Sp1 Plays a Key Role in Vasculogenic Mimicry of Human Prostate Cancer Cells
Abstract
1. Introduction
2. Results
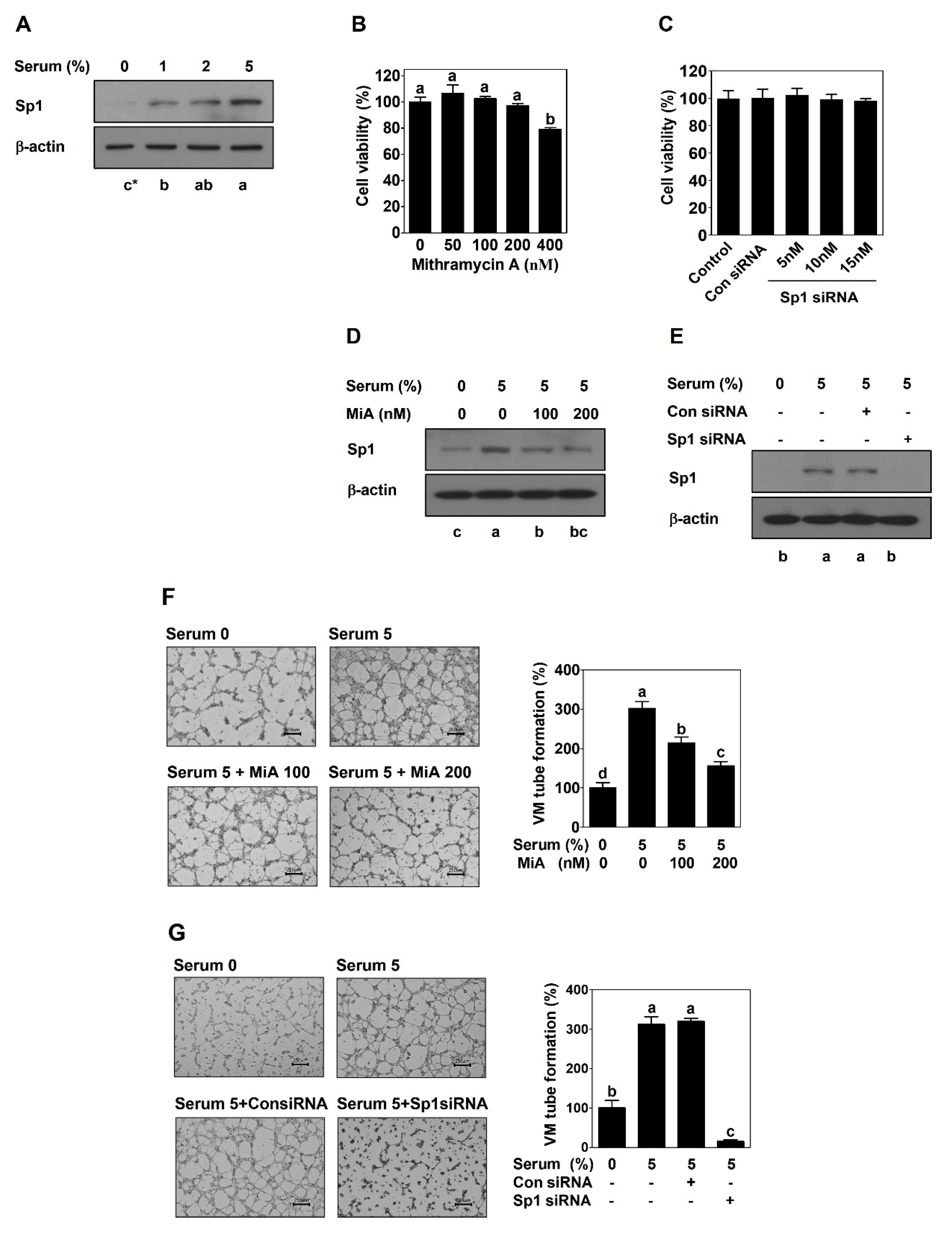
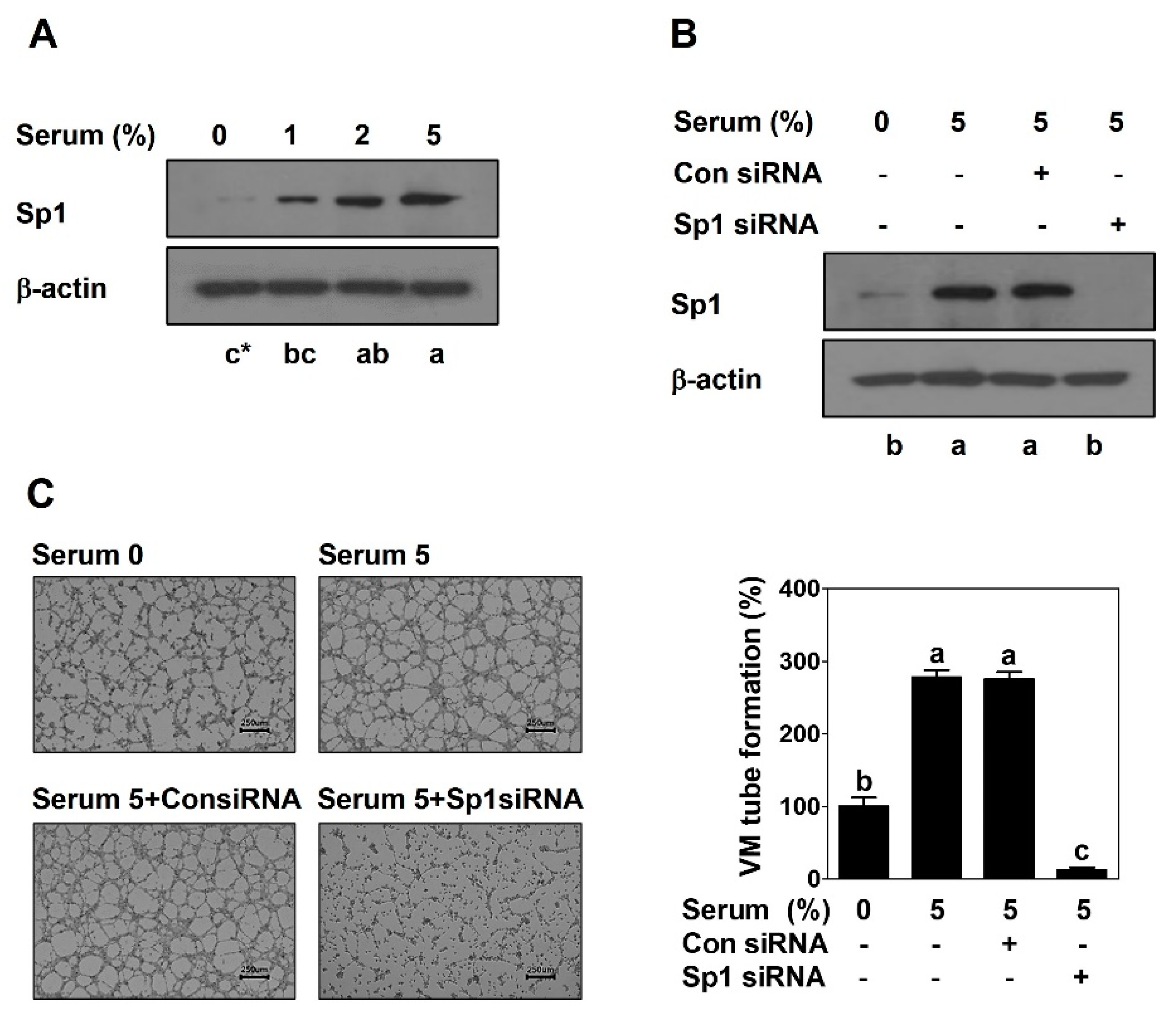
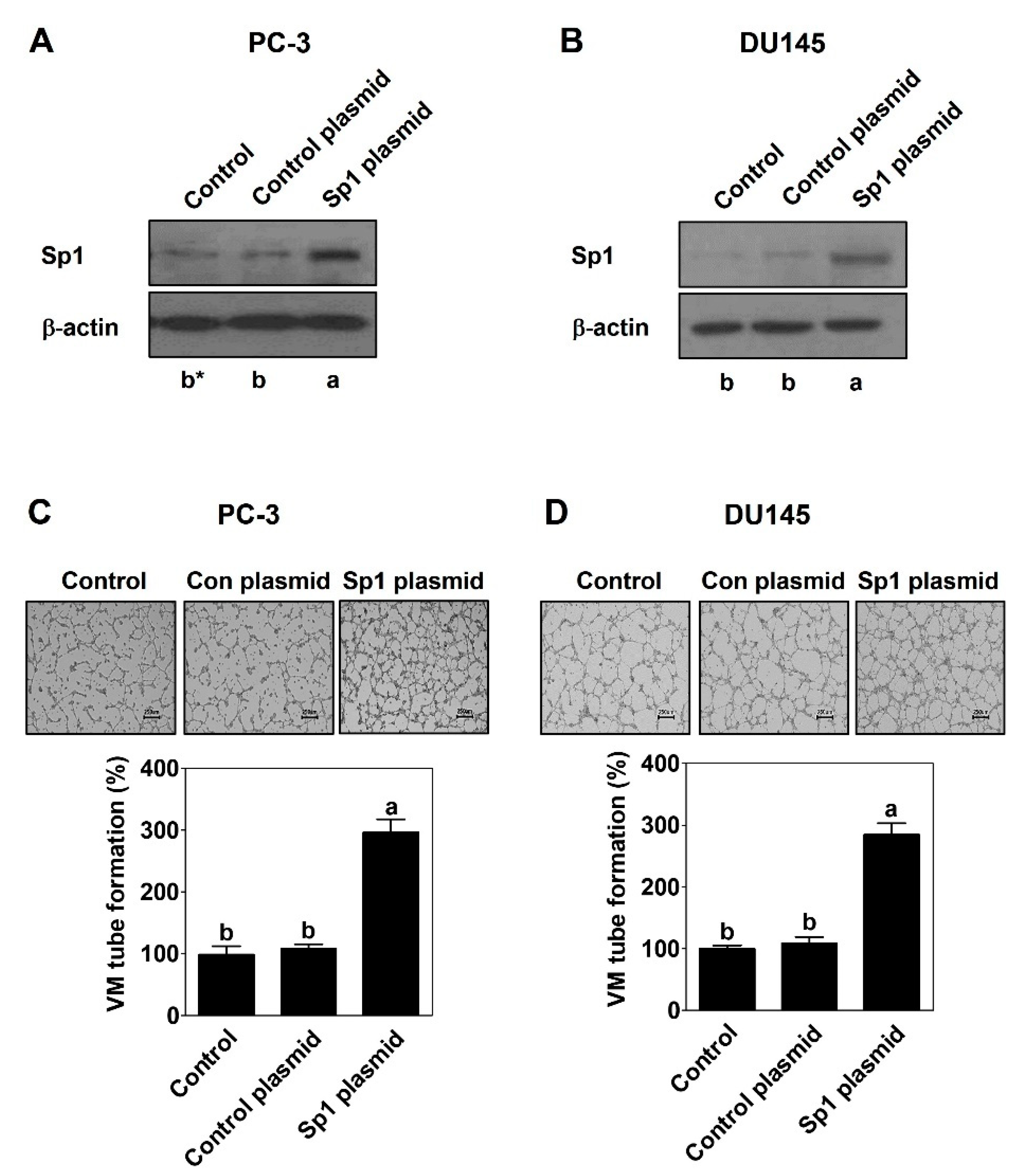
2.1. Sp1 Mediates VM Formation in PCa Cells
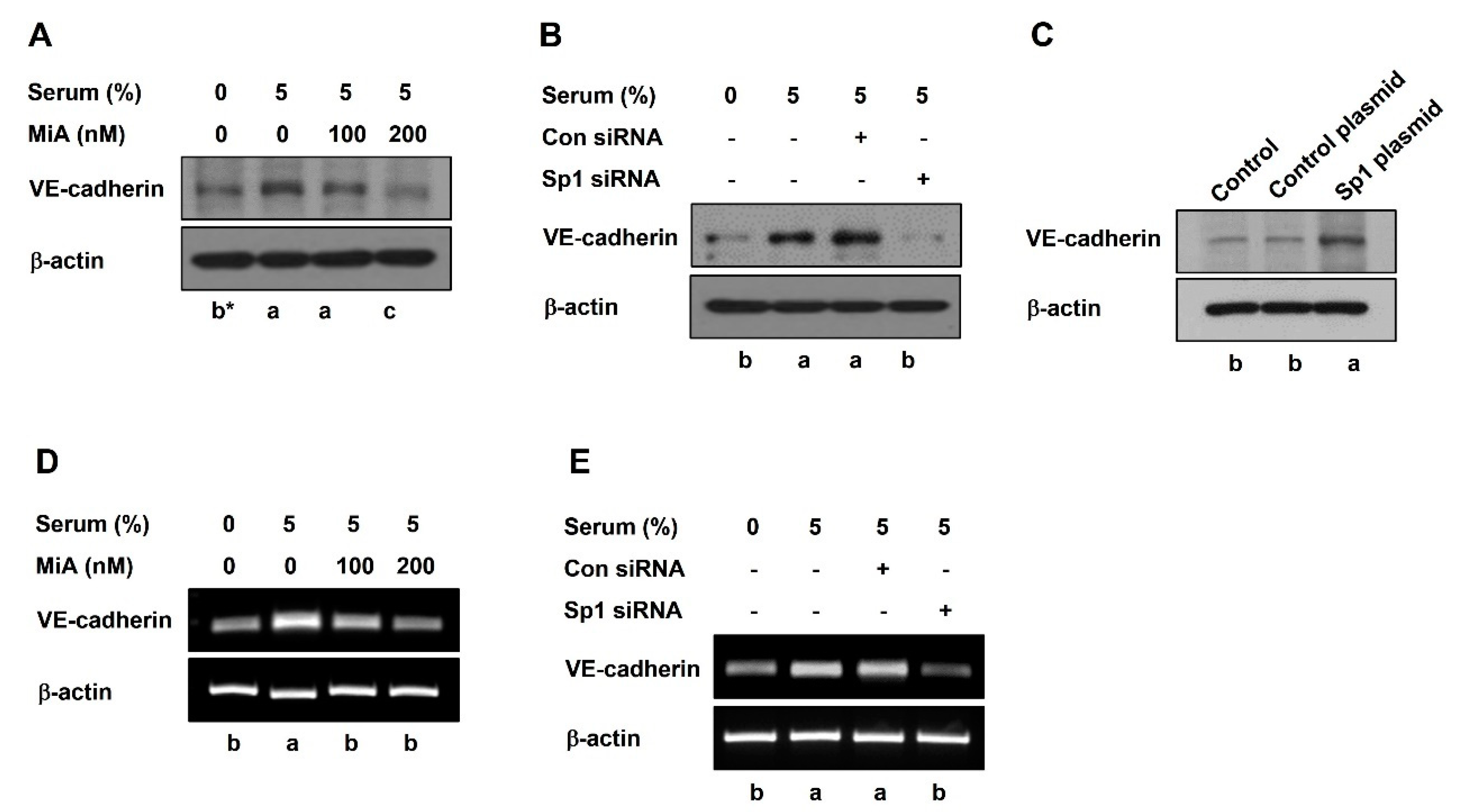
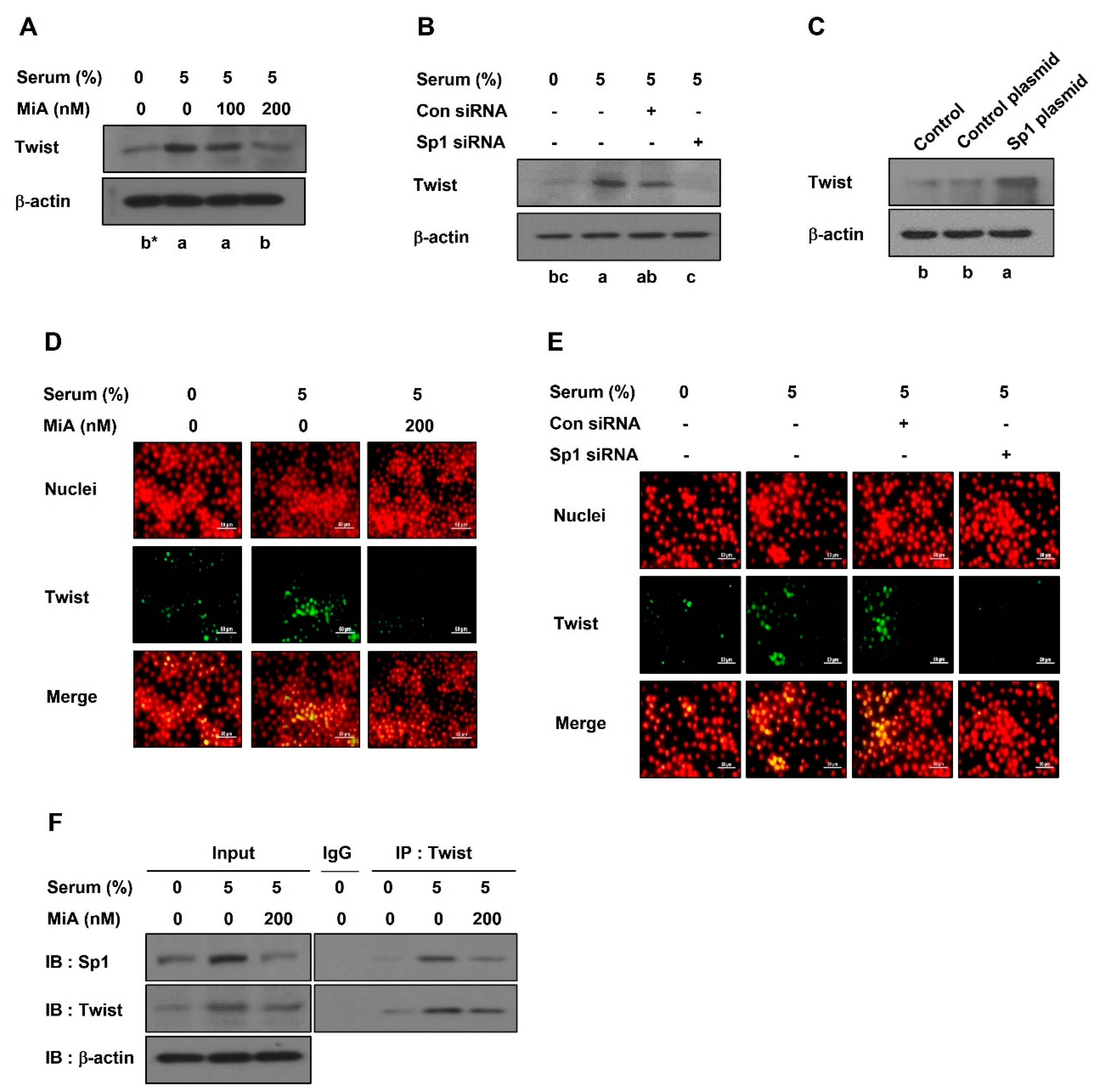
2.2. Sp1 Upregulates VE-Cadherin Expression through the Nuclear Twist in PC-3 Cells
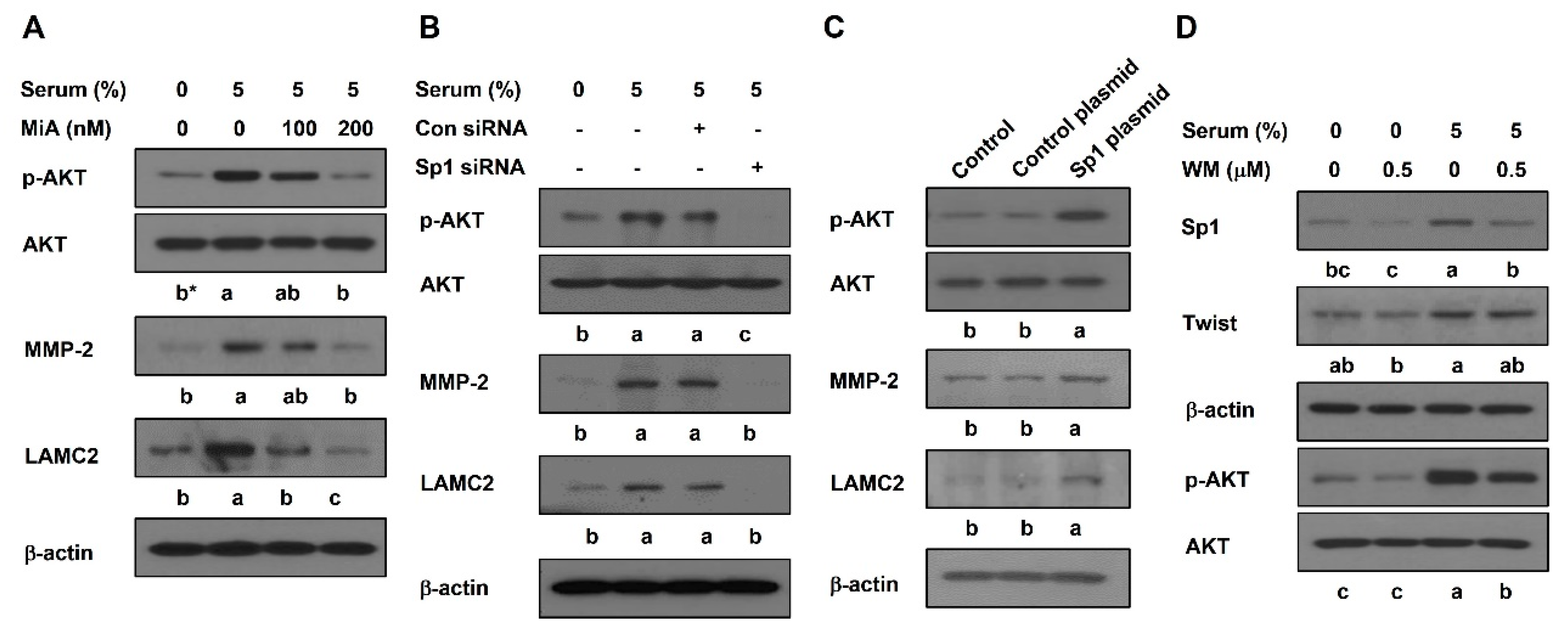
2.3. Sp1 Promotes the Activation of AKT Pathway in PC-3 Cells
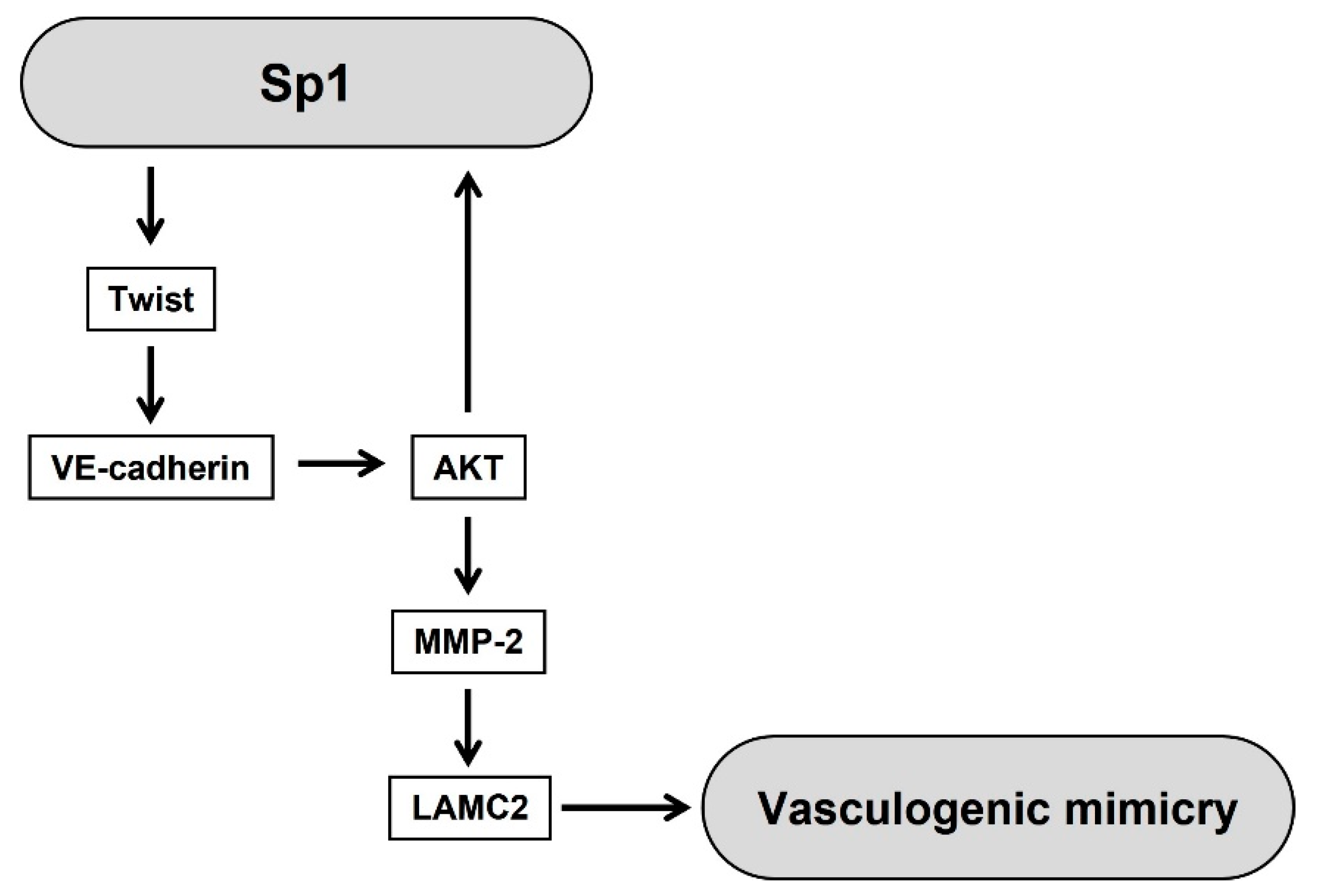
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Sp1 Silencing by Small Interfering RNA (siRNA)
4.3. Sp1 Overexpression by CRISPR Activation Plasmid
4.4. Cell Viability Assay
4.5. Three-Dimensional (3D) Culture VM Tube Formation Assay
4.6. Western Blot Analysis
4.7. Isolation of RNA and Reverse Transcriptase Polymerase Chain Reaction (RT-PCR)
4.8. Immunofluorescence Assay
4.9. Co-Immunoprecipitation (Co-IP)
4.10. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Madu, C.O.; Lu, Y. Novel diagnostic biomarkers for prostate cancer. J. Cancer 2010, 1, 150. [Google Scholar] [CrossRef] [PubMed]
- Krakhmal, N.V.; Zavyalova, M.; Denisov, E.; Vtorushin, S.; Perelmuter, V.J.A.N. Cancer invasion: Patterns and mechanisms. Acta Nat. 2015, 7, 17–28. [Google Scholar] [CrossRef]
- Lima, A.R.; de Lourdes Bastos, M.; Carvalho, M.; de Pinho, P.G. Biomarker discovery in human prostate cancer: An update in metabolomics studies. Transl. Oncol. 2016, 9, 357–370. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.S.; Santos, J.M.; Hussain, F.J.B. Aggressive prostate cancer cell nuclei have reduced stiffness. Biomicrofluidics 2018, 12, 014102. [Google Scholar] [CrossRef]
- Font-Clos, F.; Zapperi, S.; La Porta, C.A.J.I. Blood flow contributions to cancer metastasis. iScience 2020, 23, 101073. [Google Scholar] [CrossRef]
- Maniotis, A.J.; Folberg, R.; Hess, A.; Seftor, E.A.; Gardner, L.M.; Pe’er, J.; Trent, J.M.; Meltzer, P.S.; Hendrix, M.J.C. Vascular channel formation by human melanoma cells in vivo and in vitro: Vasculogenic mimicry. Am. J. Pathol. 1999, 155, 739–752. [Google Scholar] [CrossRef]
- Qiao, L.; Liang, N.; Zhang, J.; Xie, J.; Liu, F.; Xu, D.; Yu, X.; Tian, Y. Advanced research on vasculogenic mimicry in cancer. J. Cell. Mol. Med. 2015, 19, 315–326. [Google Scholar] [CrossRef]
- Seftor, R.E.; Hess, A.R.; Seftor, E.A.; Kirschmann, D.A.; Hardy, K.M.; Margaryan, N.V.; Hendrix, M.J. Tumor cell vasculogenic mimicry: From controversy to therapeutic promise. Am. J. Pathol. 2012, 181, 1115–1125. [Google Scholar] [CrossRef]
- Ge, H.; Luo, H. Overview of advances in vasculogenic mimicry—A potential target for tumor therapy. Cancer Manag. Res. 2018, 10, 2429. [Google Scholar] [CrossRef]
- Lim, D.; Do, Y.; Kwon, B.S.; Chang, W.; Lee, M.-S.; Kim, J.; Cho, J.G. Angiogenesis and vasculogenic mimicry as therapeutic targets in ovarian cancer. BMB Rep. 2020, 53, 291. [Google Scholar] [CrossRef] [PubMed]
- Itatani, Y.; Kawada, K.; Yamamoto, T.; Sakai, Y. Resistance to Anti-Angiogenic Therapy in Cancer-Alterations to Anti-VEGF Pathway. Int. J. Mol. Sci. 2018, 19, 1232. [Google Scholar] [CrossRef] [PubMed]
- Ellis, L.M.; Fidler, I.J. Finding the tumor copycat. Therapy fails, patients don’t. Nat. Med. 2010, 16, 974–975. [Google Scholar] [CrossRef] [PubMed]
- Dome, B.; Timar, J.; Dobos, J.; Meszaros, L.; Raso, E.; Paku, S.; Kenessey, I.; Ostoros, G.; Magyar, M.; Ladanyi, A. Identification and clinical significance of circulating endothelial progenitor cells in human non–small cell lung cancer. Cancer Res. 2006, 66, 7341–7347. [Google Scholar] [CrossRef]
- Zhang, J.; Qiao, L.; Liang, N.; Xie, J.; Luo, H.; Deng, G.; Zhang, J. Vasculogenic mimicry and tumor metastasis. J. BUON 2016, 21, 533–541. [Google Scholar] [PubMed]
- Cao, Z.; Bao, M.; Miele, L.; Sarkar, F.H.; Wang, Z.; Zhou, Q. Tumour vasculogenic mimicry is associated with poor prognosis of human cancer patients: A systemic review and meta-analysis. Eur. J. Cancer 2013, 49, 3914–3923. [Google Scholar] [CrossRef]
- Liu, R.; Yang, K.; Meng, C.; Zhang, Z.; Xu, Y. Vasculogenic mimicry is a marker of poor prognosis in prostate cancer. Cancer Biol. Ther. 2012, 13, 527–533. [Google Scholar] [CrossRef]
- Wang, L.; Wei, D.; Huang, S.; Peng, Z.; Le, X.; Wu, T.T.; Yao, J.; Ajani, J.; Xie, K. Transcription factor Sp1 expression is a significant predictor of survival in human gastric cancer. Clin. Cancer Res. 2003, 9, 6371–6380. [Google Scholar]
- Vellingiri, B.; Iyer, M.; Devi Subramaniam, M.; Jayaramayya, K.; Siama, Z.; Giridharan, B.; Narayanasamy, A.; Abdal Dayem, A.; Cho, S.G. Understanding the Role of the Transcription Factor Sp1 in Ovarian Cancer: From Theory to Practice. Int. J. Mol. Sci. 2020, 21, 1153. [Google Scholar] [CrossRef]
- Song, J.; Ugai, H.; Nakata-Tsutsui, H.; Kishikawa, S.; Suzuki, E.; Murata, T.; Yokoyama, K.K. Transcriptional regulation by zinc-finger proteins Sp1 and MAZ involves interactions with the same cis-elements. Int. J. Mol. Sci. 2003, 11, 547–553. [Google Scholar] [CrossRef]
- Sankpal, U.T.; Goodison, S.; Abdelrahim, M.; Basha, R. Targeting Sp1 transcription factors in prostate cancer therapy. Med. Chem. 2011, 7, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Lin, H.; Pan, J.; Mo, C.; Zhang, F.; Huang, B.; Wang, Z.; Chen, X.; Zhuang, J.; Wang, D.; et al. Vasculogenic Mimicry in Prostate Cancer: The Roles of EphA2 and PI3K. J. Cancer 2016, 7, 1114–1124. [Google Scholar] [CrossRef] [PubMed]
- Yeo, C.; Lee, H.J.; Lee, E.O. Serum promotes vasculogenic mimicry through the EphA2/VE-cadherin/AKT pathway in PC-3 human prostate cancer cells. Life Sci. 2019, 221, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, J.; Zhou, H.; Fan, G.; Li, Q. Molecular Mechanisms and Anticancer Therapeutic Strategies in Vasculogenic Mimicry. J. Cancer 2019, 10, 6327–6340. [Google Scholar] [CrossRef]
- Hendrix, M.J.; Seftor, E.A.; Meltzer, P.S.; Gardner, L.M.; Hess, A.R.; Kirschmann, D.A.; Schatteman, G.C.; Seftor, R.E. Expression and functional significance of VE-cadherin in aggressive human melanoma cells: Role in vasculogenic mimicry. Proc. Natl. Acad. Sci. USA 2001, 98, 8018–8023. [Google Scholar] [CrossRef]
- Sun, B.; Zhang, D.; Zhao, N.; Zhao, X. Epithelial-to-endothelial transition and cancer stem cells: Two cornerstones of vasculogenic mimicry in malignant tumors. Oncotarget 2017, 8, 30502. [Google Scholar] [CrossRef]
- Maniotis, A.J.; Chen, X.; Garcia, C.; DeChristopher, P.J.; Wu, D.; Pe’er, J.; Folberg, R. Control of melanoma morphogenesis, endothelial survival, and perfusion by extracellular matrix. Lab. Investig. 2002, 82, 1031–1043. [Google Scholar] [CrossRef][Green Version]
- Delgado-Bellido, D.; Serrano-Saenz, S.; Fernández-Cortés, M.; Oliver, F.J. Vasculogenic mimicry signaling revisited: Focus on non-vascular VE-cadherin. Mol. Cancer 2017, 16, 1–14. [Google Scholar] [CrossRef]
- Gory, S.; Vernet, M.; Laurent, M.; Dejana, E.; Dalmon, J.; Huber, P.J.B. The vascular endothelial-cadherin promoter directs endothelial-specific expression in transgenic mice. Blood 1999, 93, 184–192. [Google Scholar] [CrossRef]
- Gory, S.; Dalmon, J.; Prandini, M.-H.; Kortulewski, T.; de Launoit, Y.; Huber, P. Requirement of a GT box (Sp1 site) and two Ets binding sites for vascular endothelial cadherin gene transcription. J. Biol. Chem. 1998, 273, 6750–6755. [Google Scholar] [CrossRef]
- Yang, J.; Zhu, D.-M.; Zhou, X.-G.; Yin, N.; Zhang, Y.; Zhang, Z.-X.; Li, D.-C.; Zhou, J. HIF-2α promotes the formation of vasculogenic mimicry in pancreatic cancer by regulating the binding of Twist1 to the VE-cadherin promoter. Oncotarget 2017, 8, 47801. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Zhao, N.; Zhao, X.L.; Gu, Q.; Zhang, S.W.; Che, N.; Wang, X.H.; Du, J.; Liu, Y.X.; Sun, B. Expression and functional significance of Twist1 in hepatocellular carcinoma: Its role in vasculogenic mimicry. Hepatology 2010, 51, 545–556. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Sun, H.; Sun, B.; Zhu, D.; Zhao, X.; Wang, Y.; Gu, Q.; Dong, X.; Liu, F.; Zhang, Y. miR-27a-3p suppresses tumor metastasis and VM by down-regulating VE-cadherin expression and inhibiting EMT: An essential role for Twist-1 in HCC. Sci. Rep. 2016, 6, 23091. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.J.; Kwon, Y.G. Roles of YAP in mediating endothelial cell junctional stability and vascular remodeling. BMB Rep. 2015, 48, 429–430. [Google Scholar] [CrossRef]
- Szymonowicz, K.; Oeck, S.; Malewicz, N.M.; Jendrossek, V. New Insights into Protein Kinase B/Akt Signaling: Role of Localized Akt Activation and Compartment-Specific Target Proteins for the Cellular Radiation Response. Cancers 2018, 10, 78. [Google Scholar] [CrossRef]
- Li, X.; Fu, Y.; Xia, X.; Zhang, X.; Xiao, K.; Zhuang, X.; Zhang, Y. Knockdown of SP1/Syncytin1 axis inhibits the proliferation and metastasis through the AKT and ERK1/2 signaling pathways in non-small cell lung cancer. Cancer Med. 2019, 8, 5750–5759. [Google Scholar] [CrossRef]
- Pore, N.; Liu, S.; Shu, H.K.; Li, B.; Haas-Kogan, D.; Stokoe, D.; Milanini-Mongiat, J.; Pages, G.; O’Rourke, D.M.; Bernhard, E.; et al. Sp1 is involved in Akt-mediated induction of VEGF expression through an HIF-1-independent mechanism. Mol. Biol. Cell 2004, 15, 4841–4853. [Google Scholar] [CrossRef]
- Kim, J.S.; Kang, C.G.; Kim, S.H.; Lee, E.O. Rhapontigenin suppresses cell migration and invasion by inhibiting the PI3K-dependent Rac1 signaling pathway in MDA-MB-231 human breast cancer cells. J. Nat. Prod. 2014, 77, 1135–1139. [Google Scholar] [CrossRef]
- Im, E.; Yeo, C.; Lee, H.J.; Lee, E.O. Dihydroartemisinin induced caspase-dependent apoptosis through inhibiting the specificity protein 1 pathway in hepatocellular carcinoma SK-Hep-1 cells. Life Sci. 2018, 192, 286–292. [Google Scholar] [CrossRef]
- Kang, C.G.; Lee, H.J.; Kim, S.H.; Lee, E.O. Zerumbone Suppresses Osteopontin-Induced Cell Invasion Through Inhibiting the FAK/AKT/ROCK Pathway in Human Non-Small Cell Lung Cancer A549 Cells. J. Nat. Prod. 2016, 79, 156–160. [Google Scholar] [CrossRef]
- Yeo, C.; Han, D.S.; Lee, H.J.; Lee, E.O. Epigallocatechin-3-Gallate Suppresses Vasculogenic Mimicry through Inhibiting the Twist/VE-Cadherin/AKT Pathway in Human Prostate Cancer PC-3 Cells. Int. J. Mol. Sci. 2020, 21, 439. [Google Scholar] [CrossRef] [PubMed]
| Antibody | Company | Dilution | Product No. |
|---|---|---|---|
| Sp1 | Santa Cruz | 1:1000 | SC-420 |
| β-actin | Sigma-Aldrich | 1:20,000 | A5316 |
| pAKT | CST | 1:3000 | 4060 |
| AKT | CST | 1:5000 | 4691 |
| MMP-2 | Abcam | 1:1000 | ab86607 |
| LAMC2 | Abcam | 1:1000 | ab96327 |
| VE-cadherin | Abgent | 1:1000 | AP2724a |
| Twist | Abcam | 1:1000 | ab50887 |
| goat anti-rabbit IgG-HRP | CST | 1:5000 | 7074P2 |
| goat anti-mouse IgG-HRP | Bio-Rad | 1:5000 | STAR120P |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, D.-S.; Lee, E.-O. Sp1 Plays a Key Role in Vasculogenic Mimicry of Human Prostate Cancer Cells. Int. J. Mol. Sci. 2022, 23, 1321. https://doi.org/10.3390/ijms23031321
Han D-S, Lee E-O. Sp1 Plays a Key Role in Vasculogenic Mimicry of Human Prostate Cancer Cells. International Journal of Molecular Sciences. 2022; 23(3):1321. https://doi.org/10.3390/ijms23031321
Chicago/Turabian StyleHan, Deok-Soo, and Eun-Ok Lee. 2022. "Sp1 Plays a Key Role in Vasculogenic Mimicry of Human Prostate Cancer Cells" International Journal of Molecular Sciences 23, no. 3: 1321. https://doi.org/10.3390/ijms23031321
APA StyleHan, D.-S., & Lee, E.-O. (2022). Sp1 Plays a Key Role in Vasculogenic Mimicry of Human Prostate Cancer Cells. International Journal of Molecular Sciences, 23(3), 1321. https://doi.org/10.3390/ijms23031321





