Anion-Controlled Synthesis of Novel Guanidine-Substituted Oxanorbornanes
Abstract
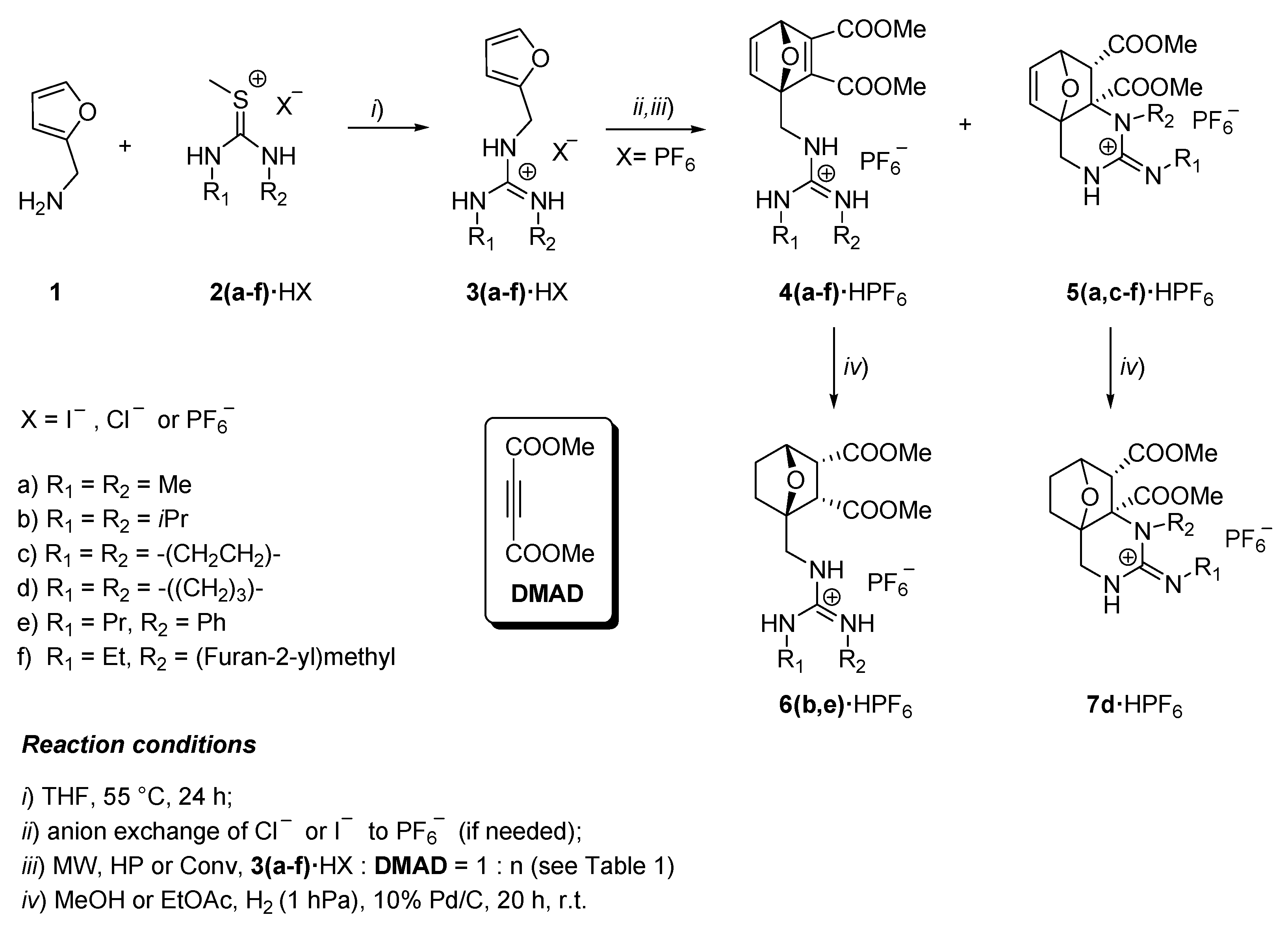
1. Introduction
2. Results and Discussion
2.1. Mechanism of the CA/IMC Tandem Process
2.2. Halides as Nucleophiles. Aza-Michael/Cyclization
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moreno-Vargas, A.J.; Vogel, P. Synthesis of 7-Oxabicyclo[2.2.1]heptane and Derivatives. In Topics in Heterocyclic Chemistry 35. Synthesis of Saturated Oxygenated Heterocycles I; Cossy, J., Ed.; Springer-Verlag: Berlin/Heidelberg, Germany, 2014. [Google Scholar] [CrossRef]
- Vogel, P. Total Asymmetric Synthesis of Glycomimetics and Polypropionates of Biological Interest. Chimia 2008, 62, 519–524. [Google Scholar] [CrossRef]
- Hackenberger, C.P.R.; Schiffers, I.; Runsink, J.; Bolm, C. General Synthesis of Unsymmetrical Norbornane Scaffolds as Inducers for Hydrogen Bond Interactions in Peptides. J. Org. Chem. 2004, 69, 739–743. [Google Scholar] [CrossRef] [PubMed]
- Saroj, S.; Janni, D.S.; Ummadi, C.R.; Manheri, M.K. Functionalizable oxanorbornane-based head-group in the design of new Non-ionic amphiphiles and their drug delivery properties. Mat. Sci. Eng. C 2020, 112, 110857. [Google Scholar] [CrossRef] [PubMed]
- Hennig, A.; Gabriel, G.J.; Tew, G.N.; Matile, S. Stimuli-Responsive Polyguanidino-Oxanorbornene Membrane Transporters as Multicomponent Sensors in Complex Matrices. J. Am. Chem. Soc. 2008, 130, 10338–10344. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Dam, M.A.; Ono, K.; Mal, A.; Shen, H.; Nutt, S.R.; Sheran, K.; Wudl, F. A thermally re-mendable cross-linked polymeric material. Science 2002, 295, 1698–1702. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.I.; Halder, S.; Gunjan, S.B.; Prasad, T. A review on Diels-Alder based self-healing polymer composites. IOP Conf. Ser. Mater. Sci. Eng. 2018, 377, 012007. [Google Scholar] [CrossRef]
- Warrener, R.N.; Margetić, D.; Sun, G.; Amarasekara, A.S.; Foley, P.; Butler, D.N.; Russell, R.A. Molecular topology: The synthesis of a new class of rigid arc-shaped spacer molecules based on syn-facially fused norbornanes and 7-heteronorbornanes in which heterobridges are used to govern backbone curvature. Tetrahedron Lett. 1999, 40, 4111–4114. [Google Scholar] [CrossRef]
- Briš, A.; Trošelj, P.; Margetić, D. Cooperativity in Binding of Aliphatic Diamines by Bis(Zn(II)porphyrin) Receptor with Moderately Flexible Linkers. Croat. Chem. Acta 2017, 90, 667–677. [Google Scholar] [CrossRef]
- Robson, R.N.; Hay, B.P.; Pfeffer, F.M. To Cooperate or Not: The Role of Central Functionality in Bisthiourea [6]polynorbornane Hosts. Eur. J. Org. Chem. 2019, 2019, 6720–6727. [Google Scholar] [CrossRef]
- Selig, P. Guanidine Organocatalysis. Synthesis 2013, 45, 703–718. [Google Scholar] [CrossRef]
- Chou, H.-C.; Leow, D.; Tan, C.-H. Recent Advances in Chiral Guanidine-Catalyzed Enantioselective Reactions. Chem. Asian J. 2019, 14, 3803–3822. [Google Scholar] [CrossRef]
- Schug, K.A.; Lindner, W. Noncovalent Binding between Guanidinium and Anionic Groups: Focus on Biological- and Synthetic-Based Arginine/Guanidinium Interactions with Phosph[on]ate and Sulf[on]ate Residues. Chem. Rev. 2005, 105, 67–113. [Google Scholar] [CrossRef]
- Schmuck, C. How to improve guanidinium cations for oxoanion binding in aqueous solution?: The design of artificial peptide receptors. Coord. Chem. Rev. 2006, 250, 3053–3067. [Google Scholar] [CrossRef]
- Hargrove, A.E.; Nieto, S.; Zhang, T.; Sessler, J.L.; Anslyn, E.V. Artificial Receptors for the Recognition of Phosphorylated Molecules. Chem. Rev. 2011, 111, 6603–6782. [Google Scholar] [CrossRef] [PubMed]
- Acheson, R.M.; Wallis, J.D. Addition reactions of heterocyclic compounds. Part 74. Products from dimethyl acetylenedicarboxylate with thiourea, thioamide, and guanidine derivatives. J. Chem. Soc. Perkin Trans. 1981, 1, 415–422. [Google Scholar] [CrossRef]
- Shikhaliev, K.S.; Kovygin, Y.A.; Potapov, A.Y.; Sabynin, A.L.; Kosheleva, E.A. Recyclization of maleimides with N-carboximide amides. Russ. Chem. Bull. Int. Ed. 2017, 66, 86–90. [Google Scholar] [CrossRef]
- Baum, J.; Scholz, D.; Tataruch, F.; Viehe, H.G. New syntheses of amino-substituted 2-azabutadienes. Chimia 1975, 29, 514–515. [Google Scholar]
- Yavari, I.; Amirahmadia, A.; Halvagar, M.R. A Synthesis of Functionalized Thiazoles and Pyrimidine-4(3H)-thiones from 1,1,3,3-Tetramethylguanidine, Acetylenic Esters, and Aryl Isothiocyanates. Synlett 2017, 28, 2629–2632. [Google Scholar] [CrossRef]
- Neochoritis, C.G.; Zarganes–Tzitzikas, T.; Stephanidou-Stephanatou, J. Dimethyl Acetylenedicarboxylate: A Versatile Tool in Organic Synthesis. Synthesis 2014, 46, 0537–0585. [Google Scholar] [CrossRef]
- Horvath, A. Catalysis and regioselectivity in the Michael addition of azoles. Kinetic vs. thermodynamic control. Tetrahedron Lett. 1996, 37, 4423–4426. [Google Scholar] [CrossRef]
- Shen, J.; Nguyen, T.T.; Goh, Y.-P.; Ye, W.; Fu, X.; Xu, J.; Tan, C.-H. Chiral Bicyclic Guanidine-Catalyzed Enantioselective Reactions of Anthrones. J. Am. Chem. Soc. 2006, 128, 13692–13693. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhao, J.; Zeng, L.; Hu, W. Organocatalytic Asymmetric Domino Aza-Michael–Mannich Reaction: Synthesis of Tetrahydroimidazopyrimidine Derivatives. J. Org. Chem. 2011, 76, 8064–8069. [Google Scholar] [CrossRef] [PubMed]
- Barešić, L.; Margetić, D.; Glasovac, Z. Cycloaddition of Thiourea- and Guanidine-Substituted Furans to Dienophiles: A Comparison of the Environmentally-Friendly Methods. Chem. Proc. 2021, 3, 57. [Google Scholar] [CrossRef]
- Barlow, M.G.; Suliman, N.N.E.; Tipping, A.E. A high-yield synthesis of 3-carboethoxy-4-trifluoromethylfuran and some Diels-Alder reactions of this furoate with acetylenic dienophiles. J. Fluor. Chem. 1995, 70, 59–69. [Google Scholar] [CrossRef]
- Zhang, P.; Liao, X.; Ma, C.; Li, Q.; Li, A.; He, Y. Chemoenzymatic Conversion of Corncob to Furfurylamine via Tandem Catalysis with Tin-Based Solid Acid and Transaminase Biocatalyst. ACS Sustain. Chem. Eng. 2019, 7, 17636–17642. [Google Scholar] [CrossRef]
- Aoyagi, N.; Endo, T. Synthesis of five- and six-membered cyclic guanidines by guanylation with isothiouronium iodides and amines under mild conditions. Synth. Commun. 2017, 47, 442–448. [Google Scholar] [CrossRef]
- Oganesyan, A.; Cruz, I.A.; Amador, R.B.; Sorto, N.A.; Lozano, J.; Godinez, C.E.; Anguiano, J.; Pace, H.; Sabih, G.; Gutierrez, C.G. High Yield Selective Acylation of Polyamines: Proton as Protecting Group. Org. Lett. 2007, 9, 4967–4970. [Google Scholar] [CrossRef]
- Huang, C.; Yin, Y.; Guo, J.; Wang, J.; Fan, B.; Yang, L. A facile synthesis of β-amino carbonyl compounds through an aza-Michael addition reaction under solvent-free conditions. RSC Adv. 2014, 4, 10188–10195. [Google Scholar] [CrossRef]
- Margetić, D. High Pressure Organic Synthesis; Verlag Walter de Gruyter: Berlin, Germany, 2019; ISBN 978-3-11-055602-5. [Google Scholar] [CrossRef]
- Kiesewetter, M.K.; Scholten, M.D.; Kirn, N.; Weber, R.L.; Hedrick, J.L.; Waymouth, R.M. Cyclic Guanidine Organic Catalysts: What Is Magic About Triazabicyclodecene? J. Org. Chem. 2009, 74, 9490–9496. [Google Scholar] [CrossRef]
- Ghobril, C.; Hammar, P.; Kodepelly, S.; Spiess, B.; Wagner, A.; Himo, F.; Baati, R. Structure–Reactivity Relationship Studies for Guanidine-Organocatalyzed Direct Intramolecular Aldolization of Ketoaldehydes. ChemCatChem 2010, 2, 1573–1581. [Google Scholar] [CrossRef]
- Kaljurand, I.; Rodima, T.; Pihl, A.; Mäemets, V.; Leito, I.; Koppel, I.A.; Mishima, M. Acid-Base Equilibria in Nonpolar Media. 4. Extension of the Self-Consistent Basicity Scale in THF Medium. Gas-Phase Basicities of Phosphazenes. J. Org. Chem. 2003, 68, 9988–9993. [Google Scholar] [CrossRef] [PubMed]
- Kaljurand, I.; Kütt, A.; Sooväli, L.; Rodima, T.; Mäemets, V.; Leito, I.; Koppel, I.A. Extension of the Self-Consistent Spectrophotometric Basicity Scale in Acetonitrile to a Full Span of 28 pKa Units: Unification of Different Basicity Scales. J. Org. Chem. 2005, 70, 1019–1028. [Google Scholar] [CrossRef]
- Zall, C.M.; Linehan, J.C.; Appel, A.M. A Molecular Copper Catalyst for Hydrogenation of CO2 to Formate. ACS Catal. 2015, 5, 5301–5305. [Google Scholar] [CrossRef]
- Yepes, D.; Valenzuela, J.; Martínez-Araya, J.I.; Pérez, P.; Jaque, P. Effect of Exchange-Correlation Functional on the Synchronicity/Nonsynchronicity in Bond Formation in Diels-Alder Reactions: A Reaction Force Constant Analysis. Phys. Chem. Chem. Phys. 2019, 21, 7412–7428. [Google Scholar] [CrossRef] [PubMed]
- Mezei, P.D.; Csonka, G.L.; Kallay, M. Accurate Diels–Alder Reaction Energies from Efficient Density Functional Calculations. J. Chem. Theory Comput. 2015, 11, 2879–2888. [Google Scholar] [CrossRef] [PubMed]
- Fan, M.-J.; Lia, G.-Q.; Liang, Y.-M. DABCO catalyzed reaction of various nucleophiles with activated alkynes leading to the formation of alkenoic acid esters, 1,4-dioxane, morpholine, and piperazinone derivatives. Tetrahedron 2006, 62, 6782–6791. [Google Scholar] [CrossRef]
- Billetter, H.; Pantenburg, I.; Ruschewitz, U. Lithium hydrogen iodo-fumarate monohydrate. Acta Cryst. 2005, E61, m1857–m1859. [Google Scholar] [CrossRef]
- Billetter, H.; Pantenburg, I.; Ruschewitz, U. Copper(II) chloro-fumarate trihydrate. Acta Cryst. 2006, E62, m881–m883. [Google Scholar] [CrossRef]
- Shestakov, A.S.; Sidorenko, O.E.; Shikhaliev, K.S. Synthesis of 2-iminoimidazolidin-4-one derivatives by cyclization of 2-aryl-1-(4,6-dimethylpyrimidin-2-yl)guanidines with ethyl bromoacetate, dimethyl acetylenedicarboxylate, and maleic anhydride. Russ. Chem. Bull. Int. Ed. 2007, 56, 1423–1430. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure and refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian09. Rev D. 01; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian16. Rev C. 01; Gaussian, Inc.: Wallingford, CT, USA, 2019. [Google Scholar]
- Glasovac, Z.; Eckert-Maksić, M.; Maksić, Z.B. Basicity of organic bases and superbases in acetonitrile by the polarized continuum model and DFT calculations. New J. Chem. 2009, 33, 588–597. [Google Scholar] [CrossRef]
- Glasovac, Z.; Kovačević, B. Modeling pKa of the Brønsted Bases as an Approach to the Gibbs Energy of the Proton in Acetonitrile. Int. J. Mol. Sci. 2022, 23, 10576. [Google Scholar] [CrossRef] [PubMed]
- Marenich, A.V.; Cramer, C.J.; Truhlar, D.G. Universal Solvation Model Based on Solute Electron Density and on a Continuum Model of the Solvent Defined by the Bulk Dielectric Constant and Atomic Surface Tensions. J. Phys. Chem. B 2009, 113, 6378–6396. [Google Scholar] [CrossRef] [PubMed]




| Entry | Reactant | Method a | Equiv./Time b | 4x·HPF6/ 5x·HPF6 | Conv./% (Yield/%) c,d |
|---|---|---|---|---|---|
| 1 | 3a·HPF6 | MW | 1:6/1 h | 85/15 | 90 (36 as 5a·HPF6) |
| 2 | 3a·HPF6 e | MW | 1:6/1 h | 57/43 | 98 (13 as 5a·HPF6) |
| 3 | 3b·HPF6 | MW | 1:3/1 h | 100/0 | 79 [57 as 4b·HPF6] f |
| 4 | 3b·HPF6 | MW | 1:3/4 h | 100/0 | 98 (70 as 4b·HPF6) |
| 5 | 3b·HPF6 | MW | 1:6/1h | 100/0 | 98 (72 as 4b·HPF6) |
| 6 | 3b·HPF6 e | MW | 1:6/1 h | 100/0 | 98 (71 as 6b·HPF6) |
| 7 | 3c·HPF6 | MW | 1:3/1 h | 91/9 | 83 (50 as 5c·HPF6) |
| 8 | 3c·HPF6 | Conv. | 1:6/1 h | 88/12 | 94 (30 as 5c·HPF6) [56 as 5c·HPF6] f |
| 9 | 3c·HPF6 e | MW | 1:6/1 h | 77/23 | 94 (70 as 5c·HPF6) |
| 10 | 3c·HPF6 | HP | 1:1.1/24 h | 5/95 | 20 (n/d) |
| 11 | 3c·HPF6 | HP | 1:1.1/30 days | 4/96 | 71 (n/d) |
| 12 | 3d·HPF6 | MW | 1:3/1 h | 88/12 | 73 (50 as 5d·HPF6) |
| 13 | 3d·HPF6 | MW | 1:6/1 h | 100/0 | 98 [61 as 4d·HPF6 ] f |
| 14 | 3d·HPF6 | MW | 1:6/1 h | 100/0 | 98 (65 as 7d·HPF6) |
| 15 | 3e·HPF6 | MW | 1:6/1 h | 93/7 | 94 (33 as 5e·HPF6) |
| 16 | 3e·HPF6 | MW | 1:6/1 h | 95/5 | 94 (64 as 6e·HPF6) |
| 17 | 3f·HPF6 | MW | 1:6/1 h | 96/4 | 97 (57 as 6f·HPF6) |
| 18 | 3f·HPF6 | HP | 1:1.1/24 h | 13/87 | 23 (n/d) |
| 19 | 3f·HPF6 | HP | 1:1.1/144 h | 13/87 | 82 (75 as 5f·HPF6) |
| Oxanorb. | NX | ACNpKa a | Model b |  rG(CA) c rG(CA) c |  rG(tot) c,d rG(tot) c,d |
|---|---|---|---|---|---|
| 4aH+ | N1 | 22.6 (22.4) | M11 | −10 | −96 |
| N2,3 | 23.0 (22.7) | B3-S (B3-L) | 16 (52) | −26 (23) | |
| 4bH+ | N1 | 23.3 (22.1) | M11 | −16 | −78 |
| N2,3 | 24.0 (23.1) | B3-S (B3-L) | 16 (52) | 6 (60) | |
| 4cH+ | N1 | 20.5 (20.5) | M11 | −3 | −98 |
| N2,3 | 21.4 (21.0) | B3-S (B3-L) | 16 (56) | −37 (16) | |
| 4dH+ | N1 | 22.3 (22.5) | M11 | −18 | −102 |
| N2,3 | 23.5 (22.8) | B3-S (B3-L) | 19 (55) | −22 (27) | |
| 4eH+ | N1 N2 N3 | 21.6 (21.5) 19.6 (18.8) 21.8 (21.9) | M11 B3-S (B3-L) | −18 18 (55) | −91 −14 (35) |
| 4fH+ | N1 N2 N3 | 21.8 (21.0) 23.4 (22.8) 22.6 (21.4) | M11 B3-S (B3-L) | −14 14 (46) | −92 −14 (36) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barešić, L.; Margetić, D.; Glasovac, Z. Anion-Controlled Synthesis of Novel Guanidine-Substituted Oxanorbornanes. Int. J. Mol. Sci. 2022, 23, 16036. https://doi.org/10.3390/ijms232416036
Barešić L, Margetić D, Glasovac Z. Anion-Controlled Synthesis of Novel Guanidine-Substituted Oxanorbornanes. International Journal of Molecular Sciences. 2022; 23(24):16036. https://doi.org/10.3390/ijms232416036
Chicago/Turabian StyleBarešić, Luka, Davor Margetić, and Zoran Glasovac. 2022. "Anion-Controlled Synthesis of Novel Guanidine-Substituted Oxanorbornanes" International Journal of Molecular Sciences 23, no. 24: 16036. https://doi.org/10.3390/ijms232416036
APA StyleBarešić, L., Margetić, D., & Glasovac, Z. (2022). Anion-Controlled Synthesis of Novel Guanidine-Substituted Oxanorbornanes. International Journal of Molecular Sciences, 23(24), 16036. https://doi.org/10.3390/ijms232416036







