Enhanced Levels of Glycosphingolipid GM3 Delay the Progression of Diabetic Nephropathy
Abstract
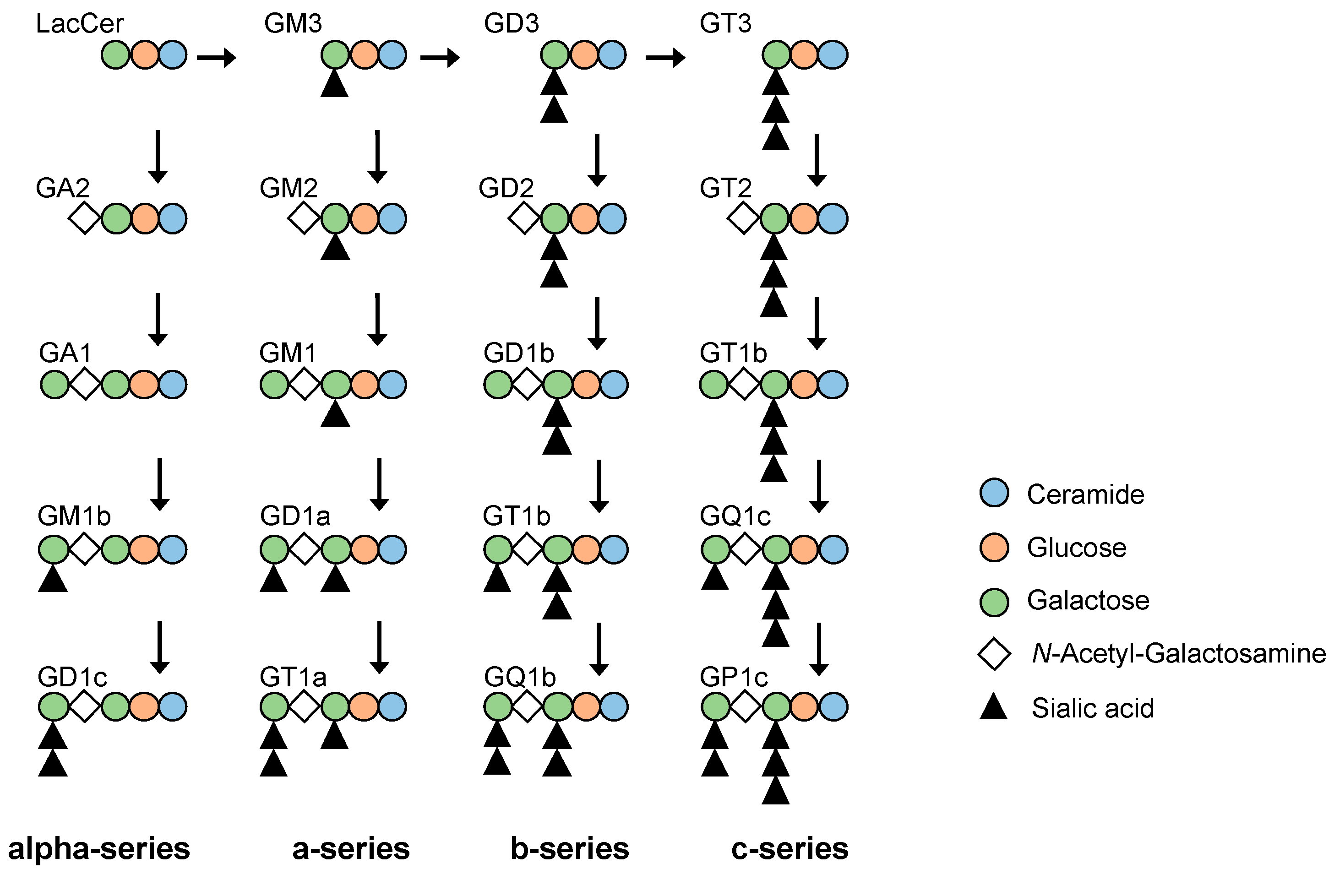
1. Introduction
2. Results
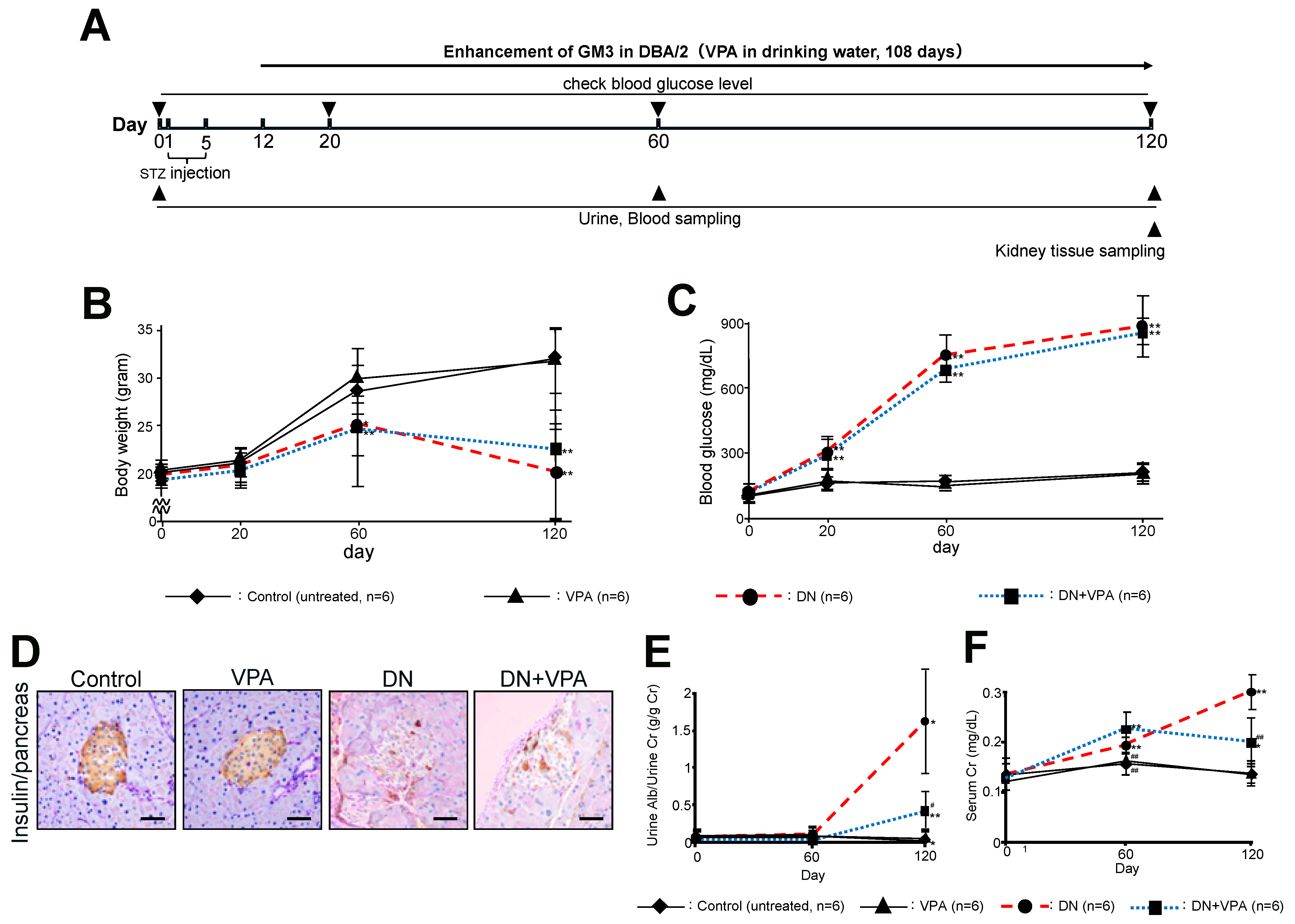
2.1. Characterization of the STZ-Induced Diabetic Model in Mice
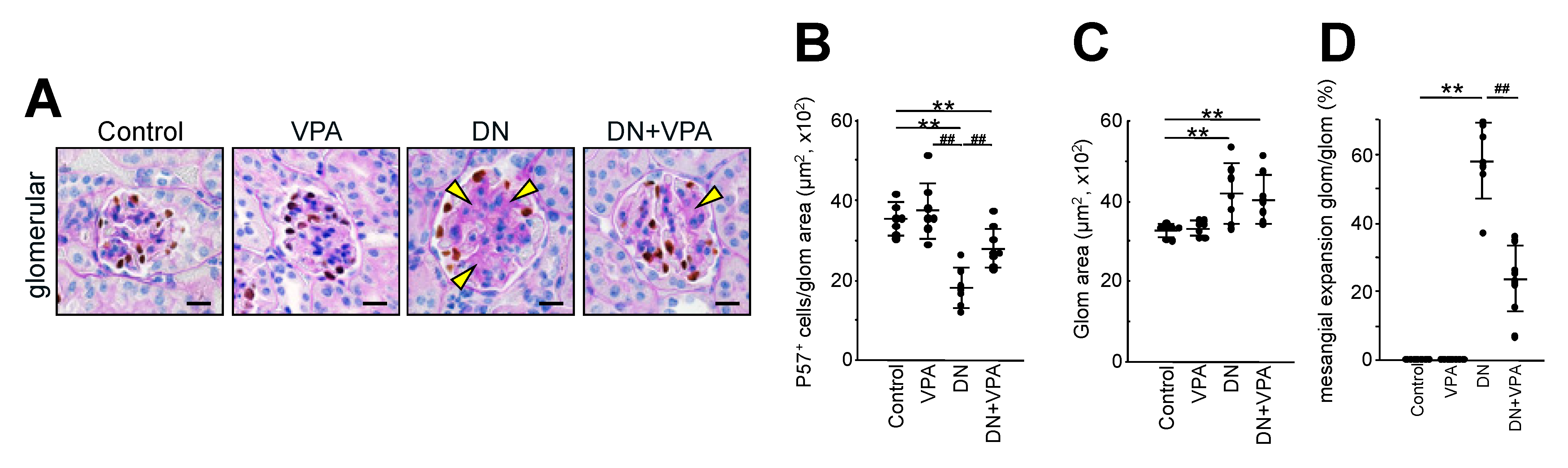
2.2. Analysis of Glomerular Morphological Changes and Podocyte Injury
2.3. The Expression of Platelet-Derived Growth Factor Receptor-β (PDGFR-B) in Glomeruli
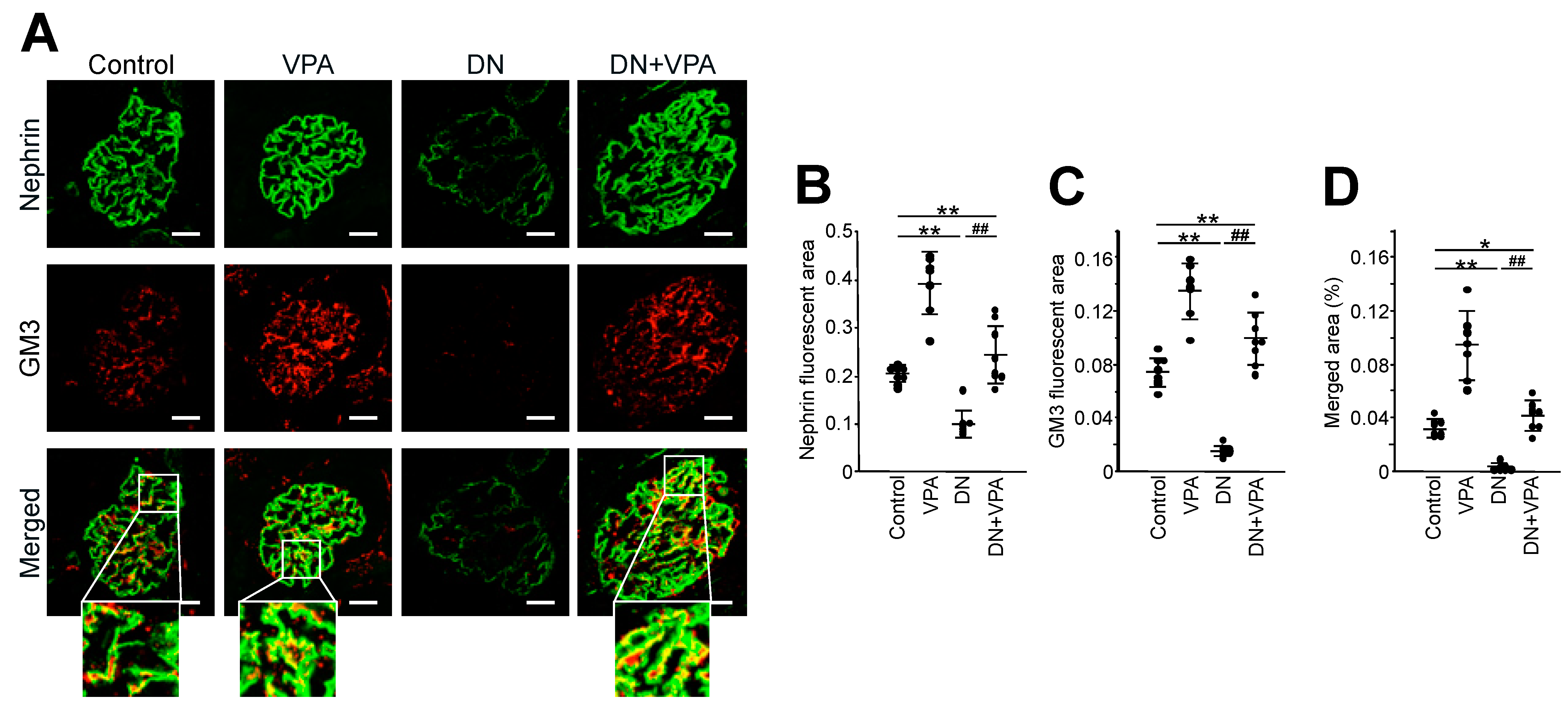
2.4. Analysis of GM3 and Nephrin Expression in Kidney Glomeruli
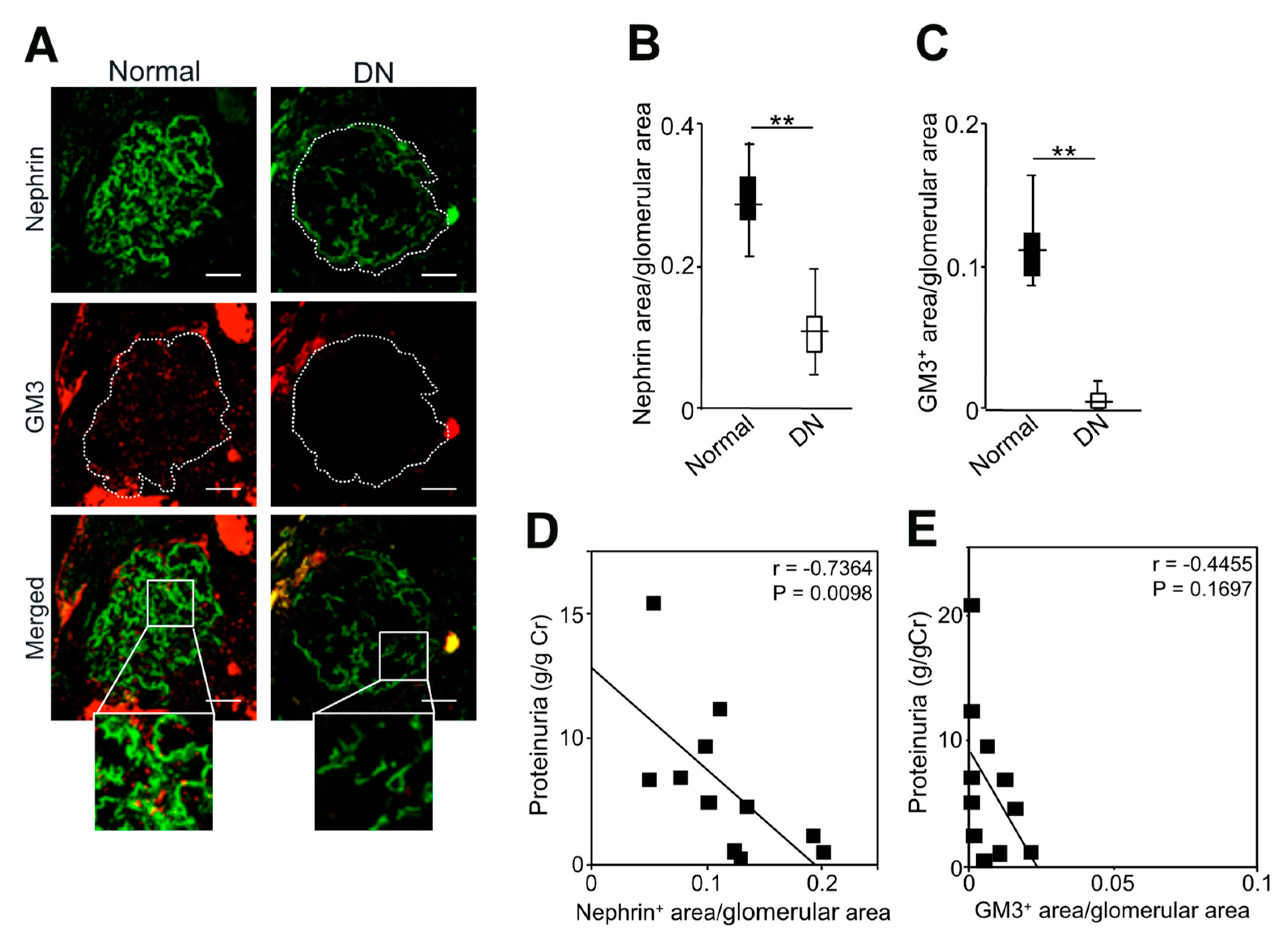
2.5. Expression of Nephrin and GM3 and Their Correlation with Albuminuria in Patients with Diabetic Nephropathy
3. Discussion
4. Materials and Methods
4.1. Reagents and Antibodies
4.2. Animal Study
4.3. Biochemical Studies
4.4. Tissue Staining
4.5. Human Study
4.6. Statistical Analysis
4.7. Study Approval
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schnaar, R.L.; Gerardy-Schahn, R.; Hildebrandt, H. Sialic acids in the brain: Gangliosides and polysialic acid in nervous system development, stability, disease, and regeneration. Physiol. Rev. 2014, 94, 461–518. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Wada, R.; Sasaki, T.; Deng, C.; Bierfreund, U.; Sandhoff, K.; Proia, R.L. A vital role for glycosphingolipid synthesis during development and differentiation. Proc. Natl. Acad. Sci. USA 1999, 96, 9142–9147. [Google Scholar] [CrossRef] [PubMed]
- Russo, D.; Della Ragione, F.; Rizzo, R.; Sugiyama, E.; Scalabri, F.; Hori, K.; Capasso, S.; Sticco, L.; Fioriniello, S.; De Gregorio, R.; et al. Glycosphingolipid metabolic reprogramming drives neural differentiation. EMBO J. 2018, 37, e97674. [Google Scholar] [CrossRef] [PubMed]
- Pukel, C.S.; Lloyd, K.O.; Travassos, L.R.; Dippold, W.G.; Oettgen, H.F.; Old, L.J. GD3, a prominent ganglioside of human melanoma. Detection and characterisation by mouse monoclonal antibody. J. Exp. Med. 1982, 155, 1133–1147. [Google Scholar] [CrossRef]
- Watanabe, T.; Pukel, C.S.; Takeyama, H.; Lloyd, K.O.; Shiku, H.; Li, L.T.; Travassos, L.R.; Oettgen, H.F.; Old, L.J. Human melanoma antigen AH is an autoantigenic ganglioside related to GD2. J. Exp. Med. 1982, 156, 1884–1889. [Google Scholar] [CrossRef]
- Hakomori, S. Cancer-associated glycosphingolipid antigens: Their structure, organization, and function. Acta Anat. 1998, 161, 79–90. [Google Scholar] [CrossRef]
- Yoon, S.J.; Nakayama, K.; Hikita, T.; Handa, K.; Hakomori, S.I. Epidermal growth factor receptor tyrosine kinase is modulated by GM3 interaction with N-linked GlcNAc termini of the receptor. Proc. Natl. Acad. Sci. USA 2006, 103, 18987–18991. [Google Scholar] [CrossRef]
- Kabayama, K.; Sato, T.; Saito, K.; Loberto, N.; Prinetti, A.; Sonnino, S.; Kinjo, M.; Igarashi, Y.; Inokuchi, J. Dissociation of the insulin receptor and caveolin-1 complex by ganglioside GM3 in the state of insulin resistance. Proc. Natl. Acad. Sci. USA 2007, 104, 13678–13683. [Google Scholar] [CrossRef]
- Kawashima, N.; Yoon, S.J.; Itoh, K.; Nakayama, K. Tyrosine kinase activity of epidermal growth factor receptor is regulated by GM3 binding through carbohydrate to carbohydrate interactions. J. Biol. Chem. 2009, 284, 6147–6155. [Google Scholar] [CrossRef]
- Kawashima, N.; Nishimiya, Y.; Takahata, S.; Nakayama, K.I. Induction of Glycosphingolipid GM3 Expression by Valproic Acid Suppresses Cancer Cell Growth. J. Biol. Chem. 2016, 291, 21424–21433. [Google Scholar] [CrossRef]
- Kwon, H.Y.; Kang, N.Y.; Dae, H.M.; Kim, K.S.; Kim, C.H.; Do, S.I.; Lee, Y. Valproic acid-mediated transcriptional regulation of human GM3 synthase (hST3Gal V) in SK-N-BE(2)-C human neuroblastoma cells. Acta Pharmacol. Sin. 2008, 29, 999–1005. [Google Scholar] [CrossRef] [PubMed]
- Song, N.; Kim, S.J.; Kwon, H.Y.; Son, S.W.; Kim, K.S.; Ahn, H.B.; Lee, Y.C. Transcriptional activation of human GM3 synthase (hST3Gal V) gene by valproic acid in ARPE-19 human retinal pigment epithelial cells. BMB Rep. 2011, 44, 405–409. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, T.; Tsubakihara, Y.; Fushimi, H.; Yamaguchi, S.; Takabatake, Y.; Rakugi, H.; Kawakami, H.; Isaka, Y. Histochemical and immunoelectron microscopic analysis of ganglioside GM3 in human kidney. Clin. Exp. Nephrol. 2015, 19, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Sison, K.; Li, C.; Tian, R.; Wnuk, M.; Sung, H.K.; Jeansson, M.; Zhang, C.; Tucholska, M.; Jones, N.; et al. Soluble FLT1 binds lipid microdomains in podocytes to control cell morphology and glomerular barrier function. Cell 2012, 151, 384–399. [Google Scholar] [CrossRef]
- Reiser, J.; Sever, S. Podocyte biology and pathogenesis of kidney disease. Annu. Rev. Med. 2013, 64, 357–366. [Google Scholar] [CrossRef]
- Yoon, S.J.; Nakayama, K.; Takahashi, N.; Yagi, H.; Utkina, N.; Wang, H.Y.; Kato, K.; Sadilek, M.; Hakomori, S.I. Interaction of N-linked glycans, having multivalent GlcNAc termini, with GM3 ganglioside. Glycoconj. J. 2006, 23, 639–649. [Google Scholar] [CrossRef]
- Naito, S.; Kawashima, N.; Ishii, D.; Fujita, T.; Iwamura, M.; Takeuchi, Y. Decreased GM3 correlates with proteinuria in minimal change nephrotic syndrome and focal segmental glomerulosclerosis. Clin. Exp. Nephrol. 2022, 26, 1078–1085. [Google Scholar] [CrossRef]
- Kawashima, N.; Naito, S.; Hanamatsu, H.; Nagane, M.; Takeuchi, Y.; Furukawa, J.I.; Iwasaki, N.; Yamashita, T.; Nakayama, K.I. Glycosphingolipid GM3 prevents albuminuria and podocytopathy induced by anti-nephrin antibody. Sci. Rep. 2022, 12, 16058. [Google Scholar] [CrossRef]
- Lin, Y.C.; Chang, Y.H.; Yang, S.Y.; Wu, K.D.; Chu, T.S. Update of pathophysiology and management of diabetic kidney disease. J. Formos. Med. Assoc. 2018, 117, 662–675. [Google Scholar] [CrossRef]
- Kopp, J.B.; Anders, H.J.; Susztak, K.; Podesta, M.A.; Remuzzi, G.; Hildebrandt, F.; Romagnani, P. Podocytopathies. Nat. Rev. Dis. Primers 2020, 6, 68. [Google Scholar] [CrossRef]
- Suzuki, H.; Usui, I.; Kato, I.; Oya, T.; Kanatani, Y.; Yamazaki, Y.; Fujisaka, S.; Senda, S.; Ishii, Y.; Urakaze, M.; et al. Deletion of platelet-derived growth factor receptor-beta improves diabetic nephropathy in Ca(2)(+)/calmodulin-dependent protein kinase IIalpha (Thr286Asp) transgenic mice. Diabetologia 2011, 54, 2953–2962. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Sudhini, Y.R.; Polat, O.K.; Reiser, J.; Altintas, M.M. Renal cell markers: Lighthouses for managing renal diseases. Am. J. Physiol. Renal. Physiol. 2021, 321, F715–F739. [Google Scholar] [CrossRef] [PubMed]
- Selby, N.M.; Taal, M.W. An updated overview of diabetic nephropathy: Diagnosis, prognosis, treatment goals and latest guidelines. Diabetes Obes. Metab. 2020, 22 (Suppl. 1), 3–15. [Google Scholar] [CrossRef]
- Pelle, M.C.; Provenzano, M.; Busutti, M.; Porcu, C.V.; Zaffina, I.; Stanga, L.; Arturi, F. Up-Date on Diabetic Nephropathy. Life 2022, 12, 1202. [Google Scholar] [CrossRef]
- Zador, I.Z.; Deshmukh, G.D.; Kunkel, R.; Johnson, K.; Radin, N.S.; Shayman, J.A. A role for glycosphingolipid accumulation in the renal hypertrophy of streptozotocin-induced diabetes mellitus. J. Clin. Investig. 1993, 91, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Kwak, D.H.; Rho, Y.I.; Kwon, O.D.; Ahan, S.H.; Song, J.H.; Choo, Y.K.; Kim, S.J.; Choi, B.K.; Jung, K.Y. Decreases of ganglioside GM3 in streptozotocin-induced diabetic glomeruli of rats. Life Sci. 2003, 72, 1997–2006. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Li, T.; Li, Z.; Wang, H.; Liu, X. Differential lipidomics of HK-2 cells and exosomes under high glucose stimulation. Int. J. Med. Sci. 2022, 19, 393–401. [Google Scholar] [CrossRef]
- Balzer, M.S.; Rohacs, T.; Susztak, K. How Many Cell Types Are in the Kidney and What Do They Do? Annu. Rev. Physiol. 2022, 84, 507–531. [Google Scholar] [CrossRef]
- Jennemann, R.; Kaden, S.; Volz, M.; Nordstrom, V.; Herzer, S.; Sandhoff, R.; Grone, H.J. Gangliosides modulate insulin secretion by pancreatic beta cells under glucose stress. Glycobiology 2020, 30, 722–734. [Google Scholar] [CrossRef]
- Mohandes, S.; Doke, T.; Hu, H.; Mukhi, D.; Dhillon, P.; Susztak, K. Molecular pathways that drive diabetic kidney disease. J. Clin. Investig. 2023, 133, 165654. [Google Scholar] [CrossRef]
- Thomas, H.Y.; Ford Versypt, A.N. Pathophysiology of mesangial expansion in diabetic nephropathy: Mesangial structure, glomerular biomechanics, and biochemical signaling and regulation. J. Biol. Eng. 2022, 16, 19. [Google Scholar] [CrossRef] [PubMed]
- Kwak, D.H.; Lee, S.; Kim, S.J.; Ahn, S.H.; Song, J.H.; Choo, Y.K.; Choi, B.K.; Jung, K.Y. Ganglioside GM3 inhibits the high glucose- and TGF-beta1-induced proliferation of rat glomerular mesangial cells. Life Sci. 2005, 77, 2540–2551. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Luo, M.; Bai, X.; Nie, P.; Zhu, Y.; Cai, H.; Li, B.; Luo, P. Cellular crosstalk of glomerular endothelial cells and podocytes in diabetic kidney disease. J. Cell Commun. Signal. 2022, 16, 313–331. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Chen, P.; Zambrano, S.; Dabaghie, D.; Hu, Y.; Moller-Hackbarth, K.; Unnersjo-Jess, D.; Korkut, G.G.; Charrin, E.; Jeansson, M.; et al. Single-cell RNA sequencing reveals the mesangial identity and species diversity of glomerular cell transcriptomes. Nat. Commun. 2021, 12, 2141. [Google Scholar] [CrossRef]
- Thomas, M.C. Targeting the Pathobiology of Diabetic Kidney Disease. Adv. Chronic. Kidney Dis. 2021, 28, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.C.; Wu, H.; Kirita, Y.; Uchimura, K.; Ledru, N.; Rennke, H.G.; Welling, P.A.; Waikar, S.S.; Humphreys, B.D. The single-cell transcriptomic landscape of early human diabetic nephropathy. Proc. Natl. Acad. Sci. USA 2019, 116, 19619–19625. [Google Scholar] [CrossRef]
- Stefansson, V.T.N.; Nair, V.; Melsom, T.; Looker, H.C.; Mariani, L.H.; Fermin, D.; Eichinger, F.; Menon, R.; Subramanian, L.; Ladd, P.; et al. Molecular programs associated with glomerular hyperfiltration in early diabetic kidney disease. Kidney Int. 2022, 102, 1345–1358. [Google Scholar] [CrossRef]
- Chen, D.; Shao, M.; Song, Y.; Ren, G.; Guo, F.; Fan, X.; Wang, Y.; Zhang, W.; Qin, G. Single-cell RNA-seq with spatial transcriptomics to create an atlas of human diabetic kidney disease. FASEB J. 2023, 37, e22938. [Google Scholar] [CrossRef]
- Yamashita, T.; Hashiramoto, A.; Haluzik, M.; Mizukami, H.; Beck, S.; Norton, A.; Kono, M.; Tsuji, S.; Daniotti, J.L.; Werth, N.; et al. Enhanced insulin sensitivity in mice lacking ganglioside GM3. Proc. Natl. Acad. Sci. USA 2003, 100, 3445–3449. [Google Scholar] [CrossRef]
- Takeuchi, K.; Naito, S.; Kawashima, N.; Ishigaki, N.; Sano, T.; Kamata, K.; Takeuchi, Y. New Anti-Nephrin Antibody Mediated Podocyte Injury Model Using a C57BL/6 Mouse Strain. Nephron 2018, 138, 71–87. [Google Scholar] [CrossRef]
- Root, H.F.; Pote, W.H., Jr.; Frehner, H. Triopathy of diabetes; sequence of neuropathy, retinopathy, and nephropathy in one hundred fifty-five patients. AMA Arch. Intern. Med. 1954, 94, 931–941. [Google Scholar] [CrossRef] [PubMed]


| Normal | DN | |
|---|---|---|
| patients | 16 | 11 |
| male:female | 11:5 | 7:4 |
| age at biopsy, years | 62.5 (50, 68.5) | 58.0 (50.5, 64.8) |
| body height, cm | 166.9 (158.1, 170.4) | 164.2 (159.5, 169.7) |
| body weight, kg | 63.7 (52.5, 69.1) | 66.9 (56.3, 79.9) |
| hemoglobin, g/dL | 15.2 (13.4, 15.5) | 10.6 (10.1, 12.4) ## |
| serum albumin, g/dL | 4.5 (4.3, 4.5) | 3.3 (2.5, 3.8) ## |
| serum urea nitrogen, mg/dL | 14.6 (12.5, 18.3) | 31.4 (23.7, 49.1) ## |
| serum creatinine, mg/dL | 0.87 (0.74, 0.93) | 2.26 (1.13, 4.21) ## |
| eGFR, mL/min/1.73 m2 | 66.5 (62, 71) | 26.0 (10.3, 51.7) ## |
| HbA1c | - | 6.0 (5.48, 6.8) |
| proteinuria, g/gCr | 0.04 (0.01, 0.06) | 4.94 (1.36, 8.79) ## |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naito, S.; Nakayama, K.; Kawashima, N. Enhanced Levels of Glycosphingolipid GM3 Delay the Progression of Diabetic Nephropathy. Int. J. Mol. Sci. 2023, 24, 11355. https://doi.org/10.3390/ijms241411355
Naito S, Nakayama K, Kawashima N. Enhanced Levels of Glycosphingolipid GM3 Delay the Progression of Diabetic Nephropathy. International Journal of Molecular Sciences. 2023; 24(14):11355. https://doi.org/10.3390/ijms241411355
Chicago/Turabian StyleNaito, Shokichi, Kenichi Nakayama, and Nagako Kawashima. 2023. "Enhanced Levels of Glycosphingolipid GM3 Delay the Progression of Diabetic Nephropathy" International Journal of Molecular Sciences 24, no. 14: 11355. https://doi.org/10.3390/ijms241411355
APA StyleNaito, S., Nakayama, K., & Kawashima, N. (2023). Enhanced Levels of Glycosphingolipid GM3 Delay the Progression of Diabetic Nephropathy. International Journal of Molecular Sciences, 24(14), 11355. https://doi.org/10.3390/ijms241411355





