Characterization and Biological Activities of Melanin from the Medicinal Fungi Ophiocordyceps sinensis
Abstract
1. Introduction
2. Results
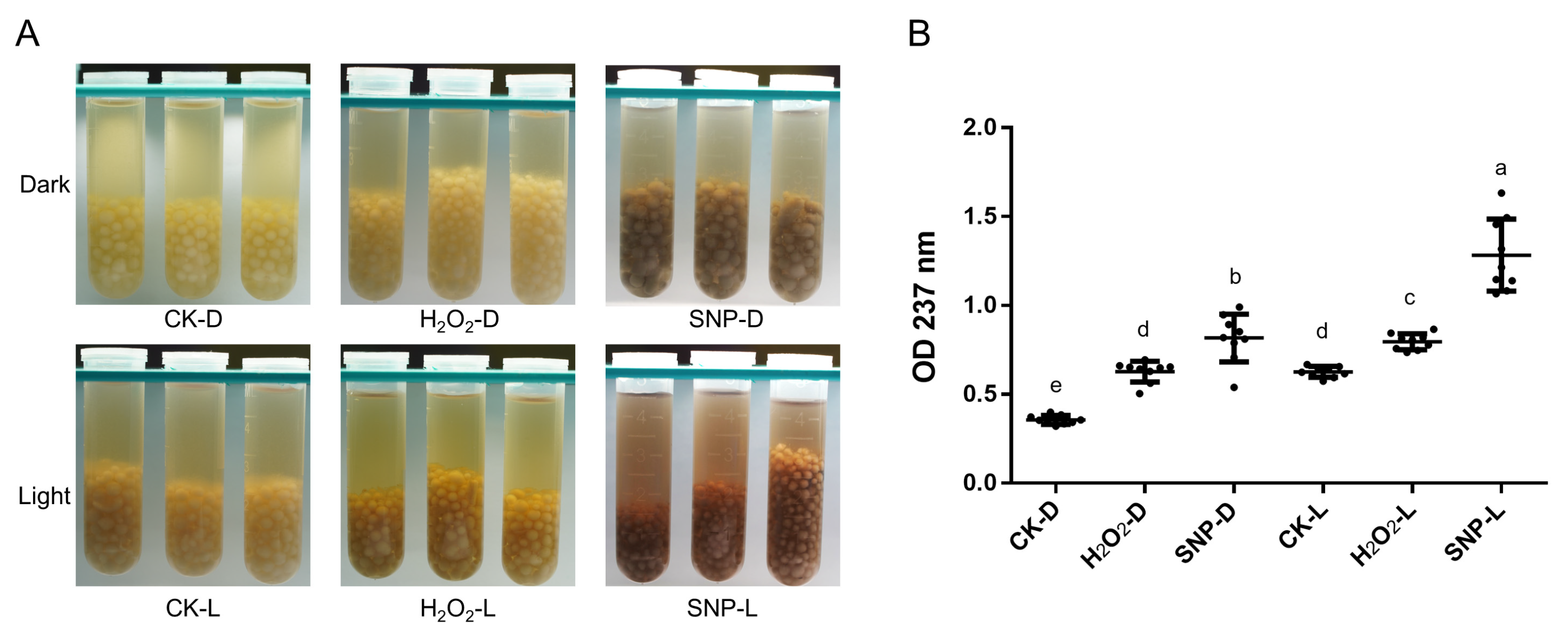
2.1. Induction Conditions of Melanin
2.2. Elemental Analysis
2.3. Spectral Characterization
2.4. Py-GCMS Analysis
2.5. Ability to Chelate Metal Ions
2.6. UV-Blocking Activity
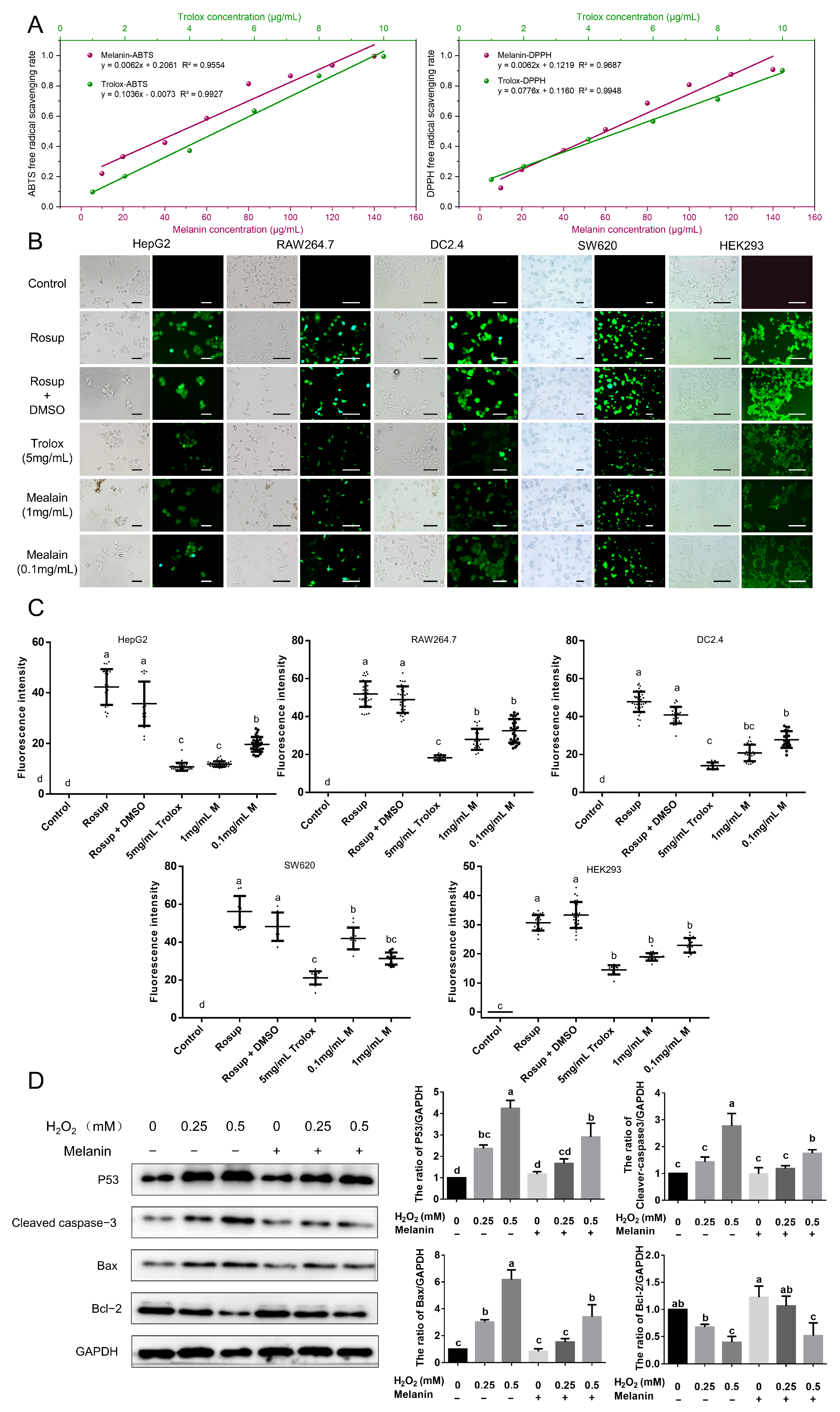
2.7. Antioxidant Activity
3. Discussion
4. Materials and Methods
4.1. Strain Cultivation and Melanin Extraction
4.2. Elemental Analysis
4.3. Spectroscopy Analysis
4.3.1. UV-Visible Absorption Spectrum
4.3.2. Fourier Transform Infrared (FTIR)
4.3.3. Electron Paramagnetic Resonance (EPR)
4.4. Pyrolysis Gas Chromatography and Mass Spectrometry (Py-GCMS)
4.5. Biological Activity
4.5.1. Ability to Chelate Metal Ions
4.5.2. UV-Blocking
4.5.3. Free-Radical-Scavenging Ability
4.5.4. Antioxidant Activity on Cells
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Choi, K.Y. Bioprocess of Microbial Melanin Production and Isolation. Front. Bioeng. Biotechnol. 2021, 9, 765110. [Google Scholar] [CrossRef] [PubMed]
- D’Alba, L.; Shawkey, M.D. Melanosomes: Biogenesis, Properties, and Evolution of an Ancient Organelle. Physiol. Rev. 2019, 99, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Li, J.; Chang, M.; Cheng, Y.; Geng, X.; Meng, J.; Zhu, M. Comparison of Physicochemical and Biochemical Properties of Natural and Arginine-Modified Melanin from Medicinal Mushroom Ganoderma lucidum. J. Basic Microbiol. 2020, 60, 1014–1028. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wu, X.; Huang, C.; Zhang, Z.; Gao, W. Isolation and Identification of Pigments from Oyster Mushrooms with Black, Yellow and Pink Caps. Food Chem. 2022, 372, 131171. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Nimse, S.B.; Mathew, D.E.; Dhimmar, A.; Sahastrabudhe, H.; Gajjar, A.; Ghadge, V.A.; Kumar, P.; Shinde, P.B. Microbial Melanin: Recent Advances in Biosynthesis, Extraction, Characterization, and Applications. Biotechnol. Adv. 2021, 53, 107773. [Google Scholar] [CrossRef]
- Leite, M.O.G.; Alves, D.A.; Lecocq, A.; Malaquias, J.B.; Delalibera, I.; Jensen, A.B. Laboratory Risk Assessment of Three Entomopathogenic Fungi Used for Pest Control toward Social Bee Pollinators. Microorganisms 2022, 10, 1800. [Google Scholar] [CrossRef]
- María Del Rosario, G.-A.; Julio, M.-A.; Sylvia Páz, D.-C.; Manuel Adrián, P.-S.; Gabriela, L.-A.; Edgar Alonso, R.-S.; Lorenzo Ulises, O.-M.; Francisco, D.-V. Soluble Melanins of the Randia Echinocarpa Fruit—Structural Characteristics and Toxicity. J. Food Biochem. 2019, 43, e13077. [Google Scholar] [CrossRef]
- Shi, F.; Li, J.; Yang, L.; Hou, G.; Ye, M. Hypolipidemic Effect and Protection Ability of Liver-Kidney Functions of Melanin from Lachnum YM226 in High-Fat Diet Fed Mice. Food Funct. 2018, 9, 880–889. [Google Scholar] [CrossRef]
- Li, X.; Wu, W.; Zhang, F.; Hu, X.; Yuan, Y.; Wu, X.; Fu, J. Differences between Water-Soluble and Water-Insoluble Melanin Derived from Inonotus hispidus Mushroom. Food Chem. X 2022, 16, 100498. [Google Scholar] [CrossRef]
- Chai, W.; Wei, Q.; Deng, W.; Zheng, Y.; Chen, X.; Huang, Q.; OuYang, C.; Peng, Y. Anti-Melanogenesis Properties of Condensed Tannins from Vigna angularis Seeds with Potent Antioxidant and DNA Damage Protection Activities. Food Funct. 2019, 10, 99–111. [Google Scholar] [CrossRef]
- Ghadge, V.; Kumar, P.; Singh, S.; Mathew, D.E.; Bhattacharya, S.; Nimse, S.B.; Shinde, P.B. Natural Melanin Produced by the Endophytic Bacillus subtilis 4NP-BL Associated with the Halophyte Salicornia brachiata. J. Agric. Food Chem. 2020, 68, 6854–6863. [Google Scholar] [CrossRef]
- Wang, L.; Jayawardena, T.U.; Yang, H.-W.; Lee, H.-G.; Jeon, Y.-J. The Potential of Sulfated Polysaccharides Isolated from the Brown Seaweed Ecklonia maxima in Cosmetics: Antioxidant, Anti-Melanogenesis, and Photoprotective Activities. Antioxidants 2020, 9, 724. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. New Insight into Melanin for Food Packaging and Biotechnology Applications. Crit. Rev. Food Sci. Nutr. 2022, 62, 4629–4655. [Google Scholar] [CrossRef]
- Tran-Ly, A.N.; Reyes, C.; Schwarze, F.W.M.R.; Ribera, J. Microbial Production of Melanin and Its Various Applications. World J. Microbiol. Biotechnol. 2020, 36, 170. [Google Scholar] [CrossRef] [PubMed]
- Martínez, L.M.; Martinez, A.; Gosset, G. Production of Melanins with Recombinant Microorganisms. Front. Bioeng. Biotechnol. 2019, 7, 285. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Song, A.; Yin, J.; Siu, K.; Wong, W.; Wu, J. Anti-Inflammation Activity of Exopolysaccharides Produced by a Medicinal Fungus Cordyceps sinensis Cs-HK1 in Cell and Animal Models. Int. J. Biol. Macromol. 2020, 149, 1042–1050. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, Q.; Li, L.; Zhou, X. Immunostimulatory Effects of the Intracellular Polysaccharides Isolated from Liquid Culture of Ophiocordyceps sinensis (Ascomycetes) on RAW264.7 Cells via the MAPK and PI3K/Akt Signaling Pathways. J. Ethnopharmacol. 2021, 275, 114130. [Google Scholar] [CrossRef]
- Lu, Q.; Li, C.; Chen, W.; Shi, Z.; Zhan, R.; He, R. Clinical Efficacy of Jinshuibao Capsules Combined with Angiotensin Receptor Blockers in Patients with Early Diabetic Nephropathy: A Meta-Analysis of Randomized Controlled Trials. Evid. Based Complement. Altern. Med. ECAM 2018, 2018, 6806943. [Google Scholar] [CrossRef]
- Tong, X.; Guo, J. High Throughput Identification of the Potential Antioxidant Peptides in Ophiocordyceps Sinensis. Molecules 2022, 27, 438. [Google Scholar] [CrossRef]
- Dong, C.; Yao, Y. Isolation, Characterization of Melanin Derived from Ophiocordyceps sinensis, an Entomogenous Fungus Endemic to the Tibetan Plateau. J. Biosci. Bioeng. 2012, 113, 474–479. [Google Scholar] [CrossRef]
- Ito, S.; Fujita, K. Microanalysis of Eumelanin and Pheomelanin in Hair and Melanomas by Chemical Degradation and Liquid Chromatography. Anal. Biochem. 1985, 144, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Surendirakumar, K.; Pandey, R.R.; Muthukumar, T.; Sathiyaseelan, A.; Loushambam, S.; Seth, A. Characterization and Biological Activities of Melanin Pigment from Root Endophytic Fungus, Phoma sp. RDSE17. Arch. Microbiol. 2022, 204, 171. [Google Scholar] [CrossRef]
- Prados-Rosales, R.; Toriola, S.; Nakouzi, A.; Chatterjee, S.; Stark, R.; Gerfen, G.; Tumpowsky, P.; Dadachova, E.; Casadevall, A. Structural Characterization of Melanin Pigments from Commercial Preparations of the Edible Mushroom Auricularia auricula. J. Agric. Food Chem. 2015, 63, 7326–7332. [Google Scholar] [CrossRef] [PubMed]
- De Souza, R.A.; Kamat, N.M.; Nadkarni, V.S. Purification and Characterisation of a Sulphur Rich Melanin from Edible Mushroom Termitomyces albuminosus Heim. Mycology 2018, 9, 296–306. [Google Scholar] [CrossRef]
- Liu, Q.; Xiao, J.; Liu, B.; Zhuang, Y.; Sun, L. Study on the Preparation and Chemical Structure Characterization of Melanin from Boletus griseus. Int. J. Mol. Sci. 2018, 19, 3736. [Google Scholar] [CrossRef]
- Dzierzega-Lecznar, A.; Kurkiewicz, S.; Stepien, K.; Chodurek, E.; Riederer, P.; Gerlach, M. Structural Investigations of Neuromelanin by Pyrolysis-Gas Chromatography/Mass Spectrometry. J. Neural Transm. 2006, 113, 729–734. [Google Scholar] [CrossRef]
- Liu, X.; Hou, R.; Wang, D.; Mai, M.; Wu, X.; Zheng, M.; Fu, J. Comprehensive Utilization of Edible Mushroom Auricularia auricula Waste Residue-Extraction, Physicochemical Properties of Melanin and Its Antioxidant Activity. Food Sci. Nutr. 2019, 7, 3774–3783. [Google Scholar] [CrossRef] [PubMed]
- Elsayis, A.; Hassan, S.W.M.; Ghanem, K.M.; Khairy, H. Optimization of Melanin Pigment Production from the Halotolerant Black Yeast Hortaea werneckii AS1 Isolated from Solar Salter in Alexandria. BMC Microbiol. 2022, 22, 92. [Google Scholar] [CrossRef]
- Suwannarach, N.; Kumla, J.; Watanabe, B.; Matsui, K.; Lumyong, S. Characterization of Melanin and Optimal Conditions for Pigment Production by an Endophytic Fungus, Spissiomyces endophytica SDBR-CMU319. PLoS ONE 2019, 14, e0222187. [Google Scholar] [CrossRef]
- Sun, W.; Yu, Y.; Chen, J.; Yu, B.; Chen, T.; Ying, H.; Zhou, S.; Ouyang, P.; Liu, D.; Chen, Y. Light Signaling Regulates Aspergillus niger Biofilm Formation by Affecting Melanin and Extracellular Polysaccharide Biosynthesis. mBio 2021, 12, e03434-20. [Google Scholar] [CrossRef]
- Zhao, Y.; Lim, J.; Xu, J.; Yu, J.-H.; Zheng, W. Nitric Oxide as a Developmental and Metabolic Signal in Filamentous Fungi. Mol. Microbiol. 2020, 113, 872–882. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Liu, M.; Huang, H.; Wen, Y.; Zhang, X.; Wei, Y. Recent Advances and Progress on Melanin-like Materials and Their Biomedical Applications. Biomacromolecules 2018, 19, 1858–1868. [Google Scholar] [CrossRef] [PubMed]
- Thaira, H.; Raval, K.; Manirethan, V.; Balakrishnan, R.M. Melanin Nano-Pigments for Heavy Metal Remediation from Water. Sep. Sci. Technol. 2019, 54, 265–274. [Google Scholar] [CrossRef]
- Rizvi, A.; Ahmed, B.; Zaidi, A.; Khan, M.S. Bioreduction of Toxicity Influenced by Bioactive Molecules Secreted under Metal Stress by Azotobacter chroococcum. Ecotoxicology 2019, 28, 302–322. [Google Scholar] [CrossRef]
- Chen, S.; Xue, C.; Wang, J.; Feng, H.; Wang, Y.; Ma, Q.; Wang, D. Adsorption of Pb(II) and Cd(II) by Squid Ommastrephes bartrami Melanin. Bioinorg. Chem. Appl. 2009, 2009, 901563. [Google Scholar] [CrossRef]
- Manirethan, V.; Raval, K.; Rajan, R.; Thaira, H.; Balakrishnan, R.M. Kinetic and Thermodynamic Studies on the Adsorption of Heavy Metals from Aqueous Solution by Melanin Nanopigment Obtained from Marine Source: Pseudomonas stutzeri. J. Environ. Manag. 2018, 214, 315–324. [Google Scholar] [CrossRef]
- Chrissian, C.; Camacho, E.; Fu, M.S.; Prados-Rosales, R.; Chatterjee, S.; Cordero, R.J.B.; Lodge, J.K.; Casadevall, A.; Stark, R.E. Melanin Deposition in Two Cryptococcus Species Depends on Cell-Wall Composition and Flexibility. J. Biol. Chem. 2020, 295, 1815–1828. [Google Scholar] [CrossRef]
- Eisenman, H.C.; Nosanchuk, J.D.; Webber, J.B.W.; Emerson, R.J.; Camesano, T.A.; Casadevall, A. Microstructure of Cell Wall-Associated Melanin in the Human Pathogenic Fungus Cryptococcus neoformans. Biochemistry 2005, 44, 3683–3693. [Google Scholar] [CrossRef]
- Perez-Dulzaides, R.; Camacho, E.; Cordero, R.J.B.; Casadevall, A. Cell-Wall Dyes Interfere with Cryptococcus neoformans Melanin Deposition. Microbiol. Read. Engl. 2018, 164, 1012–1022. [Google Scholar] [CrossRef]
- Walton, F.J.; Idnurm, A.; Heitman, J. Novel Gene Functions Required for Melanization of the Human Pathogen Cryptococcus neoformans. Mol. Microbiol. 2005, 57, 1381–1396. [Google Scholar] [CrossRef] [PubMed]
- Khemakhem, M.; Papadimitriou, V.; Sotiroudis, G.; Zoumpoulakis, P.; Arbez-Gindre, C.; Bouzouita, N.; Sotiroudis, T.G. Melanin and Humic Acid-like Polymer Complex from Olive Mill Waste Waters. Part I. Isolation and Characterization. Food Chem. 2016, 203, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Bian, Y.; Guo, J.; Majeed, H.; Zhu, K.; Guo, X.; Peng, W.; Zhou, H. Ferulic Acid Renders Protection to HEK293 Cells against Oxidative Damage and Apoptosis Induced by Hydrogen Peroxide. Vitro Cell. Dev. Biol. Anim. 2015, 51, 722–729. [Google Scholar] [CrossRef] [PubMed]


| C | H | O | N | S | C/N | C/H | C/O | |
|---|---|---|---|---|---|---|---|---|
| O. sinensis | 50.59 | 6.18 | 33.90 | 8.19 | 1.20 | 7.21 | 9.55 | 1.99 |
| Eumelanin * [21] | 56.45 | 3.15 | 31.82 | 8.49 | 0.09 | 7.76 | 20.91 | 2.37 |
| Phaeomelanin * [21] | 46.24 | 4.46 | 30.16 | 9.36 | 9.78 | 5.76 | 12.10 | 2.04 |
| Inonotus hispidus [9] | 43.67 | 6.25 | 46.58 | 3.5 | 0 | 14.56 | 8.15 | 1.25 |
| Ganoderma lucidum [3] | 54.20 | 6.24 | 33.35 | 5.14 | 1.07 | 12.30 | 10.13 | 2.17 |
| Phoma sp. RDSE17 [22] | 56.71 | 5.15 | 37.52 | 0.62 | 0 | 106.71 | 12.85 | 2.02 |
| Auricularia auricula [23] | 41.18 | 5.56 | 51.60 | 1.66 | 0 | 28.94 | 8.64 | 1.06 |
| Termitomyces albuminosus [24] | 54.68 | 3.54 | 26.92 | 2.49 | 12.36 | 25.60 | 18.00 | 2.71 |
| Boletus griseus [25] | 56.38 | 5.86 | 28.04 | 6.17 | 2.44 | 10.66 | 11.22 | 2.68 |
| Num. | RT (min) | Area (%) | Structure | Num. | RT (min) | Area (%) | Structure |
|---|---|---|---|---|---|---|---|
| 1 | 1.389 | 29.3 | 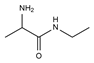 | 13 | 3.376 | 2.86 | 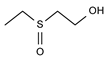 |
| 2 | 1.448 | 15.29 | 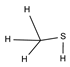 | 14 | 5.607 | 1.12 | 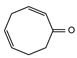 |
| 3 | 1.554 | 16.04 | 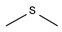 | 15 | 7.098 | 0.19 | 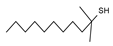 |
| 4 | 1.635 | 0.93 | 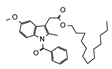 | 16 | 8.712 | 0.34 | 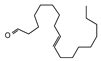 |
| 5 | 1.708 | 2.45 | 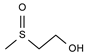 | 17 | 12.022 | 0.42 |  |
| 6 | 1.742 | 1.46 |  | 18 | 12.339 | 3.18 |  |
| 7 | 1.943 | 1.03 | 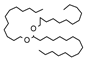 | 19 | 12.419 | 0.65 |  |
| 8 | 1.991 | 0.81 | 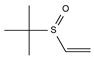 | 20 | 13.287 | 10.24 | 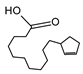 |
| 9 | 2.094 | 1.57 |  | 21 | 13.4 | 0.83 | 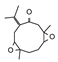 |
| 10 | 2.23 | 1.09 | 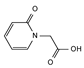 | 22 | 14.158 | 1.24 | 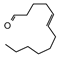 |
| 11 | 2.537 | 3.51 | 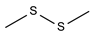 | 23 | 14.259 | 1.18 | 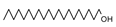 |
| 12 | 2.758 | 2.87 |  | 24 | 17.467 | 1.39 | 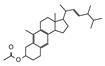 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tong, C.; Luo, J.; Xie, C.; Wei, J.; Pan, G.; Zhou, Z.; Li, C. Characterization and Biological Activities of Melanin from the Medicinal Fungi Ophiocordyceps sinensis. Int. J. Mol. Sci. 2023, 24, 10282. https://doi.org/10.3390/ijms241210282
Tong C, Luo J, Xie C, Wei J, Pan G, Zhou Z, Li C. Characterization and Biological Activities of Melanin from the Medicinal Fungi Ophiocordyceps sinensis. International Journal of Molecular Sciences. 2023; 24(12):10282. https://doi.org/10.3390/ijms241210282
Chicago/Turabian StyleTong, Chaoqun, Jian Luo, Chaolu Xie, Junhong Wei, Guoqing Pan, Zeyang Zhou, and Chunfeng Li. 2023. "Characterization and Biological Activities of Melanin from the Medicinal Fungi Ophiocordyceps sinensis" International Journal of Molecular Sciences 24, no. 12: 10282. https://doi.org/10.3390/ijms241210282
APA StyleTong, C., Luo, J., Xie, C., Wei, J., Pan, G., Zhou, Z., & Li, C. (2023). Characterization and Biological Activities of Melanin from the Medicinal Fungi Ophiocordyceps sinensis. International Journal of Molecular Sciences, 24(12), 10282. https://doi.org/10.3390/ijms241210282







