Neuronal Calcium Sensor-1 Protects Cortical Neurons from Hyperexcitation and Ca2+ Overload during Ischemia by Protecting the Population of GABAergic Neurons
Abstract
1. Introduction
2. Results
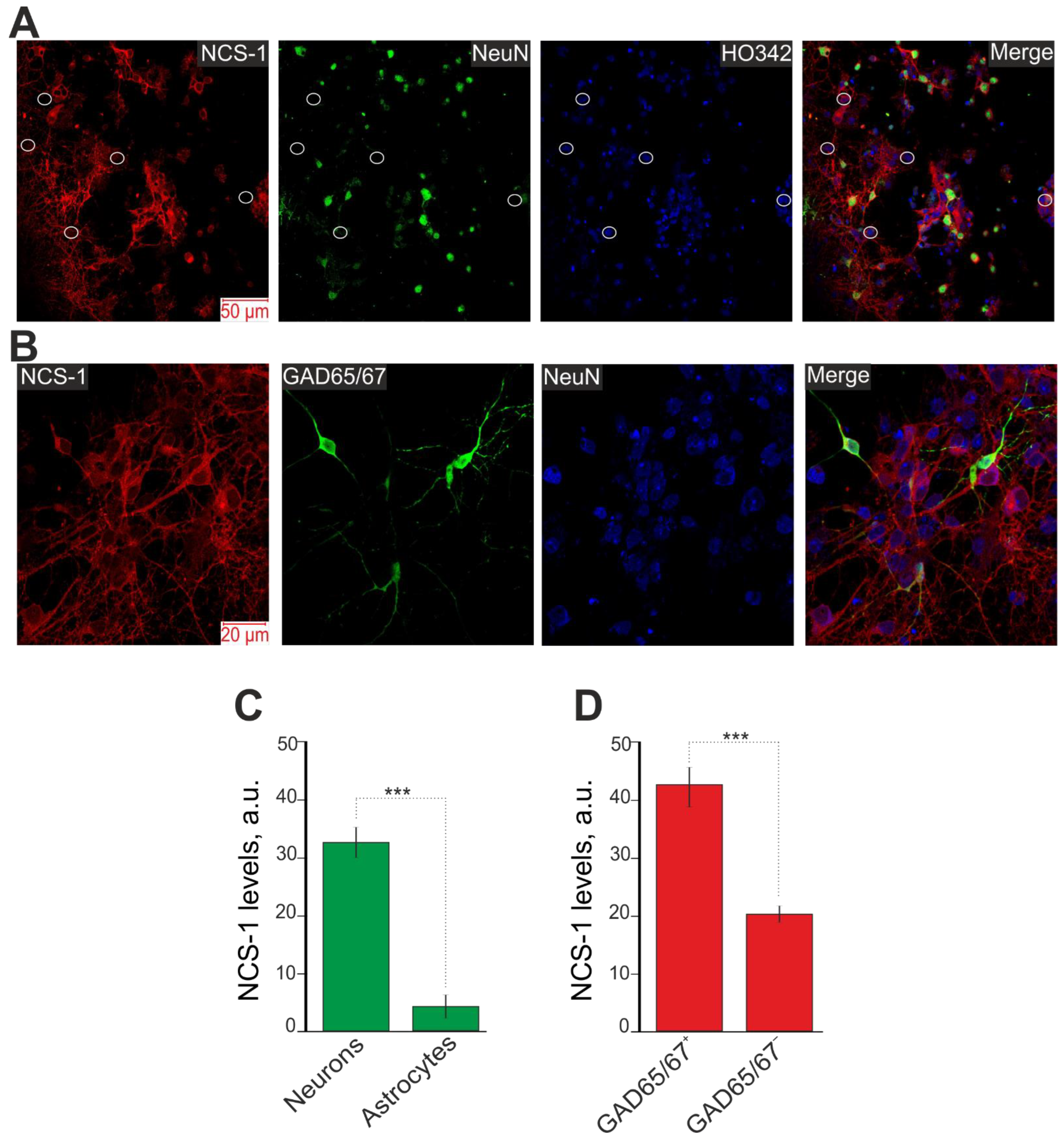
2.1. Different Expression of NCS-1 in the Cortical Cells in Culture In Vitro
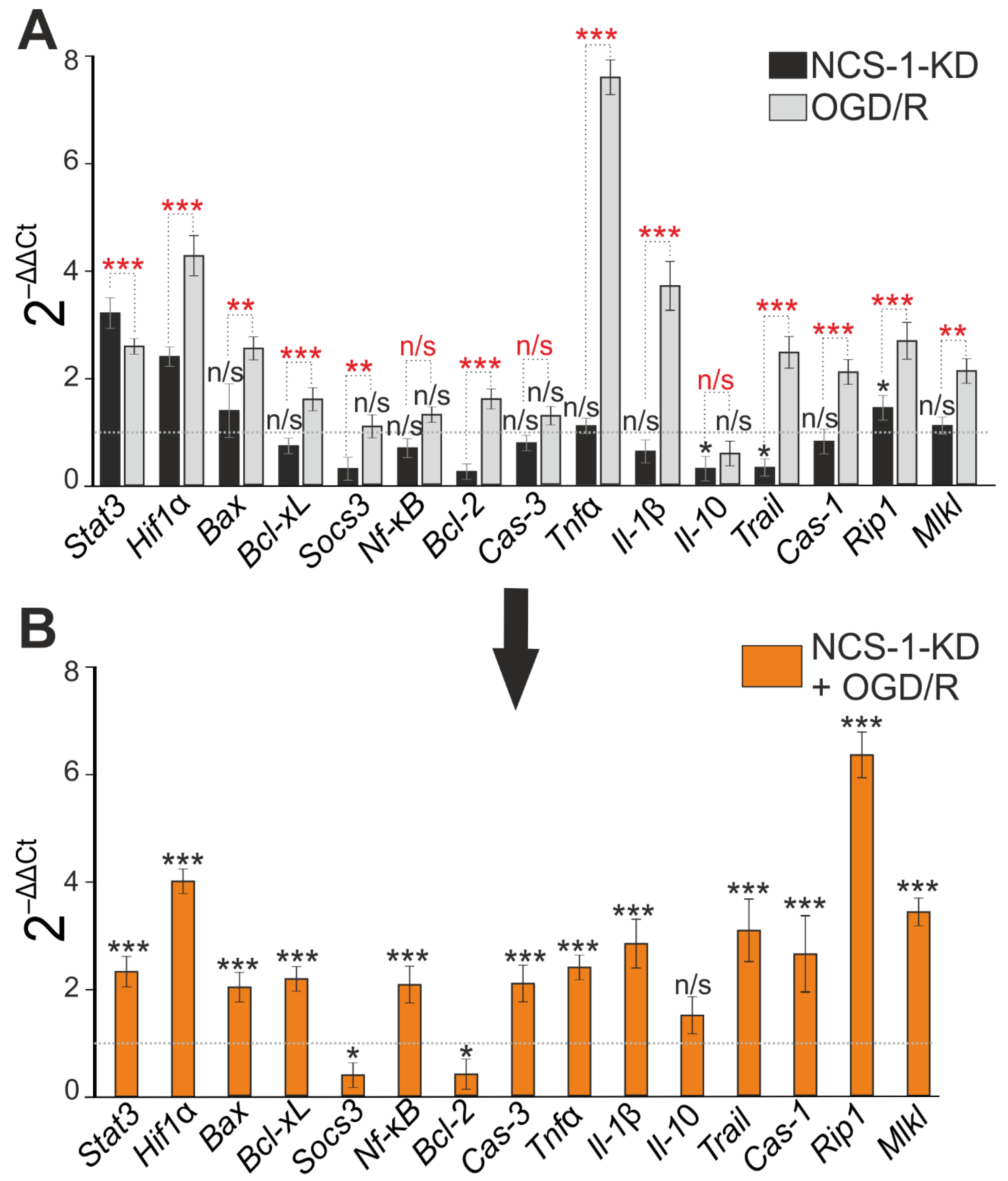
2.2. Expression of NCS-1 Protects Neurons from OGD/Reoxygenation (OGD/R)-Induced Death through the Regulation of Apoptotic and Inflammatory Protein Expression
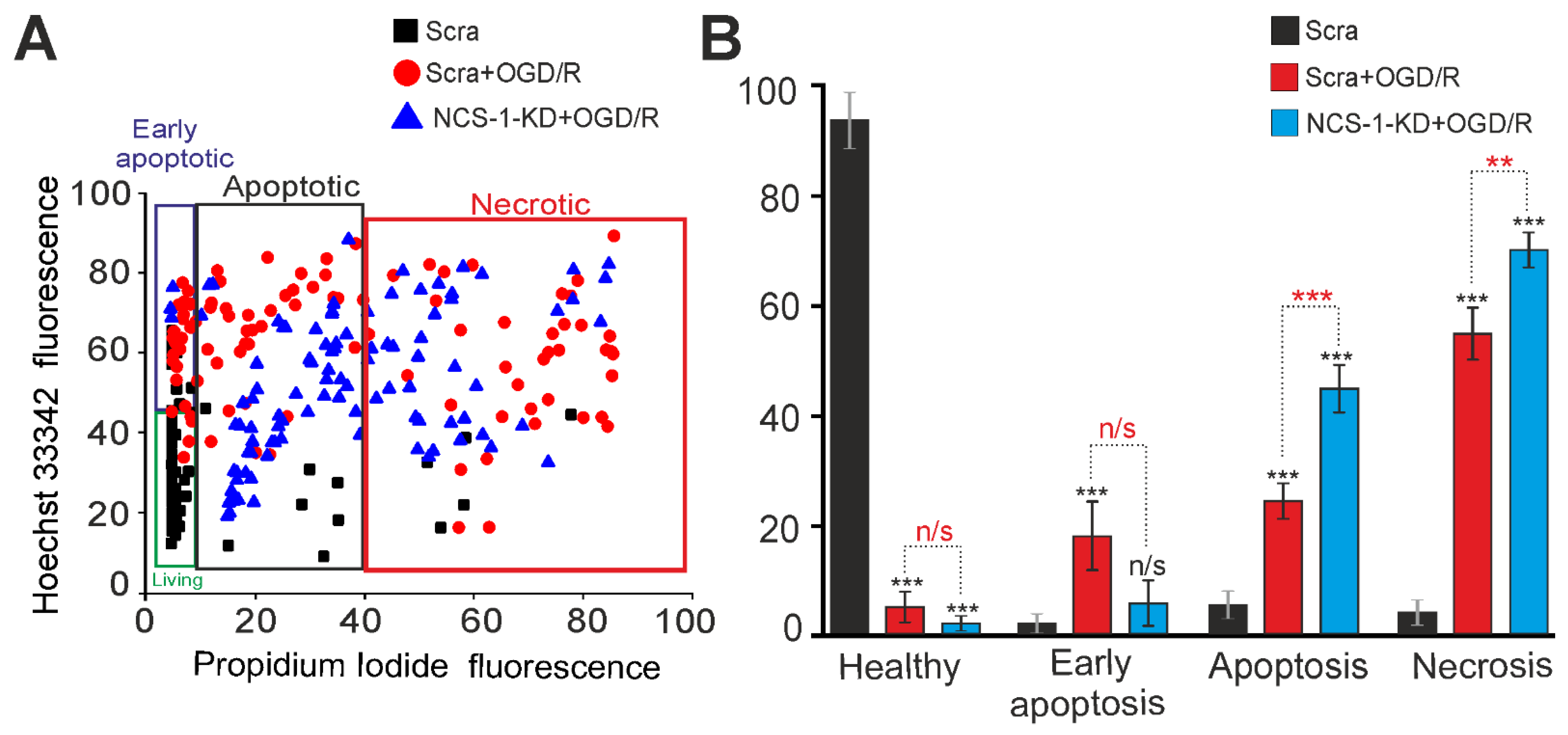
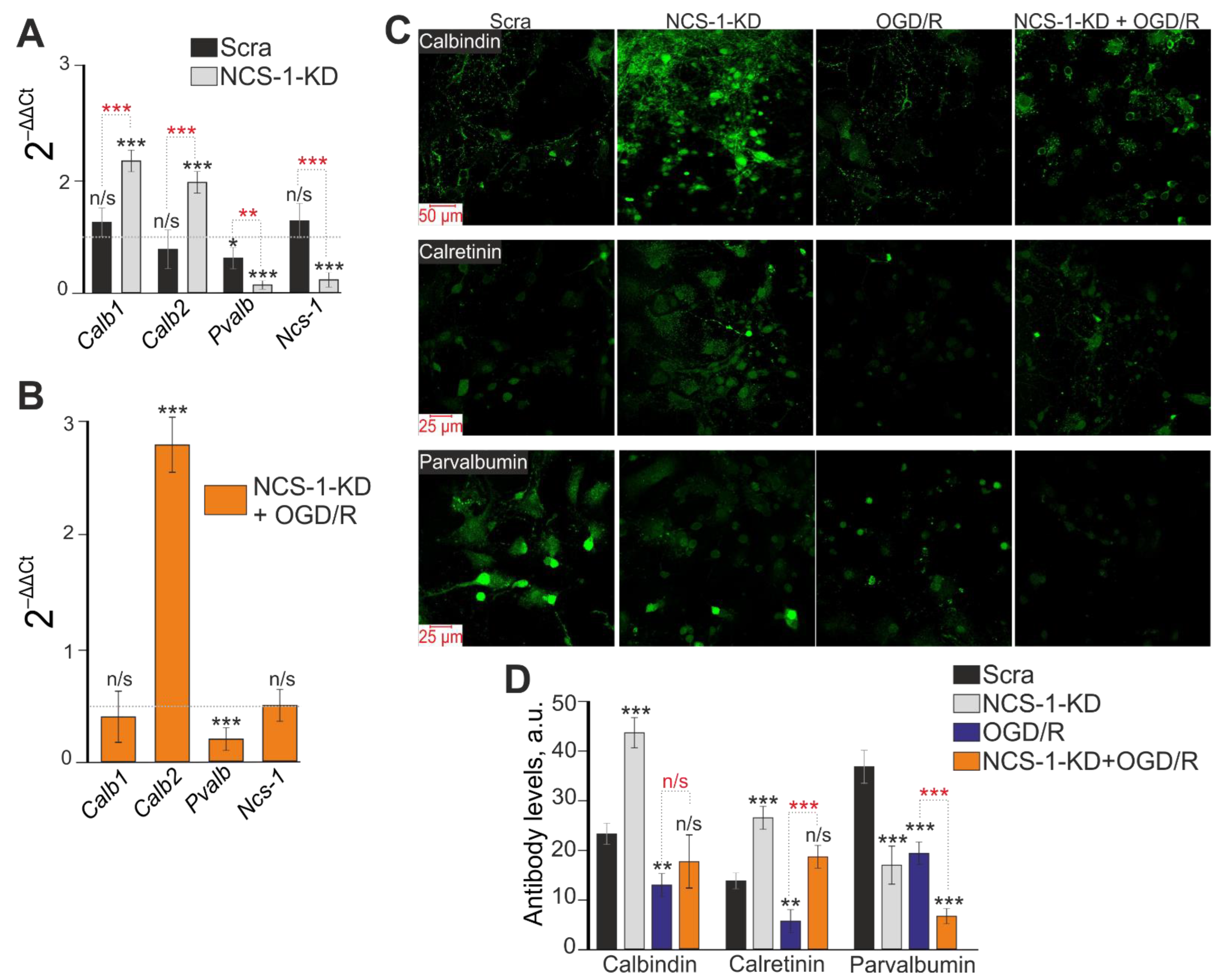
2.3. Expression of NCS-1 Protects Neurons from OGD/R-Induced Death through the Regulation of Calcium-Binding Protein Expression and Cytosolic Calcium Concentration
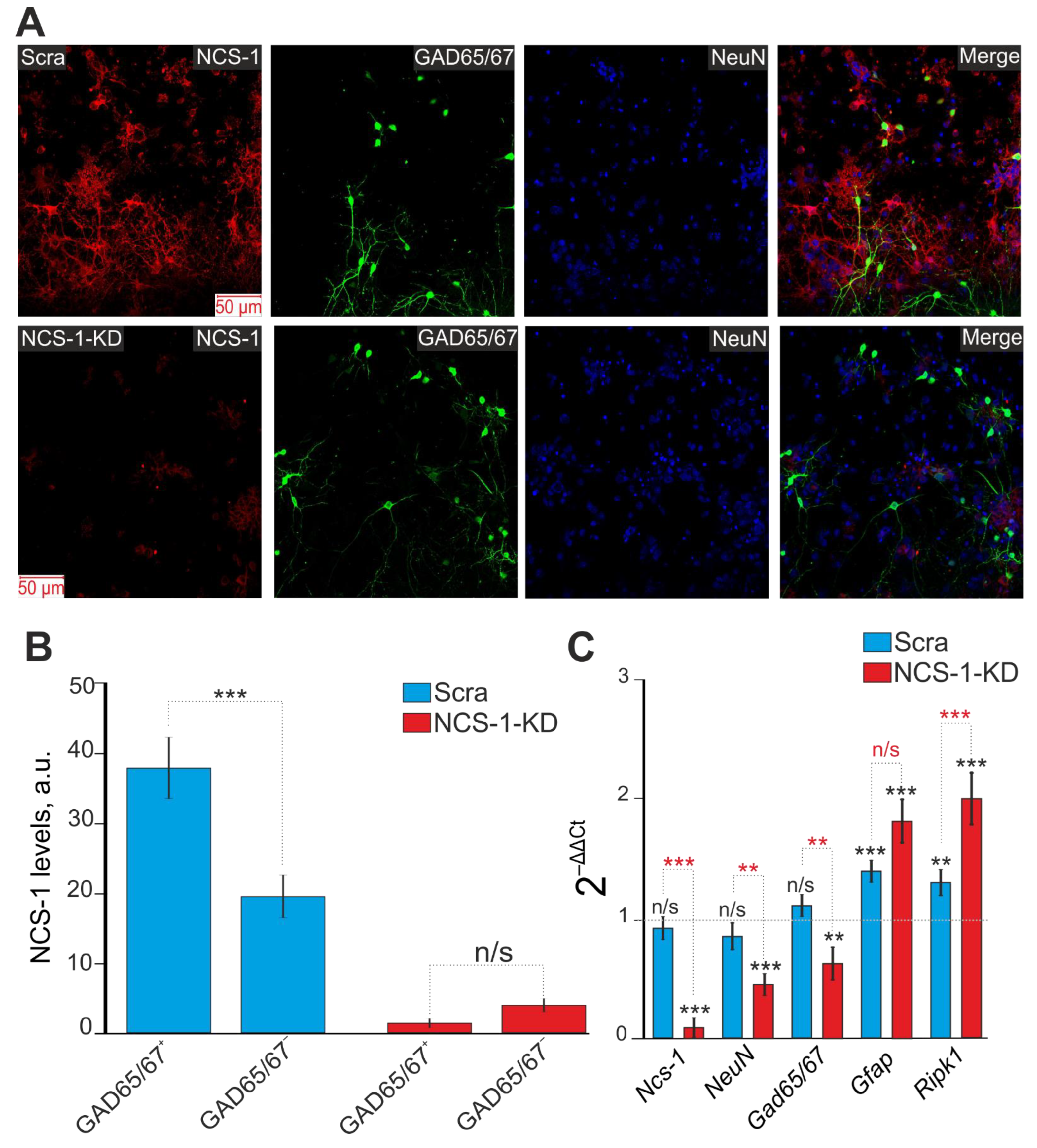
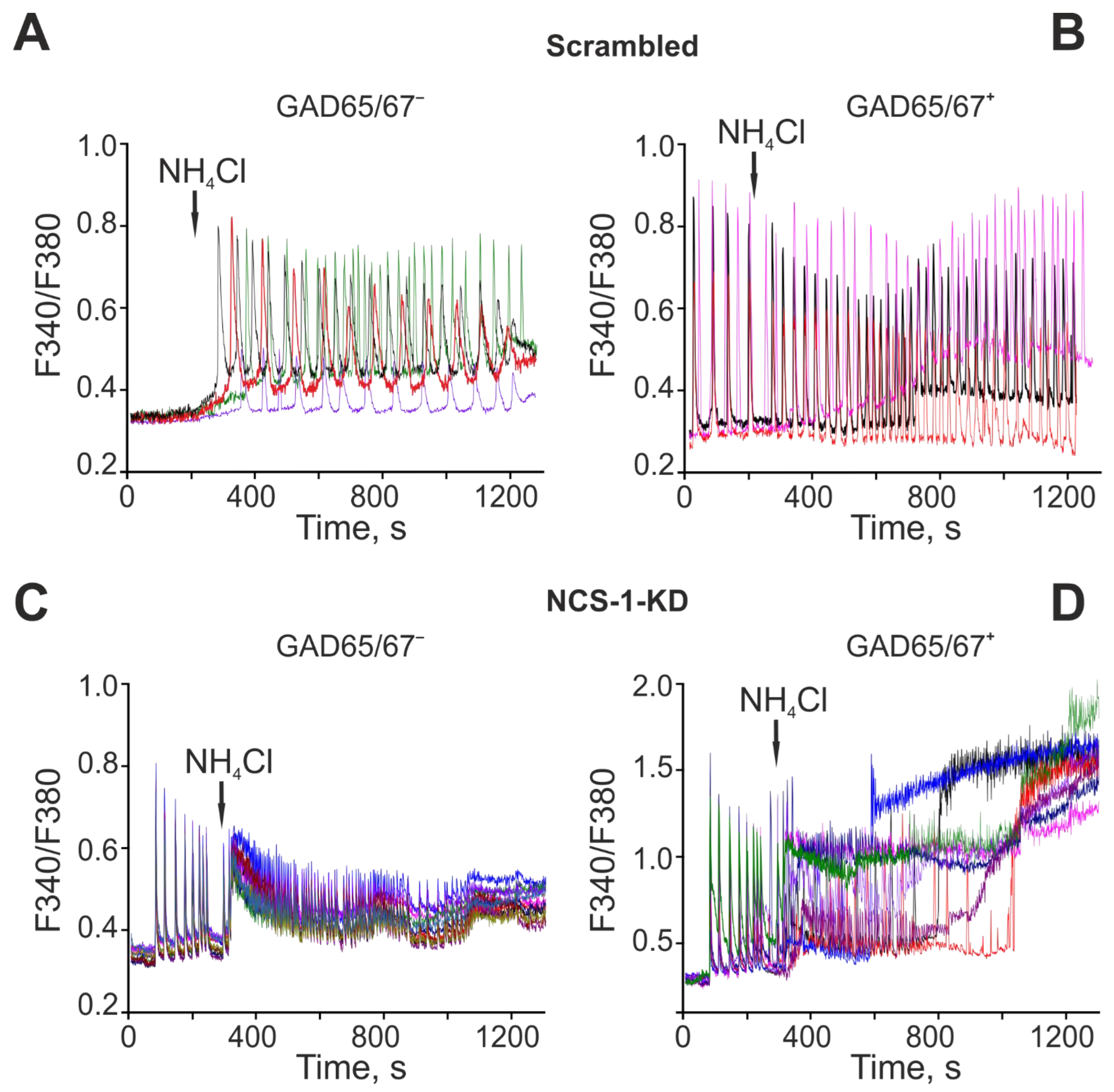
2.4. The Mechanism of the Neuroprotective Action of NCS-1 Occurs Due to the Suppression of Hyperexcitation of GABAergic Neurons and the Suppression of the Global [Ca2+]i Increase
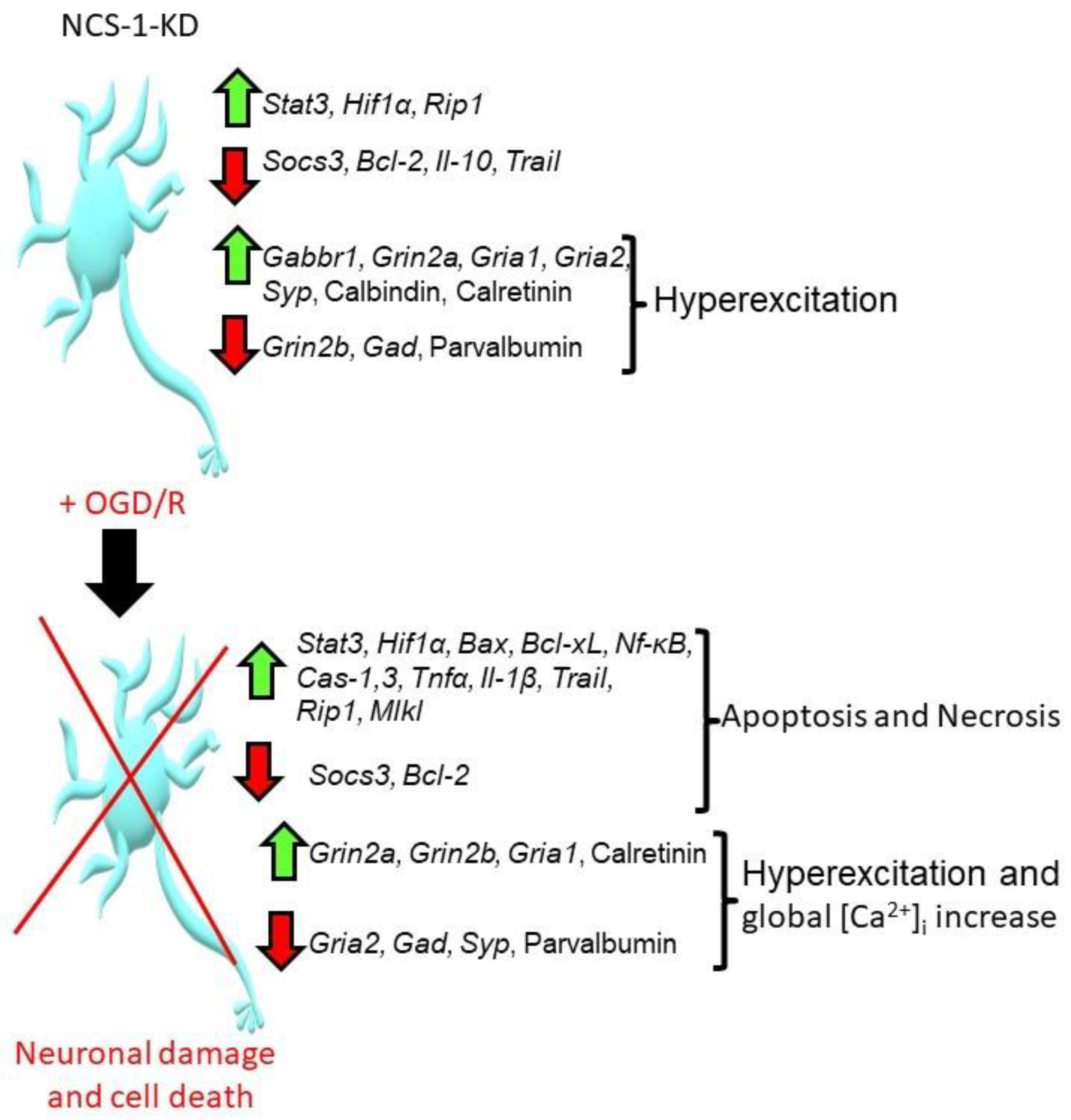
3. Discussion
4. Materials and Methods
4.1. Preparation of Mixed Neuroglial Cell Cultures
4.2. Immunocytochemistry
4.3. Transfection with Small Interfering RNA (siRNA)
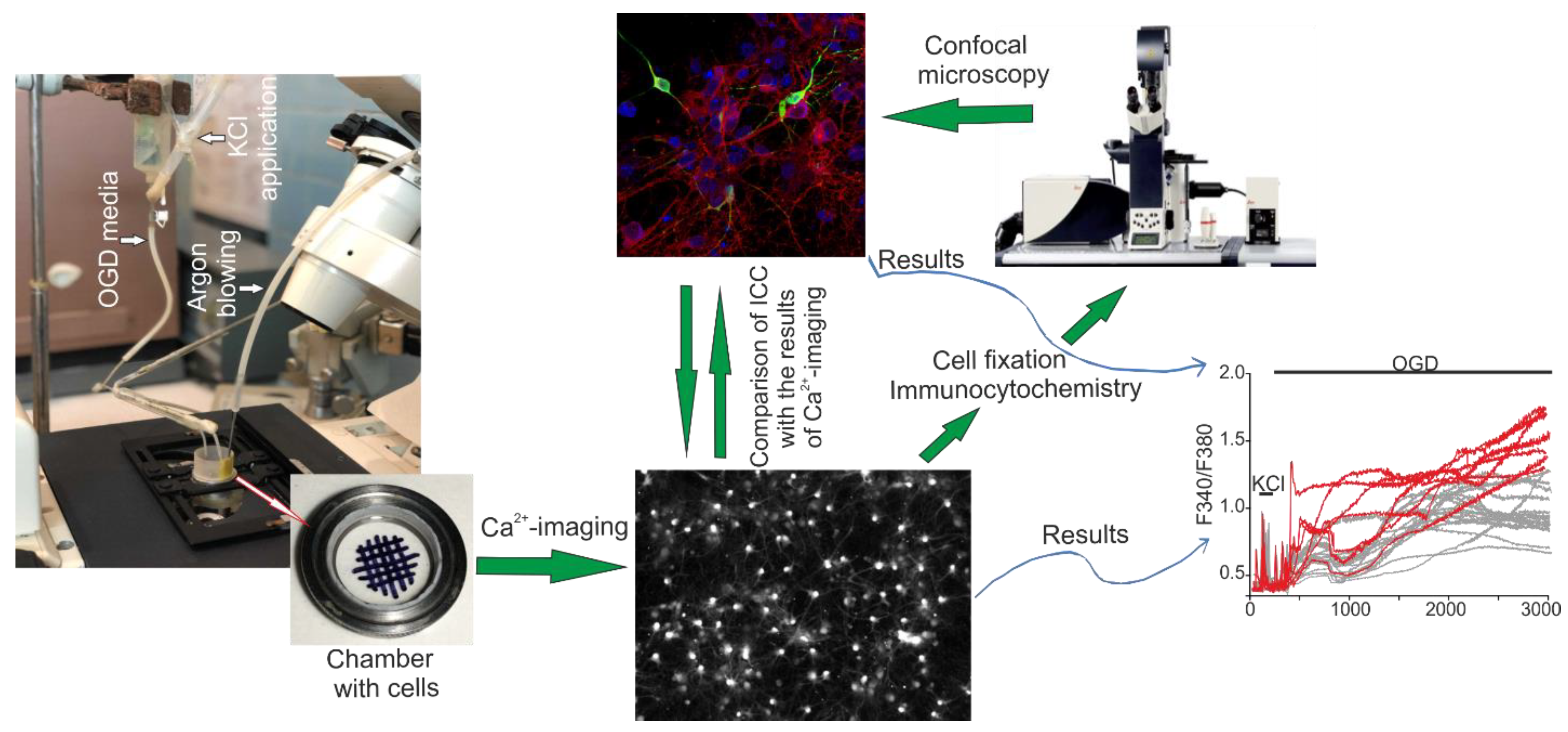
4.4. Fluorescent Ca2+ Measurements
4.5. The Technique for Simulation of Ischemia-like Conditions
4.6. Assessment of Cell Viability and Apoptosis
4.7. Extraction of RNA
4.8. Real-Time Polymerase Chain Reaction (RT-qPCR)
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Turovsky, E.A.; Turovskaya, M.V.; Kononov, A.V.; Zinchenko, V.P. Short-term episodes of hypoxia induce posthypoxic hyperexcitability and selective death of GABAergic hippocampal neurons. Exp. Neurol. 2013, 250, 1–7. [Google Scholar] [CrossRef]
- Turovskaya, M.V.; Gaidin, S.G.; Mal’tseva, V.N.; Zinchenko, V.P.; Turovsky, E.A. Taxifolin protects neurons against ischemic injury in vitro via the activation of antioxidant systems and signal transduction pathways of GABAergic neurons. Mol. Cell Neurosci. 2019, 96, 10–24. [Google Scholar] [CrossRef]
- Nakahara, I.; Kikuchi, H.; Taki, W.; Nishi, S.; Kito, M.; Yonekawa, Y.; Goto, Y.; Ogata, N. Changes in major phospholipids of mitochondria during postischemic reperfusion in rat brain. J. Neurosurg. 1992, 76, 244–250. [Google Scholar] [CrossRef]
- Orrenius, S.; McConkey, D.J.; Bellomo, G.; Nicotera, P. Role of Ca2+ in toxic cell killing. Trends Pharmacol. Sci. 1989, 10, 281–285. [Google Scholar] [CrossRef]
- Heizmann, C.W.; Braun, K. Changes in Ca2+-binding proteins in human neurodegenerative disorders. Trends Neurosci. 1992, 15, 259–264. [Google Scholar] [CrossRef]
- Burgoyne, R.D.; Haynes, L.P. Sense and specificity in neuronal calcium signaling. Biochim. Biophys. Acta. 2015, 1853, 1921–1932. [Google Scholar] [CrossRef]
- Pongs, O.; Lindemeier, J.; Zhu, X.R.; Theil, T.; Engelkamp, D.; Krah-Jentgens, I.; Lambrecht, H.G.; Koch, K.W.; Schwemer, J.; Rivosecchi, R.; et al. Frequenin--a novel calcium-binding protein that modulates synaptic efficacy in the Drosophila nervous system. Neuron 1993, 11, 15–28. [Google Scholar] [CrossRef]
- Chen, C.; Yu, L.; Zhang, P.; Jiang, J.; Zhang, Y.; Chen, X.; Wu, Q.; Wu, Q.; Zhao, S. Human neuronal calcium sensor-1 shows the highest expression level in cerebral cortex. Neurosci. Lett. 2002, 319, 67–70. [Google Scholar] [CrossRef]
- Saab, B.J.; Georgiou, J.; Nath, A.; Lee, F.J.; Wang, M.; Michalon, A.; Liu, F.; Mansuy, I.M.; Roder, J.C. NCS-1 in the dentate gyrus promotes exploration, synaptic plasticity, and rapid acquisition of spatial memory. Neuron 2009, 63, 643–656. [Google Scholar] [CrossRef]
- Nakamura, T.Y.; Nakao, S.; Nakajo, Y.; Takahashi, J.C.; Wakabayashi, S.; Yanamoto, H. Possible Signaling Pathways Mediating Neuronal Calcium Sensor-1-Dependent Spatial Learning and Memory in Mice. PLoS ONE 2017, 12, e0170829. [Google Scholar] [CrossRef]
- Burgoyne, R.D. The neuronal calcium-sensor proteins. Biochim. Biophys. Acta. 2004, 1742, 59–68. [Google Scholar] [CrossRef][Green Version]
- McFerran, B.W.; Graham, M.E.; Burgoyne, R.D. Neuronal Ca2+ sensor 1, the mammalian homologue of frequenin, is expressed in chromaffin and PC12 cells and regulates neurosecretion from dense-core granules. J. Biol. Chem. 1998, 273, 22768–22772. [Google Scholar] [CrossRef]
- Hui, K.; Fei, G.H.; Saab, B.J.; Su, J.; Roder, J.C.; Feng, Z.P. Neuronal calcium sensor-1 modulation of optimal calcium level for neurite outgrowth. Development 2007, 134, 4479–4489. [Google Scholar] [CrossRef]
- Nakamura, T.Y.; Jeromin, A.; Smith, G.; Kurushima, H.; Koga, H.; Nakabeppu, Y.; Wakabayashi, S.; Nabekura, J. Novel role of neuronal Ca2+ sensor-1 as a survival factor up-regulated in injured neurons. J. Cell Biol. 2006, 172, 1081–1091. [Google Scholar] [CrossRef]
- Yip, P.K.; Wong, L.F.; Sears, T.A.; Yáñez-Muñoz, R.J.; McMahon, S.B. Cortical overexpression of neuronal calcium sensor-1 induces functional plasticity in spinal cord following unilateral pyramidal tract injury in rat. PLoS Biol. 2010, 8, 1000399. [Google Scholar] [CrossRef]
- Sippy, T.; Cruz-Martin, A.; Jeromin, A.; Schweizer, F.E. Acute changes in short-term plasticity at synapses with elevated levels of neuronal calcium sensor-1. Nat. Neurosci. 2003, 6, 1031–1038. [Google Scholar] [CrossRef]
- Génin, A.; Davis, S.; Meziane, H.; Doyère, V.; Jeromin, A.; Roder, J.; Mallet, J.; Laroche, S. Regulated expression of the neuronal calcium sensor-1 gene during long-term potentiation in the dentate gyrus in vivo. Neuroscience 2001, 106, 571–577. [Google Scholar] [CrossRef]
- Jo, J.; Heon, S.; Kim, M.J.; Son, G.H.; Park, Y.; Henley, J.M.; Weiss, J.L.; Sheng, M.; Collingridge, G.L.; Cho, K. Metabotropic glutamate receptor-mediated LTD involves two interacting Ca2+ sensors, NCS-1 and PICK1. Neuron 2008, 60, 1095–1111. [Google Scholar] [CrossRef]
- D’Onofrio, S.; Mahaffey, S.; Garcia-Rill, E. Role of calcium channels in bipolar disorder. Curr. Psychopharmacol. 2017, 6, 122–135. [Google Scholar] [CrossRef][Green Version]
- Piton, A.; Michaud, J.L.; Peng, H.; Aradhya, S.; Gauthier, J.; Mottron, L.; Champagne, N.; Lafrenière, R.G.; Hamdan, F.F.; S2D team; et al. Mutations in the calcium-related gene IL1RAPL1 are associated with autism. Hum. Mol. Genet. 2008, 17, 3965–3974. [Google Scholar] [CrossRef]
- Multani, P.K.; Clarke, T.K.; Narasimhan, S.; Ambrose-Lanci, L.; Kampman, K.M.; Pettinati, H.M.; Oslin, D.W.; O’Brien, C.P.; Berrettini, W.H.; Lohoff, F.W. Neuronal calcium sensor-1 and cocaine addiction: A genetic association study in African-Americans and European Americans. Neurosci. Lett. 2012, 531, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Dragicevic, E.; Poetschke, C.; Duda, J.; Schlaudraff, F.; Lammel, S.; Schiemann, J.; Fauler, M.; Hetzel, A.; Watanabe, M.; Lujan, R.; et al. Cav1.3 channels control D2-autoreceptor responses via NCS-1 in substantia nigra dopamine neurons. Brain 2014, 137, 2287–2302. [Google Scholar] [CrossRef] [PubMed]
- Turovsky, E.A.; Turovskaya, M.V.; Gaidin, S.G.; Zinchenko, V.P. Cytokine IL-10, activators of PI3-kinase, agonists of α-2 adrenoreceptor and antioxidants prevent ischemia-induced cell death in rat hippocampal cultures. Arch. Biochem. Biophys. 2017, 615, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Turovsky, E.A.; Zinchenko, V.P.; Gaidin, S.G.; Turovskaya, M.V. Calcium-Binding Proteins Protect GABAergic Neurons of the Hippocampus from Hypoxia and Ischemia in vitro. Biochem. (Mosc.) Suppl. Ser. A Membr. Cell Biol. 2018, 12, 74–84. [Google Scholar] [CrossRef]
- De Rezende, V.B.; Rosa, D.V.; Comim, C.M.; Magno, L.A.; Rodrigues, A.L.; Vidigal, P.; Jeromin, A.; Quevedo, J.; Romano-Silva, M.A. NCS-1 deficiency causes anxiety and depressive-like behavior with impaired non-aversive memory in mice. Physiol. Behav. 2014, 130, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.Y.; Jeromin, A.; Mikoshiba, K.; Wakabayashi, S. Neuronal calcium sensor-1 promotes immature heart function and hypertrophy by enhancing Ca2+ signals. Circ. Res. 2011, 109, 512–523. [Google Scholar] [CrossRef]
- Jinno, S.; Jeromin, A.; Kosaka, T. Expression and possible role of neuronal calcium sensor-1 in the cerebellum. Cerebellum 2004, 3, 83–88. [Google Scholar] [CrossRef]
- Burgoyne, R.D.; Helassa, N.; McCue, H.V.; Haynes, L.P. Calcium Sensors in Neuronal Function and Dysfunction. Cold Spring Harb. Perspect. Biol. 2019, 11, 035154. [Google Scholar] [CrossRef]
- Gaidin, S.G.; Turovskaya, M.V.; Gavrish, M.S.; Babaev, A.A.; Mal’tseva, V.N.; Blinova, E.V.; Turovsky, E.A. The selective BDNF overexpression in neurons protects neuroglial networks against OGD and glutamate-induced excitotoxicity. Int. J. Neurosci. 2020, 130, 363–383. [Google Scholar] [CrossRef]
- Schwaller, B.; Durussel, I.; Jermann, D.; Herrmann, B.; Cox, J.A. Comparison of the Ca2+-binding properties of human recombinant calretinin-22k and calretinin. J. Biol. Chem. 1997, 272, 29663–29671. [Google Scholar] [CrossRef]
- Choi, W.-S.; Lee, E.; Lim, J.; Oh, Y.J. Calbindin-D28K prevents drug-induced dopaminergic neuronal death by inhibiting caspase and calpain activity. Biochem. Biophys. Res. Commun. 2008, 371, 127–131. [Google Scholar] [CrossRef]
- O’Callaghan, D.W.; Ivings, L.; Weiss, J.L.; Ashby, M.C.; Tepikin, A.V.; Burgoyne, R.D. Differential use of myristoyl groups on neuronal calcium sensor proteins as a determinant of spatio-temporal aspects of Ca2+-signal transduction. J. Biol. Chem. 2002, 277, 14227–14237. [Google Scholar] [CrossRef]
- D’Onofrio, S.; Kezunovic, N.; Hyde, J.R.; Luster, B.; Messias, E.; Urbano, F.J.; Garcia-Rill, E. Modulation of gamma oscillations in the pedunculopontine nucleus by neuronal calcium sensor protein-1: Relevance to schizophrenia and bipolar disorder. J. Neurophysiol. 2015, 113, 709–719. [Google Scholar] [CrossRef]
- Iketani, M.; Imaizumi, C.; Nakamura, F.; Jeromin, A.; Mikoshiba, K.; Goshima, Y.; Takei, K. Regulation of neurite outgrowth mediated by neuronal calcium sensor-1 and inositol 1,4,5-trisphosphate receptor in nerve growth cones. Neuroscience 2009, 161, 743–752. [Google Scholar] [CrossRef]
- Turovsky, E.A.; Mal’tseva, V.N.; Sarimov, R.M.; Simakin, A.V.; Gudkov, S.V.; Plotnikov, E.Y. Features of the cytoprotective effect of selenium nanoparticles on primary cortical neurons and astrocytes during oxygen-glucose deprivation and reoxygenation. Sci. Rep. 2022, 12, 1710. [Google Scholar] [CrossRef]
- Varlamova, E.G.; Turovsky, E.A.; Babenko, V.A.; Plotnikov, E.Y. The Mechanisms Underlying the Protective Action of Selenium Nanoparticles against Ischemia/Reoxygenation Are Mediated by the Activation of the Ca2+ Signaling System of Astrocytes and Reactive Astrogliosis. Int. J. Mol. Sci. 2021, 22, 12825. [Google Scholar] [CrossRef]
- Nakamura, T.Y.; Nakao, S.; Wakabayashi, S. Neuronal Ca2+ sensor-1 contributes to stress tolerance in cardiomyocytes via activation of mitochondrial detoxification pathways. J. Mol. Cell. Cardiol. 2016, 99, 23–34. [Google Scholar] [CrossRef]
- Nakamura, T.Y.; Nakao, S.; Wakabayashi, S. Emerging Roles of Neuronal Ca 2+ Sensor-1 in Cardiac and Neuronal Tissues: A Mini Review. Front. Mol. Neurosci. 2019, 12, 56. [Google Scholar] [CrossRef]
- Navarro, G.; Hradsky, J.; Lluís, C.; Casadó, V.; McCormick, P.J.; Kreutz, M.R.; Mikhaylova, M. NCS-1 associates with adenosine A(2A) receptors and modulates receptor function. Front. Mol. Neurosci. 2012, 5, 53. [Google Scholar] [CrossRef]
- Cohen, M.V.; Downey, J.M. Adenosine: Trigger and mediator of cardioprotection. Basic Res. Cardiol. 2008, 103, 203–215. [Google Scholar] [CrossRef]
- Fei, G.H.; Feng, Z.P. Chronic hypoxia-induced alteration of presynaptic protein profiles and neurobehavioral dysfunction are averted by supplemental oxygen in Lymnaea stagnalis. Neuroscience 2008, 153, 318–328. [Google Scholar] [CrossRef] [PubMed]
- Hendricks, K.B.; Wang, B.Q.; Schnieders, E.A.; Thorner, J. Yeast homologue of neuronal frequenin is a regulator of phosphatidylinositol-4-OH kinase. Nat. Cell Biol. 1999, 1, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Varnai, P.; Tuymetova, G.; Balla, A.; Toth, Z.E.; Oker-Blom, C.; Roder, J.; Jeromin, A.; Balla, T. Interaction of neuronal calcium sensor-1 (NCS-1) with phosphatidylinositol 4-kinase beta stimulates lipid kinase activity and affects membrane trafficking in COS-7 cells. J. Biol. Chem. 2001, 276, 40183–40189. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.Y.; Jeromin, A.; Lundstrom, K.; Yoo, S.H.; Roder, J.; Fox, A.P. Alterations in exocytosis induced by neuronal Ca2+ sensor-1 in bovine chromaffin cells. J. Neurosci. 2002, 22, 2427–2433. [Google Scholar] [CrossRef] [PubMed]
- Taverna, E.; Francolini, M.; Jeromin, A.; Hilfiker, S.; Roder, J.; Rosa, P. Neuronal calcium sensor 1 and phosphatidylinositol 4-OH kinase beta interact in neuronal cells and are translocated to membranes during nucleotide-evoked exocytosis. J. Cell Sci. 2002, 115, 3909–3922. [Google Scholar] [CrossRef] [PubMed]
- Tsujimoto, T.; Jeromin, A.; Saitoh, N.; Roder, J.C.; Takahashi, T. Neuronal calcium sensor 1 and activity-dependent facilitation of P/Q-type calcium currents at presynaptic nerve terminals. Science 2002, 295, 2276–2279. [Google Scholar] [CrossRef] [PubMed]
- Burgoyne, R.D. Neuronal calcium sensor proteins: Generating diversity in neuronal Ca2+ signalling. Nat. Rev. Neurosci. 2007, 8, 182–193. [Google Scholar] [CrossRef]
- Turovsky, E.A.; Varlamova, E.G.; Gudkov, S.V.; Plotnikov, E.Y. The Protective Mechanism of Deuterated Linoleic Acid Involves the Activation of the Ca2+ Signaling System of Astrocytes in Ischemia In Vitro. Int. J. Mol. Sci. 2021, 22, 13216. [Google Scholar] [CrossRef]
- Zinchenko, V.P.; Turovskaya, M.V.; Teplov, I.Y.; Berezhnov, A.V.; Turovsky, E.A. The role of parvalbumin-containing interneurons in the regulation of spontaneous synchronous activity of brain neurons in culture. Biophysics 2016, 61, 85–93. [Google Scholar] [CrossRef]
- Turovsky, E.A.; Varlamova, E.G. Mechanism of Ca2+-Dependent Pro-Apoptotic Action of Selenium Nanoparticles, Mediated by Activation of Cx43 Hemichannels. Biology 2021, 10, 743. [Google Scholar] [CrossRef]
- Turovsky, E.A.; Golovicheva, V.V.; Varlamova, E.G.; Danilina, T.I.; Goryunov, K.V.; Shevtsova, Y.A.; Pevzner, I.B.; Zorova, L.D.; Babenko, V.A.; Evtushenko, E.A.; et al. Mesenchymal stromal cell-derived extracellular vesicles afford neuroprotection by modulating PI3K/AKT pathway and calcium oscillations. Int. J. Biol. Sci. 2022, 18, 5345–5368. [Google Scholar] [CrossRef]
- Schmid, I.; Uittenbogaart, C.; Jamieson, B.D. Live-cell assay for detection of apoptosis by dual-laser flow cytometry using Hoechst 33342 and 7-amino-actino-mycin D. Nat Protoc. 2007, 2, 187–190. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varlamova, E.G.; Plotnikov, E.Y.; Turovsky, E.A. Neuronal Calcium Sensor-1 Protects Cortical Neurons from Hyperexcitation and Ca2+ Overload during Ischemia by Protecting the Population of GABAergic Neurons. Int. J. Mol. Sci. 2022, 23, 15675. https://doi.org/10.3390/ijms232415675
Varlamova EG, Plotnikov EY, Turovsky EA. Neuronal Calcium Sensor-1 Protects Cortical Neurons from Hyperexcitation and Ca2+ Overload during Ischemia by Protecting the Population of GABAergic Neurons. International Journal of Molecular Sciences. 2022; 23(24):15675. https://doi.org/10.3390/ijms232415675
Chicago/Turabian StyleVarlamova, Elena G., Egor Y. Plotnikov, and Egor A. Turovsky. 2022. "Neuronal Calcium Sensor-1 Protects Cortical Neurons from Hyperexcitation and Ca2+ Overload during Ischemia by Protecting the Population of GABAergic Neurons" International Journal of Molecular Sciences 23, no. 24: 15675. https://doi.org/10.3390/ijms232415675
APA StyleVarlamova, E. G., Plotnikov, E. Y., & Turovsky, E. A. (2022). Neuronal Calcium Sensor-1 Protects Cortical Neurons from Hyperexcitation and Ca2+ Overload during Ischemia by Protecting the Population of GABAergic Neurons. International Journal of Molecular Sciences, 23(24), 15675. https://doi.org/10.3390/ijms232415675






