Sm/Co Magnetic Materials: A Recycling Strategy Using Modifiable Hydrophobic Deep Eutectic Solvents Based on Trioctylphosphine Oxide
Abstract
:1. Introduction
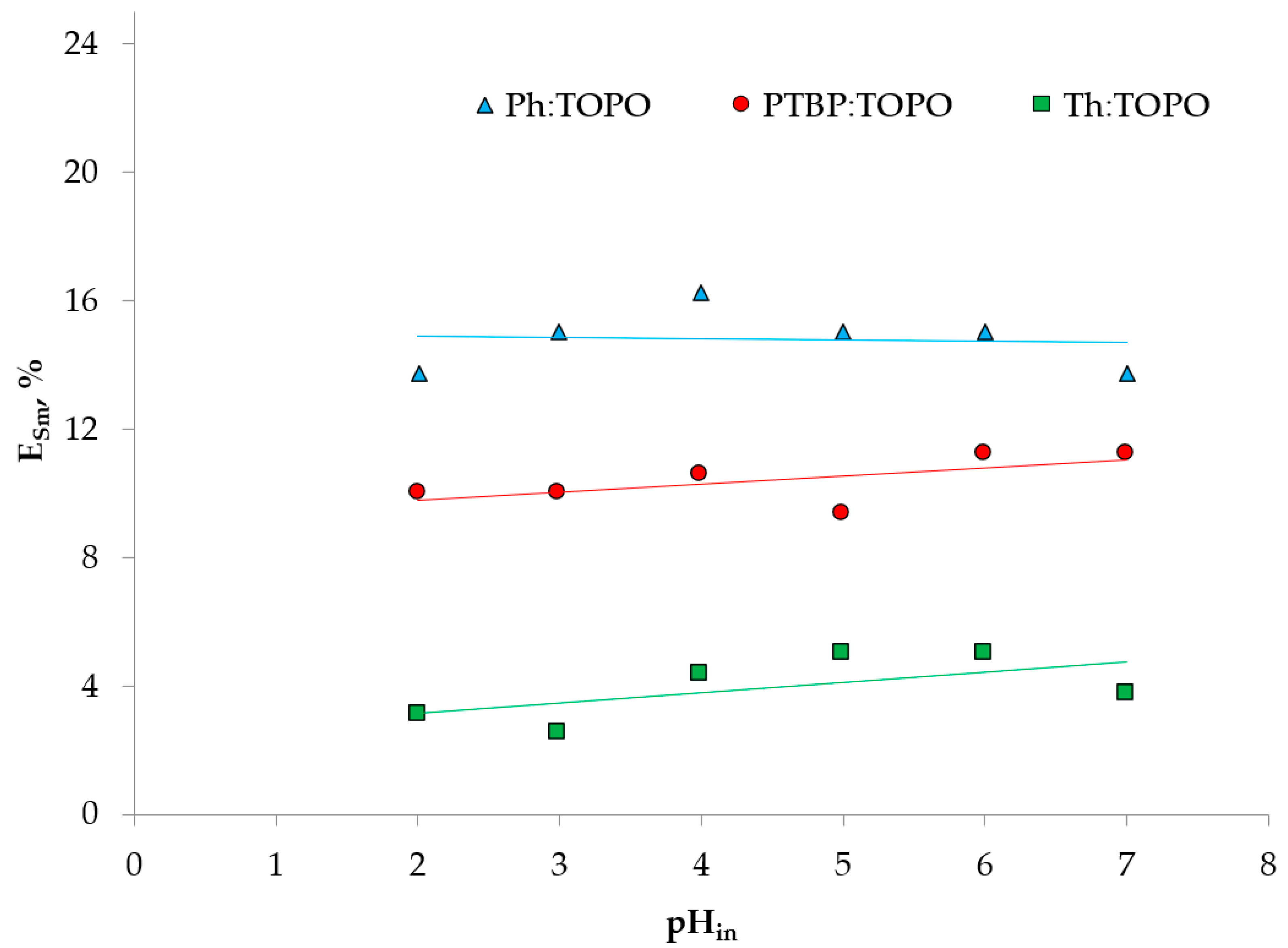
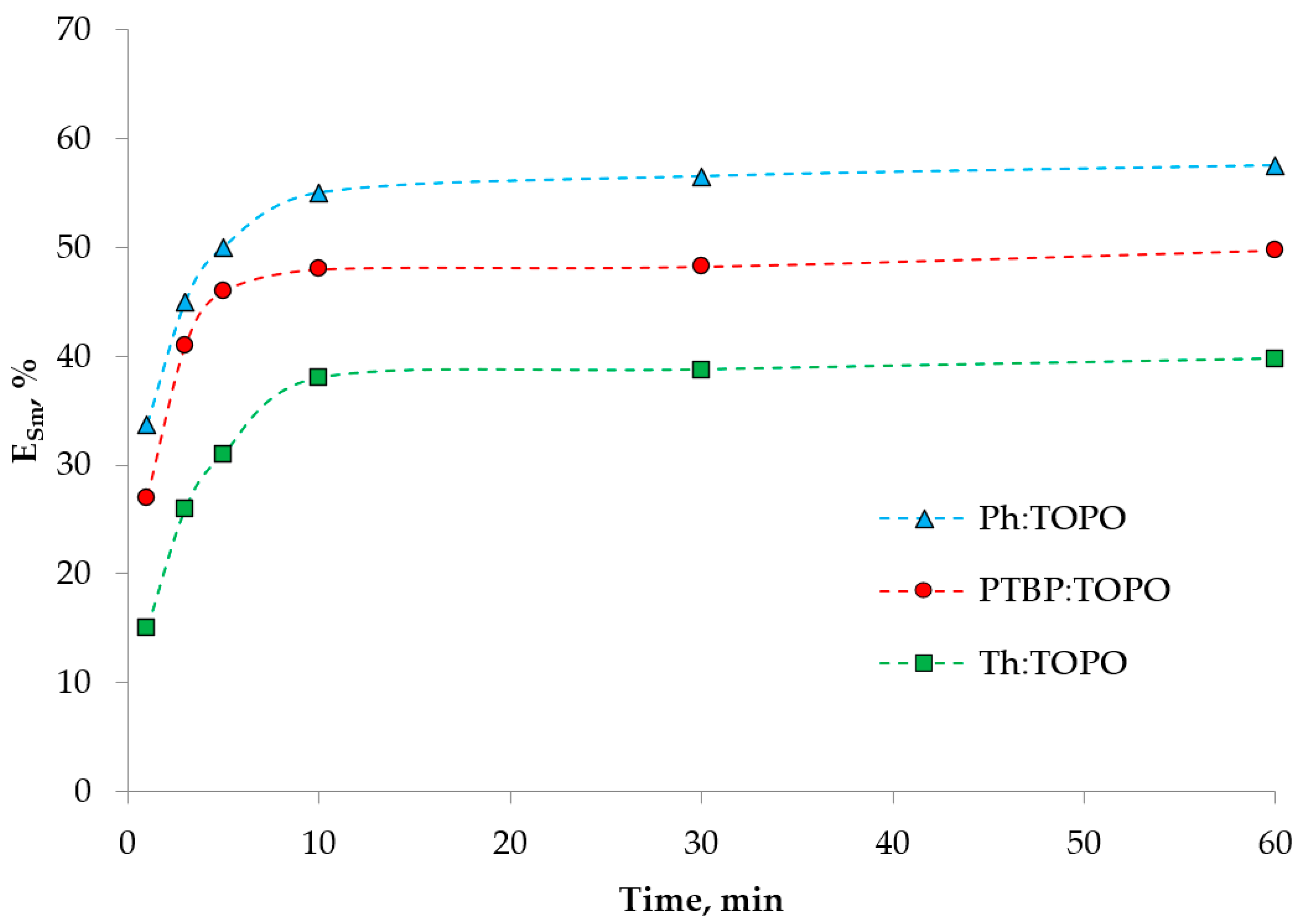
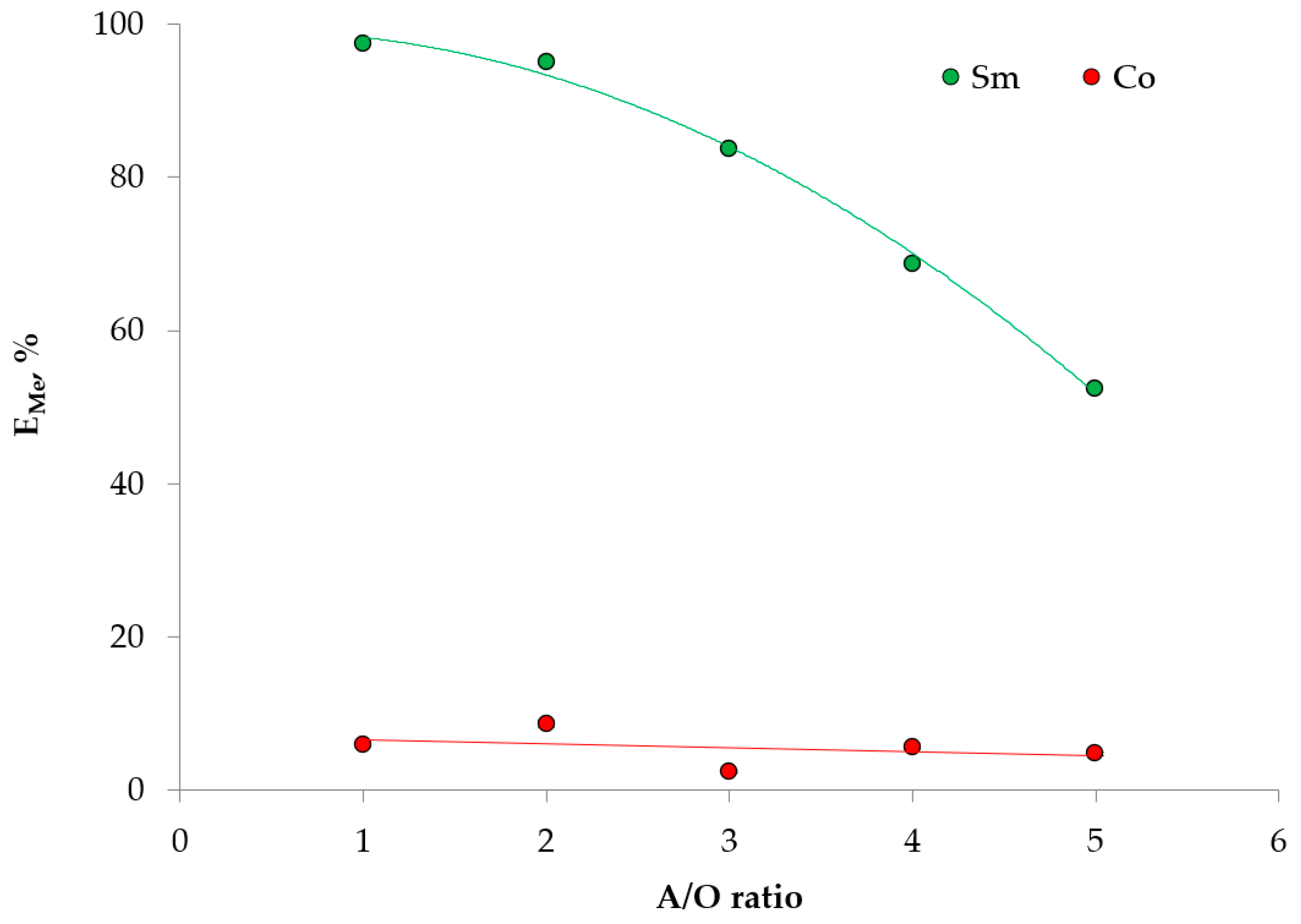
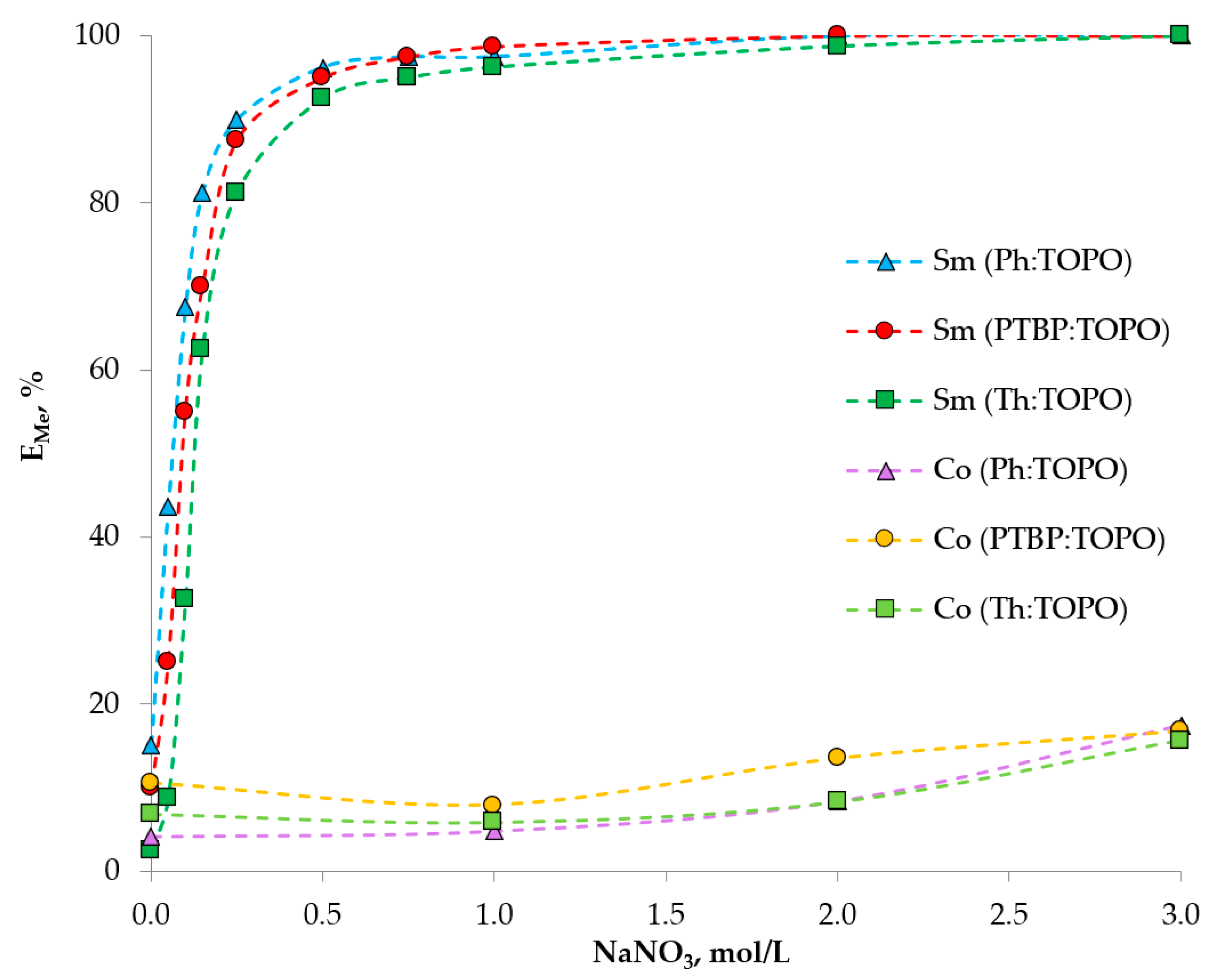
2. Results and Discussion
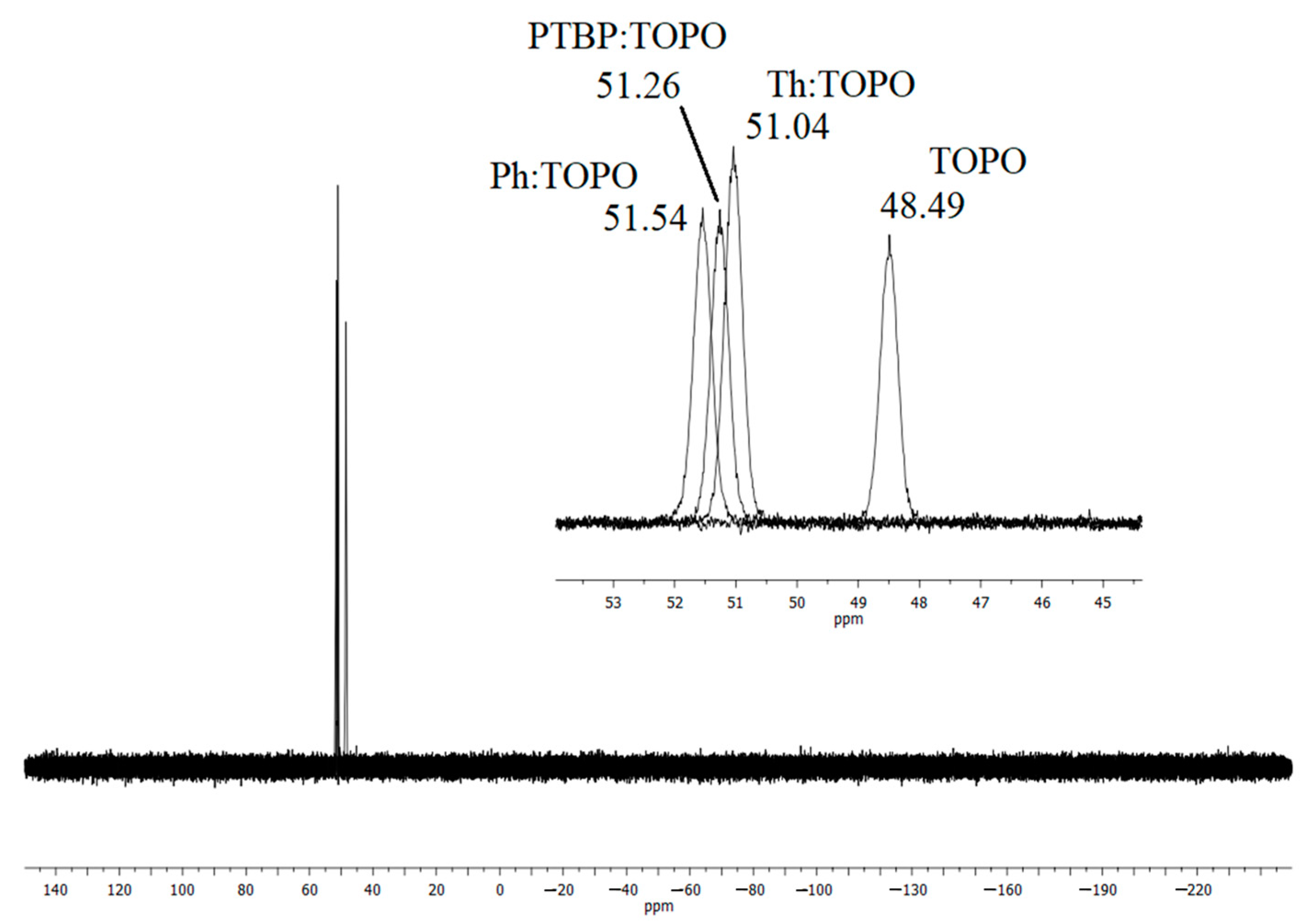
2.1. Characterization of HDES
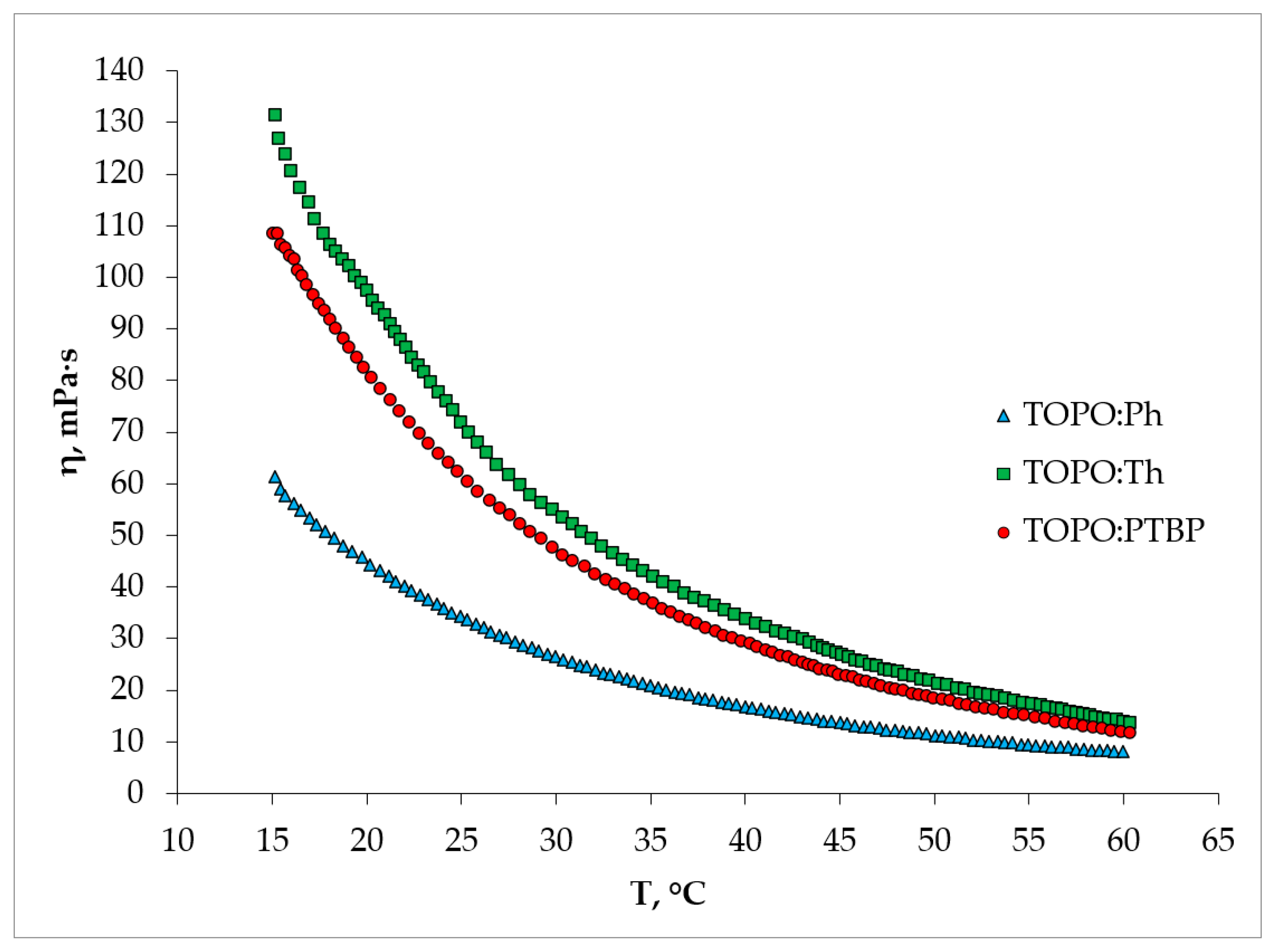
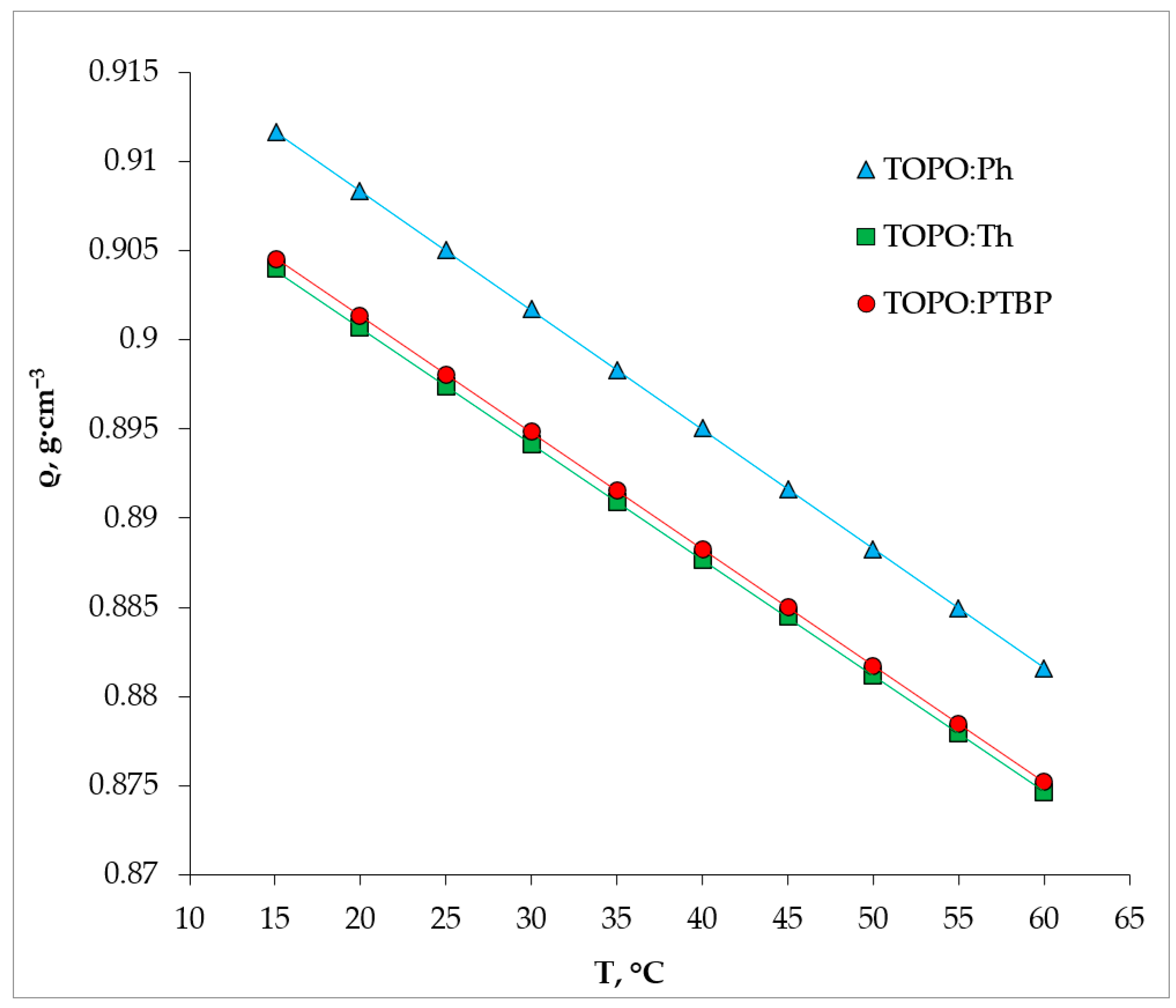
2.2. Physical Properties of HDES
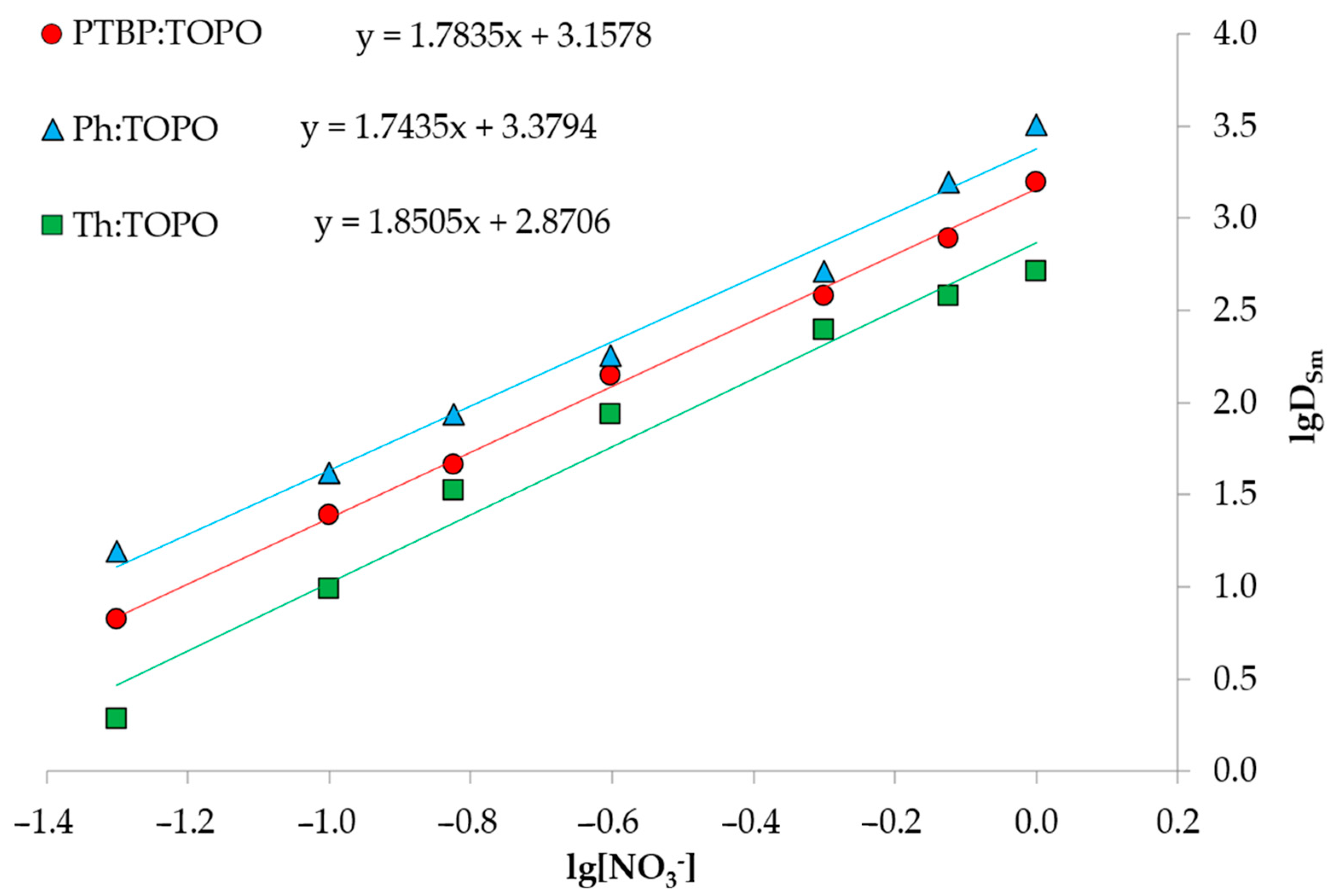
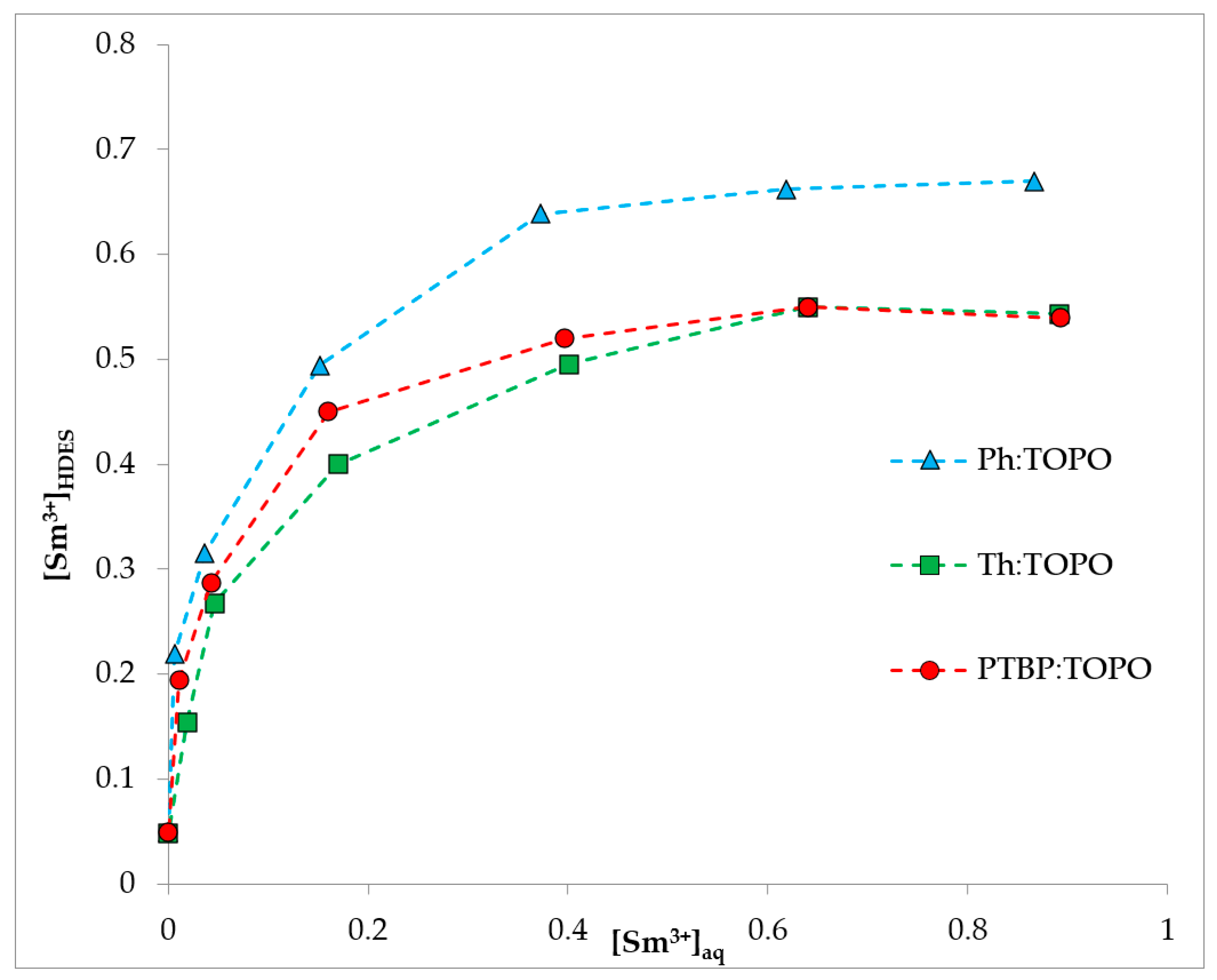
2.3. Extraction Experiment
3. Materials and Methods
3.1. Materials
3.2. Preparation of HDES
3.3. Characterization Methods of HDES
3.4. Metal Extraction
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yi, J.-H. Development of Samarium–Cobalt Rare Earth Permanent Magnetic Materials. Rare Met. 2014, 33, 633–640. [Google Scholar] [CrossRef]
- Budde, T.; Gatzen, H.H. Thin Film SmCo Magnets for Use in Electromagnetic Microactuators. J. Appl. Phys. 2006, 99, 08N304. [Google Scholar] [CrossRef]
- Binnemans, K.; Jones, P.T.; Blanpain, B.; Van Gerven, T.; Yang, Y.; Walton, A.; Buchert, M. Recycling of Rare Earths: A Critical Review. J. Clean. Prod. 2013, 51, 1–22. [Google Scholar] [CrossRef]
- Zakhodyaeva, Y.A.; Zinov’eva, I.V.; Tokar, E.S.; Voshkin, A.A. Complex Extraction of Metals in an Aqueous Two-Phase System Based on Poly(Ethylene Oxide) 1500 and Sodium Nitrate. Molecules 2019, 24, 4078. [Google Scholar] [CrossRef] [PubMed]
- Milevskiy, N.A.; Boryagina, I.V.; Karpukhina, E.A.; Kuznetsov, V.N.; Kabanova, E.G. Effect of Sodium Chloride and PH on the Composition of the Equilibrium Phases and the Partition of Palladium (II) in the Aqueous Two-Phase System PEG1500–Na2SO4–Water. J. Chem. Eng. Data 2021, 66, 1021–1031. [Google Scholar] [CrossRef]
- Quijada-Maldonado, E.; Romero, J. Solvent Extraction of Rare-Earth Elements with Ionic Liquids: Toward a Selective and Sustainable Extraction of These Valuable Elements. Curr. Opin. Green Sustain. Chem. 2021, 27, 100428. [Google Scholar] [CrossRef]
- Jiang, S.; Hu, Y.; Wang, Y.; Wang, X. Viscosity of Typical Room-Temperature Ionic Liquids: A Critical Review. J. Phys. Chem. Ref. Data 2019, 48, 033101. [Google Scholar] [CrossRef]
- Flieger, J.; Flieger, M. Ionic Liquids Toxicity—Benefits and Threats. Int. J. Mol. Sci. 2020, 21, 6267. [Google Scholar] [CrossRef]
- Zinov’eva, I.V.; Fedorov, A.Y.; Milevskii, N.A.; Zakhodyaeva, Y.A.; Voshkin, A.A. Dissolution of Metal Oxides in a Choline Chloride–Sulphosalicylic Acid Deep Eutectic Solvent. Theor. Found. Chem. Eng. 2021, 55, 663–670. [Google Scholar] [CrossRef]
- Shishov, A.; Gerasimov, A.; Bulatov, A. Deep Eutectic Solvents Based on Carboxylic Acids for Metals Separation from Plant Samples: Elemental Analysis by ICP-OES. Food Chem. 2022, 366, 130634. [Google Scholar] [CrossRef]
- Shishov, A.; Volodina, N.; Nechaeva, D.; Gagarinova, S.; Bulatov, A. An Automated Homogeneous Liquid-Liquid Microextraction Based on Deep Eutectic Solvent for the HPLC-UV Determination of Caffeine in Beverages. Microchem. J. 2019, 144, 469–473. [Google Scholar] [CrossRef]
- Yuan, Z.; Liu, H.; Yong, W.F.; She, Q.; Esteban, J. Status and Advances of Deep Eutectic Solvents for Metal Separation and Recovery. Green Chem. 2022, 24, 1895–1929. [Google Scholar] [CrossRef]
- Dwamena, A. Recent Advances in Hydrophobic Deep Eutectic Solvents for Extraction. Separations 2019, 6, 9. [Google Scholar] [CrossRef]
- Edgecomb, J.M.; Tereshatov, E.E.; Zante, G.; Boltoeva, M.; Folden Iii, C.M. Hydrophobic Amine-Based Binary Mixtures of Active Pharmaceutical and Food Grade Ingredients: Characterization and Application in Indium Extraction from Aqueous Hydrochloric Acid Media. Green Chem. 2020, 22, 7047–7058. [Google Scholar] [CrossRef]
- Hanada, T.; Goto, M. Synergistic Deep Eutectic Solvents for Lithium Extraction. ACS Sustain. Chem. Eng. 2021, 9, 2152–2160. [Google Scholar] [CrossRef]
- Shi, Y.; Xiong, D.; Zhao, Y.; Li, T.; Zhang, K.; Fan, J. Highly Efficient Extraction/Separation of Cr (VI) by a New Family of Hydrophobic Deep Eutectic Solvents. Chemosphere 2020, 241, 125082. [Google Scholar] [CrossRef]
- Milevskii, N.A.; Zinov’eva, I.V.; Zakhodyaeva, Y.A.; Voshkin, A.A. Separation of Li (I), Co (II), Ni (II), Mn (II), and Fe (III) from Hydrochloric Acid Solution Using a Menthol-Based Hydrophobic Deep Eutectic Solvent. Hydrometallurgy 2022, 207, 105777. [Google Scholar] [CrossRef]
- Mokhodoeva, O.; Maksimova, V.; Shishov, A.; Shkinev, V. Separation of Platinum Group Metals Using Deep Eutectic Solvents Based on Quaternary Ammonium Salts. Sep. Purif. Technol. 2023, 305, 122427. [Google Scholar] [CrossRef]
- Vargas, S.J.R.; Pérez-Sánchez, G.; Schaeffer, N.; Coutinho, J.A.P. Solvent Extraction in Extended Hydrogen Bonded Fluids—Separation of Pt (iv) from Pd (ii) Using TOPO-Based Type V DES. Green Chem. 2021, 23, 4540–4550. [Google Scholar] [CrossRef]
- Abranches, D.O.; Coutinho, J.A.P. Type V Deep Eutectic Solvents: Design and Applications. Curr. Opin. Green Sustain. Chem. 2022, 35, 100612. [Google Scholar] [CrossRef]
- Ni, S.; Su, J.; Zhang, H.; Zeng, Z.; Zhi, H.; Sun, X. A Cleaner Strategy for Comprehensive Recovery of Waste SmCo Magnets Based on Deep Eutectic Solvents. Chem. Eng. J. 2021, 412, 128602. [Google Scholar] [CrossRef]
- Liu, R.; Geng, Y.; Tian, Z.; Wang, N.; Wang, M.; Zhang, G.; Yang, Y. Extraction of Platinum (IV) by Hydrophobic Deep Eutectic Solvents Based on Trioctylphosphine Oxide. Hydrometallurgy 2021, 199, 105521. [Google Scholar] [CrossRef]
- Martins, M.A.R.; Crespo, E.A.; Pontes, P.V.A.; Silva, L.P.; Bülow, M.; Maximo, G.J.; Batista, E.A.C.; Held, C.; Pinho, S.P.; Coutinho, J.A.P. Tunable Hydrophobic Eutectic Solvents Based on Terpenes and Monocarboxylic Acids. ACS Sustain. Chem. Eng. 2018, 6, 8836–8846. [Google Scholar] [CrossRef]
- Crema, A.P.S.; Schaeffer, N.; Bastos, H.; Silva, L.P.; Abranches, D.O.; Passos, H.; Hespanhol, M.C.; Coutinho, J.A.P. New Family of Type V Eutectic Solvents Based on 1,10-Phenanthroline and Their Application in Metal Extraction. Hydrometallurgy 2023, 215, 105971. [Google Scholar] [CrossRef]
- Cao, J.; He, Y.; Zhang, X.; Shi, L.; Zeng, B.; Zhao, F. Development of a Tributyl Phosphate-Based Hydrophobic Deep Eutectic Solvent for Liquid-Liquid Microextraction of Total Parabens in Beverages. J. Mol. Liq. 2023, 387, 122593. [Google Scholar] [CrossRef]
- Kollau, L.J.B.M.; Vis, M.; Van Den Bruinhorst, A.; Esteves, A.C.C.; Tuinier, R. Quantification of the Liquid Window of Deep Eutectic Solvents. Chem. Commun. 2018, 54, 13351–13354. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, M.; McCourt, É.N.; Connolly, F.; Nockemann, P.; Swadźba-Kwaśny, M.; Holbrey, J.D. Hydrophobic Deep Eutectic Solvents Incorporating Trioctylphosphine Oxide: Advanced Liquid Extractants. ACS Sustain. Chem. Eng. 2018, 6, 17323–17332. [Google Scholar] [CrossRef]
- Schaeffer, N.; Conceição, J.H.F.; Martins, M.A.R.; Neves, M.C.; Pérez-Sánchez, G.; Gomes, J.R.B.; Papaiconomou, N.; Coutinho, J.A.P. Non-Ionic Hydrophobic Eutectics—Versatile Solvents for Tailored Metal Separation and Valorisation. Green Chem. 2020, 22, 2810–2820. [Google Scholar] [CrossRef]
- Frisch, M.; Trucks, G.; Schlegel, H.; Scuseria, G.; Robb, M.; Cheeseman, J.; Scalmani, G.; Barone, V.; Petersson, G.; Nakatsuji, H.; et al. Gaussian 09, Revision C.01; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305. [Google Scholar] [CrossRef] [PubMed]
- Medvedev, M.G.; Bushmarinov, I.S.; Sun, J.; Perdew, J.P.; Lyssenko, K.A. Density functional theory is straying from the path toward the exact functional. Science 2017, 355, 49–52. [Google Scholar] [CrossRef]
- Mardirossian, N.; Head-Gordon, M. Thirty years of density functional theory in computational chemistry: An overview and extensive assessment of 200 density functionals. Mol. Phys. 2017, 115, 2315–2372. [Google Scholar] [CrossRef]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 2010, 132, 154104. [Google Scholar] [CrossRef]
- Boys, S.F.; Bernardi, F. The calculation of small molecular interactions by the differences of separate total energies. Some procedures with reduced errors. Mol. Phys. 1970, 19, 553–566. [Google Scholar] [CrossRef]
- Omar, K.A.; Sadeghi, R. Physicochemical Properties of Deep Eutectic Solvents: A Review. J. Mol. Liq. 2022, 360, 119524. [Google Scholar] [CrossRef]
- Francesconi, R.; Ottani, S. Correlation of Density and Refraction Index for Liquid Binary Mixtures Containing Polyglycols. Use of the Group Contributions in the Lorentz–Lorenz, Gladstone–Dale and Vogel Equations to Evaluate the Density of Mixtures. J. Mol. Liq. 2007, 133, 125–133. [Google Scholar] [CrossRef]
- Rho, B.-J.; Sun, P.-P.; Cho, S.-Y. Recovery of Neodymium and Praseodymium from Nitrate-Based Leachate of Permanent Magnet by Solvent Extraction with Trioctylphosphine Oxide. Sep. Purif. Technol. 2020, 238, 116429. [Google Scholar] [CrossRef]
- Swain, N.; Mishra, S.; Acharya, M.R. Hydrometallurgical Route for Recovery and Separation of Samarium (III) and Cobalt (II) from Simulated Waste Solution Using Tri-n-Octyl Phosphine Oxide—A Novel Pathway for Synthesis of Samarium and Cobalt Oxides Nanoparticles. J. Alloys Compd. 2020, 815, 152423. [Google Scholar] [CrossRef]
- El-Nadi, Y.A. Lanthanum and Neodymium from Egyptian Monazite: Synergistic Extractive Separation Using Organophosphorus Reagents. Hydrometallurgy 2012, 119–120, 23–29. [Google Scholar] [CrossRef]
- Suresh, G.; Murali, M.S.; Mathur, J.N. Thermodynamics of Extraction of Am (III) and Eu (III) from Different Anionic Media with Tri-n-Octyl Phosphine Oxide. Radiochim. Acta 2003, 91, 127–134. [Google Scholar] [CrossRef]
- El-Hefny, N.E.; El-Nadi, Y.A.; Daoud, J.A. Equilibrium and Mechanism of Samarium Extraction from Chloride Medium Using Sodium Salt of CYANEX 272. Sep. Purif. Technol. 2010, 75, 310–315. [Google Scholar] [CrossRef]
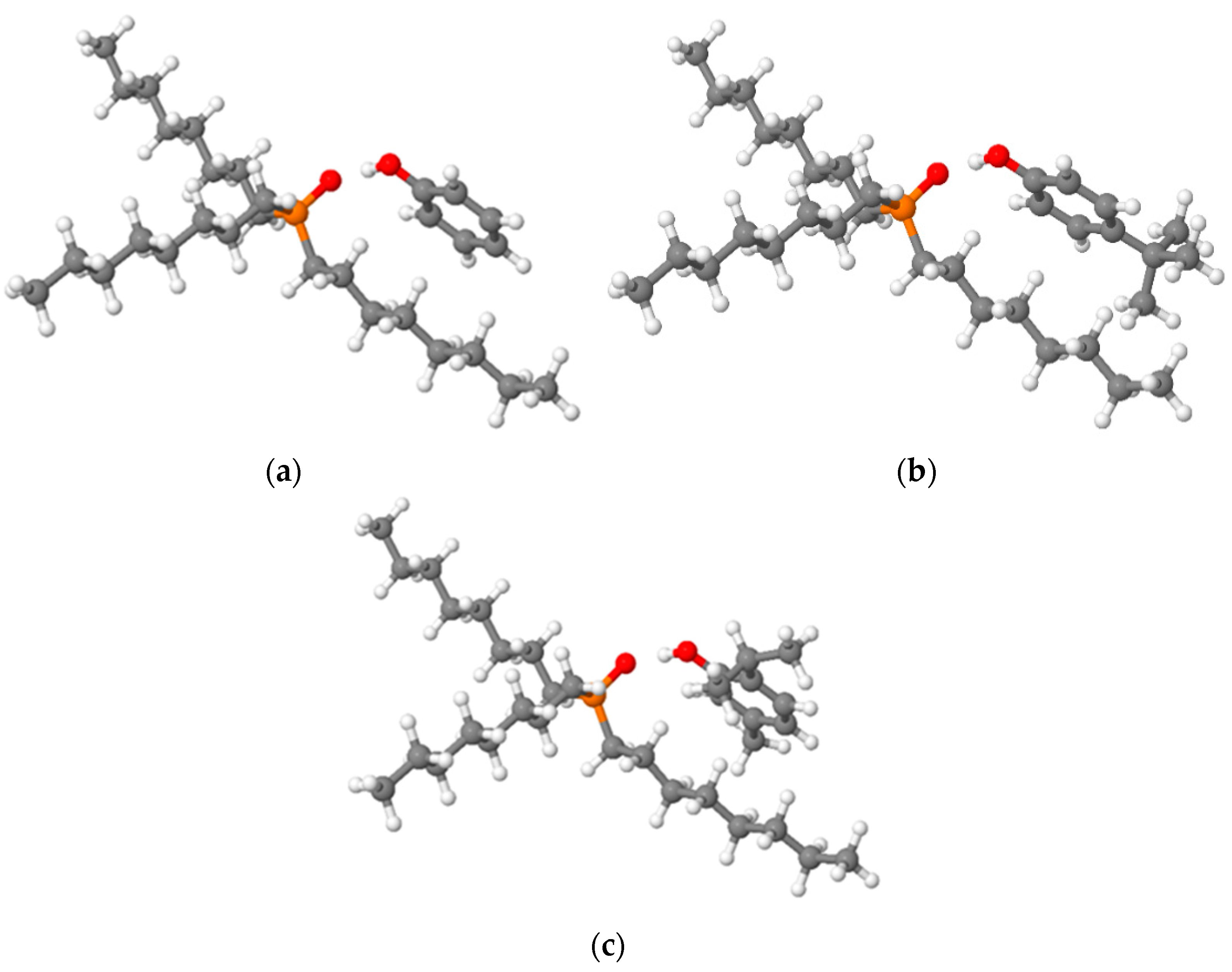
| HDES | O…H, Å | qNBO (OTOPO) | ΔEcomplexBSSE, kcal/mol |
|---|---|---|---|
| Ph:TOPO | 1.632 | −1.088 | −19.73 |
| PTBP:TOPO | 1.641 | −1.087 | −20.60 |
| Th:TOPO | 1.639 | −1.086 | −21.52 |
| T, °C | Ph:TOPO | PTBP:TOPO | Th:TOPO |
|---|---|---|---|
| 15 | 1.4827 | 1.4829 | 1.4832 |
| 25 | 1.479 | 1.4792 | 1.4795 |
| 35 | 1.4752 | 1.4756 | 1.4758 |
| 45 | 1.4715 | 1.4718 | 1.4721 |
| 55 | 1.4679 | 1.4683 | 1.4685 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Milevskii, N.A.; Zinov’eva, I.V.; Kozhevnikova, A.V.; Zakhodyaeva, Y.A.; Voshkin, A.A. Sm/Co Magnetic Materials: A Recycling Strategy Using Modifiable Hydrophobic Deep Eutectic Solvents Based on Trioctylphosphine Oxide. Int. J. Mol. Sci. 2023, 24, 14032. https://doi.org/10.3390/ijms241814032
Milevskii NA, Zinov’eva IV, Kozhevnikova AV, Zakhodyaeva YA, Voshkin AA. Sm/Co Magnetic Materials: A Recycling Strategy Using Modifiable Hydrophobic Deep Eutectic Solvents Based on Trioctylphosphine Oxide. International Journal of Molecular Sciences. 2023; 24(18):14032. https://doi.org/10.3390/ijms241814032
Chicago/Turabian StyleMilevskii, Nikita A., Inna V. Zinov’eva, Arina V. Kozhevnikova, Yulia A. Zakhodyaeva, and Andrey A. Voshkin. 2023. "Sm/Co Magnetic Materials: A Recycling Strategy Using Modifiable Hydrophobic Deep Eutectic Solvents Based on Trioctylphosphine Oxide" International Journal of Molecular Sciences 24, no. 18: 14032. https://doi.org/10.3390/ijms241814032
APA StyleMilevskii, N. A., Zinov’eva, I. V., Kozhevnikova, A. V., Zakhodyaeva, Y. A., & Voshkin, A. A. (2023). Sm/Co Magnetic Materials: A Recycling Strategy Using Modifiable Hydrophobic Deep Eutectic Solvents Based on Trioctylphosphine Oxide. International Journal of Molecular Sciences, 24(18), 14032. https://doi.org/10.3390/ijms241814032








