Ultrastable Covalent Triazine Organic Framework Based on Anthracene Moiety as Platform for High-Performance Carbon Dioxide Adsorption and Supercapacitors
Abstract
:1. Introduction
2. Results and Discussion
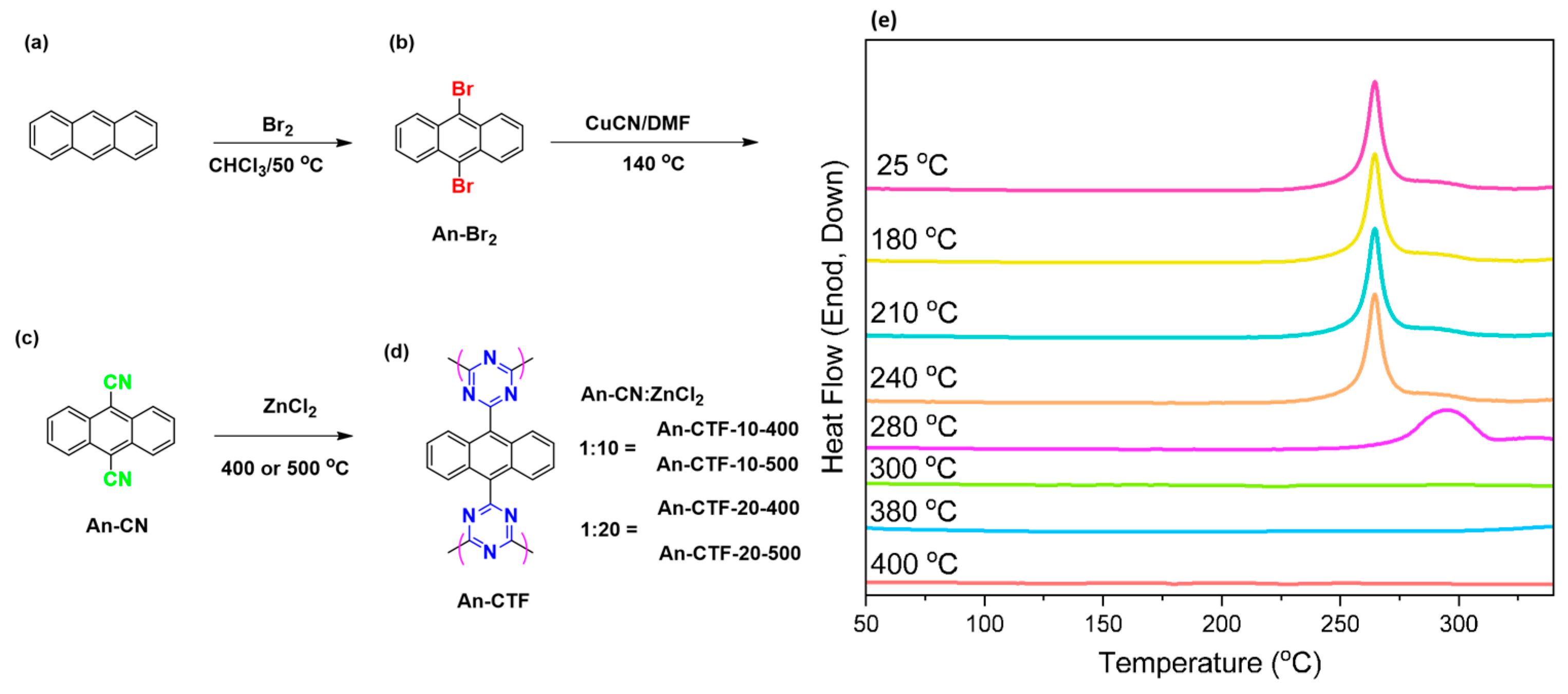
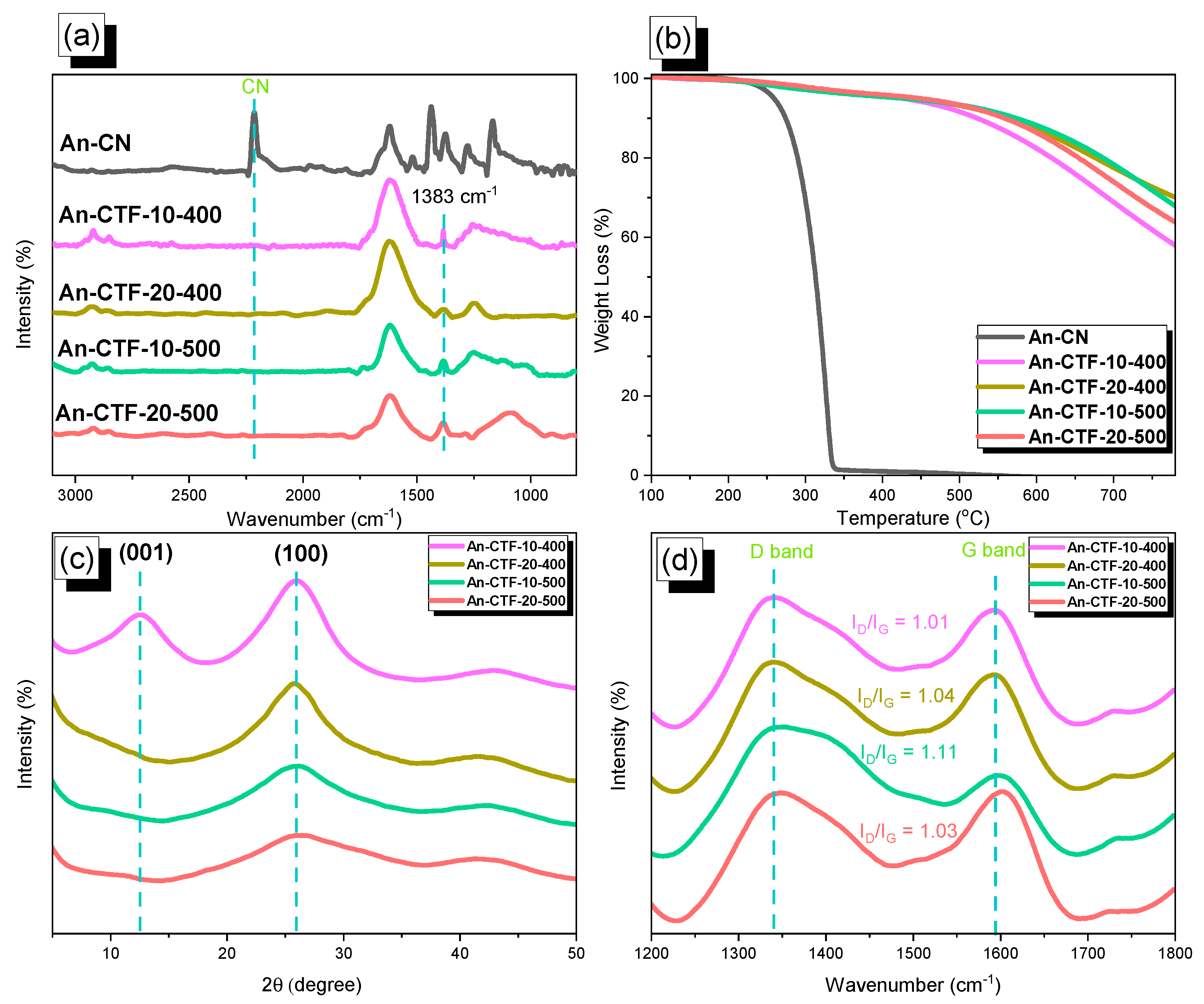
2.1. Synthesis and Characterization of An-CN and An-CTFs
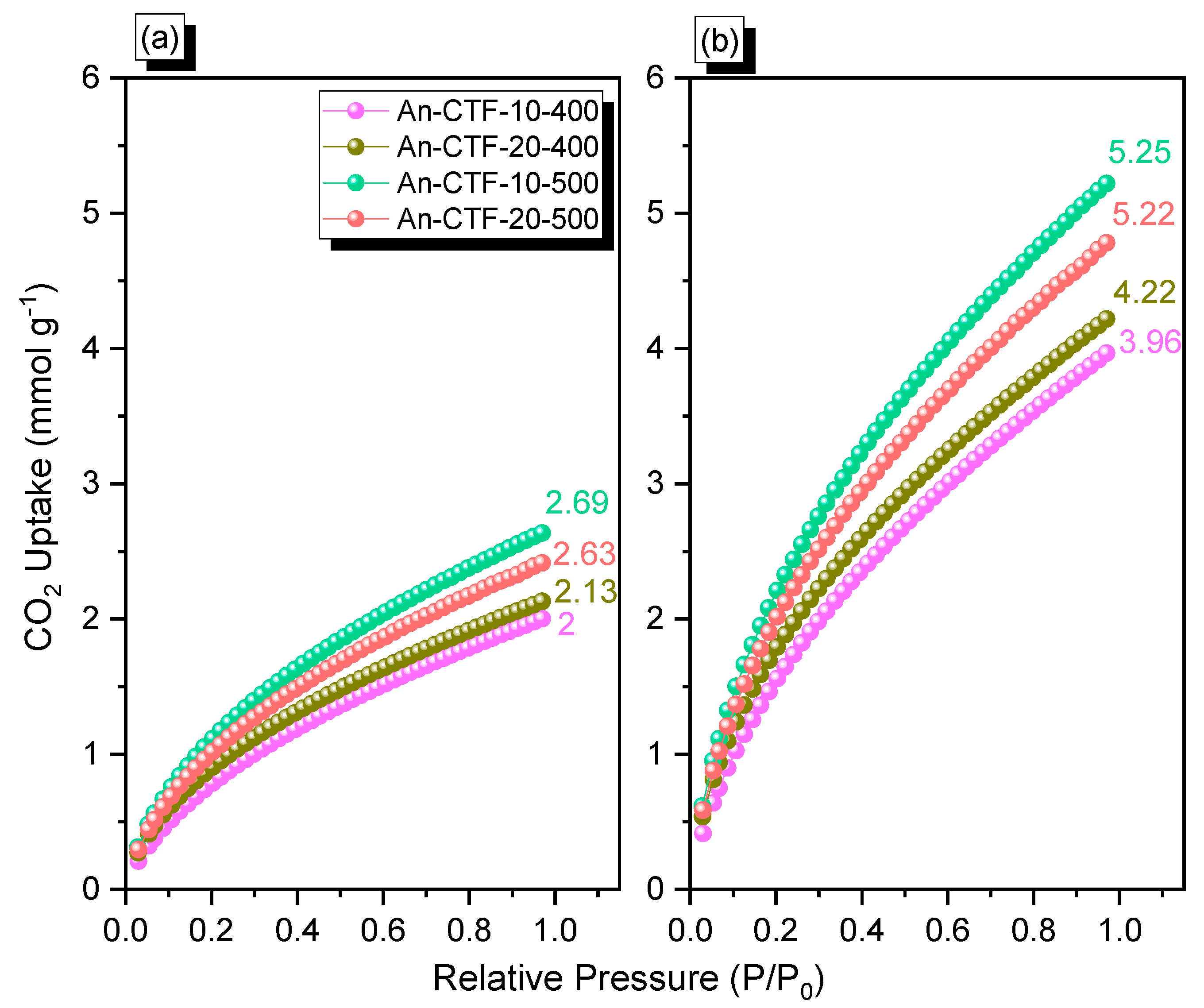
2.2. Porosity and CO2 Uptake of An-CTFs
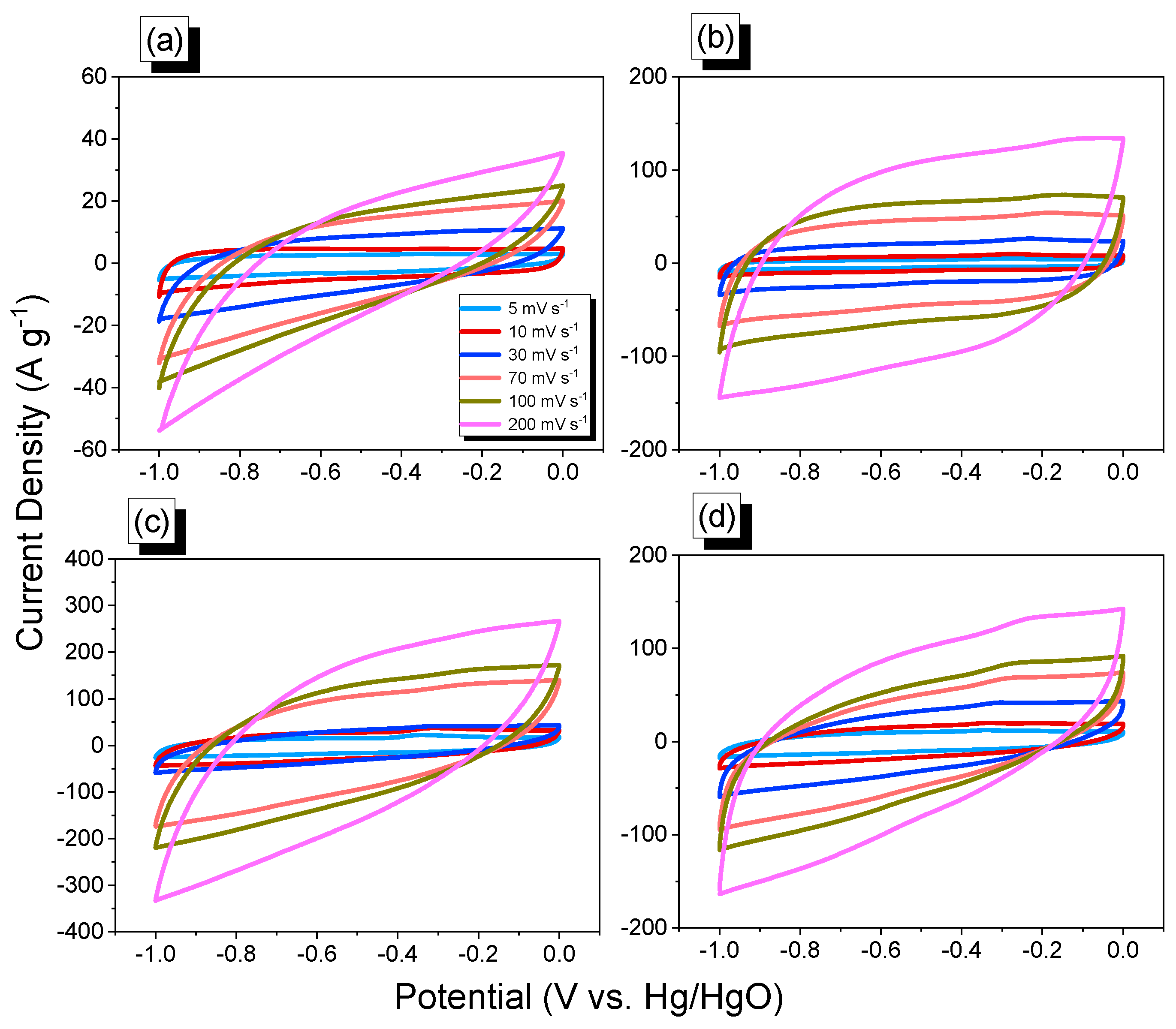
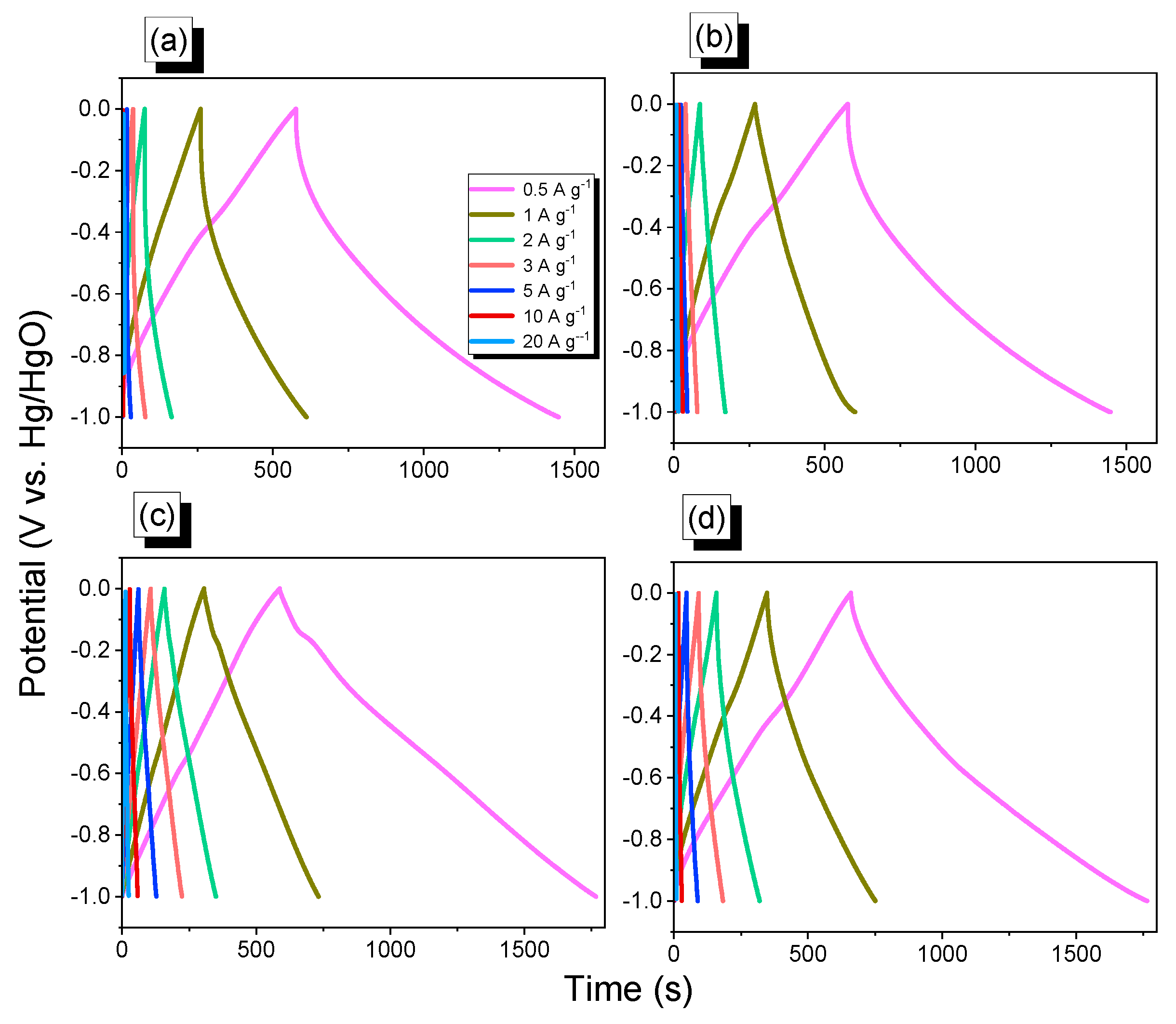
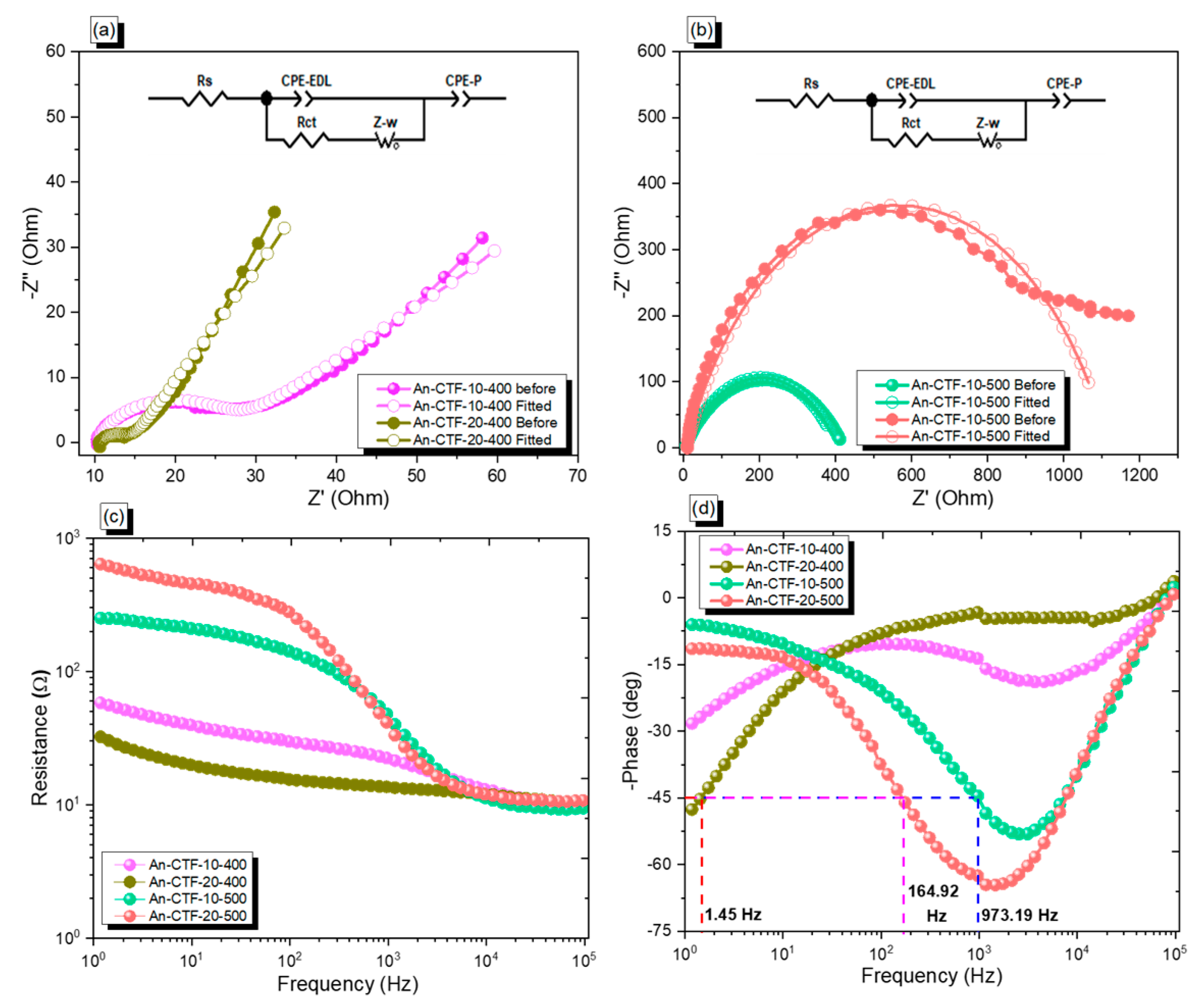
2.3. Electrochemical Performance of An-CTFs
3. Experimental Section
3.1. Materials
3.2. Preparation of 9,10-Dibromoanthracene (An-Br2)
3.3. Preparation of 9,10-Cyanoanthracene (An-CN)
3.4. Preparation of An-CTFs
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lin, Z.; Goikolea, E.; Balducci, A.; Naoi, K.; Taberna, P.L.; Salanne, M.; Yushin, G.; Simon, P. Materials for Supercapacitors: When Li-Ion Battery Power is not Enough. Mater. Today 2018, 21, 419–436. [Google Scholar] [CrossRef] [Green Version]
- EL-Mahdy, A.F.M.; Elewa, A.M.; Huang, S.W.; Chou, H.H.; Kuo, S.W. Dual-Function Fluorescent Covalent Organic Frameworks: HCl Sensing and Photocatalytic H2 Evolution from Water. Adv. Opt. Mater. 2020, 8, 2000641. [Google Scholar] [CrossRef]
- Mohamed, M.G.; EL-Mahdy, A.F.M.; Kotp, M.G.; Kuo, S.W. Advances in Porous Organic Polymers: Syntheses, Structures, and Diverse Applications. Mater. Adv. 2022, 3, 707–733. [Google Scholar] [CrossRef]
- Mohamed, M.G.; Zhang, X.; Mansoure, T.H.; El-Mahdy, A.F.M.; Huang, C.F.; Danko, M.; Xin, Z.; Kuo, S.W. Hypercrosslinked porous organic polymers based on tetraphenylanthraquinone for CO2 uptake and high-performance supercapacitor. Polymer 2020, 205, 1802369. [Google Scholar] [CrossRef]
- Chen, D.; Jiang, K.; Huang, T.; Shen, G. Recent Advances in Fiber Supercapacitors: Materials, Device Configurations, and Applications. Adv. Mater. 2020, 32, 1901806. [Google Scholar] [CrossRef]
- Zhang, G.; Xiao, P.; Ni, F.; Gu, J.; Chen, J.; Nie, Y.; Kuo, S.W.; Chen, T. Breathable and superhydrophobic photothermic fabric enables efficient interface energy management via confined heating strategy for sustainable seawater evaporation. Chem. Eng. J. 2022, 428, 131142. [Google Scholar] [CrossRef]
- Mohamed, M.G.; Tsai, M.Y.; Wang, C.F.; Huang, C.F.; Danko, M.; Dai, L.; Chen, T.; Kuo, S.W. Multifunctional Polyhedral Oligomeric Silsesquioxane (POSS) Based Hybrid Porous Materials for CO2 Uptake and Iodine Adsorption. Polymers 2021, 13, 221. [Google Scholar] [CrossRef]
- Mohamed, M.G.; Sharma, S.U.; Yang, C.H.; Samy, M.M.; Mohammed, A.A.K.; Chaganti, S.V.; Lee, J.T.; Kuo, S.W. Anthraquinone-Enriched Conjugated Microporous Polymers as Organic Cathode Materials for High-Performance Lithium-Ion Batteries. ACS Appl. Energy Mater. 2021, 4, 14628–14639. [Google Scholar] [CrossRef]
- EL-Mahdy, A.F.M.; Yu, T.C.; Mohamed, M.G.; Kuo, S.W. Secondary Structures of Polypeptide-Based Diblock Copolymers Influence the Microphase Separation of Templates for the Fabrication of Microporous Carbons. Macromolecules 2021, 54, 1030–1042. [Google Scholar] [CrossRef]
- Mohamed, M.G.; Chen, T.C.; Kuo, S.W. Solid-State Chemical Transformations to Enhance Gas Capture in Benzoxazine-Linked Conjugated Microporous Polymers. Macromolecules 2021, 54, 5866–5877. [Google Scholar] [CrossRef]
- Samy, M.M.; Mohamed, M.G.; Kuo, S.W. Pyrene-functionalized tetraphenylethylene polybenzoxazine for dispersing single-walled carbon nanotubes and energy storage. Compos. Sci. Technol. 2020, 199, 108360. [Google Scholar] [CrossRef]
- Mohamed, M.G.; Mansoure, T.H.; Takashi, Y.; Samy, M.M.; Chen, T.; Kuo, S.W. Ultrastable porous organic/inorganic polymers based on polyhedral Oligomeric silsesquioxane (POSS) hybrids exhibiting high performance for thermal property and energy storage. Microporous Mesoporous Mater. 2021, 328, 111505. [Google Scholar] [CrossRef]
- Samy, M.M.; Mohamed, M.G.; Mansoure, T.H.; Meng, T.S.; Khan, M.A.R.; Liaw, C.C.; Kuo, S.W. Solid state chemical transformations through ring-opening polymerization of ferrocene-based conjugated microporous polymers in host-guest complexes with benzoxazine-linked cyclodextrin. J. Taiwan Inst. Chem. Eng. 2022, 132, 104110. [Google Scholar] [CrossRef]
- Samy, M.M.; Mohamed, M.G.; El-Mahdy, A.F.M.; Mansoure, T.H.; Wu, K.C.W.; Kuo, S.W. High-Performance Supercapacitor Electrodes Prepared from Dispersions of Tetrabenzonaphthalene-Based Conjugated Microporous Polymers and Carbon Nanotubes. ACS Appl. Mater. Interfaces 2021, 13, 51906–51916. [Google Scholar] [CrossRef]
- Tao, Z.; Ikram, M.; Ji, H.; Zhu, Y. A Hierarchical Carbon Derived from Sponge-Templated Activation of Graphene Oxide for High Performance Supercapacitor Electrodes. Adv. Mater. 2016, 28, 5222–5228. [Google Scholar] [CrossRef]
- Yao, L.; Wu, Q.; Zhang, P.; Zhang, J.; Wang, D.; Li, Y.; Ren, X.; Mi, H.; Deng, L.; Zheng, Z. Scalable 2D Hierarchical Porous Carbon Nanosheets for Flexible Supercapacitors with Ultrahigh Energy Density. Adv. Mater. 2018, 30, 1706054. [Google Scholar] [CrossRef]
- Mohamed, M.G.; Atayde, E.C., Jr.; Matsagar, B.M.; Na, J.; Yamauchi, Y.; Wu, K.C.W.; Kuo, S.W. Construction Hierarchically Mesoporous/Microporous Materials Based on Block Copolymer and Covalent Organic Framework. J. Taiwan Inst. Chem. Eng. 2020, 112, 180–192. [Google Scholar] [CrossRef]
- Lin, T.; Chen, I.-W.; Liu, F.; Yang, C.; Bi, H.; Xu, F.; Huang, F. Nitrogen-doped Mesoporous Carbon of Extraordinary Capacitance for Electrochemical Energy Storage. Science 2015, 350, 1508–1513. [Google Scholar] [CrossRef] [Green Version]
- Zhu, Q.Y.; Wu, P.; Zhang, J.J.; Zhang, W.Y.; Zhou, Y.M.; Tang, Y.W.; Lu, T.H. Cyanogel-Derived Formation of 3 D Nanoporous SnO2–MxOy (M=Ni, Fe, Co) Hybrid Networks for High-Performance Lithium Storage. ChemSusChem 2015, 8, 131–137. [Google Scholar] [CrossRef]
- Wang, Q.; Zhu, Y.; Xue, J.; Zhao, X.; Guo, Z.; Wang, C. General Synthesis of Porous Mixed Metal Oxide Hollow Spheres with Enhanced Supercapacitive Properties. ACS Appl. Mater. Interfaces 2016, 8, 17226–17232. [Google Scholar] [CrossRef] [Green Version]
- Diaz, R.; Gisela Orcajo, M.; Botas, J.A.; Calleja, G.; Palma, J. Co8-MOF-5 as electrode for supercapacitors. Mater. Lett. 2012, 68, 126–128. [Google Scholar] [CrossRef]
- Li, W.H.; Ding, K.; Tian, H.R.; Yao, M.S.; Nath, B.; Deng, W.H.; Wang, Y.; Xu, G. Conductive Metal-Organic Framework Nanowire Array Electrodes for High-Performance Solid-State Supercapacitors. Adv. Funct. Mater. 2017, 27, 1702067. [Google Scholar] [CrossRef]
- Fuertes, A.B.; Sevilla, M. Superior Capacitive Performance of Hydrochar-Based Porous Carbons in Aqueous Electrolytes. ChemSusChem 2015, 8, 1049–1057. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, L.; Niu, Z.; Chen, J. Unconventional supercapacitors from nanocarbon-based electrode materials to device configurations. Chem. Soc. Rev. 2016, 45, 4340–4363. [Google Scholar] [CrossRef]
- Mo, R.J.; Zhao, Y.; Wu, M.; Xiao, H.M.; Kuga, S.; Huang, Y.; Li, J.P.; Fu, Y. Activated carbon from nitrogen rich watermelon rind for high-performance supercapacitors. RSC Adv. 2016, 6, 59333–59342. [Google Scholar] [CrossRef]
- Tang, J.; Wang, T.; Salunkhe, R.R.; Alshehri, S.M.; Malgras, V.; Yamauchi, Y. Three-Dimensional Nitrogen-Doped Hierarchical Porous Carbon as an Electrode for High-Performance Supercapacitors. Chem. Eur. J. 2015, 21, 17293–17298. [Google Scholar] [CrossRef]
- Zhu, Y.; Murali, S.; Stoller, M.D.; Ganesh, K.; Cai, W.; Ferreira, P.J.; Pirkle, A.; Wallace, R.M.; Cychosz, K.A.; Thommes, M. Carbon-Based Supercapacitors Produced by Activation of Graphene. Science 2011, 332, 1537–1541. [Google Scholar] [CrossRef] [Green Version]
- Samy, M.M.; Mohamed, M.G.; Kuo, S.W. Directly synthesized nitrogen-and-oxygen-doped microporous carbons derived from a bio-derived polybenzoxazine exhibiting high-performance supercapacitance and CO2 uptake. Eur. Polym. J. 2020, 138, 109954. [Google Scholar] [CrossRef]
- Gu, C.; Huang, N.; Chen, Y.C.; Zhang, H.H.; Zhang, S.T.; Li, F.H.; Ma, Y.G.; Jiang, D.L. Porous Organic Polymer Films with Tunable Work Functions and Selective Hole and Electron Flows for Energy Conversions. Angew. Chem. Int. Ed. 2016, 55, 3049–3053. [Google Scholar] [CrossRef]
- EL-Mahdy, A.F.M.; Young, C.; Kim, J.; You, J.; Yamauchi, Y.; Kuo, S.W. Hollow Microspherical and Microtubular [3+3] Carbazole-Based Covalent Organic Frameworks and Their Gas and Energy Storage Applications. ACS Appl. Mater. Interfaces 2019, 11, 9343–9354. [Google Scholar] [CrossRef]
- Mohamed, M.G.; Samy, M.M.; Mansoure, T.H.; Li, C.J.; Li, W.C.; Chen, J.H.; Zhang, K.; Kuo, S.W. Microporous Carbon and Carbon/Metal Composite Materials Derived from Bio-Benzoxazine-Linked Precursor for CO2 Capture and Energy Storage Applications. Int. J. Mol. Sci. 2022, 23, 347. [Google Scholar] [CrossRef] [PubMed]
- EL-Mahdy, A.F.M.; Liu, T.E.; Kuo, S.W. Direct synthesis of nitrogen-doped mesoporous carbons from triazine-functionalized resol for CO2 uptake and highly efficient removal of dyes. J. Hazard. Mater. 2020, 391, 122163. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.G.; Lee, C.C.; EL-Mahdy, A.F.M.; Lüder, J.; Yu, M.H.; Li, Z.; Zhu, Z.; Chueh, C.C.; Kuo, S.W. Exploitation of two-dimensional conjugated covalent organic frameworks based on tetraphenylethylene with bicarbazole and pyrene units and applications in perovskite solar cells. J. Mater. Chem. A 2020, 8, 11448–11459. [Google Scholar] [CrossRef]
- Aly, K.I.; Sayed, M.M.; Mohamed, M.G.; Kuo, S.W.; Younis, O. A facile synthetic route and dual function of network luminescent porous polyester and copolyester containing porphyrin moiety for metal ions sensor and dyes adsorption. Microporous Mesoporous Mater. 2020, 298, 110063. [Google Scholar] [CrossRef]
- Tang, J.; Salunkhe, R.R.; Liu, J.; Torad, N.L.; Imura, M.; Furukawa, S.; Yamauchi, Y. Thermal Conversion of Core-Shell Metal-Organic Frameworks: A New Method for Selectively Functionalized Nanoporous Hybrid Carbon. J. Am. Chem. Soc. 2015, 137, 1572–1580. [Google Scholar] [CrossRef] [PubMed]
- Salunkhe, R.R.; Lin, J.; Malgras, V.; Dou, S.X.; Kim, J.H.; Yamauchi, Y. Large-scale synthesis of coaxial carbon nanotube/Ni(OH)2 composites for asymmetric supercapacitor application. Nano Energy 2015, 11, 211–218. [Google Scholar] [CrossRef]
- Mike, J.F.; Lutkenhaus, J.F. Recent advances in conjugated polymer energy storage. J. Polym. Sci. B Polym. Phys. 2013, 51, 468–480. [Google Scholar] [CrossRef]
- Halder, A.; Ghosh, M.; Khayum, M.A.; Bera, S.; Addicoat, M.; Sasmal, H.S.; Karak, S.; Kurungot, S.; Banerjee, R. Interlayer Hydrogen-Bonded Covalent Organic Frameworks as High-Performance Supercapacitors. J. Am. Chem. Soc. 2018, 140, 10941–10945. [Google Scholar] [CrossRef] [Green Version]
- Gharahcheshmeh, M.H.; Gleason, K.K. Device Fabrication Based on oxidative Chemical Vapor Deposition (oCVD) Synthesis of Conducting Polymers and Related Conjugated Organic Materials. Adv. Mater. Interfaces 2019, 6, 1801564. [Google Scholar] [CrossRef] [Green Version]
- Kuo, S.W. Hydrogen bonding mediated self-assembled structures from block copolymer mixtures to mesoporous materials. Polym. Inter. 2022; preprint. [Google Scholar] [CrossRef]
- Mohamed, M.G.; Elsayed, M.H.; Elewa, A.M.; EL-Mahdy, A.F.M.; Yang, C.H.; Mohammed, A.A.K.; Chou, H.H.; Kuo, S.W. Pyrene Containing Conjugated Organic Microporous Polymers for Photocatalytic Hydrogen Evolution from Water. Catal. Sci. Technol. 2021, 11, 2229–2241. [Google Scholar] [CrossRef]
- Mohamed, M.G.; Liu, N.Y.; EL-Mahdy, A.F.M.; Kuo, S.W. Ultrastable luminescent hybrid microporous polymers based on polyhedral Oligomeric silsesquioxane for CO2 uptake and metal ion sensing. Microporous Mesoporous Mater. 2021, 311, 110695. [Google Scholar] [CrossRef]
- Elewa, A.M.; EL-Mahdy, A.F.M.; Elsayed, M.H.; Mohamed, M.G.; Kuo, S.W.; Chou, H.H. Sulfur-doped triazine-conjugated microporous polymers for achieving the robust visible-light-driven hydrogen evolution. Chem. Eng. J. 2021, 421, 129825. [Google Scholar] [CrossRef]
- Kuhn, P.; Antonietti, M.; Thomas, A. Porous, Covalent Triazine-Based Frameworks Prepared by Ionothermal Synthesis. Angew. Chem. Int. Ed. 2008, 47, 3450–3453. [Google Scholar] [CrossRef]
- Katekomol, P.; Roeser, J.; Bojdys, M.; Weber, J.; Thomas, A. Covalent Triazine Frameworks Prepared from 1,3,5-Tricyanobenzene. Chem. Mater. 2013, 25, 1542–1548. [Google Scholar] [CrossRef]
- Bhunia, A.; Vasylyeva, V.; Janiak, C. From a supramolecular tetranitrile to a porous covalent triazine-based framework with high gas uptake capacities. Chem. Commun. 2013, 49, 3961–3963. [Google Scholar] [CrossRef]
- Hao, L.; Zhang, S.; Liu, R.; Ning, J.; Zhang, G.; Zhi, L. Bottom-Up Construction of Triazine-Based Frameworks as Metal-Free Electrocatalysts for Oxygen Reduction Reaction. Adv. Mater. 2015, 27, 3190–3195. [Google Scholar] [CrossRef]
- Talapaneni, S.N.; Hwang, T.H.; Je, S.H.; Buyukcakir, O.; Choi, J.W.; Coskun, A. Elemental-Sulfur-Mediated Facile Synthesis of a Covalent Triazine Framework for High-Performance Lithium-Sulfur Batteries. Angew. Chem. Int. Ed. 2016, 55, 3106–3111. [Google Scholar] [CrossRef]
- Krishnaraj, C.; Jena, H.S.; Leus, K.; Voort, P.V.D. Covalent triazine frameworks—A sustainable perspective. Green Chem. 2020, 22, 1038–1071. [Google Scholar] [CrossRef]
- Guo, L.; Wang, X.; Zhan, Z.; Zhao, Y.; Chen, L.; Liu, T.; Tan, B.; Jin, S. Crystallization of Covalent Triazine Frameworks via a Heterogeneous Nucleation Approach for Efficient Photocatalytic Applications. Chem. Mater. 2021, 33, 1994–2003. [Google Scholar] [CrossRef]
- Vadiyar, M.M.; Liu, X.; Ye, Z. Macromolecular Polyethynylbenzonitrile Precursor-Based Porous Covalent Triazine Frameworks for Superior High-Rate High-Energy Supercapacitors. ACS Appl. Mater. Interfaces 2019, 11, 45805–45817. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zheng, S.; Liu, X.; Li, P.; Sun, L.; Yang, R.; Wang, S.; Wu, Z.S.; Bao, X.; Deng, W.Q. Conductive Microporous Covalent Triazine-Based Framework for High-Performance Electrochemical Capacitive Energy Storage. Angew. Chem. Int. Ed. 2018, 57, 7992–7996. [Google Scholar] [CrossRef] [PubMed]
- Bhanja, P.; Bhunia, K.; Das, S.K.; Pradhan, D.; Kimura, R.; Hijikata, Y.; Irle, S.; Bhaumik, A. A New Triazine-Based Covalent Organic Framework for High-Performance Capacitive Energy Storage. ChemSusChem 2017, 10, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.G.; EL-Mahdy, A.F.M.; Ahmed, M.M.M.; Kuo, S.W. Direct Synthesis of Microporous Bicarbazole-Based Covalent Triazine Frameworks for High-Performance Energy Storage and Carbon Dioxide Uptake. ChemPlusChem 2019, 84, 1767–1774. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.G.; EL-Mahdy, A.F.M.; Takashi, Y.; Kuo, S.W. Ultrastable conductive microporous covalent triazine frameworks based on pyrene moieties provide high-performance CO2 uptake and supercapacitance. New J. Chem. 2020, 44, 8241–8253. [Google Scholar] [CrossRef]
- Mukherjee, S.; Das, M.; Manna, A.; Krishna, R.; Das, S. Dual Strategic Approach to Prepare Defluorinated Triazole Embedded Covalent Triazine Frameworks with High Gas Uptake Performance. Chem. Mater. 2019, 31, 3929–3940. [Google Scholar] [CrossRef]
- Hao, L.; Ning, J.; Luo, B.; Wang, B.; Zhang, Y.; Tang, Z.; Yang, J.; Thomas, A.; Zhi, L. Structural Evolution of 2D Microporous Covalent Triazine-Based Framework toward the Study of High-Performance Supercapacitors. J. Am. Chem. Soc. 2015, 137, 219–225. [Google Scholar] [CrossRef]
- Mohamed, M.G.; Ebrahium, S.M.; Hammam, A.S.; Kuo, S.W.; Aly, K.I. Enhanced CO2 capture in nitrogen-enriched microporous carbons derived from Polybenzoxazines containing azobenzene and carboxylic acid units. J. Polym. Res. 2020, 27, 197. [Google Scholar] [CrossRef]
- Mohamed, M.G.; Tsai, M.Y.; Su, W.C.; EL-Mahdy, A.F.M.; Wang, C.F.; Huang, C.F.; Dai, L.; Chen, T.; Kuo, S.W. Nitrogen-Doped microporous carbons derived from azobenzene and nitrile-functionalized polybenzoxazines for CO2 uptake. Mater. Today Commun. 2020, 24, 101111. [Google Scholar] [CrossRef]
- Wang, G.; Leus, K.; Zhao, S.; Voort, P.V.D. Newly Designed Covalent Triazine Framework Based on Novel N-Heteroaromatic Building Blocks for Efficient CO2 and H2 Capture and Storage. ACS Appl. Mater. Interfaces 2018, 10, 1244–1249. [Google Scholar] [CrossRef]
- Abuzeid, H.R.; EL-Mahdy, A.F.M.; Ahmed, M.M.M.; Kuo, S.W. Triazine-functionalized covalent benzoxazine framework for direct synthesis of N-doped microporous carbon. Polym. Chem. 2019, 10, 6010–6020. [Google Scholar] [CrossRef]
- Gu, C.; Liu, D.; Huang, W.; Liu, J.; Yang, R. Synthesis of covalent triazine-based frameworks with high CO2 adsorption and selectivity. Polym. Chem. 2015, 6, 7410–7417. [Google Scholar] [CrossRef]
- Mohamed, M.G.; Ahmed, M.M.M.; Du, W.T.; Kuo, S.W. Meso/Microporous Carbons from Conjugated Hyper-Crosslinked Polymers Based on Tetraphenylethene for High-Performance CO2 Capture and Supercapacitor. Molecules 2021, 26, 738. [Google Scholar] [CrossRef] [PubMed]
- Pant, B.; Park, M.; Ojha, G.P.; Park, J.; Kuk, Y.S.; Lee, E.J.; Park, S.J. Carbon nanofibers wrapped with zinc oxide nano-flakes as promising electrode material for supercapacitors. J. Colloid Interface Sci. 2018, 522, 40–47. [Google Scholar] [CrossRef]
- Kazazi, M. High-performance electrode based on electrochemical polymerization of polypyrrole film on electrophoretically deposited CNTs conductive framework for supercapacitors. Solid State Ion. 2019, 336, 80–86. [Google Scholar] [CrossRef]
- Kirubasankar, B.; Narayanasamy, M.; Yang, J.; Han, M.; Zhu, W.; Su, Y.; Angaiah, S.; Yan, C. Construction of heterogeneous 2D layered MoS2/MXene nanohybrid anode material via interstratification process and its synergetic effect for asymmetric supercapacitors. Appl. Surf. Sci. 2020, 534, 147644. [Google Scholar] [CrossRef]
- Arof, A.K.; Amirudin, S.; Yusofa, S.Z.; Noor, I.M. A method based on impedance spectroscopy to determine transport properties of polymer electrolytes. Phys. Chem. Chem. Phys. 2014, 16, 1856–1867. [Google Scholar] [CrossRef] [Green Version]
- Mohamed, M.G.; Samy, M.M.; Mansoure, T.H.; Sharma, S.U.; Tsai, M.S.; Chen, J.H.; Lee, J.T.; Kuo, S.W. Dispersions of 1,3,4-Oxadiazole-Linked Conjugated Microporous Polymers with Carbon Nanotube as High-Performance Electrode for Supercapacitor. ACS Appl. Energy Mater. 2022; preprint. [Google Scholar] [CrossRef]




| Material | Td5 (°C) | Td10 (°C) | Char Yield (%) | Surface Area (m2 g−1) | Pore Size (nm) | CO2 Uptake (298 K) | CO2 Uptake (273 K) |
|---|---|---|---|---|---|---|---|
| An-CTF-10-400 | 425 | 522 | 56 | 406 | 1.1, 1.84 | 2.00 | 3.96 |
| An-CTF-20-400 | 411 | 562 | 69 | 491 | 1.02, 1.72 | 2.13 | 4.22 |
| An-CTF-10-500 | 427 | 573 | 66 | 751 | 1.18, 1.87 | 2.63 | 5.22 |
| An-CTF-20-500 | 449 | 558 | 62 | 700 | 1.06, 1.66 | 2.69 | 5.25 |
| Materials | N Species | O Species | ||||
|---|---|---|---|---|---|---|
| N-6 | N-5 | N-Q | C–O | C–OH | H2O | |
| An-CTF-10-400 | 29.51 | 41.44 | 29.05 | 27.89 | 43.57 | 28.54 |
| An-CTF-20-400 | 29.55 | 40.72 | 29.73 | 28.35 | 42.96 | 28.69 |
| An-CTF-10-500 | 29.43 | 40.62 | 29.94 | 28.28 | 43.44 | 28.28 |
| An-CTF-20-500 | 28.94 | 41.92 | 29.14 | 29.35 | 40.86 | 29.79 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohamed, M.G.; Sharma, S.U.; Liu, N.-Y.; Mansoure, T.H.; Samy, M.M.; Chaganti, S.V.; Chang, Y.-L.; Lee, J.-T.; Kuo, S.-W. Ultrastable Covalent Triazine Organic Framework Based on Anthracene Moiety as Platform for High-Performance Carbon Dioxide Adsorption and Supercapacitors. Int. J. Mol. Sci. 2022, 23, 3174. https://doi.org/10.3390/ijms23063174
Mohamed MG, Sharma SU, Liu N-Y, Mansoure TH, Samy MM, Chaganti SV, Chang Y-L, Lee J-T, Kuo S-W. Ultrastable Covalent Triazine Organic Framework Based on Anthracene Moiety as Platform for High-Performance Carbon Dioxide Adsorption and Supercapacitors. International Journal of Molecular Sciences. 2022; 23(6):3174. https://doi.org/10.3390/ijms23063174
Chicago/Turabian StyleMohamed, Mohamed Gamal, Santosh U. Sharma, Ni-Yun Liu, Tharwat Hassan Mansoure, Maha Mohamed Samy, Swetha V. Chaganti, Yu-Lung Chang, Jyh-Tsung Lee, and Shiao-Wei Kuo. 2022. "Ultrastable Covalent Triazine Organic Framework Based on Anthracene Moiety as Platform for High-Performance Carbon Dioxide Adsorption and Supercapacitors" International Journal of Molecular Sciences 23, no. 6: 3174. https://doi.org/10.3390/ijms23063174
APA StyleMohamed, M. G., Sharma, S. U., Liu, N.-Y., Mansoure, T. H., Samy, M. M., Chaganti, S. V., Chang, Y.-L., Lee, J.-T., & Kuo, S.-W. (2022). Ultrastable Covalent Triazine Organic Framework Based on Anthracene Moiety as Platform for High-Performance Carbon Dioxide Adsorption and Supercapacitors. International Journal of Molecular Sciences, 23(6), 3174. https://doi.org/10.3390/ijms23063174









