The Role of Molecular and Hormonal Factors in Obesity and the Effects of Physical Activity in Children
Abstract
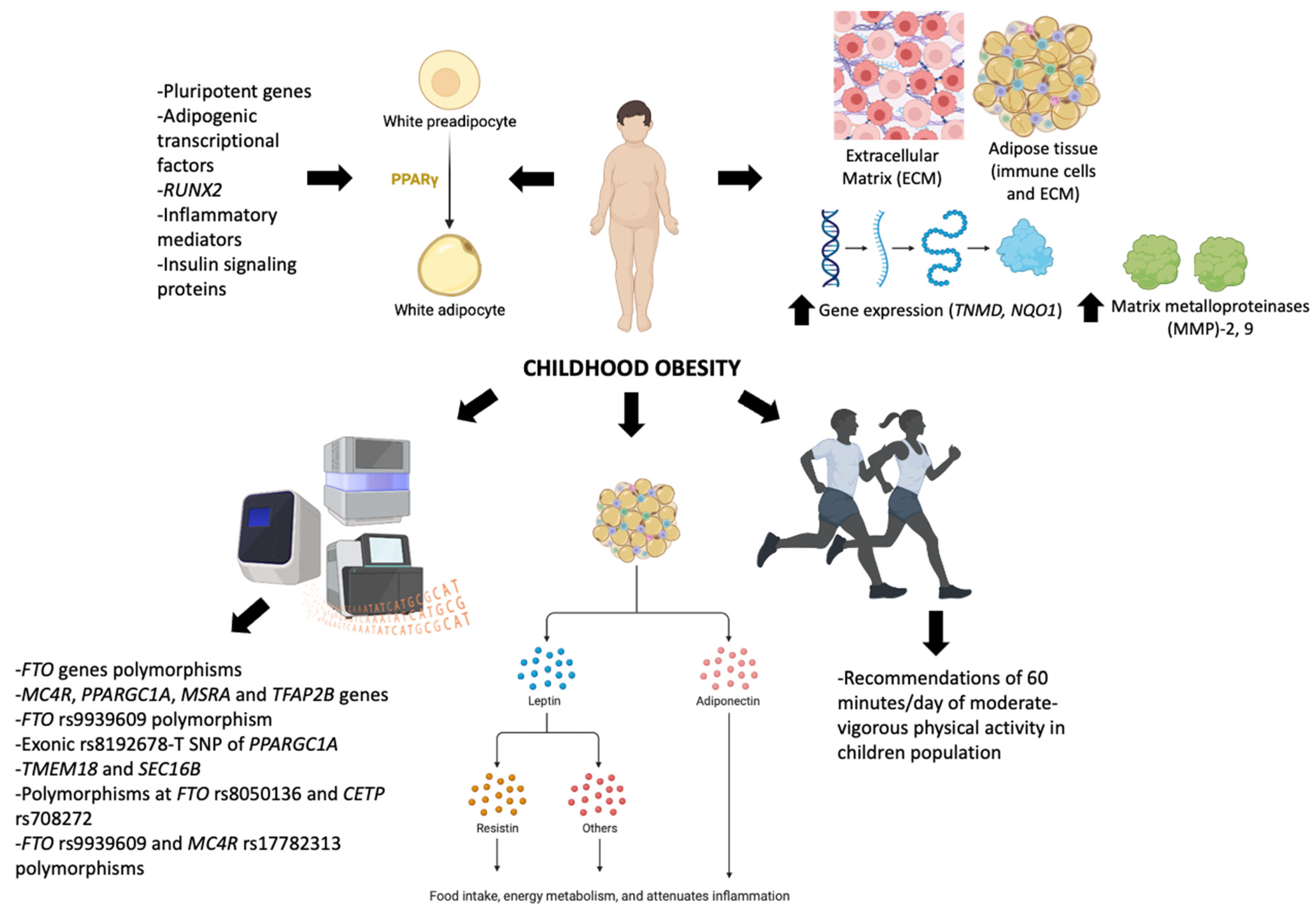
1. Introduction
2. Molecular Control in Obesity
2.1. Structure and Components of ECM Related to Obesity
2.2. Control of Differentiation in WAT
2.3. Genetics Underlying Childhood Obesity
| Gen or Polymorphism | Type of Polymorphism | Type of Study | Age | Reference |
|---|---|---|---|---|
| In a UK twin sample, the FTO gene was found to be associated with BMI beginning at age seven, and a similar finding was also noted in Danish children. In the UK study, the FTO gene explained only a small portion of the genetic variation in BMI. | - | Systematic review, nine twin and five adoption studies | up to 18 years old | [116] |
| ANRIL methylation (encoded by CDKN2A) has been associated with adiposity in the birth tissue of ethnically diverse neonates, peripheral blood of adolescents, and adipose tissue of adults. | - | Prospective study, randomized control trial | From birth to 6 years old | [118] |
| It may be assumed that MC4R, PPARGC1A, MSRA, and TFAP2B genes contribute to obesity risk in this sample of Portuguese children. | - | Prospective study with 730 children | 2 to 6 years old | [119] |
| FTO rs9939609 polymorphism was significantly associated with an increased risk of obesity. | SNP | Meta-analysis with 12 eligible studies with 5000 cases and 9853 controls | up to 18 years old | [120] |
| FAM120AOS gene was triggered by differences in BMI at 8 years (T allele of rs944990 increased BMI, with a modest association with change in BMI over time). A locus associated with childhood obesity (OLFM4) has reached genome-wide significance in relation to BMI at age 8 and/or changes over time. | SNP | GWAS meta-analysis of BMI trajectories from 1 to 17 years of age in 9377 children | 1 to 17 years of age | [121] |
| A strong association between the exonic rs8192678-T SNP of PPARGC1A and a reduction in BMI z-score. | SNP | Randomized, prospective, double-blind, placebo-controlled, multicenter trial, 134 children | aged 7 to 14 | [122] |
| TMEM18 and SEC16B were associated with an increased risk of obesity of 27% and 40%, respectively, in Hispanic/Latino children (22–88% frequency). | SNP | Genome-wide association study of childhood obesity in 1612 Hispanic/Latino children and adolescents | 2 to 18 years of age | [123] |
| Polymorphisms at FTO rs8050136 and CETP rs708272 have been identified as significant with childhood metabolic syndrome. | SNP | Prospective cohort study with 3067 children | aged 2 to 10 years | [124] |
| FTO rs9930506 and MC4R rs17782313 polymorphisms and obesity in children. | SNP | Meta-analysis with 13 studies on MC4R rs17782313 and 18 studies on FTO rs9939609 | up to 18 years old | [125] |
| FTO rs9939609 and MC4R rs17782313 polymorphisms have been associated with overweight and obesity in children. | SNP | Meta-analysis with 12 studies on MC4R rs17782313 and FTO rs9939609 | up to 18 years old | [126] |
3. Hormonal Control in Obesity
3.1. Leptin
3.2. Adiponectin
3.3. Resistin
4. Exercise and Leptin Control in Children with Obesity
5. Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fruh, S.M. Obesity: Risk factors, complications, and strategies for sustainable long-term weight management. J. Am. Assoc. Nurse Pract. 2017, 29, S3–S14. [Google Scholar] [CrossRef] [PubMed]
- Javed, A.; Jumean, M.; Murad, M.H.; Okorodudu, D.; Kumar, S.; Somers, V.K.; Sochor, O.; Lopez-Jimenez, F. Diagnostic performance of body mass index to identify obesity as defined by body adiposity in children and adolescents: A systematic review and meta-analysis. Pediatr. Obes. 2015, 10, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Hruby, A.; Hu, F.B. The Epidemiology of Obesity: A Big Picture. Pharmacoeconomics 2015, 33, 673–689. [Google Scholar] [CrossRef] [PubMed]
- Di Cesare, M.; Soric, M.; Bovet, P.; Miranda, J.J.; Bhutta, Z.; Stevens, G.A.; Laxmaiah, A.; Kengne, A.P.; Bentham, J. The epidemiological burden of obesity in childhood: A worldwide epidemic requiring urgent action. BMC Med. 2019, 17, 212. [Google Scholar] [CrossRef] [PubMed]
- Llorca-Colomer, F.; Murillo-Llorente, M.T.; Legidos-Garcia, M.E.; Palau-Ferre, A.; Perez-Bermejo, M. Differences in Classification Standards For the Prevalence of Overweight and Obesity in Children. A Systematic Review and Meta-Analysis. Clin. Epidemiol. 2022, 14, 1031–1052. [Google Scholar] [CrossRef]
- de Onis, M.; Garza, C.; Onyango, A.W.; Rolland-Cachera, M.F.; le Comité de nutrition de la Société française depédiatrie. WHO growth standards for infants and young children. Arch. Pediatr. 2009, 16, 47–53. [Google Scholar] [CrossRef]
- Ogden, C.L.; Kuczmarski, R.J.; Flegal, K.M.; Mei, Z.; Guo, S.; Wei, R.; Grummer-Strawn, L.M.; Curtin, L.R.; Roche, A.F.; Johnson, C.L. Centers for Disease Control and Prevention 2000 growth charts for the United States: Improvements to the 1977 National Center for Health Statistics version. Pediatrics 2002, 109, 45–60. [Google Scholar] [CrossRef]
- Cole, T.J.; Lobstein, T. Extended international (IOTF) body mass index cut-offs for thinness, overweight and obesity. Pediatr. Obes. 2012, 7, 284–294. [Google Scholar] [CrossRef]
- Lopez-Sanchez, G.F.; Sgroi, M.; D’Ottavio, S.; Diaz-Suarez, A.; Gonzalez-Villora, S.; Veronese, N.; Smith, L. Body Composition in Children and Adolescents Residing in Southern Europe: Prevalence of Overweight and Obesity According to Different International References. Front. Physiol. 2019, 10, 130. [Google Scholar] [CrossRef]
- Vanderwall, C.; Randall Clark, R.; Eickhoff, J.; Carrel, A.L. BMI is a poor predictor of adiposity in young overweight and obese children. BMC Pediatr. 2017, 17, 135. [Google Scholar] [CrossRef]
- Wall, C.R.; Hill, R.J.; Lovell, A.L.; Matsuyama, M.; Milne, T.; Grant, C.C.; Jiang, Y.; Chen, R.X.; Wouldes, T.A.; Davies, P.S. A multicenter, double-blind, randomized, placebo-controlled trial to evaluate the effect of consuming Growing Up Milk “Lite” on body composition in children aged 12–23 mo. Am. J. Clin. Nutr. 2019, 109, 576–585. [Google Scholar] [CrossRef]
- Wood, K.; Mantzioris, E.; Lingwood, B.; Couper, J.; Makrides, M.; Gibson, R.; Muhlhausler, B. The effect of maternal DHA supplementation on body fat mass in children at 7 years: Follow-up of the DOMInO randomized controlled trial. Prostaglandins Leukot. Essent. Fat. Acids 2018, 139, 49–54. [Google Scholar] [CrossRef]
- Jiwani, S.S.; Gatica-Dominguez, G.; Crochemore-Silva, I.; Maiga, A.; Walton, S.; Hazel, E.; Baille, B.; Bose, S.; Bosu, W.K.; Busia, K.; et al. Trends and inequalities in the nutritional status of adolescent girls and adult women in sub-Saharan Africa since 2000: A cross-sectional series study. BMJ Glob. Health 2020, 5, e002948. [Google Scholar] [CrossRef]
- Bhurosy, T.; Jeewon, R. Overweight and obesity epidemic in developing countries: A problem with diet, physical activity, or socioeconomic status? Sci. World J. 2014, 2014, 964236. [Google Scholar] [CrossRef]
- Lin, X.; Li, H. Obesity: Epidemiology, Pathophysiology, and Therapeutics. Front. Endocrinol. 2021, 12, 706978. [Google Scholar] [CrossRef]
- Camacho, S.; Ruppel, A. Is the calorie concept a real solution to the obesity epidemic? Glob. Health Action 2017, 10, 1289650. [Google Scholar] [CrossRef]
- Huang, N.C.; Kung, S.F.; Hu, S.C. The Relationship between Urbanization, the Built Environment, and Physical Activity among Older Adults in Taiwan. Int. J. Environ. Res. Public Health 2018, 15, 836. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 27 September 2022).
- Yan, J.; Liu, L.; Zhu, Y.; Huang, G.; Wang, P.P. The association between breastfeeding and childhood obesity: A meta-analysis. BMC Public Health 2014, 14, 1267. [Google Scholar] [CrossRef]
- Ruiz-Ojeda, F.J.; Mendez-Gutierrez, A.; Aguilera, C.M.; Plaza-Diaz, J. Extracellular Matrix Remodeling of Adipose Tissue in Obesity and Metabolic Diseases. Int. J. Mol. Sci. 2019, 20, 4888. [Google Scholar] [CrossRef]
- Anguita-Ruiz, A.; Bustos-Aibar, M.; Plaza-Diaz, J.; Mendez-Gutierrez, A.; Alcala-Fdez, J.; Aguilera, C.M.; Ruiz-Ojeda, F.J. Omics Approaches in Adipose Tissue and Skeletal Muscle Addressing the Role of Extracellular Matrix in Obesity and Metabolic Dysfunction. Int. J. Mol. Sci. 2021, 22, 2756. [Google Scholar] [CrossRef]
- Chen, H.J.; Yan, X.Y.; Sun, A.; Zhang, L.; Zhang, J.; Yan, Y.E. Adipose extracellular matrix deposition is an indicator of obesity and metabolic disorders. J. Nutr. Biochem. 2022, 111, 109159. [Google Scholar] [CrossRef] [PubMed]
- Ghaben, A.L.; Scherer, P.E. Adipogenesis and metabolic health. Nat. Rev. Mol. Cell Biol. 2019, 20, 242–258. [Google Scholar] [PubMed]
- Ruiz-Ojeda, F.J.; Wang, J.; Backer, T.; Krueger, M.; Zamani, S.; Rosowski, S.; Gruber, T.; Onogi, Y.; Feuchtinger, A.; Schulz, T.J.; et al. Active integrins regulate white adipose tissue insulin sensitivity and brown fat thermogenesis. Mol. Metab. 2021, 45, 101147. [Google Scholar] [CrossRef] [PubMed]
- Palmer, B.F.; Clegg, D.J. The sexual dimorphism of obesity. Mol. Cell Endocrinol. 2015, 402, 113–119. [Google Scholar] [CrossRef]
- Zamanian-Azodi, M.; Vafaee, R.; Azodi, T.; Omidi, R.; Gilanchi, S.; Azizi-Jalilian, F.; Khodarahmi, R. Molecular approaches in obesity studies. Gastroenterol. Hepatol. Bed Bench 2013, 6, S23–S31. [Google Scholar]
- Obradovic, M.; Sudar-Milovanovic, E.; Soskic, S.; Essack, M.; Arya, S.; Stewart, A.J.; Gojobori, T.; Isenovic, E.R. Leptin and Obesity: Role and Clinical Implication. Front. Endocrinol. 2021, 12, 585887. [Google Scholar] [CrossRef]
- Paquot, N.; De Flines, J.; Rorive, M. Obesity: A model of complex interactions between genetics and environment. Revue Médicale Liège 2012, 67, 332–336. [Google Scholar]
- Niemiro, G.M.; Rewane, A.; Algotar, A.M. Exercise and Fitness Effect On Obesity. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Bateman, L.A.; Slentz, C.A.; Willis, L.H.; Shields, A.T.; Piner, L.W.; Bales, C.W.; Houmard, J.A.; Kraus, W.E. Comparison of aerobic versus resistance exercise training effects on metabolic syndrome (from the Studies of a Targeted Risk Reduction Intervention Through Defined Exercise—STRRIDE-AT/RT). Am. J. Cardiol. 2011, 108, 838–844. [Google Scholar] [CrossRef]
- Radak, Z.; Zhao, Z.; Koltai, E.; Ohno, H.; Atalay, M. Oxygen consumption and usage during physical exercise: The balance between oxidative stress and ROS-dependent adaptive signaling. Antioxid Redox Signal. 2013, 18, 1208–1246. [Google Scholar] [CrossRef]
- Umpierre, D.; Ribeiro, P.A.; Kramer, C.K.; Leitao, C.B.; Zucatti, A.T.; Azevedo, M.J.; Gross, J.L.; Ribeiro, J.P.; Schaan, B.D. Physical activity advice only or structured exercise training and association with HbA1c levels in type 2 diabetes: A systematic review and meta-analysis. JAMA 2011, 305, 1790–1799. [Google Scholar] [CrossRef]
- Nambi, G.; Alghadier, M.; Elnegamy, T.E.; Basuodan, R.M.; Alwhaibi, R.M.; Vellaiyan, A.; Nwihadh, N.A.; Aldhafian, O.R.; Verma, A.; Pakkir Mohamed, S.H.; et al. Clinical (BMI and MRI) and Biochemical (Adiponectin, Leptin, TNF-alpha, and IL-6) Effects of High-Intensity Aerobic Training with High-Protein Diet in Children with Obesity Following COVID-19 Infection. Int. J. Environ. Res. Public Health 2022, 19, 7194. [Google Scholar] [CrossRef]
- Aragon-Vela, J.; Delgado-Floody, P.; Guzman-Guzman, I.P.; Salas-Sanchez, J.; Martinez-Redondo, M.; Lucena Zurita, M.; Herrador Sanchez, J.; Cardona Linares, A.J.; Consuegra Gonzalez, P.J.; Santos, E.C.M.A.; et al. Effect of COVID-19 confinement on physical activity patterns in relation to sociodemographic parameters in Spanish population. J. Sports Med. Phys. Fit. 2022, 62, 830–837. [Google Scholar] [CrossRef]
- Zachurzok, A.; Wojcik, M.; Gawlik, A.; Starzyk, J.B.; Mazur, A. An Attempt to Assess the Impact of Pandemic Restrictions on the Lifestyle, Diet, and Body Mass Index of Children with Endocrine Diseases-Preliminary Results. Nutrients 2021, 14, 156. [Google Scholar] [CrossRef]
- He, Y.; Luo, B.; Zhao, L.; Liao, S. Influences of the COVID-19 Pandemic on Obesity and Weight-Related Behaviors among Chinese Children: A Multi-Center Longitudinal Study. Nutrients 2022, 14, 3744. [Google Scholar] [CrossRef]
- Garcia-Hermoso, A.; Ramirez-Velez, R.; Saavedra, J.M. Exercise, health outcomes, and paediatric obesity: A systematic review of meta-analyses. J. Sci. Med. Sport 2019, 22, 76–84. [Google Scholar] [CrossRef]
- Rosenwald, M.; Perdikari, A.; Rülicke, T.; Wolfrum, C. Bi-directional interconversion of brite and white adipocytes. Nat. Cell Biol. 2013, 15, 659–667. [Google Scholar] [CrossRef]
- Saito, M.; Okamatsu-Ogura, Y.; Matsushita, M.; Watanabe, K.; Yoneshiro, T.; Nio-Kobayashi, J.; Iwanaga, T.; Miyagawa, M.; Kameya, T.; Nakada, K. High incidence of metabolically active brown adipose tissue in healthy adult humans: Effects of cold exposure and adiposity. Diabetes 2009, 58, 1526–1531. [Google Scholar] [CrossRef]
- Zingaretti, M.C.; Crosta, F.; Vitali, A.; Guerrieri, M.; Frontini, A.; Cannon, B.; Nedergaard, J.; Cinti, S. The presence of UCP1 demonstrates that metabolically active adipose tissue in the neck of adult humans truly represents brown adipose tissue. FASEB J. 2009, 23, 3113–3120. [Google Scholar] [CrossRef]
- Gonzalez Porras, M.A.; Stojkova, K.; Vaicik, M.K.; Pelowe, A.; Goddi, A.; Carmona, A.; Long, B.; Qutub, A.A.; Gonzalez, A.; Cohen, R.N.; et al. Integrins and extracellular matrix proteins modulate adipocyte thermogenic capacity. Sci. Rep. 2021, 11, 5442. [Google Scholar] [CrossRef]
- van der Kolk, B.W.; Saari, S.; Lovric, A.; Arif, M.; Alvarez, M.; Ko, A.; Miao, Z.; Sahebekhtiari, N.; Muniandy, M.; Heinonen, S.; et al. Molecular pathways behind acquired obesity: Adipose tissue and skeletal muscle multiomics in monozygotic twin pairs discordant for BMI. Cell Rep. Med. 2021, 2, 100226. [Google Scholar] [CrossRef]
- Muniandy, M.; Heinonen, S.; Yki-Järvinen, H.; Hakkarainen, A.; Lundbom, J.; Lundbom, N.; Kaprio, J.; Rissanen, A.; Ollikainen, M.; Pietiläinen, K. Gene expression profile of subcutaneous adipose tissue in BMI-discordant monozygotic twin pairs unravels molecular and clinical changes associated with sub-types of obesity. Int. J. Obes. 2017, 41, 1176–1184. [Google Scholar] [CrossRef]
- Pietiläinen, K.H.; Naukkarinen, J.; Rissanen, A.; Saharinen, J.; Ellonen, P.; Keränen, H.; Suomalainen, A.; Götz, A.; Suortti, T.; Yki-Järvinen, H. Global transcript profiles of fat in monozygotic twins discordant for BMI: Pathways behind acquired obesity. PLoS Med. 2008, 5, e51. [Google Scholar] [CrossRef]
- Soronen, J.; Laurila, P.-P.; Naukkarinen, J.; Surakka, I.; Ripatti, S.; Jauhiainen, M.; Olkkonen, V.M.; Yki-Järvinen, H. Adipose tissue gene expression analysis reveals changes in inflammatory, mitochondrial respiratory and lipid metabolic pathways in obese insulin-resistant subjects. BMC Med Genom. 2012, 5, 9. [Google Scholar] [CrossRef]
- Das, S.K.; Ma, L.; Sharma, N. Adipose tissue gene expression and metabolic health of obese adults. Int. J. Obes. 2015, 39, 869–873. [Google Scholar] [CrossRef]
- Van der Kolk, B.W.; Kalafati, M.; Adriaens, M.; Van Greevenbroek, M.M.; Vogelzangs, N.; Saris, W.H.; Astrup, A.; Valsesia, A.; Langin, D.; Van der Kallen, C.J. Subcutaneous adipose tissue and systemic inflammation are associated with peripheral but not hepatic insulin resistance in humans. Diabetes 2019, 68, 2247–2258. [Google Scholar] [CrossRef]
- Rydén, M.; Hrydziuszko, O.; Mileti, E.; Raman, A.; Bornholdt, J.; Boyd, M.; Toft, E.; Qvist, V.; Näslund, E.; Thorell, A. The adipose transcriptional response to insulin is determined by obesity, not insulin sensitivity. Cell Rep. 2016, 16, 2317–2326. [Google Scholar] [CrossRef]
- Wiklund, P.; Zhang, X.; Pekkala, S.; Autio, R.; Kong, L.; Yang, Y.; Keinänen-Kiukaanniemi, S.; Alen, M.; Cheng, S. Insulin resistance is associated with altered amino acid metabolism and adipose tissue dysfunction in normoglycemic women. Sci. Rep. 2016, 6, 24540. [Google Scholar] [CrossRef]
- Heinonen, S.; Saarinen, L.; Naukkarinen, J.; Rodríguez, A.; Frühbeck, G.; Hakkarainen, A.; Lundbom, J.; Lundbom, N.; Vuolteenaho, K.; Moilanen, E. Adipocyte morphology and implications for metabolic derangements in acquired obesity. Int. J. Obes. 2014, 38, 1423–1431. [Google Scholar] [CrossRef]
- Yin, Z.; Deng, T.; Peterson, L.E.; Yu, R.; Lin, J.; Hamilton, D.J.; Reardon, P.R.; Sherman, V.; Winnier, G.E.; Zhan, M. Transcriptome analysis of human adipocytes implicates the NOD-like receptor pathway in obesity-induced adipose inflammation. Mol. Cell. Endocrinol. 2014, 394, 80–87. [Google Scholar] [CrossRef]
- Kulyte, A.; Ehrlund, A.; Arner, P.; Dahlman, I. Global transcriptome profiling identifies KLF15 and SLC25A10 as modifiers of adipocytes insulin sensitivity in obese women. PLoS ONE 2017, 12, e0178485. [Google Scholar] [CrossRef]
- Mastrangelo, A.; Panadero, M.I.; Pérez, L.M.; Gálvez, B.G.; García, A.; Barbas, C.; Rupérez, F.J. New insight on obesity and adipose-derived stem cells using comprehensive metabolomics. Biochem. J. 2016, 473, 2187–2203. [Google Scholar] [CrossRef]
- Price, A.; Stunkard, A.J. Commingling analysis of obesity in twins. Hum. Hered. 1989, 39, 121–135. [Google Scholar] [CrossRef]
- Price, R.A.; Ness, R.; Laskarzewski, P. Common major gene inheritance of extreme overweight. Hum. Biol. 1990, 62, 747–765. [Google Scholar]
- Jagannadham, J.; Jaiswal, H.K.; Agrawal, S.; Rawal, K. Comprehensive Map of Molecules Implicated in Obesity. PLoS ONE 2016, 11, e0146759. [Google Scholar] [CrossRef]
- Soung, Y.-H.; Clifford, J.L.; Chung, J. Crosstalk between integrin and receptor tyrosine kinase signaling in breast carcinoma progression. BMB Rep. 2010, 43, 311–318. [Google Scholar] [CrossRef]
- Beauvais, D.M.; Rapraeger, A.C. Syndecan-1 couples the insulin-like growth factor-1 receptor to inside-out integrin activation. J. Cell Sci. 2010, 123, 3796–3807. [Google Scholar] [CrossRef]
- Baron, W.; Decker, L.; Colognato, H. Regulation of integrin growth factor interactions in oligodendrocytes by lipid raft microdomains. Curr. Biol. 2003, 13, 151–155. [Google Scholar] [CrossRef]
- Seong, J.; Huang, M.; Sim, K.M.; Kim, H.; Wang, Y. FRET-based visualization of PDGF receptor activation at membrane microdomains. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef]
- Kim, Y.; Jung, K.; Baek, D.; Hong, S.; Kim, Y. Co-targeting of EGF receptor and neuropilin-1 overcomes cetuximab resistance in pancreatic ductal adenocarcinoma with integrin β1-driven Src-Akt bypass signaling. Oncogene 2017, 36, 2543–2552. [Google Scholar] [CrossRef]
- Seguin, L.; Kato, S.; Franovic, A.; Camargo, M.F.; Lesperance, J.; Elliott, K.C.; Yebra, M.; Mielgo, A.; Lowy, A.M.; Husain, H. An integrin β3–KRAS–RalB complex drives tumour stemness and resistance to EGFR inhibition. Nat. Cell Biol. 2014, 16, 457–468. [Google Scholar] [CrossRef]
- Fujita, M.; Takada, Y.K.; Takada, Y. Insulin-like growth factor (IGF) signaling requires αvβ3-IGF1-IGF type 1 receptor (IGF1R) ternary complex formation in anchorage independence, and the complex formation does not require IGF1R and Src activation. J. Biol. Chem. 2013, 288, 3059–3069. [Google Scholar] [CrossRef]
- Hynes, R.O. Integrins: A family of cell surface receptors. Cell 1987, 48, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Hynes, R.O. Integrins: Bidirectional, allosteric signaling machines. Cell 2002, 110, 673–687. [Google Scholar] [CrossRef]
- Calderwood, D.A.; Campbell, I.D.; Critchley, D.R. Talins and kindlins: Partners in integrin-mediated adhesion. Nat. Rev. Mol. Cell Biol. 2013, 14, 503–517. [Google Scholar] [CrossRef]
- Sun, Z.; Costell, M.; Fässler, R. Integrin activation by talin, kindlin and mechanical forces. Nat. Cell biol. 2019, 21, 25–31. [Google Scholar] [CrossRef]
- Williams, A.S.; Kang, L.; Wasserman, D.H. The extracellular matrix and insulin resistance. Trends Endocrinol. Metab. 2015, 26, 357–366. [Google Scholar] [CrossRef]
- Lin, D.; Chun, T.-H.; Kang, L. Adipose extracellular matrix remodelling in obesity and insulin resistance. Biochem. Pharmacol. 2016, 119, 8–16. [Google Scholar] [CrossRef]
- Mariman, E.; Wang, P. Adipocyte extracellular matrix composition, dynamics and role in obesity. Cell. Mol. Life Sci. 2010, 67, 1277–1292. [Google Scholar] [CrossRef]
- Bauters, D.; Cobbaut, M.; Geys, L.; Van Lint, J.; Hemmeryckx, B.; Lijnen, H.R. Loss of ADAMTS5 enhances brown adipose tissue mass and promotes browning of white adipose tissue via CREB signaling. Mol. Metab. 2017, 6, 715–724. [Google Scholar] [CrossRef]
- Thrailkill, K.M.; Clay Bunn, R.; Fowlkes, J.L. Matrix metalloproteinases: Their potential role in the pathogenesis of diabetic nephropathy. Endocrine 2009, 35, 1–10. [Google Scholar] [CrossRef]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the extracellular matrix in development and disease. Nat. Rev. Mol. Cell Biol. 2014, 15, 786–801. [Google Scholar]
- Nagase, H.; Visse, R.; Murphy, G. Structure and function of matrix metalloproteinases and TIMPs. Cardiovasc. Res. 2006, 69, 562–573. [Google Scholar] [CrossRef]
- Berg, G.; Miksztowicz, V.; Schreier, L. Metalloproteinases in metabolic syndrome. Clin. Chim. Acta 2011, 412, 1731–1739. [Google Scholar] [CrossRef]
- Glowinska-Olszewska, B.; Urban, M. Elevated matrix metalloproteinase 9 and tissue inhibitor of metalloproteinase 1 in obese children and adolescents. Metabolism 2007, 56, 799–805. [Google Scholar] [CrossRef]
- Cuevas, N.; Salamanca, A.; Rodriguez-Nunez, I. Impact of obesity on autonomic control in healthy children. In Proceedings of the Pediatric Research; Nature Publishing Group: Ney York, NY, USA, 2020; p. 1136. [Google Scholar]
- Belo, V.A.; Souza-Costa, D.C.; Luizon, M.R.; Lanna, C.M.; Carneiro, P.C.; Izidoro-Toledo, T.C.; Ferraz, K.C.; Tanus-Santos, J.E. Matrix metalloproteinase-9 genetic variations affect MMP-9 levels in obese children. Int. J. Obes. 2012, 36, 69–75. [Google Scholar] [CrossRef]
- Belo, V.A.; Luizon, M.R.; Carneiro, P.C.; Gomes, V.A.; Lacchini, R.; Lanna, C.M.; Souza-Costa, D.C.; Tanus-Santos, J.E. Effect of metabolic syndrome risk factors and MMP-2 genetic variations on circulating MMP-2 levels in childhood obesity. Mol. Biol. Rep. 2013, 40, 2697–2704. [Google Scholar] [CrossRef]
- Aguilera, C.M.; Gomez-Llorente, C.; Tofe, I.; Gil-Campos, M.; Canete, R.; Gil, A. Genome-wide expression in visceral adipose tissue from obese prepubertal children. Int. J. Mol. Sci. 2015, 16, 7723–7737. [Google Scholar] [CrossRef]
- Rosen, E.D.; Spiegelman, B.M. What we talk about when we talk about fat. Cell 2014, 156, 20–44. [Google Scholar]
- Lustig, R.H.; Collier, D.; Kassotis, C.; Roepke, T.A.; Kim, M.J.; Blanc, E.; Barouki, R.; Bansal, A.; Cave, M.C.; Chatterjee, S.; et al. Obesity I: Overview and molecular and biochemical mechanisms. Biochem. Pharmacol. 2022, 199, 115012. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Lee, P.; Chisholm, D.J.; James, D.E. Control of adipocyte differentiation in different fat depots; implications for pathophysiology or therapy. Front. Endocrinol. 2015, 6, 1. [Google Scholar] [CrossRef]
- Wu, Z.; Rosen, E.D.; Brun, R.; Hauser, S.; Adelmant, G.; Troy, A.E.; McKeon, C.; Darlington, G.J.; Spiegelman, B.M. Cross-regulation of C/EBPα and PPARγ controls the transcriptional pathway of adipogenesis and insulin sensitivity. Mol. Cell 1999, 3, 151–158. [Google Scholar] [CrossRef]
- Rosen, E.D.; Hsu, C.-H.; Wang, X.; Sakai, S.; Freeman, M.W.; Gonzalez, F.J.; Spiegelman, B.M. C/EBPα induces adipogenesis through PPARγ: A unified pathway. Genes Dev. 2002, 16, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Park, B.O.; Ahrends, R.; Teruel, M.N. Consecutive positive feedback loops create a bistable switch that controls preadipocyte-to-adipocyte conversion. Cell Rep. 2012, 2, 976–990. [Google Scholar] [CrossRef]
- Moseti, D.; Regassa, A.; Kim, W.-K. Molecular regulation of adipogenesis and potential anti-adipogenic bioactive molecules. Int. J. Mol. Sci. 2016, 17, 124. [Google Scholar] [CrossRef]
- Lefterova, M.I.; Haakonsson, A.K.; Lazar, M.A.; Mandrup, S. PPARγ and the global map of adipogenesis and beyond. Trends Endocrinol. Metab. 2014, 25, 293–302. [Google Scholar] [CrossRef]
- Vimalraj, S.; Arumugam, B.; Miranda, P.; Selvamurugan, N. Runx2: Structure, function, and phosphorylation in osteoblast differentiation. Int. J. Biol. Macromol. 2015, 78, 202–208. [Google Scholar] [CrossRef]
- Huang, W.; Yang, S.; Shao, J.; Li, Y.-P. Signaling and transcriptional regulation in osteoblast commitment and differentiation. Front. Biosci. A J. Virtual Libr. 2007, 12, 3068. [Google Scholar] [CrossRef]
- Zhang, Y.-y.; Li, X.; Qian, S.-w.; Guo, L.; Huang, H.-y.; He, Q.; Liu, Y.; Ma, C.-g.; Tang, Q.-Q. Down-regulation of type I Runx2 mediated by dexamethasone is required for 3T3-L1 adipogenesis. Mol. Endocrinol. 2012, 26, 798–808. [Google Scholar] [CrossRef]
- Rawal, K.; Patel, T.P.; Purohit, K.M.; Israni, K.; Kataria, V.; Bhatt, H.; Gupta, S. Influence of obese phenotype on metabolic profile, inflammatory mediators and stemness of hADSC in adipose tissue. Clin. Nutr. 2020, 39, 3829–3835. [Google Scholar] [CrossRef]
- Ruiz-Ojeda, F.J.; Anguita-Ruiz, A.; Rico, M.C.; Leis, R.; Bueno, G.; Gil-Campos, M.; Gil, Á.; Aguilera, C.M. Serum levels of the novel adipokine isthmin-1 are associated with obesity in pubertal boys. medRxiv 2022. [Google Scholar] [CrossRef]
- Heeren, J.; Scheja, L. Isthmin 1—A novel insulin-like adipokine. Nat. Rev. Endocrinol. 2021, 17, 709–710. [Google Scholar] [CrossRef]
- Jiang, Z.; Zhao, M.; Voilquin, L.; Jung, Y.; Aikio, M.A.; Sahai, T.; Dou, F.Y.; Roche, A.M.; Carcamo-Orive, I.; Knowles, J.W. Isthmin-1 is an adipokine that promotes glucose uptake and improves glucose tolerance and hepatic steatosis. Cell Metab. 2021, 33, 1836–1852.e1811. [Google Scholar] [CrossRef]
- Sanyaolu, A.; Okorie, C.; Qi, X.; Locke, J.; Rehman, S. Childhood and Adolescent Obesity in the United States: A Public Health Concern. Glob. Pediatr. Health 2019, 6, 2333794X19891305. [Google Scholar] [CrossRef]
- Ahmad, Q.I.; Ahmad, C.B.; Ahmad, S.M. Childhood obesity. Indian J. Endocrinol. Metab. 2010, 14, 19. [Google Scholar]
- van der Klaauw, A.A.; Farooqi, I.S. The hunger genes: Pathways to obesity. Cell 2015, 161, 119–132. [Google Scholar] [CrossRef]
- Handakas, E.; Chang, K.; Khandpur, N.; Vamos, E.P.; Millett, C.; Sassi, F.; Vineis, P.; Robinson, O. Metabolic profiles of ultra-processed food consumption and their role in obesity risk in British children. Clin. Nutr. 2022, 41, 2537–2548. [Google Scholar] [CrossRef]
- Gonzalez-Dominguez, A.; Armeni, M.; Savolainen, O.; Lechuga-Sancho, A.M.; Landberg, R.; Gonzalez-Dominguez, R. Untargeted Metabolomics Based on Liquid Chromatography-Mass Spectrometry for the Analysis of Plasma and Erythrocyte Samples in Childhood Obesity. Methods Mol. Biol. 2023, 2571, 115–122. [Google Scholar] [CrossRef]
- De Spiegeleer, M.; De Paepe, E.; Van Meulebroek, L.; Gies, I.; De Schepper, J.; Vanhaecke, L. Paediatric obesity: A systematic review and pathway mapping of metabolic alterations underlying early disease processes. Mol. Med. 2021, 27, 145. [Google Scholar] [CrossRef] [PubMed]
- Hosking, J.; Pinkney, J.; Jeffery, A.; Cominetti, O.; Da Silva, L.; Collino, S.; Kussmann, M.; Hager, J.; Martin, F.P. Insulin Resistance during normal child growth and development is associated with a distinct blood metabolic phenotype (Earlybird 72). Pediatr. Diabetes 2019, 20, 832–841. [Google Scholar] [CrossRef] [PubMed]
- Martos-Moreno, G.A.; Mastrangelo, A.; Barrios, V.; Garcia, A.; Chowen, J.A.; Ruperez, F.J.; Barbas, C.; Argente, J. Metabolomics allows the discrimination of the pathophysiological relevance of hyperinsulinism in obese prepubertal children. Int. J. Obes. 2017, 41, 1473–1480. [Google Scholar] [CrossRef] [PubMed]
- Perng, W.; Tang, L.; Song, P.X.K.; Tellez-Rojo, M.M.; Cantoral, A.; Peterson, K.E. Metabolomic profiles and development of metabolic risk during the pubertal transition: A prospective study in the ELEMENT Project. Pediatr. Res. 2019, 85, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Dong, Y.; Atefi, M.; Liu, Y.; Elshimali, Y.; Vadgama, J.V. Lactate, a Neglected Factor for Diabetes and Cancer Interaction. Mediat. Inflamm. 2016, 2016, 6456018. [Google Scholar] [CrossRef] [PubMed]
- Longo, M.; Zatterale, F.; Naderi, J.; Parrillo, L.; Formisano, P.; Raciti, G.A.; Beguinot, F.; Miele, C. Adipose Tissue Dysfunction as Determinant of Obesity-Associated Metabolic Complications. Int. J. Mol. Sci. 2019, 20, 2358. [Google Scholar] [CrossRef]
- Roy, D.; Modi, A.; Ghosh, R.; Ghosh, R.; Benito-Leon, J. Visceral Adipose Tissue Molecular Networks and Regulatory microRNA in Pediatric Obesity: An In Silico Approach. Int. J. Mol. Sci. 2022, 23, 1036. [Google Scholar] [CrossRef]
- Bouchard, C. Childhood obesity: Are genetic differences involved? Am. J. Clin. Nutr. 2009, 89, 1494S–1501S. [Google Scholar] [CrossRef]
- Horikoshi, M.; Beaumont, R.N.; Day, F.R.; Warrington, N.M.; Kooijman, M.N.; Fernandez-Tajes, J.; Feenstra, B.; van Zuydam, N.R.; Gaulton, K.J.; Grarup, N.; et al. Genome-wide associations for birth weight and correlations with adult disease. Nature 2016, 538, 248–252. [Google Scholar] [CrossRef]
- Maes, H.H.; Neale, M.C.; Eaves, L.J. Genetic and environmental factors in relative body weight and human adiposity. Behav. Genet. 1997, 27, 325–351. [Google Scholar] [CrossRef] [PubMed]
- Elks, C.E.; Den Hoed, M.; Zhao, J.H.; Sharp, S.J.; Wareham, N.J.; Loos, R.J.; Ong, K.K. Variability in the heritability of body mass index: A systematic review and meta-regression. Front. Endocrinol. 2012, 3, 29. [Google Scholar] [CrossRef]
- Loos, R.J.F.; Yeo, G.S.H. The genetics of obesity: From discovery to biology. Nat. Rev. Genet. 2022, 23, 120–133. [Google Scholar] [CrossRef]
- Shungin, D.; Winkler, T.W.; Croteau-Chonka, D.C.; Ferreira, T.; Locke, A.E.; Magi, R.; Strawbridge, R.J.; Pers, T.H.; Fischer, K.; Justice, A.E.; et al. New genetic loci link adipose and insulin biology to body fat distribution. Nature 2015, 518, 187–196. [Google Scholar] [CrossRef]
- Lagou, V.; Magi, R.; Hottenga, J.J.; Grallert, H.; Perry, J.R.B.; Bouatia-Naji, N.; Marullo, L.; Rybin, D.; Jansen, R.; Min, J.L.; et al. Sex-dimorphic genetic effects and novel loci for fasting glucose and insulin variability. Nat. Commun. 2021, 12, 24. [Google Scholar] [CrossRef]
- Wang, C.; Xu, Y. Mechanisms for Sex Differences in Energy Homeostasis. J. Mol. Endocrinol. 2019, 62, R129–R143. [Google Scholar] [CrossRef] [PubMed]
- Silventoinen, K.; Rokholm, B.; Kaprio, J.; Sorensen, T.I. The genetic and environmental influences on childhood obesity: A systematic review of twin and adoption studies. Int. J. Obes. 2010, 34, 29–40. [Google Scholar] [CrossRef]
- Claussnitzer, M.; Dankel, S.N.; Kim, K.H.; Quon, G.; Meuleman, W.; Haugen, C.; Glunk, V.; Sousa, I.S.; Beaudry, J.L.; Puviindran, V.; et al. FTO Obesity Variant Circuitry and Adipocyte Browning in Humans. N. Engl. J. Med. 2015, 373, 895–907. [Google Scholar] [CrossRef] [PubMed]
- Lillycrop, K.; Murray, R.; Cheong, C.; Teh, A.L.; Clarke-Harris, R.; Barton, S.; Costello, P.; Garratt, E.; Cook, E.; Titcombe, P.; et al. ANRIL Promoter DNA Methylation: A Perinatal Marker for Later Adiposity. EBioMedicine 2017, 19, 60–72. [Google Scholar] [CrossRef]
- Albuquerque, D.; Nobrega, C.; Rodriguez-Lopez, R.; Manco, L. Association study of common polymorphisms in MSRA, TFAP2B, MC4R, NRXN3, PPARGC1A, TMEM18, SEC16B, HOXB5 and OLFM4 genes with obesity-related traits among Portuguese children. J. Hum. Genet. 2014, 59, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Quan, L.L.; Wang, H.; Tian, Y.; Mu, X.; Zhang, Y.; Tao, K. Association of fat-mass and obesity-associated gene FTO rs9939609 polymorphism with the risk of obesity among children and adolescents: A meta-analysis. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 614–623. [Google Scholar]
- Warrington, N.M.; Howe, L.D.; Paternoster, L.; Kaakinen, M.; Herrala, S.; Huikari, V.; Wu, Y.Y.; Kemp, J.P.; Timpson, N.J.; St Pourcain, B.; et al. A genome-wide association study of body mass index across early life and childhood. Int. J. Epidemiol. 2015, 44, 700–712. [Google Scholar] [CrossRef] [PubMed]
- Pastor-Villaescusa, B.; Anguita-Ruiz, A.; Cañete, M.D.; Hoyos, R.; Maldonado Lozano, J.; Bueno-Lozano, G.; Leis Trabazo, M.R.; Gil, Á.; Cañete, R.; Aguilera, C.M. Genetic polymorphism of PPAR gamma modified the effects of metformin on BMI z-score in obese children. Ann. Nutr. Metab. 2017, 71, 354. [Google Scholar] [CrossRef]
- Fernández-Rhodes, L.; Graff, M.; Bradfield, J.; Wang, Y.; Parra, E.J.; Cruz, M.; Peralta-Romero, J.; Audirac-Chalifour, A.; Hidalgo, B.; Highland, H.; et al. The association of common genetic loci with childhood obesity in 1,612 hispanic/latino children and adolescents from across the United States and Mexico. Circulation 2017, 135, AP140. [Google Scholar] [CrossRef]
- Nagrani, R.; Foraita, R.; Gianfagna, F.; Iacoviello, L.; Marild, S.; Michels, N.; Molnar, D.; Moreno, L.; Russo, P.; Veidebaum, T.; et al. Common genetic variation in obesity, lipid transfer genes and risk of Metabolic Syndrome: Results from IDEFICS/I.Family study and meta-analysis. Sci. Rep. 2020, 10, 7189. [Google Scholar] [CrossRef]
- Dastgheib, S.A.; Bahrami, R.; Setayesh, S.; Salari, S.; Mirjalili, S.R.; Noorishadkam, M.; Sadeghizadeh-Yazdi, J.; Akbarian, E.; Neamatzadeh, H. Evidence from a meta-analysis for association of MC4R rs17782313 and FTO rs9939609 polymorphisms with susceptibility to obesity in children. Diabetes Metab. Syndr. 2021, 15, 102234. [Google Scholar] [CrossRef]
- Resende, C.M.M.; Silva, H.; Campello, C.P.; Ferraz, L.A.A.; de Lima, E.L.S.; Beserra, M.A.; Muniz, M.T.C.; da Silva, L.M.P. Polymorphisms on rs9939609 FTO and rs17782313 MC4R genes in children and adolescent obesity: A systematic review. Nutrition 2021, 91–92, 111474. [Google Scholar] [CrossRef] [PubMed]
- Mateus Pellenz, F.; Crispim, D.; Silveira Assmann, T. Systems biology approach identifies key genes and related pathways in childhood obesity. Gene 2022, 830, 146512. [Google Scholar] [CrossRef] [PubMed]
- Crispim, D.; Pellenz, F.M.; Assmann, T.S. Identification of Key Genes and Pathways for Childhood Obesity Using System Biology Approach Based on Comprehensive Gene Information. J. Endocr. Soc. 2021, 5, A49–A50. [Google Scholar] [CrossRef]
- Plaza-Florido, A.; Altmae, S.; Esteban, F.J.; Lof, M.; Radom-Aizik, S.; Ortega, F.B. Cardiorespiratory fitness in children with overweight/obesity: Insights into the molecular mechanisms. Scand J. Med. Sci. Sport. 2021, 31, 2083–2091. [Google Scholar] [CrossRef] [PubMed]
- Farooqi, I.S.; Yeo, G.S.; Keogh, J.M.; Aminian, S.; Jebb, S.A.; Butler, G.; Cheetham, T.; O’Rahilly, S. Dominant and recessive inheritance of morbid obesity associated with melanocortin 4 receptor deficiency. J. Clin. Investig. 2000, 106, 271–279. [Google Scholar] [CrossRef]
- Vaisse, C.; Clement, K.; Durand, E.; Hercberg, S.; Guy-Grand, B.; Froguel, P. Melanocortin-4 receptor mutations are a frequent and heterogeneous cause of morbid obesity. J. Clin. Investig. 2000, 106, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Raskiliene, A.; Smalinskiene, A.; Kriaucioniene, V.; Lesauskaite, V.; Petkeviciene, J. Associations of MC4R, LEP, and LEPR Polymorphisms with Obesity-Related Parameters in Childhood and Adulthood. Genes 2021, 12, 949. [Google Scholar] [CrossRef]
- Locke, A.E.; Kahali, B.; Berndt, S.I.; Justice, A.E.; Pers, T.H.; Day, F.R.; Powell, C.; Vedantam, S.; Buchkovich, M.L.; Yang, J.; et al. Genetic studies of body mass index yield new insights for obesity biology. Nature 2015, 518, 197–206. [Google Scholar] [CrossRef]
- Alotaibi, M.F. Physiology of puberty in boys and girls and pathological disorders affecting its onset. J. Adolesc. 2019, 71, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, P.D.S.; Ramos-Lobo, A.M.; Rosolen Tavares, M.; Wasinski, F.; Frazao, R.; Donato, J., Jr. Characterization of the onset of leptin effects on the regulation of energy balance. J. Endocrinol. 2021, 249, 239–251. [Google Scholar] [CrossRef] [PubMed]
- Ohga, H.; Ito, K.; Kakino, K.; Mon, H.; Kusakabe, T.; Lee, J.M.; Matsuyama, M. Leptin Is an Important Endocrine Player That Directly Activates Gonadotropic Cells in Teleost Fish, Chub Mackerel. Cells 2021, 10, 3505. [Google Scholar] [CrossRef] [PubMed]
- Park, H.K.; Ahima, R.S. Physiology of leptin: Energy homeostasis, neuroendocrine function and metabolism. Metabolism 2015, 64, 24–34. [Google Scholar] [CrossRef]
- Teixeira, P.; Dos Santos, P.B.; Pazos-Moura, C.C. The role of thyroid hormone in metabolism and metabolic syndrome. Ther. Adv. Endocrinol. Metab. 2020, 11, 2042018820917869. [Google Scholar] [CrossRef]
- Isozaki, O.; Tsushima, T.; Nozoe, Y.; Miyakawa, M.; Takano, K. Leptin regulation of the thyroids: Negative regulation on thyroid hormone levels in euthyroid subjects and inhibitory effects on iodide uptake and Na+/I- symporter mRNA expression in rat FRTL-5 cells. Endocr. J. 2004, 51, 415–423. [Google Scholar] [CrossRef][Green Version]
- Rashad, N.M.; Sayed, S.E.; Sherif, M.H.; Sitohy, M.Z. Effect of a 24-week weight management program on serum leptin level in correlation to anthropometric measures in obese female: A randomized controlled clinical trial. Diabetes Metab. Syndr. 2019, 13, 2230–2235. [Google Scholar] [CrossRef]
- Bidulescu, A.; Dinh, P.C., Jr.; Sarwary, S.; Forsyth, E.; Luetke, M.C.; King, D.B.; Liu, J.; Davis, S.K.; Correa, A. Associations of leptin and adiponectin with incident type 2 diabetes and interactions among African Americans: The Jackson heart study. BMC Endocr. Disord. 2020, 20, 31. [Google Scholar] [CrossRef]
- Meek, T.H.; Morton, G.J. Leptin, diabetes, and the brain. Indian J. Endocrinol. Metab. 2012, 16, S534–S542. [Google Scholar] [CrossRef]
- Olza, J.; Ruperez, A.I.; Gil-Campos, M.; Leis, R.; Canete, R.; Tojo, R.; Gil, A.; Aguilera, C.M. Leptin Receptor Gene Variant rs11804091 Is Associated with BMI and Insulin Resistance in Spanish Female Obese Children: A Case-Control Study. Int. J. Mol. Sci. 2017, 18, 1690. [Google Scholar] [CrossRef]
- Farooqi, I.S.; Jebb, S.A.; Langmack, G.; Lawrence, E.; Cheetham, C.H.; Prentice, A.M.; Hughes, I.A.; McCamish, M.A.; O’Rahilly, S. Effects of recombinant leptin therapy in a child with congenital leptin deficiency. N. Engl. J. Med. 1999, 341, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Farooqi, I.S.; Matarese, G.; Lord, G.M.; Keogh, J.M.; Lawrence, E.; Agwu, C.; Sanna, V.; Jebb, S.A.; Perna, F.; Fontana, S. Beneficial effects of leptin on obesity, T cell hyporesponsiveness, and neuroendocrine/metabolic dysfunction of human congenital leptin deficiency. J. Clin. Investig. 2002, 110, 1093–1103. [Google Scholar] [CrossRef] [PubMed]
- Licinio, J.; Caglayan, S.; Ozata, M.; Yildiz, B.O.; De Miranda, P.B.; O’Kirwan, F.; Whitby, R.; Liang, L.; Cohen, P.; Bhasin, S. Phenotypic effects of leptin replacement on morbid obesity, diabetes mellitus, hypogonadism, and behavior in leptin-deficient adults. Proc. Natl. Acad. Sci. USA 2004, 101, 4531–4536. [Google Scholar] [CrossRef] [PubMed]
- Ozata, M.; Ozdemir, I.C.; Licinio, J. Human leptin deficiency caused by a missense mutation: Multiple endocrine defects, decreased sympathetic tone, and immune system dysfunction indicate new targets for leptin action, greater central than peripheral resistance to the effects of leptin, and spontaneous correction of leptin-mediated defects. J. Clin. Endocrinol. Metab. 1999, 84, 3686–3695. [Google Scholar]
- Simonds, S.E.; Pryor, J.T.; Ravussin, E.; Greenway, F.L.; Dileone, R.; Allen, A.M.; Bassi, J.; Elmquist, J.K.; Keogh, J.M.; Henning, E. Leptin mediates the increase in blood pressure associated with obesity. Cell 2014, 159, 1404–1416. [Google Scholar] [CrossRef]
- Friedman, J.M. Leptin and the endocrine control of energy balance. Nat. Metab. 2019, 1, 754–764. [Google Scholar] [CrossRef]
- Scherer, P.E.; Williams, S.; Fogliano, M.; Baldini, G.; Lodish, H.F. A novel serum protein similar to C1q, produced exclusively in adipocytes. J. Biol. Chem. 1995, 270, 26746–26749. [Google Scholar] [CrossRef]
- Fain, J.N.; Madan, A.K.; Hiler, M.L.; Cheema, P.; Bahouth, S.W. Comparison of the release of adipokines by adipose tissue, adipose tissue matrix, and adipocytes from visceral and subcutaneous abdominal adipose tissues of obese humans. Endocrinology 2004, 145, 2273–2282. [Google Scholar] [CrossRef]
- Ahima, R.S. Metabolic actions of adipocyte hormones: Focus on adiponectin. Obesity 2006, 14, 9S–15S. [Google Scholar] [CrossRef]
- Ohashi, K.; Parker, J.L.; Ouchi, N.; Higuchi, A.; Vita, J.A.; Gokce, N.; Pedersen, A.A.; Kalthoff, C.; Tullin, S.; Sams, A.; et al. Adiponectin promotes macrophage polarization toward an anti-inflammatory phenotype. J. Biol. Chem. 2010, 285, 6153–6160. [Google Scholar] [CrossRef]
- Becic, T.; Studenik, C.; Hoffmann, G. Exercise Increases Adiponectin and Reduces Leptin Levels in Prediabetic and Diabetic Individuals: Systematic Review and Meta-Analysis of Randomized Controlled Trials. Med. Sci. 2018, 6, 97. [Google Scholar] [CrossRef]
- Yamauchi, T.; Kamon, J.; Ito, Y.; Tsuchida, A.; Yokomizo, T.; Kita, S.; Sugiyama, T.; Miyagishi, M.; Hara, K.; Tsunoda, M. Cloning of adiponectin receptors that mediate antidiabetic metabolic effects. Nature 2003, 423, 762–769. [Google Scholar] [CrossRef] [PubMed]
- Chait, A.; den Hartigh, L.J. Adipose Tissue Distribution, Inflammation and Its Metabolic Consequences, Including Diabetes and Cardiovascular Disease. Front. Cardiovasc. Med. 2020, 7, 22. [Google Scholar] [CrossRef] [PubMed]
- Fang, H.; Judd, R. Adiponectin regulation and function. Compr. Physiol. 2018, 8, 1031–1063. [Google Scholar] [PubMed]
- Ye, R.; Wang, M.; Wang, Q.A.; Scherer, P.E. Adiponectin-mediated antilipotoxic effects in regenerating pancreatic islets. Endocrinology 2015, 156, 2019–2028. [Google Scholar] [PubMed]
- Mandal, P.; Pratt, B.T.; Barnes, M.; McMullen, M.R.; Nagy, L.E. Molecular mechanism for adiponectin-dependent M2 macrophage polarization: Link between the metabolic and innate immune activity of full-length adiponectin. J. Biol. Chem. 2011, 286, 13460–13469. [Google Scholar] [PubMed]
- Caligiuri, A.; Bertolani, C.; Guerra, C.T.; Aleffi, S.; Galastri, S.; Trappoliere, M.; Vizzutti, F.; Gelmini, S.; Laffi, G.; Pinzani, M. Adenosine monophosphate–activated protein kinase modulates the activated phenotype of hepatic stellate cells. Hepatology 2008, 47, 668–676. [Google Scholar] [CrossRef]
- Kobashi, C.; Urakaze, M.; Kishida, M.; Kibayashi, E.; Kobayashi, H.; Kihara, S.; Funahashi, T.; Takata, M.; Temaru, R.; Sato, A.; et al. Adiponectin inhibits endothelial synthesis of interleukin-8. Circ. Res. 2005, 97, 1245–1252. [Google Scholar] [CrossRef]
- Kumada, M.; Kihara, S.; Ouchi, N.; Kobayashi, H.; Okamoto, Y.; Ohashi, K.; Maeda, K.; Nagaretani, H.; Kishida, K.; Maeda, N.; et al. Adiponectin specifically increased tissue inhibitor of metalloproteinase-1 through interleukin-10 expression in human macrophages. Circulation 2004, 109, 2046–2049. [Google Scholar] [CrossRef]
- Tu, W.J.; Qiu, H.C.; Liu, Y.K.; Liu, Q.; Zeng, X.; Zhao, J. Elevated levels of adiponectin associated with major adverse cardiovascular and cerebrovascular events and mortality risk in ischemic stroke. Cardiovasc. Diabetol. 2020, 19, 125. [Google Scholar] [CrossRef]
- Liu, Z.; Liang, S.; Que, S.; Zhou, L.; Zheng, S.; Mardinoglu, A. Meta-Analysis of Adiponectin as a Biomarker for the Detection of Metabolic Syndrome. Front. Physiol. 2018, 9, 1238. [Google Scholar] [CrossRef] [PubMed]
- Achari, A.E.; Jain, S.K. Adiponectin, a Therapeutic Target for Obesity, Diabetes, and Endothelial Dysfunction. Int. J. Mol. Sci. 2017, 18, 1321. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chu, W.S.; Hemphill, C.; Elbein, S.C. Human resistin gene: Molecular scanning and evaluation of association with insulin sensitivity and type 2 diabetes in Caucasians. J. Clin. Endocrinol. Metab. 2002, 87, 2520–2524. [Google Scholar] [CrossRef] [PubMed]
- Steppan, C.M.; Bailey, S.T.; Bhat, S.; Brown, E.J.; Banerjee, R.R.; Wright, C.M.; Patel, H.R.; Ahima, R.S.; Lazar, M.A. The hormone resistin links obesity to diabetes. Nature 2001, 409, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-H.; Lee, K.; Moon, Y.S.; Sul, H.S. A cysteine-rich adipose tissue-specific secretory factor inhibits adipocyte differentiation. J. Biol. Chem. 2001, 276, 11252–11256. [Google Scholar] [CrossRef]
- Steppan, C.M.; Brown, E.J.; Wright, C.M.; Bhat, S.; Banerjee, R.R.; Dai, C.Y.; Enders, G.H.; Silberg, D.G.; Wen, X.; Wu, G.D. A family of tissue-specific resistin-like molecules. Proc. Natl. Acad. Sci. USA 2001, 98, 502–506. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, D.; Kant, S.; Pandey, S.; Ehtesham, N.Z. Resistin in metabolism, inflammation, and disease. FEBS J. 2020, 287, 3141–3149. [Google Scholar] [CrossRef]
- Trujillo, M.; Scherer, P.E. Adiponectin–journey from an adipocyte secretory protein to biomarker of the metabolic syndrome. J. Intern. Med. 2005, 257, 167–175. [Google Scholar] [CrossRef]
- Yang, H.M.; Kim, J.; Shin, D.; Kim, J.Y.; You, J.; Lee, H.C.; Jang, H.D.; Kim, H.S. Resistin impairs mitochondrial homeostasis via cyclase-associated protein 1-mediated fission, leading to obesity-induced metabolic diseases. Metabolism 2022, 138, 155343. [Google Scholar] [CrossRef]
- Zapata, J.K.; Catalan, V.; Rodriguez, A.; Ramirez, B.; Silva, C.; Escalada, J.; Salvador, J.; Calamita, G.; Azcona-Sanjulian, M.C.; Fruhbeck, G.; et al. Resting Energy Expenditure Is Not Altered in Children and Adolescents with Obesity. Effect of Age and Gender and Association with Serum Leptin Levels. Nutrients 2021, 13, 1216. [Google Scholar] [CrossRef]
- Abawi, O.; Koster, E.C.; Welling, M.S.; Boeters, S.C.M.; van Rossum, E.F.C.; van Haelst, M.M.; van der Voorn, B.; de Groot, C.J.; van den Akker, E.L.T. Resting Energy Expenditure and Body Composition in Children and Adolescents With Genetic, Hypothalamic, Medication-Induced or Multifactorial Severe Obesity. Front. Endocrinol. 2022, 13, 862817. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, M.; Leibel, R.L. Adaptive thermogenesis in humans. Int. J. Obes. 2010, 34 (Suppl. S1), S47–S55. [Google Scholar] [CrossRef] [PubMed]
- Pandit, R.; Beerens, S.; Adan, R.A.H. Role of leptin in energy expenditure: The hypothalamic perspective. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2017, 312, R938–R947. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Physical Activity. Available online: https://www.who.int/news-room/fact-sheets/detail/physical-activity (accessed on 25 October 2022).
- Chaput, J.P.; Willumsen, J.; Bull, F.; Chou, R.; Ekelund, U.; Firth, J.; Jago, R.; Ortega, F.B.; Katzmarzyk, P.T. 2020 WHO guidelines on physical activity and sedentary behaviour for children and adolescents aged 5–17 years: Summary of the evidence. Int. J. Behav. Nutr. Phys. Act. 2020, 17, 141. [Google Scholar] [CrossRef]
- Cohen, T.R.; Hazell, T.J.; Vanstone, C.A.; Rodd, C.; Weiler, H.A. Changes in eating behavior and plasma leptin in children with obesity participating in a family-centered lifestyle intervention. Appetite 2018, 125, 81–89. [Google Scholar] [CrossRef]
- Reichelt, A.C.; Westbrook, R.F.; Morris, M.J. Integration of reward signalling and appetite regulating peptide systems in the control of food-cue responses. Br. J. Pharmacol. 2015, 172, 5225–5238. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, C.; Knoll-Pientka, N.; Mwanri, A.; Erfle, C.; Onywera, V.; Tremblay, M.S.; Buhlmeier, J.; Luzak, A.; Ferland, M.; Schulz, H.; et al. Low leptin levels are associated with elevated physical activity among lean school children in rural Tanzania. BMC Public Health 2022, 22, 933. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Lyu, S.; Zhang, J. Effects of Aerobic Exercise on the Serum Leptin Level and Heart Rate Variability in the Obese Girl Children. Comput. Intell. Neurosci. 2022, 2022, 2298994. [Google Scholar] [CrossRef] [PubMed]
- Salahshoornezhad, S.; Sohrabi, Z.; Mani, A.; Abdelbasset, W.K.; Mehrabi, M.; Zare, M.; Mehrabani, S.; Gerami, S.; Haghighat, N.; Akbarzadeh, M.; et al. Effect of a multi-disciplinary program on anthropometric and biochemical parameters in obese and overweight elementary school girls: A randomized clinical trial. Nutr. Metab. Cardiovasc. Dis. 2022, 32, 1982–1989. [Google Scholar] [CrossRef]
- Cordova, A.; Villa, G.; Sureda, A.; Rodriguez-Marroyo, J.A.; Sanchez-Collado, M.P. Physical activity and cardiovascular risk factors in Spanish children aged 11-13 years. Rev. Esp. Cardiol. 2012, 65, 620–626. [Google Scholar] [CrossRef]
- Leis, R.; Jurado-Castro, J.M.; Llorente-Cantarero, F.J.; Anguita-Ruiz, A.; Iris-Ruperez, A.; Bedoya-Carpente, J.J.; Vazquez-Cobela, R.; Aguilera, C.M.; Bueno, G.; Gil-Campos, M. Cluster Analysis of Physical Activity Patterns, and Relationship with Sedentary Behavior and Healthy Lifestyles in Prepubertal Children: Genobox Cohort. Nutrients 2020, 12, 1288. [Google Scholar] [CrossRef] [PubMed]
- Ascenso, A.; Palmeira, A.; Pedro, L.M.; Martins, S.; Fonseca, H. Physical activity and cardiorespiratory fitness, but not sedentary behavior, are associated with carotid intima-media thickness in obese adolescents. Eur. J. Pediatr. 2016, 175, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Sirico, F.; Bianco, A.; D’Alicandro, G.; Castaldo, C.; Montagnani, S.; Spera, R.; Di Meglio, F.; Nurzynska, D. Effects of Physical Exercise on Adiponectin, Leptin, and Inflammatory Markers in Childhood Obesity: Systematic Review and Meta-Analysis. Child. Obes. 2018, 14, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Willis, L.H.; Slentz, C.A.; Bateman, L.A.; Shields, A.T.; Piner, L.W.; Bales, C.W.; Houmard, J.A.; Kraus, W.E. Effects of aerobic and/or resistance training on body mass and fat mass in overweight or obese adults. J. Appl. Physiol. 2012, 113, 1831–1837. [Google Scholar] [CrossRef]
- Belcher, B.R.; Chou, C.P.; Nguyen-Rodriguez, S.T.; Hsu, Y.W.; Byrd-Williams, C.E.; McClain, A.D.; Weigensberg, M.J.; Spuijt-Metz, D. Leptin predicts a decline in moderate to vigorous physical activity in minority female children at risk for obesity. Pediatr. Obes. 2013, 8, 70–77. [Google Scholar] [CrossRef]
- Jimenez-Pavon, D.; Ortega, F.B.; Artero, E.G.; Labayen, I.; Vicente-Rodriguez, G.; Huybrechts, I.; Moreno, L.A.; Manios, Y.; Beghin, L.; Polito, A.; et al. Physical activity, fitness, and serum leptin concentrations in adolescents. J. Pediatr. 2012, 160, 598–603.e592. [Google Scholar] [CrossRef]
- Miyatake, N.; Murakami, H.; Kawakami, R.; Tabata, I.; Miyachi, M.; Group, N.S. Circulating leptin levels are associated with physical activity or physical fitness in Japanese. Environ. Health Prev. Med. 2014, 19, 362–366. [Google Scholar] [CrossRef][Green Version]
- Remmel, L.; Tillmann, V.; Purge, P.; Latt, E.; Jurimae, J. Associations of serum leptin, ghrelin and peptide YY levels with physical activity and cardiorespiratory fitness in adolescent boys with different BMI values. Biol. Sport 2017, 34, 345–352. [Google Scholar] [CrossRef]
- Mangge, H.; Ciardi, C.; Becker, K.; Strasser, B.; Fuchs, D.; Gostner, J.M. Influence of Antioxidants on Leptin Metabolism and its Role in the Pathogenesis of Obesity. Adv. Exp. Med. Biol. 2017, 960, 399–413. [Google Scholar] [CrossRef]
- Gomez, S.F.; Rajmil, L. Advertising, obesity and child health: The case of Spain. BMJ Paediatr. Open 2022, 6, e001482. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Childhood Overweight & Obesity. Available online: https://www.cdc.gov/obesity/childhood/index.html (accessed on 29 October 2022).
- Garcia-Solano, M.; Gutierrez-Gonzalez, E.; Lopez-Sobaler, A.M.; Ruiz-Alvarez, M.; Bermejo Lopez, L.M.; Aparicio, A.; Garcia-Lopez, M.A.; Yusta-Boyo, M.J.; Robledo de Dios, T.; Villar Villalba, C.; et al. Weight status in the 6- to 9-year-old school population in Spain: Results of the ALADINO 2019 Study. Nutr. Hosp. 2021, 38, 943–953. [Google Scholar] [CrossRef] [PubMed]
- Pedro Angel, L.R.; Beatriz, B.A.; Jeronimo, A.V.; Antonio, P.V. Effects of a 10-week active recess program in school setting on physical fitness, school aptitudes, creativity and cognitive flexibility in elementary school children. A randomised-controlled trial. J. Sports Sci. 2021, 39, 1277–1286. [Google Scholar] [CrossRef] [PubMed]
- Santiago-Rodriguez, M.E.; Chen, J.; Pfeiffer, K.A.; Marquez, D.X.; Odoms-Young, A.; Bustamante, E.E. Developmental disparities in sedentary time by period of the day among US youth: A cross-sectional study. BMC Public Health 2022, 22, 2047. [Google Scholar] [CrossRef] [PubMed]
- Brazendale, K.; Beets, M.W.; Armstrong, B.; Weaver, R.G.; Hunt, E.T.; Pate, R.R.; Brusseau, T.A.; Bohnert, A.M.; Olds, T.; Tassitano, R.M.; et al. Children’s moderate-to-vigorous physical activity on weekdays versus weekend days: A multi-country analysis. Int. J. Behav. Nutr. Phys. Act. 2021, 18, 28. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aragón-Vela, J.; Alcalá-Bejarano Carrillo, J.; Moreno-Racero, A.; Plaza-Diaz, J. The Role of Molecular and Hormonal Factors in Obesity and the Effects of Physical Activity in Children. Int. J. Mol. Sci. 2022, 23, 15413. https://doi.org/10.3390/ijms232315413
Aragón-Vela J, Alcalá-Bejarano Carrillo J, Moreno-Racero A, Plaza-Diaz J. The Role of Molecular and Hormonal Factors in Obesity and the Effects of Physical Activity in Children. International Journal of Molecular Sciences. 2022; 23(23):15413. https://doi.org/10.3390/ijms232315413
Chicago/Turabian StyleAragón-Vela, Jerónimo, Jesús Alcalá-Bejarano Carrillo, Aurora Moreno-Racero, and Julio Plaza-Diaz. 2022. "The Role of Molecular and Hormonal Factors in Obesity and the Effects of Physical Activity in Children" International Journal of Molecular Sciences 23, no. 23: 15413. https://doi.org/10.3390/ijms232315413
APA StyleAragón-Vela, J., Alcalá-Bejarano Carrillo, J., Moreno-Racero, A., & Plaza-Diaz, J. (2022). The Role of Molecular and Hormonal Factors in Obesity and the Effects of Physical Activity in Children. International Journal of Molecular Sciences, 23(23), 15413. https://doi.org/10.3390/ijms232315413








