The Effect of a Six-Week Nordic Walking Training Cycle on Oxidative Damage of Macromolecules and Iron Metabolism in Older Patients with Multiple Myeloma in Remission—Randomized Clinical Trial
Abstract
1. Introduction
2. Results
2.1. Study Group Characteristics
2.2. Parameters of Blood Cells Count
2.3. Parameters of Iron Metabolism in Serum
2.4. Macromolecules Oxidative Damage Parameters
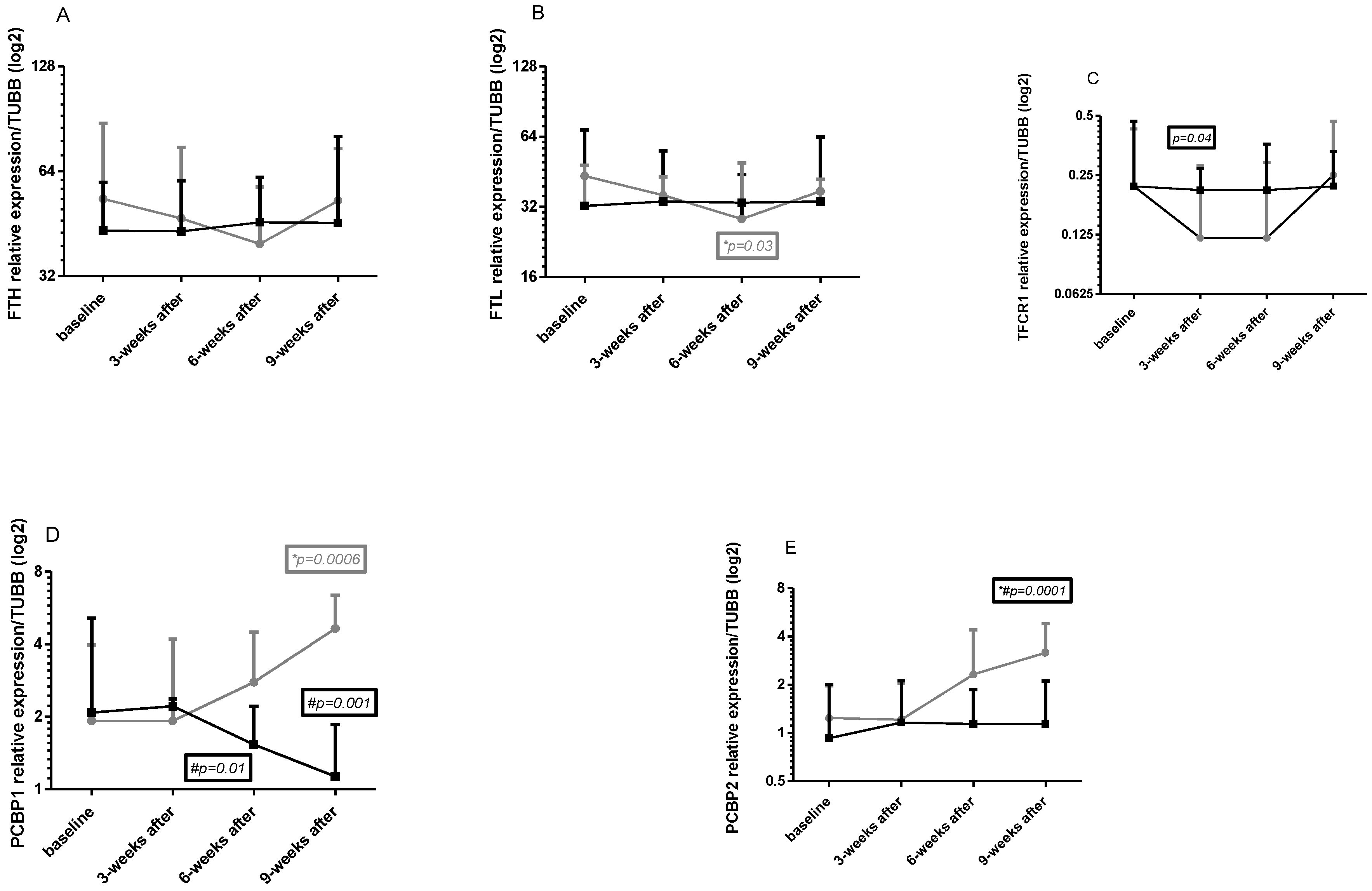
2.5. Gene Expression in Leukocytes
3. Discussion
3.1. Iron Metabolism in Multiple Myeloma
3.2. Iron Metabolism and Nordic Walking
3.3. Oxidative Stress in Multiple Myeloma
3.4. Oxidative Stress and Nordic Walking
4. Materials and Methods
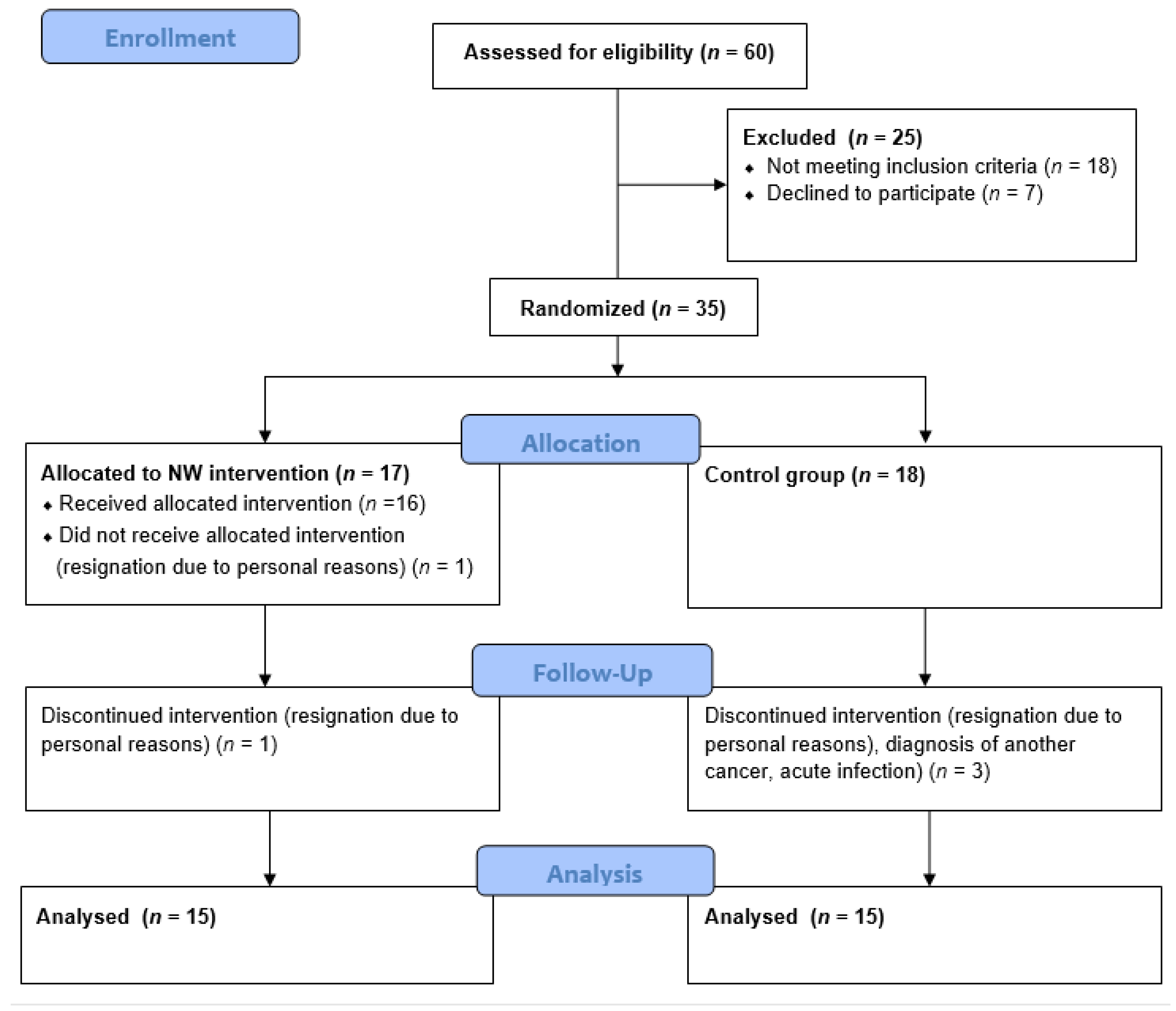
4.1. Study Group
4.2. Study Protocol
4.3. Methods
4.3.1. Venous Blood Collection
4.3.2. Gene Expression Analysis
- FTH1
- FTL
- TFRC
- PCBP1
- PCBP2
- TUBB—tubulin beta class 1 (used as the reference gene)
4.3.3. Investigation of Serum Iron Metabolism Parameters
4.3.4. Investigation of Oxidative Stress Parameters
4.4. Nordic Walking Trainings
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wojciechowska, U.; Czaderny, K.; Ciuba, A.; Didkowska, J. Nowotwory Złośliwe w Polsce w 2016 Roku; Krajowy Rejestr Nowotworó: Warsaw, Poland, 2018. [Google Scholar]
- Giannopoulos, K.; Jamroziak, K.; Usnarska-Zubkiewicz, L.; Dytfeld, D.; Jurczyszyn, A.; Walewski, J.; Lech-Marańda, E.; Walter-Croneck, A.; Pieńkowska-Grela, B.; Wróbel, T.; et al. Zalecenia Polskiej Grupy Szpiczakowej Dotyczące Rozpoznawania i Leczenia Szpiczaka Plazmocytowego Oraz Innych Dyskrazji Plazmocytowych Na Rok 2021; Polska Grupa Szpiczakowa: Krakow, Poland, 2020. [Google Scholar]
- Vanderwall, K.; Daniels-Wells, T.R.; Penichet, M.; Lichtenstein, A. Iron in Multiple Myeloma. Crit. Rev. Oncog. 2013, 18, 449–461. [Google Scholar] [CrossRef]
- Rajkumar, S.V.; Kumar, S. Multiple Myeloma: Diagnosis and Treatment. Mayo Clin. Proc. 2016, 91, 101–119. [Google Scholar] [CrossRef] [PubMed]
- Grzybkowska, A.; Anczykowska, K.; Ratkowski, W.; Aschenbrenner, P.; Antosiewicz, J.; Bonisławska, I.; Żychowska, M. Changes in Serum Iron and Leukocyte MRNA Levels of Genes Involved in Iron Metabolism in Amateur Marathon Runners—Effect of the Running Pace. Genes 2019, 10, 460. [Google Scholar] [CrossRef] [PubMed]
- Thirupathi, A.; Wang, M.; Lin, J.K.; Fekete, G.; István, B.; Baker, J.S.; Gu, Y. Effect of Different Exercise Modalities on Oxidative Stress: A Systematic Review. BioMed Res. Int. 2021, 2021, 1947928. [Google Scholar] [CrossRef] [PubMed]
- Finaud, J.; Lac, G.; Filaire, E. Oxidative Stres Relationship with Exercise and Training. Sport. Med. 2006, 36, 327–358. [Google Scholar] [CrossRef]
- Borkowska, A.; Tomczyk, M.; Żychowska, M.; Pilis, W.; Zych, M.; Antosiewicz, J. Effect of 8-Day Fasting on Leukocytes Expression of Genes and Proteins Involved in Iron Metabolism in Healthy Men. Int. J. Mol. Sci. 2021, 22, 3248. [Google Scholar] [CrossRef]
- Rahal, A.; Kumar, A.; Singh, V.; Yadav, B.; Tiwari, R.; Chakraborty, S.; Dhama, K. Oxidative Stress, Prooxidants, and Antioxidants: The Interplay. Biomed Res. Int. 2014, 2014, 761264. [Google Scholar] [CrossRef]
- Ścibior-Bentkowska, D.; Czeczot, H. Cancer Cells and Oxidative Stress. Postepy Hig. Med. Dosw. 2009, 63, 58–72. [Google Scholar]
- Hayes, J.D.; Dinkova-Kostova, A.T.; Tew, K.D. Oxidative Stress in Cancer. Cancer Cell 2020, 38, 167. [Google Scholar] [CrossRef]
- Neganova, M.; Liu, J.; Aleksandrova, Y.; Klochkov, S.; Fan, R. Therapeutic Influence on Important Targets Associated with Chronic Inflammation and Oxidative Stress in Cancer Treatment. Cancers 2021, 13, 6062. [Google Scholar] [CrossRef]
- Lodh, M.; Goswami, B.; Gupta, N.; Patra, S.K.; Saxena, A. Assessment of Oxidative Stress and Inflammatory Process in Patients of Multiple Myeloma. Indian J. Clin. Biochem. 2012, 27, 410–413. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fraile-Bermúdez, A.B.; Kortajarena, M.; Zarrazquin, I.; Maquibar, A.; Yanguas, J.J.; Sánchez-Fernández, C.E.; Gil, J.; Irazusta, A.; Ruiz-Litago, F. Relationship between Physical Activity and Markers of Oxidative Stress in Independent Community-Living Elderly Individuals. Exp. Gerontol. 2015, 70, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Żychowska, M.; Grzybkowska, A.; Wiech, M.; Urbański, R.; Pilch, W.; Piotrowska, A.; Czerwińska-Ledwig, O.; Antosiewicz, J. Exercise Training and Vitamin c Supplementation Affects Ferritin Mrna in Leukocytes without Affecting Prooxidative/Antioxidative Balance in Elderly Women. Int. J. Mol. Sci. 2020, 21, 6469. [Google Scholar] [CrossRef] [PubMed]
- Pilch, W.; Tota, Ł.; Piotrowska, A.; Śliwicka, E.; Czerwińska-Ledwig, O.; Zuziak, R.; Pilaczyńska-Szcześniak, Ł. Effects of Nordic Walking on Oxidant and Antioxidant Status: Levels of Calcidiol and Proinflammatory Cytokines in Middle-Aged Women. Oxid. Med. Cell. Longev. 2018, 2018, 6468234. [Google Scholar] [CrossRef]
- Czerwińska-Ledwig, O.; Gradek, J.; Deląg, J.; Jurczyszyn, A. P-145: The Effect of a 6-Week Nordic Walking Training Cycle on Myeloma-Related Blood Parameters, Vitamin 25(OH)D3 Serum Concentration and Peripheral Polyneuropathy Symptoms in Patients with Multiple Myeloma. Clin. Lymphoma Myeloma Leuk. 2021, 21, S114–S115. [Google Scholar] [CrossRef]
- Czerwińska-Ledwig, O.; Vesole, D.H.; Piotrowska, A.; Gradek, J.; Pilch, W.; Jurczyszyn, A. Effect of a 6-Week Cycle of Nordic Walking Training on Vitamin 25(OH)D3, Calcium-Phosphate Metabolism and Muscle Damage in Multiple Myeloma Patients-Randomized Controlled Trial. J. Clin. Med. 2022, 11, 6534. [Google Scholar] [CrossRef]
- Sawilowsky, S.S. New Effect Size Rules of Thumb. J. Mod. Appl. Stat. Methods 2009, 8, 597–599. [Google Scholar] [CrossRef]
- Xiang, F.; Shuanglun, X.; Jingfeng, W.; Ruqiong, N.; Yuan, Z.; Yongqing, L.; Jun, Z. Association of Serum 8-Hydroxy-2′-Deoxyguanosine Levels with the Presence and Severity of Coronary Artery Disease. Coron. Artery Dis. 2011, 22, 223–227. [Google Scholar] [CrossRef]
- Mueller, T.; Dieplinger, B.; Gegenhuber, A.; Haidinger, D.; Schmid, N.; Roth, N.; Ebner, F.; Landl, M.; Poelz, W.; Haltmayer, M. Serum Total 8-Iso-Prostaglandin F 2α: A New and Independent Predictor of Peripheral Arterial Disease. J. Vasc. Surg. 2004, 40, 768–773. [Google Scholar] [CrossRef][Green Version]
- Sun, Y.C.; Chang, P.Y.; Tsao, K.C.; Wu, T.L.; Sun, C.F.; Wu, L.L.; Wu, J.T. Establishment of a Sandwich ELISA Using Commercial Antibody for Plasma or Serum 3-Nitrotyrosine (3NT). Elevation in Inflammatory Diseases and Complementary between 3NT and Myeloperoxidase. Clin. Chim. Acta 2007, 378, 175–180. [Google Scholar] [CrossRef]
- Strohmaier, H.; Hinghofer-Szalkay, H.; Jörg Schaur, R. Detection of 4-Hydroxynonenal (HNE) as a Physiological Component in Human Plasma. J. Lipid Mediat. Cell Signal. 1995, 11, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yu, Z.; Cheng, H.; Mao, X.; Sui, W.; Deng, S.; Wei, X.; Lv, J.; Du, C.; Xu, J.; et al. Multiple Myeloma Hinders Erythropoiesis and Causes Anaemia Owing to High Levels of CCL3 in the Bone Marrow Microenvironment. Sci. Rep. 2020, 10, 20508. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Pan, J.; Chen, X.; Wang, L.; Chen, L.; Tian, Y.; Wang, W. Dynamic Monitoring of Serum Ferritin as an Adverse Prognostic Biomarker in Patients with Multiple Myeloma. Biomark. Med. 2021, 15, 1541–1551. [Google Scholar] [CrossRef] [PubMed]
- Bertoli, S.; Paubelle, E.; Bérard, E.; Saland, E.; Thomas, X.; Tavitian, S.; Larcher, M.V.; Vergez, F.; Delabesse, E.; Sarry, A.; et al. Ferritin Heavy/Light Chain (FTH1/FTL) Expression, Serum Ferritin Levels, and Their Functional as Well as Prognostic Roles in Acute Myeloid Leukemia. Eur. J. Haematol. 2019, 102, 131–142. [Google Scholar] [CrossRef]
- Kortas, J.; Prusik, K.; Flis, D.; Prusik, K.; Ziemann, E.; Antosiewicz, J. Effect of Nordic Walking Training on Iron Metabolism in Elderly Women. Clin. Interv. Aging 2015, 10, 1889–1896. [Google Scholar] [CrossRef]
- Kortas, J.; Ziemann, E.; Juszczak, D.; Micielska, K.; Kozłowska, M.; Prusik, K.; Prusik, K.; Antosiewicz, J. Iron Status in Elderly Women Impacts Myostatin, Adiponectin and Osteocalcin Levels Induced by Nordic Walking Training. Nutrients 2020, 12, 1129. [Google Scholar] [CrossRef] [PubMed]
- Walentukiewicz, A.; Lysak-Radomska, A.; Jaworska, J.; Prusik, K.; Prusik, K.; Kortas, J.A.; Lipí Nski, M.; Babinska, A.; Antosiewicz, J.; Ziemann, E. Vitamin D Supplementation and Nordic Walking Training Decreases Serum Homocysteine and Ferritin in Elderly Women. Int. J. Environ. Res. Public Health 2018, 15, 2064. [Google Scholar] [CrossRef]
- Lipiński, P.; Starzyński, R.R.; Styś, A.; Gajowiak, A.; Staroń, R. Heme Metabolism as an Integral Part of Iron Homeostasis. Postepy Hig. Med. Dosw. 2014, 68, 557–570. [Google Scholar] [CrossRef]
- Miceli, M.H.; Dong, L.; Grazziutti, M.L.; Fassas, A.; Thertulien, R.; Van Rhee, F.; Barlogie, B.; Anaissie, E.J. Iron Overload Is a Major Risk Factor for Severe Infection after Autologous Stem Cell Transplantation: A Study of 367 Myeloma Patients. Bone Marrow Transpl. 2006, 37, 857–864. [Google Scholar] [CrossRef]
- Schumacher, Y.O.; Schmid, A.; König, D.; Berg, A. Effects of Exercise on Soluble Transferrin Receptor and Other Variables of the Iron Status. Br. J. Sports Med. 2002, 36, 195–199. [Google Scholar] [CrossRef]
- Cichoń, J.; Ostapiuk-Karolczuk, J.; Cieślicka, M.; Dziewiecka, H.; Marcinkiewicz, A.; Tafil-Klawe, M.; Basta, P.; Maciejewski, D.; Skarpańska-Stejnborn, A. Effect of an Acute Exercise on Early Responses of Iron and Iron Regulatory Proteins in Young Female Basketball Players. BMC Sports Sci. Med. Rehabil. 2022, 14, 1–13. [Google Scholar] [CrossRef]
- Dos Santos, T.N.; Duarte, F.B.; Filho, P.A.M.; De Jesus Dos Santos, T.E.; Barbosa, M.C.; De Almeida Filho, T.P.; Cavalcanti, B.C.; De Vasconcelos, P.R.L.; Dutra, L.L.; Lopes, G.S.; et al. Association of Oxidative Stress and DNA Damage with Grafting Time in Patients with Multiple Myeloma and Lymphoma Submitted to Autologous Hematopoietic Stem Cell Transplantation. Rev. Assoc. Med. Bras. 2016, 62, 39–43. [Google Scholar] [CrossRef]
- Khadem-Ansari, M.H.; Nozari, S.; Asoudeh, M.; Rasmi, Y.; Faridvand, Y. Elevated Serum 8-Hydroxy-2’-Deoxyguanosine, Nitrite, and Nitrate in Patients with Stage I Multiple Myeloma. Int. J. Cancer Manag. 2017, 10, e8493. [Google Scholar] [CrossRef]
- Tandon, R.; Bhakar, M.; Pande, D.; Karki, K.; Khanna, H.D.; Tandon, R.; Bhakar, M.; Pande, D.; Karki, K. Oxidative Stress Induced Lipid Peroxidation and DNA Adduct Formation in the Pathogenesis of Multiple Myeloma and Lymphoma Oxidative Stress Induced Lipid Peroxidation and DNA Adduct Formation in the Pathogenesis of Multiple Myeloma and Lymphoma. J. Stress Physiol. Biochem. 2013, 9, 106–112. [Google Scholar]
- Sabuncuoǧlu, S.; Ztaş, Y.; Ukan Etinkaya, D.; Zgneş, N.; Zgneş, H. Oxidative Protein Damage with Carbonyl Levels and Nitrotyrosine Expression after Chemotherapy in Bone Marrow Transplantation Patients. Pharmacology 2012, 89, 283–286. [Google Scholar] [CrossRef]
- Kortas, J.; Kuchta, A.; Prusik, K.; Prusik, K.; Ziemann, E.; Labudda, S.; Ćwiklińska, A.; Wieczorek, E.; Jankowski, M.; Antosiewicz, J. Nordic Walking Training Attenuation of Oxidative Stress in Association with a Drop in Body Iron Stores in Elderly Women. Biogerontology 2017, 18, 517–524. [Google Scholar] [CrossRef]
- Pilch, W.; Żychowska, M.; Piotrowska, A.; Czerwińska-Ledwig, O.; Mikuľáková, W.; Sadowska-Krȩpa, E. Effects of Elevated Body Temperature on Selected Physiological Indices and Thermal Stress in Athletes and Non- Athletes. J. Hum. Kinet. 2022, 84, 112–123. [Google Scholar] [CrossRef]
- Traustadóttir, T.; Davies, S.S.; Su, Y.; Choi, L.; Brown-Borg, H.M.; Roberts, L.J.; Harman, S.M. Oxidative Stress in Older Adults: Effects of Physical Fitness. Age 2012, 34, 969–982. [Google Scholar] [CrossRef]
- Terpos, E.; Zamagni, E.; Lentzsch, S.; Drake, M.T.; García-Sanz, R.; Abildgaard, N.; Ntanasis-Stathopoulos, I.; Schjesvold, F.; de la Rubia, J.; Kyriakou, C.; et al. Treatment of Multiple Myeloma-Related Bone Disease: Recommendations from the Bone Working Group of the International Myeloma Working Group. Lancet Oncol. 2021, 22, e119–e130. [Google Scholar] [CrossRef] [PubMed]
- Chomczynski, P.; Sacchi, N. Single-Step Method of RNA Isolation by Acid Guanidinium Thiocyanate-Phenol-Chloroform Extraction. Anal. Biochem. 1987, 162, 156–159. [Google Scholar] [CrossRef]
- Nes, B.M.; Janszky, I.; Wisløff, U.; Støylen, A.; Karlsen, T. Age-Predicted Maximal Heart Rate in Healthy Subjects: The HUNT Fitness Study. Scand. J. Med. Sci. Sport. 2013, 23, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Schmittgen, T.D.; Livak, K.J. Analyzing Real-Time PCR Data by the Comparative CT Method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef] [PubMed]

| Baseline | 3 Weeks | 6 Weeks | 9 Weeks (Follow Up) | p | ||
|---|---|---|---|---|---|---|
| WBC [* 103/μL] | NW | 5.43 ± 1.23 | 5.80 ± 1.07 | 5.81 ± 1.31 * | 5.37 ± 1.16 * | * 0.02 |
| CG | 5.55 ± 0.86 | 5.54 ± 0.87 | 5.63 ± 0.86 | 5.53 ± 0.86 | >0.05 | |
| p | >0.05 | >0.05 | >0.05 | >0.05 | ||
| Cohen d | 0.11 | 0.27 | 0.16 | 0.16 | ||
| RBC [* 106/μL] | NW | 4.28 ± 0.30 *,& | 4.47 ± 0.29 * | 4.33 ± 0.27 $ | 4.47 ± 0.28 &,$ | * 0.01 |
| CG | 4.23 ± 0.32 | 4.32 ± 0.34 | 4.27 ± 0.38 | 4.26 ± 0.41 | & 0.04 | |
| p | >0.05 | >0.05 | >0.05 | >0.05 | $ 0.03 | |
| Cohen d | 0.24 | 0.63 | 0.18 | 0.60 | ||
| HGB [g/dL] | NW | 13.16 ± 1.41 | 13.73 ± 1.31 | 13.42 ± 1.03 | 13.35 ± 1.01 | >0.05 |
| CG | 13.17 ± 0.82 | 13.18 ± 0.88 | 13.14 ± 0.88 | 13.12 ± 0.89 | >0.05 | |
| p | >0.05 | >0.05 | >0.05 | >0.05 | ||
| Cohen d | 0.00 | 0.50 | 0.29 | 0.24 |
| Baseline | 3 Weeks | 6 Weeks | 9 Weeks (Follow Up) | p | ||
|---|---|---|---|---|---|---|
| Iron [μmol/L] | NW | 18.74 ± 6.38 *,# | 17.79 ± 5.58 # | 14.85 ± 2.68 * | 17.08 ± 9.22 | * 0.029 |
| CG | 14.06 ± 4.34 # | 14.36 ± 2.36 # | 16.52 ± 3.81 | 15.42 ± 2.36 | >0.05 | |
| p | 0.026 | 0.036 | >0.05 | >0.05 | ||
| Cohen d | 0.86 | 0.80 | −0.52 | 0.66 | ||
| Ferritin [ng/mL] | NW | 92.62 ± 63.63 # | 93.90 ± 70.57 # | 95.48 ± 50.58 | 84.53 ± 62.50 # | >0.05 |
| CG | 143.19 ± 68.58 # | 146.20 ± 47.82 # | 122.04 ± 42.53 | 135.49 ± 49.18 # | >0.05 | |
| p | 0.042 | 0.023 | >0.05 | 0.018 | ||
| Cohen d | −0.76 | −0.87 | −0.57 | −0.90 | ||
| Transferin [g/L] | NW | 2.41 ± 0.42 # | 2.35 ± 0.34 | 2.37 ± 0.37 * | 2.36 ± 0.32 | >0.05 |
| CG | 2.15 ± 0.25 # | 2.15 ± 0.24 | 2.28 ± 0.21 | 2.21 ± 0.23 | >0.05 | |
| p | 0.049 | >0.05 | >0.05 | >0.05 | ||
| Cohen d | 0.75 | 0.68 | 0.30 | 0.53 | ||
| TfS [%] | NW | 40.1 ± 14.4 * | 38.6 ± 12.5 | 31.9 ± 7.3 * | 40.1 ± 20.6 | * 0.037 |
| CG | 32.4 ± 8.2 | 33.3 ± 3.0 | 36.5 ± 8.5 | 35.1 ± 5.3 | >0.05 | |
| p | >0.05 | >0.05 | >0.05 | >0.05 | ||
| Cohen d | 0.66 | 0.36 | −0.58 | 3.05 |
| Inclusion Criteria: | Exclusion Criteria: |
|---|---|
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Czerwińska-Ledwig, O.; Jurczyszyn, A.; Piotrowska, A.; Pilch, W.; Antosiewicz, J.; Żychowska, M. The Effect of a Six-Week Nordic Walking Training Cycle on Oxidative Damage of Macromolecules and Iron Metabolism in Older Patients with Multiple Myeloma in Remission—Randomized Clinical Trial. Int. J. Mol. Sci. 2023, 24, 15358. https://doi.org/10.3390/ijms242015358
Czerwińska-Ledwig O, Jurczyszyn A, Piotrowska A, Pilch W, Antosiewicz J, Żychowska M. The Effect of a Six-Week Nordic Walking Training Cycle on Oxidative Damage of Macromolecules and Iron Metabolism in Older Patients with Multiple Myeloma in Remission—Randomized Clinical Trial. International Journal of Molecular Sciences. 2023; 24(20):15358. https://doi.org/10.3390/ijms242015358
Chicago/Turabian StyleCzerwińska-Ledwig, Olga, Artur Jurczyszyn, Anna Piotrowska, Wanda Pilch, Jędrzej Antosiewicz, and Małgorzata Żychowska. 2023. "The Effect of a Six-Week Nordic Walking Training Cycle on Oxidative Damage of Macromolecules and Iron Metabolism in Older Patients with Multiple Myeloma in Remission—Randomized Clinical Trial" International Journal of Molecular Sciences 24, no. 20: 15358. https://doi.org/10.3390/ijms242015358
APA StyleCzerwińska-Ledwig, O., Jurczyszyn, A., Piotrowska, A., Pilch, W., Antosiewicz, J., & Żychowska, M. (2023). The Effect of a Six-Week Nordic Walking Training Cycle on Oxidative Damage of Macromolecules and Iron Metabolism in Older Patients with Multiple Myeloma in Remission—Randomized Clinical Trial. International Journal of Molecular Sciences, 24(20), 15358. https://doi.org/10.3390/ijms242015358








