Influence of Electrospun Fibre Secondary Morphology on Antibiotic Release Kinetic and Its Impact on Antimicrobic Efficacy
Abstract
1. Introduction
2. Results
2.1. Polymer Solutions Characterization
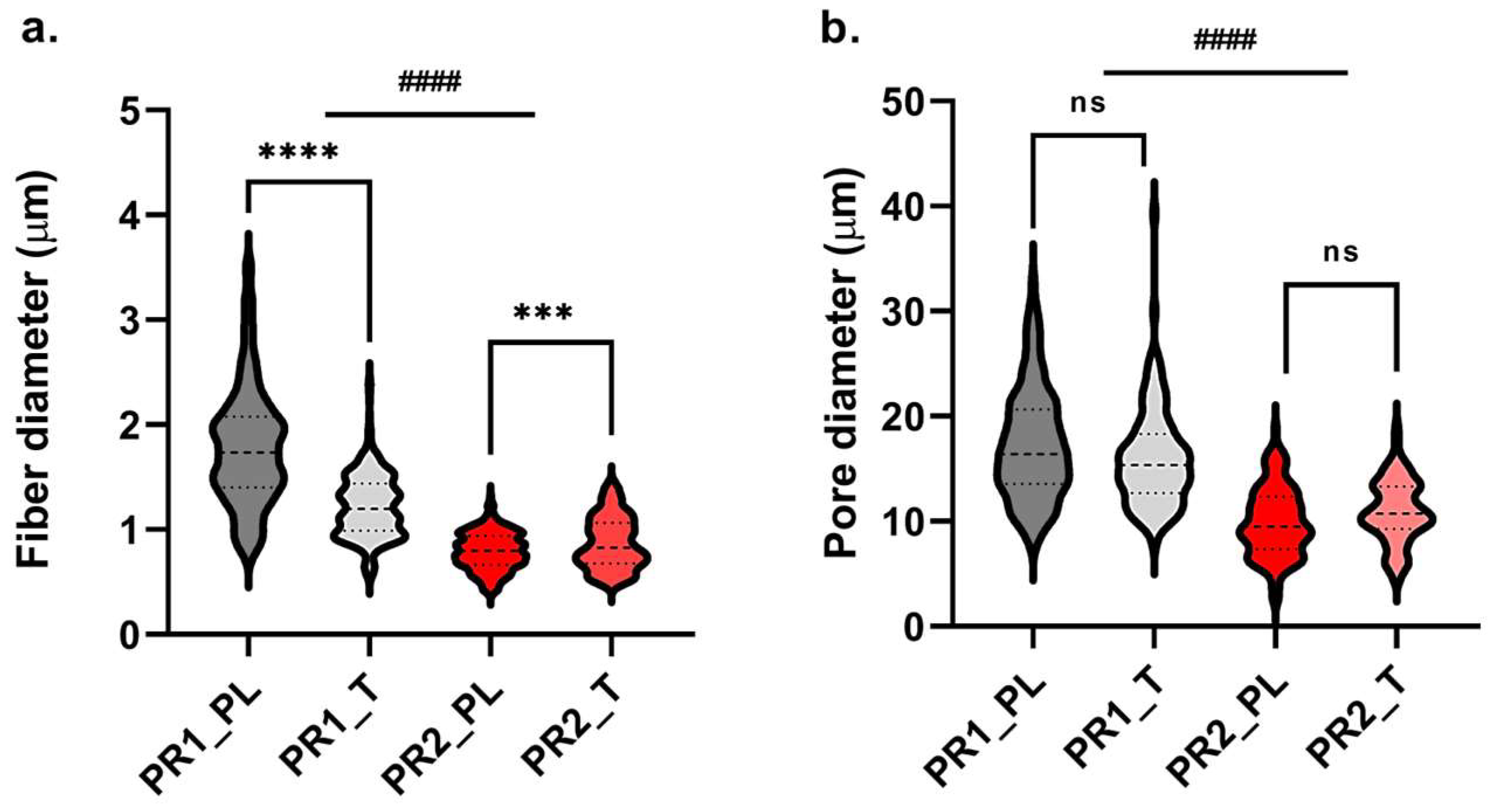
2.2. Morphological Characterization of Small-Diameter Vascular Grafts Prototypes
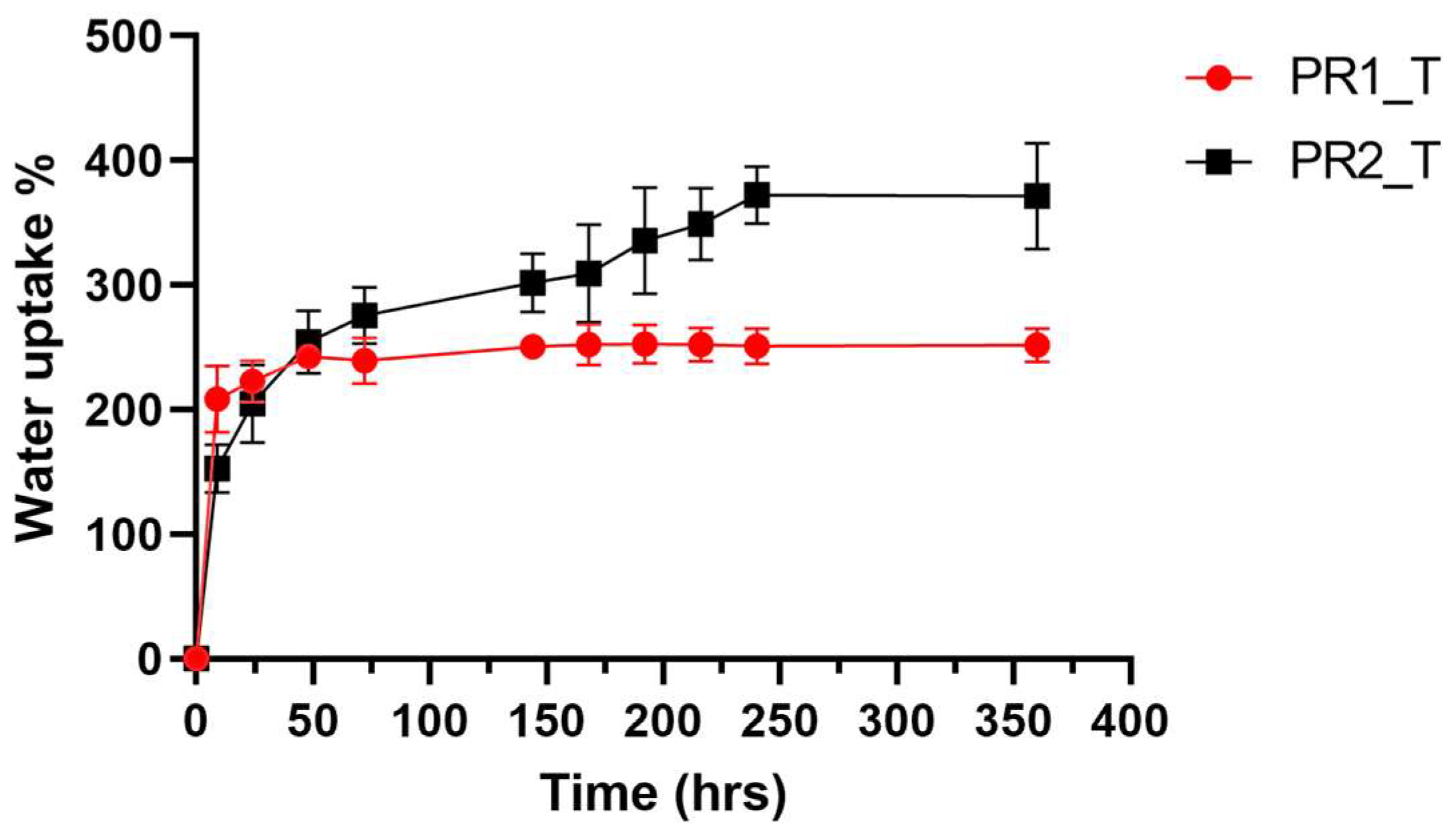
2.3. Small-Diameter Vascular Graft Prototypes Hydration Profiles
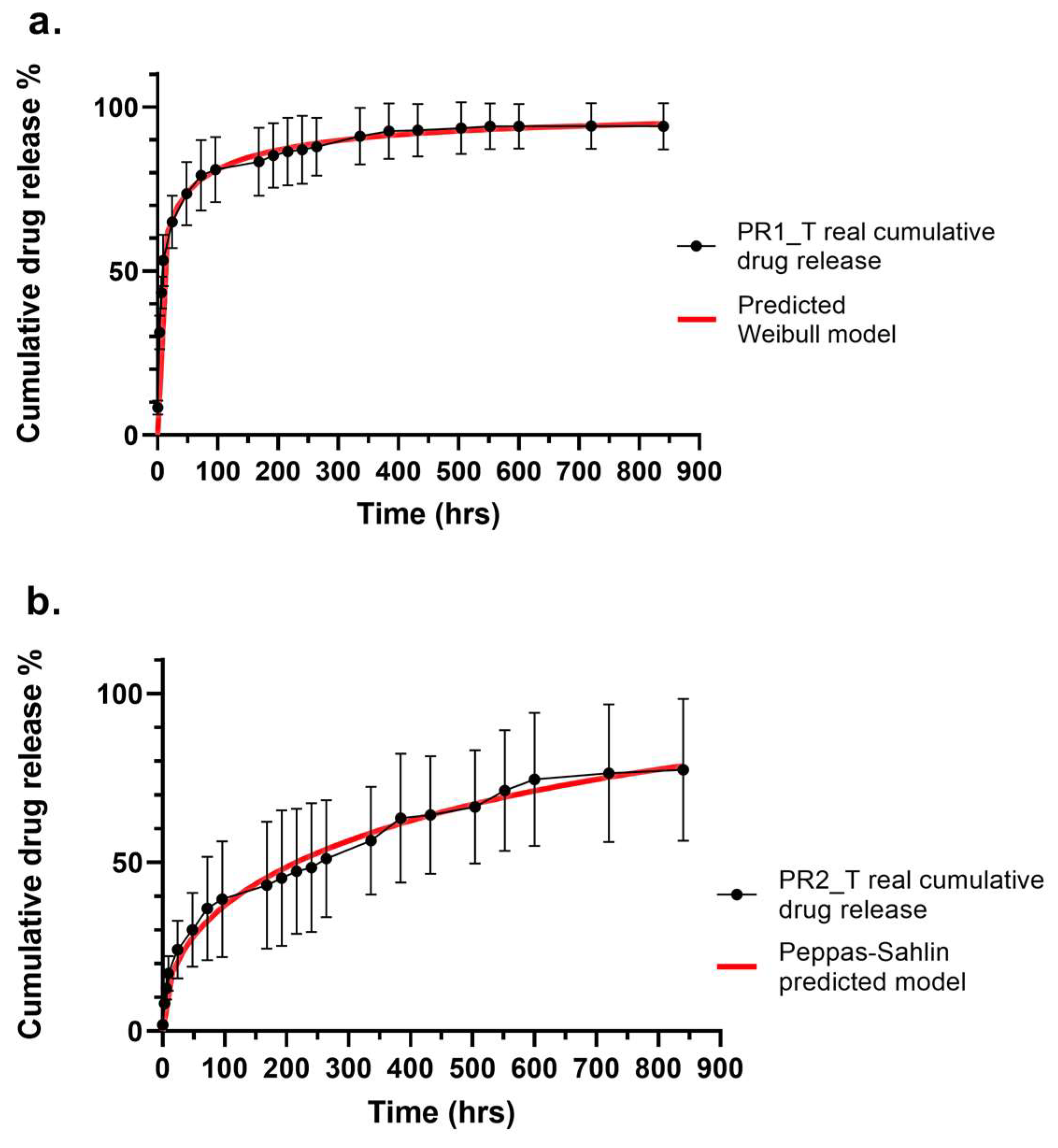
2.4. In Vitro Drug Release Profiles and Kinetics of Small-Diameter Vascular Graft Prototypes
2.5. Drug Release Kinetic Evaluation for Vasculas Graft Prototypes
2.6. Antimicrobic Activity of Small-Diameter Vascular Graft Prototypes
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Methods
4.2.1. Polymer Solutions Preparation and Characterization
4.2.2. Vascular Graft Prototypes Electrospinning and Morphological Characterization
4.2.3. Water up Take Assay
4.2.4. Drug Encapsulation Efficiency, Vascular Grafts Drug Content and In Vitro Drug Release Evaluation
4.2.5. Drug Release Kinetic Study
4.2.6. Antimicrobic Assay
4.2.7. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Matsuzaki, Y.; John, K.; Shoji, T.; Shinoka, T. The Evolution of Tissue Engineered Vascular Graft Technologies: From Preclinical Trials to Advancing Patient Care. Appl. Sci. 2019, 9, 1274. [Google Scholar] [CrossRef] [PubMed]
- He, E.; Serpelloni, S.; Alvear, P.; Rahimi, M.; Taraballi, F. Vascular Graft Infections: An Overview of Novel Treatments Using Nanoparticles and Nanofibers. Fibers 2022, 10, 12. [Google Scholar] [CrossRef]
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P.; et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990–2019: Update From the GBD 2019 Study. J. Am. Coll. Cardiol. 2020, 76, 2982–3021. [Google Scholar] [CrossRef] [PubMed]
- Costa, D.; Andreucci, M.; Ielapi, N.; Serraino, G.F.; Mastroroberto, P.; Bracale, U.M.; Serra, R. Infection of Vascular Prostheses: A Comprehensive Review. Prosthesis 2023, 5, 148–166. [Google Scholar] [CrossRef]
- Bouchet, M.; Gauthier, M.; Maire, M.; Ajji, A.; Lerouge, S. Towards compliant small-diameter vascular grafts: Predictive analytical model and experiments. Mater. Sci. Eng. C 2019, 100, 715–723. [Google Scholar] [CrossRef]
- Chandorkar, Y.; Ravikumar, K.; Basu, B. The Foreign Body Response Demystified. ACS Biomater. Sci. Eng. 2019, 5, 19–44. [Google Scholar] [CrossRef]
- Obiweluozor, F.O.; Emechebe, G.A.; Kim, D.W.; Cho, H.J.; Park, C.H.; Kim, C.S.; Jeong, I.S. Considerations in the Development of Small-Diameter Vascular Graft as an Alternative for Bypass and Reconstructive Surgeries: A Review. Cardiovasc. Eng. Technol. 2020, 11, 495–521. [Google Scholar] [CrossRef]
- Warner, M.; Nelson, R.; Lim, C. Vascular graft infection. In Oxford Textbook of Vascular Surgery; Thompson, M.M., Fitridge, R., Boyle, J., Thompson, M., Brohi, K., Hinchliffe, R.J., Cheshire, N., Naylor, A.R., Loftus, I., Davies, A.H., Eds.; Oxford University Press: Oxford, UK, 2016; ISBN 9780199658220. [Google Scholar]
- Wouthuyzen-Bakker, M.; van Oosten, M.; Bierman, W.; Winter, R.; Glaudemans, A.; Slart, R.; Toren-Wielema, M.; Tielliu, I.; Zeebregts, C.J.; Prakken, N.H.J.; et al. Diagnosis and treatment of vascular graft and endograft infections: A structured clinical approach. Int. J. Infect. Dis. 2023, 126, 22–27. [Google Scholar] [CrossRef]
- Chakfé, N.; Diener, H.; Lejay, A.; Assadian, O.; Berard, X.; Caillon, J.; Fourneau, I.; Glaudemans, A.W.J.M.; Koncar, I.; Lindholt, J.; et al. European Society for Vascular Surgery (ESVS) 2020 Clinical Practice Guidelines on the Management of Vascular Graft and Endograft Infections. Eur. J. Vasc. Endovasc. Surg. 2020, 59, 339–384. [Google Scholar] [CrossRef]
- Berard, X.; Puges, M.; Pinaquy, J.-B.; Cazanave, C.; Stecken, L.; Bordenave, L.; Pereyre, S.; M’Zali, F. Evidence of Improved Antimicrobial Efficacy of Silver and Triclosan Containing Vascular Grafts Compared with Rifampicin Soaked Grafts. Eur. J. Vasc. Endovasc. Surg. 2019, 57, 424–432. [Google Scholar] [CrossRef] [PubMed]
- Molacek, J.; Treska, V.; Houdek, K.; Opatrný, V.; Certik, B.; Baxa, J. Use of a Silver-Impregnated Vascular Graft: Single-Center Experience. Antibiotics 2022, 11, 386. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.; Mandal, B.B. Tissue-Engineered Vascular Grafts: Emerging Trends and Technologies. Adv. Funct. Mater. 2021, 31, 2100027. [Google Scholar] [CrossRef]
- Rosalia, M.; Chiesa, E.; Tottoli, E.M.; Dorati, R.; Genta, I.; Conti, B.; Pisani, S. Tobramycin Nanoantibiotics and Their Advantages: A Minireview. Int. J. Mol. Sci. 2022, 23, 14080. [Google Scholar] [CrossRef] [PubMed]
- Rosalia, M.; Ravipati, P.; Grisoli, P.; Dorati, R.; Genta, I.; Chiesa, E.; Bruni, G.; Conti, B. Tobramycin Supplemented Small-Diameter Vascular Grafts for Local Antibiotic Delivery: A Preliminary Formulation Study. Int. J. Mol. Sci. 2021, 22, 13557. [Google Scholar] [CrossRef] [PubMed]
- Leal, B.B.J.; Wakabayashi, N.; Oyama, K.; Kamiya, H.; Braghirolli, D.I.; Pranke, P. Vascular Tissue Engineering: Polymers and Methodologies for Small Caliber Vascular Grafts. Front. Cardiovasc. Med. 2021, 7, 592361. [Google Scholar] [CrossRef]
- Li, M.X.; Wei, Q.Q.; Mo, H.L.; Ren, Y.; Zhang, W.; Lu, H.J.; Joung, Y.K. Challenges and advances in materials and fabrication technologies of small-diameter vascular grafts. Biomater. Res. 2023, 27, 58. [Google Scholar] [CrossRef]
- Darie-Niță, R.N.; Râpă, M.; Frąckowiak, S. Special Features of Polyester-Based Materials for Medical Applications. Polymers 2022, 14, 951. [Google Scholar] [CrossRef]
- Hiob, M.A.; She, S.; Muiznieks, L.D.; Weiss, A.S. Biomaterials and Modifications in the Development of Small-Diameter Vascular Grafts. ACS Biomater. Sci. Eng. 2017, 3, 712–723. [Google Scholar] [CrossRef]
- Rocha, C.V.; Gonçalves, V.; da Silva, M.C.; Bañobre-López, M.; Gallo, J. PLGA-Based Composites for Various Biomedical Applications. Int. J. Mol. Sci. 2022, 23, 2034. [Google Scholar] [CrossRef]
- Miller, D.C.; Thapa, A.; Haberstroh, K.M.; Webster, T.J. Endothelial and vascular smooth muscle cell function on poly(lactic-co-glycolic acid) with nano-structured surface features. Biomaterials 2004, 25, 53–61. [Google Scholar] [CrossRef]
- Chung, J.H.; Lim, H.R.; Lee, T.H.; Lee, M.H.; Woo, Y.I.; Baek, H.S.; Lee, S.J.; Kim, J.K.; Park, J.C. Growth of Smooth Muscle Cell and Endothelial Cell on PLGA Film Containing EGCG. Key Eng. Mater. 2007, 342–343, 93–96. [Google Scholar] [CrossRef]
- Chew, C.H.; Sheu, B.-L.; Chen, A.; Huang, W.-T.; Cheng, T.-M.; Shih, C.-M.; Chang, A.; Chen, C.-C. Tissue-Engineered Vascular Graft with Co-Culture of Smooth Muscle Cells and Human Endothelial Vein Cells on an Electrospun Poly(lactic-co-glycolic acid) Microtube Array Membrane. Membranes 2021, 11, 732. [Google Scholar] [CrossRef]
- In Jeong, S.; Kim, S.Y.; Cho, S.K.; Chong, M.S.; Kim, K.S.; Kim, H.; Lee, S.B.; Lee, Y.M. Tissue-engineered vascular grafts composed of marine collagen and PLGA fibers using pulsatile perfusion bioreactors. Biomaterials 2007, 28, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Lazarovici, P.; Pomerantz, C.; Chen, X.; Wei, Y.; Lelkes, P.I. Co-Electrospun Blends of PLGA, Gelatin, and Elastin as Potential Nonthrombogenic Scaffolds for Vascular Tissue Engineering. Biomacromolecules 2011, 12, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, H.M.; Klingner, A. A review on electrospun polymeric nanofibers: Production parameters and potential applications. Polym. Test. 2020, 90, 106647. [Google Scholar] [CrossRef]
- Cao, H.; Mchugh, K.; Chew, S.Y.; Anderson, J.M. The topographical effect of electrospun nanofibrous scaffolds on the in vivo and in vitro foreign body reaction. J. Biomed. Mater. Res. Part A 2010, 93A, 1151–1159. [Google Scholar] [CrossRef]
- Barsch, F.; Mamilos, A.; Schmitt, V.H.; Babel, M.; Winter, L.; Wagner, W.; Winther, H.; Ottomann, C.; Niedermair, T.; Schreml, S.; et al. In Vivo Comparison of Synthetic Macroporous Filamentous and Sponge-like Skin Substitute Matrices Reveals Morphometric Features of the Foreign Body Reaction According to 3D Biomaterial Designs. Cells 2022, 11, 2834. [Google Scholar] [CrossRef]
- Li, Z.; Lei, I.M.; Davoodi, P.; Huleihel, L.; Huang, Y.Y.S. Solution Formulation and Rheology for Fabricating Extracellular Matrix-Derived Fibers Using Low-Voltage Electrospinning Patterning. ACS Biomater. Sci. Eng. 2019, 5, 3676–3684. [Google Scholar] [CrossRef]
- Namsheer, K.; Rout, C.S. Conducting polymers: A comprehensive review on recent advances in synthesis, properties and applications. RSC Adv. 2021, 11, 5659–5697. [Google Scholar] [CrossRef]
- Sarojini, K.G.K.; Manoj, S.V.; Singh, P.K.; Pradeep, T.; Das, S.K. Electrical conductivity of ceramic and metallic nanofluids. Colloids Surf. A Physicochem. Eng. Asp. 2013, 417, 39–46. [Google Scholar] [CrossRef]
- Haider, A.; Haider, S.; Kang, I.-K. A comprehensive review summarizing the effect of electrospinning parameters and potential applications of nanofibers in biomedical and biotechnology. Arab. J. Chem. 2018, 11, 1165–1188. [Google Scholar] [CrossRef]
- Zaarour, B.; Zhu, L.; Jin, X. A Review on the Secondary Surface Morphology of Electrospun Nanofibers: Formation Mechanisms, Characterizations, and Applications. ChemistrySelect 2020, 5, 1335–1348. [Google Scholar] [CrossRef]
- Kossyvaki, D.; Barbetta, A.; Contardi, M.; Bustreo, M.; Dziza, K.; Lauciello, S.; Athanassiou, A.; Fragouli, D. Highly Porous Curcumin-Loaded Polymer Mats for Rapid Detection of Volatile Amines. ACS Appl. Polym. Mater. 2022, 4, 4464–4475. [Google Scholar] [CrossRef]
- Zaarour, B.; Zhu, L.; Huang, C.; Jin, X. Fabrication of a polyvinylidene fluoride cactus-like nanofiber through one-step electrospinning. RSC Adv. 2018, 8, 42353–42360. [Google Scholar] [CrossRef]
- Cavanaugh, J.E.; Neath, A.A. The Akaike information criterion: Background, derivation, properties, application, interpretation, and refinements. WIREs Comput. Stat. 2019, 11, e1460. [Google Scholar] [CrossRef]
- Zhang, Y.; Huo, M.; Zhou, J.; Zou, A.; Li, W.; Yao, C.; Xie, S. DDSolver: An add-in program for modeling and comparison of drug dissolution profiles. AAPS J. 2010, 12, 263–271. [Google Scholar] [CrossRef]
- Heredia, N.S.; Vizuete, K.; Flores-Calero, M.; Katherine Pazmiño, V.; Pilaquinga, F.; Kumar, B.; Debut, A. Comparative statistical analysis of the release kinetics models for nanoprecipitated drug delivery systems based on poly(lactic-co-glycolic acid). PLoS ONE 2022, 17, e0264825. [Google Scholar] [CrossRef]
- Bruschi, M.L. Mathematical models of drug release. In Strategies to Modify the Drug Release from Pharmaceutical Systems; Woodhead Publishing: Sawston, UK, 2015; pp. 63–86. ISBN 9780081000922. [Google Scholar]
- Witzler, M.; Vermeeren, S.; Kolevatov, R.O.; Haddad, R.; Gericke, M.; Heinze, T.; Schulze, M. Evaluating Release Kinetics from Alginate Beads Coated with Polyelectrolyte Layers for Sustained Drug Delivery. ACS Appl. Bio Mater. 2021, 4, 6719–6731. [Google Scholar] [CrossRef] [PubMed]
- Reyhanoglu, G.; Reddivari, A. Tobramycin; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Shaw, M.T. On estimating the zero-shear-rate viscosity: Tests with PIB and PDMS. In Proceedings of the AIP Conference Proceedings, Graz, Austria, 21–25 September 2015; AIP Publishing LLC: Melville, NY, USA, 2016; Volume 1779, p. 70011. [Google Scholar]
- Müller, S.J.; Mirzahossein, E.; Iftekhar, E.N.; Bächer, C.; Schrüfer, S.; Schubert, D.W.; Fabry, B.; Gekle, S. Flow and hydrodynamic shear stress inside a printing needle during biofabrication. PLoS ONE 2020, 15, e0236371. [Google Scholar] [CrossRef]
- Higashi, S.; Hirai, T.; Matsubara, M.; Yoshida, H.; Beniya, A. Dynamic viscosity recovery of electrospinning solution for stabilizing elongated ultrafine polymer nanofiber by TEMPO-CNF. Sci. Rep. 2020, 10, 13427. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, T.; Rasband, W. ImageJ User Guide—IJ 1.46r. 2012. Available online: https://imagej.nih.gov/ij/docs/guide/146.html (accessed on 15 May 2023).
- Omar, M.A.; Hammad, M.A.; Nagy, D.M.; Aly, A.A. Development of spectrofluorimetric method for determination of certain aminoglycoside drugs in dosage forms and human plasma through condensation with ninhydrin and phenyl acetaldehyde. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 136, 1760–1766. [Google Scholar] [CrossRef]
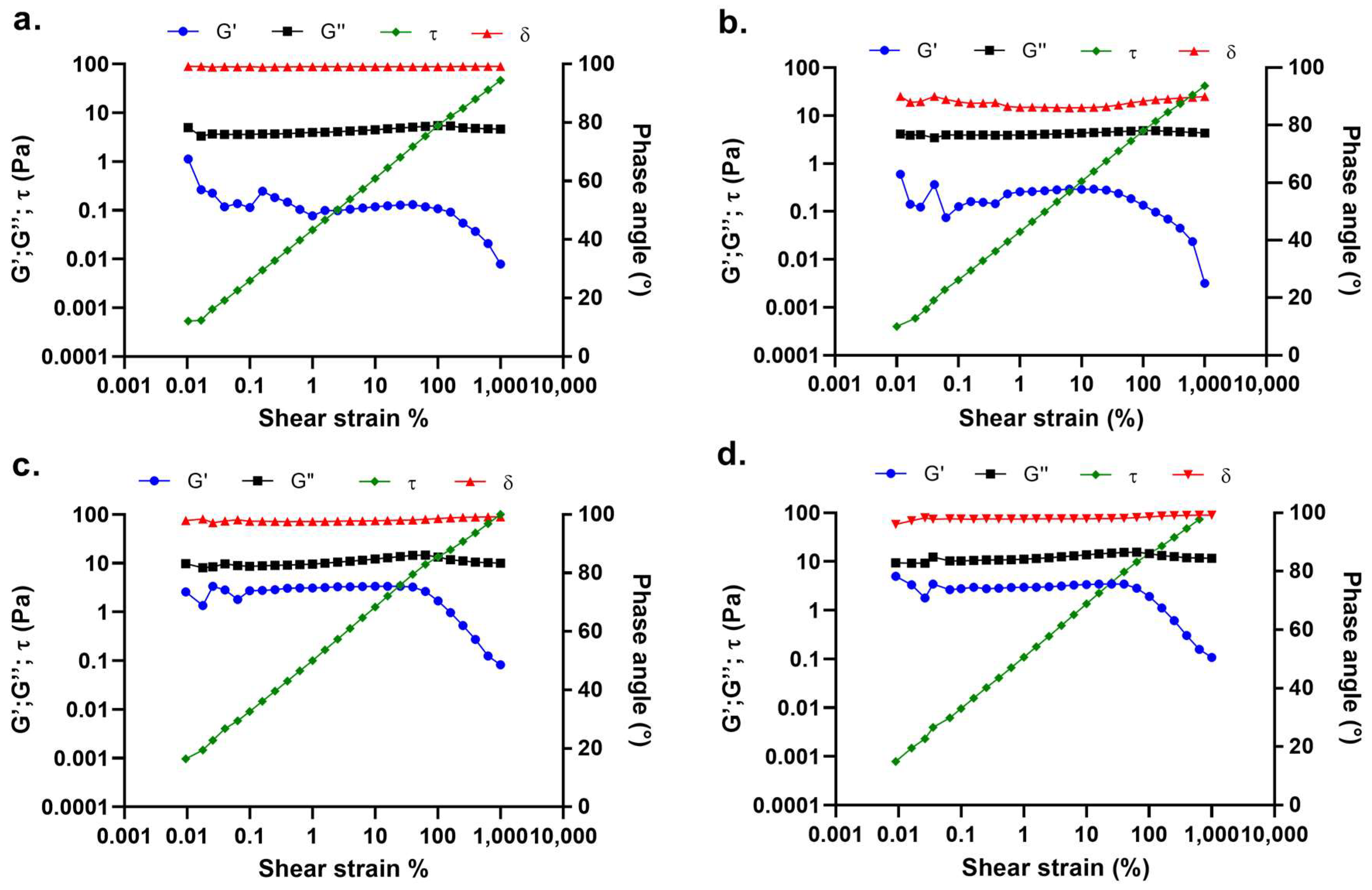
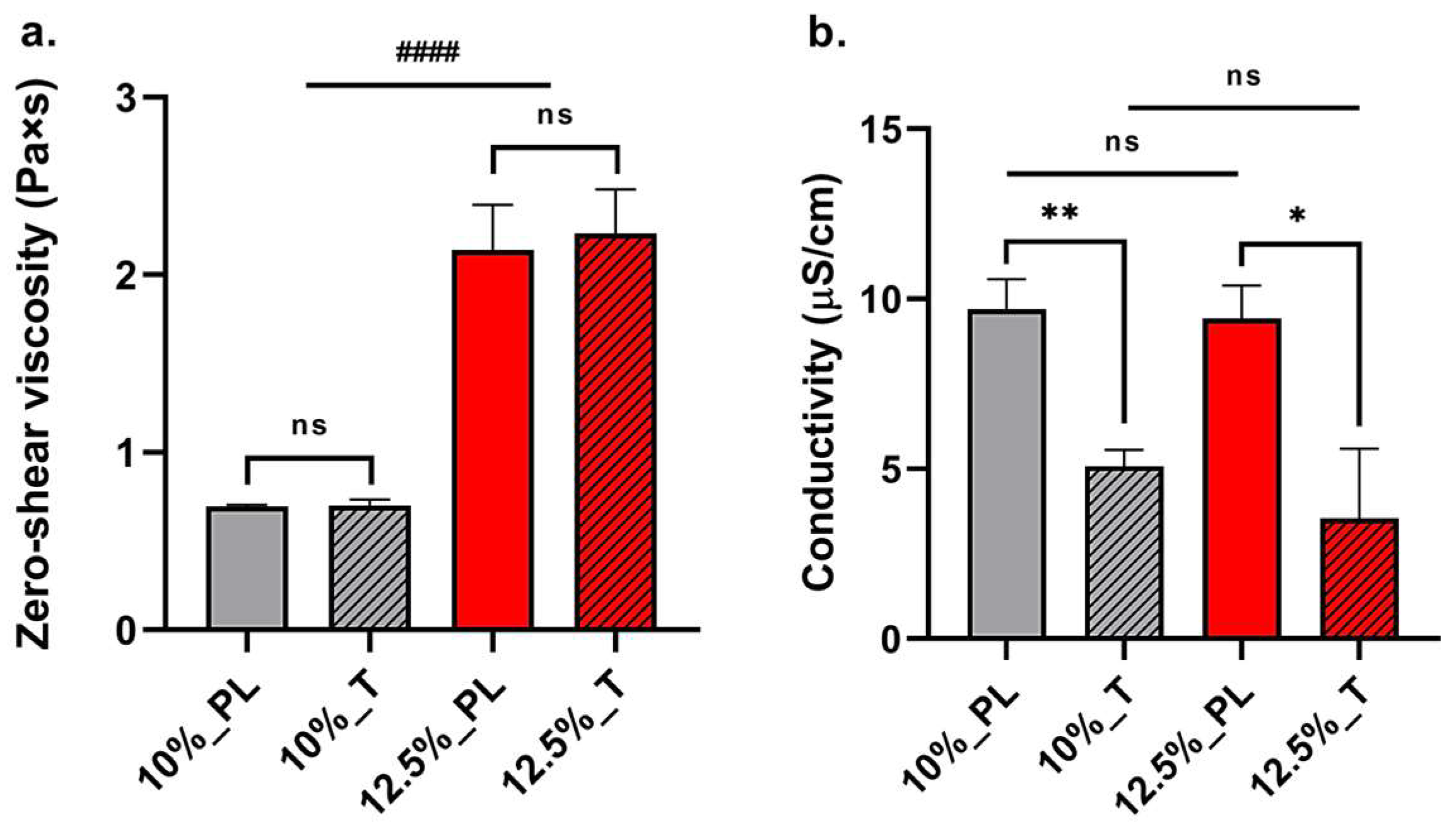


| PLGA Concentration (% w/v) | Voltage (kV) | Flow Rate (mL/h) | Temperature Range (°C) | Relative Humidity (%) |
|---|---|---|---|---|
| 10 | 18 | 0.1 | 30–35 | 20 |
| 12.5 | 20 | 0.3 | 45–37 | 20 |
| Graft Type | Wall Thickness (μm) | Fibre Diameter (μm) | Pore Diameter (μm) | Porosity (%) |
|---|---|---|---|---|
| PR1_PL | 291.95 ± 32.64 | 1.784 ± 0.561 | 17.20 ± 5.24 | 43.79 ± 0.90 |
| PR1_T | 291.00 ± 20.82 | 1.224 ± 0.301 | 16.00 ± 4.691 | 45.82 ± 0.44 |
| PR2_PL | 180.00 ± 18.00 | 0.794 ± 0.178 | 9.91 ± 3.27 | 44.00 ± 2.64 |
| PR2_T | 167.10 ± 13.08 | 0.870 ± 0.248 | 10.96 ± 3.00 | 44.49 ± 3.77 |
| Mathematical Model | Prototype 1 | Prototype 2 | ||||||
|---|---|---|---|---|---|---|---|---|
| R2 | R2-adj | AIC | MSC | R2 | R2-adj | AIC | MSC | |
| Higuchi | −0.2953 | −0.2953 | 203 | −0.3540 | 0.9386 | 0.9386 | 138 | 2.6959 |
| Korsmeyer–Peppas | 0.9332 | 0.9297 | 143 | 2.5158 | 0.9886 | 0.9874 | 107 | 4.1912 |
| Peppas–Sahlin | 0.8304 | 0.8116 | 164 | 1.4887 | 0.9909 | 0.9905 | 100 | 4.5131 |
| Weibull | 0.9978 | 0.9891 | 108 | 4.1387 | 0.9643 | 0.9580 | 133 | 2.9507 |
| Logistic | 0.9887 | 0.9874 | 109 | 4.1947 | 0.9276 | 0.9195 | 146 | 2.3396 |
| Gompertz | 0.9834 | 0.9816 | 116 | 3.8131 | 0.8914 | 0.8794 | 154 | 1.9348 |
| Probit | 0.9899 | 0.9888 | 105 | 4.4096 | 0.9356 | 0.9285 | 143 | 2.4576 |
| Prototype | Best Fitting Model | Equation | Parameters |
|---|---|---|---|
| PR1 | Weibull | F = Fmax × (1 − e[−((t − Ti)^β)/α]) | α = 2.557 ± 1.250 β = 0.295 ± 0.102 Ti = 1.500 ± 0.329 |
| PR2 | Peppas–Sahlin | F = k1 × tm + k2 × t(2m) | k1 = 5.233 ± 2.966 k2 = −0.069 ± 0.149 m = 0.45 |
| Time (h) | ME | ME |
|---|---|---|
| S. aureus | E. coli | |
| 24 | 3.98 ± 1.47 | 5.89 ± 1.16 |
| 48 | 6.31 ± 0.41 | 8.82 ± 0.07 |
| 72 | 6.30 ± 0.15 | 8.42 ± 0.40 |
| 96 | 7.55 ± 1.50 | 8.15 ± 0.53 |
| 120 | 8.58 ± 0.05 | 8.39 ± 0.31 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosalia, M.; Grisoli, P.; Dorati, R.; Chiesa, E.; Pisani, S.; Bruni, G.; Genta, I.; Conti, B. Influence of Electrospun Fibre Secondary Morphology on Antibiotic Release Kinetic and Its Impact on Antimicrobic Efficacy. Int. J. Mol. Sci. 2023, 24, 12108. https://doi.org/10.3390/ijms241512108
Rosalia M, Grisoli P, Dorati R, Chiesa E, Pisani S, Bruni G, Genta I, Conti B. Influence of Electrospun Fibre Secondary Morphology on Antibiotic Release Kinetic and Its Impact on Antimicrobic Efficacy. International Journal of Molecular Sciences. 2023; 24(15):12108. https://doi.org/10.3390/ijms241512108
Chicago/Turabian StyleRosalia, Mariella, Pietro Grisoli, Rossella Dorati, Enrica Chiesa, Silvia Pisani, Giovanna Bruni, Ida Genta, and Bice Conti. 2023. "Influence of Electrospun Fibre Secondary Morphology on Antibiotic Release Kinetic and Its Impact on Antimicrobic Efficacy" International Journal of Molecular Sciences 24, no. 15: 12108. https://doi.org/10.3390/ijms241512108
APA StyleRosalia, M., Grisoli, P., Dorati, R., Chiesa, E., Pisani, S., Bruni, G., Genta, I., & Conti, B. (2023). Influence of Electrospun Fibre Secondary Morphology on Antibiotic Release Kinetic and Its Impact on Antimicrobic Efficacy. International Journal of Molecular Sciences, 24(15), 12108. https://doi.org/10.3390/ijms241512108













