Neomycin, but Not Neamine, Blocks Angiogenic Factor Induced Nitric Oxide Release through Inhibition of Akt Phosphorylation
Abstract
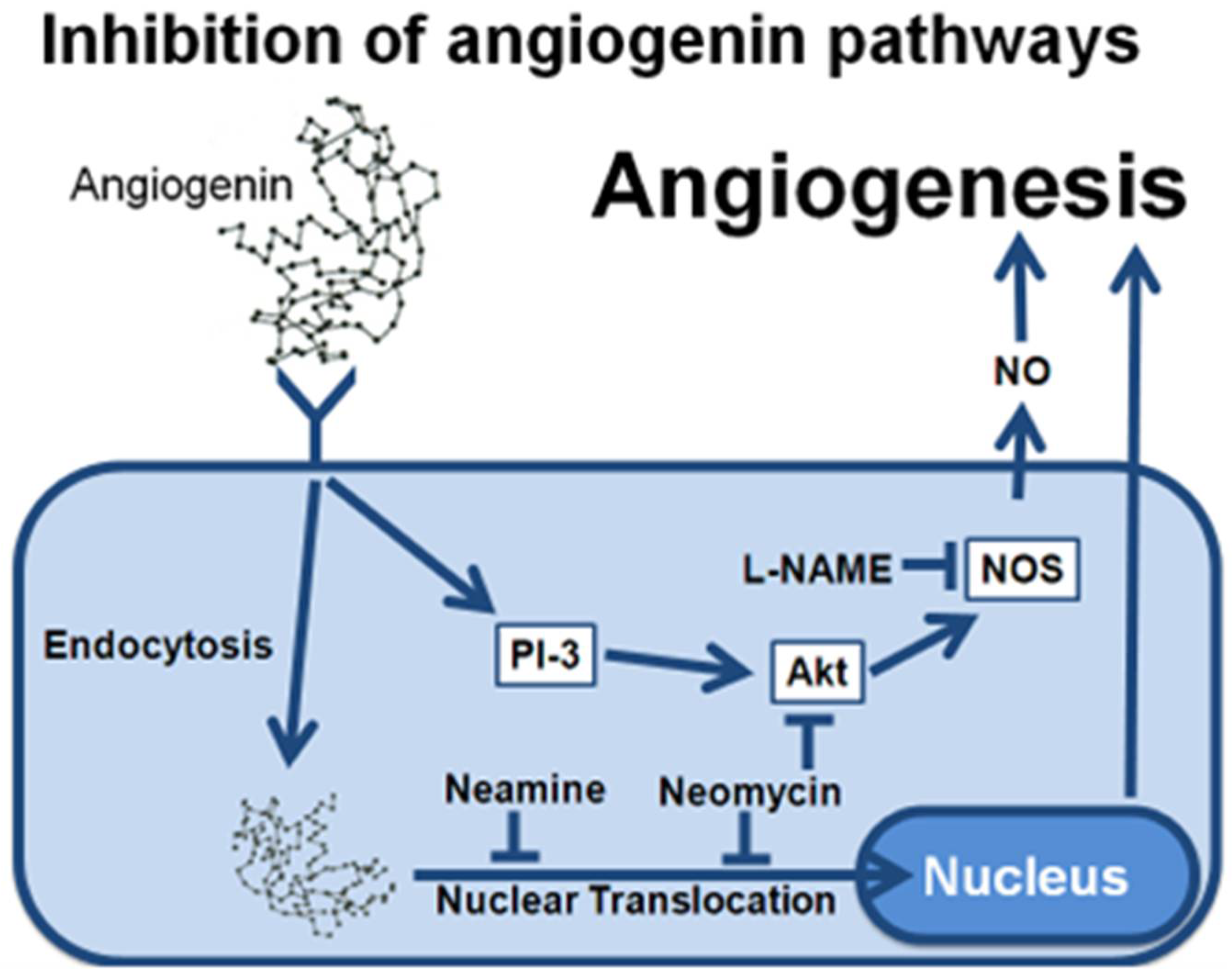
1. Introduction
2. Results
2.1. Description of the Electrochemical Assays
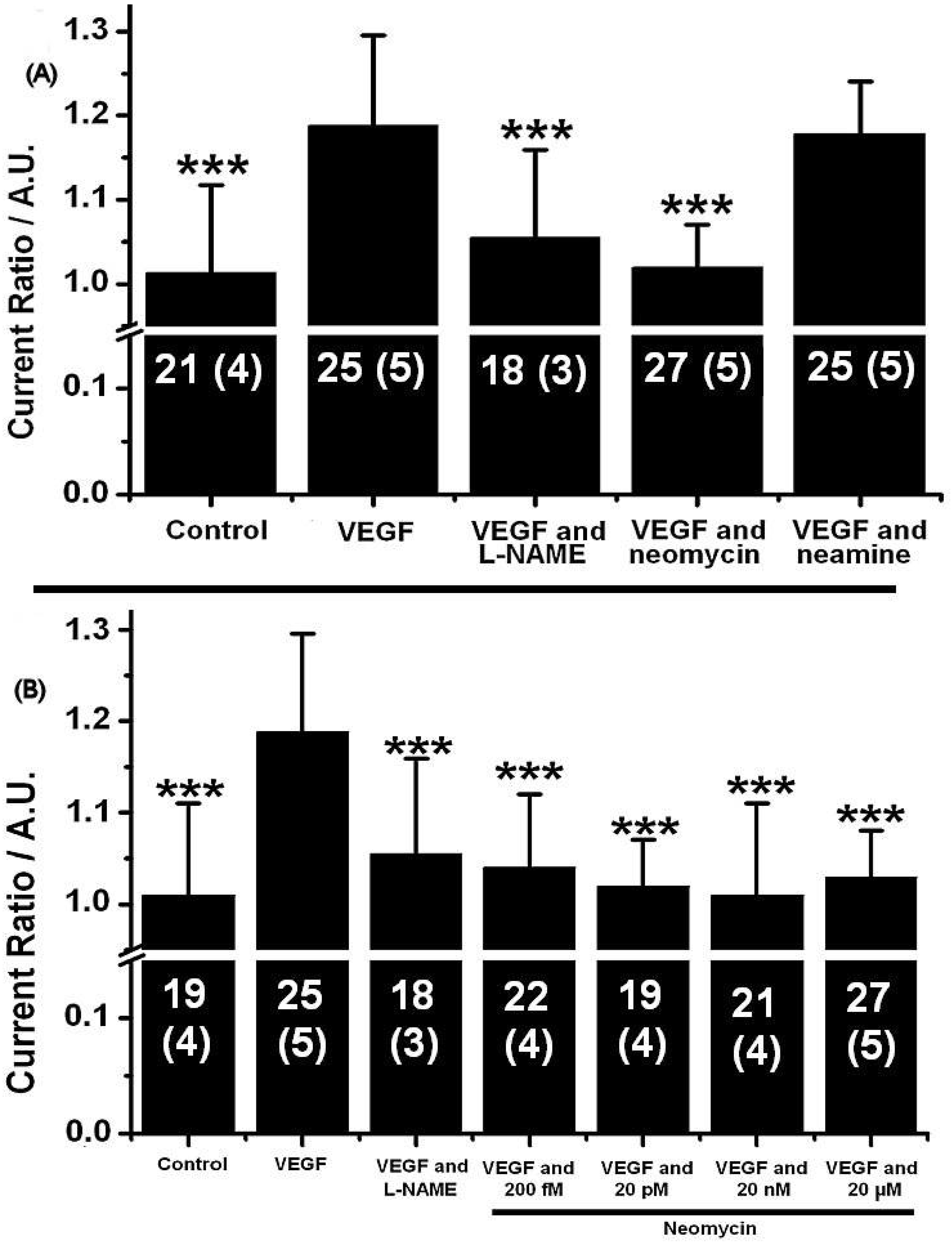
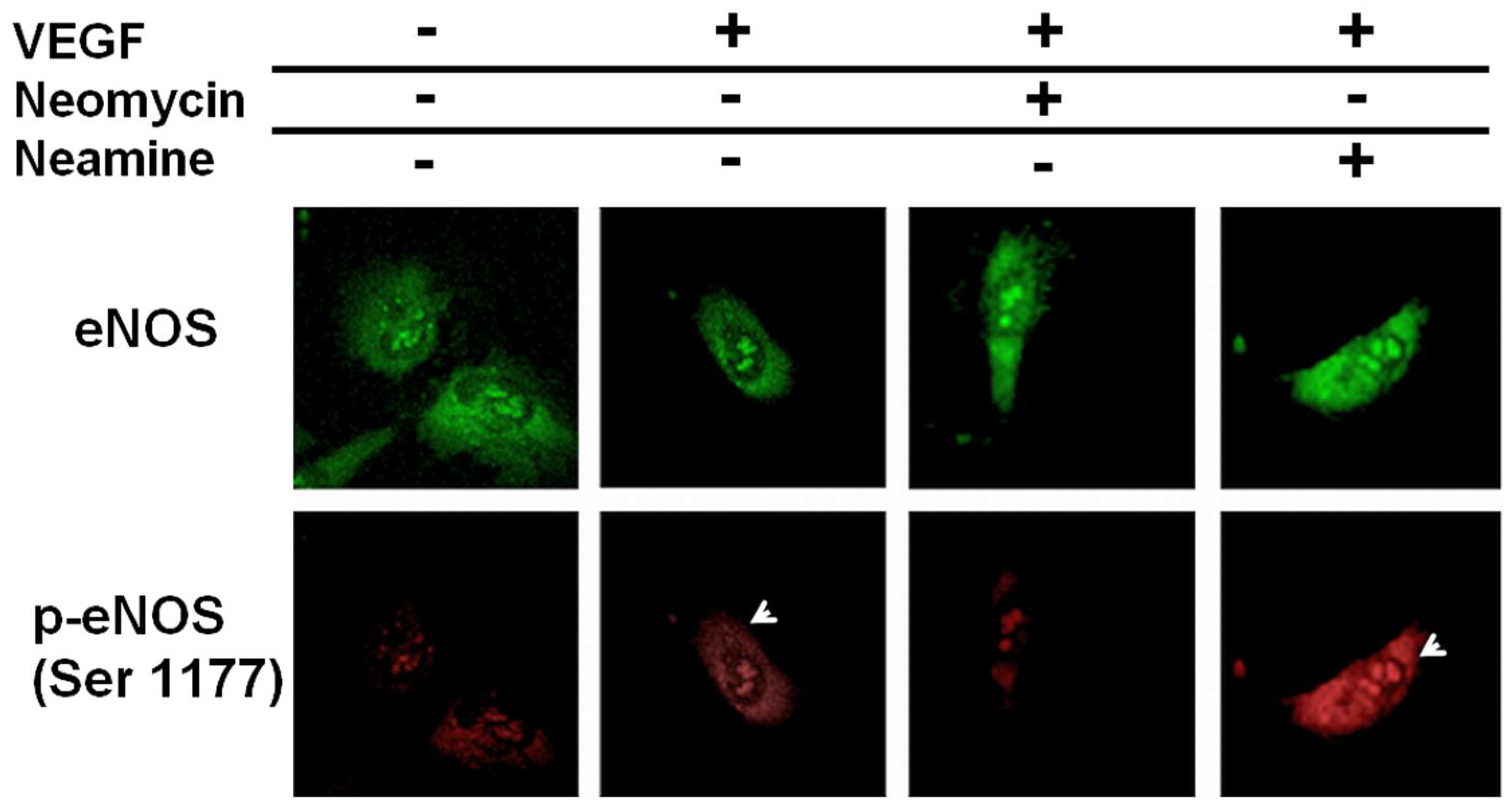
2.2. Neomycin Blocks eNOS Phosphorylation after Exposure to VEGF
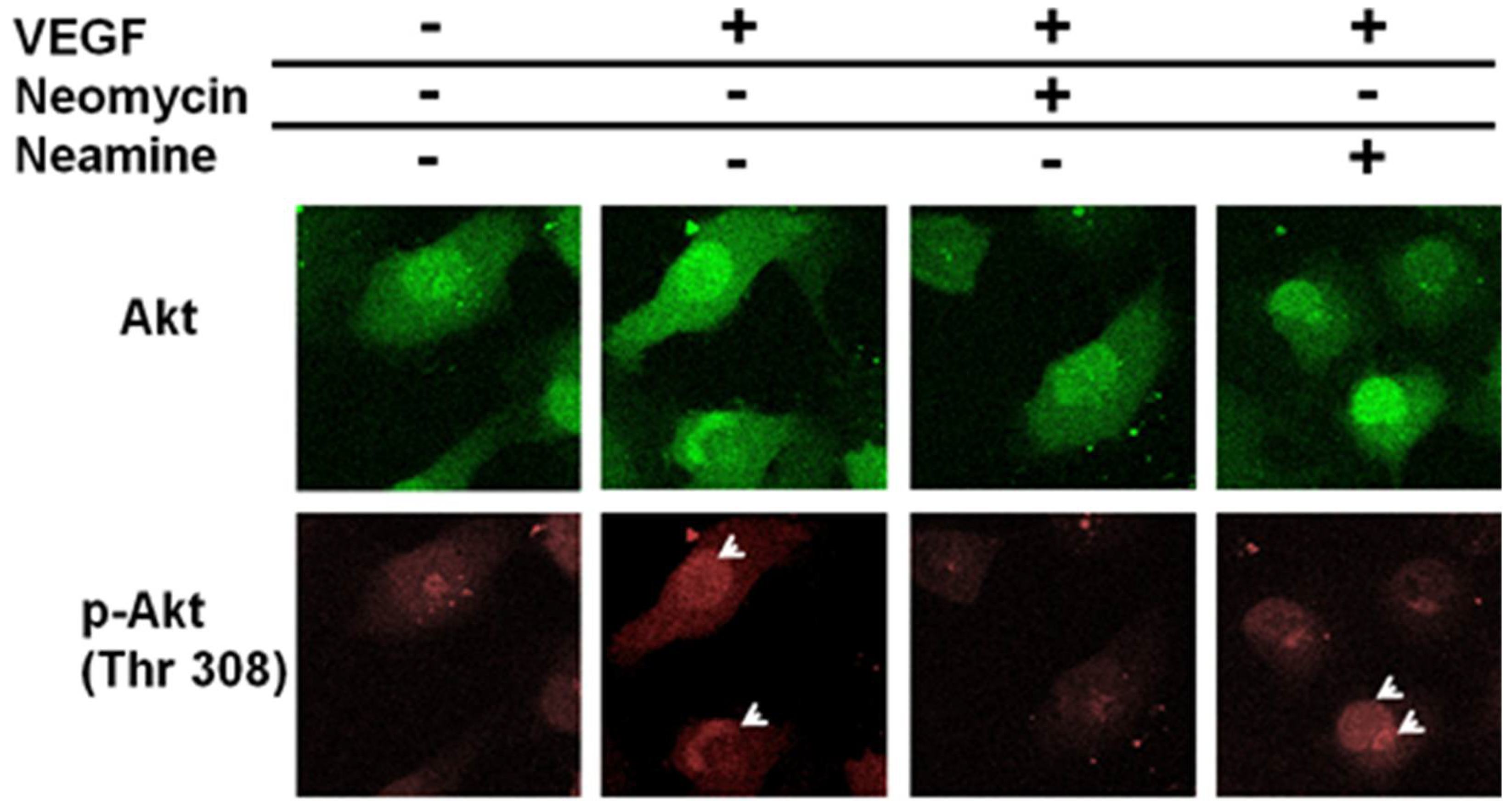
2.3. The Activation of Akt Is Inhibited by Neomycin, Not by Neamine
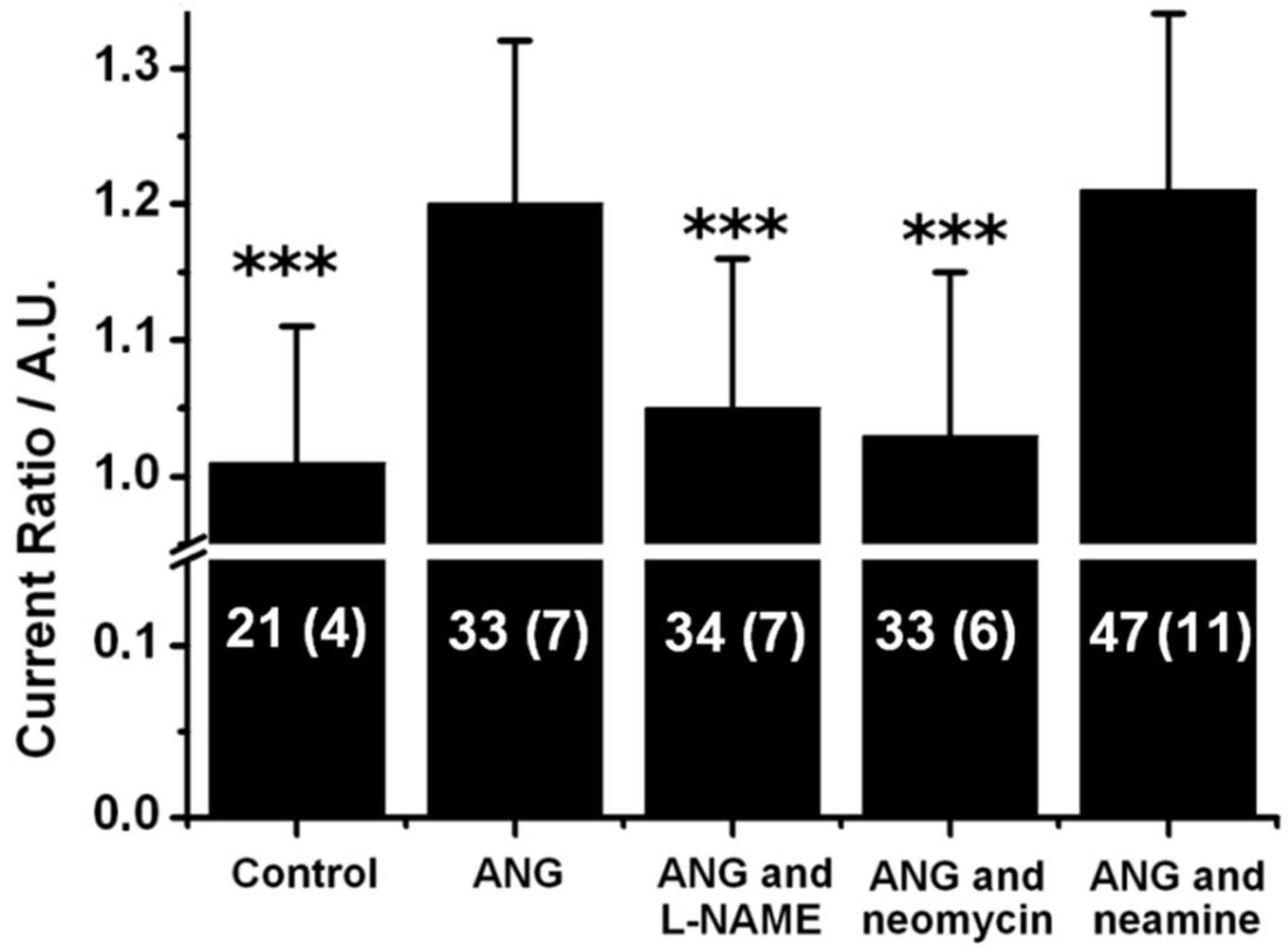
2.4. Neamine Does Not Inhibit ANG Evoked NO Release
3. Discussion
4. Materials and Methods
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Carmeliet, P.; Jain, R.K. Angiogenesis in cancer and other diseases. Nature 2000, 407, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Leung, D.W.; Cachianes, G.; Kuang, W.-J.; Goeddel, D.V.; Ferrara, N. Vascular Endothelial Growth Factor Is a Secreted Angiogenic Mitogen. Science 1989, 246, 1306–1309. [Google Scholar] [CrossRef]
- Höckel, M.; Schlenger, K.; Aral, B.; Mitze, M.; Scha¨ffer, U.; Vaupel, P. Association between tumour hypoxia and malignant progression in advanced cancer of the uterine cervix. Cancer Res. 1996, 56, 4509–4515. [Google Scholar] [PubMed]
- Höckel, M.; Vaupel, P. Biological consequences of tumour hypoxia. Semin. Oncol. 2001, 28, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Weidner, N.; Carroll, P.R.; Flax, J.; Blumenfeld, W.; Folkman, J. Tumor angiogenesis correlates with metastasis in invasive prostate carcinoma. Am. J. Pathol. 1993, 143, 401–409. [Google Scholar] [PubMed]
- Weidner, N.; Semple, J.P.; Welch, W.R.; Folkman, J. Tumor angiogenesis and metastasis—Correlation in invasive breast carcinoma. N. Engl. J. Med. 1991, 324, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Batchelor, T.T.; Duda, D.G.; di Tomaso, E.; Ancukiewicz, M.; Plotkin, S.R.; Gerstner, E.; Eichler, A.F.; Drappatz, J.; Hochberg, F.H.; Benner, T.; et al. Phase II study of Cediranib, an oral pan–vascular endothelial growth factor receptor tyrosine kinase inhibitor, in patients with recurrent glioblastoma. J. Clin. Oncol. 2010, 28, 2817–2823. [Google Scholar] [CrossRef]
- Kerbel, R.; Folkman, J. Clinical translation of angiogenesis inhibitors. Nat. Rev. Cancer 2002, 2, 727–739. [Google Scholar] [CrossRef]
- Hu, G.F. 1998 Neomycin inhibits angiogenin-induced angiogenesis. Proc. Natl. Acad. Sci. USA 1998, 95, 9791–9795. [Google Scholar] [CrossRef]
- Falco, F.G.; Smith, H.M.; Arcieri, G.M. Nephrotoxicity of aminoglycosides and gentamicin. J. Infect. Dis. 1969, 119, 406–409. [Google Scholar] [CrossRef]
- Schacht, J. Biochemistry of neomycin ototoxicity. J. Acoust. Soc. Am. 1976, 59, 940–944. [Google Scholar] [CrossRef] [PubMed]
- Hirukawa, S.; Olson, K.A.; Tsuji, T.; Hu, G.F. Neamine inhibits xenografic human tumour growth and angiogenesis in athymic mice. Clin. Cancer Res. 2005, 11, 8745–8752. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ibaragi, S.; Yoshioka, N.; Li, S.; Hu, M.G.; Hirukawa, S.; Sadow, P.M.; Hu, G.F. Neamine inhibits prostate cancer growth by suppressing angiogenin- mediated rRNA transcription. Clin. Cancer Res. 2009, 15, 1981–1988. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, K.; Liu, S.; Tsuji, T.; Olson, K.; Hu, G.F. Endogenous angiogenin in endothelial cells is a general requirement for cell proliferation and angiogenesis. Oncogene 2004, 24, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Bredt, D.S.; Hwang, P.M.; Snyder, S.H. Localization of nitric oxide synthase indicating a neural role for nitric oxide. Nature 1990, 347, 768–770. [Google Scholar] [CrossRef]
- Bogdan, C. Nitric oxide and the immune response. Nat. Immunol. 2001, 2, 907–916. [Google Scholar] [CrossRef]
- Moncada, S.; Palmer, R.M.; Higgs, E.A. Nitric oxide: Physiology, pathophysiology, and pharmacology. Pharmacol. Rev. 1991, 43, 109–142. [Google Scholar]
- Moncada, S.; Higgs, A. The L-arginine-nitric oxide pathway. N. Engl. J. Med. 1993, 329, 2002–2012. [Google Scholar]
- Rees, D.D.; Palmer, R.M.; Schulz, R.; Hodson, H.F.; Moncada, S. Characterization of three inhibitors of endothelial nitric oxide synthase in vitro and in vivo. Br. J. Pharmacol. 1990, 101, 746–752. [Google Scholar] [CrossRef]
- Huang, P.L.; Huang, Z.; Mashimo, H.; Bloch, K.D.; Moskowitz, M.A.; Bevan, J.A.; Fishman, M.C. Hypertension in mice lacking the gene for endothelial nitric oxide synthase. Nature 1995, 377, 239–242. [Google Scholar] [CrossRef]
- Buga, G.M.; Gold, M.E.; Fukuto, J.M.; Ignarro, L.J. Shear stress-induced release of nitric oxide from endothelial cells grown on beads. Hypertension 1991, 17, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Joannides, R.; Haefeli, W.E.; Linder, L.; Richard, V.; Bakkali, E.H.; Thuillez, C.; Lüscher, T.F. Nitric oxide is responsible for flow-dependent dilatation of human peripheral conduit arteries in vivo. Circulation 1995, 91, 1314–1319. [Google Scholar] [CrossRef] [PubMed]
- Trouillon, R.; Cheung, C.; Patel, B.A.; O’Hare, D. Electrochemical study of the intracellular transduction of vascular endothelial growth factor induced nitric oxide synthase activity using a multi-channel biocompatible microelectrode array. BBA-Gen. Subj. 2010, 1800, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Trouillon, R.; Kang, D.K.; Park, H.; Chang, S.I.; O’Hare, D. Angiogenin induces nitric oxide synthesis in endothelial cells through PI-3 and Akt kinases. Biochemistry 2010, 49, 3282–3288. [Google Scholar] [CrossRef] [PubMed]
- Ziche, M.; Morbidelli, L.; Choudhuri, R.; Zhang, H.T.; Donnini, S.; Granger, H.J.; Bicknell, R. Nitric oxide synthase lies downstream from vascular endothelial growth factor-induced but not basic fibroblast growth factor-induced angiogenesis. J. Clin. Investig. 1997, 99, 2625–2634. [Google Scholar] [CrossRef] [PubMed]
- Kubes, P.; Granger, D.N. Nitric oxide modulates microvascular permeability. Am. J. Physiol.-Heart Circ. Physiol. 1992, 262, H611–H615. [Google Scholar] [CrossRef]
- Gao, F.; Gao, E.; Yue, T.L.; Ohlstein, E.H.; Lopez, B.L.; Christopher, T.A.; Ma, X.L. Nitric oxide mediates the antiapoptotic effect of insulin in myocardial ischemia-reperfusion. Circulation 2002, 105, 1497–1502. [Google Scholar] [CrossRef]
- Michell, B.J.; Griffiths, J.E.; Mitchelhill, K.I.; Rodriguez-Crespo, I.; Tiganis, T.; Bozinovski, S.; De Montellano, P.R.; Kemp, B.E.; Pearson, R.B. The Akt kinase signals directly to endothelial nitric oxide synthase. Curr. Biol. 1999, 9, 845–848. [Google Scholar] [CrossRef]
- Xin, Z.; Kim, W.-S.; Hatcher, N.; Potgieter, K.; Moroz, L.L.; Gillette, R.; Sweedler, J.V. Interfering with Nitric Oxide Measurements: 4,5-diaminofluorescein reacts with dehydroascorbic acid and ascorbic acid. J. Biol. Chem. 2002, 277, 48472–48478. [Google Scholar]
- Trouillon, R.; Kang, D.K.; Chang, S.I.; O’Hare, D. Angiogenin induces nitric oxide release independently from its RNase activity. Chem. Commun. 2011, 47, 3421–3423. [Google Scholar] [CrossRef]
- Trouillon, R.; Cheung, C.; Patel, B.A.; O’Hare, D. Comparative study of poly (styrene-sulfonate)/poly (L-lysine) and fibronectin as biofouling-preventing layers in dissolved oxygen electrochemical measurements. Analyst 2009, 134, 784–793. [Google Scholar] [CrossRef] [PubMed]
- Trouillon, R.; Combs, Z.; Patel, B.A.; O’Hare, D. Comparative study of the effect of various electrode membranes on biofouling and electrochemical measurements. Electrochem. Commun. 2009, 11, 1409–1413. [Google Scholar] [CrossRef]
- Miyazaki, T.; Honda, K.; Ohata, H. Requirement of Ca2+ influx-and phosphatidylinositol 3-kinase-mediated m-calpain activity for shear stress-induced endothelial cell polarity. Am. J. Physiol.-Cell Physiol. 2007, 293, C1216–C1225. [Google Scholar] [CrossRef] [PubMed]
- Chang, F.; Lee, J.T.; Navolanic, P.M.; Steelman, L.S.; Shelton, J.G.; Blalock, W.L.; Franklin, R.A.; McCubrey, J.A. Involvement of PI3k/Akt pathway in cell cycle progression, apoptosis, and neoplastic transformation: A target for cancer chemotherapy. Leukemia 2003, 17, 590–603. [Google Scholar] [CrossRef]
- Madrid, L.V.; Wang, C.Y.; Guttridge, D.C.; Schottelius AJ, G.; Baldwin Jr, A.S.; Mayo, M.W. Akt suppresses apoptosis by stimulating the transactivation potential of the RelA/p65 subunit of NF-κ B. Mol. Cell. Biol. 2000, 20, 1626–1638. [Google Scholar] [CrossRef]
- Trouillon, R.; O’Hare, D.; Chang, S.I. An electrochemical functional assay for the sensing of nitric oxide release induced by angiogenic factors. BMB Rep. 2011, 44, 699–704. [Google Scholar] [CrossRef]
- Trouillon, R. Biological applications of the electrochemical sensing of nitric oxide: Fundamentals and recent developments. Biol. Chem. 2012, 394, 17–33. [Google Scholar] [CrossRef]
- Trouillon, R.; Williamson, E.D.; Saint, R.J.; O’Hare, D. Electrochemical detection of the binding of Bacillus anthracis protective antigen (PA) to the membrane receptor on macrophages through release of nitric oxide. Biosens. Bioelectron. 2012, 3, 138–144. [Google Scholar] [CrossRef]
- Shiojima, I.; Walsh, K. Role of Akt signaling in vascular homeostasis and angiogenesis. Circ. Res. 2002, 90, 1243–1250. [Google Scholar] [CrossRef]
- Hu, G.F.; Riordan, J.F.; Vallee, B.L. Angiogenin promotes invasiveness of cultured endothelial cells by stimulation of cell-associated proteolytic activities. Proc. Natl. Acad. Sci. USA 1994, 91, 12096–12100. [Google Scholar] [CrossRef]
- Kim, H.M.; Kang, D.K.; Kim, H.Y.; Kang, S.S.; Chang, S.I. Angiogenin-induced protein kinase B/Akt activation is necessary for angiogenesis but is independent of nuclear translocation of angiogenin in huve cells. Biochem. Biophys. Res. Commun. 2007, 352, 509–513. [Google Scholar] [CrossRef]
- Liu, S.; Yu, D.; Xu, Z.; Riordan, J.F.; Hu, G.F. Angiogenin activates ERK1/2 in human umbilical vein endothelial cells. Biochem. Biophys. Res. Commun. 2001, 287, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.; Xu, C.; Riordan, J.F. Human angiogenin is rapidly translocated to the nucleus of human umbilical vein endothelial cells and binds to DNA. J. Cell Biochem. 2000, 76, 452–462. [Google Scholar] [CrossRef]
- Chen, C.Z.; Shapiro, R. Site-specific mutagenesis reveals differences in the structural bases for tight binding of RNase inhibitor to angiogenin and RNase a. Proc. Natl. Acad. Sci. USA 1997, 94, 1761–1766. [Google Scholar] [CrossRef]
- Shapiro, R.; Vallee, B.L. Human placental ribonuclease inhibitor abolishes both angiogenic and ribonucleolytic activities of angiogenin. Proc. Natl. Acad. Sci. USA 1987, 84, 2238–2241. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Ibaragi, S.; Hu, G.F. Angiogenin as a molecular target for the treatment of prostate cancer. Curr. Cancer Ther. Rev. 2011, 7, 83–90. [Google Scholar] [CrossRef]
- Jang, S.H.; Kang, D.K.; Chang, S.I.; Scheraga, H.A.; Shin, H.C. High level production of bovine angiogenin in E. coli by an efficient refolding procedure. Biotechnol. Lett. 2004, 26, 1501–1504. [Google Scholar] [CrossRef] [PubMed]
- Ford, J.H.; Bergy, M.E.; Brooks, A.A.; Garrett, E.R.; Alberti, J.; Dyer, J.R.; Carter, H.E. Further characterization of neomycin B and neomycin C. J. Am. Chem. Soc. 1955, 77, 5311–5314. [Google Scholar] [CrossRef]
- Warboys, C.; Berson, R.; Mann, G.; Pearson, J.; Weinberg, P. Acute and chronic exposure to shear stress have opposite effects on endothelial permeability to macromolecules. Am. J. Physiol.-Heart Circ. Physiol. 2010, 298, H1850–H1856. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trouillon, R.; Kang, D.-K.; Chang, S.-I.; O’Hare, D. Neomycin, but Not Neamine, Blocks Angiogenic Factor Induced Nitric Oxide Release through Inhibition of Akt Phosphorylation. Int. J. Mol. Sci. 2022, 23, 15277. https://doi.org/10.3390/ijms232315277
Trouillon R, Kang D-K, Chang S-I, O’Hare D. Neomycin, but Not Neamine, Blocks Angiogenic Factor Induced Nitric Oxide Release through Inhibition of Akt Phosphorylation. International Journal of Molecular Sciences. 2022; 23(23):15277. https://doi.org/10.3390/ijms232315277
Chicago/Turabian StyleTrouillon, Raphaël, Dong-Ku Kang, Soo-Ik Chang, and Danny O’Hare. 2022. "Neomycin, but Not Neamine, Blocks Angiogenic Factor Induced Nitric Oxide Release through Inhibition of Akt Phosphorylation" International Journal of Molecular Sciences 23, no. 23: 15277. https://doi.org/10.3390/ijms232315277
APA StyleTrouillon, R., Kang, D.-K., Chang, S.-I., & O’Hare, D. (2022). Neomycin, but Not Neamine, Blocks Angiogenic Factor Induced Nitric Oxide Release through Inhibition of Akt Phosphorylation. International Journal of Molecular Sciences, 23(23), 15277. https://doi.org/10.3390/ijms232315277






