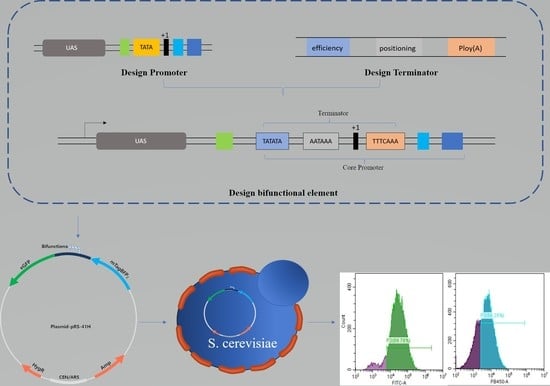
Development of Terminator–Promoter Bifunctional Elements for Application in Saccharomyces cerevisiae Pathway Engineering
Abstract
1. Introduction
2. Results and Discussion
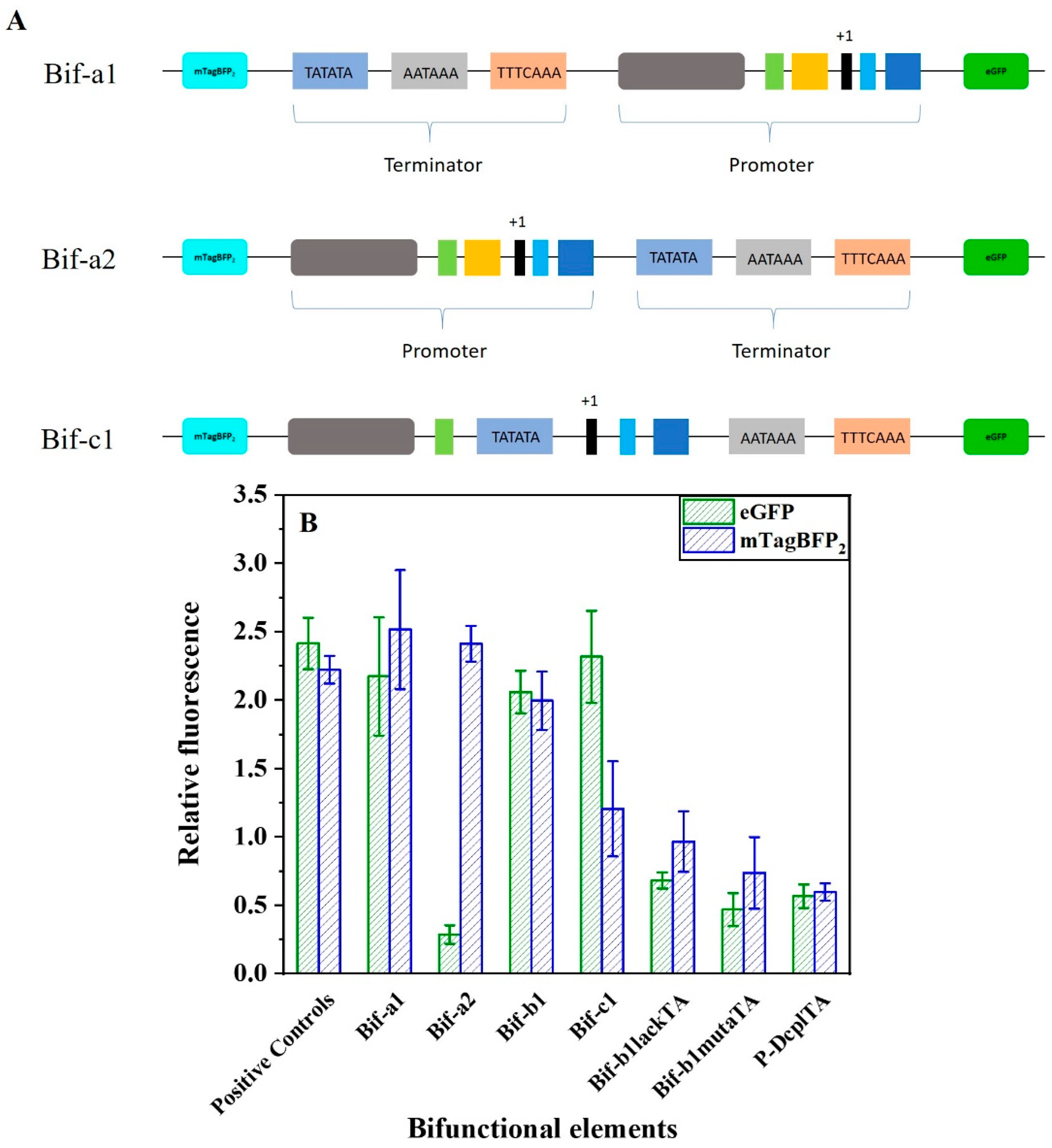
2.1. Design of Synthetic Bifunctional Element
2.2. Regulation of Promoter Activity by Spacer Sequence and UAS
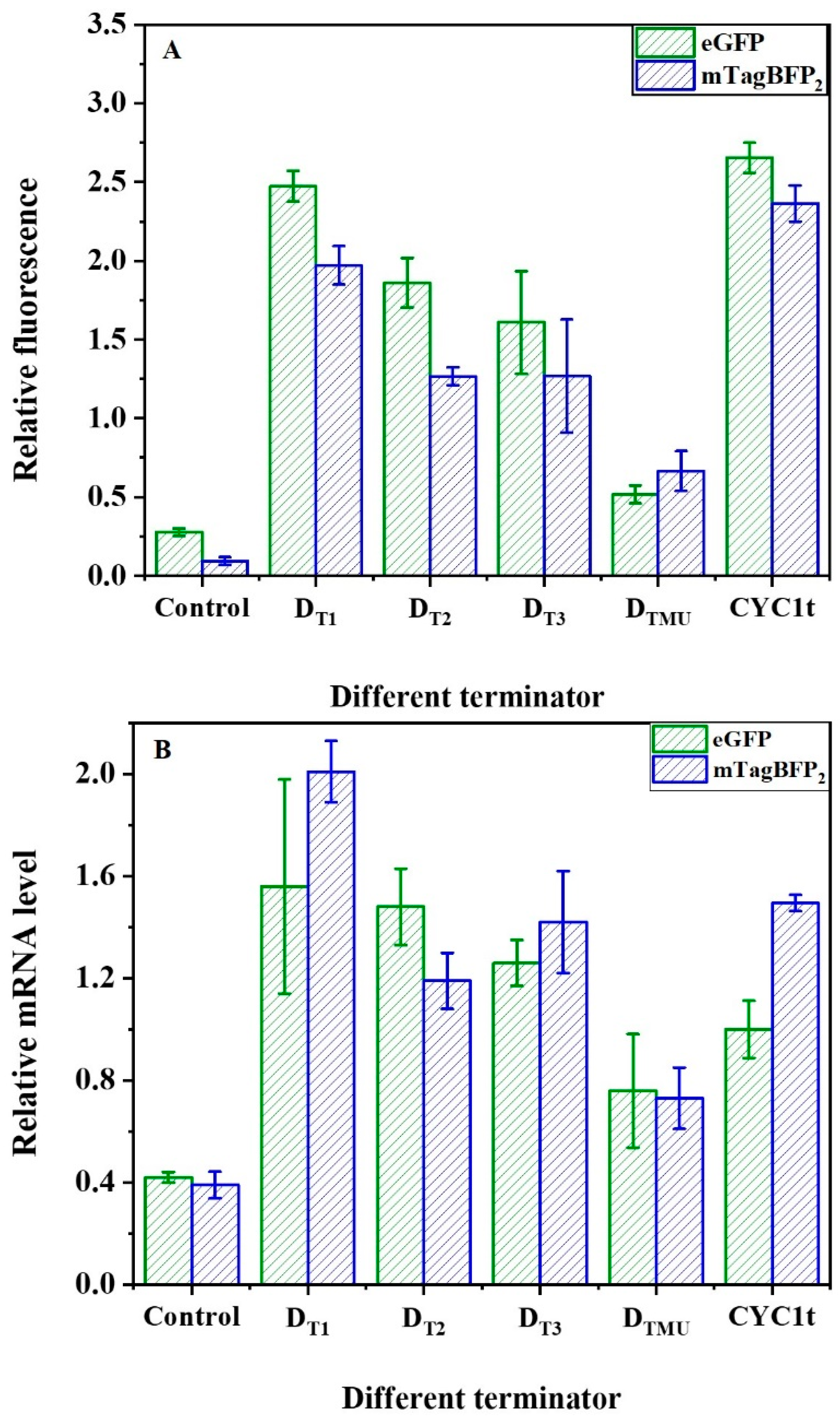
2.3. Regulation of mRNA and Fluorescent Reporter Protein Levels by the Synthetic Terminator Elements
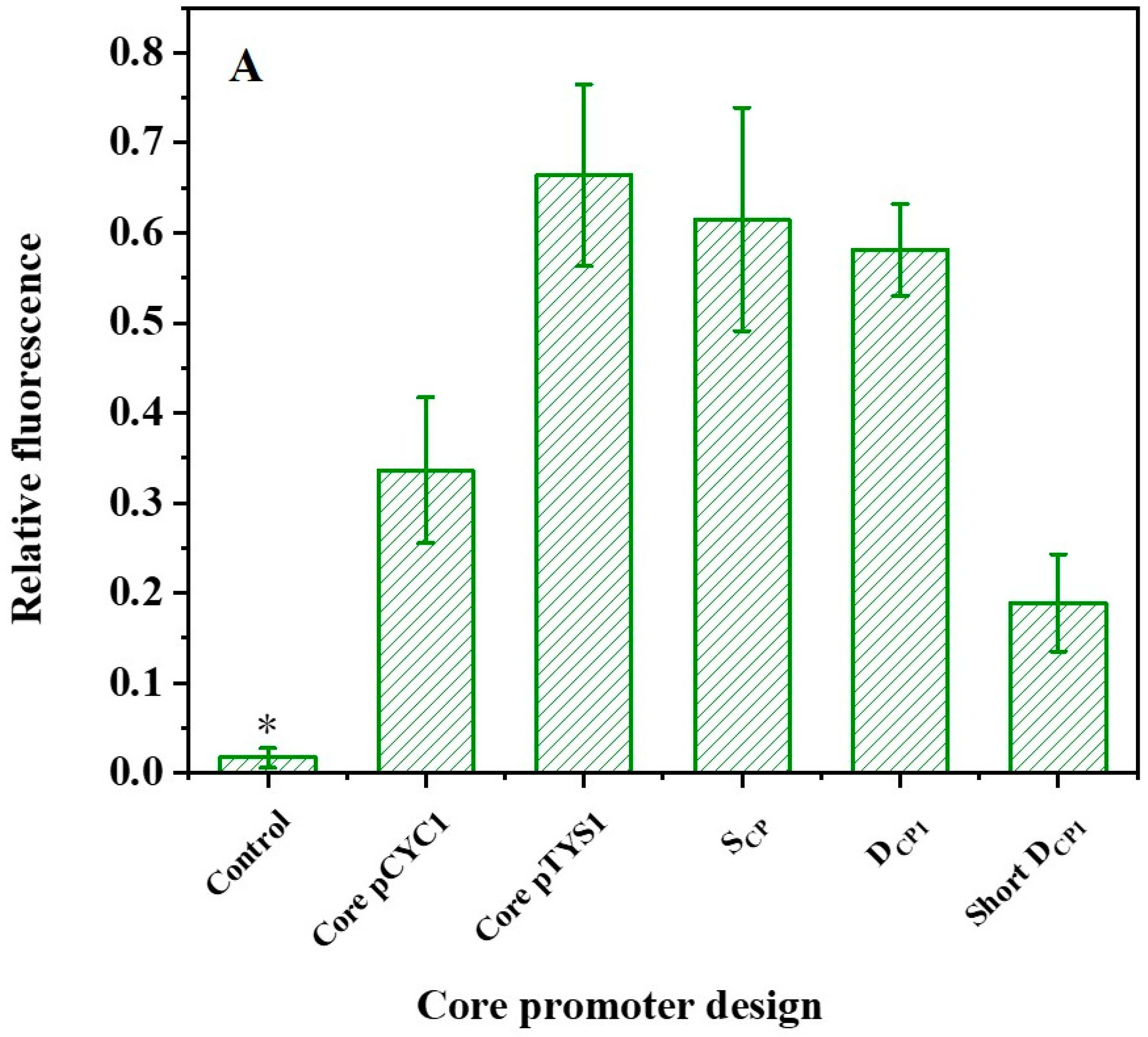
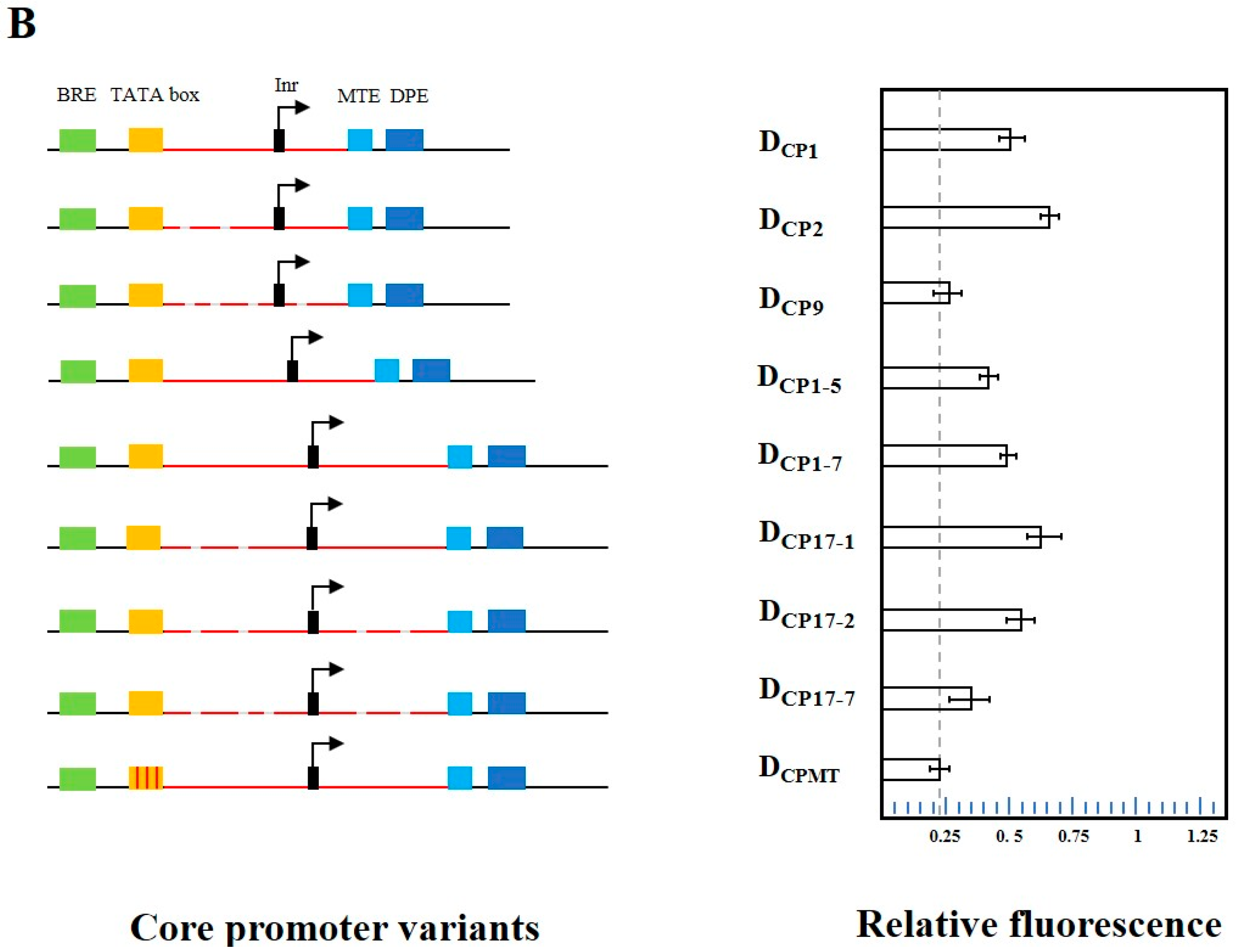
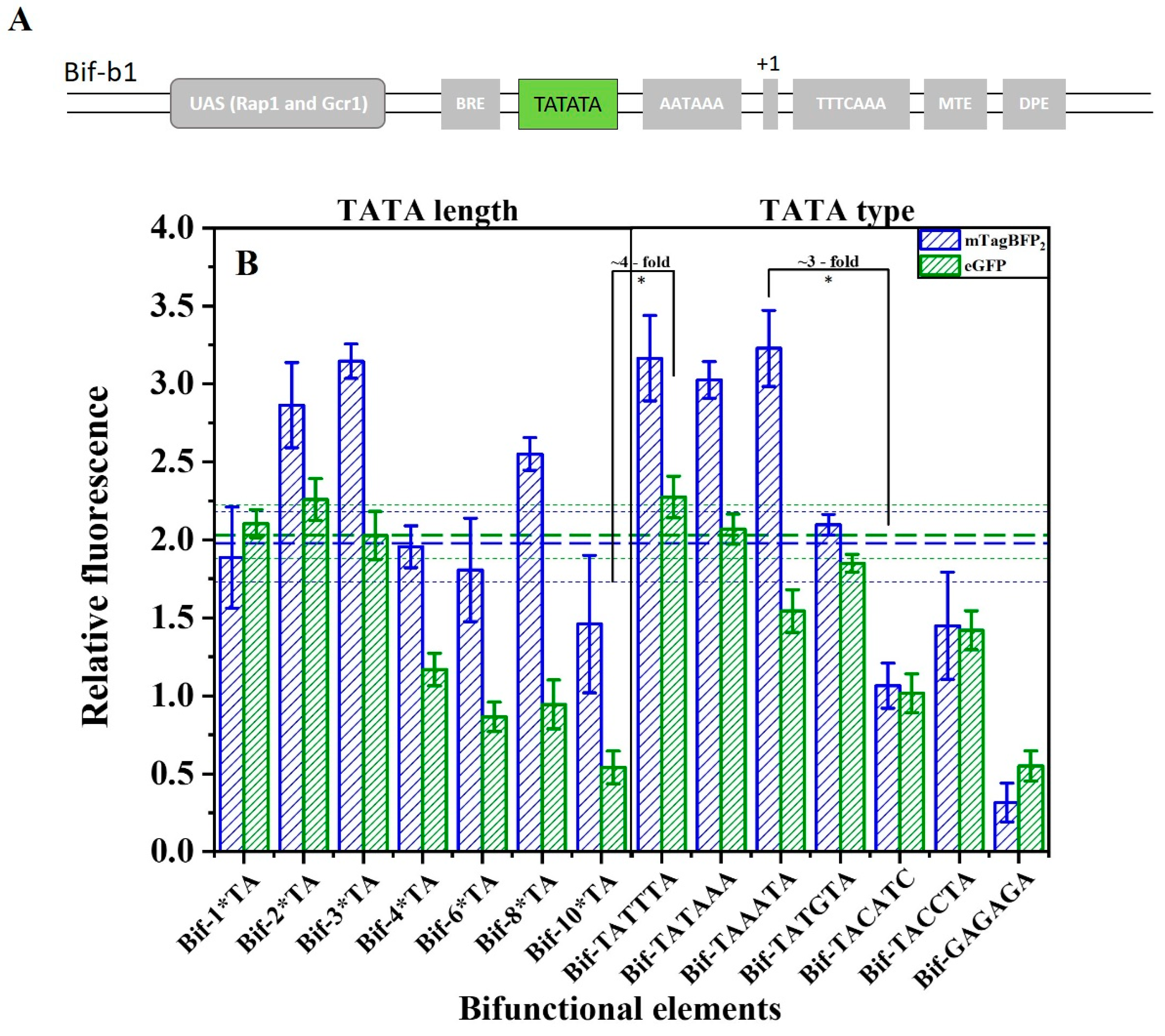
2.4. TATA Box-like Sequence Can Execute Both Functions of TATA Box and Efficiency Element
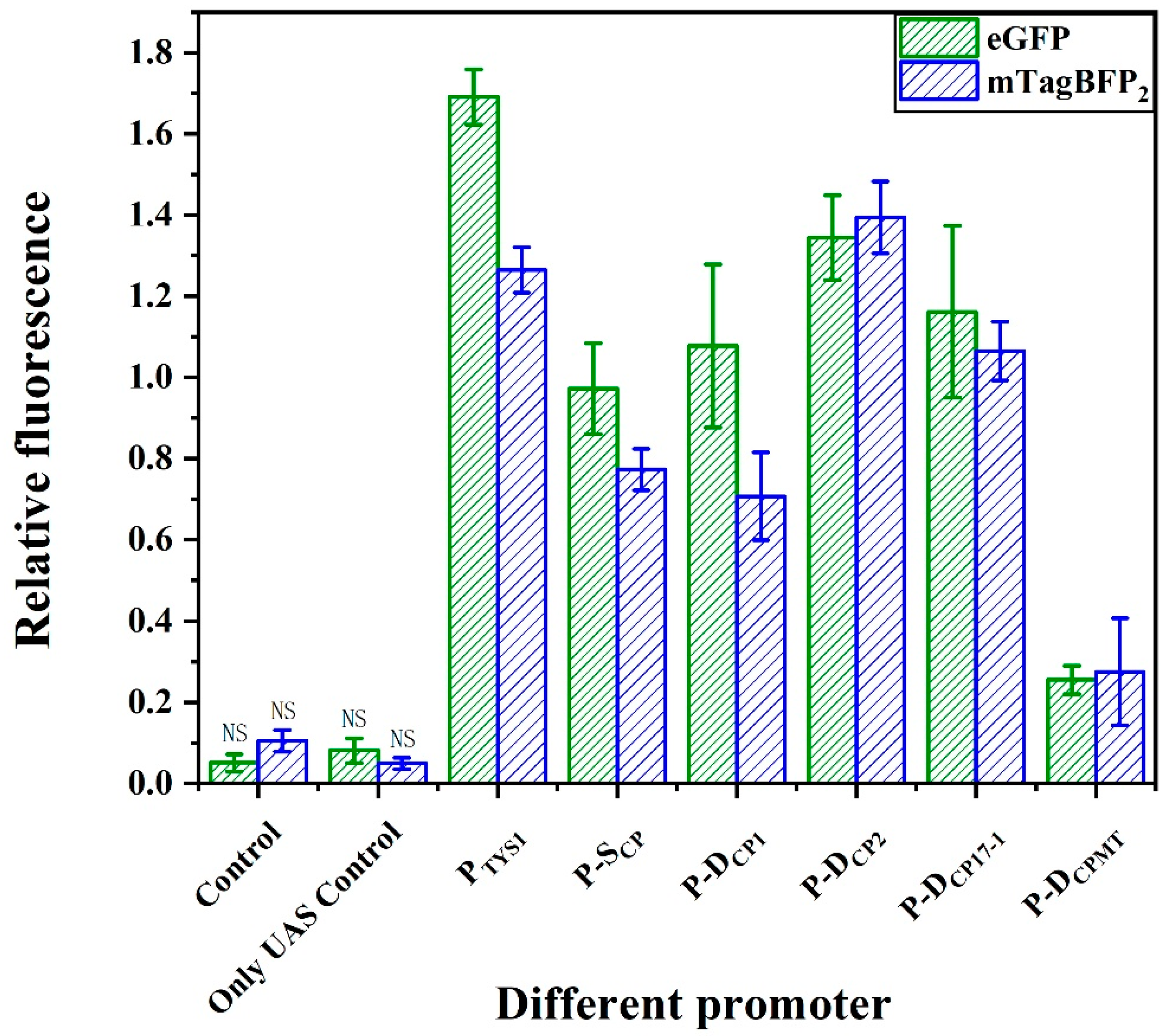
2.5. Fine-Tuning the Activities of the Bifunctional Element
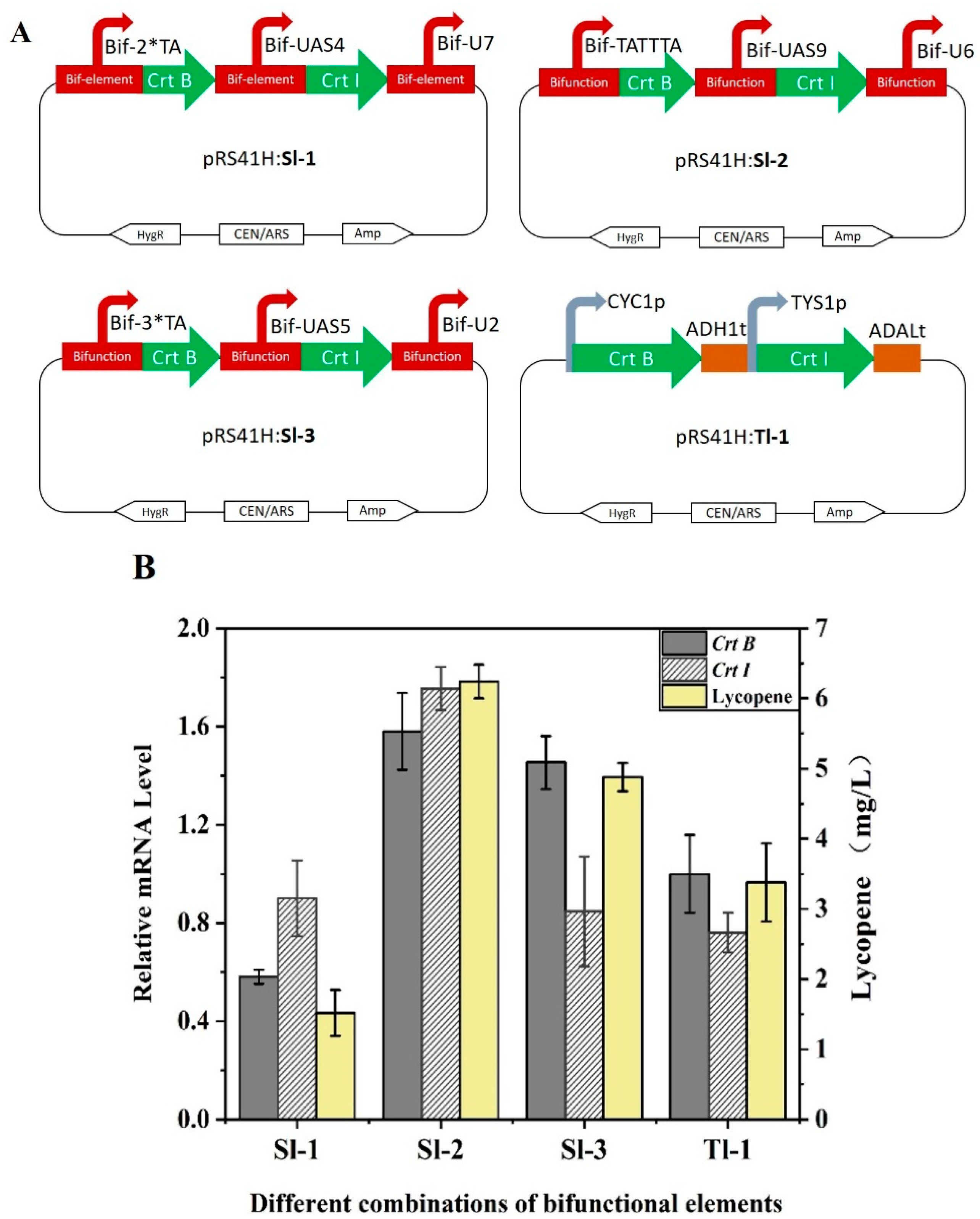
2.6. Application of the Bifunctional Element in Pathway Engineering
3. Methods
3.1. Strain, Culture Medium, and Materials
3.2. Plasmid Construction
3.3. Transformation of Yeast
3.4. Determination of Element Strength
3.5. qPCR
3.6. Statistical Analysis
3.7. Samples Preparation and Metabolite Analysis by HPLC
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| upstream activating sequence | UASs |
| super core promoter | SCP |
| initiator | Inr |
| motif ten element | MTE |
| downstream promoter element | DPE |
| enhanced green fluorescent protein | eGFP |
| blue fluorescent protein | mTagBFP2 |
| TATA box binding protein | TBP |
| Transcription Factor IIB | TFIIB |
| TFIIB recognition element | BRE |
| general transcription factors | GTFs |
| preinitiation complex | PIC |
| RNA polymerase | RNAP |
| polymerase II | Pol II |
| Designed core promoter (in this study) | DCP |
| Designed terminator (in this study) | DT |
| Designed promoter (in this study) | P-DCP |
| geranylgeranyl diphosphate | GGPP |
References
- Portela, R.M.C.; Vogl, T.; Kniely, C.; Fischer, J.E.; Oliveira, R.; Glieder, A. Synthetic Core Promoters as Universal Parts for Fine-Tuning Expression in Different Yeast Species. ACS Synth. Biol. 2017, 6, 471–484. [Google Scholar] [CrossRef]
- Ellis, T.; Adie, T.; Baldwin, G.S. DNA assembly for synthetic biology: From parts to pathways and beyond. Integr. Biol. 2011, 3, 109–118. [Google Scholar] [CrossRef]
- Hahn, S.; Young, E.T. Transcriptional Regulation in Saccharomyces cerevisiae: Transcription Factor Regulation and Function, Mechanisms of Initiation, and Roles of Activators and Coactivators. Genetics 2011, 189, 705–736. [Google Scholar] [CrossRef]
- Gertz, J.; Siggia, E.D.; Cohen, B.A. Analysis of combinatorial cis-regulation in synthetic and genomic promoters. Nature 2009, 457, 215-U113. [Google Scholar] [CrossRef]
- Guo, Z.J.; Sherman, F. 3’-end-forming signals of yeast mRNA. Trends Biochem. Sci. 1996, 21, 477–481. [Google Scholar] [CrossRef]
- Smale, S.T.; Kadonaga, J.T. The RNA polymerase II core promoter. Annu. Rev. Biochem. 2003, 72, 449–479. [Google Scholar] [CrossRef]
- Juven-Gershon, T.; Cheng, S.; Kadonaga, J.T. Rational design of a super core promoter that enhances gene expression. Nat. Methods 2006, 3, 917–922. [Google Scholar] [CrossRef] [PubMed]
- Blazeck, J.; Liu, L.Q.; Redden, H.; Alper, H. Tuning Gene Expression in Yarrowia lipolytica by a Hybrid Promoter Approach. Appl. Environ. Microbiol. 2011, 77, 7905–7914. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Liu, Q.T.; Zhang, Y.F.; Du, G.C.; Chen, J.; Kang, Z. Construction and Characterization of Broad-Spectrum Promoters for Synthetic Biology. ACS Synth. Biol. 2018, 7, 287–291. [Google Scholar] [CrossRef] [PubMed]
- Curran, K.A.; Karim, A.S.; Gupta, A.; Alper, H.S. Use of expression-enhancing terminators in Saccharomyces cerevisiae to increase mRNA half-life and improve gene expression control for metabolic engineering applications. Metab. Eng. 2013, 19, 88–97. [Google Scholar] [CrossRef]
- Guo, Z.J.; Sherman, F. Signals sufficient for 3’-end formation of yeast mRNA. Mol. Cell. Biol. 1996, 16, 2772–2776. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.X.; Wei, L.N.; Sheng, Y.; Zhang, G.L. Yeast Synthetic Terminators: Fine Regulation of Strength through Linker Sequences. ChemBioChem 2019, 20, 2383–2389. [Google Scholar] [CrossRef] [PubMed]
- Kang, W.; Ha, K.S.; Uhm, H.; Park, K.; Lee, J.Y.; Hohng, S.; Kang, C. Transcription reinitiation by recycling RNA polymerase that diffuses on DNA after releasing terminated RNA. Nat. Commun. 2020, 11, 450. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.G.; Young, L.; Chuang, R.-Y.; Venter, J.C.; Hutchison, C.A., III; Smith, H.O. Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat. Methods 2009, 6, 343-U41. [Google Scholar] [CrossRef]
- Engler, C.; Kandzia, R.; Marillonnet, S. A One Pot, One Step, Precision Cloning Method with High Throughput Capability. PLoS ONE 2008, 3, e3647. [Google Scholar] [CrossRef]
- Berrow, N.S.; Alderton, D.; Sainsbury, S.; Nettleship, J.; Assenberg, R.; Rahman, N.; Stuart, D.I.; Owens, R.J. A versatile ligation-independent cloning method suitable for high-throughput expression screening applications. Nucleic Acids Res. 2007, 35, 12. [Google Scholar] [CrossRef]
- Ma, H.; Kunes, S.; Schatz, P.J.; Botstein, D. Plasmid construction by homologous recombination in yeast. Gene 1987, 58, 201–216. [Google Scholar] [CrossRef]
- MacPherson, M.; Saka, Y. Short Synthetic Terminators for Assembly of Transcription Units in Vitro and Stable Chromosomal Integration in Yeast S. cerevisiae. ACS Synth. Biol. 2017, 6, 130–138. [Google Scholar] [CrossRef][Green Version]
- Purnick, P.E.; Weiss, R. The second wave of synthetic biology: From modules to systems. Nat. Rev. Mol. Cell Biol. 2009, 10, 410–422. [Google Scholar] [CrossRef]
- MacDonald, J.T.; Barnes, C.; Kitney, R.I.; Freemont, P.S.; Stan, G.-B.V. Computational design approaches and tools for synthetic biology. Integr. Biol. 2011, 3, 97–108. [Google Scholar] [CrossRef]
- Basehoar, A.D.; Zanton, S.J.; Pugh, B.F. Identification and distinct regulation of yeast TATA box-containing genes. Cell 2004, 117, 847. [Google Scholar] [CrossRef]
- Lee, S.; Hahn, S. Model for binding of transcription factor TFIIB to the TBP-DNA complex. Nature 1995, 376, 609–612. [Google Scholar] [CrossRef]
- Lagrange, T.; Kim, T.K.; Orphanides, G.; Ebright, Y.W.; Ebright, R.H.; Reinberg, D. High-resolution mapping of nucleoprotein complexes by site-specific protein-DNA photocrosslinking: Organization of the human TBP-TFIIA-TFIIB-DNA quaternary complex. Proc. Natl. Acad. Sci. USA 1996, 93, 10620–10625. [Google Scholar] [CrossRef]
- Cramer, P. Organization and regulation of gene transcription. Nature 2019, 573, 45–54. [Google Scholar] [CrossRef]
- Kadonaga, J.T. The DPE, a core promoter element for transcription by RNA polymerase II. Exp. Mol. Med. 2002, 34, 259–264. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lim, C.Y.; Santoso, B.; Boulay, T.; Dong, E.; Ohler, U.; Kadonaga, J.T. The MTE, a new core promoter element for transcription by RNA polymerase II. Genes Dev. 2004, 18, 1606–1617. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Blazeck, J.; Alper, H.S. Promoter engineering: Recent advances in controlling transcription at the most fundamental level. Biotechnol. J. 2013, 8, 46. [Google Scholar] [CrossRef] [PubMed]
- Juven-Gershon, T.; Kadonaga, J.T. Regulation of gene expression via the core promoter and the basal transcriptional machinery. Dev. Biol. 2010, 339, 225–229. [Google Scholar] [CrossRef]
- van Helden, J.; del Olmo, M.; Perez-Ortin, J.E. Statistical analysis of yeast genomic downstream sequences reveals putative polyadenylation signals. Nucleic Acids Res. 2000, 28, 1000–1010. [Google Scholar] [CrossRef] [PubMed]
- Argaman, L.; Hershberg, R.; Vogel, J.; Bejerano, G.; Wagner, E.G.H.; Margalit, H.; Altuvia, S. Novel small RNA-encoding genes in the intergenic regions of Escherichia coli. Curr. Biol. 2001, 11, 941–950. [Google Scholar] [CrossRef]
- Curran, K.A.; Morse, N.J.; Markham, K.A.; Wagman, A.M.; Gupta, A.; Alper, H.S. Short Synthetic Terminators for Improved Heterologous Gene Expression in Yeast. ACS Synth. Biol. 2015, 4, 824–832. [Google Scholar] [CrossRef]
- Dye, M.J.; Proudfoot, N.J. Terminal exon definition occurs contrascriptionally and promotes termination of RNA polymerase II. Mol. Cell 1999, 3, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, S.; Young, E.M.; Jones, T.S.; Densmore, D.; Voigt, C.A. Genetic circuit design automation for yeast. Nat. Microbiol. 2020, 5, 1349–1360. [Google Scholar] [CrossRef] [PubMed]
- Blount, B.; Weenink, T.; Vasylechko, S.; Ellis, T. Rational Diversification of a Promoter Providing Fine-Tuned Expression and Orthogonal Regulation for Synthetic Biology. PLoS ONE 2012, 7, e33279. [Google Scholar] [CrossRef]
- Kaiser, C.; Michaelis, S.; Mitchell, A. Methods in Yeast Genetics: A Cold Spring Harbor Course Manual; CHSL Press: Plainview, NY, USA, 1994. [Google Scholar]
- Raser, J.M.; O’Shea, E.K. Control of stochasticity in eukaryotic gene expression. Science 2004, 304, 1811–1814. [Google Scholar] [CrossRef]
- Harbison, C.T.; Gordon, D.B.; Lee, T.I.; Rinaldi, N.J.; Macisaac, K.D.; Danford, T.W.; Hannett, N.M.; Tagne, J.B.; Reynolds, D.B.; Yoo, J.; et al. Transcriptional regulatory code of a eukaryotic genome. Nature 2004, 431, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.Z.; Killion, P.J.; Iyer, V.R. Genetic reconstruction of a functional transcriptional regulatory network. Nat. Genet. 2007, 39, 683–687. [Google Scholar] [CrossRef]
- Casini, A.; Christodoulou, G.; Freemont, P.S.; Baldwin, G.S.; Ellis, T.; MacDonald, J.T. R2oDNA Designer: Computational Design of Biologically Neutral Synthetic DNA Sequences. ACS Synth. Biol. 2014, 3, 525–528. [Google Scholar] [CrossRef]
- Zhang, G.; Cao, Q.; Liu, J.; Liu, B.; Li, J.; Li, C. Refactoring β-amyrin synthesis in Saccharomyces cerevisiae. AIChE J. 2015, 61, 3172–3179. [Google Scholar] [CrossRef]
- Li, X.; Wang, Z.; Zhang, G.; Yi, L. Improving lycopene production in Saccharomyces cerevisiae through optimizing pathway and chassis metabolism. Chem. Eng. Sci. 2019, 193, 364–369. [Google Scholar] [CrossRef]
- Wei, L.; Wang, Z.; Zhang, G.; Ye, B. Characterization of Terminators in Saccharomyces cerevisiae and an Exploration of Factors Affecting Their Strength. ChemBioChem 2017, 18, 2422–2427. [Google Scholar] [CrossRef]
- Cambray, G.; Guimaraes, J.C.; Mutalik, V.K.; Lam, C.; Mai, Q.-A.; Thimmaiah, T.; Carothers, J.M.; Arkin, A.P.; Endy, D. Measurement and modeling of intrinsic transcription terminators. Nucleic Acids Res. 2013, 41, 5139–5148. [Google Scholar] [CrossRef] [PubMed]
- Yamanishi, M.; Ito, Y.; Kintaka, R.; Imamura, C.; Katahira, S.; Ikeuchi, A.; Moriya, H.; Matsuyama, T. A Genome-Wide Activity Assessment of Terminator Regions in Saccharomyces cerevisiae Provides a “Terminatome” Toolbox. ACS Synth. Biol. 2013, 2, 337–347. [Google Scholar] [CrossRef] [PubMed]
- West, S.; Proudfoot, N.J. Transcriptional Termination Enhances Protein Expression in Human Cells. Mol. Cell 2009, 33, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Jing, L.; Qiang, L. Can terminators be used as insulators into yeast synthetic gene circuits? J. Biol. Eng. 2016, 10, 19. [Google Scholar] [CrossRef] [PubMed]
- Mogno, I.; Vallania, F.; Mitra, R.D.; Cohen, B.A. TATA is a modular component of synthetic promoters. Genome Res. 2010, 20, 1391–1397. [Google Scholar] [CrossRef] [PubMed]
- Sugihara, F.; Kasahara, K.; Kokubo, T. Highly redundant function of multiple AT-rich sequences as core promoter elements in the TATA-less RPS5 promoter of Saccharomyces cerevisiae. Nucleic Acids Res. 2011, 39, 59–75. [Google Scholar] [CrossRef]
- Misawa, N.; Nakagawa, M.; Kobayashi, K.; Yamano, S.; Izawa, Y.; Nakamura, K.; Harashima, K. Elucidation of the Erwinia uredovora carotenoid biosynthetic pathway by functional analysis of gene products expressed in Escherichia coli. J. Bacteriol. 1990, 172, 6704–6712. [Google Scholar] [CrossRef]
- Lubliner, S.; Keren, L.; Segal, E. Sequence features of yeast and human core promoters that are predictive of maximal promoter activity. Nucleic Acids Res. 2013, 41, 5569–5581. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ni, X.; Liu, Z.; Guo, J.; Zhang, G. Development of Terminator–Promoter Bifunctional Elements for Application in Saccharomyces cerevisiae Pathway Engineering. Int. J. Mol. Sci. 2023, 24, 9870. https://doi.org/10.3390/ijms24129870
Ni X, Liu Z, Guo J, Zhang G. Development of Terminator–Promoter Bifunctional Elements for Application in Saccharomyces cerevisiae Pathway Engineering. International Journal of Molecular Sciences. 2023; 24(12):9870. https://doi.org/10.3390/ijms24129870
Chicago/Turabian StyleNi, Xiaoxia, Zhengyang Liu, Jintang Guo, and Genlin Zhang. 2023. "Development of Terminator–Promoter Bifunctional Elements for Application in Saccharomyces cerevisiae Pathway Engineering" International Journal of Molecular Sciences 24, no. 12: 9870. https://doi.org/10.3390/ijms24129870
APA StyleNi, X., Liu, Z., Guo, J., & Zhang, G. (2023). Development of Terminator–Promoter Bifunctional Elements for Application in Saccharomyces cerevisiae Pathway Engineering. International Journal of Molecular Sciences, 24(12), 9870. https://doi.org/10.3390/ijms24129870