Anti-Melanogenic and Antioxidant Activity of Bifidobacterium longum Strain ZJ1 Extracts, Isolated from a Chinese Centenarian
Abstract
1. Introduction
2. Results
2.1. Preparation of ZJ1 Extracts
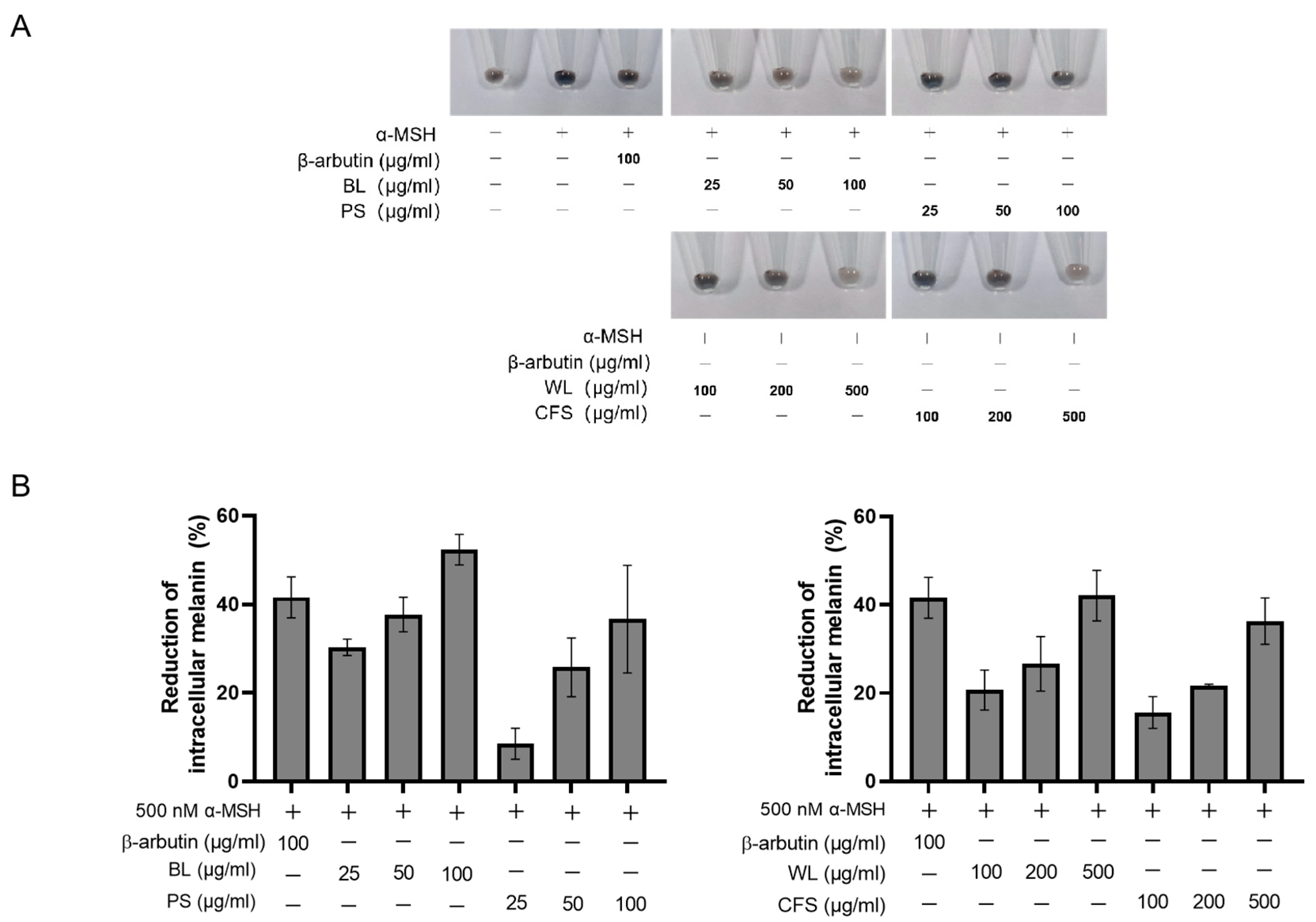
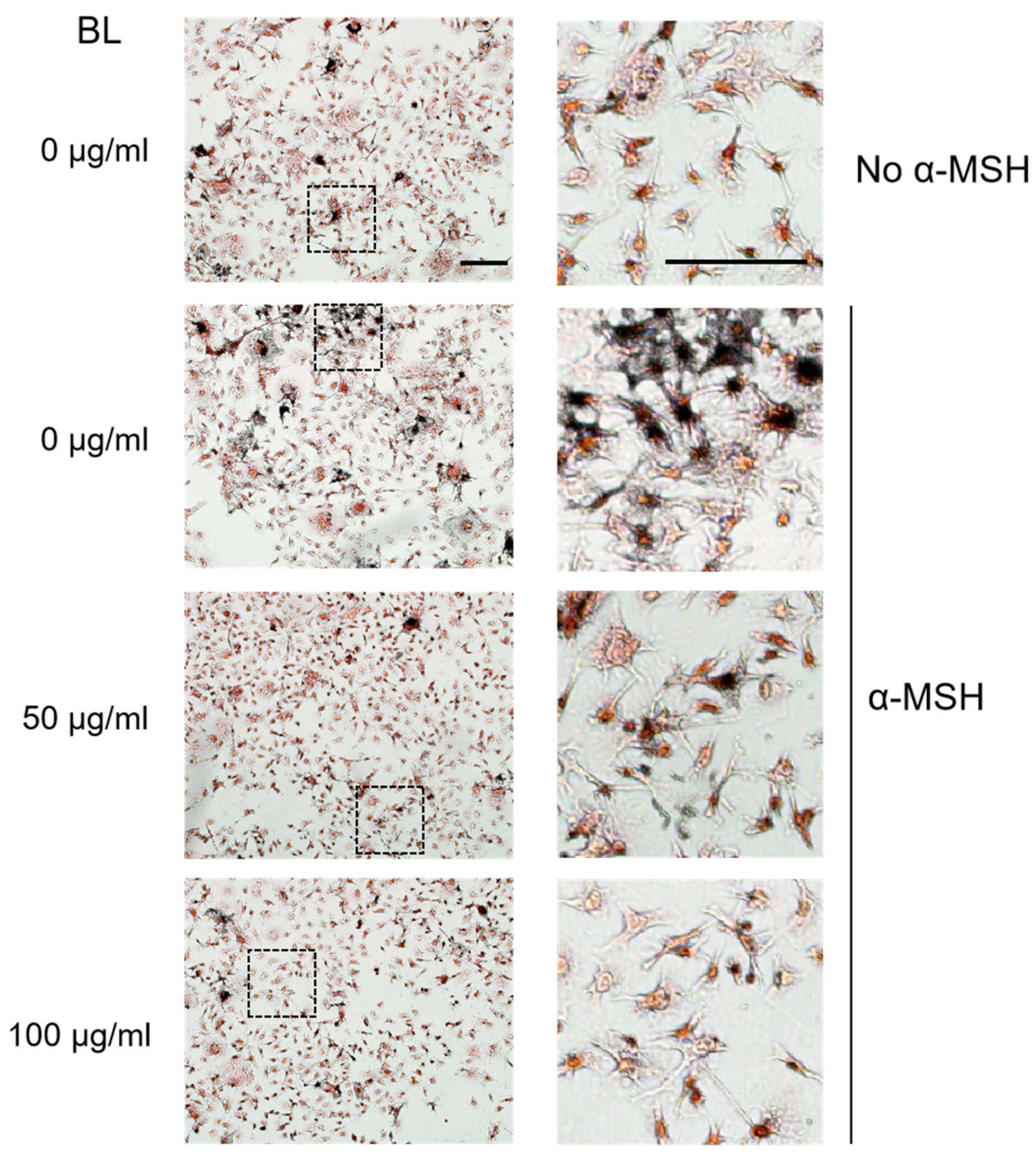
2.2. Anti-Melanogenic Effect of ZJ1 Extracts on B16-F10 Cells
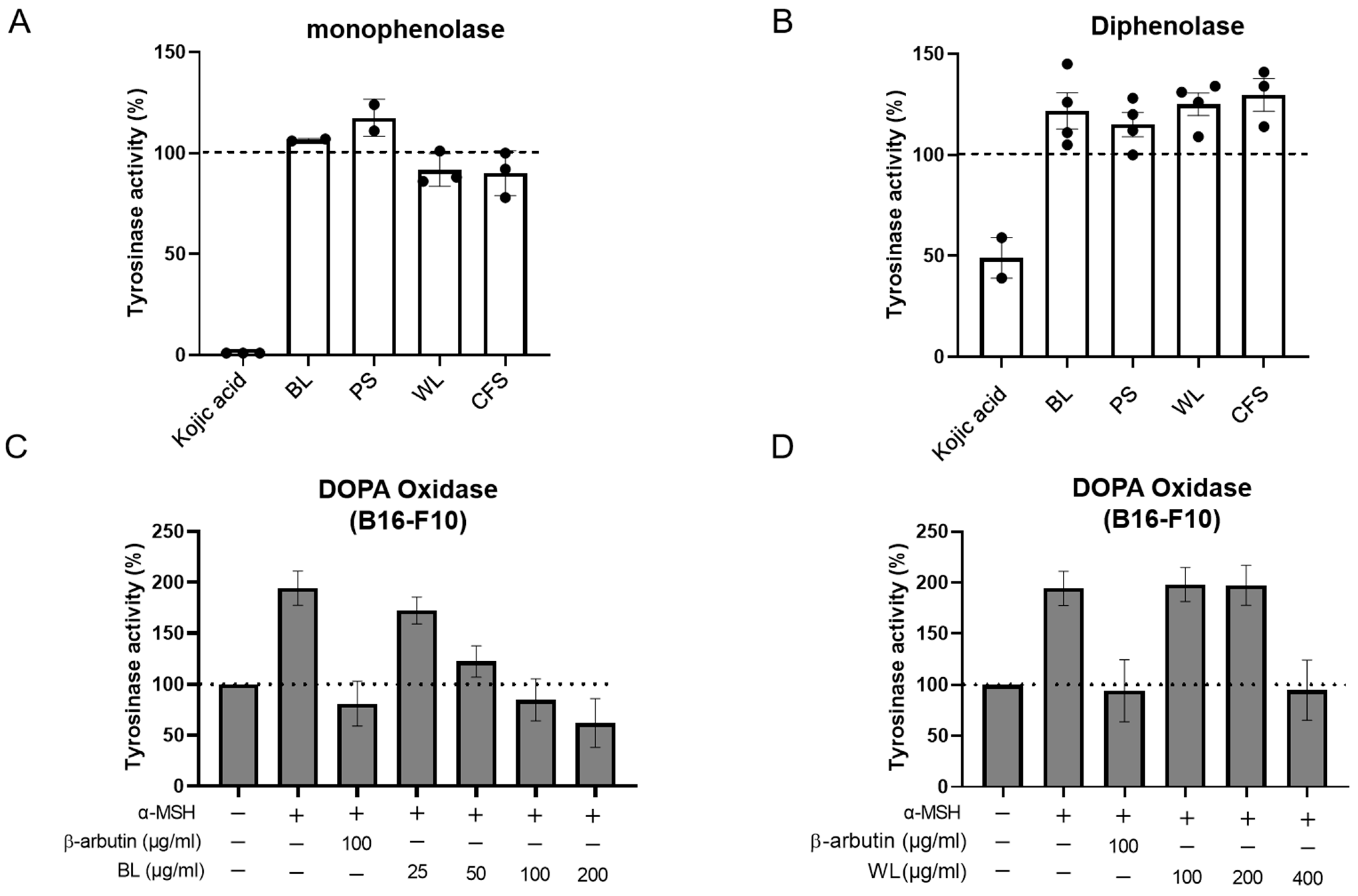
2.3. BL and WL Reduce Intracellular Tyrosinase Activity
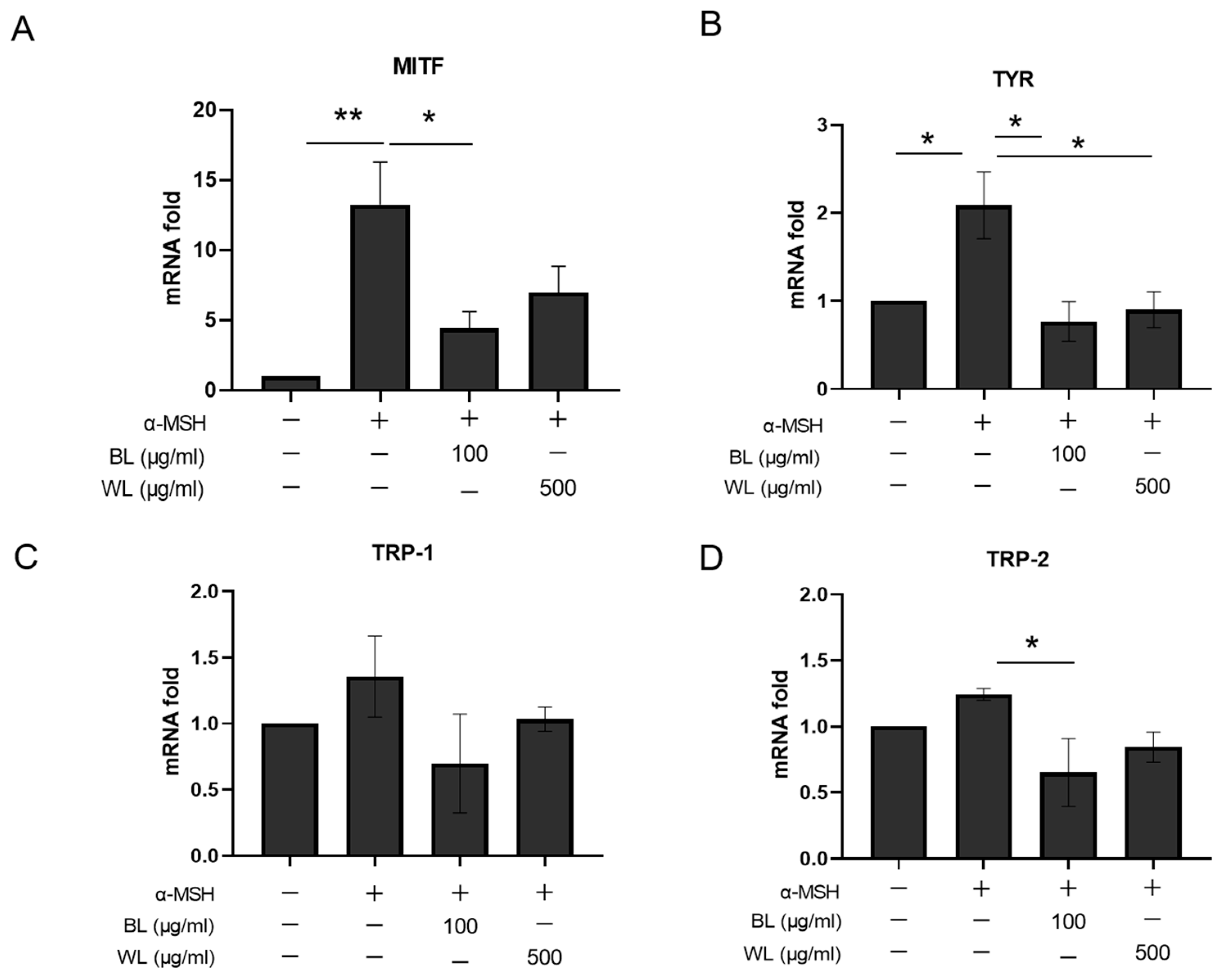
2.4. BL and WL Downregulate Melanogenesis-Related Gene Expression
2.5. BL and WL Inhibit Melanogenesis in Zebrafish Embryos
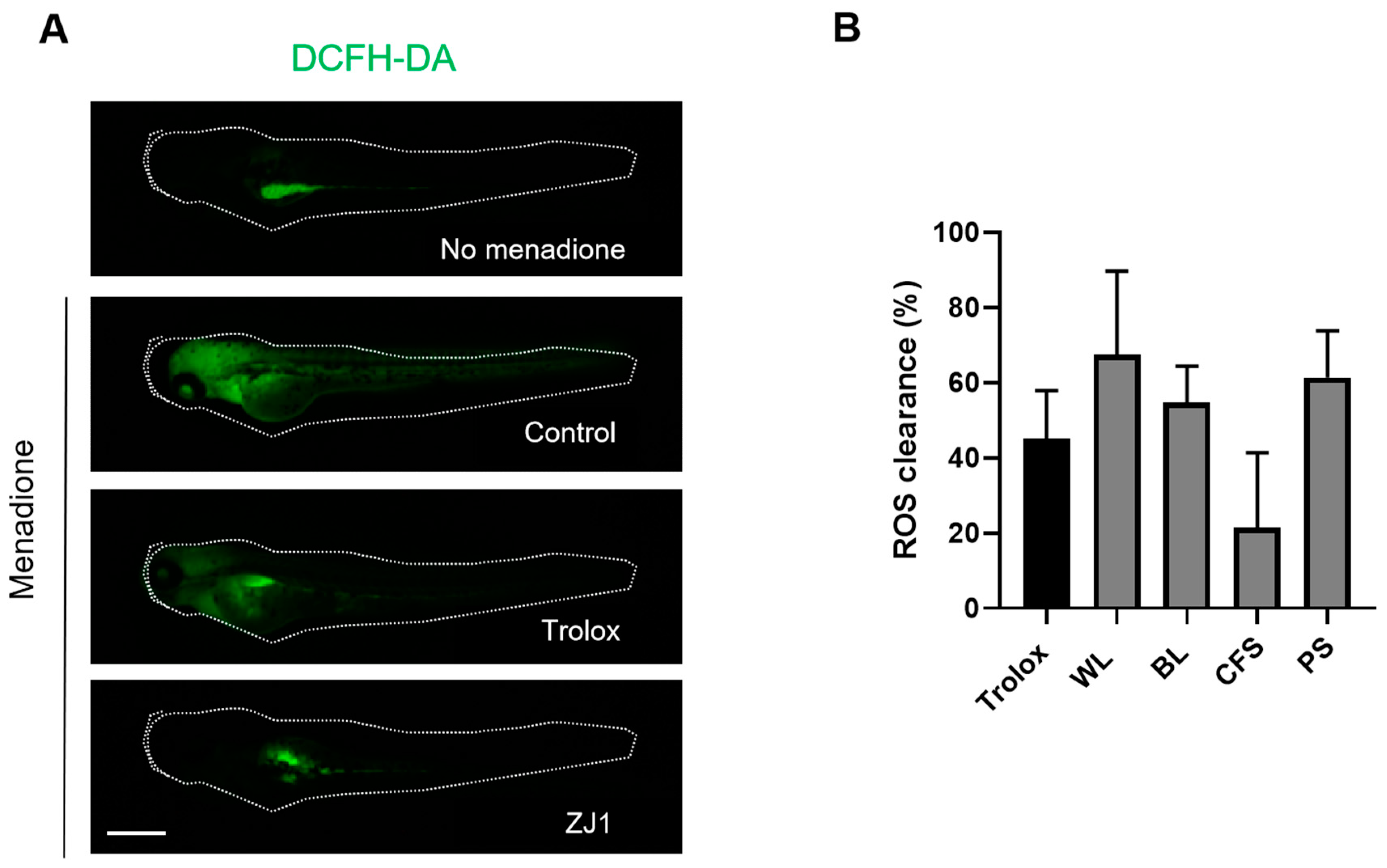
2.6. Antioxidant Property of ZJ1 Extracts
3. Discussion
4. Materials and Methods
4.1. Materials and Reagents
4.2. Preparation of Bacterial Lysates, Whole Lysates, Cell-Free Supernatant and Polysaccharides
4.3. Cell Culture
4.4. Cell Viability Assay
4.5. Assay of Melanin Contents in Melanoma Cells B16-F10
4.6. Assay of Cell-Free Mushroom Tyrosinase Activity
4.7. Assay of Intracellular Tyrosinase Activity
4.8. Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR) and Quantitative Real-Time Polymerase Chain Reaction
4.9. ABTS Assay
4.10. Zebrafish Maintenance and Embryo Collection
4.11. Assay of Melanin Content and Tyrosinase Activity in Zebrafish Embryos
4.12. Assay of Cellular Reactive Oxygen Species (ROS) in Zebrafish Embryo Model
4.13. Statistics
5. Conclusions
6. Limitations of the Study
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ohbayashi, N.; Fukuda, M. Recent advances in understanding the molecular basis of melanogenesis in melanocytes. F1000Research 2020, 9, 608. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Tobin, D.J.; Shibahara, S.; Wortsman, J. Melanin pigmentation in mammalian skin and its hormonal regulation. Physiol. Rev. 2004, 84, 1155–1228. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Wortsman, J.; Luger, T.; Paus, R.; Solomon, S. Corticotropin Releasing Hormone and Proopiomelanocortin Involvement in the Cutaneous Response to Stress. Physiol. Rev. 2000, 80, 979–1020. [Google Scholar]
- Moreiras, H.; Seabra, M.C.; Barral, D.C. Melanin Transfer in the Epidermis: The Pursuit of Skin Pigmentation Control Mechanisms. Int. J. Mol. Sci. 2021, 22, 4466. [Google Scholar] [CrossRef]
- Serre, C.; Busuttil, V.; Botto, J.-M. Intrinsic and extrinsic regulation of human skin melanogenesis and pigmentation. Int. J. Cosmet. Sci. 2018, 40, 328–347. [Google Scholar] [CrossRef]
- Slominski, A.; Moellmann, G.; Kuklinska, E. L-Tyrosine, L-DOPA, and Tyrosinase as Positive Regulators of the Subcellular Apparatus of Melanogenesis in Bomirski Ab Amelanotic Melanoma Cells. Pigment. Cell Res. 1989, 2, 109–116. [Google Scholar] [CrossRef]
- Boo, Y.C. Arbutin as a Skin Depigmenting Agent with Antimelanogenic and Antioxidant Properties. Antioxidants 2021, 10, 1129. [Google Scholar] [CrossRef]
- Kim, K.; Huh, Y.; Lim, K.-M. Anti-Pigmentary Natural Compounds and Their Mode of Action. Int. J. Mol. Sci. 2021, 22, 6206. [Google Scholar] [CrossRef]
- Leyden, J.; Shergill, B.; Micali, G.; Downie, J.; Wallo, W. Natural options for the management of hyperpigmentation. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 1140–1145. [Google Scholar] [CrossRef] [PubMed]
- Bento-Lopes, L.; Cabaço, L.C.; Charneca, J.; Neto, M.V.; Seabra, M.C.; Barral, D.C. Melanin’s Journey from Melanocytes to Keratinocytes: Uncovering the Molecular Mechanisms of Melanin Transfer and Processing. Int. J. Mol. Sci. 2023, 24, 11289. [Google Scholar] [CrossRef]
- Hakozaki, T.; Minwalla, L.; Zhuang, J.; Chhoa, M.; Matsubara, A.; Miyamoto, K.; Greatens, A.; Hillebrand, G.; Bissett, D.; Boissy, R. The effect of niacinamide on reducing cutaneous pigmentation and suppression of melanosome transfer. Br. J. Dermatol. 2002, 147, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Hwang, J.S.; Kim, H.-S. N-Nicotinoyl dopamine inhibits skin pigmentation by suppressing of melanosome transfer. Eur. J. Pharmacol. 2015, 769, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, K.; Tsukamoto, K.; Okazaki, M.; Virador, V.M.; Lei, T.-C.; Suzuki, Y.; Uchida, G.; Kitano, Y.; Harii, K. Effects of all-trans retinoic acid on melanogenesis in pigmented skin equivalents and monolayer culture of melanocytes. J. Dermatol. Sci. 2001, 27, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Gigli, V.; Piccinino, D.; Avitabile, D.; Antiochia, R.; Capecchi, E.; Saladino, R. Laccase Mediator Cocktail System as a Sustainable Skin Whitening Agent for Deep Eumelanin Decolorization. Int. J. Mol. Sci. 2022, 23, 6238. [Google Scholar] [CrossRef]
- Sung, H.J.; Khan, M.F.; Kim, Y.H. Recombinant lignin peroxidase-catalyzed decolorization of melanin using in-situ generated H2O2 for application in whitening cosmetics. Int. J. Biol. Macromol. 2019, 136, 20–26. [Google Scholar] [CrossRef]
- Boyajian, J.L.; Ghebretatios, M.; Schaly, S.; Islam, P.; Prakash, S. Microbiome and Human Aging: Probiotic and Prebiotic Potentials in Longevity, Skin Health and Cellular Senescence. Nutrients 2021, 13, 4550. [Google Scholar] [CrossRef]
- Mahmud, R.; Akter, S.; Tamanna, S.K.; Mazumder, L.; Esti, I.Z.; Banerjee, S.; Akter, S.; Hasan, R.; Acharjee, M.; Hossain, S.; et al. Impact of gut microbiome on skin health: Gut-skin axis observed through the lenses of therapeutics and skin diseases. Gut Microbes 2022, 14, 2096995. [Google Scholar] [CrossRef]
- De Pessemier, B.; Grine, L.; Debaere, M.; Maes, A.; Paetzold, B.; Callewaert, C. Gut–Skin Axis: Current Knowledge of the Interrelationship between Microbial Dysbiosis and Skin Conditions. Microorganisms 2021, 9, 353. [Google Scholar] [CrossRef] [PubMed]
- Salminen, S.; Collado, M.C.; Endo, A.; Hill, C.; Lebeer, S.; Quigley, E.M.M.; Sanders, M.E.; Shamir, R.; Swann, J.R.; Szajewska, H.; et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 649–667. [Google Scholar] [CrossRef]
- Duarte, M.; Oliveira, A.L.; Oliveira, C.; Pintado, M.; Amaro, A.; Madureira, A.R. Current postbiotics in the cosmetic market—An update and development opportunities. Appl. Microbiol. Biotechnol. 2022, 106, 5879–5891. [Google Scholar] [CrossRef]
- Piqué, N.; Berlanga, M.; Miñana-Galbis, D. Health Benefits of Heat-Killed (Tyndallized) Probiotics: An Overview. Int. J. Mol. Sci. 2019, 20, 2534. [Google Scholar] [CrossRef] [PubMed]
- Turroni, F.; Peano, C.; Pass, D.A.; Foroni, E.; Severgnini, M.; Claesson, M.J.; Kerr, C.; Hourihane, J.; Murray, D.; Fuligni, F.; et al. Diversity of Bifidobacteria within the Infant Gut Microbiota. PLoS ONE 2012, 7, e36957. [Google Scholar] [CrossRef] [PubMed]
- Mills, S.; Yang, B.; Smith, G.J.; Stanton, C.; Ross, R.P. Efficacy of Bifidobacterium longum alone or in multi-strain probiotic formulations during early life and beyond. Gut Microbes 2023, 15, 2186098. [Google Scholar] [CrossRef] [PubMed]
- Fang, Z.; Pan, T.; Li, L.; Wang, H.; Zhu, J.; Zhang, H.; Zhao, J.; Chen, W.; Lu, W. Bifidobacterium longum mediated tryptophan metabolism to improve atopic dermatitis via the gut-skin axis. Gut Microbes 2022, 14, 2044723. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Li, W.; Wang, W.; Sun, B. Draft Genome Sequence of Bifidobacterium longum ZJ1, Isolated from a Centenarian in Anhui, China. Genome Announc. 2019, 8, e00878-19. [Google Scholar] [CrossRef]
- Guéniche, A.; Bastien, P.; Ovigne, J.M.; Kermici, M.; Courchay, G.; Chevalier, V.; Breton, L.; Castiel-Higounenc, I. Bifidobacterium longum lysate, a new ingredient for reactive skin. Exp. Dermatol. 2010, 19, e1–e8. [Google Scholar] [CrossRef]
- Ferreira, A.M.; de Souza, A.A.; Koga, R.d.C.R.; Sena, I.d.S.; Matos, M.d.J.S.; Tomazi, R.; Ferreira, I.M.; Carvalho, J.C.T. Anti-Melanogenic Potential of Natural and Synthetic Substances: Application in Zebrafish Model. Molecules 2023, 28, 1053. [Google Scholar] [CrossRef]
- Neupane, R.; Boddu, S.H.; Renukuntla, J.; Babu, R.J.; Tiwari, A.K. Alternatives to Biological Skin in Permeation Studies: Current Trends and Possibilities. Pharmaceutics 2020, 12, 152. [Google Scholar] [CrossRef]
- Ma, X.; Pan, Y.; Zhao, W.; Sun, P.; Zhao, J.; Yan, S.; Wang, R.; Han, Y.; Liu, W.; Tan, S.; et al. Bifidobacterium infantis strain YLGB-1496 possesses excellent antioxidant and skin barrier-enhancing efficacy in vitro. Exp. Dermatol. 2022, 31, 1089–1094. [Google Scholar] [CrossRef]
- Huang, H.-C.; Chang, T.-M. Antioxidative properties and inhibitory effect of Bifidobacterium adolescentis on melanogenesis. World J. Microbiol. Biotechnol. 2012, 28, 2903–2912. [Google Scholar] [CrossRef]
- Huang, H.-C.; Huang, W.-Y.; Chiu, S.-H.; Ke, H.-J.; Chiu, S.-W.; Wu, S.-Y.; Kuo, F.-S.; Chang, T.-M. Antimelanogenic and antioxidative properties of Bifidobacterium bifidum. Arch. Dermatol. Res. 2011, 303, 527–531. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-C.; Lee, I.J.; Huang, C.; Chang, T.-M. Lactic Acid Bacteria and Lactic Acid for Skin Health and Melanogenesis Inhibition. Curr. Pharm. Biotechnol. 2020, 21, 566–577. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.R.; Kim, H.; Jung, B.J.; You, G.E.; Jang, S.; Chung, D.K. Lipoteichoic Acid Isolated from Lactobacillus plantarum Inhibits Melanogenesis in B16F10 Mouse Melanoma Cells. Mol. Cells 2015, 38, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.; Truong, V.-L.; Lee, M.; Kim, D.; Kim, M.S.; Cho, H.; Jung, Y.H.; Yang, J.; Jeong, W.S.; Kim, Y. Investigation of phenyllactic acid as a potent tyrosinase inhibitor produced by probiotics. Curr. Res. Food Sci. 2023, 6, 100413. [Google Scholar] [CrossRef]
- Liu, F.; Qu, L.; Li, H.; He, J.; Wang, L.; Fang, Y.; Yan, X.; Yang, Q.; Peng, B.; Wu, W.; et al. Advances in Biomedical Functions of Natural Whitening Substances in the Treatment of Skin Pigmentation Diseases. Pharmaceutics 2022, 14, 2308. [Google Scholar] [CrossRef]
- Andaloussi, S.A.; Talbaoui, H.; Marczak, R.; Bonaly, R. Isolation and characterization of exocellular polysaccharides produced by Bifidobacterium Iongum. Appl. Microbiol. Biotechnol. 1995, 43, 995–1000. [Google Scholar] [CrossRef]
- Lee, S.; Park, H.-O.; Yoo, W. Anti-Melanogenic and Antioxidant Effects of Cell-Free Supernatant from Lactobacillus gasseri BNR17. Microorganisms 2022, 10, 788. [Google Scholar] [CrossRef]
- Li, X.; Lin, J.; Gao, Y.; Han, W.; Chen, D. Antioxidant activity and mechanism of Rhizoma Cimicifugae. Chem. Central J. 2012, 6, 140. [Google Scholar] [CrossRef]

| Fraction | Bacterial Lysate (BL) (mg/g, n = 9) | Cell-Free Supernatant (CFS) (mg/mL, n = 5 or 8) | Whole Lysate (WL) (mg/mL, n = 7) |
|---|---|---|---|
| Polysaccharide | n.d | 0.16 ± 0.03 | n.d |
| Protein | 9.56 ± 1.12 | 11.82 ± 0.94 | 11.75 ± 0.93 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, J.; Zhang, F.; Yu, H.; Qi, S.; Wu, Y.; Xiao, W. Anti-Melanogenic and Antioxidant Activity of Bifidobacterium longum Strain ZJ1 Extracts, Isolated from a Chinese Centenarian. Int. J. Mol. Sci. 2023, 24, 12810. https://doi.org/10.3390/ijms241612810
Wu J, Zhang F, Yu H, Qi S, Wu Y, Xiao W. Anti-Melanogenic and Antioxidant Activity of Bifidobacterium longum Strain ZJ1 Extracts, Isolated from a Chinese Centenarian. International Journal of Molecular Sciences. 2023; 24(16):12810. https://doi.org/10.3390/ijms241612810
Chicago/Turabian StyleWu, Jing, Funa Zhang, Haixia Yu, Shimei Qi, Yu Wu, and Weihua Xiao. 2023. "Anti-Melanogenic and Antioxidant Activity of Bifidobacterium longum Strain ZJ1 Extracts, Isolated from a Chinese Centenarian" International Journal of Molecular Sciences 24, no. 16: 12810. https://doi.org/10.3390/ijms241612810
APA StyleWu, J., Zhang, F., Yu, H., Qi, S., Wu, Y., & Xiao, W. (2023). Anti-Melanogenic and Antioxidant Activity of Bifidobacterium longum Strain ZJ1 Extracts, Isolated from a Chinese Centenarian. International Journal of Molecular Sciences, 24(16), 12810. https://doi.org/10.3390/ijms241612810






