Oncolytic Effect of Zika Virus in Neuroendocrine Pancreatic Tumors: New Perspectives for Therapeutic Approaches
Abstract
1. Introduction
2. Results
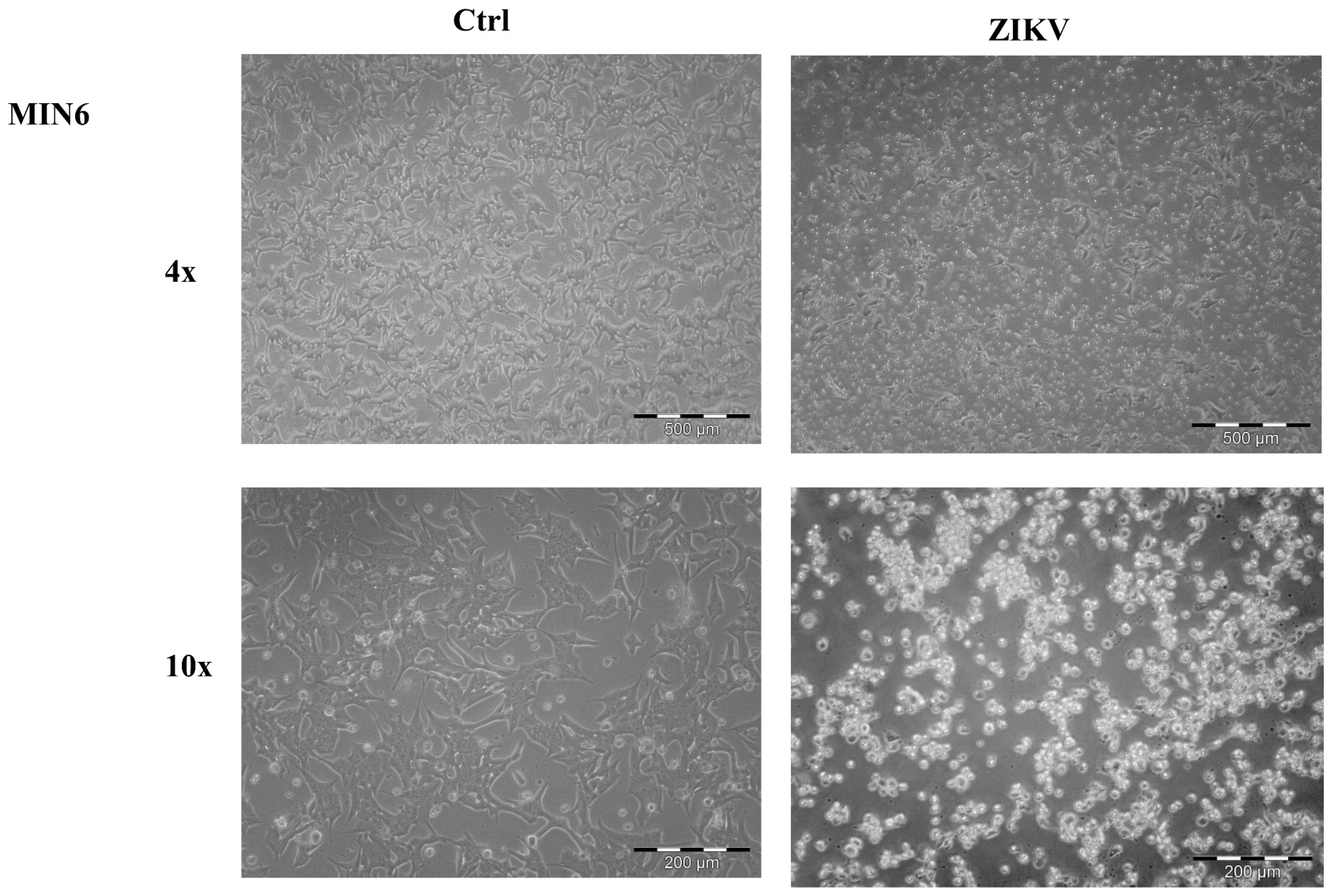
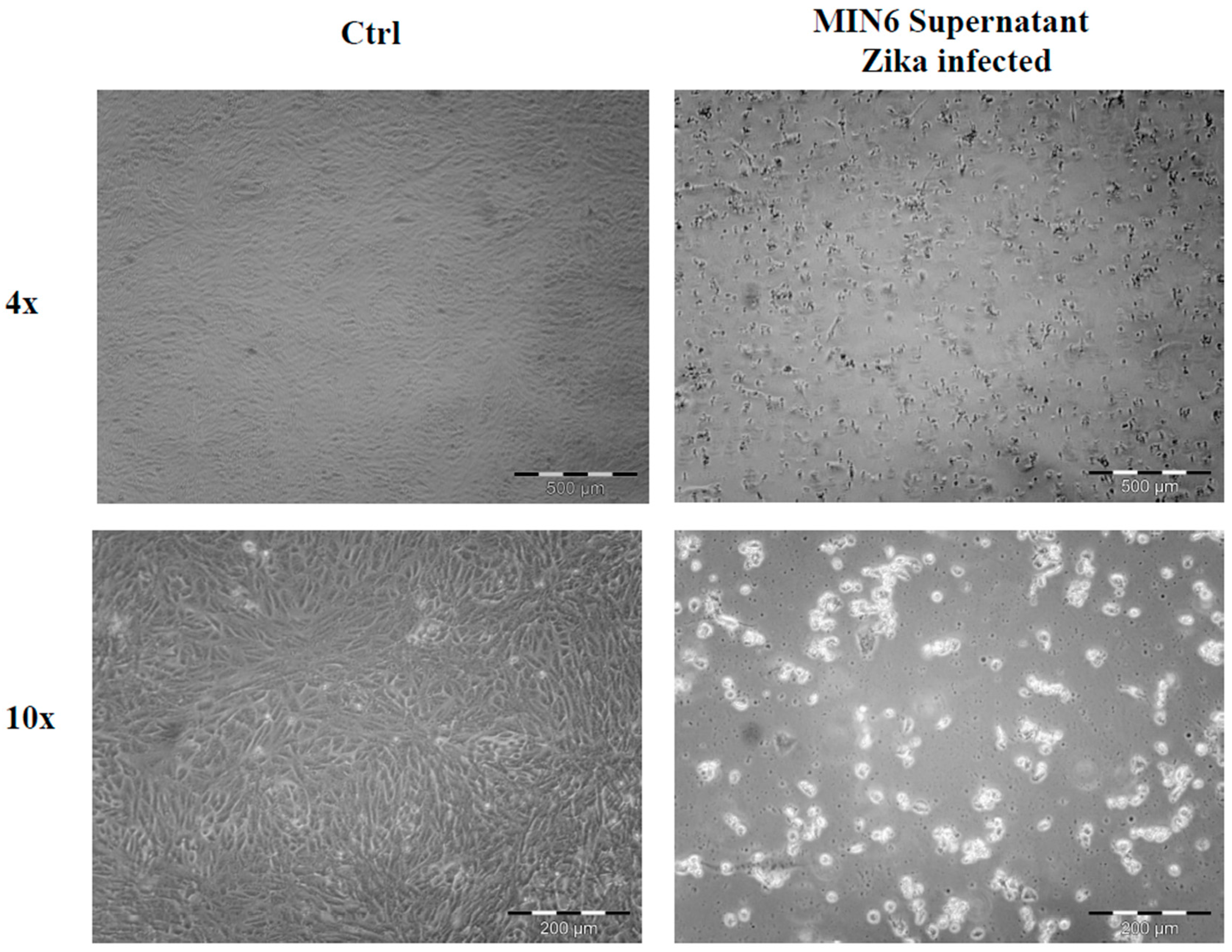
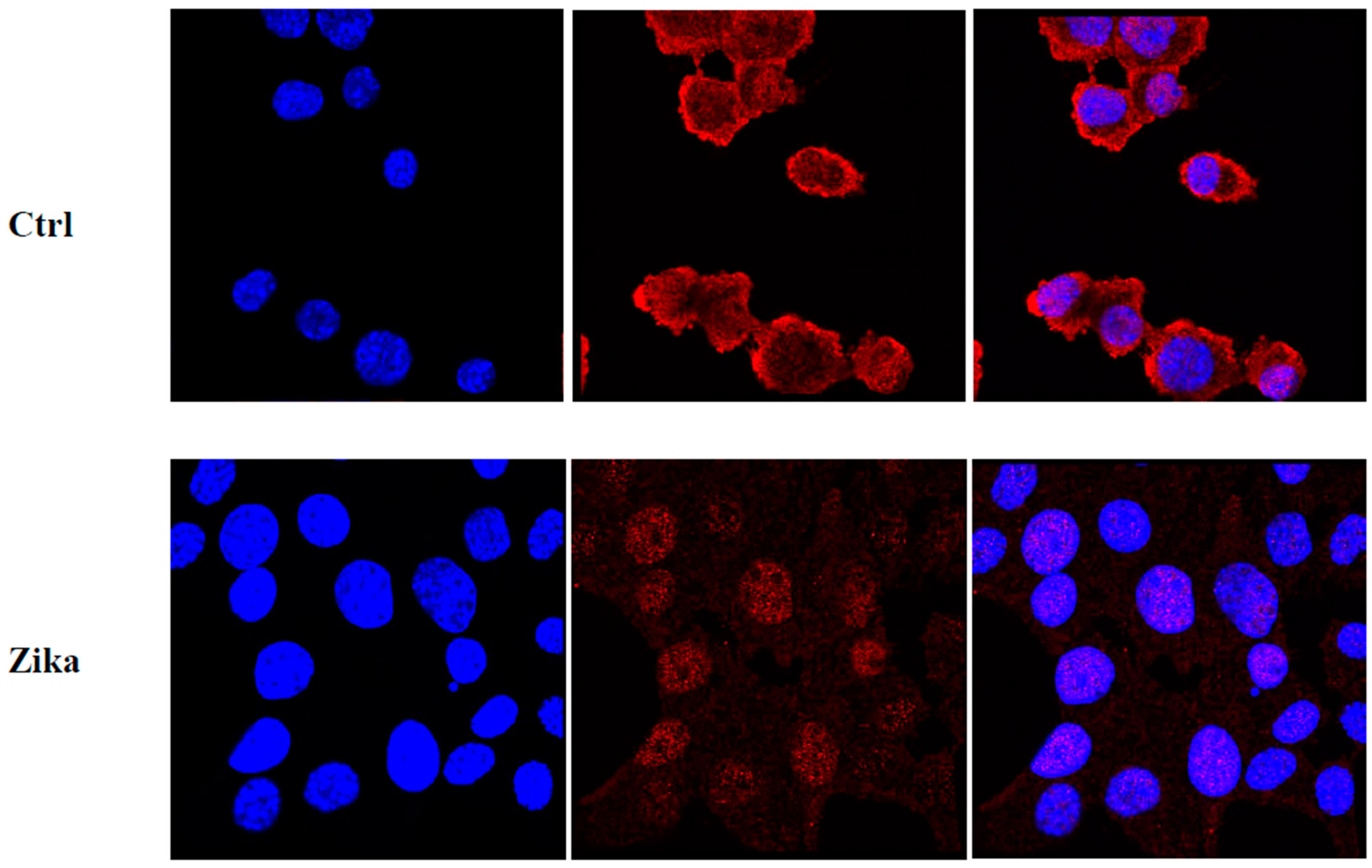
2.1. Cell Morphological Evaluation after ZIKV Infection
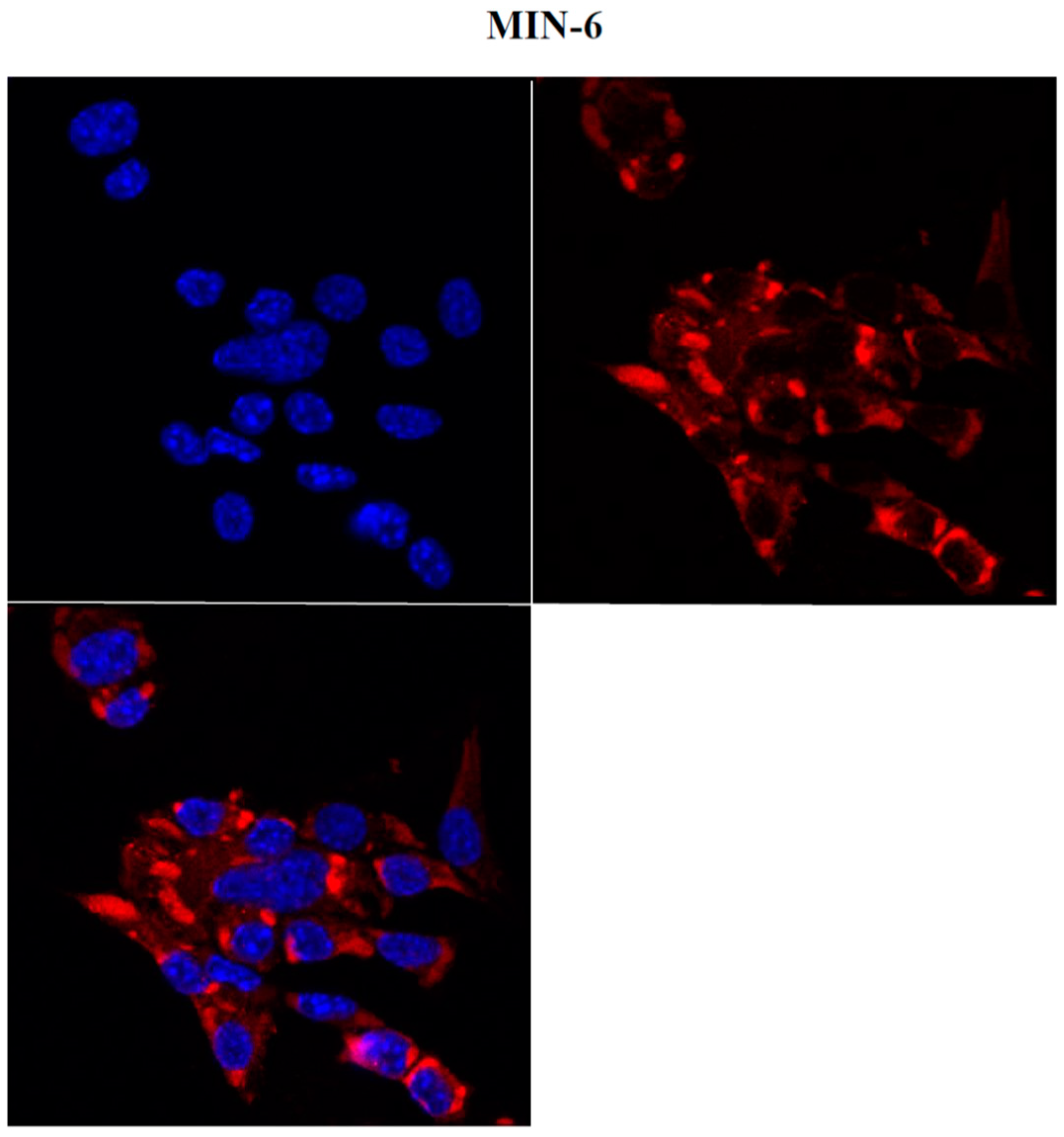
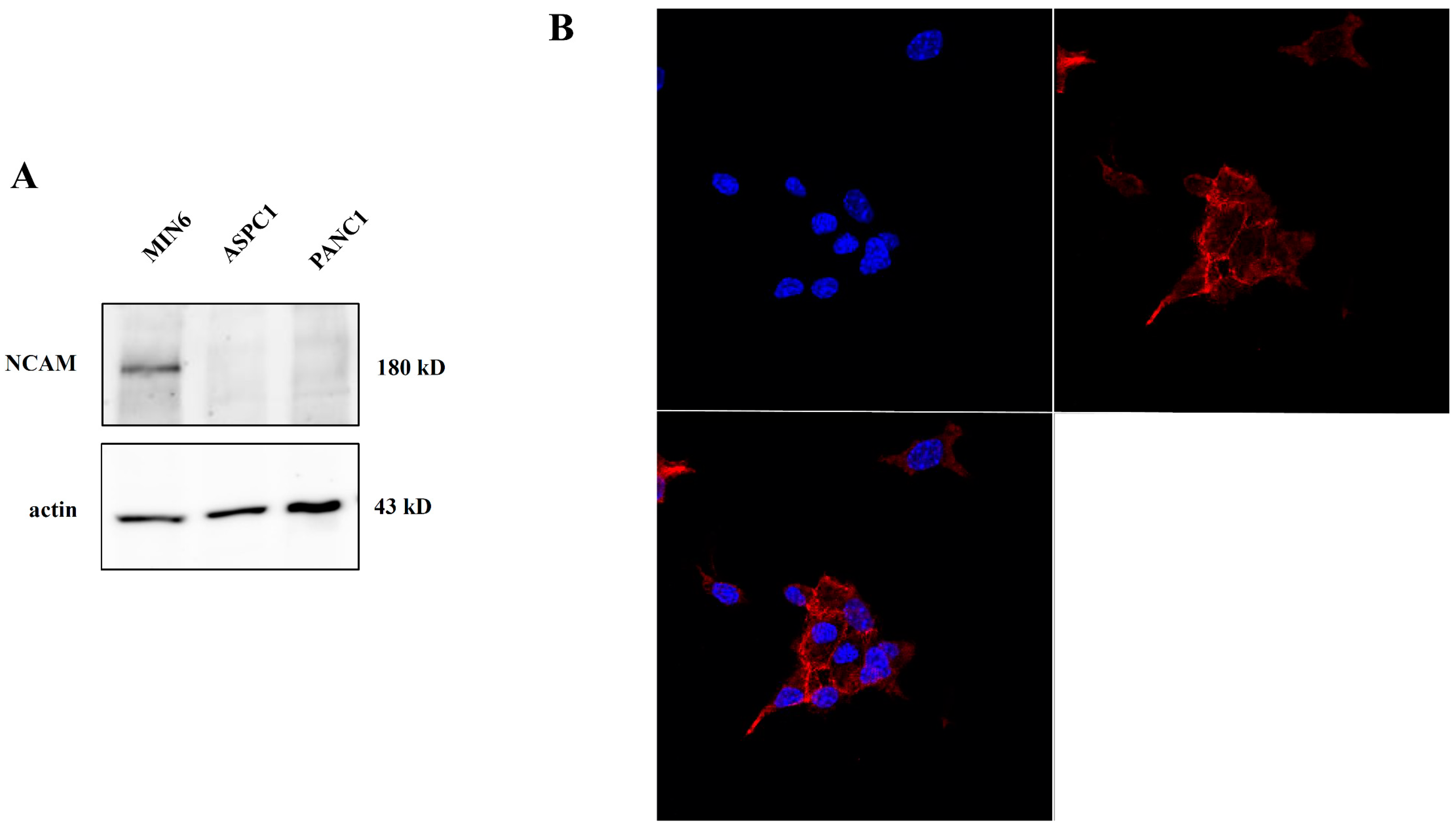
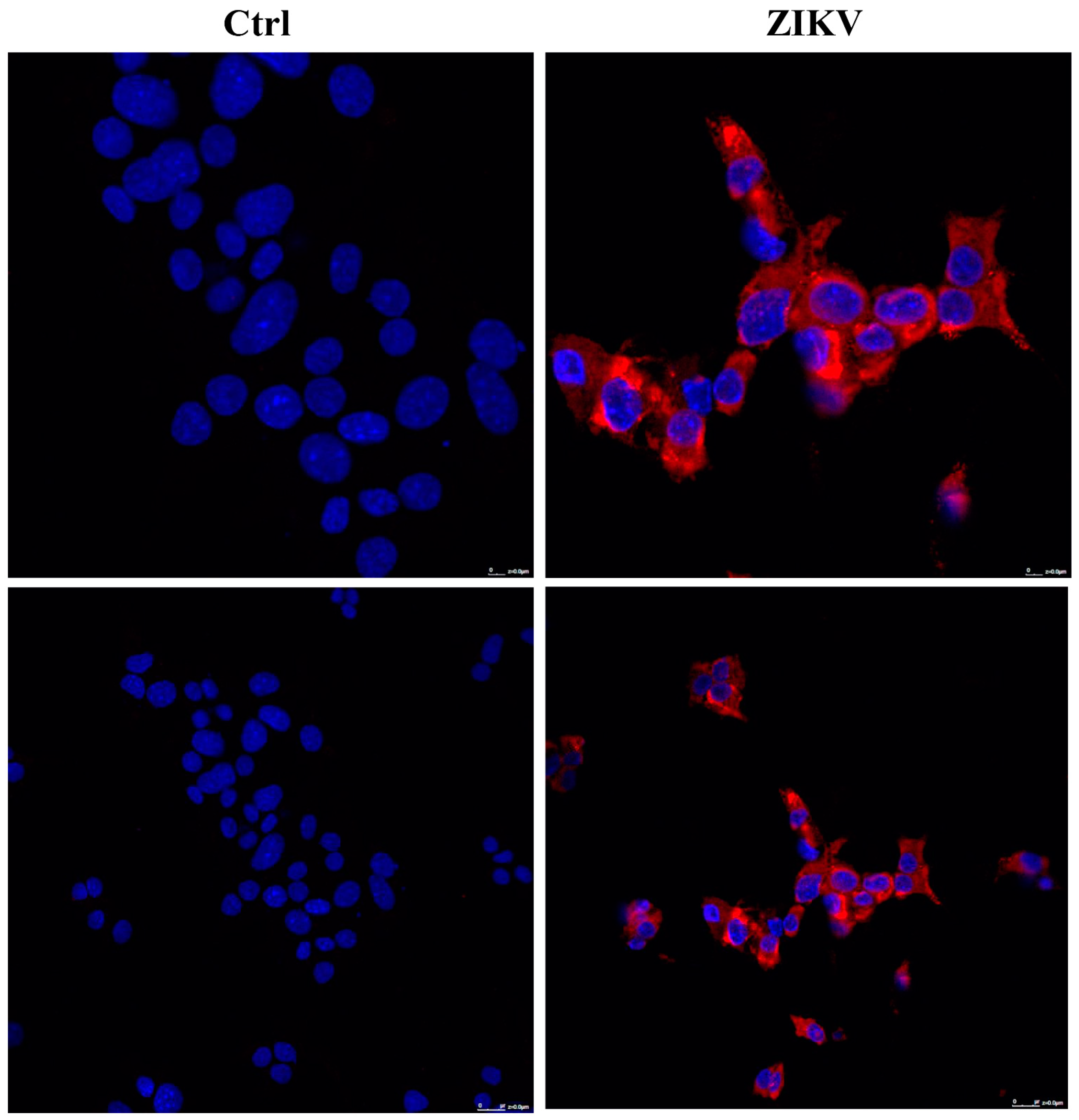
2.2. NCAM Expression in Pancreatic Tumor Cells
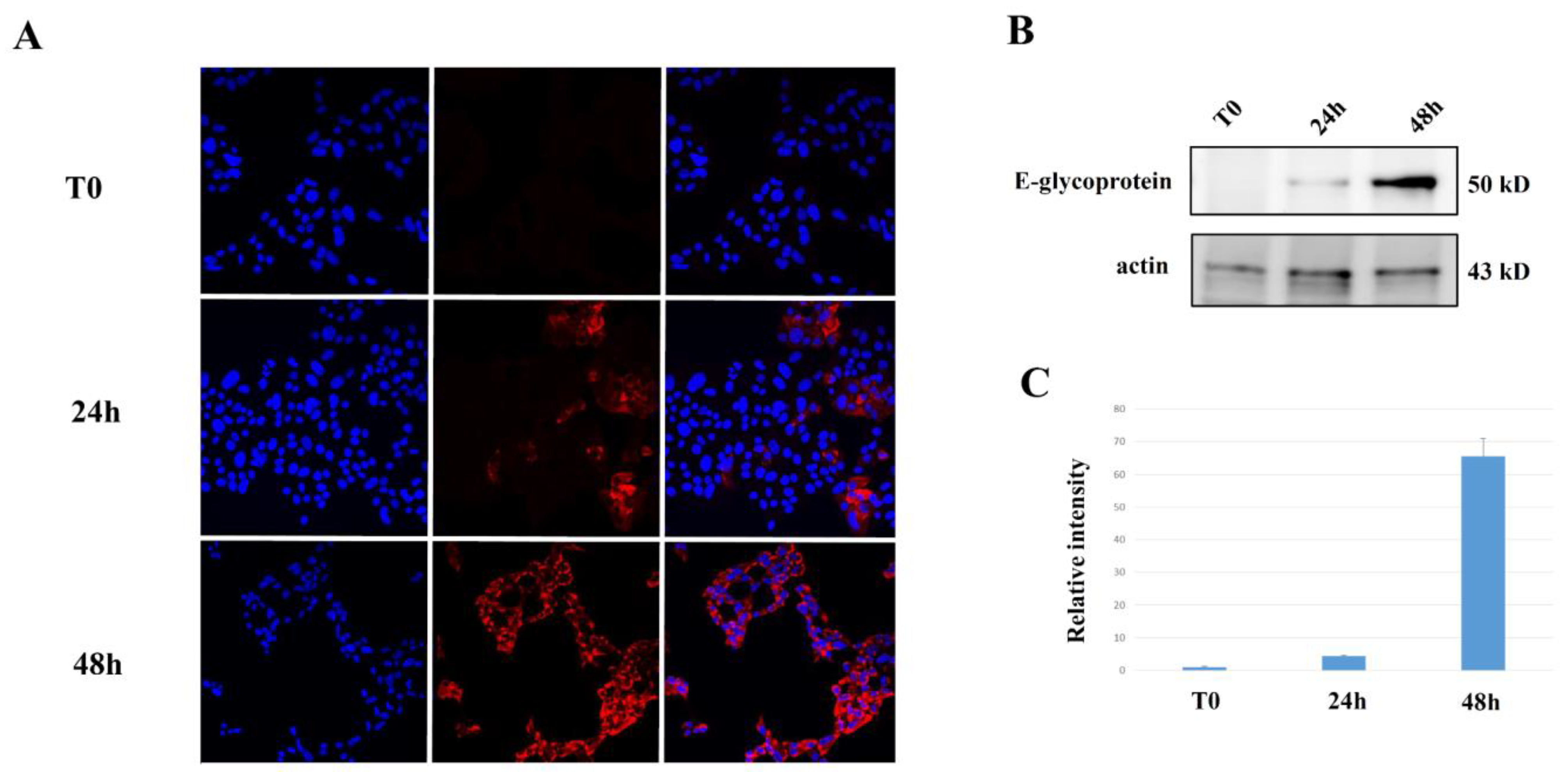
2.3. Evaluation of ZIKV Replication in NET Cells
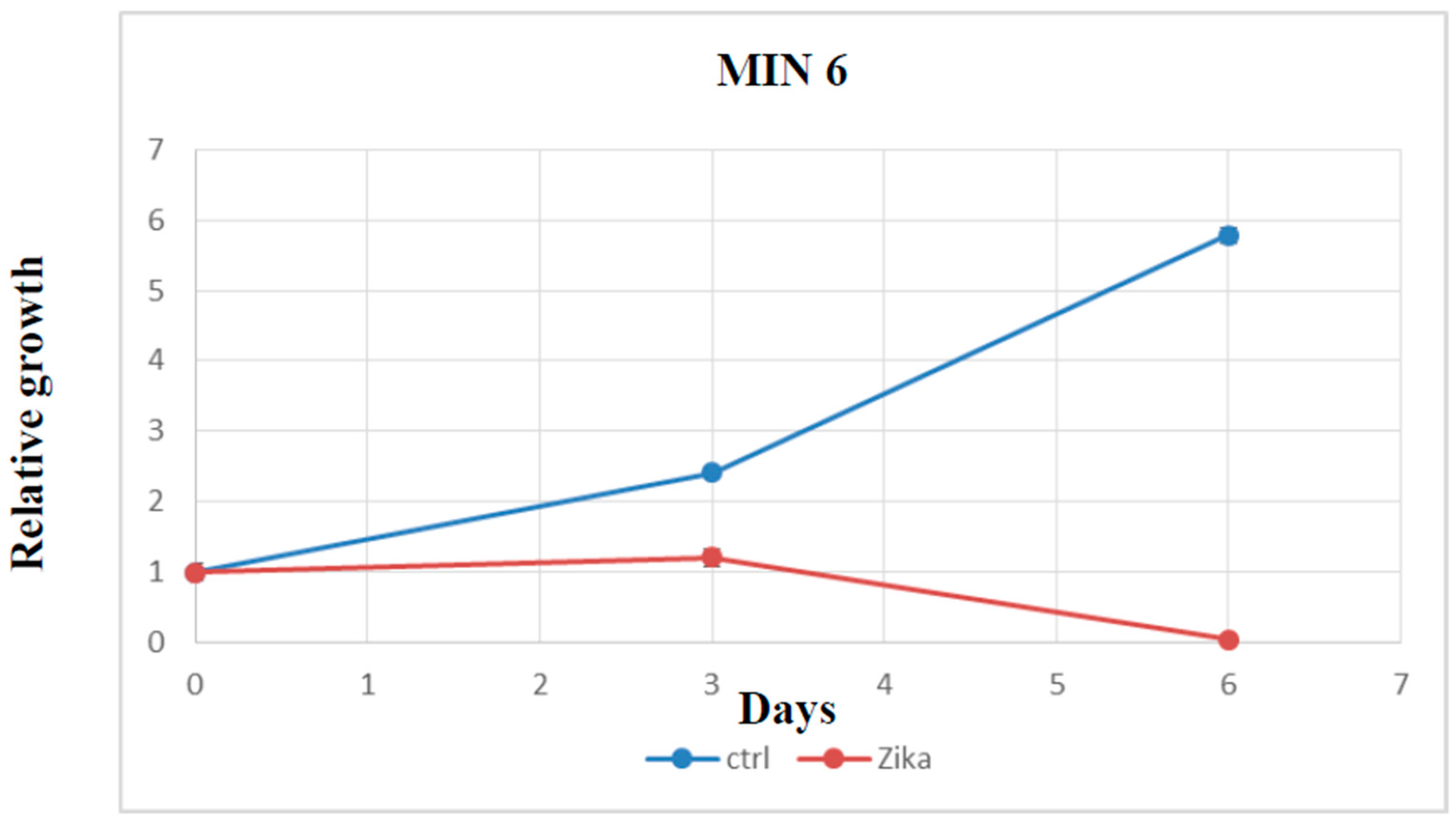
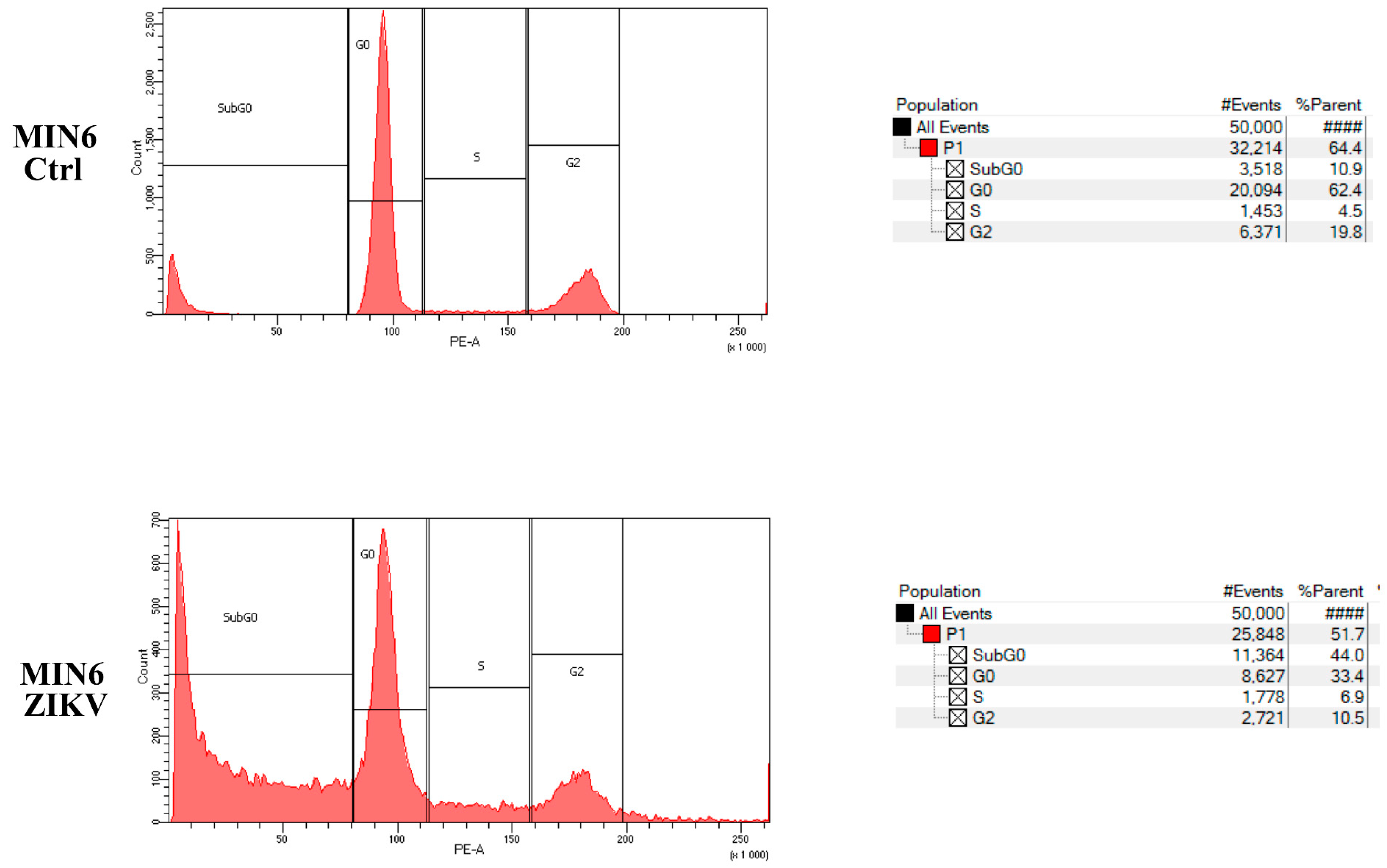
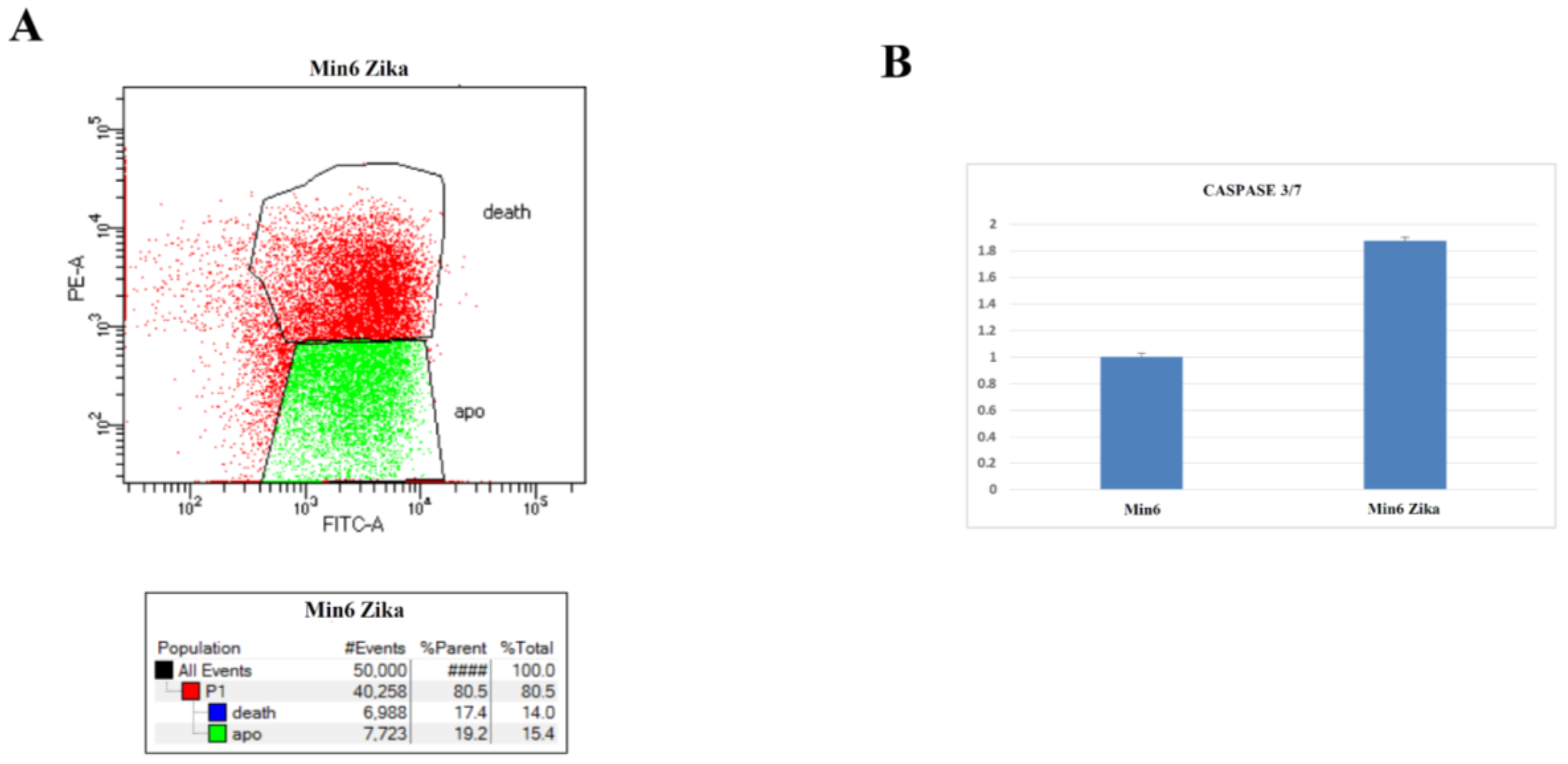
2.4. Evaluation of Cell Growth, Cell Cycle, and Cell Death after ZIKV Infection
3. Discussion
4. Materials and Methods
4.1. Cell Culture, Transfection, and Infection
4.2. RNA Extraction and RT-PCR
4.3. Immunofluorescence
4.4. Immunoblotting
4.5. Propidium Iodide Cell Cycle Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Halbrook, C.J.; Lyssiotis, C.A.; Pasca di Magliano, M.; Maitra, A. Pancreatic cancer: Advances and challenges. Cell 2023, 186, 1729–1754. [Google Scholar] [CrossRef] [PubMed]
- Oronsky, B.; Ma, P.C.; Morgensztern, D.; Carter, C.A. Nothing But NET: A Review of Neuroendocrine Tumors and Carcinomas. Neoplasia 2017, 19, 991–1002. [Google Scholar] [CrossRef] [PubMed]
- Nagtegaal, I.D.; Odze, R.D.; Klimstra, D.; Paradis, V.; Rugge, M.; Schirmacher, P.; Washington, K.M.; Carneiro, F.; Cree, I.A.; The WHO Classification of Tumours Editorial Board. The 2019 WHO classification of tumours of the digestive system. Histopathology 2020, 76, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, J.; He, C.; Sun, T.; Yan, Y.; Che, G.; Li, X.; Sun, H.; Ma, H. Trends in Incidence and Survival of Patients with Pancreatic Neuroendocrine Neoplasm, 1987–2016. J. Oncol. 2021, 2021, 4302675. [Google Scholar] [CrossRef] [PubMed]
- Yao, H.; Hu, G.; Jiang, C.; Fan, M.; Yuan, L.; Shi, H.; Lin, R. Epidemiologic trends and survival of early-onset gastroenteropancreatic neuroendocrine neoplasms. Front. Endocrinol. 2023, 14, 1241724. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, B.; Gustafsson, B.I.; Chan, A.; Svejda, B.; Kidd, M.; Modlin, I.M. The epidemiology of gastroenteropancreatic neuroendocrine tumors. Endocrinol. Metab. Clin. N. Am. 2011, 40, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Guo, X.; Huang, H.; Wang, C.; Yang, F.; Zhang, Y.; Wang, J.; Han, L.; Jin, Z.; Cai, T.; et al. A Switch in Tissue Stem Cell Identity Causes Neuroendocrine Tumors in Drosophila Gut. Cell Rep. 2020, 30, 1724–1734.e4. [Google Scholar] [CrossRef]
- Modlin, I.M.; Oberg, K.; Chung, D.C.; Jensen, R.T.; de Herder, W.W.; Thakker, R.V.; Caplin, M.; Delle Fave, G.; Kaltsas, G.A.; Krenning, E.P.; et al. Gastroenteropancreatic neuroendocrine tumours. Lancet Oncol. 2008, 9, 61–72. [Google Scholar] [CrossRef]
- Devhare, P.; Meyer, K.; Steele, R.; Ray, R.B.; Ray, R. Zika virus infection dysregulates human neural stem cell growth and inhibits differentiation into neuroprogenitor cells. Cell Death Dis. 2017, 8, e3106. [Google Scholar] [CrossRef]
- Iannolo, G.; Sciuto, M.R.; Cuscino, N.; Pallini, R.; Douradinha, B.; Ricci Vitiani, L.; De Maria, R.; Conaldi, P.G. Zika virus infection induces MiR34c expression in glioblastoma stem cells: New perspectives for brain tumor treatments. Cell Death Dis. 2019, 10, 263. [Google Scholar] [CrossRef]
- Zhu, Z.; Gorman, M.J.; McKenzie, L.D.; Chai, J.N.; Hubert, C.G.; Prager, B.C.; Fernandez, E.; Richner, J.M.; Zhang, R.; Shan, C.; et al. Zika virus has oncolytic activity against glioblastoma stem cells. J. Exp. Med. 2017, 214, 2843–2857. [Google Scholar] [CrossRef]
- Francipane, M.G.; Douradinha, B.; Chinnici, C.M.; Russelli, G.; Conaldi, P.G.; Iannolo, G. Zika Virus: A New Therapeutic Candidate for Glioblastoma Treatment. Int. J. Mol. Sci. 2021, 22, 10996. [Google Scholar] [CrossRef]
- Dick, G.W.; Kitchen, S.F.; Haddow, A.J. Zika virus. I. Isolations and serological specificity. Trans. R. Soc. Trop. Med. Hyg. 1952, 46, 509–520. [Google Scholar] [CrossRef] [PubMed]
- Hamel, R.; Dejarnac, O.; Wichit, S.; Ekchariyawat, P.; Neyret, A.; Luplertlop, N.; Perera-Lecoin, M.; Surasombatpattana, P.; Talignani, L.; Thomas, F.; et al. Biology of Zika Virus Infection in Human Skin Cells. J. Virol. 2015, 89, 8880–8896. [Google Scholar] [CrossRef] [PubMed]
- Petersen, E.; Wilson, M.E.; Touch, S.; McCloskey, B.; Mwaba, P.; Bates, M.; Dar, O.; Mattes, F.; Kidd, M.; Ippolito, G.; et al. Rapid Spread of Zika Virus in The Americas--Implications for Public Health Preparedness for Mass Gatherings at the 2016 Brazil Olympic Games. Int. J. Infect. Dis. 2016, 44, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, O.J.M.; da Silva, I.R.F. Guillain-Barre syndrome and Zika virus outbreaks. Curr. Opin. Neurol. 2017, 30, 500–507. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, M.; Zhang, Y.; Chen, J.; Sirohi, D.; Miller, A.; Zhang, Y.; Chen, Z.; Lu, H.; Xu, J.; Kuhn, R.J.; et al. Chemical proteomics tracks virus entry and uncovers NCAM1 as Zika virus receptor. Nat. Commun. 2020, 11, 3896. [Google Scholar] [CrossRef]
- Portier, B.P.; Taglialatela, G. Bcl-2 localized at the nuclear compartment induces apoptosis after transient overexpression. J. Biol. Chem. 2006, 281, 40493–40502. [Google Scholar] [CrossRef]
- Lin, B.; Kolluri, S.K.; Lin, F.; Liu, W.; Han, Y.H.; Cao, X.; Dawson, M.I.; Reed, J.C.; Zhang, X.K. Conversion of Bcl-2 from protector to killer by interaction with nuclear orphan receptor Nur77/TR3. Cell 2004, 116, 527–540. [Google Scholar] [CrossRef]
- Garber, K. China approves world’s first oncolytic virus therapy for cancer treatment. J. Natl. Cancer Inst. 2006, 98, 298–300. [Google Scholar] [CrossRef]
- Gozzoli Podolsky-Gondim, G.; Gomes Da Silva, M.V.; Alves Esposito, D.L.; Carlotti, C.G.; Maris Peria, F.; Neder Serafini, L.; dos Santos, A.C.; Scridelli, C.A.; Ussem, L.; Colli, B.O.; et al. Intracranial Zika Virus Inoculation in a Patient with Recurrent Glioblastoma: A Brief Report of the Clinical and Technical Aspects in a Case of Compassionate Use. Available online: https://www.snola2022.com/evento/snola2022/trabalhosaprovados/naintegra/1104635 (accessed on 11 February 2022).
- Tinnirello, R.; Chinnici, C.M.; Miceli, V.; Busa, R.; Bulati, M.; Gallo, A.; Zito, G.; Conaldi, P.G.; Iannolo, G. Two Sides of The Same Coin: Normal and Tumoral Stem Cells, The Relevance of In Vitro Models and Therapeutic Approaches: The Experience with Zika Virus in Nervous System Development and Glioblastoma Treatment. Int. J. Mol. Sci. 2023, 24, 13550. [Google Scholar] [CrossRef] [PubMed]
- Garcez, P.P.; Loiola, E.C.; Madeiro da Costa, R.; Higa, L.M.; Trindade, P.; Delvecchio, R.; Nascimento, J.M.; Brindeiro, R.; Tanuri, A.; Rehen, S.K. Zika virus impairs growth in human neurospheres and brain organoids. Science 2016, 352, 816–818. [Google Scholar] [CrossRef] [PubMed]
- Garcez, P.P.; Guasti, A.; Ventura, N.; Higa, L.M.; Andreiuolo, F.; de Freitas, G.P.A.; Ribeiro, L.J.; Maia, R.A.; de Lima, S.M.B.; de Souza Azevedo, A.; et al. Case report: Regression of Glioblastoma after flavivirus infection. Front. Med. 2023, 10, 1192070. [Google Scholar] [CrossRef] [PubMed]
- Jakovcevski, I.; Mo, Z.; Zecevic, N. Down-regulation of the axonal polysialic acid-neural cell adhesion molecule expression coincides with the onset of myelination in the human fetal forebrain. Neuroscience 2007, 149, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Franceschini, I.; Angata, K.; Ong, E.; Hong, A.; Doherty, P.; Fukuda, M. Polysialyltransferase ST8Sia II (STX) polysialylates all of the major isoforms of NCAM and facilitates neurite outgrowth. Glycobiology 2001, 11, 231–239. [Google Scholar] [CrossRef][Green Version]
- Jensen, M.; Berthold, F. Targeting the neural cell adhesion molecule in cancer. Cancer Lett. 2007, 258, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Zhang, M.; Shi, L.; Yang, X.; Chen, L.; Cao, N.; Lei, A.; Cao, Y. Neural stemness contributes to cell tumorigenicity. Cell Biosci. 2021, 11, 21. [Google Scholar] [CrossRef] [PubMed]
- Lenharo, M. Scientists deliberately gave women Zika—Here’s why. Nature 2023, 623, 18. [Google Scholar] [CrossRef]
- Santoro, R.; Zanotto, M.; Carbone, C.; Piro, G.; Tortora, G.; Melisi, D. MEKK3 Sustains EMT and Stemness in Pancreatic Cancer by Regulating YAP and TAZ Transcriptional Activity. Anticancer Res. 2018, 38, 1937–1946. [Google Scholar]
- Miyazaki, J.; Araki, K.; Yamato, E.; Ikegami, H.; Asano, T.; Shibasaki, Y.; Oka, Y.; Yamamura, K. Establishment of a pancreatic beta cell line that retains glucose-inducible insulin secretion: Special reference to expression of glucose transporter isoforms. Endocrinology 1990, 127, 126–132. [Google Scholar] [CrossRef]

| Diagnosis | Grading | Mitotic Index (HPF) * | Ki-67 (%) † | Morphology |
|---|---|---|---|---|
| NET | 1 | <2/10 | ≤3 | Well differentiated |
| NET | 2 | 2–20/10 | 3–20 | Well differentiated |
| NET | 3 | >20/10 | >20 | Well differentiated |
| NEC - Small cell - Large cell | 3 | >20/10 | >20 | Poorly differentiated |
| MiNEN | - | - | - | Well—Poor differentiated |
| Tumor-like lesions | - | - | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cocco, M.M.; Carcione, C.; Miceli, V.; Tinnirello, R.; Chinnici, C.M.; Carbone, C.; Zito, G.; Conaldi, P.G.; Iannolo, G. Oncolytic Effect of Zika Virus in Neuroendocrine Pancreatic Tumors: New Perspectives for Therapeutic Approaches. Int. J. Mol. Sci. 2023, 24, 17271. https://doi.org/10.3390/ijms242417271
Cocco MM, Carcione C, Miceli V, Tinnirello R, Chinnici CM, Carbone C, Zito G, Conaldi PG, Iannolo G. Oncolytic Effect of Zika Virus in Neuroendocrine Pancreatic Tumors: New Perspectives for Therapeutic Approaches. International Journal of Molecular Sciences. 2023; 24(24):17271. https://doi.org/10.3390/ijms242417271
Chicago/Turabian StyleCocco, Martina Maria, Claudia Carcione, Vitale Miceli, Rosaria Tinnirello, Cinzia Maria Chinnici, Carmine Carbone, Giovanni Zito, Pier Giulio Conaldi, and Gioacchin Iannolo. 2023. "Oncolytic Effect of Zika Virus in Neuroendocrine Pancreatic Tumors: New Perspectives for Therapeutic Approaches" International Journal of Molecular Sciences 24, no. 24: 17271. https://doi.org/10.3390/ijms242417271
APA StyleCocco, M. M., Carcione, C., Miceli, V., Tinnirello, R., Chinnici, C. M., Carbone, C., Zito, G., Conaldi, P. G., & Iannolo, G. (2023). Oncolytic Effect of Zika Virus in Neuroendocrine Pancreatic Tumors: New Perspectives for Therapeutic Approaches. International Journal of Molecular Sciences, 24(24), 17271. https://doi.org/10.3390/ijms242417271








