New Ethynylphenylborasilsesquioxanes—Their Reactivity and Behavior during Thermal Decomposition
Abstract
:1. Introduction
2. Results and Discussion
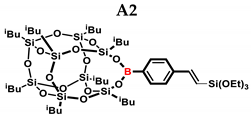
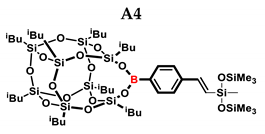
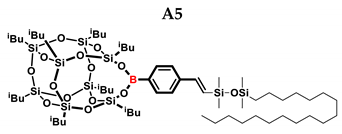
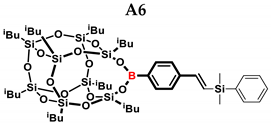
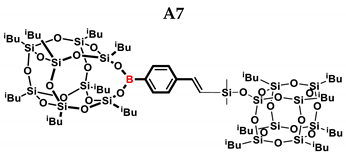
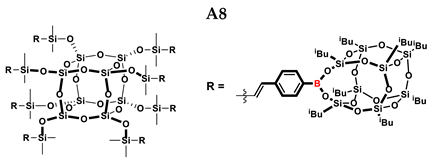
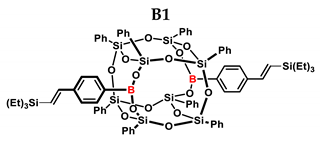
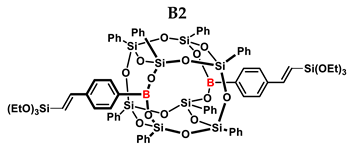
2.1. Reactivity Tests
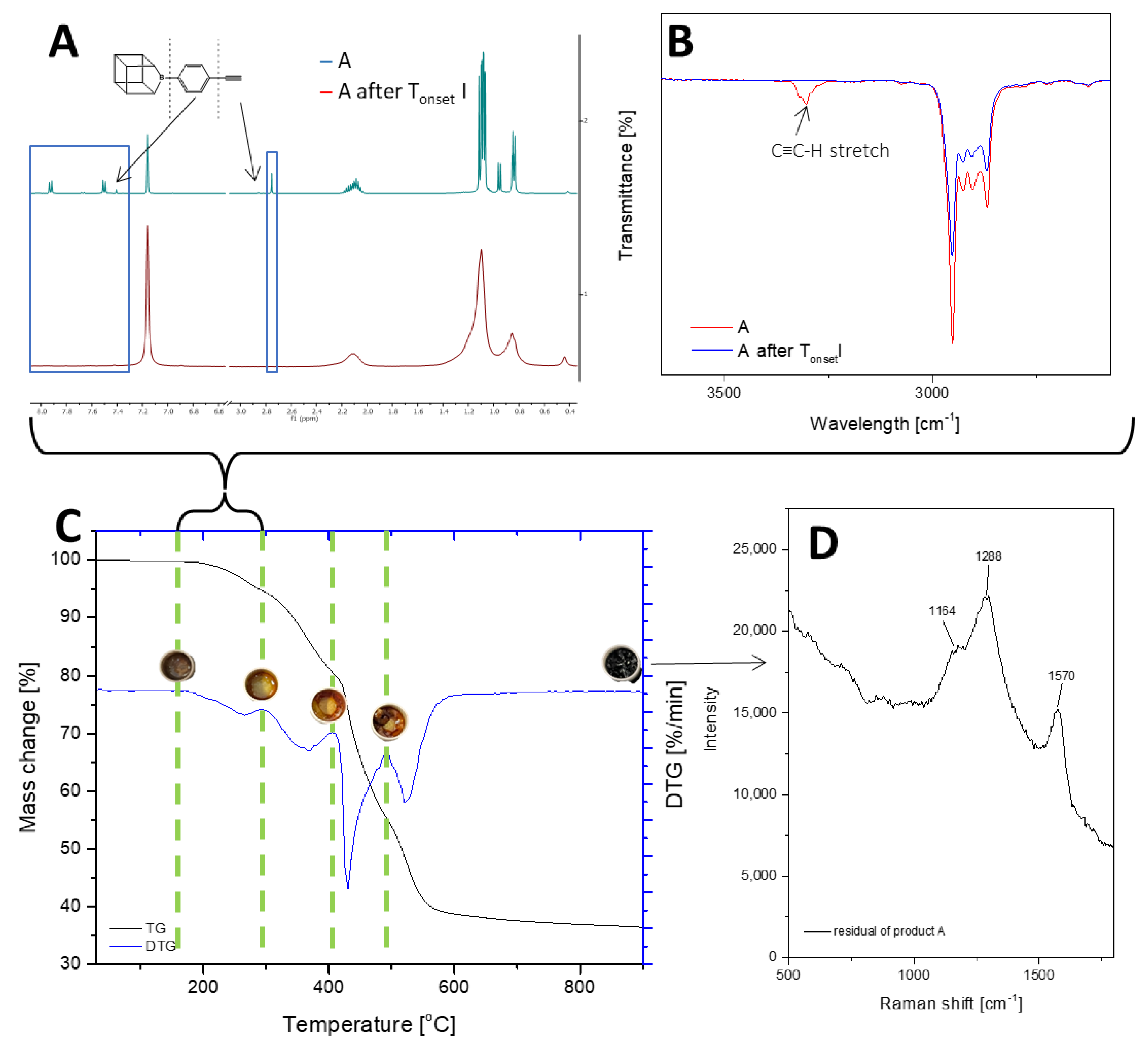
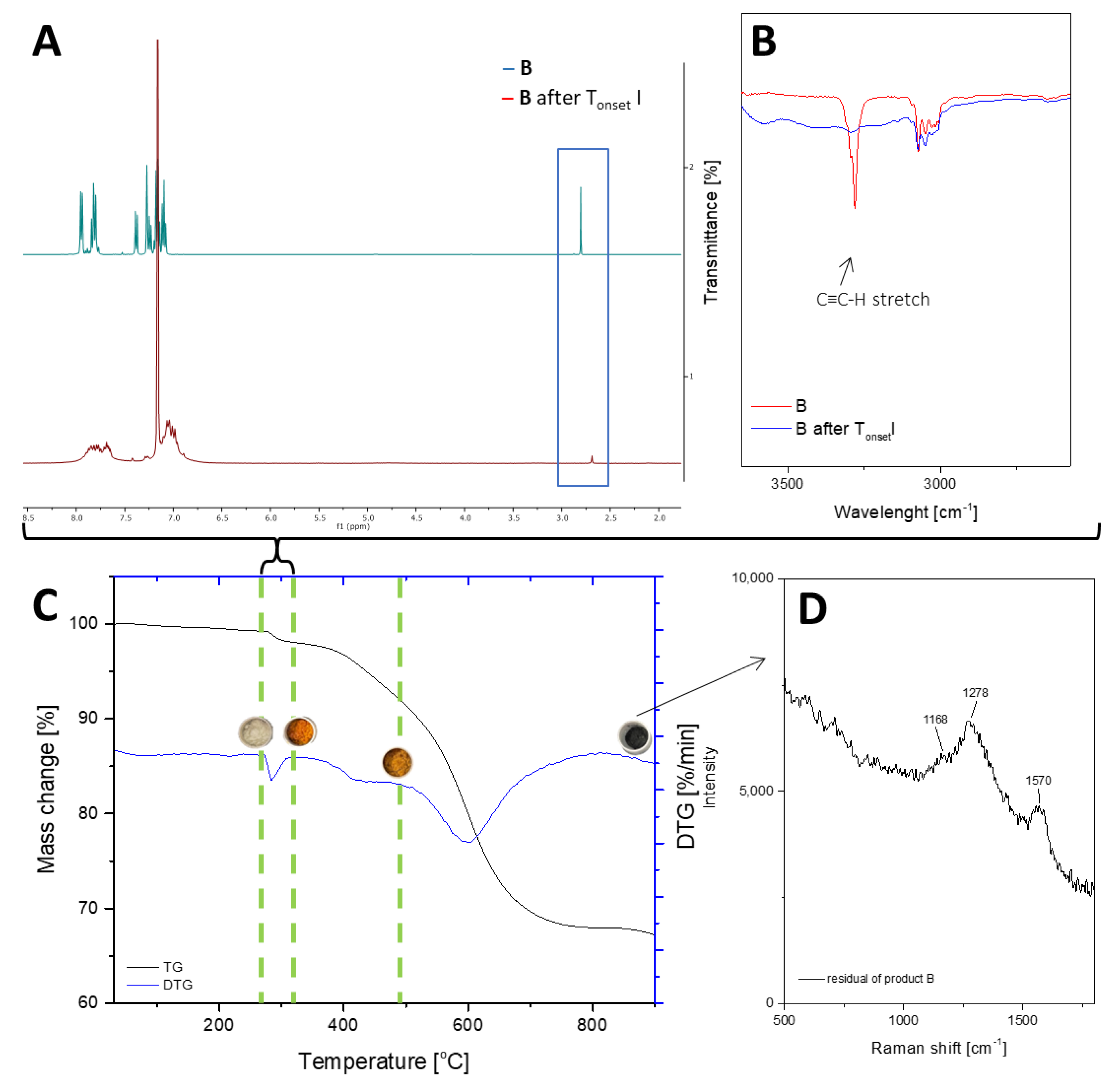
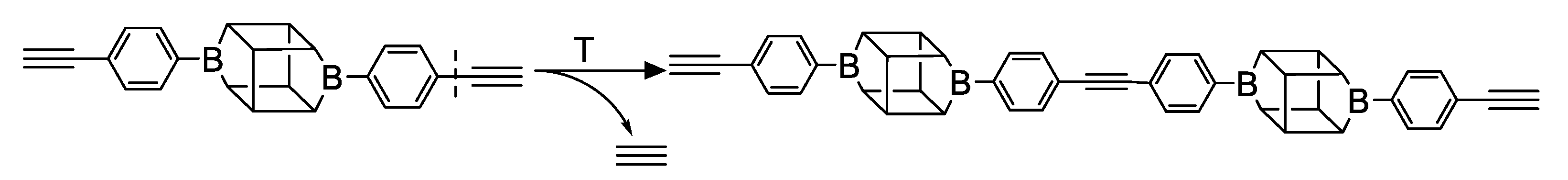
2.2. Thermal Decomposition Analysis
3. Materials and Methods
3.1. Materials
3.2. Synthesis of Organosilicon Precursors
3.2.1. General Procedure for Hydrosilylation of Ethynylphenylborasilsesquioxane with Compounds Bearing Si–H Moiety
3.2.2. General Procedure for Hydrosilylation of Diethynylphenylborasilsesquioxane with Compounds Bearing Si–H Moiety
3.3. NMR Spectroscopy Analysis




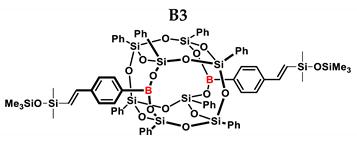
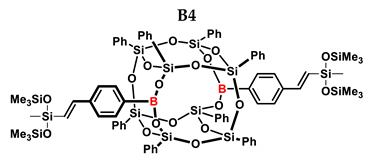
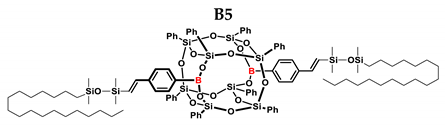
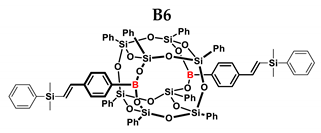

3.4. Analytical Methods
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, C.; Babonneau, F.; Bonhomme, C.; Laine, R.M.; Soles, C.L.; Hristov, H.; Yee, A.F. Highly Porous Polyhedral Silsesquioxane Polymers. Synthesis and Characterization. J. Am. Chem. Soc. 1998, 120, 8380–8391. [Google Scholar] [CrossRef]
- Voronkov, M.G.; Lavrent’yev, V.I. Polyhedral oligosilsesquioxanes and their homo derivatives. In Inorganic Ring Systems; Topics in Current Chemistry; Springer: Berlin/Heidelberg, Germany, 1982; Volume 102. [Google Scholar] [CrossRef]
- Edelmann, F.T. Advances in Organometallic Chemistry; Elsevier: Amsterdam, The Netherlands, 2008; Volume 55, pp. 1–329. [Google Scholar]
- Li, G.; Wang, L.; Ni, H.; Pittman, C.U., Jr. Polyhedral Oligomeric Silsesquioxane (POSS) Polymers and Copolymers: A Review. J. Inorg. Organomet. Polym. 2001, 11, 123–154. [Google Scholar] [CrossRef]
- Sztorch, B.; Nowak, K.; Frydrych, M.; Leśniewska, J.; Krysiak, K.; Przekop, R.E.; Olejnik, A. Improving the Dispersibility of TiO2 in the Colloidal System Using Trifunctional Spherosilicates. Materials 2023, 16, 1442. [Google Scholar] [CrossRef]
- Brząkalski, D.; Przekop, R.E.; Sztorch, B.; Frydrych, M.; Pakuła, D.; Jałbrzykowski, M.; Markiewicz, G.; Marciniec, B. Why POSS-Type Compounds Should Be Considered Nanomodifiers, Not Nanofillers—A Polypropylene Blends Case Study. Polymers 2021, 13, 2124. [Google Scholar] [CrossRef] [PubMed]
- Brząkalski, D.; Przekop, R.E.; Sztorch, B.; Jakubowska, P.; Jałbrzykowski, M.; Marciniec, B. Silsesquioxane Derivatives as Functional Additives for Preparation of Polyethylene-Based Composites: A Case of Trisilanol Melt-Condensation. Polymers 2020, 12, 2269. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Chi, H.; Joshy, K.S.; Wang, F. Progress in the Synthesis of Bifunctionalized Polyhedral Oligomeric Silsesquioxane. Polymers 2019, 11, 2098. [Google Scholar] [CrossRef] [PubMed]
- Thankachan, A.P.; Afsina, C.M.A.; Shamna, S.; Anilkumar, G. Recent Advances in Ruthenium-Catalyzed Hydrosilylation of Unsaturated Compounds: Applications and Mechanistic Studies. Results Chem. 2022, 4, 100511. [Google Scholar] [CrossRef]
- Zhao, B.; Hang, G.; Li, L.; Zheng, S. Nanocomposites of Polyethylene with Polyhedral Oligomeric Silsesquioxane: From Thermoplastics to Vitrimers through Silyl Ether Metathesis. Mater. Today Chem. 2022, 24, 100759. [Google Scholar] [CrossRef]
- Żak, P.; Pietraszuk, C. Application of Olefin Metathesis in the Synthesis of Functionalized Polyhedral Oligomeric Silsesquioxanes (POSS) and POSS-Containing Polymeric Materials. Beilstein J. Org. Chem. 2019, 15, 310–332. [Google Scholar] [CrossRef] [PubMed]
- Żak, P.; Dudziec, B.; Kubicki, M.; Marciniec, B. Silylative Coupling versus Metathesis-Efficient Methods for the Synthesis of Difunctionalized Double-Decker Silsesquioxane Derivatives. Chem. A Eur. J. 2014, 20, 9387–9393. [Google Scholar] [CrossRef]
- Kannan, R.Y.; Salacinski, H.J.; Butler, P.E.M.; Seifalian, A.M. Polyhedral Oligomeric Silsesquioxane Nanocomposites: The next Generation Material for Biomedical Applications. Acc. Chem. Res. 2005, 38, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.; Lu, L.; Ha, C.-S. Silsesquioxane-Containing Hybrid Nanomaterials: Fascinating Platforms for Advanced Applications. Macromol. Chem. Phys. 2019, 220, 1800324. [Google Scholar] [CrossRef]
- Zhou, H.; Ye, Q.; Xu, J. Polyhedral Oligomeric Silsesquioxane-Based Hybrid Materials and Their Applications. Mater. Chem. Front. 2017, 1, 212–230. [Google Scholar] [CrossRef]
- Duchateau, R. Incompletely Condensed Silsesquioxanes: Versatile Tools in Developing Silica-Supported Olefin Polymerization Catalysts. Chem. Rev. 2002, 102, 3525–3542. [Google Scholar] [CrossRef]
- Bivona, L.A.; Fichera, O.; Fusaro, L.; Giacalone, F.; Buaki-Sogo, M.; Gruttadauria, M.; Aprile, C. A Polyhedral Oligomeric Silsesquioxane-Based Catalyst for the Efficient Synthesis of Cyclic Carbonates. Catal. Sci. Technol. 2015, 5, 5000–5007. [Google Scholar] [CrossRef]
- Seidi, F.; Jouyandeh, M.; Taghizadeh, A.; Taghizadeh, M.; Habibzadeh, S.; Jin, Y.; Xiao, H.; Zarrintaj, P.; Saeb, M.R. Polyhedral Oligomeric Silsesquioxane/Epoxy Coatings: A Review. Surf. Innov. 2021, 9, 3–16. [Google Scholar] [CrossRef]
- Zhang, W.; Camino, G. Polymer/Polyhedral Oligomeric Silsesquioxane (POSS) Nanocomposites: An Overview of Fire Retardance. Prog. Polym. Sci. 2017, 67, 77–125. [Google Scholar] [CrossRef]
- Chen, H.; Jiao, X.; Zhou, J.-T. The Research Progress of Polyhedral Oligomeric Silsesquioxane (POSS) Applied to Electrical Energy Storage Elements. Funct. Mater. Lett. 2017, 10, 1730001. [Google Scholar] [CrossRef]
- Sun, J.; Chen, Y.; Zhao, L.; Chen, Y.; Qi, D.; Choi, K.; Shin, D.-S.; Jiang, J. Porphyrin-POSS Molecular Hybrids. Chem. Eur. J. 2013, 19, 12613–12618. [Google Scholar] [CrossRef] [PubMed]
- Chechelska-Noworyta, A.; Mrówka, J.; Owińska, M.; Hasik, M. Application of Spectroscopic Methods in the Studies of Polysiloxanes, Cubic Oligomeric Silsesquioxanes, and Spherosilicates Modified by Organic Functional Groups via Hydrosilylation; Springer eBooks: Berlin/Heidelberg, Germany, 2018; pp. 361–400. [Google Scholar]
- Soldatov, M.A.; Liu, H. Hybrid Porous Polymers Based on Cage-like Organosiloxanes: Synthesis, Properties and Applications. Prog. Polym. Sci. 2021, 119, 101419. [Google Scholar] [CrossRef]
- Du, Y.; Liu, H. Cage-like Silsesquioxanes-Based Hybrid Materials. Dalton Trans. 2020, 49, 5396–5405. [Google Scholar] [CrossRef] [PubMed]
- Hartmann-Thompson, C. (Ed.) Applications of Polyhedral Oligomeric Silsesquioxanes; Advances in Silicon Science; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2011. [Google Scholar] [CrossRef]
- Białek, M.; Czaja, K. Application of Silsesquioxanes in the Preparation of Polyolefin-Based Materials. Materials 2023, 16, 1876. [Google Scholar] [CrossRef]
- Kannan, A.; Muthuraj, C.; Mayavan, A.; Gandhi, S. Multifaceted Applications of Polyhedral Oligomeric Silsesquioxane and Their Composites. Mater. Today Chem. 2023, 30, 101568. [Google Scholar] [CrossRef]
- Zhang, K.; Huang, S.; Zhang, Q.; Zhu, H.; Zhu, S. Polyhedral Oligomeric Silsesquioxane-based Functional Coatings: A Review. Can. J. Chem. Eng. 2023, 101, 4979–4991. [Google Scholar] [CrossRef]
- Feher, F.J.; Soulivong, D.; Nguyen, F.; Ziller, J.W. A New Route to Heterosilsesquioxane Frameworks. Angew. Chem. Int. Ed. 1998, 37, 2663–2666. [Google Scholar] [CrossRef]
- Feher, F.J.; Newman, D.A.; Walzer, J.F. Silsesquioxanes as Models for Silica Surfaces. J. Am. Chem. Soc. 1989, 111, 1741–1748. [Google Scholar] [CrossRef]
- Fina, A.; Abbenhuis, H.C.L.; Tabuani, D.; Frache, A.; Camino, G. Polypropylene Metal Functionalised POSS Nanocomposites: A Study by Thermogravimetric Analysis. Polym. Degrad. Stab. 2006, 91, 1064–1070. [Google Scholar] [CrossRef]
- Carniato, F.; Boccaleri, E.; Marchese, L.; Fina, A.; Tabuani, D.; Camino, G. Synthesis and Characterisation of Metal Isobutylsilsesquioxanes and Their Role as Inorganic–Organic Nanoadditives for Enhancing Polymer Thermal Stability. Eur. J. Inorg. Chem. 2007, 2007, 585–591. [Google Scholar] [CrossRef]
- Garg, S.; Unruh, D.K.; Krempner, C. Zirconium and Hafnium Polyhedral Oligosilsesquioxane Complexes—Green Homogeneous Catalysts in the Formation of Bio-Derived Ethers via a MPV/Etherification Reaction Cascade. Catal. Sci. Technol. 2021, 11, 211–218. [Google Scholar] [CrossRef]
- Feher, F.J.; Budzichowski, T.A.; Ziller, J.W. Synthesis, Reactivity, and Dynamic Behavior of a Boron-Containing Silsesquioxane. Inorg. Chem. 1992, 31, 5100–5105. [Google Scholar] [CrossRef]
- Gerritsen, G.; Duchateau, R.; Santen, V.R.R.; Yap, G. Boron, Aluminum, and Gallium Silsesquioxane Compounds, Homogeneous Models for Group 13 Element-Containing Silicates and Zeolites. Organometallics 2002, 22, 100–110. [Google Scholar] [CrossRef]
- Barry, B.-D.; Dannatt, J.E.; King, A.K.; Lee, A.; Maleczka, R.E. A General Diversity Oriented Synthesis of Asymmetric Double-Decker Shaped Silsesquioxanes. Chem. Commun. 2019, 55, 8623–8626. [Google Scholar] [CrossRef]
- Frydrych, M.; Sztorch, B.; Brząkalski, D.; Pakuła, D.; Frąckowiak, D.; Majchrzak, M.; Marciniec, B. New Functionalized Borasilsesquioxanes Obtained via Metathetical Transformation of 4-Vinylphenylborasilsesquioxanes. Eur. J. Inorg. Chem. 2020, 2020, 3597–3600. [Google Scholar] [CrossRef]
- Frydrych, M.; Sztorch, B.; Brząkalski, D.; Pakuła, D.; Przekop, R.E.; Srishailam, K.; Reddy, B.V.; Marciniec, B. Silane-Bearing Borasilsesquioxanes: Synthetic Protocol and Unsuspected Redistribution Reactions. Collect. Czechoslov. Chem. Commun. 2023, 88, e202200295. [Google Scholar] [CrossRef] [PubMed]
- Brząkalski, D.; Przekop, R.E.; Dobrosielska, M.; Sztorch, B.; Marciniak, P.; Marciniec, B. Highly Bulky Spherosilicates as Functional Additives for Polyethylene Processing—Influence on Mechanical and Thermal Properties. Polym. Compos. 2020, 41, 3389–3402. [Google Scholar] [CrossRef]
- Pakuła, D.; Przekop, R.E.; Brząkalski, D.; Frydrych, M.; Sztorch, B.; Marciniec, B. Sulfur-Containing Silsesquioxane Derivatives Obtained by the Thiol-ene Reaction: Synthesis and Thermal Degradation. Collect. Czechoslov. Chem. Commun. 2022, 87, e202200099. [Google Scholar] [CrossRef] [PubMed]
- Frydrych, M.; Pakuła, D.; Sztorch, B.; Brząkalski, D.; Przekop, R.E.; Marciniec, B. Novel Silsesquioxane-Derived Boronate Esters—Synthesis and Thermal Properties. Molecules 2021, 26, 4107. [Google Scholar] [CrossRef]
- Skoczeń, A.; Frąckowiak, D.; Przekop, R.E.; Frydrych, M.; Kasperkowiak, M.; Jeleń, P.; Sitarz, M.; Marciniec, B. New Ceramics Precursors Containing SI and GE Atoms—Cubic Germasilsesquioxanes—Synthesis, Thermal Decomposition and Spectroscopic Analysis. Molecules 2022, 27, 1441. [Google Scholar] [CrossRef]
- Pan, Z.; Liu, M.; Zheng, C.; Gao, D.; Huang, W. Study of Karstedt’s Catalyst for Hydrosilylation of a Wide Variety of Functionalized Alkenes with Triethoxysilane and Trimethoxysilane. Chin. J. Chem. 2017, 35, 1227–1230. [Google Scholar] [CrossRef]
- Peng, L.; Xu, F.; Shinohara, K.; Nishida, T.; Wakamatsu, K.; Orita, A.; Otera, J. Remarkable Electron-Withdrawing Effect of the Ph2P(O)-Ethynyl Group: Ph2P(O)-Ethynyl-Substituted Aryl Halides and Copper Acetylides for Tailor-Made Sonogashira Couplings. Org. Chem. Front. 2015, 2, 248–252. [Google Scholar] [CrossRef]
- Oliveira, C.; Galindo, R.E.; Palacio, C.; Calderon, S.; Almeida, B.G.; Henriques, M.; Espinosa, A.; Carvalho, S. Surface Characterization of Ti-Si-C-ON Coatings for Orthopedic Devices: XPS and Raman Spectroscopy. Solid State Sci. 2011, 13, 95–100. [Google Scholar] [CrossRef]
- Liang, E.; Zhang, J.; Leme, J.; Moura, C.; Cunha, L. Raman Analysis of Si–C–N Films Grown by Reactive Magnetron Sputtering. Thin Solid Films 2004, 469–470, 410–415. [Google Scholar] [CrossRef]
- Hashimoto, H.; Koyama, Y. The C C Stretching Raman Lines of [β-Carotene Isomers in the S1 State as Detected by Pump-Probe Resonance Raman Spectroscopy. Chem. Phys. Lett. 1989, 154, 321–325. [Google Scholar] [CrossRef]
- Moore, L.M.J.; Zavala, J.J.; Lamb, J.T.; Reams, J.T.; Yandek, G.R.; Guenthner, A.J.; Haddad, T.S.; Ghiassi, K.B. Bis-Phenylethynyl Polyhedral Oligomeric Silsesquioxanes: New High-Temperature, Processable Thermosetting Materials. RSC Adv. 2018, 8, 27400–27405. [Google Scholar] [CrossRef] [PubMed]
- Fina, A.; Tabuani, D.; Carniato, F.; Frache, A.; Boccaleri, E.; Camino, G. Polyhedral Oligomeric Silsesquioxanes (POSS) Thermal Degradation. Thermochim. Acta 2006, 440, 36–42. [Google Scholar] [CrossRef]
- Xu, Y.; Ma, Y.; Deng, Y.; Yang, C.; Chen, J.; Dai, L. Morphology and Thermal Properties of Organic–Inorganic Hybrid Material Involving Monofunctional-Anhydride POSS and Epoxy Resin. Mater. Chem. Phys. 2011, 125, 174–183. [Google Scholar] [CrossRef]
- Filho, N.L.D.; De Aquino, H.A.; Pires, G.; Caetano, L. Relationship between the Dielectric and Mechanical Properties and the Ratio of Epoxy Resin to Hardener of the Hybrid Thermosetting Polymers. J. Braz. Chem. Soc. 2006, 17, 533–541. [Google Scholar] [CrossRef]
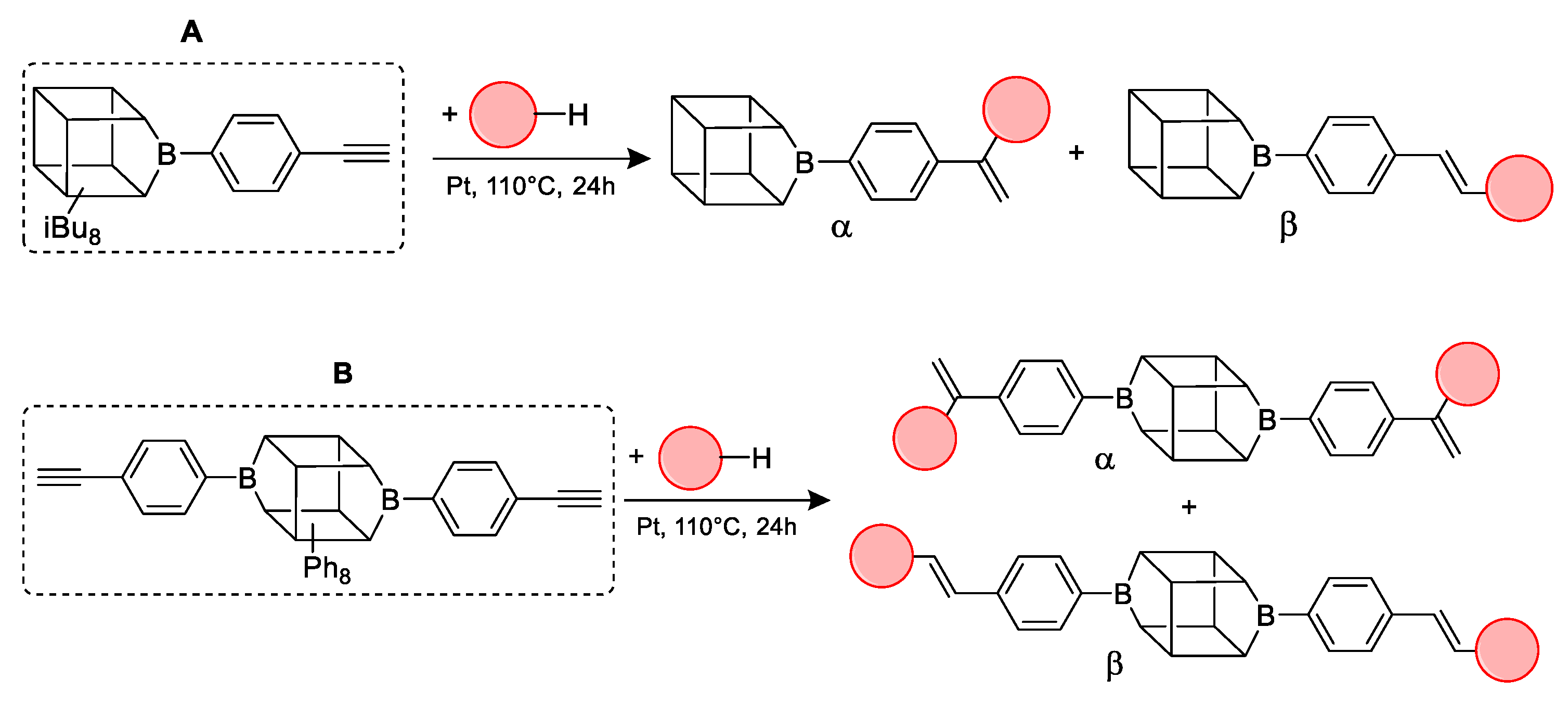


| Entry | Silane Compound | Molar Ratio [a] | Conversion/Yield [b] | α:β Ratio [b] |
|---|---|---|---|---|
| A1 | Et3SiH | 1:1 | 99% | 34:66 |
| A2 | (EtO)3SiH | 1:1 | ~65% | ~62:38 |
| A3 | PMDS | 1:1 | 84% | 43:57 |
| 1:1.5 | 99% | |||
| A4 | HMTS | 1:1 | 99% | 52:48 |
| A5 | Me2PhSiH | 1:1 | 99% | 29:71 |
| A6 | TMDS-OD | 1:1 | 80% | 47:53 |
| 1:1.5 | 99% | |||
| A7 | SSQ-OSiH | 1:1 | 65% | 55:45 |
| A8 | SS-8H | 8:1 | 77% | 4:96 |
| Entry | Silane Compound | Molar Ratio [a] | Conversion/Yield [b] | α:β Ratio [b] |
|---|---|---|---|---|
| B1 | Et3SiH | 1:1 | 97% | 29:71 |
| B2 | (EtO)3SiH | 1:1 | ~99% | - |
| B3 | PMDS | 1:1 | 98% | 40:60 |
| B4 | HMTS | 1:1 | 99% | 37:63 |
| B5 | Me2PhSiH | 1:1 | 99% | 25:75 |
| B6 | TMDS-OD | 1:1 | 90% | 44:56 |
| 1:1.5 | 99% | |||
| B7 | SSQ-OSiH | 1:1 | 92% | 31:69 |
| Sample | 1% Mass Loss (°C) T1% | Onset Temperature (°C) Tonset | Temperature at the Maximum Rate of Mass Loss (°C) Tmax | ||||||
|---|---|---|---|---|---|---|---|---|---|
| - | I | II | III | IV | I | II | III | IV | |
| A | 216.2 | 224.2 | 330.0 | 422.6 | 512.4 | 266.8 | 365.8 | 429 | 521.8 |
| B | 283.1 | 278.1 | 389.0 | 504.4 | 560.5 | 284.4 | 441.5 | 602.1 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frydrych, M.; Sztorch, B.; Przekop, R.E.; Marciniec, B. New Ethynylphenylborasilsesquioxanes—Their Reactivity and Behavior during Thermal Decomposition. Int. J. Mol. Sci. 2023, 24, 13960. https://doi.org/10.3390/ijms241813960
Frydrych M, Sztorch B, Przekop RE, Marciniec B. New Ethynylphenylborasilsesquioxanes—Their Reactivity and Behavior during Thermal Decomposition. International Journal of Molecular Sciences. 2023; 24(18):13960. https://doi.org/10.3390/ijms241813960
Chicago/Turabian StyleFrydrych, Miłosz, Bogna Sztorch, Robert E. Przekop, and Bogdan Marciniec. 2023. "New Ethynylphenylborasilsesquioxanes—Their Reactivity and Behavior during Thermal Decomposition" International Journal of Molecular Sciences 24, no. 18: 13960. https://doi.org/10.3390/ijms241813960
APA StyleFrydrych, M., Sztorch, B., Przekop, R. E., & Marciniec, B. (2023). New Ethynylphenylborasilsesquioxanes—Their Reactivity and Behavior during Thermal Decomposition. International Journal of Molecular Sciences, 24(18), 13960. https://doi.org/10.3390/ijms241813960








