Pulmonary Fibrosis as a Result of Acute Lung Inflammation: Molecular Mechanisms, Relevant In Vivo Models, Prognostic and Therapeutic Approaches
Abstract
1. Introduction
2. Acute Lung Inflammation as a Precursor of Pulmonary Fibrosis: Etiology, Pathogenesis, Morphological Characteristics, Outcomes
2.1. Acute Lung Injury (ALI) as One of the Etiological Factors of Pulmonary Fibrosis
2.2. Pathogenesis of ALI
2.3. Pathomorphological Changes in the Lungs during ALI Development
2.4. Outcomes of Acute Lung Inflammation
3. Pathomorphological Changes in the Lungs during Fibrosis Development
3.1. Pathogenesis of Pulmonary Fibrosis Development
3.2. Pathomorphological Changes in the Lungs during Fibrosis Development
3.2.1. Histological Classification
3.2.2. Multidisciplinary Classification of American Thoracic Society and European Respiratory Society
4. Molecular Mechanisms of Pulmonary Fibrosis Development
4.1. Main Effector Cells of Pulmonary Fibrosis: Fibroblasts, Myofibroblasts and Fibrocytes
4.1.1. Fibrocytes Characteristics and Their Role in Pulmonary Fibrosis Development
4.1.2. Differentiation of Fibroblasts to Myofibroblasts and Their Role in Pulmonary Fibrosis Development
4.1.3. Lipofibroblasts in Pulmonary Fibrosis Development
4.2. Possible Role of EMT in Pulmonary Fibrosis Development
4.3. Signaling Pathways in Pulmonary Fibrosis Development
4.3.1. TGF-β Signaling Pathway
4.3.2. Wnt/β-Catenin Signaling Pathway
- Wnt/β-catenin signaling induces the anti-apoptotic and pro-fibrotic phenotype in lung fibroblasts, leading to fibroblast proliferation and their differentiation into myofibroblasts, exacerbating lung tissue fibrosis [181];
- Wnt/β-catenin activation of AEC II increases IL-1β production, stimulating inflammatory and pro-fibrotic responses [182];
- Non-canonical activation of Wnt also stimulates fibroblast proliferation and increases the synthesis of ECM components [178].
4.3.3. VEGF Signaling Pathway
4.3.4. Hedgehog Signaling Pathway
4.3.5. Notch Signaling Pathway
4.3.6. Fibroblast Growth Factor Signaling Pathway
4.4. Role of Cytokines in Pulmonary Fibrosis Development
4.5. Role of Immune Cells in Pulmonary Fibrosis Development
4.5.1. T-Lymphocytes
4.5.2. Macrophages
4.5.3. Autoimmunity
4.6. Role of Reactive Oxygen Species in the Development of Pulmonary Fibrosis
5. Relevant Murine Models of Acute Lung Injury and Pulmonary Fibrosis
5.1. Animal Models of Acute Lung Injury
5.1.1. LPS-Induced Acute Lung Injury
5.1.2. Hyperoxia-Induced Acute Lung Injury
5.1.3. Oleic Acid-Induced Acute Lung Injury
5.1.4. Acid Aspiration-Induced Acute Lung Injury
5.1.5. Mechanical Ventilation-Induced Acute Lung Injury
5.2. Animal Models of Pulmonary Fibrosis
5.2.1. Bleomycin-Induced Pulmonary Fibrosis
5.2.2. Radiation-Induced Pulmonary Fibrosis
5.2.3. Silica Particle-Induced Pulmonary Fibrosis
5.2.4. FITC-Induced Pulmonary Fibrosis
6. Prognostic Markers of Acute Lung Injury and Pulmonary Fibrosis
6.1. Biomarkers of Acute Lung Injury
6.1.1. Exudative Phase
6.1.2. Proliferative Phase
6.2. Biomarkers of Pulmonary Fibrosis
7. Therapeutic Approaches to Suppress Transition from Acute Lung Inflammation to Fibrosis
7.1. Approved Fibrosis Therapeutics
7.2. Anti-Fibrotic Therapeutic in Phase II and III Clinical Trials
7.3. Gene Therapy in Pulmonary Fibrosis Treatment
7.3.1. Enhancement of Gene Expression
7.3.2. Suppression of Gene Expression
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, C.; Zhao, H.; Li, B.L.; Gao, F.; Liu, H.; Cai, J.M.; Zheng, M. CpG-oligodeoxynucleotides may be effective for preventing ionizing radiation induced pulmonary fibrosis. Toxicol. Lett. 2018, 292, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Lin, Z.; Zhou, Q.; Chang, M.; Wang, Y.; Guan, Y.; Li, H.; Zhao, Y.; Liu, N.; Jin, Y.; et al. Metformin alleviates crystalline silica-induced pulmonary fibrosis by remodeling endothelial cells to mesenchymal transition via autophagy signaling. Ecotoxicol. Environ. Saf. 2022, 245, 114100. [Google Scholar] [CrossRef]
- Noble, P.W.; Barkauskas, C.E.; Jiang, D. Pulmonary fibrosis: Patterns and perpetrators. J. Clin. Investig. 2012, 122, 2756–2762. [Google Scholar] [CrossRef]
- Piotrowski, W.J.; Martusewicz-Boros, M.M.; Białas, A.J.; Barczyk, A.; Batko, B.; Błasińska, K.; Boros, P.W.; Górska, K.; Grzanka, P.; Jassem, E.; et al. Guidelines of the Polish Respiratory Society on the Diagnosis and Treatment of Progressive Fibrosing Interstitial Lung Diseases Other than Idiopathic Pulmonary Fibrosis. Adv. Respir. Med. 2022, 90, 425–450. [Google Scholar] [CrossRef]
- De Matteis, S.; Murgia, N. Work-related interstitial lung disease: What is the true burden? Int. J. Tuberc. Lung Dis. 2022, 26, 1001–1005. [Google Scholar] [CrossRef] [PubMed]
- Andreikos, D.; Karampitsakos, T.; Tzouvelekis, A.; Stratakos, G. Statins’ still controversial role in pulmonary fibrosis: What does the evidence show? Pulm. Pharmacol. Ther. 2022, 77, 102168. [Google Scholar] [CrossRef] [PubMed]
- Maher, T.M.; Bendstrup, E.; Dron, L.; Langley, J.; Smith, G.; Khalid, J.M.; Patel, H.; Kreuter, M. Global incidence and prevalence of idiopathic pulmonary fibrosis. Respir. Res. 2021, 22, 197. [Google Scholar] [CrossRef] [PubMed]
- Duchemann, B.; Annesi-Maesano, I.; De Naurois, C.J.; Sanyal, S.; Brillet, P.Y.; Brauner, M.; Kambouchner, M.; Huynh, S.; Naccache, J.M.; Borie, R.; et al. Prevalence and incidence of interstitial lung diseases in a multi-ethnic county of Greater Paris. Eur. Respir. J. 2017, 50, 1602419. [Google Scholar] [CrossRef] [PubMed]
- Tzouvelekis, A.; Bouros, D. Estrogen Signaling and MicroRNAs in Lung Fibrosis. Sex, Hormones, and Rock Scars. Am. J. Respir. Crit. Care Med. 2019, 200, 1199–1200. [Google Scholar] [CrossRef]
- Dwyer, D.N.O.; Ashley, S.L.; Gurczynski, S.J.; Xia, M.; Wilke, C.; Falkowski, N.R.; Norman, K.C.; Arnold, K.B.; Huffnagle, G.B.; Salisbury, M.L.; et al. Lung Microbiota Contribute to Pulmonary Inflammation and Disease Progression in Pulmonary Fibrosis. Am. J. Respir. Crit. Care Med. 2019, 199, 1127–1138. [Google Scholar] [CrossRef]
- Pardo, A.; Selman, M. The interplay of the genetic architecture, aging, and environmental factors in the pathogenesis of idiopathic pulmonary fibrosis. Am. J. Respir. Cell Mol. Biol. 2021, 64, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Ley, B.; Collard, H.R.; King, T.E. Clinical course and prediction of survival in idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 2011, 183, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Uegami, W.; Bychkov, A.; Ozasa, M.; Uehara, K.; Kataoka, K.; Johkoh, T.; Kondoh, Y.; Sakanashi, H.; Fukuoka, J. MIXTURE of human expertise and deep learning-Developing an explainable model for predicting pathological diagnosis and survival in patients with interstitial lung disease. Mod. Pathol. 2022, 35, 1083–1091. [Google Scholar] [CrossRef]
- Ryerson, C.J.; Urbania, T.H.; Richeldi, L.; Mooney, J.J.; Lee, J.S.; Jones, K.D.; Elicker, B.M.; Koth, L.L.; King, T.E.; Wolters, P.J.; et al. Prevalence and prognosis of unclassifiable interstitial lung disease. Eur. Respir. J. 2013, 42, 750–757. [Google Scholar] [CrossRef] [PubMed]
- Denham, J.W.; Hauer-Jensen, M. The radiotherapeutic injury—A complex “wound”. Radiother. Oncol. 2002, 63, 129–145. [Google Scholar] [CrossRef] [PubMed]
- Fubini, B.; Hubbard, A. Reactive oxygen species (ROS) and reactive nitrogen species (RNS) generation by silica in inflammation and fibrosis. Free Radic. Biol. Med. 2003, 34, 1507–1516. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Stubbe, J.A. Bleomycins: Towards better therapeutics. Nat. Rev. Cancer 2005, 5, 102–112. [Google Scholar] [CrossRef]
- Kligerman, S. Pathogenesis, Imaging, and Evolution of Acute Lung Injury. Radiol. Clin. N. Am. 2022, 60, 925–939. [Google Scholar] [CrossRef]
- Saito, S.; Deskin, B.; Rehan, M.; Yadav, S.; Matsunaga, Y.; Lasky, J.A.; Thannickal, V.J. Novel mediators of idiopathic pulmonary fibrosis. Clin. Sci. 2022, 136, 1229–1240. [Google Scholar] [CrossRef]
- Kelly, B.G.; Lok, S.S.; Hasleton, P.S.; Egan, J.J.; Stewart, J.P. A rearranged form of Epstein-Barr virus DNA is associated with idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 2002, 166, 510–513. [Google Scholar] [CrossRef]
- Reddy, C.A.; Wakwaya, Y.T. Impact of gastroesophageal reflux disease on idiopathic pulmonary fibrosis and lung transplant recipients. Curr. Opin. Gastroenterol. 2022, 38, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.; Zhong, B.; Fan, Z.; Zhang, H.; Xu, M.; Zhang, X.; Sanders, Y.Y. DNA methylation in pulmonary fibrosis and lung cancer. Expert Rev. Respir. Med. 2022, 16, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, A.; Flores-Gonzalez, J.; Buendia-Roldan, I.; Chavez-Galan, L. Telomere Shortening and Its Association with Cell Dysfunction in Lung Diseases. Int. J. Mol. Sci. 2022, 23, 425. [Google Scholar] [CrossRef] [PubMed]
- Ruaro, B.; Pozzan, R.; Confalonieri, P.; Tavano, S.; Hughes, M.; Matucci Cerinic, M.; Baratella, E.; Zanatta, E.; Lerda, S.; Geri, P.; et al. Gastroesophageal Reflux Disease in Idiopathic Pulmonary Fibrosis: Viewer or Actor? To Treat or Not to Treat? Pharmaceuticals 2022, 15, 1033. [Google Scholar] [CrossRef]
- Bédard Méthot, D.; Leblanc, É.; Lacasse, Y. Meta-analysis of Gastroesophageal Reflux Disease and Idiopathic Pulmonary Fibrosis. Chest 2019, 155, 33–43. [Google Scholar] [CrossRef]
- Crisan-Dabija, R.; Pavel, C.A.; Popa, I.V.; Tarus, A.; Burlacu, A. “A ChainOnly as Strong as Its Weakest Link”: An Up-to-Date Literature Review on the Bidirectional Interaction ofPulmonary Fibrosis and COVID-19. J. Proteome Res. 2020, 19, 4327. [Google Scholar] [CrossRef] [PubMed]
- Kessler, A.; Heightman, M.; Brennan, E. Post-COVID-19 respiratory problems: Burden and management. Curr. Opin. Support. Palliat. Care 2022, 16, 203–209. [Google Scholar] [CrossRef]
- King, T.E.; Pardo, A.; Selman, M. Idiopathic pulmonary fibrosis. Lancet 2011, 378, 1949–1961. [Google Scholar] [CrossRef] [PubMed]
- Raghu, G.; Selman, M. Nintedanib and pirfenidone: New antifibrotic treatments indicated for idiopathic pulmonary fibrosis offer hopes and raises questions. Am. J. Respir. Crit. Care Med. 2015, 191, 252–254. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Jia, A. Protective effect of carvacrol on acute lung injury induced by lipopolysaccharide in mice. Inflammation 2014, 37, 1091–1101. [Google Scholar] [CrossRef]
- Lee, J.W.; Chun, W.; Kwon, O.K.; Park, H.A.; Lim, Y.; Lee, J.H.; Kim, D.Y.; Kim, J.H.; Lee, H.K.; Ryu, H.W.; et al. 3,4,5-Trihydroxycinnamic acid attenuates lipopolysaccharide (LPS)-induced acute lung injury via downregulating inflammatory molecules and upregulating HO-1/AMPK activation. Int. Immunopharmacol. 2018, 64, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Chen, S.; Zeng, M.; Lin, D.; Wang, Y.; Wen, X.; Xu, C.; Yang, L.; Fan, X.; Gong, Y.; et al. Apelin-13 Administration Protects Against LPS-Induced Acute Lung Injury by Inhibiting NF-κB Pathway and NLRP3 Inflammasome Activation. Cell. Physiol. Biochem. 2018, 49, 1918–1932. [Google Scholar] [CrossRef]
- Yang, J.; Li, S.; Wang, L.; Du, F.; Zhou, X.; Song, Q.; Zhao, J.; Fang, R. Ginsenoside Rg3 attenuates lipopolysaccharide-induced acute lung injury via MerTK-dependent activation of the PI3K/AKT/mTOR pathway. Front. Pharmacol. 2018, 9, 850. [Google Scholar] [CrossRef] [PubMed]
- Bellani, G.; Laffey, J.G.; Pham, T.; Fan, E.; Brochard, L.; Esteban, A.; Gattinoni, L.; Van Haren, F.M.P.; Larsson, A.; McAuley, D.F.; et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA J. Am. Med. Assoc. 2016, 315, 788–800. [Google Scholar] [CrossRef] [PubMed]
- Fan, E.; Brodie, D.; Slutsky, A.S. Acute respiratory distress syndrome advances in diagnosis and treatment. JAMA J. Am. Med. Assoc. 2018, 319, 698–710. [Google Scholar] [CrossRef] [PubMed]
- Jamal, M.; Bangash, H.I.; Habiba, M.; Lei, Y.; Xie, T.; Sun, J.; Wei, Z.; Hong, Z.; Shao, L.; Zhang, Q. Immune dysregulation and system pathology in COVID-19. Virulence 2021, 12, 918–936. [Google Scholar] [CrossRef]
- Ramasamy, S.; Subbian, S. Critical determinants of cytokine storm and type i interferon response in COVID-19 pathogenesis. Clin. Microbiol. Rev. 2021, 34, e00299-20. [Google Scholar] [CrossRef]
- Ksiazek, T.G.; Erdman, D.; Goldsmith, C.S.; Zaki, S.R.; Peret, T.; Emery, S.; Tong, S.; Urbani, C.; Comer, J.A.; Lim, W.; et al. A Novel Coronavirus Associated with Severe Acute Respiratory Syndrome. N. Engl. J. Med. 2003, 348, 1953–1966. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, J.; Chakraborty, T.; Romero, M.; Mraheil, M.A.; Kutlar, A.; Pace, B.; Lucas, R. Streptococcus pneumoniae and Its Virulence Factors H2O2 and Pneumolysin Are Potent Mediators of the Acute Chest Syndrome in Sickle Cell Disease. Toxins 2021, 13, 157. [Google Scholar] [CrossRef]
- Lucas, R.; Hadizamani, Y.; Gonzales, J.; Gorshkov, B.; Bodmer, T.; Berthiaume, Y.; Moehrlen, U.; Lode, H.; Huwer, H.; Hudel, M.; et al. Impact of bacterial toxins in the lungs. Toxins 2020, 12, 223. [Google Scholar] [CrossRef]
- Klomp, M.; Ghosh, S.; Mohammed, S.; Nadeem Khan, M. From virus to inflammation, how influenza promotes lung damage. J. Leukoc. Biol. 2021, 110, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.D.; Wunderink, R.G. Viral Pneumonia and Acute Respiratory Distress Syndrome. Clin. Chest Med. 2017, 38, 113–125. [Google Scholar] [CrossRef]
- Zhang, H.; Dong, W.; Li, S.; Zhang, Y.; Lv, Z.; Yang, L.; Jiang, L.; Wu, T.; Wang, Y. Salidroside protects against ventilation-induced lung injury by inhibiting the expression of matrix metalloproteinase-9. Pharm. Biol. 2021, 59, 760–768. [Google Scholar] [CrossRef] [PubMed]
- Goligher, E.C.; Douflé, G.; Fan, E. Update in mechanical ventilation, sedation, and outcomes 2014. Am. J. Respir. Crit. Care Med. 2015, 191, 1367–1373. [Google Scholar] [CrossRef]
- Agrawal, D.K.; Smith, B.J.; Sottile, P.D.; Albers, D.J. A Damaged-Informed Lung Ventilator Model for Ventilator Waveforms. Front. Physiol. 2021, 12, 724046. [Google Scholar] [CrossRef] [PubMed]
- Radbel, J.; Laskin, D.L.; Laskin, J.D.; Kipen, H.M. Disease-modifying treatment of chemical threat agent–induced acute lung injury. Ann. N. Y. Acad. Sci. 2020, 1480, 14–29. [Google Scholar] [CrossRef]
- Pauluhn, J. Phosgene inhalation toxicity: Update on mechanisms and mechanism-based treatment strategies. Toxicology 2021, 450, 152682. [Google Scholar] [CrossRef]
- Laskin, D.L.; Malaviya, R.; Laskin, J.D. Role of Macrophages in Acute Lung Injury and Chronic Fibrosis Induced by Pulmonary Toxicants. Toxicol. Sci. 2019, 168, 287–301. [Google Scholar] [CrossRef]
- Triantafyllou, G.A.; Tiberio, P.J.; Zou, R.H.; Lamberty, P.E.; Lynch, M.J.; Kreit, J.W.; Gladwin, M.T.; Morris, A.; Chiarchiaro, J. Vaping-associated acute lung injury: A case series. Am. J. Respir. Crit. Care Med. 2019, 200, 1430–1431. [Google Scholar] [CrossRef]
- Kasson, E.; Singh, A.K.; Huang, M.; Wu, D.; Cavazos-Rehg, P. Using a mixed methods approach to identify public perception of vaping risks and overall health outcomes on Twitter during the 2019 EVALI outbreak. Int. J. Med. Inform. 2021, 155, 104574. [Google Scholar] [CrossRef]
- Zhang, C.N.; Li, F.J.; Zhao, Z.L.; Zhang, J.N. The role of extracellular vesicles in traumatic brain injury-induced acute lung injury. Am. J. Physiol. Lung Cell. Mol. Physiol. 2021, 321, L885–L891. [Google Scholar] [CrossRef] [PubMed]
- Ziaka, M.; Exadaktylos, A. Brain–lung interactions and mechanical ventilation in patients with isolated brain injury. Crit. Care 2021, 25, 358. [Google Scholar] [CrossRef] [PubMed]
- Sever, I.H.; Ozkul, B.; Erisik Tanriover, D.; Ozkul, O.; Elgormus, C.S.; Gur, S.G.; Sogut, I.; Uyanikgil, Y.; Cetin, E.O.; Erbas, O. Protective effect of oxytocin through its anti-inflammatory and antioxidant role in a model of sepsis-induced acute lung injury: Demonstrated by CT and histological findings. Exp. Lung Res. 2021, 47, 426–435. [Google Scholar] [CrossRef]
- Jiao, Y.; Zhang, T.; Zhang, C.; Ji, H.; Tong, X.; Xia, R.; Wang, W.; Ma, Z.; Shi, X. Exosomal miR-30d-5p of neutrophils induces M1 macrophage polarization and primes macrophage pyroptosis in sepsis-related acute lung injury. Crit. Care 2021, 25, 356. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.; Deng, J.; Zhou, X.; Cai, B.; Zhang, B.; Chen, X.; Chen, Z.; Wang, W. Sitagliptin activates the p62–Keap1–Nrf2 signalling pathway to alleviate oxidative stress and excessive autophagy in severe acute pancreatitis-related acute lung injury. Cell Death Dis. 2021, 12, 928. [Google Scholar] [CrossRef] [PubMed]
- He, Y.Q.; Zhou, C.C.; Yu, L.Y.; Wang, L.; Deng, J.L.; Tao, Y.L.; Zhang, F.; Chen, W.S. Natural product derived phytochemicals in managing acute lung injury by multiple mechanisms. Pharmacol. Res. 2021, 163, 105224. [Google Scholar] [CrossRef]
- Blázquez-Prieto, J.; López-Alonso, I.; Huidobro, C.; Albaiceta, G.M. The emerging role of neutrophils in repair after acute lung injury. Am. J. Respir. Cell Mol. Biol. 2018, 59, 289–294. [Google Scholar] [CrossRef]
- Chen, T.; Zhu, G.; Meng, X.; Zhang, X. Recent developments of small molecules with anti-inflammatory activities for the treatment of acute lung injury. Eur. J. Med. Chem. 2020, 207, 112660. [Google Scholar] [CrossRef]
- Hussell, T.; Bell, T.J. Alveolar macrophages: Plasticity in a tissue-specific context. Nat. Rev. Immunol. 2014, 14, 81–93. [Google Scholar] [CrossRef]
- Gwyer Findlay, E.; Hussell, T. Macrophage-mediated inflammation and disease: A focus on the lung. Mediat. Inflamm. 2012, 2012, 140937. [Google Scholar] [CrossRef]
- Byrne, A.J.; Mathie, S.A.; Gregory, L.G.; Lloyd, C.M. Pulmonary macrophages: Key players in the innate defence of the airways. Thorax 2015, 70, 1189–1196. [Google Scholar] [CrossRef] [PubMed]
- Garbi, N.; Lambrecht, B.N. Location, function, and ontogeny of pulmonary macrophages during the steady state. Pflug. Arch. Eur. J. Physiol. 2017, 469, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Meyer, N.J.; Gattinoni, L.; Calfee, C.S. Acute respiratory distress syndrome. Lancet 2021, 398, 622–637. [Google Scholar] [CrossRef] [PubMed]
- Mulugeta, S.; Nureki, S.I.; Beers, M.F. Lost after translation: Insights from pulmonary surfactant for understanding the role of alveolar epithelial dysfunction and cellular quality control in fibrotic lung disease. Am. J. Physiol. Lung Cell. Mol. Physiol. 2015, 309, L507–L525. [Google Scholar] [CrossRef]
- Puntorieri, V.; Hiansen, J.Q.; McCaig, L.A.; Yao, L.J.; Veldhuizen, R.A.W.; Lewis, J.F. The effects of exogenous surfactant administration on ventilation-induced inflammation in mouse models of lung injury. BMC Pulm. Med. 2013, 13, 67. [Google Scholar] [CrossRef]
- Zebialowicz Ahlström, J.; Massaro, F.; Mikolka, P.; Feinstein, R.; Perchiazzi, G.; Basabe-Burgos, O.; Curstedt, T.; Larsson, A.; Johansson, J.; Rising, A. Synthetic surfactant with a recombinant surfactant protein C analogue improves lung function and attenuates inflammation in a model of acute respiratory distress syndrome in adult rabbits. Respir. Res. 2019, 20, 245. [Google Scholar] [CrossRef]
- Zemans, R.L.; Matthay, M.A. Bench-to-bedside review: The role of the alveolar epithelium in the resolution of pulmonary edema in acute lung injury. Crit. Care 2004, 8, 469–477. [Google Scholar] [CrossRef]
- Zarrilli, G.; Angerilli, V.; Businello, G.; Sbaraglia, M.; Traverso, G.; Fortarezza, F.; Rizzo, S.; De Gaspari, M.; Basso, C.; Calabrese, F.; et al. The immunopathological and histological landscape of COVID-19-mediated lung injury. Int. J. Mol. Sci. 2021, 22, 974. [Google Scholar] [CrossRef]
- Gouda, M.M.; Shaikh, S.B.; Bhandary, Y.P. Inflammatory and Fibrinolytic System in Acute Respiratory Distress Syndrome. Lung 2018, 196, 609–616. [Google Scholar] [CrossRef]
- Borczuk, A.C. Pulmonary pathology of COVID-19: A review of autopsy studies. Curr. Opin. Pulm. Med. 2021, 27, 184–192. [Google Scholar] [CrossRef]
- Mohanty, S.K.; Satapathy, A.; Naidu, M.M.; Mukhopadhyay, S.; Sharma, S.; Barton, L.M.; Stroberg, E.; Duval, E.J.; Pradhan, D.; Tzankov, A.; et al. Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) and coronavirus disease 19 (COVID-19)-A natomic pathology perspective on current knowledge. Diagn. Pathol. 2020, 15, s13000–s13020. [Google Scholar] [CrossRef] [PubMed]
- Distler, J.H.W.; Györfi, A.H.; Ramanujam, M.; Whitfield, M.L.; Königshoff, M.; Lafyatis, R. Shared and distinct mechanisms of fibrosis. Nat. Rev. Rheumatol. 2019, 15, 705–730. [Google Scholar] [CrossRef] [PubMed]
- Morgenstern, D.E.; Gifford, M.A.C.; Li, L.L.; Doerschuk, C.M.; Dinauer, M.C. Absence of respiratory burst in X-linked chronic granulomatous disease mice leads to abnormalities in both host defense and inflammatory response to Aspergillus fumigatus. J. Exp. Med. 1997, 185, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Leung, C.C.; Yu, I.T.S.; Chen, W. Silicosis. Lancet 2012, 379, 2008–2018. [Google Scholar] [CrossRef] [PubMed]
- Hannah, J.R.; D’Cruz, D.P. Pulmonary Complications of Systemic Lupus Erythematosus. Semin. Respir. Crit. Care Med. 2019, 40, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Ahmadinejad, F.; Møller, S.G.; Hashemzadeh-Chaleshtori, M.; Bidkhori, G.; Jami, M.S. Molecular mechanisms behind free radical scavengers function against oxidative stress. Antioxidants 2017, 6, 51. [Google Scholar] [CrossRef]
- Oczypok, E.A.; Perkins, T.N.; Oury, T.D. All the “RAGE” in lung disease: The receptor for advanced glycation endproducts (RAGE) is a major mediator of pulmonary inflammatory responses. Paediatr. Respir. Rev. 2017, 23, 40–49. [Google Scholar] [CrossRef]
- Yousuf, A.; Ibrahim, W.; Greening, N.J.; Brightling, C.E. T2 Biologics for Chronic Obstructive Pulmonary Disease. J. Allergy Clin. Immunol. Pract. 2019, 7, 1405–1416. [Google Scholar] [CrossRef]
- Wynn, T.A. Common and unique mechanisms regulate fibrosis in various fibroproliferative diseases. J. Clin. Investig. 2007, 117, 524–529. [Google Scholar] [CrossRef]
- Bochaton-Piallat, M.L.; Gabbiani, G.; Hinz, B. The myofibroblast in wound healing and fibrosis: Answered and unanswered questions. F1000Research 2016, 5, F1000. [Google Scholar] [CrossRef]
- Upagupta, C.; Shimbori, C.; Alsilmi, R.; Kolb, M. Matrix abnormalities in pulmonary fibrosis. Eur. Respir. Rev. 2018, 27, 180033. [Google Scholar] [CrossRef] [PubMed]
- Gaggar, A.; Weathington, N. Bioactive extracellular matrix fragments in lung health and disease. J. Clin. Investig. 2016, 126, 3176–3184. [Google Scholar] [CrossRef]
- Usman, K.; Hsieh, A.; Hackett, T.L. The role of mirnas in extracellular matrix repair and chronic fibrotic lung diseases. Cells 2021, 10, 1706. [Google Scholar] [CrossRef] [PubMed]
- Thunnissen, E.; Motoi, N.; Minami, Y.; Matsubara, D.; Timens, W.; Nakatani, Y.; Ishikawa, Y.; Baez-Navarro, X.; Radonic, T.; Blaauwgeers, H.; et al. Elastin in pulmonary pathology: Relevance in tumours with a lepidic or papillary appearance. A comprehensive understanding from a morphological viewpoint. Histopathology 2022, 80, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Olczyk, P.; Mencner, Ł.; Komosinska-Vassev, K. The role of the extracellular matrix components in cutaneous wound healing. Biomed. Res. Int. 2014, 2014, 747584. [Google Scholar] [CrossRef] [PubMed]
- Herrera, J.; Henke, C.A.; Bitterman, P.B. Extracellular matrix as a driver of progressive fibrosis. J. Clin. Investig. 2018, 128, 45–53. [Google Scholar] [CrossRef]
- Parker, M.W.; Rossi, D.; Peterson, M.; Smith, K.; Sikstrom, K.; White, E.S.; Connett, J.E.; Henke, C.A.; Larsson, O.; Bitterman, P.B. Fibrotic extracellular matrix activates a profibrotic positive feedback loop. J. Clin. Investig. 2014, 124, 1622–1635. [Google Scholar] [CrossRef]
- Liu, F.; Lagares, D.; Choi, K.M.; Stopfer, L.; Marinković, A.; Vrbanac, V.; Probst, C.K.; Hiemer, S.E.; Sisson, T.H.; Horowitz, J.C.; et al. Mechanosignaling through YAP and TAZ drives fibroblast activation and fibrosis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2015, 308, L344–L357. [Google Scholar] [CrossRef]
- Burgstaller, G.; Oehrle, B.; Gerckens, M.; White, E.S.; Schiller, H.B.; Eickelberg, O. The instructive extracellular matrix of the lung: Basic composition and alterations in chronic lung disease. Eur. Respir. J. 2017, 50, 1601805. [Google Scholar] [CrossRef]
- Annoni, R.; Lanças, T.; Tanigawa, R.Y.; Matsushita, M.D.M.; Fernezlian, S.D.M.; Bruno, A.; Da Silva, L.F.F.; Roughley, P.J.; Battaglia, S.; Dolhnikoff, M.; et al. Extracellular matrix composition in COPD. Eur. Respir. J. 2012, 40, 1362–1373. [Google Scholar] [CrossRef] [PubMed]
- Ito, J.T.; Lourenço, J.D.; Righetti, R.F.; Tibério, I.F.L.C.; Prado, C.M.; Lopes, F.D.T.Q.S. Extracellular Matrix Component Remodeling in Respiratory Diseases: What Has Been Found in Clinical and Experimental Studies? Cells 2019, 8, 342. [Google Scholar] [CrossRef] [PubMed]
- Pardo, A.; Selman, M. Lung fibroblasts, aging, and idiopathic pulmonary fibrosis. Ann. Am. Thorac. Soc. 2016, 13, S417–S421. [Google Scholar] [CrossRef] [PubMed]
- Prasse, A.; Pechkovsky, D.V.; Toews, G.B.; Jungraithmayr, W.; Kollert, F.; Goldmann, T.; Vollmer, E.; Müller-Quernheim, J.; Zissel, G. A vicious circle of alveolar macrophages and fibroblasts perpetuates pulmonary fibrosis via CCL18. Am. J. Respir. Crit. Care Med. 2006, 173, 781–792. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.M.; Lederer, D.J.; Borczuk, A.C.; Kawut, S.M. Pulmonary hypertension in idiopathic pulmonary fibrosis. Chest 2007, 132, 998–1006. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Hong, W.; Gao, M.; Yi, E.; Zhang, J.; Hao, B.; Liang, C.; Li, X.; Li, C.; Ye, X.; et al. Long noncoding RNA COPDA1 promotes airway smooth muscle cell proliferation in chronic obstructive pulmonary disease. Am. J. Respir. Cell Mol. Biol. 2019, 61, 584–596. [Google Scholar] [CrossRef] [PubMed]
- de Almeida, L.G.N.; Thode, H.; Eslambolchi, Y.; Chopra, S.; Young, D.; Gill, S.; Devel, L.; Dufour, A. Matrix Metalloproteinases: From Molecular Mechanisms to Physiology, Pathophysiology, and Pharmacology. Pharmacol. Rev. 2022, 74, 712–768. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.E.; Parks, W.C. Metalloproteinases and their inhibitors: Regulators of wound healing. Int. J. Biochem. Cell Biol. 2008, 40, 1334–1347. [Google Scholar] [CrossRef]
- Parks, W.C.; Wilson, C.L.; López-Boado, Y.S. Matrix metalloproteinases as modulators of inflammation and innate immunity. Nat. Rev. Immunol. 2004, 4, 617–629. [Google Scholar] [CrossRef]
- Wynn, T.A. Integrating mechanisms of pulmonary fibrosis. J. Exp. Med. 2011, 208, 1339–1350. [Google Scholar] [CrossRef]
- Majo, J.; Klinkhammer, B.M.; Boor, P.; Tiniakos, D. Pathology and natural history of organ fibrosis. Curr. Opin. Pharmacol. 2019, 49, 82–89. [Google Scholar] [CrossRef]
- Travis, W.D.; King, T.E.; Bateman, E.D.; Lynch, D.A.; Capron, F.; Center, D.; Colby, T.V.; Cordier, J.F.; DuBois, R.M.; Galvin, J.; et al. American thoracic society/European respiratory society international multidisciplinary consensus classification of the idiopathic interstitial pneumonias. Am. J. Respir. Crit. Care Med. 2002, 165, 277–304. [Google Scholar] [CrossRef]
- Smith, M.L. Update on Pulmonary Fibrosis: Not All Fibrosis Is Created Equally. Arch. Pathol. Lab. Med. 2016, 140, 221–229. [Google Scholar] [CrossRef]
- Travis, W.D.; Costabel, U.; Hansell, D.M.; King, T.E.; Lynch, D.A.; Nicholson, A.G.; Ryerson, C.J.; Ryu, J.H.; Selman, M.; Wells, A.U.; et al. An official American Thoracic Society/European Respiratory Society statement: Update of the international multidisciplinary classification of the idiopathic interstitial pneumonias. Am. J. Respir. Crit. Care Med. 2013, 188, 733–748. [Google Scholar] [CrossRef]
- Heukels, P.; Moor, C.C.; von der Thüsen, J.H.; Wijsenbeek, M.S.; Kool, M. Inflammation and immunity in IPF pathogenesis and treatment. Respir. Med. 2019, 147, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, N.; Jin, H.; Liu, T.; Chensue, S.W.; Phan, S.H. Bone marrow-derived progenitor cells in pulmonary fibrosis. J. Clin. Investig. 2004, 113, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Bucala, R.; Spiegel, L.A.; Chesney, J.; Hogan, M.; Cerami, A. Circulating fibrocytes define a new leukocyte subpopulation that mediates tissue repair. Mol. Med. 1994, 1, 71–81. [Google Scholar] [CrossRef]
- Kuwana, M.; Okazaki, Y.; Kodama, H.; Izumi, K.; Yasuoka, H.; Ogawa, Y.; Kawakami, Y.; Ikeda, Y. Human circulating CD14 + monocytes as a source of progenitors that exhibit mesenchymal cell differentiation. J. Leukoc. Biol. 2003, 74, 833–845. [Google Scholar] [CrossRef]
- Postlethwaite, A.E.; Shigemitsu, H.; Kanangat, S. Cellular origins of fibroblasts: Possible implications for organ fibrosis in systemic sclerosis. Curr. Opin. Rheumatol. 2004, 16, 733–738. [Google Scholar] [CrossRef]
- Willis, B.C.; Liebler, J.M.; Luby-Phelps, K.; Nicholson, A.G.; Crandall, E.D.; Du Bois, R.M.; Borok, Z. Induction of epithelial-mesenchymal transition in alveolar epithelial cells by transforming growth factor-β1: Potential role in idiopathic pulmonary fibrosis. Am. J. Pathol. 2005, 166, 1321–1332. [Google Scholar] [CrossRef]
- Foxman, E.F.; Campbell, J.J.; Butcher, E.C. Multistep navigation and the combinatorial control of leukocyte chemotaxis. J. Cell Biol. 1997, 139, 1349–1360. [Google Scholar] [CrossRef]
- Pilling, D.; Fan, T.; Huang, D.; Kaul, B.; Gomer, R.H. Identification of markers that distinguish monocyte-derived fibrocytes from monocytes, macrophages, and fibroblasts. PLoS ONE 2009, 4, e7475. [Google Scholar] [CrossRef] [PubMed]
- Phillips, R.J.; Burdick, M.D.; Hong, K.; Lutz, M.A.; Murray, L.A.; Xue, Y.Y.; Belperio, J.A.; Keane, M.P.; Strieter, R.M. Circulating fibrocytes traffic to the lungs in response to CXCL12 and mediate fibrosis. J. Clin. Investig. 2004, 114, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Russo, F.P.; Alison, M.R.; Bigger, B.W.; Amofah, E.; Florou, A.; Amin, F.; Bou-Gharios, G.; Jeffery, R.; Iredale, J.P.; Forbes, S.J. The Bone Marrow Functionally Contributes to Liver Fibrosis. Gastroenterology 2006, 130, 1807–1821. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Zhang, Z.; Jia, L.; Wang, Y. Role of bone marrow-derived fibroblasts in renal fibrosis. Front. Physiol. 2016, 7, 61. [Google Scholar] [CrossRef] [PubMed]
- Haudek, S.B.; Xia, Y.; Huebener, P.; Lee, J.M.; Carlson, S.; Crawford, J.R.; Pilling, D.; Gomer, R.H.; Trial, J.A.; Frangogiannis, N.G.; et al. Bone marrow-derived fibroblast precursors mediate ischemic cardiomyopathy in mice. Proc. Natl. Acad. Sci. USA 2006, 103, 18284–18289. [Google Scholar] [CrossRef] [PubMed]
- Varcoe, R.L.; Mikhail, M.; Guiffre, A.K.; Pennings, G.; Vicaretti, M.; Hawthorne, W.J.; Fletcher, J.P.; Medbury, H.J. The role of the fibrocyte in intimal hyperplasia. J. Thromb. Haemost. 2006, 4, 1125–1133. [Google Scholar] [CrossRef]
- Mori, L.; Bellini, A.; Stacey, M.A.; Schmidt, M.; Mattoli, S. Fibrocytes contribute to the myofibroblast population in wounded skin and originate from the bone marrow. Exp. Cell Res. 2005, 304, 81–90. [Google Scholar] [CrossRef]
- Lassance, L.; Marino, G.K.; Medeiros, C.S.; Thangavadivel, S.; Wilson, S.E. Fibrocyte migration, differentiation and apoptosis during the corneal wound healing response to injury. Exp. Eye Res. 2018, 170, 177–187. [Google Scholar] [CrossRef]
- Metz, C.N. Fibrocytes: A unique cell population implicated in wound healing. Cell. Mol. Life Sci. 2003, 60, 1342–1350. [Google Scholar] [CrossRef]
- Kisseleva, T.; Von Köckritz-Blickwede, M.; Reichart, D.; McGillvray, S.M.; Wingender, G.; Kronenberg, M.; Glass, C.K.; Nizet, V.; Brenner, D.A. Fibrocyte-like cells recruited to the spleen support innate and adaptive immune responses to acute injury or infection. J. Mol. Med. 2011, 89, 997–1013. [Google Scholar] [CrossRef]
- Collins, D.; Fry, C.; Moore, B.B.; Nemzek, J.A. Phagocytosis by Fibrocytes as a Mechanism to Decrease Bacterial Burden and Increase Survival in Sepsis. Shock 2019, 51, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Smadja, D.M.; Dorfmüller, P.; Guerin, C.L.; Bieche, I.; Badoual, C.; Boscolo, E.; Kambouchner, M.; Cazes, A.; Mercier, O.; Humbert, M.; et al. Cooperation between human fibrocytes and endothelial colony-forming cells increases angiogenesis via the CXCR4 pathway. Thromb. Haemost. 2014, 112, 1002–1013. [Google Scholar] [CrossRef] [PubMed]
- Chong, S.G.; Sato, S.; Kolb, M.; Gauldie, J. Fibrocytes and fibroblasts—Where are we now. Int. J. Biochem. Cell Biol. 2019, 116, 105595. [Google Scholar] [CrossRef]
- Higashiyama, R.; Moro, T.; Nakao, S.; Mikami, K.; Fukumitsu, H.; Ueda, Y.; Ikeda, K.; Adachi, E.; Bou-Gharios, G.; Okazaki, I.; et al. Negligible Contribution of Bone Marrow-Derived Cells to Collagen Production During Hepatic Fibrogenesis in Mice. Gastroenterology 2009, 137, 1459–1466.e1. [Google Scholar] [CrossRef]
- Kleaveland, K.R.; Moore, B.B.; Kim, K.K. Paracrine functions of fibrocytes to promote lung fibrosis. Expert Rev. Respir. Med. 2014, 8, 163–172. [Google Scholar] [CrossRef][Green Version]
- Hartlapp, I.; Abe, R.; Saeed, R.W.; Peng, T.; Voelter, W.; Bucala, R.; Metz, C.N. Fibrocytes induce an angiogenic phenotype in cultured endothelial cells and promote angiogenesis in vivo. FASEB J. 2001, 15, 2215–2224. [Google Scholar] [CrossRef]
- Hong, K.M.; Burdick, M.D.; Phillips, R.J.; Heber, D.; Strieter, R.M. Characterization of human fibrocytes as circulating adipocyte progenitors and the formation of human adipose tissue in SCID mice. FASEB J. 2005, 19, 2029–2031. [Google Scholar] [CrossRef] [PubMed]
- García-de-Alba, C.; Becerril, C.; Ruiz, V.; González, Y.; Reyes, S.; García-Alvarez, J.; Selman, M.; Pardo, A. Expression of matrix metalloproteases by fibrocytes: Possible role in migration and homing. Am. J. Respir. Crit. Care Med. 2010, 182, 1144–1152. [Google Scholar] [CrossRef] [PubMed]
- Chiang, H.Y.; Chu, P.H.; Lee, T.H. R1R2 peptide ameliorates pulmonary fibrosis in mice through fibrocyte migration and differentiation. PLoS ONE 2017, 12, e0185811. [Google Scholar] [CrossRef]
- Tomasek, J.J.; Gabbiani, G.; Hinz, B.; Chaponnier, C.; Brown, R.A. Myofibroblasts and mechano: Regulation of connective tissue remodelling. Nat. Rev. Mol. Cell Biol. 2002, 3, 349–363. [Google Scholar] [CrossRef]
- Hinz, B. Formation and function of the myofibroblast during tissue repair. J. Investig. Dermatol. 2007, 127, 526–537. [Google Scholar] [CrossRef] [PubMed]
- Hinz, B. Masters and servants of the force: The role of matrix adhesions in myofibroblast force perception and transmission. Eur. J. Cell Biol. 2006, 85, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Hinz, B.; Pittet, P.; Smith-Clerc, J.; Chaponnier, C.; Meister, J.J. Myofibroblast development is characterized by specific cell-cell adherens junctions. Mol. Biol. Cell 2004, 15, 4310–4320. [Google Scholar] [CrossRef]
- Rehan, V.K.; Torday, J.S. The lung alveolar lipofibroblast: An evolutionary strategy against neonatal hyperoxic lung injury. Antioxid. Redox Signal. 2014, 21, 1893–1904. [Google Scholar] [CrossRef] [PubMed]
- Habiel, D.M.; Hogaboam, C.M. Heterogeneity of Fibroblasts and Myofibroblasts in Pulmonary Fibrosis. Curr. Pathobiol. Rep. 2017, 5, 101–110. [Google Scholar] [CrossRef]
- Rehan, V.K.; Torday, J.S. Hyperoxia augments pulmonary lipofibroblast-to-myofibroblast transdifferentiation. Cell Biochem. Biophys. 2003, 38, 239–249. [Google Scholar] [CrossRef]
- Rehan, V.K.; Wang, Y.; Sugano, S.; Romero, S.; Chen, X.; Santos, J.; Khazanchi, A.; Torday, J.S. Mechanism of nicotine-induced pulmonary fibroblast transdifferentiation. Am. J. Physiol. Lung Cell. Mol. Physiol. 2005, 289, 667–676. [Google Scholar] [CrossRef]
- Hecker, L.; Jagirdar, R.; Jin, T.; Thannickal, V.J. Reversible differentiation of myofibroblasts by MyoD. Exp. Cell Res. 2011, 317, 1914–1921. [Google Scholar] [CrossRef]
- Kheirollahi, V.; Wasnick, R.M.; Biasin, V.; Vazquez-Armendariz, A.I.; Chu, X.; Moiseenko, A.; Weiss, A.; Wilhelm, J.; Zhang, J.S.; Kwapiszewska, G.; et al. Metformin induces lipogenic differentiation in myofibroblasts to reverse lung fibrosis. Nat. Commun. 2019, 10, 2987. [Google Scholar] [CrossRef]
- Bartis, D.; Mise, N.; Mahida, R.Y.; Eickelberg, O.; Thickett, D.R. Epithelial–mesenchymal transition in lung development and disease: Does it exist and is it important? Thorax 2014, 69, 760–765. [Google Scholar] [CrossRef]
- Nieto, M.A.; Huang, R.Y.Y.J.; Jackson, R.A.A.; Thiery, J.P.P. EMT: 2016. Cell 2016, 166, 21–45. [Google Scholar] [CrossRef] [PubMed]
- Onder, T.T.; Gupta, P.B.; Mani, S.A.; Yang, J.; Lander, E.S.; Weinberg, R.A. Loss of E-cadherin promotes metastasis via multiple downstream transcriptional pathways. Cancer Res. 2008, 68, 3645–3654. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Jones, M.; Davies, D.; Wang, Y. Epithelial-Mesenchymal Transition Contributes to Pulmonary Fibrosis via Aberrant Epithelial/Fibroblastic Cross-Talk. J. Lung Health Dis. 2019, 3, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Marmai, C.; Sutherland, R.E.; Kim, K.K.; Dolganov, G.M.; Fang, X.; Kim, S.S.; Jiang, S.; Golden, J.A.; Hoopes, C.W.; Matthay, M.A.; et al. Alveolar epithelial cells express mesenchymal proteins in patients with idiopathic pulmonary fibrosis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2011, 301, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Rock, J.R.; Barkauskas, C.E.; Cronce, M.J.; Xue, Y.; Harris, J.R.; Liang, J.; Noble, P.W.; Hogan, B.L.M. Multiple stromal populations contribute to pulmonary fibrosis without evidence for epithelial to mesenchymal transition. Proc. Natl. Acad. Sci. USA 2011, 108, E1475. [Google Scholar] [CrossRef] [PubMed]
- Degryse, A.L.; Tanjore, H.; Xu, X.C.; Polosukhin, V.V.; Jones, B.R.; Boomershine, C.S.; Ortiz, C.; Sherrill, T.P.; McMahon, F.B.; Gleaves, L.A.; et al. TGFβ signaling in lung epithelium regulates bleomycin-induced alveolar injury and fibroblast recruitment. Am. J. Physiol. Lung Cell. Mol. Physiol. 2011, 300, L887. [Google Scholar] [CrossRef]
- Yao, L.; Conforti, F.; Hill, C.; Bell, J.; Drawater, L.; Li, J.; Liu, D.; Xiong, H.; Alzetani, A.; Chee, S.J.; et al. Paracrine signalling during ZEB1-mediated epithelial–mesenchymal transition augments local myofibroblast differentiation in lung fibrosis. Cell Death Differ. 2019, 26, 943–957. [Google Scholar] [CrossRef]
- Chanda, D.; Kurundkar, A.; Rangarajan, S.; Locy, M.; Bernard, K.; Sharma, N.S.; Logsdon, N.J.; Liu, H.; Crossman, D.K.; Horowitz, J.C.; et al. Developmental Reprogramming in Mesenchymal Stromal Cells of Human Subjects with Idiopathic Pulmonary Fibrosis. Sci. Rep. 2016, 6, 37445. [Google Scholar] [CrossRef]
- Lehmann, M.; Baarsma, H.A.; Königshoff, M. WNT signaling in lung aging and disease. Ann. Am. Thorac. Soc. 2016, 13, 411–416. [Google Scholar] [CrossRef]
- Herriges, M.; Morrisey, E.E. Lung development: Orchestrating the generation and regeneration of a complex organ. Development 2014, 141, 502–513. [Google Scholar] [CrossRef]
- Morrisey, E.E.; Hogan, B.L.M. Preparing for the First Breath: Genetic and Cellular Mechanisms in Lung Development. Dev. Cell 2010, 18, 8–23. [Google Scholar] [CrossRef]
- Königshoff, M.; Balsara, N.; Pfaff, E.M.; Kramer, M.; Chrobak, I.; Seeger, W.; Eickelberg, O. Functional Wnt signaling is increased in idiopathic pulmonary fibrosis. PLoS ONE 2008, 3, e2142. [Google Scholar] [CrossRef] [PubMed]
- Selman, M.; Pardo, A.; Kaminski, N. Idiopathic pulmonary fibrosis: Aberrant recapitulation of developmental programs? PLoS Med. 2008, 5, e62. [Google Scholar] [CrossRef] [PubMed]
- Landi, C.; Carleo, A.; Vantaggiato, L.; Bergantini, L.; D’Alessandro, M.; Cameli, P.; Sebastiani, G.; Dotta, F.; Bargagli, E. Common molecular pathways targeted by nintedanib in cancer and IPF: A bioinformatic study. Pulm. Pharmacol. Ther. 2020, 64, 101941. [Google Scholar] [CrossRef]
- Aschner, Y.; Downey, G.P. Transforming Growth Factor-β: Master Regulator of the Respiratory System in Health and Disease. Am. J. Respir. Cell Mol. Biol. 2016, 54, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Juban, G.; Saclier, M.; Yacoub-Youssef, H.; Kernou, A.; Arnold, L.; Boisson, C.; Ben Larbi, S.; Magnan, M.; Cuvellier, S.; Théret, M.; et al. AMPK Activation Regulates LTBP4-Dependent TGF-β1 Secretion by Pro-inflammatory Macrophages and Controls Fibrosis in Duchenne Muscular Dystrophy. Cell Rep. 2018, 25, 2163–2176.e6. [Google Scholar] [CrossRef] [PubMed]
- Meyer, A.; Wang, W.; Qu, J.; Croft, L.; Degen, J.L.; Coller, B.S.; Ahamed, J. Platelet TGF-β1 contributions to plasma TGF-β1, cardiac fibrosis, and systolic dysfunction in a mouse model of pressure overload. Blood 2012, 119, 1064–1074. [Google Scholar] [CrossRef]
- Celada, L.J.; Kropski, J.A.; Herazo-Maya, J.D.; Luo, W.; Creecy, A.; Abad, A.T.; Chioma, O.S.; Lee, G.; Hassell, N.E.; Shaginurova, G.I.; et al. PD-1 up-regulation on CD4+ T cells promotes pulmonary fibrosis through STAT3-mediated IL-17A and TGF-β1 production. Sci. Transl. Med. 2018, 10, eaar8356. [Google Scholar] [CrossRef]
- Falcone, D.J.; McCaffrey, T.A.; Haimovitz-Friedman, A.; Vergilio, J.A.; Nicholson, A.C. Macrophage and foam cell release of matrix-bound growth factors. Role of plasminogen activation. J. Biol. Chem. 1993, 268, 11951–11958. [Google Scholar] [CrossRef]
- Frangogiannis, N.G. Transforming growth factor–β in tissue fibrosis. J. Exp. Med. 2020, 217, e20190103. [Google Scholar] [CrossRef]
- Russo, I.; Cavalera, M.; Huang, S.; Su, Y.; Hanna, A.; Chen, B.; Shinde, A.V.; Conway, S.J.; Graff, J.; Frangogiannis, N.G. Protective Effects of Activated Myofibroblasts in the Pressure-Overloaded Myocardium Are Mediated Through Smad-Dependent Activation of a Matrix-Preserving Program. Circ. Res. 2019, 124, 1214–1227. [Google Scholar] [CrossRef]
- Krafts, K.P. Tissue repair: The hidden drama. Organogenesis 2010, 6, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, I.E.; Eickelberg, O. The impact of TGF-β on lung fibrosis: From targeting to biomarkers. Proc. Am. Thorac. Soc. 2012, 9, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A.; Vannella, K.M. Macrophages in Tissue Repair, Regeneration, and Fibrosis. Immunity 2016, 44, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Travis, M.A.; Sheppard, D. TGF-β activation and function in immunity. Annu. Rev. Immunol. 2014, 32, 51–82. [Google Scholar] [CrossRef] [PubMed]
- Nevers, T.; Salvador, A.M.; Velazquez, F.; Ngwenyama, N.; Carrillo-Salinas, F.J.; Aronovitz, M.; Blanton, R.M.; Alcaide, P. Th1 effector T cells selectively orchestrate cardiac fibrosis in nonischemic heart failure. J. Exp. Med. 2017, 214, 3311–3329. [Google Scholar] [CrossRef]
- Wilson, M.S.; Wynn, T.A. Pulmonary fibrosis: Pathogenesis, etiology and regulation. Mucosal Immunol. 2009, 2, 103–121. [Google Scholar] [CrossRef]
- Degryse, A.L.; Tanjore, H.; Xu, X.C.; Polosukhin, V.V.; Jones, B.R.; McMahon, F.B.; Gleaves, L.A.; Blackwell, T.S.; Lawson, W.E. Repetitive intratracheal bleomycin models several features of idiopathic pulmonary fibrosis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2010, 299, 442–452. [Google Scholar] [CrossRef]
- Logan, C.Y.; Nusse, R. The Wnt signaling pathway in development and disease. Annu. Rev. Cell Dev. Biol. 2004, 20, 781–810. [Google Scholar] [CrossRef]
- Nusse, R.; Clevers, H. Wnt/β-Catenin Signaling, Disease, and Emerging Therapeutic Modalities. Cell 2017, 169, 985–999. [Google Scholar] [CrossRef]
- Pongracz, J.E.; Stockley, R.A. Wnt signalling in lung development and diseases. Respir. Res. 2006, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Crampton, S.P.; Hughes, C.C.W. Wnt Signaling Induces Matrix Metalloproteinase Expression and Regulates T Cell Transmigration. Immunity 2007, 26, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Qu, B.; Liu, B.R.; Du, Y.J.; Chen, J.; Cheng, Y.Q.; Xu, W.; Wang, X.H. Wnt/β-catenin signaling pathway may regulate the expression of angiogenic growth factors in hepatocellular carcinoma. Oncol. Lett. 2014, 7, 1175–1178. [Google Scholar] [CrossRef] [PubMed]
- Pandur, P.; Maurus, D.; Kühl, M. Increasingly complex: New players enter the Wnt signaling network. BioEssays 2002, 24, 881–884. [Google Scholar] [CrossRef]
- Zhang, Y.; Goss, A.M.; Cohen, E.D.; Kadzik, R.; Lepore, J.J.; Muthukumaraswamy, K.; Yang, J.; DeMayo, F.J.; Whitsett, J.A.; Parmacek, M.S.; et al. A Gata6-Wnt pathway required for epithelial stem cell development and airway regeneration. Nat. Genet. 2008, 40, 862–870. [Google Scholar] [CrossRef]
- Flozak, A.S.; Lam, A.P.; Russell, S.; Jain, M.; Peled, O.N.; Sheppard, K.A.; Beri, R.; Mutlu, G.M.; Budinger, G.R.S.; Gottardi, C.J. β-Catenin/T-cell factor signaling is activated during lung injury and promotes the survival and migration of alveolar epithelial cells. J. Biol. Chem. 2010, 285, 3157–3167. [Google Scholar] [CrossRef]
- Xu, W.; Xu, B.; Zhao, Y.; Yang, N.; Liu, C.; Wen, G.; Zhang, B. Wnt5a reverses the inhibitory effect of hyperoxia on transdifferentiation of alveolar epithelial type II cells to type I cells. J. Physiol. Biochem. 2015, 71, 823–838. [Google Scholar] [CrossRef]
- Vuga, L.J.; Ben-Yehudah, A.; Kovkarova-Naumovski, E.; Oriss, T.; Gibson, K.F.; Feghali-Bostwick, C.; Kaminski, N. WNT5A is a regulator of fibroblast proliferation and resistance to apoptosis. Am. J. Respir. Cell Mol. Biol. 2009, 41, 583–589. [Google Scholar] [CrossRef]
- Chilosi, M.; Poletti, V.; Zamò, A.; Lestani, M.; Montagna, L.; Piccoli, P.; Pedron, S.; Bertaso, M.; Scarpa, A.; Murer, B.; et al. Aberrant Wnt/β-catenin pathway activation in idiopathic pulmonary fibrosis. Am. J. Pathol. 2003, 162, 1495–1502. [Google Scholar] [CrossRef]
- Kim, T.H.; Kim, S.H.; Seo, J.Y.; Chung, H.; Kwak, H.J.; Lee, S.K.; Yoon, H.J.; Shin, D.H.; Park, S.S.; Sohn, J.W. Blockade of the Wnt/β-Catenin Pathway Attenuates Bleomycin-Induced Pulmonary Fibrosis. Tohoku J. Exp. Med. 2011, 223, 45–54. [Google Scholar] [CrossRef]
- Hamburg-Shields, E.; Dinuoscio, G.J.; Mullin, N.K.; Lafayatis, R.; Atit, R.P. Sustained β-catenin activity in dermal fibroblasts promotes fibrosis by up-regulating expression of extracellular matrix protein-coding genes. J. Pathol. 2015, 235, 686–697. [Google Scholar] [CrossRef] [PubMed]
- Aumiller, V.; Balsara, N.; Wilhelm, J.; Günther, A.; Königshoff, M. WNT/β-catenin signaling induces IL-1β expression by alveolar epithelial cells in pulmonary fibrosis. Am. J. Respir. Cell Mol. Biol. 2013, 49, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Liu, Y.; Kahn, M.; Ann, D.K.; Han, A.; Wang, H.; Nguyen, C.; Flodby, P.; Zhong, Q.; Krishnaveni, M.S.; et al. Interactions between β-catenin and transforming growth factor-β signaling pathways mediate epithelial- mesenchymal transition and are dependent on the transcriptional co-activator cAMP-response element-binding protein (CREB)-binding protein (CBP). J. Biol. Chem. 2012, 287, 7026–7038. [Google Scholar] [CrossRef] [PubMed]
- Hasaneen, N.A.; Cao, J.; Pulkoski-Gross, A.; Zucker, S.; Foda, H.D. Extracellular Matrix Metalloproteinase Inducer (EMMPRIN) promotes lung fibroblast proliferation, survival and differentiation to myofibroblasts. Respir. Res. 2016, 17, 169–183. [Google Scholar] [CrossRef]
- Ferrara, N.; Gerber, H.P.; LeCouter, J. The biology of VEGF and its receptors. Nat. Med. 2003, 9, 669–676. [Google Scholar] [CrossRef]
- Koch, S.; Claesson-Welsh, L. Signal transduction by vascular endothelial growth factor receptors. Cold Spring Harb. Perspect. Med. 2012, 2, a006502. [Google Scholar] [CrossRef] [PubMed]
- Kaner, R.J.; Crystal, R.G. Compartmentalization of vascular endothelial growth factor to the epithelial surface of the human lung. Mol. Med. 2001, 7, 240–246. [Google Scholar] [CrossRef]
- Chanda, D.; Otoupalova, E.; Smith, S.R.; Volckaert, T.; De Langhe, S.P.; Thannickal, V.J. Developmental pathways in the pathogenesis of lung fibrosis. Mol. Aspects Med. 2019, 65, 56–69. [Google Scholar] [CrossRef]
- Medford, A.R.L.; Millar, A.B. Vascular endothelial growth factor (VEGF) in acute lung injury (ALI) and acute respiratory distress syndrome (ARDS): Paradox or paradigm? Thorax 2006, 61, 621–626. [Google Scholar] [CrossRef]
- Varet, J.; Douglas, S.K.; Gilmartin, L.; Medford, A.R.L.; Bates, D.O.; Harper, S.J.; Millar, A.B. VEGF in the lung: A role for novel isoforms. Am. J. Physiol. Lung Cell. Mol. Physiol. 2010, 298, 768–774. [Google Scholar] [CrossRef]
- Chen, C.M.; Wang, L.F. High-dose vascular endothelial growth factor increases surfactant protein gene expressions in preterm rat lung. Early Hum. Dev. 2007, 83, 581–584. [Google Scholar] [CrossRef] [PubMed]
- Leung, D.W.; Cachianes, G.; Kuang, W.J.; Goeddel, D.V.; Ferrara, N. Vascular endothelial growth factor is a secreted angiogenic mitogen. Science 1989, 246, 1306–1309. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, H.; Krüger, S.; Hammerschmidt, S.; Wirtz, H. High concentrations of vascular endothelial growth factor reduce stretch-induced apoptosis of alveolar type II cells. Respirology 2010, 15, 343–348. [Google Scholar] [CrossRef]
- Mura, M.; Binnie, M.; Han, B.; Li, C.; Andrade, C.F.; Shiozaki, A.; Zhang, Y.; Ferrara, N.; Hwang, D.; Waddell, T.K.; et al. Functions of type II pneumocyte-derived vascular endothelial growth factor in alveolar structure, acute inflammation, and vascular permeability. Am. J. Pathol. 2010, 176, 1725–1734. [Google Scholar] [CrossRef] [PubMed]
- Le Cras, T.D.; Spitzmiller, R.E.; Albertine, K.H.; Greenberg, J.M.; Whitsett, J.A.; Akeson, A.L. VEGF causes pulmonary hemorrhage, hemosiderosis, and air space enlargement in neonatal mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 2004, 287, 10. [Google Scholar] [CrossRef]
- Ando, M.; Miyazaki, E.; Ito, T.; Hiroshige, S.; Nureki, S.I.; Ueno, T.; Takenaka, R.; Fukami, T.; Kumamoto, T. Significance of serum vascular endothelial growth factor level in patients with idiopathic pulmonary fibrosis. Lung 2010, 188, 247–252. [Google Scholar] [CrossRef]
- Arai, T.; Hirose, M.; Kagawa, T.; Hatsuda, K.; Inoue, Y. Platelet-derived growth factor can predict survival and acute exacerbation in patients with idiopathic pulmonary fibrosis. J. Thorac. Dis. 2022, 14, 278–294. [Google Scholar] [CrossRef]
- Murray, L.A.; Habiel, D.M.; Hohmann, M.; Camelo, A.; Shang, H.; Zhou, Y.; Coelho, A.L.; Peng, X.; Gulati, M.; Crestani, B.; et al. Antifibrotic role of vascular endothelial growth factor in pulmonary fibrosis. JCI Insight 2017, 2, e92192. [Google Scholar] [CrossRef]
- Amano, H.; Mastui, Y.; Ito, Y.; Shibata, Y.; Betto, T.; Eshima, K.; Ogawa, F.; Satoh, Y.; Shibuya, M.; Majima, M. The role of vascular endothelial growth factor receptor 1 tyrosine kinase signaling in bleomycin-induced pulmonary fibrosis. Biomed. Pharmacother. 2019, 117, 109067. [Google Scholar] [CrossRef]
- Probst, C.K.; Montesi, S.B.; Medoff, B.D.; Shea, B.S.; Knipe, R.S. Vascular permeability in the fibrotic lung. Eur. Respir. J. 2020, 56, 1900100. [Google Scholar] [CrossRef]
- Barratt, S.L.; Blythe, T.; Jarrett, C.; Ourradi, K.; Shelley-Fraser, G.; Day, M.J.; Qiu, Y.; Harper, S.; Maher, T.M.; Oltean, S.; et al. Differential expression of VEGF-Axxx isoforms is critical for development of pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 2017, 196, 479–493. [Google Scholar] [CrossRef] [PubMed]
- Ebina, M.; Shimizukawa, M.; Shibata, N.; Kimura, Y.; Suzuki, T.; Endo, M.; Sasano, H.; Kondo, T.; Nukiwa, T. Heterogeneous increase in CD34-positive alveolar capillaries in idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 2004, 169, 1203–1208. [Google Scholar] [CrossRef] [PubMed]
- Tzouvelekis, A.; Anevlavis, S.; Bouros, D. Angiogenesis in interstitial Lung Diseases: A pathogenetic hallmark or a bystander? Respir. Res. 2006, 7, 82. [Google Scholar] [CrossRef] [PubMed]
- Ingham, P.W.; McMahon, A.P. Hedgehog signaling in animal development: Paradigms and principles. Genes Dev. 2001, 15, 3059–3087. [Google Scholar] [CrossRef]
- Nüsslein-Volhard, C.; Wieschaus, E. Mutations affecting segment number and polarity in drosophila. Nature 1980, 287, 795–801. [Google Scholar] [CrossRef]
- Fernandes-Silva, H.; Correia-Pinto, J.; Moura, R.S. Canonical sonic hedgehog signaling in early lung development. J. Dev. Biol. 2017, 5, 3. [Google Scholar] [CrossRef]
- Taipale, J.; Cooper, M.K.; Maiti, T.; Beachy, P.A. Patched acts catalytically to suppress the activity of smoothened. Nature 2002, 418, 892–897. [Google Scholar] [CrossRef]
- Humke, E.W.; Dorn, K.V.; Milenkovic, L.; Scott, M.P.; Rohatgi, R. The output of Hedgehog signaling is controlled by the dynamic association between Suppressor of Fused and the Gli proteins. Genes Dev. 2010, 24, 670–682. [Google Scholar] [CrossRef]
- Sasaki, H.; Hui, C.C.; Nakafuku, M.; Kondoh, H. A binding site for Gli proteins is essential for HNF-3β floor plate enhancer activity in transgenics and can respond to Shh in vitro. Development 1997, 124, 1313–1322. [Google Scholar] [CrossRef]
- Oliver, T.G.; Grasfeder, L.L.; Carroll, A.L.; Kaiser, C.; Gillingham, C.L.; Lin, S.M.; Wickramasinghe, R.; Scott, M.P.; Wechsler-Reya, R.J. Transcriptional profiling of the Sonic hedgehog response: A critical role for N-myc in proliferation of neuronal precursors. Proc. Natl. Acad. Sci. USA 2003, 100, 7331–7336. [Google Scholar] [CrossRef]
- Astorga, J.; Carlsson, P. Hedgehog induction of murine vasculogenesis is mediated by Foxf1 and Bmp4. Development 2007, 134, 3753–3761. [Google Scholar] [CrossRef] [PubMed]
- Bolaños, A.L.; Milla, C.M.; Lira, J.C.; Ramírez, R.; Checa, M.; Barrera, L.; García-Alvarez, J.; Carbajal, V.; Becerril, C.; Gaxiola, M.; et al. Role of Sonic Hedgehog in idiopathic pulmonary fibrosis. Am. J. Physiol. Lung Cell. Mol. SPhysiol. 2012, 303, 11. [Google Scholar] [CrossRef]
- Cigna, N.; Farrokhi Moshai, E.; Brayer, S.; Marchal-Somme, J.; Wémeau-Stervinou, L.; Fabre, A.; Mal, H.; Lesèche, G.; Dehoux, M.; Soler, P.; et al. The hedgehog system machinery controls transforming growth factor-β-dependent myofibroblastic differentiation in humans: Involvement in idiopathic pulmonary fibrosis. Am. J. Pathol. 2012, 181, 2126–2137. [Google Scholar] [CrossRef] [PubMed]
- Horn, A.; Palumbo, K.; Cordazzo, C.; Dees, C.; Akhmetshina, A.; Tomcik, M.; Zerr, P.; Avouac, J.; Gusinde, J.; Zwerina, J.; et al. Hedgehog signaling controls fibroblast activation and tissue fibrosis in systemic sclerosis. Arthritis Rheum. 2012, 64, 2724–2733. [Google Scholar] [CrossRef]
- Liu, L.; Kugler, M.C.; Loomis, C.A.; Samdani, R.; Zhao, Z.; Chen, G.J.; Brandt, J.P.; Brownell, I.; Joyner, A.L.; Rom, W.N.; et al. Hedgehog signaling in neonatal and adult lung. Am. J. Respir. Cell Mol. Biol. 2013, 48, 703–710. [Google Scholar] [CrossRef] [PubMed]
- Moshai, E.F.; Wémeau-Stervinou, L.; Cigna, N.; Brayer, S.; Sommé, J.M.; Crestani, B.; Mailleux, A.A. Targeting the Hedgehog-Glioma-associated oncogene homolog pathway inhibits bleomycin-induced lung fibrosis in mice. Am. J. Respir. Cell Mol. Biol. 2014, 51, 11–25. [Google Scholar] [CrossRef]
- Xu, K.; Moghal, N.; Egan, S.E. Notch signaling in lung development and disease. In Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2012; Volume 727, pp. 89–98. ISBN 9781461408987. [Google Scholar]
- Bray, S.J. Notch signalling in context. Nat. Rev. Mol. Cell Biol. 2016, 17, 722–735. [Google Scholar] [CrossRef] [PubMed]
- Kavian, N. New Insights into the Mechanism of Notch Signalling in Fibrosis. Open Rheumatol. J. 2012, 6, 96–102. [Google Scholar] [CrossRef]
- Noseda, M.; Fu, Y.X.; Niessen, K.; Wong, F.; Chang, L.; McLean, G.; Karsan, A. Smooth muscle α-actin is a direct target of Notch/CSL. Circ. Res. 2006, 98, 1468–1470. [Google Scholar] [CrossRef]
- Hu, B.; Wu, Z.; Bai, D.; Liu, T.; Ullenbruch, M.R.; Phan, S.H. Mesenchymal deficiency of notch1 attenuates bleomycin-induced pulmonary fibrosis. Am. J. Pathol. 2015, 185, 3066–3075. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, G.; Mo, B.; Wang, C. Artesunate ameliorates lung fibrosis via inhibiting the notch signaling pathway. Exp. Ther. Med. 2017, 14, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Ornitz, D.M.; Itoh, N. The fibroblast growth factor signaling pathway. Wiley Interdiscip. Rev. Dev. Biol. 2015, 4, 215–266. [Google Scholar] [CrossRef] [PubMed]
- Kranenburg, A.R.; Willems-Widyastuti, A.; Mooi, W.J.; Saxena, P.R.; Sterk, P.J.; de Boer, W.I.; Sharma, H.S. Chronic obstructive pulmonary disease is associated with enhanced bronchial expression of FGF-1, FGF-2, and FGFR-1. J. Pathol. 2005, 206, 28–38. [Google Scholar] [CrossRef]
- Guzy, R.D.; Stoilov, I.; Elton, T.J.; Mecham, R.P.; Ornitz, D.M. Fibroblast growth factor 2 is required for epithelial recovery, but not for pulmonary fibrosis, in response to bleomycin. Am. J. Respir. Cell Mol. Biol. 2015, 52, 116–128. [Google Scholar] [CrossRef]
- MacKenzie, B.A.; Korfei, M.; Henneke, I.; Sibinska, Z.; Tian, X.; Hezel, S.; Dilai, S.; Wasnick, R.; Schneider, B.; Wilhelm, J.; et al. Increased FGF1-FGFRc expression in idiopathic pulmonary fibrosis. Respir. Res. 2015, 16, 83. [Google Scholar] [CrossRef] [PubMed]
- Volckaert, T.; Dill, E.; Campbell, A.; Tiozzo, C.; Majka, S.; Bellusci, S.; De Langhe, S.P. Parabronchial smooth muscle constitutes an airway epithelial stem cell niche in the mouse lung after injury. J. Clin. Investig. 2011, 121, 4409–4419. [Google Scholar] [CrossRef]
- Stevens, N.C.; Edwards, P.C.; Tran, L.M.; Ding, X.; Van Winkle, L.S.; Fiehn, O. Metabolomics of Lung Microdissections Reveals Region- And Sex-Specific Metabolic Effects of Acute Naphthalene Exposure in Mice. Toxicol. Sci. 2021, 184, 214–222. [Google Scholar] [CrossRef]
- Shimbori, C.; Bellaye, P.S.; Xia, J.; Gauldie, J.; Ask, K.; Ramos, C.; Becerril, C.; Pardo, A.; Selman, M.; Kolb, M. Fibroblast growth factor-1 attenuates TGF-β1-induced lung fibrosis. J. Pathol. 2016, 240, 197–210. [Google Scholar] [CrossRef]
- Cutroneo, K.R.; White, S.L.; Phan, S.H.; Ehrlich, H.P. Therapies for bleomycin induced lung fibrosis through regulation of TGF-β1 induced collagen gene expression. J. Cell. Physiol. 2007, 211, 585–589. [Google Scholar] [CrossRef]
- Sheppard, D. Transforming growth factor β: A central modulator of pulmonary and airway inflammation and fibrosis. Proc. Am. Thorac. Soc. 2006, 3, 413–417. [Google Scholar] [CrossRef]
- Biernacka, A.; Dobaczewski, M.; Frangogiannis, N.G. TGF-β signaling in fibrosis. Growth Factors 2011, 29, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Ihn, H. Pathogenesis of fibrosis: Role of TGF-β and CTGF. Curr. Opin. Rheumatol. 2002, 14, 681–685. [Google Scholar] [CrossRef] [PubMed]
- Bonniaud, P.; Margetts, P.J.; Kolb, M.; Haberberger, T.; Kelly, M.; Robertson, J.; Gauldie, J. Adenoviral gene transfer of connective tissue growth factor in the lung induces transient fibrosis. Am. J. Respir. Crit. Care Med. 2003, 168, 770–778. [Google Scholar] [CrossRef] [PubMed]
- Hyldgaard, C.; Hilberg, O.; Muller, A.; Bendstrup, E. A cohort study of interstitial lung diseases in central Denmark. Respir. Med. 2014, 108, 793–799. [Google Scholar] [CrossRef]
- Bonner, J.C. Regulation of PDGF and its receptors in fibrotic diseases. Cytokine Growth Factor Rev. 2004, 15, 255–273. [Google Scholar] [CrossRef]
- Abdollahi, A.; Li, M.; Ping, G.; Plathow, C.; Domhan, S.; Kiessling, F.; Lee, L.B.; McMahon, G.; Gröne, H.J.; Lipson, K.E.; et al. Inhibition of platelet-derived growth factor signaling attenuates pulmonary fibrosis. J. Exp. Med. 2005, 201, 925–935. [Google Scholar] [CrossRef]
- Hetzel, M.; Bachem, M.; Anders, D.; Trischler, G.; Faehling, M. Different effects of growth factors on proliferation and matrix production of normal and fibrotic human lung fibroblasts. Lung 2005, 183, 225–237. [Google Scholar] [CrossRef]
- Osornio-Vargas, A.R.; Goodell, A.L.; Hernández-Rodríguez, N.A.; Brody, A.R.; Coin, P.G.; Badgett, A.; Bonner, J.C. Platelet-derived growth factor (PDGF)-AA, -AB, and -BB induce differential chemotaxis of early-passage rat lung fibroblasts in vitro. Am. J. Respir. Cell Mol. Biol. 1995, 12, 33–40. [Google Scholar] [CrossRef]
- Hsu, E.; Feghali-Bostwick, C.A. Insulin-like growth factor-II is increased in systemic sclerosis-associated pulmonary fibrosis and contributes to the fibrotic process via jun N-terminal kinase- and phosphatidylinositol-3 kinase-dependent pathways. Am. J. Pathol. 2008, 172, 1580–1590. [Google Scholar] [CrossRef]
- Maeda, A.; Hiyama, K.; Yamakido, H.; Ishioka, S.; Yamakido, M. Increased expression of platelet-derived growth factor A and insulin-like growth factor-I in BAL cells during the development of bleomycin-induced pulmonary fibrosis in mice. Chest 1996, 109, 780–786. [Google Scholar] [CrossRef]
- Atamas, S.P. Complex cytokine regulation of tissue fibrosis. Life Sci. 2002, 72, 631–643. [Google Scholar] [CrossRef] [PubMed]
- Jakubzick, C.; Kunkel, S.L.; Puri, R.K.; Hogaboam, C.M. Therapeutic targeting of IL-4- And IL-13-responsive cells in pulmonary fibrosis. Immunol. Res. 2004, 30, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Fuschiotti, P. Role of IL-13 in systemic sclerosis. Cytokine 2011, 56, 544–549. [Google Scholar] [CrossRef] [PubMed]
- Joshi, B.H.; Hogaboam, C.; Dover, P.; Husain, S.R.; Puri, R.K. Role of Interleukin-13 in Cancer, Pulmonary Fibrosis, and Other TH2-Type Diseases. Vitam. Horm. 2006, 74, 479–504. [Google Scholar] [CrossRef]
- King, T.E.; Albera, C.; Bradford, W.Z.; Costabel, U.; Hormel, P.; Lancaster, L.; Noble, P.W.; Sahn, S.A.; Szwarcberg, J.; Thomeer, M.; et al. Effect of interferon gamma-1b on survival in patients with idiopathic pulmonary fibrosis (INSPIRE): A multicentre, randomised, placebo-controlled trial. Lancet 2009, 374, 222–228. [Google Scholar] [CrossRef]
- Raghu, G.; Brown, K.K.; Bradford, W.Z.; Starko, K.; Noble, P.W.; Schwartz, D.A.; King, T.E. A Placebo-Controlled Trial of Interferon Gamma-1b in Patients with Idiopathic Pulmonary Fibrosis. N. Engl. J. Med. 2004, 350, 125–133. [Google Scholar] [CrossRef]
- Hoshino, T.; Okamoto, M.; Sakazaki, Y.; Kato, S.; Young, H.A.; Aizawa, H. Role of proinflammatory cytokines IL-18 and IL-1β in bleomycin-induced lung injury in humans and mice. Am. J. Respir. Cell Mol. Biol. 2009, 41, 661–670. [Google Scholar] [CrossRef]
- Kolb, M.; Margetts, P.J.; Anthony, D.C.; Pitossi, F.; Gauldie, J. Transient expression of IL-1β induces acute lung injury and chronic repair leading to pulmonary fibrosis. J. Clin. Investig. 2001, 107, 1529–1536. [Google Scholar] [CrossRef]
- Raghu, G.; Brown, K.K.; Costabel, U.; Cottin, V.; Du Bois, R.M.; Lasky, J.A.; Thomeer, M.; Utz, J.P.; Khandker, R.K.; McDermott, L.; et al. Treatment of idiopathic pulmonary fibrosis with etanercept: An exploratory, placebo-controlled trial. Am. J. Respir. Crit. Care Med. 2008, 178, 948–955. [Google Scholar] [CrossRef]
- Altintas, N.; Erboga, M.; Aktas, C.; Bilir, B.; Aydin, M.; Sengul, A.; Ates, Z.; Topcu, B.; Gurel, A. Protective Effect of Infliximab, a Tumor Necrosis Factor-Alfa Inhibitor, on Bleomycin-Induced Lung Fibrosis in Rats. Inflammation 2016, 39, 65–78. [Google Scholar] [CrossRef]
- Guo, J.; Luo, Y.; Yin, F.; Huo, X.; Niu, G.; Song, M.; Chen, S.; Zhang, X. Overexpression of Tumor Necrosis Factor-Like Ligand 1 A in Myeloid Cells Aggravates Liver Fibrosis in Mice. J. Immunol. Res. 2019, 2019, 7657294. [Google Scholar] [CrossRef]
- Wilson, M.S.; Madala, S.K.; Ramalingam, T.R.; Gochuico, B.R.; Rosas, I.O.; Cheever, A.W.; Wynn, T.A. Bleomycin and IL-1β-mediated pulmonary fibrosis is IL-17A dependent. J. Exp. Med. 2010, 207, 535–552. [Google Scholar] [CrossRef] [PubMed]
- Simonian, P.L.; Roark, C.L.; Wehrmann, F.; Lanham, A.K.; Diaz del Valle, F.; Born, W.K.; O’Brien, R.L.; Fontenot, A.P. Th17-Polarized Immune Response in a Murine Model of Hypersensitivity Pneumonitis and Lung Fibrosis. J. Immunol. 2009, 182, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, A.; Brewer, A.W.; Trueblood, E.S.; Luzina, I.G.; Todd, N.W.; Atamas, S.P.; Arnett, H.A. Mechanisms of Oncostatin M-Induced Pulmonary Inflammation and Fibrosis. J. Immunol. 2008, 181, 7243–7253. [Google Scholar] [CrossRef] [PubMed]
- Ihn, H.; Tamaki, K. Oncostatin M Stimulates the Growth of Dermal Fibroblasts Via a Mitogen-Activated Protein Kinase-Dependent Pathway. J. Immunol. 2000, 165, 2149–2155. [Google Scholar] [CrossRef] [PubMed]
- Scaffidi, A.K.; Mutsaers, S.E.; Moodley, Y.P.; McAnulty, R.J.; Laurent, G.J.; Thompson, P.J.; Knight, D.A. Oncostatin M stimulates proliferation, induces collagen production and inhibits apoptosis of human lung fibroblasts. Br. J. Pharmacol. 2002, 136, 793–801. [Google Scholar] [CrossRef]
- Sun, L.; Louie, M.C.; Vannella, K.M.; Wilke, C.A.; Levine, A.M.; Moore, B.B.; Shanley, T.P. New concepts of IL-10-induced lung fibrosis: Fibrocyte recruitment and M2 activation in a CCL2/CCR2 axis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2011, 300, L341–L353. [Google Scholar] [CrossRef]
- García-Prieto, E.; González-López, A.; Cabrera, S.; Astudillo, A.; Gutiérrez-Fernández, A.; Fanjul-Fernandez, M.; Batalla-Solís, E.; Puente, X.S.; Fueyo, A.; López-Otín, C.; et al. Resistance to bleomycin-induced lung fibrosis in MMP-8 deficient mice is mediated by interleukin-10. PLoS ONE 2010, 5, e13242. [Google Scholar] [CrossRef]
- Schrier, D.J.; Phan, S.H.; McGarry, B.M. The effects of the nude (nu/nu) mutation on bleomycin-induced pulmonary fibrosis. A biochemical evaluation. Am. Rev. Respir. Dis. 1983, 127, 614–617. [Google Scholar] [CrossRef]
- Okazaki, T.; Nakao, A.; Nakano, H.; Takahashi, F.; Takahashi, K.; Shimozato, O.; Takeda, K.; Yagita, H.; Okumura, K. Impairment of Bleomycin-Induced Lung Fibrosis in CD28-Deficient Mice. J. Immunol. 2001, 167, 1977–1981. [Google Scholar] [CrossRef]
- Piguet, P.F.; Collart, M.A.; Grau, G.E.; Kapanci, Y.; Vassalli, P. Tumor necrosis factor/cachectin plays a key role in bleomycin-induced pneumopathy and fibrosis. J. Exp. Med. 1989, 170, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; MacLean, J.A.; Pinto, C.; Kradin, R.L. The effect of an anti-CD3 monoclonal antibody on bleomycin-induced lymphokine production and lung injury. Am. J. Respir. Crit. Care Med. 1996, 154, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Maggi, E.; Cosmi, L.; Liotta, F.; Romagnani, P.; Romagnani, S.; Annunziato, F. Thymic regulatory T cells. Autoimmun. Rev. 2005, 4, 579–586. [Google Scholar] [CrossRef]
- Re, S.L.; Lecocq, M.; Uwambayinema, F.; Yakoub, Y.; Delos, M.; Demoulin, J.B.; Lucas, S.; Sparwasser, T.; Renauld, J.C.; Lison, D.; et al. Platelet-derived growth factor-producing CD4 + foxp3 +regulatory T lymphocytes promote lung fibrosis. Am. J. Respir. Crit. Care Med. 2011, 184, 1270–1281. [Google Scholar] [CrossRef]
- Daniil, Z.; Kitsanta, P.; Kapotsis, G.; Mathioudaki, M.; Kollintza, A.; Karatza, M.; Milic-Emili, J.; Roussos, C.; Papiris, S.A. CD8+ T lymphocytes in lung tissue from patients with idiopathic pulmonary fibrosis. Respir. Res. 2005, 6, 2971–2977. [Google Scholar] [CrossRef] [PubMed]
- Parra, E.R.; Kairalla, R.A.; Ribeiro De Carvalho, C.R.; Eher, E.; Capelozzi, V.L. Inflammatory cell phenotyping of the pulmonary interstitium in idiopathic interstitial pneumonia. Respiration 2007, 74, 159–169. [Google Scholar] [CrossRef]
- Fireman, E.; Vardinon, N.; Burke, M.; Spizer, S.; Levin, S.; Endler, A.; Stav, D.; Topilsky, M.; Mann, A.; Schwarz, Y.; et al. Predictive value of response to treatment of T-lymphocyte subpopulations in idiopathic pulmonary fibrosis. Eur. Respir. J. 1998, 11, 706–711. [Google Scholar] [CrossRef]
- Marchal-Sommé, J.; Uzunhan, Y.; Marchand-Adam, S.; Valeyre, D.; Soumelis, V.; Crestani, B.; Soler, P. Cutting Edge: Nonproliferating Mature Immune Cells Form a Novel Type of Organized Lymphoid Structure in Idiopathic Pulmonary Fibrosis. J. Immunol. 2006, 176, 5735–5739. [Google Scholar] [CrossRef]
- Reynolds, H.Y. Lung inflammation and fibrosis: An alveolar macrophage-centered perspective from the 1970s to 1980s. Am. J. Respir. Crit. Care Med. 2005, 171, 98–102. [Google Scholar] [CrossRef]
- Bocchino, M.; Agnese, S.; Fagone, E.; Svegliati, S.; Grieco, D.; Vancheri, C.; Gabrielli, A.; Sanduzzi, A.; Avvedimento, E.V. Reactive oxygen species are required for maintenance and differentiation of primary lung fibroblasts in idiopathic pulmonary fibrosis. PLoS ONE 2010, 5, e14003. [Google Scholar] [CrossRef]
- Schaberg, T.; Rau, M.; Stephan, H.; Lode, H. Increased number of alveolar macrophages expressing surface molecules of the CD11/CD18 family in sarcoidosis and idiopathic pulmonary fibrosis is related to the production of superoxide anions by these cells. Am. Rev. Respir. Dis. 1993, 147, 1507–1513. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q.; Dalavanga, Y.; Poulakis, N.; Sixt, S.U.; Guzman, J.; Costabel, U. Decreased expression of haem oxygenase-1 by alveolar macrophages in idiopathic pulmonary fibrosis. Eur. Respir. J. 2008, 31, 1030–1036. [Google Scholar] [CrossRef] [PubMed]
- Scotton, C.J.; Krupiczojc, M.A.; Königshoff, M.; Mercer, P.F.; Lee, Y.C.G.; Kaminski, N.; Morser, J.; Post, J.M.; Maher, T.M.; Nicholson, A.G.; et al. Increased local expression of coagulation factor X contributes to the fibrotic response in human and murine lung injury. J. Clin. Investig. 2009, 119, 2550–2563. [Google Scholar] [CrossRef] [PubMed]
- Howell, D.C.J.; Johns, R.H.; Lasky, J.A.; Shan, B.; Scotton, C.J.; Laurent, G.J.; Chambers, R.C. Absence of proteinase-activated receptor-1 signaling affords protection from bleomycin-induced lung inflammation and fibrosis. Am. J. Pathol. 2005, 166, 1353–1365. [Google Scholar] [CrossRef] [PubMed]
- Martinet, Y.; Rom, W.N.; Grotendorst, G.R.; Martin, G.R.; Crystal, R.G. Exaggerated Spontaneous Release of Platelet-Derived Growth Factor by Alveolar Macrophages from Patients with Idiopathic Pulmonary Fibrosis . N. Engl. J. Med. 1987, 317, 202–209. [Google Scholar] [CrossRef]
- Migliaccio, C.T.; Buford, M.C.; Jessop, F.; Holian, A. The IL-4Rα pathway in macrophages and its potential role in silica-induced pulmonary fibrosis. J. Leukoc. Biol. 2008, 83, 630–639. [Google Scholar] [CrossRef]
- Mora, A.L.; Torres-González, E.; Rojas, M.; Corredor, C.; Ritzenthaler, J.; Xu, J.; Roman, J.; Brigham, K.; Stecenko, A. Activation of alveolar macrophages via the alternative pathway in herpesvirus-induced lung fibrosis. Am. J. Respir. Cell Mol. Biol. 2006, 35, 466–473. [Google Scholar] [CrossRef]
- Murray, L.A.; Chen, Q.; Kramer, M.S.; Hesson, D.P.; Argentieri, R.L.; Peng, X.; Gulati, M.; Homer, R.J.; Russell, T.; Van Rooijen, N.; et al. TGF-beta driven lung fibrosis is macrophage dependent and blocked by Serum amyloid P. Int. J. Biochem. Cell Biol. 2011, 43, 154–162. [Google Scholar] [CrossRef]
- Murray, L.A.; Rosada, R.; Moreira, A.P.; Joshi, A.; Kramer, M.S.; Hesson, D.P.; Argentieri, R.L.; Mathai, S.; Gulati, M.; Herzog, E.L.; et al. Serum amyloid P therapeutically attenuates murine bleomycin-induced pulmonary fibrosis via its effects on macrophages. PLoS ONE 2010, 5, e9683. [Google Scholar] [CrossRef]
- Shum, A.K.; Alimohammadi, M.; Tan, C.L.; Cheng, M.H.; Metzger, T.C.; Law, C.S.; Lwin, W.; Perheentupa, J.; Bour-Jordan, H.; Carel, J.C.; et al. BPIFB1 is a lung-specific autoantigen associated with interstitial lung disease. Sci. Transl. Med. 2013, 5, 206ra139. [Google Scholar] [CrossRef]
- Jongerius, I.; Porcelijn, L.; van Beek, A.E.; Semple, J.W.; van der Schoot, C.E.; Vlaar, A.P.J.; Kapur, R. The Role of Complement in Transfusion-Related Acute Lung Injury. Transfus. Med. Rev. 2019, 33, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Wu, H.; Wang, C.; Xiao, Z.; Xu, F. Regulatory T cells and acute lung injury: Cytokines, uncontrolled inflammation, and therapeutic implications. Front. Immunol. 2018, 9, 1545. [Google Scholar] [CrossRef] [PubMed]
- Bartz, R.R.; Piantadosi, C.A. Clinical review: Oxygen as a signaling molecule. Crit. Care 2010, 14, 234. [Google Scholar] [CrossRef] [PubMed]
- Kinnula, V.L.; Crapo, J.D. Superoxide dismutases in the lung and human lung diseases. Am. J. Respir. Crit. Care Med. 2003, 167, 1600–1619. [Google Scholar] [CrossRef]
- Carnesecchi, S.; Deffert, C.; Donati, Y.; Basset, O.; Hinz, B.; Preynat-Seauve, O.; Guichard, C.; Arbiser, J.L.; Banfi, B.; Pache, J.C.; et al. A key role for NOX4 in epithelial cell death during development of lung fibrosis. Antioxid. Redox Signal. 2011, 15, 607–619. [Google Scholar] [CrossRef]
- Park, W.Y.; Goodman, R.B.; Steinberg, K.P.; Ruzinski, J.T.; Radella, F.; Park, D.R.; Pugin, J.; Skerrett, S.J.; Hudson, L.D.; Martin, T.R. Cytokine balance in the lungs of patients with acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 2001, 164, 1896–1903. [Google Scholar] [CrossRef]
- Berthiaume, Y.; Matthay, M.A. Alveolar edema fluid clearance and acute lung injury. Respir. Physiol. Neurobiol. 2007, 159, 350–359. [Google Scholar] [CrossRef]
- Mikacenic, C.; Moore, R.; Dmyterko, V.; West, T.E.; Altemeier, W.A.; Liles, W.C.; Lood, C. Neutrophil extracellular traps (NETs) are increased in the alveolar spaces of patients with ventilator-associated pneumonia. Crit. Care 2018, 22, 358. [Google Scholar] [CrossRef]
- Jiang, K.; Guo, S.; Yang, C.; Yang, J.; Chen, Y.; Shaukat, A.; Zhao, G.; Wu, H.; Deng, G. Barbaloin protects against lipopolysaccharide (LPS)-induced acute lung injury by inhibiting the ROS-mediated PI3K/AKT/NF-κB pathway. Int. Immunopharmacol. 2018, 64, 140–150. [Google Scholar] [CrossRef]
- Soromou, L.W.; Chen, N.; Jiang, L.; Huo, M.; Wei, M.; Chu, X.; Millimouno, F.M.; Feng, H.; Sidime, Y.; Deng, X. Astragalin attenuates lipopolysaccharide-induced inflammatory responses by down-regulating NF-κB signaling pathway. Biochem. Biophys. Res. Commun. 2012, 419, 256–261. [Google Scholar] [CrossRef]
- Zhang, B.; Liu, Z.Y.; Li, Y.Y.; Luo, Y.; Liu, M.L.; Dong, H.Y.; Wang, Y.X.; Liu, Y.; Zhao, P.T.; Jin, F.G.; et al. Antiinflammatory effects of matrine in LPS-induced acute lung injury in mice. Eur. J. Pharm. Sci. 2011, 44, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Ye, R.; Liu, Z. ACE2 exhibits protective effects against LPS-induced acute lung injury in mice by inhibiting the LPS-TLR4 pathway. Exp. Mol. Pathol. 2020, 113, 104350. [Google Scholar] [CrossRef] [PubMed]
- Domscheit, H.; Hegeman, M.A.; Carvalho, N.; Spieth, P.M. Molecular Dynamics of Lipopolysaccharide-Induced Lung Injury in Rodents. Front. Physiol. 2020, 11, 36. [Google Scholar] [CrossRef] [PubMed]
- Nova, Z.; Skovierova, H.; Calkovska, A. Alveolar-capillary membrane-related pulmonary cells as a target in endotoxin-induced acute lung injury. Int. J. Mol. Sci. 2019, 20, 831. [Google Scholar] [CrossRef] [PubMed]
- Al-Ani, B.; ShamsEldeen, A.M.; Kamar, S.S.; Haidara, M.A.; Al-Hashem, F.; Alshahrani, M.Y.; Al-Hakami, A.M.; Kader, D.H.A.; Maarouf, A. Lipopolysaccharide induces acute lung injury and alveolar haemorrhage in association with the cytokine storm, coagulopathy and AT1R/JAK/STAT augmentation in a rat model that mimics moderate and severe COVID-19 pathology. Clin. Exp. Pharmacol. Physiol. 2022, 49, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Memar, M.Y.; Yekani, M.; Alizadeh, N.; Baghi, H.B. Hyperbaric oxygen therapy: Antimicrobial mechanisms and clinical application for infections. Biomed. Pharmacother. 2019, 109, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, N.R.; Brower, R.G.; Hager, D.N.; Thompson, B.T.; Netzer, G.; Shanholtz, C.; Lagakos, A.; Checkley, W. Oxygen Exposure Resulting in Arterial Oxygen Tensions Above the Protocol Goal Was Associated with Worse Clinical Outcomes in Acute Respiratory Distress Syndrome. Crit. Care Med. 2018, 46, 517–524. [Google Scholar] [CrossRef]
- Amarelle, L.; Quintela, L.; Hurtado, J.; Malacrida, L. Hyperoxia and Lungs: What We Have Learned From Animal Models. Front. Med. 2021, 8, 235. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, Y.; Wang, Z.; Chen, Y.; Li, R. Intermittent hyperbaric oxygen exposure mobilizing peroxiredoxin 6 to prevent oxygen toxicity. J. Physiol. Sci. 2019, 69, 779–790. [Google Scholar] [CrossRef]
- Schuster, D.P. ARDS: Clinical lessons from the oleic acid model of acute lung injury. Am. J. Respir. Crit. Care Med. 1994, 149, 245–260. [Google Scholar] [CrossRef]
- Derks, C.M.; Jacobovitz Derks, D. Embolic pneumopathy induced by oleic acid: A systematic morphologic study. Am. J. Pathol. 1977, 87, 143–158. [Google Scholar] [PubMed]
- Ulrich, K.; Stern, M.; Goddard, M.E.; Williams, J.; Zhu, J.; Dewar, A.; Painter, H.A.; Jeffery, P.K.; Gill, D.R.; Hyde, S.C.; et al. Keratinocyte growth factor therapy in murine oleic acid-induced acute lung injury. Am. J. Physiol. Lung Cell. Mol. Physiol. 2005, 288, 1179–1192. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gonçalves-De-Albuquerque, C.F.; Silva, A.R.; Burth, P.; Castro-Faria, M.V.; Castro-Faria-Neto, H.C. Acute respiratory distress syndrome: Role of oleic acid-triggered lung injury and inflammation. Mediators Inflamm. 2015, 2015, 260465. [Google Scholar] [CrossRef]
- Glasbey, J.C.; Bibi, S.; Pata, F.; Ozkan, B.B.; Van Straten, S.; Hodson, J.; Chapman, S.J.; Blanco-Colino, R.; Chapman, S.J.; Pellino, G.; et al. Timing of nasogastric tube insertion and the risk of postoperative pneumonia: An international, prospective cohort study. Color. Dis. 2020, 22, 2288–2297. [Google Scholar] [CrossRef]
- Suresh, M.V.; Yalamanchili, G.; Rao, T.C.; Aktay, S.; Kralovich, A.; Shah, Y.M.; Raghavendran, K. Hypoxia-inducible factor (HIF)-1α-induced regulation of lung injury in pulmonary aspiration is mediated through NF-kB. FASEB Bioadv. 2022, 4, 309. [Google Scholar] [CrossRef] [PubMed]
- Puri, G.; Naura, A.S. Critical role of mitochondrial oxidative stress in acid aspiration induced ALI in mice. Toxicol. Mech. Methods 2020, 30, 266–274. [Google Scholar] [CrossRef]
- Yano, T.; Deterding, R.R.; Simonet, W.S.; Shannon, J.M.; Mason, R.J. Keratinocyte Growth Factor Reduces Lung Damage Due to Acid Instillation in Rats. Am. J. Respir. Cell Mol. Biol. 1996, 15, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Folkesson, H.G.; Matthay, M.A.; Hebert, C.A.; Broaddus, V.C. Acid aspiration-induced lung injury in rabbits is mediated by interleukin- 8-dependent mechanisms. J. Clin. Investig. 1995, 96, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Dreyfuss, D.; Saumon, G. Ventilator-induced lung injury: Lessons from experimental studies. Am. J. Respir. Crit. Care Med. 1998, 157, 294–323. [Google Scholar] [CrossRef]
- Dos Santos, C.C.; Slutsky, A.S. Invited review: Mechanisms of ventilator-induced lung injury: A perspective. J. Appl. Physiol. 2000, 89, 1645–1656. [Google Scholar] [CrossRef]
- Network, T.A.R.D.S. Ventilation with Lower Tidal Volumes as Compared with Traditional Tidal Volumes for Acute Lung Injury and the Acute Respiratory Distress Syndrome. N. Engl. J. Med. 2000, 342, 1301–1308. [Google Scholar] [CrossRef]
- Wolthius, E.K. Lung protective mechanical ventilation. In Lung Protective Mechanical Ventilation; UvA-DARE (Digital Academic Repository): Amsterdam, The Netherlands, 2008; pp. 86–94. [Google Scholar]
- Wang, H.M.; Bodenstein, M.; Duenges, B.; Ganatti, S.; Boehme, S.; Ning, Y.; Roehrig, B.; Markstaller, K. Ventilator-associated lung injury superposed to oleic acid infusion or surfactant depletion: Histopathological characteristics of two porcine models of acute lung injury. Eur. Surg. Res. 2010, 45, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Kamm, R.D.; Lee, R.T. Cell mechanics and mechanotransduction: Pathways, probes, and physiology. Am. J. Physiol. Cell Physiol. 2004, 287, C1–C11. [Google Scholar] [CrossRef] [PubMed]
- Kuiper, J.W.; Plötz, F.B.; Groeneveld, A.J.; Haitsma, J.J.; Jothy, S.; Vaschetto, R.; Zhang, H.; Slutsky, A.S. High tidal volume mechanical ventilation-induced lung injury in rats is greater after acid instillation than after sepsis-induced acute lung injury, but does not increase systemic inflammation: An experimental study. BMC Anesthesiol. 2011, 11, 26. [Google Scholar] [CrossRef]
- Degryse, A.L.; Lawson, W.E. Progress toward improving animal models for idiopathic pulmonary fibrosis. Am. J. Med. Sci. 2011, 341, 444–449. [Google Scholar] [CrossRef]
- Brandt, J.; Gerrites, V. Bleomycin. In xPharm: The Comprehensive Pharmacology Reference; Elsevier Inc.: Amsterdam, The Netherlands, 2007; pp. 1–6. ISBN 9780080552323. [Google Scholar]
- Moeller, A.; Ask, K.; Warburton, D.; Gauldie, J.; Kolb, M. The bleomycin animal model: A useful tool to investigate treatment options for idiopathic pulmonary fibrosis? Int. J. Biochem. Cell Biol. 2008, 40, 362–382. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; De Los Santos, F.G.; Phan, S.H. The bleomycin model of pulmonary fibrosis. Methods Mol. Biol. 2017, 1627, 27–42. [Google Scholar] [CrossRef]
- Matute-Bello, G.; Frevert, C.W.; Martin, T.R. Animal models of acute lung injury. Am. J. Physiol. Lung Cell. Mol. Physiol. 2008, 295, 379–399. [Google Scholar] [CrossRef]
- McDonald, S.; Rubin, P.; Chang, A.Y.C.; Penney, D.P.; Finkelstein, J.N.; Grossberg, S.; Feins, R.; Gregory, P.K. Pulmonary changes induced by combined mouse β-interferon (rMuIFN-β) and irradiation in normal mice-toxic versus protective effects. Radiother. Oncol. 1993, 26, 212–218. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Y.; Han, J.; Mei, H.; Yu, D.; Ding, Q.; Zhang, T.; Wu, G.; Peng, G.; Lin, Z. Metformin Attenuates Radiation-Induced Pulmonary Fibrosis in a Murine Model. Radiat. Res. 2017, 188, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Lu, L.; Liu, B.; Qin, S. Effects of phycocyanin on pulmonary and gut microbiota in a radiation-induced pulmonary fibrosis model. Biomed. Pharmacother. 2020, 132, 110826. [Google Scholar] [CrossRef]
- Andersson-Sjöland, A.; Nihlberg, K.; Eriksson, L.; Bjermer, L.; Westergren-Thorsson, G. Fibrocytes and the tissue niche in lung repair. Respir. Res. 2011, 12, 76. [Google Scholar] [CrossRef]
- Groves, A.M.; Johnston, C.J.; Williams, J.P.; Finkelstein, J.N. Role of Infiltrating Monocytes in the Development of Radiation-Induced Pulmonary Fibrosis. Radiat. Res. 2018, 189, 300–311. [Google Scholar] [CrossRef]
- Ghobadi, G.; Bartelds, B.; Van Der Veen, S.J.; Dickinson, M.G.; Brandenburg, S.; Berger, R.M.F.; Langendijk, J.A.; Coppes, R.P.; Van Luijk, P. Lung irradiation induces pulmonary vascular remodelling resembling pulmonary arterial hypertension. Thorax 2012, 67, 334–341. [Google Scholar] [CrossRef]
- Wu, X.; Kaner, R.J.; Martinez, F.J. Idiopathic Pulmonary Fibrosis: What Is the Best Treatment? Barcelona Respir. Netw. 2017, 3, 86–101. [Google Scholar] [CrossRef]
- Karkale, S.; Khurana, A.; Saifi, M.A.; Godugu, C.; Talla, V. Oropharyngeal administration of silica in Swiss mice: A robust and reproducible model of occupational pulmonary fibrosis. Pulm. Pharmacol. Ther. 2018, 51, 32–40. [Google Scholar] [CrossRef]
- Li, C.; Lu, Y.; Du, S.; Li, S.; Zhang, Y.; Liu, F.; Chen, Y.; Weng, D.; Chen, J. Dioscin exerts protective effects against crystalline silica-induced pulmonary fibrosis in mice. Theranostics 2017, 7, 4255–4275. [Google Scholar] [CrossRef]
- Barbarin, V.; Nihoul, A.; Misson, P.; Arras, M.; Delos, M.; Leclercq, I.; Lison, D.; Huaux, F. The role of pro- and anti-inflammatory responses in silica-induced lung fibrosis. Respir. Res. 2005, 6, 112. [Google Scholar] [CrossRef]
- Moore, B.B.; Lawson, W.E.; Oury, T.D.; Sisson, T.H.; Raghavendran, K.; Hogaboam, C.M. Animal models of fibrotic lung disease. Am. J. Respir. Cell Mol. Biol. 2013, 49, 167–179. [Google Scholar] [CrossRef]
- Riggs, J.L.; Seiwald, R.J.; Burckhalter, J.H.; Downs, C.M.; Metcalf, T.G. Isothiocyanate compounds as fluorescent labeling agents for immune serum. Am. J. Pathol. 1958, 34, 1081–1097. [Google Scholar]
- Roberts, S.N.; Howie, S.E.M.; Wallace, W.A.H.; Brown, D.M.; Lamb, D.; Ramage, E.A.; Donaldson, K. A novel model for human interstitial lung disease: Hapten-driven lung fibrosis in rodents. J. Pathol. 1995, 176, 309–318. [Google Scholar] [CrossRef]
- Moore, B.B.; Hogaboam, C.M. Murine models of pulmonary fibrosis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2008, 294, 152–160. [Google Scholar] [CrossRef]
- Cross, L.J.M.; Matthay, M.A. Biomarkers in Acute Lung Injury: Insights into the Pathogenesis of Acute Lung Injury. Crit. Care Clin. 2011, 27, 355–377. [Google Scholar] [CrossRef]
- Spadaro, S.; Park, M.; Turrini, C.; Tunstall, T.; Thwaites, R.; Mauri, T.; Ragazzi, R.; Ruggeri, P.; Hansel, T.T.; Caramori, G.; et al. Biomarkers for Acute Respiratory Distress syndrome and prospects for personalised medicine. J. Inflamm. 2019, 16, 1. [Google Scholar] [CrossRef]
- Rowaiye, A.B.; Okpalefe, O.A.; Adejoke, O.O.; Ogidigo, J.O.; Oladipo, O.H.; Ogu, A.C.; Oli, A.N.; Olofinase, S.; Onyekwere, O.; Abubakar, A.R.; et al. Attenuating the effects of novel COVID-19 (SARS-CoV-2) infection-induced cytokine storm and the implications. J. Inflamm. Res. 2021, 14, 1487–1510. [Google Scholar] [CrossRef]
- Ramirez, J.A.; Wiemken, T.L.; Peyrani, P.; Arnold, F.W.; Kelley, R.; Mattingly, W.A.; Nakamatsu, R.; Pena, S.; Guinn, B.E.; Furmanek, S.P.; et al. Adults Hospitalized with Pneumonia in the United States: Incidence, Epidemiology, and Mortality. Clin. Infect. Dis. 2017, 65, 1806–1812. [Google Scholar] [CrossRef]
- Storms, A.D.; Chen, J.; Jackson, L.A.; Nordin, J.D.; Naleway, A.L.; Glanz, J.M.; Jacobsen, S.J.; Weintraub, E.S.; Klein, N.P.; Gargiullo, P.M.; et al. Rates and risk factors associated with hospitalization for pneumonia with ICU admission among adults. BMC Pulm. Med. 2017, 17, 208. [Google Scholar] [CrossRef]
- Bellani, G.; Laffey, J.G.; Pham, T.; Fan, E. The LUNG SAFE study: A presentation of the prevalence of ARDS according to the Berlin Definition! Crit. Care 2016, 20, 268. [Google Scholar] [CrossRef]
- Martínez, R.; Menéndez, R.; Reyes, S.; Polverino, E.; Cillóniz, C.; Martínez, A.; Esquinas, C.; Filella, X.; Ramírez, P.; Torres, A. Factors associated with inflammatory cytokine patterns in community-acquired pneumonia. Eur. Respir. J. 2011, 37, 393–399. [Google Scholar] [CrossRef]
- Calfee, C.S.; Delucchi, K.; Parsons, P.E.; Thompson, B.T.; Ware, L.B.; Matthay, M.A. Subphenotypes in acute respiratory distress syndrome: Latent class analysis of data from two randomised controlled trials. Lancet Respir. Med. 2014, 2, 611–620. [Google Scholar] [CrossRef]
- Calfee, C.S.; Janz, D.R.; Bernard, G.R.; May, A.K.; Kangelaris, K.N.; Matthay, M.A.; Ware, L.B. Distinct molecular phenotypes of direct vs indirect ARDS in single-center and multicenter studies. Chest 2015, 147, 1539–1548. [Google Scholar] [CrossRef] [PubMed]
- Shieh, J.-M.; Tseng, H.-Y.; Jung, F.; Yang, S.-H.; Lin, J.-C. Elevation of IL-6 and IL-33 Levels in Serum Associated with Lung Fibrosis and Skeletal Muscle Wasting in a Bleomycin-Induced Lung Injury Mouse Model. Mediators Inflamm. 2019, 2019, 7947596. [Google Scholar] [CrossRef] [PubMed]
- Papiris, S.A.; Tomos, I.P.; Karakatsani, A.; Spathis, A.; Korbila, I.; Analitis, A.; Kolilekas, L.; Kagouridis, K.; Loukides, S.; Karakitsos, P.; et al. High levels of IL-6 and IL-8 characterize early-on idiopathic pulmonary fibrosis acute exacerbations. Cytokine 2018, 102, 168–172. [Google Scholar] [CrossRef]
- Santa Cruz, A.; Mendes-Frias, A.; Oliveira, A.I.; Dias, L.; Matos, A.R.; Carvalho, A.; Capela, C.; Pedrosa, J.; Castro, A.G.; Silvestre, R. Interleukin-6 Is a Biomarker for the Development of Fatal Severe Acute Respiratory Syndrome Coronavirus 2 Pneumonia. Front. Immunol. 2021, 12, 263. [Google Scholar] [CrossRef]
- Lee, Y.L.; Chen, W.; Chen, L.Y.; Chen, C.H.; Lin, Y.C.; Liang, S.J.; Shih, C.M. Systemic and bronchoalveolar cytokines as predictors of in-hospital mortality in severe community-acquired pneumonia. J. Crit. Care 2010, 25, 176.e7–176.e13. [Google Scholar] [CrossRef]
- Horspool, A.M.; Kieffer, T.; Russ, B.P.; DeJong, M.A.; Wolf, M.A.; Karakiozis, J.M.; Hickey, B.J.; Fagone, P.; Tacker, D.H.; Bevere, J.R.; et al. Interplay of Antibody and Cytokine Production Reveals CXCL13 as a Potential Novel Biomarker of Lethal SARS-CoV-2 Infection. Msphere 2021, 6, e01324-20. [Google Scholar] [CrossRef]
- Perreau, M.; Suffiotti, M.; Marques-Vidal, P.; Wiedemann, A.; Levy, Y.; Laouénan, C.; Ghosn, J.; Fenwick, C.; Comte, D.; Roger, T.; et al. The cytokines HGF and CXCL13 predict the severity and the mortality in COVID-19 patients. Nat. Commun. 2021, 12, 4888. [Google Scholar] [CrossRef]
- Parsons, P.E.; Eisner, M.D.; Thompson, B.T.; Matthay, M.A.; Ancukiewicz, M.; Bernard, G.R.; Wheeler, A.P. Lower tidal volume ventilation and plasma cytokine markers of inflammation in patients with acute lung injury. Crit. Care Med. 2005, 33, 1–6. [Google Scholar] [CrossRef]
- Frenzel, J.; Gessner, C.; Sandvoss, T.; Hammerschmidt, S.; Schellenberger, W.; Sack, U.; Eschrich, K.; Wirtz, H. Outcome prediction in pneumonia induced ALI/ARDS by clinical features and peptide patterns of BALF determined by mass spectrometry. PLoS ONE 2011, 6, e25544. [Google Scholar] [CrossRef]
- Dolinay, T.; Kim, Y.S.; Howrylak, J.; Hunninghake, G.M.; An, C.H.; Fredenburgh, L.; Massaro, A.F.; Rogers, A.; Gazourian, L.; Nakahira, K.; et al. Inflammasome-regulated cytokines are critical mediators of acute lung injury. Am. J. Respir. Crit. Care Med. 2012, 185, 1225–1234. [Google Scholar] [CrossRef]
- Kaplanski, G. Interleukin-18: Biological properties and role in disease pathogenesis. Immunol. Rev. 2018, 281, 138–153. [Google Scholar] [CrossRef]
- Tong, M.; Xiong, Y.; Zhu, C.; Xu, H.; Zheng, Q.; Jiang, Y.; Zou, L.; Xiao, X.; Chen, F.; Yan, X.; et al. Serum surfactant protein D in COVID-19 is elevated and correlated with disease severity. BMC Infect. Dis. 2021, 21, 737. [Google Scholar] [CrossRef]
- Naderi, N.; Rahimzadeh, M. Krebs von den Lungen-6 (KL-6) as a clinical marker for severe COVID-19: A systematic review and meta-analyses. Virology 2022, 566, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Thickett, D.R.; Armstrong, L.; Millar, A.B. A role for vascular endothelial growth factor in acute and resolving lung injury. Am. J. Respir. Crit. Care Med. 2002, 166, 1332–1337. [Google Scholar] [CrossRef] [PubMed]
- Kong, Y.; Han, J.; Wu, X.; Zeng, H.; Liu, J.; Zhang, H. VEGF-D: A novel biomarker for detection of COVID-19 progression. Crit. Care 2020, 24, 373. [Google Scholar] [CrossRef]
- Stern, J.-B.; Fierobe, L.; Paugam, C.; Rolland, C.; Dehoux, M.; Petiet, A.; Dombret, M.-C.; Mantz, J.; Aubier, M.; Crestani, B. Keratinocyte growth factor and hepatocyte growth factor/scatter factor are. Crit. Care Med. 2000, 28, 2326–2333. [Google Scholar] [CrossRef]
- Agrawal, A.; Zhuo, H.; Brady, S.; Levitt, J.; Steingrub, J.; Siegel, M.D.; Soto, G.; Peterson, M.W.; Chesnutt, M.S.; Matthay, M.A.; et al. Pathogenetic and predictive value of biomarkers in patients with ALI and lower severity of illness: Results from two clinical trials. Am. J. Physiol. Lung Cell. Mol. Physiol. 2012, 303, 634–639. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, A.C.C.; Flori, H.; Dahmer, M.K.; Sim, M.S.; Quasney, M.W.; Curley, M.A.Q.; Matthay, M.A.; Sapru, A.; Monteiro, A.C.C.; Dahmer, M.K.; et al. Thrombomodulin is associated with increased mortality and organ failure in mechanically ventilated children with acute respiratory failure: Biomarker analysis from a multicenter randomized controlled trial. Crit. Care 2021, 25, 271. [Google Scholar] [CrossRef] [PubMed]
- Chandel, N.S.; Budinger, G.R.S.; Mutlu, G.M.; Varga, J.; Synenki, L.; Donnelly, H.K.; Zirk, A.; Eisenbart, J.; Jovanovic, B.; Jain, M. Keratinocyte growth factor expression is suppressed in early acute lung injury/acute respiratory distress syndrome by smad and c-Abl pathways. Crit. Care Med. 2009, 37, 1678–1684. [Google Scholar] [CrossRef] [PubMed]
- Brunetta, E.; Folci, M.; Bottazzi, B.; De Santis, M.; Gritti, G.; Protti, A.; Mapelli, S.N.; Bonovas, S.; Piovani, D.; Leone, R.; et al. Macrophage expression and prognostic significance of the long pentraxin PTX3 in COVID-19. Nat. Immunol. 2021, 22, 19–24. [Google Scholar] [CrossRef]
- Hästbacka, J.; Linko, R.; Tervahartiala, T.; Varpula, T.; Hovilehto, S.; Parviainen, I.; Vaara, S.T.; Sorsa, T.; Pettilä, V. Serum MMP-8 and TIMP-1 in critically ill patients with acute respiratory failure: TIMP-1 is associated with increased 90-day mortality. Anesth. Analg. 2014, 118, 790–798. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.L.; Chen, D.; Yan, J.; Yang, Q.; Han, Q.Q.; Li, S.S.; Cheng, L. Proteomic characteristics of bronchoalveolar lavage fluid in critical COVID-19 patients. FEBS J. 2021, 288, 5190–5200. [Google Scholar] [CrossRef] [PubMed]
- da Silva-Neto, P.V.; Do Valle, V.B.; Fuzo, C.A.; Fernandes, T.M.; Toro, D.M.; Fraga-Silva, T.F.C.; Basile, P.A.; de Carvalho, J.C.S.; Pimentel, V.E.; Pérez, M.M.; et al. Matrix Metalloproteinases on Severe COVID-19 Lung Disease Pathogenesis: Cooperative Actions of MMP-8/MMP-2 Axis on Immune Response through HLA-G Shedding and Oxidative Stress. Biomolecules 2022, 12, 604. [Google Scholar] [CrossRef] [PubMed]
- Van Oort, P.M.; Bos, L.D.; Póvoa, P.; Ramirez, P.; Torres, A.; Artigas, A.; Schultz, M.J.; Martin-Loeches, I. Soluble urokinase plasminogen activator receptor for the prediction of ventilator-associated pneumonia. ERJ Open Res. 2019, 5, 00212-2018. [Google Scholar] [CrossRef] [PubMed]
- Conrad, C.; Yildiz, D.; Cleary, S.J.; Margraf, A.; Cook, L.; Schlomann, U.; Panaretou, B.; Bowser, J.L.; Karmouty-Quintana, H.; Li, J.; et al. ADAM8 signaling drives neutrophil migration and ARDS severity. JCI Insight 2022, 7, e149870. [Google Scholar] [CrossRef]
- Ohshimo, S.; Ishikawa, N.; Horimasu, Y.; Hattori, N.; Hirohashi, N.; Tanigawa, K.; Kohno, N.; Bonella, F.; Guzman, J.; Costabel, U. Baseline KL-6 predicts increased risk for acute exacerbation of idiopathic pulmonary fibrosis. Respir. Med. 2014, 108, 1031–1039. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, T.; Otsuka, M.; Chiba, H.; Ikeda, K.; Mori, Y.; Umeda, Y.; Nishikiori, H.; Kuronuma, K.; Takahashi, H. Surfactant protein A as a biomarker of outcomes of anti-fibrotic drug therapy in patients with idiopathic pulmonary fibrosis. BMC Pulm. Med. 2020, 20, 27. [Google Scholar] [CrossRef]
- Hasegawa, M.; Fujimoto, M.; Hamaguchi, Y.; Matsushita, T.; Inoue, K.; Sato, S.; Takehara, K. Use of serum Clara cell 16-kDa (CC16) Levels as a potential indicator of active pulmonary fibrosis in systemic sclerosis. J. Rheumatol. 2011, 38, 877–884. [Google Scholar] [CrossRef]
- Rosas, I.O.; Richards, T.J.; Konishi, K.; Zhang, Y.; Gibson, K.; Lokshin, A.E.; Lindell, K.O.; Cisneros, J.; MacDonald, S.D.; Pardo, A.; et al. MMP1 and MMP7 as potential peripheral blood biomarkers in idiopathic pulmonary fibrosis. PLoS Med. 2008, 5, e93. [Google Scholar] [CrossRef]
- Bauer, Y.; White, E.S.; de Bernard, S.; Cornelisse, P.; Leconte, I.; Morganti, A.; Roux, S.; Nayler, O. MMP-7 is a predictive biomarker of disease progression in patients with idiopathic pulmonary fibrosis. ERJ Open Res. 2017, 3, 00074-2016. [Google Scholar] [CrossRef]
- Todd, J.L.; Vinisko, R.; Liu, Y.; Neely, M.L.; Overton, R.; Flaherty, K.R.; Noth, I.; Newby, L.K.; Lasky, J.A.; Olman, M.A.; et al. Circulating matrix metalloproteinases and tissue metalloproteinase inhibitors in patients with idiopathic pulmonary fibrosis in the multicenter IPF-PRO Registry cohort. BMC Pulm. Med. 2020, 20, 64. [Google Scholar] [CrossRef]
- Ge, J.; Tang, L.; Mu, P.; Zhu, F.; Xie, L.; Tang, Y. Association of ADAM17 Expression Levels in Patients with Interstitial Lung Disease. Immunol. Investig. 2020, 49, 134–145. [Google Scholar] [CrossRef]
- Tajiri, M.; Okamoto, M.; Fujimoto, K.; Johkoh, T.; Ono, J.; Tominaga, M.; Azuma, K.; Kawayama, T.; Ohta, S.; Izuhara, K.; et al. Serum level of periostin can predict long-term outcome of idiopathic pulmonary fibrosis. Respir. Investig. 2015, 53, 73–81. [Google Scholar] [CrossRef]
- Fujiwara, A.; Kobayashi, H.; Masuya, M.; Maruyama, M.; Nakamura, S.; Ibata, H.; Fujimoto, H.; Ohnishi, M.; Urawa, M.; Naito, M.; et al. Correlation between circulating fibrocytes, and activity and progression of interstitial lung diseases. Respirology 2012, 17, 693–698. [Google Scholar] [CrossRef]
- Odackal, J.; Yu, V.; Gomez-Manjerres, D.; Field, J.J.; Burdick, M.D.; Mehrad, B. Circulating fibrocytes as prognostic biomarkers of autoimmune interstitial lung disease. ERJ Open Res. 2020, 6, 00481-2020. [Google Scholar] [CrossRef]
- Gui, X.; Qiu, X.; Xie, M.; Tian, Y.; Min, C.; Huang, M.; Hongyan, W.; Chen, T.; Zhang, X.; Chen, J.; et al. Prognostic Value of Serum Osteopontin in Acute Exacerbation of Idiopathic Pulmonary Fibrosis. Biomed. Res. Int. 2020, 2020, 3424208. [Google Scholar] [CrossRef]
- Vadasz, Z.; Balbir Gurman, A.; Meroni, P.; Farge, D.; Levi, Y.; Ingegnoli, F.; Braun-Moscovici, Y.; Rosner, I.; Slobodin, G.; Rozenbaum, M.; et al. Lysyl oxidase-a possible role in systemic sclerosis-associated pulmonary hypertension: A multicentre study. Rheumatol. 2019, 58, 1547–1555. [Google Scholar] [CrossRef]
- Guiot, J.; Bondue, B.; Henket, M.; Corhay, J.L.; Louis, R. Raised serum levels of IGFBP-1 and IGFBP-2 in idiopathic pulmonary fibrosis. BMC Pulm. Med. 2016, 16, 86. [Google Scholar] [CrossRef]
- Guiot, J.; Njock, M.S.; André, B.; Gester, F.; Henket, M.; de Seny, D.; Moermans, C.; Malaise, M.G.; Louis, R. Serum IGFBP-2 in systemic sclerosis as a prognostic factor of lung dysfunction. Sci. Rep. 2021, 11, 10882. [Google Scholar] [CrossRef]
- Silva, E.M.; Mariano, V.S.; Aguiar Pastrez, P.R.; Pinto, M.C.; Castro, A.G.; Syrjanen, K.J.; Longatto-Filho, A. High systemic IL-6 is associated with worse prognosis in patients with non-small cell lung cancer. PLoS ONE 2017, 12, e0181125. [Google Scholar] [CrossRef]
- Adegunsoye, A.; Alqalyoobi, S.; Linderholm, A.; Bowman, W.S.; Lee, C.T.; Pugashetti, J.V.; Sarma, N.; Ma, S.F.; Haczku, A.; Sperling, A.; et al. Circulating Plasma Biomarkers of Survival in Antifibrotic-Treated Patients With Idiopathic Pulmonary Fibrosis. Chest 2020, 158, 1526–1534. [Google Scholar] [CrossRef] [PubMed]
- Bhargava, M.; Wendt, C.H. Biomarkers in acute lung injury. Transl. Res. 2012, 159, 205–217. [Google Scholar] [CrossRef] [PubMed]
- Jalkanen, V.; Yang, R.; Linko, R.; Huhtala, H.; Okkonen, M.; Varpula, T.; Pettilä, V.; Tenhunen, J. SuPAR and PAI-1 in critically ill, mechanically ventilated patients. Intensive Care Med. 2013, 39, 489–496. [Google Scholar] [CrossRef] [PubMed]
- González-López, A.; Albaiceta, G.M. Repair after acute lung injury: Molecular mechanisms and therapeutic opportunities. Crit. Care 2012, 16, 209. [Google Scholar] [CrossRef]
- Singh, K.P.; Lawyer, G.; Muthumalage, T.; Maremanda, K.P.; Khan, N.A.; McDonough, S.R.; Ye, D.; McIntosh, S.; Rahman, I. Systemic biomarkers in electronic cigarette users: Implications for noninvasive assessment of vaping-associated pulmonary injuries. ERJ Open Res. 2019, 5, 00182-2019. [Google Scholar] [CrossRef]
- Zhang, Y.; Tedrow, J.; Nouraie, M.; Li, X.; Chandra, D.; Bon, J.; Kass, D.J.; Fuhrman, C.R.; Leader, J.K.; Duncan, S.R.; et al. Elevated plasma level of Pentraxin 3 is associated with emphysema and mortality in smokers. Thorax 2021, 76, 335–342. [Google Scholar] [CrossRef]
- D’Alonzo, D.; De Fenza, M.; Pavone, V. COVID-19 and pneumonia: A role for the uPA/uPAR system. Drug Discov. Today 2020, 25, 1528–1534. [Google Scholar] [CrossRef]
- Salisbury, M.L.; Xia, M.; Zhou, Y.; Murray, S.; Tayob, N.; Brown, K.K.; Wells, A.U.; Schmidt, S.L.; Martinez, F.J.; Flaherty, K.R. Idiopathic pulmonary fibrosis: Gender-age-physiology index stage for predicting future lung function decline. Chest 2016, 149, 491–498. [Google Scholar] [CrossRef]
- Ray, P.; Manach, Y.L.; Riou, B.; Houle, T.T. Statistical Evaluation of a Biomarker. Anesthesiology 2010, 112, 1023–1040. [Google Scholar] [CrossRef]
- Kohno, N.; Kyoizumi, S.; Awaya, Y.; Fukuhara, H.; Yamakido, M.; Akiyama, M. New serum indicator of interstitial pneumonitis activity. Sialylated carbohydrate antigen KL-6. Chest 1989, 96, 68–73. [Google Scholar] [CrossRef]
- Aloisio, E.; Braga, F.; Puricelli, C.; Panteghini, M. Prognostic role of Krebs von den Lungen-6 (KL-6) measurement in idiopathic pulmonary fibrosis: A systematic review and meta-analysis. Clin. Chem. Lab. Med. 2021, 59, 1400–1408. [Google Scholar] [CrossRef] [PubMed]
- Ishii, H.; Mukae, H.; Kadota, J.; Kaida, H.; Nagata, T.; Abe, K.; Matsukura, S.; Kohno, S. High serum concentrations of surfactant protein A in usual interstitial pneumonia compared with non-specific interstitial pneumonia. Thorax 2003, 58, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Hant, F.N.; Ludwicka-Bradley, A.; Wang, H.J.; Li, N.; Elashoff, R.; Tashkin, D.P.; Silver, R.M. Surfactant protein D and KL-6 as serum biomarkers of interstitial lung disease in patients with scleroderma. J. Rheumatol. 2009, 36, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Katyal, S.L. Clara Cells and Clara Cell 10 kD Protein (CC10). Am. J. Respir. Cell Mol. Biol. 1997, 17, 141–143. [Google Scholar] [CrossRef]
- Olewicz-Gawlik, A.; Trzybulska, D.; Kuznar-Kaminska, B.; Katulska, K.; Danczak-Pazdrowska, A.; Batura-Gabryel, H.; Hrycaj, P. Serum Clara cell 16-kDa protein levels and lung impairment in systemic sclerosis patients. Rev. Bras. Reumatol. 2016, 56, 309–313. [Google Scholar] [CrossRef][Green Version]
- Jessen, H.; Hoyer, N.; Prior, T.S.; Frederiksen, P.; Karsdal, M.A.; Leeming, D.J.; Bendstrup, E.; Sand, J.M.B.; Shaker, S.B. Turnover of type I and III collagen predicts progression of idiopathic pulmonary fibrosis. Respir. Res. 2021, 22, 205. [Google Scholar] [CrossRef]
- Zhong, S.; Khalil, R.A. A Disintegrin and Metalloproteinase (ADAM) and ADAM with thrombospondin motifs (ADAMTS) family in vascular biology and disease. Biochem. Pharmacol. 2019, 164, 188–204. [Google Scholar] [CrossRef]
- Shaffiq, A.; Haitchi, H.M.; Pang, Y.Y.; Alangari, A.A.; Jones, M.G.; Marshall, B.G.; Howarth, P.H.; Davies, D.E.; O’Reilly, K.M.A. A disintegrin and metalloprotease (ADAM) 33 protein in patients with pulmonary sarcoidosis. Respirology 2012, 17, 342–349. [Google Scholar] [CrossRef]
- Conway, S.J.; Izuhara, K.; Kudo, Y.; Litvin, J.; Markwald, R.; Ouyang, G.; Arron, J.R.; Holweg, C.T.J.; Kudo, A. The role of periostin in tissue remodeling across health and disease. Cell. Mol. Life Sci. 2014, 71, 1279–1288. [Google Scholar] [CrossRef]
- Okamoto, M.; Izuhara, K.; Ohta, S.; Ono, J.; Hoshino, T. Ability of Periostin as a New Biomarker of Idiopathic Pulmonary Fibrosis. In Periostin; Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2019; Volume 1132, pp. 79–87. [Google Scholar]
- Vetrone, S.A.; Montecino-Rodriguez, E.; Kudryashova, E.; Kramerova, I.; Hoffman, E.P.; Liu, S.D.; Miceli, M.C.; Spencer, M.J. Osteopontin promotes fibrosis in dystrophic mouse muscle by modulating immune cell subsets and intramuscular TGF-β. J. Clin. Investig. 2009, 119, 1583–1594. [Google Scholar] [CrossRef]
- Lamort, A.S.; Giopanou, I.; Psallidas, I.; Stathopoulos, G.T. Osteopontin as a Link between Inflammation and Cancer: The Thorax in the Spotlight. Cells 2019, 8, 815. [Google Scholar] [CrossRef] [PubMed]
- Chopra, V.; Sangarappillai, R.M.; Romero-Canelón, I.; Jones, A.M. Lysyl Oxidase Like-2 (LOXL2): An Emerging Oncology Target. Adv. Ther. 2020, 3, 1900119. [Google Scholar] [CrossRef]
- Kumar, P.; Smith, T.; Raeman, R.; Chopyk, D.M.; Brink, H.; Liu, Y.; Sulchek, T.; Anania, F.A. Periostin promotes liver fibrogenesis by activating lysyl oxidase in hepatic stellate cells. J. Biol. Chem. 2018, 293, 12781–12792. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.I.; Clemmons, D.R. Insulin-like growth factors and their binding proteins: Biological actions. Endocr. Rev. 1995, 16, 3–34. [Google Scholar] [CrossRef] [PubMed]
- Ruan, W.; Ying, K. Abnormal expression of IGF-binding proteins, an initiating event in idiopathic pulmonary fibrosis? Pathol. Res. Pract. 2010, 206, 537–543. [Google Scholar] [CrossRef]
- Oku, H.; Shimizu, T.; Kawabata, T.; Nagira, M.; Hikita, I.; Ueyama, A.; Matsushima, S.; Torii, M.; Arimura, A. Antifibrotic action of pirfenidone and prednisolone: Different effects on pulmonary cytokines and growth factors in bleomycin-induced murine pulmonary fibrosis. Eur. J. Pharmacol. 2008, 590, 400–408. [Google Scholar] [CrossRef]
- Azuma, A.; Nukiwa, T.; Tsuboi, E.; Suga, M.; Abe, S.; Nakata, K.; Taguchi, Y.; Nagai, S.; Itoh, H.; Ohi, M.; et al. Double-blind, placebo-controlled trial of pirfenidone in patients with idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 2005, 171, 1040–1047. [Google Scholar] [CrossRef]
- King, T.E.; Bradford, W.Z.; Castro-Bernardini, S.; Fagan, E.A.; Glaspole, I.; Glassberg, M.K.; Gorina, E.; Hopkins, P.M.; Kardatzke, D.; Lancaster, L.; et al. A Phase 3 Trial of Pirfenidone in Patients with Idiopathic Pulmonary Fibrosis. N. Engl. J. Med. 2014, 370, 2083–2092. [Google Scholar] [CrossRef]
- Wollin, L.; Maillet, I.; Quesniaux, V.; Holweg, A.; Ryffel, B. Antifibrotic and anti-inflammatory activity of the Tyrosine Kinase inhibitor Nintedanib in Experimental Models of Lung Fibrosiss. J. Pharmacol. Exp. Ther. 2014, 349, 209–220. [Google Scholar] [CrossRef]
- Hilberg, F.; Roth, G.J.; Krssak, M.; Kautschitsch, S.; Sommergruber, W.; Tontsch-Grunt, U.; Garin-Chesa, P.; Bader, G.; Zoephel, A.; Quant, J.; et al. BIBF 1120: Triple angiokinase inhibitor with sustained receptor blockade and good antitumor efficacy. Cancer Res. 2008, 68, 4774–4782. [Google Scholar] [CrossRef]
- Richeldi, L.; Costabel, U.; Selman, M.; Kim, D.S.; Hansell, D.M.; Nicholson, A.G.; Brown, K.K.; Flaherty, K.R.; Noble, P.W.; Raghu, G.; et al. Efficacy of a Tyrosine Kinase Inhibitor in Idiopathic Pulmonary Fibrosis. N. Engl. J. Med. 2011, 365, 1079–1087. [Google Scholar] [CrossRef]
- Richeldi, L.; du Bois, R.M.; Raghu, G.; Azuma, A.; Brown, K.K.; Costabel, U.; Cottin, V.; Flaherty, K.R.; Hansell, D.M.; Inoue, Y.; et al. Efficacy and Safety of Nintedanib in Idiopathic Pulmonary Fibrosis. N. Engl. J. Med. 2014, 370, 2071–2082. [Google Scholar] [CrossRef] [PubMed]
- Pilling, D.; Buckley, C.D.; Salmon, M.; Gomer, R.H. Inhibition of Fibrocyte Differentiation by Serum Amyloid, P.J. Immunol. 2003, 171, 5537–5546. [Google Scholar] [CrossRef]
- Pilling, D.; Roife, D.; Wang, M.; Ronkainen, S.D.; Crawford, J.R.; Travis, E.L.; Gomer, R.H. Reduction of Bleomycin-Induced Pulmonary Fibrosis by Serum Amyloid, P. J. Immunol. 2007, 179, 4035–4044. [Google Scholar] [CrossRef] [PubMed]
- Raghu, G.; Van Den Blink, B.; Hamblin, M.J.; Whitney Brown, A.; Golden, J.A.; Ho, L.A.; Wijsenbeek, M.S.; Vasakova, M.; Pesci, A.; Antin-Ozerkis, D.E.; et al. Effect of recombinant human pentraxin 2 vs placebo on change in forced vital capacity in patients with idiopathic pulmonary fibrosis a randomized clinical trial. JAMA J. Am. Med. Assoc. 2018, 319, 2299–2307. [Google Scholar] [CrossRef] [PubMed]
- Kono, M.; Nakamura, Y.; Suda, T.; Kato, M.; Kaida, Y.; Hashimoto, D.; Inui, N.; Hamada, E.; Miyazaki, O.; Kurashita, S.; et al. Plasma CCN2 (connective tissue growth factor; CTGF) is a potential biomarker in idiopathic pulmonary fibrosis (IPF). Clin. Chim. Acta 2011, 412, 2211–2215. [Google Scholar] [CrossRef] [PubMed]
- Bonniaud, P.; Martin, G.; Margetts, P.J.; Ask, K.; Robertson, J.; Gauldie, J.; Kolb, M. Connective tissue growth factor is crucial to inducing a profibrotic environment in “fibrosis-resistant” Balb/c mouse lungs. Am. J. Respir. Cell Mol. Biol. 2004, 31, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wu, G.; Gou, L.; Liu, Z.; Wang, X.; Fan, X.; Wu, L.; Liu, N. A novel single-chain-Fv antibody against connective tissue growth factor attenuates bleomycin-induced pulmonary fibrosis in mice. Respirology 2011, 16, 500–507. [Google Scholar] [CrossRef]
- Gorina, E.; Richeldi, L.; Raghu, G.; Fernandez Perez, E.; Costabel, U.; Albera, C.; Lederer, D.; Flaherty, K.; Ettinger, N.; Bercz, P.; et al. PRAISE, a randomized, placebo-controlled, double-blind Phase 2 clinical trial of pamrevlumab (FG-3019) in IPF patients. Eur. Respir. J. 2017, 50, OA3400. [Google Scholar] [CrossRef]
- Gagnon, L.; Leduc, M.; Thibodeau, J.F.; Zhang, M.Z.; Grouix, B.; Sarra-Bournet, F.; Gagnon, W.; Hince, K.; Tremblay, M.; Geerts, L.; et al. A Newly Discovered Antifibrotic Pathway Regulated by Two Fatty Acid Receptors: GPR40 and GPR84. Am. J. Pathol. 2018, 188, 1132–1148. [Google Scholar] [CrossRef]
- Kolilekas, L.; Papiris, S.; Bouros, D. Existing and emerging treatments for idiopathic pulmonary fibrosis. Expert Rev. Respir. Med. 2019, 13, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Aoki, J.; Inoue, A.; Okudaira, S. Two pathways for lysophosphatidic acid production. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2008, 1781, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Montesi, S.B.; Mathai, S.K.; Brenner, L.N.; Gorshkova, I.A.; Berdyshev, E.V.; Tager, A.M.; Shea, B.S. Docosatetraenoyl LPA is elevated in exhaled breath condensate in idiopathic pulmonary fibrosis. BMC Pulm. Med. 2014, 14, 5. [Google Scholar] [CrossRef] [PubMed]
- Maher, T.M.; van der Aar, E.M.; Van de Steen, O.; Allamassey, L.; Desrivot, J.; Dupont, S.; Fagard, L.; Ford, P.; Fieuw, A.; Wuyts, W. Safety, tolerability, pharmacokinetics, and pharmacodynamics of GLPG1690, a novel autotaxin inhibitor, to treat idiopathic pulmonary fibrosis (FLORA): A phase 2a randomised placebo-controlled trial. Lancet Respir. Med. 2018, 6, 627–635. [Google Scholar] [CrossRef]
- Prelich, G. Gene overexpression: Uses, mechanisms, and interpretation. Genetics 2012, 190, 841–854. [Google Scholar] [CrossRef]
- Ruigrok, M.J.R.; Frijlink, H.W.; Melgert, B.N.; Olinga, P.; Hinrichs, W.L.J. Gene therapy strategies for idiopathic pulmonary fibrosis: Recent advances, current challenges, and future directions. Mol. Ther. Methods Clin. Dev. 2021, 20, 483–496. [Google Scholar] [CrossRef]
- Epperly, M.W.; Bray, J.A.; Krager, S.; Berry, L.M.; Gooding, W.; Engelhardt, J.F.; Zwacka, R.; Travis, E.L.; Greenberger, J.S. Intratracheal injection of adenovirus containing the human MnSOD transgene protects athymic nude mice from irradiation-induced organizing alveolitis. Int. J. Radiat. Oncol. Biol. Phys. 1999, 43, 169–181. [Google Scholar] [CrossRef]
- Epperly, M.W.; Travis, E.L.; Sikora, C.; Greenberger, J.S. Magnesium superoxide dismutase (MnSOD) plasmid/liposome pulmonary radioprotective gene therapy: Modulation of irradiation-induced mRNA for IL-I, TNF-α, and TGF-β correlates with delay of organizing alveolitis/fibrosis. Biol. Blood Marrow Transplant. 1999, 5, 204–214. [Google Scholar] [CrossRef]
- Nakao, A.; Fujii, M.; Matsumura, R.; Kumano, K.; Saito, Y.; Miyazono, K.; Iwamoto, I. Transient gene transfer and expression of Smad7 prevents bleomycin- induced lung fibrosis in mice. J. Clin. Investig. 1999, 104, 5–11. [Google Scholar] [CrossRef]
- de Ceuninck van Capelle, C.; Spit, M.; ten Dijke, P. Current perspectives on inhibitory SMAD7 in health and disease. Crit. Rev. Biochem. Mol. Biol. 2020, 55, 691–715. [Google Scholar] [CrossRef]
- Sisson, T.H.; Hattori, N.; Xu, Y.; Simon, R.H. Treatment of bleomycin-induced pulmonary fibrosis by transfer of urokinase-type plasminogen activator genes. Hum. Gene Ther. 1999, 10, 2315–2323. [Google Scholar] [CrossRef]
- Povedano, J.M.; Martinez, P.; Serrano, R.; Tejera, Á.; Gómez-López, G.; Bobadilla, M.; Flores, J.M.; Bosch, F.; Blasco, M.A. Therapeutic effects of telomerase in mice with pulmonary fibrosis induced by damage to the lungs and short telomeres. eLife 2018, 7, 31299. [Google Scholar] [CrossRef]
- Fichtner-Feigl, S.; Strober, W.; Kawakami, K.; Puri, R.K.; Kitani, A. IL-13 signaling through the IL-13α2 receptor is involved in induction of TGF-β1 production and fibrosis. Nat. Med. 2006, 12, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Ji, Q.; Ji, J.; Ju, S.; Xu, C.; Yong, X.; Xu, X.; Muddassir, M.; Chen, X.; Xie, J.; et al. Co-delivery of siPTPN13 and siNOX4 via (myo)fibroblasttargeting polymeric micelles for idiopathic pulmonary fibrosis therapy. Theranostics 2021, 11, 3244–3261. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Shi, W.; Wang, Y.L.; Chen, H.; Bringas, P.; Datto, M.B.; Frederick, J.P.; Wang, X.F.; Warburton, D. Smad3 deficiency attenuates bleomycin-induced pulmonary fibrosis in mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 2002, 282, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.-S.; Hu, X.-B.; Liu, W.; Sun, Y.; Liu, Z. Effects of RNA interference-induced Smad3 gene silencing on pulmonary fibrosis caused by paraquat in mice. Exp. Biol. Med. 2012, 237, 548–555. [Google Scholar] [CrossRef]
- Garbuzenko, O.B.; Ivanova, V.; Kholodovych, V.; Reimer, D.C.; Reuhl, K.R.; Yurkow, E.; Adler, D.; Minko, T. Combinatorial treatment of idiopathic pulmonary fibrosis using nanoparticles with prostaglandin E and siRNA(s). Nanomed. Nanotechnol. Biol. Med. 2017, 13, 1983–1992. [Google Scholar] [CrossRef]
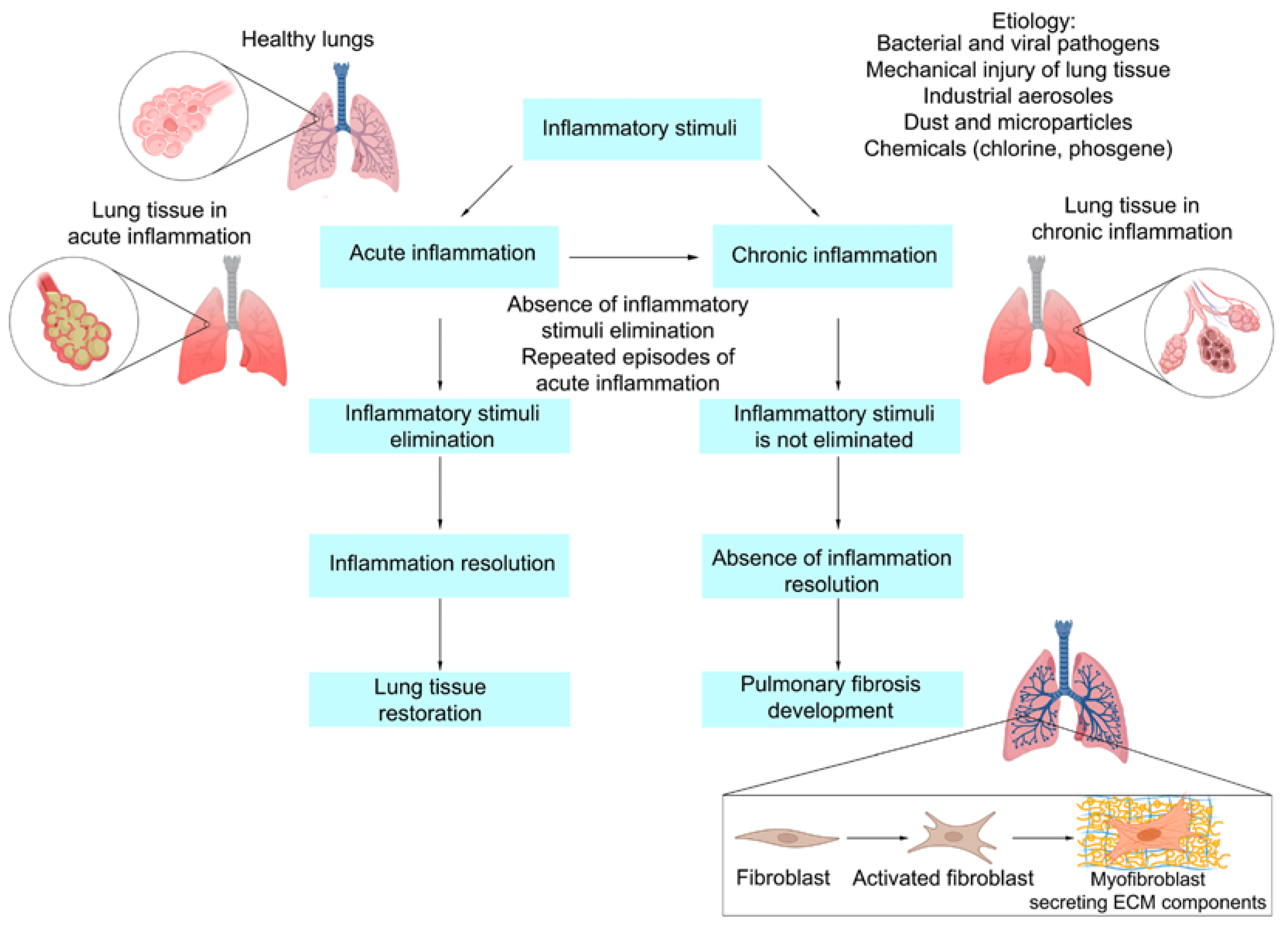
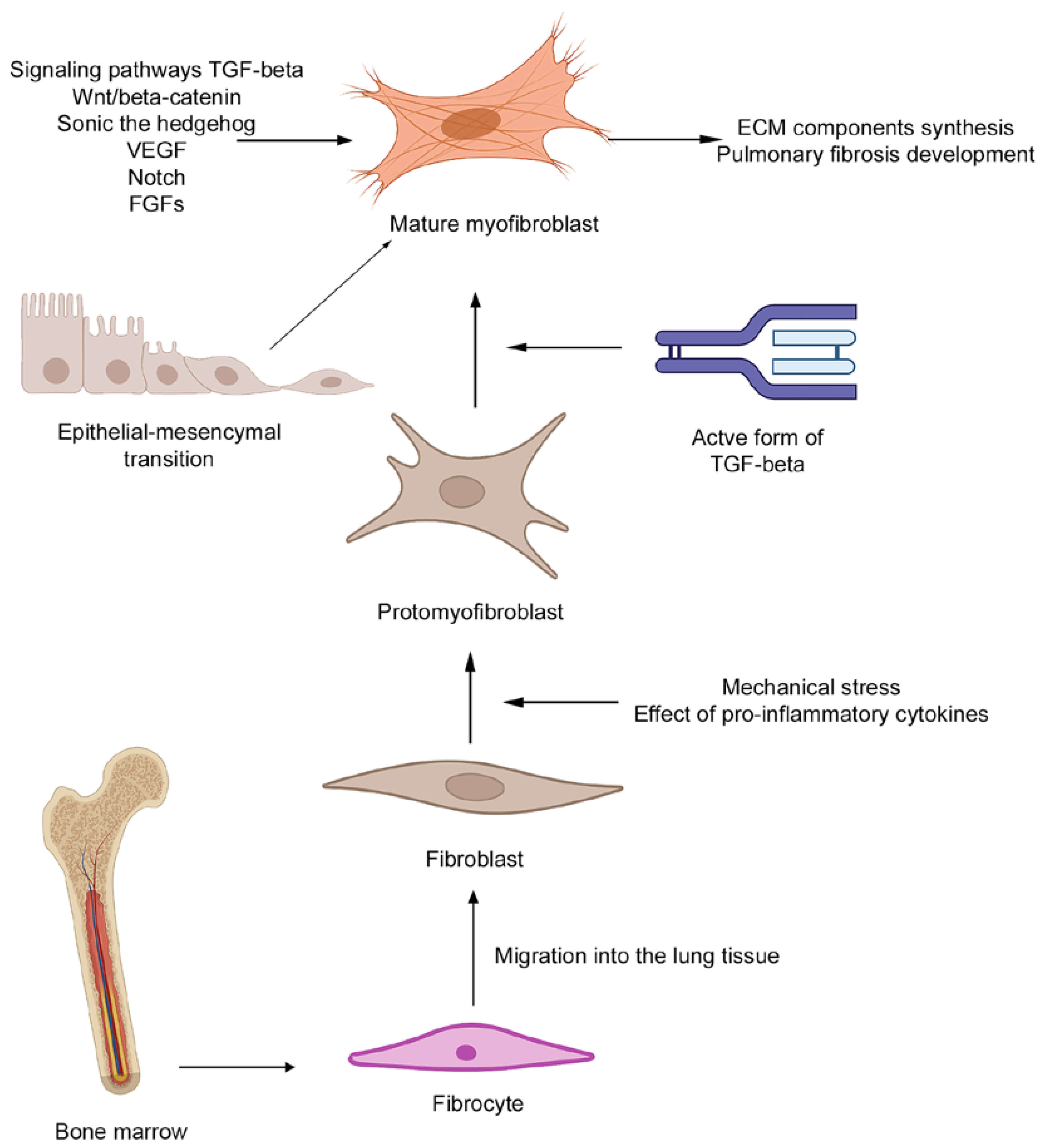
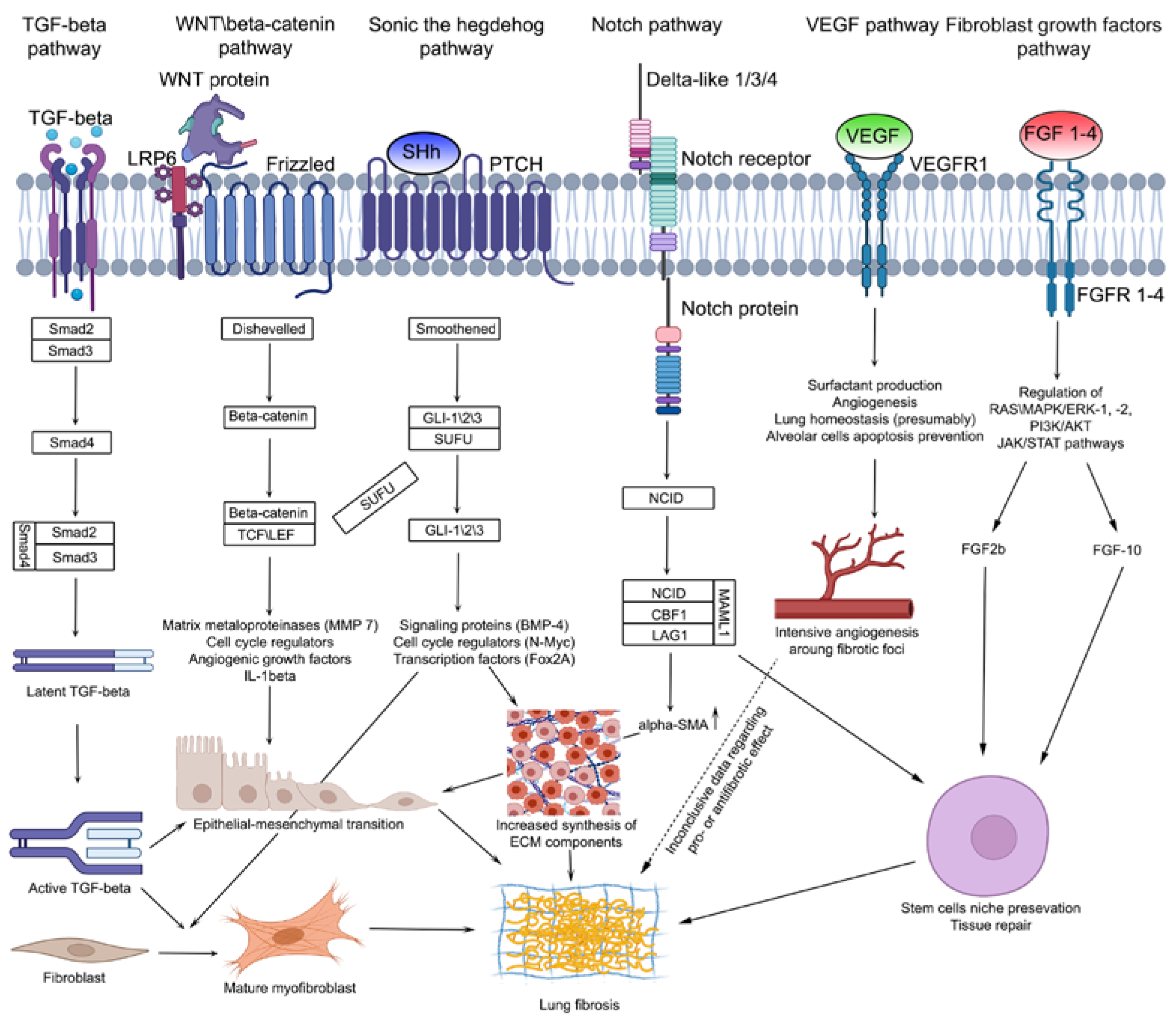
| Model | Histological Characteristics | Modeling Object | Advantages and Disadvantages | References |
|---|---|---|---|---|
| Lipopolysaccharide (LPS)-induced acute lung injury Inducing agent—LPS | Neutrophil-associated inflammatory infiltration with the admixture of lymphocytes and macrophages, circulatory disruption, microvascular thrombosis, hemorrhages, interstitial and alveolar edemas | Inflammatory response similar to bacterial infection | +Multiplicity of administration routes (intraperitoneal, intranasal, intratracheal) +Reproducibility −Variability of inflammation severity −Dependence on LPS quality/purity/bacteria type | [290,291,292,293,294,295,296] |
| Hyperoxia-induced acute lung injury Inducing agent—100% oxygen | Thickening of alveolar walls, alveolar and interstitial edemas, hemorrhages and inflammatory infiltration | Exudative and proliferative phases of ALI/ARDS; lung restoration after injury | +Reproducibility +Clinical relevance regarding ICU patients receiving oxygen support or mechanical ventilation −Relevance regarding healthy lungs | [297,298,299,300] |
| Oleic acid-induced acute lung injury Inducing agent—Cis-9-octadecenoic acid | Early necrotic foci and microvascular thrombosis followed by AEC II proliferation and connective tissue growth in sub-pleural areas; polymorphism of damaged sites in the lung tissue | Morphological changes in the lung tissue caused by fat embolism of pulmonary vessels caused by severe trauma or bone fractures | +Reproducibility +Reflection of reversible ALI phases −Dissolution in ethyl alcohol −Few cases of ALI caused by fat embolism in humans | [301,302,303,304] |
| Acid aspiration-induced acute lung injury Inducing agent—hydrochloric acid | Neutrophil inflammatory infiltration and alveolar hemorrhages followed by fibroproliferative response; interstitial and alveolar edemas | Morphological changes in the lung tissue caused by gastric contents aspiration; neutrophil migration mechanisms in ALI/ARDS | +Multiple altered parameters in the lungs +Combination with other ALI induction methods −Narrow induction dose of hydrochloric acid −Clinical relevance due to complex mixture of gastric contents in humans | [305,306,307,308,309] |
| Mechanical ventilation-induced acute lung injury Inducing agent—mechanical ventilation | Interstitial edema, infiltration of alveolar walls by mononuclear cells, hemorrhages and fibrin deposition | Mechanical ventilation strategies in critical ALI patients support; lung biomechanics during ALI/ARDS development | +Clinical relevance +Combination with other lung damaging factors −Complexity/special equipment | [310,311,312,313,314,315,316] |
| Model | Histological Characteristics | Modeling Object | Advantages and Disadvantages | References |
|---|---|---|---|---|
| Bleomycin-induced pulmonary fibrosis Inducing agent—bleomycin | Mononuclear infiltration of lung tissue, thickening of alveolar walls, congestion of blood vessels, destruction of alveolar epithelium and deposition of newly synthesized collagen fibers in alveolar walls and around blood vessels and bronchi | Mechanisms of pulmonary fibrosis development after inflammation/ARDS; estimation of anti-fibrotic therapy efficacy | +Multiplicity of administration routes (intranasal, intratracheal, intraperitoneal, subcutaneous, intravenous, via inhalations) +Simplicity of induction +Reproducibility/standardization of effects +Clinical relevance regarding ARDS −Self-limiting character of fibrosis −Relevance of induction route regarding human fibrosis | [317,318,319,320,321] |
| Radiation-induced pulmonary fibrosis Inducing agent—ionizing radiation | Subpleural fibrotic foci, increased collagen deposition in collapsed alveoli and inflammatory infiltration of surrounding fibrotic tissue | Radiation-triggered pulmonary fibrosis; lung vessels remodeling during pulmonary hypertension | +Clinical relevance −Long duration −High coast (ionizing radiation source, personal protection) | [99,322,323,324,325,326,327] |
| Silica particle-induced pulmonary fibrosis Inducing agent—silica particles | Connective tissue growth and formation of fibrotic nodes around silica particles, low inflammation intensity | Silica-associated nodular pulmonary fibrosis, silicosis and silica fibrosis | +Multiplicity of administration routes (aerosol inhalation, intratracheal or oropharyngeal instillation) +Constant fibrotic response +Possibility of long-term investigations −Dependence of fibrosis formation on administration route (intratracheal instillation—two to four weeks, aerosol inhalation—one to three months) −Low reproducibility −Low clinical relevance −Absence of some pulmonary fibrosis characteristics (local heterogeneity, hyperplastic changes in alveolar epithelium) | [328,329,330,331,332] |
| Fluorescein isothiocyanate (FITC)-induced lung fibrosis Inducing agent—FITC | Infiltration of lung tissue with mononuclear cells and neutrophils, edema and epithelial cell hyperplasia, culminating in fibrosis development | Fibrosis detection; investigation of anti-fibrotic drugs regarding already formed fibrosis | +Usage of different mouse lines +Constant fibrotic response +Possibility of long-term investigations +Non-self-limiting character −Dependence on FITC quality and size of particles −Narrow difference between effective and toxic doses −Clinical relevance | [332,333,334,335] |
| Lung Pathology | Marker | Source | Design of the Study | Proposed Usage in Clinical Setting | References |
|---|---|---|---|---|---|
| ALI (exudative phase) | IL-6 | Serum, BALF | Murine model; single-center prospective cohort studies | Increased concentration is associated with early exacerbations of pulmonary fibrosis, fatal SARS-CoV-2-induced pneumonia and higher chance of fatal ALI | [345,346,347,348] |
| CXCL13 | Serum and plasma | Single center prospective cohort studies | Increased concentration is a prognostic marker of SARS-CoV-2-induced ALI mortality, admission to intensive care unit (ICU) and ICU mortality | [349,350] | |
| IL-8 | Plasma, BALF | Single and multiple-center randomized clinical trial | Increased concentration is a marker of severity and increased mortality in ALI patients | [351,352] | |
| IL-18 | Peripheral blood, plasma, lung tissue | Murine model; single-center randomized study | Increased concentration indicates morbidity, increased severity and ICU mortality of ALI patients | [353,354] | |
| IL-1β | BALF | Single-center observational study | Increased concentration correlates with high ICU mortality | [348] | |
| Tumor necrosis factor α | Serum, BALF | Single-center prospective study | Increased concentration indicates high ICU mortality | [348] | |
| Surfactant proteins A and D | Serum | Single-center observational study | Increased concentration is a prognostic marker of early severe course of SARS-CoV-2-induced ALI | [355] | |
| Krebs von den Lungen-6 | Serum | Literature meta-analysis | Increased concentration predicts severe COVID-19 stratification | [356] | |
| Vascular endothelial growth factor | BAL fluid | Single-center observational and retrospective studies | Increased concentration is a prognostic marker of resolving ALI | [357,358] | |
| Keratinocyte growth factor | BAL fluid | Single-center prospective study | Increased concentration is a prognostic marker of severity and poor outcome of ALI | [359] | |
| Plasminogen activator inhibitor | Plasma | Prospective multi-center observational study | Increased concentration is a predictor of ALI severity | [360] | |
| Thrombomodulin | Plasma | Prospective multi-center observational study | Increased concentration predicts increased ALI mortality in first 90 days after invasive mechanical ventilation | [361] | |
| ALI (proliferative phase) | Hepatocyte growth factor | BAL fluid | Single-center cohort study | Increased concentration is a prognostic marker of ALI development | [362] |
| Pentraxin-3 | Peripheral blood mononuclear cells | Single-center observational study | Increased concentration is a prognostic marker of short term mortality in COVID-19 | [363] | |
| Tissue inhibitor of metalloproteinase-1 | Serum | Multi-center observational study | Increased concentration is associated with worse outcome in mechanically ventilated ALI patients | [364] | |
| Tenascin-C | BAL fluid | Single-center observational study | Increased concentration is a prognostic marker of severe SARS-CoV-2 induced ALI | [365] | |
| Matrix metalloproteinase-8 | BALF | Single-center observational and prospective study | Increased concentration in BALF is a prognostic marker of fatal ALI | [366] | |
| Urokinase plasminogen activator | Plasma | Multi-center observational prospective study | Increased concentration in plasma is diagnostic and prognostic marker of mechanically ventilated ALI patients | [367] | |
| A disintegrin and metalloproteinase-8 | BAL fluid | Mice model; single-center observational study | Increased concentration is a prognostic marker of ALI onset and severity | [368] | |
| Pulmonary fibrosis | Krebs von den Lungen-6 | Serum | Single-center prospective study | Increased concentration is a prognostic factor of acute exacerbation in pulmonary fibrosis | [369] |
| Surfactant protein A | Serum | Single-center observational study | Decreased concentration is a prognostic marker of anti-fibrotic therapy effectiveness | [370] | |
| Clara cell protein 16 | Serum | Single-center retrospective longitudinal study | Increased concentration is a prognostic marker of active pulmonary fibrosis in systemic sclerosis patients | [371] | |
| Matrix metalloproteinase 1 | Peripheral blood, lung tissue | Single-center observational study | Increased peripheral blood concentration is a diagnostic marker of pulmonary fibrosis, differentiating it from other ILDs. | [372] | |
| Matrix metalloproteinase 7 | Serum | Multi-center, prospective, randomized, double-blind, placebo-controlled trial | Increased serum concentration is a prognostic marker of high risk of worsening and decline of lung functions | [373] | |
| Matrix metalloproteinase-9 | Serum | Multi-center observational study | Increased concentration is a prognostic marker of severe course and worse outcome of pulmonary fibrosis | [374] | |
| A disintegrin and metalloproteinase-17 | Peripheral blood mononuclear cells | Single-center observational study | Increased expression is associated with more active disease development and severity | [375] | |
| Periostin | Serum | Single-center retrospective study | Increased concentration is a prognostic marker of increased mortality in pulmonary fibrosis patients | [376] | |
| Circulating fibrocytes | Serum | Single-center observational study | Increased concentration is a prognostic marker of pulmonary fibrosis activity and increased mortality | [377,378] | |
| Osteopontin | Serum | Single-center observational study | Increased concentration is a prognostic marker of exacerbation of pulmonary fibrosis | [379] | |
| Lysyl oxidase-like 2 | Serum | Single-center observational study | Increased concentration is a prognostic marker of pulmonary hypertension and worse disease outcome in pulmonary fibrosis | [380] | |
| Insulin-like growth factor binding proteins | Serum | Single-center observational studies | Increased concentration is associated with worse disease outcome of systemic sclerosis-associated pulmonary fibrosis | [381,382] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savin, I.A.; Zenkova, M.A.; Sen’kova, A.V. Pulmonary Fibrosis as a Result of Acute Lung Inflammation: Molecular Mechanisms, Relevant In Vivo Models, Prognostic and Therapeutic Approaches. Int. J. Mol. Sci. 2022, 23, 14959. https://doi.org/10.3390/ijms232314959
Savin IA, Zenkova MA, Sen’kova AV. Pulmonary Fibrosis as a Result of Acute Lung Inflammation: Molecular Mechanisms, Relevant In Vivo Models, Prognostic and Therapeutic Approaches. International Journal of Molecular Sciences. 2022; 23(23):14959. https://doi.org/10.3390/ijms232314959
Chicago/Turabian StyleSavin, Innokenty A., Marina A. Zenkova, and Aleksandra V. Sen’kova. 2022. "Pulmonary Fibrosis as a Result of Acute Lung Inflammation: Molecular Mechanisms, Relevant In Vivo Models, Prognostic and Therapeutic Approaches" International Journal of Molecular Sciences 23, no. 23: 14959. https://doi.org/10.3390/ijms232314959
APA StyleSavin, I. A., Zenkova, M. A., & Sen’kova, A. V. (2022). Pulmonary Fibrosis as a Result of Acute Lung Inflammation: Molecular Mechanisms, Relevant In Vivo Models, Prognostic and Therapeutic Approaches. International Journal of Molecular Sciences, 23(23), 14959. https://doi.org/10.3390/ijms232314959





