Chitosan-Enriched Solution Blow Spun Poly(Ethylene Oxide) Nanofibers with Poly(Dimethylsiloxane) Hydrophobic Outer Layer for Skin Healing and Regeneration
Abstract
:1. Introduction
2. Results and Discussion
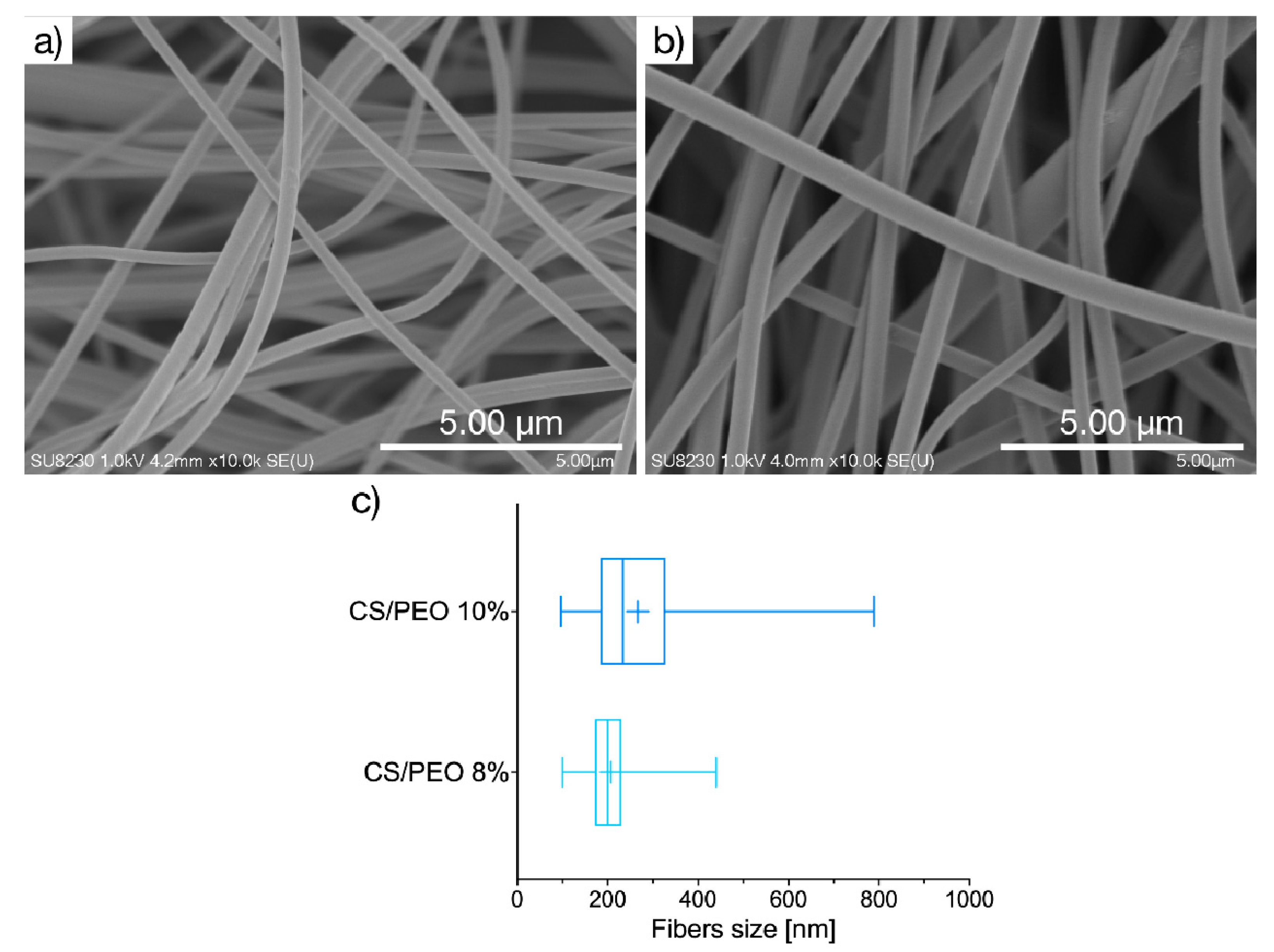
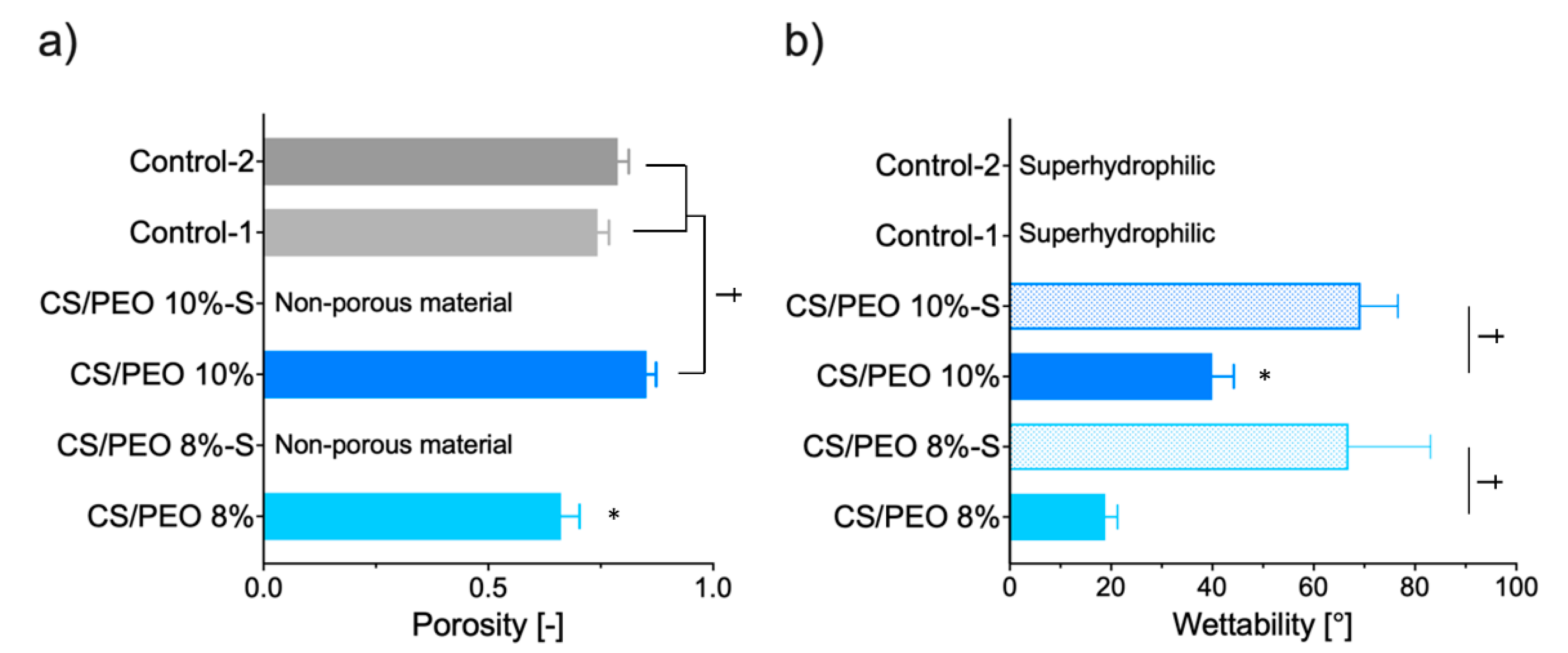
2.1. Physicochemical Characteristics
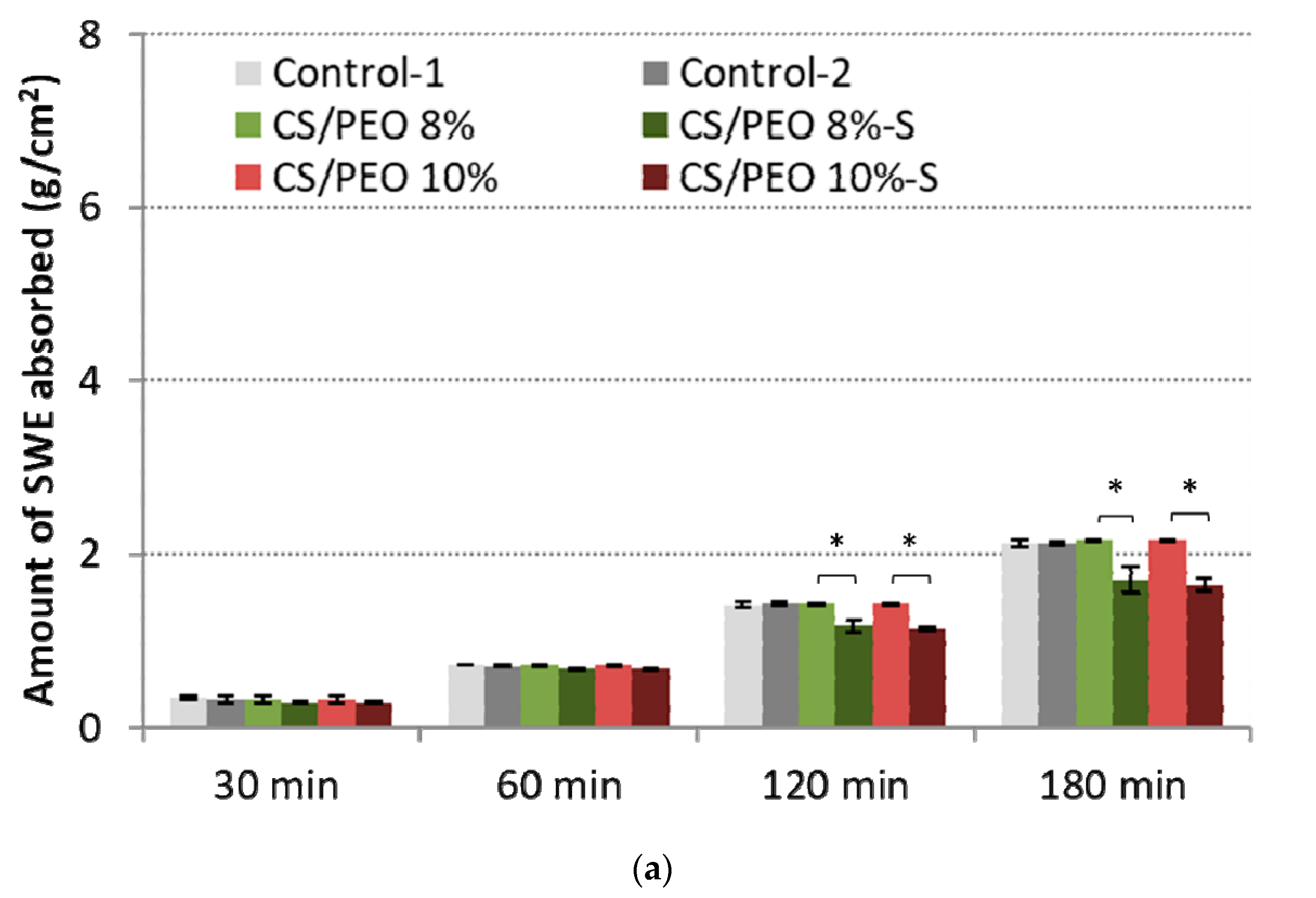
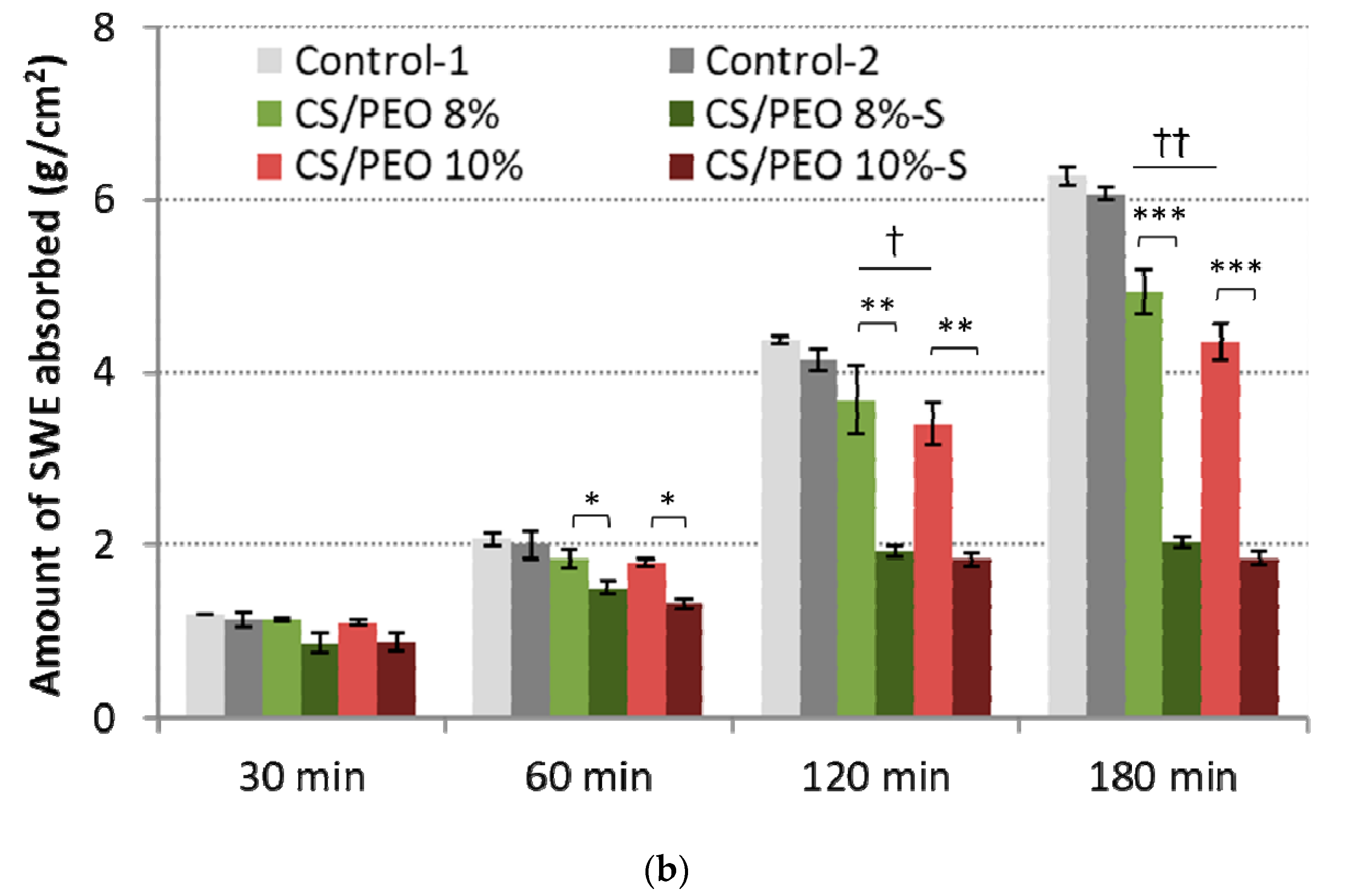
2.2. Mechanical Properties and Absorption Capacity
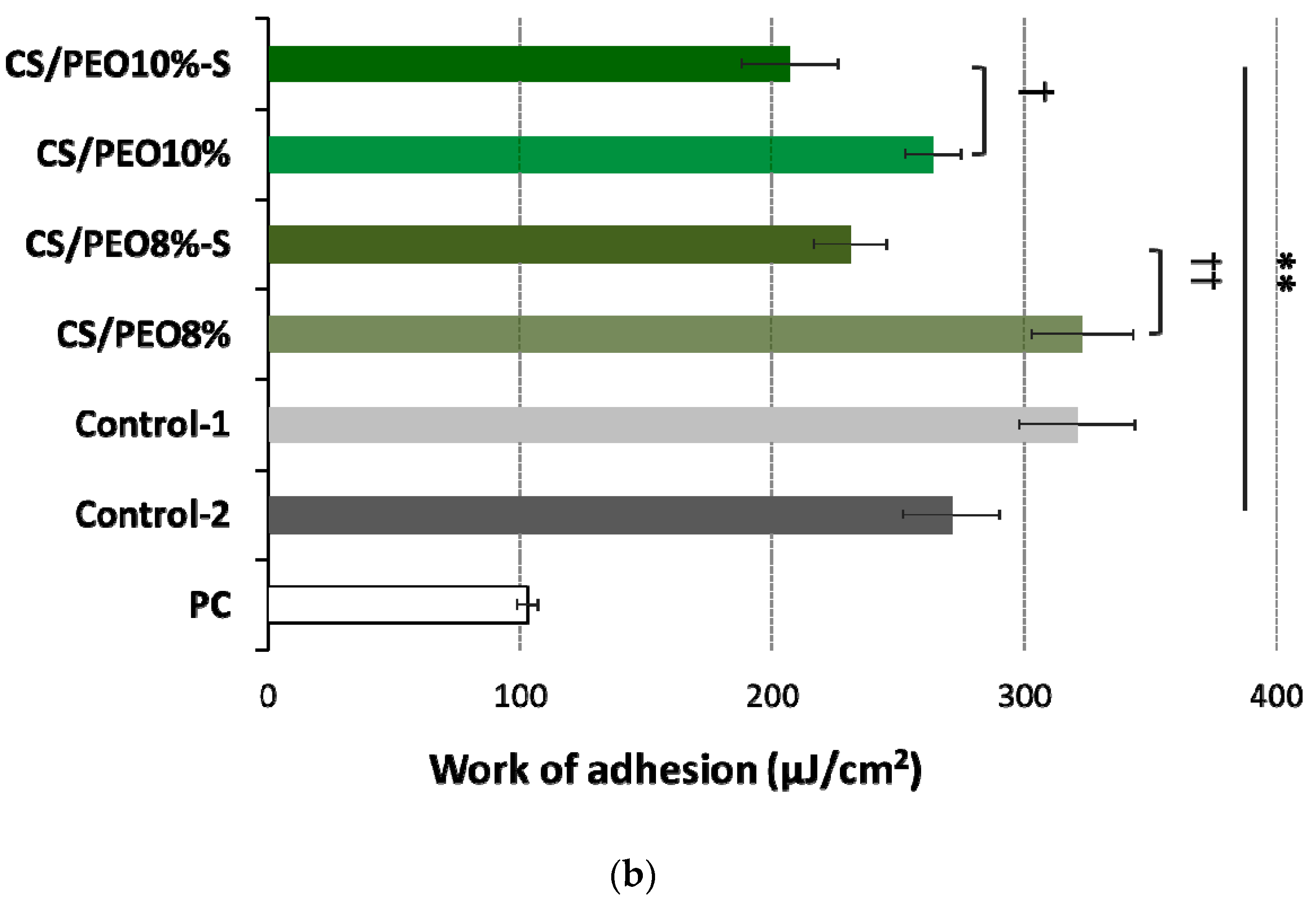
2.3. Bioadhesive Behavior
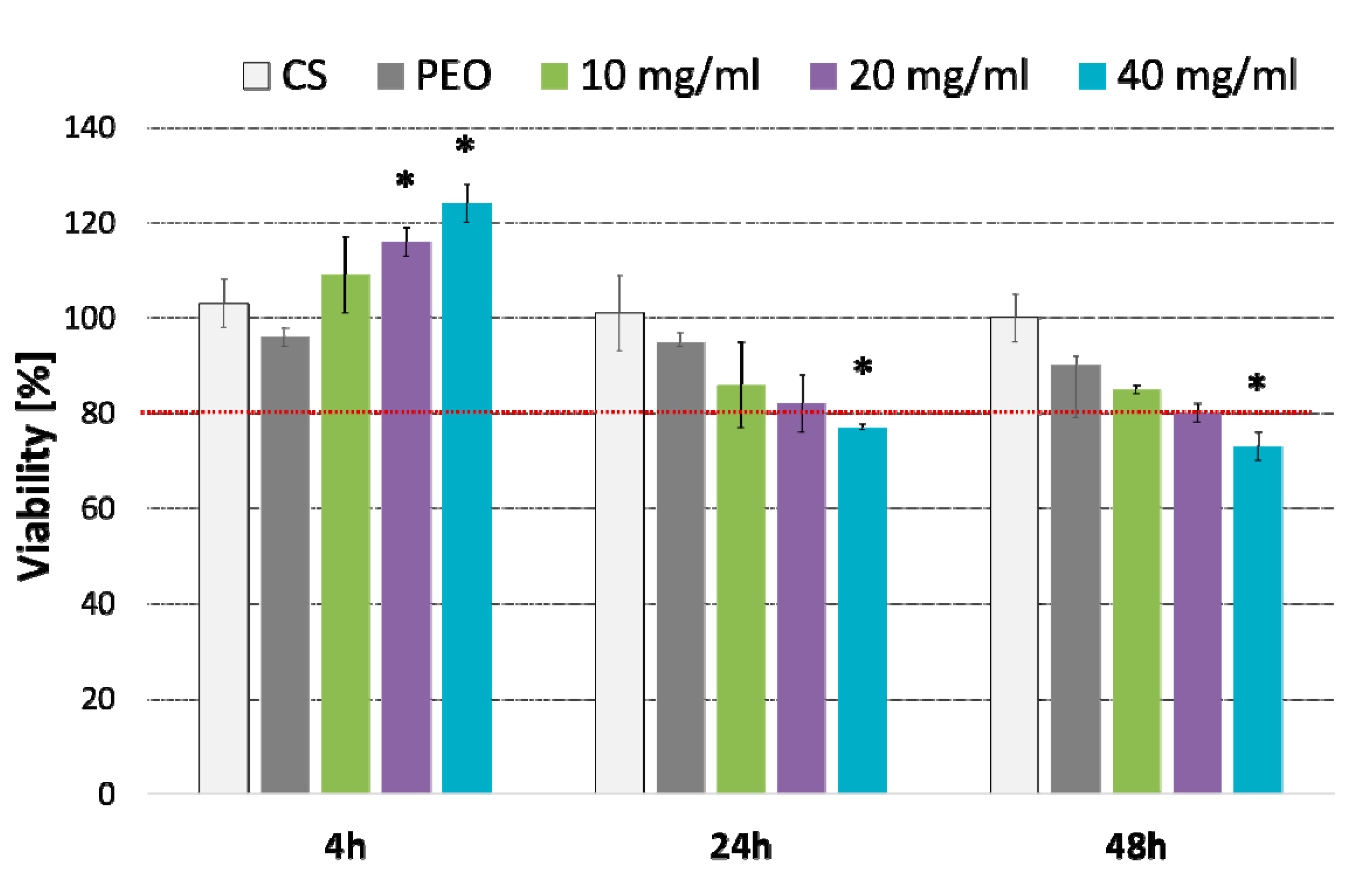
2.4. In Vitro Cytotoxicity Studies and Irritant Test with EpiDermTM
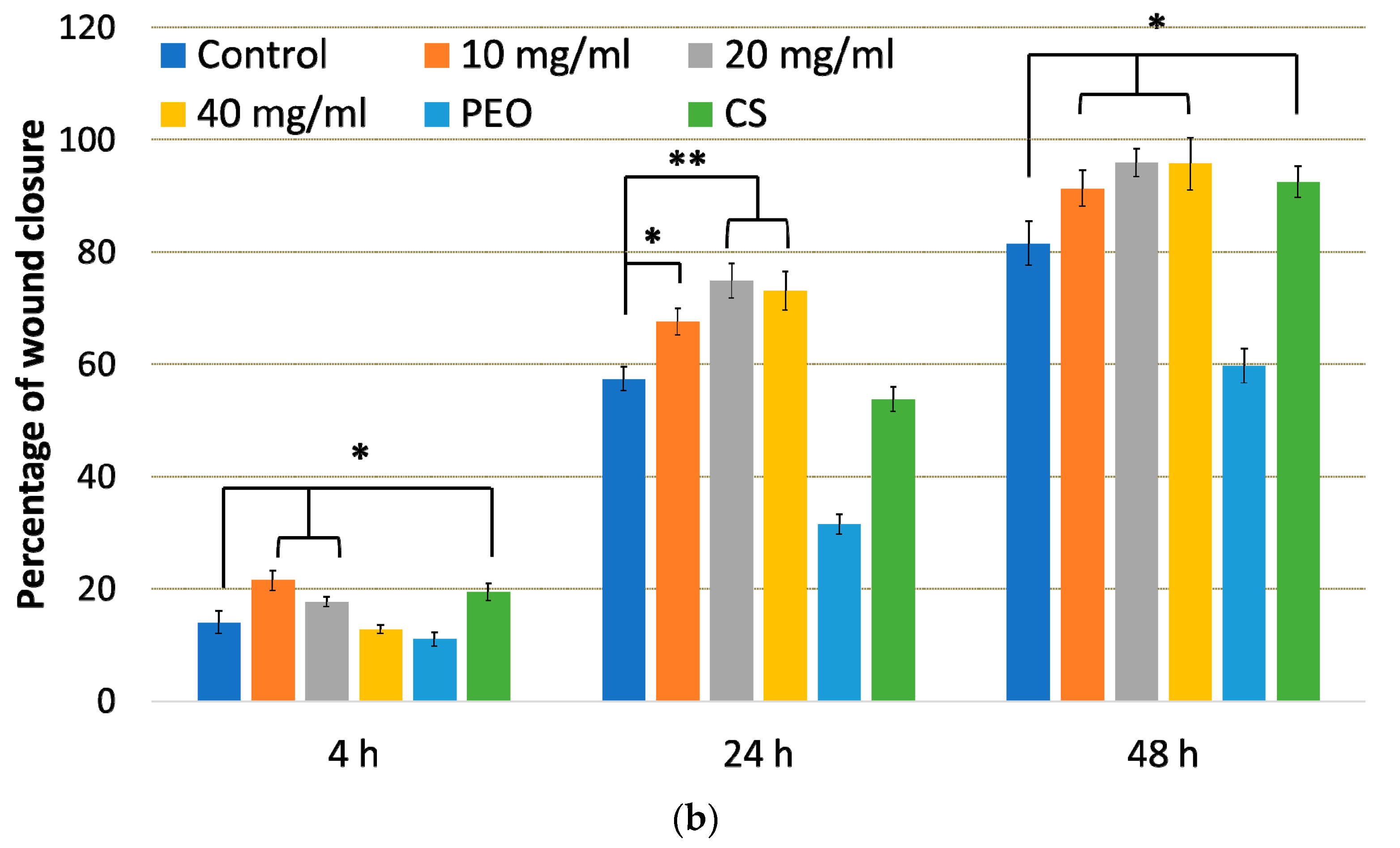
2.5. In Vitro Scratch Assay
3. Materials and Methods
3.1. Materials
3.2. Preparation of Polymer Blend Solutions for Blow Spinning
3.3. Solution Blow Spinning Technique
3.4. Scanning Electron Microscopy
3.5. Porosity
3.6. Wetting Properties
3.7. Attenuated Total Reflection Fourier Transformed Infrared Spectroscopy (ATR-FTIR)
3.8. Mechanical Characteristics
3.9. Simulate Wound Exudate Absorption Capacity
3.10. Bioadhesive Behavior in Contact with Excised Human Skin
3.11. Cell Culture and Treatment
3.12. Cytotoxicity Assays
3.12.1. MTT Assay
3.12.2. JC-1 Assay
3.12.3. EpiDerm™ Skin Irritation Test
3.13. In Vitro Scratch Assay
3.14. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Brabazon, D. Nanostructured Materials in Reference Module. In Materials Science and Materials Engineering; Hashmi, S., Ed.; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Jeevanandam, J.; Barhoum, A.; Chan, Y.S.; Alain Dufresne, A.; Danquah, M.K. Review on Nanoparticles and Nanostructured Materials: History, Sources, Toxicity and Regulations. Beilstein J. Nanotechnol. 2018, 9, 1050–1074. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nemati, S.; Kim, S.J.; Shin, Y.M.; Shin, H. Current Progress in Application of Polymeric Nanofibers to Tissue Engineering. Nano Converg. 2019, 6, 36–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, M.; Duan, X.P.; Li, Y.M.; Yang, D.P.; Long, Y.Z. Electrospun Nanofibers for Wound Healing. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 76, 1413–1423. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ding, B.; Li, B. Biomimetic electrospun nanofibrous structures for tissue engineering. Mater. Today (Kidlington) 2013, 16, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.M.; Zhang, Y.Z.; Kotaki, M.; Ramakrishna, S. A Review on Polymer Nanofibers by Electrospinning and their Applications in Nanocomposites. Compos. Sci. Technol. 2003, 63, 2223–2253. [Google Scholar] [CrossRef]
- Kopeć, K.; Wojasiński, M.; Ciach, T. Superhydrophilic Polyurethane/Polydopamine Nanofibrous Materials Enhancing Cell Adhesion for Application in Tissue Engineering. Int. J. Mol. Sci. 2020, 21, 6798. [Google Scholar] [CrossRef]
- Jia, C.; Li, L.; Song, J.; Li, Z.; Wu, H. Mass Production of Ultrafine Fibers by a Versatile Solution Blow Spinning Method. Acc. Mater. Res. 2021, 2, 432–446. [Google Scholar] [CrossRef]
- Daristotle, J.L.; Behrens, A.M.; Sandler, A.D.; Kofinas, P. A Review of the Fundamental Principles and Applications of Solution Blow Spinning. ACS Appl. Mater. Interfaces 2016, 8, 34951–34963. [Google Scholar] [CrossRef] [Green Version]
- Luo, X.; Li, L. Chitosan’s Wide Profile from Fibre to Fabrics: An Overview. In Chitin-Chitosan—Myriad Functionalities in Science and Technology; Dongre, R.S., Ed.; IntechOpen: London, UK, 2018; Available online: https://www.intechopen.com/chapters/60859 (accessed on 5 April 2022). [CrossRef] [Green Version]
- Jiménez-Gómez, C.P.; Cecilia, J.A. Chitosan: A Natural Biopolymer with a Wide and Varied Range of Applications. Molecules 2020, 25, 3981. [Google Scholar] [CrossRef]
- Sahariah, P.; Másson, M. Antimicrobial Chitosan and Chitosan Derivatives: A Review of the Structure-Activity Relationship. Biomacromolecules 2017, 18, 3846–3868. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, L.; Li, P.; Hao, X.; Yang, X.; Xi, G.; Liu, W.; Feng, Y.; He, H.; Shi, C. Polysaccharide Based Hemostatic Strategy for Ultrarapid Hemostasis. Macromol. Biosci. 2020, 20, e1900370–e1900384. [Google Scholar] [CrossRef] [PubMed]
- Cheng, F.; Gao, J.; Wang, L.; Hu, X. Composite Chitosan/Poly(Ethylene Oxide) Electrospun Nanofibrous Mats as Novel Wound Dressing Matrixes for the Controlled Release of Drugs. J. Appl. Polym. Sci. 2015, 132, 42060. [Google Scholar] [CrossRef]
- Szymańska, E.; Czarnomysy, R.; Jacyna, J.; Basa, A.; Wilczewska, A.Z.; Markuszewski, M.; Winnicka, K. Could Spray-Dried Microbeads with Chitosan Glutamate Be Considered as Promising Vaginal Microbicide Carriers? The Effect of Process Variables on the In Vitro Functional and Physicochemical Characteristics. Int. J. Pharm. 2019, 568, 118558–118570. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I.H.; An, X.; Dai, L.; Li, H.; Khan, A.; Ni, Y. Chitosan-Based Polymer Matrix for Pharmaceutical Excipients and Drug Delivery. Curr. Med. Chem. 2019, 26, 2502–2513. [Google Scholar] [CrossRef] [PubMed]
- Tao, F.; Cheng, Y.; Shi, X.; Zheng, H.; Du, Y.; Xiang, W.; Deng, H. Applications of Chitin and Chitosan Nanofibers in Bone Regenerative Engineering. Carbohydr. Polym. 2020, 230, 115658. [Google Scholar] [CrossRef]
- Akpan, E.I.; Gbeneborn, O.P.; Adeosun, S.O.; Cletus, O. Chapter Chitin and Chitosan Composites for Bone Tissue Regeneration. In Handbook of Chitin and Chitosan; Chitin and Chitosan Based Polymer Materials for Various Applications; Elsevier: Amsterdam, The Netherlands, 2020; Volume 3, pp. 499–553. [Google Scholar]
- Buschmann, M.D.; Merzouki, A.; Lavertu, M.; Thibault, M.; Jean, M.; Darras, V. Chitosans for Delivery of Nucleic Acids. Adv. Drug Del. Rev. 2013, 65, 1234–1270. [Google Scholar] [CrossRef] [PubMed]
- Szymańska, E.; Sosnowska, K.; Miltyk, W.; Rusak, M.; Basa, A.; Winnicka, K. The Effect of Beta-Glycerophosphate Crosslinking on Chitosan Cytotoxicity and Physicochemical Properties of Hydrogels for Vaginal Application. Polymers 2015, 7, 2223–2244. [Google Scholar] [CrossRef] [Green Version]
- Szymańska, E.; Szekalska, M.; Czarnomysy, R.; Lavrič, Z.; Srčič, S.; Miltyk, W.; Winnicka, K. Novel Spray Dried Glycerol 2-Phosphate Cross-Linked Chitosan Microparticulate Vaginal Delivery System-Development, Characterization and Cytotoxicity Studies. Mar. Drugs 2016, 14, 174. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.H.; Wang, L.S.; Chen, S.Y.; Huang, M.C.; Li, Y.H.; Lin, Y.C.; Chen, P.F.; Shaw, J.F.; Huang, K.S. Microfluidic Assisted Synthesis of Silver Nanoparticle–Chitosan Composite Microparticles for Antibacterial Applications. Int. J. Pharm. 2016, 510, 493–500. [Google Scholar] [CrossRef] [Green Version]
- Rowe, R.C.; Sheskey, P.J.; Quinn, M.E. Handbook of Pharmaceutical Excipients, 6th ed.; Pharmaceutical Press: London, UK, 2009; pp. 522–524. [Google Scholar]
- Bahlouli, M.I.; Bekkour, K.; Benchabane, A.; Hemar, Y.; Nemdili, A. The Effect of Temperature on the Rheological Behavior of Polyethylene Oxide (Peo) Solutions. Appl. Rheol. 2013, 23, 13435–13450. [Google Scholar]
- Grothe, T.; Brikmann, J.; Ehrmann, A. PEO as Spinnable Polymer and Spinning Agent for Non-Spinnable Materials (Conference paper). In Proceedings of the International Textile Conference, Dresden, Germany, 24–25 November 2016. [Google Scholar]
- Klossner, R.R.; Quenn, H.A.; Coughlin, A.J.; Krause, W.E. Correlation of Chitosan’s Rheological Properties and Its Ability to Electrospin. Biomacromolecules 2008, 9, 2947–2953. [Google Scholar] [CrossRef] [PubMed]
- Kuntzler, S.G.; Costa, J.A.V.; de Morais, M.G. Development of Electrospun Nanofibers Containing Chitosan/PEO Blend and Phenolic Compounds with Antibacterial Activity. Int. J. Biol. Macromol. 2018, 117, 800–806. [Google Scholar] [CrossRef] [PubMed]
- Mengistu Lemma, S.; Bossard, F.; Rinaudo, M. Preparation of Pure and Stable Chitosan Nanofibers by Electrospinning in the Presence of Poly(Ethylene Oxide). Int. J. Mol. Sci. 2016, 17, 1790. [Google Scholar] [CrossRef] [Green Version]
- Liu, R.; Xu, X.; Zhuang, X.; Cheng, B. Solution Blowing of Chitosan/PVA Hydrogel Nanofiber Mat. Carbohydr. Polym. 2014, 101, 1116–1121. [Google Scholar] [CrossRef]
- Bienek, D.R.; Hoffman, K.M.; Tutak, W. Blow-Spun Chitosan/PEG/PLGA Nanofibers as a Novel Tissue Engineering Scaffold with Antibacterial Properties. J. Mater. Sci. Mater. Med. 2016, 27, 146. [Google Scholar] [CrossRef]
- Pucić, I.; Jurkin, T. FTIR Assessment of Poly(Ethylene Oxide) Irradiated in Solid State, Melt and Aqueous Solution. Radiat. Phys. Chem. 2012, 81, 1426–1429. [Google Scholar] [CrossRef]
- Tarus, B.K.; Fadel, N.; Al-Oufy, A.; El-Messiry, M. Investigation of mechanical propertiesof electrospun poly (vinyl chloride) polymer nanoengineered composite. J. Eng. Fibers Fabr. 2020, 15, 1558925020982569. [Google Scholar]
- Okan, D.; Woo, K.; Ayello, E.A.; Sibbald, G. The Role of Moisture Balance in Wound Healing. Adv. Skin Wound Care. 2007, 20, 39–53. [Google Scholar] [CrossRef]
- Rashid, T.U.; Gorga, R.E.; Krause, W.E. Mechanical Properties of Electrospun Fibers—A Critical Review. Adv. Eng. Mater. 2021, 23, 2100153. [Google Scholar] [CrossRef]
- Conte, A.A.; Sun, K.; Hu, X.; Beachley, V.Z. Effects of Fiber Density and Strain Rate on the Mechanical Properties of Electrospun Polycaprolactone Nanofiber Mats. Front. Chem. 2020, 8, 610–620. [Google Scholar] [CrossRef]
- Wong, S.C.; Baji, A.; Leng, S. Effect of Fiber Diameter on Tensile Properties of Electrospun Poly(caprolactone). Polymer 2008, 49, 4713–4722. [Google Scholar] [CrossRef]
- Nitti, P.; Gallo, N.; Natta, L.; Scalera, F.; Palazzo, B.; Sannino, A.; Gervaso, F. Influence of Nanofiber Orientation on Morphological and Mechanical Properties of Electrospun Chitosan Mats. J. Healthc. Eng. 2018, 2018, 3651480. [Google Scholar] [CrossRef]
- Niculescu, A.G.; Grumezescu, A.M. An Up-to-Date Review of Biomaterials Application in Wound Management. Polymers 2022, 14, 421. [Google Scholar] [CrossRef]
- Dabiri, G.; Damstetter, E.; Phillips, T. Choosing a Wound Dressing Based on Common Wound Characteristics. Adv. Wound Care 2016, 5, 32–41. [Google Scholar] [CrossRef] [Green Version]
- Grande, A.; Sivakumaran, D. Postoperative Wound Care: The Effect of Variability in Exudate Flow Rate on Dressing Uptake in an In Vitro Wound Model. In Proceedings of the Association of periOperative Registered Nurses (AORN) Surgical Conference & Expo, Anaheim, CA, USA, 2–6 April 2016. [Google Scholar]
- Rippon, M.; White, R.; Davies, P. Skin Adhesives and Their Role in Wound Dressings. Wounds UK 2007, 3, 76–85. [Google Scholar]
- Zillmer, R.; Agren, M.S.; Gottrup, F.; Karlsmark, T. Biophysical Effects of Repetitive Removal of Adhesive Dressings on Peri-Ulcer Skin. J. Wound Care 2006, 15, 187–191. [Google Scholar] [CrossRef]
- Singh, R.; Shitiz, K.; Singh, A. Chitin and Chitosan: Biopolymers for Wound Management. Int. Wound J. 2017, 14, 1276–1289. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, J.; Li, P.; Ning, N. The Effect of Chitosan in Wound Healing: A Systematic Review. Adv. Skin Wound Care 2021, 34, 262–266. [Google Scholar] [CrossRef]
- ISO 10993-5 Biological Evaluation of Medical Devices. Part 15: Tests for In Vitro Cytotoxicity. Int. Stand. ISO 10993-5 2009. Available online: https://www.iso.org/standard/36406.html (accessed on 14 March 2022).
- Kandárová, H.; Hayden, P.; Klausner, M.; Kubilus, J.; Kearney, P.; Sheasgreen, J. In Vitro Skin Irritation Testing: Improving the Sensitivity of the EpiDerm Skin Irritation Test Protocol. Altern. Lab. Anim. 2009, 37, 671–689. [Google Scholar] [CrossRef]
- Kandárová, H.; Hayden, P.; Klausner, M.; Kubilus, J.; Sheasgreen, J. An In Vitro Skin Irritation Test (SIT) Using the EpiDerm Reconstructed Human Epidermal (RHE) Model. J. Vis. Exp. 2009, 13, 1366. [Google Scholar] [CrossRef] [Green Version]
- EN ISO 10993-10:2013 Biological Evaluation of Medical Devices-Part 10: Tests for Irritation and Skin Sensitization (ISO 10993-10:2010). Available online: https://www.iso.org/standard/75279.html (accessed on 14 March 2022).
- Howling, G.I.; Dettmar, P.W.; Goddard, P.A.; Hampson, F.C.; Dornish, M.; Wood, E.J. The Effect of Chitin and Chitosan on the Proliferation of Human Skin Fibroblasts and Keratinocytes in Vitro. Biomaterials 2001, 22, 2959–2966. [Google Scholar] [CrossRef]
- Huang, J.; Ren, J.; Chen, G.; Li, Z.; Liu, Y.; Wang, G.; Wu, X. Tunable Sequential Drug Delivery System Based on Chitosan/Hyaluronic Acid Hydrogels and PLGA Microspheres for Management of Non-Healing Infected Wounds. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 89, 213–222. [Google Scholar] [CrossRef]
- Liang, C.C.; Park, A.Y.; Guan, J.L. In Vitro Scratch Assay: A Convenient and Inexpensive Method for Analysis of Cell Migration In Vitro. Nat. Protoc. 2007, 2, 329–333. [Google Scholar] [CrossRef] [Green Version]
- Bagheri, M.; Validi, M.; Gholipour, A.; Makvandi, P.; Sharifi, E. Chitosan Nanofiber Biocomposites for Potential Wound Healing Applications: Antioxidant Activity with Synergic Antibacterial Effect. Bioeng. Transl. Med. 2021, 7, e10254–e10269. [Google Scholar] [CrossRef]
- Salerno, S.; De Santo, M.P.; Drioli, E.; De Bartolo, L. Nano- and Micro-Porous Chitosan Membranes for Human Epidermal Stratification and Differentiation. Membranes 2021, 11, 394. [Google Scholar] [CrossRef]
- Morganti, P.; Fusco, A.; Paoletti, I.; Perfetto, B.; Del Ciotto, P.; Palombo, M.; Chianese, A.; Baroni, A.; Donnarumma, G. Anti-Inflammatory, Immunomodulatory, and Tissue Repair Activity on Human Keratinocytes by Green Innovative Nanocomposites. Materials 2017, 10, 843. [Google Scholar] [CrossRef] [Green Version]
- Czechowska-Biskup, R.; Jarosińska, D.; Rokita, B.; Ulański, P.; Rosiak, J.M. Determination Degree of Deacetylation of Chitosan: Comparison of Methods. Prog. Chem. Appl. Chitin Its Deriv. 2012, 17, 5–20. [Google Scholar]
- Bradford, C.; Freeman, R.; Percival, S.L. In Vitro Study of Sustained Antimicrobial Activity of a New Silver Alginate Dressing. J. Am. Col. Certif. Wound Spec. 2009, 1, 117–120. [Google Scholar] [CrossRef] [Green Version]
- Rinaudo, M.; Pavlov, G.; Desbrières, J. Influence of Acetic Acid Concentration on the Solubilization of Chitosan. Polymer 1999, 40, 7029–7032. [Google Scholar] [CrossRef]
- Czarnecka, K.; Wojasiński, M.; Ciach, T.; Sajkiewicz, P. Solution Blow Spinning of Polycaprolactone—Rheological Determination of Spinnability and the Effect of Processing Conditions on Fiber Diameter and Alignment. Materials 2021, 14, 1463. [Google Scholar] [CrossRef]
- Zhang, X.; Lv, J.; Yin, X.; Li, Z.; Lin, Q.; Zhu, L. Nanofibrous Polystyrene Membranes Prepared through Solution Blow Spinning with an Airbrush and the Facile Application in Oil Recovery. Appl. Phys. A 2018, 124, 362. [Google Scholar] [CrossRef]
- Potaś, J.; Szymańska, E.; Basa, A.; Hafner, A.; Winnicka, K. Tragacanth Gum/Chitosan Polyelectrolyte Complexes-Based Hydrogels Enriched with Xanthan Gum as Promising Materials for Buccal Application. Materials 2020, 14, 86. [Google Scholar] [CrossRef]
- Thomas, S.; Fram, P. The Development of a Novel Technique for Predicting the Exudate Handling Properties of Modern Wound Dressings. J. Tissue Viability 2001, 11, 145–160. [Google Scholar] [CrossRef]
- Szymańska, E.; Krzyżowska, M.; Cal, K.; Mikolaszek, B.; Tomaszewski, J.; Wołczyński, S.; Winnicka, K. Potential of Mucoadhesive Chitosan Glutamate Microparticles as Microbicide Carriers—Antiherpes Activity and Penetration Behavior Across the Human Vaginal Epithelium. Drug Deliv. 2021, 28, 2278–2288. [Google Scholar] [CrossRef]
- Khorshid, F.A. The Effect of the Medium Viscosity on the Cells Morphology in Reaction of Cells to Topography-I. Proc. 2nd Saudi Sci. Conf. 2005, 1, 67–98. [Google Scholar]
- Carmichael, J.; DeGraff, W.G.; Gazdar, A.F.; Minna, J.D.; Mitchell, J.B. Evaluation of a Tetrazolium-Based Semiautomated Colorimetric Assay: Assessment of Chemosensitivity Testing. Cancer Res. 1987, 47, 936–942. [Google Scholar]
- Mattek EpiDerm Protocole. Available online: https://www.mattek.com/wp-content/uploads/EPI-200-SIT-Skin-Irritation-MK-24-007-0023.pdf (accessed on 16 February 2022).
- OECD Guidelines for the Testing of Chemicals, In Vitro Skin Irritation: Reconstructed Human Epidermis Test Method, OECD/OCDE 439. Available online: https://www.oecd.org/env/ehs/testing/tg439-revised-in-vitro-skin-irritation-reconstructed-human-epidermis-test-method.pdf (accessed on 16 February 2022).





| Sample | Viability ± S.D. (%) | Coefficient of Variance (%) | In Vivo Prediction |
|---|---|---|---|
| PBS (negative control) | 100.0 ± 7.0 | 7.0 | Non-irritant |
| 5% SDS (positive control) | 2.5 ± 0.3 | 10.5 | Irritant |
| CS/PEO 10% | 100.2 ± 2.2 | 2.2 | Non-irritant |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szymańska, E.; Wojasiński, M.; Czarnomysy, R.; Dębowska, R.; Łopianiak, I.; Adasiewicz, K.; Ciach, T.; Winnicka, K. Chitosan-Enriched Solution Blow Spun Poly(Ethylene Oxide) Nanofibers with Poly(Dimethylsiloxane) Hydrophobic Outer Layer for Skin Healing and Regeneration. Int. J. Mol. Sci. 2022, 23, 5135. https://doi.org/10.3390/ijms23095135
Szymańska E, Wojasiński M, Czarnomysy R, Dębowska R, Łopianiak I, Adasiewicz K, Ciach T, Winnicka K. Chitosan-Enriched Solution Blow Spun Poly(Ethylene Oxide) Nanofibers with Poly(Dimethylsiloxane) Hydrophobic Outer Layer for Skin Healing and Regeneration. International Journal of Molecular Sciences. 2022; 23(9):5135. https://doi.org/10.3390/ijms23095135
Chicago/Turabian StyleSzymańska, Emilia, Michał Wojasiński, Robert Czarnomysy, Renata Dębowska, Iwona Łopianiak, Kamil Adasiewicz, Tomasz Ciach, and Katarzyna Winnicka. 2022. "Chitosan-Enriched Solution Blow Spun Poly(Ethylene Oxide) Nanofibers with Poly(Dimethylsiloxane) Hydrophobic Outer Layer for Skin Healing and Regeneration" International Journal of Molecular Sciences 23, no. 9: 5135. https://doi.org/10.3390/ijms23095135
APA StyleSzymańska, E., Wojasiński, M., Czarnomysy, R., Dębowska, R., Łopianiak, I., Adasiewicz, K., Ciach, T., & Winnicka, K. (2022). Chitosan-Enriched Solution Blow Spun Poly(Ethylene Oxide) Nanofibers with Poly(Dimethylsiloxane) Hydrophobic Outer Layer for Skin Healing and Regeneration. International Journal of Molecular Sciences, 23(9), 5135. https://doi.org/10.3390/ijms23095135









