Development, Establishment, and Validation of a Model for the Mineralization of Periodontium Remodelling Cells: Cementoblasts
Abstract
1. Introduction
2. Results
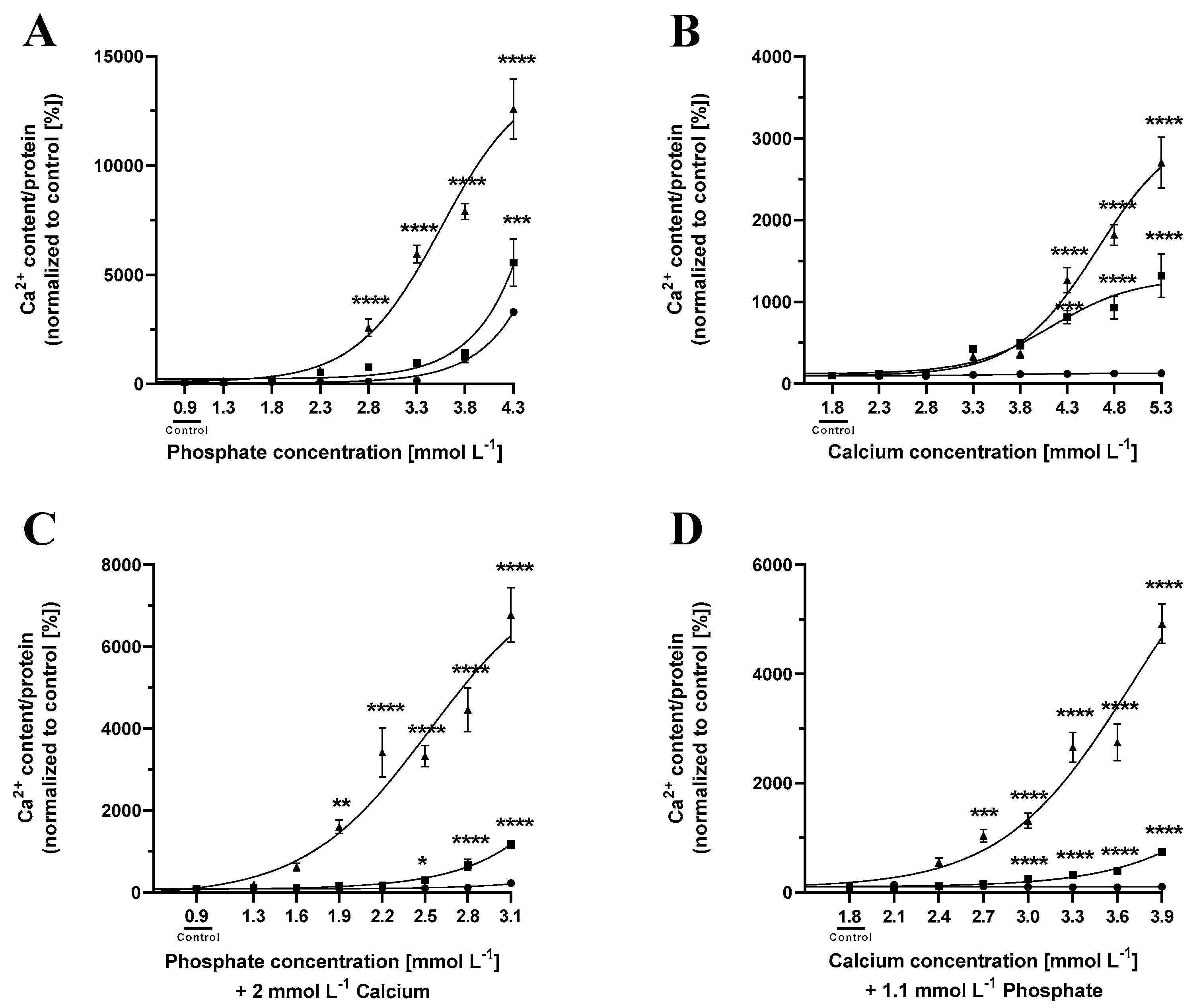
2.1. Cementoblast Calcification Increases with Increasing Phosphate Concentrations
2.2. Cementoblast Calcification Increases with Increasing Calcium Concentrations
2.3. Increasing Phosphate Concentrations in Combination with Increased Calcium Concentrations Augment Cementoblast Calcification
2.4. Increasing Calcium Concentrations in Combination with Increased Phosphate Concentration Augment Cementoblast Calcification
2.5. Cementoblast Calcification Decreases with Increasing Fetal Calf Serum Concentrations
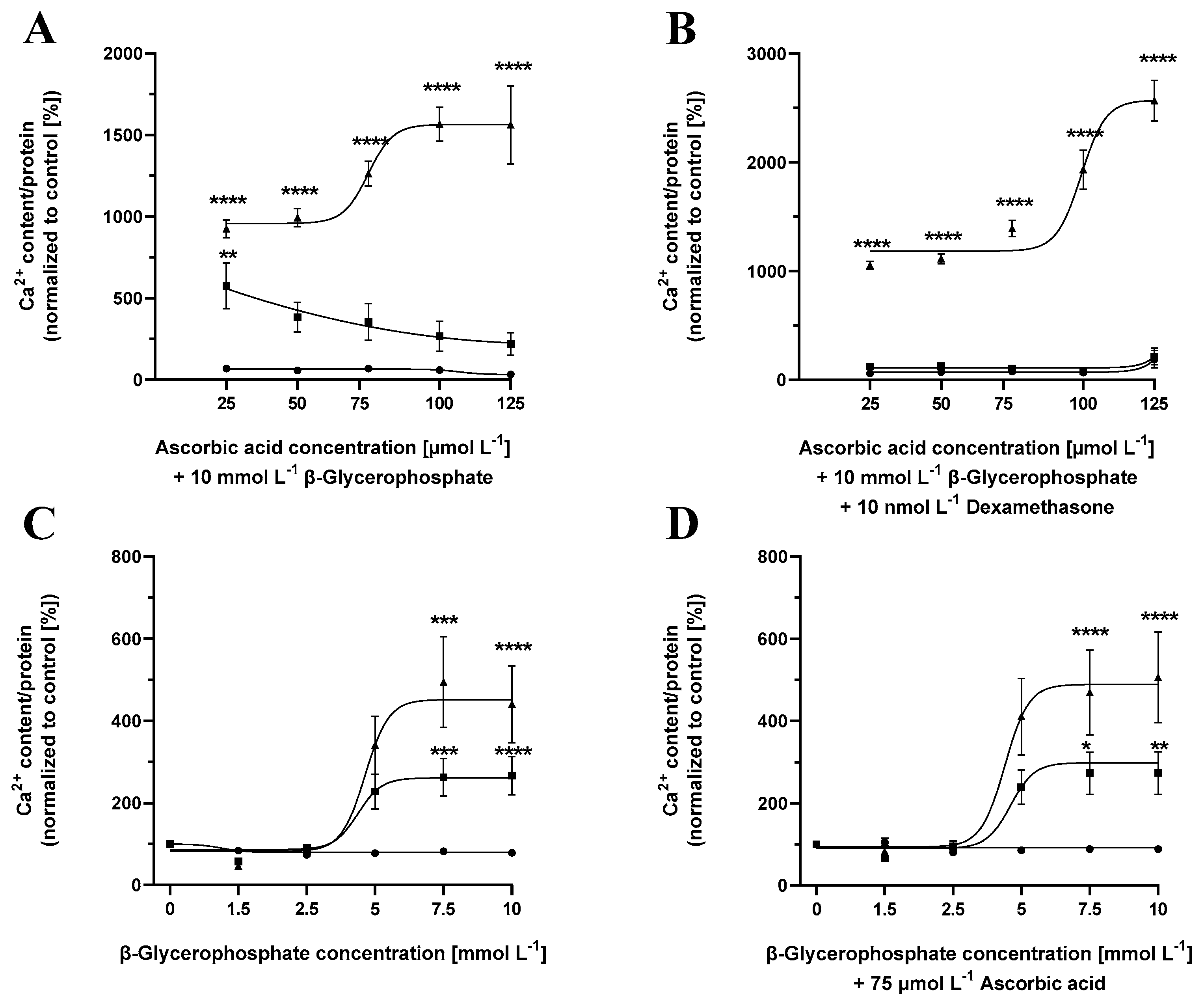
2.6. Cementoblast Calcification Is Dependent on Ascorbic Acid Concentrations, Independent of Dexamethasone Supplementation
2.7. Cementoblast Calcification Is Dependent on β-Glycerolphosphate Concentrations
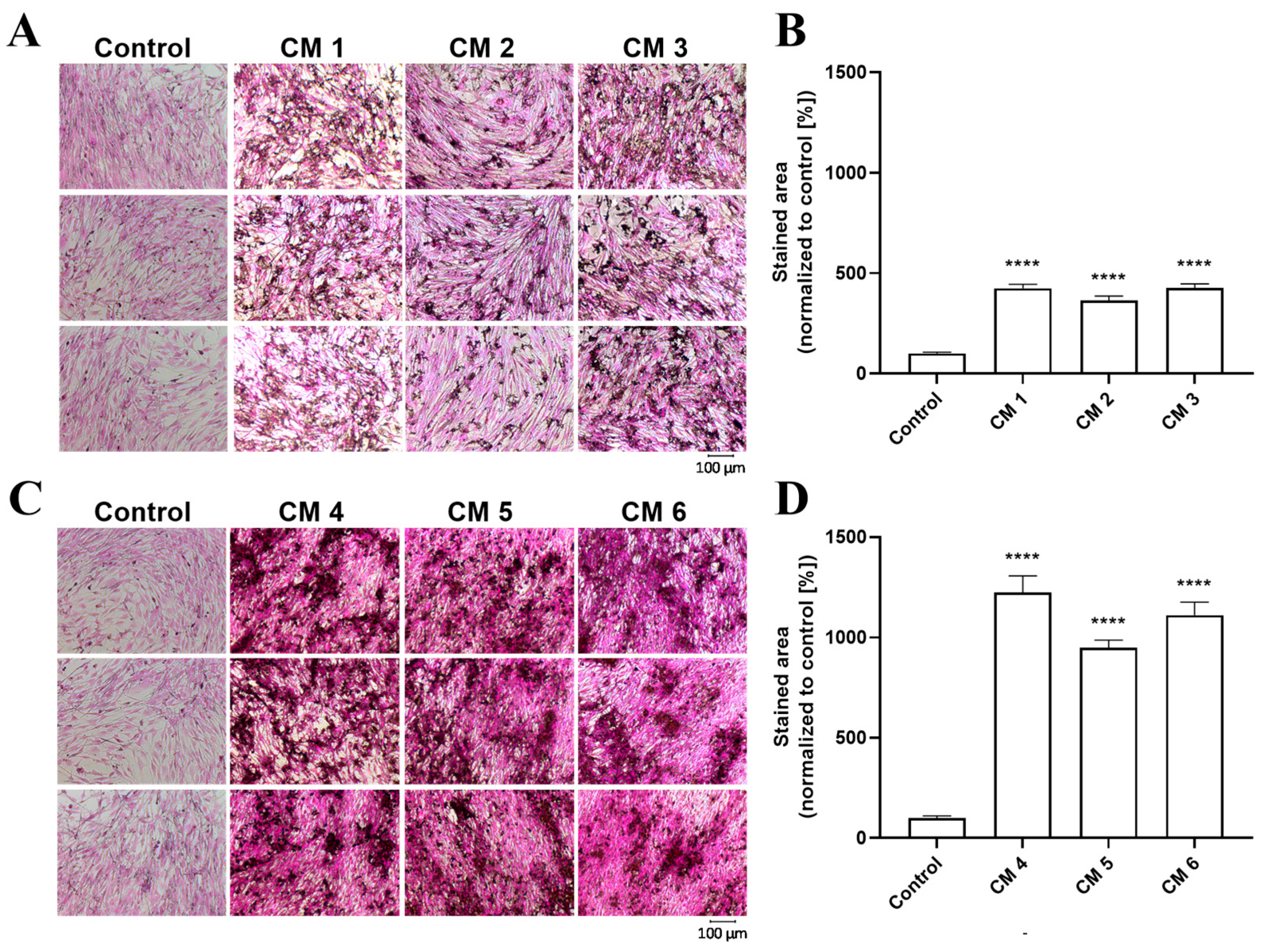
2.8. Visualization and Quantification of the Calcification of Cementoblasts by Von Kossa Staining and Alizarin Red Staining
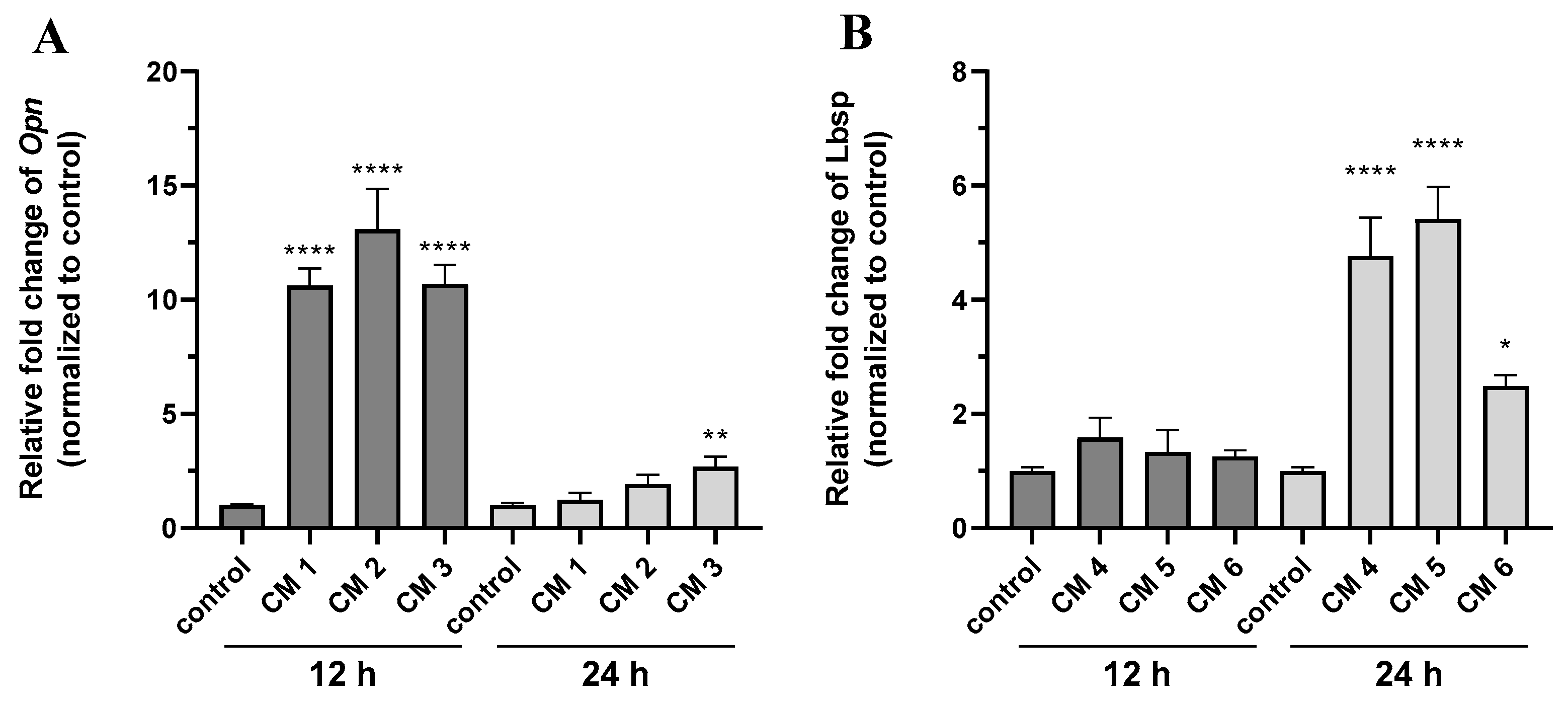
2.9. Different Calcification Pathways Are Triggered by Different Calcification-Inducing Supplements
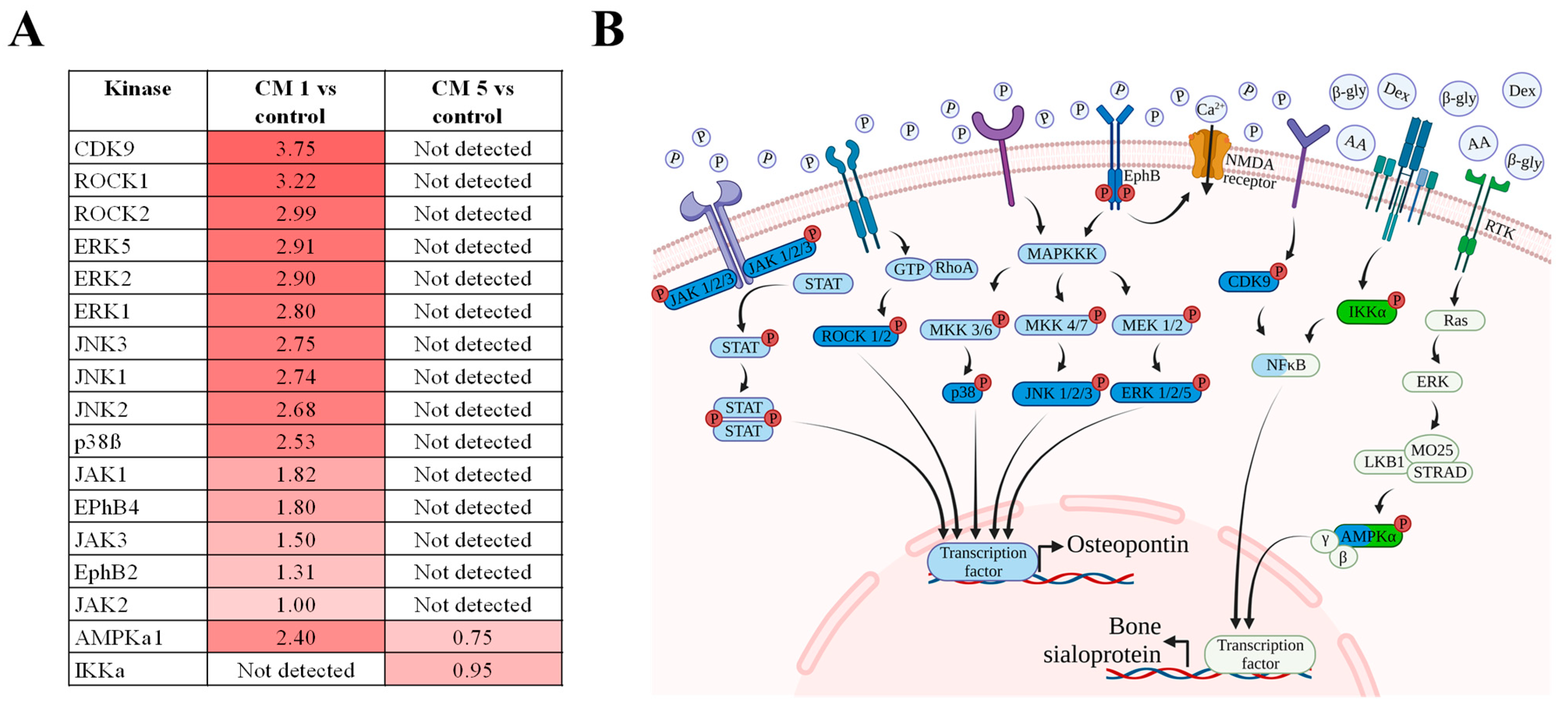
2.10. Kinases Influencing Osteogenic Pathways Are Phosphorylated by the Calcification-Inducing Supplements
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Basic Incubation Medium of Cementoblasts
4.3. Inducers of Calcification Processes in Cementoblasts for Experiments
4.4. Quantification of the Calcium Concentration of Cementoblasts
4.5. Quantification of Protein Concentration of Cementoblasts
4.6. Visualization and Quantification of the Calcification of Cementoblasts by Von Kossa Staining
4.7. Visualization and Quantification of the Calcification of Cementoblasts by Alizarin Red Staining
4.8. Reverse Transcription-Quantitative Polymerase Chain Reaction (RT-qPCR) Analyses of mRNA Expression
4.9. Kinase Assay
4.10. Statistics
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yamamoto, S.; Fukagawa, M. Uremic Toxicity and Bone in CKD. J. Nephrol. 2017, 30, 623–627. [Google Scholar] [CrossRef] [PubMed]
- Savica, V.; Calo, L.A.; Caldarera, R.; Cavaleri, A.; Granata, A.; Santoro, D.; Savica, R.; Muraca, U.; Mallamace, A.; Bellinghieri, G. Phosphate salivary secretion in hemodialysis patients: Implications for the treatment of hyperphosphatemia. Nephron Physiol. 2007, 105, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Sanz, M.; Marco Del Castillo, A.; Jepsen, S.; Gonzalez-Juanatey, J.R.; D’Aiuto, F.; Bouchard, P.; Chapple, I.; Dietrich, T.; Gotsman, I.; Graziani, F.; et al. Periodontitis and cardiovascular diseases: Consensus report. J. Clin. Periodontol. 2020, 47, 268–288. [Google Scholar] [CrossRef] [PubMed]
- Serni, L.; Caroti, L.; Barbato, L.; Nieri, M.; Serni, S.; Cirami, C.L.; Cairo, F. Association between chronic kidney disease and periodontitis. A systematic review and metanalysis. Oral Dis. 2023, 29, 40–50. [Google Scholar] [CrossRef]
- Mohamed, F.F.; Amadeu de Oliveira, F.; Kinoshita, Y.; Yalamanchili, R.R.; Eltilib, L.A.; Andras, N.L.; Narisawa, S.; Tani, T.; Chu, E.Y.; Millan, J.L.; et al. Dentoalveolar Alterations in an Adenine-Induced Chronic Kidney Disease Mouse Model. J. Bone Miner. Res. 2023, 38, 1192–1207. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Y.; Shao, P.; Wang, L.; Bao, X.; Hu, M. IL1beta inhibits differentiation of cementoblasts via microRNA-325-3p. J. Cell. Biochem. 2020, 121, 2606–2617. [Google Scholar] [CrossRef]
- Bosshardt, D.D.; Stadlinger, B.; Terheyden, H. Cell-to-cell communication—Periodontal regeneration. Clin. Oral Implants Res. 2015, 26, 229–239. [Google Scholar] [CrossRef]
- Iordanishvili, A.K.; Belskikh, O.A.; Pikhur, O.L. Characteristics of dental hard tissues in chronic kidney disease: Morphology, chemical composition, possibilities of remineralizing therapy. Stomatologiia 2019, 98, 25–30. [Google Scholar] [CrossRef]
- Tsang, H.G.; Clark, E.L.; Markby, G.R.; Bush, S.J.; Hume, D.A.; Corcoran, B.M.; MacRae, V.E.; Summers, K.M. Expression of Calcification and Extracellular Matrix Genes in the Cardiovascular System of the Healthy Domestic Sheep (Ovis aries). Front. Genet. 2020, 11, 919. [Google Scholar] [CrossRef]
- Liu, S.; Yang, W.; Li, Y.; Sun, C. Fetal bovine serum, an important factor affecting the reproducibility of cell experiments. Sci. Rep. 2023, 13, 1942. [Google Scholar] [CrossRef]
- Zhu, D.; Deng, X.; Han, X.F.; Sun, X.X.; Pan, T.W.; Zheng, L.P.; Liu, Y.Q. Wedelolactone Enhances Osteoblastogenesis through ERK- and JNK-mediated BMP2 Expression and Smad/1/5/8 Phosphorylation. Molecules 2018, 23, 561. [Google Scholar] [CrossRef] [PubMed]
- Miyata, Y.; Obata, Y.; Mochizuki, Y.; Kitamura, M.; Mitsunari, K.; Matsuo, T.; Ohba, K.; Mukae, H.; Nishino, T.; Yoshimura, A.; et al. Periodontal Disease in Patients Receiving Dialysis. Int. J. Mol. Sci. 2019, 20, 3805. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, T.; Sharma, P.; Walter, C.; Weston, P.; Beck, J. The epidemiological evidence behind the association between periodontitis and incident atherosclerotic cardiovascular disease. J. Clin. Periodontol. 2013, 40 (Suppl. 14), S70–S84. [Google Scholar] [CrossRef] [PubMed]
- Mathur, V.P.; Dhillon, J.K. Dental Caries: A Disease Which Needs Attention. Indian J. Pediatr. 2018, 85, 202–206. [Google Scholar] [CrossRef]
- Abou Neel, E.A.; Aljabo, A.; Strange, A.; Ibrahim, S.; Coathup, M.; Young, A.M.; Bozec, L.; Mudera, V. Demineralization-remineralization dynamics in teeth and bone. Int. J. Nanomed. 2016, 11, 4743–4763. [Google Scholar] [CrossRef]
- Holmar, J.; Uhlin, F.; Fernstrom, A.; Luman, M.; Jankowski, J.; Fridolin, I. An optical method for serum calcium and phosphorus level assessment during hemodialysis. Toxins 2015, 7, 719–727. [Google Scholar] [CrossRef]
- Razzaque, M.S. Salivary phosphate as a biomarker for human diseases. FASEB Bioadv. 2022, 4, 102–108. [Google Scholar] [CrossRef]
- Bae, W.J.; Park, J.S.; Kang, S.K.; Kwon, I.K.; Kim, E.C. Effects of Melatonin and Its Underlying Mechanism on Ethanol-Stimulated Senescence and Osteoclastic Differentiation in Human Periodontal Ligament Cells and Cementoblasts. Int. J. Mol. Sci. 2018, 19, 1742. [Google Scholar] [CrossRef]
- Matthews, B.G.; Roguljic, H.; Franceschetti, T.; Roeder, E.; Matic, I.; Vidovic, I.; Joshi, P.; Kum, K.Y.; Kalajzic, I. Gene-expression analysis of cementoblasts and osteoblasts. J. Periodontal. Res. 2016, 51, 304–312. [Google Scholar] [CrossRef]
- Derici, U.; El Nahas, A.M. Vascular calcifications in uremia: Old concepts and new insights. Semin. Dial. 2006, 19, 60–68. [Google Scholar] [CrossRef]
- Peter, S.J.; Liang, C.R.; Kim, D.J.; Widmer, M.S.; Mikos, A.G. Osteoblastic phenotype of rat marrow stromal cells cultured in the presence of dexamethasone, beta-glycerolphosphate, and L-ascorbic acid. J. Cell Biochem. 1998, 71, 55–62. [Google Scholar] [CrossRef]
- An, S.; Gao, Y.; Huang, Y.; Jiang, X.; Ma, K.; Ling, J. Short-term effects of calcium ions on the apoptosis and onset of mineralization of human dental pulp cells in vitro and in vivo. Int. J. Mol. Med. 2015, 36, 215–221. [Google Scholar] [CrossRef]
- Rahman, H.; Qasim, M.; Schultze, F.C.; Oellerich, M.; Asif, A.R. Fetal calf serum heat inactivation and lipopolysaccharide contamination influence the human T lymphoblast proteome and phosphoproteome. Proteome Sci. 2011, 9, 71. [Google Scholar] [CrossRef] [PubMed]
- Bohets, H.H.; Nouwen, E.J.; De Broe, M.E.; Dierickx, P.J. Effects of foetal calf serum on cell viability, cytotoxicity and detoxification in the two kidney-derived cell lines LLC-PK1 and MDCK. Toxicol. Vitro 1994, 8, 559–561. [Google Scholar] [CrossRef] [PubMed]
- Braam, L.; Dissel, P.; Gijsbers, B.; Spronk, H.; Hamulyak, K.; Soute, B.; Debie, W.; Vermeer, C. Assay for human matrix gla protein in serum: Potential applications in the cardiovascular field. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1257–1261. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Shimokawa, H.; Takagi, Y.; Sasaki, S. Calcium-binding properties of fetuin in fetal bovine serum. J. Exp. Zool. 1994, 270, 501–507. [Google Scholar] [CrossRef]
- Chen, H.Y.; Chiu, Y.L.; Hsu, S.P.; Pai, M.F.; Yang, J.Y.; Peng, Y.S. Relationship between fetuin A, vascular calcification and fracture risk in dialysis patients. PLoS ONE 2016, 11, e0158789. [Google Scholar] [CrossRef]
- Vater, C.; Kasten, P.; Stiehler, M. Culture media for the differentiation of mesenchymal stromal cells. Acta Biomater. 2011, 7, 463–477. [Google Scholar] [CrossRef]
- Alesutan, I.; Moritz, F.; Haider, T.; Shouxuan, S.; Gollmann-Tepekoylu, C.; Holfeld, J.; Pieske, B.; Lang, F.; Eckardt, K.U.; Heinzmann, S.S.; et al. Impact of beta-glycerophosphate on the bioenergetic profile of vascular smooth muscle cells. J. Mol. Med. 2020, 98, 985–997. [Google Scholar] [CrossRef]
- Ivanov, V.; Ivanova, S.; Niedzwiecki, A.; Rath, M. Vitamin C inhibits the calcification process in human vascular smooth muscle cells. Am. J. Cardiovasc. Dis. 2020, 10, 108–116. [Google Scholar]
- D’Aniello, C.; Cermola, F.; Patriarca, E.J.; Minchiotti, G. Vitamin C in Stem Cell Biology: Impact on Extracellular Matrix Homeostasis and Epigenetics. Stem Cells Int. 2017, 2017, 8936156. [Google Scholar] [CrossRef]
- Wang, Y.H.; Liu, Y.; Maye, P.; Rowe, D.W. Examination of mineralized nodule formation in living osteoblastic cultures using fluorescent dyes. Biotechnol. Prog. 2006, 22, 1697–1701. [Google Scholar] [CrossRef]
- Chen, Q.; Shou, P.; Zhang, L.; Xu, C.; Zheng, C.; Han, Y.; Li, W.; Huang, Y.; Zhang, X.; Shao, C.; et al. An osteopontin-integrin interaction plays a critical role in directing adipogenesis and osteogenesis by mesenchymal stem cells. Stem Cells 2014, 32, 327–337. [Google Scholar] [CrossRef]
- Thi Nguyen, N.; Thi Nguyen, T.; Nguyen, H.T.; Lee, J.M.; Kim, M.J.; Qi, X.F.; Cha, S.K.; Lee, I.K.; Park, K.S. Inhibition of mitochondrial phosphate carrier prevents high phosphate-induced superoxide generation and vascular calcification. Exp. Mol. Med. 2023, 55, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Paudel, R.; Fusi, L.; Schmidt, M. The MEK5/ERK5 Pathway in Health and Disease. Int. J. Mol. Sci. 2021, 22, 7594. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Li, L.; Gao, J.; Chen, X.; Sang, Q. miR-214 suppresses the osteogenic differentiation of bone marrow-derived mesenchymal stem cells and these effects are mediated through the inhibition of the JNK and p38 pathways. Int. J. Mol. Med. 2017, 39, 71–80. [Google Scholar] [CrossRef]
- Matsuzaki, E.; Hirose, H.; Fujimasa, S.; Yoshimoto, S.; Yanagi, T.; Matsumoto, K.; Nikaido, M.; Minakami, M.; Matsumoto, N.; Anan, H. Sphingosine-1-phosphate receptor 2 agonist induces bone formation in rat apicoectomy and alveolar bone defect model. J. Dent. Sci. 2022, 17, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Tao, G.; Guan, Y.; Chen, S.; He, Y.; Li, T.; Zou, S.; Li, Y. The role of ephrinB2-EphB4 signalling in bone remodelling during orthodontic tooth movement. Orthod. Craniofac. Res. 2023, 26, 107–116. [Google Scholar] [CrossRef]
- Cai, B.; Chang, S.H.; Becker, E.B.; Bonni, A.; Xia, Z. p38 MAP kinase mediates apoptosis through phosphorylation of BimEL at Ser-65. J. Biol. Chem. 2006, 281, 25215–25222. [Google Scholar] [CrossRef] [PubMed]
- Cagnol, S.; Chambard, J.C. ERK and cell death: Mechanisms of ERK-induced cell death—Apoptosis, autophagy and senescence. FEBS J. 2010, 277, 2–21. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Wei, L. Rho kinase in the regulation of cell death and survival. Arch. Immunol. Ther. Exp. 2007, 55, 61–75. [Google Scholar] [CrossRef]
- Ogasawara, T.; Katagiri, M.; Yamamoto, A.; Hoshi, K.; Takato, T.; Nakamura, K.; Tanaka, S.; Okayama, H.; Kawaguchi, H. Osteoclast differentiation by RANKL requires NF-kappaB-mediated downregulation of cyclin-dependent kinase 6 (Cdk6). J. Bone Miner. Res. 2004, 19, 1128–1136. [Google Scholar] [CrossRef]
- Brasier, A.R. Expanding role of cyclin dependent kinases in cytokine inducible gene expression. Cell Cycle 2008, 7, 2661–2666. [Google Scholar] [CrossRef][Green Version]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-kappaB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Jang, W.G.; Kim, E.J.; Bae, I.H.; Lee, K.N.; Kim, Y.D.; Kim, D.K.; Kim, S.H.; Lee, C.H.; Franceschi, R.T.; Choi, H.S.; et al. Metformin induces osteoblast differentiation via orphan nuclear receptor SHP-mediated transactivation of Runx2. Bone 2011, 48, 885–893. [Google Scholar] [CrossRef] [PubMed]
- D’Errico, J.A.; Berry, J.E.; Ouyang, H.; Strayhorn, C.L.; Windle, J.J.; Somerman, M.J. Employing a transgenic animal model to obtain cementoblasts in vitro. J. Periodontol. 2000, 71, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Niederau, C.; Craveiro, R.B.; Azraq, I.; Brockhaus, J.; Bastian, A.; Kirschneck, C.; Wolf, M. Selection and validation of reference genes by RT-qPCR for murine cementoblasts in mechanical loading experiments simulating orthodontic forces in vitro. Sci. Rep. 2020, 10, 10893. [Google Scholar] [CrossRef] [PubMed]
- Azraq, I.; Craveiro, R.B.; Niederau, C.; Brockhaus, J.; Bastian, A.; Knaup, I.; Neuss, S.; Wolf, M. Gene expression and phosphorylation of ERK and AKT are regulated depending on mechanical force and cell confluence in murine cementoblasts. Ann. Anat. 2021, 234, 151668. [Google Scholar] [CrossRef]
- Minihane, A.M. Fish oil omega-3 fatty acids and cardio-metabolic health, alone or with statins. Eur. J. Clin. Nutr. 2013, 67, 536–540. [Google Scholar] [CrossRef]


| CM 1 | CM 2 | CM 3 | CM 4 | CM 5 | CM 6 | |
|---|---|---|---|---|---|---|
| FCS [Vol%] | 2.5 | 2.5 | 5 | 5 | 5 | |
| NaH2PO4 [mmol L−1] | 0.95 | 0.65 | ||||
| Na2HPO4 [mmol L−1] | 0.95 | 0.65 | ||||
| CaCl2 [mmol L−1] | 2.5 | 0.2 | ||||
| L-ascorbic acid [µmol L−1] | 75 | 75 | 75 | |||
| β-glycerolphosphate [mmol L−1] | 10 | 12.5 | 7.5 | |||
| Dexamethasone [nmol L−1] | 10 | 5 | 5 |
| Experiment | DMEM | P/S [%] | Na2HPO4: NaH2PO4 (1:1) [mmol L−1] Final Conc. | CaCl2 [mmol L−1] Final Conc. | FCS [%] | β-Glycerol-Phosphate [mmol L−1] | Ascorbic Acid [µmol L−1] | Dexame-Thasone [nmol L−1] |
|---|---|---|---|---|---|---|---|---|
| Phosphate or calcium concentration dependency | ||||||||
| Control | + | 1 | 0.9 | 1.8 | 2.5 | – | – | – |
| CM (Phosphate) | + | 1 | 1.3, 1.8, 2.3, 2.8, 3.3, 3.8, 4.3 | 1.8 | 2.5 | – | – | – |
| CM (CaCl2) | + | 1 | 0.9 | 2.3, 2.8, 3.3, 3.8, 4.3, 4.8, 5.3 | 2.5 | – | – | – |
| Phosphate and calcium concentration dependency | ||||||||
| Control | + | 1 | 0.9 | 1.8 | 2.5 | – | – | – |
| CM (Phosphate & fixed CaCl2) | + | 1 | 1.3, 1.6, 1.9, 2.2, 2.5, 2.8, 3.1 | 2 | 2.5 | – | – | – |
| CM (Fixed phosphate & CaCl2) | + | 1 | 1.1 | 2.1, 2.4, 2.7, 3.0, 3.3, 3.6, 3.9 | 2.5 | – | – | – |
| FCS concentration dependency | ||||||||
| Control | + | 1 | 0.9 | 1.8 | 1, 2.5, 5, 7.5, 10, 12.5, 15 | – | – | – |
| CM | + | 1 | 0.9 | 1.8 | 1, 2.5, 5, 7.5, 10, 12.5, 15 | 10 | 75 | – |
| Ascorbic acid concentration dependency | ||||||||
| Control | + | 1 | 0.9 | 1.8 | 5 | – | – | – |
| CM | + | 1 | 0.9 | 1.8 | 5 | 10 | 25, 50, 75, 100, 125, 250 | – |
| CM with dexamethasone | + | 1 | 0.9 | 1.8 | 5 | 10 | 25, 50, 75, 100, 125, 250 | 10 |
| β-glycerolphosphate concentration dependency | ||||||||
| Control | + | 1 | 0.9 | 1.8 | 5 | – | – | – |
| CM | + | 1 | 0.9 | 1.8 | 5 | 1.5, 2.5, 5, 7.5, 10, 12.5, 15 | 75 | – |
| CM with dexamethasone | + | 1 | 0.9 | 1.8 | 5 | 1.5, 2.5, 5, 7.5, 10, 12.5, 15 | 75 | 10 |
| Gene | Nucleotide Sequence | |
|---|---|---|
| β-actin | Forward | 5′-CAACGAGCGGTTCCGATG-3′ |
| Reverse | 5′-GCCACAGGATTCCATACCCAA-3′ | |
| Bone sialoprotein (Lbsp) | Forward | 5′-TGAGTGACAGCCGGGAGAAC-3′ |
| Reverse | 5′-AAGAAAGTAGCGTGGCCGGT-3′ | |
| Osteopontin (Opn) | Forward | 5′-CTTTCACTCCAATCGTCCCTAC-3′ |
| Reverse | 5′-GCTCTCTTTGGAATGCTCAAGT-3′ | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhargava, S.; Jankowski, J.; Merckelbach, E.; Roth, C.E.; Craveiro, R.B.; Wolf, M. Development, Establishment, and Validation of a Model for the Mineralization of Periodontium Remodelling Cells: Cementoblasts. Int. J. Mol. Sci. 2023, 24, 13829. https://doi.org/10.3390/ijms241813829
Bhargava S, Jankowski J, Merckelbach E, Roth CE, Craveiro RB, Wolf M. Development, Establishment, and Validation of a Model for the Mineralization of Periodontium Remodelling Cells: Cementoblasts. International Journal of Molecular Sciences. 2023; 24(18):13829. https://doi.org/10.3390/ijms241813829
Chicago/Turabian StyleBhargava, Shruti, Joachim Jankowski, Erik Merckelbach, Charlotte Elisa Roth, Rogerio Bastos Craveiro, and Michael Wolf. 2023. "Development, Establishment, and Validation of a Model for the Mineralization of Periodontium Remodelling Cells: Cementoblasts" International Journal of Molecular Sciences 24, no. 18: 13829. https://doi.org/10.3390/ijms241813829
APA StyleBhargava, S., Jankowski, J., Merckelbach, E., Roth, C. E., Craveiro, R. B., & Wolf, M. (2023). Development, Establishment, and Validation of a Model for the Mineralization of Periodontium Remodelling Cells: Cementoblasts. International Journal of Molecular Sciences, 24(18), 13829. https://doi.org/10.3390/ijms241813829






