg. Med. Chem. Lett. 2013, 23, 6459–6462. [Google Scholar] [CrossRef]
- O’Neill, M.J.; Bogaert, L.; Hicks, C.A.; Bond, A.; Ward, M.A.; Ebinger, G.; Ornstein, P.L.; Michotte, Y.; Lodge, D. LY377770, a novel iGlu5 kainate receptor antagonist with neuroprotective effects in global and focal cerebral ischaemia. Neuropharmacology 2000, 39, 1575–1588. [Google Scholar] [CrossRef]
- Smolders, I.; Bortolotto, Z.A.; Clarke, V.R.; Warre, R.; Khan, G.M.; O’Neill, M.J.; Ornstein, P.L.; Bleakman, D.; Ogden, A.; Weiss, B.; et al. Antagonists of GLU(K5)-containing kainate receptors prevent pilocarpine-induced limbic seizures. Nat. Neurosci. 2002, 5, 796–804. [Google Scholar] [CrossRef]
- Simmons, R.M.; Li, D.L.; Hoo, K.H.; Deverill, M.; Ornstein, P.L.; Iyengar, S. Kainate GluR5 receptor subtype mediates the nociceptive response to formalin in the rat. Neuropharmacology 1998, 37, 25–36. [Google Scholar] [CrossRef]
- Palecek, J.; Neugebauer, V.; Carlton, S.M.; Iyengar, S.; Willis, W.D. The effect of a kainate GluR5 receptor antagonist on responses of spinothalamic tract neurons in a model of peripheral neuropathy in primates. Pain 2004, 111, 151–161. [Google Scholar] [CrossRef]
- Filla, S.A.; Winter, M.A.; Johnson, K.W.; Bleakman, D.; Bell, M.G.; Bleisch, T.J.; Castaño, A.M.; Clemens-Smith, A.; Del Prado, M.; Dieckman, D.K.; et al. Ethyl (3s,4aR,6S,8aR)-6-(4-ethoxycar-bonylimidazol-1-ylmethyl) decahydroiso-quinoline-3-carboxylic ester: A prodrug of a GluR5 kainate receptor antagonist active in two animal models of acute migraine. J. Med. Chem. 2002, 45, 4383–4386. [Google Scholar] [CrossRef]
- Weiss, B.; Alt, A.; Ogden, A.M.; Gates, M.; Dieckman, D.K.; Clemens-Smith, A.; Ho, K.H.; Jarvie, K.; Rizkalla, G.; Wright, R.A.; et al. Pharmacological characterization of the competitive GLUK5 receptor antagonist decahydroisoquinoline LY466195 in vitro and in vivo. J. Pharmacol. Exp. Ther. 2006, 318, 772–781. [Google Scholar] [CrossRef]
- Alushin, G.M.; Jane, D.; Mayer, M.L. Binding site and ligand flexibility revealed by high resolution crystal structures of GluK1 competitive antagonists. Neuropharmacology 2011, 60, 126–134. [Google Scholar] [CrossRef]
- Martinez-Perez, J.A.; Iyengar, S.; Shannon, H.E.; Bleakman, D.; Alt, A.; Clawson, D.K.; Arnold, B.M.; Bell, M.G.; Bleisch, T.J.; Castaño, A.M.; et al. GluK1 antagonists from 6-(tetrazolyl)phenyl decahydroisoquinoline derivatives: In vitro profile and in vivo analgesic efficacy. Bioorg. Med. Chem. Lett. 2013, 23, 6463–6466. [Google Scholar] [CrossRef]
- Chappell, A.S.; Iyengar, S.; Lobo, E.D.; Prucka, W.R. Results from clinical trials of a selective ionotropic glutamate receptor 5 (iGluR5) antagonist, LY5454694 tosylate, in 2 chronic pain conditions. Pain 2014, 155, 1140–1149. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, M.J.; Bond, A.; Ornstein, P.L.; Ward, M.A.; Hicks, C.A.; Hoo, K.; Bleakman, D.; Lodge, D. Decahydroisoquinolines: Novel competitive AMPA/kainate antagonists with neuroprotective effects in global cerebral ischaemia. Neuropharmacology 1998, 37, 1211–1222. [Google Scholar] [CrossRef]
- Madsen, U.; Bang-Andersen, B.; Brehm, L.; Christensen, I.T.; Ebert, B.; Kristoffersen, I.T.; Lang, Y.; Krogsgaard-Larsen, P. Synthesis and pharmacology of highly selective carboxy and phosphono isoxazole amino acid AMPA receptor antagonists. J. Med. Chem. 1996, 39, 1682–1691. [Google Scholar] [CrossRef]
- Moller, E.H.; Egebjerg, J.; Brehm, L.; Stensbol, T.B.; Johansen, T.N.; Madsen, U.; Krogsgaard-Larsen, P. Resolution, absolute stereochemistry, and enantiopharmacology of the GluR1-4 and GluR5 antagonist 2-amino-3-[5-tert-butyl-3-(phosphonomethoxy)-4-isoxazolyl]propionic acid. Chirality 1999, 11, 752–759. [Google Scholar] [CrossRef]
- Hald, H.; Naur, P.; Pickering, D.S.; Sprogøe, D.; Madsen, U.; Timmermann, D.B.; Ahring, P.K.; Liljefors, T.; Schousboe, A.; Egebjerg, J.; et al. Partial agonism and antagonism of the ionotropic glutamate receptor iGLuR5: Structures of the ligand-binding core in complex with domoic acid and 2-amino-3-[5-tert-butyl-3-(phosphonomethoxy)-4-isoxazolyl]propionic acid. J. Biol. Chem. 2007, 282, 25726–25736. [Google Scholar] [CrossRef] [PubMed]
- Szymańska, E.; Frydenvang, K.; Contreras-Sanz, A.; Pickering, D.S.; Frola, E.; Serafimoska, Z.; Nielsen, B.; Kastrup, J.S.; Johansen, T.N. A new phenylalanine derivative acts as an antagonist at the AMPA receptor GluA2 and introduces partial domain closure: Synthesis, resolution, pharmacology, and crystal structure. J. Med. Chem. 2011, 54, 7289–7298. [Google Scholar] [CrossRef]
- Venskutonyte, R.; Frydenvang, K.; Valades, E.A.; Szymańska, E.; Johansen, T.N.; Kastrup, J.S.; Pickering, D.S. Structural and pharmacological characterization of phenylalanine-based AMPA receptor antagonists at kainate receptors. ChemMedChem 2012, 7, 1793–1798. [Google Scholar] [CrossRef]
- Szymanska, E.; Pickering, D.S.; Nielsen, B.; Johansen, T.N. 3-Substituted phenylalanines as selective AMPA- and kainate receptor ligands. Bioorg. Med. Chem. 2009, 17, 6390–6401. [Google Scholar] [CrossRef]
- Szymańska, E.; Frydenvang, K.; Pickering, D.S.; Krintel, C.; Nielsen, B.; Kooshki, A.; Zachariassen, L.G.; Olsen, L.; Kastrup, J.S.; Johansen, T.N. Studies on aryl-substituted phenylalanines: Synthesis, activity, and different binding modes at AMPA receptors. J. Med. Chem. 2016, 59, 448–461. [Google Scholar] [CrossRef]
- Szymańska, E.; Chałupnik, P.; Szczepańska, K.; Cuñado Moral, A.M.; Pickering, D.S.; Nielsen, B.; Johansen, T.N.; Kieć-Kononowicz, K. Design, synthesis and structure-activity relationships of novel phenylalanine-based amino acids as kainate receptors ligands. Bioorg. Med. Chem. Lett. 2016, 26, 5568–5572. [Google Scholar] [CrossRef]
- Larsen, A.M.; Venskutonytè, R.; Valadés, E.A.; Nielsen, B.; Pickering, D.S.; Bunch, L. Discovery of a new class of ionotropic glutamate receptor antagonists by the rational design of (2 S,3 R)-3-(3-carboxyphenyl)-pyrrolidine-2-carboxylic acid. ACS Chem. Neurosci. 2011, 2, 107–114. [Google Scholar] [CrossRef]
- Krogsgaard-Larsen, N.; Delgar, C.G.; Koch, K.; Brown, P.M.G.E.; Møller, C.; Han, L.; Huynh, T.H.V.; Hansen, S.W.; Nielsen, B.; Bowie, D.; et al. Design and synthesis of a series of l-trans-4-substituted prolines as selective antagonists for the ionotropic glutamate receptors including functional and X-ray crystallographic studies of new subtype selective kainic acid receptor subtype 1 (GluK1) antagonist (2S,4R)-4-(2-carboxyphenoxy)pyrrolidine-2-carboxylic acid. J. Med. Chem. 2017, 60, 441–457. [Google Scholar] [CrossRef]
- Bunch, L. Pyrrolidine-2-Carboxylic Acid Derivatives as iGluR Antagonists. US20140235576A1, 21 August 2014. [Google Scholar]
- Bunch, L. Substituted 4-Proline Derivatives as iGluR Antagonists. WO2015003723A1, 15 January 2015. [Google Scholar]
- Sanders, J.M.; Ito, K.; Settimo, L.; Pentikäinen, O.T.; Shoji, M.; Sasaki, M.; Johnson, M.S.; Sakai, R.; Swanson, G.T. Divergent pharmacological activity of novel marine-derived excitatory amino acids on glutamate receptors. J. Pharmacol. Exp. Ther. 2005, 314, 1068–1078. [Google Scholar] [CrossRef]
- Sanders, J.M.; Pentikäinen, O.T.; Settimo, L.; Pentikäinen, U.; Shoji, M.; Sasaki, M.; Sakai, R.; Johnson, M.S.; Swanson, G.T. Determination of binding site residues responsible for the subunit selectivity of novel marine-derived compounds on kainate receptors. Mol. Pharmacol. 2006, 69, 1849–1860. [Google Scholar] [CrossRef]
- Frydenvang, K.; Lash, L.L.; Naur, P.; Postila, P.A.; Pickering, D.S.; Smith, C.M.; Gajhede, M.; Sasaki, M.; Sakai, R.; Pentikänen, O.T.; et al. Full domain closure of the ligand-binding core of the ionotropic glutamate receptor iGluR5 induced by the high affinity agonist dysiherbaine and the functional antagonist 8,9-dideoxyneodysiherbaine. J. Biol. Chem. 2009, 284, 14219–14229. [Google Scholar] [CrossRef]
- Qiu, C.-S.; Lash-Van Wyhe, L.; Sasaki, M.; Sakai, R.; Swanson, G.T.; Gereau, R.W. Antinociceptive effects of MSVIII-19, a functional antagonist of the GluK1 kainate receptor. Pain 2011, 152, 1052–1060. [Google Scholar] [CrossRef]
- Lash, L.L.; Sanders, J.M.; Akiyama, N.; Shoji, M.; Postila, P.; Pentikäinen, O.T.; Sasaki, M.; Sakai, R.; Swanson, G.T. Novel analogs and stereoisomers of the marine toxin neodysiherbaine with specificity for kainate receptors. J. Pharmacol. Exp. Ther. 2008, 324, 484–496. [Google Scholar] [CrossRef]
- Swanson, G.T.; Lash, L.; Sakai, R. Kainate Receptor-Selective Epimeric Analogs of Dysiherbaine. U.S. Patent US20090118358A1, 5 July 2009. [Google Scholar]
- Brogi, S.; Brindisi, M.; Butini, S.; Kshirsagar, G.U.; Maramai, S.; Chemi, G.; Gemma, S.; Campiani, G.; Novellino, E.; Fiorenzani, P.; et al. (S)-2-Amino-3-(5-methyl-3-hydroxyisoxazol-4-yl)propanoic acid (AMPA) and kainate receptor ligands: Further exploration of bioisosteric replacements and structural and biological investigation. J. Med. Chem. 2018, 61, 2124–2130. [Google Scholar] [CrossRef]
- Venskutonyte, R.; Butini, S.; Coccone, S.S.; Gemma, S.; Brindisi, M.; Kumar, V.; Guarino, E.; Maramai, S.; Valenti, S.; Amir, A.; et al. Selective kainate receptor (GluK1) ligands structurally based upon 1H-cyclopentapyrimidin-2,4(1H3H)- dione: Synthesis, molecular modeling, and pharmacological and biostructural characterization. J. Med. Chem. 2011, 54, 4793–4805. [Google Scholar] [CrossRef]
- Poulie, C.B.M.; Larsen, Y.; Leteneur, C.; Barthet, G.; Bjørn-Yoshimoto, W.E.; Malhaire, F.; Nielsen, B.; Pin, J.-P.; Mulle, C.; Pickering, D.S.; et al. (S)-2-Mercaptohistidine: A first selective orthosteric GluK3 antagonist. ACS Chem. Neurosci. 2022, 13, 1580–1587. [Google Scholar] [CrossRef]
- Mula, M. Emerging drugs for focal epilepsy. Expert Opin. Emerg. Drugs 2013, 18, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Faught, E. BGG492 (selurampanel), an AMPA/kainate receptor antagonist drug for epilepsy. Expert Opin. Investig. Drugs 2014, 23, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Orain, D.; Tasdelen, E.; Haessig, S.; Koller, M.; Picard, A.; Dubois, C.; Lingenhoehl, K.; Desrayaud, S.; Floersheim, P.; Carcache, D.; et al. Design and synthesis of Selurampanel, a novel orally active and competitive AMPA receptor antagonist. ChemMedChem 2017, 12, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, E.O.; Varming, T.; Mathiesen, C.; Jensen, L.H.; Moller, A.; Gouliaev, A.H.; Wätjen, F.; Drejer, J. SPD 502: A water-soluble and in vivo long-lasting AMPA antagonist with neuroprotective activity. J. Pharmacol. Exp. Ther. 1999, 289, 1492–1501. [Google Scholar] [PubMed]
- Keppel Hesselink, J.M. NS1209/SPD 502, a novel selective AMPA antagonist for stroke, neuropathic pain or epilepsy? Drug development lessons learned. Drug Dev. Res. 2017, 78, 75–80. [Google Scholar] [CrossRef]
- Mignani, S.; Bohme, G.A.; Birraux, G.; Boireau, A.; Jimonet, P.; Damour, D.; Genevois-Borella, A.; Debono, M.W.; Pratt, J.; Vuilhorgne, M.; et al. 9-Carboxymethyl-5H,10H-imidazo[1,2-a]indeno[1,2-e]pyrazin-4-one-2-carbocylic acid (RPR117824): Selective anticonvulsive and neuroprotective AMPA antagonist. Bioorg. Med. Chem. 2002, 10, 1627–1637. [Google Scholar] [CrossRef]
- Krampfl, K.; Schlesinger, F.; Cordes, A.L.; Bufler, J. Molecular analysis of the interaction of the pyrazine derivatives RPR119990 and RPR117824 with human AMPA-type glutamate receptor channels. Neuropharmacology 2006, 50, 479–490. [Google Scholar] [CrossRef]
- Gormsen, L.; Finnerup, N.B.; Almqvist, P.M.; Jensen, T.S. The efficacy of the AMPA receptor antagonist ns1209 and lidocaine in nerve injury pain: A randomized, double-blind, placebo-controlled, three-way crossover study. Anesth. Analg. 2009, 108, 1311–1319. [Google Scholar] [CrossRef]
- Colotta, V.; Catarzi, D.; Varano, F.; Lenzi, O.; Filacchioni, G.; Costagli, C.; Galli, A.; Ghelardini, C.; Galeotti, N.; Gratteri, P.; et al. Structural investigation of the 7-chloro-3-hydroxy-1H-quinazoline-2,4-dione scaffold to obtain AMPA and kainate receptor selective antagonists. Synthesis, pharmacological, and molecular modeling studies. J. Med. Chem. 2006, 49, 6015–6026. [Google Scholar] [CrossRef]
- Colotta, V.; Lenzi, O.; Catarzi, D.; Varano, F.; Squarcialupi, L.; Costagli, C.; Galli, A.; Ghelardini, C.; Pugliese, A.M.; Maraula, G.; et al. 3-Hydroxy-1H-quinazoline-2,4-dione derivatives as new antagonists at ionotropic glutamate receptors: Molecular modeling and pharmacological studies. Eur. J. Med. Chem. 2012, 54, 470–482. [Google Scholar] [CrossRef]
- Kasper, C.; Pickering, D.S.; Mirza, O.; Olsen, L.; Kristensen, A.S.; Greenwood, J.R.; Liljefors, T.; Schousboe, A.; Wätjen, F.; Gajhede, M.; et al. The structure of a mixed GluR2 ligand-binding core dimer in complex with (S)-glutamate and the antagonist (S)-NS1209. J. Mol. Biol. 2006, 357, 1184–1201. [Google Scholar] [CrossRef]
- Wilding, T.J.; Huettner, J.E. Activation and desensitization of hippocampal kainate receptors. J. Neurosci. 1997, 17, 2713–2721. [Google Scholar] [CrossRef]
- Bleakman, D.; Ballyk, B.A.; Schoepp, D.D.; Palmer, A.J.; Bath, C.P.; Sharpe, E.F.; Woolley, M.L.; Bufton, H.R.; Kamboj, R.K.; Tarnawa, I.; et al. Activity of 2,3-benzodiazepines at native rat and recombinant human glutamate receptors in vitro: Stereospecificity and selectivity profiles. Neuropharmacology 1996, 35, 1689–1702. [Google Scholar] [CrossRef]
- Christensen, J.K.; Varming, T.; Ahring, P.K.; Jørgensen, T.D.; Nielsen, E. In vitro characterization of 5-carboxyl-2,4-di-benzamidobenzoic acid (NS3763), a noncompetitive antagonist of GLUK5 receptors. J. Pharmacol. Exp. Ther. 2004, 309, 1003–1010. [Google Scholar] [CrossRef]
- Anna Kaczor, A.; Kronbach, C.; Unverferth, K.; Pihlaja, K.; Wiinamaki, K.; Sinkkonen, J.; Kijkowska-Murak, U.; Wrobel, T.; Stachal, T.; Matosiuk, D. Novel non-competitive antagonists of kainate GluK1/GluK2 receptors. Lett. Drug Des. Discov. 2012, 9, 891–898. [Google Scholar] [CrossRef]
- Shvarts, V.; Chung, S. Perampanel: Newly approved, novel antiepileptic medication for partial-onset seizures. Expert Rev. Neurother. 2013, 13, 131–134. [Google Scholar] [CrossRef]
- Fukushima, K.; Hatanaka, K.; Sagane, K.; Ido, K. Inhibitory effect of anti-seizure medications on ionotropic glutamate receptors: Special focus on AMPA receptor subunits. Epilepsy Res. 2020, 167, 106452. [Google Scholar] [CrossRef]
- Taniguchi, S.; Stolz, J.R.; Swanson, G.T. The antiseizure drug Perampanel is a subunit-selective negative allosteric modulator of kainate receptors. J. Neurosci. 2022, 42, 5499–5509. [Google Scholar] [CrossRef]
- Kanemura, H.; Sano, F.; Aihara, M. Usefulness of perampanel with concomitant levetiracetam for patients with drug-resistant epilepsy. Eur. J. Paediatr. Neurol. 2019, 23, 197–203. [Google Scholar] [CrossRef]
- Trigg, A.; Brohan, E.; Cocks, K.; Jones, A.; Tahami Monfared, A.A.; Chabot, I.; Meier, G.; Campbell, R.; Li, H.; Ngo, L.Y. Health-related quality of life in pediatric patients with partial onset seizures or primary generalized tonic-clonic seizures receiving adjunctive perampanel. Epilepsy Behav. 2021, 118, 107938. [Google Scholar] [CrossRef]
- Mehndiratta, M.M.; Manoj, G.; Jabeen, S.A.; Patten, A. Efficacy and safety of adjunctive perampanel in patients with focal seizures or generalized tonic-clonic seizures: Post hoc analysis of Phase II and Phase III double-blind and open-label extension studies in India. Epilepsia Open 2021, 8, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Fogarasi, A.; Flamini, R.; Milh, M.; Phillips, S.; Yoshitomi, S.; Patten, A.; Takase, T.; Laurenza, A.; Ngo, L.Y. Open-label study to investigate the safety and efficacy of adjunctive perampanel in pediatric patients. Epilepsia 2020, 61, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Nishida, T.; Lee, S.K.; Wu, T.; Tiamkao, S.; Dash, A. Efficacy and safety of perampanel in generalized and focal to bilateral tonic-clonic seizures: A comparative study of Asian and non-Asian populations. Epilepsia 2019, 60, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Renfroe, J.B.; Mintz, M.; Davis, R.; Ferreira, J.; Dispoto, S.; Ferry, J.; Umetsu, Y.; Rege, B.; Majid, O.; Hussein, Z.; et al. Adjunctive Perampanel oral suspension in pediatric patients from 2 to <12 years of age with epilepsy: Pharmacokinetics, safety, tolerability, and efficacy. J. Child Neurol. 2019, 34, 284–294. [Google Scholar] [CrossRef]
- Usui, N.; Akamatsu, N.; Nakasato, N.; Ohnishi, A.; Kaneko, S.; Hiramatsu, H.; Saeki, K.; Miyagishi, H.; Inoue, Y. Long-term tolerability, safety and efficacy of adjunctive perampanel in the open-label, dose-ascending Study 231 and extension Study 233 in Japanese patients with epilepsy. Seizure 2018, 62, 26–32. [Google Scholar] [CrossRef]
- Lin, K.L.; Lin, J.J.; Chou, M.L.; Hung, P.C.; Hsieh, M.Y.; Chou, I.J.; Lim, S.N.; Wu, T.; Wang, H.S. Efficacy and tolerability of perampanel in children and adolescents with pharmacoresistant epilepsy: The first real-world evaluation in Asian pediatric neurology clinics. Epilepsy Behav. 2018, 85, 188–194. [Google Scholar] [CrossRef]
- Piña-Garza, J.E.; Lagae, L.; Villanueva, V.; Renfroe, J.B.; Laurenza, A.; Williams, B.; Kumar, D.; Meador, K.J. Long-term effects of adjunctive perampanel on cognition in adolescents with partial seizures. Epilepsy Behav. 2018, 83, 50–58. [Google Scholar] [CrossRef]
- Eggert, K.; Squillacote, D.; Barone, P.; Dodel, R.; Katzenschlager, R.; Emre, M.; Lees, A.J.; Rascol, O.; Poewe, W.; Tolosa, E.; et al. Safety and efficacy of perampanel in advanced parkinson’s disease: A randomized, placebo-controlled study. Mov. Disord. 2010, 25, 896–905. [Google Scholar] [CrossRef]
- Rascol, O.; Barone, P.; Behari, M.; Emre, M.; Giladi, N.; Olanow, C.W.; Ruzicka, E.; Bibbiani, F.; Squillacote, D.; Patten, A.; et al. Perampanel in Parkinson disease fluctuations: A double-blind randomized trial with placebo and entacapone. Clin. Neuropharmacol. 2012, 35, 15–20. [Google Scholar] [CrossRef]
- Oskarsson, B.; Mauricio, E.A.; Shah, J.S.; Li, Z.; Rogawski, M.A. Cortical excitability threshold can be increased by the AMPA blocker Perampanel in amyotrophic lateral sclerosis. Muscle Nerve 2021, 64, 215–219. [Google Scholar] [CrossRef]
- Aizawa, H.; Kato, H.; Oba, K.; Kawahara, T.; Okubo, Y.; Saito, T.; Naito, M.; Urushitani, M.; Tamaoka, A.; Nakamagoe, K.; et al. Randomized phase 2 study of perampanel for sporadic amyotrophic lateral sclerosis. J. Neurol. 2022, 269, 885–896. [Google Scholar] [CrossRef]
- Wilding, T.J.; Chai, Y.H.; Huettner, J.E. Inhibition of rat neuronal kainate receptors by cis-unsaturated fatty acids. J. Physiol. 1998, 513 Pt 2, 331–339. [Google Scholar] [CrossRef]
- Wilding, T.J.; Zhou, Y.; Huettner, J.E. Q/R site editing controls kainate receptor inhibition by membrane fatty acids. J. Neurosci. 2005, 25, 9470–9478. [Google Scholar] [CrossRef]
- Wilding, T.J.; Fulling, E.; Zhou, Y.; Huettner, J.E. Amino acid substitutions in the pore helix of GluR6 control inhibition by membrane fatty acids. J. Gen. Physiol. 2008, 132, 85–99. [Google Scholar] [CrossRef]
- Christensen, J.K.; Paternain, A.V.; Selak, S.; Ahring, P.K.; Lerma, J. A mosaic of functional kainate receptors in hippocampal interneurons. J. Neurosci. 2004, 24, 8986–8993. [Google Scholar] [CrossRef]
- Xu, J.; Liu, Y.; Zhang, G.Y. Neuroprotection of GluR5-containing kainate receptor activation against ischemic brain injury through decreasing tyrosine phosphorylation of N-methyl-D-aspartate receptors mediated by Src kinase. J. Biol. Chem. 2008, 283, 29355–29366. [Google Scholar] [CrossRef]
- Valgeirsson, J.; Nielsen, E.; Peters, D.; Varming, T.; Mathiesen, C.; Kristensen, A.S.; Madsen, U. 2-arylureidobenzoic acids: Selective noncompetitive antagonists for the homomeric kainate receptor subtype GluR5. J. Med. Chem. 2003, 46, 5834–5843. [Google Scholar] [CrossRef]
- Valgeirsson, J.; Nielsen, E.; Peters, D.; Mathiesen, C.; Kristensen, A.S.; Madsen, U. Bioisosteric modifications of 2-arylureidobenzoic acids: Selective noncompetitive antagonists for the homomeric kainate receptor subtype GluR5. J. Med. Chem. 2004, 47, 6948–6957. [Google Scholar] [CrossRef]
- Kaczor, A.A.; Karczmarzyk, Z.; Fruziński, A.; Pihlaja, K.; Sinkkonen, J.; Wiinämaki, K.; Kronbach, C.; Unverferth, K.; Poso, A.; Matosiuk, D. Structural studies, homology modeling and molecular docking of novel non-competitive antagonists of GluK1/GluK2 receptors. Bioorg. Med. Chem. 2014, 22, 787–795. [Google Scholar] [CrossRef]
- Bartyzel, A.; Kaczor, A.A.; Mahmoudi, G.; Masoudiasl, A.; Wróbel, T.M.; Pitucha, M.; Matosiuk, D. Experimental and computational structural studies of 2,3,5-trisubstituted and 1,2,3,5-tetrasubstituted indoles as non-competitive antagonists of GluK1/GluK2 receptors. Molecules 2022, 27, 2479. [Google Scholar] [CrossRef]
- Ben-Menachem, E. Topiramate: Current status and therapeutic potential. Expert Opin. Investig. Drugs 1997, 6, 1085–1094. [Google Scholar] [CrossRef] [PubMed]
- Kaminski, R.M.; Banerjee, M.; Rogawski, M.A. Topiramate selectively protects against seizures induced by ATPA, a GluR5 kainate receptor agonist. Neuropharmacology 2004, 46, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Braga, M.F.M.; Aroniadou-Anderjaska, V.; Li, H.; Rogawski, M.A. Topiramate reduces excitability in the basolateral amygdala by selectively inhibiting GluK1 (GluR5) kainate receptors on interneurons and positively modulating GABA_A receptors on principal neurons. J. Pharmacol. Exp. Ther. 2009, 330, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Gryder, D.S.; Rogawski, M.A. Selective antagonism of GluR5 kainate-receptor-mediated synaptic currents by topiramate in rat basolateral amygdala neurons. J. Neurosci. 2003, 23, 7069–7074. [Google Scholar] [CrossRef] [PubMed]
- Strømgaard, K.; Andersen, K.; Krogsgaard-Larsen, P.; Jaroszewski, J.W. Recent advances in the medicinal chemistry of polyamine toxins. Mini-Rev. Med. Chem. 2001, 1, 317–338. [Google Scholar] [CrossRef]
- Brown, P.M.; Aurousseau, M.R.; Musgaard, M.; Biggin, P.C.; Bowie, D. Kainate receptor pore-forming and auxiliary subunits regulate channel block by a novel mechanism. J. Physiol. 2016, 594, 1821–1840. [Google Scholar] [CrossRef]
- Lomeli, H.; Wisden, W.; Köhler, M.; Keinänen, K.; Sommer, B.; Seeburg, P.H. High-affinity kainate and domoate receptors in rat brain. FEBS Lett. 1992, 307, 139–143. [Google Scholar] [CrossRef]
- Bowie, D. Polyamine-mediated channel block of ionotropic glutamate receptors and its regulation by auxiliary proteins. J. Biol. Chem. 2018, 293, 18789–18802. [Google Scholar] [CrossRef]
- Mott, D.D.; Washburn, M.S.; Zhang, S.; Dingledine, R.J. Subunit-dependent modulation of kainate receptors by extracellular protons and polyamines. J. Neurosci. 2003, 23, 1179–1188. [Google Scholar] [CrossRef]
- Quistad, G.B.; Suwanrumpha, S.; Jarema, M.A.; Shapiro, M.J.; Skinner, W.S.; Jamieson, G.C.; Lui, A.; Fu, E.W. Structure of paralytic acylpolyamines from the spider Agelenopsisaperta. Biochem. Biophys. Res. Commun. 1990, 169, 51–56. [Google Scholar] [CrossRef]
- Eldefrawi, A.T.; Eldefrawi, M.E.; Konnot, K.; Mansour, N.A.; Nakanishit, K.; Oltzt, E.; Usherwood, P.N.R. Structure and synthesis of a potent glutamate receptor antagonist in wasp venom (wasp venom toxin/quisqualate receptor). Proc. Natl. Acad. Sci. USA 1988, 85, 4910–4913. [Google Scholar] [CrossRef]
- Kachel, H.S.; Franzyk, H.; Mellor, I.R. Philanthotoxin analogues that selectively inhibit ganglionic nicotinic acetylcholine receptors with exceptional potency. J. Med. Chem. 2019, 62, 6214–6222. [Google Scholar] [CrossRef]
- Vassileiou, C.; Kalantzi, S.; Vachlioti, E.; Athanassopoulos, C.M.; Koutsakis, C.; Piperigkou, Z.; Karamanos, N.; Stivarou, T.; Lymberi, P.; Avgoustakis, K.; et al. New analogs of polyamine toxins from spiders and wasps: Liquid phase fragment synthesis and evaluation of antiproliferative activity. Molecules 2022, 27, 447. [Google Scholar] [CrossRef]
- Verdoni, M.; Roudaut, H.; De Pomyers, H.; Gigmes, D.; Bertin, D.; Luis, J.; Bengeloune, A.H.; Mabrouk, K. ArgTX-636, a polyamine isolated from spider venom: A novel class of melanogenesis inhibitors. Bioorg. Med. Chem. 2016, 24, 5685–5692. [Google Scholar] [CrossRef]
- Kromann, H.; Krikstolaityte, S.; Andersen, A.J.; Andersen, K.; Krogsgaard-Larsen, P.; Jaroszewski, J.W.; Egebjerg, J.; Strømgaard, K. Solid-phase synthesis of polyamine toxin analogues: Potent and selective antagonists of Ca2+-permeable AMPA receptors. J. Med. Chem. 2002, 45, 5745–5754. [Google Scholar] [CrossRef]
- Kachel, H.S.; Patel, R.N.; Franzyk, H.; Mellor, I.R. Block of nicotinic acetylcholine receptors by philanthotoxins is strongly dependent on their subunit composition. Sci. Rep. 2016, 6, 38116. [Google Scholar] [CrossRef]
- Matavel, A.; Estrada, G.; De Marco Almeida, F. Spider venom and drug discovery: A review. In Spider Venoms; Springer: Dordrecht, The Netherlands, 2016; pp. 273–292. [Google Scholar] [CrossRef]
- Magazanik, L.G.; Buldakova, S.L.; Samoilova, M.V.; Gmiro, V.E.; Mellor, I.R.; Usherwood, P.N.R. Block of open channels of recombinant AMPA receptors and native AMPA/kainate receptors by adamantane derivatives. J. Physiol. 1997, 505 Pt 3, 655–663. [Google Scholar] [CrossRef]
- Koike, M.; Iino, M.; Ozawa, S. Blocking effect of 1-naphthyl acetyl spermine on Ca2+-permeable AMPA receptors in cultured rat hippocampal neurons. Neurosci. Res. 1997, 29, 27–36. [Google Scholar] [CrossRef]
- Nelson, J.K.; Frølund, S.U.; Tikhonov, D.B.; Kristensen, A.S.; Strømgaard, K. Synthesis and biological activity of argiotoxin 636 and analogues: Selective antagonists for ionotropic glutamate receptors. Angew. Chem. Int. Ed. 2009, 48, 3087–3091. [Google Scholar] [CrossRef]
- Xiong, X.F.; Poulsen, M.H.; Hussein, R.A.; Nørager, N.G.; Strømgaard, K. Structure-activity relationship study of spider polyamine toxins as inhibitors of ionotropic glutamate receptors. ChemMedChem 2014, 9, 2661–2670. [Google Scholar] [CrossRef]
- Nørager, N.G.; Poulsen, M.H.; Jensen, A.G.; Jeppesen, N.S.; Kristensen, A.S.; Strømgaard, K. Structure−activity relationship studies of n-methylated and n-hydroxylated spider polyamine toxins as inhibitors of ionotropic glutamate receptors. J. Med. Chem. 2014, 57, 4940–4949. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, M.H.; Lucas, S.; Bach, T.B.; Barslund, A.F.; Wenzler, C.; Jensen, C.B.; Kristensen, A.S.; Strømgaard, K. Structure−Activity relationship studies of argiotoxins: Selective and potent inhibitors of ionotropic glutamate receptors. J. Med. Chem. 2013, 56, 1171–1181. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Niu, L. Developing RNA aptamers for potential treatment of neurological diseases. Future Med. Chem. 2019, 11, 551–565. [Google Scholar] [CrossRef] [PubMed]
- Germer, K.; Leonard, M.; Zhang, X. RNA aptamers and their therapeutic and diagnostic applications. Int. J. Biochem. Mol. Biol. 2013, 4, 27–40. [Google Scholar]
- Huang, Z.; Niu, L. RNA aptamers for AMPA receptors. Neuropharmacology 2021, 199, 108761. [Google Scholar] [CrossRef]
- Huang, Z.; Wen, W.; Wu, A.; Niu, L. Chemically modified, α-amino-3-hydroxy-5-methyl-4-isoxazole (AMPA) receptor RNA aptamers designed for in vivo use. ACS Chem. Neurosci. 2017, 8, 2437–2445. [Google Scholar] [CrossRef]
- Jaremko, W.; Huang, Z.; Karl, N.; Pierce, V.D.; Lynch, J.; Niu, L. A kainate receptor-selective RNA aptamer. J. Biol. Chem. 2020, 295, 6280–6288. [Google Scholar] [CrossRef]
- Ni, S.; Yao, H.; Wang, L.; Lu, J.; Jiang, F.; Lu, A.; Zhang, G. Chemical modifications of nucleic acid aptamers for therapeutic purposes. Int. J. Mol. Sci. 2017, 18, 1683. [Google Scholar] [CrossRef]
- Niu, L.; Huang, Z. Chemically Modified AMPA Receptor RNA Aptamers. U.S. Patent US10815486B2, 27 October 2020. [Google Scholar]
- Jaremko, W.J.; Huang, Z.; Wen, W.; Wu, A.; Karl, N.; Niu, L. One aptamer, two functions: The full-length aptamer inhibits AMPA receptors, while the short one inhibits both AMPA and kainate receptors. RNA Dis. 2017, 4, e1560. [Google Scholar]
- Braga, M.F.; Aroniadou-Anderjaska, V. Methods for Treating Neurological Conditions and Exposure to Nerve Agents. WO2017218344A1, 21 December 2017. [Google Scholar]
- Chenard, B.L.; Menniti, F.S.; Saltarelli, M.D. Pharmaceutical Combinations for the Treatment of Stroke and Traumatic Brain Injury. U.S. Patent US20020123510A1, 23 November 2004. [Google Scholar]
- Krissansen, G.W.; Kanwar, J.R. Treatment of Demyelinating Diseases by Administering GPE. WO2002030447, 18 April 2002. [Google Scholar]
- Olney John, W. Safener Drug Combinations for Use with NMDA Antagonist Drugs. U.S. Patent US20120232025A1, 8 September 2011. [Google Scholar]
- Ikonomidou, H. New Use of Glutamate Antagonists for the Treatment of Cancer. WO2000024395A1, 4 May 2000. [Google Scholar]
- Kozachuk, W.E. Methods of Producing Weight Loss and Treatment of Obesity. U.S. Patent US6191117B1, 20 February 2001. [Google Scholar]
- Martynyuk, A.E.; Dennis, D.M.; Gravenstein, N.; Glushakov, A.V.; Yarotskyy, V.; Sumners, C.; Seubert, C. Methods and Materials for Treating Mental Illness. U.S. Patent US20080096870, 24 April 2008. [Google Scholar]
- Steppuhn Karin, G.; Bressler, K.; Gieseler, M.; Stephens David, N.; Turski, L. Pharmaceutical Agent for Treatment of Withdrawal Symptoms. U.S. Patent US5385903A, 31 January 1995. [Google Scholar]


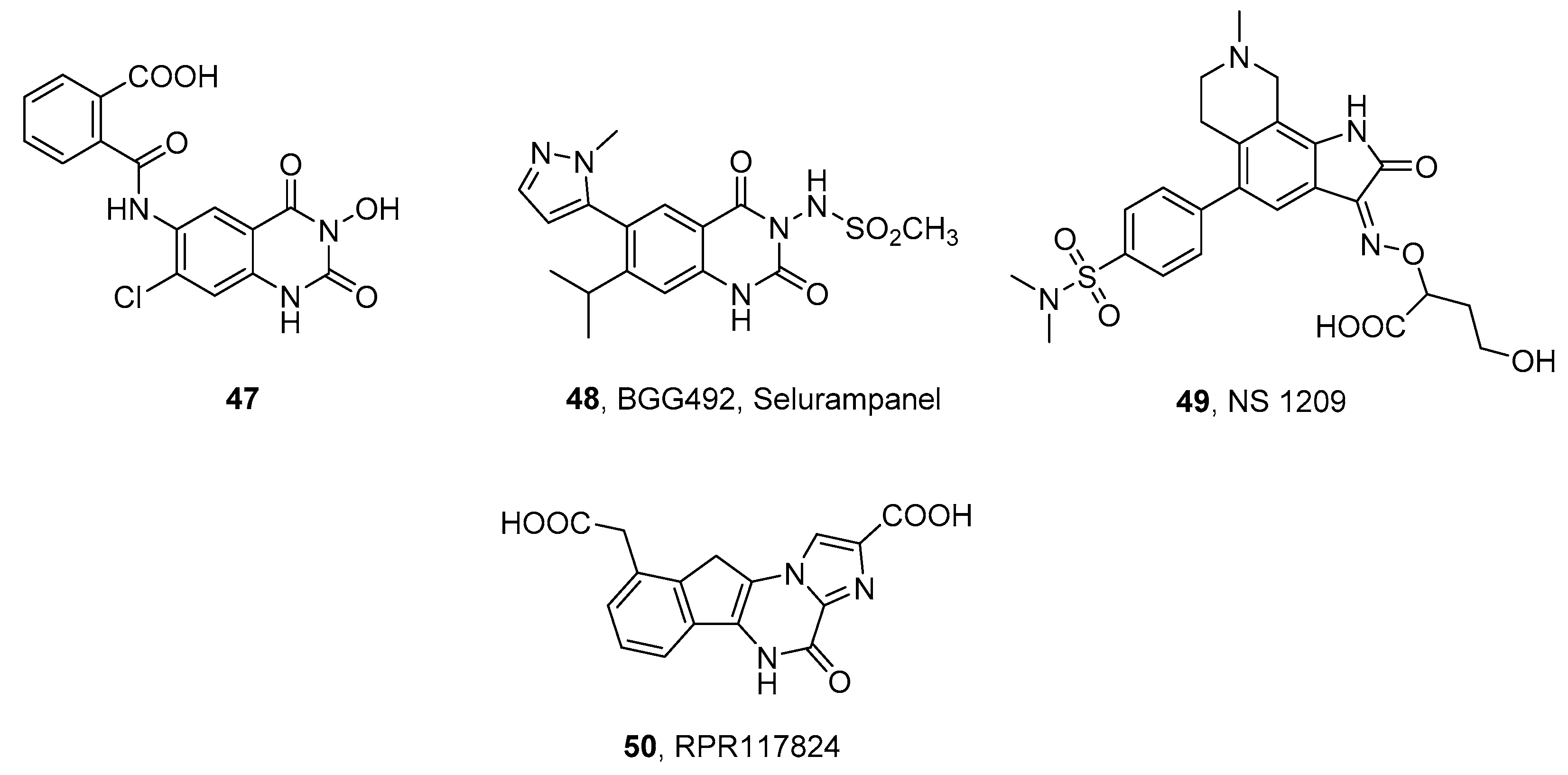

| cmpd | Native iGluRs | Recombinant Homomeric iGluRs | |||
|---|---|---|---|---|---|
| AMPA | KA | GluA2 | GluK1 | GluK3 | |
| 47a | 16 (Ki) | 0.62 (IC50) | nd | nd | nd |
| 48, selurampanel b | 0.19 (IC50) | >100 (IC50) | nd | nd | nd |
| 49, NS 1209 c | 0.043 (IC50) c | 81 (IC50) c | 0.030 (Ki) d | 0.74 (Ki) d | nd |
| 50, RPR117824 e | 0.018 (IC50) | nd | nd | nd | nd |
| cmpd | IC50 [μM] | |||
|---|---|---|---|---|
| Recombinant Homomeric iGluRs | ||||
| GluA1 | GluA2 | GluK1 | GluK2 | |
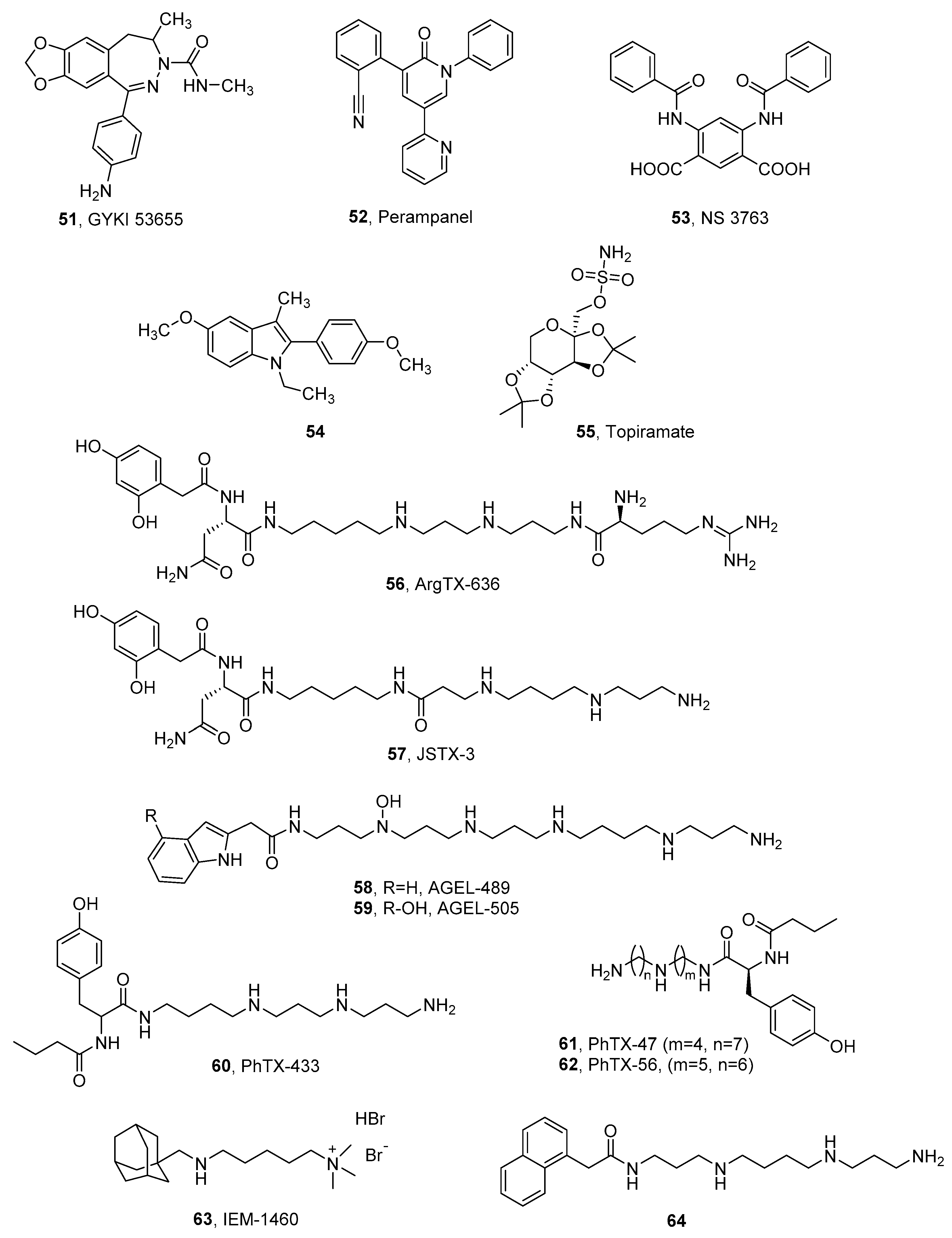
| 51, GYKI 53655 | 5.9 ± 0.1 a | 1.9 ± 0.8 b | >100 b | 198 ± 53 b |
| 53, NS 3763 c | nd | nd | 1.6 ± 0.2 | >30 |
| 54d | nd | nd | 4.0 | 0.7 |
| Receptor | IC50 [μM] |
|---|---|
| GluA4 | 0.56 ± 0.12 |
| GluK1 | 19 ± 5 |
| GluK2 | 26 ± 10 |
| GluK3 | 41 ± 6 |
| GluK1/5 | 2.8 ± 0.3 |
| GluK2/5 | 0.85 ± 0.09 |
| GluK3/5 | 14 ± 8 |
| GluK1/NETO1 | 11 ± 4 |
| GluK1/NETO2 | 19 ± 4 |
| GluK2/NETO1 | 5.4 ± 0.9 |
| GluK2/NETO2 | 5.8 ± 1.0 |
| GluK1/5/NETO2 | 0.99 ± 0.15 |
| GluK2/5/NETO1 | 0.69 ± 0.16 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chałupnik, P.; Szymańska, E. Kainate Receptor Antagonists: Recent Advances and Therapeutic Perspective. Int. J. Mol. Sci. 2023, 24, 1908. https://doi.org/10.3390/ijms24031908
Chałupnik P, Szymańska E. Kainate Receptor Antagonists: Recent Advances and Therapeutic Perspective. International Journal of Molecular Sciences. 2023; 24(3):1908. https://doi.org/10.3390/ijms24031908
Chicago/Turabian StyleChałupnik, Paulina, and Ewa Szymańska. 2023. "Kainate Receptor Antagonists: Recent Advances and Therapeutic Perspective" International Journal of Molecular Sciences 24, no. 3: 1908. https://doi.org/10.3390/ijms24031908
APA StyleChałupnik, P., & Szymańska, E. (2023). Kainate Receptor Antagonists: Recent Advances and Therapeutic Perspective. International Journal of Molecular Sciences, 24(3), 1908. https://doi.org/10.3390/ijms24031908