Ratiometric Zinc Biosensor Based on Bioluminescence Resonance Energy Transfer: Trace Metal Ion Determination with Tunable Response
Abstract
1. Introduction
2. Results
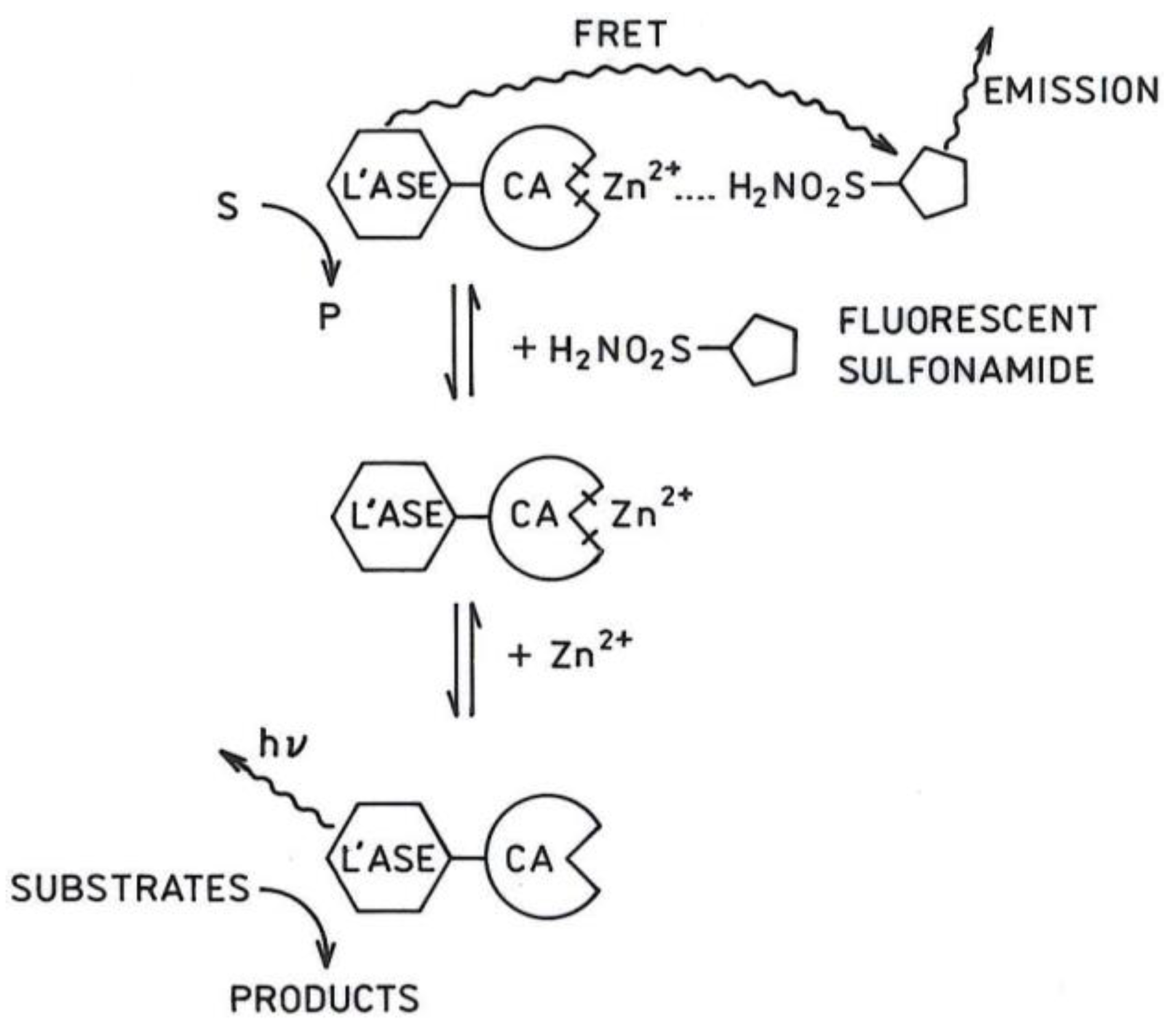
2.1. Principle of the Sensor
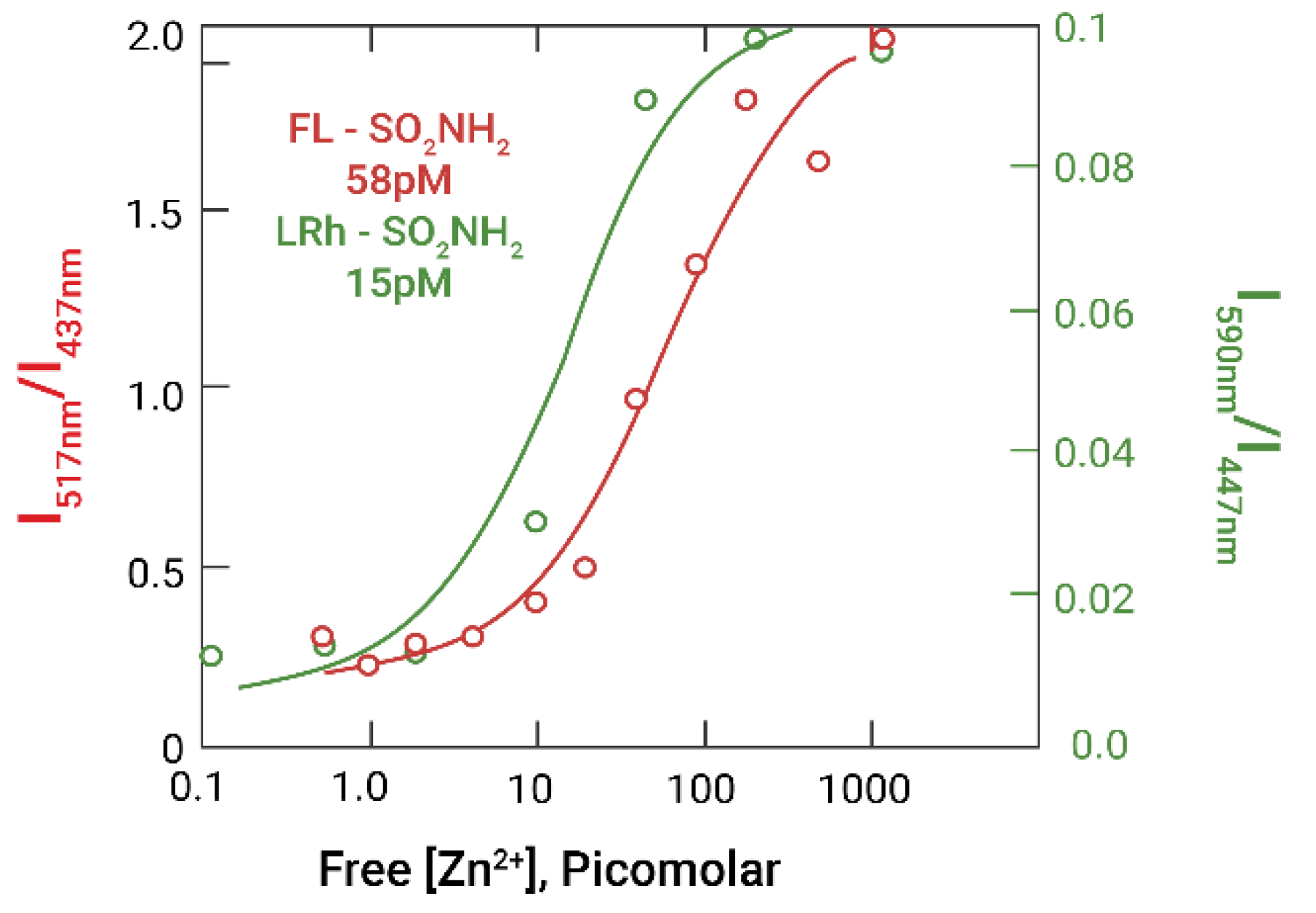
2.2. Experimental Results
3. Discussion
4. Materials and Methods
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Krezel, A.; Maret, W. Zinc-buffering capacity of a eukaryotic cell at physiological pZn. J. Biol. Inorg. Chem. 2006, 11, 1049–1062. [Google Scholar] [CrossRef]
- Stumm, W.; Morgan, J.J. Aquatic Chemistry: Chemical Equilibria and Rates in Natural Waters, 3rd ed.; Wiley-Interscience: New York, NY, USA, 1996. [Google Scholar]
- Sokalski, T.; Ceresa, A.; Zwickl, A.T.; Pretsch, E. Large Improvement of the Lower Detection Limit of Ion-Selective Polymer Membrane Electrodes. J. Am. Chem. Soc. 1997, 119, 11347–11348. [Google Scholar] [CrossRef]
- Sherrell, R.M.; Boyle, E.A. Zinc, chromium, vanadium, and iron in the Mediterranenan Sea. Deep. Sea Res. 1988, 35, 1319–1334. [Google Scholar] [CrossRef]
- Donat, J.R.; Bruland, K.W. A comparison of two voltammetric techniques for determining zinc speciation in Northeast Pacific Ocean waters. Mar. Chem. 1990, 28, 301–323. [Google Scholar] [CrossRef]
- Frederickson, C.J.; Kasarskis, E.J.; Ringo, D.; Frederickson, R.E. A quinoline fluorescence method for visualizing and assaying the histochemically reactive zinc (bouton zinc) in the brain. J. Neurosci. Methods 1987, 20, 91–103. [Google Scholar] [CrossRef]
- Budde, T.; Minta, A.; White, J.; Kay, A. Imaging free zinc in synaptic terminals in live hippocampal slices. Neuroscience 1997, 79, 347–358. [Google Scholar] [CrossRef] [PubMed]
- Burdette, S.C.; Walkup, G.K.; Spingler, B.; Tsien, R.Y.; Lippard, S.J. Fluorescent sensors for Zn2+ based on a fluorescein platform: Synthesis, properties and intracellular distribution. J. Am. Chem. Soc. 2001, 123, 7831–7841. [Google Scholar] [CrossRef]
- Gee, K.; Zhou, Z.-L.; Ton-That, D.; Sensi, S.; Weiss, J. Measuring zinc in living cells.: A new generation of sensitive and selective fluorescent probes. Cell Calcium 2002, 31, 245–251. [Google Scholar] [CrossRef]
- Maruyama, S.; Kikuchi, K.; Hirano, T.; Urano, Y.; Nagano, T. A Novel, Cell-Permeable, Fluorescent Probe for Ratiometric Imaging of Zinc Ion. J. Am. Chem. Soc. 2002, 124, 10650. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Guo, Z. Fluorescent detection of zinc in biological systems: Recent development on the design of chemosensors and biosensors. Co ord. Chem. Rev. 2004, 248, 205–229. [Google Scholar] [CrossRef]
- Lau, Y.H.; Rutledge, P.J.; Watkinson, M.; Todd, M.H. Chemical sensors that incorporate click-derived triazoles. Chem. Soc. Rev. 2011, 40, 2848–2866. [Google Scholar] [CrossRef]
- Carter, K.P.; Young, A.M.; Palmer, A.E. Fluorescent Sensors for Measuring Metal Ions in Living Systems. Chem. Rev. 2014, 114, 4564–4601. [Google Scholar] [CrossRef]
- Thompson, R.B.; Jones, E.R. Enzyme-based fiber optic zinc biosensor. Anal. Chem. 1993, 65, 730–734. [Google Scholar] [CrossRef]
- Godwin, H.A.; Berg, J.M. A Fluorescent Zinc Probe Based on Metal-Induced Peptide Folding. J. Am. Chem. Soc. 1996, 118, 6514–6515. [Google Scholar] [CrossRef]
- Pearce, L.L.; Gandley, R.E.; Han, W.; Wasserloos, K.; Stitt, M.; Kanai, A.J.; McLaughlin, M.K.; Pitt, B.R.; Levitan, E.S. Role of metallothionein in nitric oxide signaling as revealed by a green fluorescent fusion protein. Proc. Natl. Acad. Sci. USA 2000, 97, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.-H.; Maret, W. A fluorescence resonance energy transfer sensor for the beta-domain of metallothionein. Proc. Natl. Acad. Sci. USA 2003, 100, 2255–2260. [Google Scholar] [CrossRef]
- van Dongen, E.M.W.M.; Evers, T.H.; Dekkers, L.M.; Meijer, E.W.; Klomp, A.L.W.J.; Merkx, M. Variation of Linker Length in Ratiometric Fluorescent Sensor Proteins Allows Rational Tuning of Zn(II) Affinity in the Picomolar to Femtomolar Range. J. Am. Chem. Soc. 2007, 129, 3494–3495. [Google Scholar] [CrossRef] [PubMed]
- Dittmer, P.; Miranda, J.G.; Gorski, J.A.; Palmer, A.E. Genetically Encoded Sensors to Elucidate Spatial Distribution of Cellular Zinc. J. Biol. Chem. 2009, 284, 16289–16297. [Google Scholar] [CrossRef]
- Wang, D.; Hurst, T.K.; Thompson, R.B.; Fierke, C.A. Genetically encoded ratiometric biosensors to measure extracellular exchangeable zinc in Escherichia coli. J. Biomed. Opt. 2011, 16, 087011-1–087011-11. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.B.; Fierke, C.A. Enzymes as Sensors, 1st ed.; Elsevier: Amsterdam, The Netherlands; Academic Press: Cambridge, MA, USA, 2017; Volume 589, pp. xviii + 511. [Google Scholar]
- Thompson, R.B.; Fierke, C.A. Measuring and Imaging Metal Ions with Fluorescence-Based Biosensors: Speciation, Selectivity, Kinetics, and Other Issues. In Enzymes as Sensors; Thompson, R.B., Fierke, C.A., Eds.; Elsevier: Amsterdam, The Netherlands; Academic Press: Cambridge, MA, USA, 2017; Volume 589, pp. 281–299. [Google Scholar]
- De Acha, N.; Elosúa, C.; Corres, J.M.; Arregui, F.J. Fluorescent Sensors for the Detection of Heavy Metal Ions in Aqueous Media. Sensors 2019, 19, 599. [Google Scholar] [CrossRef]
- Malmstadt, H.V.; Enke, C.G.; Crouch, S.R. Electronic Measurements for Scientists; W.A. Benjamin, Inc.: Menlo Park, CA, USA, 1974; p. 906. [Google Scholar]
- Buser, J.R.; Holstein, C.A.; Yager, P.A. Microfluidic Diagnostics for Low-Resource Settings: Improving Global Health without a Power Cord. In Microfluidics for Medicial Applications; Berg, A.v.d., Segerink, L., Eds.; Royal Society of Chemistry: London, UK, 2015; pp. 151–190. [Google Scholar] [CrossRef]
- Ntziachristos, V. Fluorescence molecular imaging. Annu. Rev. Biomed. Eng. 2006, 8, 1–33. [Google Scholar] [CrossRef] [PubMed]
- Negrin, R.S.; Contag, C.H. In vivo imaging using bioluminescence: A tool for probing graft vs. host disease. Nat. Rev. Immunol. 2006, 6, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Deluca, M.; McElroy, W.D. Purification and properties of firefly luciferase. In Bioluminescence and Chemiluminescence; DeLuca, M., Ed.; Academic Press: Cambridge, MA, USA, 1978; Volume 57, pp. 3–15. [Google Scholar]
- Fernandez-Gutierrez, A.; de la Pena, A.M. Determinations of inorganic substances by luminescence methods. In Molecular Luminescence Spectroscopy, Part I: Methods and Applications; Schulman, S.G., Ed.; Wiley-Interscience: New York, NY, USA, 1985; Volume 77, pp. 371–546. [Google Scholar]
- Ziegler, M.M.; Baldwin, T.O. Bioluminescence and Chemiluminescence: Part C; Academic Press: Cambridge, MA, USA, 2000. [Google Scholar]
- Fierke, C.A.; Thompson, R.B. Fluorescence-based biosensing of zinc using carbonic anhydrase. BioMetals 2001, 14, 205–222. [Google Scholar] [CrossRef]
- Hurst, T.K.; Wang, D.; Thompson, R.B.; Fierke, C.A. Carbonic anhydrase II-based metal ion sensing: Advances and new perspectives. Biochim. et Biophys. Acta (BBA) Proteins Proteom. 2010, 1804, 393–403. [Google Scholar] [CrossRef] [PubMed]
- McCall, K.A. Metal Ion Specificity and Avidity in Carbonic Anhydrase Variants. Ph.D. Thesis, Duke University, Durham, NC, USA, 2000. [Google Scholar]
- Hunt, J.A.; Ahmed, M.; Fierke, C.A. Metal binding specificity in carbonic anhydrase is influenced by conserved hydrophobic amino acids. Biochemistry 1999, 38, 9054–9060. [Google Scholar] [CrossRef] [PubMed]
- McCall, K.A.; Fierke, C.A. Probing Determinants of the Metal Ion Selectivity in Carbonic Anhydrase Using Mutagenesis. Biochemistry 2004, 43, 3979–3986. [Google Scholar] [CrossRef]
- Zeng, H.-H.; Thompson, R.B.; Maliwal, B.P.; Fones, G.R.; Moffett, J.W.; Fierke, C.A. Real-Time Determination of Picomolar Free Cu(II) in Seawater Using a Fluorescence-Based Fiber Optic Biosensor. Anal. Chem. 2003, 75, 6807–6812. [Google Scholar] [CrossRef] [PubMed]
- Bozym, R.A.; Thompson, R.B.; Stoddard, A.K.; Fierke, C.A. Measuring Picomolar Intracellular Exchangeable Zinc in PC-12 Cells Using a Ratiometric Fluorescence Biosensor. ACS Chem. Biol. 2006, 1, 103–111. [Google Scholar] [CrossRef] [PubMed]
- McCranor, B.J.; Szmacinski, H.; Zeng, H.H.; Stoddard, A.K.; Hurst, T.; Fierke, C.A.; Lakowicz, J.R.; Thompson, R.B. Fluorescence lifetime imaging of physiological free Cu(ii) levels in live cells with a Cu(ii)-selective carbonic anhydrase-based biosensor. Metallomics 2014, 6, 1034–1042. [Google Scholar] [CrossRef][Green Version]
- Huang, C.-C.; Lesburg, C.A.; Kiefer, L.L.; Fierke, C.A.; Christianson, D.W. Reversal of the Hydrogen Bond to Zinc Ligand Histidine-119 Dramatically Diminishes Catalysis and Enhances Metal Equilibration Kinetics in Carbonic Anhydrase II. Biochemistry 1996, 35, 3439–3446. [Google Scholar] [CrossRef]
- Grynkiewicz, G.; Poenie, M.; Tsien, R.Y. A new generation of calcium indicators with greatly improved fluorescence properties. J. Biol. Chem. 1985, 260, 3440–3450. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.B.; Maliwal, B.P.; Fierke, C.A. Expanded Dynamic Range of Free Zinc Ion Determination by Fluorescence Anisotropy. Anal. Chem. 1998, 70, 1749–1754. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.; Patchan, M. Lifetime-Based Fluorescence Energy Transfer Biosensing of Zinc. Anal. Biochem. 1995, 227, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.B.; Cramer, M.L.; Bozym, R.; Fierke, C.A. Excitation ratiometric fluorescent biosensor for zinc ion at picomolar levels. J. Biomed. Opt. 2002, 7, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Maren, T.H. Use of inhibitors in physiological studies of carbonic anhydrase. Am. J. Physiol. 1977, 232, F291–F297. [Google Scholar] [CrossRef] [PubMed]
- Hall, M.P.; Unch, J.; Binkowski, B.F.; Valley, M.P.; Butler, B.L.; Wood, M.G.; Otto, P.; Zimmerman, K.; Vidugiris, G.; Machleidt, T.; et al. Engineered Luciferase Reporter from a Deep Sea Shrimp Utilizing a Novel Imidazopyrazinone Substrate. ACS Chem. Biol. 2012, 7, 1848–1857. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.B.; Matveeva, E.G.; Fierke, C.A.; Bourne, L.; Franke, G. Bioluminescent Metal Ion Assay. U.S. Patent 9193990 B2, 24 November 2015. [Google Scholar]
- Forster, T. Intermolecular energy migration and fluorescence (Ger.). Ann. Der Phys. 1948, 2, 55–75. [Google Scholar]
- Bozym, R.; Hurst, T.K.; Westerberg, N.; Stoddard, A.; Fierke, C.A.; Frederickson, C.J.; Thompson, R.B. Determination of zinc using carbonic anhydrase-based fluorescence biosensors. In Fluorescence Spectroscopy; Brand, L., Johnson, M., Eds.; Academic Press: San Diego, CA, USA, 2008; Volume 450, pp. 287–309. [Google Scholar] [CrossRef]
- Bjerrum, N.Z. Dissoziationskonstanten von mehrbasischen Säuren und ihre Anwendung zur Berechnung molekularer Dimensionen. Phys. Chem. 1923, 106, 219–242. [Google Scholar]
- Weber, G. Photoelectric Method for the Measurement of the Polarization of the Fluorescence of Solutions. J. Opt. Soc. Am. 1956, 46, 962–970. [Google Scholar] [CrossRef]
- Chen, R.F.; Kernohan, J.C. Combination of Bovine Carbonic Anhydrase with a Fluorescent Sulfonamide. J. Biol. Chem. 1967, 242, 5813–5823. [Google Scholar] [CrossRef]
- Cheong, W.-F.; Prahl, S.A.; Welch, A.J. A review of the optical properties of biological tissues. IEEE J. Quantum Electron. 1990, 26, 2166–2185. [Google Scholar] [CrossRef]
- Marshall, M.V.; Rasmussen, J.C.; Tan, I.-C.; Aldrich, M.B.; Adams, K.E.; Wang, X.; Fife, C.E.; Maus, E.A.; Smith, L.A.; Sevick-Muraca, E.M. Near-infrared fluorescence imaging in humans with Indocyanine Green: A review and update. Open Surg. Oncol. J. 2010, 2, 12–25. [Google Scholar] [CrossRef] [PubMed]
- Kovar, J.L.; Xu, X.; Draney, D.; Cupp, A.; Simpson, M.A.; Olive, D.M. Near-infrared-labeled tetracycline derivative is an effective marker of bone deposition in mice. Anal. Biochem. 2011, 416, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.-H.; Bozym, R.A.; Rosenthal, R.E.; Fiskum, G.; Cotto-Cumba, C.; Westerberg, N.; Fierke, C.A.; Stoddard, A.; Cramer, M.L.; Frederickson, C.J.; et al. In Situ Measurement of Free Zinc in an Ischemia Model and Cell Culture Using a Ratiometric Fluorescence-Based Biosensor; SPIE Conference on Advanced Biomedical and CLinical Diagnostic Systems III; Vo-Dinh, T., Grundfest, W.S., Benaron, D.A., Cohn, G.E., Eds.; SPIE: San Jose, CA, USA, 2005; pp. 51–59. [Google Scholar] [CrossRef]
- McCranor, B.J.; Bozym, R.A.; Vitolo, M.I.; Fierke, C.A.; Bambrick, L.; Polster, B.M.; Fiskum, G.; Thompson, R.B. Quantitative imaging of mitochondrial and cytosolic free zinc levels in an in vitro model of ischemia/reperfusion. J. Bioenerg. Biomembr. 2012, 44, 253–263. [Google Scholar] [CrossRef]
- Supuran, C.T.; Scozzafava, A. Carbonic anhydrase inhibitors and their therapeutic potential. Expert Opin. Ther. Patents 2000, 10, 575–600. [Google Scholar] [CrossRef]
- Zeng, H.H.; Matveeva, E.G.; Stoddard, A.K.; Fierke, C.A.; Thompson, R.B. Long Wavelength Fluorescence Ratiometric Zinc Biosensor. J. Fluoresc. 2013, 23, 375–379. [Google Scholar] [CrossRef][Green Version]
- McCranor, B.; Thompson, R.B. Long Wavelength Fluorescence Lifetime Standards for Front-Face Fluorometry. J. Fluoresc. 2010, 20, 435–440. [Google Scholar] [CrossRef][Green Version]
- Aper, S.J.A.; Dierickx, P.; Merkx, M. Dual Readout BRET/FRET Sensors for Measuring Intracellular Zinc. ACS Chem. Biol. 2016, 11, 2854–2864. [Google Scholar] [CrossRef]
- Michielsen, C.M.S.; van Aalen, E.A.; Merkx, M. Ratiometric Bioluminescent Zinc Sensor Proteins to Quantify Serum and Intracellular Free Zn2+. ACS Chem. Biol. 2022, 17, 1567–1576. [Google Scholar] [CrossRef]
- Pocker, Y.; Fong, C.T.O. Inactivation of bovine carbonic anhydrase by dipicolinate: Kinetic studies and mechanistic implications. Biochemistry 1983, 22, 813–818. [Google Scholar] [CrossRef] [PubMed]

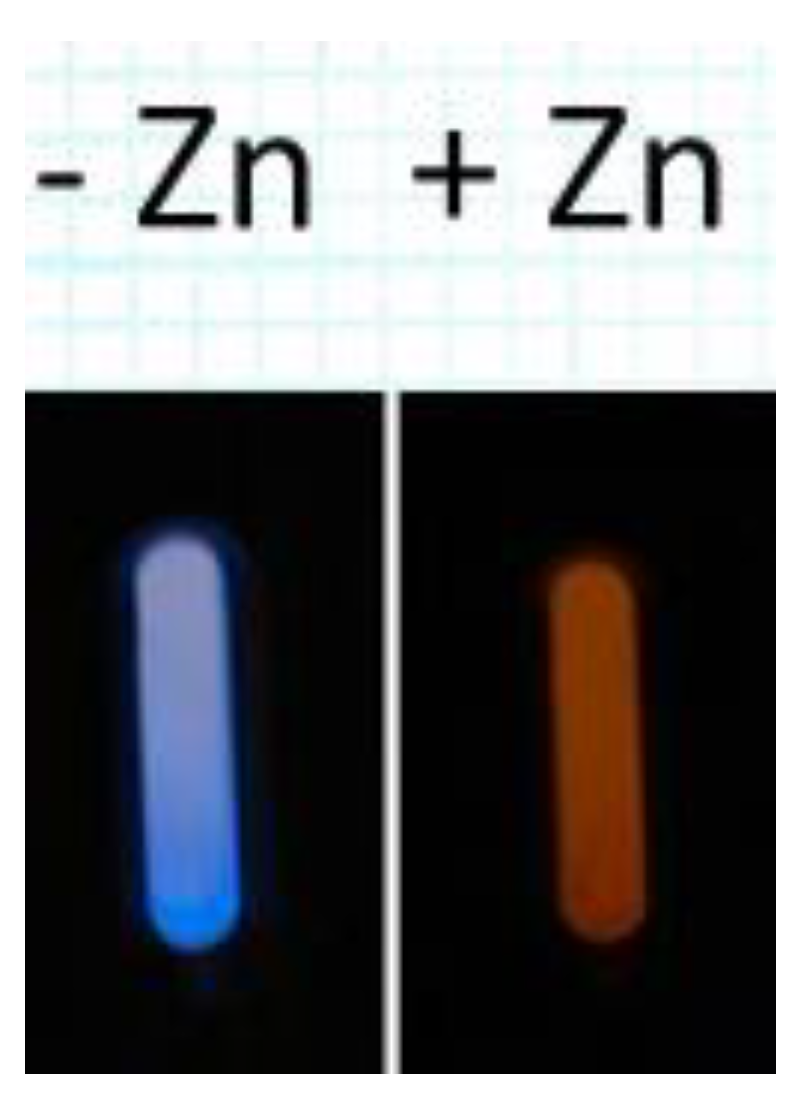
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matveeva, E.G.; Stoddard, A.K.; Zeng, H.-H.; Franke, G.; Bourne, L.; Fierke, C.A.; Thompson, R.B. Ratiometric Zinc Biosensor Based on Bioluminescence Resonance Energy Transfer: Trace Metal Ion Determination with Tunable Response. Int. J. Mol. Sci. 2022, 23, 14936. https://doi.org/10.3390/ijms232314936
Matveeva EG, Stoddard AK, Zeng H-H, Franke G, Bourne L, Fierke CA, Thompson RB. Ratiometric Zinc Biosensor Based on Bioluminescence Resonance Energy Transfer: Trace Metal Ion Determination with Tunable Response. International Journal of Molecular Sciences. 2022; 23(23):14936. https://doi.org/10.3390/ijms232314936
Chicago/Turabian StyleMatveeva, Evgenia G., Andrea K. Stoddard, Hui-Hui Zeng, Graham Franke, Leslie Bourne, Carol A. Fierke, and Richard B. Thompson. 2022. "Ratiometric Zinc Biosensor Based on Bioluminescence Resonance Energy Transfer: Trace Metal Ion Determination with Tunable Response" International Journal of Molecular Sciences 23, no. 23: 14936. https://doi.org/10.3390/ijms232314936
APA StyleMatveeva, E. G., Stoddard, A. K., Zeng, H.-H., Franke, G., Bourne, L., Fierke, C. A., & Thompson, R. B. (2022). Ratiometric Zinc Biosensor Based on Bioluminescence Resonance Energy Transfer: Trace Metal Ion Determination with Tunable Response. International Journal of Molecular Sciences, 23(23), 14936. https://doi.org/10.3390/ijms232314936





