Cone Photoreceptor Loss in Light-Damaged Albino Rats
Abstract
:1. Introduction
2. Results
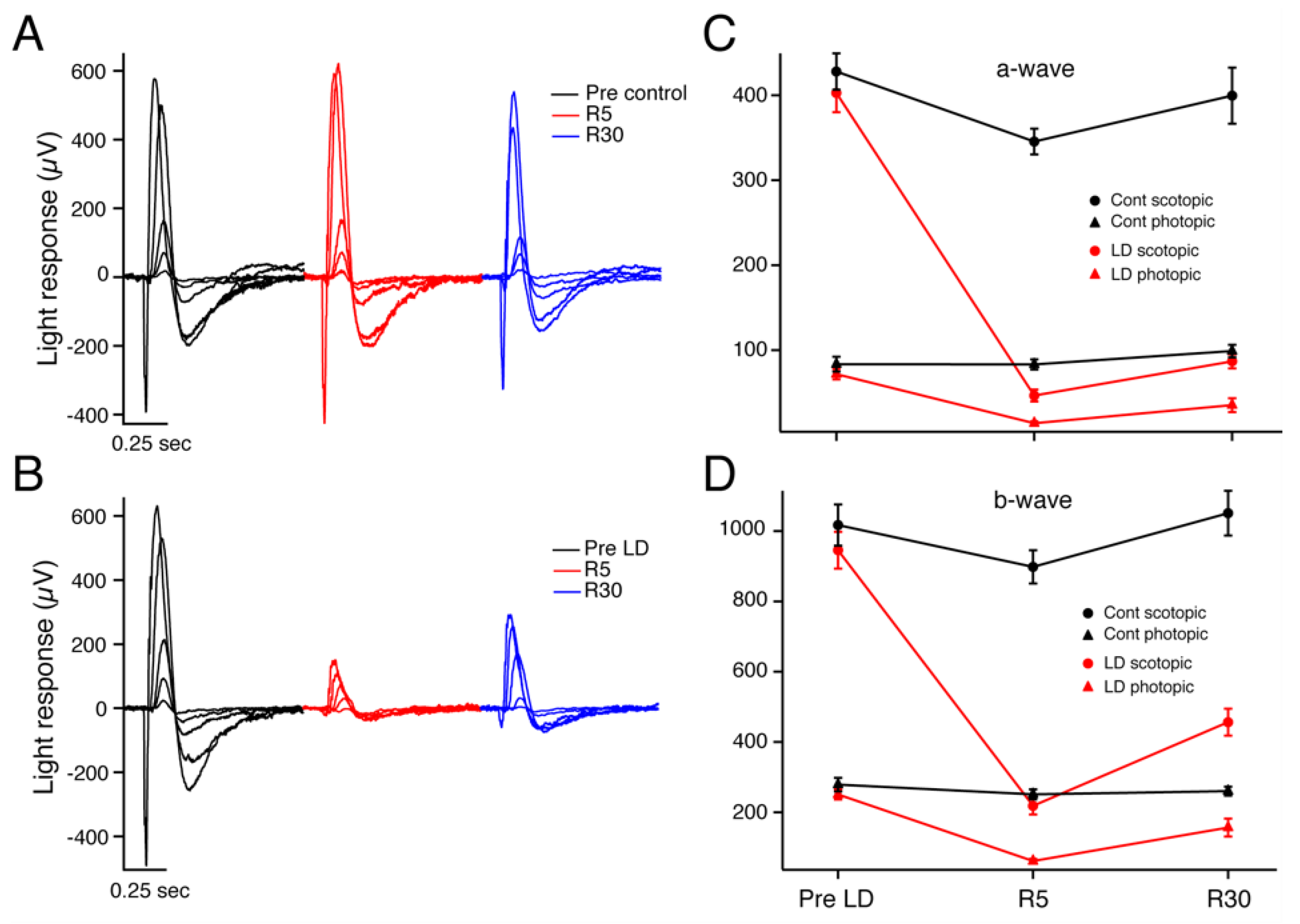
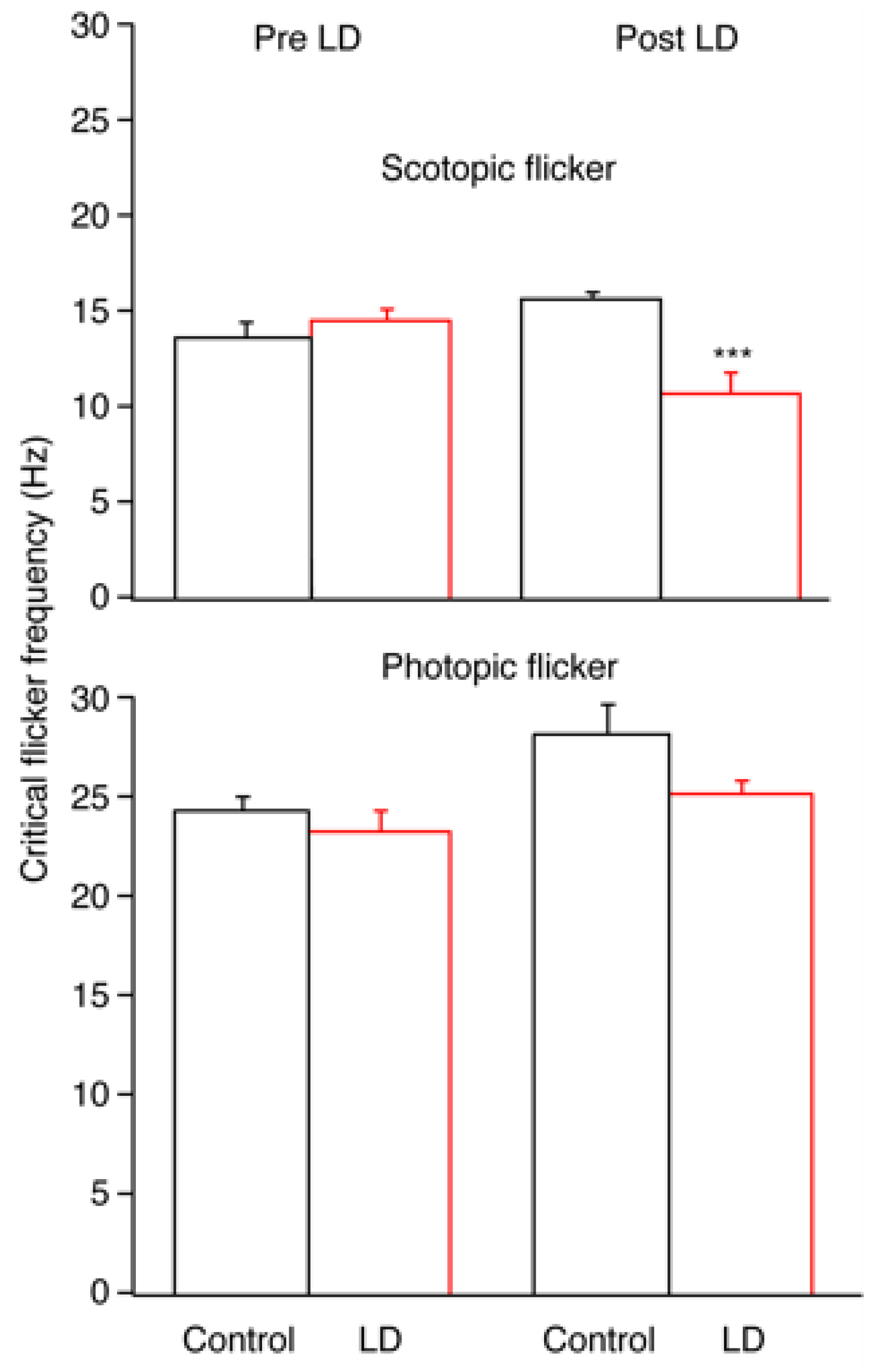
2.1. Electroretinography
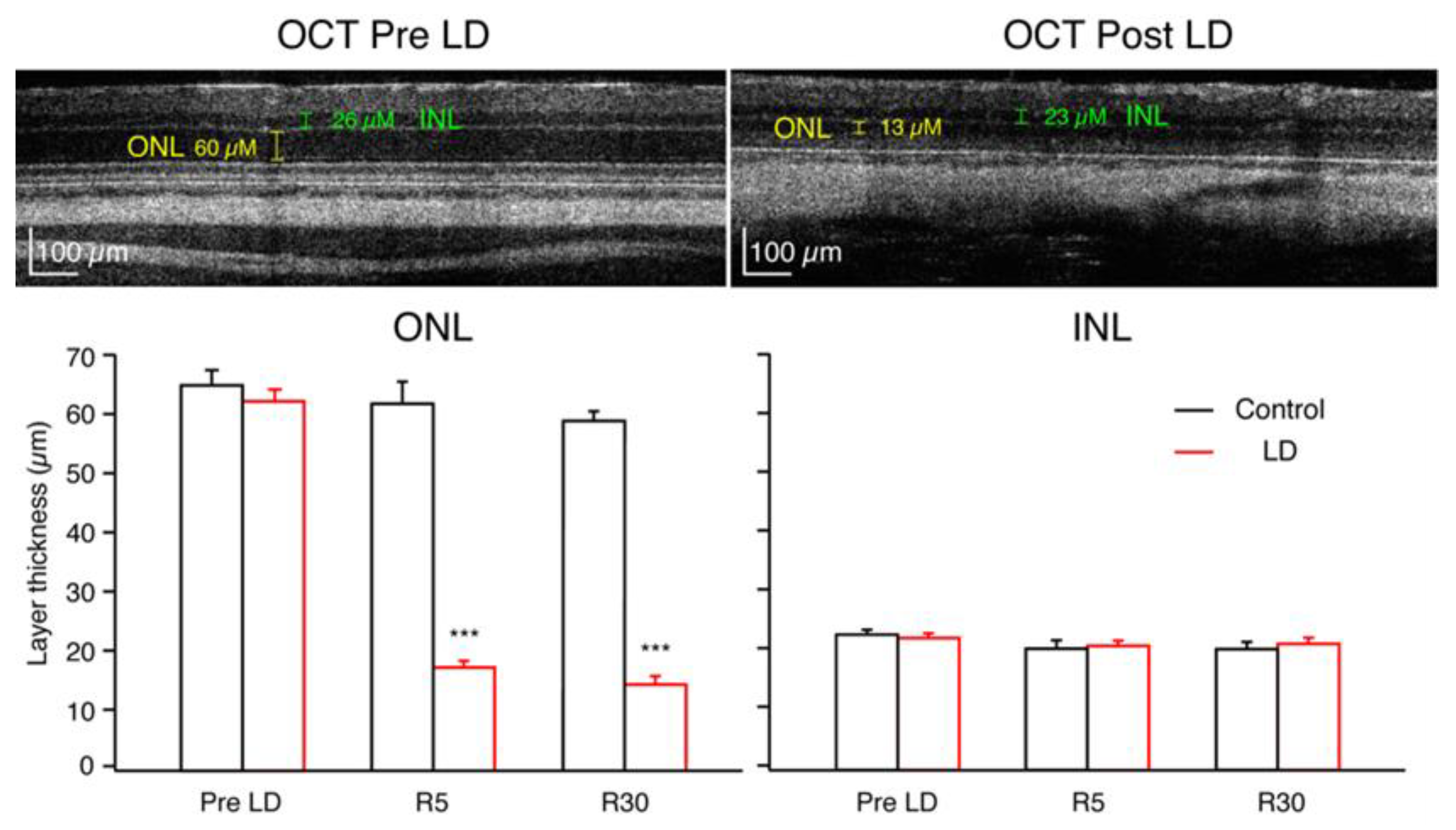
2.2. Optical Coherence Tomography
2.3. Immunohistochemistry
3. Discussion
3.1. Electrophysiologic Assessments of Visual Function
3.2. In Vivo and In Vitro Imaging of Retinal Layers
4. Materials and Methods
4.1. Light Damage
4.2. Electroretinography
4.3. Optical Coherence Tomography
4.4. Histology
4.5. Immunohistochemistry
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Gal, A.; Li, Y.; Thompson, D.A.; Weir, J.; Orth, U.; Jacobson, S.G.; Apfelstedt-Sylvia, E.; Vollrath, D. Mutations in MERTK, the human orthologue of the RCS rat retinal dystrophy gene, cause retinitis pigmentosa. Nat. Genet. 2000, 26, 270–271. [Google Scholar] [CrossRef] [PubMed]
- Mohand-Said, S.; Hicks, D.; Leveillard, T.; Picaud, S.; Porto, F.; Sahel, J.A. Rod-cone interactions: Developmental and clinical significance. Prog. Retin. Eye Res. 2001, 20, 451–467. [Google Scholar] [CrossRef]
- Delyfer, M.N.; Leveillard, T.; Mohand-Said, S.; Hicks, D.; Picaud, S.; Sahel, J.A. Inherited retinal degenerations: Therapeutic prospects. Biol. Cell 2004, 96, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.Y. Gene mutations in Retinitis Pigmentosa and their clinical implications. Clin. Chim. Acta 2005, 351, 5–16. [Google Scholar] [CrossRef]
- Mohand-Said, S.; Hicks, D.; Simonutti, M.; Tran-Minh, D.; Deudon-Combe, A.; Dreyfus, H.; Silverman, M.S.; Ogilvie, J.M.; Tenkova, T.; Sahel, J.A. Photoreceptor transplants increase host cone survival in the retinal degeneration (rd) mouse. Ophthalmic Res. 1997, 29, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Mohand-Said, S.; Deudon-Combe, A.; Hicks, D.; Simonutti, M.; Forster, V.; Fintz, A.-C.; Leveillard, T.; Dreyfus, H.; Sahel, J.-A. Normal retina releases a diffusible factor stimulating cone survival in the retinal degeneration mouse. Proc. Natl. Acad. Sci. USA 2004, 95, 8357–8362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sahel, J.A. Saving cone cells in hereditary rod diseases: A possible role for rod-derived cone viability factor (RdCVF) therapy. Retina 2005, 25, S38–S39. [Google Scholar] [CrossRef] [PubMed]
- Di Pierdomenico, J.; Martínez-Vacas, A.; Hernández-Muñoz, D.; Gómez-Ramírez, A.M.; Valiente-Soriano, F.J.; Agudo-Barriuso, M.; Vidal-Sanz, M.; Villegas-Pérez, M.P.; García-Ayuso, D. Coordinated Intervention of Microglial and Müller Cells in Light-Induced Retinal Degeneration. Investig. Opthalmology Vis. Sci. 2020, 61, 47. [Google Scholar] [CrossRef] [Green Version]
- Xu, S.; Zhang, P.; Zhang, M.; Wang, X.; Li, G.; Xu, G.; Ni, Y. Synaptic changes and the response of microglia in a light-induced photoreceptor degeneration model. Mol. Vis. 2021, 27, 206–220. [Google Scholar]
- Marie, M.; Forster, V.; Fouquet, S.; Berto, P.; Barrau, C.; Ehrismann, C.; Sahel, J.-A.; Tessier, G.; Picaud, S. Phototoxic damage to cone photoreceptors can be independent of the visual pigment: The porphyrin hypothesis. Cell Death Dis. 2020, 11, 711. [Google Scholar] [CrossRef]
- Keeler, C. Retinal degeneration in the mouse is rodless retina. J. Hered. 1966, 57, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Noell, W.K.; Walker, V.S.; Kang, B.S.; Berman, S. Retinal damage by light in rats. Investig. Ophthalmol. 1966, 5, 450–473. [Google Scholar]
- D’Cruz, P.M.; Yasumura, D.; Weir, J.; Matthes, M.T.; Abderrahim, H.; LaVail, M.M.; Vollrath, D. Mutation of the receptor tyrosine kinase gene Mertk in the retinal dystrophic RCS rat. Hum. Mol. Genet. 2000, 9, 645–651. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Polosa, A.; Bessaklia, H.; Lachapelle, P. Strain Differences in Light-Induced Retinopathy. PLoS ONE 2016, 11, e0158082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miralles de Imperial-Ollero, J.A.; Gallego-Ortega, A.; Ortín-Martínez, A.; Villegas-Pérez, M.P.; Valiente-Soriano, F.J.; Vidal-Sanz, M. Animal Models of LED-Induced Phototoxicity. Short- and Long-Term in vivo and ex vivo Retinal Alterations. Life 2021, 11, 1137. [Google Scholar] [CrossRef] [PubMed]
- Krigel, A.; Berdugo, M.; Picard, E.; Levy-Boukris, R.; Jaadane, I.; Jonet, L.; Dernigoghossian, M.; Andrieu-Soler, C.; Torriglia, A.; Behar-Cohen, F. Light-induced retinal damage using different light sources, protocols and rat strains reveals LED phototoxicity. Neuroscience 2016, 339, 296–307. [Google Scholar] [CrossRef] [Green Version]
- Turnock, S.; Gerbeix, C.; Thirion-Delalande, C.; Pearson, N.; Forster, R. Assessment of phototoxicity in pigmented Long-Evans rat: Sparfloxacin and 8-methoxypsoralen. Regul. Toxicol. Pharmacol. 2018, 92, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Ranchon, I.; Chen, S.; Alvarez, K.; Anderson, R. Systemic administration of phenyl-N-tert-butylnitrone protects the retina from light damage. Investig. Ophthalmol. Vis. Sci. 2001, 42, 1375–1379. [Google Scholar]
- Rubin, G.R.; Wen, Y.; Loop, M.S.; Kraft, T.W. ERG and Behavioral CFF in Light-Damaged Albino Rats. Int. J. Mol. Sci. 2022; in press. [Google Scholar]
- Anderson, K.V.; O’Steen, W.K. Black-white and pattern discrimination in rats without photoreceptors. Exp. Neurol. 1972, 34, 446–454. [Google Scholar] [CrossRef]
- Bennett, M.H.; Dyer, R.F.; Dunn, J.D. Visual dysfuction after long-term continuous light exposure. Exp. Neurol. 1973, 40, 652–660. [Google Scholar] [CrossRef]
- Bennett, M.H.; Dyer, R.F.; Dunn, J.D. Visual-deficit following long-term continuous light exposure. Exp. Neurol. 1973, 38, 80–89. [Google Scholar] [CrossRef]
- Sugawara, T.; Sieving, P.A.; Bush, R.A. Quantitative relationship of the scotopic and photopic ERG to photoreceptor cell loss in light damaged rats. Exp. Eye Res. 2000, 70, 693–705. [Google Scholar] [CrossRef] [PubMed]
- Riccitelli, S.; Di Paolo, M.; Ashley, J.; Bisti, S.; Di Marco, S. The Timecourses of Functional, Morphological, and Molecular Changes Triggered by Light Exposure in Sprague-Dawley Rat Retinas. Cells 2021, 10, 1561. [Google Scholar] [CrossRef] [PubMed]
- Michon, J.J.; Li, Z.L.; Shioura, N.; Anderson, R.J.; Tso, M.O. A comparative study of methods of photoreceptor morphometry. Investig. Ophthalmol. Vis. Sci. 1991, 32, 280–284. [Google Scholar]
- Miralles de Imperial-Ollero, J.A.; Gallego-Ortega, A.; Norte-Muñoz, M.; Di Pierdomenico, J.; Bernal-Garro, J.M.; Valiente-Soriano, F.J.; Vidal-Sanz, M. Short- and Long-Term Study of the Impact of Focal Blue Light-Emitting Diode-Induced Phototoxicity in Adult Albino Rats. Int. J. Mol. Sci. 2021, 22, 9742. [Google Scholar] [CrossRef] [PubMed]
- Montalbán-Soler, L.; Alarcón-Martínez, L.; Jiménez-López, M.; Salinas-Navarro, M.; Galindo-Romero, C.; Bezerra de Sá, F.; García-Ayuso, D.; Avilés-Trigueros, M.; Vidal-Sanz, M.; Agudo-Barriuso, M.; et al. Retinal compensatory changes after light damage in albino mice. Mol. Vis. 2012, 18, 675–693. [Google Scholar]
- Swinkels, D.; Das, Y.; Kocherlakota, S.; Vinckier, S.; Wever, E.; van Kampen, A.H.C.; Vaz, F.M.; Baes, M. Cell Type-Selective Loss of Peroxisomal β-Oxidation Impairs Bipolar Cell but Not Photoreceptor Survival in the Retina. Cells 2022, 11, 161. [Google Scholar] [CrossRef]
- Rao, A.; Dallman, R.; Henderson, S.; Chen, C.K. Gbeta5 is required for normal light responses and morphology of retinal ON-bipolar cells. J. Neurosci. 2007, 27, 14199–14204. [Google Scholar] [CrossRef] [Green Version]
- Burkhardt, D.A.; Fahey, P.K.; Sikora, M.A. Retinal bipolar cells: Contrast encoding for sinusoidal modulation and steps of luminance contrast. Vis. Neurosci. 2004, 21, 883–893. [Google Scholar] [CrossRef]
- Penn, J.S.; Williams, T.P. Photostasis: Regulation of daily photon-catch by rat retinas in response to various cyclic illuminances. Exp. Eye Res. 1986, 43, 915–928. [Google Scholar] [CrossRef]
- Wen, Y.E.; Niculescu, D.M.; Wotring, V.E.; Kraft, T.W. Patho-Physiology of Rod Photoreceptors Under Stress -Retinal Degeneration Reshape the Light Responses of Surviving Rod Photoreceptors. Investig. Ophthalmol. Vis. Sci. 2009, 50, 1046. [Google Scholar]
- Li, Q.; Timmers, A.M.; Hunter, K.; Gonzalez-Pola, C.; Lewin, A.S.; Reitze, D.H.; Hauswirth, W. Noninvasive imaging by optical coherence tomography to monitor retinal degeneration in the mouse. Investig. Ophthalmol. Vis. Sci. 2001, 42, 2981–2989. [Google Scholar]
- Grytz, R.; El Hamdaoui, M.; Fuchs, P.A.; Fazio, M.A.; McNabb, R.P.; Kuo, A.N.; Girkin, C.A.; Samuels, B.C. Nonlinear distortion correction for posterior eye segment optical coherence tomography with application to tree shrews. Biomed. Opt. Express 2022, 13, 1070. [Google Scholar] [CrossRef]
- Ortín-Martínez, A.; Nadal-Nicolás, F.M.; Jiménez-López, M.; Alburquerque-Béjar, J.J.; Nieto-López, L.; García-Ayuso, D.; Villegas-Pérez, M.P.; Vidal-Sanz, M.; Agudo-Barriuso, M. Number and distribution of mouse retinal cone photoreceptors: Differences between an albino (Swiss) and a pigmented (C57/BL6) strain. PLoS ONE 2014, 9, e102392. [Google Scholar] [CrossRef]
- Ortín-Martínez, A.; Valiente-Soriano, F.J.; García-Ayuso, D.; Alarcón-Martínez, L.; Jiménez-López, M.; Bernal-Garro, J.M.; Nieto-López, L.; Nadal-Nicolás, F.M.; Villegas-Pérez, M.P.; Wheeler, L.A.; et al. A novel in vivo model of focal light emitting diode-induced cone-photoreceptor phototoxicity: Neuroprotection afforded by brimonidine, BDNF, PEDF or bFGF. PLoS ONE 2014, 9, e113798. [Google Scholar] [CrossRef]
- Wu, J.; Seregard, S.; Algvere, P.V. Photochemical damage of the retina. Surv. Ophthalmol. 2006, 51, 461–481. [Google Scholar] [CrossRef]
- Valiente-Soriano, F.J.; Ortín-Martínez, A.; Di Pierdomenico, J.; García-Ayuso, D.; Gallego-Ortega, A.; de Imperial-Ollero, J.A.M.; Jiménez-López, M.; Villegas-Pérez, M.P.; Wheeler, L.A.; Vidal-Sanz, M. Topical Brimonidine or Intravitreal BDNF, CNTF, or bFGF Protect Cones against Phototoxicity. Transl. Vis. Sci. Technol. 2019, 8, 36. [Google Scholar] [CrossRef] [Green Version]
- Curcio, C.A.; Medeiros, N.E.; Millican, C.L. Photoreceptor loss in age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 1996, 37, 1236–1249. [Google Scholar]
- Nigalye, A.K.; Hess, K.; Pundlik, S.J.; Jeffrey, B.G.; Cukras, C.A.; Husain, D. Dark Adaptation and Its Role in Age-Related Macular Degeneration. J. Clin. Med. 2022, 11, 1358. [Google Scholar] [CrossRef]
- Marmor, M.F.; Holder, G.E.; Seeliger, M.W.; Yamamoto, S. Standard for clinical electroretinography. Doc. Ophthalmol. 2004, 108, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Clark, M.E.; Kraft, T.W. Measuring rodent electroretinograms to assess retinal function. Methods Mol. Biol. 2012, 884, 265–276. [Google Scholar] [CrossRef] [PubMed]
- Clark, M.E. Comparisons Between Electrophysiologic Assessments of Visual Function and Histological Findings of Cone Photoreceptors in Light-Damage Albino Rats. Master’s Thesis, University of Alabama at Birmingham, Birmingham, AL, USA, 2013. [Google Scholar]



| Dark Adapted Amplitude (μV) | Light Adapted Amplitude (μV) | |||
|---|---|---|---|---|
| a-Wave | b-Wave | a-Wave | b-Wave | |
| Pre-LD | ||||
| Control | 428 ± 21 | 1017 ± 58 | 83.5 ± 8.7 | 279 ± 19 |
| LD | 403 ± 23 | 945 ± 52 | 71.7 ± 6.2 | 250 ± 14 |
| p value n.s. | ||||
| R5 | ||||
| Control | 345 ± 15 | 898 ± 47 | 83.2 ± 6.0 | 251.0 ± 4.0 |
| LD | 46.6 ± 7.1 | 218 ± 25 | 13.9 ± 2.2 | 61.0 ± 6.1 |
| 1p < 0.001 | ||||
| R30 | ||||
| Control 2 | 400 ± 33 | 1051 ± 64 | 98.9 ± 7.3 | 260 ± 13 |
| LD | 86.7 ± 8.3 | 456 ± 38 | 30.8 ± 5.0 | 157 ± 25 |
| p < 0.001 | p < 0.001 | p < 0.01 | p < 0.01 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benthal, M.C.; McKeown, A.S.; Kraft, T.W. Cone Photoreceptor Loss in Light-Damaged Albino Rats. Int. J. Mol. Sci. 2022, 23, 3978. https://doi.org/10.3390/ijms23073978
Benthal MC, McKeown AS, Kraft TW. Cone Photoreceptor Loss in Light-Damaged Albino Rats. International Journal of Molecular Sciences. 2022; 23(7):3978. https://doi.org/10.3390/ijms23073978
Chicago/Turabian StyleBenthal, Molly C., Alex S. McKeown, and Timothy W. Kraft. 2022. "Cone Photoreceptor Loss in Light-Damaged Albino Rats" International Journal of Molecular Sciences 23, no. 7: 3978. https://doi.org/10.3390/ijms23073978
APA StyleBenthal, M. C., McKeown, A. S., & Kraft, T. W. (2022). Cone Photoreceptor Loss in Light-Damaged Albino Rats. International Journal of Molecular Sciences, 23(7), 3978. https://doi.org/10.3390/ijms23073978






