Enterococcal Membrane Vesicles as Vaccine Candidates
Abstract
1. Introduction
2. Results
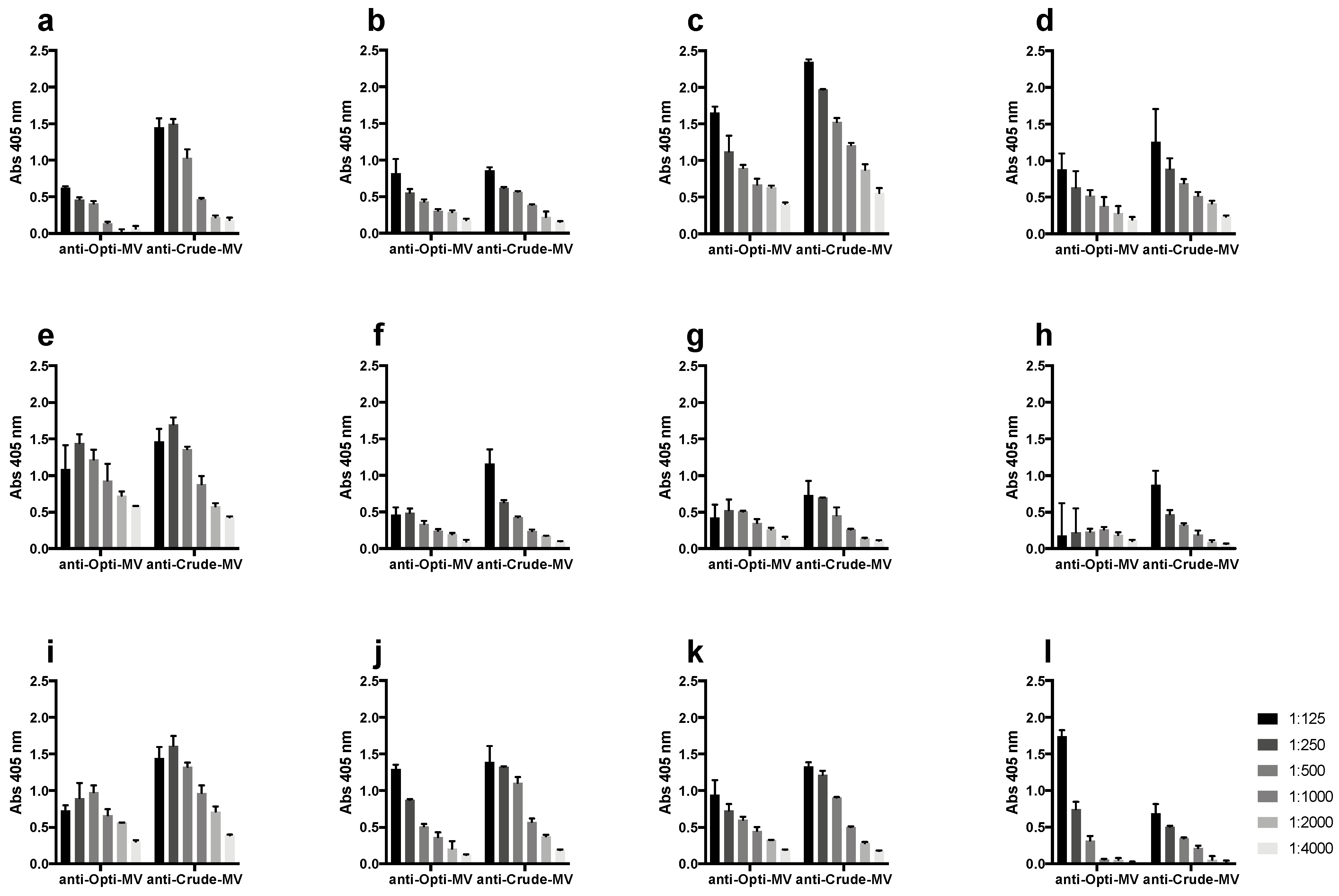
2.1. Immunization with Enterococcal Membrane Vesicles Triggers an Immune Response
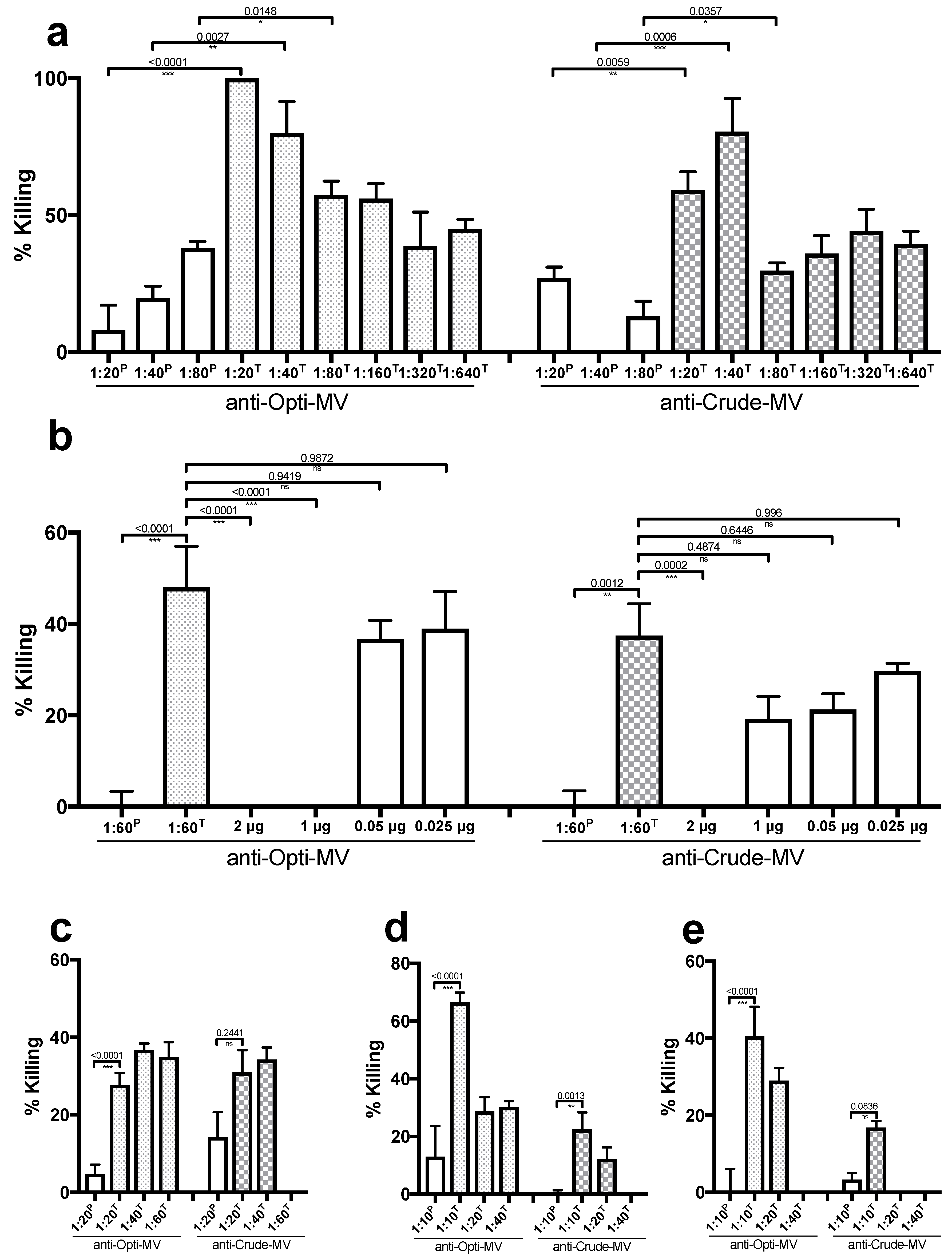
2.2. Anti-Enterococcal Membrane Vesicle Sera Mediate Opsonophagocytic Killing
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains
4.2. Isolation and Characterization of Membrane Vesicles
4.3. Rabbit Immunizations
4.4. Specific Titer Quantification
4.5. IgG and IgM Titer Quantification
4.6. Whole-Bacterial-Cell ELISA
4.7. OPA/OPIA
4.8. Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO Regional Office for Europe; European Centre for Disease Prevention and Control. Antimicrobial Resistance Surveillance in Europe 2022–2020 Data; WHO Regional Office for Europe: Copenhagen, Denmark, 2022. [Google Scholar] [CrossRef]
- Pidot, S.J.; Gao, W.; Buultjens, A.H.; Monk, I.R.; Guerillot, R.; Carter, G.P.; Lee, J.Y.H.; Lam, M.M.C.; Grayson, M.L.; Ballard, S.A.; et al. Increasing Tolerance of Hospital Enterococcus faecium to Handwash Alcohols. Sci. Transl. Med. 2018, 10, eaar6115. [Google Scholar] [CrossRef]
- Arias, C.A.; Murray, B.E. The Rise of the Enterococcus: Beyond Vancomycin Resistance. Nat. Rev. Microbiol. 2012, 10, 266–278. [Google Scholar] [CrossRef]
- Wagenvoort, J.H.T.; De Brauwer, E.I.G.B.; Penders, R.J.R.; van der Linden, C.J.; Willems, R.J.; Top, J.; Bonten, M.J. Environmental Survival of Vancomycin-Sensitive Ampicillin-Resistant Enterococcus faecium (AREfm). Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 1901–1903. [Google Scholar] [CrossRef][Green Version]
- Kalfopoulou, E.; Laverde, D.; Miklic, K.; Romero-Saavedra, F.; Malic, S.; Carboni, F.; Adamo, R.; Lenac Rovis, T.; Jonjic, S.; Huebner, J. Development of Opsonic Mouse Monoclonal Antibodies against Multidrug Resistant Enterococci. Infect. Immun. 2019, 87, e00276-19. [Google Scholar] [CrossRef]
- Kropec, A.; Sava, I.G.; Vonend, C.; Sakinc, T.; Grohmann, E.; Huebner, J. Identification of SagA as a Novel Vaccine Target for the Prevention of Enterococcus faecium Infections. Microbiology 2011, 157, 3429–3434. [Google Scholar] [CrossRef]
- Romero-Saavedra, F.; Laverde, D.; Budin-Verneuil, A.; Muller, C.; Bernay, B.; Benachour, A.; Hartke, A.; Huebner, J. Characterization of Two Metal Binding Lipoproteins as Vaccine Candidates for Enterococcal Infections. PLoS ONE 2015, 10, e0136625. [Google Scholar] [CrossRef][Green Version]
- Romero-Saavedra, F.; Laverde, D.; Wobser, D.; Michaux, C.; Budin-Verneuil, A.; Bernay, B.; Benachour, A.; Hartke, A.; Huebner, J. Identification of Peptidoglycan-Associated Proteins as Vaccine Candidates for Enterococcal Infections. PLoS ONE 2014, 9, e0111880. [Google Scholar] [CrossRef]
- Laverde, D.; Romero-Saavedra, F.; Argunov, D.A.; Enotarpi, J.; Krylov, V.B.; Kalfopoulou, E.; Martini, C.; Torelli, R.; Van Der Marel, G.A.; Sanguinetti, M.; et al. Synthetic Oligomers Mimicking Capsular Polysaccharide Diheteroglycan Are Potential Vaccine Candidates against Encapsulated Enterococcal Infections. ACS Infect. Dis. 2020, 6, 1816–1826. [Google Scholar] [CrossRef]
- Theilacker, C.; Kaczyński, Z.; Kropec, A.; Sava, I.; Ye, L.; Bychowska, A.; Holst, O.; Huebner, J. Serodiversity of Opsonic Antibodies against Enterococcus faecalis—Glycans of the Cell Wall Revisited. PLoS ONE 2011, 6, e17839. [Google Scholar] [CrossRef][Green Version]
- Wang, Y.; Huebner, J.; Tzianabos, A.O.; Martirosian, G.; Kasper, D.L.; Pier, G.B. Structure of an Antigenic Teichoic Acid Shared by Clinical Isolates of Enterococcus faecalis and Vancomycin-Resistant Enterococcus faecium. Carbohydr. Res. 1999, 316, 155–160. [Google Scholar] [CrossRef]
- Wagner, T.; Joshi, B.; Janice, J.; Askarian, F.; Škalko-Basnet, N.; Hagestad, O.C.; Mekhlif, A.; Wai, S.N.; Hegstad, K.; Johannessen, M. Enterococcus faecium Produces Membrane Vesicles Containing Virulence Factors and Antimicrobial Resistance Related Proteins. J. Proteom. 2018, 187, 28–38. [Google Scholar] [CrossRef]
- Kaparakis-Liaskos, M.; Ferrero, R.L. Immune Modulation by Bacterial Outer Membrane Vesicles. Nat. Rev. Immunol. 2015, 15, 375–387. [Google Scholar] [CrossRef]
- Van der Pol, L.; Stork, M.; van der Ley, P. Outer Membrane Vesicles as Platform Vaccine Technology. Biotechnol. J. 2015, 10, 1689–1706. [Google Scholar] [CrossRef]
- Vesikari, T.; Esposito, S.; Prymula, R.; Ypma, E.; Kohl, I.; Toneatto, D.; Dull, P.; Kimura, A.; EU Meningococcal B Infant Vaccine Study group. Immunogenicity and Safety of an Investigational Multicomponent, Recombinant, Meningococcal Serogroup B Vaccine (4CMenB) Administered Concomitantly with Routine Infant and Child Vaccinations: Results of Two Randomised Trials. Lancet 2013, 381, 825–835. [Google Scholar] [CrossRef]
- Bjune, G.; Høiby, E.A.; Grønnesby, J.K.; Arnesen, O.; Fredriksen, J.H.; Halstensen, A.; Holten, E.; Lindbak, A.K.; Nøkleby, H.; Rosenqvist, E. Effect of Outer Membrane Vesicle Vaccine against Group B Meningococcal Disease in Norway. Lancet 1991, 338, 1093–1096. [Google Scholar] [CrossRef]
- Choi, C.-W.; Park, E.C.; Yun, S.H.; Lee, S.-Y.; Kim, S.I.; Kim, G.-H. Potential Usefulness of Streptococcus pneumoniae Extracellular Membrane Vesicles as Antibacterial Vaccines. Available online: https://www.hindawi.com/journals/jir/2017/7931982/ (accessed on 11 December 2017).
- Choi, S.J.; Kim, M.-H.; Jeon, J.; Kim, O.Y.; Choi, Y.; Seo, J.; Hong, S.-W.; Lee, W.-H.; Jeon, S.G.; Gho, Y.S.; et al. Active Immunization with Extracellular Vesicles Derived from Staphylococcus aureus Effectively Protects against Staphylococcal Lung Infections, Mainly via Th1 Cell-Mediated Immunity. PLoS ONE 2015, 10, e0136021. [Google Scholar] [CrossRef]
- Askarian, F.-; Lapek, J.J.; Dongre, M.; Tsai, C.-M.; Kumaraswamy, M.; Kousha, A.; Valderrama, J.A.; Ludviksen, J.A.; Cavanagh, P.; Uchiyama, S.; et al. Staphylococcus aureus Membrane-Derived Vesicles Promote Bacterial Virulence and Confer Protective Immunity in Murine Infection Models. Front. Microbiol. 2018, 9, 262. [Google Scholar] [CrossRef]
- Cossarizza, A.; Chang, H.-D.; Radbruch, A.; Akdis, M.; Andrä, I.; Annunziato, F.; Bacher, P.; Barnaba, V.; Battistini, L.; Bauer, W.M.; et al. Guidelines for the Use of Flow Cytometry and Cell Sorting in Immunological Studies. Eur. J. Immunol. 2017, 47, 1584–1797. [Google Scholar] [CrossRef]
- Romero-Saavedra, F.; Laverde, D.; Kalfopoulou, E.; Martini, C.; Torelli, R.; Martinez-Matamoros, D.; Sanguinetti, M.; Huebner, J. Conjugation of Different Immunogenic Enterococcal Vaccine Target Antigens Leads to Extended Strain Coverage. J. Infect. Dis. 2019, 220, 1589–1598. [Google Scholar] [CrossRef]
- Bender, J.K.; Cattoir, V.; Hegstad, K.; Sadowy, E.; Coque, T.M.; Westh, H.; Hammerum, A.M.; Schaffer, K.; Burns, K.; Murchan, S.; et al. Update on Prevalence and Mechanisms of Resistance to Linezolid, Tigecycline and Daptomycin in Enterococci in Europe: Towards a Common Nomenclature. Drug Resist. Updat. 2018, 40, 25–39. [Google Scholar] [CrossRef]
- Poudel, A.N.; Zhu, S.; Cooper, N.; Little, P.; Tarrant, C.; Hickman, M.; Yao, G. The Economic Burden of Antibiotic Resistance: A Systematic Review and Meta-Analysis. PLoS ONE 2023, 18, e0285170. [Google Scholar] [CrossRef] [PubMed]
- DiazGranados, C.A.; Zimmer, S.M.; Klein, M.; Jernigan, J.A. Comparison of Mortality Associated with Vancomycin-Resistant and Vancomycin-Susceptible Enterococcal Bloodstream Infections: A Meta-Analysis. Clin. Infect. Dis. 2005, 41, 327–333. [Google Scholar] [CrossRef]
- Suchomel, M.; Lenhardt, A.; Kampf, G.; Grisold, A. Enterococcus hirae, Enterococcus faecium and Enterococcus faecalis Show Different Sensitivities to Typical Biocidal Agents Used for Disinfection. J. Hosp. Infect. 2019, 103, 435–440. [Google Scholar] [CrossRef]
- Geraldes, C.; Tavares, L.; Gil, S.; Oliveira, M. Enterococcus Virulence and Resistant Traits Associated with Its Permanence in the Hospital Environment. Antibiotics 2022, 11, 857. [Google Scholar] [CrossRef]
- Kalfopoulou, E.; Huebner, J. Advances and Prospects in Vaccine Development against Enterococci. Cells 2020, 9, 2397. [Google Scholar] [CrossRef] [PubMed]
- Lieberman, L.A. Outer Membrane Vesicles: A Bacterial-Derived Vaccination System. Front. Microbiol. 2022, 13, 1029146. [Google Scholar] [CrossRef] [PubMed]
- Micoli, F.; MacLennan, C.A. Outer Membrane Vesicle Vaccines. Semin. Immunol. 2020, 50, 101433. [Google Scholar] [CrossRef] [PubMed]
- Bottero, D.; Gaillard, M.E.; Errea, A.; Moreno, G.; Zurita, E.; Pianciola, L.; Rumbo, M.; Hozbor, D. Outer Membrane Vesicles Derived from Bordetella parapertussis as an Acellular Vaccine against Bordetella parapertussis and Bordetella pertussis Infection. Vaccine 2013, 31, 5262–5268. [Google Scholar] [CrossRef] [PubMed]
- Schild, S.; Nelson, E.J.; Bishop, A.L.; Camilli, A. Characterization of Vibrio cholerae Outer Membrane Vesicles as a Candidate Vaccine for Cholera. Infect. Immun. 2009, 77, 472–484. [Google Scholar] [CrossRef]
- Leitner, D.R.; Lichtenegger, S.; Temel, P.; Zingl, F.G.; Ratzberger, D.; Roier, S.; Schild-Prüfert, K.; Feichter, S.; Reidl, J.; Schild, S. A Combined Vaccine Approach against Vibrio cholerae and ETEC Based on Outer Membrane Vesicles. Front. Microbiol. 2015, 6, 823. [Google Scholar] [CrossRef][Green Version]
- McCaig, W.D.; Loving, C.L.; Hughes, H.R.; Brockmeier, S.L. Characterization and Vaccine Potential of Outer Membrane Vesicles Produced by Haemophilus parasuis. PLoS ONE 2016, 11, e0149132. [Google Scholar] [CrossRef]
- Zare Banadkoki, E.; Rasooli, I.; Ghazanfari, T.; Siadat, S.D.; Shafiee Ardestani, M.; Owlia, P. Pseudomonas aeruginosa PAO1 Outer Membrane Vesicles-Diphtheria Toxoid Conjugate as a Vaccine Candidate in a Murine Burn Model. Sci. Rep. 2022, 12, 22324. [Google Scholar] [CrossRef]
- McConnell, M.J.; Rumbo, C.; Bou, G.; Pachón, J. Outer Membrane Vesicles as an Acellular Vaccine against Acinetobacter baumannii. Vaccine 2011, 29, 5705–5710. [Google Scholar] [CrossRef]
- Gasperini, G.; Alfini, R.; Arato, V.; Mancini, F.; Aruta, M.G.; Kanvatirth, P.; Pickard, D.; Necchi, F.; Saul, A.; Rossi, O.; et al. Salmonella Paratyphi A Outer Membrane Vesicles Displaying Vi Polysaccharide as a Multivalent Vaccine against Enteric Fever. Infect. Immun. 2021, 89, e00699-20. [Google Scholar] [CrossRef]
- Behrens, F.; Funk-Hilsdorf, T.C.; Kuebler, W.M.; Simmons, S. Bacterial Membrane Vesicles in Pneumonia: From Mediators of Virulence to Innovative Vaccine Candidates. Int. J. Mol. Sci. 2021, 22, 3858. [Google Scholar] [CrossRef]
- König, E.; Gagliardi, A.; Riedmiller, I.; Andretta, C.; Tomasi, M.; Irene, C.; Frattini, L.; Zanella, I.; Berti, F.; Grandi, A.; et al. Multi-Antigen Outer Membrane Vesicle Engineering to Develop Polyvalent Vaccines: The Staphylococcus aureus Case. Front. Immunol. 2021, 12, 752168. [Google Scholar] [CrossRef]
- Afonina, I.; Tien, B.; Nair, Z.; Matysik, A.; Lam, L.N.; Veleba, M.; Jie, A.K.J.; Rashid, R.; Cazenave-Gassiot, A.; Wenk, M.; et al. The Composition and Function of Enterococcus faecalis Membrane Vesicles. Microlife 2021, 2, uqab002. [Google Scholar] [CrossRef]
- Teng, F.; Kawalec, M.; Weinstock, G.M.; Hryniewicz, W.; Murray, B.E. An Enterococcus faecium Secreted Antigen, SagA, Exhibits Broad-Spectrum Binding to Extracellular Matrix Proteins and Appears Essential for E. faecium Growth. Infect. Immun. 2003, 71, 5033–5041. [Google Scholar] [CrossRef]
- Micoli, F.; Adamo, R.; Costantino, P. Protein Carriers for Glycoconjugate Vaccines: History, Selection Criteria, Characterization and New Trends. Molecules 2018, 23, 1451. [Google Scholar] [CrossRef]
- Werner, G.; Neumann, B.; Weber, R.E.; Kresken, M.; Wendt, C.; Bender, J.K.; Becker, K.; Borgmann, S.; Diefenbach, A.; Hamprecht, A.; et al. Thirty Years of VRE in Germany—“Expect the Unexpected”: The View from the National Reference Centre for Staphylococci and Enterococci. Drug Resist. Updates 2020, 53, 100732. [Google Scholar] [CrossRef]
- de Been, M.; van Schaik, W.; Cheng, L.; Corander, J.; Willems, R.J. Recent Recombination Events in the Core Genome Are Associated with Adaptive Evolution in Enterococcus faecium. Genome Biol. Evol. 2013, 5, 1524–1535. [Google Scholar] [CrossRef]
- Huebner, J.; Wang, Y.; Krueger, W.A.; Madoff, L.C.; Martirosian, G.; Boisot, S.; Goldmann, D.A.; Kasper, D.L.; Tzianabos, A.O.; Pier, G.B. Isolation and Chemical Characterization of a Capsular Polysaccharide Antigen Shared by Clinical Isolates of Enterococcus faecalis and Vancomycin-Resistant Enterococcus faecium. Infect. Immun. 1999, 67, 1213–1219. [Google Scholar] [CrossRef]
- Palmer, K.L.; Carniol, K.; Manson, J.M.; Heiman, D.; Shea, T.; Young, S.; Zeng, Q.; Gevers, D.; Feldgarden, M.; Birren, B.; et al. High-Quality Draft Genome Sequences of 28 Enterococcus Sp. Isolates. J. Bacteriol. 2010, 192, 2469–2470. [Google Scholar] [CrossRef]
- Rossmann, F.S.; Laverde, D.; Kropec, A.; Romero-Saavedra, F.; Meyer-Buehn, M.; Huebner, J. Isolation of Highly Active Monoclonal Antibodies against Multiresistant Gram-Positive Bacteria. PLoS ONE 2015, 10, e0118405. [Google Scholar] [CrossRef]
| IgM (mg/mL) | IgG (mg/mL) | ||||
|---|---|---|---|---|---|
| Day | Bleed | Anti-OptiPrep | Anti-Crude | Anti-OptiPrep | Anti-Crude |
| 0 | 1st Pre | 0.70 | 0.40 | 8.39 | 7.15 |
| 7 | 2nd Pre | 0.46 | 0.57 | 3.85 | 5.32 |
| 42 | Test | 0.90 | 0.37 | 7.42 | 5.53 |
| 56 | Terminal | 0.65 | 0.32 | 9.4 | 10.91 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wagner, T.M.; Romero-Saavedra, F.; Laverde, D.; Johannessen, M.; Hübner, J.; Hegstad, K. Enterococcal Membrane Vesicles as Vaccine Candidates. Int. J. Mol. Sci. 2023, 24, 16051. https://doi.org/10.3390/ijms242216051
Wagner TM, Romero-Saavedra F, Laverde D, Johannessen M, Hübner J, Hegstad K. Enterococcal Membrane Vesicles as Vaccine Candidates. International Journal of Molecular Sciences. 2023; 24(22):16051. https://doi.org/10.3390/ijms242216051
Chicago/Turabian StyleWagner, Theresa Maria, Felipe Romero-Saavedra, Diana Laverde, Mona Johannessen, Johannes Hübner, and Kristin Hegstad. 2023. "Enterococcal Membrane Vesicles as Vaccine Candidates" International Journal of Molecular Sciences 24, no. 22: 16051. https://doi.org/10.3390/ijms242216051
APA StyleWagner, T. M., Romero-Saavedra, F., Laverde, D., Johannessen, M., Hübner, J., & Hegstad, K. (2023). Enterococcal Membrane Vesicles as Vaccine Candidates. International Journal of Molecular Sciences, 24(22), 16051. https://doi.org/10.3390/ijms242216051







