Abstract
Peptide-based strategies have received an enormous amount of attention because of their specificity and applicability. Their specificity and tumor-targeting ability are applied to diagnosis and treatment for cancer patients. In this review, we will summarize recent advancements and future perspectives on peptide-based strategies for cancer treatment. The literature search was conducted to identify relevant articles for peptide-based strategies for cancer treatment. It was performed using PubMed for articles in English until June 2023. Information on clinical trials was also obtained from ClinicalTrial.gov. Given that peptide-based strategies have several advantages such as targeted delivery to the diseased area, personalized designs, relatively small sizes, and simple production process, bioactive peptides having anti-cancer activities (anti-cancer peptides or ACPs) have been tested in pre-clinical settings and clinical trials. The capability of peptides for tumor targeting is essentially useful for peptide–drug conjugates (PDCs), diagnosis, and image-guided surgery. Immunomodulation with peptide vaccines has been extensively tested in clinical trials. Despite such advantages, FDA-approved peptide agents for solid cancer are still limited. This review will provide a detailed overview of current approaches, design strategies, routes of administration, and new technological advancements. We will highlight the success and limitations of peptide-based therapies for cancer treatment.
1. Introduction
Cancer represents a profound worldwide public health challenge, demanding significant attention and resources [1]. According to a recent study conducted in 21 countries across five continents, cancer emerges as the primary reason for mortality in numerous nations [2]. Major challenges in cancer treatment include the emergence of multidrug resistance and the scarcity of tumor-specific therapies that exhibit minimal side effects. Cancer therapy has undergone significant advancements, but the need for more effective and targeted treatments remains [3]. One of the key aspects of cancer therapy is the targeted delivery of chemotherapeutic agents to cancer cells, maximizing treatment effectiveness while minimizing harm to healthy tissue. Bioactive peptides have gained attention due to their potential anticancer properties. Peptide-based approaches offer several advantages in cancer treatment, including enhanced specificity, reduced toxicity to normal tissues, and versatility in targeting various molecular pathways involved in cancer progression [4,5,6,7].
Diverse natural and modified peptides have been extensively studied and acquired, spanning numerous therapeutic areas. Therapeutic peptides act as hormones, growth factors, neurotransmitters, ion channel ligands, or anti-infective agents. They possess high affinity and specificity when binding to cell surface receptors, triggering specific intracellular effects. In terms of their mode of action, therapeutic peptides share similarities with biologics like therapeutic proteins and antibodies, offering targeted and specific therapeutic approaches. Peptides have certain drawbacks compared to antibodies, including a shorter half-life due to rapid excretion and susceptibility to protease degradation. However, peptides offer advantages such as low cost, the ability to penetrate deep tissue, efficient internalization into cells, lower immunogenicity and reduced toxicity towards bone marrow and the liver, and ease of modification using chemical methods, setting them apart from antibodies [8,9].
In this review, we will describe the peptide-based approaches for cancer diagnosis and treatment, the design strategies, and summarize peptide-based-cancer diagnosis and therapies in clinical and pre-clinical to provide an overview of peptide-based agents for cancer treatment.
2. Peptide-Based Approaches for Cancer Treatment
Peptide-based therapeutic approaches for cancer treatment encompass a wide range of strategies that leverage the unique properties of peptides to target and combat cancer cells.
2.1. Anti-Cancer Peptides (ACPs)
ACPs have undergone recent enhancements to be transformed into drugs and vaccines, subsequently undergoing evaluation through different stages of clinical trials [10]. ACPs are short bioactive peptides composed of 10–60 amino acids, which exert their therapeutic effects through various mechanisms that specifically target cancer cells while sparing normal cells. Common mechanisms of action for anti-cancer peptides are apoptosis induction, membrane disruption, angiogenesis inhibition, signaling pathway modulation, and immunomodulation. In addition, ACPs can interfere with key signaling pathways that promote cancer cell survival, proliferation, and metastasis. These peptides may target proteins involved in cell cycle regulation, growth factor signaling, or survival pathways, leading to the inhibition of cancer cell growth and survival.
2.1.1. Induction of Apoptosis
ACPs can trigger programmed cell death, known as apoptosis, in cancer cells. They can directly target specific intracellular components involved in apoptosis regulation, such as mitochondrial membranes or caspases, leading to the activation of apoptotic pathways. This results in the controlled elimination of cancer cells. Out of the various bioactivities, the apoptosis pathway is recognized as the most efficacious strategy in non-surgical cancer therapies because of its ability to cause minimal inflammation and damage to the targeted regions [11,12]. The anti-apoptotic B-cell lymphoma 2 (BCL-2) family proteins primarily regulate the intrinsic apoptosis pathway, whereas the extrinsic apoptosis pathway involves Tumor necrosis factor (TNF) receptor (TNFR), FAS (CD95), and Death receptor 3 (DR3)/WSL [13] (Figure 1A). Peptide-based cancer therapy can target both apoptosis pathways. Antimicrobial peptides from Anabas testudineus AtMP1 and AtMP2 (Table 1 for peptides tested in pre-clinical settings) induce apoptosis of breast cancer cells MCF7 and MDA-MB-231 by down-regulating BCL-2 gene [14]. In contrast, another antimicrobial peptide from Nile tilapia (Oreochromis niloticus), MSP-4, induces the apoptosis of osteosarcoma MG63 cells through a Fas/FasL-mediated pathway [15].
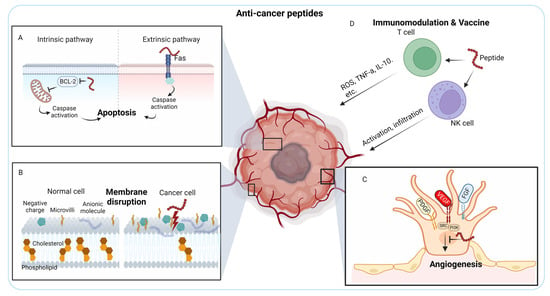
Figure 1.
Mechanisms of anti-cancer peptides, highlighted for each mechanism indicated by boxes and their magnified boxes with schemes. (A) Apoptosis induction. (B) Membrane disruption. (C) Inhibition of tumor angiogenesis. (D) Immunomodulation and peptide vaccine.
A peptide fragment, p28 (NSC745104) (CDG Therapeutics Inc., Elk Grove Village, IL, USA) (Table 2 for peptides tested in clinical trials) derived from bacterial protein azurin also induces cell cycle arrest in various types of human cancer cells [16,17]. Cupredoxin azurin is secreted by the opportunistic pathogen Pseudomonas aeruginosa in response to increasing numbers of and proximity to cancer cells [18,19,20,21,22]. Secreted azurin preferentially enters a variety of solid tumor cell lines including breast cancers and induces p53-mediated apoptosis [18,19,23,24,25]. A fragment of azurin, amino acids 50 to 77 (p28), is responsible for azurin’s preferential penetration and anti-proliferative activity [16,26]. As a single therapeutic agent, p28 (NSC745104) was tested in two Phase I clinical trials (NCT00914914, NCT01975116) and granted the FDA Orphan Drug and Rare Pediatric Disease Designations since it showed preliminary efficacy without apparent adverse effects, toxicity, or immunogenicity in patients with advanced solid tumors and in pediatric patients with recurrent and refractory central nervus system (CNS) tumors (NCI and Pediatric Brain Tumor Consortium) [27,28,29].


Table 1.
Examples of peptides in pre-clinical tests.
Table 1.
Examples of peptides in pre-clinical tests.
| Name | Sequence | Effects | Refs. |
|---|---|---|---|
| AtMP1 | THPPTTTTTTTTTTTTTAAPATTT | Apoptosis | [14] |
| AtMP2 | TGIATSGLATFTLHTGSLAPAT | Apoptosis | [14] |
| MSP-4 | FIHHIIGGLFSAGKAIHRLIRRRRR | Apoptosis | [15] |
| peptide 8.6 | Ac-MCTIDFDEYRFRKT-NH2 | Apoptosis | [30] |
| HPRP-A1-TAT | Ac-FKKLKKLFSKLWN WK-RKKRRQRRR-NH2 | Membrane disruption | [31] |
| melittin | NH2-GIGAVLKVLTTGLPALISWIKRKRQQ-NH2 | Membrane disruption | [32] |
| QR-KLU | QKRKRKKSRY-KLUKLUKKLUKLUK | Angiogenesis inhibition | [33] |
| KV11 | YTMNPRKLFDY | Angiogenesis inhibition | [34] |
| KAI | SRGTPVDERLFLIVRVTVQLSHP-NH2 | Angiogenesis inhibition | [35,36,37] |
| LFcinB26–36 | RRWQWRMKKLG | Immunomodulation | [38,39] |
| LFcinB17–41 | FKCRRWQWRMKKLGAPSITCVRRAF | Immunomodulation | [38,39] |
| CREKA | CREKA | Tumor-homing | [40] |
| RGD-4C | CDCRGDCFCG | Tumor-homing | [41] |
| c(RGDyK) | c(RGDyK) | Tumor-homing | [42] |
| CCK8 | DY(SO3H)MGWMDF-NH2 | Tumor-homing | [43] |
| TAT | YGRKKRRQRRR | CPP | [44] |
| Penetratin | RQIKIWFQNRRMKWKK | CPP | [45,46] |
| Transportan | GWTLNSAGYLLGKINLKALAALAKKIL-NH2 | CPP | [47] |
| M13 | AGYLLGKINLKACAALAKKCL | CPP | [48,49] |
| pA | cNGQGEQc | Targeting integrins | [50] |
| NKTP-3 | kkRRuPLBIUBDPVdRRKrgerppr | Inhibits tumor growth | [51] |
| KRpep-2d | Ac-RRRRCPLYISYDPVCRRRR-NH2 | Inhibits tumor growth | [52] |
| HVGGSSV | HVGGSSV | Imaging | [53,54] |
| X4-2-6 | LLFVITLPFWAVDAVANWYFGNDD-PEG27 | Preventing metastasis | [55] |
| CLT1 | CGLIIQKNEC | Tumor-homing | [56] |
| C5C2 | SSVVQSTGHMQSKVYESVLALSAEVQAAR-NH2 | BBB permeabilization | [57] |
| HAV6 | Ac-SHAVSS-NH2 | BBB permeabilization | [58,59,60] |
| K16ApoE | KKKKKKKKKKKKKKKKLRVRLASHLRKLRKRLLRDA | RMT | [61] |
| AEP | LRKLRKRLLR | RMT | [62] |
| RAP12 | EAKIEKHNHYQK | RMT | [63] |
| melanotransferrin (MTf)-derived peptide | DSSHAFTLDELR | RMT | [64] |
| Peptide-22 (VH434) | Ac-[CMPRLRGC]c-NH2 | RMT | [49,65] |
| L57 | TWPKHFDKHTFYSILKLGKH-OH | RMT | [66] |
| M1 | TFYGGRPKRNNFLRGIR | RMT | [67] |
| LRPep2 | HPWCCGLRLDLR | RMT | [68] |
| TfR-T12 | THRPPMWSPVWP | RMT | [69] |
| T7 | HAIYPRH | RMT | [69] |
| B6 | G GHKAKGPRKLGS | RMT | [70] |
| CRT peptide | CRTIGPSVC | RMT | [71] |
| NanoLigand Carriers (NLC) | CGYRPVHNIRGHWAPG | RMT | [72] |
| Leptin30 | YQQILTSMPSRNVIQISNDLENLRDLLHVL | RMT | [73,74] |
| g21 | TLIKTIVTRINDISHTQSVSA | RMT | [75] |
| A7R | ATWLPPR | RMT | [76,77] |
| IL-13p | TAMRAVDKLLLHLKKLFREGQFNRNFESIIICRDRT | RMT | [78] |
| Pep-1 | CGEMGWVRC | RMT | [79,80] |
| G7 | GFtGFLS | RMT | [81,82] |
| RVG-9R | YTIWMPENPRPGTPCDIFTNSRGKRASNGGGGRRRRRRRR | RMT | [83] |
| RDP | MGKSVRTWNEIIPSKGCLRVGGRCHPHVNGGG-RRRRRRRRR | RMT | [84] |
| 39 mer RDP | KSVRTWNEIIPSKGCLR VGGRCHPH VNGGGRRRRRRRRR | RMT | [85] |
| KC2S | YTKTWCDGFCSSRGKRIDLG | RMT | [86] |
| CDX | FKESWREARGTRIERG | RMT | [87,88] |
| MiniAp-4 | H-[Dap]KAPETALD-NH2 | RMT | [89] |
| TGN | TGNYKALHPHNG | RMT | [90] |
| SynB1 | RGGRLSYSRRRFSTSTGR | AMT | [91] |
| CAQK | CAQK | AMT | [92] |
| G23 | HLNILSTLWKYR | AMT | [93,94] |
| PepH3 | AGILKRW | AMT | [95] |
| N-methyl phenylalanine-rich peptide * | N-MePhe-(N-MePhe)3-CONH2 | AMT | [96] |
| phenylproline tetrapeptide * | (PhPro)4 | AMT | [97] |
| NegPep | SGTQEEY | AMT | [98] |
| Porphyrin | AGILKRWK-NH2 | AMT | [99] |
| NFL-TBS.40–63 | YSSYSAPVSSSLSVRRSYSSSSGS | AMT | [100,101] |
| LMWP | CVSRRRRRRGGRRRR | AMT | [102] |
Small letter indicates D amino acids. C indicates cyclized peptide. * indicates peptides described as 3 letter code and functional groups. Ph, Me, CONH2, NH2, SO3H, OH, Ac are functional groups. Some modifications may not be depicted here such as disulfide bond or PEGylation.

Table 2.
Examples of peptides tested in clinical trials.
ATSP-7041 (Aileron Therapeutics, Watertown, MA, USA) is also activating the p53 pathway by interacting and inhibiting mouse double minute 2 (MDM2) and MDMX (MDM4) [131]. MDM2 is an E3 ubiquitin ligase that inhibits p53 by targeting it for degradation. MDMX inhibits p53’s transactivation activity and promotes MDM2 activity via direct protein–protein interactions. ATSP-7041 and its derivative ALRN-6924 were developed from N-terminal α-helical domain of the p53 tumor suppressor protein, which directly binds to MDM2/MDMX [132] and inhibits growth of solid tumors and lymphomas [133,134,135]. ALRN-6924 was tested in Phase I/II clinical trial for patients with solid tumors and lymphomas (NCT02264613). ALRN-6924 was well-tolerated as there was evidence of single-agent anti-tumor activity, including complete and partial responses [103,104]. However, Phase Ib chemoprotection trial in patients with p53-mutated breast cancer (NCT05622058) was terminated as patients experienced severe, grade 4 neutropenia and alopecia, failing to meet the main end points of the trial [136].
Additionally, some ACPs can interact with DNA molecules within cancer cells. They can bind to the DNA helix, induce conformational changes, or interfere with DNA replication, transcription, or repair processes. This disruption of DNA integrity can lead to DNA damage, genomic instability, and subsequent cell cycle arrest or cell death. For instance, White et al. discovered a peptide that specifically targets the C-terminal domain of breast cancer-associated protein 1 (BRCA1) in breast cancer, which altered the DNA damage response [30].
2.1.2. Membrane Disruption
Negatively charged, low cholesterol, and aberrant microvilli content cancer cell surfaces could facilitate the specific activity of ACPs against cancer cells [137,138]. (i) In healthy cells, negatively charged phospholipids are mainly found in the inner membrane leaflets, but in cancer cells, this asymmetry is disrupted, causing overexpression of negatively charged phosphatidylserine on the cell membrane surface (Figure 1B). Additionally, other anionic molecules, such as O-glycosylated mucins, and the glycosaminoglycan side chains mainly in the form of heparin sulfate, further increasing the negative charges on cancer cells. (ii) Furthermore, in healthy cell membranes, cholesterol serves as a crucial regulator of fluidity, contributing to the inhibition of cationic peptide entry or translocation. The lower cholesterol content in cancer cell membranes compromises this protective mechanism, thereby augmenting their vulnerability to the cytolytic effects of ACPs (Figure 1B). Most membrane-disrupting ACPs possess amphipathic properties, meaning they have both hydrophobic and hydrophilic regions. These peptides can interact with the lipid bilayer of cancer cell membranes, causing disruption and destabilization. This disruption can lead to increased permeability, leakage of intracellular components, and, ultimately, cell lysis. (iii) Moreover, the increased abundance and aberrant morphology of microvilli on cancer cells augment the cell surface area and contact with ACPs, further enhancing their interaction with these peptides.
One example of ACPs targeting cancer cell membranes is HPRP-A1-TAT [31], a hybrid peptide that can destroy the cell membrane to cause rapid leakage of cytoplasmic contents and has a strong anti-cancer activity. The IC50 value of this ACP in melanoma, gastric, liver, and cervical cancer cells is less than 10 µM [31]. Another example of membrane-disrupting peptide is melittin [32]. To reduce the hemolytic side effect of melittin, encapsulating melittin in hydrogel by conjugating with hydrogel self-assembling peptide RADA32 and loading doxorubicin reduced side effects and selectively inhibited tumor growth, recruited activated natural killer (NK) cells in the primary melanoma tumor, activated dendritic cells, and generated cytotoxic T-cells against remaining tumors [139].
2.1.3. Inhibition of Tumor Angiogenesis
Angiogenesis is the process where new capillaries are formed from pre-existing blood vessels, and it has a vital function in cancer by providing oxygen and nutrients necessary for tumor growth and metastasis [140,141]. Angiogenesis is controlled by various signaling pathways, such as vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), platelet-derived growth factor (PDGF), and angiopoietins [141]. Of these pathways, VEGF holds particular significance. In both preclinical tumor models and human cancer patients, the exclusive inhibition of VEGF by antibodies or small molecules has demonstrated notable effectiveness in antiangiogenic therapy, yielding positive outcomes [142]. However, resistance to VEGF inhibitors occurs through various mechanisms [143]. ACPs are studied as alternative approaches which suppress angiogenesis by targeting and inhibiting specific signaling pathways involved in angiogenesis, thereby reducing the blood supply to tumors and impeding their progression (Figure 1C). Wang et al. developed a VEGFR targeting PDC, which suppresses tumor angiogenesis in transcatheter arterial chemoembolization (TACE) model for hepatocellular carcinoma therapy [33]. KV11, an 11-amino acid peptide derived from apolipoprotein A (ApoA), inhibits angiogenesis both in vitro and in vivo, specifically targeting the c-Src/ERK signaling pathways [34].
Another example of the small peptide angiogenesis inhibitor, KAI, was designed to inhibit KIF13B-mediated VEGFR2 trafficking to the cell surface, thereby inhibiting receiving VEGF [144,145]. The peptide KAI, a kinesin-derived angiogenesis inhibitor, inhibits trafficking of VEGFR2 from the Golgi apparatus and recycling of internalized VEGFR2 and inhibits pathological angiogenesis and vascular leakage in wet age-related macular degeneration, blinding eye disease [35,36]. KAI also successfully penetrates the cell membrane by utilizing cationic residues, resulting in inhibiting tumor angiogenesis and tumor growth [144] and metastasis in cancer models in mice [37].
2.1.4. Immunomodulation
Cancer cells can evade the immune system’s detection and response mechanisms, allowing them to escape elimination [146,147]. Immunotherapies employ various approaches to target immune cells and enhance their ability to kill cancer [148]. ACPs can modulate the immune response against cancer cells by stimulating the activation and proliferation of immune cells, such as T cells and NK cells, leading to enhanced recognition and elimination of cancer cells by the immune system (Figure 1D). Additionally, ACPs may modulate immune checkpoints, enhancing the antitumor immune response [149,150]. Bovine lactoferrin (LfcinB), a peptide derived from lactoferrin, hinders the growth of head and neck squamous cell carcinoma by inducing increased lymphocyte infiltration to inhibit head and neck squamous cell carcinoma in vivo [38,39].
2.1.5. Peptide Vaccine
Among immunotherapies, in addition to immunomodulators, peptide vaccines play a crucial role in educating the immune system to generate anti-cancer activity. By stimulating specific immune responses against cancer cells, peptide vaccines enhance the body’s capacity to recognize and effectively target them. The efficacy of cancer vaccines is closely linked to the recognition of tumor antigens by T lymphocytes. The ideal antigen for cancer vaccines should exhibit exclusive expression in cancer cells and possess high immunogenicity [151]. Peptide-based cancer vaccines typically consist of 20–30 amino acids containing specific epitopes from highly immunogenic antigens, aiming to induce the desired immune response. Compared to other vaccine types, peptide vaccines offer several benefits, particularly regarding safety and production simplicity [152]. The E75 peptide breast cancer vaccine (Her2 p369–p377) [153] with polyactin A has been demonstrated to increase CD4+ and CD8+ T lymphocytes, enhance splenocyte proliferation, and elevate interferon-γ levels [154]. Nelipepimut-S (E75, HER2/Neu, NeuVax) (SELLAS Life Sciences, New York, NY, USA) has been tested in clinical trials (NCT00841399 [106], NCT00091286 [107], NCT00791037 [107], NCT01532960 [108]). However, the Phase III clinical trial (NCT01479244) failed to show the difference in disease-free survival between Nelipepimut-S and placebo [109]. Combination therapies with trastuzumab Phase IIb (NCT01570036) showed clinical benefit in patients with triple-negative breast cancer (TNBC) [110].
2.2. Tumor-Homing Peptides
Tumor-homing peptides (THPs) are oligopeptides, usually consisting of 30 or fewer amino acids that are efficiently and specifically incorporated into tumor cells [155]. They are designed to be tumor cell-specific to enhance the internalization of small-molecule drugs or chemotherapeutic agents by creating PDC, enabling targeted delivery of therapeutic payloads to cancer cells [156]. Additionally, nanoparticles or liposomes can be modified with peptides to deliver the chemotherapeutic agents loaded in the nanoparticles/liposomes into tumor cells. [156]. CREKA (CREKA) can recognize clotted plasma proteins and selectively homes to tumors, where it binds to vessel walls and tumor stroma [40]. This peptide successfully amplified nanoparticles home to tumors in vivo [40].
RGD peptide (Arg-Gly-Asp), derived from integrin-binding motif from extracellular matrix proteins [157], specifically targets integrin αVβ3 receptors, which is highly expressed in several types of tumors. RGD is widely tested for potential use in diagnostic imaging [158,159] and therapies by conjugating with drugs or coating nanoparticles/liposomes [160]. The αv integrin-specific internalizing RGD (iRGD) (CRGD R/K GP D/E C) [161] (was fused with exosome membrane protein Lamp2 to make tumor-targeting exosome [162]. This strategy enabled effective and targeted delivery of drugs (e.g., doxorubicin) to breast cancer cells expressing αv integrin, resulting in the inhibition of tumor growth in mice [162]. The iRGD (Cend-1, Cend Therapeutics, San Diego, CA, USA) has been tested in clinical trials for the treatment of metastatic pancreatic cancer [111]. RGD peptides, including RGD-4C (ACDCRGDCFCG) peptide [41], c(RGDyK) [42], and small molecules Cilengitide™ (cRGDfV, EMD 121974) (ICENI Pharma, Edinburgh, UK) [163], target tumor vasculature by binding overexpressed αvβ3 integrin in the angiogenic endothelial cells and inhibit angiogenesis [164,165,166]. Cilengitide has been tested in clinical trials. However, a multicenter randomized open-label Phase III clinical trial (NCT00689221) failed to show any improvement. Thus, Cilengitide is not further developed as an anti-cancer [112].
THPs are also used for peptide receptor radionuclide therapy (PRRT), which combines a tumor-homing peptide with a radionuclide or radioactive isotope as the therapeutic substance [167]. Cholecystokinin (CCK) receptors bind to gastrin, a 33 amino acid peptide hormone, and CCK2 receptor is abundant in tumors [168]. Human colorectal and pancreatic tumors have been treated using 111In-labeled CCK8 and minigastrin (LEEEEEAYGWMDF), a peptide specifically designed to target CCK-2 receptors, in mice [43].
PRRT is used in clinics. [177Lu]-DOTATATE (Lutathera) (Novartis, Basel, Switzerland) (Table 3 for peptides in clinics) was approved by FDA in 2018 for PRRT for gastroenteropancreatic neuroendocrine tumors (GEP-NET) after showing improved survival in NETTER-1 Phase III clinical trial (NCT01578239) [169,170]. DOTATATE [DOTA-(Tyr3)-octreotate] is an 8 aa cyclic disulfide peptide with a covalently bonded DOTA bifunctional chelator [2,2′,2″,2‴-(1,4,7,10-tetraazacyclododecane-1,4,7,10-tetral) tetraacetic acid, tetraxetan]. The peptide octreotate (octreotide acid) mimics natural somatostatin targeting the somatostatin receptors (SSTR).
Another example of recently approved PRRT is [177Lu] Lu-PSMA-617 (Pluvicto) (Novartis), which targets prostate-specific membrane antigen (PSMA), thus showing efficacy in inhibiting prostate cancer growth [171,172].


Table 3.
Peptides in clinics for cancer treatment and diagnosis.
Table 3.
Peptides in clinics for cancer treatment and diagnosis.
| Name | Company | Year | Targets | Used for | Refs. |
|---|---|---|---|---|---|
| Leuprorelin, Lupron, Viadur, Eligard, Fensolvi | Abbott Laboratories, Abbott Park, IL, USA | 1985 | GnRH receptor | Prostate cancer, breast cancer | [173,174] |
| Goserelin, Zoladex | TerSera therapeutics, Deerfield, IL, USA | 1997 | GnRH receptor | Prostate cancer, breast cancer, endometriosis | [175,176] |
| Octreotide, Sandostatin | Novartis, Basel, Switzerland | 1998 | Reduction of growth hormone | treat diarrhea or diarrhea associated with some types of cancer | [177] |
| Cetrorelix, Cetrotide | Merck Serono, Darmstadt, Germany | 2000 | GnRH receptor | In vitro fertilization | [175] |
| Abarelix, Plenaxis | Praecis Pharmaceuticals, Waltham, MA, USA | 2003 | GnRH receptor | Advanced prostate cancer | [9] |
| Degarelix, Firmagon | Ferring pharmaceuticals, Saint-Prex, Switzerland | 2008 | GnRH receptor | Advanced prostate cancer | [173,175,178] |
| Carfilzomib, Kyprolis | Onyx Pharmaceuticals, Newbury Park, CA, USA, and Amgen, Thousand Oaks, CA, USA | 2012 | Proteasome inhibitor | multiple myeloma | [179] |
| Netspot, 68Ga DOTATATE | Novartis, Basel, Switzerland | 2016 | SSTR | PET diagnostics of neuroendocrine tumor | [180,181] |
| Lutathera, 177Lu-DOTATATE | Novartis, Basel, Switzerland | 2018 | SSTR | PRRT for gastroenteropancreatic neuroendocrine tumors | [169,170] |
| Edotreotide gallium, 68Ga-DOTATOC | ITM Radiopharma, München, Germany | 2019 | SSTR | PET for neuroendocrine tumors | [182] |
| Detectnet, 64Cu-DOTATATE | RadioMedix, Houston, TX, USA | 2020 | SSTR | PET for neuroendocrine tumors | [183,184] |
| Gallium gozetotide, 68Ga-PSMA-11 | Novartis, Basel, Switzerland | 2020 | PSMA | PET for recurrent prostate cancer | [185,186,187] |
| Pylarify, piflufolastat F18, | Lantheus, Billerica, MA, USA | 2021 | PSMA | PET for recurrent prostate cancer | [188,189] |
| Pluvicto, 177Lu-PSMA-617 | Novartis, Basel, Switzerland | 2022 | PSMA | PRRT for metastatic castration-resistant prostate cancer | [171,172] |
SSTR: somatostatin receptors, PSMA: the prostate-specific membrane antigen.
3. Peptide Design Strategies
In recent decades, there has been a notable increase in the availability of peptide drugs in the market. However, peptide drugs face challenges in formulation and delivery compared to small molecules, limiting their development. Factors such as shorter circulation half-lives, lower cell permeability, enzymatic degradation, and limitations in oral delivery hinder the efficient administration and absorption of therapeutic peptides [190,191]. Indeed, therapeutic peptides offer advantages such as high target specificity and low toxicity, making them promising candidates for drug development [192]. Overcoming the current limitations associated with their formulation and delivery would unlock their full potential, leading to the development of safer and more effective drugs. By improving the delivery mechanisms, enhancing stability, prolonging circulation half-lives, and optimizing routes of administration, the limitations of therapeutic peptides can be addressed, resulting in the development of advanced cancer therapies with improved clinical outcomes. Various design strategies have been developed to improve the effectiveness of bioactive peptides [193].
3.1. Cell-Penetrating Peptides
Cell-penetrating peptides (CPPs) are short peptides containing fewer than 30 amino acid residues, with a high content of basic amino acids like arginine and lysine. These peptides possess the remarkable ability to transport various cargo across cellular membranes while maintaining their functional integrity. CPPs can be utilized as either ACPs or THPs [194].
The first CPP, human immunodeficiency virus (HIV) TAT, can deliver the biologically active fusion protein to all tissue in mice [44]. Penetratin is a 16 aa CPP derived from Drosophila Antennapedia homeodomain and widely used in preclinical settings [45,46]. Transportan is a CPP derived from galanin, a natural peptide distributed throughout the nervous system [47].
The aforementioned p28 is also a CPP, which preferentially penetrates into cancer cells [16]. CPP p28 enhances the cytotoxic activity of temozolomide in the glioblastoma multiforme model [195]. Additionally, p28 can cross the blood–brain barrier (BBB) to enhance the efficacy of DNA-damaging agents by activating the p53-p21 axis [196].
3.2. Peptide Cyclization
Peptide cyclization involves the transformation of linear peptides into cyclic peptides, which helps mitigate proteolytic degradation caused by amino- and carboxypeptidases. This process masks both the N-terminal amino group and the C-terminal carboxyl group, resulting in enhanced stability against enzymatic degradation. Cyclic peptides also have a limited number of conformations in solution, allowing them to bind more efficiently to the active site of the desired target [197]. For example, cyclic peptides with the sequence cNGXGXXc, specifically targeting integrin α3β1, were found to enhance cell adhesion by selectively binding to the over-expressed integrin α3β1 in non-small lung cancer cells [50].
Cyclic peptides have been used for tough targets such as the Ras family GTPases [198]. NKTP-3, a cyclic D-peptide, inhibited the growth of A427 cells carrying the KRASG12D mutation by specifically targeting NRP1 and KRASG12D. It also exhibited potent antitumor activity in xenograft models derived from A427 cells and primary lung cancer models driven by KRASG12D, all while displaying no apparent toxicity [51]. KRpep-2d (Takeda, Tokyo, Japan) was developed from screening of cyclic peptides on phage displaying interaction to recombinant K-RasG12D [52]. LUNA18 (Chugai Pharmaceutical, Tokyo, Japan) is also a cyclic peptide targeting KRAS [105]. LUNA18 is currently tested in a Phase I clinical trial for solid tumors (NCT05012618) and is likely for progressing into Phase II [199].
3.3. Manipulation of the Amino Acid Sequence
Replacing partly or fully the L-amino acids with D-amino acids in peptide structures is a viable approach to enhance stability and decrease immunogenicity [200]. Examples include octreotide, a modified version of somatostatin with all L-amino acids replaced by D-amino acids, resulting in increased enzymatic stability and a longer plasma half-life [201]. Octreotide (Sandostatin) (Novartis) was first approved by FDA to reduce growth hormone in patients with acromegaly in 1998. Octreotide is used in clinics to treat the symptoms associated with metastatic carcinoid tumors (flushing and diarrhea) and vasoactive intestinal peptide (VIP) secreting adenomas (watery diarrhea). Octreotide acid (octreotate) is used as [177Lu]-DOTATATE (Lutathera) (Novartis) for PRRT in clinics. Octreotide conjugated with DOTA, DOTATOC (DOTA-[Tyr3]-octreotide), is extensively tested in clinical trials for diagnostic imaging as described in Section 4.2.
The use of D-amino acids in antifouling peptide biosensors and antimicrobial peptides has shown enhanced stability and activity. However, complete substitution with D-amino acids may lead to reduction in vivo activity and potential toxicity [202,203].
3.4. Peptides Conjugated with Polymers
The bioavailability and stability of peptide drugs can be improved through polymer–peptide conjugations. This approach involves attaching therapeutic peptides to polymers such as polyethylene glycol (PEG), poly(amidoamine) (PAMAM), poly(β-amino ester) (PAE), and natural polysaccharides, which leads to nanoscale self-assemblies and larger sizes that prevent renal filtration [204,205,206]. For example, A 40 kD PEG linked to the HVGGSSV peptide efficiently targets Tax-Interacting Protein 1 (TIP1), which is known to be overexpressed in human-invasive breast cancer cells [53,54].
3.5. Peptide-Assembled Nanoparticles
Peptides can be designed to self-assemble or be combined with polymeric molecules to create nanoparticles through non-covalent bonds. These nanoparticles have demonstrated attractive properties, including improved recognition of targeted cells, responsiveness to microenvironments, facilitation of internalization, and enhanced therapeutic effects [207]. X4-2-6, a PEG-modified 24-amino acid peptide analog of the second transmembrane helix of CXCR4, forms nanoparticles that inhibit CXCR4 function, prevent bone metastasis, and serve as a drug delivery system [55].
4. Peptides in Applications
4.1. Routes of Administration
Recent advancements in biopharmaceutical engineering have resulted in the development of various drugs based on peptides or proteins [7,167,194,208,209]. The route of drug administration significantly affects its effectiveness as a treatment [210,211]. Currently, several peptide-based drugs are on the market. For instance, Leuprorelin (Abbott Laboratories, Abbott Park, IL, USA), Goserelin (TerSera therapeutics, Deerfield, IL, USA), and Cetrorelix (Merck Serono, Darmstadt, Germany) have been used for breast cancer and prostate cancer patients. They are designed as hormone analogs and rapidly absorbed following subcutaneous injection [173,174,175,176]. Although the traditional route of administering protein and peptide-based drugs using a needle and syringe is commonly practiced, it has certain limitations, such as patient comfort, cost, sterility, etc. In this section, we review alternative routes that have been proposed for peptides or proteins (Figure 2).
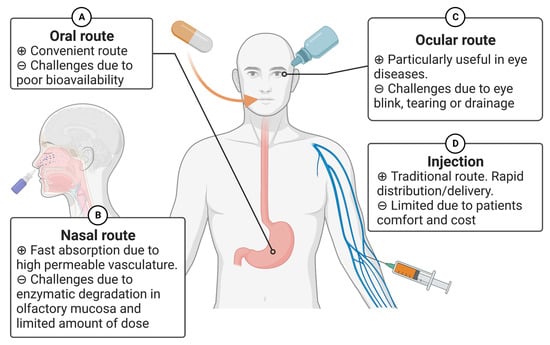
Figure 2.
Routes of administration in humans. Comparison of four different routes of drug administration: (A) oral, (B) nasal, (C) ocular and (D) intravenous injection.
4.1.1. Oral Route
This is one of the most convenient routes for administration of common drugs (Figure 2A). However, the administration of peptide drugs through this route poses challenges due to poor membrane permeability, denaturation of peptides due to an acidic environment in the stomach, and the susceptibility of peptides to enzymatic degradation in the gastrointestinal tract (GIT) [212,213]. In general, as the molecular weight increases, the permeability decreases. To overcome these limitations, several formulation technologies have been proposed. For example, to enhance the stability of peptide- or protein-based drugs, co-administered enzyme inhibitor(s) have been studied. Codelivery of therapeutic peptides with enzyme/protease inhibitors can inhibit degradation in the intestinal lumen, leading to greater absorption and bioavailability. Some examples of such inhibitors are soybean trypsin inhibitors, aprotinin, and leupeptin [214]. Aprotinin is currently withdrawn from the market due to the negative mortality trend [215]. To improve therapeutic peptide stability without undesirable adverse effects, it is crucial to select appropriate protease inhibitors based on the sequence/structure of therapeutic peptide drugs and to optimize a balance between the efficacy and safety of peptidase inhibitors.
The other type of co-administrative materials for peptide oral delivery have also been studied. Chitosan nanoparticles are proposed to use as a carrier for orally delivering peptides and vaccines. They possess remarkable biocompatibility, controlled release of peptides, and promote absorption in the GIT. Also, enclosed peptides within these nanoparticles can endure enzymatic degradation and withstand different pH conditions [216].
Various types of compounds with diverse chemical properties, including surfactants, fatty acids, medium chain glycerides, steroidal detergents, acylcarnitine and alkanoylcholines, N-acetylated α-amino acids and N-acetylated non-α-amino acids, and mucoadhesive polymers have been studied to enhance the intestinal absorption of large polypeptide drugs. Although some of these have been tested in clinical trials [217], the challenge of low bioavailability in peptide drugs persists, and a comprehensive breakthrough with wide-ranging applicability to various peptides has not been fully achieved yet.
4.1.2. Nasal Route
The epithelium of the nose is loosely packed with high permeable vasculature. This route can deliver peptides by transport mechanisms, including passive diffusion, carrier-mediated transport, and transcytosis [208,218,219] (Figure 2B). In general, peptides administered via the nasal route demonstrate increased permeation and faster absorption when peptides have a lower molecular weight. Clinically, IM862 (Cytran Inc., Kirkland WA, USA), a synthetic dipeptide (L-glutamine L-tryptophan), was previously tested. IM862 is a naturally occurring peptide with antiangiogenic properties by reducing the production of VEGF [220,221]. In the animal models, the antitumor activity of IM862 was similar, irrespective of the route of administration, including intranasal, subcutaneous, intravenous, and intramuscular [117]. Due to its convenient administration and relatively high bioavailability (71%) after intranasal administration, the nasal route was chosen for human trials Phase III (NCT00002445) [117].
Despite the potential of this delivery route, there are several challenges associated with the nasal route of administration for peptides or proteins [219]. After administration, bioavailability is significantly affected by the peptides’ surface, size, lipophilicity, and pI (isoelectric point). The enzymatic degradation in the mammalian olfactory mucosa is also an important factor [219]. The limitation of this route is associated with the restricted amount of dosage that can be administered. Consequently, it still requires the development and formulation of a drug with a high dose capacity [222]. In spite of such limitations, several advantages of this pain-free and non-invasive administration are evident. Hence, delivering the peptides through the nasal route is a potential alternative to drug delivery strategies.
4.1.3. Ocular Route
This route is particularly useful in treating ocular malignant tumors such as intraocular melanoma (uveal) (Figure 2C) [192]. Drug administration through the eye presents challenges due to its natural processes, such as blinking, tearing, and drainage.
As mentioned above, the bioavailability of peptides or proteins is generally low in most of the non-invasive routes (e.g., oral and nasal) [223]. The other current limitation in delivering peptide- and protein-based drugs through noninvasive routes may arise from the considerable expense, which could restrict the number of economically feasible options. Although the dosage form of the non-invasive routes can be self-administered by patients, the manufacturing cost of peptide and protein drugs would be less compared to the traditional injections at healthcare facilities. While considerable progress has been made previously, further advancements in formulation technology, such as the development of new penetration enhancers, enzyme inhibitors, etc., are still needed.
4.2. Diagnostics—Imaging
Cancer detection and management have been facilitated by the utilization of various imaging modalities. Molecular imaging with targeted probes or contrast agents has proven to be a valuable tool in diagnosing various types of cancer. Advances in molecular imaging technology may increase the precision with which therapies can be implemented [224]. However, the efficacy of molecular imaging is dependent on the imaging modalities and probes/contrast agents employed to specifically target, detect, and visualize cancer biomarkers. Typical imaging modalities in oncology include magnetic resonance imaging (MRI), positron emission tomography (PET), computed tomography (CT), optical fluorescence imaging, and ultrasound sonography. Imaging probes or contrast agents are predominantly composed of target-specific molecules that are designed to recognize and bind to tumors, enabling their visibility in various imaging modalities [225]. They can also participate in metabolic pathways. In many cases, they are labeled with metals, radioactive, or fluorescent materials. Although hardware such as highly sensitive detection sensors or scanners are also important factors in molecular imaging, in this review, we focus on imaging probes, particularly peptide-based agents on MRI, and nuclear medicine techniques such as single-photon emission computed tomography (SPECT) and PET, and optical/near-infrared (NIR) imaging.
4.2.1. MRI
MRI utilizes a high magnetic field and creates images by applying specific radiofrequency pulses, resulting in distinct signal patterns across various tissues. These patterns depend on the composition of the tissues, specifically the types and concentrations of nuclei within them. MRI is a powerful non-invasive imaging technique that takes advantage of a very high spatial and temporal resolution and can provide detailed molecular/cellular information when combined with a contrast agent. MRI contrast agents are molecules or particles that influence the relaxation of water protons. They can be T1- or T2-weighted, affecting the longitudinal or transverse relaxation times, respectively. T1-weighted contrast agents are mostly small gadolinium (Gd3+) or manganese (Mn2+) paramagnetic complexes, while the majority of the T2-weighted ones are iron oxide-based superparamagnetic nanoparticles [226,227]. Due to the lack of sensitivity of MRI, relatively high concentrations of a contrast agent (mM range) need to be injected to generate local variation in the signal intensity. To achieve a specific binding to the target biomarker, different approaches (e.g., nanoparticles, antibodies, or peptides) have been studied [228,229,230,231]. In general, the use of peptides instead of small molecules to target the same biomarker leads to stronger binding affinity and selectivity [232,233]. In addition, compared to proteins, peptides have advantages such as low immunogenicity, more resistance to enzymatic degradation, favorable pharmacokinetics, and biodistribution [232,234].
Gadolinium (III) is often used as an MRI contrast agent since it has seven unpaired electrons and a high magnetic moment [235]. Presently, to avoid free Gd (III) and nephrotoxicity, the approved Gd contrast agents are chelated with a total of eight nitrogen and oxygen from the chelator function as Gd3+-binding ligands [236]. Two of the most common Gd-based contrast agents are Gd-DTPA (diethylene triamine pentaacetic acid, Magnevist) and Gd-DOTA (Dotarem) [237]. Although they are useful and have been used clinically, their elimination half-life is relatively short (~1.3 h in humans) [238]. In addition to delivery strategies, by attaching targeting moieties, optimizing the half-life and pharmacokinetic properties of these contrast agents can substantially improve the imaging quality and eliminate repeated dose injections. The modifications with several antibodies [e.g., targeting VEGF, epidermal growth factor receptor (EGFR), or human epidermal growth factor receptor 2 (HER2)] have been proposed to improve the MRI contrast agents [239]. The Gd modifications with peptides for MRI applications have also been studied. For instance, peptides conjugated with DTPA (Gd) or DOTA (Gd) by a PEG linker improved the activity suggesting their potential application as diagnostic agents for MRI [56,240,241]. Tumor-homing peptide, such as CREKA [40], is promising for detecting cancer by MRI in vivo [240,241].
4.2.2. SPECT
Radionuclides are unstable nuclides that emit α, β+/− charged particles, Auger electrons, and/or γ rays with radioactive decay processes. Radionuclides used in medical imaging can be categorized into two types based on their emissions; the first type consists of radionuclides that emit γ rays, which can be detected using SPECT. The second type includes radionuclides that primarily emit β+ rays, which can be detected through techniques such as PET [242]. Although the sensitivity, resolution, and fast acquisition of SPECT due to the solid-state detector technology provide a great impact on nuclear diagnosis [243], various carrier molecules to target tumors have been proposed. As proteins, radiolabeled trastuzumab and pertuzumab antibodies have shown high accumulation in tumor tissues [244]. In this case, the optimum timeframe for evaluating antibody-based imaging with favorable tumor-to-organ ratios is typically 3–5 days following administration. Given that the smaller molecular size can alter the clearance pathway and peptides have relatively shorter circulation time, peptides can be suitable molecules for imaging procedures [245]. Peptide-based probes for SPECT have been developed in several preclinical and clinical studies [246,247]. For example, RGD tri-amino acids peptide can specifically bind to the integrin αVβ3 receptors, which is highly expressed in several types of tumors. 99mTc-PEG4-E[PEG4-c(RGDfK)]2 (99mTc-3PRGD2) is proposed as an RGD containing SPECT radiotracer. Multicenter studies suggest that imaging with 99mTc-3PRGD2 is sensitive for the detection of lung malignancies [248].
4.2.3. PET
As noted above, the basic principle of PET is that proton-rich radionuclides decay by emitting positrons (β+), which subsequently travel a short distance and annihilate with an electron (β−) to create two 511-kilo electron volt photons that arise almost exactly 180 degrees apart [249]. The properties of some radionuclides that decay via β+ decay are shown in Table 4 [250,251].

Table 4.
Properties of commonly used PET radionuclides.
Similar to SPECT, peptides have been used as carrier molecules to deliver these radionuclides for PET. Various labeling methods have been designed [209]. In clinical studies, various peptides have been explored for tumor diagnosis [232,252]. For example, peptides targeting somatostatin receptors (SSTR), the prostate-specific membrane antigen (PSMA), integrins, chemokines, urokinase-type plasminogen activator receptors (uPAR), and cholecystokinin receptors (CCK2-R) serve as notable examples [253,254,255,256,257,258]. Among them, Netspot (68Ga-DOTATATE) (Novartis) [180,181], Edotreotide gallium (68Ga-DOTATOC) (ITM Radiopharma, München, Germany) [182,259], and Detectnet (64Cu-DOTATATE) (Radio-Medix, Houston, TX, USA) [183,184] target SSTR and approved by FDA for PET in 2016, 2019, and 2020, respectively. Gallium gozetotide (Locametz, Illuccix, 68Ga-PSMA-11) (Novartis) [185,186,187] and Pylarify (piflufolastat F18, 18F-DCFPyL) (Lantheus, Billerica, MA, USA) [188,189,260,261,262] targets PSMA and is approved by FDA in 2020 and 2022, respectively.
Radionuclides-labeled peptide probes are important tools in molecular imaging using SPECT and PET. Given the extensive exploration of novel labeling strategies and the ongoing optimization of associated peptides, the advancement of peptide-based tracers will continue to be a pivotal area of focus in imaging research with radionuclides.
4.2.4. Optical/NIR Imaging
Optical imaging incorporates multiple modalities, including bioluminescence imaging (BLI), fluorescence, and chemiluminescence [263]. They are often used in preclinical studies. Among them, fluorescence imaging has been used in clinical studies. In particular, fluorescent agents that are emitted in the NIR region (700–900 nm) enable sufficient depth of light penetration allowing for real-time surgical guidance. Imaging in the NIR region offers several advantages, such as the presence of minimal tissue absorbance, scattering, and autofluorescence. These unique features establish a minimal background, providing an ideal basis for incorporating tissue-specific contrast. Due to these properties, such as high sensitivity, contrast, and resolution, intraoperative fluorescence imaging is particularly well-studied for surgical applications. Accurate detection of visually hidden tumor lesions and tumor margins during surgery can lead to a significant impact on overall cancer survival/outcomes. Among various types of solid tumors, the highest positive margin rate following tumor resection in the US is oral cavity tumors [264]. Ovarian and prostate cancers had the highest positive margin rate prevalence in women and men, respectively. Bladder, thyroid, colorectal, kidney, lung cancer, and breast cancer are common types of tumors with the highest positive margin rates [264,265]. The substantial increase in the number of early-phase clinical trials for image-guided surgery with NIR agents reflects a significant potential to develop novel methods for visualizing and accurately identifying tumor margins in specific cancer types. Indocyanine green (ICG) is one of the most commonly used NIR dyes. While it has an excellent safety profile in humans, ICG is a non-specific agent that is rapidly cleared by the liver and excreted in bile [266,267]. Currently, most strategies for NIR dyes, including ICG, use conjugation technologies with targeting motifs such as peptides [268].
Because the positive margin rate following breast-conserving surgery (lumpectomy) is unignorably high, we developed a new near-infrared fluorescence imaging probe ICG-p28 by utilizing ICG labeled with the cell-penetrating peptide p28 carrying a tumor-targeting motif [16]. As described earlier in the sections of ACP and CPP, p28 crosses the BBB and selectively enters cancer cells in mouse models [195,196]. Due to such unique tumor-targeting properties, ICG-p28 was developed and tested in clinically relevant preclinical settings [269,270]. These studies demonstrated that intraoperative imaging with ICG-p28 accurately identified the tumor margins, improving tumor recurrence rate in multiple breast cancer animal models independent of the receptor expression status [269,270]. These results emphasize the importance of the imaging approach and its translational potential.
4.3. Targeting Delivery
Drug delivery is critically important for optimizing the efficacy of drugs while reducing toxic adverse effects. Several approaches have been proposed to deliver active compounds to the target sites [194,208]. In this section, we focus on peptide-based targeting delivery.
In passive targeting, peptides can accumulate at tumor sites by intrinsic characteristics of peptides such as size and charge and due to distinctive properties of the targeted sites such as local vasculature and lymphatic drainage. At tumor sites, nearby vasculature is generally leaky, and lymphatic drainage is impaired or absent [271]. Under such conditions, the enhanced permeation and retention (EPR) effect allows preferential accumulation of peptides within the tumor tissue. The concept of the EPR effect was first proposed in 1986 [272], and the EPR effect with a universal mechanism is a unique feature of solid tumors [273,274,275,276]. In hypoxic conditions or inflammation, blood vessels become more permeable. New blood vessels (neovascularization) in hypoxic tumors are generally leaky as they have large openings (200–2000 nm) [277,278].
In contrast, receptor-mediated approaches are active targeting strategies [279,280,281]. The abundant expression of peptide-binding receptors in human tumors highlights their potential as promising targets for selective anti-cancer drug delivery. Synthetic analog peptides of natural ligands are of significant interest as receptor-targeting entities due to their ability to exhibit high affinity, rapid internalization rates, and low immunogenicity. Several target candidates for this group of peptides, such as integrin receptors, HER2, and EGFR [280,281].
In addition, CPPs are also very efficient in delivering various molecules into cells [194]. Although the entry mechanism of CPPs into cells is still a matter of some debate, direct translocation and/or endocytotic cellular entry are often described as the entry mechanism of CPPs [282,283].
As described in the previous section, CPPs have significant potential to target delivery vehicles, and currently, ~2000 CPPs have been identified based on the CPPsite 2.0 database. An important challenge in cancer therapy involves the limitations of drug delivery to the target lesions by barriers such as the tumor microenvironment or the BBB. CPPs opened a new avenue to overcome such limitations.
4.4. Crossing the BBB with Peptides
The BBB is a unique microvasculature of the CNS, protecting the brain from harmful agents in circulation [284] (Figure 3). Due to the tightest protection by the BBB, drug delivery to the brain tumor is one of the big challenges of cancer therapies. Having tumor-targeting or vascular-targeting abilities of peptides, potential drug delivery to brain cancer (glioma and CNS lymphoma), and brain-metastasized cancer have been tested.
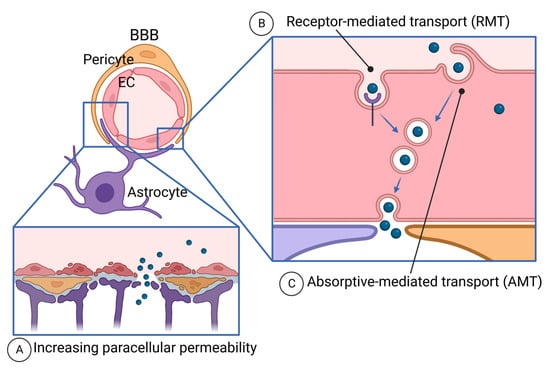
Figure 3.
Routes of drug delivery crossing the BBB. (A) The BBB is formed by a tightly connected EC layer covered by pericytes and astrocyte end foot. To deliver the drugs via paracellular routes, junctions are disrupted by various methods described in Section 4.4.1. (B) While keeping the barrier intact, drugs can be delivered by receptor-mediated transport by conjugating drugs with ligand-mimicking peptides, as described in Section 4.4.2. (C) Drugs can be delivered by absorptive-mediated transport. CPPs and BBB shuttles use this pathway as described in Section 4.4.3.
Drug delivery beyond the BBB is mediated by paracellular diffusion and transcellular route [285]. Paracellular diffusion is largely eliminated by tight junctions (TJs) and disruption of the BBB or temporal regulation of the BBB is necessary, as discussed below. The transcellular route uses the movement of molecules through the cells, passing through apical and basolateral membranes [286].
4.4.1. Paracellular Diffusion and the BBB Disruption
To facilitate drug delivery to the brain, BBB disruption (BBBD) has been tested since the 1970s in animal models and clinical trials [218,287] (Figure 3A). Bradykinin is an endogenous peptide that opens the BBB for small molecules, such as sodium fluorescein, but not for bigger molecules, such as albumin [288]. Bradykinin and its analog RMP-7 selectively induce permeability in tumor vasculature in the brain, not in the normal brain, as its B2 receptor is abundant in tumor vasculature [289,290]. RMP-7 (lobradimil and cereport) (Alkermes, Dublin, Ireland) has been tested as a combination with carboplatin in clinical trials [113,114,115,116]; however, it failed to show improvement of the efficacy of carboplatin in a randomized controlled Phase II trial [115]. RMP-7 is not currently being investigated for BBBD purposes [287].
Non-peptide molecules such as hypertonic mannitol [291,292,293,294], alkylglycerols [295], regadenoson [296,297], and other device-based strategies such as cranial implantable ultrasound [298,299], hyperthermia [300,301], low-level laser treatment (LLLT) [302], magnetic resonance-guided laser ablation (MRgLA) [303], MR-guided focused ultrasound (MRgFUS) [304,305] has been tested, but progress has not been satisfactory [287].
More recently, targeted approaches of reversible regulation of the barrier and more selectively to the BBB have been taken. The BBB is characterized by a tightly packed monolayer of non-fenestrated endothelial cells (ECs) connected by TJs, adherens junctions (AJs), and gap junctions [284]. Brain ECs have a higher expression of TJ proteins compared to arterial and venous ECs [306]. Especially, Claudin-5 has gained attention as a gatekeeper of the BBB [307]. A peptidomimetic derived from Claudin-5 induces transient BBB permeabilization and facilitates brain uptake of MRI contrast agents in mice [57].
Cadherins are important adherens junction proteins. E-cadherin-derived peptides (e.g., HAV6) induce endothelial permeability and inhibit resealing of tight junctions [60]. These E-cadherin-derived peptides facilitate drug delivery to the brain in animal models [58] and inhibit the growth of medulloblastoma in mice [59]. But these peptides also open junctions in intestinal epithelial cells in vitro [308].
4.4.2. Transcellular Route—Receptor-Mediated Transport (RMT)
The transcellular route of drug delivery is endocytosis-mediated transcytosis, which is absorptive-mediated transport (AMT) and receptor-mediated transport (RMT) (Figure 3B,C). AMT mediates the uptake of cationic molecules at the luminal surface of EC and the exocytosis of the molecules at the abluminal surface [309]. We will discuss this below regarding CPP. RMT is mediated by the interaction of ligands to the receptors on the luminal surface of the BBB, thereby, ligand-derived peptides have been extensively developed [310]. For this purpose, the receptors need to be abundantly expressed at the luminal surface of ECs, and selectively to the BBB [311,312,313]. Such receptors include the low-density lipoprotein receptor (LDLRs) [314], transferrin receptor (TfR1) [315], insulin receptor [316], and leptin receptor [317,318].
Among them, LDLR has been most studied for RMT [319,320]. LDLR plays an important role in lipoprotein transport across the BBB for the delivery of essential lipids to the brain [314]. Their natural protein ligands are apolipoproteins (Apo) A [321], ApoB [322], and ApoE [323]. The peptides derived from ApoB and ApoE have been tested for drug delivery across the BBB [61,322,323,324,325].
LDLR-related proteins (LRPs) mediate transcytosis of lactoferrin [326], melanotransferrin (p97) [64,327], receptor-associated protein (RAP) [328], tissue plasminogen activator (tPA) [329], and β-amyloid precursor (APP) [330]. Aprotinin is known to interact with LRPs [331] and 19 amino acid peptide Angiopep-2 (AngioChem Inc., Montreal, QC, Canada) was designed from the LRP-binding domain of aprotinin [332,333]. Angiopep-2 was conjugated with FDA-approved chemotherapeutic agents, such as doxorubicin (ANG1007) [334], etoposide (ANG1009) [334], paclitaxel (ANG1005) [335], and bioactive peptides [336]. Its conjugate with paclitaxel (ANG1005 or GRN1005) [337] showed good tolerance in Phase I clinical trials [118,119] and tested for the treatment with recurrent high-grade glioma in combination with bevacizumab (NCT01480583). Dual-targeting of liposomes modified with Angiopep-2 and tumor-targeting peptide derived from neuropilin (tLyP-1) showed successful drug delivery of VEGF siRNA and docetaxel for glioma-bearing mice [338].
Other peptides binding to LDLRs are the ApoE-derived peptide K16ApoE [61], AEP [62], RAP-derived peptide RAP12 [63], and melanotransferrin (MTf)-derived peptide [64]. Screening of peptides using phage display has been applied to find the peptides binding to receptors. Peptide-22 (VH434), found by phage display, interacts with LDLR and facilitates drug delivery to the brain in animal models [49,65]. Other peptides found by phage display for binding to LDLR or LRP1 are L57 [66], M1 [67], and LRPep2 [68].
Phage display has been widely used to define TfR-binding peptides. TfR-T12 and T7 were identified by phage display [69] and tested for drug delivery to the brains of rodents [339,340] and zebrafish [341]. The other peptides found in the phage display are B6 [70], CRT peptide [71], and NanoLigand Carriers (NLC) [72]. TfRB1G3 was designed from natural peptides called Cystine-dense peptides (CDPs), a mini-protein class with high affinity and low immunogenicity [342].
Leptin receptor is highly expressed on the BBB [317,318]. In the same year, Leptin30 (aa 82–111 of human leptin P41159) [74] and peptide Lep70–89 (aa 91–110 of P41159) were defined as a brain-targeting peptide from leptin. Lep70–89-modified liposomes exhibit cellular uptake via macropinocytosis in mouse brain endothelial cells [343]. Leptin30 showed gene delivery to the brain in mice [73]. Another peptide, g21 (aa 33–53 of mouse leptin P41160), also facilitates the delivery of nanoparticles modified with g21 to the brain in rats [75].
RGD peptide, the most widely studied adhesive peptide found as an integrin-binding motif from extracellular matrix proteins [157], also provides targeted delivery to tumor vasculature as the αvβ3 integrin is highly expressed in angiogenic endothelial cells in tumors [344,345,346,347]. Cyclic RGD (cRGD) or stapled RGD (sRGD) have been used to modify micelles or liposomes and have shown accumulation in orthotopic glioma in mice [348,349]. Liposomes modified with cRGD could deliver anti-cancer drugs such as paclitaxel, oxaliplatin, and doxorubicin across the BBB, inhibited glioma growth, and improved the survival of glioma-bearing mice [348,349,350]. Such tumor-targeting and brain-penetrating property of RGD peptide has been tested as imaging agents in clinical trials [351]. 18F Fluciclatide (AH111585) (GE Healthcare, Chicago, IL, USA) was tested for PET imaging following i.v. injection to detect solid tumors, including high-grade glioma, in the Phase II trial (NCT00565721). 18F-RGD-K5 (Siemens Molecular Imaging Inc., Knoxville, TN, USA) is also an RGD-based radiotracer for PET imaging. Biodistribution and safety of i.v.-injected 18F-RGD-K5 was tested in monkeys and humans in Early Phase I study (NCT00743353) [120]. Other RGD-based radiotracers tested in Early Phase I for PET/CT imaging are 68Ga-BNOTA-PRGD2 (NCT01806675), 18F-FPPRGD2 (NCT01806675), and 68Ga-RM26-RGD (NCT05549024).
For therapeutic purposes, DNX-2401 (Delta-24-RGD, tasadenoturev) (DNAtrix, Inc., Houston, TX, USA) has been tested for glioma patients in clinical trials. DNX-2401 is a tumor-selective, replication-competent oncolytic adenovirus. A Phase I, dose-escalation, biologic-end-point clinical trial was conducted by intratumoral injection (NCT00805376) [127]. In another Phase I trial (NCT01582516), DNX-2401 was administered by convection-enhanced delivery (CED), which is a local drug delivery to bypass the BBB. Successful tumor targeting and safety in the brain were confirmed [125]. In the Phase I trial (NCT03178032), infusion of DNX-2401 through a catheter placed in the cerebellar peduncle followed by radiotherapy showed promising reduction/stabilization of tumor size of pediatric patients with diffuse intrinsic pontine glioma (DIPG) [128]. DNX-2401 was further tested in the Phase I trial by intra-arterial infusion (NCT 03896568) [129], and the Phase I/II study as a combination of intratumoral delivery of DNX-2401 followed by i.v. anti-PD-1 antibody pembrolizumab in recurrent glioblastoma (NCT02798406) [126]. Although RGD potentially crosses the BBB, the BBB penetration is not the focus of these studies.
VEGFR2 and Neuropilin-1 (NRP1) are important co-receptors for VEGF to mediate angiogenesis. A peptide screened for binding to the VEGF receptors, A7R [77] interacts with NRP1 but not with NRP2 or VEGFR2 [352,353]. Glycosylated A7R derivative is stable in serum, able to cross the BBB to deliver paclitaxel to glioma in mice, and improves survival of glioma-bearing mice [76].
Interleukin (IL)-13 receptor (IL-13R) α2 is highly expressed in glioma, thus is considered as a target for drug delivery. A peptide derived from IL-13 (IL-13p) is used to modify docetaxel-loaded nanoparticles and showed suppression of the growth of s.c. glioma in mice [78]. Phage display found another peptide, Pep-1, which binds to IL-13Rα2 [79]. Following i.v. injection, Pep-1 homes to both s.c. and orthotopic GBM xenografts in mice [79] and facilitates delivery of chemotherapy agent, cilengitide (CGL) loaded in Pep-1 conjugated liposome, to suppress the growth of s.c. glioma [80].
A peptide G7 was derived from opioid peptide MMP-2200 [81,82]. G7-modified nanoparticles (NPs) cross the BBB by several endocytotic vesicles and macropinocytotic processes [81].
Neurotropic viruses, snake neurotoxins, and bee venoms have received attention as they interfere with brain cells. Rabies virus glycoprotein (RVG) interacts with nicotinic acetylcholine receptor (nAChR) on neuronal cells to enable viral entry into neuronal cells [354]. 29 mer RVG-derived peptide [355] was conjugated with 9R to enable siRNA binding (RVG-9R), which enabled the delivery of siRNA to the brain [83]. An independently identified 43 mer RVG-derived peptide (RDP) [84] and 39 mer RDP [85] also successfully delivered the fused protein to the brain in mice. KC2S is a synthetic peptide derived from snake neurotoxins that bind to nAChRs [86]. Paclitaxel-encapsulated KC2S-micelles afforded robust inhibition of intracranial glioblastoma in mice [86]. CDX is derived from snake neurotoxin candoxin, which also binds to nAChR [87]. CDX-micelle could deliver paclitaxel and improve the survival of glioblastoma-bearing mice [87]. To improve the stability, replacing with D amino acid (resulting in DCDX) could deliver doxorubicin for glioblastoma-bearing mice [88]. Apamin is a peptide found in bee venom, crossing the BBB [356,357], and MiniAp-4 is a shorter peptide derived from apamin [89].
Several peptide shuttles have been found through in vivo phage display biopanning without aiming for a particular receptor. The most prominent example is that of TGN [90]. This sequence is actively transported across brain endothelial cells, and its brain selectivity suggests that the mechanism is receptor mediated. The brain delivery capacity of this shuttle is supported by enhanced therapeutic effects in glioblastoma and Alzheimer’s mouse models [358,359].
4.4.3. Transcellular Route—Absorptive-Mediated Transport (AMT) and Brain-Penetrant Peptides
The mechanisms of drug delivery to the brain using cell-permeable peptides (CPPs) are not fully understood. It is believed that cationic CPPs bind to the luminal surface of the EC membrane and are endocytosed via AMT and exocytosed at the abluminal surface [309] (Figure 3C). Some CPPs have the ability to penetrate the BBB and are used for drug delivery [360,361].
TAT can deliver the biologically active fusion protein to all tissues in mice, including the brain, beyond the BBB [362,363]. After that, more selective CPPs were found and tested for drug delivery.
Penetratin is a 16 aa CPP, derived from the Drosophila Antennapedia homeodomain [45,46]. Doxorubicin conjugated with penetratin [91] or encapsulating receptor-targeted liposomes modified with transferrin and penetratin [361] cross the BBB after i.v. injection in rodents.
Transportan is a CPP derived from galanin, a natural peptide distributed throughout the nervous system [47]. M13 is a derivative of transportan, showing the delivery of M13-conjugated cisplatin into the brain in GBM-bearing mice [48].
Protegrins are small peptides with antimicrobial activity found in porcine leukocytes [364]. SynB1 and SynB3 are peptides derived from Progrin-1 (PG1) and can deliver conjugated doxorubicin to the brain via absorptive-mediated transport [365,366].
Glutathione crosses the BBB via carrier-mediated transport [367]. Glutathione PEGylated (GSH-PEG) liposomes (G-Technology) [368,369]and glutathione-coated nanoparticles [370] have been used for drug delivery across the BBB. 2B3-101 (2-BBB, Leiden, Netherlands) is a glutathione PEGylated liposomal methylprednisolone, an anti-inflammation drug, showing efficacy in reducing the severity of encephalomyelitis in mice [130]. 2B3-101 is further tested in an Open-label, Phase I/IIa, dose-escalating study in patients with solid tumors, brain metastases, or recurrent malignant glioma (NCT01386580).
A peptide CAQK, found by in vivo phage display screening in mice with acute brain injury, interacts with chondroitin sulfate proteoglycans, upregulated in the injured brain [92]. CAQK-coated nanoparticle successfully delivered siRNA to the injured site in the brain after i.v. injection [92]. Peptides interacting with lipids on the membrane can also mediate drug delivery to the brain. Gangliosides are glycosphingolipids, abundant in neuronal cells. G23 peptide, found by phage display targeting gangliosides [93,94], could promote the transport of nanoparticles across the BBB, and provide a targeting effect [371].
A peptide named PepH3, derived from Dengue virus type 2 capsid protein (DEN2C), crosses the BBB by receptor-independent AMT [95]. Other peptides promising for drug delivery across the BBB are N-methyl phenylalanine-rich peptide [96], phenyl proline tetrapeptide [97], non-canonical anionic peptide NegPep [98], Porphyrin [99], and a neurofilament-derived peptide NFL-TBS.40–63 [100,101].
Finally, bypassing the BBB by intratumoral injection, implantation of drug-releasing polymers, convection-enhanced drug delivery [372], and intranasal delivery [222,373]. Low-molecular-weight protamine (LMWP) is a cell-penetrating peptide used as a conjugate with nanoparticles to facilitate drug delivery to the brain via intranasal administration in rats [102].
5. Future Perspectives
In this review, we described peptide-based approaches for cancer treatment. Many of these peptides were identified from natural sources such as proteins from bacteria, plants, or animals/humans since nature generates functional materials in living systems in the form of proteins and peptides. Although they are quite useful, artificial intelligence (AI) and machine learning-based strategies without human bias have the potential to revolutionize and accelerate the discovery and design of peptide-based drugs. In recent years, AI and deep machine learning-based strategies have shown immense potential in the medical field (e.g., drug discovery) [374,375]. These models have the capability to generate data that extends beyond what we have in the training samples, enabling an effective and fast tool for exploring the extensive search space of high-dimensional data, such as peptide/protein sequences. Although we are currently in the process of enhancing our understanding of utilizing AI safely and its widespread adoption in clinical settings currently remains limited, a new generation of peptide-based agents may soon be among the most important elements in clinical management as the utilization of AI is rapidly developing and appeared to help to minimize human errors.
6. Conclusions
In this review, we reviewed original research articles, recent review articles, and information from ClinicalTrial.gov to summarize the uses of peptide-based agents as anti-cancer therapeutics, drug delivery as peptide-drug conjugates or modifying liposomes/nanoparticles, and diagnostics imaging. New strategies to develop and design peptides are still being tested in preclinical models for future development of peptide-based diagnostics or therapies for cancer patients [7,9,194,376,377].
Author Contributions
Conceptualization, N.T.T.N., T.Y. and K.H.Y.; writing—original draft preparation, N.T.T.N., T.Y. and K.H.Y.; writing—review and editing, N.T.T.N., T.Y. and K.H.Y.; funding acquisition, T.Y. and K.H.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported in part by the National Institutes of Health/National Cancer Institute (R21CA252370, R21CA280814, and R01CA272564) and the National Institutes of Health/National Institute of Biomedical Imaging and Bioengineering (R01EB023924) to T.Y. and the National Institutes of Health/National Eye Institute (R01EY029339) to K.H.Y.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Dagenais, G.R.; Leong, D.P.; Rangarajan, S.; Lanas, F.; Lopez-Jaramillo, P.; Gupta, R.; Diaz, R.; Avezum, A.; Oliveira, G.B.F.; Wielgosz, A.; et al. Variations in common diseases, hospital admissions, and deaths in middle-aged adults in 21 countries from five continents (PURE): A prospective cohort study. Lancet 2020, 395, 785–794. [Google Scholar] [CrossRef] [PubMed]
- Marqus, S.; Pirogova, E.; Piva, T.J. Evaluation of the use of therapeutic peptides for cancer treatment. J. Biomed. Sci. 2017, 24, 21. [Google Scholar] [CrossRef] [PubMed]
- Faraji, N.; Arab, S.S.; Doustmohammadi, A.; Daly, N.L.; Khosroushahi, A.Y. ApInAPDB: A database of apoptosis-inducing anticancer peptides. Sci. Rep. 2022, 12, 21341. [Google Scholar] [CrossRef]
- Zhang, X.X.; Eden, H.S.; Chen, X. Peptides in cancer nanomedicine: Drug carriers, targeting ligands and protease substrates. J. Control. Release 2012, 159, 2–13. [Google Scholar] [CrossRef]
- Tyagi, A.; Tuknait, A.; Anand, P.; Gupta, S.; Sharma, M.; Mathur, D.; Joshi, A.; Singh, S.; Gautam, A.; Raghava, G.P. CancerPPD: A database of anticancer peptides and proteins. Nucleic Acids Res. 2015, 43, D837–D843. [Google Scholar] [CrossRef]
- Karami Fath, M.; Babakhaniyan, K.; Zokaei, M.; Yaghoubian, A.; Akbari, S.; Khorsandi, M.; Soofi, A.; Nabi-Afjadi, M.; Zalpoor, H.; Jalalifar, F.; et al. Anti-cancer peptide-based therapeutic strategies in solid tumors. Cell. Mol. Biol. Lett. 2022, 27, 33. [Google Scholar] [CrossRef]
- Blanco-Miguez, A.; Gutiérrez-Jácome, A.; Pérez-Pérez, M.; Pérez-Rodríguez, G.; Catalán-García, S.; Fdez-Riverola, F.; Lourenço, A.; Sánchez, B. From amino acid sequence to bioactivity: The biomedical potential of antitumor peptides. Protein. Sci. 2016, 25, 1084–1095. [Google Scholar] [CrossRef]
- Wang, L.; Wang, N.; Zhang, W.; Cheng, X.; Yan, Z.; Shao, G.; Wang, X.; Wang, R.; Fu, C. Therapeutic peptides: Current applications and future directions. Signal Transduct. Target. Ther. 2022, 7, 48. [Google Scholar] [CrossRef]
- Chiangjong, W.; Chutipongtanate, S.; Hongeng, S. Anticancer peptide: Physicochemical property, functional aspect and trend in clinical application (Review). Int. J. Oncol. 2020, 57, 678–696. [Google Scholar] [CrossRef]
- Baig, S.; Seevasant, I.; Mohamad, J.; Mukheem, A.; Huri, H.Z.; Kamarul, T. Potential of apoptotic pathway-targeted cancer therapeutic research: Where do we stand? Cell Death Dis. 2016, 7, e2058. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, C.M.; Singh, A.T.K. Apoptosis: A Target for Anticancer Therapy. Int. J. Mol. Sci. 2018, 19, 448. [Google Scholar] [CrossRef] [PubMed]
- Montero, J.; Letai, A. Why do BCL-2 inhibitors work and where should we use them in the clinic? Cell Death Differ. 2018, 25, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Najm, A.A.K.; Azfaralariff, A.; Dyari, H.R.E.; Othman, B.A.; Shahid, M.; Khalili, N.; Law, D.; Alwi, S.S.S.; Fazry, S. Anti-breast cancer synthetic peptides derived from the Anabas testudineus skin mucus fractions. Sci. Rep. 2021, 11, 23182. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.M.; Tseng, C.-C.; Chen, N.-F.; Tai, M.-H.; Hung, H.-C.; Feng, C.-W.; Cheng, S.-Y.; Huang, S.-Y.; Jean, Y.-H.; Wen, Z.-H. MSP-4, an Antimicrobial Peptide, Induces Apoptosis via Activation of Extrinsic Fas/FasL- and Intrinsic Mitochondria-Mediated Pathways in One Osteosarcoma Cell Line. Mar. Drugs 2018, 16, 8. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Mehta, R.R.; Lekmine, F.; Christov, K.; King, M.L.; Majumdar, D.; Shilkaitis, A.; Green, A.; Bratescu, L.; Beattie, C.W.; et al. A peptide fragment of azurin induces a p53-mediated cell cycle arrest in human breast cancer cells. Mol. Cancer Ther. 2009, 8, 2947–2958. [Google Scholar] [CrossRef]
- Signorelli, S.; Santini, S.; Yamada, T.; Bizzarri, A.R.; Beattie, C.W.; Cannistraro, S. Binding of Amphipathic Cell Penetrating Peptide p28 to Wild Type and Mutated p53 as studied by Raman, Atomic Force and Surface Plasmon Resonance spectroscopies. Biochim. Biophys. Acta (BBA) Gen. Subj. 2017, 1861, 910–921. [Google Scholar] [CrossRef]
- Yamada, T.; Goto, M.; Punj, V.; Zaborina, O.; Chen, M.L.; Kimbara, K.; Majumdar, D.; Cunningham, E.; Das Gupta, T.K.; Chakrabarty, A.M. Bacterial redox protein azurin, tumor suppressor protein p53, and regression of cancer. Proc. Natl. Acad. Sci. USA 2002, 99, 14098–14103. [Google Scholar] [CrossRef]
- Yamada, T.; Goto, M.; Punj, V.; Zaborina, O.; Kimbara, K.; Das Gupta, T.K.; Chakrabarty, A.M. The bacterial redox protein azurin induces apoptosis in J774 macrophages through complex formation and stabilization of the tumor suppressor protein p53. Infect. Immun. 2002, 70, 7054–7062. [Google Scholar] [CrossRef]
- Yamada, T.; Hiraoka, Y.; Ikehata, M.; Kimbara, K.; Avner, B.S.; Das Gupta, T.K.; Chakrabarty, A.M. Apoptosis or growth arrest: Modulation of tumor suppressor p53’s specificity by bacterial redox protein azurin. Proc. Natl. Acad. Sci. USA 2004, 101, 4770–4775. [Google Scholar] [CrossRef]
- Taglialegna, A. Pseudomonas against cancer. Nat. Rev. Microbiol. 2023, 21, 131. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.K.; Naffouje, S.A.; Goto, M.; Wang, J.; Christov, K.; Rademacher, D.J.; Green, A.; Stecenko, A.A.; Chakrabarty, A.M.; Das Gupta, T.K.; et al. Cross-talk between cancer and Pseudomonas aeruginosa mediates tumor suppression. Commun. Biol. 2023, 6, 16. [Google Scholar] [CrossRef] [PubMed]
- Sinha, G. Bacterial battalions join war against cancer. Nat. Med. 2003, 9, 1229. [Google Scholar] [CrossRef] [PubMed]
- Pseudomonas Gives Cancer Cells the Blues. Sci. STKE 2002, 2002, tw416. [CrossRef]
- Senior, K. Bacterial protein causes cancer cells to self-destruct. Lancet Oncol. 2002, 3, 712. [Google Scholar] [CrossRef]
- Yamada, T.; Fialho, A.M.; Punj, V.; Bratescu, L.; Das Gupta, T.K.; Chakrabarty, A.M. Internalization of bacterial redox protein azurin in mammalian cells: Entry domain and specificity. Cell. Microbiol. 2005, 7, 1418–1431. [Google Scholar] [CrossRef]
- Razzak, M. Targeted therapies: One step closer to drugging p53. Nat. Rev. Clin. Oncol. 2013, 10, 246. [Google Scholar] [CrossRef]
- Lulla, R.R.; Goldman, S.; Yamada, T.; Beattie, C.W.; Bressler, L.; Pacini, M.; Pollack, I.F.; Fisher, P.G.; Packer, R.J.; Dunkel, I.J.; et al. Phase I trial of p28 (NSC745104), a non-HDM2-mediated peptide inhibitor of p53 ubiquitination in pediatric patients with recurrent or progressive central nervous system tumors: A Pediatric Brain Tumor Consortium Study. Neuro-Oncology 2016, 18, 1319–1325. [Google Scholar] [CrossRef]
- Warso, M.A.; Richards, J.M.; Mehta, D.; Christov, K.; Schaeffer, C.; Bressler, L.R.; Yamada, T.; Majumdar, D.; Kennedy, S.A.; Beattie, C.W.; et al. A first-in-class, first-in-human, phase I trial of p28, a non-HDM2-mediated peptide inhibitor of p53 ubiquitination in patients with advanced solid tumours. Br. J. Cancer 2013, 108, 1061–1070. [Google Scholar] [CrossRef]
- White, E.R.; Sun, L.; Ma, Z.; Beckta, J.M.; Danzig, B.A.; Hacker, D.E.; Huie, M.; Williams, D.C.; Edwards, R.A.; Valerie, K.; et al. Peptide library approach to uncover phosphomimetic inhibitors of the BRCA1 C-terminal domain. ACS Chem. Biol. 2015, 10, 1198–1208. [Google Scholar] [CrossRef]
- Hao, X.; Yan, Q.; Zhao, J.; Wang, W.; Huang, Y.; Chen, Y. TAT Modification of α-Helical Anticancer Peptides to Improve Specificity and Efficacy. PLoS ONE 2015, 10, e0138911. [Google Scholar] [CrossRef] [PubMed]
- Habermann, E. Bee and wasp venoms. Science 1972, 177, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Liu, J.; Li, T.; Wang, Y.; Liu, X.; Bai, Y.; Wang, C.; Ju, S.; Huang, S.; Yang, C.; et al. A VEGFR targeting peptide-drug conjugate (PDC) suppresses tumor angiogenesis in a TACE model for hepatocellular carcinoma therapy. Cell Death Discov. 2022, 8, 411. [Google Scholar] [CrossRef] [PubMed]
- Yi, Z.F.; Cho, S.-G.; Zhao, H.; Wu, Y.-Y.; Luo, J.; Li, D.; Yi, T.; Xu, X.; Wu, Z.; Liu, M. A novel peptide from human apolipoprotein(a) inhibits angiogenesis and tumor growth by targeting c-Src phosphorylation in VEGF-induced human umbilical endothelial cells. Int. J. Cancer 2009, 124, 843–852. [Google Scholar] [CrossRef]
- Cho, H.D.; Nhàn, N.T.T.; Zhou, C.; Tu, K.; Nguyen, T.; Sarich, N.A.; Yamada, K.H. KIF13B mediates VEGFR2 recycling to modulate vascular permeability. Cell. Mol. Life Sci. 2023, 80, 91. [Google Scholar] [CrossRef]
- Waters, S.B.; Zhou, C.; Nguyen, T.; Zelkha, R.; Lee, H.; Kazlauskas, A.; Rosenblatt, M.I.; Malik, A.B.; Yamada, K.H. VEGFR2 Trafficking by KIF13B Is a Novel Therapeutic Target for Wet Age-Related Macular Degeneration. Investig. Ophthalmol. Vis. Sci. 2021, 62, 5. [Google Scholar] [CrossRef]
- Yamada, K.H. A novel approach in preventing vascular leakage and angiogenesis in wet age-related macular degeneration. Neural Regen. Res. 2022, 17, 1751–1752. [Google Scholar] [CrossRef]
- Wolf, J.S.; Li, G.; Varadhachary, A.; Petrak, K.; Schneyer, M.; Li, D.; Ongkasuwan, J.; Zhang, X.; Taylor, R.J.; Strome, S.E.; et al. Oral lactoferrin results in T cell-dependent tumor inhibition of head and neck squamous cell carcinoma in vivo. Clin. Cancer Res. 2007, 13, 1601–1610. [Google Scholar] [CrossRef]
- Zhang, Y.; Nicolau, A.; Lima, C.F.; Rodrigues, L.R. Bovine lactoferrin induces cell cycle arrest and inhibits mTOR signaling in breast cancer cells. Nutr. Cancer 2014, 66, 1371–1385. [Google Scholar] [CrossRef]
- Simberg, D.; Duza, T.; Park, J.H.; Essler, M.; Pilch, J.; Zhang, L.; Derfus, A.M.; Yang, M.; Hoffman, R.M.; Bhatia, S.; et al. Biomimetic amplification of nanoparticle homing to tumors. Proc. Natl. Acad. Sci. USA 2007, 104, 932–936. [Google Scholar] [CrossRef]
- Pasqualini, R.; Koivunen, E.; Ruoslahti, E. αv integrins as receptors for tumor targeting by circulating ligands. Nat. Biotechnol. 1997, 15, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Haubner, R.; Wester, H.J.; Burkhart, F.; Senekowitsch-Schmidtke, R.; Weber, W.; Goodman, S.L.; Kessler, H.; Schwaiger, M. Glycosylated RGD-containing peptides: Tracer for tumor targeting and angiogenesis imaging with improved biokinetics. J. Nucl. Med. 2001, 42, 326–336. [Google Scholar] [PubMed]
- Laverman, P.; Roosenburg, S.; Gotthardt, M.; Park, J.; Oyen, W.J.G.; de Jong, M.; Hellmich, M.R.; Rutjes, F.P.J.T.; van Delft, F.L.; Boerman, O.C. Targeting of a CCK2 receptor splice variant with 111In-labelled cholecystokinin-8 (CCK8) and 111In-labelled minigastrin. Eur. J. Nucl. Med. Mol. Imaging 2008, 35, 386–392. [Google Scholar] [CrossRef]
- Frankel, A.D.; Pabo, C.O. Cellular uptake of the tat protein from human immunodeficiency virus. Cell 1988, 55, 1189–1193. [Google Scholar] [CrossRef] [PubMed]
- Dupont, E.; Prochiantz, A.; Joliot, A. Penetratin story: An overview. Methods Mol. Biol. 2011, 683, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Derossi, D.; Joliot, A.; Chassaing, G.; Prochiantz, A. The third helix of the Antennapedia homeodomain translocates through biological membranes. J. Biol. Chem. 1994, 269, 10444–10450. [Google Scholar] [CrossRef] [PubMed]
- Pooga, M.; Lindgren, M.; Hallbrink, M.; Brakenhielm, E.; Langel, U. Galanin-based peptides, galparan and transportan, with receptor-dependent and independent activities. Ann. N. Y. Acad. Sci. 1998, 863, 450–453. [Google Scholar] [CrossRef]
- Jimenez-Macias, J.L.; Lee, Y.-C.; Miller, E.; Finkelberg, T.; Zdioruk, M.; Berger, G.; Farquhar, C.; Nowicki, M.; Cho, C.-F.; Fedeles, B.; et al. A Pt(IV)-conjugated brain penetrant macrocyclic peptide shows pre-clinical efficacy in glioblastoma. J. Control. Release 2022, 352, 623–636. [Google Scholar] [CrossRef]
- Fadzen, C.M.; Wolfe, J.M.; Cho, C.-F.; Chiocca, E.A.; Lawler, S.E.; Pentelute, B.L. Perfluoroarene-Based Peptide Macrocycles to Enhance Penetration Across the Blood-Brain Barrier. J. Am. Chem. Soc. 2017, 139, 15628–15631. [Google Scholar] [CrossRef]
- Lau, D.; Guo, L.; Liu, R.; Marik, J.; Lam, K. Peptide ligands targeting integrin α3β1 in non-small cell lung cancer. Lung Cancer 2006, 52, 291–297. [Google Scholar] [CrossRef]
- Zhou, Y.; Zou, Y.; Yang, M.; Mei, S.; Liu, X.; Han, H.; Zhang, C.-D.; Niu, M.-M. Highly Potent, Selective, Biostable, and Cell-Permeable Cyclic d-Peptide for Dual-Targeting Therapy of Lung Cancer. J. Am. Chem. Soc. 2022, 144, 7117–7128. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, K.; Kamada, Y.; Sameshima, T.; Yaguchi, M.; Niida, A.; Sasaki, S.; Miwa, M.; Ohkubo, S.; Sakamoto, J.-I.; Kamaura, M.; et al. K-Ras(G12D)-selective inhibitory peptides generated by random peptide T7 phage display technology. Biochem. Biophys. Res. Commun. 2017, 484, 605–611. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; Fu, A.; Wang, H.; Diaz, R.; Geng, L.; Onishko, H.; Hallahan, D.E. Noninvasive assessment of cancer response to therapy. Nat. Med. 2008, 14, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, V.; Singh, A.K.; Rogers, B.E.; Thotala, D.; Hallahan, D.E. PEGylated peptide to TIP1 is a novel targeting agent that binds specifically to various cancers in vivo. J. Control. Release 2019, 298, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Tarasov, S.G.; Gaponenko, V.; Howard, O.M.Z.; Chen, Y.; Oppenheim, J.J.; Dyba, M.A.; Subramaniam, S.; Lee, Y.; Michejda, C.; Tarasova, N.I. Structural plasticity of a transmembrane peptide allows self-assembly into biologically active nanoparticles. Proc. Natl. Acad. Sci. USA 2011, 108, 9798–9803. [Google Scholar] [CrossRef]
- Tan, M.; Wu, X.; Jeong, E.-K.; Chen, Q.; Lu, Z.-R. Peptide-targeted Nanoglobular Gd-DOTA monoamide conjugates for magnetic resonance cancer molecular imaging. Biomacromolecules 2010, 11, 754–761. [Google Scholar] [CrossRef]
- Dithmer, S.; Staat, C.; Müller, C.; Ku, M.-C.; Pohlmann, A.; Niendorf, T.; Gehne, N.; Fallier-Becker, P.; Kittel, Á.; Walter, F.R.; et al. Claudin peptidomimetics modulate tissue barriers for enhanced drug delivery. Ann. N. Y. Acad. Sci. 2017, 1397, 169–184. [Google Scholar] [CrossRef]
- Tabanor, K.; Lee, P.; Kiptoo, P.; Choi, I.-Y.; Sherry, E.B.; Eagle, C.S.; Williams, T.D.; Siahaan, T.J. Brain Delivery of Drug and MRI Contrast Agent: Detection and Quantitative Determination of Brain Deposition of CPT-Glu Using LC-MS/MS and Gd-DTPA Using Magnetic Resonance Imaging. Mol. Pharm. 2016, 13, 379–390. [Google Scholar] [CrossRef]
- Sajesh, B.V.; On, N.H.; Omar, R.; Alrushaid, S.; Kopec, B.M.; Wang, W.-G.; Sun, H.-D.; Lillico, R.; Lakowski, T.M.; Siahaan, T.J.; et al. Validation of Cadherin HAV6 Peptide in the Transient Modulation of the Blood-Brain Barrier for the Treatment of Brain Tumors. Pharmaceutics 2019, 11, 481. [Google Scholar] [CrossRef]
- Kiptoo, P.; Sinaga, E.; Calcagno, A.M.; Zhao, H.; Kobayashi, N.; Tambunan, U.S.F.; Siahaan, T.J. Enhancement of drug absorption through the blood-brain barrier and inhibition of intercellular tight junction resealing by E-cadherin peptides. Mol. Pharm. 2011, 8, 239–249. [Google Scholar] [CrossRef]
- Sarkar, G.; Curran, G.L.; Mahlum, E.; Decklever, T.; Wengenack, T.M.; Blahnik, A.; Hoesley, B.; Lowe, V.J.; Poduslo, J.F.; Jenkins, R.B. A carrier for non-covalent delivery of functional β-galactosidase and antibodies against amyloid plaques and IgM to the brain. PLoS ONE 2011, 6, e28881. [Google Scholar] [CrossRef] [PubMed]
- Al-Azzawi, S.; Masheta, D.; Guildford, A.; Phillips, G.; Santin, M. Designing and Characterization of a Novel Delivery System for Improved Cellular Uptake by Brain Using Dendronised Apo-E-Derived Peptide. Front. Bioeng. Biotechnol. 2019, 7, 49. [Google Scholar] [CrossRef] [PubMed]
- Ruan, H.; Chai, Z.; Shen, Q.; Chen, X.; Su, B.; Xie, C.; Zhan, C.; Yao, S.; Wang, H.; Zhang, M.; et al. A novel peptide ligand RAP12 of LRP1 for glioma targeted drug delivery. J. Control. Release 2018, 279, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Thom, G.; Tian, M.-M.; Hatcher, J.P.; Rodrigo, N.; Burrell, M.; Gurrell, I.; Vitalis, T.Z.; Abraham, T.; Jefferies, W.A.; Webster, C.I.; et al. A peptide derived from melanotransferrin delivers a protein-based interleukin 1 receptor antagonist across the BBB and ameliorates neuropathic pain in a preclinical model. J. Cereb. Blood Flow Metab. 2019, 39, 2074–2088. [Google Scholar] [CrossRef] [PubMed]
- Malcor, J.D.; Payrot, N.; David, M.; Faucon, A.; Abouzid, K.; Jacquot, G.; Floquet, N.; Debarbieux, F.; Rougon, G.; Martinez, J.; et al. Chemical optimization of new ligands of the low-density lipoprotein receptor as potential vectors for central nervous system targeting. J. Med. Chem. 2012, 55, 2227–2241. [Google Scholar] [CrossRef]
- Sakamoto, K.; Shinohara, T.; Adachi, Y.; Asami, T.; Ohtaki, T. A novel LRP1-binding peptide L57 that crosses the blood brain barrier. Biochem. Biophys. Rep. 2017, 12, 135–139. [Google Scholar] [CrossRef]
- Chen, L.; Zeng, D.; Xu, N.; Li, C.; Zhang, W.; Zhu, X.-J.; Gao, Y.; Chen, P.R.; Lin, J. Blood-Brain Barrier- and Blood-Brain Tumor Barrier-Penetrating Peptide-Derived Targeted Therapeutics for Glioma and Malignant Tumor Brain Metastases. ACS Appl. Mater. Interfaces 2019, 11, 41889–41897. [Google Scholar] [CrossRef]
- André, S.; Larbanoix, L.; Verteneuil, S.; Stanicki, D.; Nonclercq, D.; Vander Elst, L.; Laurent, S.; Muller, R.N.; Burtea, C. Development of an LDL Receptor-Targeted Peptide Susceptible to Facilitate the Brain Access of Diagnostic or Therapeutic Agents. Biology 2020, 9, 161. [Google Scholar] [CrossRef]
- Lee, J.H.; Engler, J.A.; Collawn, J.F.; Moore, B.A. Receptor mediated uptake of peptides that bind the human transferrin receptor. Eur. J. Biochem. 2001, 268, 2004–2012. [Google Scholar] [CrossRef]
- Xia, H.; Anderson, B.; Mao, Q.; Davidson, B.L. Recombinant human adenovirus: Targeting to the human transferrin receptor improves gene transfer to brain microcapillary endothelium. J. Virol. 2000, 74, 11359–11366. [Google Scholar] [CrossRef]
- Staquicini, F.I.; Ozawa, M.G.; Moya, C.A.; Driessen, W.H.; Barbu, E.M.; Nishimori, H.; Soghomonyan, S.; Flores, L.G.; Liang, X.; Paolillo, V.; et al. Systemic combinatorial peptide selection yields a non-canonical iron-mimicry mechanism for targeting tumors in a mouse model of human glioblastoma. J. Clin. Investig. 2011, 121, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.P.; Ahmadvand, D.; Su, J.; Hall, A.; Tan, X.; Farhangrazi, Z.S.; Moghimi, S.M. Crossing the blood-brain-barrier with nanoligand drug carriers self-assembled from a phage display peptide. Nat. Commun. 2019, 10, 4635. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, J.; Shao, K.; Huang, R.; Ye, L.; Lou, J.; Jiang, C. A leptin derived 30-amino-acid peptide modified pegylated poly-L-lysine dendrigraft for brain targeted gene delivery. Biomaterials 2010, 31, 5246–5257. [Google Scholar] [CrossRef] [PubMed]
- Barrett, G.L.; Trieu, J.; Naim, T. The identification of leptin-derived peptides that are taken up by the brain. Regul. Pept. 2009, 155, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Tosi, G.; Badiali, L.; Ruozi, B.; Vergoni, A.V.; Bondioli, L.; Ferrari, A.; Rivasi, F.; Forni, F.; Vandelli, M.A. Can leptin-derived sequence-modified nanoparticles be suitable tools for brain delivery? Nanomedicine 2012, 7, 365–382. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, X.; Xie, C.; Zhang, M.; Ruan, H.; Wang, S.; Jiang, K.; Wang, F.; Zhan, C.; Lu, W.; et al. Nanodisk-based glioma-targeted drug delivery enabled by a stable glycopeptide. J. Control. Release 2018, 284, 26–38. [Google Scholar] [CrossRef]
- Binetruy-Tournaire, R.; Demangel, C.; Malavaud, B.; Vassy, R.; Rouyre, S.; Kraemer, M.; Plouët, J.; Derbin, C.; Perret, G.; Mazié, J.C. Identification of a peptide blocking vascular endothelial growth factor (VEGF)-mediated angiogenesis. EMBO J. 2000, 19, 1525–1533. [Google Scholar] [CrossRef]
- Gao, H.; Yang, Z.; Zhang, S.; Cao, S.; Pang, Z.; Yang, X.; Jiang, X. Glioma-homing peptide with a cell-penetrating effect for targeting delivery with enhanced glioma localization, penetration and suppression of glioma growth. J. Control. Release 2013, 172, 921–928. [Google Scholar] [CrossRef]
- Pandya, H.; Gibo, D.M.; Garg, S.; Kridel, S.; Debinski, W. An interleukin 13 receptor α2-specific peptide homes to human Glioblastoma multiforme xenografts. Neuro-Oncology 2012, 14, 6–18. [Google Scholar] [CrossRef]
- Jiao, Z.; Li, Y.; Pang, H.; Zheng, Y.; Zhao, Y. Pep-1 peptide-functionalized liposome to enhance the anticancer efficacy of cilengitide in glioma treatment. Colloids Surf. B Biointerfaces 2017, 158, 68–75. [Google Scholar] [CrossRef]
- Tosi, G.; Fano, R.A.; Bondioli, L.; Badiali, L.; Benassi, R.; Rivasi, F.; Ruozi, B.; Forni, F.; Vandelli, M.A. Investigation on mechanisms of glycopeptide nanoparticles for drug delivery across the blood-brain barrier. Nanomedicine 2011, 6, 423–436. [Google Scholar] [CrossRef] [PubMed]
- Tosi, G.; Costantino, L.; Rivasi, F.; Ruozi, B.; Leo, E.; Vergoni, A.; Tacchi, R.; Bertolini, A.; Vandelli, M.; Forni, F. Targeting the central nervous system: In vivo experiments with peptide-derivatized nanoparticles loaded with Loperamide and Rhodamine-123. J. Control. Release 2007, 122, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Wu, H.; McBride, J.L.; Jung, K.-E.; Kim, M.H.; Davidson, B.L.; Lee, S.K.; Shankar, P.; Manjunath, N. Transvascular delivery of small interfering RNA to the central nervous system. Nature 2007, 448, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Zhou, R.; Fu, A.; Xu, X.; Huang, Y.; Hu, C. Targeted delivery of large fusion protein into hippocampal neurons by systemic administration. J. Drug Target. 2011, 19, 632–636. [Google Scholar] [CrossRef] [PubMed]
- Fu, A.; Wang, Y.; Zhan, L.; Zhou, R. Targeted delivery of proteins into the central nervous system mediated by rabies virus glycoprotein-derived peptide. Pharm. Res. 2012, 29, 1562–1569. [Google Scholar] [CrossRef]
- Zhan, C.; Yan, Z.; Xie, C.; Lu, W. Loop 2 of Ophiophagus hannah toxin b binds with neuronal nicotinic acetylcholine receptors and enhances intracranial drug delivery. Mol. Pharm. 2010, 7, 1940–1947. [Google Scholar] [CrossRef]
- Zhan, C.; Li, B.; Hu, L.; Wei, X.; Feng, L.; Fu, W.; Lu, W. Micelle-based brain-targeted drug delivery enabled by a nicotine acetylcholine receptor ligand. Angew. Chem. Int. Ed. 2011, 50, 5482–5485. [Google Scholar] [CrossRef]
- Wei, X.; Zhan, C.; Shen, Q.; Fu, W.; Xie, C.; Gao, J.; Peng, C.; Zheng, P.; Lu, W. A D-peptide ligand of nicotine acetylcholine receptors for brain-targeted drug delivery. Angew. Chem. Int. Ed. 2015, 54, 3023–3027. [Google Scholar] [CrossRef]
- Oller-Salvia, B.; Sánchez-Navarro, M.; Ciudad, S.; Guiu, M.; Arranz-Gibert, P.; Garcia, C.; Gomis, R.R.; Cecchelli, R.; García, J.; Giralt, E.; et al. MiniAp-4: A Venom-Inspired Peptidomimetic for Brain Delivery. Angew. Chem. Int. Ed. 2016, 55, 572–575. [Google Scholar] [CrossRef]
- Li, J.; Feng, L.; Fan, L.; Zha, Y.; Guo, L.; Zhang, Q.; Chen, J.; Pang, Z.; Wang, Y.; Jiang, X.; et al. Targeting the brain with PEG–PLGA nanoparticles modified with phage-displayed peptides. Biomaterials 2011, 32, 4943–4950. [Google Scholar] [CrossRef]
- Rousselle, C.; Clair, P.; Lefauconnier, J.-M.; Kaczorek, M.; Scherrmann, J.-M.; Temsamani, J. New advances in the transport of doxorubicin through the blood-brain barrier by a peptide vector-mediated strategy. Mol. Pharmacol. 2000, 57, 679–686. [Google Scholar] [CrossRef]
- Mann, A.P.; Scodeller, P.; Hussain, S.; Joo, J.; Kwon, E.; Braun, G.B.; Mölder, T.; She, Z.-G.; Kotamraju, V.R.; Ranscht, B.; et al. A peptide for targeted, systemic delivery of imaging and therapeutic compounds into acute brain injuries. Nat. Commun. 2016, 7, 11980. [Google Scholar] [CrossRef] [PubMed]
- Stojanov, K.; Georgieva, J.V.; Brinkhuis, R.P.; van Hest, J.C.; Rutjes, F.P.; Dierckx, R.A.J.O.; de Vries, E.F.J.; Zuhorn, I.S. In vivo biodistribution of prion- and GM1-targeted polymersomes following intravenous administration in mice. Mol. Pharm. 2012, 9, 1620–1627. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.K.; Teng, Q.; Garrity-Moses, M.; Federici, T.; Tanase, D.; Imperiale, M.J.; Boulis, N.M. A novel peptide defined through phage display for therapeutic protein and vector neuronal targeting. Neurobiol. Dis. 2005, 19, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Neves, V.; Aires-Da-Silva, F.; Morais, M.; Gano, L.; Ribeiro, E.; Pinto, A.; Aguiar, S.; Gaspar, D.; Fernandes, C.; Correia, J.D.G.; et al. Novel Peptides Derived from Dengue Virus Capsid Protein Translocate Reversibly the Blood–Brain Barrier through a Receptor-Free Mechanism. ACS Chem. Biol. 2017, 12, 1257–1268. [Google Scholar] [CrossRef] [PubMed]
- Malakoutikhah, M.; Prades, R.; Teixidó, M.; Giralt, E. N-Methyl phenylalanine-rich peptides as highly versatile blood−brain barrier shuttles. J. Med. Chem. 2010, 53, 2354–2363. [Google Scholar] [CrossRef] [PubMed]
- Arranz-Gibert, P.; Guixer, B.; Malakoutikhah, M.; Muttenthaler, M.; Guzmán, F.; Teixidó, M.; Giralt, E. Lipid bilayer crossing—The gate of symmetry. Water-soluble phenylproline-based blood-brain barrier shuttles. J. Am. Chem. Soc. 2015, 137, 7357–7364. [Google Scholar] [CrossRef] [PubMed]
- Neves-Coelho, S.; Eleutério, R.P.; Enguita, F.J.; Neves, V.; Castanho, M.A.R.B. A New Noncanonical anionic peptide that translocates a cellular blood–brain barrier model. Molecules 2017, 22, 1753. [Google Scholar] [CrossRef]
- Mendonça, D.A.; Bakker, M.; Cruz-Oliveira, C.; Neves, V.; Jiménez, M.A.; Defaus, S.; Cavaco, M.; Veiga, A.S.; Cadima-Couto, I.; Castanho, M.A.R.B.; et al. Penetrating the Blood-Brain Barrier with New Peptide–Porphyrin Conjugates Having anti-HIV Activity. Bioconjug. Chem. 2021, 32, 1067–1077. [Google Scholar] [CrossRef]
- Lépinoux-Chambaud, C.; Eyer, J. The NFL-TBS.40–63 peptide targets and kills glioblastoma stem cells derived from human patients and also targets nanocapsules into these cells. Int. J. Pharm. 2019, 566, 218–228. [Google Scholar] [CrossRef]
- Balzeau, J.; Pinier, M.; Berges, R.; Saulnier, P.; Benoit, J.-P.; Eyer, J. The effect of functionalizing lipid nanocapsules with NFL-TBS.40-63 peptide on their uptake by glioblastoma cells. Biomaterials 2013, 34, 3381–3389. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Gao, X.; Gu, G.; Liu, Z.; Zeng, N.; Hu, Q.; Song, Q.; Yao, L.; Pang, Z.; Jiang, X.; et al. Low molecular weight protamine-functionalized nanoparticles for drug delivery to the brain after intranasal administration. Biomaterials 2011, 32, 9888–9898. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Singh, M.; Santos, G.S.; Guerlavais, V.; Carvajal, L.A.; Aivado, M.; Zhan, Y.; Oliveira, M.M.; Westerberg, L.S.; Annis, D.A.; et al. Pharmacologic Activation of p53 Triggers Viral Mimicry Response Thereby Abolishing Tumor Immune Evasion and Promoting Antitumor Immunity. Cancer Discov. 2021, 11, 3090–3105. [Google Scholar] [CrossRef]
- Saleh, M.N.; Patel, M.R.; Bauer, T.M.; Goel, S.; Falchook, G.S.; Shapiro, G.I.; Chung, K.Y.; Infante, J.R.; Conry, R.M.; Rabinowits, G.; et al. Phase 1 Trial of ALRN-6924, a Dual Inhibitor of MDMX and MDM2, in Patients with Solid Tumors and Lymphomas Bearing Wild-type TP53. Clin. Cancer Res. 2021, 27, 5236–5247. [Google Scholar] [CrossRef] [PubMed]
- Nomura, K.; Hashimoto, S.; Takeyama, R.; Tamiya, M.; Kato, T.; Muraoka, T.; Kage, M.; Nii, K.; Kotake, K.; Iida, S.; et al. Broadly Applicable and Comprehensive Synthetic Method for N-Alkyl-Rich Drug-like Cyclic Peptides. J. Med. Chem. 2022, 65, 13401–13412. [Google Scholar] [CrossRef] [PubMed]
- Mittendorf, E.A.; Clifton, G.T.; Holmes, J.P.; Schneble, E.; van Echo, D.; Ponniah, S.; Peoples, G.E. Final report of the phase I/II clinical trial of the E75 (nelipepimut-S) vaccine with booster inoculations to prevent disease recurrence in high-risk breast cancer patients. Ann. Oncol. 2014, 25, 1735–1742. [Google Scholar] [CrossRef]
- Lynch, K.T.; Squeo, G.C.; Kane, W.J.; Meneveau, M.O.; Petroni, G.; Olson, W.C.; Chianese-Bullock, K.A.; Slingluff, C.L., Jr.; Foley, E.F.; Friel, C.M. A pilot trial of vaccination with Carcinoembryonic antigen and Her2/neu peptides in advanced colorectal cancer. Int. J. Cancer 2022, 150, 164–173. [Google Scholar] [CrossRef]
- Dillon, P.M.; Petroni, G.R.; Smolkin, M.E.; Brenin, D.R.; Chianese-Bullock, K.A.; Smith, K.T.; Olson, W.C.; Fanous, I.S.; Nail, C.J.; Brenin, C.M.; et al. A pilot study of the immunogenicity of a 9-peptide breast cancer vaccine plus poly-ICLC in early stage breast cancer. J. Immunother. Cancer 2017, 5, 92. [Google Scholar] [CrossRef]
- Mittendorf, E.A.; Lu, B.; Melisko, M.; Hiller, J.P.; Bondarenko, I.; Brunt, A.M.; Sergii, G.; Petrakova, K.; Peoples, G.E. Efficacy and Safety Analysis of Nelipepimut-S Vaccine to Prevent Breast Cancer Recurrence: A Randomized, Multicenter, Phase III Clinical Trial. Clin. Cancer Res. 2019, 25, 4248–4254. [Google Scholar] [CrossRef]
- Clifton, G.T.; Hale, D.; Vreeland, T.J.; Hickerson, A.T.; Litton, J.K.; Alatrash, G.; Murthy, R.K.; Qiao, N.; Philips, A.V.; Lukas, J.J.; et al. Results of a Randomized Phase IIb Trial of Nelipepimut-S + Trastuzumab versus Trastuzumab to Prevent Recurrences in Patients with High-Risk HER2 Low-Expressing Breast Cancer. Clin. Cancer Res. 2020, 26, 2515–2523. [Google Scholar] [CrossRef]
- Dean, A.; Gill, S.; McGregor, M.; Broadbridge, V.; Järveläinen, H.A.; Price, T. Dual αV-integrin and neuropilin-1 targeting peptide CEND-1 plus nab-paclitaxel and gemcitabine for the treatment of metastatic pancreatic ductal adenocarcinoma: A first-in-human, open-label, multicentre, phase 1 study. Lancet Gastroenterol. Hepatol. 2022, 7, 943–951. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Hegi, M.E.; Gorlia, T.; Erridge, S.C.; Perry, J.; Hong, Y.-K.; Aldape, K.D.; Lhermitte, B.; Pietsch, T.; Grujicic, D.; et al. Cilengitide combined with standard treatment for patients with newly diagnosed glioblastoma with methylated MGMT promoter (CENTRIC EORTC 26071-22072 study): A multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2014, 15, 1100–1108. [Google Scholar] [CrossRef] [PubMed]
- Warren, K.E.; Patel, M.C.; Aikin, A.A.; Widemann, B.; Libucha, M.; Adamson, P.C.; Neuwirth, R.; Benziger, D.; O’Toole, T.; Ford, K.; et al. Phase I trial of lobradimil (RMP-7) and carboplatin in children with brain tumors. Cancer Chemother. Pharmacol. 2001, 48, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Warren, K.; Jakacki, R.; Widemann, B.; Aikin, A.; Libucha, M.; Packer, R.; Vezina, G.; Reaman, G.; Shaw, D.; Krailo, M.; et al. Phase II trial of intravenous lobradimil and carboplatin in childhood brain tumors: A report from the Children’s Oncology Group. Cancer Chemother. Pharmacol. 2006, 58, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Prados, M.D.; Schold, S.C., Jr.; Fine, H.A.; Jaeckle, K.; Hochberg, F.; Mechtler, L.; Fetell, M.R.; Phuphanich, S.; Feun, L.; Janus, T.J.; et al. A randomized, double-blind, placebo-controlled, phase 2 study of RMP-7 in combination with carboplatin administered intravenously for the treatment of recurrent malignant glioma. Neuro-Oncology 2003, 5, 96–103. [Google Scholar] [CrossRef]
- Gregor, A.; Lind, M.; Newman, H.; Grant, R.; Hadley, D.; Barton, T.; Osborn, C. Phase II studies of RMP-7 and carboplatin in the treatment of recurrent high grade glioma. RMP-7 European Study Group. J. Neurooncol. 1999, 44, 137–145. [Google Scholar] [CrossRef]
- Noy, A.; Scadden, D.T.; Lee, J.; Dezube, B.J.; Aboulafia, D.; Tulpule, A.; Walmsley, S.; Gill, P. Angiogenesis inhibitor IM862 is ineffective against AIDS-Kaposi’s sarcoma in a phase III trial, but demonstrates sustained, potent effect of highly active antiretroviral therapy: From the AIDS Malignancy Consortium and IM862 Study Team. J. Clin. Oncol. 2005, 23, 990–998. [Google Scholar] [CrossRef]
- Kurzrock, R.; Gabrail, N.; Chandhasin, C.; Moulder, S.; Smith, C.; Brenner, A.; Sankhala, K.; Mita, A.; Elian, K.; Bouchard, D.; et al. Safety, pharmacokinetics, and activity of GRN1005, a novel conjugate of angiopep-2, a peptide facilitating brain penetration, and paclitaxel, in patients with advanced solid tumors. Mol. Cancer Ther. 2012, 11, 308–316. [Google Scholar] [CrossRef]
- Drappatz, J.; Brenner, A.; Wong, E.T.; Eichler, A.; Schiff, D.; Groves, M.D.; Mikkelsen, T.; Rosenfeld, S.; Sarantopoulos, J.; Meyers, C.A.; et al. Phase I study of GRN1005 in recurrent malignant glioma. Clin. Cancer Res. 2013, 19, 1567–1576. [Google Scholar] [CrossRef]
- Doss, M.; Kolb, H.C.; Zhang, J.J.; Bélanger, M.-J.; Stubbs, J.B.; Stabin, M.G.; Hostetler, E.D.; Alpaugh, R.K.; von Mehren, M.; Walsh, J.C.; et al. Biodistribution and radiation dosimetry of the integrin marker 18F-RGD-K5 determined from whole-body PET/CT in monkeys and humans. J. Nucl. Med. 2012, 53, 787–795. [Google Scholar] [CrossRef]
- Zheng, K.; Liang, N.; Zhang, J.; Lang, L.; Zhang, W.; Li, S.; Zhao, J.; Niu, G.; Li, F.; Zhu, Z.; et al. 68Ga-NOTA-PRGD2 PET/CT for Integrin Imaging in Patients with Lung Cancer. J. Nucl. Med. 2015, 56, 1823–1827. [Google Scholar] [CrossRef] [PubMed]
- Toriihara, A.; Duan, H.; Thompson, H.M.; Park, S.; Hatami, N.; Baratto, L.; Fan, A.C.; Iagaru, A. 18F-FPPRGD2 PET/CT in patients with metastatic renal cell cancer. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1518–1523. [Google Scholar] [CrossRef] [PubMed]
- Minamimoto, R.; Jamali, M.; Barkhodari, A.; Mosci, C.; Mittra, E.; Shen, B.; Chin, F.; Gambhir, S.S.; Iagaru, A. Biodistribution of the 18F-FPPRGD2 PET radiopharmaceutical in cancer patients: An atlas of SUV measurements. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 1850–1858. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Niu, G.; Fan, X.; Lang, L.; Hou, G.; Chen, L.; Wu, H.; Zhu, Z.; Li, F.; Chen, X. PET Using a GRPR Antagonist 68Ga-RM26 in Healthy Volunteers and Prostate Cancer Patients. J. Nucl. Med. 2018, 59, 922–928. [Google Scholar] [CrossRef] [PubMed]
- Van Putten, E.H.; Kleijn, A.; Van Beusechem, V.W.; Noske, D.; Lamers, C.H.; De Goede, A.L.; Idema, S.; Hoefnagel, D.; Kloezeman, J.J.; Fueyo, J.; et al. Convection Enhanced Delivery of the Oncolytic Adenovirus Delta24-RGD in Patients with Recurrent GBM: A Phase I Clinical Trial Including Correlative Studies. Clin. Cancer Res. 2022, 28, 1572–1585. [Google Scholar] [CrossRef] [PubMed]
- Nassiri, F.; Patil, V.; Yefet, L.S.; Singh, O.; Liu, J.; Dang, R.M.; Yamaguchi, T.N.; Daras, M.; Cloughesy, T.F.; Colman, H.; et al. Oncolytic DNX-2401 virotherapy plus pembrolizumab in recurrent glioblastoma: A phase 1/2 trial. Nat. Med. 2023, 29, 1370–1378. [Google Scholar] [CrossRef]
- Lang, F.F.; Conrad, C.; Gomez-Manzano, C.; Yung, W.K.A.; Sawaya, R.; Weinberg, J.S.; Prabhu, S.S.; Rao, G.; Fuller, G.N.; Aldape, K.D.; et al. Phase I Study of DNX-2401 (Delta-24-RGD) Oncolytic Adenovirus: Replication and Immunotherapeutic Effects in Recurrent Malignant Glioma. J. Clin. Oncol. 2018, 36, 1419–1427. [Google Scholar] [CrossRef]
- Pérez-Larraya, J.G.; Garcia-Moure, M.; Labiano, S.; Patiño-García, A.; Dobbs, J.; Gonzalez-Huarriz, M.; Zalacain, M.; Marrodan, L.; Martinez-Velez, N.; Puigdelloses, M.; et al. Oncolytic DNX-2401 Virus for Pediatric Diffuse Intrinsic Pontine Glioma. N. Engl. J. Med. 2022, 386, 2471–2481. [Google Scholar] [CrossRef]
- Chen, S.R.; Chen, M.M.; Ene, C.; Lang, F.F.; Kan, P. Perfusion-guided endovascular super-selective intra-arterial infusion for treatment of malignant brain tumors. J. Neurointerv.Surg. 2022, 14, 533–538. [Google Scholar] [CrossRef]
- Lee, D.-H.; Rötger, C.; Appeldoorn, C.C.; Reijerkerk, A.; Gladdines, W.; Gaillard, P.J.; Linker, R.A. Glutathione PEGylated liposomal methylprednisolone (2B3-201) attenuates CNS inflammation and degeneration in murine myelin oligodendrocyte glycoprotein induced experimental autoimmune encephalomyelitis. J. Neuroimmunol. 2014, 274, 96–101. [Google Scholar] [CrossRef]
- Chang, Y.S.; Graves, B.; Guerlavais, V.; Tovar, C.; Packman, K.; To, K.-H.; Olson, K.A.; Kesavan, K.; Gangurde, P.; Mukherjee, A.; et al. Stapled α-helical peptide drug development: A potent dual inhibitor of MDM2 and MDMX for p53-dependent cancer therapy. Proc. Natl. Acad. Sci. USA 2013, 110, E3445–E3454. [Google Scholar] [CrossRef] [PubMed]
- Guerlavais, V.; Sawyer, T.K.; Carvajal, L.; Chang, Y.S.; Graves, B.; Ren, J.G.; Sutton, D.; Olson, K.A.; Packman, K.; Darlak, K.; et al. Discovery of Sulanemadlin (ALRN-6924), the First Cell-Permeating, Stabilized α-Helical Peptide in Clinical Development. J. Med. Chem. 2023, 66, 9401–9417. [Google Scholar] [CrossRef]
- Ng, S.Y.; Yoshida, N.; Christie, A.L.; Ghandi, M.; Dharia, N.V.; Dempster, J.; Murakami, M.; Shigemori, K.; Morrow, S.N.; Van Scoyk, A.; et al. Targetable vulnerabilities in T- and NK-cell lymphomas identified through preclinical models. Nat. Commun. 2018, 9, 2024. [Google Scholar] [CrossRef] [PubMed]
- Carvajal, L.A.; Ben Neriah, D.; Senecal, A.; Benard, L.; Thiruthuvanathan, V.; Yatsenko, T.; Narayanagari, S.-R.; Wheat, J.C.; Todorova, T.I.; Mitchell, K.; et al. Dual inhibition of MDMX and MDM2 as a therapeutic strategy in leukemia. Sci. Transl. Med. 2018, 10, eaao3003. [Google Scholar] [CrossRef] [PubMed]
- Pairawan, S.; Zhao, M.; Yuca, E.; Annis, A.; Evans, K.; Sutton, D.; Carvajal, L.; Ren, J.G.; Santiago, S.; Guerlavais, V.; et al. First in class dual MDM2/MDMX inhibitor ALRN-6924 enhances antitumor efficacy of chemotherapy in TP53 wild-type hormone receptor-positive breast cancer models. Breast Cancer Res. 2021, 23, 29. [Google Scholar] [CrossRef]
- Phase 1b Trial of ALRN-6924 for p53-Mutated Breast Cancer Terminated. Targeted Oncology, 22 February 2023. Available online: https://www.targetedonc.com/view/phase-1b-trial-of-alrn-6924-for-p53-mutated-breast-cancer-terminated (accessed on 30 June 2023).
- Tan, J.; Tay, J.; Hedrick, J.; Yang, Y.Y. Synthetic macromolecules as therapeutics that overcome resistance in cancer and microbial infection. Biomaterials 2020, 252, 120078. [Google Scholar] [CrossRef]
- Lin, L.; Chi, J.; Yan, Y.; Luo, R.; Feng, X.; Zheng, Y.; Xian, D.; Li, X.; Quan, G.; Liu, D.; et al. Membrane-disruptive peptides/peptidomimetics-based therapeutics: Promising systems to combat bacteria and cancer in the drug-resistant era. Acta Pharm. Sin. B 2021, 11, 2609–2644. [Google Scholar] [CrossRef]
- Jin, H.; Wan, C.; Zou, Z.; Zhao, G.; Zhang, L.; Geng, Y.; Chen, T.; Huang, A.; Jiang, F.; Feng, J.-P.; et al. Tumor Ablation and Therapeutic Immunity Induction by an Injectable Peptide Hydrogel. ACS Nano 2018, 12, 3295–3310. [Google Scholar] [CrossRef]
- De Palma, M.; Biziato, D.; Petrova, T.V. Microenvironmental regulation of tumour angiogenesis. Nat. Rev. Cancer 2017, 17, 457–474. [Google Scholar] [CrossRef]
- Carmeliet, P. Angiogenesis in life, disease and medicine. Nature 2005, 438, 932–936. [Google Scholar] [CrossRef]
- Saharinen, P.; Eklund, L.; Pulkki, K.; Bono, P.; Alitalo, K. VEGF and angiopoietin signaling in tumor angiogenesis and metastasis. Trends Mol. Med. 2011, 17, 347–362. [Google Scholar] [CrossRef] [PubMed]
- Bergers, G.; Hanahan, D. Modes of resistance to anti-angiogenic therapy. Nat. Rev. Cancer 2008, 8, 592–603. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.H.; Kang, H.; Malik, A.B. Antiangiogenic Therapeutic Potential of Peptides Derived from the Molecular Motor KIF13B that Transports VEGFR2 to Plasmalemma in Endothelial Cells. Am. J. Pathol. 2017, 187, 214–224. [Google Scholar] [CrossRef]
- Yamada, K.H.; Nakajima, Y.; Geyer, M.; Wary, K.K.; Ushio-Fukai, M.; Komarova, Y.; Malik, A.B. KIF13B regulates angiogenesis through Golgi to plasma membrane trafficking of VEGFR2. J. Cell Sci. 2014, 127 Pt 20, 4518–4530. [Google Scholar] [CrossRef]
- Gonzalez, H.; Hagerling, C.; Werb, Z. Roles of the immune system in cancer: From tumor initiation to metastatic progression. Genes Dev. 2018, 32, 1267–1284. [Google Scholar] [CrossRef] [PubMed]
- Palucka, A.K.; Coussens, L.M. The Basis of Oncoimmunology. Cell 2016, 164, 1233–1247. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, Z. The history and advances in cancer immunotherapy: Understanding the characteristics of tumor-infiltrating immune cells and their therapeutic implications. Cell. Mol. Immunol. 2020, 17, 807–821. [Google Scholar] [CrossRef]
- Tripathi, A.K.; Vishwanatha, J.K. Role of Anti-Cancer Peptides as Immunomodulatory Agents: Potential and Design Strategy. Pharmaceutics 2022, 14, 2686. [Google Scholar] [CrossRef]
- Furukawa, N.; Popel, A.S. Peptides that immunoactivate the tumor microenvironment. Biochim. Biophys. Acta (BBA) Rev. Cancer 2021, 1875, 188486. [Google Scholar] [CrossRef]
- Coulie, P.G.; Van den Eynde, B.J.; Van Der Bruggen, P.; Boon, T. Tumour antigens recognized by T lymphocytes: At the core of cancer immunotherapy. Nat. Rev. Cancer 2014, 14, 135–146. [Google Scholar] [CrossRef]
- Abd-Aziz, N.; Poh, C.L. Development of Peptide-Based Vaccines for Cancer. J. Oncol. 2022, 2022, 9749363. [Google Scholar] [CrossRef] [PubMed]
- Fisk, B.; Blevins, T.L.; Wharton, J.T.; Ioannides, C.G. Identification of an immunodominant peptide of HER-2/neu protooncogene recognized by ovarian tumor-specific cytotoxic T lymphocyte lines. J. Exp. Med. 1995, 181, 2109–2117. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Li, Y.; Wang, Y.; Ren, S.; Li, Y.; Wang, B. Polyactin A is a novel and potent immunological adjuvant for peptide-based cancer vaccine. Int. Immunopharmacol. 2018, 54, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Kondo, E.; Iioka, H.; Saito, K. Tumor-homing peptide and its utility for advanced cancer medicine. Cancer Sci. 2021, 112, 2118–2125. [Google Scholar] [CrossRef]
- Vadevoo, S.M.P.; Gurung, S.; Lee, H.-S.; Gunassekaran, G.R.; Lee, S.-M.; Yoon, J.-W.; Lee, Y.-K.; Lee, B. Peptides as multifunctional players in cancer therapy. Exp. Mol. Med. 2023, 55, 1099–1109. [Google Scholar] [CrossRef]
- Ruoslahti, E.; Pierschbacher, M.D. New perspectives in cell adhesion: RGD and integrins. Science 1987, 238, 491–497. [Google Scholar] [CrossRef]
- Niu, G.; Chen, X. RGD PET: From Lesion Detection to Therapy Response Monitoring. J. Nucl. Med. 2016, 57, 501–502. [Google Scholar] [CrossRef]
- Li, L.; Chen, X.; Yu, J.; Yuan, S. Preliminary Clinical Application of RGD-Containing Peptides as PET Radiotracers for Imaging Tumors. Front. Oncol. 2022, 12, 837952. [Google Scholar] [CrossRef]
- Battistini, L.; Bugatti, K.; Sartori, A.; Curti, C.; Zanardi, F. RGD Peptide-Drug Conjugates as Effective Dual Targeting Platforms: Recent Advances. Eur. J. Org. Chem. 2021, 17, 2506–2528. [Google Scholar] [CrossRef]
- Sugahara, K.N.; Teesalu, T.; Karmali, P.P.; Kotamraju, V.R.; Agemy, L.; Girard, O.M.; Hanahan, D.; Mattrey, R.F.; Ruoslahti, E. Tissue-penetrating delivery of compounds and nanoparticles into tumors. Cancer Cell 2009, 16, 510–520. [Google Scholar] [CrossRef]
- Tian, Y.; Li, S.; Song, J.; Ji, T.; Zhu, M.; Anderson, G.J.; Wei, J.; Nie, G. A doxorubicin delivery platform using engineered natural membrane vesicle exosomes for targeted tumor therapy. Biomaterials 2014, 35, 2383–2390. [Google Scholar] [CrossRef] [PubMed]
- Pfaff, M.; Tangemann, K.; Müller, B.; Gurrath, M.; Müller, G.; Kessler, H.; Timpl, R.; Engel, J. Selective recognition of cyclic RGD peptides of NMR defined conformation by αIIbβ3, αVβ3, and α5β1 integrins. J. Biol. Chem. 1994, 269, 20233–20238. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Jia, L.; Zhu, G.; Wang, W.; Geng, M.; Lu, H.; Zhang, Y.; Zhou, M.; Zhang, F.; Cheng, X. Sortase A-mediated cyclization of novel polycyclic RGD peptides for ανβ3 integrin targeting. Bioorg. Med. Chem. Lett. 2022, 73, 128888. [Google Scholar] [CrossRef]
- Echigo, H.; Mishiro, K.; Munekane, M.; Fuchigami, T.; Kitamura, Y.; Kinuya, S.; Ogawa, K. Development and evaluation of a theranostic probe with RGD peptide introduced platinum complex to enable tumor-specific accumulation. Bioorg. Med. Chem. 2022, 70, 116919. [Google Scholar] [CrossRef] [PubMed]
- Kato, N.; Sato, T.; Fuchigami, Y.; Suga, T.; Geng, L.; Tsurumaru, M.; Hagimori, M.; Mukai, H.; Kawakami, S. Synthesis and evaluation of a novel adapter lipid derivative for preparation of cyclic peptide-modified PEGylated liposomes: Application of cyclic RGD peptide. Eur. J. Pharm. Sci. 2022, 176, 106239. [Google Scholar] [CrossRef]
- Merola, E.; Grana, C.M. Peptide Receptor Radionuclide Therapy (PRRT): Innovations and Improvements. Cancers 2023, 15, 2975. [Google Scholar] [CrossRef]
- Reubi, J.C.; Schaer, J.C.; Waser, B. Cholecystokinin(CCK)-A and CCK-B/gastrin receptors in human tumors. Cancer Res. 1997, 57, 1377–1386. [Google Scholar]
- Strosberg, J.R.; Caplin, M.E.; Kunz, P.L.; Ruszniewski, P.B.; Bodei, L.; Hendifar, A.E.; Mittra, E.; Wolin, E.M.; Yao, J.C.; Pavel, M.E.; et al. Final overall survival in the phase 3 NETTER-1 study of lutetium-177-DOTATATE in patients with midgut neuroendocrine tumors. JCO 2021, 39 (Suppl. 15), 4112. [Google Scholar] [CrossRef]
- Strosberg, J.; El-Haddad, G.; Wolin, E.; Hendifar, A.; Yao, J.; Chasen, B.; Mittra, E.; Kunz, P.L.; Kulke, M.H.; Jacene, H.; et al. Phase 3 Trial of 177Lu-Dotatate for Midgut Neuroendocrine Tumors. N. Engl. J. Med. 2017, 376, 125–135. [Google Scholar] [CrossRef]
- Hope, T.A.; Antonarakis, E.S.; Bodei, L.; Calais, J.; Iravani, A.; Jacene, H.; Koo, P.J.; Morgans, A.K.; Osborne, J.R.; Tagawa, S.T.; et al. SNMMI Consensus Statement on Patient Selection and Appropriate Use of 177Lu-PSMA-617 Radionuclide Therapy. J. Nucl. Med. 2023, 64, jnumed.123.265952. [Google Scholar] [CrossRef]
- Hennrich, U.; Eder, M. [177Lu]Lu-PSMA-617 (PluvictoTM): The First FDA-Approved Radiotherapeutical for Treatment of Prostate Cancer. Pharmaceuticals 2022, 15, 1292. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Li, C.; Zhang, X.; Cheng, L.; Ding, R.; Zhang, L. Degarelix vs. leuprorelin for the treatment of prostate cancer in China: A cost-utility analysis. Front. Public Health 2022, 10, 942800. [Google Scholar] [CrossRef] [PubMed]
- Plosker, G.L.; Brogden, R.N. Leuprorelin: A review of its pharmacology and therapeutic use in prostatic cancer, endometriosis and other sex hormone-related disorders. Drugs 1994, 48, 930–967. [Google Scholar] [CrossRef]
- Garrido, M.P.; Hernandez, A.; Vega, M.; Araya, E.; Romero, C. Conventional and new proposals of GnRH therapy for ovarian, breast, and prostatic cancers. Front. Endocrinol. 2023, 14, 1143261. [Google Scholar] [CrossRef]
- Cheer, S.M.; Plosker, G.L.; Simpson, D.; Wagstaff, A.J. Goserelin: A review of its use in the treatment of early breast cancer in premenopausal and perimenopausal women. Drugs 2005, 65, 2639–2655. [Google Scholar] [CrossRef]
- Chapman, J.-A.W.; Costantino, J.P.; Dong, B.; Margolese, R.G.; Pritchard, K.I.; Shepherd, L.E.; Gelmon, K.A.; Wolmark, N.; Pollak, M.N. Octreotide LAR and tamoxifen versus tamoxifen in phase III randomize early breast cancer trials: NCIC CTG MA.14 and NSABP B-29. Breast Cancer Res. Treat. 2015, 153, 353–360. [Google Scholar] [CrossRef]
- Zengerling, F.; Jakob, J.J.; Schmidt, S.; Meerpohl, J.J.; Blümle, A.; Schmucker, C.; Mayer, B.; Kunath, F. Degarelix for treating advanced hormone-sensitive prostate cancer. Cochrane Database Syst. Rev. 2021, 8, CD012548. [Google Scholar] [CrossRef] [PubMed]
- Jayaweera, S.P.E.; Kanakanamge, S.P.W.; Rajalingam, D.; Silva, G.N. Carfilzomib: A Promising Proteasome Inhibitor for the Treatment of Relapsed and Refractory Multiple Myeloma. Front. Oncol. 2021, 11, 740796. [Google Scholar] [CrossRef]
- Chauhan, A.; El-Khouli, R.; Waits, T.; Agrawal, R.; Siddiqui, F.; Tarter, Z.; Horn, M.; Weiss, H.; Oates, E.; Evers, B.M.; et al. Post FDA approval analysis of 200 gallium-68 DOTATATE imaging: A retrospective analysis in neuroendocrine tumor patients. Oncotarget 2020, 11, 3061–3068. [Google Scholar] [CrossRef]
- Gallium Ga 68 Dotatate. In Drugs and Lactation Database (LactMed®) [Internet]; National Library of Medicine, National Center for Biotechnology Information: Bethesda, MD, USA, 2006. Available online: https://www.ncbi.nlm.nih.gov/books/NBK500826/ (accessed on 30 June 2023).
- Poeppel, T.D.; Binse, I.; Petersenn, S.; Lahner, H.; Schott, M.; Antoch, G.; Brandau, W.; Bockisch, A.; Boy, C. 68Ga-DOTATOC versus 68Ga-DOTATATE PET/CT in functional imaging of neuroendocrine tumors. J. Nucl. Med. 2011, 52, 1864–1870. [Google Scholar] [CrossRef]
- Loft, M.; Carlsen, E.A.; Johnbeck, C.B.; Johannesen, H.H.; Binderup, T.; Pfeifer, A.; Mortensen, J.; Oturai, P.; Loft, A.; Berthelsen, A.K.; et al. 64Cu-DOTATATE PET in Patients with Neuroendocrine Neoplasms: Prospective, Head-to-Head Comparison of Imaging at 1 Hour and 3 Hours After Injection. J. Nucl. Med. 2021, 62, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Carlsen, E.A.; Johnbeck, C.B.; Loft, M.; Pfeifer, A.; Oturai, P.; Langer, S.W.; Knigge, U.; Ladefoged, C.N.; Kjaer, A. Semiautomatic Tumor Delineation for Evaluation of 64Cu-DOTATATE PET/CT in Patients with Neuroendocrine Neoplasms: Prognostication Based on Lowest Lesion Uptake and Total Tumor Volume. J. Nucl. Med. 2021, 62, 1564–1570. [Google Scholar] [CrossRef] [PubMed]
- Pratt, E.C.; Lopez-Montes, A.; Volpe, A.; Crowley, M.J.; Carter, L.M.; Mittal, V.; Pillarsetty, N.; Ponomarev, V.; Udías, J.M.; Grimm, J.; et al. Simultaneous quantitative imaging of two PET radiotracers via the detection of positron-electron annihilation and prompt gamma emissions. Nat. Biomed. Eng. 2023, 7, 1028–1039. [Google Scholar] [CrossRef] [PubMed]
- Montorsi, F.; Gandaglia, G.; Robesti, D.; Dehò, F.; Briganti, A. Re: Kathia De Man, Nick Van Laeken, Vanessa Schelfhout; et al. Re: Kathia De Man, Nick Van Laeken, Vanessa Schelfhout; et al. 18F-PSMA-11 Versus 68Ga-PSMA-11 Positron Emission Tomography/Computed Tomography for Staging and Biochemical Recurrence of Prostate Cancer: A Prospective Double-blind Randomised Cross-over Trial. Eur Urol. 2022;82:501-509. Eur. Urol. 2022, 82, e143. [Google Scholar] [CrossRef]
- Ceci, F.; Rovera, G.; Iorio, G.C.; Guarneri, A.; Chiofalo, V.; Passera, R.; Oderda, M.; Dall’armellina, S.; Liberini, V.; Grimaldi, S.; et al. Event-free survival after 68Ga-PSMA-11 PET/CT in recurrent hormone-sensitive prostate cancer (HSPC) patients eligible for salvage therapy. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 3257–3268. [Google Scholar] [CrossRef]
- Pienta, K.J.; Gorin, M.A.; Rowe, S.P.; Carroll, P.R.; Pouliot, F.; Probst, S.; Saperstein, L.; Preston, M.A.; Alva, A.S.; Patnaik, A.; et al. A Phase 2/3 Prospective Multicenter Study of the Diagnostic Accuracy of Prostate Specific Membrane Antigen PET/CT with 18F-DCFPyL in Prostate Cancer Patients (OSPREY). J. Urol. 2021, 206, 52–61. [Google Scholar] [CrossRef]
- Morris, M.J.; Rowe, S.P.; Gorin, M.A.; Saperstein, L.; Pouliot, F.; Josephson, D.; Wong, J.Y.; Pantel, A.R.; Cho, S.Y.; Gage, K.L.; et al. Diagnostic Performance of 18F-DCFPyL-PET/CT in Men with Biochemically Recurrent Prostate Cancer: Results from the CONDOR Phase III, Multicenter Study. Clin. Cancer Res. 2021, 27, 3674–3682. [Google Scholar] [CrossRef]
- Bruno, B.J.; Miller, G.D.; Lim, C.S. Basics and recent advances in peptide and protein drug delivery. Ther. Deliv. 2013, 4, 1443–1467. [Google Scholar] [CrossRef]
- Hamman, J.H.; Enslin, G.M.; Kotze, A.F. Oral delivery of peptide drugs: Barriers and developments. BioDrugs 2005, 19, 165–177. [Google Scholar] [CrossRef]
- Nhàn, N.T.T.; Maidana, D.E.; Yamada, K.H. Ocular Delivery of Therapeutic Agents by Cell-Penetrating Peptides. Cells 2023, 12, 1071. [Google Scholar] [CrossRef]
- Li, C.M.; Haratipour, P.; Lingeman, R.G.; Perry, J.J.P.; Gu, L.; Hickey, R.J.; Malkas, L.H. Novel Peptide Therapeutic Approaches for Cancer Treatment. Cells 2021, 10, 2908. [Google Scholar] [CrossRef] [PubMed]
- Bottens, R.A.; Yamada, T. Cell-Penetrating Peptides (CPPs) as Therapeutic and Diagnostic Agents for Cancer. Cancers 2022, 14, 5546. [Google Scholar] [CrossRef] [PubMed]
- Mander, S.; Naffouje, S.A.; Gao, J.; Li, W.; Christov, K.; Green, A.; Bongarzone, E.R.; Das Gupta, T.K.; Yamada, T. Tumor-targeting cell-penetrating peptide, p28, for glioblastoma imaging and therapy. Front. Oncol. 2022, 12, 940001. [Google Scholar] [CrossRef]
- Mander, S.; Gorman, G.S.; Coward, L.U.; Christov, K.; Green, A.; Das Gupta, T.K.; Yamada, T. The brain-penetrant cell-cycle inhibitor p28 sensitizes brain metastases to DNA-damaging agents. Neuro-Oncol. Adv. 2023, 5, vdad042. [Google Scholar] [CrossRef]
- Roxin, A.; Zheng, G. Flexible or fixed: A comparative review of linear and cyclic cancer-targeting peptides. Future Med. Chem. 2012, 4, 1601–1618. [Google Scholar] [CrossRef]
- Upadhyaya, P.; Qian, Z.; Selner, N.G.; Clippinger, S.R.; Wu, Z.; Briesewitz, R.; Pei, D. Inhibition of Ras signaling by blocking Ras-effector interactions with cyclic peptides. Angew. Chem. Int. Ed. 2015, 54, 7602–7606. [Google Scholar] [CrossRef] [PubMed]
- LUNA-18 by Chugai Pharmaceutical for Solid Tumor: Likelihood of Approval. 2023. Available online: https://www.pharmaceutical-technology.com/data-insights/luna-18-chugai-pharmaceutical-solid-tumor-likelihood-of-approval/ (accessed on 9 August 2023).
- Papo, N.; Braunstein, A.; Eshhar, Z.; Shai, Y. Suppression of human prostate tumor growth in mice by a cytolytic D-, L-amino Acid Peptide: Membrane lysis, increased necrosis, and inhibition of prostate-specific antigen secretion. Cancer Res. 2004, 64, 5779–5786. [Google Scholar] [CrossRef]
- Harris, A.G. Somatostatin and somatostatin analogues: Pharmacokinetics and pharmacodynamic effects. Gut 1994, 35 (Suppl. 3), S1–S4. [Google Scholar] [CrossRef]
- Lu, J.; Xu, H.; Xia, J.; Ma, J.; Xu, J.; Li, Y.; Feng, J. D- and Unnatural Amino Acid Substituted Antimicrobial Peptides with Improved Proteolytic Resistance and Their Proteolytic Degradation Characteristics. Front. Microbiol. 2020, 11, 563030. [Google Scholar] [CrossRef]
- Zhao, S.; Liu, N.; Wang, W.; Xu, Z.; Wu, Y.; Luo, X. An electrochemical biosensor for α-fetoprotein detection in human serum based on peptides containing isomer D-Amino acids with enhanced stability and antifouling property. Biosens. Bioelectron. 2021, 190, 113466. [Google Scholar] [CrossRef]
- Gupta, V.; Bhavanasi, S.; Quadir, M.; Singh, K.; Ghosh, G.; Vasamreddy, K.; Ghosh, A.; Siahaan, T.J.; Banerjee, S.; Banerjee, S.K. Protein PEGylation for cancer therapy: Bench to bedside. J. Cell Commun. Signal. 2019, 13, 319–330. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Z.Y.; Hou, C.Y.; Zhang, D.; Liu, Y.; Lin, Y.X.; An, H.W.; Li, X.J.; Wang, H. Self-assembly of cytotoxic peptide conjugated poly(β-amino ester)s for synergistic cancer chemotherapy. J. Mater. Chem. B 2015, 3, 2943–2953. [Google Scholar] [CrossRef] [PubMed]
- Suga, T.; Fuchigami, Y.; Hagimori, M.; Kawakami, S. Ligand peptide-grafted PEGylated liposomes using HER2 targeted peptide-lipid derivatives for targeted delivery in breast cancer cells: The effect of serine-glycine repeated peptides as a spacer. Int. J. Pharm. 2017, 521, 361–364. [Google Scholar] [CrossRef]
- Zhang, M.; Xu, H. Peptide-assembled nanoparticles targeting tumor cells and tumor microenvironment for cancer therapy. Front. Chem. 2023, 11, 1115495. [Google Scholar] [CrossRef] [PubMed]
- Cesaro, A.; Lin, S.; Pardi, N.; de la Fuente-Nunez, C. Advanced delivery systems for peptide antibiotics. Adv. Drug Deliv. Rev. 2023, 196, 114733. [Google Scholar] [CrossRef] [PubMed]
- Failla, M.; Floresta, G.; Abbate, V. Peptide-based positron emission tomography probes: Current strategies for synthesis and radiolabelling. RSC Med. Chem. 2023, 14, 592–623. [Google Scholar] [CrossRef]
- Homayun, B.; Lin, X.; Choi, H.-J. Challenges and Recent Progress in Oral Drug Delivery Systems for Biopharmaceuticals. Pharmaceutics 2019, 11, 129. [Google Scholar] [CrossRef]
- Benet, L.Z. Effect of route of administration and distribution on drug action. J. Pharmacokinet. Biopharm. 1978, 6, 559–585. [Google Scholar] [CrossRef]
- Linker, S.M.; Schellhaas, C.; Kamenik, A.S.; Veldhuizen, M.M.; Waibl, F.; Roth, H.-J.; Fouché, M.; Rodde, S.; Riniker, S. Lessons for Oral Bioavailability: How Conformationally Flexible Cyclic Peptides Enter and Cross Lipid Membranes. J. Med. Chem. 2023, 66, 2773–2788. [Google Scholar] [CrossRef]
- Drucker, D.J. Advances in oral peptide therapeutics. Nat. Rev. Drug Discov. 2020, 19, 277–289. [Google Scholar] [CrossRef]
- Yamamoto, A.; Taniguchi, T.; Rikyuu, K.; Tsuji, T.; Fujita, T.; Murakami, M.; Muranishi, S. Effects of various protease inhibitors on the intestinal absorption and degradation of insulin in rats. Pharm. Res. 1994, 11, 1496–1500. [Google Scholar] [CrossRef] [PubMed]
- Hébert, P.C.; Fergusson, D.A.; Hutton, B.; Mazer, C.D.; Fremes, S.; Blajchman, M.; MacAdams, C.; Wells, G.; Robblee, J.; Bussières, J.; et al. Regulatory decisions pertaining to aprotinin may be putting patients at risk. CMAJ 2014, 186, 1379–1386. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.Y.; Al-Salami, H.; Dass, C.R. The role of chitosan on oral delivery of peptide-loaded nanoparticle formulation. J. Drug Target. 2018, 26, 551–562. [Google Scholar] [CrossRef] [PubMed]
- Aungst, B.J. Intestinal permeation enhancers. J. Pharm. Sci. 2000, 89, 429–442. [Google Scholar] [CrossRef]
- Pardridge, W.M. A Historical Review of Brain Drug Delivery. Pharmaceutics 2022, 14, 1283. [Google Scholar] [CrossRef]
- Alabsi, W.; Eedara, B.B.; Encinas-Basurto, D.; Polt, R.; Mansour, H.M. Nose-to-Brain Delivery of Therapeutic Peptides as Nasal Aerosols. Pharmaceutics 2022, 14, 1870. [Google Scholar] [CrossRef]
- Tulpule, A.; Scadden, D.T.; Espina, B.M.; Cabriales, S.; Howard, W.; Shea, K.; Gill, P.S. Results of a randomized study of IM862 nasal solution in the treatment of AIDS-related Kaposi’s sarcoma. J. Clin. Oncol. 2000, 18, 716–723. [Google Scholar] [CrossRef]
- Voelker, R. Cancer Drug in Nasal Drops. JAMA 1999, 281, 2277. [Google Scholar] [CrossRef]
- Samaridou, E.; Alonso, M.J. Nose-to-brain peptide delivery—The potential of nanotechnology. Bioorg. Med. Chem. 2018, 26, 2888–2905. [Google Scholar] [CrossRef]
- Jitendra; Sharma, P.K.; Bansal, S.; Banik, A. Noninvasive routes of proteins and peptides drug delivery. Indian J. Pharm. Sci. 2011, 73, 367–375. [Google Scholar] [CrossRef]
- Mondal, S.B.; O’brien, C.M.; Bishop, K.; Fields, R.C.; Margenthaler, J.A.; Achilefu, S. Repurposing Molecular Imaging and Sensing for Cancer Image-Guided Surgery. J. Nucl. Med. 2020, 61, 1113–1122. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Lu, Z.R. Molecular imaging of the tumor microenvironment. Adv. Drug Deliv. Rev. 2017, 113, 24–48. [Google Scholar] [CrossRef] [PubMed]
- Kastelik-Hryniewiecka, A.; Jewula, P.; Bakalorz, K.; Kramer-Marek, G.; Kuźnik, N. Targeted PET/MRI Imaging Super Probes: A Critical Review of Opportunities and Challenges. Int. J. Nanomed. 2021, 16, 8465–8483. [Google Scholar] [CrossRef] [PubMed]
- Hanaoka, K.; Lubag, A.J.M.; Castillo-Muzquiz, A.; Kodadek, T.; Sherry, A.D. The detection limit of a Gd3+-based T1 agent is substantially reduced when targeted to a protein microdomain. Magn. Reson. Imaging 2008, 26, 608–617. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Peng, J.; Li, J.; Huang, L.; Yang, J.; Huang, K.; Li, H.; Jiang, N.; Zheng, S.; Zhang, X.; et al. Tumor-Targeted and Clearable Human Protein-Based MRI Nanoprobes. Nano. Lett. 2017, 17, 4096–4100. [Google Scholar] [CrossRef]
- Zhang, P.; Cui, Y.; Anderson, C.F.; Zhang, C.; Li, Y.; Wang, R.; Cui, H. Peptide-based nanoprobes for molecular imaging and disease diagnostics. Chem. Soc. Rev. 2018, 47, 3490–3529. [Google Scholar] [CrossRef]
- Vithanarachchi, S.M.; Allen, M.J. Strategies for Target-Specific Contrast Agents for Magnetic Resonance Imaging. Curr. Mol. Imaging 2012, 1, 12–25. [Google Scholar] [CrossRef]
- Koudrina, A.; DeRosa, M.C. Advances in Medical Imaging: Aptamer- and Peptide-Targeted MRI and CT Contrast Agents. ACS Omega 2020, 5, 22691–22701. [Google Scholar] [CrossRef]
- Sun, X.; Li, Y.; Liu, T.; Li, Z.; Zhang, X.; Chen, X. Peptide-based imaging agents for cancer detection. Adv. Drug Deliv. Rev. 2017, 110–111, 38–51. [Google Scholar] [CrossRef]
- Ding, C.; Wu, K.; Wang, W.; Guan, Z.; Wang, L.; Wang, X.; Wang, R.; Liu, L.; Fan, J. Synthesis of a cell penetrating peptide modified superparamagnetic iron oxide and MRI detection of bladder cancer. Oncotarget 2017, 8, 4718–4729. [Google Scholar] [CrossRef]
- Jaspers, K.; Aerts, H.J.; Leiner, T.; Oostendorp, M.; van Riel, N.A.; Post, M.J.; Backes, W.H. Reliability of pharmacokinetic parameters: Small vs. medium-sized contrast agents. Magn. Reson. Med. 2009, 62, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Hao, D.; Ai, T.; Goerner, F.; Hu, X.; Runge, V.M.; Tweedle, M. MRI contrast agents: Basic chemistry and safety. J. Magn. Reson. Imaging 2012, 36, 1060–1071. [Google Scholar] [CrossRef] [PubMed]
- Xue, S.; Qiao, J.; Jiang, J.; Hubbard, K.; White, N.; Wei, L.; Li, S.; Liu, Z.-R.; Yang, J.J. Design of ProCAs (protein-based Gd3+ MRI contrast agents) with high dose efficiency and capability for molecular imaging of cancer biomarkers. Med. Res. Rev. 2014, 34, 1070–1099. [Google Scholar] [CrossRef] [PubMed]
- Wahsner, J.; Gale, E.M.; Rodríguez-Rodríguez, A.; Caravan, P. Chemistry of MRI Contrast Agents: Current Challenges and New Frontiers. Chem. Rev. 2019, 119, 957–1057. [Google Scholar] [CrossRef] [PubMed]
- Joffe, P.; Thomsen, H.S.; Meusel, M. Pharmacokinetics of gadodiamide injection in patients with severe renal insufficiency and patients undergoing hemodialysis or continuous ambulatory peritoneal dialysis. Acad. Radiol. 1998, 5, 491–502. [Google Scholar] [CrossRef]
- Bai, J.W.; Qiu, S.Q.; Zhang, G.J. Molecular and functional imaging in cancer-targeted therapy: Current applications and future directions. Signal Transduct. Target. Ther. 2023, 8, 89. [Google Scholar] [CrossRef]
- Zhou, Z.; Wu, X.; Kresak, A.; Griswold, M.; Lu, Z.-R. Peptide targeted tripod macrocyclic Gd(III) chelates for cancer molecular MRI. Biomaterials 2013, 34, 7683–7693. [Google Scholar] [CrossRef]
- Zhang, N.; Ru, B.; Hu, J.; Xu, L.; Wan, Q.; Liu, W.; Cai, W.; Zhu, T.; Ji, Z.; Guo, R.; et al. Recent advances of CREKA peptide-based nanoplatforms in biomedical applications. J. Nanobiotechnol. 2023, 21, 77. [Google Scholar] [CrossRef]
- Benfante, V.; Stefano, A.; Ali, M.; Laudicella, R.; Arancio, W.; Cucchiara, A.; Caruso, F.; Cammarata, F.P.; Coronnello, C.; Russo, G.; et al. An Overview of In Vitro Assays of 64Cu-, 68Ga-, 125I-, and 99mTc-Labelled Radiopharmaceuticals Using Radiometric Counters in the Era of Radiotheranostics. Diagnostics 2023, 13, 1210. [Google Scholar] [CrossRef]
- Duatti, A. Review on 99mTc radiopharmaceuticals with emphasis on new advancements. Nucl. Med. Biol. 2021, 92, 202–216. [Google Scholar] [CrossRef]
- Cai, W.; Niu, G.; Chen, X. Multimodality imaging of the HER-kinase axis in cancer. Eur. J. Nucl. Med. Mol. Imaging 2008, 35, 186–208. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Xie, J.; Chen, X. Peptides and peptide hormones for molecular imaging and disease diagnosis. Chem. Rev. 2010, 110, 3087–3111. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.R.; Deutscher, S.L. 111In-labeled galectin-3-targeting peptide as a SPECT agent for imaging breast tumors. J. Nucl. Med. 2008, 49, 796–803. [Google Scholar] [CrossRef] [PubMed]
- Ahmadpour, S.; Hosseinimehr, S.J. Recent developments in peptide-based SPECT radiopharmaceuticals for breast tumor targeting. Life Sci. 2019, 239, 116870. [Google Scholar] [CrossRef]
- Zhu, Z.; Miao, W.; Li, Q.; Dai, H.; Ma, Q.; Wang, F.; Yang, A.; Jia, B.; Jing, X.; Liu, S.; et al. 99mTc-3PRGD2 for integrin receptor imaging of lung cancer: A multicenter study. J. Nucl. Med. 2012, 53, 716–722. [Google Scholar] [CrossRef]
- Scroggie, K.R.; Perkins, M.V.; Chalker, J.M. Reaction of [18F]Fluoride at Heteroatoms and Metals for Imaging of Peptides and Proteins by Positron Emission Tomography. Front. Chem. 2021, 9, 6876789. [Google Scholar] [CrossRef]
- Pimlott, S.L.; Sutherland, A. Molecular tracers for the PET and SPECT imaging of disease. Chem. Soc. Rev. 2011, 40, 149–162. [Google Scholar] [CrossRef]
- Nolting, D.D.; Nickels, M.L.; Guo, N.; Pham, W. Molecular imaging probe development: A chemistry perspective. Am. J. Nucl. Med. Mol. Imaging 2012, 2, 273–306. [Google Scholar]
- Jackson, I.M.; Scott, P.J.H.; Thompson, S. Clinical Applications of Radiolabeled Peptides for PET. Semin Nucl. Med. 2017, 47, 493–523. [Google Scholar] [CrossRef]
- Wu, C.; Yue, X.; Lang, L.; Kiesewetter, D.O.; Li, F.; Zhu, Z.; Niu, G.; Chen, X. Longitudinal PET imaging of muscular inflammation using 18F-DPA-714 and 18F-Alfatide II and differentiation with tumors. Theranostics 2014, 4, 546–555. [Google Scholar] [CrossRef]
- Schottelius, M.; Poethko, T.; Herz, M.; Reubi, J.-C.; Kessler, H.; Schwaiger, M.; Wester, H.-J. First 18F-labeled tracer suitable for routine clinical imaging of sst receptor-expressing tumors using positron emission tomography. Clin. Cancer Res. 2004, 10, 3593–3606. [Google Scholar] [CrossRef] [PubMed]
- Persson, M.; Skovgaard, D.; Brandt-Larsen, M.; Christensen, C.; Madsen, J.; Nielsen, C.H.; Thurison, T.; Klausen, T.L.; Holm, S.; Loft, A.; et al. First-in-human uPAR PET: Imaging of Cancer Aggressiveness. Theranostics 2015, 5, 1303–1316. [Google Scholar] [CrossRef] [PubMed]
- Kircher, M.; Herhaus, P.; Schottelius, M.; Buck, A.K.; Werner, R.A.; Wester, H.-J.; Keller, U.; Lapa, C. CXCR4-directed theranostics in oncology and inflammation. Ann. Nucl. Med. 2018, 32, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Benešová, M.; Schäfer, M.; Bauder-Wüst, U.; Afshar-Oromieh, A.; Kratochwil, C.; Mier, W.; Haberkorn, U.; Kopka, K.; Eder, M. Preclinical Evaluation of a Tailor-Made DOTA-Conjugated PSMA Inhibitor with Optimized Linker Moiety for Imaging and Endoradiotherapy of Prostate Cancer. J. Nucl. Med. 2015, 56, 914–920. [Google Scholar] [CrossRef]
- Behe, M.; Behr, T.M. Cholecystokinin-B (CCK-B)/gastrin receptor targeting peptides for staging and therapy of medullary thyroid cancer and other CCK-B receptor expressing malignancies. Biopolymers 2002, 66, 399–418. [Google Scholar] [CrossRef]
- Manoharan, P.; Lamarca, A.; Navalkissoor, S.; Calero, J.; Chan, P.S.; Julyan, P.; Sierra, M.; Caplin, M.; Valle, J. Safety, tolerability and clinical implementation of ‘ready-to-use’ 68gallium-DOTA0-Tyr3-octreotide (68Ga-DOTATOC) (SomaKIT TOC) for injection in patients diagnosed with gastroenteropancreatic neuroendocrine tumours (GEP-NETs). ESMO Open 2020, 5, e000650. [Google Scholar] [CrossRef]
- Werner, R.A.; Derlin, T.; Lapa, C.; Sheikbahaei, S.; Higuchi, T.; Giesel, F.L.; Behr, S.; Drzezga, A.; Kimura, H.; Buck, A.K.; et al. 18F-Labeled, PSMA-Targeted Radiotracers: Leveraging the Advantages of Radiofluorination for Prostate Cancer Molecular Imaging. Theranostics 2020, 10, 1–16. [Google Scholar] [CrossRef]
- Tan, N.; Oyoyo, U.; Bavadian, N.; Ferguson, N.; Mukkamala, A.; Calais, J.; Davenport, M.S. PSMA-targeted Radiotracers versus 18F Fluciclovine for the Detection of Prostate Cancer Biochemical Recurrence after Definitive Therapy: A Systematic Review and Meta-Analysis. Radiology 2020, 296, 44–55. [Google Scholar] [CrossRef]
- Mena, E.; Lindenberg, M.L.; Turkbey, I.B.; Shih, J.H.; Harmon, S.A.; Lim, I.; Lin, F.; Adler, S.; Eclarinal, P.; McKinney, Y.L.; et al. 18F-DCFPyL PET/CT Imaging in Patients with Biochemically Recurrent Prostate Cancer after Primary Local Therapy. J. Nucl. Med. 2020, 61, 881–889. [Google Scholar] [CrossRef]
- Serkova, N.J.; Glunde, K.; Haney, C.R.; Farhoud, M.; De Lille, A.; Redente, E.F.; Simberg, D.; Westerly, D.C.; Griffin, L.; Mason, R.P. Preclinical Applications of Multi-Platform Imaging in Animal Models of Cancer. Cancer Res. 2021, 81, 1189–1200. [Google Scholar] [CrossRef]
- Orosco, R.K.; Tapia, V.J.; Califano, J.A.; Clary, B.; Cohen, E.E.W.; Kane, C.; Lippman, S.M.; Messer, K.; Molinolo, A.; Murphy, J.D.; et al. Positive Surgical Margins in the 10 Most Common Solid Cancers. Sci. Rep. 2018, 8, 5686. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, E.L.; Warram, J.M.; Bland, K.I.; Zinn, K.R.D. The status of contemporary image-guided modalities in oncologic surgery. Ann. Surg. 2015, 261, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Sutton, P.A.; van Dam, M.A.; Cahill, R.A.; Mieog, S.; Polom, K.; Vahrmeijer, A.L.; van der Vorst, J. Fluorescence-guided surgery: Comprehensive review. BJS Open 2023, 7, zrad049. [Google Scholar] [CrossRef] [PubMed]
- Boekestijn, I.; van Oosterom, M.N.; Dell’oglio, P.; van Velden, F.H.P.; Pool, M.; Maurer, T.; Rietbergen, D.D.D.; Buckle, T.; van Leeuwen, F.W.B. The current status and future prospects for molecular imaging-guided precision surgery. Cancer Imaging 2022, 22, 48. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.R.; Schroeder, A.B.; Grudzinski, J.J.; Rosenthal, E.L.; Warram, J.M.; Pinchuk, A.N.; Eliceiri, K.W.; Kuo, J.S.; Weichert, J.P. Beyond the margins: Real-time detection of cancer using targeted fluorophores. Nat. Rev. Clin. Oncol. 2017, 14, 347–364. [Google Scholar] [CrossRef]
- Naffouje, S.A.; Goto, M.; Coward, L.U.; Gorman, G.S.; Christov, K.; Wang, J.; Green, A.; Shilkaitis, A.; Das Gupta, T.K.; Yamada, T. Nontoxic Tumor-Targeting Optical Agents for Intraoperative Breast Tumor Imaging. J. Med. Chem. 2022, 65, 7371–7379. [Google Scholar] [CrossRef]
- Goto, M.; Ryoo, I.; Naffouje, S.; Mander, S.; Christov, K.; Wang, J.; Green, A.; Shilkaitis, A.; Das Gupta, T.K.; Yamada, T. Image-guided surgery with a new tumour-targeting probe improves the identification of positive margins. EBioMedicine 2022, 76, 103850. [Google Scholar] [CrossRef]
- Bazak, R.; Houri, M.; EL Achy, S.; Hussein, W.; Refaat, T. Passive targeting of nanoparticles to cancer: A comprehensive review of the literature. Mol. Clin. Oncol. 2014, 2, 904–908. [Google Scholar] [CrossRef]
- Matsumura, Y.; Maeda, H. A new concept for macromolecular therapeutics in cancer chemotherapy: Mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986, 46 Pt 1, 6387–6392. [Google Scholar]
- Maeda, H. Polymer therapeutics and the EPR effect. J. Drug Target. 2017, 25, 781–785. [Google Scholar] [CrossRef]
- Maeda, H. The link between infection and cancer: Tumor vasculature, free radicals, and drug delivery to tumors via the EPR effect. Cancer Sci. 2013, 104, 779–789. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H. Tumor-selective delivery of macromolecular drugs via the EPR effect: Background and future prospects. Bioconjug. Chem. 2010, 21, 797–802. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Nakamura, H.; Maeda, H. The EPR effect: Unique features of tumor blood vessels for drug delivery, factors involved, and limitations and augmentation of the effect. Adv. Drug Deliv. Rev. 2011, 63, 136–151. [Google Scholar] [CrossRef]
- Jain, R.K. The next frontier of molecular medicine: Delivery of therapeutics. Nat. Med. 1998, 4, 655–657. [Google Scholar] [CrossRef]
- Gavas, S.; Quazi, S.; Karpinski, T.M. Nanoparticles for Cancer Therapy: Current Progress and Challenges. Nanoscale Res. Lett. 2021, 16, 173. [Google Scholar] [CrossRef] [PubMed]
- Samec, T.; Boulos, J.; Gilmore, S.; Hazelton, A.; Alexander-Bryant, A. Peptide-based delivery of therapeutics in cancer treatment. Mater. Today Bio 2022, 14, 100248. [Google Scholar] [CrossRef] [PubMed]
- Cheetham, A.G.; Keith, D.; Zhang, P.; Lin, R.; Su, H.; Cui, H. Targeting Tumors with Small Molecule Peptides. Curr. Cancer Drug Targets 2016, 16, 489–508. [Google Scholar] [CrossRef] [PubMed]
- Worm, D.J.; Els-Heindl, S.; Beck-Sickinger, A.G. Targeting of peptide-binding receptors on cancer cells with peptide-drug conjugates. Pept. Sci. 2020, 112, e24171. [Google Scholar] [CrossRef]
- Xie, J.; Bi, Y.; Zhang, H.; Dong, S.; Teng, L.; Lee, R.J.; Yang, Z. Cell-Penetrating Peptides in Diagnosis and Treatment of Human Diseases: From Preclinical Research to Clinical Application. Front. Pharmacol. 2020, 11, 697. [Google Scholar] [CrossRef]
- Vives, E.; Schmidt, J.; Pelegrin, A. Cell-penetrating and cell-targeting peptides in drug delivery. Biochim. Biophys. Acta (BBA) Rev. Cancer 2008, 1786, 126–138. [Google Scholar] [CrossRef]
- Arvanitis, C.D.; Ferraro, G.B.; Jain, R.K. The blood-brain barrier and blood-tumour barrier in brain tumours and metastases. Nat. Rev. Cancer 2020, 20, 26–41. [Google Scholar] [CrossRef] [PubMed]
- Kucharz, K.; Kutuzov, N.; Zhukov, O.; Janiurek, M.M.; Lauritzen, M. Shedding Light on the Blood-Brain Barrier Transport with Two-Photon Microscopy In Vivo. Pharm. Res. 2022, 39, 1457–1468. [Google Scholar] [CrossRef] [PubMed]
- Wettschureck, N.; Strilic, B.; Offermanns, S. Passing the Vascular Barrier: Endothelial Signaling Processes Controlling Extravasation. Physiol. Rev. 2019, 99, 1467–1525. [Google Scholar] [CrossRef] [PubMed]
- Karmur, B.S.; Philteos, J.; Abbasian, A.; Zacharia, B.E.; Lipsman, N.; Levin, V.; Grossman, S.; Mansouri, A. Blood-Brain Barrier Disruption in Neuro-Oncology: Strategies, Failures, and Challenges to Overcome. Front. Oncol. 2020, 10, 563840. [Google Scholar] [CrossRef] [PubMed]
- Unterberg, A.; Wahl, M.; Baethmann, A. Effects of bradykinin on permeability and diameter of pial vessels in vivo. J. Cereb. Blood Flow Metab. 1984, 4, 574–585. [Google Scholar] [CrossRef]
- Nakano, S.; Matsukado, K.; Black, K.L. Increased brain tumor microvessel permeability after intracarotid bradykinin infusion is mediated by nitric oxide. Cancer Res. 1996, 56, 4027–4031. [Google Scholar]
- Black, K.L.; Cloughesy, T.; Huang, S.-C.; Gobin, Y.P.; Zhou, Y.; Grous, J.; Nelson, G.; Farahani, K.; Hoh, C.K.; Phelps, M.; et al. Intracarotid infusion of RMP-7, a bradykinin analog, and transport of gallium-68 ethylenediamine tetraacetic acid into human gliomas. J. Neurosurg. 1997, 86, 603–609. [Google Scholar] [CrossRef]
- Neuwelt, E.A.; Maravilla, K.R.; Frenkel, E.P.; Rapaport, S.I.; Hill, S.A.; Barnett, P.A. Osmotic blood-brain barrier disruption. Computerized tomographic monitoring of chemotherapeutic agent delivery. J. Clin. Investig. 1979, 64, 684–688. [Google Scholar] [CrossRef]
- Kuitunen, H.K.; Rönkä, A.L.K.; Sonkajärvi, E.M.; Isokangas, J.-M.; Pyörälä, M.; Palosaari, K.A.A.; Jokimäki, A.S.; Partanen, A.E.; Littow, H.J.; Vakkala, M.A.; et al. Blood–Brain Barrier Disruption (BBBD)-Based Immunochemotherapy for Primary Central Nervous System Lymphoma (PCNSL), Early Results of a Phase II Study. Cancers 2023, 15, 1341. [Google Scholar] [CrossRef]
- Iorio-Morin, C.; Gahide, G.; Morin, C.; Vanderweyen, D.; Roy, M.A.; St-Pierre, I.; Massicotte-Tisluck, K.; Fortin, D. Management of Primary Central Nervous System Lymphoma Using Intra-Arterial Chemotherapy with Osmotic Blood-Brain Barrier Disruption: Retrospective Analysis of the Sherbrooke Cohort. Front. Oncol. 2020, 10, 543648. [Google Scholar] [CrossRef]
- Angelov, L.; Doolittle, N.D.; Kraemer, D.F.; Siegal, T.; Barnett, G.H.; Peereboom, D.M.; Stevens, G.; McGregor, J.; Jahnke, K.; Lacy, C.A.; et al. Blood-brain barrier disruption and intra-arterial methotrexate-based therapy for newly diagnosed primary CNS lymphoma: A multi-institutional experience. J. Clin. Oncol. 2009, 27, 3503–3509. [Google Scholar] [CrossRef] [PubMed]
- Jendrossek, V.; Erdlenbruch, B.; Lakomek, M.; Eibl, H. Transient and controllable opening of the blood-brain barrier to cytostatic and antibiotic agents by alkylglycerols in rats. Exp. Brain Res. 2000, 135, 417–422. [Google Scholar] [CrossRef]
- Jackson, S.; Weingart, J.; Nduom, E.K.; Harfi, T.T.; George, R.T.; McAreavey, D.; Ye, X.; Anders, N.M.; Peer, C.; Figg, W.D.; et al. The effect of an adenosine A2A agonist on intra-tumoral concentrations of temozolomide in patients with recurrent glioblastoma. Fluids Barriers CNS 2018, 15, 2. [Google Scholar] [CrossRef]
- Jackson, S.; George, R.T.; Lodge, M.A.; Piotrowski, A.; Wahl, R.L.; Gujar, S.K.; Grossman, S.A. The effect of regadenoson on the integrity of the human blood-brain barrier, a pilot study. J. Neurooncol. 2017, 132, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Beccaria, K.; Canney, M.; Goldwirt, L.; Fernandez, C.; Adam, C.; Piquet, J.; Autret, G.; Clément, O.; Lafon, C.; Chapelon, J.-Y.; et al. Opening of the blood-brain barrier with an unfocused ultrasound device in rabbits. J. Neurosurg. 2013, 119, 887–898. [Google Scholar] [CrossRef]
- Idbaih, A.; Canney, M.; Belin, L.; Desseaux, C.; Vignot, A.; Bouchoux, G.; Asquier, N.; Law-Ye, B.; Leclercq, D.; Bissery, A.; et al. Safety and Feasibility of Repeated and Transient Blood-Brain Barrier Disruption by Pulsed Ultrasound in Patients with Recurrent Glioblastoma. Clin. Cancer Res. 2019, 25, 3793–3801. [Google Scholar] [CrossRef]
- Patel, B.; Yang, P.H.; Kim, A.H. The effect of thermal therapy on the blood-brain barrier and blood-tumor barrier. Int. J. Hyperth. 2020, 37, 35–43. [Google Scholar] [CrossRef]
- Kiyatkin, E.A.; Sharma, H.S. Permeability of the blood-brain barrier depends on brain temperature. Neuroscience 2009, 161, 926–939. [Google Scholar] [CrossRef]
- Semyachkina-Glushkovskaya, O.; Bragin, D.; Bragina, O.; Socolovski, S.; Shirokov, A.; Fedosov, I.; Ageev, V.; Blokhina, I.; Dubrovsky, A.; Telnova, V.; et al. Low-Level Laser Treatment Induces the Blood-Brain Barrier Opening and the Brain Drainage System Activation: Delivery of Liposomes into Mouse Glioblastoma. Pharmaceutics 2023, 15, 567. [Google Scholar] [CrossRef]
- Leuthardt, E.C.; Duan, C.; Kim, M.J.; Campian, J.L.; Kim, A.H.; Miller-Thomas, M.M.; Shimony, J.S.; Tran, D.D. Hyperthermic Laser Ablation of Recurrent Glioblastoma Leads to Temporary Disruption of the Peritumoral Blood Brain Barrier. PLoS ONE 2016, 11, e0148613. [Google Scholar] [CrossRef]
- Sheikov, N.; McDannold, N.; Jolesz, F.; Zhang, Y.-Z.; Tam, K.; Hynynen, K. Brain arterioles show more active vesicular transport of blood-borne tracer molecules than capillaries and venules after focused ultrasound-evoked opening of the blood-brain barrier. Ultrasound. Med. Biol. 2006, 32, 1399–1409. [Google Scholar] [CrossRef] [PubMed]
- Mainprize, T.; Lipsman, N.; Huang, Y.; Meng, Y.; Bethune, A.; Ironside, S.; Heyn, C.; Alkins, R.; Trudeau, M.; Sahgal, A.; et al. Blood-Brain Barrier Opening in Primary Brain Tumors with Non-invasive MR-Guided Focused Ultrasound: A Clinical Safety and Feasibility Study. Sci. Rep. 2019, 9, 321. [Google Scholar] [CrossRef] [PubMed]
- Ronaldson, P.T.; Davis, T.P. Targeting blood-brain barrier changes during inflammatory pain: An opportunity for optimizing CNS drug delivery. Ther. Deliv. 2011, 2, 1015–1041. [Google Scholar] [CrossRef] [PubMed]
- Greene, C.; Hanley, N.; Campbell, M. Claudin-5: Gatekeeper of neurological function. Fluids Barriers CNS 2019, 16, 3. [Google Scholar] [CrossRef]
- Bocsik, A.; Walter, F.R.; Gyebrovszki, A.; Fülöp, L.; Blasig, I.; Dabrowski, S.; Ötvös, F.; Tóth, A.; Rákhely, G.; Veszelka, S.; et al. Reversible Opening of Intercellular Junctions of Intestinal Epithelial and Brain Endothelial Cells with Tight Junction Modulator Peptides. J. Pharm. Sci. 2016, 105, 754–765. [Google Scholar] [CrossRef]
- Herve, F.; Ghinea, N.; Scherrmann, J.M. CNS delivery via adsorptive transcytosis. AAPS J. 2008, 10, 455–472. [Google Scholar] [CrossRef]
- Lajoie, J.M.; Shusta, E.V. Targeting receptor-mediated transport for delivery of biologics across the blood-brain barrier. Annu. Rev. Pharmacol. Toxicol. 2015, 55, 613–631. [Google Scholar] [CrossRef]
- Uchida, Y.; Tachikawa, M.; Obuchi, W.; Hoshi, Y.; Tomioka, Y.; Ohtsuki, S.; Terasaki, T. A study protocol for quantitative targeted absolute proteomics (QTAP) by LC-MS/MS: Application for inter-strain differences in protein expression levels of transporters, receptors, claudin-5, and marker proteins at the blood-brain barrier in ddY, FVB, and C57BL/6J mice. Fluids Barriers CNS 2013, 10, 21. [Google Scholar] [CrossRef]
- Uchida, Y.; Ohtsuki, S.; Katsukura, Y.; Ikeda, C.; Suzuki, T.; Kamiie, J.; Terasaki, T. Quantitative targeted absolute proteomics of human blood-brain barrier transporters and receptors. J. Neurochem. 2011, 117, 333–345. [Google Scholar] [CrossRef]
- Daneman, R.; Zhou, L.; Agalliu, D.; Cahoy, J.D.; Kaushal, A.; Barres, B.A. The mouse blood-brain barrier transcriptome: A new resource for understanding the development and function of brain endothelial cells. PLoS ONE 2010, 5, e13741. [Google Scholar] [CrossRef]
- Dehouck, B.; Fenart, L.; Dehouck, M.-P.; Pierce, A.; Torpier, G.; Cecchelli, R. A new function for the LDL receptor: Transcytosis of LDL across the blood-brain barrier. J. Cell Biol. 1997, 138, 877–889. [Google Scholar] [CrossRef]
- Qian, Z.M.; Li, H.; Sun, H.; Ho, K. Targeted drug delivery via the transferrin receptor-mediated endocytosis pathway. Pharmacol. Rev. 2002, 54, 561–587. [Google Scholar] [CrossRef] [PubMed]
- Duffy, K.R.; Pardridge, W.M. Blood-brain barrier transcytosis of insulin in developing rabbits. Brain Res. 1987, 420, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Tu, H.; Pan, W.; Feucht, L.; Kastin, A.J. Convergent trafficking pattern of leptin after endocytosis mediated by ObRa-ObRd. J. Cell. Physiol. 2007, 212, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Banks, W.A.; Kastin, A.J.; Huang, W.; Jaspan, J.B.; Maness, L.M. Leptin enters the brain by a saturable system independent of insulin. Peptides 1996, 17, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Notarnicola, M.; Linsalata, M.; Caruso, M.G.; Cavallini, A.; Di Leo, A. Low density lipoprotein receptors and polyamine levels in human colorectal adenocarcinoma. J. Gastroenterol. 1995, 30, 705–709. [Google Scholar] [CrossRef] [PubMed]
- Chung, N.S.; Wasan, K.M. Potential role of the low-density lipoprotein receptor family as mediators of cellular drug uptake. Adv. Drug Deliv. Rev. 2004, 56, 1315–1334. [Google Scholar] [CrossRef]
- Kreuter, J.; Hekmatara, T.; Dreis, S.; Vogel, T.; Gelperina, S.; Langer, K. Covalent attachment of apolipoprotein A-I and apolipoprotein B-100 to albumin nanoparticles enables drug transport into the brain. J. Control. Release 2007, 118, 54–58. [Google Scholar] [CrossRef]
- Spencer, B.J.; Verma, I.M. Targeted delivery of proteins across the blood-brain barrier. Proc. Natl. Acad. Sci. USA 2007, 104, 7594–7599. [Google Scholar] [CrossRef]
- Wang, D.; El-Amouri, S.S.; Dai, M.; Kuan, C.-Y.; Hui, D.Y.; Brady, R.O.; Pan, D. Engineering a lysosomal enzyme with a derivative of receptor-binding domain of apoE enables delivery across the blood-brain barrier. Proc. Natl. Acad. Sci. USA 2013, 110, 2999–3004. [Google Scholar] [CrossRef]
- Wang, X.-S.; Ciraolo, G.; Morris, R.; Gruenstein, E. Identification of a neuronal endocytic pathway activated by an apolipoprotein E (apoE) receptor binding peptide. Brain Res. 1997, 778, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Clayton, D.; Brereton, I.M.; Kroon, P.A.; Smith, R. NMR studies of the low-density lipoprotein receptor-binding peptide of apolipoprotein E bound to dodecylphosphocholine micelles. Protein. Sci. 1999, 8, 1797–1805. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fillebeen, C.; Descamps, L.; Dehouck, M.-P.; Fenart, L.; Benaıssa, M.; Spik, G.; Cecchelli, R.; Pierce, A. Receptor-mediated transcytosis of lactoferrin through the blood-brain barrier. J. Biol. Chem. 1999, 274, 7011–7017. [Google Scholar] [CrossRef] [PubMed]
- Demeule, M.; Poirier, J.; Jodoin, J.; Bertrand, Y.; Desrosiers, R.R.; Dagenais, C.; Nguyen, T.; Lanthier, J.; Gabathuler, R.; Kennard, M.; et al. High transcytosis of melanotransferrin (P97) across the blood-brain barrier. J. Neurochem. 2002, 83, 924–933. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Kastin, A.J.; Zankel, T.C.; van Kerkhof, P.; Terasaki, T.; Bu, G. Efficient transfer of receptor-associated protein (RAP) across the blood-brain barrier. J. Cell Sci. 2004, 117 Pt 21, 5071–5078. [Google Scholar] [CrossRef] [PubMed]
- Benchenane, K.; Berezowski, V.; Fernández-Monreal, M.; Brillault, J.; Valable, S.; Dehouck, M.-P.; Cecchelli, R.; Vivien, D.; Touzani, O.; Ali, C.; et al. Oxygen glucose deprivation switches the transport of tPA across the blood-brain barrier from an LRP-dependent to an increased LRP-independent process. Stroke 2005, 36, 1059–1064. [Google Scholar] [CrossRef] [PubMed]
- Kounnas, M.Z.; Moir, R.D.; Rebeck, G.W.; Bush, A.I.; Argraves, W.S.; Tanzi, R.E.; Hyman, B.T.; Strickland, D.K. LDL receptor-related protein, a multifunctional ApoE receptor, binds secreted β-amyloid precursor protein and mediates its degradation. Cell 1995, 82, 331–340. [Google Scholar] [CrossRef]
- Moestrup, S.K.; Cui, S.; Vorum, H.; Bregengård, C.; Bjørn, S.E.; Norris, K.; Gliemann, J.; Christensen, E.I. Evidence that epithelial glycoprotein 330/megalin mediates uptake of polybasic drugs. J. Clin. Investig. 1995, 96, 1404–1413. [Google Scholar] [CrossRef]
- Demeule, M.; Régina, A.; Ché, C.; Poirier, J.; Nguyen, T.; Gabathuler, R.; Castaigne, J.-P.; Béliveau, R. Identification and design of peptides as a new drug delivery system for the brain. J. Pharmacol. Exp. Ther. 2008, 324, 1064–1072. [Google Scholar] [CrossRef]
- Demeule, M.; Currie, J.-C.; Bertrand, Y.; Ché, C.; Nguyen, T.; Régina, A.; Gabathuler, R.; Castaigne, J.-P.; Béliveau, R. Involvement of the low-density lipoprotein receptor-related protein in the transcytosis of the brain delivery vector angiopep-2. J. Neurochem. 2008, 106, 1534–1544. [Google Scholar] [CrossRef]
- Ché, C.; Yang, G.; Thiot, C.; Lacoste, M.-C.; Currie, J.-C.; Demeule, M.; Régina, A.; Béliveau, R.; Castaigne, J.-P. New Angiopep-modified doxorubicin (ANG1007) and etoposide (ANG1009) chemotherapeutics with increased brain penetration. J. Med. Chem. 2010, 53, 2814–2824. [Google Scholar] [CrossRef] [PubMed]
- Régina, A.; Demeule, M.; Ché, C.; Lavallée, I.; Poirier, J.; Gabathuler, R.; Béliveau, R.; Castaigne, J.-P. Antitumour activity of ANG1005, a conjugate between paclitaxel and the new brain delivery vector Angiopep-2. Br. J. Pharmacol. 2008, 155, 185–197. [Google Scholar] [CrossRef]
- Demeule, M.; Beaudet, N.; Régina, A.; Besserer-Offroy, É.; Murza, A.; Tétreault, P.; Belleville, K.; Ché, C.; Larocque, A.; Thiot, C.; et al. Conjugation of a brain-penetrant peptide with neurotensin provides antinociceptive properties. J. Clin. Investig. 2014, 124, 1199–1213. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, Y.; Currie, J.-C.; Poirier, J.; Demeule, M.; Abulrob, A.; Fatehi, D.; Stanimirovic, D.; Sartelet, H.; Castaigne, J.-P.; Béliveau, R. Influence of glioma tumour microenvironment on the transport of ANG1005 via low-density lipoprotein receptor-related protein 1. Br. J. Cancer 2011, 105, 1697–1707. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.-Z.; Li, J.-Q.; Wang, Z.-Z.; Dong, D.-W.; Qi, X.-R. Tumor-targeting dual peptides-modified cationic liposomes for delivery of siRNA and docetaxel to gliomas. Biomaterials 2014, 35, 5226–5239. [Google Scholar] [CrossRef]
- Wängler, C.; Nada, D.; Höfner, G.; Maschauer, S.; Wängler, B.; Schneider, S.; Schirrmacher, E.; Wanner, K.T.; Schirrmacher, R.; Prante, O. In vitro and initial in vivo evaluation of 68Ga-labeled transferrin receptor (TfR) binding peptides as potential carriers for enhanced drug transport into TfR expressing cells. Mol. Imaging Biol. 2011, 13, 332–341. [Google Scholar] [CrossRef] [PubMed]
- Mu, L.M.; Bu, Y.Z.; Liu, L.; Xie, H.J.; Ju, R.J.; Wu, J.S.; Zeng, F.; Zhao, Y.; Zhang, J.Y.; Lu, W.L. Lipid vesicles containing transferrin receptor binding peptide TfR-T12 and octa-arginine conjugate stearyl-R8 efficiently treat brain glioma along with glioma stem cells. Sci. Rep. 2017, 7, 3487. [Google Scholar] [CrossRef]
- Kang, Z.; Zeng, C.; Tian, L.; Wang, T.; Yang, S.; Cheng, Q.; Zhang, J.; Meng, Q.; Zhang, C.; Meng, Z. Transferrin receptor targeting segment T7 containing peptide gene delivery vectors for efficient transfection of brain tumor cells. Drug Deliv. 2022, 29, 2375–2385. [Google Scholar] [CrossRef]
- Crook, Z.R.; Girard, E.; Sevilla, G.P.; Merrill, M.; Friend, D.; Rupert, P.B.; Pakiam, F.; Nguyen, E.; Yin, C.; Ruff, R.O. A TfR-Binding Cystine–Dense Peptide Promotes Blood–Brain Barrier Penetration of Bioactive Molecules. J. Mol. Biol. 2020, 432, 3989–4009. [Google Scholar] [CrossRef]
- Tamaru, M.; Akita, H.; Fujiwara, T.; Kajimoto, K.; Harashima, H. Leptin-derived peptide, a targeting ligand for mouse brain-derived endothelial cells via macropinocytosis. Biochem. Biophys. Res. Commun. 2010, 394, 587–592. [Google Scholar] [CrossRef]
- Pulous, F.E.; Petrich, B.G. Integrin-dependent regulation of the endothelial barrier. Tissue Barriers 2019, 7, 1685844. [Google Scholar] [CrossRef] [PubMed]
- Max, R.; Gerritsen, R.R.; Nooijen, P.T.; Goodman, S.L.; Sutter, A.; Keilholz, U.; Ruiter, D.J.; De Waal, R.M. Immunohistochemical analysis of integrin αvβ3 expression on tumor-associated vessels of human carcinomas. Int. J. Cancer 1997, 71, 320–324. [Google Scholar] [CrossRef]
- Brooks, P.C.; Strömblad, S.; Klemke, R.; Visscher, D.; Sarkar, F.H.; Cheresh, D.A. Antiintegrin αvβ3 blocks human breast cancer growth and angiogenesis in human skin. J. Clin. Investig. 1995, 96, 1815–1822. [Google Scholar] [CrossRef] [PubMed]
- Brooks, P.C.; Montgomery, A.M.; Rosenfeld, M.; Reisfeld, R.A.; Hu, T.; Klier, G.; Cheresh, D.A. Integrin αvβ3 antagonists promote tumor regression by inducing apoptosis of angiogenic blood vessels. Cell 1994, 79, 1157–1164. [Google Scholar] [CrossRef] [PubMed]
- Miura, Y.; Takenaka, T.; Toh, K.; Wu, S.; Nishihara, H.; Kano, M.R.; Ino, Y.; Nomoto, T.; Matsumoto, Y.; Koyama, H.; et al. Cyclic RGD-linked polymeric micelles for targeted delivery of platinum anticancer drugs to glioblastoma through the blood-brain tumor barrier. ACS Nano 2013, 7, 8583–8592. [Google Scholar] [CrossRef]
- Liu, Y.; Ran, R.; Chen, J.; Kuang, Q.; Tang, J.; Mei, L.; Zhang, Q.; Gao, H.; Zhang, Z.; He, Q. Paclitaxel loaded liposomes decorated with a multifunctional tandem peptide for glioma targeting. Biomaterials 2014, 35, 4835–4847. [Google Scholar] [CrossRef]
- Wei, X.; Gao, J.; Zhan, C.; Xie, C.; Chai, Z.; Ran, D.; Ying, M.; Zheng, P.; Lu, W. Liposome-based glioma targeted drug delivery enabled by stable peptide ligands. J. Control. Release 2015, 218, 13–21. [Google Scholar] [CrossRef]
- Gaertner, F.C.; Kessler, H.; Wester, H.-J.; Schwaiger, M.; Beer, A.J. Radiolabelled RGD peptides for imaging and therapy. Eur. J. Nucl. Med. Mol. Imaging 2012, 39 (Suppl. 1), S126–S138. [Google Scholar] [CrossRef]
- Tirand, L.; Frochot, C.; Vanderesse, R.; Thomas, N.; Trinquet, E.; Pinel, S.; Viriot, M.-L.; Guillemin, F.; Barberi-Heyob, M. A peptide competing with VEGF165 binding on neuropilin-1 mediates targeting of a chlorin-type photosensitizer and potentiates its photodynamic activity in human endothelial cells. J. Control. Release 2006, 111, 153–164. [Google Scholar] [CrossRef]
- Perret, G.Y.; Starzec, A.; Hauet, N.; Vergote, J.; Le Pecheur, M.; Vassy, R.; Léger, G.; Verbeke, K.A.; Bormans, G.; Nicolas, P. In vitro evaluation and biodistribution of a 99mTc-labeled anti-VEGF peptide targeting neuropilin-1. Nucl. Med. Biol. 2004, 31, 575–581. [Google Scholar] [CrossRef]
- Lentz, T.L.; Burrage, T.G.; Smith, A.L.; Crick, J.; Tignor, G.H. Is the acetylcholine receptor a rabies virus receptor? Science 1982, 215, 182–184. [Google Scholar] [CrossRef] [PubMed]
- Lentz, T.L. Rabies virus binding to an acetylcholine receptor α-subunit peptide. J. Mol. Recognit. 1990, 3, 82–88. [Google Scholar] [CrossRef]
- Oller-Salvia, B.; Teixido, M.; Giralt, E. From venoms to BBB shuttles: Synthesis and blood-brain barrier transport assessment of apamin and a nontoxic analog. Biopolymers 2013, 100, 675–686. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Jiang, H.; Bi, Q.; Luo, Q.; Li, J.; Zhang, Y.; Chen, Z.; Li, C. Apamin-mediated actively targeted drug delivery for treatment of spinal cord injury: More than just a concept. Mol. Pharm. 2014, 11, 3210–3222. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wan, X.; Zheng, X.; Shao, X.; Liu, Q.; Zhang, Q.; Qian, Y. Dual-functional nanoparticles targeting amyloid plaques in the brains of Alzheimer’s disease mice. Biomaterials 2014, 35, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Qian, J.; Cao, S.; Yang, Z.; Pang, Z.; Pan, S.; Fan, L.; Xi, Z.; Jiang, X.; Zhang, Q. Precise glioma targeting of and penetration by aptamer and peptide dual-functioned nanoparticles. Biomaterials 2012, 33, 5115–5123. [Google Scholar] [CrossRef]
- Zou, L.-L.; Ma, J.-L.; Wang, T.; Yang, T.-B.; Liu, C.-B. Cell-penetrating Peptide-mediated therapeutic molecule delivery into the central nervous system. Curr. Neuropharmacol. 2013, 11, 197–208. [Google Scholar] [CrossRef]
- Sharma, G.; Modgil, A.; Zhong, T.; Sun, C.; Singh, J. Influence of short-chain cell-penetrating peptides on transport of doxorubicin encapsulating receptor-targeted liposomes across brain endothelial barrier. Pharm. Res. 2014, 31, 1194–1209. [Google Scholar] [CrossRef]
- Schwarze, S.R.; Ho, A.; Vocero-Akbani, A.; Dowdy, S.F. In vivo protein transduction: Delivery of a biologically active protein into the mouse. Science 1999, 285, 1569–1572. [Google Scholar] [CrossRef]
- Rizzuti, M.; Nizzardo, M.; Zanetta, C.; Ramirez, A.; Corti, S. Therapeutic applications of the cell-penetrating HIV-1 Tat peptide. Drug Discov. Today 2015, 20, 76–85. [Google Scholar] [CrossRef]
- Kokryakov, V.N.; Harwig, S.S.; Panyutich, E.A.; Shevchenko, A.A.; Aleshina, G.M.; Shamova, O.V.; Korneva, H.A.; Lehrer, R.I. Protegrins: Leukocyte antimicrobial peptides that combine features of corticostatic defensins and tachyplesins. FEBS Lett. 1993, 327, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Rousselle, C.; Smirnova, M.; Clair, P.; Lefauconnier, J.M.; Chavanieu, A.; Calas, B.; Scherrmann, J.M.; Temsamani, J. Enhanced delivery of doxorubicin into the brain via a peptide-vector-mediated strategy: Saturation kinetics and specificity. J. Pharmacol. Exp. Ther. 2001, 296, 124–131. [Google Scholar] [PubMed]
- Drin, G.; Rousselle, C.; Scherrmann, J.-M.; Rees, A.R.; Temsamani, J. Peptide delivery to the brain via adsorptive-mediated endocytosis: Advances with SynB vectors. AAPS PharmSci 2002, 4, 26. [Google Scholar] [CrossRef] [PubMed]
- Kannan, R.; Kuhlenkamp, J.F.; Jeandidier, E.; Trinh, H.; Ookhtens, M.; Kaplowitz, N. Evidence for carrier-mediated transport of glutathione across the blood-brain barrier in the rat. J. Clin. Investig. 1990, 85, 2009–2013. [Google Scholar] [CrossRef]
- Gaillard, P.J.; Appeldoorn, C.C.; Rip, J.; Dorland, R.; van der Pol, S.M.; Kooij, G.; de Vries, H.E.; Reijerkerk, A. Enhanced brain delivery of liposomal methylprednisolone improved therapeutic efficacy in a model of neuroinflammation. J. Control. Release 2012, 164, 364–369. [Google Scholar] [CrossRef]
- Rip, J.; Chen, L.; Hartman, R.; Heuvel, A.v.D.; Reijerkerk, A.; van Kregten, J.; van der Boom, B.; Appeldoorn, C.; de Boer, M.; Maussang, D.; et al. Glutathione PEGylated liposomes: Pharmacokinetics and delivery of cargo across the blood-brain barrier in rats. J. Drug Target. 2014, 22, 460–467. [Google Scholar] [CrossRef]
- Mdzinarishvili, A.; Sutariya, V.; Talasila, P.K.; Geldenhuys, W.J.; Sadana, P. Engineering triiodothyronine (T3) nanoparticle for use in ischemic brain stroke. Drug Deliv. Transl. Res. 2013, 3, 309–317. [Google Scholar] [CrossRef]
- Georgieva, J.V.; Brinkhuis, R.P.; Stojanov, K.; Weijers, C.A.G.M.; Zuilhof, H.; Rutjes, F.P.J.T.; Hoekstra, D.; van Hest, J.C.M.; Zuhorn, I.S. Peptide-mediated blood-brain barrier transport of polymersomes. Angew. Chem. Int. Ed. 2012, 51, 8339–8342. [Google Scholar] [CrossRef]
- Barua, N.U.; Gill, S.S.; Love, S. Convection-enhanced drug delivery to the brain: Therapeutic potential and neuropathological considerations. Brain Pathol. 2014, 24, 117–127. [Google Scholar] [CrossRef]
- Van Woensel, M.; Wauthoz, N.; Rosière, R.; Amighi, K.; Mathieu, V.; Lefranc, F.; Van Gool, S.W.; De Vleeschouwer, S. Formulations for Intranasal Delivery of Pharmacological Agents to Combat Brain Disease: A New Opportunity to Tackle GBM? Cancers 2013, 5, 1020–1048. [Google Scholar] [CrossRef]
- Nuritas. Rapid and Unrivalled AI Enabled Peptide Discovery the First Artificial Intelligence Drug Discovery Company with an In-Market Commercially Validated Platform. Available online: https://www.nature.com/articles/d43747-020-00948-x (accessed on 30 June 2023).
- Batra, R.; Loeffler, T.D.; Chan, H.; Srinivasan, S.; Cui, H.; Korendovych, I.V.; Nanda, V.; Palmer, L.C.; Solomon, L.A.; Fry, H.C.; et al. Machine learning overcomes human bias in the discovery of self-assembling peptides. Nat. Chem. 2022, 14, 1427–1435. [Google Scholar] [CrossRef] [PubMed]
- Parrasia, S.; Szabò, I.; Zoratti, M.; Biasutto, L. Peptides as Pharmacological Carriers to the Brain: Promises, Shortcomings and Challenges. Mol. Pharm. 2022, 19, 3700–3729. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, J.; Sheng, Y.; Wang, W. Recent Advances of Studies on Cell-Penetrating Peptides Based on Molecular Dynamics Simulations. Cells 2022, 11, 4016. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).