Unilateral Hypofunction of the Masseter Leads to Molecular and 3D Morphometric Signs of Atrophy in Ipsilateral Agonist Masticatory Muscles in Adult Mice
Abstract
:1. Introduction
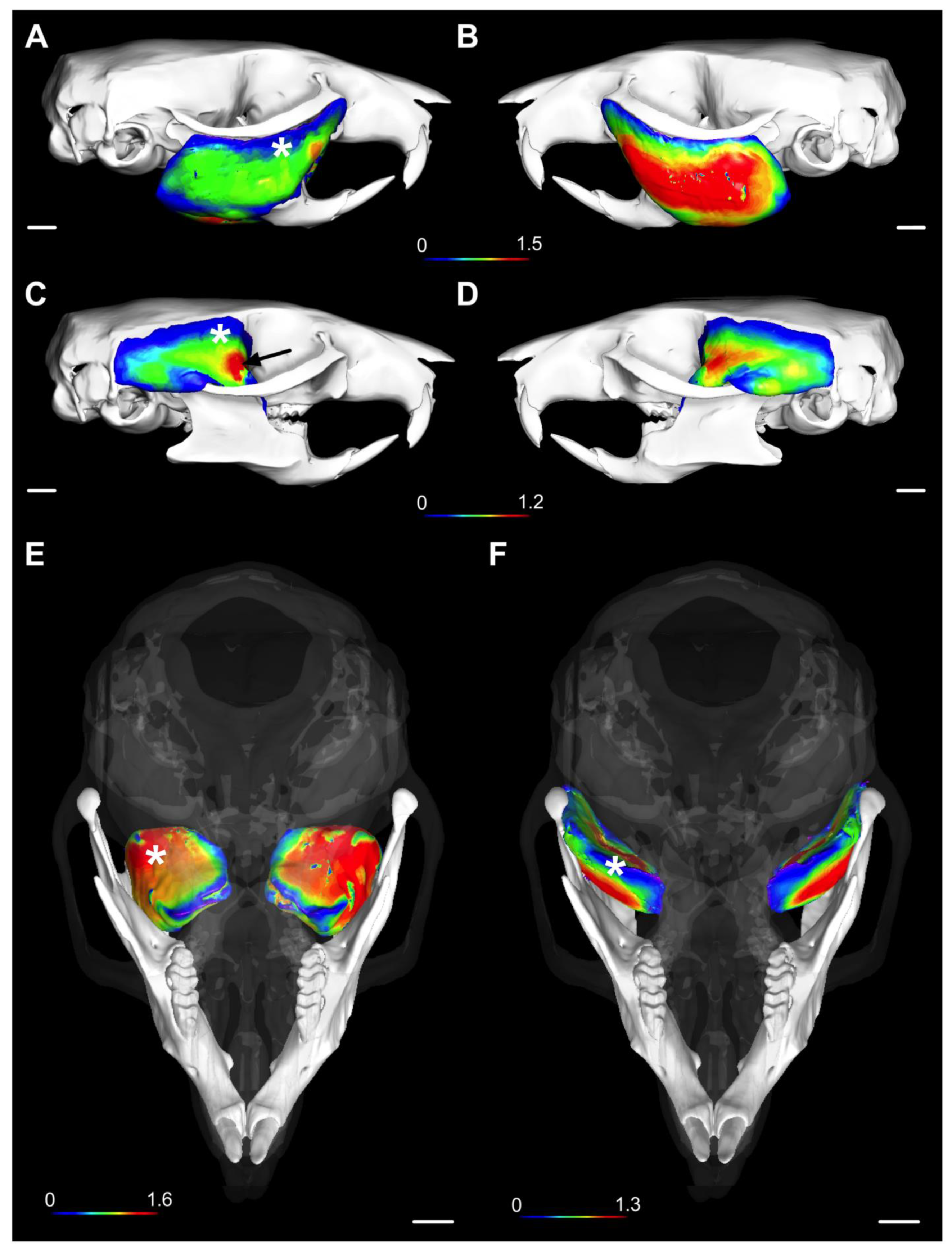
2. Results
3. Discussion
4. Materials and Methods
4.1. Unilateral Masseter Hypofunction: Adult Mouse Model
4.2. Relative Levels of mRNA of Atrophy-Related Molecular Markers in Mouse Masticatory Muscles
4.3. The Three-Dimensional Phenotyping of Mouse Masticatory Muscles with X-ray Microtomography (µCT)
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yasuda, G.; Moriuchi, E.; Hamanaka, R.; Fujishita, A.; Yoshimi, T.; Yamamoto, K.; Hayashida, K.; Koga, Y.; Yoshida, N. Visualization of mandibular movement relative to the maxilla during mastication in mice: Integration of kinematic analysis and reconstruction of a three-dimensional model of the maxillofacial structure. BMC Oral Health 2021, 21, 527. [Google Scholar] [CrossRef] [PubMed]
- Yoshimi, T.; Koga, Y.; Nakamura, A.; Fujishita, A.; Kohara, H.; Moriuchi, E.; Yoshimi, K.; Tsai, C.Y.; Yoshida, N. Mechanism of motor coordination of masseter and temporalis muscles for increased masticatory efficiency in mice. J. Oral. Rehabil. 2017, 44, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Baverstock, H.; Jeffery, N.S.; Cobb, S.N. The morphology of the mouse masticatory musculature. J. Anat. 2013, 223, 46–60. [Google Scholar] [CrossRef] [PubMed]
- Okayasu, I.; Yamada, Y.; Kohno, S.; Yoshida, N. New animal model for studying mastication in oral motor disorders. J. Dent. Res. 2003, 82, 318–321. [Google Scholar] [CrossRef] [PubMed]
- Moriuchi, E.; Hamanaka, R.; Koga, Y.; Fujishita, A.; Yoshimi, T.; Yasuda, G.; Kohara, H.; Yoshida, N. Development and evaluation of a jaw-tracking system for mice: Reconstruction of three-dimensional movement trajectories on an arbitrary point on the mandible. Biomed. Eng. Online 2019, 18, 59. [Google Scholar] [CrossRef] [PubMed]
- Munoz Lora, V.R.M.; Del Bel Cury, A.A.; Jabbari, B.; Lackovic, Z. Botulinum Toxin Type A in Dental Medicine. J. Dent. Res. 2019, 98, 1450–1457. [Google Scholar] [CrossRef]
- Pirazzini, M.; Rossetto, O.; Eleopra, R.; Montecucco, C. Botulinum Neurotoxins: Biology, Pharmacology, and Toxicology. Pharmacol. Rev. 2017, 69, 200–235. [Google Scholar] [CrossRef]
- Balanta-Melo, J.; Toro-Ibacache, V.; Torres-Quintana, M.A.; Kupczik, K.; Vega, C.; Morales, C.; Hernandez-Moya, N.; Arias-Calderon, M.; Beato, C.; Buvinic, S. Early molecular response and microanatomical changes in the masseter muscle and mandibular head after botulinum toxin intervention in adult mice. Ann. Anat. Anat. Anz. 2018, 216, 112–119. [Google Scholar] [CrossRef]
- Balanta-Melo, J.; Torres-Quintana, M.A.; Bemmann, M.; Vega, C.; Gonzalez, C.; Kupczik, K.; Toro-Ibacache, V.; Buvinic, S. Masseter muscle atrophy impairs bone quality of the mandibular condyle but not the alveolar process early after induction. J. Oral. Rehabil. 2019, 46, 233–241. [Google Scholar] [CrossRef]
- Balanta-Melo, J.; Bemmann, M.; Ibacache, V.T.; Kupczik, K.; Buvinic, S. Three-dimensional assessment of enamel and dentine in mouse molar teeth during masseter muscle hypofunction. Rev. Estomatol. 2019, 26, 30–37. [Google Scholar] [CrossRef]
- Pereira, I.N.; Hassan, H. Botulinum toxin A in dentistry and orofacial surgery: An evidence-based review—Part 1: Therapeutic applications. Evid. Based Dent. 2022. [Google Scholar] [CrossRef] [PubMed]
- Klasser, G.D.; Goulet, J.P.; Moreno-Hay, I. Classification and Diagnosis of Temporomandibular Disorders and Temporomandibular Disorder Pain. Dent. Clin. N. Am. 2023, 67, 211–225. [Google Scholar] [CrossRef] [PubMed]
- Zagury, J.G.; Ananthan, S.; Quek, S.Y.P.; Subramanian, G. Myofascial Temporomandibular Disorders at a Turning Point: Pragmatic or Evidence-Based Management? Dent. Clin. N. Am. 2023, 67, 335–348. [Google Scholar] [CrossRef] [PubMed]
- Yost, O.; Liverman, C.T.; English, R.; Mackey, S.; Bond, E.C. (Eds.) Temporomandibular Disorders: Priorities for Research and Care; The National Academies Collection: Reports funded by National Institutes of Health; National Academies Press: Washington, DC, USA, 2020. [Google Scholar]
- Valesan, L.F.; Da-Cas, C.D.; Reus, J.C.; Denardin, A.C.S.; Garanhani, R.R.; Bonotto, D.; Januzzi, E.; de Souza, B.D.M. Prevalence of temporomandibular joint disorders: A systematic review and meta-analysis. Clin. Oral Investig. 2021, 25, 441–453. [Google Scholar] [CrossRef]
- Haddad, C.; Sayegh, S.M.; El Zoghbi, A.; Lawand, G.; Nasr, L. The Prevalence and Predicting Factors of Temporomandibular Disorders in COVID-19 Infection: A Cross-Sectional Study. Cureus 2022, 14, e28167. [Google Scholar] [CrossRef]
- Minervini, G.; Franco, R.; Marrapodi, M.M.; Mehta, V.; Fiorillo, L.; Badnjevic, A.; Cervino, G.; Cicciu, M. The Association between COVID-19 Related Anxiety, Stress, Depression, Temporomandibular Disorders, and Headaches from Childhood to Adulthood: A Systematic Review. Brain Sci. 2023, 13, 481. [Google Scholar] [CrossRef] [PubMed]
- Ginszt, M.; Zielinski, G.; Szkutnik, J.; Wojcicki, M.; Baszczowski, M.; Litko-Rola, M.; Zielinska, D.; Rozylo-Kalinowska, I. The Difference in Electromyographic Activity While Wearing a Medical Mask in Women with and without Temporomandibular Disorders. Int. J. Environ. Res. Public Health 2022, 18, 15559. [Google Scholar] [CrossRef]
- Qamar, Z.; Alghamdi, A.M.S.; Haydarah, N.K.B.; Balateef, A.A.; Alamoudi, A.A.; Abumismar, M.A.; Shivakumar, S.; Cicciu, M.; Minervini, G. Impact of temporomandibular disorders on oral health-related quality of life: A systematic review and meta-analysis. J. Oral Rehabil. 2023, 50, 706–714. [Google Scholar] [CrossRef]
- Nowak, Z.; Checinski, M.; Nitecka-Buchta, A.; Bulanda, S.; Ilczuk-Rypula, D.; Postek-Stefanska, L.; Baron, S. Intramuscular Injections and Dry Needling within Masticatory Muscles in Management of Myofascial Pain. Systematic Review of Clinical Trials. Int. J. Environ. Res. Public Health 2021, 18, 9552. [Google Scholar] [CrossRef]
- De la Torre Canales, G.; Poluha, R.L.; Alvarez Pinzon, Y.N.; Rodrigues Conti, P.C.; Manfredini, D.; Sanchez-Ayala, A.; Rizzatti-Barbosa, C.M. Effects of Botulinum Toxin Type A on the Psychosocial Features of Myofascial Pain TMD Subjects: A Randomized Controlled Trial. J. Oral Facial Pain Headache 2021, 35, 288–296. [Google Scholar] [CrossRef]
- De la Torre Canales, G.; Poluha, R.L.; Pinzon, N.A.; Da Silva, B.R.; Almeida, A.M.; Ernberg, M.; Manso, A.C.; Bonjardim, L.R.; Rizzatti-Barbosa, C.M. Efficacy of Botulinum Toxin Type-A I in the Improvement of Mandibular Motion and Muscle Sensibility in Myofascial Pain TMD Subjects: A Randomized Controlled Trial. Toxins 2022, 14, 441. [Google Scholar] [CrossRef] [PubMed]
- Agren, M.; Sahin, C.; Pettersson, M. The effect of botulinum toxin injections on bruxism: A systematic review. J. Oral Rehabil. 2020, 47, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Balanta-Melo, J.; Vargas, J.P.; Bendersky, J.; Villanueva, J. Toxina Botulínica tipo A para el bruxismo del sueño en adultos. Int. J. Interdiscip. Dent. 2022, 15, 101–107. [Google Scholar] [CrossRef]
- Sendra, L.A.; Azeredo Alves Antunes, L.; Barboza, E.P. Use of botulinum neurotoxin Type A in the management of primary bruxism in adults: An updated systematic review. J. Prosthet. Dent. 2022. [Google Scholar] [CrossRef]
- Rauso, R.; Lo Giudice, G.; Tartaro, G.; Zerbinati, N.; Nicoletti, G.F.; Fragola, R. Botulinum toxin type A injections for masticatory muscles hypertrophy: A systematic review. J. Cranio-Maxillofac. Surg. 2022, 50, 7–18. [Google Scholar] [CrossRef]
- Libouban, H.; Guintard, C.; Minier, N.; Aguado, E.; Chappard, D. Long-Term Quantitative Evaluation of Muscle and Bone Wasting Induced by Botulinum Toxin in Mice Using Microcomputed Tomography. Calcif. Tissue Int. 2018, 102, 695–704. [Google Scholar] [CrossRef]
- Ma, W.; Cai, Y.; Shen, Y.; Chen, X.; Zhang, L.; Ji, Y.; Chen, Z.; Zhu, J.; Yang, X.; Sun, H. HDAC4 Knockdown Alleviates Denervation-Induced Muscle Atrophy by Inhibiting Myogenin-Dependent Atrogene Activation. Front. Cell Neurosci. 2021, 15, 663384. [Google Scholar] [CrossRef]
- Macpherson, P.C.; Wang, X.; Goldman, D. Myogenin regulates denervation-dependent muscle atrophy in mouse soleus muscle. J. Cell Biochem. 2011, 112, 2149–2159. [Google Scholar] [CrossRef]
- Moresi, V.; Williams, A.H.; Meadows, E.; Flynn, J.M.; Potthoff, M.J.; McAnally, J.; Shelton, J.M.; Backs, J.; Klein, W.H.; Richardson, J.A.; et al. Myogenin and class II HDACs control neurogenic muscle atrophy by inducing E3 ubiquitin ligases. Cell 2010, 143, 35–45. [Google Scholar] [CrossRef]
- Chen, L.; Huang, H.W.; Gu, S.H.; Xu, L.; Gu, Y.D.; Xu, J.G. The study of myogenin expression in denervated human skeletal muscles. J. Int. Med. Res. 2011, 39, 378–387. [Google Scholar] [CrossRef]
- Hale, M.; Riding, S.; Singh, B.R. Near-infrared imaging of balb/c mice injected with a detoxified botulinum neurotoxin A. Botulinum J. 2010, 1, 431–441. [Google Scholar] [CrossRef]
- Buvinic, S.; Balanta-Melo, J.; Kupczik, K.; Vasquez, W.; Beato, C.; Toro-Ibacache, V. Muscle-Bone Crosstalk in the Masticatory System: From Biomechanical to Molecular Interactions. Front. Endocrinol. 2020, 11, 606947. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Michels, L.V.; Pitsillides, A.A.; Greeves, J.; Plotkin, L.I.; Cardo, V.; Sims, N.A.; Clarkin, C.E. Sexing Bones: Improving Transparency of Sex Reporting to Address Bias Within Preclinical Studies. J. Bone Miner. Res. 2023, 38, 5–13. [Google Scholar] [CrossRef]
- Tsouknidas, A.; Jimenez-Rojo, L.; Karatsis, E.; Michailidis, N.; Mitsiadis, T.A. A Bio-Realistic Finite Element Model to Evaluate the Effect of Masticatory Loadings on Mouse Mandible-Related Tissues. Front. Physiol. 2017, 8, 273. [Google Scholar] [CrossRef] [PubMed]
- Van Spronsen, P.H. Long-Face Craniofacial Morphology: Cause or Effect of Weak Masticatory Musculature? Semin. Orthod. 2010, 16, 99–117. [Google Scholar] [CrossRef]
- Plotkin, L.I.; Buvinic, S.; Balanta-Melo, J. In vitro and in vivo studies using non-traditional bisphosphonates. Bone 2020, 134, 115301. [Google Scholar] [CrossRef]
- Botzenhart, U.U.; Keil, C.; Tsagkari, E.; Zeidler-Rentzsch, I.; Gredes, T.; Gedrange, T. Influence of botulinum toxin A on craniofacial morphology after injection into the right masseter muscle of dystrophin deficient (mdx-) mice. Ann. Anat. 2021, 236, 151715. [Google Scholar] [CrossRef]
- Dutra, E.H.; Yadav, S. The effects on the mandibular condyle of Botox injection into the masseter are not transient. Am. J. Orthod. Dentofac. Orthop. 2019, 156, 193–202. [Google Scholar] [CrossRef]
- Balanta-Melo, J.; Toro-Ibacache, V.; Kupczik, K.; Buvinic, S. Mandibular Bone Loss after Masticatory Muscles Intervention with Botulinum Toxin: An Approach from Basic Research to Clinical Findings. Toxins 2019, 11, 84. [Google Scholar] [CrossRef]
- Moussa, M.S.; Bachour, D.; Komarova, S.V. Adverse effect of botulinum toxin-A injections on mandibular bone: A systematic review and meta-analysis. J. Oral Rehabil. 2023. [Google Scholar] [CrossRef]
- De la Torre Canales, G.; Alvarez-Pinzon, N.; Munoz-Lora, V.R.M.; Vieira Peroni, L.; Farias Gomes, A.; Sanchez-Ayala, A.; Haiter-Neto, F.; Manfredini, D.; Rizzatti-Barbosa, C.M. Efficacy and Safety of Botulinum Toxin Type A on Persistent Myofascial Pain: A Randomized Clinical Trial. Toxins 2020, 12, 395. [Google Scholar] [CrossRef]
- Morales-Jimenez, C.; Balanta-Melo, J.; Arias-Calderon, M.; Hernandez, N.; Gomez-Valenzuela, F.; Escobar, A.; Jaimovich, E.; Buvinic, S. Mechanical Disturbance of Osteoclasts Induces ATP Release That Leads to Protein Synthesis in Skeletal Muscle through an Akt-mTOR Signaling Pathway. Int. J. Mol. Sci. 2022, 23, 9444. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.T.; Chee, A.; Trieu, J.; Naim, T.; Lynch, G.S. Importance of functional and metabolic impairments in the characterization of the C-26 murine model of cancer cachexia. Dis. Model. Mech. 2012, 5, 533–545. [Google Scholar] [CrossRef] [PubMed]
- Church, J.E.; Trieu, J.; Sheorey, R.; Chee, A.Y.; Naim, T.; Baum, D.M.; Ryall, J.G.; Gregorevic, P.; Lynch, G.S. Functional beta-adrenoceptors are important for early muscle regeneration in mice through effects on myoblast proliferation and differentiation. PLoS ONE 2014, 9, e101379. [Google Scholar] [CrossRef] [PubMed]
- Bustamante, M.; Fernandez-Verdejo, R.; Jaimovich, E.; Buvinic, S. Electrical stimulation induces IL-6 in skeletal muscle through extracellular ATP by activating Ca2+ signals and an IL-6 autocrine loop. Am. J. Physiol. Endocrinol. Metab. 2014, 306, E869–E882. [Google Scholar] [CrossRef] [PubMed]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Lösel, P.; Heuveline, V. Enhancing a Diffusion Algorithm for 4D Image Segmentation Using Local Information. In Proceedings of the Medical Imaging 2016: Image Processing, San Diego, CA, USA, 27 February–3 March 2016. [Google Scholar]
- Losel, P.D.; van de Kamp, T.; Jayme, A.; Ershov, A.; Farago, T.; Pichler, O.; Tan Jerome, N.; Aadepu, N.; Bremer, S.; Chilingaryan, S.A.; et al. Introducing Biomedisa as an open-source online platform for biomedical image segmentation. Nat. Commun. 2020, 11, 5577. [Google Scholar] [CrossRef]


| BoNTA Side–Control Side | |||||||
|---|---|---|---|---|---|---|---|
| Muscle | Days | Gene | Control Side Mean ± SEM | BoNTA Side Mean ± SEM | p Value * | CI 95% ** | Cohen’s dz *** |
| Masseter | 2 | Atrogin-1 | 1.15 ± 0.28 | 6.55 ± 1.11 | 0.016 | [2.363, 8.429] | 1.867 |
| Murf1 | 0.82 ± 0.17 | 5.26 ± 1.21 | 0.016 | [1.278, 7.612] | 1.473 | ||
| Myogenin | 0.79 ± 0.20 | 2.36 ± 0.68 | 0.016 | [−0.181, 3.331] | 0.941 | ||
| 7 | Atrogin-1 | 1.15 ± 0.25 | 8.88 ± 1.86 | 0.016 | [2.611, 12.840] | 1.585 | |
| Murf1 | 1.02 ± 0.23 | 6.24 ± 2.30 | 0.016 | [−0.678, 11.120] | 0.929 | ||
| Myogenin | 0.63 ± 0.20 | 42.72 ± 12.35 | 0.016 | [10.220, 73.950] | 1.386 | ||
| 14 | Atrogin-1 | 0.84 ± 0.21 | 4.49 ± 0.78 | 0.016 | [1.535, 5.754] | 1.813 | |
| Murf1 | 0.80 ± 0.31 | 2.54 ± 0.69 | 0.016 | [0.696, 2.775] | 1.751 | ||
| Myogenin | 0.87 ± 0.12 | 77.13 ± 26.59 | 0.016 | [7.825, 144.700] | 1.169 | ||
| Temporalis | 2 | Atrogin-1 | 0.68 ± 0.16 | 10.22 ± 3.49 | 0.016 | [0.845, 18.250] | 1.151 |
| Murf1 | 1.72 ± 0.45 | 12.01 ± 5.04 | 0.031 | [−1.640, 22.210] | 0.906 | ||
| Myogenin | 1.37 ± 0.62 | 4.67 ± 2.06 | 0.016 | [−0.410, 7.024] | 0.934 | ||
| 7 | Atrogin-1 | 1.02 ± 0.09 | 9.05 ± 4.49 | 0.047 | [−3.473, 19.540] | 0.732 | |
| Murf1 | 3.13 ± 1.78 | 26.37 ± 6.41 | 0.031 | [4.173–42.320] | 1.514 | ||
| Myogenin | 1.61 ± 1.13 | 10.11 ± 2.87 | 0.031 | [−0.165, 17.170] | 1.218 | ||
| 14 | Atrogin-1 | 1.07 ± 0.18 | 7.21 ± 2.77 | 0.031 | [−1.187, 13.470] | 1.041 | |
| Murf1 | 0.78 ± 0.27 | 2.41 ± 0.44 | 0.031 | [0.9810, 2.287] | 3.107 | ||
| Myogenin | 1.24 ± 0.20 | 10.70 ± 0.68 | 0.031 | [−5.984, 24.900] | 0.760 | ||
| Medial pterygoid | 2 | Atrogin-1 | 1.52 ± 0.50 | 5.56 ± 2.98 | 0.063 | [−3.262, 11.340] | 0.687 |
| Murf1 | 1.28 ± 0.47 | 4.35 ± 1.32 | 0.125 | [−0.811, 6.957] | 0.982 | ||
| Myogenin | 0.66 ± 0.18 | 4.89 ± 1.28 | 0.031 | [0.5235, 7.918] | 1.417 | ||
| 7 | Atrogin-1 | 0.83 ± 0.44 | 5.98 ± 2.49 | 0.016 | [−0.634, 10.930] | 0.935 | |
| Murf1 | 1.38 ± 0.49 | 3.21 ± 1.35 | 0.109 | [−1.302, 4.949] | 0.612 | ||
| Myogenin | 1.28 ± 0.34 | 39.23 ± 9.00 | 0.016 | [5.120, 60.790] | 1.744 | ||
| 14 | Atrogin-1 | 0.81 ± 0.20 | 1.35 ± 0.52 | 0.219 | [−0.619, 1.699] | 0.489 | |
| Murf1 | 0.79 ± 0.42 | 0.49 ± 0.19 | 0.500 | [−1.725, 1.122] | 0.302 | ||
| Myogenin | 1.23 ± 0.39 | 25.89 ± 12.39 | 0.016 | [−10.50, 59.82] | 0.870 | ||
| Lateral pterygoid | 2 | Atrogin-1 | 1.23 ± 0.31 | 0.59 ± 0.15 | 0.094 | [−1.674, 0.394] | −0.769 |
| Murf1 | 1.38 ± 0.52 | 0.41 ± 0.10 | 0.016 | [−2.327, 0.390] | −0.748 | ||
| Myogenin | 0.83 ± 0.33 | 1.42 ± 0.57 | 0.281 | [−0.838, 2.001] | 0.43 | ||
| 7 | Atrogin-1 | 1.05 ± 0.22 | 4.23 ± 2.53 | 0.313 | [−3.831, 10.190] | 0.563 | |
| Murf1 | 1.11 ± 0.24 | 2.68 ± 1.47 | 0.281 | [−2.589, 5.272] | 0.396 | ||
| Myogenin | 0.86 ± 0.25 | 1.56 ± 0.63 | 0.281 | [−0.967, 2.377] | 0.443 | ||
| 14 | Atrogin-1 | 1.29 ± 0.33 | 1.12 ± 0.41 | 0.156 | [−0.558, 0.2178] | −0.460 | |
| Murf1 | 1.18 ± 0.31 | 0.90 ± 0.17 | 0.344 | [−1.060, 0.503] | −0.367 | ||
| Myogenin | 0.96 ± 0.42 | 2.45 ± 1.21 | 0.156 | [−1.905, 4.902] | 0.462 | ||
| Volume (mm3) | Thickness (mm) | |||||
|---|---|---|---|---|---|---|
| Muscles | Control Side Mean ± SEM | BoNTA Side Mean ± SEM | Mean Difference | Control Side Mean ± SEM | BoNTA Side Mean ± SEM | Mean Difference |
| Masseter | 43.6 ± 1.3 | 27.4 ± 0.4 | −16.3 * | 0.87 ± 0.01 | 0.64 ± 0.01 | −0.23 * |
| Temporalis | 21.3 ± 0.6 | 18.6 ± 0.7 | −2.6 * | 0.57 ± 0.00 | 0.56 ± 0.00 | −0.01 * |
| Medial pterygoid | 13.7 ± 0.6 | 11.5 ± 0.5 | −2.2 * | 0.97 ± 0.02 | 0.88 ± 0.02 | −0.09 * |
| Lateral pterygoid | 7.4 ± 0.2 | 7.9 ± 0.2 | +0.5 * | 0.72 ± 0.01 | 0.73 ± 0.01 | +0.01 ns |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balanta-Melo, J.; Eyquem-Reyes, A.; Blanco, N.; Vásquez, W.; Kupczik, K.; Toro-Ibacache, V.; Buvinic, S. Unilateral Hypofunction of the Masseter Leads to Molecular and 3D Morphometric Signs of Atrophy in Ipsilateral Agonist Masticatory Muscles in Adult Mice. Int. J. Mol. Sci. 2023, 24, 14740. https://doi.org/10.3390/ijms241914740
Balanta-Melo J, Eyquem-Reyes A, Blanco N, Vásquez W, Kupczik K, Toro-Ibacache V, Buvinic S. Unilateral Hypofunction of the Masseter Leads to Molecular and 3D Morphometric Signs of Atrophy in Ipsilateral Agonist Masticatory Muscles in Adult Mice. International Journal of Molecular Sciences. 2023; 24(19):14740. https://doi.org/10.3390/ijms241914740
Chicago/Turabian StyleBalanta-Melo, Julián, Andrea Eyquem-Reyes, Noelia Blanco, Walter Vásquez, Kornelius Kupczik, Viviana Toro-Ibacache, and Sonja Buvinic. 2023. "Unilateral Hypofunction of the Masseter Leads to Molecular and 3D Morphometric Signs of Atrophy in Ipsilateral Agonist Masticatory Muscles in Adult Mice" International Journal of Molecular Sciences 24, no. 19: 14740. https://doi.org/10.3390/ijms241914740
APA StyleBalanta-Melo, J., Eyquem-Reyes, A., Blanco, N., Vásquez, W., Kupczik, K., Toro-Ibacache, V., & Buvinic, S. (2023). Unilateral Hypofunction of the Masseter Leads to Molecular and 3D Morphometric Signs of Atrophy in Ipsilateral Agonist Masticatory Muscles in Adult Mice. International Journal of Molecular Sciences, 24(19), 14740. https://doi.org/10.3390/ijms241914740









